Abstract
STUDY QUESTION
What is the effect of uterine bathing with sonography gel prior to IVF/ICSI-treatment on live birth rates after fresh embryo transfer in patients with endometriosis?
SUMMARY ANSWER
After formal interim analysis and premature ending of the trial, no significant difference between uterine bathing using a pharmacologically neutral sonography gel compared to a sham procedure on live birth rate after fresh embryo transfer in endometriosis patients (26.7% vs. 15.4%, relative risk (RR) 1.73, 95% confidence interval (CI) 0.81–3.72; P-value 0.147) could be found, although the trial was underpowered to draw definite conclusions.
WHAT IS KNOWN ALREADY
Impaired implantation receptivity contributes to reduced clinical pregnancy rates after IVF/ICSI-treatment in endometriosis patients. Previous studies have suggested a favourable effect of tubal flushing with Lipiodol® on natural conceptions. This benefit might also be explained by enhancing implantation through endometrial immunomodulation. Although recent studies showed no beneficial effect of endometrial scratching, the effect of mechanical stress by intrauterine infusion on the endometrium in endometriosis patients undergoing IVF/ICSI-treatment has not been investigated yet.
STUDY DESIGN, SIZE, DURATION
We performed a multicentre, patient-blinded, randomised controlled trial in which women were randomly allocated to either a Gel Infusion Sonography (GIS, intervention group) or a sham procedure (control group) prior to IVF/ICSI-treatment. Since recruitment was slow and completion of the study was considered unfeasible, the study was halted after inclusion of 112 of the planned 184 women.
PARTICIPANTS/MATERIALS, SETTING, METHODS
We included infertile women with surgically confirmed endometriosis ASRM stage I–IV undergoing IVF/ICSI-treatment. After informed consent, women were randomised to GIS with intrauterine instillation of ExEm-gel® or sonography with gel into the vagina (sham). This was performed in the cycle preceding the embryo transfer, on the day GnRH analogue treatment was started. The primary endpoint was live birth rate after fresh embryo transfer. Analysis was performed by both intention-to-treat and per-protocol.
MAIN RESULTS AND THE ROLE OF CHANCE
Between July 2014 to September 2018, we randomly allocated 112 women to GIS (n = 60) or sham procedure (n = 52). The live birth rate after fresh embryo transfer was 16/60 (26.7%) after GIS versus 8/52 (15.4%) after the sham (RR 1.73, 95% CI 0.81–3.72; P-value 0.147). Ongoing pregnancy rate was 16/60 (26.7%) after GIS versus 9/52 (17.3%) in the controls (RR 1.54, 95% CI 0.74–3.18). Miscarriage occurred in 1/60 (1.7%) after GIS versus 5/52 (9.6%) in the controls (RR 0.17, 95% CI 0.02–1.44) women. Uterine bathing resulted in a higher pain score compared with a sham procedure (visual analogue scale score 2.7 [1.3–3.5] vs. 1.0 [0.0–2.0], P < 0.001). There were two adverse events after GIS compared with none after sham procedures.
LIMITATIONS, REASONS FOR CAUTION
The study was terminated prematurely due to slow recruitment and trial fatigue. Therefore, the trial is underpowered to draw definite conclusions regarding the effect of uterine bathing with sonography gel on live birth rate after fresh embryo transfer in endometriosis patients undergoing IVF/ICSI-treatment.
WIDER IMPLICATIONS OF THE FINDINGS
We could not demonstrate a favourable effect of uterine bathing procedures with sonography gel prior to IVF/ICSI-treatment in patients with endometriosis.
STUDY FUNDING/COMPETING INTEREST(S)
Investigator initiated study. IQ Medical Ventures provided the ExEm FOAM® kits free of charge, they were not involved in the study design, data management, statistical analyses and/or manuscript preparation, etc. C.B.L. reports receiving grants from Ferring, Merck and Guerbet, outside the submitted work. C.B.L. is Editor-in-Chief of Human Reproduction. V.M. reports grants and other from Guerbet, outside the submitted work. B.W.M. reports grants from NHMRC (GNT1176437), personal fees from ObsEva, Merck and Merck KGaA, Guerbet and iGenomix, outside the submitted work. N.P.J. reports research funding from Abb-Vie and Myovant Sciences and consultancy for Vifor Pharma, Guerbet, Myovant Sciences and Roche Diagnostics, outside the submitted work. K.D. reports personal fees from Guerbet, outside the submitted work. The other authors do not report any conflicts of interest. No financial support was provided.
TRIAL REGISTRATION NUMBER
NL4025 (NTR4198)
TRIAL REGISTRATION DATE
7 October 2013
DATE OF FIRST PATIENT’S ENROLMENT
22 July 2014
Keywords: endometriosis, IVF/ICSI, ART, implantation, uterine bathing, endometrium, pregnancy, live birth
WHAT DOES THIS MEAN FOR PATIENTS?
In women with endometriosis, pregnancy rates after fertility treatment with IVF/ICSI are reduced. This is probably related to an impaired implantation receptivity of the endometrium (attachment of a fertilised oocyte to the inner lining of the uterine cavity).
We investigated whether intrauterine infusion of sonography gel prior to IVF/ICSI treatment would have a beneficial effect on live birth rate after fresh embryo transfer in patients with endometriosis by improving implantation. Participants were randomly allocated to receive intrauterine infusion with gel or a sham procedure, in which gel was infused into the vagina.
Although the study was stopped prematurely due to slow recruitment and trial fatigue, the researchers could not demonstrate a favourable effect of the intervention in comparison to the sham procedure.
Introduction
In patients with endometriosis, pregnancy and live birth rates after IVF or ICSI treatment are reduced compared to patients without endometriosis (Barnhart et al., 2002; Harb et al., 2013; Muteshi et al., 2018). This could be caused by a decreased quality of oocytes, a reduced implantation capacity of the embryo and/or a diminished endometrial receptivity (Pellicer et al., 2001; Brosens et al., 2012; Lessey and Kim, 2017). Suggested explanations for the impaired endometrial receptivity of endometriosis patients undergoing IVF/ICSI-treatment are progesterone resistance, inflammation and inadequate decidualisation as well as the presence of concomitant adenomyosis (Brosens et al., 2012; Revel, 2012; Vercellini et al., 2014; Muteshi et al., 2018).
For nearly two decades, it has been hypothesised that applying local endometrial injury might induce a beneficial effect on endometrial receptivity, especially in patients with recurrent implantation failure (RIF) after IVF/ICSI-treatment (Barash et al., 2003; Zhou et al., 2008, Potdar et al., 2012; Nastri et al., 2015). Although multiple randomised controlled trials (RCTs) have investigated the effect of endometrial scratching prior to IVF/ICSI-treatment, the actual role of applying endometrial injury in women undergoing ART remains unclear (van Hoogenhuijze et al., 2019). A critical assessment of the quality of papers on endometrial scratching suggests that methodological problems may have biased the results of the trials (Li et al., 2019).
As an alternative to scratching the endometrium, its receptivity might be modulated by infusing fluids into the uterine cavity (‘uterine bathing’). In patients with minimal to mild endometriosis, undergoing hysterosalpingography with Lipiodol®, an ethiodised oil derived from poppy seed, showed improved naturally conceived clinical pregnancy rates post-procedure (Johnson et al., 2004). Although this result is possibly explained by a flushing effect on the Fallopian tubes, a direct (therapeutic) effect on the endometrial receptivity was suggested as well. This hypothesis is supported by data from a randomised controlled animal study (Johnson et al., 2005) in which treatment with Lipiodol® (vs. infusion with saline vs. sham treatment) altered the dendritic cell population of the endometrium in mice, suggesting a Lipiodol-induced favourable uterine immune response. Endometrial gene transcript regulation during Lipiodol® uterine bathing has also demonstrated a downregulation of endometrial Osteopontin, a molecule that could be interfering with implantation receptivity in women with endometriosis (Johnson et al, 2019). In order to investigate the effectiveness of pre-IVF uterine bathing with Lipiodol® in women with endometriosis or RIF, the IVF-LUBE trial was performed (Reilly et al., 2019). The study was, however, not sufficiently powered to show any difference between the intervention and control arm.
Besides a possible pharmacological mechanism of Lipiodol®, the favourable effects of uterine bathing could also be explained as the direct result of mechanical stress on the endometrium. In the available evidence, a control group treated with intrauterine saline or gel was not included and endometrial biopsies confirming the drug-therapeutic effect of Lipiodol® were absent. This hypothesis is partly supported by the observations in limited quality trials that screening hysteroscopy, using intrauterine infusion, may increase IVF/ICSI pregnancy rates in some subpopulations (Bosteels et al., 2010; Karayalçin et al., 2012; Kamath et al., 2019).
In order to investigate the effect of mechanical pressure by intrauterine infusion of a pharmacological neutral sonography gel on the endometrium in endometriosis patients (ASRM I–IV) undergoing IVF/ICSI treatment, we designed the TUBIE trial (Trial on Uterine Bathing before IVF/ICSI treatment in patients with Endometriosis). In the intervention arm of this randomised trial, we performed a Gel Infusion Sonography (GIS) prior to IVF/ICSI treatment, and in the control arm, we applied a sham procedure. In both arms, we used ExEm-gel®, a pharmacologically neutral gel, not containing any components which may enhance endometrial receptivity (Exalto et al., 2007, 2014; Emanuel et al., 2012).
We hypothesised that in endometriosis patients scheduled for IVF/ICSI-treatment, uterine bathing with sonography gel would be superior to a sham procedure in terms of live birth rates after fresh embryo transfer, through a mechanical pressure mechanism inducing local injury of the endometrium.
Material and methods
A multicentre, patient-blinded, parallel two-arm RCT was performed in four centres in the Netherlands and Belgium with special care for endometriosis. Participating centres were: Amsterdam UMC (Location VUmc), Amsterdam, the Netherlands; OLVG, Amsterdam, the Netherlands; Isala, Zwolle, the Netherlands and Ghent University Hospital, Ghent, Belgium. The study was approved by the institutional review board of the Amsterdam UMC (Location VUmc); (METC VUmc reference number: 2013.242). The study had the acronym TUBIE trial (Trial on Uterine Bathing before IVF-/ICSI-treatment in patients with Endometriosis) and was registered in the Dutch Trial Register (NL4025 (NTR4198), 7 October 2013).
Patients
Infertile women aged 18–42 years with surgically confirmed endometriosis (American Society of Reproductive Medicine (ASRM), stage I–IV), scheduled for IVF/ICSI-treatment, were asked to participate by the investigators before the start of their IVF/ICSI treatment. Women with known anomalies of the uterus, a malignancy or a pregnancy were excluded.
Randomisation and allocation
After written informed consent, women were randomly assigned (1:1) to either a GIS procedure (intervention) or sham procedure (control). Randomisation was performed without stratification in randomly permuted blocks of 2, 4 or 6, using a computer-generated randomisation list. This randomisation list was rendered by a statistician (P.M.v.d.V). All study procedures (intervention and control) were performed by investigators who were not involved in the IVF- or ICSI-treatment. Patients, IVF clinic staff, embryologists and IVF technicians were blinded for the outcomes of randomisation i.e. the performed study procedure and the subsequent IVF/ICSI-treatment.
Interventions
The study procedures (intervention or sham) were performed in the cycle preceding IVF/ICSI stimulation and embryo transfer, on the day GnRH analogue treatment was started (van Hoogenhuijze et al., 2019) according to a standard long agonist protocol. Both procedures were performed by investigators who were not involved in the IVF/ICSI-treatment. For the GIS procedure, pharmacologically neutral ExEm-gel (GynaecologIQ/GISKIT, IQ Medical Ventures BV, Rotterdam, the Netherlands) was used (Exalto et al., 2014). A flexible catheter was placed through the internal ostium infusing a maximum amount of 10 ml gel in the uterine cavity. A transvaginal ultrasound, which was blinded for the patient, was performed immediately after infusing the gel. After performing the ultrasound, the catheter was removed and the procedure was ended.
The sham procedure was performed in a similar way to the GIS procedure with the exception that the tip of the flexible catheter was placed in the vaginal posterior fornix, in order to avoid unintentional endometrial injury. After infusing a maximum amount of 10 ml ExEm-gel® intravaginally, a transvaginal ultrasound was performed. Again the ultrasound was blinded for the patient. After performing this, the procedure was ended by removal of the catheter.
IVF/ICSI procedure
IVF/ICSI treatment procedures were performed by local protocol. In summary, at baseline (cycle Day 2 or 3), a transvaginal sonography was performed. Patients started with oral contraceptives on the third day of the cycle for the total duration of 3 weeks. After 2 weeks, pituitary downregulation by administration of GnRH agonists was started. GnRH agonists were continued till and including the day of hCG administration. Ovarian stimulation was performed according to the centres local protocol by recombinant FSH or highly purified HMG (dosage individually determined) and was started on cycle Day 3. Patients were monitored routinely by a standard protocol, depending on the follicle growth. Monitoring was continued until the criteria for urinary hCG injection (Pregnyl®) or recombinant hCG injection (Ovitrelle®) were met. Oocyte retrieval was carried out 34–37 h after hCG injection. IVF/ICSI incubation procedures were performed according to the clinics standard practice. Embryo transfer (≤2 embryos) was performed 72–120 h after oocyte retrieval. Remaining good quality embryos were cryopreserved in nitrogen vapour tanks or in liquid nitrogen. Luteal support was accomplished by two capsules of 100 mg Utrogestan per vaginam, three times a day, for 15 days, or in case of pregnancy, 22 days. All medication was registered and marketed in the Netherlands and Belgium.
Outcomes
The primary outcome was live birth rate after fresh embryo transfer. Secondary outcomes were biochemical pregnancy (increase in serum hCG), clinical pregnancy (gestational sac, with or without heartbeat, visible on ultrasound), ongoing pregnancy (viable intrauterine pregnancy that progresses beyond 12 weeks of gestation), multiple pregnancy (two or more gestational sacs, with or without heartbeat, visible on ultrasound), miscarriage (the presence of non-vitality on ultrasound or spontaneous loss of pregnancy before 12 weeks of gestation) and ectopic pregnancy (defined histologically if treated surgically or by clinical diagnosis if managed conservatively), all after the first cycle. If a pregnancy was terminated (medical-assisted termination of pregnancy before 24 weeks of gestation), this was reported separately and not counted as live birth. Furthermore, adverse events of the procedure (such as infection and bleeding), pain experienced during the procedure (recorded using the visual analogue scale (VAS 0.0–10.0 cm)) and complications of the IVF/ICSI treatment, as well as complications during the pregnancy and postpartum period, and neonatal outcomes were registered.
Sample size
The sample size calculation was based on the data of the interim analysis of the IVF-LUBE trial (Reilly et al., 2011). To demonstrate an absolute difference of 20% in live birth rate between the two groups (20% in the sham group vs. 40% in the GIS group) with a two-sided significance level of 5% and a power of 80%, 164 patients needed to be included in this trial. Considering a 10% loss to follow-up, the trial needed to include 92 patients per study group (184 patients in total).
Statistical analysis
The chi-square test or the Fishers’ exact test was used to compare dichotomous outcomes between the two arms of the trial. The relative risk (RR) is calculated as the effect size together with its 95% confidence interval (CI). The Student’s t-test or Mann–Whitney U test (depending on the data distribution) was used to compare continuous outcomes between the arms. All statistics on primary and secondary outcomes were undertaken for the intention-to-treat (ITT) and per-protocol (PP) analysis. The statistical analysis was performed in IBM SPSS Statistics version 22.0 using a two-sided significance level of 5%.
Because of the slow accrual of this trial and the IVF-LUBE trial concluding that uterine bathing with Lipiodol® did not add to the success of IVF (Reilly et al., 2019), it was decided to perform an interim analysis as soon as half of the planned study population had completed the study protocol. Details of the interim analysis were added to the trial protocol and approved by the institutional review board of the Amsterdam UMC (Location VUmc) before the required data became available. The interim analysis consisted of calculation of the conditional power which was defined as the probability of concluding live birth rate to be significantly higher in the GIS arm at the end of the trial given the planned sample size of 184 patients and the data observed so far. The conditional power was calculated under the assumption that the live birth rates in future participants in each of the two arms equalled those observed in the participants already included in the interim analysis. An independent statistician, who had no further involvement in the trial, performed the interim analysis as unblinding was considered necessary in order to avoid continuing the trial in case live birth rate was considerably smaller in GIS arm.
Results
Between July 2014 and September 2018, when the interim analysis was performed, a total of 113 women were included in this trial. After randomisation, one patient was excluded from the trial as she did not meet the inclusion criteria (Fergusson et al., 2002). She underwent a short stimulation IVF/ICSI protocol instead of the long stimulation protocol.
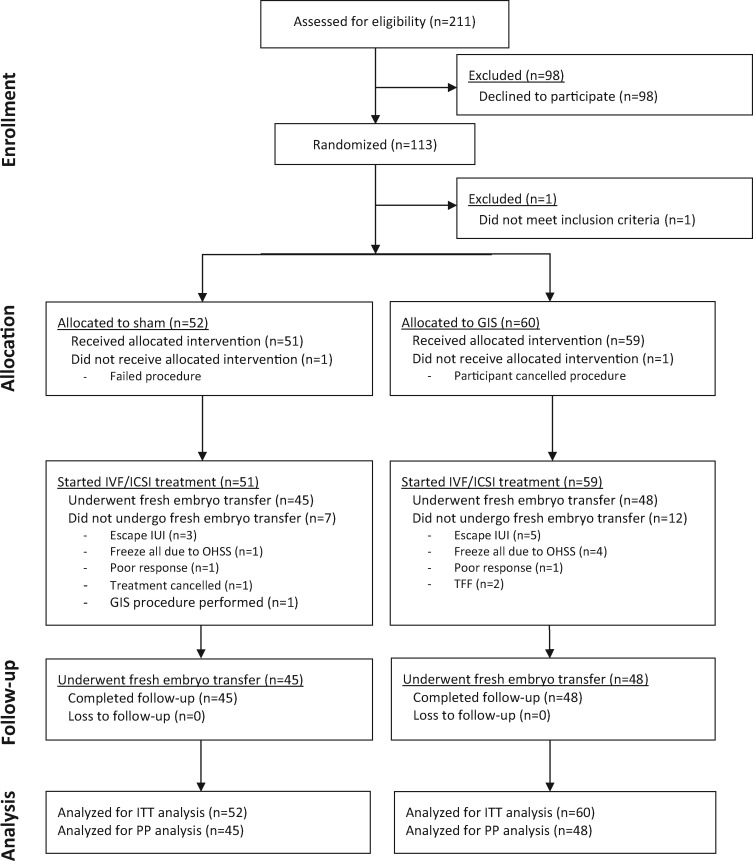
This trial started as a single-centre study in Amsterdam. From the start, recruitment of participants was progressing slowly. Fewer women than initially expected fulfilled the in- and exclusion criteria of the study. This resulted in an adaptation of the inclusion criteria allowing all ASRM stages of endometriosis to be included (amendment to protocol approved by institutional review board of the Amsterdam UMC (Location VUmc)). Unfortunately, this was not enough to reach a sufficient number of inclusions and lead to engaging collaboration with three other large endometriosis care centres in the Netherlands and Belgium. However, even with this expansion, the inclusion rate remained slow, therefore it was decided to perform an interim analysis where conditional power would be used to decide on continuing or stopping the trial. At the time of interim analysis, the live birth rate was 24% (12/49) in the GIS group compared to 19% (8/43) in the sham group. Conditional power in case of continuing the trial was calculated as 4%. The conditional power was considered too low to warrant continuation of the trial and the trial was stopped for futility. Consequently, 112 women allocated to either uterine bathing by GIS (n = 60) or a sham procedure (n = 52) were included in the final analysis (Fig. 1).
Figure 1.
Consort flowchart TUBIE trial. GIS, Gel Infusion Sonography; OHSS, ovarian hyperstimulation syndrome; TFF, total fertilisation failure; ITT, intention-to-treat; PP, per-protocol.
Baseline characteristics
Baseline characteristics are shown in Table I. In all participants, endometriosis was surgically confirmed: 65 women (58%) were diagnosed with minimal to mild endometriosis (ASRM stage I–II), 47 women (42%) with moderate to severe endometriosis (ASRM stage III–IV). Besides endometriosis, 52 couples (46.6%) had concomitant reasons for infertility: male factor (total motile sperm count <1 × 106 ml, n = 27 (24.1%)), tubal factor (one- or two-sided, n = 23 (20.5%)), anovulation (n = 7 (6.3%)) and cervical factor (n = 2 (1.8%)). Seven couples (6.3%) had more than two reasons for infertility.
Table I.
Baseline characteristics.
| GIS | Sham | |||
|---|---|---|---|---|
| (n = 60) | (n = 52) | |||
| Age (years), median (IQR) | 35 [33–39] | 36 [33–38] | ||
| BMI (kg/m2), median (IQR) | 22.3 [20.2–23.9] | 21.6 [20.8–24.2] | ||
| Smoking, n (%) | 3 (5.0) | 1 (1.9) | ||
| Ethnicity, n (%) | ||||
| Caucasian | 57 (95.0) | 42 (80.8) | ||
| Non-Caucasian | 3 (5.0) | 10 (19.2) | ||
| Gravidity, n (%) | ||||
| 0 | 23 (38.3) | 25 (48.1) | ||
| ≥1 | 37 (61.7) | 27 (51.9) | ||
| Parity, n (%) | ||||
| 0 | 39 (65.0) | 35 (67.3) | ||
| ≥1 | 21 (35.0) | 17 (32.7) | ||
| Miscarriage in obstetric history, n (%) | 22 (36.7) | 17 (32.7) | ||
| Curettage in obstetric history, n (%) | 4 (6.7) | 6 (11.5) | ||
| EUG in obstetric history, n (%) | 2 (3.3) | 2 (3.8) | ||
| Stage of endometriosis (rASRM), n (%) | ||||
| Minimal / mild (I/II) | 30 (50.0) | 35 (67.3) | ||
| Moderate / severe (III/IV) | 30 (50.0) | 17 (32.7) | ||
| Average cycle duration (days), median (IQR) | 28 [28–30] | 28 [28–30] | ||
| Total number of AFC on cycle Day 2–4, mean (± SD) | 15 ± 6 | 11 ± 6 | ||
| Months of infertility, mean (± SD) | 44.2 ± 26.8 | 35.3 ± 18.1 | ||
| Previous MAR in current child wish, n (%) | 54 (90.0) | 48 (92.3) | ||
| IVF/ICSI cycle number, median (IQR) | 2 [1–2] | 1 [1–2] | ||
AFC, antral follicle count; EUG, extra uterine gravidity; GIS, Gel Infusion Sonography; IQR, interquartile range; MAR, medically assisted reproduction; n, number; rASRM, revised American Society for Reproductive Medicine; SD, standard deviation.
Of the women randomised for GIS, one participant cancelled the procedure and did not receive the allocated treatment. In the sham group, one procedure failed as gel was infused into the uterine cavity during the procedure. Of the 59 women who underwent a GIS procedure, 12 did not undergo fresh embryo transfer after completing the IVF/ICSI-treatment, including 5 with a poor response, 4 with a hyper response or 2 women with total fertilisation failure. In the group with the sham procedure, five women had a poor response and one a hyper response resulting in a cancelled fresh embryo transfer. As a result, 48/60 women (80%) in the GIS group and 45/52 women (86.5%) in sham procedure group underwent a fresh embryo transfer. There was no loss to follow-up.
Pain scores
The median pain scores, measured by VAS (0.0–10.0 cm) directly after the procedure, were significantly higher following GIS 2.7 cm (25th to 75th percentiles 1.3–3.5) than following the sham procedure 1.0 cm (0.0–2.0, P < 0.001). There were two adverse events after a GIS procedure. One woman fainted during the procedure because of excessive pain (VAS 10.0 cm), another woman suffered from non-severe vaginal blood loss for a day after the procedure (Table II).
Table II.
Investigational procedure characteristics.
| GIS | Sham | P -values | |
|---|---|---|---|
| (n = 60) | (n = 52) | ||
| Amount gel infused (ml), median (IQR) | 6.0 [5.0–7.0] | 5.0 [4.0–5.5] | 0.003* |
| Procedure time (min), mean (± SD) | 9.3 ± 2.7 | 8.5 ± 2.0 | 0.089 |
| Cervical leakage, n (%) | 13 (21.7) | n/a | – |
| VAS score (cm), median (IQR) | 2.7 [1.3–3.5] | 1.0 [0.0–2.0] | <0.001* |
| Adverse event, n (%) | 2 (3.3) | 0 (0.0) | 0.173 |
IQR, interquartile range; n, number; n/a, not applicable; SD, standard deviation; VAS, visual analogue scale.
P-value < 0.05.
IVF/ICSI-treatment
The IVF- or ICSI-treatment characteristics are presented in Table III. There were no significant differences between the groups. Six women randomised for a GIS procedure had a complication of the IVF/ICSI-treatment: four women were diagnosed with mild-to-moderate ovarian hyperstimulation syndrome (OHSS) (Navot and Bergh, 1993); one woman was diagnosed with an ectopic pregnancy for which a laparoscopic tubotomy was undertaken; and one woman was diagnosed with an ovarian torsion in pregnancy at 6 weeks and 6 days of gestation then underwent a laparoscopy and the pregnancy continued successfully. In the sham group, only one woman had a complication of the IVF/ICSI-treatment, due to showing signs of a mild-to-moderate OHSS (P = 0.081).
Table III.
IVF/ICSI-treatment characteristics.
| GIS | Sham | P -values | |
|---|---|---|---|
| (n = 60) | (n = 52) | ||
| Treatment type, n (%) | 0.286 | ||
| IVF | 42 (70.0) | 41 (78.8) | |
| ICSI | 18 (30.0) | 11 (21.2) | |
| Cycle number, median (IQR) | 2 [1–2] | 1 [1–2] | 0.029* |
| Endometrial thickness at day of hCG (mm), median (IQR) | 3.9 [2.8–5.9] | 4.0 [2.9–5.3] | 0.751 |
| Dose FSH (U), median (IQR) | 2225 [1663–2925] | 2475 [1650–2925] | 0.460 |
| Semen, gain after processing (*106), median (IQR) | 14.0 [4.5–49] | 20.0 [8.0–49.0] | 0.339 |
| Number of retrieved oocytes, median (IQR) | 10 [7–14] | 8 [5–13] | 0.160 |
| Number of fertilised oocytes, median (IQR) | 6 [4–8] | 5 [3–9] | 0.333 |
| Number of embryos for transfer, median (IQR) | 5 [4–8] | 4 [2–8] | 0.333 |
| Number of transferred embryos, n (%) | 0.126 | ||
| No embryo transferred | 12 (20) | 6 (11.5) | |
| SET | 40 (66.7) | 43 (82.7) | |
| DET | 8 (13.3) | 3 (5.8) | |
| Top-quality of transferred embryo, n (%) | |||
| Embryo 1 | 18 (45.0) | 17 (39.5) | 0.895 |
| Embryo 2 (in case of DET) | 2 (25.0) | 0 (0.0) | 0.338 |
| Blastocyst transfer, n (%) | 2 (3.3) | 1 (1.9) | 0.645 |
| Cryopreservation, n (%) | 43 (71.7) | 34 (65.4) | 0.097 |
| Number of frozen embryos, median (IQR) | 4 [2–7] | 4 [1–7] | 0.277 |
| Complications, n (%) | 6 (10.0) | 1 (1.9) | 0.081 |
| OHSS (mild/moderate) | 4 (6.7) | 1 (1.9) | |
| Ectopic pregnancy | 1 (1.7) | 0 (0.0) | |
| Ovarian torsion | 1 (1.7) | 0 (0.0) |
DET, double embryo transfer; GIS, Gel Infusion Sonography; mm, millimetre; n, number; OHSS, ovarian hyperstimulation syndrome; SET, single embryo transfer; U, units.
P-value < 0.05.
Pregnancy outcomes
ITT analysis
The primary outcome, live birth rate per patient, was 16/60 (26.7%) in the GIS groups compared to 8/52 (15.4%) in the sham group (RR 1.73, 95% CI 0.81–3.72, P = 0.147) (Table IV). Secondary outcome measures are listed in Table IV. Adjustment for baseline characteristics (ethnicity, antral follicle count, duration infertility and cycle number) had no impact on the results (not reported). Two sensitivity analyses were performed: a first sensitivity analysis in which the excluded patient (n = 1) was included in the analysis and a second sensitivity analysis in which we counted the woman in the control group who had her pregnancy terminated because of trisomy 21 as live birth. Conclusion of the ITT analysis remained unchanged for both the sensitivity analyses.
Table IV.
Primary and secondary outcome measures; pregnancy outcomes after fresh embryo transfer (ITT analysis).
| GIS (n = 60) | Sham (n = 52) | P-values | Relative risk (RR) | 95% CI for the RR | |
|---|---|---|---|---|---|
| Primary outcome, n (%) | |||||
| Live birth | 16 (26.7) | 8 (15.4) | 0.147 | 1.73 | (0.81–3.72) |
| Secondary outcome, n (%) | |||||
| Biochemical pregnancy | 24 (40.0) | 18 (34.6) | 0.557 | 1.16 | (0.71–1.88) |
| Clinical pregnancy | 17 (28.3) | 14 (26.9) | 0.868 | 1.05 | (0.58–1.92) |
| Ongoing pregnancy | 16 (26.7) | 9 (17.3) | 0.236 | 1.54 | (0.74–3.18) |
| Multiple pregnancy | 1 (1.7) | 0 (0.0) | – | – | – |
| Miscarriage | 1 (1.7) | 5 (9.6) | – | – | – |
| Ectopic pregnancy | 1 (1.7) | 0 (0.0) | – | – | – |
| Termination of pregnancy | 0 (0.0) | 1 (1.9) | – | – | – |
CI, confidence interval; GIS, Gel Infusion Sonography; ITT, intention-to-treat; n, number; RR, relative risk.
PP analysis
When analysed PP, live birth rate after fresh embryo transfer in the GIS group was 16/48 (33.3%) versus 8/45 (17.8%) (RR 1.80, 95% CI 0.84–3.82, P-value 0.117). Secondary outcome measures of the PP analysis are shown in Table V.
Table V.
Primary and secondary outcome measures; pregnancy outcomes after fresh embryo transfer (PP analysis).
| GIS (n = 48) | Sham (n = 45) | P -values | Relative risk (RR) | 95% CI for the RR | |
|---|---|---|---|---|---|
| Primary outcome, n (%) | |||||
| Live birth | 16 (33.3) | 8 (17.8) | 0.117 | 1.80 | (0.84–3.82) |
| Secondary outcome, n (%) | |||||
| Biochemical pregnancy | 24 (50) | 15 (33) | 0.104 | 1.50 | (0.91–2.48) |
| Clinical pregnancy | 17 (35.4) | 12 (26.7) | 0.363 | 1.33 | (0.72–2.46) |
| Ongoing pregnancy | 16 (33.3) | 8 (17.8) | 0.087 | 1.88 | (0.89–3.95) |
| Multiple pregnancy | 1 (2.1) | 0 (0.0) | – | – | – |
| Miscarriage | 1 (2.1) | 4 (8.9) | – | – | – |
| Ectopic pregnancy | 1 (2.1) | 0 (0.0) | – | – | – |
| Termination of pregnancy | 0 (0.0) | 0 (0.0) | – | – | – |
CI, confidence interval; GIS, Gel Infusion Sonography; n, number; PP, per-protocol; RR, relative risk.
Pregnancy, postpartum and neonatal outcomes
Pregnancy outcomes and postpartum period are shown in Table VI. One woman in the control group had her pregnancy terminated because of trisomy 21. In the intervention group, five pregnancy complications were reported: two women developed hypothyroidism; two women were diagnosed with gestational diabetes and a placenta previa and one woman was diagnosed with pre-eclampsia. In the sham group, two complications of pregnancy were reported: one woman had gestational diabetes and one woman with fetomaternal transfusion. There were four postpartum complications in the intervention group: three postpartum haemorrhage and one bladder lesion during caesarean section; and one woman in the sham group had a postpartum curettage because of retained products of conception. There was no significant difference in neonatal outcomes between the groups. In the intervention group, one child was admitted to the neonatal intensive care unit because of respiratory distress syndrome after premature birth.
Table VI.
Follow-up of pregnancy and postpartum period.
| GIS | Sham | P -values | |
|---|---|---|---|
| (n = 16) | (n = 8) | ||
| Complications during pregnancy, n (%) | 5 (31.3) | 2 (25.0) | 0.750 |
| Postpartum complications, n (%) | 4 (25.0) | 1 (12.5) | 0.477 |
| Gestational age (weeks + days), median (IQR) | 39 + 3 [37 + 6 – 40 + 1] | 40 + 0 [37 + 5 – 40 + 2] | 0.787 |
| Prematurity, n (%) | 1 (6.3) | 0 (0.0) | – |
| NICU admittance, n (%) | 1 (6.3) | 0 (0.0) | – |
GIS, Gel Infusion Sonography; IQR, interquartile range; n, number; NICU, neonatal intensive care unit.
Discussion
The TUBIE trial investigated the effect of uterine bathing using ExEm-gel® on live birth rates after fresh embryo transfer in endometriosis patients undergoing IVF/ICSI-treatment. Regarding the primary outcome, no significant differences were found between patients randomised for intervention and sham procedures (26.7% vs. 15.4%, RR 1.73, 95% CI 0.81–3.72; P-value 0.147). The study was terminated prematurely after a formal interim analysis was performed when 50% of the planned study population had completed the study protocol.
The main strength of the TUBIE trial was the patient-blinded randomised controlled study design and the analysis on both ITT and PP basis. All study procedures (intervention and control) were performed with a pharmacologically neutral gel by investigators who were not involved in the IVF/ICSI-treatment procedures. Additionally, patients were blinded for the received treatment, since a sham procedure was performed in the control group. Despite this blinding, it should be noted that the GIS procedure and sham procedure might have been experienced differently by the patients, given the fact that the median pain scores measured immediately after the procedure, were significantly higher after GIS compared to that after the sham procedure.
The main weakness was the unplanned interim analysis and the insufficient power of the trial. When initiating the TUBIE trial, we stated that a 20% improvement in live birth rate in patients receiving GIS procedure was needed to accept the intervention. However, at the time of the interim analysis, a 5% improvement was shown in the GIS group (GIS vs. Sham; 24% vs. 19%). Conditional power in case of continuing the trial was calculated as 4%, which was considerably less than the 90% which was the preset minimum value for further continuation of the trial. A high threshold of 90% was chosen and described in an amendment to protocol, approved by the institutional review board, because of the considerable trial fatigue which made it only feasible to continue the trial in case of very promising interim results. It must be noted that generally much lower thresholds for the conditional power, around 20–30%, are used to guide stopping for futility. However, also if a lower threshold for the conditional power was chosen, the trial would still have been stopped early for futility since the conditional power at the interim analysis was only 4%.
After terminating the trial, there was a difference of 11.3% in live birth rate between the groups (26.7% vs. 15.4%). However, this difference in live birth rate was not statistically significant. Since the study is underpowered, drawing firm conclusions regarding the effect of uterine bathing with a pharmacologically neutral gel on live birth rates after fresh embryo transfer in patients with endometriosis undergoing IVF/ICSI-treatment is impossible. The 20% improvement used in the original power analysis was based on interim data of the IVF-LUBE trial (Reilly et al., 2011). In hindsight, this improvement was over-optimistic.
The idea that local endometrial injury might induce a positive effect on endometrial receptivity was first suggested by Granot et al. (2000). Since then, multiple studies have been performed to investigate this effect, reporting either positive, negative or neutral trial results (Nastri et al., 2015; Panagiotopoulou et al., 2015). Up until now, a convincing beneficial effect of endometrial scratching has not been established. The most recent systematic review still reports high clinical and statistical heterogeneity in the included trials with no differences between scratch and controls with respect to live birth and clinical pregnancy rates (van Hoogenhuijze et al., 2019). In addition, the recently published randomised trial of Lensen et al. (2019a) including 1364 women undergoing endometrial scratching prior to IVF treatment is in line with these findings and did not report higher live birth rates after scratching. Among the study population of this trial, 7.9% were endometriosis patients (n = 108). Subgroup analysis also showed no beneficial effect of endometrial scratching prior to IVF/ICSI-treatment on live birth rate in the endometriosis patients as well. Although endometrial scratching is currently the most offered IVF add-on in the UK (Spencer et al., 2016), the risk of harm should also be considered. Therefore, this add-on is currently rated as ‘amber’ by the Human Fertilisation and Embryology Authority (HFEA) (Lensen et al., 2019b).
Theoretically, local injury of the endometrium might also be applied by infusion of intrauterine fluids or gels. Since previous studies have suggested that uterine bathing with Lipiodol® might have an effect on endometrial receptivity by immunomodulation (Johnson et al., 2005; Johnson, 2014; Johnson et al., 2019), we aimed to investigate the effect of mechanical stress on the endometrium by uterine bathing with an pharmacologically neutral non-embryotoxic gel (ExEm-gel®). ExEm-gel® is frequently used during fertility work-up and is registered and marketed in the Netherlands for GIS procedures (Exalto et al., 2014). To the best of our knowledge, ExEm-gel® does not contain potential therapeutic components which may enhance endometrial receptivity (Exalto et al., 2007, 2014; Emanuel et al., 2012). And although observations in limited quality trials suggest that screening hysteroscopy might improve IVF/ICSI-treatment results (Bosteels et al., 2010; Karayalçin et al., 2012; Kamath et al., 2019), performing uterine bathing with ExEm-gel® is not endorsed by our study results. This is in accordance to the final report of the LUBE trial (Reilly et al., 2019) in which no difference in success rates for IVF following uterine bathing with Lipiodol for women with endometriosis or RIF could be found. The trail of Salehpour et al. (2016) in which intrauterine saline infusion as a form of endometrial injury was performed during IVF treatment even showed a negative effect on clinical pregnancy numbers and implantation rates among patients with RIF.
The recent insights that applying endometrial injury does not positively affect reproductive outcomes in patients undergoing IVF/ICSI-treatment is supported by the results of our TUBIE trial. Overall, the biological plausibility of performing endometrial injury procedures to improve endometrial receptivity can be questioned (Simón and Bellver, 2014). The various hypotheses that might explain the beneficial effect of applying endometrial injury have never been shown nor proven by a plausible biological influence on the endometrium and its effect on implantation. Evidence is derived from trials with high clinical and statistical heterogeneity and the investigated procedures (scratching, uterine bathing and screening hysteroscopy) have injured the endometrium in different ways. Additionally, methodological issues are common in the majority of RCTs reporting on endometrial scratching, leading to biased trial results (Li et al., 2019). Therefore the results cannot simply be merged and compared together in a straight forward way. Furthermore, these invasive procedures can be painful and may lead to adverse events.
Conclusion
In conclusion, in this prematurely terminated and therefore underpowered multicentre RCT, we found no favourable effect of uterine bathing using a pharmacologically neutral gel compared to a sham procedure prior to IVF or ICSI treatment on live birth rates after fresh embryo transfer in patients with endometriosis. Uterine bathing procedures were associated with higher pain scores and a small number of adverse events. Therefore, uterine bathing procedures are not advised as a routine add-on to IVF/ICSI treatment in women with endometriosis.
Authors’ roles
B.W.M., C.B.L., K.D., L.E.E.v.d.H., M.C.I.L., N.P.J., P.M.v.d.V. and V.M. played a role in conception and design of the trial. A.M.F.S., F.V., H.R.V., H.Ö., M.C.I.L. and W.K. took part in collection and assembly of data. A.M.F.S., C.B.L., H.Ö., M.C.I.L., P.M.v.d.V. and V.M. analysed and interpreted the data and drafted the manuscript. All authors critically reviewed the manuscript and made appropriate correction. All authors have read and approved the final version of the manuscript.
Funding
This was an investigator initiated study. IQ Medical Ventures provided the ExEm FOAM® kits free of charge. No financial support was provided.
Conflict of interest
Investigator initiated study. IQ Medical Ventures provided the ExEm FOAM® kits free of charge, they were not involved in the study design, data management, statistical analyses and/or manuscript preparation, etc. C.B.L. reports receiving grants from Ferring, Merck and Guerbet, outside the submitted work. C.B.L. is Editor-in-Chief of Human Reproduction. V.M. reports grants and other from Guerbet, outside the submitted work. B.W.M. reports grants from NHMRC (GNT1176437), personal fees from ObsEva, Merck and Merck KGaA, Guerbet and iGenomix, outside the submitted work. N.P.J. reports research funding from Abb-Vie and Myovant Sciences and consultancy for Vifor Pharma, Guerbet, Myovant Sciences and Roche Diagnostics, outside the submitted work. K.D. reports personal fees from Guerbet, outside the submitted work. The other authors do not report any conflicts of interest. No financial support was provided.
References
- Barash A, Dekel N, Fieldust S, Segal I, Schechtman E, Granot I. Local injury to the endometrium doubles the incidence of successful pregnancies in patients undergoing in vitro fertilization. Fertil Steril 2003;79:1317–1322. [DOI] [PubMed] [Google Scholar]
- Barnhart K, Dunsmoor-Su R, Coutifaris C. Effect of endometriosis on in vitro fertilization. Fertil Steril 2002;77:1148–1155. [DOI] [PubMed] [Google Scholar]
- Bosteels J, Weyers S, Puttemans P, Panayotidis C, Van Herendael B, Gomel V, Mol BW, Mathieu C, D'Hooghe T. The effectiveness of hysteroscopy in improving pregnancy rates in subfertile women without other gynaecological symptoms: a systematic review. Hum Reprod Update 2010;16:1–11. [DOI] [PubMed] [Google Scholar]
- Brosens I, Brosens JJ, Benagiano G. The eutopic endometrium in endometriosis: are the changes of clinical significance? Reprod Biomed Online 2012;24:496–502. [DOI] [PubMed] [Google Scholar]
- Emanuel MH, van Vliet M, Weber M, Exalto N. First experiences with hysterosalpingo-foam sonography (HyFoSy) for office tubal patency testing. Hum Reprod 2012;27:114–117. [DOI] [PubMed] [Google Scholar]
- Exalto N, Stappers C, van Raamsdonk LAM, Emanuel MH. Gel instillation sonohysterography: first experience with a new technique. Fertil Steril 2007;87:152–155. [DOI] [PubMed] [Google Scholar]
- Exalto N, Stassen M, Emanuel MH. Safety aspects and side-effects of ExEm-gel and foam for uterine cavity distension and tubal patency testing. Reprod Biomed Online 2014;29:534–540. [DOI] [PubMed] [Google Scholar]
- Fergusson D, Aaron SD, Guyatt G, Hébert P. Post-randomisation exclusions: the intention to treat principle and excluding patients from analysis. BMJ 2002;325:652–654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Granot I, Dekel N, Bechor E, Segal I, Fieldust S, Barash A. Temporal analysis of connexin43 protein and gene expression throughout the menstrual cycle in human endometrium. Fertil Steril 2000;73:381–386. [DOI] [PubMed] [Google Scholar]
- Harb HM, Gallos ID, Chu J, Harb M, Coomarasamy A. The effect of endometriosis on in vitro fertilisation outcome: a systematic review and meta-analysis. BJOG 2013;120:1308–1320. [DOI] [PubMed] [Google Scholar]
- Johnson NP. Review of lipiodol treatment for infertility - an innovative treatment for endometriosis-related infertility? Aust N Z J Obstet Gynaecol 2014;54:9–12. [DOI] [PubMed] [Google Scholar]
- Johnson NP, Baidya S, Jessup SO, Print CG, Muthukaruppan A, Chamley LW, Hadden WE, Hull ML, Mehta S, Shelling AN. Randomised trial of Lipiodol Uterine Bathing Effect (LUBE) in women with endometriosis-related infertility. FandR 2019;1:57–64. [Google Scholar]
- Johnson NP, Bhattu S, Wagner A, Blake DA, Chamley LW. Lipiodol alters murine uterine dendritic cell populations: a potential mechanism for the fertility-enhancing effect of lipiodol. Fertil Steril 2005;83:1814–1821. [DOI] [PubMed] [Google Scholar]
- Johnson NP, Farquhar CM, Hadden WE, Suckling J, Yu Y, Sadler L. The FLUSH trial–flushing with lipiodol for unexplained (and endometriosis-related) subfertility by hysterosalpingography: a randomized trial. Hum Reprod 2004;19:2043–2051. [DOI] [PubMed] [Google Scholar]
- Kamath MS, Bosteels J, D'Hooghe TM, Seshadri S, Weyers S, Mol BWJ, Broekmans FJ, Sunkara SK. Screening hysteroscopy in subfertile women and women undergoing assisted reproduction. Cochrane Database Syst Rev 2019;4:CD012856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karayalçin R, Ozyer S, Ozcan S, Uzunlar O, Gürlek B, Moraloğlu O, Batioğlu S. Office hysteroscopy improves pregnancy rates following IVF. Reprod Biomed Online 2012;25:261–266. [DOI] [PubMed] [Google Scholar]
- Lensen S, Osavlyuk D, Armstrong S, Stadelmann C, Hennes A, Napier E, Wilkinson J, Sadler L, Gupta D, Strandell A. et al. A randomized trial of endometrial scratching before in vitro fertilization. N Engl J Med 2019. a;380:325–334. [DOI] [PubMed] [Google Scholar]
- Lensen S, Shreeve N, Barnhart KT, Gibreel A, Ng EHY, Moffett A. In vitro fertilization add-ons for the endometrium: it doesn't add-up. Fertil Steril 2019. b;112:987–993. [DOI] [PubMed] [Google Scholar]
- Lessey BA, Kim JJ. Endometrial receptivity in the eutopic endometrium of women with endometriosis: it is affected, and let me show you why. Fertil Steril 2017;108:19–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W, Suke S, Wertaschnigg D, Lensen S, Wang R, Gurrin L, Mol BW. Randomised controlled trials evaluating endometrial scratching: assessment of methodological issues. Hum Reprod 2019;34:2372–2380. [DOI] [PubMed] [Google Scholar]
- Muteshi CM, Ohuma EO, Child T, Becker CM. The effect of endometriosis on live birth rate and other reproductive outcomes in ART cycles: a cohort study. Hum Reprod Open 2018;2018:hoy016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nastri CO, Lensen SF, Gibreel A, Raine-Fenning N, Ferriani RA, Bhattacharya S, Martins WP. Endometrial injury in women undergoing assisted reproductive techniques. Cochrane Database Syst Rev 2015;3:CD009517. [DOI] [PubMed] [Google Scholar]
- Navot D, Bergh PA. Ovarian hyperstimulation syndrome: a practical approach. In: Ovarian endocrinopathies. The Proceedings of the 8th Reinier de Graaf Symposium Amsterdam, 2-4 September 1993; p.215–225.
- Panagiotopoulou N, Karavolos S, Choudhary M. Endometrial injury prior to assisted reproductive techniques for recurrent implantation failure: a systematic literature review. Eur J Obstet Gynecol Reprod Biol 2015;193:27–33. [DOI] [PubMed] [Google Scholar]
- Pellicer A, Navarro J, Bosch E, Garrido N, Garcia-Velasco JA, Remohí J, Simón C. Endometrial quality in infertile women with endometriosis. Ann N Y Acad Sci 2001;943:122–130. [DOI] [PubMed] [Google Scholar]
- Potdar N, Gelbaya T, Nardo LG. Endometrial injury to overcome recurrent embryo implantation failure: a systematic review and meta-analysis. Reprod Biomed Online 2012;25:561–571. [DOI] [PubMed] [Google Scholar]
- Reilly SJ, Glanville EJ, Dhorepatil B, Prentice LR, Mol BW, Johnson NP. The IVF-LUBE trial—a randomized trial to assess Lipiodol(®) uterine bathing effect in women with endometriosis or repeat implantation failure undergoing IVF. Reprod Biomed Online 2019;38:380–386. [DOI] [PubMed] [Google Scholar]
- Reilly SJ, Stewart AW, Prentice LR, Johnson NP. The IVF-Lube Trial: Lipiodol uterine bathing effect for enhancing the results of in vitro fertilisation, a pilot randomised trial. In: Abstract of the 11th World Congress on Endometriosis 2011;S10–S15. [Google Scholar]
- Revel A. Defective endometrial receptivity. Fertil Steril 2012;97:1028–1032. [DOI] [PubMed] [Google Scholar]
- Salehpour S, Zamaniyan M, Saharkhiz N, Zadeh Modares S, Hosieni S, Seif S, Malih N, Rezapoor P, Sohrabi MR, Preventive Gynecology Research Center (PGRC), Shahid Beheshti University of Medical Sciences, Tehran, Iran. Does intrauterine saline infusion by intrauterine insemination (IUI) catheter as endometrial injury during IVF cycles improve pregnancy outcomes among patients with recurrent implantation failure?: an RCT. Int J Reprod Biomed 2016;14:583–588. [PMC free article] [PubMed] [Google Scholar]
- Simón C, Bellver J. Scratching beneath ‘The Scratching Case’: systematic reviews and meta-analyses, the back door for evidence-based medicine. Hum Reprod 2014;29:1618–1621. [DOI] [PubMed] [Google Scholar]
- Spencer EA, Mahtani KR, Goldacre B, Heneghan C. Claims for fertility interventions: a systematic assessment of statements on UK fertility centre websites. BMJ Open 2016;6:e013940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Hoogenhuijze NE, Kasius JC, Broekmans FJM, Bosteels J, Torrance HL. Endometrial scratching prior to IVF; does it help and for whom? A systematic review and meta-analysis. Hum Reprod Open 2019;2019:hoy025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vercellini P, Consonni D, Dridi D, Bracco B, Frattaruolo MP, Somigliana E. Uterine adenomyosis and in vitro fertilization outcome: a systematic review and meta-analysis. Hum Reprod 2014;29:964–977. [DOI] [PubMed] [Google Scholar]
- Zhou L, Li R, Wang R, Huang HX, Zhong K. Local injury to the endometrium in controlled ovarian hyperstimulation cycles improves implantation rates. Fertil Steril 2008;89:1166–1176. [DOI] [PubMed] [Google Scholar]