Supplemental Digital Content is available in the text
Keywords: coronavirus disease 2019, D-dimer, (MESH) adult, venous thromboembolism
Abstract
Coronavirus disease 2019 (COVID-19) has been associated with increased incidence of venous thromboembolic events (VTE) as well as mortality. D-dimer is a marker of fibrinolysis and has been used as a diagnostic and prognostic marker in VTE among other diseases. The purpose of our study is to describe outcomes from out center and to examine trends in D-dimer levels as it relates to VTE and mortality.
Patients admitted with confirmed COVID-19 cases to Emory Healthcare from March 12, 2020 through April 6, 2020 with measured plasma D-dimer levels were included in our retrospective analysis. Relevant data about comorbidities, hospitalization course, laboratory results, and outcomes were analyzed.
One hundred fifteen patients were included in our study. Mean age was 64 ± 15 years, 47 (41%) females and 84 (73%) African-American. Hypertension was present in 83 (72%) and diabetes in 60 (52%). Mean duration of hospitalization was 19 ± 11 days with 62 (54%) patients intubated (mean duration of 13 ± 8 days). VTE was diagnosed in 27 (23%) patients (mean time to diagnosis 14 ± 9 days). Median D-dimer within the first 7 days of hospitalization was higher (6450 vs. 1596 ng/mL, p < 0.001) in VTE cases compared to non-VTE cases, and was predictive of VTE (area under the curve [AUC] = 0.72, optimal threshold 2500 ng/mL) although not of mortality (AUC 0.55, P = .34). Change in D-dimer level (AUC = 0.72 P = .004) and rate of D-dimer rise (AUC = 0.75 P = .001) were also predictive of VTE, though neither predicted death (P > .05 for all). Within the first 7 days of hospitalization, peak D-dimer level of >2500 ng/mL and a rate of change exceeding 150 ng/mL/d were predictive of future diagnosis of VTE. Rise in D-dimer >2000 ng/mL within any 24 hour period through hospital day 10 had 75% sensitivity and 74% specificity for diagnosis of VTE.
We found that both magnitude and rate of rise in d-dimer within the first 10 days of hospitalization are predictive of diagnosis of VTE but not mortality. These parameters may aid in identifying individuals with possible underlying VTE or at high risk for VTE, thereby guiding risk stratification and anticoagulation policies in COVID-19 patients.
Key Points
Question: Can d-dimer trends in hospitalized patients with COVID-19 assist with prediction of adverse clinical outcomes such as VTE?
Findings: Median, magnitude, and rate of rise in d-dimer within 10 days of hospitalization, were predictive of a diagnosis of VTE. A d-dimer level of >2500 ng/mL, a rate of change that exceeds 150 ng/mL/d, or a rise in d-dimer level within any 24 hour period of >2000 ng/mL were predictive of a future diagnosis of VTE.
Meaning: D-dimer parameters can guide risk stratification of VTE and help direct evaluation and treatment in patients with COVID-19.
1. Introduction
Coronavirus disease 2019 (COVID-19) continues to be a significant source of morbidity and mortality worldwide.[1] Although descriptions of adverse outcomes and associated clinical characteristics have been reported, biomarkers which may serve as early predictors of adverse outcomes have yet to be fully characterized.[2,3] Amongst biomarkers trends in plasma D-dimer are of particular interest, given reports of hypercoagulability in critically ill patients with COVID-19. Venous thromboembolic events (VTE) events have been shown to be highly prevalent in COVID-19, with cited incidence rates as high as 20% to 25% in hospitalized patients and higher in critically ill patients with prolonged hospitalizations.[4,5] D-dimer has additionally been reported in cohort studies to be associated with poor outcomes in hospitalized patients with COVID-19.[6–8]
Historically, D-dimer has been used primarily as a biomarker of fibrinolysis in the diagnostic evaluation of VTE, as well as a prognostic marker for adverse outcomes in various diseases such as pulmonary embolism (PE), malignancy, cardiovascular disease, and sepsis.[9,10] Recently published cohort studies have suggested that D-dimer in isolation or in combination with measures of clinical severity of organ dysfunction, is a potential predictor of mortality in hospitalized patients with COVID-19.[8] A comprehensive description of trajectories of change in D-dimer levels in COVID-19 patients is lacking, and whether early levels and/or the early rate of change in D-dimer levels are predictive of risk of VTE or death remain unknown.
In an attempt to inform clinical practice, we examined trends in D-dimer levels in a cohort of 128 consecutive hospitalized patients with COVID-19 across our healthcare system, and examined the relationships between D-dimer levels, VTE, and mortality.
2. Methods
2.1. Study design
This was a retrospective cohort analysis occurring in an urban, academic, tertiary referral center. Patients 18 years of age or older with diagnosis of COVID-19, confirmed via polymerase chain reaction, were identified from a survey of all hospitalized patients admitted to Emory Healthcare March 12, 2020 thru April 6, 2020. Electronic medical records were reviewed from admission through discharge or until the censor date of April 30, 2020. This study was approved by the Emory University Institutional Review Board.
2.2. Data sources and definitions
Basic demographics including age, sex, race, body mass index, and comorbid diseases were collected. Length of stay, intensive care unit (ICU) admission rates and length of ICU stay, intubation, and duration of mechanical ventilation, and final disposition were additionally recorded. Peak, median, and delta D-dimer along with utilization of therapeutic anticoagulation, and clinical endpoints were additionally examined.
The primary endpoint was vascular events – collectively termed as VTE – and included deep venous thrombosis (DVT), PE, and central venous or dialysis access clots. Hospital mortality was a separate co-primary endpoint. DVT was diagnosed by duplex ultrasound and PE by a combination of CT, CT angiogram, ventilation/perfusion scans, and echocardiography. Venous access and renal replacement therapy circuit clot presence was determined by the bedside providers. Secondary outcomes were major bleeding occurrences, discharge, and readmission greater than 7 days from discharge.
Plasma D-dimer levels were measured using high sensitivity latex dimer assay (Instrumentation Laboratories, Bedford, MA). Assays were performed using 3.2% citrated plasma. Most patients had a single D-dimer level on a given day; however, the mean value was used if more than 1 D-dimer was measured on a single day. Missing data were not imputed.
2.3. Statistical analysis
Descriptive statistics were used to summarize the data; results are reported as percentages for categorical variables and as ranges, means or medians with standard deviations and interquartile ranges for continuous or ordinal variables as based on their distribution. Comparisons between means and medians of continuous variables were assessed with independent T-tests (2-tailed) or by the Mann–Whitney U non-parametric test for skewed variables respectively. Categorical variables were compared with Pearson Chi-square and Fisher Exact tests. Significance for all descriptive analyses was set at P < .05. Univariable analyses were conducted using binary logistic regression for binary outcome variables (VTE or death) to identify predictors of these outcomes and included variables such as age, race, body mass index, sex, disease severity, D-dimer parameters, ICU admission, intubation status, and comorbid conditions. Univariate predictors (P < .05) were then considered in multivariable binary logistic regression analyses to identify independent predictors of the outcome variables (adjusted P < .05).
D-dimer was analyzed using several approaches. Individual patient D-dimer levels throughout hospitalization were summarized as median and maximum D-dimer values. The number of D-dimer tests performed on each day of hospitalization for the entire cohort was assessed. D-dimer trends were examined in a subset of patients who had at least 3 D-dimer values reported within the first 7 days of hospitalization. A linear regression model was used to calculate the rate of early change in D-dimer levels for each patient. The maximum D-dimer level and the slope or rate of rise in D-dimer within the first 7 days were analyzed as predictors of VTE or mortality using binary logistic regression.
An additional subset analysis of D-dimer trends was examined to assess for relative and absolute changes in D-dimer before diagnosis of VTE. Patients who were anticoagulated or diagnosed with VTE within the first 3 days were excluded from this (and all) subset analyses. For this analysis, ceiling D-dimer in VTE patients was defined as the maximum D-dimer value within 48 hours before VTE diagnosis. In the non-VTE group, the ceiling D-dimer was calculated as the D-dimer value measured on day 10 (+–2 days) of hospitalization, since the majority of VTE diagnoses were made after this day in our cohort. Specific to this sub-analysis, for both VTE and non-VTE groups, the baseline D-dimer (or admission D-dimer level) was defined as the mean within the first 5 days of hospitalization in order to overcome inconsistencies in daily D-dimer measurements across patients especially within the early admission period. Analysis was done using binary logistic regression in relation to the end points of VTE or death. A third analysis of D-dimer trends was performed to assess whether the magnitude of rise in D-dimer levels in any given 24 hour period within the first 10 days of hospitalization, was predictive of VTE diagnosis or mortality.
Receiver operating characteristic (ROC) curve analysis was performed to determine the area under the curve (AUC) as a measure of discriminative power, and sensitivity, specificity, positive predictive value, and negative predictive values at each cut off were assessed to identify optimal thresholds of D-dimer parameters as predictors of outcome variables. ROC analyses were performed using hospitalization-wide D-dimer levels as well as subset analysis to examine D-dimer parameters within the first 7 days (maximum and rate of change), absolute and relative changes in D-dimer, and maximal consecutive rises in D-dimer. Statistical analyses were performed using SPSS (Ver. 25) and figures were generated using either SPSS and Graphpad Prism (Ver. 6).
3. Results
3.1. Patient characteristics
A total of 115 hospitalized and confirmed COVID-19 cases with D-dimers measured were identified across Emory Healthcare during this study period. The mean age of the cohort was 64 ± 16 years; 47 (41%) were females and 84 (73%) were African-American. Significantly less people with VTE were female compared to without VTE (22% vs 47%, P = .024). Mean body mass index was 30 ± 8 kg/m2. Common comorbidities included hypertension (n = 83, 72%) and diabetes (n = 60, 52%). Significantly more people in the VTE group had hypertension compared to the non-VTE group (93% vs 66%, P < .006). Overall, 79 (69%) of patients were classified as having severe COVID, with higher rates in the VTE group versus non-VTE group (85% vs 64%, P = .036).
The mean duration of hospitalization was 19 ± 11 days with the VTE group being admitted significantly longer than those without VTE (28 ± 9 days vs 17 ± 10 days, P < .001) .77 (67%) patients were admitted to the ICU during their hospitalization with a significantly higher admission rate in the VTE versus non-VTE group (89% vs 60%, P < .005) as well as a longer mean length of stay in the ICU (21 ± 9 vs 12 ± 8, P < .001); 62 patients (54%) were intubated and the mean duration of mechanical ventilation was 13 ± 8 days. A significantly higher rate of those with VTE versus non-VTE were intubated (85% vs 44%, P < .001) as well as a longer mean length of intubation (17 ± 9 vs 11 ± 7, P < .001). Amongst the cohort, 98 (85%) patients were either discharged (n = 61, 53% discharged home) or died (n = 21, 18%) during the study period, while 17 (15%) remained hospitalized and were censored on the last day of the study period (Table 1).
Table 1.
Patient characteristics and outcomes. Demographics and outcomes.
| Baseline characteristics | Total Cohort (n = 115) | VTE (N = 27) | No VTE (N = 88) | P-value |
| Age, mean (SD) | 64 (15) | 64 (14) | 65 (16) | .83 |
| Race, n (%) AA | 84 (73) | 23 (85) | 61 (69) | .14 |
| Female, n (%) | 47 (41) | 6 (22) | 41 (47) | .02 |
| Smoking Ever, n (%) | 28 (24) | 6 (22) | 22 (25) | .77 |
| Obesity, n (%) | 50 (43) | 12 (44) | 38 (43) | .91 |
| BMI, Mean (SD) | 30 (8) | 32 (11) | 30 (7) | .21 |
| H/o HTN, n (%) | 83 (72) | 25 (93) | 58 (66) | .006 |
| H/o DM, n (%) | 60 (52) | 17 (63) | 43 (49) | .20 |
| ESRD on HD, n (%) | 10 (9) | 2 (7) | 8 (9) | 1 |
| H/o VTE, n (%) | 8 (7) | 2 (7) | 6 7) | 1 |
| H/o CAD, n (%) | 17 (15) | 4 (15) | 13 (15) | 1 |
| H/o CVA, n (%) | 17 (15) | 3 (11) | 14 (16) | .76 |
| H/o Afib, n (%) | 9 (8) | 1 (4) | 8 (9) | .68 |
| Known Active Cancer, n (%) | 6 (5) | 2 (7) | 4 (5) | .62 |
| Mean Days from onset to Presentation (SD) | 6 (4) | 7 (5) | 6 (4) | .13 |
| Severe COVID, n (%) | 79 (69) | 23 (85) | 56 (64) | .036 |
| Hospitalization Details | ||||
| Duration of Admission, mean days (SD) | 19 (11) | 28 (9) | 17 (10) | <.001 |
| ICU, n (%) | 77 (67) | 24 (89) | 53 (60) | .005 |
| Length of ICU Stay, mean days (SD), n = 77 | 15 (9) | 21 (9) | 12 (8) | <.001 |
| Intubation, n (%) | 62 (54) | 23 (85) | 39 (44) | <.001 |
| Length of intubation, mean days (SD), n = 62 | 13 (8) | 17 (9) | 11 (7) | <.001 |
| D dimer >2000 Any, n (%) | 83 (72) | 26 (96) | 57 (65) | .001 |
| D dimer >3000 Any, n (%) | 74 (64) | 26 (96) | 48 (55) | <.001 |
| D dimer median (IQR), n = 115 | 2031 (1078-6084) | 6450 (4171-17285) | 1596 (910-4166) | <.001 |
| CRP median (IQR), n = 113 | 135 (82-195) | 138 (83-208) | 132 (78-193) | .55 |
| Non-prophylactic anticoagulation, n (%) | 59 (51) | 25 (93) | 34 (39) | <.001 |
| Outcomes | ||||
| Any venous vascular event, n (%) | 27 (23) | N/A | N/A | N/A |
| DVT, n (%) | 12 (10) | 12 (44) | N/A | N/A |
| PE, n (%) | 5 (4) | 5 (19) | N/A | N/A |
| Other vascular conditions, n (%) | 1 (1) | 1 (4) | N/A | N/A |
| Death, n (%) | 21 (18) | 6 (22) | 15 (17) | 0.54 |
| Venous line clots, n (%) | 14 (12) | 14 (52) | N/A | N/A |
| Days until VTE diagnosis, mean (SD) | 14 (9) | N/A | N/A | N/A |
| Days until line clot diagnosis, mean (SD) | 10 (5) | N/A | N/A | N/A |
| Days until starting anticoagulation, mean (SD) (n = 59) | 8 (6) | 10 (6) | 7 (6) | .055 |
| Readmission (>7 d after discharge), n (%) | 1 (1) | 0 | 1 (1) | 1 |
| Major bleeding, n (%) | 5 (4) | 2 (7) | 3 (3) | .34 |
| Discharge or death, n (%) | 98 (85) | 20 (74) | 77 (88) | .09 |
| Discharge disposition home, n (%) | 61 (53) | 10 (37) | 51 (58) | .06 |
3.2. VTE frequency and therapeutic anticoagulation in COVID19 patients
A diagnosis of VTE was made in 27 (23%) patients. Mean time to diagnosis of VTE was 14 ± 9 days. The most common forms of VTE included DVT (n = 12), PE (n = 5), and venous line clots (n = 14) (Table 1). Fifty nine patients (51%) received therapeutic anticoagulation primarily using intravenous unfractionated heparin (71%), low molecular weight heparin (17%), and direct thrombin inhibitors (61%) (Supplemental Table 1). Therapeutic anticoagulation initiation was significantly higher in the VTE group compared to the non-VTE group (93% vs 39%, P < .001). The mean time to initiation of therapeutic anticoagulation was 8 ± 6 days and was not significantly higher in anticoagulated patients diagnosed with VTE compared to those without a confirmed VTE (VTE: mean 10 ± 6 days vs no VTE: mean 7 ± 6 days, P = .055).
3.3. D-dimer trends in hospitalized COVID19 patients
A total of 1457 daily D-dimer measurements were performed [median d-dimer measurements per patient 11 (IQR 5–19). Predominance of all D-dimer tests (80%) were performed on or before day 21 (Supplemental Fig. 1). For the entire cohort, median D-dimer for the entire hospitalization was 2031 (IQR 1078–6089 ng/mL) and followed a right-ward skewed distribution. Median D-dimer was significantly higher in the VTE group (6450[IQR 4171–17285] vs 1596 [910–4166] P < .001). Basedon peak hospitalization-wide D-dimer levels, 74 had peak D-dimer >3000 and 83 had peak D-dimer >2000 ng/mL. D-dimer >2000 ng/mL (96% vs 65%, P = .001) and D-dimer >3000 ng/mL (96% vs 55%, P < .001) was significantly higher in the VTE group. We assessed trajectories of D-dimer over the first 25 days of hospitalization and observed that D-dimer values increased in a nearly-linear fashion from day 1 of admission to day 10, after which levels plateaued. Patients with VTE – as compared to patients without VTE – had a higher peak D-dimer (12,954 IQR 5386–28,425 vs 2095 IQR 1148–8885 ng/mL, P = .01) and a steeper slope of increase (487 vs 202 ng/mL/d, P = .002) over the first 10 days (Fig. 1). In contrast, trajectories of D-dimer were similar in the patients who died versus those who survived (D-dimer slope 228 vs 269 ng/mL/d, P = .74).
Figure 1.
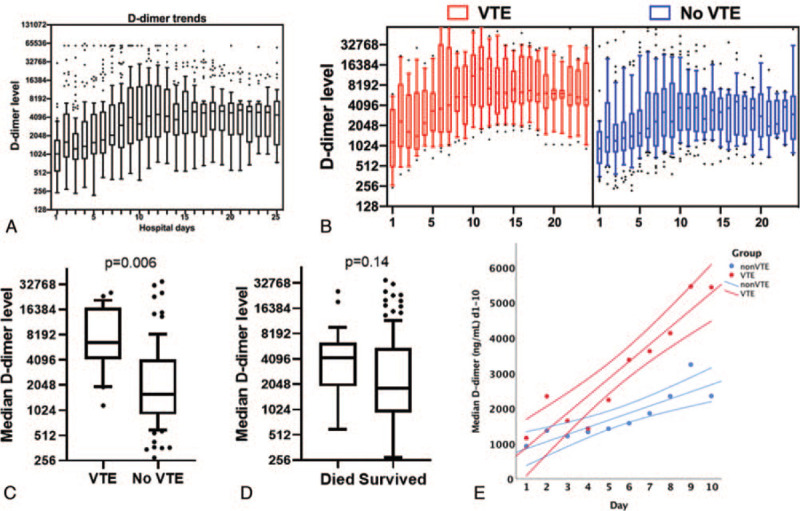
Trajectories of D-dimer changes in hospitalized COVID-19 patients and association with VTE and mortality. (A) Summary of D-dimer trajectories for 115 hospitalized COVID-19 patients over the first 25 d of hospitalization (box plots with median values for each day are shown). (B) Summary of D-dimer trajectories for patients diagnosed with VTE (red) and without VTE (blue). (C) Comparison of hospitalization-wide median D-dimer in VTE and non-VTE groups. (D) Comparison of hospitalization-wide median D-dimer in patients who died and those who survived. Mann–Whitney U test P-value shown for C and D. (E) Comparison of rate of rise in median D-dimer values during the first 10 d of hospitalization in VTE (red) and non-VTE patients. Linear slope and 95% confidence intervals of the mean are shown. COVID-19 = coronavirus disease 2019, VTE = venous thromboembolism.
3.4. Association between D-dimer levels, diagnosis of VTE and in-patient mortality
Median D-dimer levels were significantly higher in patients with VTE compared to those without VTE (6540 IQR 4171–17,285 vs 1596 IQR 910–4166 ng/mL, P < .001). ROC analyses demonstrated median D-dimer levels had moderate discriminative power to detect VTE (AUC = 0.82), but not mortality (AUC = 0.64) (Supplemental Fig. 2). In unadjusted analyses, any peak D-dimer >2000 ng/mL or D-dimer >3000 ng/mL was associated with VTE diagnosis (odds ratio [OR] 14.140, 95% confidence interval [CI] 1.830–109.253, P = .011 and OR 21.667, 95% CI 2.815–166.780, P = .003, respectively). Other univariate predictors of VTE were female sex (OR 0.328, 95% CI 0.121–0.890, P = .029), history of hypertension (OR 6.466, 95% CI 1.434–29.157, P = .015), ICU admission (OR 5.283, 95% CI 1.478–18.885, P = .010), and intubation (OR 7.224: 95% CI 2.306–22.635, P = .001) (Supplemental Table 2). After adjusting for sex, hypertension, and ICU admission, both D-dimer thresholds were independently associated with VTE diagnosis; no other variables achieved significance (OR 9.592, 95% CI 1.110–82.866 P = .040 and OR 15.595, 95% CI 1.902–127.848, P = .010, respectively for D-dimer 2000 ng/mL and 3000 ng/mL thresholds) (Supplemental Table 3). Univariate predictors of death were age (OR 1.087, 95% CI 1.042–1.133, P < .001), history of COPD (OR 7.667, 95% CI 1.195–49.206, P = .032), ICU admission (OR 5.897, 95% CI 1.296–26.831, P = .022), intubation (OR 3.339, 95% CI 1.131–9.858, P = .029), and D-dimer >3000 ng/mL (OR 4.071, 95% CI 1.121–14.788, P = .033), but not D-dimer >2000 ng/mL (OR 4.453, 95% CI 0.974–20.365, P = .054). After adjusting for age, COPD, and ICU admission, D-dimer >3000 ng/mL was no longer significant (OR 4.847, 95% CI 0.933–25.167, P = .060).
Since VTE for our study included DVT/PE and vascular access thrombosis, we examined whether the association between D-dimer with VTE was confounded by type of VTE. Of the 27 patients diagnosed with VTE, 12 were DVT, 5 were PE, and 14 were venous line clots. Hospitalization-wide median D-dimer levels were similar in patients with line-associated VTE or with DVT/PE (6700 IQR 4719–17539 ng/mL vs 6375 IQR 3906–9660 ng/mL, respectively, P = .8). Hospitalization-wide median D-dimer >3000 ng/mL was equally predictive of non-line associated (AUC = 0.79) and line-associated VTE (0.78), indicating that the association between D-dimer levels and VTE was not related to VTE subtype.
3.5. Early changes in D-dimer levels are predictive of VTE in hospitalized COVID19 patients.
While our observed associations between hospitalization-wide median D-dimer values and VTE in COVID19 patients support a possible relationship between underlying hypercoagulability and VTE, it does not provide temporality. We assessed whether absolute and relative change in D-dimer levels before diagnosis of VTE were associated with VTE diagnosis by limiting the analysis to a subset of patients who had at least 2 D-dimer levels measured in the first 10 days of hospitalization, were not therapeutically anti-coagulated within the first 3 days and were not diagnosed with a VTE within the first 3 days of hospitalization. Based on these criteria, 25 VTE cases and 55 non-VTE cases were included in this analysis. Baseline and ceiling D-dimer (maximum D-dimer level within the specified timeframe) values were assessed before VTE diagnosis in VTE positive patients, and before10 (+–2 days) in the non-VTE group. Baseline D-dimer was not associated with VTE (AUC = 0.49, P = .86) while pre-VTE ceiling D-dimer (AUC = 0.70, P = .008), delta increase in D-dimer (absolute difference) (AUC = 0.72, P = .004) and rate of D-dimer rise (delta D-dimer/days between baseline and ceiling levels) (AUC = 0.75, P = .001) were all predictive of VTE (Fig. 2). None of these parameters were predictive of death (P > .05 for all); thresholds for these parameters to rule out VTE presented in Table 2A.
Figure 2.
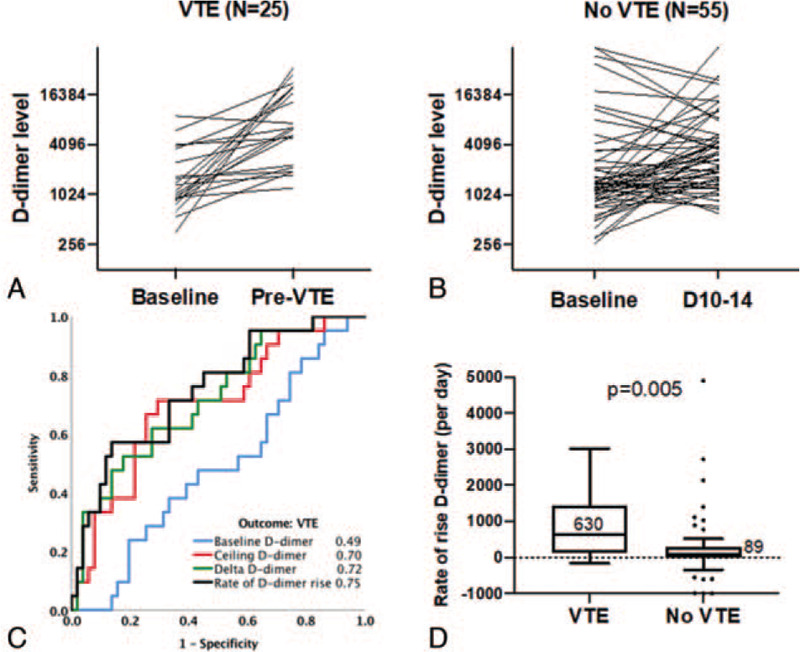
Early changes in D-dimer predict VTE in hospitalized COVID-19 patients. (A) Individual patient-level trajectories of change in D-dimer in VTE patients, from baseline (average d-dimer between days 1–3) and ceiling levels (day before VTE diagnosis). (B) Individual patient-level trajectories of change in D-dimer in non-VTE patients, from baseline (average d-dimer between days 1–3) and ceiling levels (average D-dimer level between day 10–14). (C) ROC analysis with AUC values for baseline D-dimer, ceiling D-dimer, delta rise in D-dimer (ceiling minus baseline) and rate of change in D-dimer as predictors of VTE. AUC values are indicated. (D) Comparison of median rates of rise in D-dimer in VTE and non-VTE patients (Mann–Whitney U P-value is indicated). AUC = area under the curve, COVID-19 = coronavirus disease 2019, ROC = receiver operator characteristic curve, VTE = venous thromboembolism.
Table 2.
Early D-dimer trends and associations with VTE.
| A. Changes in D-dimer pre-VTE and association with VTE (N = 80) | ||||
| 1. Variable: Ceiling D-dimer (ng/mL) (pre-VTE or d10-14) | ||||
| Threshold | Sensitivity | Specificity | PPV | NPV |
| 1500 | 92 | 21.8 | 34.8 | 85.7 |
| 2000 | 80 | 32.7 | 35.1 | 78.2 |
| 3000 | 72 | 47.3 | 38.3 | 78.8 |
| 4000 | 72 | 58.2 | 43.9 | 82.1 |
| 5000 | 68 | 70.9 | 51.5 | 83 |
| 2. Variable: Delta D-dimer (ng/mL) | ||||
| Threshold | Sensitivity | Specificity | PPV | NPV |
| 100 | 96 | 32.7 | 39.3 | 94.7 |
| 250 | 92 | 34.5 | 39 | 90.5 |
| 500 | 84 | 40 | 38.9 | 84.6 |
| 1000 | 76 | 50.9 | 41.3 | 82.3 |
| 1500 | 64 | 56.4 | 40 | 77.5 |
| 2500 | 64 | 67.3 | 47.1 | 80.4 |
| 4500 | 96 | 32.7 | 56.8 | 79 |
| 3. Variable: Rate of D-dimer rise (delta d-dimer/d) | ||||
| Threshold | Sensitivity | Specificity | PPV | NPV |
| 90 | 84 | 50.9 | 44.5 | 87.5 |
| 150 | 72 | 61.8 | 46.1 | 82.9 |
| 300 | 60 | 74.5 | 51.7 | 80.4 |
| 500 | 56 | 83.6 | 60.1 | 80.7 |
| B. D-dimer trends within the first 7 d of hospitalization and association with VTE (N = 78) | ||||
| 1. Variable: D-dimer (ng/mL) max d1–7 | ||||
| Threshold | Sensitivity | Specificity | PPV | NPV |
| 1500 | 95 | 37.9 | 34.5 | 95.7 |
| 2000 | 75 | 53.4 | 35.7 | 86.1 |
| 2500 | 75 | 62.1 | 40.6 | 87.8 |
| 3000 | 70 | 63.8 | 40 | 86.1 |
| 4000 | 60 | 65.5 | 37.5 | 82.6 |
| 5000 | 55 | 69 | 38 | 81.6 |
| 2. Variable: D-dimer slope (ng/mL/d) d1–7 | ||||
| Threshold | Sensitivity | Specificity | PPV | NPV |
| 100 | 85 | 50 | 37 | 90.6 |
| 150 | 85 | 60 | 42.3 | 92.1 |
| 200 | 70 | 66 | 41.5 | 86.5 |
| 300 | 50 | 74 | 40 | 81.1 |
| 1000 | 45 | 83 | 47.7 | 81.4 |
Given the apparently linear rise in d-dimer levels within the first 7 days of hospitalization, we next hypothesized that maximum D-dimer levels and the rate of rise in D-dimer levels within the first 7 days of admission predict VTE and death. 78 patients without therapeutic anticoagulation therapy within the first 3 days of admission or VTE diagnoses within the first 3 days of admission, and with at least 3 D-dimer levels collected within the first 7 days of hospitalization--were included and among these. Twenty had a VTE diagnosed (mean day of diagnosis 11.3 ± 5.7 days) and 16 patients died (mean day of death 20 ± 10 days). We used 2 parameters to describe early changes in D-dimer: (1) maximum D-dimer within the first 7 days, and (2) rate of change in D-dimer within the first 7 days, calculated as the slope of a linear equation (rate of change in D-dimer per day). The median of D-dimermaxd1-7 was 2290 (IQR 1350–3517 ng/mL) and was significantly higher (P = .005) in VTE cases (median 6200 IQR 2262–31,056 ng/mL) compared to non-VTE cases (median 1933 IQR 1144–6686 ng/mL). D-dimermaxd1-7 was predictive of VTE (AUC = 0.72) with an optimal threshold of 2500 ng/mL (Table 2B), but was not predictive of death (AUC 0.55, P = .34). A D-dimermax d1-7 >2500 ng/mL was associated with higher odds of VTE (OR = 3.2, 95% CI 1.19.2, P = .029) in unadjusted analyses, but not after adjustment for age, sex, ICU admission, and hypertension (ORadj = 2.14, 95% CI 0.0.69–6.7, P = .19). The median D-dimer slope was 162.7 (IQR 41.8–968.4) ng/mL/d for the entire cohort and was significantly higher in patients with VTE (518 IQR 171–6,968 ng/mL/d) compared to those without VTE (103 IQR 36.3–1606.2 ng/mL/d) (Fig. 3, P = .004). D-dimer slope was predictive of VTE (AUC = 0.71) with an optimal threshold of 150 ng/mL/d, but was not predictive of death (AUC 0.57, P = .51). A D-dimer slope >150 ng/mL/d was associated with higher odds of VTE (OR = 8.6, 95% CI 2.27–32.8, P = .002) in unadjusted analyses as well as after adjustment for age, sex, ICU admission, and hypertension (ORadj = 6.7, 95% CI 1.65–27.3, P = .008). Neither a D-dimer max >2500 ng/mL nor a D-dimer slope >150 ng/mL/d were predictors of death (P = .35 and .66, respectively).
Figure 3.
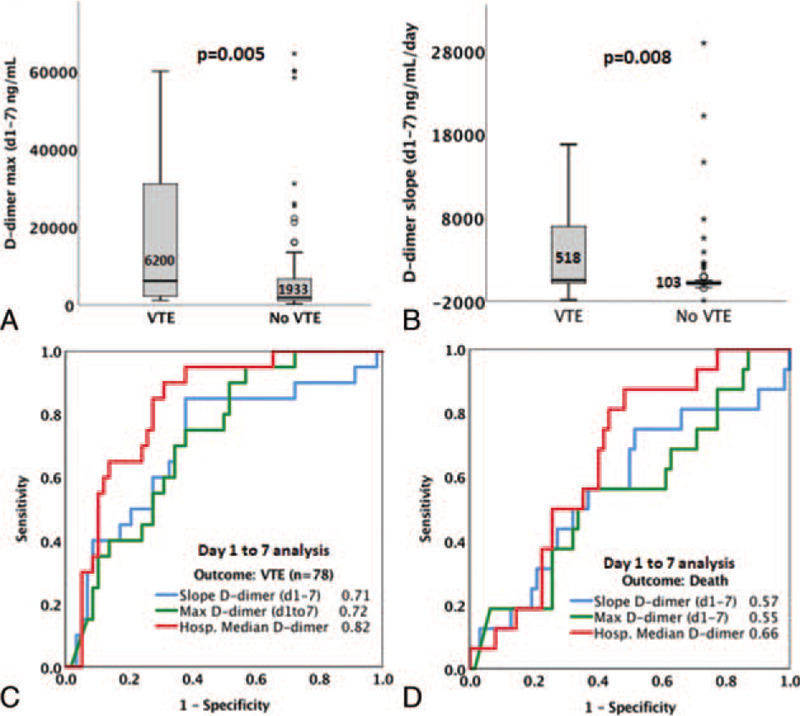
D-dimer changes within the first 7 d of hospitalization predict VTE in COVID-19 patients. (A) Comparison of medians of maximum D-dimer levels attained within the first 7 d of admission in VTE and non-VTE patients. (B) Comparison of medians of D-dimer slopes (rate of change in D-dimer per day) within the first 7 d of admission in VTE and non-VTE patients. For A and B, Mann–Whitney U P-values are indicated. (C, D) ROC analyses comparing discriminative power (AUC) for changes in D-dimer within 7 d of hospitalization to predict VTE (C) or mortality (D). AUCs are indicated for each predictor. AUC = area under the curve, COVID-19 = coronavirus disease 2019, VTE = venous thromboembolism.
Based on the observed increasing trend of D-dimer levels in the first 10 days of hospitalization, we also determined whether maximum rise in D-dimer within any 24 hour period is predictive of VTE or of death. We included 80 patients who were not therapeutically anticoagulated or diagnosed with VTE within the first 3 days of admission in this analysis. Maximum rise in D-dimer in a 24 hour period within the first 10 days was predictive of VTE (AUC = 0.75) and rise >2000 ng/mL within a 24 hour period had a 75% sensitivity and 74% specificity for diagnosis of VTE. Additionally, a rise in D-dimer >2000 ng/mL over 24 hours was significantly associated with VTE in unadjusted (OR = 6.56, 95% CI 2.16–19.99) and adjusted analyses (OR = 4.61, 95% CI 1.42–14.9) after controlling for hypertension and ICU admission (Supplemental Fig. 3).
4. Discussion
Our comprehensive analysis of D-dimer trajectories in this cohort identified several trends that can be used to aid in VTE diagnosis and risk stratification. Median levels, maximum, and rate of rise in D-dimer within the first 7 to 10 days of hospitalization were all predictive of a diagnosis of VTE. A D-dimer level of >2500 ng/mL within the first 7 days of admission, a rate of change that exceeds 150 ng/mL/d over the first 7 days, or a rise in D-dimer level within any 24 hour period of >2000 ng/mL were highly predictive of a future diagnosis of VTE. While the role and the intensity of empiric anticoagulation based on D-dimer levels in COVID-19 patients remains to be clarified, it is possible that the D-dimer thresholds we have identified can guide anticoagulation regimens in hospitalized COVID-19 patients.
Our results confirm observations from other recent publications on COVID-19. The observed frequency of VTE (23%) in our cohort was consistent with rates published by others.[4,5] Significant differences in D-dimer levels amongst subsets of patients diagnosed with VTE, compared to those without VTE were also consistent with the existing literature.[4] Additionally, we observed a significant association between hypertension and VTE risk; consistent with reported associations between COVID-19 disease severity and hypertension.[11] Unlike other reports, we did not find a significant association between any of the D-dimer metrics with mortality.[12] We suspect this may be related to nearly 50% of patients in our cohort being on therapeutic anticoagulation, which may have reduced mortality as reported in prior studies.[13,14]
The limitations of our study include its retrospective nature as well as rapidly evolving literature and treatment paradigms surrounding COVID-19. Anticoagulation guidelines and regimens were optimized based on emerging literature, potentially leading to the lack of association between D-dimer levels and mortality. While we show a correlation between D-dimer and VTE, potential confounders include the presence of comorbidities and other baseline demographics which could also impact D-dimer levels. Observations by us and others do suggest an increased incidence of vascular and hypercoagulable events in hospitalized COVID-19 patients, suggesting the validity of our findings although the lack of a control (non-COVID19) group limits our ability to confirm this. Another limitation was inconsistency in daily D-dimer testing in all patients due to lack of a universal policy for daily D-dimer testing. However, a median of 40 tests were performed per day with most tests concentrated within the first 25 days of hospitalization, making our results more generalizable to COVID-19 patients. Patients were censored through the last day of the study. As some patients were still hospitalized at time of study completion, this may have impacted our results. Lastly, since diagnostic testing for VTE was based on clinical suspicion rather than based on a universal screening approach, it is possible that some VTE's may have gone undiagnosed. Strengths of our study include a relatively large study cohort and a large set of over 1,400 D-dimer values that were used in our analysis over the study period. We also performed statistically robust analyses of early changes in D-dimer levels using 3 different analytic approaches, increasing the validity of our findings.
In summary, our results confirm a high rate of VTE in hospitalized COVID-19 patients and emphasize the prognostic and diagnostic value of daily D-dimer values in these patients. Based on the trends and the magnitude of D-dimer change within the first 7-10 days, it is possible to identify individuals with potential underlying VTE or at high risk of VTE development. Our results suggest the utility of D-dimer levels in guiding risk stratification, with additional implications for early anticoagulation policy development for patients with COVID-19. Additional exploration of mortality predictors and characterization of thrombotic events in patients with COVID is needed to further guide the direction of therapy and management guidelines.
Author contributions
Conceptualization: Christina Creel-Bulos, Michael Liu, Sara C. Auld, Manila Gaddh, Christine L. Kempton, Milad Sharifpour, Roman M. Sniecinski, Cheryl L. Maier, Fadi B. Nahab, Srikant Rangaraju.
Data curation: Michael Liu, Fadi B. Nahab, Srikant Rangaraju.
Formal analysis: Michael Liu, Srikant Rangaraju.
Investigation: Christina Creel-Bulos, Michael Liu, Sara C. Auld, Manila Gaddh, Christine L. Kempton, Milad Sharifpour, Roman M. Sniecinski, Cheryl L. Maier, Fadi B. Nahab, Srikant Rangaraju.
Project administration: Christina Creel-Bulos, Fadi B. Nahab.
Supervision: Fadi B. Nahab, Srikant Rangaraju.
Writing – original draft: Christina Creel-Bulos, Michael Liu, Sara C. Auld, Manila Gaddh, Christine L. Kempton, Milad Sharifpour, Roman M. Sniecinski, Cheryl L. Maier, Fadi B. Nahab, Srikant Rangaraju.
Writing – review & editing: Christina Creel-Bulos, Michael Liu, Sara C. Auld, Manila Gaddh, Christine L. Kempton, Milad Sharifpour, Roman M. Sniecinski, Cheryl L. Maier, Fadi B. Nahab, Srikant Rangaraju.
Supplementary Material
Supplementary Material
Supplementary Material
Supplementary Material
Supplementary Material
Supplementary Material
Footnotes
Abbreviations: AUC = area under the curve, COVID-19 = coronavirus disease 2019, DVT = deep vein thrombosis, ICU = intensive care unit, OR = odds ratio, PE = pulmonary embolism, ROC = receiver operator characteristic curve, VTE = venous thromboembolism.
How to cite this article: Creel-Bulos C, Liu M, Auld SC, Gaddh M, Kempton CL, Sharifpour M, Sniecinski RM, Maier CL, Nahab FB, Rangaraju S. Trends and diagnostic value of D-dimer levels in patients hospitalized with coronavirus disease 2019. Medicine. 2020;99:46(e23186).
CC-B and ML contributed equally to this study as first authors.
FBN and SR contributed equally to this study as senior and co-corresponding authors.
This work was performed at the Emory University Hospital in Atlanta, Georgia
Reprints will not be ordered.
This submission is not under consideration elsewhere. The content of this submission has not been previously published. The authors have read and approved the manuscript.
The authors have read and approved the manuscript (no other authors).
This work was supported in part by the following grants: NIH/NIAID K23 AI134182 (SCA), 1K08NS099474-01 (SR) and NIH/CTSA UL1TR002378.
The authors have no conflicts of interest to disclose.
Data access, responsibility and analysis: ML and SR had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
AA = African American, BMI = body mass index, CRP = C reactive protein, H/o = history of.
NPV = Negative predictive value, VTE = venous thromboembolism, PPV = Positive predictive value.
References
- [1].COVID-19 Map. Johns Hopkins Coronavirus Resource Center. Available at: https://coronavirus.jhu.edu/map.html [access May 12, 2020]. [Google Scholar]
- [2].Bhatraju PK, Ghassemieh BJ, Nichols M, et al. Covid-19 in critically ill patients in the seattle region - case series. N Engl J Med 2020;382:2012–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Young BE, Ong SWX, Kalimuddin S, et al. Epidemiologic features and clinical course of patients infected with SARS-CoV-2 in Singapore. JAMA 2020;323:1488–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Cui S, Chen S, Li X, et al. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost 2020;18:1421–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Middeldorp S, Coppens M, van Haaps TF, et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost 2020;18:1995–2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Ruan Q, Yang K, Wang W, et al. Correction to: clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Medicine 2020;46:1294–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Ruan Q, Yang K, Wang W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med 2020;46:846–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020;395:1054–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Halaby R, Popma CJ, Cohen A, et al. D-Dimer elevation and adverse outcomes. J Thromb Thrombolysis 2015;39:55–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Querol-Ribelles JM, Tenias JM, Grau E, et al. Plasma d-dimer levels correlate with outcomes in patients with community-acquired pneumonia. Chest 2004;126:1087–92. [DOI] [PubMed] [Google Scholar]
- [11].Lippi G, Wong J, Henry BM. Hypertension in patients with coronavirus disease 2019 (COVID-19): a pooled analysis. Pol Arch Intern Med 2020;130:304–9. [DOI] [PubMed] [Google Scholar]
- [12].Zhang L, Yan X, Fan Q, et al. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J Thromb Haemost 2020;18:1324–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Tang N, Bai H, Chen X, et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost 2020;18:1094–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Paranjpe I, Fuster V, Lala A, et al. Association of treatment dose anticoagulation with in-hospital survival among hospitalized patients with COVID-19. J Am Coll Cardiol 2020;76:122–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


