Abstract
Most cases of autoimmune hypothyroidism are due to Hashimoto thyroiditis (HT). It is sometimes difficult to distinguish HT from mucosa associated lymphoid tissue (MALT) lymphoma using cytology or histology alone. This has necessitated the use of immunohistochemistry and other molecular techniques. A descriptive study was undertaken to determine the prevalence of MALT lymphoma and other associations of HT using histopathological and immunohistochemical methods. For testing of kappa and lambda antibodies, paraffin sections were prepared for immunohistochemical staining using a peroxidase-antiperoxidase immune complex method. Immunostaining in HT, demonstrated a polyclonal lymphoid population, as evidenced by dual positivity for kappa and lambda staining cells, whereas MALT lymphoma revealed a monoclonal lymphoid population, with strong positivity for kappa immunostaining and lack of lambda light chain expression. The proportion of MALT lymphoma in surgically treated cases of HT in the present study was 3.5% (95% CI: 0%–8.3%). The common malignancy associated with HT was papillary carcinoma of the thyroid (8.8%). Among the benign conditions, nodular colloid goiter (7%) was more commonly associated. Although modern molecular techniques are available for the confirmation of lymphomas, they are more expensive, time consuming and are available only in a few centers. Strict morphological criteria can differentiate HT from MALT lymphoma, but in suspicious cases, paraffin section immunohistochemistry using light chain restriction can offer comparable and reliable results. Since papillary carcinoma and MALT lymphoma have been associated with HT, these patients require careful surveillance.
Keywords: Hashimoto thyroiditis, MALT lymphoma, morphology, immunohistochemistry, associations and variants, nodular Hashimoto thyroiditis
Introduction
Most cases of autoimmune hypothyroidism are due to Hashimoto thyroiditis (HT). Induction of thyroid autoimmunity is accompanied by a progressive depletion of thyroid epithelial cells by apoptosis and replacement of the thyroid parenchyma by mononuclear cells and fibrosis. The mononuclear cell infiltrate is mainly lymphoplasmacytic with formation of well-developed germinal centers. Besides the clinical sequelae of hypothyroidism, the presence of HT increases the risk of development of thyroid neoplasms, including MALT lymphoma and papillary carcinoma. The lymphoid tissue present in the thyroid has been considered part of the mucosa associated lymphoid tissue (MALT). The coexistence of reactive and neoplastic processes in HT may pose difficulty in diagnosing MALT lymphoma using cytology or histology alone.1 This study was undertaken to ascertain the importance of light chain restriction for confirming the diagnosis of MALT lymphoma and to determine its proportion in diagnosed cases of HT. Variants of HT and its benign and malignant associations were also identified.
Materials and methods
Study design
Descriptive study
The study population included all consecutive cases of HT specimens received during the study period. The study tools used were histopathological and immunohistochemical methods.
Histopathology
Formalin-fixed, paraffin embedded tissue sections were prepared and stained with hematoxylin and eosin stain for histopathological diagnosis.
Immunohistochemistry
Tumor cells were tested with primary antibodies against CD20 (L26, PathnSitu Biotechnologies), lambda (EP172, PathnSitu Biotechnologies), and kappa (EP171, PathnSitu Biotechnologies). For testing of kappa and lambda antibodies, sections were prepared for immunohistochemical staining using a peroxidase-antiperoxidase immune complex method. After inhibition of endogenous peroxidases with 3% hydrogen peroxide, the slides were washed in Tris-buffered saline (TBS) and incubated with primary antibodies (Ig kappa and Ig lambda) for 30 min at 37°C in a humid chamber. After rewashing in TBS, they were incubated with Poly Excel target binder reagent for 12–15 min. The slides were again washed in TBS and incubated with Poly Excel horse radish peroxidase (HRP detection system 2 steps, PathnSitu Biotechnologies) for 12–15 min. After washing in buffer, the reaction products were developed for 2–5 min by adding diamino benzaldehyde (DAB) as the chromogen and the sections were lightly counterstained with Mayer’s hematoxylin.
Ethical conduct: This study conformed to the tenets of World Medical Association Declaration of Helsinki.
Statistics
The statistical method used was descriptive statistics.
Results
Specimen details
The present study was performed using 57 thyroidectomy specimens of clinically diagnosed HT cases. The age of the patients ranged from 26 to 66 years, with a mean of 43.0 ± 9.02 years (Table 1). Among these, 55 were females and two were males (females—96.5% and males—3.5%). All the patients presented with goiter. Diffuse enlargement of the thyroid gland was seen in 36 cases (63.2%) and nodular enlargement in 21 cases (36.8%). Total thyroidectomy was performed in 50 cases (87.6%), subtotal thyroidectomy in three cases (5.3%), hemi-thyroidectomy in three cases (5.3%), and completion thyroidectomy in one case (1.8%).
Table 1.
Age distribution of cases.
| Age in years | Frequency | % |
|---|---|---|
| 21–30 | 8 | 14.0 |
| 31–40 | 13 | 22.8 |
| 41–50 | 24 | 42.1 |
| 51–60 | 11 | 19.3 |
| 61–70 | 1 | 1.8 |
Gross examination
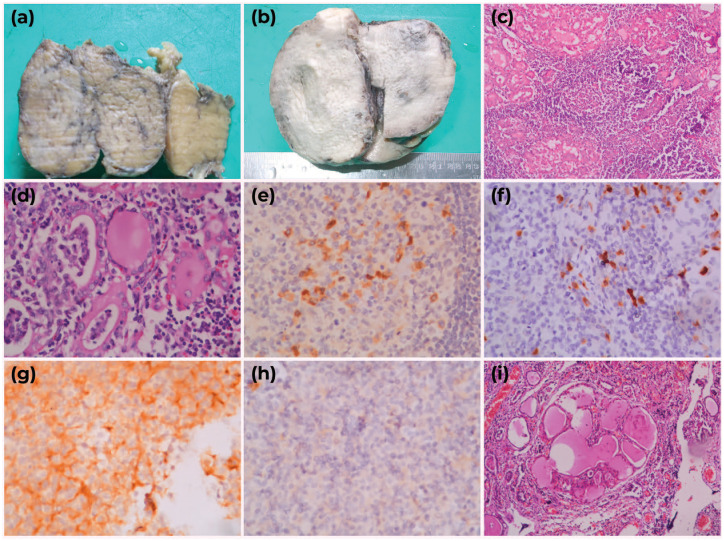
Grossly, the cut surface of all specimens of HT had a gray white, lobulated appearance with a firm consistency (Figure 1(a)), whereas thyroid lymphomas exhibited a homogeneous, gray white, bulging appearance with a soft consistency (Figure 1(b)).
Figure 1.
(a) Gross specimen of Hashimoto thyroiditis; cut surface showing a gray white, lobulated appearance, (b) gross specimen of thyroid lymphoma; cut surface showing a white fleshy and bulging appearance, (c) Hashimoto thyroiditis demonstrating lymphoid follicle formation and atrophic thyroid follicles H-E stain ×100, (d) MALT lymphoma demonstrating lymphoepithelial lesions. H-E stain ×400, (e) Hashimoto thyroiditis demonstrating positive kappa immunostaining ×400, (f) Hashimoto thyroiditis demonstrating positive lambda immunostaining ×400, (g) MALT lymphoma demonstrating strong and diffuse positive kappa immunostaining ×400, (h) MALT lymphoma demonstrating lack of lambda light chain expression immunostaining ×400, and (i) nodular variant of Hashimoto thyroiditis H-E stain ×100.
Histopathological findings
Hashimoto thyroiditis
Among the 55 cases diagnosed as HT by histopathology, 53 showed lymphoid infiltration of the stroma forming lymphoid follicles. However, two cases showed diffuse lymphoplasmacytic infiltration, which raised a suspicion of primary thyroid lymphoma. Most of the lymphoid follicles had a well-defined germinal center and a mantle zone. Intervening atrophic thyroid follicles were seen, many of which were lined by Hürthle cells (Figure 1(c)).
Primary thyroid lymphoma (MALT lymphoma)
Among the 57 thyroidectomy specimens, two cases were diagnosed as primary thyroid lymphoma by histopathology. Extensive lymphoplasmacytoid infiltrate with effacement of architecture by atypical cells were seen. The infiltrate was composed of lymphoid cells with small, slightly irregular, folded or centrocyte-like nuclear contours, condensed nuclear chromatin, and inconspicuous nucleoli. Centroblasts with round, regular nuclear membranes and one to two nucleoli were also noted. Lymphoepithelial lesions were frequent which had a distinctive appearance of rounded balls or masses filling and distending the lumen of the thyroid follicles (MALT balls). Few areas with plasmacytoid differentiation were also noted. Residual thyroid epithelium was present in the lymphoid infiltrate; this took the form of small follicles, many of which were lined by Hürthle cells (Figure 1(d)).
Immunohistochemical findings
Hashimoto thyroiditis
Among the 55 cases, 53 cases which were diagnosed histologically as HT, demonstrated polyclonal lymphoid population on immunohistochemical examination, confirming the histopathologic diagnosis. The two cases that histologically exhibited diffuse lymphoplasmacytic infiltration and raised a suspicion of primary thyroid lymphoma, also illustrated a polyclonal lymphoid population. Thus, in all the 55 cases of HT, the lymphoid population was polyclonal, as evidenced by dual positivity for kappa (Figure 1(e)) and lambda (Figure 1(f)) immunostaining. In these polyclonal infiltrates, the ratio of kappa to lambda staining cells never exceeded 5:1. Hence all the 55 cases of HT diagnosed by histopathology, including the two suspicious cases, were confirmed as HT by immunohistochemistry.
Primary thyroid lymphoma (MALT lymphoma)
The two cases of MALT lymphoma were characterized by the predominance of B-cells that were not confined to the germinal centers. The small and large neoplastic cells expressed CD20 and were negative for CD3, confirming the B-cell lineage. It demonstrated a strong and diffuse positive kappa immunostaining (Figure 1(g)) and lack of lambda light chain expression (Figure 1(h)). Immunostaining for kappa and lambda antibodies confirmed the morphologic diagnosis, where the lymphoid population was clearly monoclonal for kappa with a ⩾10:1 ratio, as counted in 10 high-power microscopic fields. Monoclonality was assumed when the ratio was ⩾10:1 between kappa and lambda staining cells.
Among the 57 thyroidectomy specimens, 55 cases (96.5%) were confirmed as HT by histopathological and immunohistochemical methods. Fibrosing and nodular variants of HT were also identified (Figure 1(i)). The various other associations of HT identified in this study include Hürthle cell nodule, papillary micro-carcinoma, nodular colloid goiter, follicular adenoma, and papillary carcinoma. Two cases were confirmed as MALT lymphoma by histopathological and immunohistochemical methods. MALT lymphoma constituted 3.5% of the cases (95% CI: 0%–8.3%). The different variants of HT and its benign and malignant associations are summarized in Table 2.
Table 2.
Variants and associations of HT.
| Microscopy | Frequency | % |
|---|---|---|
| Hashimoto thyroiditis | 40 | 70.1 |
| Hashimoto thyroiditis and Hürthle cell nodule | 2 | 3.5 |
| Hashimoto thyroiditis and papillary micro-carcinoma | 3 | 5.3 |
| Hashimoto thyroiditis and nodular colloid goiter | 4 | 7.0 |
| Fibrosing variant of Hashimoto thyroiditis | 2 | 3.5 |
| Hashimoto thyroiditis and follicular adenoma | 1 | 1.8 |
| Hashimoto thyroiditis and papillary carcinoma | 2 | 3.5 |
| Nodular Hashimoto thyroiditis | 1 | 1.8 |
| MALT lymphoma | 2 | 3.5 |
Discussion
The current study was performed on 57 thyroidectomy specimens which included 55 cases of HT and two cases of MALT lymphoma. The age of the patients ranged from 26 to 66 years, with a mean age of 43.0 ± 9.02 years. There were 55 females and two males with a predominant female gender predilection. In all the 55 cases of HT, including the two cases with extensive lymphoplasmacytoid infiltrates, the lymphoid population was polyclonal, as evidenced by dual positivity for kappa and lambda immunostaining and the kappa lambda ratio never exceeded 5:1. However, in the two cases of MALT lymphoma (3.5%), the lymphoid population was clearly monoclonal for kappa, with more than 10:1 ratio between kappa and lambda staining cells. Monoclonality was assumed, based on the criteria proposed by Burg et al., when the ratio between kappa and lambda staining cells was 10:1 or greater.2 Thus, immunostaining offers a reliable method to distinguish between HT with extensive lymphoplasmacytoid infiltrate and MALT lymphoma, in concurrence with earlier studies.3–5
HT is an established risk factor for the development of primary thyroid lymphoma (PTL).6 Indeed, lymphocytic thyroiditis is present in the background, in 94% of thyroid lymphomas.7,8 Distinguishing severe chronic lymphocytic thyroiditis from lymphoma can at times, pose great difficulty.9 PTL manifests in only 0.5% of HT cases and hence it should not be viewed as a premalignant condition.10
Most PTLs are of B-cell origin and predominantly of the diffuse large-cell type. The second-most common histologic type is MALT lymphoma. Other less frequently encountered types include Hodgkin’s disease, Burkitt’s lymphoma and plasmacytoma. True follicle center lymphomas of the thyroid and T-cell lymphomas are extremely rare.7,11
Microscopically, the majority of PTLs are diffuse large B-cell lymphoma (DLBCL) and marginal zone B-cell lymphoma (MZBCL), which includes MALT lymphoma. About 50%–80% of PTLs are DLBCLs and 20%–30% are MALT lymphomas of extranodal marginal type.7,12,13 Some DLBCLs might result from the transformation of pre-existing low-grade MALT lymphomas, suggesting a morphological progression from chronic lymphocytic thyroiditis to low-grade MALT lymphoma and subsequently to high-grade large cell lymphoma.11 Various studies have reported an association between PTLs and HT, recognized HT as a risk factor6–13 and suggested a strong link between HT and MALT lymphoma.14 This close relationship is attributed to the chronic antigenic stimulation that leads to malignant transformation.15
MALT is a term proposed by Isaacson and Wright for the component of the immune system that has evolved to protect the freely permeable surface of the gastrointestinal tract and other mucosal membranes, directly exposed to the external environment.16 MALT lymphomas are extranodal, low-grade marginal zone B-cell lymphomas and are a distinct subtype of non-Hodgkin lymphoma.17–19
Immunohistochemically, the immuno-phenotype in MALT lymphoma involves B-cells, positive for CD19, CD20, CD22, CD79a, Bcl-2, anti-kappa, and anti-lambda antibodies, whilst CD3, CD5, CD10, Bcl-6, and CD23 are usually negative.7,17,20 Individual B-cells in lymphoid tissue possess either kappa or lambda immunoglobulin light chains, but never both together. Clonality assessment can be accomplished through evaluation of kappa and lambda light chain expression. Anti-kappa and anti-lambda antibodies detect light chain immunoglobulins on normal and neoplastic B-cells in human lymphoid tissue. In normal lymphoid tissue, the kappa and lambda cell ratio is approximately 2:1. However, this ratio is altered in reactive tissues and malignant conditions. The essential difference between a reactive and neoplastic lymphoid infiltrate is the presence of light chain restriction in the latter.11,17 The lymphoid population is polyclonal in HT, suggesting a reactive population with no light chain restriction. Polyclonality is assumed by dual positivity for kappa and lambda immunostaining, with the ratio of kappa to lambda-staining cells never exceeding 5:1. In thyroid lymphoma, kappa and lambda immunostaining is clearly monoclonal. Monoclonality is assumed when there is a ratio of 10:1 or greater between kappa and lambda staining cells.2 Majority of the positively staining cells in MALT lymphoma are plasmacytes, with an intracytoplasmic kappa light chain belonging predominantly to IgG class.4 However, there are some exceptions that report marginal zone lymphomas with kappa and lambda dual light chain expression, kappa restricted B cells, or B cells with negative light chain expression.20,21
Paraffin section immunohistochemistry using a new set of anti-kappa and anti-lambda antibodies to detect immunoglobulin light chain has been reported in this study. The present study revealed monotypic kappa light chain expression, in both the cases morphologically diagnosed as MALT lymphoma, suggesting a high degree of correlation between morphology and immunohistochemistry.
In the present study, the variants of HT noted were fibrosing and nodular types. The fibrosing variant of HT constituted only 3.5% of cases as against 10%–13% reported earlier. In this variant, the thyroid architecture is destroyed with severe follicular atrophy, dense keloid-like fibrosis and prominent squamoid metaplasia of the follicular epithelium.22
Although HT typically exhibits a diffuse appearance microscopically, in some cases a distinct nodularity is evident. When the epithelial components of such nodules exhibit a hyperplastic quality, it is categorized as nodular HT. Nodular HT comprises Hürthle cells having a solid configuration, along with lymphoplasmacytic infiltration and atretic thyroid follicles in the background. In contrast, the nodular colloid goiter demonstrates nodules composed of colloid-rich follicles and fail to exhibit other features of nodular HT. Also, nodular HT should not be confused with Hürthle cell nodule, as the latter is an associated benign condition lacking features of HT in its setting. In the present study, one case demonstrated features of nodular HT, which is found to be rare in literature review.
The benign associations of HT included follicular adenoma, Hürthle cell nodule, and nodular colloid goiter. HT was seen to be associated more with nodular colloid goiter (7%).
Papillary carcinoma was found to be the most common malignancy associated with HT in the current study. Among the 55 cases of HT, five cases were associated with papillary carcinoma (8.8%), of which three were papillary micro-carcinoma (5.3%), and two were conventional papillary carcinoma (3.5%). These findings are in concurrence with previous studies, that reported papillary carcinoma of thyroid as the most common type of thyroid carcinoma in long standing cases of HT.5,23
In a similar study that discussed 254 cases of papillary micro-carcinoma of the thyroid, HT was noted in the background of 100 cases (39.4%).24 The present study also revealed an association with papillary micro-carcinoma (5.3%), but the low percentage may be due to racial or ethnic differences, the small sample size, and the prospective design of the study.
Review of the literature shows a wide variation in the rate of coexistence of HT and papillary carcinoma.25–30 Co-occurrence of papillary carcinoma and MALT lymphoma is exceedingly rare in HT patients.23 As papillary carcinoma and MALT lymphoma are associated with HT, these patients warrant careful surveillance.
This study outlines an audit of our experience with HT and a comprehensive discussion regarding its variants and associations. The prevalence of MALT lymphoma and papillary carcinoma in diagnosed cases of HT has been reported in various populations and races. It ranged between 0.5% to 6.3% for MALT lymphomas, and 0.4% to 9.5% for papillary carcinomas in different studies. The prevalence of MALT lymphoma and papillary carcinoma in symptomatic, surgically treated cases of HT has been reported as 3.5% and 8.8% respectively in the present study. This high percentage is not a true reflection of prevalence, as the study does not include all the cases of HT in the general population.
Although modern molecular techniques are available for the confirmation of lymphomas, they are more expensive, time consuming and are available only in a few centers. A drawback associated with paraffin-section immunohistochemistry is the variation in the intensity of immunostaining, probably due to fixation issues and other intrinsic factors. Strict morphological criteria can differentiate between HT and MALT lymphoma; but in suspicious cases, paraffin-section immunohistochemistry can offer comparable and reliable results.
Conclusion
The current study was performed on 57 thyroidectomy specimens, which included 55 cases of HT and two cases of MALT lymphoma. The proportion of MALT lymphoma in surgically treated cases of HT constituted 3.5%of the cases (95% CI: 0%–8.3%). Immunostaining offers a reliable method to distinguish between HT with extensive lymphoplasmacytoid infiltrate and MALT lymphomas. Immunostaining in HT demonstrated a polyclonal lymphoid population, as evidenced by dual positivity for kappa and lambda staining. MALT lymphoma revealed a monoclonal lymphoid population, with strong positivity for kappa immunostaining and lack of lambda light chain expression. Hence, light chain restriction was useful in distinguishing HT from MALT lymphoma. The variants and the benign conditions associated with HT included the fibrosing variant, the nodular variant, Hürthle cell nodule, follicular adenoma, and nodular colloid goiter. Among the benign conditions, nodular colloid goiter (7%) was found to be more commonly associated with HT. The prevalence of MALT lymphoma and papillary carcinoma in surgically treated cases of HT is being reported as 3.5% and 8.8% respectively in the present study. Hence patients with HT require careful surveillance for both the disease entities.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: CP Dain  https://orcid.org/0000-0002-6325-1427
https://orcid.org/0000-0002-6325-1427
References
- 1. Baloch ZW, Livolsi VA. Pathology of thyroid and parathyroid disease. In: SE Mills. (ed.) Sternberg’s diagnostic surgical pathology. 5th ed. Philadelphia: Lippincott Williams and Wilkins, 2010, pp.1508–1653. [Google Scholar]
- 2. Burg G, Kerl H, Kaudewitz P, et al. Immunoenzymatic typing of lymphoplasmacytoid skin infiltrates. J Dermatol Surg Oncol 1984; 10: 284–290. [DOI] [PubMed] [Google Scholar]
- 3. Amani HK. Histopathologic and immunohistochemical features of Hashimoto thyroiditis. Indian J Pathol Microbiol 2011; 54: 464–471. [DOI] [PubMed] [Google Scholar]
- 4. Hyjek E, Isaacson PG. Primary B cell lymphoma of the thyroid and its relationship to Hashimoto’s thyroiditis. Hum Pathol 1988; 19: 1315–1326. [DOI] [PubMed] [Google Scholar]
- 5. Chen HI, Akpolat I, Mody DR, et al. Restricted kappa lambda light chain ratio by flow cytometry in germinal centre B cells in Hashimoto thyroiditis. Am J Clin Pathol 2006; 125: 42–48. [PubMed] [Google Scholar]
- 6. Holm L, Blomgren H, Lowhagen T. Cancer risks in patients with chronic lymphocytic thyroiditis. N Engl J Med 1985; 312: 601–604. [DOI] [PubMed] [Google Scholar]
- 7. Derringer GA, Thompson LD, Frommelt RA, et al. Malignant lymphoma of the thyroid gland: a clinicopathologic study of 108 cases. Am J Surg Pathol 2000; 24: 623–639. [DOI] [PubMed] [Google Scholar]
- 8. Widder S, Pasieka JL. Primary thyroid lymphomas. Curr Treat Options Oncol 2004; 5: 307–313. [DOI] [PubMed] [Google Scholar]
- 9. Thieblemont C, Mayer A, Dumontet C, et al. Primary thyroid lymphoma is a heterogeneous disease. J Clin Endocrinol Metab 2002; 87: 105–111. [DOI] [PubMed] [Google Scholar]
- 10. Hengjeerajarus N, Klaisuwan T, Norasetthada L, et al. MALT lymphoma with large cell transformation on the background of Hashimoto’s thyroiditis: a case report and review literature. J Med Assoc Thai 2015; 98: 514–519. [PubMed] [Google Scholar]
- 11. Sarinah B, Hisham AN. Primary lymphoma of the thyroid: diagnostic and therapeutic considerations. Asian J Surg 2010; 33: 20–24. [DOI] [PubMed] [Google Scholar]
- 12. Peixoto R, Pinto JC, Soares V, et al. Primary thyroid lymphoma: a case report and review of literature. Ann Med Surg 2017; 13: 29–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chen C, Yang Y, Jin L, et al. Primary thyroid T-lymphoblastic lymphoma: a case report and review of the literature. Int J clin Exp Pathol 2014; 7: 443–450. [PMC free article] [PubMed] [Google Scholar]
- 14. Teixeira Mendes LS, Wotherspoon A. Marginal zone lymphoma: associated autoimmunity and auto-immune disorders. Best Pract Res Clin Haematol 2017; 30: 65–76. [DOI] [PubMed] [Google Scholar]
- 15. Kossev P, Livolsi V. Lymphoid lesions of the thyroid: review in light of the revised European-American lymphoma classification and upcoming World Health Organisation classification. Thyroid 1999; 9: 1273–1280. [DOI] [PubMed] [Google Scholar]
- 16. Isaacson P, Wright DH. Malignant lymphoma of mucosa-associated lymphoid tissue. A distinctive type of B-cell lymphoma. Cancer 1983; 52: 1410–1416. [DOI] [PubMed] [Google Scholar]
- 17. Bacon CM, Du MQ, Dogan A. Mucosa-associated lymphoid tissue (MALT) lymphoma: a practical guide for pathologists. J Clin Pathol 2007; 60: 361–372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Maes B, De Wolf-Peeters C. Marginal zone B-cell lymphoma-an update on recent advances. Histopathology 2002; 40: 117–126. [DOI] [PubMed] [Google Scholar]
- 19. Troch M, Woehrer S, Strenbel B, et al. Chronic autoimmune thyroiditis (Hashimoto thyroiditis) in patients with MALT lymphoma. Ann Oncol 2008; 19: 1336–1339. [DOI] [PubMed] [Google Scholar]
- 20. Tanaka T, Kitabatake K, Lino M, et al. Immunohistochemical comparison of CD5, lambda, and kappa expression in primary and recurrent buccal mucosa- associated lymphoid tissue (MALT) lymphomas. Diagn Pathol 2011; 6: 82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Weiss LM, Loera S, Bacchi CE. Immunoglobulin light chain immunohistochemistry revisited, with emphasis on reactive follicular hyperplasia vs. follicular lymphoma. Appl Immunohistochem Mol Morphol 2010; 18: 199–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Harach HR, Williams ED. Fibrous thyroiditis - an immunopathological study. Histopathology 1983; 7: 739–751. [DOI] [PubMed] [Google Scholar]
- 23. Cheng V, Brainard J, Nasr C. Co-occurrence of papillary thyroid carcinoma and primary lymphoma of the thyroid in a patient with long standing Hashimoto thyroiditis. Thyroid 2012; 22: 647–650. [DOI] [PubMed] [Google Scholar]
- 24. Fu X, Lou S, Shi H, et al. Clinicopathologic analysis of 254 cases of papillary thyroid microcarcinoma. Zhonghua Bing Li Xue Za Zhi 2015; 44: 258–261. (In Chinese) [PubMed] [Google Scholar]
- 25. Matesa-Anic D, Matesa N, Dabelic N, et al. Coexistence of papillary carcinoma and Hashimoto thyroiditis. Acta Clin Croat 2009; 48: 9–12. [PubMed] [Google Scholar]
- 26. Konturek A, Braczynski M, Wierchowski W, et al. Coexistence of papillary thyroid cancer with Hashimoto thyroiditis. Langenbecks Arch Surg 2013; 398: 389–394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Girardi FM, Barra MB, Zettler CG. Papillary thyroid carcinoma: does the association with Hashimoto thyroiditis affect the clinicopathologic characteristics of the disease? Braz J Otorhinolaryngol 2015; 81:283–287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Jeong JS, Kim HK, Lee CR, et al. Coexistence of chronic lymphocytic thyroiditis with papillary thyroid carcinoma: clinical manifestation and prognostic outcome. J Korean Med Sci 2012; 27: 883–889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Yoon YH, Kim HJ, Lee JW, et al. The clinicopathologic differences in papillary thyroid carcinoma with or without coexisting chronic lymphocytic thyroiditis. Eur Arch Otorhinolaryngol 2012; 269: 1013–1017. [DOI] [PubMed] [Google Scholar]
- 30. Noureldine SI, Tufano RP. Association of Hashimoto’s thyroiditis and thyroid cancer. Curr Opin Oncol 2015; 27: 21–25. [DOI] [PubMed] [Google Scholar]