Abstract
Background
Antegrade cerebral perfusion (ACP), including unilateral and bilateral, is most commonly used for cerebral protection in aortic surgery. There is still no consensus on the superiority of the two methods. Our research aimed to investigate the clinical effects of u-ACP and b-ACP.
Methods
321 of 356 patients with type A aortic dissection were studied retrospectively. 124 patients (38.6%) received u-ACP, and 197 patients (61.4%) received b-ACP. We compared the incidence of postoperative neurological complications and other collected data between two groups. Besides, we also analyzed perioperative variables to find the potential associated factors for neurological dysfunction (ND).
Results
For u-ACP group, 54 patients (43.5%) had postoperative neurological complications, including 22 patients (17.7%) with permanent neurologic dysfunction (PND) and 32 patients (25.8%) with temporary neurologic dysfunction (TND). For b-ACP group, 47 patients (23.8%) experienced postoperative neurological complications, including 16 patients (8.1%) of PND and 31 patients (15.7%) of TND. The incidence of PND and TND were significantly different between two groups along with shorter CPB time (p = 0.016), higher nasopharyngeal temperature (p≦0.000), shorter ventilation time (p = 0.018), and lower incidence of hypoxia (p = 0.022). Furthermore, multivariate stepwise logistic regression analysis confirmed that preoperative neurological dysfunction (OR = 1.20, p = 0.028), CPB duration (OR = 3.21, p = 0.002), and type of cerebral perfusion (OR = 1.48, p = 0.017) were strongly associated with postoperative ND.
Conclusions
In our study, it was observed that b-ACP procedure exhibited shorter CPB time, milder hypothermia, shorter ventilation time, lower incidence of postoperative hypoxia, and neurological dysfunction compared to u-ACP. Meanwhile, the incidence of ND was independently associated with three factors: preoperative neurological dysfunction, CPB time, and type of cerebral perfusion.
Keywords: Cerebral protection, Unilateral antegrade cerebral perfusion, Bilateral antegrade cerebral perfusion, Type A aortic dissection, Neurological complications
Background
Aortic dissection (AD) is one of the most serious cardiac emergencies owing to associated high mortality rates, especially Stanford type A [1, 2]. Presently, surgery is the primary treatment method. The frozen elephant trunk technique is increasingly being used to repair dissections extending over the entire aortic arch. Despite immense improvements in surgical techniques, the operative mortality and complications of type A aortic dissection (AD) remain considerably high [3]. Neurological dysfunction (ND) is a common complication, with a reported case rate ranging from 5.5 to 33.3% [4]. Consequently, it is crucial to implement appropriate measures to prevent cerebral injury.
In recent decades, various kinds of protocols to avoid cerebral damage have been utilized. Advances in these protocols have subsequently improved neurological outcomes for type A AD. Currently, antegrade cerebral perfusion (ACP) has become the standard method of cerebral support. However, there is still a controversy regarding the superiority of either the unilateral ACP (u-ACP) or bilateral ACP (b-ACP) [5, 6].
Generally, the feasibility of u-ACP for keeping the brain perfused during the circulatory arrest period is based on the integrity of the circle of Willis. Nevertheless, there are several anatomical variations of the circle. Particularly, b-ACP is an approach of providing cerebral perfusion through both sides simultaneously to mimic the physiological conditions. Therefore, we postulated that b-ACP may be more advantageous for cerebral support or other aspects related to mortality, neurological outcome, and other systemic complications, compared to u-ACP.
We also retrospectively gathered potential variables according to the Society of Thoracic Surgeons (STS) National Database, aiming at identifying risk factors for postoperative neurological dysfunction in patients with type A AD undergoing total aortic arch replacement.
Methods
Study populations
We retroactively studied 356 patients with type A AD who were admitted to our cardiac surgical intensive care unit after total aortic arch replacement from January 1, 2014 to December 31, 2018. 321 patients undergoing total arch replacement with one of the following manifestations shown on CTA: arch tear, carotid dissection or occlusion, or the presence of an aortic arch aneurysm were included into our research, of whom 124 underwent a u-ACP (38.6%) and 197 underwent a b-ACP (61.4%). The experimental protocol and informed consent were approved by the Institutional Review Board of our hospital, and all subjects gave informed consent. Among the 321 patients, there were 224 males and 97 females. The mean age was 51.98 ± 9.78 years. All the cases were diagnosed using preoperative computed tomography angiography (CTA) and therapy was conducted within 2 weeks of onset. Preoperative echocardiography was also necessary to evaluate the cardiac function, the exact position of intima rupture, and to assess the condition of the aortic and mitral valves. Every patient underwent total arch replacements. According to the surgery time from onset, we divided patients into three groups: hyper-acute (< 24 h), acute (24–72 h) and subacute (> 72 h). The concomitant procedures included Bentall procedure, coronary artery bypass grafting (CABG) and Bentall + CABG. The relevant demographic data and surgical strategies were illustrated in Table 1. Preoperative neurological complications were defined as acute neurological dysfunctions from dissection, which included one patient with coma, three with somnolence, one with hemiplegia, and four with monoplegia.
Table 1.
Demographic and preoperative characteristics
| Variable | u-ACP (n = 124) | b-ACP (n = 197) | p value |
|---|---|---|---|
| Age (year, ± s) | 55.38 ± 10.40 | 54.00 ± 9.37 | 0.646 |
| Male (n, %) | 85 (68.5%) | 139 (70.6%) | 0.822 |
| BMI (kg/m2, ± s) | 24.58 ± 1.25 | 25.21 ± 1.44 | 0.148 |
| Hypertension (n, %) | 94 (76.3%) | 157 (80.1%) | 0.615 |
| Diabetes mellitus (n, %) | 16 (12.9%) | 43 (21.8%) | 0.433 |
| COPD (n, %) | 3 (2.4%) | 7 (3.6%) | 0.746 |
| Cerebrovascular disease history (n, %) | 23 (18.5%) | 37 (18.8%) | 0.958 |
| Renal dysfunction (n, %)a | 5 (4.0%) | 12 (6.1%) | 0.423 |
| Hemodynamic instability | 4 (3.2%) | 9 (4.6%) | 0.552 |
| Results of CT scan | |||
| Arch tear | 105 | 152 | 0.361 |
| Carotid dissection | 6 | 8 | |
| Aortic arch aneurysm | 13 | 37 | |
| Preoperative neurological complication (n, %) | 5 (4.0%) | 11 (5.6%) | 0.534 |
| Preoperative intubation (n, %) | 6 (4.8%) | 10 (5.1%) | 0.666 |
| LVEF < 0.4 (n, %) | 4 (3.2%) | 10 (5.1%) | 0.578 |
p < 0.05 is considered as statistically significant
u-ACP unilateral antegrade cerebral perfusion, b-ACP bilateral antegrade cerebral perfusion, COPD chronic obstructive pulmonary disease
aA serum creatinine level > 97 μmol/L is considered to indicate renal dysfunction
The anesthetic regimen was kept standard. Intravenous (IV) injections of 2.5–5 mg midazolam, 0.2–0.6 mg/kg etomidate, 0.1–5 μg/kg sufentanil, and 0.6 mg/kg rocuronium were performed as the standard anesthetic induction in these patients. Subsequently, tracheal intubations were executed. Anesthesia was maintained by continuous inhalation of sevoflurane, IV injections of dexmedetomidine and rocuronium, combined with theadditional administration of midazolam and sufentanil when necessary.
Operative technique
All operations were conducted by the same surgical staff. Our operative techniques are composed of cardiopulmonary bypass (CPB), moderate hypothermia (24–28 ℃), circulatory arrest, and ACP. Monitoring data included left radial and dorsalis pedis arterial pressures, main arterial pressure (MAP), central venous pressure (CVP), electrocardiography (ECG), blood oxygen saturation (SaO2), and arterial blood gas (ABG) analysis. Cerebral saturation was monitored with near-infrared spectroscope (NIRS). Prior to December 2015, u-ACP with moderate hypothermia circulatory arrest (24–26 ℃) was routinely performed to maintain cerebral perfusion. From January 2016, b-ACP was initiated until the end of the study. The temperature of circulatory arrest was gradually increased to 26–28 ℃. In both groups, right axillary artery with right femoral artery cannulations were conducted to establish CPB. Besides, cannulation of the right axillary artery was used for cerebral perfusion in the u-ACP group. However, for patients with dissection extending from the innominate artery and into right subclavian, we chose to cut off the right common carotid artery and cannulated directly into the true lumen where dissection was relatively less involved. In the b-ACP group, right axillary artery cannulation was employed along with a 12-F or 14-F balloon-tip catheter in the left common carotid artery.
After CPB was established, we cross-clamped the ascending aorta after the nasopharyngeal temperature dropped to 34 ℃ or lower. Consequently, cold blood cardioplegia was injected into the coronary Ostia antegrade or the coronary sinus retrograde directly to stop the heart. Subsequently, we made a longitudinal incision on the ascending aorta and performed the aortic root procedure depending on the severity and extent of the disease, including aortic root formation or Bentall procedure with or without CABG.
When the temperature reached 24–28 ℃, all three branches of the aortic arch were separately clamped, and the systemic circulation stopped. In the u-ACP group, cerebral perfusion was provided only by the right axillary artery cannulation. Moreover, 12 of 124 patients, who immediately switched to b-ACP, were classified as b-ACP group patients. In the b-ACP group, both the right axillary artery and left common carotid artery were used for cerebral protection. Flow rates of 5–10 ml/kg/min were used for ACP, with a perfusion pressure range of 50–80 mmHg. A stented elephant trunk was inserted into the proximal descending aorta and attached to the distal end of the graft. Subsequently, the systemic circulation of the lower body was restarted through the right femoral artery. For the u-ACP group, anastomosis of the left common carotid artery was first performed to restore left cerebral perfusion. Afterwards, the proximal aortic root was anastomosed to the prosthetic graft to restore systemic circulation, and the temperature increased progressively. The left subclavian artery and innominate artery were sequentially anastomosed to the prosthetic graft. For the b-ACP group, the left subclavian artery was initially anastomosed to the prosthetic graft, followed by the left common carotid artery, proximal aortic root, and innominate artery.
Statistical analysis
All of the perioperative data were analyzed distinctively between two groups (see Tables 2, 3). The postoperative variables consisted of 30-day mortality rate, ventilation time (h), permanent neurological dysfunction (PND), temporary neurological dysfunction (TND), acute kidney injury (AKI, referring to elevated serum creatinine concentration > 1.5 times of baseline or urine volume < 0.5 ml/kg/h for 6 h within 48 h after surgery), hypoxia (PaO2/FiO2 < 200 with PEEP ≥ 5cm H2O within 72 h after surgery), and postoperative bleeding volume within 24 h of surgery. Patients with neurological symptoms had to be examined by CT scans to confirm the diagnosis. PND was defined as the presence of permanent neurological deficits persisting after being discharged with focal or global cerebral lesions confirmed by CT or MRI, including monoplegia, hemiplegia, paraplegia and coma, confirmed by CT or MRI. TND included transient ischemic attack (TIA) and reversible neurological deficits such as delirium, confusion and agitation with no new lesions on CT. All the diagnoses of stroke were adjudicated by a neurologist who was blinded.
Table 2.
Intraoperative data
| Variable | u-ACP (n = 124) | b-ACP (n = 197) | p |
|---|---|---|---|
| CPB duration (min, ± s) | 260.07 ± 76.79 | 235.79 ± 46.60 | 0.016 |
| Aortic cross-clamping duration (min, ± s) | 154.53 ± 36.50 | 154.94 ± 33.75 | 0.946 |
| Circulatory arrest time (min, ± s) | 26.60 ± 6.78 | 25.62 ± 6.31 | 0.384 |
| Nasopharyngeal temperature (℃, ± s) | 24.92 ± 0.28 | 27.09 ± 1.22 | 0.000 |
| Concomitant procedures (n, %) | |||
| Bentall | 25 (20.2%) | 47 (23.9%) | 0.703 |
| CABG | 9 (7.3%) | 18 (9.1%) | |
| Bentall + CABG | 7 (5.6%) | 13 (6.6%) | |
| Surgery time (n, %) | |||
| Hyper-acute (< 24 h) | 24 (19.4%) | 35 (17.8%) | 0.914 |
| Acute (24-72 h) | 82 (66.1%) | 131 (66.5%) | |
| Subacute (> 72 h) | 18 (14.5%) | 31 (15.7%) | |
p < 0.05 is considered as statistically significant
u-ACP unilateral antegrade cerebral perfusion, b-ACP bilateral antegrade cerebral perfusion, CPB cardiopulmonary bypass, CABG coronary artery bypass grafting
Table 3.
Postoperative data
| Variable | u-ACP (n = 124) | b-ACP (n = 197) | p |
|---|---|---|---|
| Drainage volume within 24 h > 1 L (n, %) | 22 (17.7%) | 27 (13.7%) | 0.328 |
| Ventilation time (h, ± s) | 62.26 ± 46.42 | 44.03 ± 28.74 | 0.018 |
| Hpoxia (n, %) | 34 (27.4%) | 33 (16.8%) | 0.022 |
| AKI (n, %) | 18 (14.5%) | 22 (11.1%) | 0.376 |
| PND (n, %) | 22 (17.7%) | 16 (8.1%) | 0.009 |
| TND (n, %) | 32 (25.8%) | 31 (15.7%) | 0.027 |
| ICU stay (days, ± s) | 18.73 ± 5.67 | 17.10 ± 5.18 | 0.447 |
| 30-day mortality (n, %) | 12 (9.7%) | 10 (5.1%) | 0.112 |
p < 0.05 is considered as statistically significant
u-ACP unilateral antegrade cerebral perfusion, b-ACP bilateral antegrade cerebral perfusion, AKI acute kidney injury, PND permanent neurologic dysfunction, TND temporary neurologic dysfunction
Continuous variables were expressed as mean ± SD; categorical data were expressed as proportions. The T test or Mann–Whitney test were used to compare continuous variables while categorical data were compared using the χ2 test or the Fisher exact test. We divided the patients into two groups (ND vs non-ND). All potential risk variables were analyzed for significance. For factors with p values < 0.05, the multivariable logistic regression model was employed to further identify independent risk factors.
Results
Intraoperative data
There were no significant differences regarding rates of concomitant surgery, including the Bentall procedure, CABG, and Bentall + CABG between the two groups (Table 2). However, we observed that the CPB durations and nasopharyngeal temperatures during the circulatory arrest period were considerably different.
Mortality rates and morbidities
After meticulous comparisons of the postoperative data, we did not discover differences in the 30-day mortality rates, incidence of postoperative renal failure, or bleeding volume > 1 L within 24 h of surgery. Compared to the u-ACP group, the patients in b-ACP group exhibited shorter ventilation times (44.03 ± 28.74 vs. 62.26 ± 46.42; p = 0.018) and overall lower incidence of hypoxia (16.4% vs. 27.9%; p = 0.037) (see Table 3).
Neurological events
We compared the postoperative neurological complications between the two groups (see Fig. 1). For the u-ACP group, PND was observed in 22 patients (17.7%): paraplegia (n = 2), monoplegia (n = 4), hemiplegia (n = 11), and coma (n = 5). TND was observed in 32 patients (25.8%): delirium (n = 23), TIA (n = 2), and confusion and agitation (n = 7). Meanwhile, a total of 16 patients (8.1%) with PND were observed in the b-ACP group: paraplegia (n = 1), hemiplegia (n = 9), monoplegia (n = 4), and coma (n = 2); whilst 31 patients (15.7%) with TND were observed: delirium (n = 25), confusion and agitation (n = 6), and no TIA. The incidence of PND and TND were undoubtedly different between the two groups (see Table 4).
Fig. 1.
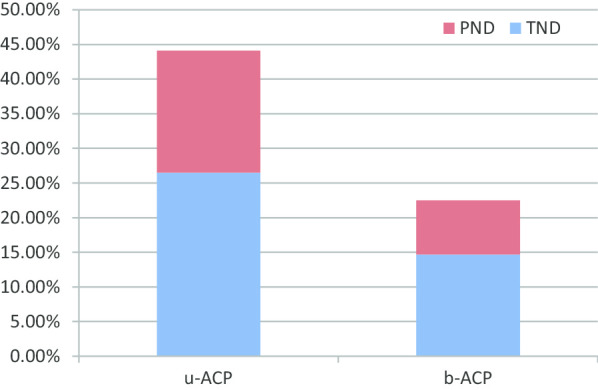
Postoperative neurological dysfunction in the u-ACP and b-ACP groups
Table 4.
Description of ND between u-ACP and b-ACP groups
| ND | u-ACP (n = 124) | b-ACP (n = 197) |
|---|---|---|
| PND | 22 (17.7%) | 16 (8.1%) |
| Paraplegia | 2 (1.6%) | 1 (0.5%) |
| Monoplegia | 4 (3.2%) | 4 (2.0%) |
| Hemiplegia | 11 (8.9%) | 9 (4.6%) |
| Coma | 5 (4.0%) | 2 (1.0%) |
| TND | 32 (25.8%) | 31 (15.7%) |
| Delirium | 23 (18.5%) | 25 (12.7%) |
| TIA | 2 (1.6%) | 0 (0%) |
| Confusion and agitation | 7 (5.6%) | 6 (3.0%) |
u-ACP unilateral antegrade cerebral perfusion, b-ACP bilateral antegrade cerebral perfusion, PND permanent neurologic dysfunction, TND temporary neurologic dysfunction
Furthermore, we divided the patients into different groups based on the incidence of postoperative neurological dysfunction: ND group and non-ND group. The results of univariate analyses of all the potential risk variables were listed in Table 5. All variables with p values < 0.05 were included in the multivariable analysis model. Our study demonstrated that preoperative neurological dysfunction (OR = 1.20, p = 0.028), CPB duration (OR = 3.21, p = 0.002) and type of cerebral perfusion (OR = 1.48, p = 0.017) were independent risk factors for genesis of ND (Table 6).
Table 5.
Univariate analysis of perioperative risk factors of postoperative ND in patients with type A aortic dissection
| Variable | ND (n = 101) | Non-ND (n = 220) | p |
|---|---|---|---|
| Preoperative variables | |||
| Age (year, ± s) | 56.70 ± 9.91 | 53.83 ± 9.75 | 0.422 |
| Male (n, %) | 73 (73.7%) | 155 (69.8%) | 0.475 |
| Education level (year, ± s) | 10.61 ± 5.30 | 11.23 ± 4.85 | 0.651 |
| Tobacco use (n, %) | 52 (52.5%) | 108 (48.6%) | 0.521 |
| Alcohol use (n, %) | 20 (20.2%) | 37 (16.7%) | 0.444 |
| Barrier of hearing or language (n, %) | 3 (3.0%) | 7 (3.2%) | 1.000 |
| Hypertension (n, %) | 87 (87.9%) | 168 (75.7%) | 0.012 |
| Diabetes mellitus (n, %) | 23 (23.2%) | 36 (16.2%) | 0.134 |
| COPD (n, %) | 4 (4.0%) | 6 (2.7%) | 0.505 |
| Cerebrovascular disease history (n, %) | 26 (26.3%) | 34 (15.3%) | 0.020 |
| Peripheral arterial disease (n, %) | 7 (7.1%) | 11 (5.0%) | 0.447 |
| Renal dysfunction (n, %) | 9 (9.1%) | 8 (3.6%) | 0.043 |
| LVEF < 0.4 (n, %) | 5 (5.1%) | 9 (4.1%) | 0.684 |
| Preoperative neurological dysfunction (n, %) | 10 (9.9%) | 6 (2.7%) | 0.005 |
| Preoperative intubation (n, %) | 9 (8.9%) | 7 (3.2%) | 0.024 |
| Preoperative cardiac arrhythmia (n, %) | 8 (7.9%) | 11 (5%) | 0.273 |
| Intraoperative variables | |||
| Concomitant procedures (n, %) | |||
| Bentall | 29 (28.7%) | 43 (19.5%) | 0.358 |
| CABG | 15 (14.9%) | 12 (5.5%) | |
| Bentall + CABG | 10 (9.9%) | 10 (4.5%) | |
| CPB duration (min, ± s) | 259.93 ± 69.94 | 234.48 ± 49.24 | 0.009 |
| Aortic cross-clamping duration (min, ± s) | 157.96 ± 37.17 | 153.25 ± 33.11 | 0.418 |
| Circulatory arrest time (min, ± s) | 27.06 ± 6.05 | 25.33 ± 6.59 | 0.112 |
| Type of cerebral perfusion (u-ACP, %) | 50 (49.5%) | 74 (33.6%) | 0.007 |
| Nasopharyngeal temperature (℃, ± s) | 26.11 ± 1.49 | 26.60 ± 1.38 | 0.039 |
| Blood transfusion > 1 L (n, %) | 25 (24.8%) | 34 (15.5%) | 0.046 |
| Postoperative variables | |||
| Postoperative drainage volume within 24 h (> 1 L) (n, %) | 15 (14.9%) | 16 (7.3%) | 0.033 |
| AKI (n, %) | 16 (15.8%) | 25 (11.4%) | 0.264 |
| Hemoglobin (g/L, ± s) | 93.25 ± 10.39 | 100.50 ± 9.35 | 0.180 |
| Cardiac arrhythmia (n, %) | 21 (50.0%) | 36 (19.7%) | 0.335 |
p < 0.05 is considered as statistically significant
Table 6.
Multivariate stepwise logistic regression analysis for predictors of postoperative ND in patients with type A aortic dissection
| Variable | Regression coefficient | OR (95% CI) | p |
|---|---|---|---|
| Preoperative neurological dysfunctiona | 0.391 | 1.20 (1.07–1.36) | 0.028 |
| CPB durationb | 1.501 | 3.21 (1.43–5.72) | 0.002 |
| Type of cerebral perfusionb | 0.660 | 1.48 (1.07–2.04) | 0.017 |
aPreoperative variables
bIntraoperative variables
Discussion
We compared the two cerebral perfusion methods concerning a set of clinical outcomes. We discovered that patients in the b-ACP group had shorter CPB times, higher permissive arrest temperatures, shorter ventilation times, and lower incidences of postoperative hypoxia and ND. ND is a serious postoperative complication of acute type A AD [7], with an incidence range of 0–32.8% [8]. Nevertheless, its major risk factors remain uncertain, despite the relatively frequent occurrence of ND in type A AD patients. Therefore, we analyzed various potential factors retrospectively and revealed that the preoperative neurological function, CPB time, and type of cerebral perfusion chosen were independent risk factors for postoperative neurological dysfunction. Consequently, we may reduce the occurrence of postoperative ND by shortening the CPB time and choosing the most efficient cerebral perfusion method. Besides, the relationship between postoperative neurological complications and the intraoperative ACP method used is further discussed in this report.
Hypothermic circulatory arrest combined with ACP has been recognized as the first choice for cerebral protection in aortic arch surgery and was used worldwide [9, 10]. This technique yielded the best results and mostly improved short-term or long-term outcomes. Nevertheless, the use of u-ACP vs b-ACP remains a subject of debate. We routinely used right axillary artery cannulation for u-ACP in type A AD operations. Its main advantage is that it involves less risk of dissection or atherosclerosis in the right axillary artery. Besides, the isolation and cannulation procedure is much easier in this way. Nowadays, along with the tremendous improvements in operative technology, intubation methods, and clearer determination of anatomical locations, a substantial body of evidence suggests the exact effect of b-ACP with advantages of shorter CPB time, permissive milder hypothermia, lower incidence of hypoxia and ND [11, 12]. The possible reasons can be summarized as follows.
We previously explained the surgical procedures with u-ACP or b-ACP in detail. The anastomosis sequence of the three branches of the aortic arch in the two groups is different. For the u-ACP group, the left common carotid artery was initially anastomosed to restore the physiological status of the left cerebral perfusion, followed by the anastomosis of the left subclavian artery. The initial anastomosis of the left common carotid artery can hinder the subsequent operation on the left subclavian artery by reducing the surgical field, making the procedure more challenging and time-consuming. Consequently, the incidence of anastomotic bleeding of the left subclavian artery rose, and our surgeons needed more time and attention to stop the bleeding. The mean CPB time of the u-ACP group was longer than that of the b-ACP group. Although the sequence of anastomosis of supra-arotic branches mostly depended on surgeons' personal choice, we preferred to first anastomose left common carotid artery for patients in the u-ACP group since it provided 2/3 blood-supply of left hemicerebrum. Besides, the sequence of anastomosis of branches mainly depended on the importance of cerebral perfusion. For patients in the b-ACP group, the bilateral cannulation for cerebral perfusion made us more confident to first anastomose the left subclavian artery to make the procedure easier regardless of cerebral blood-supply.
The integrity of the circle of Willis is the key for sufficient cerebral perfusion under u-ACP. Nonetheless, there are several anatomical variations of the circle, not to mention cases of vascular dysplasia or deficiency of vascular branches. Studies have verified that the proportion of individuals with intact circles of Willis comprise 21–25% of the population, and the variation rate can be as high as 50%, especially in the posterior circulation [13, 14]. However, some patients cannot undergo CTA, magnetic resonance angiography (MRA), or digital subtraction angiography (DSA) to determine the integrity of the circle of Willis before surgery due to the severity of the disease itself or for other reasons. Therefore, b-ACP can assure continuous bilateral cerebral perfusion regardless of the integrity of the circle of Willis. Harrer et al. [15] reported that the left cerebral oxygen saturation increased by about 19% when the perfusion approach was switched from u-ACP to b-ACP.
With the advent of deep hypothermic circulatory arrest (DHAC) into clinical practice by Griepp and associates in the 1970s, the outcome of arch surgery was remarkably improved. Hypothermia provides neurological protection essentially by decreasing the global cerebral metabolic rate and increasing the cerebral tolerance to circulatory arrest. Hypothermia can also reduce temperature-dependent release and extracellular levels of excitatory neurotransmitters such as glutamate, contributing to inhibiting pro-apoptotic activity and reducing the level of free radicals and inflammatory cytokines [16]. Earlier protocols have utilized temperatures as low as 14℃ with the belief that lower temperature can sufficiently diminish cerebral metabolic demands. In recent years, surgeons began to question the limitations of DHAC. Ehrlich et al. reported that further reduction of temperature beyond 18℃ did not further decrease the oxygen requirement in the brain. It led to greater cerebrovascular homeostasis impairment and reduced cerebral perfusion [17]. Several experts have cautioned against DHAC because of its related hazards of prolonged CPB time, greater coagulopathy, and aggravated inflammatory responses. Recent studies from high-volume aortic centres have demonstrated excellent results of moderate levels of hypothermia combined with ACP [18, 19]. With the increase in the circulatory arrest temperature, the tolerance of cerebral tissue to ischemia or hypoxia reduced. In the b-ACP group, both cerebral hemispheres were perfused directly through cannulation, permitting better perfusion and a higher arrest temperature compared with the u-ACP group. Particularly, the higher circulatory arrest temperature caused less impaired cerebrovascular homeostasis, avoiding hypo-cerebral or hyper-cerebral perfusion through the physiological regulation of the cerebral vessels themselves. Finally, the higher temperature during circulatory arrest enabled shorter cooling and rewarming time, substantially reducing the CPB time.
Study limitations
This study has some inevitable limitations. First, the study is a retrospective review for 5 years, and we cannot exclude the influence of era bias. Second, this study only represents a single-centre experience instead of a multicenter experience. Thus, we need to design a multicenter prospective analysis in the future to further verify our results.
Conclusions
The b-ACP procedure had some advantages compared to u-ACP, including reduced incidence of postoperative neurological dysfunction, shorter ventilation time, shorter CBP time, and lower incidence of postoperative hypoxia, according to our research. Furthermore, we analyzed the potential risk factors for ND and revealed that preoperative neurological dysfunction, CPB duration, and perfusion method were independent risk factors for the generation of ND. Therefore, we may reduce the incidence of ND by modifying the adjustable factors including CPB time and more appropriate cerebral perfusion method.
Acknowledgements
In writing this paper, I have benefited from the presence of my teachers and colleagues. They generously helped me collect materials I needed and made many invaluable suggestions. I hereby extend my grateful thanks to them for their kind help. Particularly, I am deeply indebted to Professor Xinyan Pang, who guided me throughout my writing of this thesis. He carefully read the whole draft and offered painstaking and precious criticism. His standards of academic excellence have made my revision and exciting and gratifying experience. I also wish to sincerely thank my director, whose brilliant ideas and perceptive observations have proved immensely constructive. Furthermore, none of this would have been possible without the help of those individuals and organizations hereafter mentioned with gratitude: our department and its staff, the department of cardiovascular surgery of Qilu Hospital of Shandong University and its staff.
Abbreviations
- AD
Aortic dissection
- ND
Neurological dysfunction
- ACP
Antegrade cerebral perfusion
- u-ACP
Unilateral antegrade cerebral perfusion
- b-ACP
Bilateral antegrade cerebral perfusion
- CPB
Cardiopulmonary bypass
- MAP
Mean arterial pressure
- CVP
Central venous pressure
- ECG
Electrocardiography
- SaO2
Blood oxygen saturation
- ABG
Arterial blood gas
- NIRS
Near-infrared spectroscope
- PND
Permanent neurological dysfunction
- TND
Temporary neurological dysfunction
- AKI
Acute kidney injury
- TIA
Transient ischemic attack
- DHAC
Deep hypothermic circulatory arrest
Authors’ contributions
LZQ conceptualization, data curation, formal analysis, investigation, methodology and writing. WC conceptualization, data curation, investigation and methodology. ZXQ conceptualization, methodology and review. WSM conceptualization and review. FCC data curation, investigation and methodology. PXY conceptualization, formal analysis, investigation, methodology and writing. All authors have read and approved the manuscript.
Funding
No funding.
Availability of data and materials
The datasets analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The experimental protocol and informed consent were approved by the Institutional Review Board of our hospital-Qilu Hospital of Shandong University. And all patients involved in this study gave their informed consent verbally. Since our research was a retrospective one and we just collected patients’ data retrospectively without any intervention that could influence patients’ clinical outcome.
Consent for publication
Not applicable.
Competing interests
We declare that there are no conflicts of interest to the manuscript submitted. We do not have any commercial or personal relationships with other people or organizations in connection with our work.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zhengqin Liu and Chen Wang contributed equally to this work and should be considered co-first authors
References
- 1.Nienaber CA, Eagla KA. Aortic dissection: new frontiers in diagnosis and management: part I: from etiology to diagnostic strategies. Circulation. 2003;108(5):628–635. doi: 10.1161/01.CIR.0000087009.16755.E4. [DOI] [PubMed] [Google Scholar]
- 2.Mehta RH, Suzuki T, Hagan PG, Bossone E, Gilon D, Llovet A, Maroto LC, Cooper JV, Smith DE, Armstrong WF, Nienaber CA, Eagle KA. Predicting death in patients with acute type A aortic dissection. Circulation. 2002;105(2):200–206. doi: 10.1161/hc0202.102246. [DOI] [PubMed] [Google Scholar]
- 3.Tsai TT, Trimarchi S, Nienaber CA. Acute aortic dissection: perspectives from the International Registry of Acute Aortic Dissection (IRAD) Eur J Vasc Endovasc Surg. 2009;37:149–159. doi: 10.1016/j.ejvs.2008.11.032. [DOI] [PubMed] [Google Scholar]
- 4.Apostolakis E, Koletsis EN, Dedeilias P, Kokotsakis JN, Sakellaropoulos G, Psevdi A, et al. Antegrade versus retrograde cerebral perfusion in relation to postoperative complications following aortic arch surgery for acute aortic dissection type A. J Card Surg. 2008;23:480–487. doi: 10.1111/j.1540-8191.2008.00587.x. [DOI] [PubMed] [Google Scholar]
- 5.Kazui T, Yamashita K, Washiyama N, Terada H, Bashar AH, Suzuki K, et al. Aortic arch replacement using selective cerebral perfusion. Ann Thorac Surg. 2007;83:S796–S798. doi: 10.1016/j.athoracsur.2006.10.082. [DOI] [PubMed] [Google Scholar]
- 6.Olsson C, Thelin S. Antegrade cerebral perfusion with a simplified technique: unilateral versus bilateral perfusion. Ann Thorac Surg. 2006;81:868–874. doi: 10.1016/j.athoracsur.2005.08.079. [DOI] [PubMed] [Google Scholar]
- 7.Peterson MD, Mazine A, El-Hamamsy I, Manlhiot C, Ouzounian M, MacArthur RG, et al. Knowledge, attitudes, and practice preferences of Canadian cardiac surgeons toward the management of acute type A aortic dissection. Thorac Cardiovasc Surg. 2015;150:824–831. doi: 10.1016/j.jtcvs.2015.07.026. [DOI] [PubMed] [Google Scholar]
- 8.Conzelmann LO, Hoffmann I, Blettner M, Kallenbach K, Karck M, Dapunt O, et al. Analysis of risk factors for neurological dysfuction in patients with acute aortic dissection type A: data from the German Registry for Acute Aortic Dissection Type A (GERAADA) Eur J Cardiothorac Surg. 2012;42:557–565. doi: 10.1093/ejcts/ezs025. [DOI] [PubMed] [Google Scholar]
- 9.Usui A, Ueda Y. Arch first technique under deep hypothermic circulatory arrest with retrograde cerebral perfusion. Multimed Man Cardiothorac Surg. 2007 doi: 10.1510/mmcts.2006.001974. [DOI] [PubMed] [Google Scholar]
- 10.Milewski RK, Pacini D, Moser GW, Moeller P, Cowie D, Szeto WY, et al. Retrograde and antegrade cerebral perfusion: results in short elective arch reconstructive times. Ann Thorac Surg. 2010;89:1448–1457. doi: 10.1016/j.athoracsur.2010.01.056. [DOI] [PubMed] [Google Scholar]
- 11.Di Eusanio M, Schepens MA, Morshuis WJ, Dossche KM, Di Bartolomeo R, Pacini D, et al. Brain protection using antegrade selective cerebral perfusion: a multicenter study. Ann Thorac Surg. 2003;76:1181–1189. doi: 10.1016/S0003-4975(03)00824-5. [DOI] [PubMed] [Google Scholar]
- 12.Kamiya H, Hagl C, Kropivnitskaya I, Böthig D, Kallenbach K, Khaladj N, et al. The safety of moderate hypothermic lower body circulatory arrest with selective cerebral perfusion: a propensity score analysis. Thorac Cardiovasc Surg. 2007;133:501–509. doi: 10.1016/j.jtcvs.2006.09.045. [DOI] [PubMed] [Google Scholar]
- 13.Okahara M, Kiyosue H, Mori H, Tanoue S, Sainou M, Nagatomi H. Anatomic variations of the cerebral arteries and their embryology: a pictorial review. Eur Radiol. 2002;12:2548–2561. doi: 10.1007/s00330-001-1286-x. [DOI] [PubMed] [Google Scholar]
- 14.Klimek-Piotrowska W, Rybicka M, Wojnarska A, Wjtowicz A, Koziej M, Hołda MK. A multitude of variations in the configuration of the circle of Willis: an autopsy study. Anat Sci Int. 2016;91:325–333. doi: 10.1007/s12565-015-0301-2. [DOI] [PubMed] [Google Scholar]
- 15.Harrer M, Waldenberger FR, Weiss G, Folkmann S, Gorlitzer M, Moidl R, et al. Aortic arch surgery using bilateral antegrade selective cerebral perfusion in combination with near-infrared spectroscopy. Eur J Cardiothorac Surg. 2010;38(5):561–567. doi: 10.1016/j.ejcts.2010.03.016. [DOI] [PubMed] [Google Scholar]
- 16.Gonzalez-Ibarra FP, Varon J, Lopez-Meza EG. Therapeutic hypothermia: critical review of the molecular mechanisms of action. Front Neurol. 2011;2:4. doi: 10.3389/fneur.2011.00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ehrlich MP, McCullough JN, Zhang N, Weisz DJ, Juvonen T, Bodian CA, et al. Effect of hypothermia on cerebral blood flow and metabolism in the pig. Ann Thorac Surg. 2002;73(1):191–197. doi: 10.1016/S0003-4975(01)03273-8. [DOI] [PubMed] [Google Scholar]
- 18.Zierer A, El-Sayed Ahmad A, Papadopoulos N, Moritz A, Diegeler A, Urbanski PP, et al. Selective antegrade cerebral perfusion and mild (28 ℃–30 ℃) systemic hypothermic circulatory arrest for aortic arch replacement: results from 1002 patients. J Thorac Cardiovasc Surg. 2012;144:1042–1049. doi: 10.1016/j.jtcvs.2012.07.063. [DOI] [PubMed] [Google Scholar]
- 19.Comas GM, Leshnower BG, Halkos ME, Thourani VH, Puskas JD, Guyton RA, et al. Acute type A dissection: impact of antegrade cerebral perfusion under moderate hypothermia. Ann Thorac Surg. 2013;96:2135–2141. doi: 10.1016/j.athoracsur.2013.06.085. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets analysed during the current study are available from the corresponding author on reasonable request.


