Abstract
Background
The stability of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) on human skin remains unknown, considering the hazards of viral exposure to humans. We generated a model that allows the safe reproduction of clinical studies on the application of pathogens to human skin and elucidated the stability of SARS-CoV-2 on human skin.
Methods
We evaluated the stability of SARS-CoV-2 and influenza A virus (IAV), mixed with culture medium or upper respiratory mucus, on human skin surfaces and the dermal disinfection effectiveness of 80% (weight/weight) ethanol against SARS-CoV-2 and IAV.
Results
SARS-CoV-2 and IAV were inactivated more rapidly on skin surfaces than on other surfaces (stainless steel/glass/plastic); the survival time was significantly longer for SARS-CoV-2 than for IAV (9.04 hours [95% confidence interval, 7.96– 10.2 hours] vs 1.82 hours [1.65–2.00 hours]). IAV on other surfaces was inactivated faster in mucus versus medium conditions, while SARS-CoV-2 showed similar stability in the mucus and medium; the survival time was significantly longer for SARS-CoV-2 than for IAV (11.09 hours [10.22–12.00 hours] vs 1.69 hours [1.57–1.81 hours]). Moreover, both SARS-CoV-2 and IAV in the mucus/medium on human skin were completely inactivated within 15 seconds by ethanol treatment.
Conclusions
The 9-hour survival of SARS-CoV-2 on human skin may increase the risk of contact transmission in comparison with IAV, thus accelerating the pandemic. Proper hand hygiene is important to prevent the spread of SARS-CoV-2 infections.
Keywords: SARS-CoV-2, human skin, stability, influenza A virus, hand hygiene
The survival time of SARS-CoV-2 on the human skin was ~9 hours, significantly longer than that of influenza A virus (~1.8 hours). The longer survival of SARS-CoV-2 on skin increases contact-transmission risk; however, hand hygiene can reduce this risk.
Studies on the control of the novel coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), have progressed rapidly worldwide. Numerous studies on the stability of SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV) indicate that these coronaviruses have relatively higher stability compared with that of enveloped viruses, such as influenza A virus (IAV) [1–5]. Furthermore, the stability of SARS-CoV-2 on various surfaces was reported recently, providing essential information regarding the control of infection [6, 7]. Contact transmission through human skin is considered a significant risk factor in the spread of SARS-CoV-2 [8, 9]; hence, it is critical to have information about the stability (survival time) of SARS-CoV-2 on human skin to develop approaches to prevent contact transmission. However, it is dangerous to apply highly pathogenic and infectious agents, including SARS-CoV-2, directly to human skin. Moreover, even though skin samples can be procured from surgical procedures, the amount of tissue obtained is small, which is a deterrent in the construction of models to conduct experiments with high reproducibility [10]. Due to these limitations, the stability of highly pathogenic and infectious agents on human skin remains unknown.
To overcome the abovementioned limitations, we generated a model to evaluate the stability of pathogens on human skin obtained from forensic autopsy specimens. Human skin (particularly the epidermis) is characterized by slower deterioration after death compared with other organs, and the collected skin can be used for grafting even 24 hours after death [11–13]. Therefore, this evaluation model, using skin collected from autopsy specimens (~1 day after death), could preserve skin functions and successfully model the in vivo conditions. Furthermore, at institutions performing forensic autopsy, fresh skin samples collected up to 1 day after death can be stably supplied for research, thus facilitating the generation of a high-quality and reproducible model [14].
In this study, we compared the stability of SARS-CoV-2 on human skin with that of IAV, which is a common virus transmitted through droplets and contact transmission worldwide. First, we confirmed whether the constructed model could replicate the in vivo skin conditions accurately. Thereafter, we evaluated the stability of SARS-CoV-2 and IAV in culture medium and on the surface of stainless steel, borosilicate glass, polystyrene, and human skin using this model. Moreover, the stability of SARS-CoV-2 and IAV, mixed with mucus from the upper respiratory tract, was evaluated because the viral particles of SARS-CoV-2 and IAV that adhere to objects and the skin are vehiculated by infectious body fluids (mucus). Finally, we evaluated the effectiveness of 80% (weight/weight [w/w]) ethanol in the disinfection of human skin exposed to SARS-CoV-2 or IAV.
METHODS
Viruses and Cells
Madin-Darby canine kidney (MDCK) cells were purchased from the RIKEN BioResource Center Cell Bank (Ibaragi, Japan) and were cultured in minimal essential medium (Sigma Aldrich, St Louis, MO) supplemented with 10% fetal bovine serum and standard antibiotics (penicillin/streptomycin). Influenza A virus (PR8; A/Puerto Rico/8/1934; H1N1) was cultured in MDCK cells and stored as a working stock at −80°C. Virus titers were measured via focus-forming assays in MDCK cells, as previously described, and expressed as focus-forming units (FFU) [15, 16]. Specifically, at 12 hours postinfection, cells were fixed with phosphate-buffered saline (PBS) containing 4% paraformaldehyde and 0.1% Triton X-100 for 30 minutes at room temperature. To detect influenza virus antigens, the cells were then stained with a rabbit polyclonal antibody that recognizes influenza virus NP and M1 proteins. Antibody binding to viral proteins was detected with an Alexa Fluor 488–conjugated secondary antibody (Molecular Probes, Carlsbad, CA) diluted 1:500 in PBS, and cells containing virus antigens were counted under a fluorescence microscope.
VeroE6/TMPRSS2 cells, expressing the transmembrane serine protease TMPRSS2, were purchased from the Japanese Collection of Research Bioresources Cell Bank (Osaka, Japan) and were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Sigma Aldrich) supplemented with 5% fetal bovine serum and G418 (Nacalai Tesque, Kyoto, Japan) [17, 18]. SARS-CoV-2 (JPN/TY/WK-521) was generously provided by the National Institute of Infectious Diseases (Tokyo, Japan). The virus was cultured in VeroE6/TMPRSS2 cells and stored as a working stock at −80°C. The titers of the virus were measured in terms of 50% tissue culture infectious dose (TCID50) in VeroE6/TMPRSS2 cells. Specifically, 4 days after inoculation, the cytopathic effect in each well was scored under a microscope and the TCID50 was calculated [3, 6, 17].
Both viruses were concentrated and purified as follows: 96 hours postinfection, the culture medium was harvested and centrifuged for 10 minutes at 2500 × g at 4°C to eliminate the cellular debris. Virions in the supernatant were sedimented through a 20% (w/w) sucrose cushion in PBS through ultracentrifugation at 28 000 rpm for 2.5 hours at 4°C in a Beckman SW28 rotor (Beckman Coulter Inc., Brea, CA) [19].
Collection of Sputum Samples and Preparation of Mucus
Mucus samples (sputum samples ≥2 g) were obtained from 3 individuals diagnosed with an acute upper respiratory tract infection in 2019. Individuals younger than 20 years of age with chronic respiratory illness or those taking expectorants were excluded. The titration of viruses was performed on MDCK and VeroE6/TMPRSS2 cells to confirm that no active viruses (IAV or SARS-CoV-2) were present. Furthermore, to completely inactivate the infectious pathogens derived from the individuals, the samples were irradiated with ultraviolet light (15 mJ/cm2). Bubbles were eliminated from the irradiated samples through centrifugation at a low speed to avoid the destruction of the mucous structure [20, 21].
Collection of Human Skin From Autopsy Specimens and Preparation of the Virus Stability Evaluation Model
Human skin was sampled from forensic autopsy specimens obtained from the Department of Forensic Medicine, Kyoto Prefectural University of Medicine. Abdominal skin autopsy samples from subjects aged 20–70 years, with a postmortem duration of approximately 1 day, were excised into rectangles with dimensions greater than 4 × 8 cm2 and used for subsequent evaluation [14].
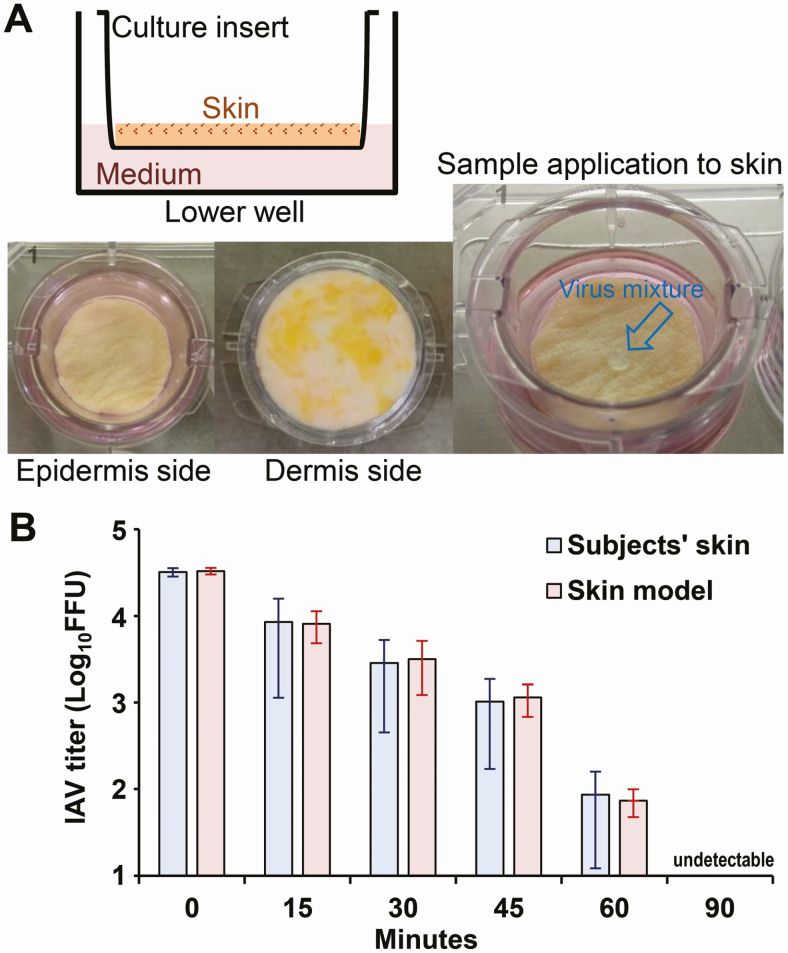
Thereafter, we developed a model to accurately evaluate the stability of the viruses on human skin. This model was designed such that the skin sample did not deteriorate because of drying even after long-term incubation. Most subcutaneous adipose tissue was rapidly obliterated. The treated skin (mainly the epidermal and dermal layers) was rinsed with PBS and placed onto a culture insert with a membrane of 8.0-µm pore size (Corning, Corning, NY). The culture inserts were placed into 6- or 12-well plates containing 1.0 mL of DMEM (Figure 1A).
Figure 1.
Outline of the pathogen stability evaluation model and its reproducibility. The pathogen stability evaluation model was constructed using human skin collected from forensic autopsy specimens (A). To evaluate the reproducibility of the model, IAV was applied to the 6 model skin samples and to the hand skin of 6 subjects (amount of virus: 1.0 × 105 FFU), and the titer of the remaining viruses on the skin was measured. The 95% confidence interval (red bars) of the viable virus titer on the model skin at each elapsed time was within the 95% confidence interval (blue bars) of the viable virus titer on the skin of live individuals (B). Abbreviations: FFU, focus-forming units; IAV, influenza A virus.
Evaluation of Viral Stability on Various Surfaces
Viral survival was assessed on the surface of stainless steel, borosilicate glass, polystyrene, and the human skin model. Influenza A virus or SARS-CoV-2 were mixed with DMEM or mucus and applied in 5-µL aliquots to each surface (1.0 × 105 FFU or 1.0 × 105 TCID50, respectively). Each sample was incubated in a controlled environment (25°C and 45–55% relative humidity) for 0–96 hours. Thereafter, the residual viruses on the surface were recovered in 1 mL of DMEM and titrated [3, 22–25]. Three independent experiments were performed for each measurement, and the results are expressed as the mean ± standard error of the mean.
To evaluate the reproducibility of the human skin model, the stability of IAV on the hands of 6 subjects, who provided informed consent, was analyzed. Influenza A virus was mixed with DMEM and applied in 5-µL aliquots to the skin of the subjects’ hands (1.0 × 105 FFU). Each sample was incubated under a controlled room environment (25°C and 45–55% relative humidity) for 0–1.5 hours. Thereafter, the residual viruses were recovered from the surface in 1 mL of DMEM and titrated [26].
Time measurements were started immediately after the virus aliquots were applied to the surfaces; zero-hour incubation indicates virus recovery immediately after applying the virus mixture to each surface.
Evaluation of the Efficacy of an Ethanol-based Disinfectant Against SARS-CoV-2 and IAV on Human Skin
SARS-CoV-2 or IAV were mixed with DMEM or mucus and applied in 5-µL aliquots to human skin (1.0 × 105 TCID50 or 1.0 × 105 FFU, respectively). Skin samples were then incubated at 25°C, under a relative humidity of 45–55%, for 30 minutes. Thereafter, 95 µL of 80% (w/w) ethanol was applied to the skin and incubated for 15 seconds prior to neutralization by dilution with 900 µL of DMEM; the remaining viruses were then titrated [20]. Three independent experiments were performed for each measurement and the results are expressed as the mean ± standard error of the mean.
Ethical Considerations
The study protocol, including the sample collection procedures, was reviewed and approved by the Institutional Review Board of the Kyoto Prefectural University of Medicine (ERB-C-1593).
Statistical Analysis
Data were analyzed using the GraphPad Prism 7 software (GraphPad, Inc, La Jolla, CA). The elapsed time was defined as the explanatory variable (X-axis), and the log virus titers of IAV or SARS-CoV-2 were defined as the explained variable (Y-axis). A least-squares linear regression analysis with a logarithmic link function was performed to generate regression curves for both viruses. The measurement limits of the titers of IAV and SARS-CoV-2 were 101 FFU and 100.5 TCID50, respectively; therefore, the survival times of IAV and SARS-CoV-2 were defined as the X values when the Y values of the regression curves were 1.0 and 0.5, respectively. The half-life time of each log virus titer was determined from the slope of the respective regression line.
RESULTS
To validate the model, first we evaluated the stability of IAV on the skin using the newly generated skin model and compared it with that obtained in the context of live subjects’ hand skin (Figure 1A). The 95% confidence interval of the viable virus titers on the model skin at each elapsed time (15, 30, 45, and 60 minutes) was within the 95% confidence interval of the viable virus titers on the subjects’ skin: 3.91 log10 FFU (95% confidence interval, 3.68–4.06 log10 FFU) vs 3.93 log10 FFU (3.06–4.20 log10 FFU), 3.50 log10 FFU (3.09–3.71 log10 FFU) vs 3.46 log10 FFU (2.65–3.72 log10 FFU), 3.06 log10 FFU (2.83–3.21 log10 FFU) vs 3.01 log10 FFU (2.23–3.27 log10 FFU), and 1.87 log10 FFU (1.68–2.00 log10 FFU) vs 1.93 log10 FFU (1.08–2.20 log10 FFU), respectively (Figure 1B). These results indicate that the present evaluation model can replicate data obtained using the skin from live subjects’ hands accurately.
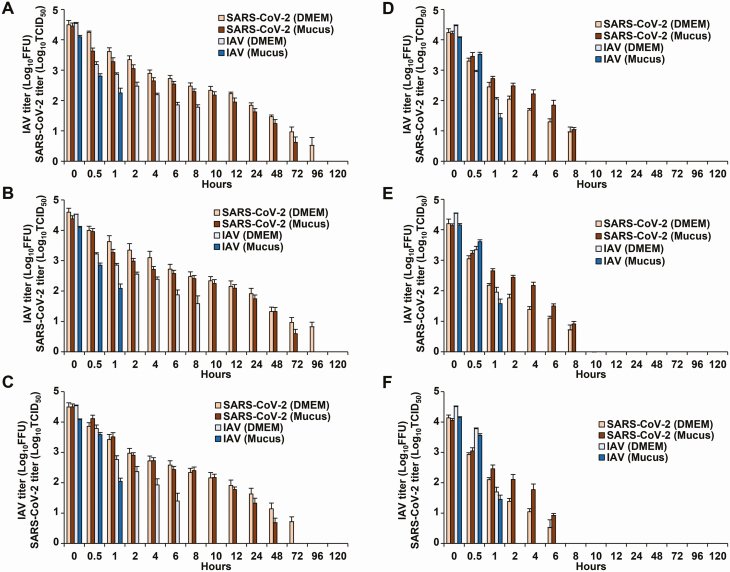
Next, the stability of SARS-CoV-2 and IAV, mixed in DMEM, was evaluated on the surface of stainless steel, borosilicate glass, and polystyrene. The survival time of SARS-CoV-2 was approximately 8-fold the survival time of IAV on these surfaces; SARS-CoV-2 also displayed higher stability. However, SARS-CoV-2 and IAV were more rapidly inactivated on all human skin surfaces (HS1, HS2, HS3) than on stainless steel, borosilicate glass, and polystyrene surfaces; the survival time and half-life time of SARS-CoV-2 and IAV were significantly shorter on human skin than those on stainless steel, borosilicate glass, and polystyrene (Figure 2, Table 1, Supplementary Figures 1–3). Moreover, SARS-CoV-2 on all human skin surfaces (HS1, HS2, HS3) displayed higher stability than IAV, and both the survival and half-life times of SARS-CoV-2 on skin were significantly longer than those of IAV (9.04 hours [95% confidence interval, 7.96–10.20 hours] vs 1.82 hours [1.65–2.00 hours] and 3.53 hours [3.02–4.16 hours] vs .80 hours [.72–.90 hours], respectively) (Table 1, Supplementary Figure 3).
Figure 2.
A–F, Fluctuations in the titer of SARS-CoV-2 and IAV surviving on the surface of stainless steel (A), borosilicate glass (B), polystyrene (C), and 3 skin samples (HS1 [D], HS2 [E], and HS3 [F]). SARS-CoV-2/IAV was mixed with DMEM or mucus and applied in 5-µL aliquots to each surface (amount of virus: 1.0 × 105 FFU or 1.0 × 105 TCID50, respectively). Each surface was incubated in a constant environment (temperature: 25°C; humidity: 45–55%) for 0–120 hours. The remaining viruses on the surface were then recovered in 1 mL of culture medium and titrated. For each measurement, 3 independent experiments were performed, and the results are expressed as the mean ± standard error of the mean. Bars referring to the data below the detection limit were omitted. See Supplementary Figures 1 and 2 for raw data. Abbreviations: DMEM, Dulbecco’s modified Eagle’s medium; FFU, focus-forming units; IAV, influenza A virus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TCID50, 50% tissue culture infectious dose.
Table 1.
Survival Time and Half-life Time of Viruses on Each Surface
| Survival Time,a Median (95% CI), Hours | Half-life Time,b Median (95% CI), Hours | |||||||
|---|---|---|---|---|---|---|---|---|
| IAV (DMEM) | SARS-CoV-2 (DMEM) | IAV (Mucus) | SARS-CoV-2 (Mucus) | IAV (DMEM) | SARS-CoV-2 (DMEM) | IAV (Mucus) | SARS-CoV-2 (Mucus) | |
| Stainless steel | 11.56 (10.11–13.22) | 84.29 (54.01–119.56) | 1.73 (1.57–1.91) | 64.51 (52.35–77.73) | 6.78 (5.84–7.97) | 32.62 (16.80–56.68) | .86 (.76–.98) | 25.53 (18.45–34.24) |
| Borosilicate glass | 10.61 (9.18–12.27) | 85.74 (56.27–119.80) | 1.73 (1.58–1.88) | 61.23 (49.03–74.44) | 6.13 (5.22–7.29) | 33.24 (17.59–56.49) | .85 (.76–.96) | 23.63 (17.16–31.86) |
| Polystyrene | 6.07 (5.05–7.27) | 58.07 (37.76–81.95) | 1.96 (1.76–2.18) | 35.92 (29.58–42.67) | 3.04 (2.40–3.87) | 22.58 (11.64–41.24) | .91 (.80–1.04) | 13.17 (10.26–17.35) |
| Human skin (HS total) | 1.82 (1.65–2.00) | 9.04 (7.96–10.22) | 1.69 (1.57–1.81) | 11.09 (10.22–12.00) | .80 (.72–.90) | 3.53 (3.02–4.16) | .77 (.71–.84) | 4.16 (3.79–4.58) |
| HS1 | 1.81 (1.64–2.00) | 10.93 (8.95–13.10) | 1.66 (1.47–1.88) | 12.24 (10.64–13.94) | .82 (.73–.93) | 4.13 (3.29–5.28) | .77 (.66–.89) | 4.47 (3.83–5.26) |
| HS2 | 1.79 (1.50–2.13) | 9.45 (7.72–11.38) | 1.71 (1.51–1.94) | 12.2 (11.10–13.34) | .78 (.64–.98) | 3.75 (2.93–4.86) | .78 (.67–.91) | 4.51 (4.06–5.03) |
| HS3 | 1.86 (1.50–2.27) | 6.14 (4.91–7.53) | 1.69 (1.49–1.91) | 8.13 (6.85–9.51) | .79 (.63–1.04) | 2.36 (1.73–3.21) | .77 (.67–.90) | 3.13 (2.56–3.86) |
The elapsed time was defined as an explanatory variable (X-axis), and the log virus titer of IAV or SARS-CoV-2 was defined as an explained variable (Y-axis). A linear regression analysis with logarithmic link function was performed for each virus to create a curve of regression (see also Supplementary Figure 3).
Abbreviations: CI, confidence interval; DMEM, Dulbecco’s modified Eagle’s medium; FFU, focus-forming units; IAV, influenza A virus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TCID50, 50% tissue culture infectious dose.
aThe measurement limits of the titers of IAV and SARS-CoV-2 were 101 FFU and 100.5 TCID50, respectively; therefore, the survival times of IAV and SARS-CoV-2 were defined as the X values when the Y values of the regression curves were 1.0 and 0.5, respectively.
bThe half-life time of each log virus titer was calculated from the slope of each regression line.
Stability analysis of the viruses mixed with mucus from the upper respiratory tract revealed that, on stainless steel, borosilicate glass, and polystyrene surfaces, IAV was more rapidly inactivated in mucus than in DMEM, whereas the stability of SARS-CoV-2 in mucus and DMEM was similar (Figure 2, Supplementary Figures 1 and 2). Importantly, in the analysis of viruses mixed with mucus, the survival and half-life times of SARS-CoV-2 were also significantly longer than those of IAV on human skin (11.09 hours [10.22–12.00 hours] vs 1.69 hours [1.57–1.81 hours] and 4.16 hours [3.79–4.58 hours] vs .77 hours [.71–.84 hours], respectively) (Table 1, Supplementary Figure 3).
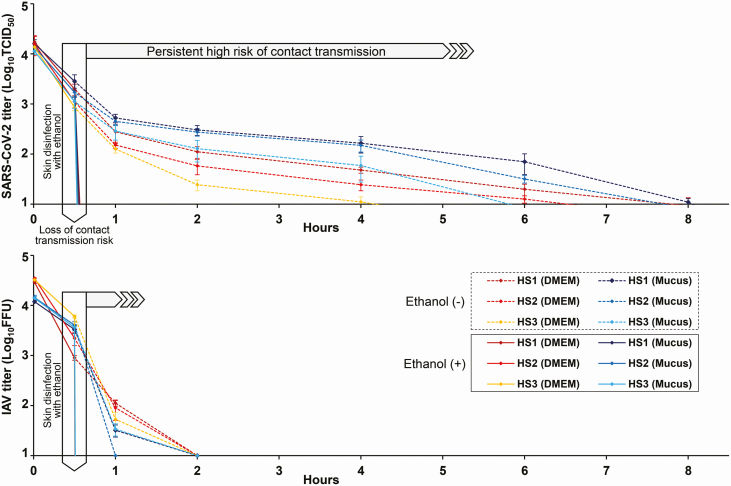
Finally, SARS-CoV-2 both in the context of mucus and DMEM was completely inactivated on all human skin surfaces (HS1, HS2, HS3) within 15 seconds upon treatment with 80% (w/w) ethanol. Furthermore, IAV was completely inactivated under the same evaluation conditions (Figure 3).
Figure 3.
Evaluation of the disinfection effectiveness of 80% (w/w) ethanol against SARS-CoV-2 (upper panel) and IAV (lower panel) on human skin. Thirty minutes after the mixture of the DMEM/mucus and SARS-CoV-2/IAV was applied to each skin surface (HS1/HS2/HS3), 80% ethanol was further applied to the skin surfaces for 15 seconds, followed by disinfectant inactivation via dilution with culture medium. The surviving viruses on the skin surfaces were then titrated. For comparison, the surviving viruses on the skin surfaces in the absence of ethanol were also titrated over time. For each measurement, 3 independent experiments were performed, and the results are expressed as mean ± standard error values. Abbreviations: DMEM, Dulbecco’s modified Eagle’s medium; IAV, influenza A virus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TCID50, 50% tissue culture infectious dose; w/w, weight/weight.
DISCUSSION
Since the collected skin can be used for skin grafting even 24 hours after death [11–13], this evaluation model, using skin autopsy specimens collected approximately 1 day after death, has the potential to preserve skin function and to faithfully model in vivo conditions. In actuality, the 95% confidence interval of the viable virus titers on the model skin at each elapsed time point was within the corresponding 95% confidence interval of the viable virus titers in the context of the live subjects’ hand skin, indicating that the constructed evaluation model can accurately replicate the in vivo conditions. Moreover, IAV was inactivated on the skin in approximately 1 hour, in concordance with previous reports [26, 27]. This model is, therefore, quite helpful for the evaluation of the stability of highly pathogenic and highly infectious agents, such as SARS-CoV-2, on human skin, as well as for the evaluation of their disinfection efficacy; we believe this new method has the potential to contribute greatly to the development of infection-control strategies in the future.
The survival time of SARS-CoV-2 was approximately 8-fold that of IAV on the surface of stainless steel, borosilicate glass, and polystyrene, displaying consequently higher stability than IAV. These results are similar to those of previous studies [2, 3, 6]. However, the survival and half-life times of SARS-CoV-2 and IAV were significantly shorter on human skin than those on the other surfaces, indicating that human skin is less suitable for the survival of viruses. The survival time of SARS-CoV-2 on skin was approximately 9 hours and was significantly longer than that of IAV (~1.8 hours). These results indicate that SARS-CoV-2 has a markedly higher stability on human skin than that of IAV.
Influenza A virus was inactivated more rapidly in mucus versus DMEM on the surface of stainless steel, borosilicate glass, and polystyrene; in fact, its survival time was drastically reduced. SARS-CoV-2 displayed the same level of stability in both mucus and DMEM, and the survival time and half-life were not significantly different. These results suggest that viral inactivators in the mucus from the upper respiratory tract are less effective against SARS-CoV-2 than they are against IAV.
Taken together, the determined long 9-hour survival time of SARS-CoV-2 on human skin may increase the risk of viral invasion in the body or its transmission from the skin to other surfaces, with a potential impact in the acceleration of the SARS-CoV-2 pandemic. However, SARS-CoV-2 in the mucus and DMEM was completely inactivated within 15 seconds of exposure to 80% (w/w) ethanol. Thus, appropriate hand hygiene using ethanol-based disinfectants leads to quick viral inactivation and may reduce the high risk of contact infections. On the contrary, since not only the virus stability but also the infectious dose and transmission route may greatly affect the risk of contact transmission, future research needs to focus on factors other than virus stability.
In this study, the analysis was performed using a least-squares linear regression analysis with a logarithmic link function instead of the normal linear regression. There may be multiple phases in the context of loss of infectivity, with a recognized multifactorial character. The speed of infectivity loss may change over time due to many different factors; in fact, we speculate that this is the main reason because the regular linear regression did not fit well. We will focus on some of these potential factors, such as humidity and temperature, and carry out further research in the future.
There were 3 main limitations to this study. First, only 1 SARS-CoV-2 strain and only 1 influenza strain were used. Furthermore, the PR8 strain was used as the influenza strain instead of a relevant clinical isolate. It is necessary to further increase the number of virus strains and proceed with the research in the future to understand if these results can be translated to human influenza and COVID-19. Second, only 3 skin samples from autopsy specimens and 3 mucus samples were used in this study. Since the number of samples is small, in future clinical studies we plan to increase the number of samples and evaluate the clinical background of patients. Third, an evaluation model using human skin harvested in the context of a forensic autopsy was used for the evaluation of virus stability and disinfecting effectiveness. It is necessary to demonstrate in the future that the skin surface in this model is similar to that of live skin. However, the application of highly pathogenic and infectious agents to the skin of subjects is dangerous and not feasible clinically, and there is no evaluation system at this stage that is more accurate and reproducible than this model. Of note, the inactivation of IAV in the context of autopsy skin samples and of live skin was similar, supporting the validity of our model.
In conclusion, this study shows that SARS-CoV-2 may have a higher risk of contact transmission than IAV because the first is much more stable on human skin than the former. These findings support the hypothesis that proper hand hygiene is important for the prevention of the spread of SARS-CoV-2. Thus, this study may contribute to the development of better control strategies in the context of COVID-19 to prevent the occurrence of the second or third waves of this pandemic.
Supplementary Material
Notes
Author contributions. Study concept and design, drafting of the manuscript, statistical analysis, and secured funding: R. H. Data acquisition: R. H., H. I., N. W., T. Y., R. B., and T. D. Data analysis and interpretation: R. H., Y. N., Y. I., and T. N. Administrative/technical/material support: R. H. and H. I. Study supervision: R. H. and T. N.
Acknowledgments. The authors thank Satista Co, Ltd, for assistance in the statistical analysis and Editage (www.editage.com) for English-language editing.
Financial support. This research was supported by Japan Agency for Medical Research and Development (AMED) (grant numbers JP20fk0108077 and JP20fk0108270) and JSPS KAKENHI (grant numbers JP18K16183 and JP18H03040). This research was also supported by grants from the Mochida Memorial Foundation for Medical and Pharmaceutical Research, the Uehara Memorial Foundation, the Ichiro Kanehara Foundation, the Takeda Science Foundation, and the Daiwa Securities Health Foundation.
Potential conflicts of interest. Y. I. reports grants and personal fees from Merck, Sharp & Dohme; Gilead Sciences Inc; AbbVie Inc; Mitsubishi Tanabe Pharma Corporation; Daiichi Sankyo Company, Ltd; Astellas Pharma Inc; EA Pharma Co, Ltd; Eisai Co, Ltd; Otsuka Pharmaceutical Co, Ltd; GlaxoSmithKline K.K.; Sumitomo Dainippon Pharma Co, Ltd; Takeda Pharmaceutical Company, Ltd; Chugai Pharmaceutical Co, Ltd; AstraZeneca K.K.; ASKA Pharmaceutical Co, Ltd; Asahi Kasei Pharma Corporation; Fujifilm Medical Co, Ltd; and Bristol-Myers Squibb, outside the submitted work; additionally, Y. I. reports grants from Novo Nordisk Pharma Ltd; Kyorin Pharmaceutical Company, Ltd; Merck Serono Co, Ltd; Nissan Chemical Industries Ltd; Shionogi & Co; Ltd; Japan Agency for Medical Research and Development; Japan Society for the Promotion of Science; and a Health and Labor Sciences Research Grant, outside the submitted work. Y. I. also reports personal fees from Janssen Pharmaceutical K.K.; Mylan Inc; Kowa Pharmaceutical Company, Ltd; Mochida Pharmaceutical Co, Ltd; Taiho Pharmaceutical Co, Ltd; Tsumura & Co; Zeria Pharmaceutical Co, Ltd; Taisho Toyama Pharmaceutical Co, Ltd; outside the submitted work; Y. I. also reports donation-funded department from NichiNichi Pharmaceutical Co, Ltd, outside the submitted work. R. H. reports the conclusion of a collaboration research contract and receipt of a collaboration research fund from Kao Corporation and Fuso Pharmaceutical Industries, outside the submitted work. All other authors report no potential conflicts. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1. Kampf G, Todt D, Pfaender S, Steinmann E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J Hosp Infect 2020; 104:246–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Otter JA, Donskey C, Yezli S, Douthwaite S, Goldenberg SD, Weber DJ. Transmission of SARS and MERS coronaviruses and influenza virus in healthcare settings: the possible role of dry surface contamination. J Hosp Infect 2016; 92:235–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. van Doremalen N, Bushmaker T, Munster VJ. Stability of Middle East respiratory syndrome coronavirus (MERS-CoV) under different environmental conditions. Euro Surveill 2013; 18:20590. [DOI] [PubMed] [Google Scholar]
- 4. Duan SM, Zhao XS, Wen RF, et al. ; SARS Research Team Stability of SARS coronavirus in human specimens and environment and its sensitivity to heating and UV irradiation. Biomed Environ Sci 2003; 16:246–55. [PubMed] [Google Scholar]
- 5. Weber TP, Stilianakis NI. Inactivation of influenza A viruses in the environment and modes of transmission: a critical review. J Infect 2008; 57:361–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med 2020; 382:1564–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ong SWX, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA 2020; 323:1610–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. World Health Organization. Infection prevention and control during health care when novel coronavirus (nCoV) infection is suspected. Available at: https://www.who.int/publications/i/item/10665-331495. Accessed 6 June 2020.
- 9. US Centers for Disease Control and Prevention. Prevention and control recommendations for patients with suspected or confirmed coronavirus disease 2019 (COVID-19) in healthcare settings. Available at: https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Finfection-control%2Fcontrol-recommendations.html. Accessed 6 June 2020.
- 10. Graham ML, Springthorpe VS, Sattar SA. Ex vivo protocol for testing virus survival on human skin: experiments with herpesvirus 2. Appl Environ Microbiol 1996; 62:4252–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Boekema BK, Boekestijn B, Breederveld RS. Evaluation of saline, RPMI and DMEM/F12 for storage of split-thickness skin grafts. Burns 2015; 41:848–52. [DOI] [PubMed] [Google Scholar]
- 12. Franchini M, Zanini D, Bosinelli A, et al. Evaluation of cryopreserved donor skin viability: the experience of the regional tissue bank of Verona. Blood Transfus 2009; 7:100–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hirel B, Watier E, Chesne C, Patoux-Pibouin M, Guillouzo A. Culture and drug biotransformation capacity of adult human keratinocytes from post-mortem skin. Br J Dermatol 1996; 134:831–6. [PubMed] [Google Scholar]
- 14. Akutsu T, Ikegaya H, Watanabe K, Miyasaka S. Immunohistochemical staining of skin-expressed proteins to identify exfoliated epidermal cells for forensic purposes. Forensic Sci Int 2019; 303:109940. [DOI] [PubMed] [Google Scholar]
- 15. Hirose R, Nakaya T, Naito Y, et al. Mechanism of human influenza virus RNA persistence and virion survival in feces: mucus protects virions from acid and digestive juices. J Infect Dis 2017; 216:105–9. [DOI] [PubMed] [Google Scholar]
- 16. Daidoji T, Watanabe Y, Ibrahim MS, et al. Avian influenza virus infection of immortalized human respiratory epithelial cells depends upon a delicate balance between hemagglutinin acid stability and endosomal pH. J Biol Chem 2015; 290:10627–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Matsuyama S, Nao N, Shirato K, et al. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc Natl Acad Sci USA 2020; 117:7001–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Matsuyama S, Nagata N, Shirato K, Kawase M, Takeda M, Taguchi F. Efficient activation of the severe acute respiratory syndrome coronavirus spike protein by the transmembrane protease TMPRSS2. J Virol 2010; 84:12658–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Bárcena M, Oostergetel GT, Bartelink W, et al. Cryo-electron tomography of mouse hepatitis virus: insights into the structure of the coronavirion. Proc Natl Acad Sci USA 2009; 106:582–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hirose R, Nakaya T, Naito Y, et al. Situations leading to reduced effectiveness of current hand hygiene against infectious mucus from influenza virus-infected patients. mSphere 2019; 4:e00474–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Hirose R, Nakaya T, Naito Y, et al. Viscosity is an important factor of resistance to alcohol-based disinfectants by pathogens present in mucus. Sci Rep 2017; 7:13186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Noyce JO, Michels H, Keevil CW. Inactivation of influenza A virus on copper versus stainless steel surfaces. Appl Environ Microbiol 2007; 73:2748–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Thomas Y, Vogel G, Wunderli W, et al. Survival of influenza virus on banknotes. Appl Environ Microbiol 2008; 74:3002–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Chan KH, Peiris JS, Lam SY, Poon LL, Yuen KY, Seto WH. The effects of temperature and relative humidity on the viability of the SARS coronavirus. Adv Virol 2011; 2011:734690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Rabenau HF, Cinatl J, Morgenstern B, Bauer G, Preiser W, Doerr HW. Stability and inactivation of SARS coronavirus. Med Microbiol Immunol 2005; 194:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Thomas Y, Boquete-Suter P, Koch D, Pittet D, Kaiser L. Survival of influenza virus on human fingers. Clin Microbiol Infect 2014; 20:O58–64. [DOI] [PubMed] [Google Scholar]
- 27. Mukherjee DV, Cohen B, Bovino ME, Desai S, Whittier S, Larson EL. Survival of influenza virus on hands and fomites in community and laboratory settings. Am J Infect Control 2012; 40:590–4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.