Abstract
Eukaryotic 5-methylcytosine RNA methyltransferases catalyze the transfer of a methyl group to the fifth carbon of a cytosine base in RNA sequences to produce 5-methylcytosine (m5C). m5C RNA methyltransferases play a crucial role in the maintenance of functionality and stability of RNA. Viruses have developed a number of strategies to suppress host innate immunity and ensure efficient transcription and translation for the replication of new virions. One such viral strategy is to use host m5C RNA methyltransferases to modify viral RNA and thus to affect antiviral host responses. Here, we summarize the latest findings concerning the roles of m5C RNA methyltransferases, namely, NOL1/NOP2/SUN domain (NSUN) proteins and DNA methyltransferase 2/tRNA methyltransferase 1 (DNMT2/TRDMT1) during viral infections. Moreover, the use of m5C RNA methyltransferase inhibitors as an antiviral therapy is discussed.
Keywords: 5-methylcytosine, NSUN proteins, DNMT2/TRDMT1, viruses, antiviral drugs
1. Introduction
The development of molecular techniques has allowed for research intensification in the field of RNA modifications. Up to now, over a hundred of RNA modifications have been identified including the methylation of fifth carbon in cytosine catalyzed by a number of m5C RNA methyltransferases [1]. Since the first report demonstrating chromatography-based detection of methylated cytosines in poly(A)-messenger RNA [2], m5C was also detected in other types of RNA such as transfer RNA (tRNA), ribosomal RNA (rRNA), small nuclear RNA (snRNA), microRNA (miRNA), long noncoding RNA (lncRNA), transactivation response element (TAR), small vault RNA (vtRNA), and enhancer RNA (eRNA) of many organisms across all phylogenetic groups [3,4,5,6] Thanks to advances in RNA bisulfite sequencing, methylated RNA immunoprecipitation sequencing, and 5-azacytidine-mediated RNA immunoprecipitation or methylation-induced cross-linking immunoprecipitation, new qualitative and quantitative information about the target sites of RNA cytosine methyltransferases may be provided [7,8,9,10]. Molecular studies have also shown that m5C can regulate many aspects of RNA metabolism, namely, RNA export, ribosome assembly, translation, and RNA stability [11]. However, the impact of RNA cytosine methylation on transcriptional and translational efficiency during viral infection is still far from being understood. More information is available concerning the effects of N6-methyladenosine (m6A) and 2′O-methylated (Nm) ribonucleotides. For instance, m6A may stabilize the viral RNA and efficiency of viral protein translation [12,13]. Moreover, the 2′-O-methylation on the first nucleotide of the viral RNA may protect the viral RNA from the innate immune response pathway by camouflaging the viral RNA as a cellular mRNA [14]. Despite the role of m6A and 2′O-methylated (Nm) ribonucleotides in viral RNA metabolism is well characterized, the function of m5C during viral infection is less explored, and important issues remain to be resolved [15]. Therefore, here, we summarize the latest findings concerning the roles of m5C RNA methyltransferases during viral infections and suggest the use of m5C RNA methyltransferase inhibitors in antiviral therapies.
2. m5C RNA Methyltransferases and Their RNA Targets
It is widely accepted that in humans, m5C is incorporated into different RNA molecules by the action of seven members of the NOL1/NOP2/SUN domain (NSUN) family of proteins, namely, NSUN1, NSUN2, NSUN3, NSUN4, NSUN5, NSUN6, and NSUN7, and the DNA methyltransferase 2/tRNA methyltransferase 1 (DNMT2/TRDMT1) [16]. Moreover, selected NSUN proteins such as NSUN1, NSUN2, and NSUN5 are evolutionally conserved and, e.g., the yeast Saccharomyces cerevisiae orthologues are available, namely, Nop2, Trm4, and Rcm1, respectively [6]. m5C RNA methyltransferases rely on S-adenosylmethionine (SAM) as a donor of the methyl group, and NSUN proteins utilize two catalytic cysteines in the active site, whereas DNMT2 uses a single cysteine in the active site that is similar to the mechanism of action of other DNA methyltransferases [17,18]. Regardless of mechanism involved, a covalent intermediate is created between a protein cysteine and the cytosine in RNA to stimulate the electron-deficient pyrimidine heterocycle for the nucleophilic attack of fifth carbon on the methyl group of SAM that is located in two conserved motifs IV and VI in NSUN family members [17].
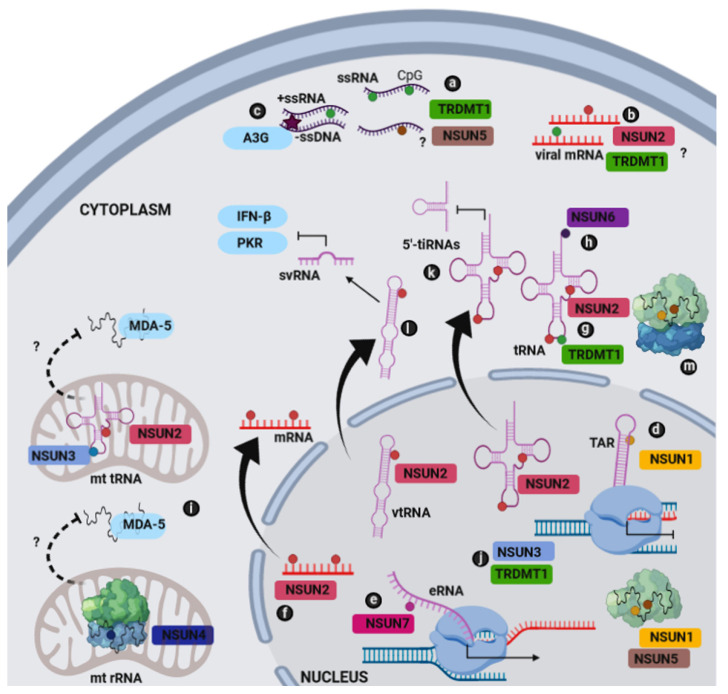
Different subcellular localization and RNA targets of human m5C RNA methyltransferases have been documented (Figure 1) [6]. Nucleolar NSUN1 (NOP2/nucleolar antigen p120) methylates the single C5 position of cytosine (4447nt) in 28S rRNA [19]. Nuclear/nucleolar NSUN2 introduces m5C at two positions of intron-containing tRNA(Leu)(CAA) precursors (34 and 48nt) and at three positions of tRNA(Gly)(GCC) precursors (48, 49, and 50nt) [20,21] as well as in mitochondrial tRNAs [22]. NSUN2 may be also detected in the cytoplasm. NSUN2 also methylates cytosine to produce m5C in various RNAs such as mRNAs, lncRNAs, and small non-coding RNAs, such as vault RNAs (vtRNAs) [20,21,23,24]. Mitochondrial NSUN3 mediates methylation of cytosine to yield m5C at the nucleotide 34 of mt-tRNA(Met) [25,26]. Mitochondrial NSUN4 methylates mt-12S rRNA at the nucleotide 841 [27]. Nucleolar NSUN5 methylates the single C(5) position of cytosine in 28S rRNA (3782nt) [28,29,30]. Cytoplasmic and Golgi apparatus located NSUN6 methylates the C5 position of cytosine 72 in tRNA(Thr)(TGT) and tRNA(Cys)(GCA) [31,32,33]. Nuclear NSUN7 methylates enhancer RNAs (eRNAs) at different positions [34]. Cytoplasmic, and to a less extent nuclear, DNMT2 (TRDMT1) methylates the aspartic acid, glycine, and valine tRNA at the cytosine-38 residue in the anticodon loop [35,36].
Figure 1.
Overview of human m5C RNA methyltransferases and their RNA targets during viral infections. (a) The methylation of 5′C—phosphate—G3′ (CpG) islands in viral genomic RNA by DNMT2 (TRDMT1) and perhaps NSUN5 may promote viral heterogeneity. (b) The modification of viral transcriptome by NSUN2 and perhaps TRDMT1 may result in increased transcript stability and efficient translation. (c) The cytosine deamination by A3G may lead to dC-to-dU conversions in viral (–)ssDNA that may cause G-to-A hypermutations in progeny viral genomes. (d) Induction of viral latency by NSUN1-mediated m5C methylation of TAR. (e) NSUN7-mediated m5C methylation of eRNA, a transcriptional coactivator of PGC-1α (peroxisome proliferator-activated receptor gamma coactivator 1-alpha) and an activator of RNA polymerase II. (f) NSUN2-mediated methylation of viral mRNA promotes its splicing and transport into the cytoplasm (a black arrow). (g,h) NSUN2-, TRDMT1-, and NSUN6-mediated methylation of tRNA molecules protects them against stress-induced degradation and supports efficient translation. (i) Putative protective role of NSUN2-, NSUN3-, and NSUN4-mediated methylation of tRNA and rRNA in the mitochondria. These modification events prevent cytoplasmic translocation of tRNA and rRNA and binding of stress-based degraded form of tRNA and rRNA to a pattern-recognition receptor for RNA (MDA-5) that in turn counteracts the induction of a type I interferon response. (j) NSUN3 and TRDMT1 as putative regulators of RNA polymerase II-mediated gene transcription during cellular stress response. (k) NSUN2-mediated biogenesis of tiRNAs. (l) NSUN2-mediated methylation of vtRNA may promote its conversion to svRNA that, in turn, may inhibit the translation of PKR and IFN-β production. (m) NSUN1- and NSUN5-mediated methylation of rRNA supports ribosome biogenesis and efficient translation. rRNA, ribosomal RNA; tRNA, transfer RNA; mt tRNA, mitochondrial tRNA; eRNA, enhancer RNA; svRNA, specific vtRNA-derived small non-coding RNA; vtRNA, vault RNA; TAR, transactivation response element; tiRNA; tRNA-derived stress-induced RNA, mRNA, messenger RNA; ssRNA, single-stranded RNA; MDA-5, melanoma differentiation-associated protein 5; PKR, RNA-dependent protein kinase; IFN-β, interferon beta; ?-putative action, A3G, apolipoprotein B mRNA editing enzyme catalytic subunit 3G (cytidine deaminase); *- dC-to-dU conversion in viral negative-sense single-stranded DNA (–ssDNA).
Diverse phenotypic features have been reported in mice lacking selected NSun proteins. NSun1 deletion induced apoptotic cell death, affected lineage specification and decreased the pools of RNA during mouse embryonic development [37]. In NSun2−/− mice, testis size is decreased and sperm differentiation is inhibited [38]. Moreover, Dnmt2−/−NSun2−/− embryos were smaller and lighter compared to their wild-type littermates. Newborn double-knockout mice also appeared smaller at birth, failed to develop visible milk spots, showed a reduced thickness and organization of the cerebral cortex and an immature skeleton with incomplete ossification [39]. Dnmt2−/−NSun2−/− mice were also characterized by changes in cellular lipid storage [39]. It has been documented that Nsun4 is essential for embryonic development in the mouse [27]. The growth of mutant embryos (Nsun4−/−) was significantly retarded [27]. Moreover, knockout of Nsun4 in the mouse heart resulted in mitochondrial dysfunction, namely, mitoribosome assembly and mitochondrial translation were diminished [27]. In contrast, adult Nsun5-KO mice displayed spatial cognitive deficits with impairment of oligodendrocyte precursor cells [40]. Nsun5 knockout in mice also resulted in a decrease in body weight and lean mass and limited protein synthesis and growth [30]. It has been suggested that NSun6 is dispensable for mouse embryonic development, and loss of NSUN6-associated methylation lowers mRNA stability in embryonic cells [41]. Mice with mutated Nsun7 gene showed male sterility as a consequence of affected sperm motility [42].
3. Host-Based Methylation of the Fifth Position of Cytosine Residues in Viral RNA
In 1975, the presence of 5-methylcytosine (m5C) in the Sindbis virus 26S RNA obtained from infected host cells was revealed and the putative role of m5C during viral infection was postulated [43,44]. Similar observations were noticed in the case of adenovirus 2 [45]. In the RNA of cells infected with adenovirus 2, N6-methyladenosine (m6A) and a minor fraction of m5C were detected. However, the authors were unable to identify the presence of m5C in the viral RNA particles.
Recent studies using ultrahigh-performance liquid chromatography (UHPLC), tandem mass spectrometry (UPLC-MS/MS), and photo-cross-linking-assisted m5C sequencing have revealed that genomic RNA of murine leukemia virus (MLV) contains higher levels of m6A, m5C, and 2′O-methylated (Nm) ribonucleotides than cellular mRNA [46]. Similarly, 10-fold higher levels of m5C and 2′O-methylations have been observed in human immunodeficiency virus 1 (HIV-1) RNA compared to these noticed in typical cellular mRNAs [47]. Courtney et al. determined that m5C accounted for 0.6–1.4% of cytosines in HIV-1 RNA (approximately 11 m5Cs per HIV-1 genomic RNA) [47].
It is worthwhile to mention that methylation occurs the most frequently in CpG regions of DNA and RNA viruses [48,49,50]. More recently, it has been shown that CpG may be also over-represented in dsRNA viruses at the location across codon boundaries compared to negative-sense single-stranded RNA (−ssRNA) and positive-sense single-stranded RNA (+ssRNA) viruses [51]. It is postulated that the changes in retroviral CpG RNA may be a result of cytidine deaminase activity. However, the mechanism that may contribute to the elimination of CpG in riboviruses is still unknown. Perhaps it is not based on cytidine deaminase activity as riboviral RNA does not form the DNA intermediate that is essential for cytidine deaminase activity [51,52].
The analysis of RNA modification using direct RNA sequencing also revealed m5C signals in viral RNA of Coronaviridae family [53,54]. More recently, a comprehensive study on severe acute respiratory syndrome coronaviruses (SARS-CoV) using direct RNA sequencing documented that 42 positions with predicted m5C modification occur at consistent positions between subgenome-length mRNAs [55]. Thus, one may ask a question if the occurrence of cytosine methylation at fifth position may be a source of genetic heterogeneity in early steps of coronavirus infection. This may rely on the discontinuous extension for synthesis of subgenome-length negative strands [56], namely, a nested set of 5′- and 3′-coterminal subgenomic (sg) mRNAs is produced and this set is characterized by a common 5′ leader sequence, which is identical to the 5′-end of the viral genome [57,58]. However, the role of 5-methylcytosine in viral genomic RNA, especially in Coronaviridae family, is not well established and requires further comprehensive studies.
It is known that experimental increase in the pools of CpG dinucleotides in CpG-deficient viral genomes may lead to significant decrease in viral replication and virulence [59,60,61]. Thus, it may suggest that m5C RNA methyltransferases may play an important role during the recognition of viral CpG and the inhibition of replication process of selected types of viruses. For example, fruit fly m5C RNA methyltransferases Dnmt2 has been reported to directly interact with Drosophila C virus (DCV) RNA being a part of antiviral defense strategy as an evolutionarily conserved innate immune response [62,63] (Figure 1).
It has been shown that NSUN5 can also bind to the core protein of HCV, a positive-strand RNA virus [64] (Figure 1). On the other hand, it has been reported that the methyltransferase NSUN2 may serve as the primary writer for m5C on HIV-1 genomic RNA [47]. Inactivation of NSUN2 resulted in limited addition of m5C to HIV-1 transcripts and inhibited viral replication [47]. Thus, we suggest that this modification may be important also for genomic RNA stabilization, RNA transport to host cellular compartments, replication regulation, protection against degradation, and promotion of viral genetic heterogeneity that is based on cytidine deaminase activity.
To the best of our knowledge, little is known about the effects of m5C RNA methyltransferases on the activity of cytidine deaminase-based antiviral defense system. The evolutionarily conserved APOBEC family of proteins (apolipoprotein B mRNA editing enzymes) are deaminase enzymes (cytidine-to-uridine editing enzymes) that allow for editing of RNA/ssDNA sequences and may promote diversity is mRNA editing [65]. Apolipoprotein B editing complex 3 (APOBEC3) consists of one gene in rodents and up to seven genes in primates, namely, APOBEC3A, APOBEC3B, APOBEC3C, APOBECDE, APOBEC3F, APOBEC3G, and APOBEC3H [65]. APOBEC3 subfamily has important role during viral infections as it can inhibit a number of viruses, e.g., HIV-1, human T-lymphotropic virus (HTLV), hepatitis C virus (HCV), hepatitis B virus (HBV), human papillomavirus (HPV), herpes simplex virus 1 (HSV-1) and Epstein-Barr virus (EBV) by editing-dependent and independent mechanisms [65]. In particular, APOBEC3G (A3G) promotes cytidine-to-uridine hypermutations during reverse transcription and deaminates C residues in CC motifs and other members of APOBEC3 group provide modifications in CT motifs [66,67]. APOBEC3 may promote beneficial mutations of HIV type-1 that may result in adaptation and evolution in natural infection [68]. APOBEC3A can be considered as a potent deamination factor of both C and m5C, while APOBEC3G is much weaker in its ability to deaminate m5C [69]. More recently, the N2-C271A NSUN2 mutant was considered to study the proteins, which are packaged into HIV-1 virions [47]. This mutagenic event is based on the substitution of second conserved cysteine to alanine that may result in spontaneous cross-links to target cytosines. Interestingly, APOBEC3G and NSUN2 were shown to be packaged into HIV-1 virions in the N2-C271A NSUN2 mutant [47]. This may suggest putative interactions between NSUN2 and APOBEC3G. However, the consequences of such interactions need to be determined in the future (Figure 1). Perhaps host m5C RNA methyltransferases may protect some viruses against mutagenic activity of cytidine deaminase that may limit cytidine deaminase-mediated lethality.
4. The Modulation of Host Cellular Metabolism via Virus Hijacking of RNA Processing
The transcriptome of host cells infected with several RNA viruses, e.g., Zika virus (ZIKV), dengue virus (DENV), HCV, poliovirus, and HIV-1, has been reported to be post-transcriptionally modified [70]. This may suggest that post-transcriptional modifications such as 5-methylcytosine may be considered as a new layer of regulation by which RNA viruses subvert the host and evade cellular surveillance systems [70].
Viral infections are also accompanied by cellular stress responses in the host cells that may affect proper transcription and translation of viral mRNA and proteins, respectively [71]. Thus, m5C RNA methyltransferases can be exploited to sustain viral transcriptional and translational activity by the incorporation of 5-methylcytosine into viral mRNA and/or host tRNA and rRNA [20]. Indeed, there are numerous examples of the involvement of host m5C RNA methyltransferases in RNA modification-based regulation of transcription and translation upon viral infections.
It has been postulated that NSUN1 (NOP2), a member of ribosomal large subunit assembly complex, may play the role in the regulation of the cell cycle and in the nucleolar activity associated with cell proliferation [72]. Kong et al. have recently shown that NOP2 restricts HIV-1 replication, suppresses HIV-1 transcription, and promotes viral latency via m5C methylation of TAR RNA [73] (Figure 1).
Depletion of NSUN7 decreased the levels of m5C in eRNA. Methylated eRNA is considered to be a transcriptional coactivator associated with peroxisome proliferator-activated receptor-gamma co-activator 1 alpha (PGC-1α) that modulates cellular metabolic responses. eRNA methylation stabilizes eRNA-bound protein complex and enhances RNA polymerase II activity [34]. This may suggest the involvement of NSUN7 during metabolic reprogramming of infected cells as PGC-1α is robustly induced upon HCV infection [74] (Figure 1).
NSUN2 is involved in various processes such as cellular differentiation by controlling protein synthesis, promoting tRNA stability and preventing mRNA decay [21]. It has been shown that HIV-1 mRNA in infected cells is highly modified by the addition of m5C by the host NSUN2 methyltransferase. Inactivation of NSUN2 resulted in the loss of m5C addition and inhibition of HIV-1 mRNA translation as well as reduced ribosome binding to viral mRNAs and the dysregulation of alternative splicing of viral RNAs [47] (Figure 1). It was also shown that downregulation of the m5C writer NSUN2 inhibits MLV replication [46].
It was also reported that human DNMT2 participates in RNA processing during cellular stress [75]. DNMT2 interacts with proteins involved in RNA processing and DNMT2 is translocated from the nucleus into the cytoplasm during cellular stress [75]. DNMT2 also promoted survival of HIV-1 RNA in infected host cells by RNA cytosine methylation activity [76]. HIV-1 induces DNMT2 translocation from the nucleus to the stress granules, and then, DNMT2 methylates HIV-1 RNA [76]. These suggest that HIV-1 ensures its own survival in the host cells via hijacking of the RNA processing machinery and stress granule promotion.
In contrast to DNMT2- and NSUN2-mediated tRNA modifications, the role of NSUN6 during viral infection remains elusive due to the lack of comprehensive research data. However, selected transcriptomic analyses showed that the changes in NSUN6 expression occur upon infection with the SARS-CoV [77] (Figure 1).
Thus far, there is no information regarding the direct associations between NSUN3 or NSUN4 and viral infection. However, it is evident that at least one role of these mitochondrial m5C rRNA methyltransferases is to maintain mitochondrial activity during infection-associated intense energy consumption [71] (Figure 1).
Interestingly, mRNA expression microarray datasets from the Gene Expression Omnibus (GEO) database showed that expression levels of both 5-methylcytosine rRNA methyltransferase genes in cells infected with Ebola virus (EBOV) (GSE17509), influenza A virus (IAV) subtype H5N1 (A/H5N1) (GSE43302), SARS-BatS RBD (GSE47960), and HCV JFH-1 strain (GSE20948) were altered suggesting that NSUN3 and NSUN4 proteins are active members of the cellular viral response pathway.
Here, we hypothesize that mitochondrial NSUNs are essential for the maintenance of mitochondrial RNA stability and loss of methylation may lead to the transfer of nonfunctional mtRNAs into the cytoplasm. mtRNAs can be recognized by MDA5 and bind to MDA5, a well-known pattern-recognition receptor for RNA that induces a type I interferon response [78]. This hypothesis is supported by a recent work of Dhir et al. (2018) showing that mitochondrial RNA could be a source of self-nucleic acids and an initiator of an interferon response via the cytoplasmic receptor MDA5 as a consequence of RNA metabolism [79] (Figure 1).
Moreover, NSUN3 may have distinct roles beyond the methylation of mt-tRNA(Met) I [25,26,80]. Cheng et al. have found that NSUN3 can form a complex with heterogeneous nuclear ribonucleoprotein K (hnRNPK), DNMT2, and positive transcription elongation factor b (P-TEFb) at elongating RNA polymerase II sites [80]. Thus, it is possible that DNMT2 and NSUN3 can be also considered as new regulatory proteins in transcriptional process in the nucleus during cancer progression or chronic cellular stress [80] (Figure 1).
5. Attenuation of Host Antiviral Response by Virus-Mediated Activation of NSUN2
NSUN2, a multifunctional methyltransferase, is also required for biogenesis of tRNA-derived non-coding fragments (tRFs) [20] (Figure 1). tRFs inhibit protein synthesis via several mechanisms including direct inhibition of the ribosome and displacement of RNA-binding proteins [81,82,83]. Interestingly, some viruses can use tRFs to modulate host immune responses [84]. Deng et al. showed that respiratory syncytial virus (RSV) induces activation of ribonuclease angiogenin (ANG) and cleaves mature cytoplasmic tRNAGlu (CTC) in its anticodon loop to produce two halves: 5-half (tRF5-GluCTC or 5-tiRNAGlu) and 3-half (3-tiRNAGlu) that suppress the expression of anti-RSV protein APOER2 [84].
In addition, C(5)-methylation of mRNAs regulates mRNA export (Figure 1). Methylated transcripts are specifically recognized by THOC4/ALYREF that mediates mRNA nucleocytoplasmic shuttling [85]. THOC4/ALYREF are essential for the export of Kaposi’s sarcoma-associated herpesvirus (KSHV) intronless mRNAs and infectious virus production via recruitment of the TREX complex [86].
NSUN2 controls processing of vault ncRNAs into small regulatory RNAs with microRNA functions such as the three vtRNAs [87]. vtRNA contains a cytosine methylation site, and the methylation of vtRNA1.1 promoted its processing into smaller fragments (svRNAs). The lack of NSUN2 protein leads to the loss of cytosine-5 methylation in vault RNAs causing aberrant processing into Argonaute-associated small RNA fragments [87]. vtRNAs can be also used by viruses to silence host antiviral responses [88]. The double-stranded (ds) RNA-dependent protein kinase (PKR), a member of host innate immune responses, mediates the activation of signal transduction pathways leading to interferon beta (IFN-β) gene induction during viral infection or RNA transfection [89] (Figure 1). It was shown that vtRNAs promoted viral replication in A549 cells and mouse lungs after infection with IAV through repressing PKR activation and the subsequent antiviral interferon response [88].
During SARS-CoV infection, small noncoding RNAs were also detected. The analysis of deep sequenced RNA isolated from the lungs of infected mice showed three 18–22 nt small viral RNAs (svRNAs). The three svRNAs were derived from the nsp3 (svRNA-nsp3.1 and -nsp3.2) and N (svRNA-N) genomic regions of SARS-CoV [90]. These observations suggest that the biogenesis of small viral RNA could be also a target of host NSUN2, but additional studies are required to confirm such assumptions.
6. Inhibitors of m5C RNA Methyltransferases as Antiviral Drugs
Taking into account the involvement of m5C RNA methyltransferases in the antiviral response, one can ask whether the inhibitors of these enzymes can be considered as antiviral drugs. Nucleotide and nucleoside analogs are examples of broad-spectrum antiviral drugs that inhibit transcription and/or replication of different RNA and DNA viruses [91]. Pharmacologic inhibition of DNA methylation has been successfully used as an anticancer therapy using the cytosine analogs 5-azacytidine (azacytidine) and the closely related compound 2-deoxy-5-azacytidine (decitabine) [92].
Interestingly, DNA demethylation responses of azacytidine and decitabine drugs are not restricted to the inhibition of DNA methylation [93]. Schaefer et al., using RNA bisulfite sequencing, showed that azacytidine, but not decitabine, inhibited cytosine-38 methylation of tRNA(Asp), a major substrate of DNMT2 [93]. They found that azacytidine caused a substantially stronger metabolic effect than decitabine in all cancer cell lines tested that is consistent with the effect of this drug on RNA metabolism [93]. In this context, it is interesting that 5-azacytidine has been shown to inhibit HIV-1 infectivity [94]. Molecular examination of the mechanism by which 5-azacytidine inhibits HIV-1 revealed that 5-azacytidine exerts antiviral activity through its incorporation into both viral RNA and DNA [95]. The antiviral activity of the derivative 5-aza-2′-deoxycytidine was detected using other retroviruses such as HTLV-1 [96]. Decitabine was successfully used in a trial to reduce HIV infectivity [97]. Moreover, azacytidine showed antiviral activity against Rift Valley fever virus (RVFV) [91], human metapneumovirus (HMPV) [98], A/H5N1 [99], IAV subtype H3N2 (A/H3N2), laboratory-adapted IAV H1N1 strains [100], EBV [101], and HSV-1 [102].
Zebularine (pyrimidin-2-one ribonucleoside), a cytidine analog that acts as a DNA demethylase inhibitor as well as a cytidine deaminase inhibitor, can be also considered as an antiviral drug [89]. However, the complex metabolism of zebularine and its limited DNA incorporation may restrain its applications in antiviral therapies. Indeed, zebularine is less potent than 5-azacytidine and its successful action requires higher doses for inhibition of DNMT activity [103].
Sinefungin (A9145) and its related metabolite, A9145C, are analogs of the methyl donor SAM and inhibitors of RNA and DNA methyltransferases [104]. Sinefungin (A9145) and its related metabolite, A9145C, were also found to be potent inhibitors of Newcastle disease virus (NDV), currently named Avian avulavirus 1 (AAvV-1) [105], Feline herpesvirus type 1 (FeHV-1) [106], vesicular stomatitis virus (VSV) [107], ZIKV [108], DENV [109], and the West Nile virus (WNV) [110]. It is worthwhile to mention that sinefungin has been identified as an inhibitor of 2′-O-RNA SARS-CoV-2 methyltransferase [111] and may electrostatically interact with the 2′-OH and N3 groups of adenosine moiety of RNA substrate thus mimicking the methyl transfer reaction of SAM substrate [112].
Tea polyphenol epigallocatechin-3-gallate (EGCG) is also another promising inhibitor of DNA/RNA methyltransferases. It was shown that EGCG can inhibit DNA methyltransferase and reactivate methylation-silenced genes in cancer cell lines [113].
Halby et al. developed a convergent synthetic pathway, starting from a protected bromomethylcytosine derivative, to synthesize transition state analogs of DNA methyltransferases [114]. However, they found low activity of seven 5-methylcytosine-adenosine compounds against hDNMT1, hDNMT3Acat, TRDMT1, and other human and viral RNA methyltransferases. Nevertheless, this research paves the way for the conception of new inhibitors based on the m5C scaffold.
7. Conclusions and Perspectives
In summary, we hypothesize that inhibitors of m5C RNA methyltransferases can be used in the future to modulate virus-mediated effects in host cells. However, several issues need to be addressed, namely, efficiency, selectivity, bioavailability, and biocompatibility of putative inhibitors in biological systems. The side effects of their use can be also a serious problem since these compounds can inhibit the enzyme activity of both virus-infected and virus-free cells. For instance, the use of 5-aza-2′-deoxycytidine can result in thrombocytopenia and anemia and less frequently can lead to congestive cardiac failure and tachycardia (http://sideeffects.embl.de). Moreover, it has been documented that high dose of azacytidine or 5-aza-2′-deoxycytidine may cause renal dysfunction [115] and spermatogenesis abnormalities [116] as well as may promote teratogenic effects during early embryogenesis [117]. Efficiency can be improved by molecular modeling of analogs in such way that the analogs are more effective than m5C in the binding to the catalytic sites of enzymes and the analogs are better matched structurally that may result in permanent inhibition and degradation of these enzymes [114]. Another problem may be the degradation of the analogs by extracellular and intracellular enzymes. Therefore, it is also important to develop systems that would prevent the degradation of the analogs before their targeted delivery. The selectivity of the inhibitors of m5C RNA methyltransferases can be improved by the addition of functional groups to the analogs [118]. A good solution would be to use the conjugates of m5C analogs based on the mechanism of cationic carriers that would allow for more efficient transport. Techniques for obtaining the oligonucleotide analogs with cationic backbone linkages were previously proposed [119]. Another well-established approach for nucleic acid delivery is based on cationic lipid-nucleic acid complexes [120,121]. The advantages of these systems for therapeutic applications include low toxicity and non-immunogenicity, ease of production, and the potential of transferring large amount of nucleic acids or oligonucleotides into the cells. Moreover, the addition of chemical groups to analogs can improve their bioavailability, e.g., during oral administration [122]. Improving the effectiveness of 5-azacytidine by increasing its bioavailability to human cells was also achieved by elaidic acid esterification [123]. An alternative delivery system for m5C analogs was also proposed, namely mesoporous silica nanoparticles [124]. In this view, the delivery of m5C RNA methyltransferase inhibitors can be improved by the use of nanocapsules that may serve as a promising technology for future antiviral therapies. The unique structure of nanocapsules may allow for proper loading of nucleoside analogs, oligonucleotides with nucleoside analogs, tag-like ligands, receptors, additional dyes, or other compounds that may support the monitoring and control of drug release. More studies are needed to better characterize the molecular bases of the biological action and therapeutic effects of m5C RNA methyltransferase inhibitors and their putative side effects in vitro and in vivo.
Abbreviations
| A/H5N1 | influenza A virus subtype H5N1 |
| A3G | apolipoprotein B mRNA editing enzyme catalytic subunit 3G (cytidine deaminase) |
| AAvV-1 | avian avulavirus 1 |
| ANG | angiogenin |
| APOBEC | apolipoprotein B editing complex |
| APOER2 | apolipoprotein E receptor 2 |
| CpG | 5′C—phosphate—G3′ |
| Cys | cysteine |
| DCV | Drosophila C virus |
| DENV | dengue virus |
| DNA | deoxyrybonucleic acid |
| DNMT2 | DNA methyltransferase 2 |
| dsRNA | double-stranded RNA |
| EBOV | Ebola virus |
| EBV | Epstein-Barr virus |
| EGCG | epigallocatechin-3-gallate |
| eRNA | enhancer RNA |
| FeHV-1 | feline herpesvirus type 1 |
| GEO | Gene Expression Omnibus |
| Glu | glutamic acid |
| Gly | glycine |
| HBV | hepatitis B virus |
| HCV | hepatitis C virus |
| hDNMT1 | human DNA methyltransferase 1 |
| hDNMT3Acat | human DNA methyltransferase 3A catalytic domain |
| HIV-1 | human immunodeficiency virus 1 |
| HMPV | human metapneumovirus |
| hnRNPK | heterogeneous nuclear ribonucleoprotein K |
| HPV | human papillomavirus |
| HSV-1 | herpes simplex virus 1 |
| HTLV | human T-lymphotropic virus |
| IAV | influenza A virus |
| IFN-β | interferon beta |
| KSHV | Kaposi’s sarcoma-associated herpesvirus |
| Leu | leucine |
| lncRNA | long non-coding RNA |
| m5C | 5-methylcytosine |
| m6A | N6-methyladenosine |
| MDA-5 | melanoma differentiation-associated protein 5 |
| Met | methionine |
| miRNA | microRNA |
| MLV | murine leukemia virus |
| mRNA | messenger RNA |
| mt tRNA | mitochondrial tRNA |
| NDV | Newcastle disease virus |
| Nop2 | nucleolar protein 2 |
| Nsp3 | non-structural protein 3 |
| NSUN | NOL1/NOP2/SUN domain |
| nt | nucleotide |
| PGC-1α | peroxisome proliferator-activated receptor gamma coactivator 1-alpha |
| PKR | RNA-dependent protein kinase |
| P-TEFb | positive transcription elongation factor b |
| Rcm1 | rRNA (cytosine-C5-)-methyltransferase |
| RNA | ribonucleic acid |
| rRNA | ribosomal RNA |
| RSV | respiratory syncytial virus |
| RVFV | Rift Valley fever virus |
| SAM | S-adenosylmethionine |
| SARS-Bat SRBD | SARS-CoV-like virus isolated from bats that contains the spike-protein receptor-binding domain from wild type SARS-CoV Urbani |
| SARS-CoV | severe acute respiratory syndrome coronavirus |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| snRNA | small nuclear RNA |
| +ssRNA | positive-sense single-stranded RNA |
| -ssRNA | negative-sense single-stranded RNA |
| ssRNA | single-stranded RNA |
| svRNA | small non-coding RNA fragments derived from vtRNA |
| svRNA | small viral RNA |
| TAR | transactivation response element |
| THOC4/ALYREF | THO complex subunit 4/Aly/REF export factor |
| Thr | threonine |
| tiRNA | tRNA-derived stress-induced RNA |
| TRDMT1 | tRNA methyltransferase 1 |
| TREX | transcription export complex |
| tRFs | tRNA-derived non-coding fragments |
| Trm4 | multisite-specific tRNA:m5C-methyltransferase |
| tRNA | transfer RNA |
| UHPLC | ultrahigh-performance liquid chromatography |
| UPLC-MS/MS | ultra-performance liquid chromatography and tandem mass spectrometry |
| VSV | vesicular stomatitis virus |
| vtRNA | small vault RNA |
| WNV | West Nile virus |
| ZIKV | Zika virus |
Author Contributions
Conceptualization, M.W. and A.L.; writing—original draft preparation, M.W., P.S., M.D., and A.L.; writing—review and editing, M.W. and A.L.; visualization, M.W.; supervision, M.W., A.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Boccaletto P., A Machnicka M., Purta E., Piątkowski P., Bagiński B., Wirecki T.K., De Crécy-Lagard V., Ross R., A Limbach P., Kotter A., et al. MODOMICS: A database of RNA modification pathways. 2017 update. Nucleic Acids Res. 2018;46:D303–D307. doi: 10.1093/nar/gkx1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Desrosiers R., Friderici K., Rottman F. Identification of Methylated Nucleosides in Messenger RNA from Novikoff Hepatoma Cells. Proc. Natl. Acad. Sci. USA. 1974;71:3971–3975. doi: 10.1073/pnas.71.10.3971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Amort T., Soulière M.F., Wille A., Jia X.-Y., Fiegl H., Wörle H., Micura R., Lusser A. Long non-coding RNAs as targets for cytosine methylation. RNA Biol. 2013;10:1002–1008. doi: 10.4161/rna.24454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Amort T., Rieder D., Wille A., Khokhlova-Cubberley D., Riml C., Trixl L., Jia X.-Y., Micura R., Lusser A. Distinct 5-methylcytosine profiles in poly(A) RNA from mouse embryonic stem cells and brain. Genome Biol. 2017;18:1–16. doi: 10.1186/s13059-016-1139-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lewis C.J., Pan T., Kalsotra A. RNA modifications and structures cooperate to guide RNA–protein interactions. Nat. Rev. Mol. Cell Biol. 2017;18:202–210. doi: 10.1038/nrm.2016.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bohnsack M.T., Höbartner C., Bohnsack M.T. Eukaryotic 5-methylcytosine (m5C) RNA Methyltransferases: Mechanisms, Cellular Functions, and Links to Disease. Genes. 2019;10:102. doi: 10.3390/genes10020102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schaefer M., Pollex T., Hanna K., Lyko F. RNA cytosine methylation analysis by bisulfite sequencing. Nucleic Acids Res. 2008;37:e12. doi: 10.1093/nar/gkn954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Edelheit S., Schwartz S., Mumbach M.R., Wurtzel O., Sorek R. Transcriptome-Wide Mapping of 5-methylcytidine RNA Modifications in Bacteria, Archaea, and Yeast Reveals m5C within Archaeal mRNAs. PLoS Genet. 2013;9:e1003602. doi: 10.1371/journal.pgen.1003602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Khoddami V., Cairns B.R. Identification of direct targets and modified bases of RNA cytosine methyltransferases. Nat. Biotechnol. 2013;31:458–464. doi: 10.1038/nbt.2566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hussain S., Aleksic J., Blanco S., Dietmann S., Frye M. Characterizing 5-methylcytosine in the mammalian epitranscriptome. Genome Biol. 2013;14:215. doi: 10.1186/gb4143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Trixl L., Lusser A. The dynamic RNA modification 5-methylcytosine and its emerging role as an epitranscriptomic mark. Wiley Interdiscip. Rev. RNA. 2018;10:e1510. doi: 10.1002/wrna.1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dang W., Xie Y., Cao P., Xin S., Wang J., Li S., Li Y., Lu J. N6-Methyladenosine and Viral Infection. Front. Microbiol. 2019;10:417. doi: 10.3389/fmicb.2019.00417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Manners O., Baquero-Perez B., Whitehouse A. m6A: Widespread regulatory control in virus replication. Biochim. Biophys. Acta (BBA)-Bioenerg. 2019;1862:370–381. doi: 10.1016/j.bbagrm.2018.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Daffis S., Szretter K.J., Schriewer J., Li J., Youn S., Errett J., Lin T.-Y., Schneller S.W., Zust R., Dong H., et al. 2′-O methylation of the viral mRNA cap evades host restriction by IFIT family members. Nat. Cell Biol. 2010;468:452–456. doi: 10.1038/nature09489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pereira-Montecinos C., Valiente-Echeverría F., Soto-Rifo R. Epitranscriptomic regulation of viral replication. Biochim. Biophys. Acta (BBA)-Bioenerg. 2017;1860:460–471. doi: 10.1016/j.bbagrm.2017.02.002. [DOI] [PubMed] [Google Scholar]
- 16.Reid R., Greene P.J., Santi D.V. Exposition of a family of RNA m5C methyltransferases from searching genomic and proteomic sequences. Nucleic Acids Res. 1999;27:3138–3145. doi: 10.1093/nar/27.15.3138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu Y., Santi D.V. m5C RNA and m5C DNA methyl transferases use different cysteine residues as catalysts. Proc. Natl. Acad. Sci. USA. 2000;97:8263–8265. doi: 10.1073/pnas.97.15.8263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.King M.Y., Redman K.L. RNA Methyltransferases Utilize Two Cysteine Residues in the Formation of 5-Methylcytosine. Biochemistry. 2002;41:11218–11225. doi: 10.1021/bi026055q. [DOI] [PubMed] [Google Scholar]
- 19.Bourgeois G., Ney M., Gaspar I., Aigueperse C., Schaefer M., Kellner S., Helm M., Motorin Y. Eukaryotic rRNA Modification by Yeast 5-Methylcytosine-Methyltransferases and Human Proliferation-Associated Antigen p120. PLoS ONE. 2015;10:e0133321. doi: 10.1371/journal.pone.0133321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gkatza N.A., Castro C., Harvey R.F., Heiß M., Popis M.C., Blanco S., Bornelöv S., Sajini A.A., Gleeson J.G., Griffin J.L., et al. Cytosine-5 RNA methylation links protein synthesis to cell metabolism. PLoS Biol. 2019;17:e3000297. doi: 10.1371/journal.pbio.3000297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Auxilien S., Guérineau V., Szweykowska-Kulinska Z., Golinelli-Pimpaneau B. The human tRNA m5C methyltransferase Misu is multisite-specific. RNA Biol. 2012;9:1331–1338. doi: 10.4161/rna.22180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Van Haute L., Lee S.-Y., McCann B.J., A Powell C., Bansal D., Vasiliauskaitė L., Garone C., Shin S., Kim J.-S., Frye M., et al. NSUN2 introduces 5-methylcytosines in mammalian mitochondrial tRNAs. Nucleic Acids Res. 2019;47:8720–8733. doi: 10.1093/nar/gkz559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brzezicha B., Schmidt M., Makałowska I., Jarmołowski A., Pieńkowska J., Szweykowska-Kulińska Z. Identification of human tRNA:m5C methyltransferase catalysing intron-dependent m5C formation in the first position of the anticodon of the. Nucleic Acids Res. 2006;34:6034–6043. doi: 10.1093/nar/gkl765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen X., Li A., Sun B.-F., Yang Y., Han Y.-N., Yuan X., Chen R.-X., Wei W.-S., Liu Y., Gao C.-C., et al. 5-methylcytosine promotes pathogenesis of bladder cancer through stabilizing mRNAs. Nat. Cell Biol. 2019;21:978–990. doi: 10.1038/s41556-019-0361-y. [DOI] [PubMed] [Google Scholar]
- 25.Haag S., Sloan K.E., Ranjan N., Warda A.S., Kretschmer J., Blessing C., Hubner B., Seikowski J., Dennerlein S., Rehling P., et al. NSUN3 and ABH1 modify the wobble position of mt-tRNAMet to expand codon recognition in mitochondrial translation. EMBO J. 2016;35:2104–2119. doi: 10.15252/embj.201694885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nakano S., Suzuki T., Kawarada L., Iwata H., Asano K. NSUN3 methylase initiates 5-formylcytidine biogenesis in human mitochondrial tRNA(Met) Nat. Chem. Biol. 2016;12:546–551. doi: 10.1038/nchembio.2099. [DOI] [PubMed] [Google Scholar]
- 27.Metodiev M.D., Spåhr H., Polosa P.L., Meharg C., Becker C., Altmueller J., Habermann B., Larsson N.-G., Ruzzenente B. NSUN4 Is a Dual Function Mitochondrial Protein Required for Both Methylation of 12S rRNA and Coordination of Mitoribosomal Assembly. PLoS Genet. 2014;10:e1004110. doi: 10.1371/journal.pgen.1004110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sharma S., Yang J., Watzinger P., Kötter P., Entian K.-D. Yeast Nop2 and Rcm1 methylate C2870 and C2278 of the 25S rRNA, respectively. Nucleic Acids Res. 2013;41:9062–9076. doi: 10.1093/nar/gkt679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Janin M., Ortiz-Barahona V., De Moura M.C., Martínez-Cardús A., Llinàs-Arias P., Soler M., Nachmani D., Pelletier J., Schumann U., Calleja-Cervantes M.E., et al. Epigenetic loss of RNA-methyltransferase NSUN5 in glioma targets ribosomes to drive a stress adaptive translational program. Acta Neuropathol. 2019;138:1053–1074. doi: 10.1007/s00401-019-02062-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Heissenberger C., Liendl L., Nagelreiter F., Gonskikh Y., Yang G., Stelzer E.M., Krammer T.L., Micutkova L., Vogt S., Kreil D.P., et al. Loss of the ribosomal RNA methyltransferase NSUN5 impairs global protein synthesis and normal growth. Nucleic Acids Res. 2019;47:11807–11825. doi: 10.1093/nar/gkz1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Haag S., Warda A.S., Kretschmer J., Günnigmann M.A., Höbartner C., Bohnsack M.T. NSUN6 is a human RNA methyltransferase that catalyzes formation of m5C72 in specific tRNAs. RNA. 2015;21:1532–1543. doi: 10.1261/rna.051524.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu R.-J., Long T., Li J., Li H., Wang E.-D. Structural basis for substrate binding and catalytic mechanism of a human RNA:m5C methyltransferase NSun6. Nucleic Acids Res. 2017;45:6684–6697. doi: 10.1093/nar/gkx473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Long T., Li J., Li H., Zhou M., Zhou X.-L., Liu R.-J., Wang E.-D. Sequence-specific and Shape-selective RNA Recognition by the Human RNA 5-Methylcytosine Methyltransferase NSun6. J. Biol. Chem. 2016;291:24293–24303. doi: 10.1074/jbc.M116.742569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Aguilo F., Li S., Balasubramaniyan N., Sancho A., Benko S., Zhang F., A Vashisht A., Rengasamy M., Andino B., Chen C.-H., et al. Deposition of 5-Methylcytosine on Enhancer RNAs Enables the Coactivator Function of PGC-1α. Cell Rep. 2016;14:479–492. doi: 10.1016/j.celrep.2015.12.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Goll M.G., Kirpekar F., Maggert K.A., Yoder J.A., Hsieh C.-L., Zhang X., Golic K.G., Jacobsen S.E., Bestor T.H. Methylation of tRNAAsp by the DNA Methyltransferase Homolog Dnmt2. Science. 2006;311:395–398. doi: 10.1126/science.1120976. [DOI] [PubMed] [Google Scholar]
- 36.Schaefer M., Lyko F. Lack of evidence for DNA methylation of Invader4 retroelements in Drosophila and implications for Dnmt2-mediated epigenetic regulation. Nat. Genet. 2010;42:920–921. doi: 10.1038/ng1110-920. [DOI] [PubMed] [Google Scholar]
- 37.Cui W., Pizzollo J., Han Z., Marcho C., Zhang K., Mager J. Nop2 is required for mammalian preimplantation development. Mol. Reprod. Dev. 2015;83:124–131. doi: 10.1002/mrd.22600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hussain S., Tuorto F., Menon S., Blanco S., Cox C., Flores J.V., Watt S., Kudo N.R., Lyko F., Frye M. The Mouse Cytosine-5 RNA Methyltransferase NSun2 Is a Component of the Chromatoid Body and Required for Testis Differentiation. Mol. Cell. Biol. 2013;33:1561–1570. doi: 10.1128/MCB.01523-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tuorto F., Liebers R., Musch T., Schaefer M., Hofmann S., Kellner S., Frye M., Helm M., Stoecklin G., Lyko F. RNA cytosine methylation by Dnmt2 and NSun2 promotes tRNA stability and protein synthesis. Nat. Struct. Mol. Biol. 2012;19:900–905. doi: 10.1038/nsmb.2357. [DOI] [PubMed] [Google Scholar]
- 40.Zhang T., Chen P., Li W., Sha S., Wang Y., Yuan Z., Shen B., Chen L. Cognitive deficits in mice lacking Nsun5, a cytosine-5 RNA methyltransferase, with impairment of oligodendrocyte precursor cells. Glia. 2019;67:688–702. doi: 10.1002/glia.23565. [DOI] [PubMed] [Google Scholar]
- 41.Selmi T., Hussain S., Dietmann S., Heiss M., Carter J.-M., Dennison R., Flad S., Huang Y.-L., Kellner S., Borneloev S., et al. Sequence- and structure-specific cytosine-5 mRNA methylation by NSUN6. bioRxiv. 2020 doi: 10.1101/2020.10.01.320036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Harris T., Marquez B., Suarez S., Schimenti J. Sperm Motility Defects and Infertility in Male Mice with a Mutation in Nsun7, a Member of the Sun Domain-Containing Family of Putative RNA Methyltransferases1. Biol. Reprod. 2007;77:376–382. doi: 10.1095/biolreprod.106.058669. [DOI] [PubMed] [Google Scholar]
- 43.Dubin D.T., Stollar V. Methylation of Sindbis virus “26S” messenger RNA. Biochem. Biophys. Res. Commun. 1975;66:1373–1379. doi: 10.1016/0006-291X(75)90511-2. [DOI] [PubMed] [Google Scholar]
- 44.Dubin D.T., Stollar V., HsuChen C.-C., Timko K., Guild G.M. Sindbis virus messenger RNA: The 5′-termini and methylated residues of 26 and 42 S RNA. Virology. 1977;77:457–470. doi: 10.1016/0042-6822(77)90471-8. [DOI] [PubMed] [Google Scholar]
- 45.Sommer S., Salditt-Georgieff M., Bachenheimer S., Darnell J., Furuichi Y., Morgan M., Shatkin A. The methylation of adenovirus-specific nuclear and cytoplasmic RNA. Nucleic Acids Res. 1976;3:749–766. doi: 10.1093/nar/3.3.749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Courtney D.G., Chalem A., Bogerd H.P., Law B.A., Kennedy E.M., Holley C.L., Cullen B.R. Extensive Epitranscriptomic Methylation of A and C Residues on Murine Leukemia Virus Transcripts Enhances Viral Gene Expression. mBio. 2019;10:e01209-19. doi: 10.1128/mBio.01209-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Courtney D., Tsai K., Bogerd H.P., Kennedy E.M., Law B.A., Emery A., Swanstrom R., Holley C.L., Cullen B.R. Epitranscriptomic Addition of m5C to HIV-1 Transcripts Regulates Viral Gene Expression. SSRN Electron. J. 2019;26:217. doi: 10.1016/j.chom.2019.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rima B.K., McFerran N.V. Dinucleotide and stop codon frequencies in single-stranded RNA viruses. J. Gen. Virol. 1997;78:2859–2870. doi: 10.1099/0022-1317-78-11-2859. [DOI] [PubMed] [Google Scholar]
- 49.Karlin S., Doerfler W., Cardon L.R. Why is CpG suppressed in the genomes of virtually all small eukaryotic viruses but not in those of large eukaryotic viruses? J. Virol. 1994;68:2889–2897. doi: 10.1128/JVI.68.5.2889-2897.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hoelzer K., Shackelton L.A., Parrish C.R. Presence and role of cytosine methylation in DNA viruses of animals. Nucleic Acids Res. 2008;36:2825–2837. doi: 10.1093/nar/gkn121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cheng X., Virk N., Chen W., Ji S., Ji S., Sun Y., Wu X. CpG Usage in RNA Viruses: Data and Hypotheses. PLoS ONE. 2013;8:e74109. doi: 10.1371/journal.pone.0074109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Matyasek R., Kovarik A. Mutation patterns of human SARS-COV-2 and Bat RATG13 coronavirus genomes are strongly biased towards C>U transitions, indicating rapid evolution in their hosts. Genes. 2020;11:761. doi: 10.3390/genes11070761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Viehweger A., Krautwurst S., Lamkiewicz K., Madhugiri R., Ziebuhr J., Hölzer M., Marz M. Direct RNA nanopore sequencing of full-length coronavirus genomes provides novel insights into structural variants and enables modification analysis. Genome Res. 2019;29:1545–1554. doi: 10.1101/gr.247064.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chiang C.-M., D K., Jy L., Js Y., Jw K., Vn K., H C. Faculty Opinions recommendation of The Architecture of SARS-CoV-2 Transcriptome. Fac. Opin. Post Publ. Peer Rev. Biomed. Lit. 2020;181:914. doi: 10.3410/f.737803948.793574094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Taiaroa G., Rawlinson D., Featherstone L., Pitt M., Caly L., Druce J., Purcell D., Harty L., Tran T., Roberts J., et al. Direct RNA sequencing and early evolution of SARS-CoV-2 2020. bioRxiv. 2020;2020:976167. [Google Scholar]
- 56.Sawicki S.G., Sawicki D.L. Coronaviruses use Discontinuous Extension for Synthesis of Subgenome-Length Negative Strands. Adv. Exp. Med. Biol. 1995;380:499–506. doi: 10.1007/978-1-4615-1899-0_79. [DOI] [PubMed] [Google Scholar]
- 57.Zúñiga S., Sola I., Alonso S., Enjuanes L. Sequence Motifs Involved in the Regulation of Discontinuous Coronavirus Subgenomic RNA Synthesis. J. Virol. 2004;78:980–994. doi: 10.1128/JVI.78.2.980-994.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sawicki S.G., Sawicki D.L., Siddell S.G. A Contemporary View of Coronavirus Transcription. J. Virol. 2006;81:20–29. doi: 10.1128/JVI.01358-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Antzin-Anduetza I., Mahiet C., Granger L.A., Odendall C., Swanson C.M. Increasing the CpG dinucleotide abundance in the HIV-1 genomic RNA inhibits viral replication. Retrovirology. 2017;14:1–18. doi: 10.1186/s12977-017-0374-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wasson M.K., Borkakoti J., Kumar A., Biswas B., Vivekanandan P. The CpG dinucleotide content of the HIV-1 envelope gene may predict disease progression. Sci. Rep. 2017;7:1–8. doi: 10.1038/s41598-017-08716-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Trus I., Udenze D., Berube N., Wheler C., Martel M.-J., Gerdts V., Karniychuk U. CpG-Recoding in Zika Virus Genome Causes Host-Age-Dependent Attenuation of Infection With Protection Against Lethal Heterologous Challenge in Mice. Front. Immunol. 2020;10:3077. doi: 10.3389/fimmu.2019.03077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Durdevic Z., Schaefer M. Dnmt2 methyltransferases and immunity: An ancient overlooked connection between nucleotide modification and host defense? BioEssays. 2013;35:1044–1049. doi: 10.1002/bies.201300088. [DOI] [PubMed] [Google Scholar]
- 63.Durdevic Z., Mobin M.B., Hanna K., Lyko F., Schaefer M. The RNA Methyltransferase Dnmt2 Is Required for Efficient Dicer-2-Dependent siRNA Pathway Activity in Drosophila. Cell Rep. 2013;4:931–937. doi: 10.1016/j.celrep.2013.07.046. [DOI] [PubMed] [Google Scholar]
- 64.Lee J.S., Tabata K., Twu W.-I., Rahman S., Kim H.S., Yu J.B., Jee M.H., Bartenschlager R., Jang S.K. RACK1 mediates rewiring of intracellular networks induced by hepatitis C virus infection. PLoS Pathog. 2019;15:e1008021. doi: 10.1371/journal.ppat.1008021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Stavrou S., Ross S.R. APOBEC3 Proteins in Viral Immunity. J. Immunol. 2015;195:4565–4570. doi: 10.4049/jimmunol.1501504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bishop K.N., Holmes R.K., Sheehy A.M., Davidson N.O., Cho S.-J., Malim M.H. Cytidine Deamination of Retroviral DNA by Diverse APOBEC Proteins. Curr. Biol. 2004;14:1392–1396. doi: 10.1016/j.cub.2004.06.057. [DOI] [PubMed] [Google Scholar]
- 67.Beale R.C., Petersen-Mahrt S.K., Watt I.N., Harris R.S., Rada C., Neuberger M.S. Comparison of the Differential Context-dependence of DNA Deamination by APOBEC Enzymes: Correlation with Mutation Spectra in vivo. J. Mol. Biol. 2004;337:585–596. doi: 10.1016/j.jmb.2004.01.046. [DOI] [PubMed] [Google Scholar]
- 68.Kim E.-Y., Lorenzo-Redondo R., Little S.J., Chung Y.-S., Phalora P.K., Berry I.M., Archer J., Penugonda S., Fischer W., Richman D.D., et al. Human APOBEC3 Induced Mutation of Human Immunodeficiency Virus Type-1 Contributes to Adaptation and Evolution in Natural Infection. PLoS Pathog. 2014;10:e1004281. doi: 10.1371/journal.ppat.1004281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wijesinghe P., Bhagwat A.S. Efficient deamination of 5-methylcytosines in DNA by human APOBEC3A, but not by AID or APOBEC3G. Nucleic Acids Res. 2012;40:9206–9217. doi: 10.1093/nar/gks685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.McIntyre W., Netzband R., Bonenfant G., Biegel J.M., Miller C., Fuchs G., Henderson E., Arra M., Canki M., Fabris D., et al. Positive-sense RNA viruses reveal the complexity and dynamics of the cellular and viral epitranscriptomes during infection. Nucleic Acids Res. 2018;46:5776–5791. doi: 10.1093/nar/gky029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schwarz K.B. Oxidative stress during viral infection: A review. Free Radic. Biol. Med. 1996;21:641–649. doi: 10.1016/0891-5849(96)00131-1. [DOI] [PubMed] [Google Scholar]
- 72.Sloan K.E., Bohnsack M.T., Watkins N.J. The 5S RNP Couples p53 Homeostasis to Ribosome Biogenesis and Nucleolar Stress. Cell Rep. 2013;5:237–247. doi: 10.1016/j.celrep.2013.08.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kong W., Biswas A., Zhou D., Fiches G., Fujinaga K., Santoso N., Zhu J. Nucleolar protein Nop2/Nsun1 suppresses HIV-1 transcription and promotes viral latency by competing with TAT for TAR binding and methylation. PLoS Pathog. 2020;16:e1008430. doi: 10.1371/journal.ppat.1008430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Shlomai A., Rechtman M.M., Burdelova E.O., Zilberberg A., Hoffman S., Solar I., Fishman S., Halpern Z., Sklan E.H. The metabolic regulator PGC-1α links hepatitis C virus infection to hepatic insulin resistance. J. Hepatol. 2012;57:867–873. doi: 10.1016/j.jhep.2012.06.021. [DOI] [PubMed] [Google Scholar]
- 75.Thiagarajan D., Dev R.R., Khosla S. The DNA methyltranferase Dnmt2 participates in RNA processing during cellular stress. Epigenetics. 2011;6:103–113. doi: 10.4161/epi.6.1.13418. [DOI] [PubMed] [Google Scholar]
- 76.Dev R.R., Ganji R., Singh S.P., Mahalingam S., Banerjee S., Khosla S. Cytosine methylation by DNMT2 facilitates stability and survival of HIV-1 RNA in the host cell during infection. Biochem. J. 2017;474:2009–2026. doi: 10.1042/BCJ20170258. [DOI] [PubMed] [Google Scholar]
- 77.Mitchell H.D., Eisfeld A.J., Sims A.C., McDermott J.E., Matzke M.M., Webb-Robertson B.J., Tilton S.C., Tchitchek N., Josset L., Li C., et al. A network integration approach to predict conserved regulators related to pathogenicity of influenza and SARS-COV respiratory viruses. PLoS ONE. 2013;8:e69374. doi: 10.1371/journal.pone.0069374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Linder A., Hornung V. Mitochondrial dsRNA: A New DAMP for MDA5. Dev. Cell. 2018;46:530–532. doi: 10.1016/j.devcel.2018.08.019. [DOI] [PubMed] [Google Scholar]
- 79.Dhir A., Dhir S., Borowski L.S., Jimenez L., Teitell M., Rötig A., Crow Y.J., Rice G.I., Duffy D., Tamby C., et al. Mitochondrial double-stranded RNA triggers antiviral signalling in humans. Nat. Cell Biol. 2018;560:238–242. doi: 10.1038/s41586-018-0363-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cheng J.X., Chen L., Li Y., Cloe A., Yue M., Wei J., Watanabe K.A., Shammo J.M., Anastasi J., Shen Q.J., et al. RNA cytosine methylation and methyltransferases mediate chromatin organization and 5-azacytidine response and resistance in leukaemia. Nat. Commun. 2018;9:1163. doi: 10.1038/s41467-018-03513-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ivanov P., Emara M.M., Villen J., Gygi S.P., Anderson P. Angiogenin-Induced tRNA Fragments Inhibit Translation Initiation. Mol. Cell. 2011;43:613–623. doi: 10.1016/j.molcel.2011.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Goodarzi H., Liu X., Nguyen H.C., Zhang S., Fish L., Tavazoie S.F. Endogenous tRNA-Derived Fragments Suppress Breast Cancer Progression via YBX1 Displacement. Cell. 2015;161:790–802. doi: 10.1016/j.cell.2015.02.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gebetsberger J.V., Wyss L., Mleczko A.M., Reuther J., Polacek N. A tRNA-derived fragment competes with mRNA for ribosome binding and regulates translation during stress. RNA Biol. 2017;14:1364–1373. doi: 10.1080/15476286.2016.1257470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Deng J., Ptashkin R.N., Chen Y., Cheng Z., Liu G., Phan T., Deng X., Zhou J., Lee I., Lee Y.S., et al. Respiratory Syncytial Virus Utilizes a tRNA Fragment to Suppress Antiviral Responses Through a Novel Targeting Mechanism. Mol. Ther. 2015;23:1622–1629. doi: 10.1038/mt.2015.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Yang X., Yang Y., Sun B.-F., Chen Y.-S., Xu J.-W., Lai W.-Y., Li A., Wang X., Bhattarai D.P., Xiao W., et al. 5-methylcytosine promotes mRNA export — NSUN2 as the methyltransferase and ALYREF as an m5C reader. Cell Res. 2017;27:606–625. doi: 10.1038/cr.2017.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Boyne J.R., Colgan K.J., Whitehouse A. Recruitment of the complete HTREX complex is required for Kaposi’s sarcoma-associated herpesvirus intronless mRNA nuclear export and virus replication. PLoS Pathog. 2008;4:e1000194. doi: 10.1371/journal.ppat.1000194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hussain S., Sajini A.A., Blanco S., Dietmann S., Lombard P., Sugimoto Y., Paramor M., Gleeson J.G., Odom D.T., Ule J., et al. NSun2-Mediated Cytosine-5 Methylation of Vault Noncoding RNA Determines Its Processing into Regulatory Small RNAs. Cell Rep. 2013;4:255–261. doi: 10.1016/j.celrep.2013.06.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Li F., Chen Y., Zhang Z., Ouyang J., Wang Y., Yan R., Huang S., Gao G.F., Guo G., Chen J.-L. Robust expression of vault RNAs induced by influenza a virus plays a critical role in suppression of PKR-mediated innate immunity. Nucleic Acids Res. 2015;43:10321–10337. doi: 10.1093/nar/gkv1078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.McAllister C.S., Taghavi N., Samuel C.E. Protein Kinase PKR Amplification of Interferon β Induction Occurs through Initiation Factor eIF-2α-mediated Translational Control. J. Biol. Chem. 2012;287:36384–36392. doi: 10.1074/jbc.M112.390039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Morales L., Oliveros J.C., Fernandez-Delgado R., Tenoever B.R., Enjuanes L., Sola I. SARS-CoV-Encoded Small RNAs Contribute to Infection-Associated Lung Pathology. Cell Host Microbe. 2017;21:344–355. doi: 10.1016/j.chom.2017.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Ianevski A., Zusinaite E., Kuivanen S., Strand M., Lysvand H., Teppor M., Kakkola L., Paavilainen H., Laajala M., Kallio-Kokko H., et al. Novel activities of safe-in-human broad-spectrum antiviral agents. Antivir. Res. 2018;154:174–182. doi: 10.1016/j.antiviral.2018.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Šorm F., Pískala A., Čihák A., Veselý J. 5-Azacytidine, a new, highly effective cancerostatic. Cell. Mol. Life Sci. 1964;20:202–203. doi: 10.1007/BF02135399. [DOI] [PubMed] [Google Scholar]
- 93.Schaefer M., Hagemann S., Hanna K., Lyko F. Azacytidine Inhibits RNA Methylation at DNMT2 Target Sites in Human Cancer Cell Lines. Cancer Res. 2009;69:8127–8132. doi: 10.1158/0008-5472.CAN-09-0458. [DOI] [PubMed] [Google Scholar]
- 94.Bouchard J., Walker M.C., Leclerc J.M., Lapointe N., Beaulieu R., Thibodeau L. 5-azacytidine and 5-azadeoxycytidine inhibit human immunodeficiency virus type 1 replication in vitro. Antimicrob. Agents Chemother. 1990;34:206–209. doi: 10.1128/AAC.34.2.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Dapp M.J., Clouser C.L., Patterson S., Mansky L.M. 5-Azacytidine Can Induce Lethal Mutagenesis in Human Immunodeficiency Virus Type 1. J. Virol. 2009;83:11950–11958. doi: 10.1128/JVI.01406-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Diamantopoulos P.T., Michael M., Benopoulou O., Bazanis E., Tzeletas G., Meletis J., Vayopoulos G., Viniou N.-A. Antiretroviral activity of 5-azacytidine during treatment of a HTLV-1 positive myelodysplastic syndrome with autoimmune manifestations. Virol. J. 2012;9:1. doi: 10.1186/1743-422X-9-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Clouser C.L., Patterson S.E., Mansky L.M. Exploiting Drug Repositioning for Discovery of a Novel HIV Combination Therapy. J. Virol. 2010;84:9301–9309. doi: 10.1128/JVI.01006-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bösl K., Ianevski A., Than T.T., Andersen P.I., Kuivanen S., Teppor M., Zusinaite E., Dumpis U., Vitkauskiene A., Cox R.J., et al. Common Nodes of Virus–Host Interaction Revealed Through an Integrated Network Analysis. Front. Immunol. 2019;10:2186. doi: 10.3389/fimmu.2019.02186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Huang F., Zhang C., Liu Q., Zhao Y., Zhang Y., Qin Y., Li X., Li C., Zhou C., Jin N., et al. Identification of amitriptyline HCl, flavin adenine dinucleotide, azacitidine and calcitriol as repurposing drugs for influenza A H5N1 virus-induced lung injury. PLoS Pathog. 2020;16:e1008341. doi: 10.1371/journal.ppat.1008341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pauly M.D., Lauring A.S. Effective Lethal Mutagenesis of Influenza Virus by Three Nucleoside Analogs. J. Virol. 2015;89:3584–3597. doi: 10.1128/JVI.03483-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chan A.T.C., Tao Q., Robertson K.D., Flinn I.W., Mann R.B., Klencke B., Kwan W.H., Leung T.W.-T., Johnson P.J., Ambinder R.F. Azacitidine Induces Demethylation of the Epstein-Barr Virus Genome in Tumors. J. Clin. Oncol. 2004;22:1373–1381. doi: 10.1200/JCO.2004.04.185. [DOI] [PubMed] [Google Scholar]
- 102.Takimoto K. Reactivation and mutagenesis of herpes virus in 5-azacytidine-treated monkey kidney cells. Mutat. Res. Repair Rep. 1985;146:9–13. doi: 10.1016/0167-8817(85)90049-5. [DOI] [PubMed] [Google Scholar]
- 103.Rao S.P., Rechsteiner M.P., Berger C., Sigrist J.A., Nadal D., Bernasconi M. Zebularine reactivates silenced E-cadherin but unlike 5-azacytidine does not induce switching from latent to lytic Epstein-Barr virus infection in Burkitt’s lymphoma Akata cells. Mol. Cancer. 2007;6:3. doi: 10.1186/1476-4598-6-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Yebra M.J., Sánchez J., Martin C.G., Hardisson C., Barbes C. The effect of sinefungin and synthetic analogues on RNA and DNA methyltransferases from Streptomyces. J. Antibiot. 1991;44:1141–1147. doi: 10.7164/antibiotics.44.1141. [DOI] [PubMed] [Google Scholar]
- 105.Pugh C.S., Borchardt R.T., O Stone H. Sinefungin, a potent inhibitor of virion mRNA(guanine-7-)-methyltransferase, mRNA(nucleoside-2′-)-methyltransferase, and viral multiplication. J. Biol. Chem. 1978;253:4075–4077. [PubMed] [Google Scholar]
- 106.Kuroda Y., Yamagata H., Nemoto M., Inagaki K., Tamura T., Maeda K. Antiviral effect of sinefungin on in vitro growth of feline herpesvirus type 1. J. Antibiot. 2019;72:981–985. doi: 10.1038/s41429-019-0234-4. [DOI] [PubMed] [Google Scholar]
- 107.Li J., Chorba J.S., Whelan S.P. Vesicular Stomatitis Viruses Resistant to the Methylase Inhibitor Sinefungin Upregulate RNA Synthesis and Reveal Mutations That Affect mRNA Cap Methylation. J. Virol. 2007;81:4104–4115. doi: 10.1128/JVI.02681-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hercik K., Brynda J., Nencka R., Boura E. Structural basis of Zika virus methyltransferase inhibition by sinefungin. Arch. Virol. 2017;162:2091–2096. doi: 10.1007/s00705-017-3345-x. [DOI] [PubMed] [Google Scholar]
- 109.Lim S.P., Sonntag L.S., Noble C., Nilar S.H., Ng R.H., Zou G., Monaghan P., Chung K.Y., Dong H., Liu B., et al. Small Molecule Inhibitors That Selectively Block Dengue Virus Methyltransferase. J. Biol. Chem. 2010;286:6233–6240. doi: 10.1074/jbc.M110.179184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chen H., Liu L., Jones S.A., Banavali N., Kass J., Li Z., Zhang J., Kramer L.D., Ghosh A.K., Li H. Selective inhibition of the West Nile virus methyltransferase by nucleoside analogs. Antivir. Res. 2013;97:232–239. doi: 10.1016/j.antiviral.2012.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Krafcikova P., Silhan J., Nencka R., Boura E. Structural analysis of the SARS-COV-2 methyltransferase complex involved in RNA cap creation bound to sinefungin. Nat. Commun. 2020;11:3717. doi: 10.1038/s41467-020-17495-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Mahalapbutr P., Kongtaworn N., Rungrotmongkol T. Structural insight into the recognition of S-adenosyl-L-homocysteine and sinefungin in SARS-COV-2 NSP16/NSP10 RNA cap 2′-O-methyltransferase. Comput. Struct. Biotechnol. J. 2020;18:2757–2765. doi: 10.1016/j.csbj.2020.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Fang M.Z., Wang Y., Ai N., Hou Z., Sun Y., Lu H., Welsh W., Yang C.S. Tea polyphenol (-)-epigallocatechin-3-gallate inhibits DNA methyltransferase and reactivates methylation-silenced genes in cancer cell lines. Cancer Res. 2003;63:7563–7570. [PubMed] [Google Scholar]
- 114.Halby L., Marechal N., Pechalrieu D., Cura V., Franchini D.-M., Faux C., Alby F., Troffer-Charlier N., Kudithipudi S., Jeltsch A., et al. Hijacking DNA methyltransferase transition state analogues to produce chemical scaffolds for PRMT inhibitors. Philos. Trans. R. Soc. B Biol. Sci. 2018;373:20170072. doi: 10.1098/rstb.2017.0072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kintzel P.E. Anticancer Drug—Induced Kidney Disorders. Drug Saf. 2001;24:19–38. doi: 10.2165/00002018-200124010-00003. [DOI] [PubMed] [Google Scholar]
- 116.Oakes C.C., Kelly T.L.J., Robaire B., Trasler J.M. Adverse Effects of 5-Aza-2′-Deoxycytidine on Spermatogenesis Include Reduced Sperm Function and Selective Inhibition of de Novo DNA Methylation. J. Pharmacol. Exp. Ther. 2007;322:1171–1180. doi: 10.1124/jpet.107.121699. [DOI] [PubMed] [Google Scholar]
- 117.Branch S., Francis B.M., Brownie C.F., Chernoff N. Teratogenic effects of the demethylating agent 5-aza-2′-deoxycytidine in the Swiss Webster mouse. Toxicology. 1996;112:37–43. doi: 10.1016/0300-483X(96)88183-2. [DOI] [PubMed] [Google Scholar]
- 118.Valderrama J.A., González M.F., Colonelli P., Vásquez-Velásquez D. Design and Synthesis of Angucyclinone 5-Aza Analogues. Synlett. 2006;2006:2777–2780. doi: 10.1055/s-2006-950254. [DOI] [Google Scholar]
- 119.Meng M., Ducho C. Oligonucleotide analogues with cationic backbone linkages. Beilstein J. Org. Chem. 2018;14:1293–1308. doi: 10.3762/bjoc.14.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Wang J., Ayano E., Maitani Y., Kanazawa H. Enhanced cellular uptake and gene silencing activity of siRNA using temperature-responsive polymer-modified liposome. Int. J. Pharm. 2017;523:217–228. doi: 10.1016/j.ijpharm.2017.03.035. [DOI] [PubMed] [Google Scholar]
- 121.Safinya C.R., Ewert K.K., Majzoub R.N., Leal C. Cationic liposome–nucleic acid complexes for gene delivery and gene silencing. New J. Chem. 2014;38:5164–5172. doi: 10.1039/C4NJ01314J. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Ziemba A., Hayes E., Freeman B.B., Ye T., Pizzorno G. Development of an Oral Form of Azacytidine: 2′3′5′Triacetyl-5-Azacytidine. Chemother. Res. Pract. 2011;2011:1–9. doi: 10.1155/2011/965826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Brueckner B., Rius M., Markelova M.R., Fichtner I., Hals P.-A., Sandvold M.L., Lyko F. Delivery of 5-Azacytidine to Human Cancer Cells by Elaidic Acid Esterification Increases Therapeutic Drug Efficacy. Mol. Cancer Ther. 2010;9:1256–1264. doi: 10.1158/1535-7163.MCT-09-1202. [DOI] [PubMed] [Google Scholar]
- 124.Cheng J., Ding Q., Wang J., Deng L., Yang L., Tao L., Lei H., Lu S. 5-Azacytidine delivered by mesoporous silica nanoparticles regulates the differentiation of P19 cells into cardiomyocytes. Nanoscale. 2016;8:2011–2021. doi: 10.1039/C5NR08560H. [DOI] [PubMed] [Google Scholar]