Abstract
Objectives
There was an outbreak of pediatric multisystem inflammation syndrome (PMIS) was observed in multiple countries recently, and this syndrome was suspected to be associated with SARS-CoV-2 infection. At present, there is still no standardized diagnostic criteria and treatment regimen for PMIS, while the etiology and pathogenesis still remain unclear.
Methods
We performed a systematic review on PubMed and Embase from the time of inception to June 24th 2020 in order to find relevant cases.
Results
There are seven studies included, and 80% of patients suffered persistent fever and 90% appeared gastrointestinal symptoms. IgG antibody against SARS-CoV-2 was positive on 81% of patients, while 37% of the patients were nucleic acid positive. C-reactive protein, IL-6 and PCT were elevated and intravenous immunoglobulin was a routine treatment for PMIS. There were more than half of patients required inotropic supports and mechanical ventilation were applied to 33% of patients. The median length of hospital stay was 10.66 days and 74% had admitted to accept intensive care.
Conclusions
Our study documented three common types of PMIS clinical presentation: persistent fever and gastrointestinal symptoms, shocked with heart dysfunction and Kawasaki disease-like syndrome. PMIS patients proved with a marked inflammatory state were possibly associated with SARS-CoV-2 infection.
Keywords: Pediatric Multi-system Inflammatory Syndrome, SARS-CoV-2, COVID-19
Background
The early situation of coronavirus disease 2019 (COVID-19) seemed to appear milder symptoms and lower fatality on younger patients (Juan et al., 2020, Tagarro et al., 2020, Zheng et al., 2020). As the pandemic continues, clusters of children presented with toxic shock symptoms and incomplete Kawasaki features were observed across Europe and the United States (Belhadjer et al., 2020, Riphagen et al., 2020, Toubiana et al., 2020a, 2020). This novel syndrome was named pediatric multi-system inflammatory syndrome (PMIS) or multisystem inflammatory syndrome in children (MIS-C).
There are many organizations like the World Health Organization (WHO), US Centers for Disease Control and Prevention (CDC), the Royal College of Paediatrics and Child Health (RCPCH) and the European Centre for Disease Prevention and Control, have defined and created criteria for this childhood inflammatory disorder as the COVID-19 pandemic was rampant over various countries (Whittaker et al., 2020). Since then, PMIS has been reported in New York, Paris and San Diego (Belhadjer et al., 2020, Toubiana et al., 2020a, Toubiana et al., 2020b, Whittaker et al., 2020). However, as the diagnostic criteria has not been unified, misdiagnosis and missed diagnosis still remain possible, meanwhile unclear definitions could bring confusion and difficult management. In addition, molecular evidence in pathogenesis remains highly insufficient in this recent onset of COVID-19 pediatric complication. To address the above-mentioned questions, we aim to collect current evidence on pediatric PMIS and provide insights into pathogenesis, hopefully, providing possible clues for further research and treatment at this moment.
Methods
We conducted a systematic literature search in PubMed and Embase according to the following terms: “coronavirus or COVID-19 or 2019-nCoV or SARS-CoV-2” and “Pediatric Multi-system Inflammatory Syndrome or Kawasaki-like disease”. Since PMIS was identified in May 2020, the time period was restricted to but not included the literature published prior to this time point. Available full texts and reference lists of relevant studies were reviewed. There was no language restriction to our research. The last update of the study was on June 24th, 2020.
Inclusion and exclusion criteria
There were two independent reviewers (Hua Zou, Juan Lu) screened all titles and abstracts in term of eligibility. Studies are eligible for inclusion if they met the following criteria: 1) patients were less than 18 years old, 2) patients with elevated inflammatory markers, 3) patients with no other obvious microbial causes of inflammation, including bacterial sepsis, staphylococcal or streptococcal shock syndromes. We excluded the following criterion: editorials, correspondence letters, review, qualitative studies and non-full text articles.
Data collection and quality assessment
After full-text screening of eligibility and review, there were three authors (Jingjing Liu, Josiah Hiu-yuen Wong, Chunli Li) extracted data solely. The following items were extracted from each study, including: author, journal, date of publication, study design, country, contact or travel history, clinical symptoms, laboratory results and prognosis. The quality of studies presented in meta-analysis was assessed by the Newcastle-Ottawa Scale (NOS). Articles at poor quality (score Z 0-3) were excluded.
Data synthesis and analysis
We presented the data with descriptive statistics and pooled available data for overall demonstration. Stata 15.0 was used for meta-analysis and drawing graph, and Q test was used to evaluate heterogeneity. If P > 0.1 and I2 <50%, a fixed effect model was used, otherwise a random effect model was chosen. Categorical variables adopted a double-arc sine conversion method and the combined effect was expressed by rates. Continuous variables were calculated by the average and standard errors and the combined effects were the average.
Assessment of bias
Egger precision weighted linear regression tests and funnel plots were used to test potential publication bias. If publication bias was presented, the trim and fill method would be performed and Rosenthal’s Failsafe N would be calculated to evaluate the effects.
Results
In our literature research, there are 26 potentially relevant articles identified, of which three were immediately excluded due to the duplication. After removing editorials, reviews and irrelevant studies, there were studies whose full texts were screened. The report to individual case were ruled out, which resulted in seven studies that were coincident with all criteria (Fig.1 ).
Fig. 1.
Flow diagram of the study selection process.
The summary to the included studies was presented in Table 1 , in which a total of 182 PMIS patients were included. All studies were retrospectively conducted and the majority of them were studied in France and England. The risks of bias ranged from 1.71 to 2.57; five studies scored >2, indicating a low risk of bias (Table 2 ). There was no difference in gender of research objects among these studies and the mean age was about 8.99 (95%CI 7.99–9.98).
Table 1.
Characteristics of the included studies.
| Study name | Country | Type of study | Sample size | Gender (Male) | Age, median (IQR), y | Shock cases for study | SARS-CoV-2 infection | ICU admission | Length of hospital stays | Mortality |
|---|---|---|---|---|---|---|---|---|---|---|
| Elizabeth Whittaker,2020 | England | Retrospective study | 58 | 25 | 9 (5.7-14) | 29 | 45 | 29 | - | 1 |
| Zahra Belhadjer,2020 | France and Switzerland | Retrospective study | 35 | 18 | 10 | 28 | 30 | 35 | 10(8-14) | 0 |
| Marion Grimaud,2020 | France | Retrospective study | 20 | 10 | 10(2.5-15) | 20 | 19 | 15 | - | 0 |
| Julie Toubiana,2020 | France | Retrospective study | 21 | 9 | 7.9(3.7-16.6) | 15 | 19 | 17 | 8(5-17) | 0 |
| Tristan Ramcharan,2020 | England | Retrospective study | 15 | 11 | 8.8(6.4-11.2) | 10 | 14 | 10 | 12(9-13) | 0 |
| Marie Pouletty,2020 | France | Retrospective study | 16 | 8 | 10(4.7-12.5) | 11 | 12 | 7 | 14(10-20) | 0 |
| Julie Toubiana,2020 | France | Retrospective study | 17 | 7 | 7.5(3.7-16.6) | 11 | 14 | 13 | 8(5-17) | 0 |
Table 2.
Quality assessment.
| First author | Population representative | Sample size adequate |
Confounders | Statistical analysis |
Missing data |
Methodology of the outcome |
Objective assessment |
OVERALL (0-3, higher score = lower risk of bias) |
|---|---|---|---|---|---|---|---|---|
| Elizabeth Whittaker,2020(Whittaker et al., 2020) | 3 | 3 | 3 | 3 | 2 | 1 | 3 | 2.57 |
| Zahra Belhadjer,2020(Belhadjer et al., 2020) | 1 | 3 | 2 | 3 | 2 | 3 | 2 | 2.28 |
| Marion Grimaud,2020(Grimaud et al., 2020) | 2 | 2 | 2 | 2 | 2 | 1 | 2 | 1.85 |
| Julie Toubiana,2020(Toubiana et al., 2020a) | 3 | 2 | 2 | 2 | 2 | 3 | 2 | 2.28 |
| Tristan Ramcharan,2020(Ramcharan et al., 2020) | 3 | 2 | 2 | 2 | 2 | 3 | 2 | 2.28 |
| Marie Pouletty,2020(Pouletty et al., 2020) | 3 | 2 | 3 | 3 | 2 | 2 | 3 | 2.57 |
| Julie Toubiana,2020(Toubiana et al., 2020a) | 2 | 2 | 0 | 2 | 2 | 2 | 2 | 1.71 |
Clinical Characteristics
In Table 3 , we summarized epidemiological and clinical features of the reported PMIS patients. The average age of the patients was 8.99 years (95% CI 7.99-9.98, I2 <0.01, P = 0.92). Major symptoms occurred to patients could be divided into three categories according to their clinical manifestation. The first category, into which most patients fell, was persistent fever (80%, 95% CI 0.44-1, I2 = 95.75, P < 0.01) and gastrointestinal symptoms (90%, 95% CI 0.71-1, I2 = 87.62, P < 0.01). The second category of symptoms was shock (74%, 95% CI 0.57-.088, I2 = 80.03, P < 0.01) and left ventricular dysfunction (70%, 95% CI 0.44-.091, I2 = 91.19, P < 0.01). The third category was the fulfilled diagnostic criteria for Kawasaki disease (KD) (17%, 95% CI 0.02-0.40, I2 = 90.92, P < 0.01). There was only 9% of patients appeared respiratory symptoms (95% CI 0-0.29, I2 = 91.08, P < 0.01) (Fig. 2 ).<-- -->
Table 3.
Characteristics of Patients through meta analysis.
| Variable | Total Patients(n) | Average | 95% CI | I2% | P value |
|---|---|---|---|---|---|
| Clinical Characteristics | |||||
| Age (years) | 147 | 8.99 | 7.99-9.98 | <0.01 | 0.92 |
| Gastrointestinal symptoms (n%) | 182 | 0.9 | 0.71-1 | 87.62 | <0.01 |
| Febrile and inflammatory (n%) | 182 | 0.8 | 0.44-1 | 95.75 | <0.01 |
| Shock (n%) | 182 | 0.74 | 0.57-.088 | 80.03 | <0.01 |
| left ventricular dysfunction (n%) | 182 | 0.70 | 0.44-.091 | 91.19 | <0.01 |
| KD (n%) | 182 | 0.17 | 0.02-0.40 | 90.92 | <0.01 |
| respiratory symptom (n%) | 182 | 0.09 | 0-0.29 | 91.08 | <0.01 |
| Risk factors and Laboratory Results | |||||
| Contact with Viral (n%) | 182 | 0.15 | 0.01-0.40 | 92.02 | <0.01 |
| Onset of Kawa-COVID-19 to hospitalization (days) | 71 | 5.9 | 5.55-6.25 | 31.30 | 0.23 |
| Nucleic acid positive of SARS Cov-2 (n%) | 182 | 0.37 | 0.26-0.50 | 59.94 | <0.01 |
| lgG of SARS Cov-2 (n%) | 182 | 0.81 | 0.74-0.86 | 34.25 | 0.17 |
| Lymphocyte (*10^9/l) | 98 | 0.92 | 0.74-1.11 | 6 | 0.36 |
| Platelet (*10^9/l) | 132 | 166.84 | 150.54-183.14 | 75.30 | 03 |
| CRP (mg/l) | 182 | 210.80 | 183-238.59 | 47.40 | 0.08 |
| IL-6 (pg/ml) | 49 | 138.63 | 103.78-173.48 | <0.01 | 0.71 |
| PCT (ng/ml) | 80 | 35.51 | 10.64-60.37 | <0.01 | 0.99 |
| Albumin (g/l) | 125 | 21.96 | 20.01-23.91 | 65.40 | 0.02 |
| Troponin (ng/l) | 180 | 60.21 | 21.88-95.54 | 15.70 | 0.31 |
| NT-proBNP (pg/ml) | 141 | 6526.48 | 1142.31-11910.66 | 95.20 | <0.01 |
| D-dimer (ng/ml) | 124 | 3588.13 | 1968.50-5207.77 | 76.80 | <0.01 |
| Treatments | |||||
| Intravenous immunoglobulin (n%) | 182 | 0.9 | 0.76-0.99 | 82.85 | <0.01 |
| Anticoagulation (n%) | 87 | 0.77 | 0.48-0.97 | 85.78 | <0.01 |
| Inotropic support (n%) | 182 | 0.67 | 0.51-0.82 | 78.46 | <0.01 |
| Intubation (n%) | 182 | 0.33 | 0.13-0.57 | 89.34 | <0.01 |
| Corticosteroids (n%) | 182 | 0.35 | 0.21-0.51 | 76.54 | <0.01 |
| Outcome | |||||
| Length of hospital stay (days) | 99 | 10.66 | 8.86-12.46 | 61.30 | 0.03 |
| Admitted to intensive care unit (n%) | 182 | 0.74 | 0.52-0.91 | 88.13 | <0.01 |
| Dilatation of the coronary arteries (n%) | 182 | 0.17 | 0.09-0.28 | 61.40 | 0.02 |
| Acute kidney injury (n%). | 182 | 0.13 | 0-0.37 | 91.97 | <0.01 |
Fig. 2.
Pooled clinical characteristics of PMIS. A) Gastrointestinal symptoms B) Febrile and inflammatory C) shock D) left ventricular dysfunction E) KD F) respiratory symptom.
Risk factors and Laboratory Results
Among all patients hit with PMIS, of which 15% patients had obvious contacts with SARS Cov-2 infected individuals (95% CI 0.01-0.40, I2 = 92.02, P < 0.01). The median time from the onset of PMIS to hospitalization was 5.9 days (95% CI 5.55-6.25, I2 = 31.30, P = 0.23). IgG antibody against SARS-CoV-2 was positive on 81% of patients (95% CI 0.74-0.86, I2 = 34.25, P = 0.17), 37% of nucleic acid was positive by RT-PCR (95% CI 0.26-0.50, I2 = 59.94, P < 0.01) (Fig. 3 ).
Fig. 3.
Pooled risk factors of PMIS. A) Contact with viral B) Onset of Kawa-COVID-19 to hospitalization C) Nucleic acid positive of SARS Cov-2 D) lgG of SARS Cov-2.
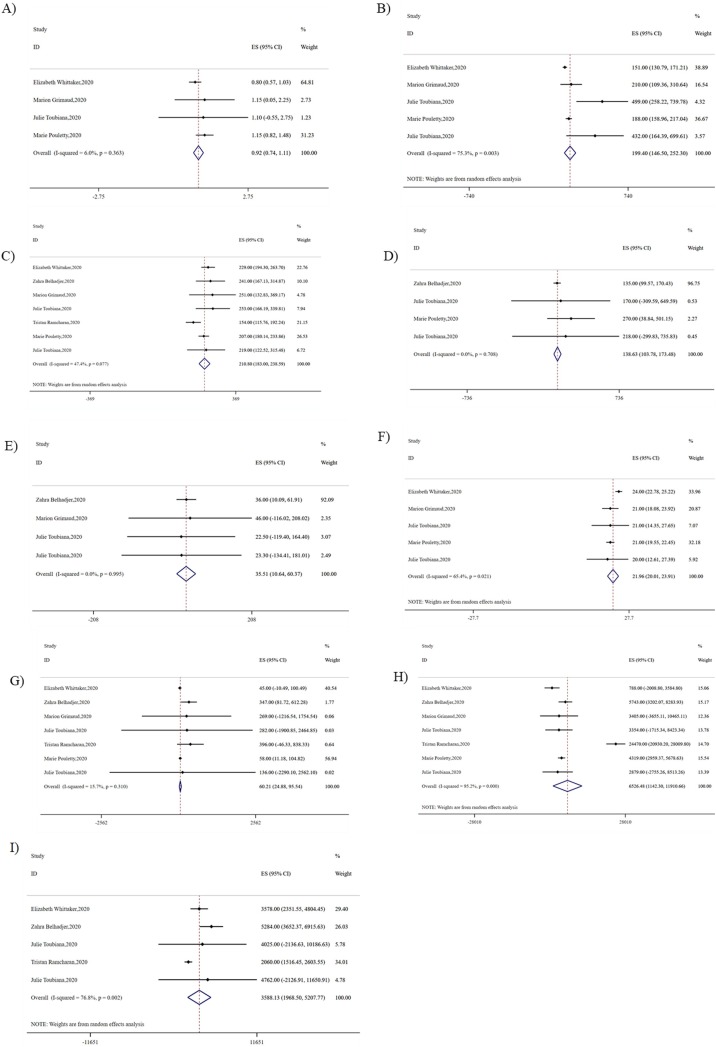
There was a severe inflammatory state with the elevated C-reactive protein (210.80 mg/l, 95% CI 183-238.59, I2 = 47.40, P = 0.08), IL-6 (138.63 pg/l, 95% CI 103.78-173.48, I2 <0.01, P = 0.71) and PCT (35.51 ng/l, 95% CI 10.64-60.37, I2 <0.01, P = 0.99). Myocardial markers troponin (60.21 ng/l, 95% CI 21.88-95.54, I2 = 15.70, P = 0.31) and NT-proBNP (6526.48 pg/l, 95% CI 1142.31-11910.66, I2 = 95.20, P < 0.01) concentrations were also elevated. In addition, albumin concentrations (21.96 g/l, 95% CI 20.01-23.91, I2 = 65.40, P = 0.02) and lymphocyte count (0.92*10^9/l, 95% CI 0.74-1.11, I2 = 6, P = 0.36) of PMIS patients were frequently reduced (Fig. 4 ).
Fig. 4.
Pooled laboratory results of PMIS. A) Lymphocyte B) Platelet C) CRP D) IL-6 E) PCT F) Albumin G) Troponin H) NT-proBNP I) D-dimer.
Treatments
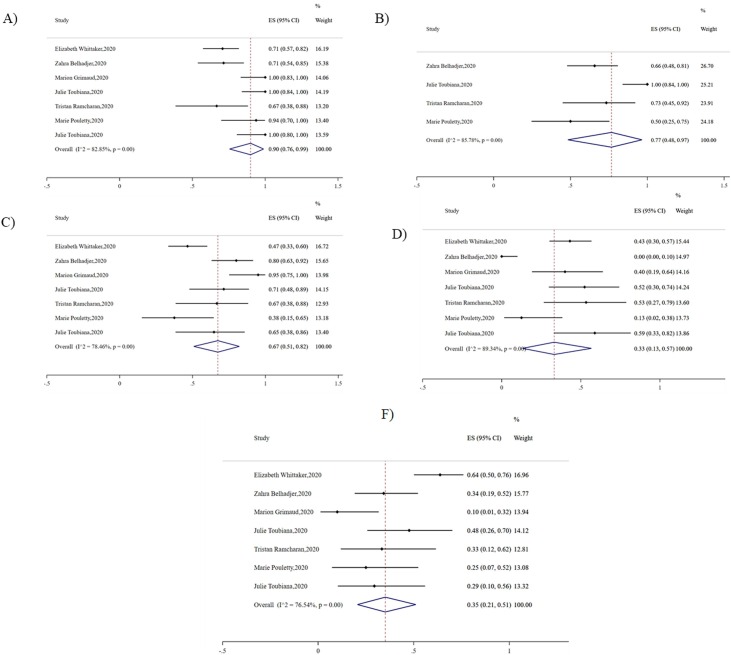
Intravenous immunoglobulin was a routine treatment for PMIS (90%, 95% CI 0.76-0.99, I2 = 82.25, P < 0.01). Approximately, there were 77% of patients received anticoagulant therapy (95% CI 0.48-0.97, I2 = 85.78, P < 0.01) during their hospitalization. More than half of patients were required inotropic supports (67%, 95% CI 0.51–0.82, I2 = 78.46, P < 0.01), and corticosteroids were used on 35% of patients (95% CI 0.21-0.51, I2 = 76.54, P < 0.01). Although respiratory symptoms were rare to see on PMIS patients, mechanical ventilation was required on 33% of patients (95% CI 0.13–0.57, I2 = 89.34, P < 0.01) (Fig. 5 ).
Fig. 5.
Pooled treatment of PMIS. A) Intravenous immunoglobulin B) Anticoagulation C) Inotropic support D) Intubation E) Corticosteroids.
Outcome
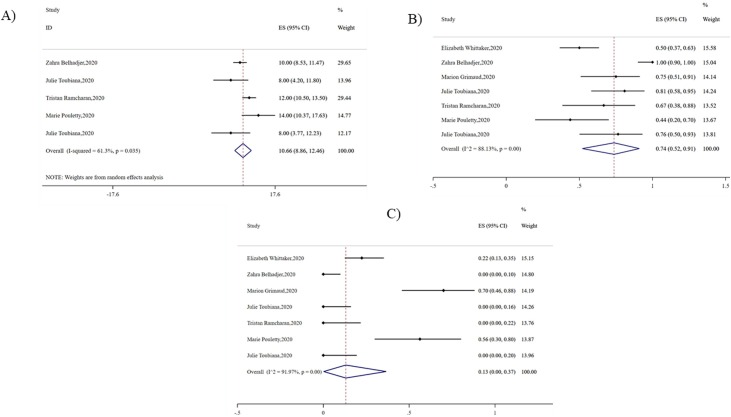
The median length of hospital stay was 10.66 days (95% CI 8.86-12.46, I2 = 61.30, P = 0.03), and 74% of patients had been admitted to intensive care unit (95% CI 0.52-0.91, I2 = 88.13, P < 0.01). The dilatation of coronary arteries (17%, 95% CI 0.09-0.28, I2 = 61.40, P = 0.02) and acute kidney injury (13%, 95% CI 0-0.37, I2 = 91.97, P < 0.01) occurred to some patients (Fig. 6 ).
Fig. 6.
Pooled outcome of PMIS. A) Length of hospital stay B) Admitted to intensive care unit C) Dilatation of the coronary arteries
Discussion
Clusters of patients with PMIS have been reported in several countries and regions. To date, the number of confirmed cases in France (Belhadjer et al., 2020, Juan et al., 2020, Pouletty et al., 2020, Toubiana et al., 2020a, 2020), the UK (Ramcharan et al., 2020, Whittaker et al., 2020) and Italy (Verdoni et al., 2020) has exceeded 2000, however the reported clinical manifestations to every patient were not consistent. WHO, CDC and Royal College of Paediatrics and Child Health havd issued their own diagnostic criteria for PMIS. Due to the inconsistent standards underestimated and overestimated, more patients with PMIS beyond the current diagnostic criteria was underestimated (Levin, 2020), and patients with other childhood infection or inflammatory infections without PMIS meeting the criteria was overestimated (Rowley, 2020). Hence, this systematic review containing currently available observational studies on PMIS is crucial to lay a foundation for establishing a unified diagnostic standard.
We found that patients who suffered from PMIS experienced three types of clinical manifestation: 1) persistent fever and gastrointestinal symptoms, 2) shocked with heart dysfunction and 3) symptoms coincident with the diagnostic criteria for KD. Among them, fever and gastrointestinal symptoms were the most common clinical features. In fact, PMIS shared similar clinical symptoms with KD disease. However, KD usually occured to the children under five years old (Hu et al., 2019, Pinna et al., 2008), whereas the median age of the patients with PMIS was 8.99 years. KD shock syndrome (KDSS), one of the most serious manifestations in KD, was usually also presented with myocardial ischemia, resulting in a moderate elevation to myocardial markers (Soni et al., 2020); while the troponin of patients with PMIS increased by 400 times on average and NT-proBNP increased by about 65 times. In addition, though inflammatory markers were elevated on both KDSS patients and PMIS patients, the elevation on KDSS patients was moderate (Hu et al., 2019, Taddio et al., 2017), while PCT, IL-6 and CRP were 350 times, 16 times and 3 times higher on PMIS patients, respectively.
At present, there was no standard treatment for PMIS. This meta-analysis showed that intravenous immunoglobulin was the most common treatment. Inotropic support and anticoagulant therapy were needed on some but not all patients in preventing shocks. Despite respiratory symptoms were rare to see on PMIS patients, mechanical ventilation in some cases was also required. This study provides evidence for clinicians to choose empirical treatment for PMIS.
Children suffered with SARS-COV2 infection presented relatively mild symptoms and good prognosis (Heavey et al., 2020, Zheng et al., 2020). Although patients hit with PMIS presented few and mild respiratory symptoms, most of them would still need to stay at ICU for further treatment and the average duration of hospital stay climbed up. At the same time, Marion Grimaud had found that patients admitted to the ICU had higher levels of inflammatory biomarkers, such as CRP, PCT, fibrinogen and neutrophils (Grimaud et al., 2020). Marie Pouletty found patients with older age, haemodynamic failure, higher ferritinaemia, higher urea and creatinine were more susceptible to severe PMIS (Pouletty et al., 2020). Fortunately, the currently observed mortality caused by PMIS was extremely low, there was only one death reported related to PMIS (Whittaker et al., 2020). However, we recommended a long-term follow-up for patients at high risks for aneurysm, as our analysis showed that more than half of the patients had arterial dilatation throughout the illness.
With limited understanding of the molecular pathogenesis of PMIS, we put forward several potential hypotheses in this discussion in hope of shedding light for future research. First, in our analysis, the positive rate of SARS-CoV-2-specific antibody detection was higher than that of RT-PCR detection (81% vs 37%). It is therefore possible that antibodies developed against SARS-CoV-2 could be one of the triggers for PMIS. Of note, the recently described syndrome seems to be a delayed inflammatory response that probably relates to adaptive immunity and neutralized antibodies (NAbs) after initial encounter with the COVID-19 infection. Although adaptive immunity is crucial to clear the invaded virus, direct infection of T cells by SARS-CoV-2 might contribute to lymphopenia and impair antiviral response. Moreover, some NAbs targeting non-receptor-binding domains of spike S protein could appear an antibody-dependent enhancement (ADE) effect that accelerates viral replication and results in cellular destruction. Virus-antibody complex following the interaction with Fc receptors, as well as subsequent complement activation, might also mediate dysregulated response and increase cytokine secretion (Katzelnick et al., 2017). Overproduction of these inflammatory mediators could lead to a stimulated risk of vascular hyperpermeability, high fever, shock, and even evolve severe multiorgan damages.
Second, ACE2 which is the functional receptor of SARS-CoV-2, might be involved in the pathogenesis of the syndrome that we presented here, though the positive rate of SARS-COV-2 was low. Myocardial injury, one of the most severe symptoms of this emerging syndrome, seems to occur directly, by viral destruction of cardiac endothelial cells; and indirectly, through ACE2-associated signaling pathways (Hoffmann et al., 2020). Furthermore, releasing these inflammatory cytokines also contributes to the destruction of vascular matrix proteins in part as well as the loss of structural integrity of the vessels, culminating in coronary dilatation and aneurysm formation linked to Kawasaki-like features of this novel syndrome.
Limitations
Most of literature included in this study are single-arm case review studies, and lack of control groups, and the characteristics of PMIS and KD disease cannot be accurately compared. No publication bias analysis was conducted due to the mentioned studies is few. In addition, the ethnic distributions of PMIS were not included in this study.
Conclusions
Patients suffered from PMIS could present with either persistent fever and gastrointestinal symptoms, the shock of heart dysfunction, KD-like symptoms or either combination mentioned above. Future studies are required to elucidate the exact role of children’s immune response to the pathogenesis of this phenomenon, so as to characterize the risk factors affecting the development of PMIS, and to determine whether SARS-CoV-2 could directly secrete toxins or coinfect with other microbial agents to trigger this syndrome.
Declaration of interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics approval and consent to participate
Not applicable
Consent for publication
Not applicable
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare that they have no competing interests
Funding statement
No funding.
Authors’ contributions
Hua Zou, Juan Lu and Josiah Hiu-yuen Wong, designed the study, Jingjing Liu, Si Cheng, Yan Shen and Chunli Li performed study. Hua Zou, Qiuhong Li and Juan Lu analyzed data. Hua Zou and Xiaojiong Jia wrote and revised this manuscript.
Acknowledgements
Not applicable
References
- Belhadjer Z., Meot M., Bajolle F., Khraiche D., Legendre A., Abakka S. Acute heart failure in multisystem inflammatory syndrome in children (MIS-C) in the context of global SARS-CoV-2 pandemic. Circulation. 2020 doi: 10.1161/CIRCULATIONAHA.120.048360. [DOI] [PubMed] [Google Scholar]
- Grimaud M., Starck J., Levy M., Marais C., Chareyre J., Khraiche D. Acute myocarditis and multisystem inflammatory emerging disease following SARS-CoV-2 infection in critically ill children. Ann Intensive Care. 2020;10(1):69. doi: 10.1186/s13613-020-00690-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heavey L., Casey G., Kelly C., Kelly D., McDarby G. No evidence of secondary transmission of COVID-19 from children attending school in Ireland, 2020. Euro Surveill. 2020;25(21) doi: 10.2807/1560-7917.ES.2020.25.21.2000903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181(2) doi: 10.1016/j.cell.2020.02.052. 271-80 e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu Y.C., Liu H.M., Lin M.T., Chen C.A., Chiu S.N., Lu C.W. Outcomes of Kawasaki Disease Children With Spontaneous Defervescence Within 10 Days. Front Pediatr. 2019;7:158. doi: 10.3389/fped.2019.00158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juan J., Gil M.M., Rong Z., Zhang Y., Yang H., Poon L.C. Effects of coronavirus disease 2019 (COVID-19) on maternal, perinatal and neonatal outcomes: a systematic review. Ultrasound Obstet Gynecol. 2020 doi: 10.1002/uog.22088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katzelnick L.C., Gresh L., Halloran M.E., Mercado J.C., Kuan G., Gordon A. Antibody-dependent enhancement of severe dengue disease in humans. Science. 2017;358(6365):929–932. doi: 10.1126/science.aan6836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin M. Childhood Multisystem Inflammatory Syndrome - A New Challenge in the Pandemic. N Engl J Med. 2020 doi: 10.1056/NEJMe2023158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinna G.S., Kafetzis D.A., Tselkas O.I., Skevaki C.L. Kawasaki disease: an overview. Curr Opin Infect Dis. 2008;21(3):263–270. doi: 10.1097/QCO.0b013e3282fbf9cd. [DOI] [PubMed] [Google Scholar]
- Pouletty M., Borocco C., Ouldali N., Caseris M., Basmaci R., Lachaume N. Paediatric multisystem inflammatory syndrome temporally associated with SARS-CoV-2 mimicking Kawasaki disease (Kawa-COVID-19): a multicentre cohort. Ann Rheum Dis. 2020 doi: 10.1136/annrheumdis-2020-217960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramcharan T., Nolan O., Lai C.Y., Prabhu N., Krishnamurthy R., Richter A.G. Paediatric Inflammatory Multisystem Syndrome: Temporally Associated with SARS-CoV-2 (PIMS-TS): Cardiac Features, Management and Short-Term Outcomes at a UK Tertiary Paediatric Hospital. Pediatr Cardiol. 2020 doi: 10.1007/s00246-020-02391-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riphagen S., Gomez X., Gonzalez-Martinez C., Wilkinson N., Theocharis P. Hyperinflammatory shock in children during COVID-19 pandemic. Lancet. 2020;395(10237):1607–1608. doi: 10.1016/S0140-6736(20)31094-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowley A.H. Understanding SARS-CoV-2-related multisystem inflammatory syndrome in children. Nat Rev Immunol. 2020 doi: 10.1038/s41577-020-0367-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soni P.R., Noval Rivas M., Arditi M. A Comprehensive Update on Kawasaki Disease Vasculitis and Myocarditis. Curr Rheumatol Rep. 2020;22(2):6. doi: 10.1007/s11926-020-0882-1. [DOI] [PubMed] [Google Scholar]
- Taddio A., Rossi E.D., Monasta L., Pastore S., Tommasini A., Lepore L. Describing Kawasaki shock syndrome: results from a retrospective study and literature review. Clin Rheumatol. 2017;36(1):223–228. doi: 10.1007/s10067-016-3316-8. [DOI] [PubMed] [Google Scholar]
- Tagarro A., Epalza C., Santos M., Sanz-Santaeufemia F.J., Otheo E., Moraleda C. Screening and Severity of Coronavirus Disease 2019 (COVID-19) in Children in Madrid, Spain. JAMA Pediatr. 2020 doi: 10.1001/jamapediatrics.2020.1346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toubiana J., Poirault C., Corsia A., Bajolle F., Fourgeaud J., Angoulvant F. Kawasaki-like multisystem inflammatory syndrome in children during the covid-19 pandemic in Paris, France: prospective observational study. BMJ. 2020;369:m2094. doi: 10.1136/bmj.m2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toubiana J., Poirault C., Corsia A., Bajolle F., Fourgeaud J., Angoulvant F. Outbreak of Kawasaki disease in children during COVID-19 pandemic: a prospective observational study in Paris, France. medRxiv. 2020 doi: 10.1136/bmj.m2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verdoni L., Mazza A., Gervasoni A., Martelli L., Ruggeri M., Ciuffreda M. An outbreak of severe Kawasaki-like disease at the Italian epicentre of the SARS-CoV-2 epidemic: an observational cohort study. Lancet. 2020;395(10239):1771–1778. doi: 10.1016/S0140-6736(20)31103-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whittaker E., Bamford A., Kenny J., Kaforou M., Jones C.E., Shah P. Clinical Characteristics of 58 Children With a Pediatric Inflammatory Multisystem Syndrome Temporally Associated With SARS-CoV-2. JAMA. 2020 doi: 10.1001/jama.2020.10369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng G., Wang B., Zhang H., Xie C., Zhang Y., Wen Z. Clinical Characteristics of Acute Respiratory Syndrome with SARS-CoV-2 Infection in Children in South China. Pediatr Pulmonol. 2020 doi: 10.1002/ppul.24921. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.