Abstract
Background
Telfairia occidentalis (TO) has many biological activities including blood glucose regulation. Thus, it is being used in the treatment of diabetes mellitus. TO has been shown to cause insulin-mediated hypoglycaemia, which leads to post-hypoglycaemic hyperglycaemia. However, the mechanism involved in the post-hypoglycaemic hyperglycaemia is still poorly understood.
Objective
This research was designed to determine the response of glucoregulatory hormones and enzymes to TO treatment.
Methods
Thirty-five male Wistar rats were divided into seven oral treatment groups (n = 5/group), which received either of 100 mg/kg or 200 mg/kg TO for 7-, 10- or 14 days.
Results
The 7-day treatment with TO significantly increased the levels of insulin, glucagon, and glucose-6-phosphatase (G6Pase) activity but decreased the levels of glucose, adrenaline, and glucokinase (GCK) activity. The 10-day treatment with 100 mg/kg TO increased glucose and decreased GCK activity while 200 mg/kg for the same duration increased glucose, insulin, GCK and G6Pase activities but reduced glucagon. The 14-day treatment with 100 mg/kg TO decreased glucose and glucagon but increased cortisol, while 200 mg/kg TO for same duration increased insulin, but reduced glucagon and GCK activity.
Conclusion
The TO’s post-hypoglycaemic hyperglycaemia results from increased glucagon and G6Pase activity, and reduced GCK activity. Moreover, the glucagon response mainly depends on glucose rather than insulin.
Keywords: Plasma glucose, Insulin, Counter-regulatory hormones, Glucoregulatory enzymes, Telfairia occidentalis
Abbreviations: TO, Telfairia occidentalis; LDH, Lactate dehydrogenase; G6Pase, Glucose-6-phosphatase; NIH, National Institutes of Health; GCK, Glucokinase; NAD, Nicotinamide adenine dinucleotide; G6PD, Glucose-6-phosphate dehydrogenase; ELISA, Enzyme-linked immunosorbent assay; Avidin-HRP, Avidin-Horseradish Peroxidase; SPSS, Statistical Package for the Social Sciences; SEM, Standard error of mean; ANOVA, Analysis of Variance; LSD, Least Significance Difference; cAMP, Cyclic adenosine monophosphate; G6P, Glucose-6-phosphate; EGP, Endogenous glucose production
1. Introduction
Telfairia occidentalis Hook.f. (TO), commonly called fluted pumpkin or fluted gourd, is a plant belonging to the family of Cucurbitaceae that is consumed mostly in West African countries such as Nigeria, Ghana and Sierra Leone [1,2]. In Nigeria, TO is known by different names among various tribes such as “iroko” or “apiroko” in Yoruba, “ubong” in Efik, “ugu” in Igbo, “umeke” in Edo, and “umee” in Urhobo [2]. It has been used in traditional medicine for the treatment of malaria and convulsion attack [3]; anaemia [2]; blood and skin diseases [4]; and in the prevention of andropause [5], among other uses.
The effect of TO on blood glucose level is contradictory, as both hypo- and hyperglycaemic effects have been reported. For instance, its hypoglycaemic effect and use for the treatment of blood glucose disorders such as diabetes mellitus have been well-reported [[6], [7], [8], [9], [10]]. Contrarily, a few studies also reported its hyperglycaemic effect [11,12]. However, despite the numerous studies on the glucoregulatory effects of TO, the hypoglycaemic and/or hyperglycaemic mechanism(s) of TO have not been well-investigated. We previously investigated the glycaemic effects of administration of two doses (100- and 200 mg/kg) of TO for one- and two weeks. The results showed reduced plasma glucose, increased levels of insulin and cortisol as well as increased liver lactate dehydrogenase (LDH) activity after one week of treatment but reduced insulin, increased glucose and further increases in cortisol and LDH activity after two weeks. Thus, we concluded that the TO-induced lowering of blood glucose might be mediated through an increase in plasma insulin [13]. The results also showed that TO actually produced hypoglycaemic effect and that the hyperglycaemia seen after prolonged treatment for two weeks [13] or more [11] was probably due to counter-regulation as evidenced by the increased plasma cortisol concentration and LDH activity.
We recently investigated the effects of TO on plasma lactate and glucose, as well as liver glycogen and glucose-6-phosphatase (G6Pase) activity [14]. The results showed that blood glucose also decreased after one week of treatment but increased after two weeks [14] similar to our previous findings [13]. However, lactate, glycogen and G6Pase activity reduced in a dose- and duration-dependent manner after 1 and 2 weeks of treatment; suggesting that TO may be acting through mechanisms that involve regulation of glycolysis, lactate oxidation/gluconeogenesis, and glycogenolysis to reduce or increase the blood glucose.
Hormones and enzymes are known to play critical roles in glucose metabolism. While insulin has been shown to mediate the hypoglycaemic effect of TO, information on the counter-regulatory response viz-a-viz the role of counter-regulatory hormones; and the hormonal and enzymatic interplay during the counter-regulatory response to TO remains poorly understood. The only counterregulatory hormone that has been linked with the increase in blood glucose following long-term treatment with TO is cortisol [13]. However, it is difficult and unreasonable to conclude that cortisol is the only hormone involved in the increased blood glucose following long-term treatment with TO because there are other crucial counter-regulatory hormones such as glucagon and adrenaline whose responses to the TO-induced lowering of blood glucose need to be investigated.
This study aimed at investigating the responses of glucoregulatory hormones and enzymes to the administration of TO. Our findings will provide a better insight into the physiological events leading to the TO-induced changes in blood glucose.
2. Methods
2.1. Preparation of TO aqueous extract
Fresh leaves of TO were sourced locally from a vegetable seller at Yoruba Road, Ilorin, Kwara State, Nigeria. The plant was taken to the Department of Plant Biology, University of Ilorin for authentication and identification by Mr Bolu Ajayi, a curator. The sample was deposited in the Herbarium of the University, and a voucher number (UILH/001/1063) was issued for the specimen. The leaves were washed with water to remove sand and other debris, air-dried and pulverised into powder using an electric blender. A sample weighing 110 g was soaked in 1.5 L of distilled water for about 72 h and stirred at 1-h intervals. The mixture was sieved with a clean white cloth, and the resultant filtrate evaporated in a water bath at 40 °C to obtain a solid greasy extract that yielded 27.27%. The extract was properly labelled and stored in a refrigerator set at 4 °C. The solid extract was dissolved in normal saline and used for the study.
2.2. Experimental animals
Thirty-five (35) male Wistar rats weighing between 150 and 200 g were obtained from a private breeder in Ogbomosho, Oyo State, Nigeria, and housed at room temperature in a wire-wooden cage placed inside the Central Animal House of the Faculty of Basic Medical Sciences, College of Health Sciences, University of Ilorin, Nigeria. The animals had free access to normal pelleted standard rat diet and water ad libitum and maintained on the daily light/dark cycle. “Principles of laboratory animal care (NIH publication No. 85–23, revised 1985)” were followed. All experiments were examined and approved by our institutional ethics committee. All necessary protocols were strictly followed to ensure the humane treatment of the animals.
2.3. Experimental protocol
Following a previously established method for the determination of animal sample size [15], thirty-five (35) male rats were randomly allocated by an invited neutral person who knew nothing about the study into 7 oral treatment groups (n = 5/group) as follows:
Group 1 (control): received only standard diet and 0.2 ml of normal saline for 14 days.
Group 2 (100 mg/kg/7days): each rat received 100 mg/kg of TO for 7 days.
Group 3 (100 mg/kg/10days): each rat received 100 mg/kg of TO for 10 days.
Group 4 (100 mg/kg/14days): each rat received 100 mg/kg of TO for 14 days.
Group 5 (200 mg/kg/7days): each rat received 200 mg/kg of TO for 7 days.
Group 6 (200 mg/kg/10days): each rat received 200 mg/kg of TO for 10 days.
Group 7 (200 mg/kg/14days): each rat received 200 mg/kg of TO for 14 days.
The doses and duration of treatment used in this study have been previously reported [14] except for the duration of 10 days. After the last treatment for each group, the rats were fasted overnight for 12 h. On the following day, they were sacrificed under 0.2 ml/kg of ketamine hydrochloride anaesthesia. Blood samples were collected by cardiac puncture into heparinised bottles and centrifuged at 3000 rpm for 10 min. Plasma was then collected into plain bottles, preserved at −20 °C and later used for the determination of glucose, insulin, glucagon, adrenalin and cortisol concentrations. Liver homogenates were made from the liver tissue samples of each rat and used for the estimation of GCK and G6Pase activities.
2.4. Determination of plasma glucose concentration
Plasma glucose was determined spectrophotometrically (Beckman Coulter DTX 880 Multimode Detector) using Fortress assay kit (product code: BXC0102; Fortress Diagnostics Limited, Unit 2C Antrim Technology Park, Antrim, BT41 1QS, United Kingdom) following the manufacturer’s procedure and based on the glucose oxidase method [16].
2.5. Estimation of plasma hormones concentrations
Insulin (product code: IN374S) and cortisol (product code: CO103S) kits were purchased from the Calbiotech Inc., 1935 CordellCt., El Cajon, CA 92020. Glucagon (product code: E1066Ra) and adrenaline (product code: E0545Ra) kits were purchased from the Bioassay Technology Laboratory, 1713 Junjiang International Bldg, 218 Ningguo Rd, Yangpu Dist, Shanghai, China). All assays were done spectrophotometrically (Beckman Coulter DTX 880 Multimode Detector) following manufacturers’ procedures.
2.6. Estimation of liver enzymes activities
The GCK activity assay was based upon the reduction of NAD+ through a coupled reaction with glucose-6-phosphate dehydrogenase (G6PD) and was determined spectrophotometrically (Beckman Coulter DTX 880 Multimode Detector) by measuring the change in absorbance at 340 nm. One unit of activity of GCK reduced 1 μmol of NAD+ per minute at 30 °C and pH 8.0 under the specified conditions.
Glucose-6-phosphatase (G6Pase) activity was estimated using a kit from Elabscience Biotechnology Co. Ltd, Wuhan, Hubei Province, China, which uses Sandwich-ELISA as the method. Briefly, the micro ELISA plate provided in the kit was pre-coated with an antibody specific to G6Pase. Standards or samples were added to the appropriate micro-ELISA plate wells and combined to the specific antibody. Then a biotinylated detection antibody specific for G6Pase and Avidin-Horseradish Peroxidase (Avidin-HRP) conjugate were added to each microplate well and incubated. Free components were washed away. The substrate was added to each well. Only those wells that contain G6Pase, biotinylated detection antibody and Avidin-HRP conjugate appeared blue and were read spectrophotometrically at 450 nm [14].
2.7. Data analysis
Data were analysed using SPSS version 20.0 for Windows (IBM Corporation, Armonk, NY, USA). All values were expressed as the Mean ± S.E.M. of the variables measured. The normality of the data was assessed using the Shapiro-Wilk test, and the confirmation of the data’s normality led to the use of parametric test to assess the significant difference among groups. Significance was assessed by the one-way analysis of variance (ANOVA), followed by a post-hoc Least Significance Difference (LSD) test for multiple comparisons. p-Values < 0.05 were taken as statistically significant.
3. Results
3.1. Effects of TO on plasma glucose concentration
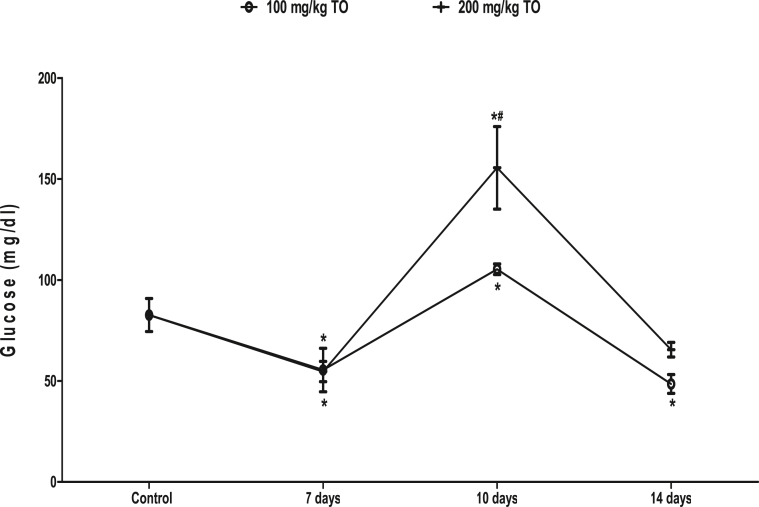
The plasma glucose concentration was reduced in rats that received 100 mg/kg and 200 mg/kg of TO for 7 (55.43 ± 10.79 mg/dl; 54.66 ± 5.05 mg/dl respectively) and 14 days (48.53 ± 4.66 mg/dl; 65.50 ± 3.60 mg/dl respectively) when compared to the control (82.62 ± 8.16 mg/dl). However, the concentration was increased by both 100 mg/kg (105.30 ± 2.63 mg/dl) and 200 mg/kg (155.50 ± 20.43 mg/dl) TO at 10 days when compared to the control. When compared, the increase in plasma glucose caused by 200 mg/kg TO was higher than that caused by 100 mg/kg TO at 10 days (Fig. 1).
Fig. 1.
Effects of Telfairia occidentalis on plasma glucose levels in rats. ∗p < 0.05 vs. control; #p < 0.05 vs. animals treated with 100 mg/kg Telfairia occidentalis (TO) for same duration.
3.2. Effects of TO on plasma insulin concentration
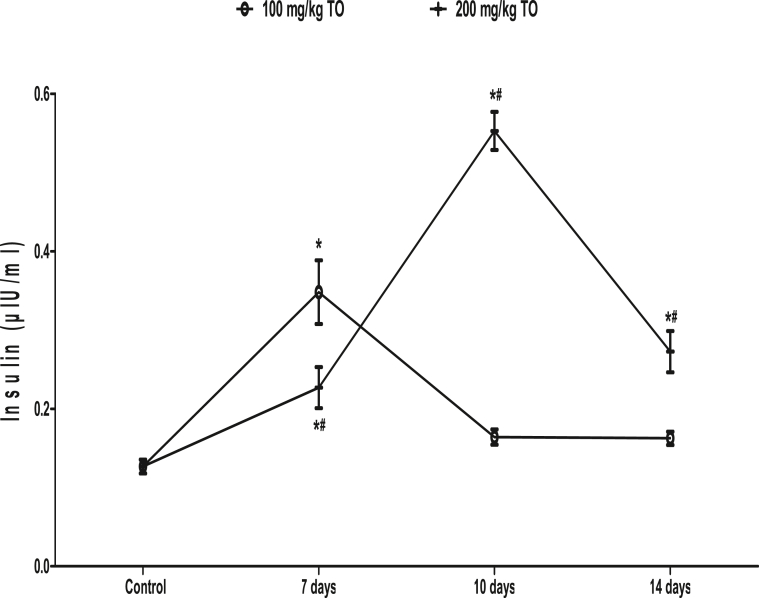
In rats that received 100 mg/kg TO, the insulin concentration increased at 7 days (0.35 ± 0.04 μIU/ml) and returned to values similar to the control (0.13 ± 0.01 μIU/ml) at 10 (0.16 ± 0.01 μIU/ml) and 14 (0.16 ± 0.01 μIU/ml) days. In rats that received the 200 mg/kg TO, the insulin concentration increased at 7 (0.23 ± 0.03 μIU/ml), 10 (0.55 ± 0.02 μIU/ml), and 14 (0.27 ± 0.03 μIU/ml) days when compared to the control (0.13 ± 0.01 μIU/ml). When compared, rats that received 200 mg/kg TO had higher insulin concentration than those that received 100 mg/kg TO at 10 and 14 days, but lower insulin at 7 days (Fig. 2).
Fig. 2.
Effects of Telfairia occidentalis on plasma insulin levels in rats. ∗p < 0.05 vs. control; #p < 0.05 vs. animals treated with 100 mg/kg Telfairia occidentalis (TO) for same duration.
3.3. Effects of TO on plasma glucagon concentration
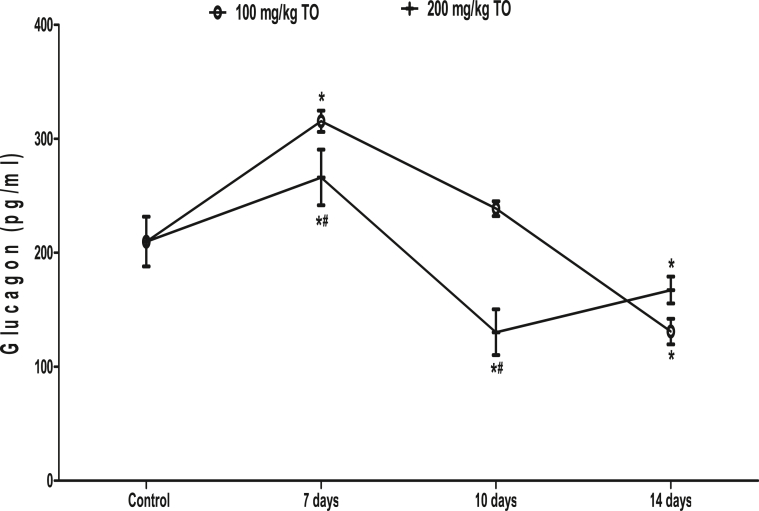
In rats that received 100 mg/kg TO, the glucagon concentration increased at 7 days (315.40 ± 9.30 pg/ml), but returned to a level similar to the control (209.80 ± 21.77 pg/ml) at 10 days (238.80 ± 6.51 pg/ml) and significantly reduced further at 14 days (130.90 ± 11.18 pg/ml). In rats that received 200 mg/kg TO, the glucagon was also increased at 7 days (266.10 ± 24.40 pg/ml) but reduced at 10 (130.30 ± 20.07 pg/ml) and 14 (167.30 ± 11.83 pg/ml) days when compared to the control (209.80 ± 21.77 pg/ml). When compared, rats that received 200 mg/kg TO had lower glucagon concentration than those that received 100 mg/kg TO at 7 and 10 days only (Fig. 3).
Fig. 3.
Effects of Telfairia occidentalis on plasma glucagon levels in rats. ∗p < 0.05 vs. control; #p < 0.05 vs. animals treated with 100 mg/kg Telfairia occidentalis (TO) for same duration.
3.4. Effects of TO on plasma adrenaline concentration
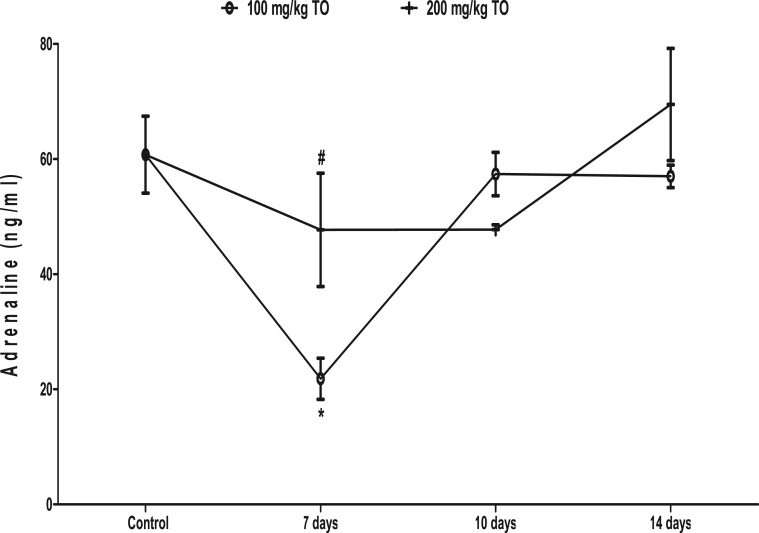
In rats that received 100 mg/kg TO, the adrenaline concentration decreased at 7 days (21.84 ± 3.59 ng/ml), but returned to a level similar to the control (60.77 ± 6.69 ng/ml) at 10 (57.41 ± 3.78 ng/ml) and 14 (57.01 ± 1.95 ng/ml) days. The 200 mg/kg TO did not cause any significant change in the adrenaline concentration during the administration periods when compared to control. When compared, rats that received 200 mg/kg TO had higher adrenaline concentration than those that received 100 mg/kg TO at 7 days only (Fig. 4).
Fig. 4.
Effects of Telfairia occidentalis on plasma adrenaline levels in rats. ∗p < 0.05 vs. control; #p < 0.05 vs. animals treated with 100 mg/kg Telfairia occidentalis (TO) for same duration.
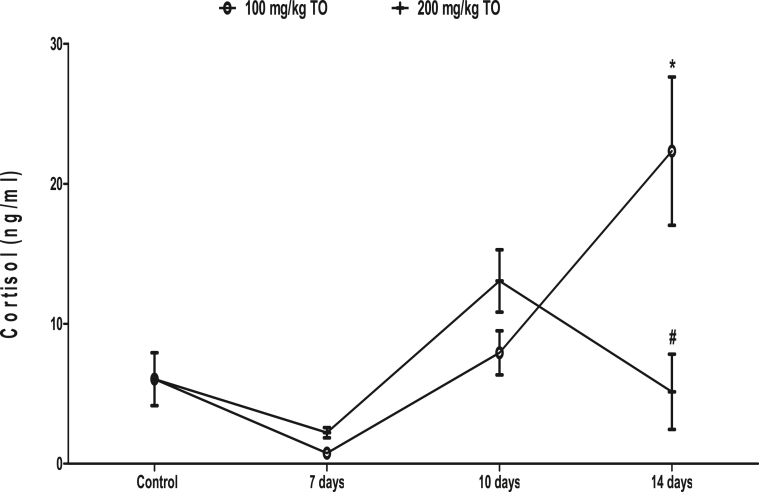
3.5. Effects of TO on plasma cortisol concentration
In rats that received 100 mg/kg TO, the cortisol concentration did not change at 7 (0.76 ± 0.21 ng/ml) and 10 (7.92 ± 1.57 ng/ml) days but increased at 14 days (22.33 ± 5.30 ng/ml) when compared to the control (6.04 ± 1.89 ng/ml). In rats that received 200 mg/kg TO, there was no significant change in the cortisol concentrations during the administration periods when compared to control. When compared, rats that received 200 mg/kg TO had lower cortisol concentration than those that received 100 mg/kg TO at 14 days only (Fig. 5).
Fig. 5.
Effects of Telfairia occidentalis on plasma cortisol levels in rats. ∗p < 0.05 vs. control; #p < 0.05 vs. animals treated with 100 mg/kg Telfairia occidentalis (TO) for same duration.
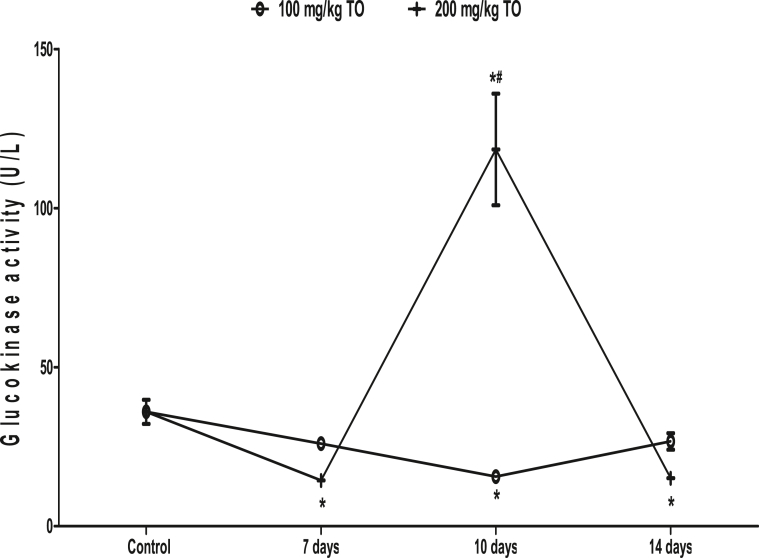
3.6. Effects of TO on liver GCK activity
In rats that received 100 mg/kg TO, the GCK activity was lower at 10 days (15.58 ± 0.76 U/L) but similar when compared to the control (35.96 ± 3.80 U/L) at 7 (25.96 ± 0.83 U/L) and 14 (26.66 ± 2.66 U/L) days. In rats that received 200 mg/kg TO, the GCK activity was higher at 10 days (118.44 ± 17.55 U/L) but lower at 7 (14.37 ± 1.53 U/L) and 14 (15.11 ± 1.32 U/L) days than the control. When compared, rats that received 200 mg/kg TO had higher GCK activity than those that received 100 mg/kg TO at 10 days only (Fig. 6).
Fig. 6.
Effects of Telfairia occidentalis on liver glucokinase activity in rats. ∗p < 0.05 vs. control; #p < 0.05 vs. animals treated with 100 mg/kg Telfairia occidentalis (TO) for same duration.
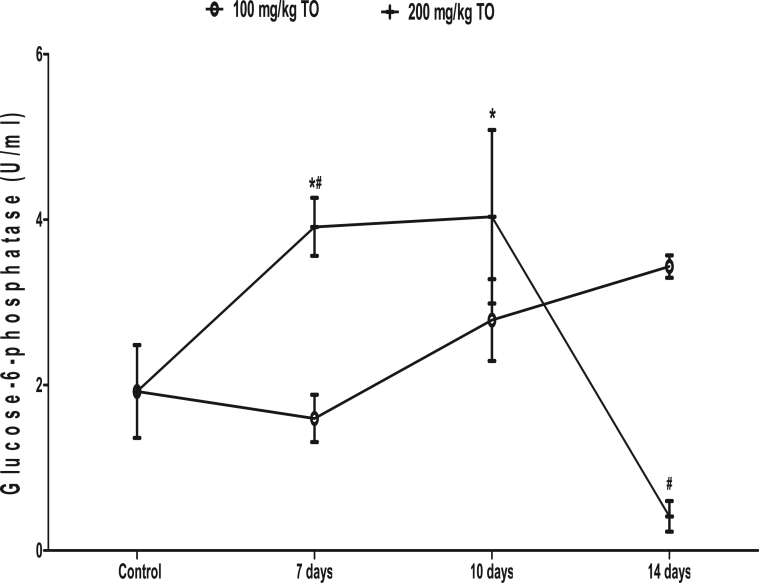
3.7. Effects of TO on G6Pase activity
In rats that received 100 mg/kg TO, there was no significant change in the G6Pase activity during the administration periods when compared to control. In rats that received 200 mg/kg TO, the G6Pase activity increased at 7 (3.91 ± 0.35 U/ml) and 10 (4.04 ± 1.05 U/ml) days, but insignificantly decreased at 14 days (0.41 ± 0.19 U/ml) when compared to control (1.92 ± 0.56 U/ml). When compared, rats that received 200 mg/kg had higher G6Pase activity at 7 days but lower G6Pase actvity at 14 days than those that received 100 mg/kg of TO (Fig. 7).
Fig. 7.
Effects of Telfairia occidentalis on liver glucose-6-phosphatase activity in rats. ∗p < 0.05 vs. control; #p < 0.05 vs. animals treated with 100 mg/kg Telfairia occidentalis (TO) for same duration.
4. Discussion
In the present study, there were significant reductions in the plasma glucose of rats that received both doses of TO for 7 days and 100 mg/kg (but not 200 mg/kg) of TO for 14 days. These findings are consistent with the previously-reported hypoglycaemic effect of TO [7,13,14,[17], [18], [19]]. We also observed surges of insulin and glucagon and simultaneous suppression, albeit insignificant, of 2 key hyperglycaemic hormones (adrenaline and cortisol) after 7-day treatment with both doses of TO. The insulin-glucagon contest observed during the 7-day treatment with both doses of TO led to hypoglycaemia in favour of insulin but not hyperglycaemia in favour of glucagon. The domineering effect of insulin was likely because insulin increased by 180% while glucagon increased by 50% in the 100 mg/kg treatment for 7 days, while the increases were 70% and 30% respectively in the 200 mg/kg treatment for 7 days. This suggests that hypoglycaemia occurred even in the presence of hyperglucagonaemia because of the increases in the insulin/glucagon ratio, which made the insulin effect to dominate over that of glucagon. The suppression of adrenaline and cortisol after 7 days was probably because of the inhibitory and response-delaying effects of insulin on both hormones [[20], [21], [22]].
Our observation of reduced plasma glucose in rats that received 100 mg/kg TO for 14 days in this study contradicts our previous findings [13,14] where we observed an increase in plasma glucose in rats that received the same dose for the same period. This conflict led us to investigate the plasma glucose of rats after 10-day treatment (between 7th – 14th day) with both doses. Interestingly, glucose level increased in these groups (even when compared with the control) with accompanying return of insulin, glucagon, adrenaline and cortisol to levels similar to control in animals that received 100 mg/kg TO. In rats that received 200 mg/kg of TO for 10 days, the increase in blood glucose was associated with an increase in insulin and decrease in glucagon, but the maintenance of adrenaline and cortisol concentrations at levels similar to the control. These suggest that there was an activation of the counter-regulatory response to insulin-induced hypoglycaemia, which accelerates the release of blood-sugar-raising hormones from the adrenal-pituitary system, setting off a running contest between the system and excessive insulin action. Under the recurrent stress of hypoglycaemia, the insulin-opposing factors have been shown to gain ascendency over insulin action and thus produce hyperglycaemia despite hyperinsulinism [23]. Thus, the TO-induced changes in glucose depends mainly on the level of blood glucose itself and the response of the counter-regulatory hormones. Also, these findings suggest that the effect of TO is primarily hypoglycaemic while the post-hypoglycaemic hyperglycaemia seen after prolonged treatment with TO is due to counter-regulation aimed at restoring glucose homeostasis.
What led to the simultaneous insulin-glucagon surge after 7-day treatment with TO? Just like β-cell secretion, pancreatic α-cell secretion is also regulated by glucose and insulin through GCK as a sensor [[24], [25], [26]]. Glucagon suppression as insulin increases appears to relate to insulin sensitivity in normal individuals [27,28] while hyperglucagonaemia in the presence of hyperinsulinaemia and hyperglycaemia is due to insulin resistance/reduced insulin sensitivity in α-cells [29]. Moreover, the increased glucagon inhibits high insulin secretion [30] and ameliorates the expected hyperinsulinaemia-induced hypoglycaemia to maintain glucose homeostasis [31]. Rizza et al. [32] had earlier reported that elevated glucagon concentrations without accompanying appropriate increase in insulin secretion can result in sustained hyperglycaemia. Thus, there is also no doubt that sustained hyperinsulinaemia, when unaccompanied with appropriate increases in glucagon secretion, can result in sustained hypoglycaemia. The hyperglucagonaemia observed on the 7th day of treatment with both doses of TO (simultaneously with insulin-induced hypoglycaemia) was meant to initiate counter-regulatory responses, which is consistent with previous reports that high plasma glucagon opposes insulin secretion and action [30], and increases adrenalin/sympathetic activity [[33], [34], [35], [36]] and cortisol level [37,38]. All these countered the activities of insulin and probably inhibited tissue glucose uptake, stimulated hepatic and kidney gluconeogenesis and glycogenolysis, which could have led to the increased plasma glucose at 10 days and also apparent increases in adrenaline and cortisol later seen on the 10th and 14th days compared to 7th day.
Is the simultaneous insulin-glucagon increase against the β-cell “switch-off” hypothesis”? The hypothesis proposes that sudden cessation of insulin secretion from β-cells into the portal circulation of the islet during hypoglycaemia is a necessary signal for the glucagon response from downstream α-cells (which is the first line of defence against hypoglycaemia) [39]. Banarer et al. [40] hypothesised that defective glucagon secretion during hypoglycaemia in diabetic patients might be due to the lack of a switch-off signal from the β-cell. Contrarily, Bolli et al. [41] rejected the hypothesis by concluding that hypoglycaemia is the primary signal for glucagon secretion independent of insulin levels. Therefore, the present finding in which plasma glucagon rose in response to hypoglycaemia while hyperinsulinaemia persists after 7 days of treatment with both 100- and 200 mg/kg TO agrees with the observation of Bolli et al. [41] and suggests that hypoglycaemia but not β-cell switch-off of insulin secretion is the main factor that stimulated the glucagon response. This view is supported by the recent study of Yu et al. [42] which showed that glucose controls glucagon secretion by directly modulating cAMP in α-cells such that glucose-induced increase in subplasmalemmal cAMP concentration in the α-cells during hypoglycaemia will stimulate glucagon secretion while a decrease in cAMP concentration during hyperglycaemia will inhibit glucagon secretion.
The GCK, present in the liver and pancreatic β- and α–cells, plays a critical role in hepatic glucose metabolism [43,44], especially by regulating insulin and glucagon secretion [24,26]. After being facilitated into the liver cells [45,46], the free glucose is phosphorylated by GCK to form G6P whose production increases as blood glucose increases and then used in glycogenesis, the pentose phosphate pathway, or glycolysis. Since insulin is reportedly the primary up-regulator of the liver GCK [47,48] even independent of blood glucose concentration [47], GCK activity was expected to have been increased (to promote glycogenesis and prevent hyperglycaemia) by the hyperinsulinaemia observed with 7-day treatment with both doses and 14-day treatment with 200 mg/kg TO. In this study, GCK activity did not only decrease when there was hyperinsulinaemia in these groups but also decreased in rats that received 100 mg/kg TO for 10 days that had similar plasma levels of insulin with the control. Moreover, GCK activity was decreased by 100 mg/kg TO but increased by 200 mg/kg TO while insulin and glucagon were respectively increased and decreased by 200 mg/kg at 10 days. These suggest that other factor(s) aside insulin might be responsible for the modulation of GCK activity. Our data showed that the increased plasma glucagon after 7-day treatment with both doses of TO is responsible for the reduction in GCK activity in these groups while the decrease in glucagon in rats that received 200 mg/kg for 10 days also led to an increase in GCK activity in this group. These assertions are supported by a previous report that glucagon is the primary down-regulator of GCK [47].
A reduction in GCK activity normally increases counter-regulatory responses to hypoglycaemia in mice and humans with diabetes [49]. A GCK-dependent glucose-sensing mechanism operates during hypoglycaemia in both humans and mice to facilitate critical protective responses to falling glucose, which leads to hypoglycaemia-associated reduction in insulin secretion and release of the counter-regulatory hormones such as glucagon and epinephrine [49]. Moreover, GCK is responsible for sensing blood glucose not only by β-cells but also by other cells involved in regulating blood glucose. In the case of α-cells, deletion of GCK from the α-cells in mice resulted in substantial loss of glucose-induced suppression of glucagon release [24]. Similarly, GCK within the brain glucose-sensing areas contributes to epinephrine responses to hypoglycaemia [49]. Therefore, the reduction in liver GCK activity both during the decrease and increase in plasma glucose suggests that liver GCK also plays a crucial role in counter-regulation.
A major limitation of this study is the absence of data on α-cell GCK activity, which would have been useful in establishing whether there is any form of cross-talk/synergy between the liver and α-cell GCK during counter-regulation. However, the existence of a feedback loop between the hepatocytes (expressing the glucagon receptor) and the pancreatic α-cells had been proposed [[50], [51], [52], [53], [54], [55]]. Further studies that take into cognisance the expression and activity of both liver and α-cell GCK during counter-regulation will shed light on the possibility of a cross-talk between them.
Liver G6Pase is a central enzyme in liver glucose production as it catalyzes the terminal reaction of liver glucose production, the hydrolysis of G6P generated from glycogenolysis and gluconeogenesis [[56], [57], [58]] into phosphate and free glucose, which is consecutively released into the circulation [[59], [60], [61]]. Following increasing blood glucose, such as after a meal, glucose uptake is stimulated by insulin [62] mainly in the liver and muscle. The free glucose in the liver is rapidly phosphorylated to glucose-6-phosphate (G6P) by GCK and the G6P is subsequently used in glycogenesis, the pentose phosphate pathway, or glycolysis. Thus, the increase in G6Pase activity observed with 200 mg/kg TO for 7 and 10 days in this study might be due to its activation by hypoglycaemia and glucagon earlier observed in 7 days. This is consistent with a previous report that the G6P hydrolytic flux is stimulated by glucagon infusion in-vivo in the rat and isolated hepatocytes and that this activation may account by itself for a concomitant increase in endogenous glucose production (EGP) [63].
Glucose and insulin have been reported to reduce G6Pase gene and mRNA expressions and these reductions dominate the over stimulatory effect of glucocorticoids [[64], [65], [66], [67], [68], [69]]. We noticed a sharp fall (compared with 7 and 10 days) in the G6Pase activity of the group treated with 200 mg/kg TO for 14 days, which we speculate to be due to the combined effects of hyperinsulinaemia and hypoglucagonaemia noticed on 10th and 14th days. Furthermore, the reduction in plasma glucose towards normal after 14-day treatment with 200 mg/kg TO might be associated with the presence of hyperinsulinaemia, hypoglucagonaemia and reduction in G6Pase activity in this group.
The increase in plasma insulin and consequent reduction in blood glucose caused by TO might be connected with the TO’s tannins and saponins; both of which have well-documented hypoglycaemic property [[70], [71], [72], [73], [74]]. In fact, saponins exert an anti-hyperglycaemic function by increasing plasma insulin levels [75], restoring insulin response [76], activating glycogen synthesis [77] and inhibiting α-glucosidase activity [78]. While the present study is limited by non-identification of the specific tannins and saponins present in our TO extract, isolation and investigation of the compounds on their effects and mechanisms on glucose metabolism will be worthwhile in future studies.
5. Conclusion
The present study showed that the hypoglycaemic effect of TO was mediated by insulin and that the post-hypoglycaemic hyperglycaemia results from the counter-regulatory effects of increased glucagon level and G6Pase activity, and reduced GCK activity. Moreover, plasma glucagon response to TO-induced hypoglycaemia is mainly dependent on plasma glucose rather than insulin.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data statement
The data that support the findings of this study are available from the corresponding author, [SOS], upon reasonable request.
CRediT authorship contribution statement
Toyin Mohammed Salman: Conceptualization, Supervision, Data interpretation, Manuscript drafting. Isiaka Abdullateef Alagbonsi: Conceptualization, Formal analysis, Data interpretation, Manuscript drafting. Sheu Oluwadare Sulaiman: Formal analysis, Data interpretation, Manuscript drafting, All authors read and approved the final manuscript to be published.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
We acknowledge Mr A.A. Feyitimi for his technical assistance during the research.
Contributor Information
Toyin Mohammed Salman, Email: drsalman111@yahoo.com.
Isiaka Abdullateef Alagbonsi, Email: easylat@gmail.com.
Sheu Oluwadare Sulaiman, Email: abdulameedsheu@yahoo.com.
References
- 1.Okoli B.E., Mgbeogu C.M. Fluted pumpkin, Telfairia occidentalis: West African vegetable crop. Econ Bot. 1983;37:145–149. doi: 10.2307/4254473. [DOI] [Google Scholar]
- 2.Akoroda M.O. Seed production and breeding potential of the fluted pumpkin, Telfairia occidentalis. Euphytica. 1990;49(1):25–32. doi: 10.1007/BF00024127. [DOI] [Google Scholar]
- 3.Gbile Z.O. Ethnobotany, taxonomy and conservation of medicinal plants. In: Sofowora A.O., editor. The state of medicinal plants research in Nigeria. University of Ibadan Press; 1986. p. 19. [Google Scholar]
- 4.Alade G.O., Okpako E., Ajibesin K.K., Omobuwajo O.R. Indigenous knowledge of herbal medicines among adolescents in amassoma, Bayelsa state, Nigeria. Global J Health Sci. 2015;8(1):217. doi: 10.5539/gjhs.v8n1p217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sofowora A., Ogunbodede E., Onayade A. The role and place of medicinal plants in the strategies for disease prevention. Afr J Tradit, Complementary Altern Med. 2013;10(5):210–229. doi: 10.4314/ajtcam.v10i5.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eseyin O.A., Ebong P., Ekpo A., Igboasoiyi A., Oforah E. Hypoglycemic effect of the seed extract of Telfairia occidentalis in rat. Pakistan J Biol Sci. 2007;10(3):498–501. doi: 10.3923/pjbs.2007.498.501. [DOI] [PubMed] [Google Scholar]
- 7.Eseyin O.A., Ebong P., Eyong E.U., Umoh E., Agboke A. Hypoglycaemic activity of ethyl acetate fraction of the leaf extract of Telfairia occidentalis. Pak J Pharm Sci. 2010;23(3):341–343. http://www.ncbi.nlm.nih.gov/pubmed/20566451 [PubMed] [Google Scholar]
- 8.Jimoh T.O. Enzymes inhibitory and radical scavenging potentials of two selected tropical vegetable ( Moringa oleifera and Telfairia occidentalis ) leaves relevant to type 2 diabetes mellitus. Rev Bras Farmacogn. 2018;28(1):73–79. doi: 10.1016/j.bjp.2017.04.003. [DOI] [Google Scholar]
- 9.Okonkwo C.O., Egesie G.U., Maduka S.O., Oguaka V.N. Hypoglycaemic effect of aqueous extract of Telfairia occidentalis leaf extract in alloxan induced diabetic Wistar rats. Am J Physiol Biochem Pharmacol. 2018;7:42–47. doi: 10.5455/ajpbp.20180430035035. 2 (1) [DOI] [Google Scholar]
- 10.Usar I., Ekwere E., Aguiyi J., Francis O. Synergistic blood sugar lowering effect of the combined leaf extract of vernonia amygdalina (Del), Telfairia occidentalis and ocimum gratissimum in alloxan induced diabetic rats. J Nat Sci Res. 2017;7(12):100–105. [Google Scholar]
- 11.Adisa W.A., Otamere H.O., Osifo C.U., Ediangbe A.P., Ogarah P.A., Aigbogun T.O. Plasma glucose in Telfairia occidentalis treated rats. Adv Appl Sci Res. 2012;3(3):1551–1553. [Google Scholar]
- 12.Chukwunonso E.C.C.E., Udensi A H., Ezean L U.S. Dietary incorporation of boiled fluted pumpkin (Telfairia occidentalis Hook F.) seeds 2: alterations in serum lipid profile and blood glucose concentration of rats. Res J Biol Sci. 2010;5(2):146–149. doi: 10.3923/rjbsci.2010.146.149. [DOI] [Google Scholar]
- 13.Salman T.M., Alagbonsi I., Biliaminu S., Ayandele O., Oladejo O., Adeosun O. Blood glucose-lowering effect of Telfairia Occidentalis: a preliminary study on the underlying mechanism and responses. Biokemistri. 2013;25(3):133–139. https://tspace.library.utoronto.ca/handle/1807/44821 Accessed July 16, 2019. [Google Scholar]
- 14.Salman T.M., Lawal O.R., Sulaiman S.O., Feyitimi A.A., Alada A.R.A. Effects of Telfairia occidentalis leaf extract on plasma lactate and liver glycogen in rats. Niger J Physiol Sci. 2018;33(2):169–175. http://www.ncbi.nlm.nih.gov/pubmed/30837771 Accessed July 16, 2019. [PubMed] [Google Scholar]
- 15.Charan J., Kantharia N. How to calculate sample size in animal studies? J Pharmacol Pharmacother. 2013;4:303–306. doi: 10.4103/0976-500X.119726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Trinder P. Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann Clin Biochem An Int J Biochem Lab Med. 1969;6(1):24–27. doi: 10.1177/000456326900600108. [DOI] [Google Scholar]
- 17.Emudianughe T., Aderibigbe A. The hypoglycemic effect of Telfairia occidentalis aqueous leaf extract in rats. W Afr J Pharmacol Drug Res. 2002;18:14–16. [Google Scholar]
- 18.Nwozo S., Adaramoye O., Ajaiyeoba E. Antidiabetic and hypolipidemic studies of Telfairia occidentalis on Alloxan-induced diabetic rat. Niger J Nat Prod Med. 2004;8:45–47. [Google Scholar]
- 19.Salman T.M., Olayaki L.A., Oyeyemi W.A. Aqueous extract of Telfairia occidentalis leaves reduces blood sugar and increases haematological and reproductive indices in male rats. Afr J Biotechnol. 2008;7(14):2299–2303. doi: 10.5897/AJB08.328. [DOI] [Google Scholar]
- 20.Moan A., Nordby G., Rostrup M., Eide I., Kjeldsen S. Insulin sensitivity, sympathetic activity, and cardiovascular reactivity in young men. Am J Hypertens. 1995;8(3):268–275. doi: 10.1016/0895-7061(94)00206-Q. [DOI] [PubMed] [Google Scholar]
- 21.Høieggen A., Fossum E., Moan A., Eide I., Kjeldsen S. vol. 7. Blood Press; 1998. pp. 291–298. (Insulin modifies the glucose response to mental stress independently of forearm blood flow). 5-6. [DOI] [PubMed] [Google Scholar]
- 22.Thienel M., Wilhelm I., Benedict C., Born J., Hallschmid M. Intranasal insulin decreases circulating cortisol concentrations during early sleep in elderly humans. Neurobiol Aging. 2017;54:170–174. doi: 10.1016/j.neurobiolaging.2017.03.006. [DOI] [PubMed] [Google Scholar]
- 23.Somogyi M. Exacerbation of diabetes by excess insulin action. Am J Med. 1959;26(2):169–191. doi: 10.1016/0002-9343(59)90307-9. [DOI] [PubMed] [Google Scholar]
- 24.Basco D., Zhang Q., Salehi A. α-cell glucokinase suppresses glucose-regulated glucagon secretion. Nat Commun. 2018;9(1):546. doi: 10.1038/s41467-018-03034-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Matschinsky F.M., Wilson D.F. The central role of glucokinase in glucose homeostasis: a perspective 50 Years after demonstrating the presence of the enzyme in islets of langerhans. Front Physiol. 2019;10(148):1–15. doi: 10.3389/fphys.2019.00148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wilson D.F., Cember A.T.J., Matschinsky F.M. The thermodynamic basis of glucose-stimulated insulin release: a model of the core mechanism. Phys Rep. 2017;5(12):e13327. doi: 10.14814/phy2.13327. 1-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Larsson H., Ahren B. Islet dysfunction in insulin resistance involves impaired insulin secretion and increased glucagon secretion in postmenopausal women with impaired glucose tolerance. Diabetes Care. 2000;23(5):650–657. doi: 10.2337/diacare.23.5.650. [DOI] [PubMed] [Google Scholar]
- 28.Ahrén B. Glucagon secretion in relation to insulin sensitivity in healthy subjects. Diabetologia. 2006;49(1):117–122. doi: 10.1007/s00125-005-0056-8. [DOI] [PubMed] [Google Scholar]
- 29.Ahrén B. Glucagon – early breakthroughs and recent discoveries. Peptides. 2015;67:74–81. doi: 10.1016/j.peptides.2015.03.011. [DOI] [PubMed] [Google Scholar]
- 30.Szablewski L. In: Glucose homeostasis and insulin resistance. Szablewski L., editor. Bentham Science Publishers; 2011. [DOI] [Google Scholar]
- 31.Quesada I., Tudurí E., Ripoll C., Nadal Á. Physiology of the pancreatic α-cell and glucagon secretion: role in glucose homeostasis and diabetes. J Endocrinol. 2008;199(1):5–19. doi: 10.1677/JOE-08-0290. [DOI] [PubMed] [Google Scholar]
- 32.Rizza R.A., Cryer P.E., Gerich J.E. Role of glucagon, catecholamines, and growth hormone in human glucose counterregulation. J Clin Invest. 1979;64(1):62–71. doi: 10.1172/JCI109464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Porte D., Williams R.H. Inhibition of insulin release by norepinephrine in man. Science. 1966;152(3726):1248–1250. doi: 10.1126/science.152.3726.1248. 80- [DOI] [PubMed] [Google Scholar]
- 34.Fan W., Dinulescu D.M., Butler A.A., Zhou J., Marks D.L., Cone R.D. The central melanocortin system can directly regulate serum insulin levels∗. Endocrinology. 2000;141(9):3072–3079. doi: 10.1210/endo.141.9.7665. [DOI] [PubMed] [Google Scholar]
- 35.Malaisse W., Malaisse-Lagae F., Wright P.H., Ashmore J. Effects of adrenergic and cholinergic agents upon insulin secretion in vitro. Endocrinology. 1967;80(5):975–978. doi: 10.1210/endo-80-5-975. [DOI] [PubMed] [Google Scholar]
- 36.Imai Y., Patel H.R., Hawkins E.J., Doliba N.M., Matschinsky F.M., Ahima R.S. Insulin secretion is increased in pancreatic islets of neuropeptide Y-deficient mice. Endocrinology. 2007;148(12):5716–5723. doi: 10.1210/en.2007-0404. [DOI] [PubMed] [Google Scholar]
- 37.Livingston J.N., Lockwood D.H. Effect of glucocorticoids on the glucose transport system of isolated fat cells. J Biol Chem. 1975;250(21):8353–8360. http://www.ncbi.nlm.nih.gov/pubmed/1194258 [PubMed] [Google Scholar]
- 38.Smith O.L.K., Wong C.Y., Gelfand R.A. Influence of glucocorticoids on skeletal muscle proteolysis in normal and diabetic-adrenalectomized eviscerated rats. Metabolism. 1990;39(6):641–646. doi: 10.1016/0026-0495(90)90033-9. [DOI] [PubMed] [Google Scholar]
- 39.Zhou H., Tran P.O.T., Yang S. Regulation of -cell function by the -cell during hypoglycemia in Wistar rats: the “switch-off” hypothesis. Diabetes. 2004;53(6):1482–1487. doi: 10.2337/diabetes.53.6.1482. [DOI] [PubMed] [Google Scholar]
- 40.Banarer S., McGregor V.P., Cryer P.E. Intraislet hyperinsulinemia prevents the glucagon response to hypoglycemia despite an intact autonomic response. Diabetes. 2002;51(4):958–965. doi: 10.2337/diabetes.51.4.958. [DOI] [PubMed] [Google Scholar]
- 41.Bolli G., De Feo P., Perriello G. Mechanisms of glucagon secretion during insulin-induced hypoglycemia in man. Role of the beta cell and arterial hyperinsulinemia. J Clin Invest. 1984;73(4):917–922. doi: 10.1172/JCI111315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yu Q., Shuai H., Ahooghalandari P., Gylfe E., Tengholm A. Glucose controls glucagon secretion by directly modulating cAMP in alpha cells. Diabetologia. 2019;62(7):1212–1224. doi: 10.1007/s00125-019-4857-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Salas M., Vinula E., Sols A. Insulin-dependent synthesis of liver glucokinase in the rat. J Biol Chem. 1963;238(11):3535–3538. [PubMed] [Google Scholar]
- 44.Sharma C., Manjeshwar R., Weinhouse S. Effects of diet and insulin on glucose-adenosine triphosphate phosphotransferases of rat liver. J Biol Chem. 1963;238:3840–3845. http://www.ncbi.nlm.nih.gov/pubmed/14086716 [PubMed] [Google Scholar]
- 45.Matschinsky F.M., Kauffman F.C., Ellerman J.E. Effect of hyperglycemia on the hexose monophosphate shunt in islets of Langerhans. Diabetes. 1968;17(8):475–480. doi: 10.2337/diab.17.8.475. [DOI] [PubMed] [Google Scholar]
- 46.Pullen T.J., da Silva Xavier G., Kelsey G., Rutter G.A. miR-29a and miR-29b contribute to pancreatic -Cell-Specific silencing of monocarboxylate transporter 1 (Mct1) Mol Cell Biol. 2011;31(15):3182–3194. doi: 10.1128/MCB.01433-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Iynedjian P.B. Mammalian glucokinase and its gene. Biochem J. 1993;293(1):1–13. doi: 10.1042/bj2930001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Iynedjian P.B., Gjinovci A., Renold A.E. Stimulation by insulin of glucokinase gene transcription in liver of diabetic rats. J Biol Chem. 1988;263:740–744. [PubMed] [Google Scholar]
- 49.Chakera A.J., Hurst P.S., Spyer G. Molecular reductions in glucokinase activity increase counter-regulatory responses to hypoglycemia in mice and humans with diabetes. Mol Metabol. 2018;17:17–27. doi: 10.1016/j.molmet.2018.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Solloway M.J., Madjidi A., Gu C. Glucagon couples hepatic amino acid catabolism to mTOR-dependent regulation of α-cell mass. Cell Rep. 2015;12(3):495–510. doi: 10.1016/j.celrep.2015.06.034. [DOI] [PubMed] [Google Scholar]
- 51.Kim J., Okamoto H., Huang Z. Amino acid transporter Slc38a5 controls glucagon receptor inhibition-induced pancreatic α cell hyperplasia in mice. Cell Metabol. 2017;25(6):1348–1361. doi: 10.1016/j.cmet.2017.05.006. e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dean E.D., Li M., Prasad N. Interrupted glucagon signaling reveals hepatic α cell Axis and role for L-glutamine in α cell proliferation. Cell Metabol. 2017;25(6):1362–1373. doi: 10.1016/j.cmet.2017.05.011. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Galsgaard K.D., Winther-Sørensen M., Ørskov C. Disruption of glucagon receptor signaling causes hyperaminoacidemia exposing a possible liver-alpha-cell axis. Am J Physiol Metabol. 2018;314(1):E93–E103. doi: 10.1152/ajpendo.00198.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wewer Albrechtsen N.J., Færch K., Jensen T.M. Evidence of a liver–alpha cell axis in humans: hepatic insulin resistance attenuates relationship between fasting plasma glucagon and glucagonotropic amino acids. Diabetologia. 2018;61(3):671–680. doi: 10.1007/s00125-017-4535-5. [DOI] [PubMed] [Google Scholar]
- 55.Galsgaard K.D., Winther-Sørensen M., Pedersen J. Glucose and amino acid metabolism in mice depend mutually on glucagon and insulin receptor signaling. Am J Physiol Metabol. 2019;316(4):E660–E673. doi: 10.1152/ajpendo.00410.2018. [DOI] [PubMed] [Google Scholar]
- 56.Mithieux G. New knowledge regarding glucose-6 phosphatase gene and protein and their roles in the regulation of glucose metabolism. Eur J Endocrinol. 1997;136(2):137–145. doi: 10.1530/eje.0.1360137. [DOI] [PubMed] [Google Scholar]
- 57.van de Werve G., Lange A., Newgard C., Méchin M.-C., Li Y., Berteloot A. New lessons in the regulation of glucose metabolism taught by the glucose 6-phosphatase system. Eur J Biochem. 2000;267(6):1533–1549. doi: 10.1046/j.1432-1327.2000.01160.x. [DOI] [PubMed] [Google Scholar]
- 58.Foster J.D., Pederson B.A., Nordlie R.C. Glucose-6-Phosphatase structure, regulation, and function: an update. Exp Biol Med. 1997;215(4):314–332. doi: 10.3181/00379727-215-44142. [DOI] [PubMed] [Google Scholar]
- 59.Barthel A., Schmoll D., Unterman T.G. FoxO proteins in insulin action and metabolism. Trends Endocrinol Metabol. 2005;16(4):183–189. doi: 10.1016/j.tem.2005.03.010. [DOI] [PubMed] [Google Scholar]
- 60.Nordlie R.C., Foster J.D., Lange A.J. Regulation of glucose production by the liver. Annu Rev Nutr. 1999;19(1):379–406. doi: 10.1146/annurev.nutr.19.1.379. [DOI] [PubMed] [Google Scholar]
- 61.Schmoll D., Walker K., Alessi D. Regulation of glucose-6-phosphatase gene expression by protein kinase Balpha and the forkhead transcription factor FKHR. Evidence for insulin response unit-dependent and -independent effects of insulin on promoter activity. J Biol Chem. 2000;275(46):36324–36333. doi: 10.1074/jbc.M003616200. [DOI] [PubMed] [Google Scholar]
- 62.Bogan J.S. Regulation of glucose transporter translocation in Health and diabetes. Annu Rev Biochem. 2012;81(1):507–532. doi: 10.1146/annurev-biochem-060109-094246. [DOI] [PubMed] [Google Scholar]
- 63.Ichai C., Guignot L., El-Mir M.Y. Glucose 6-phosphate hydrolysis is activated by glucagon in a low temperature-sensitive manner. J Biol Chem. 2001;276(30):28126–28133. doi: 10.1074/jbc.M010186200. [DOI] [PubMed] [Google Scholar]
- 64.Lange A.J., Argaud D., Elmaghrabi M.R., Pan W.S., Maitra S.R., Pilkis S.J. Isolation of a cDNA for the catalytic subunit of rat liver glucose-6-phosphatase: regulation of gene expression in FAO hepatoma cells by insulin, dexamethasone and cAMP. Biochem Biophys Res Commun. 1994;201(1):302–309. doi: 10.1006/bbrc.1994.1702. [DOI] [PubMed] [Google Scholar]
- 65.Schmoll D., Allan B.B., Burchell A. Cloning and sequencing of the 5′ region of the human glucose-6-phosphatase gene: transcriptional regulation by cAMP, insulin and glucocorticoids in H4IIE hepatoma cells. FEBS Lett. 1996;383(1-2):63–66. doi: 10.1016/0014-5793(96)00224-4. [DOI] [PubMed] [Google Scholar]
- 66.Pagliassotti M.J., Wei Y., Bizeau M.E. Glucose-6-Phosphatase activity is not suppressed but the mRNA level is increased by a sucrose-enriched meal in rats. J Nutr. 2003;133(1):32–37. doi: 10.1093/jn/133.1.32. [DOI] [PubMed] [Google Scholar]
- 67.Guignot L., Mithieux G. Mechanisms by which insulin, associated or not with glucose, may inhibit hepatic glucose production in the rat. Am J Physiol Metabol. 1999;277(6):E984–E989. doi: 10.1152/ajpendo.1999.277.6.E984. [DOI] [PubMed] [Google Scholar]
- 68.Minassian C., Tarpin S., Mithieux G. Role of glucose-6 phosphatase, glucokinase, and glucose-6 phosphate in liver insulin resistance and its correction by metformin. Biochem Pharmacol. 1998;55(8):1213–1219. doi: 10.1016/S0006-2952(97)00576-5. [DOI] [PubMed] [Google Scholar]
- 69.Mithieux G., Daniele N., Payrastre B., Zitoun C. Liver microsomal glucose-6-phosphatase is competitively inhibited by the lipid products of phosphatidylinositol 3-kinase. J Biol Chem. 1998;273(1):17–19. doi: 10.1074/jbc.273.1.17. [DOI] [PubMed] [Google Scholar]
- 70.Liu X., Kim J., Li Y., Li J., Liu F., Chen X. Tannic acid stimulates glucose transport and inhibits adipocyte differentiation in 3T3-L1 cells. J Nutr. 2005;135(2):165–171. doi: 10.1093/jn/135.2.165. [DOI] [PubMed] [Google Scholar]
- 71.Morada N.J., Metillo E.B., Uy M.M., Oclarit J.M. Toxicity and hypoglycemic effect of tannin-containing extract from the mangrove tree Sonneratia alba Sm. Bull Env Pharmacol Life Sci. 2016;5(6):58–64. [Google Scholar]
- 72.Nkiru U. Antifungal effect and phytochemical screening of Telfairia occidentalis (hook f.) Leaf extracts. J Plant Biotechnol Microbiol. 2018;1(1):1–4. [Google Scholar]
- 73.Barky A El, Hussein S.A. Saponins-and-Their-Potential-Role-in-Diabetes-Mellitus. Diabetes Manag. 2017;7(1):148–158. [Google Scholar]
- 74.Xu J., Wang S., Feng T., Chen Y., Yang G. Hypoglycemic and hypolipidemic effects of total saponins from Stauntonia chinensis in diabetic db/db mice. J Cell Mol Med. 2018;22(12):6026–6038. doi: 10.1111/jcmm.13876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Metwally N. Chemical constituents of the Egyptian Plant Anabasis articulata (Forssk) Moq and its antidiabetic effects on rats with streptozotocin-induced diabetic hepatopathy. J Appl Pharmaceut Sci. 2012;2:54–65. doi: 10.7324/JAPS.2012.2403. [DOI] [Google Scholar]
- 76.Zheng T., Shu G., Yang Z., Mo S., Zhao Y., Mei Z. Antidiabetic effect of total saponins from Entada phaseoloides (L.) Merr. in type 2 diabetic rats. J Ethnopharmacol. 2012;139(3):814–821. doi: 10.1016/j.jep.2011.12.025. [DOI] [PubMed] [Google Scholar]
- 77.Xiong W.-T., Gu L., Wang C., Sun H.-X., Liu X. Anti-hyperglycemic and hypolipidemic effects of Cistanche tubulosa in type 2 diabetic db/db mice. J Ethnopharmacol. 2013;150(3):935–945. doi: 10.1016/j.jep.2013.09.027. [DOI] [PubMed] [Google Scholar]
- 78.Hemalatha T., Pulavendran S., Balachandran C., Manohar B.M., Puvanakrishnan R. Arjunolic acid: a novel phytomedicine with multifunctional therapeutic applications. Indian J Exp Biol. 2010;48(3):238–247. http://www.ncbi.nlm.nih.gov/pubmed/21046976 [PubMed] [Google Scholar]