Abstract
A 10-year-old girl presented with ileus, urinary retention, dry mouth, lack of tears, fixed dilated pupils, and diffuse anhidrosis 7-days after a febrile illness. We hypothesized that her syndrome was due to autoimmunity against muscarinic acetylcholine receptors, blocking their activation. Using an indirect enzyme-linked immunosorbent assay for all five muscarinic receptors (M1-M5) we identified in the patient’s serum antibodies that selectively bound to M3 receptors. In-vitro functional studies confirmed that these autoantibodies selectively blocked M3 receptor activation. Thus, autoantibodies against M3 acetylcholine receptors cause acute postganglionic cholinergic dysautonomia.
Keywords: Autonomic, Autoimmune, Muscarinic, Cholinergic, Dysautonomia
INTRODUCTION
Postganglionic cholinergic dysautonomia (PCD) is a severe disorder characterized by acute onset of urinary retention, decreased gastrointestinal motility, diffuse anhidrosis, dry eyes, dry mouth, and internal ophthalmoplegia (fixed dilated pupils). All the affected autonomic functions, i.e., parasympathetic and sympathetic sudomotor, require acetylcholine neurotransmission, and signs and symptoms are due to a lack of activation of muscarinic acetylcholine receptors in peripheral tissues (Fig.1). Nicotinic acetylcholine receptors in autonomic ganglia and skeletal muscle, however, are unaffected, and patients have neither orthostatic hypotension nor muscle weakness.1–5 First recognized in 1972, PCD was thought to resemble cases of restricted botulism and believed to be caused by “..an impaired or abolished synthesis or release of acetylcholine in postganglionic autonomic nerves.”2 Because it frequently followed a viral infection, an autoimmune etiology, such as autoantibodies against postganglionic cholinergic nerve terminals inhibiting acetylcholine release, was suspected.4 However, autoimmunity against muscarinic acetylcholine receptors (mAChRs) in target tissues, rather than against nerve terminals, is also possible. We tested this hypothesis in a patient with PCD using an indirect enzyme-linked immunosorbent assay (ELISA) for all five types of mAChRs. The patient’s serum showed increased binding only against the M3 receptor, and in-vitro functional studies confirmed that these autoantibodies were blocking M3 receptor activation. Accordingly, an autoimmune-mediated disorder caused selective muscarinic failure in this patient. This is the first conclusive demonstration of antibodies blocking M3 acetylcholine receptors causing a clinical syndrome.
Figure 1: Autonomic and somatic efferent neurons and target organs.
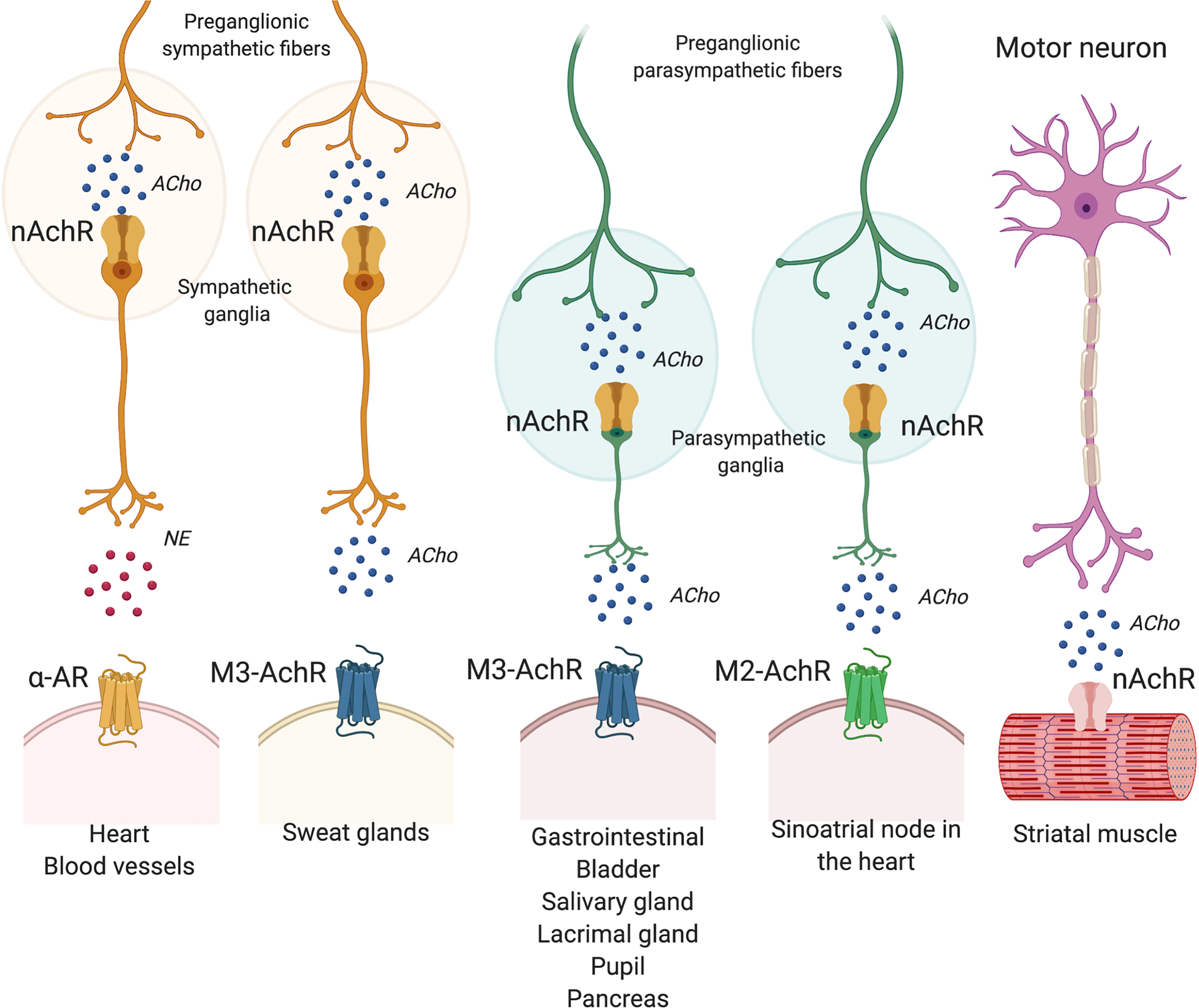
Nicotinic acetylcholine receptors (nAchR) are located in the sympathetic and parasympathetic ganglia and in striatal muscle. Muscarinic acetylcholine (ACho) receptors (mAchR) are located in sweat glands and organs and glands throughout the body (sinoatrial node in the heart, gastrointestinal system, detrusor muscle of the bladder, salivary glands, lacrimal glands, pupillary constrictor muscles, and pancreas). Given the lack of orthostatic hypotension and muscle weakness, the phenotype of postganglionic cholinergic dysautonomia is consistent with selective muscarinic failure, sparing nicotinic acetylcholine and α-adrenergic (α-AR) receptors. NE: norepinephrine.
METHODS
Case description
A week after a febrile illness with a sore throat and facial erythema that resolved spontaneously, a previously healthy 10-year-old girl acutely developed severe recurrent emesis, dysphagia, abdominal pain, and blurry vision. Within 48-hours, she developed urinary retention and was admitted to the pediatric intensive care unit. Her appetite had been minimal, she had had no bowel movements for 7 days, and her physical examination showed parched tongue (Fig.2A), dry skin and dilated pupils unresponsive to light with normal extraocular eye movements, sensory and motor function, and deep tendon reflexes. Her abdomen was painfully distended, with no bowel sounds. Her blood pressure was normal, and her heart rate was 119 bpm. Amylase was 237 U/l, and lipase was 236 U/l. A nasogastric tube was placed, and intravenous hydration started. An abdominal ultrasound disclosed urinary retention and hydronephrosis. Urodynamic testing confirmed an atonic bladder with a post-void residual volume of 401 ml requiring an indwelling catheter. A brain MRI, anti-acetylcholine ganglionic receptor, anti-Ro/SSA, and anti-La/SSB antibodies, rheumatoid factor, immunoglobulin levels, plasma and stool botulinum toxin, viral serologies, urine toxicology, and chest and abdominal computerized tomography were normal or negative. Parotid gland biopsy showed no evidence of lymphocytic, plasma cell or eosinophilic infiltrates. Clinical course and treatment. With the suspicion of an autoimmune-mediated disorder, the patient received a 5-day course of methylprednisolone (30 mg/kg/day) followed by a 5-day course of intravenous immunoglobulins (15 g per day). She also began treatment with the oral muscarinic receptor agonist bethanechol (5 mg every 6 hours), erythromycin (200 mg every 6 hours) as prokinetic, and artificial tears for alacrima. Treatment resulted in moderate symptomatic improvement: she was able to urinate without a catheter, had daily bowel movements, and resumed oral nutrition. Amylase and lipase normalized. She was discharged 40-days after admission on bethanechol, erythromycin, and artificial tears. Her weight was 32kg (35th percentile for age), 5kg less than before admission. She received no additional steroids or other immunomodulatory treatments. At a 6-month follow-up visit, she was back in school, denied urinary or gastric problems, had normal tears, but her pupils remained dilated and unresponsive to light. Her weight remained similar (34th percentile). She had similar findings during the 12- and 18-month follow-up visits but had regained the weight lost during her illness (48th and 64th percentiles respectively).
Figure 2. Oral and ocular manifestations of selective muscarinic failure.

A. Extremely dry tongue during the acute phase. B. Upon discharge her tongue had improved but still remained significantly dry. C. At the 6-month follow-up visit, her tongue had normal appearance. D. During the acute phase, pupils were mydriatic and non-reactive to light (Video) and lacrimation was absent (Schirmer test < 5 mm/5 min). E. To asses for the presence of parasympathetic denervation supersensitivity, we evaluated pupillary responses to pilocarpine instillation 0.125%. After pilocarpine 0.125 % both pupils constricted tonically (2-mm diameter) remaining miotic for 4-hours, consistent with parasympathetic denervation supersensitivity.
Autonomic function testing
Detailed autonomic reflex testing assessing cardiovascular parasympathetic and sympathetic adrenergic, sympathetic cholinergic (sudomotor), parasympathetic corneal, and parasympathetic pancreatic functions, was performed during the acute phase as previously described.6 The Schirmer test was performed to assess lacrimation.
Enzyme-linked immunosorbent assay (ELISA)
Serum was obtained from the patient during the acute phase of the disease and at 6-months follow-up and from a healthy age- and sex-matched control (34.15μg protein/μl). We developed an indirect ELISA to detect the presence of autoantibodies against muscarinic receptors. Chinese hamster ovary (CHO) cells were grown and transfected to express the human muscarinic M1, M2, M3, M4 or M5 receptors as a tagged protein with the N-terminal myc-tag (Origene, Rockville, MD, USA). Membranes were prepared from CHO cells expressing myc-tagged human muscarinic cholinergic receptors (M1 to M5). We used an anti-myc antibody to confirm the abundance of receptor in all five cell lines (data not shown).
ELISAs were carried out as described previously.7, 8 Briefly, cells were plated on poly-D-lysine-coated 24-well plates (2×105 cells/well). The next day, cells were fixed, washed and incubated with 3% BSA in PBS for 1-h at 37°C, followed by overnight incubation at 4°C with of 1:1000 myc-mAb, a 1:30,000 dilution of human primary antisera (control, acute phase, or 6-month follow-up; 0.01μl serum/300μl vol/well), or 1:500 dilution of commercial anti-M3 antibody (US Biological Life Sciences, Salem, MA, USA; cat No.324719) in 1% BSA in PBS. The wells were then washed three times followed by a 1-h incubation at 37°C with a 1:1000 dilution of either secondary anti-mouse or 1:10,000 secondary anti-human antibody coupled to horseradish peroxidase (1:1000 dilution of this antibody was used for commercial anti-M3 antibodies). The wells were washed three times with PBS (5-min each wash), and color was developed by the addition of o-phenylenediamine (5mg/10ml in 0.1M citrate phosphate buffer, pH5.0, containing 20μl of H2O2). Absorbance at 490 nm was measured with a Bio-Rad ELISA reader. Data were expressed in mean ± standard error of the mean (SEM) of three independent experiments.
Functional studies
To pharmacologically characterize the interaction between the patient’s serum and the muscarinic M3 receptor, we monitored the binding of the radio-labeled GTP analog, [35S]guanosine-5’-O-(3-thio)triphosphate ([35S]GTPγS), in response to the patient’s serum as described.9 Membranes (20μg) from CHO cells expressing myc-tagged M3 human muscarinic cholinergic receptors were pre-incubated with 1μg antisera from the patient (acute phase, and 6-month follow-up; 0.029μl serum/50μl membrane) and the control (0.029μl serum/50μl membrane) for 10-min at room temperature, followed by treatment with 1 or 10 μM pilocarpine HCl. [35S]GTPγS binding was determined as described.9 Data were expressed in mean±SEM of three independent experiments.
To test the hypothesis that antibodies against the M3 receptor in the patient’s serum obtained during the acute phase would block the M3 receptor signaling we performed a calcium mobilization assay. CHO cells expressing Myc-tagged muscarinic M3 receptors (10,000 cells/well) were plated into black 384-well clear-bottom plates. On the next day, cells were incubated with calcium-6 dye in the absence or presence of 1:250 dilution of serum from the patient (acute phase and 6-month follow-up; 0.4μl serum/100μl assay buffer/well) and the control (0.4μl serum/100μl assay buffer/well) for 1-hour at 37°C. Cells were treated with increasing concentrations of pilocarpine (0–10 μM), an M3-cholinergic agonist, and increases in intracellular Ca2+ levels were measured for 210-seconds using the fluorescent imaging plate reader (FLIPR) work station. Adenosine triphosphate (ATP) was used as a positive control for the measurement of intracellular Ca2+ levels. Data were expressed in mean±SEM of three independent experiments.
RESULTS
Autonomic evaluation
Schirmer test showed absent lacrimation (Fig.2D). Pupils at baseline were mydriatic and non-reactive to light or accommodation (Video). Pilocarpine administration resulted in tonic pupillary constriction, consistent with parasympathetic denervation supersensitivity (Fig.2E). Pancreatic polypeptide levels were 67 pg/ml and increased to only 90 pg/ml after a sham-meal procedure, consistent with impaired parasympathetic pancreatic function.10 Sympathetic skin response (SSR) and electrochemical skin conductance (ESC) were extremely low suggesting impaired sympathetic cholinergic (sudomotor) function.11 Blood pressure in the supine position was 99/55 mmHg with a heart rate of 77 bpm. Valsalva maneuver had a blood pressure overshoot indicating preserved sympathetic adrenergic function. Measures of cardiac parasympathetic function were slightly below the normal for age (E:I ratio of 1.29 compared to 1.45±0.2 in healthy aged-matched controls). Blood pressure was well maintained during 60-degree tilt with an appropriate increase in heart rate. Plasma norepinephrine levels in the supine position were normal (484 pg/ml).
ELISA
Only cells expressing the M3 receptor had significantly higher receptor recognition in the presence of the patient’s serum compared to the control’s (P<0.01) and the follow-up serum (P<0.05) indicating that the patient’s serum obtained in the acute phase was abundant in autoantibodies against the cholinergic M3 receptor, but not against the other muscarinic receptors tested (M1, M2, M4, M5) (Fig.3A). To support the presence of specific anti-M3 antibodies in the patient’s serum during the acute phase, we used a commercially available anti-M3 antibody as a positive control, that we previously tested at different concentrations in CHO cells expressing the M3 receptor and control CHO cells (data not shown). The commercial anti-M3 antibody (1:500) was highly selective for CHO cells expressing M3 receptors, but not other muscarinic receptors (M1, M2, M4 and M5)(Fig.3B). Finally, we compared in CHO cells expressing M3 receptors the signal obtained with the patient’s serum (acute phase, and 6-month follow-up), healthy control serum, and the commercial anti-M3 antibody. The receptor recognition signal with the patient’s serum during the acute phase was similar to that of the commercial anti-M3 antibody (P=n.s.); in both cases it was significantly higher than that of the healthy control and the 6-month patient’s sera (Fig.3C). This supports the presence of anti-M3 antibodies in the patient’s serum with the same activity as commercially available anti-M3 antibodies.
Figure 3. Patient’s serum from the acute phase contains antibodies blocking M3 muscarinic receptors.
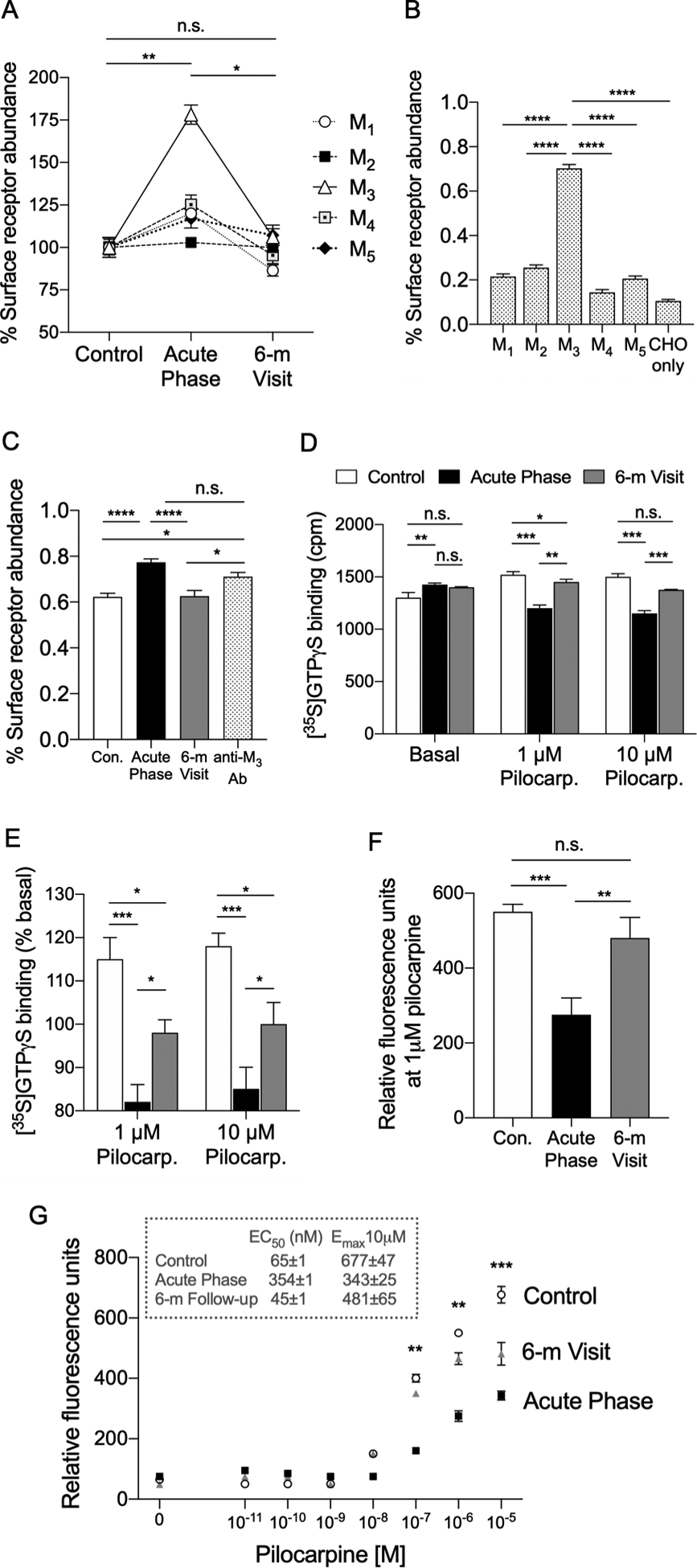
(A) ELISA results after addition of the patient’s sera. We detected a significant signal when the patient’s serum during the acute phase was exposed to cells expressing cholinergic M3 muscarinic receptors (P<0.01 vs. healthy control serum), but no difference when the serum was exposed to cells expressing M1, M2, M4 or M5 muscarinic receptors (n.s. vs. healthy control serum, not indicated in the figure). This signal was significantly reduced to levels similar to those observed when we used serum from an age- and sex-matched health control when we used the patient’s 6-month follow-up visit (6-m) serum (P<0.05 compared to the acute phase; and P=n.s. compared to the healthy control) Data are mean±SEM of three independent experiments. *P<0.05; **P<0.01; One-Way ANOVA. n.s.=not significant. (B) ELISA results after addition of a commercially available anti-M3 antibody. As expected, we detected a significant binding only in cells expressing M3 receptors, but not in the cells expressing the other muscarinic receptors, or in CHO cells devoid of muscarinic receptors (CHO only) when we used a commercial, specific anti-M3 antibody. Data are mean ± SEM of 3 independent experiments. ****P<0.0001; One-Way ANOVA. (C) ELISA results comparing anti-M3 antibody binding to cell expressing M3 receptors in the presence or absence of the patient’s serum. Both the commercially available anti-M3 antibody (anti-M3 Ab) and the patient’s serum during the acute phase (Acute Phase) showed similar binding activity to the M3 cells (P=n.s.), with higher signal compared to the sample from the healthy control (Con.) and the patient’s serum at the 6-month follow-up visit (6-m Visit). This supports the presence of specific, biologically active, anti-M3 receptors in the patient’s serum during the acute phase. Data are mean ± SEM of 3 independent experiments. *P<0.05; ****P<0.0001; One-Way ANOVA. n.s.=not significant. (D) [35S]GTPγS binding assay. The [35S]GTPγS binding assay showed blockade of M3 receptor activation induced by increasing concentrations of pilocarpine, a known M3 cholinergic agonist, in the presence of serum from the patient (Acute Phase) compared to serum obtained from a healthy control (P<0.001, acute phase vs. control). The activation of the M3 receptor was less impaired during the 6-month follow-up visit (6-m Visit), but still significantly inferior to the control serum (P<0.05 6-month vs. control). **P<0.01; ***P<0.001 One-Way ANOVA. n.s.=not significant. (E) Panel E shows data in panel D normalized (percentage) to the corresponding basal values. Data are mean±SEM of 3 independent experiments. *P<0.05; **P<0.01; ***P<0.001; One-Way ANOVA. n.s.=not significant. (F) Intracellular Ca+2 mobilization assay with 1 μM pilocarpine. In the presence of 1 μM pilocarpine, a known muscarinic agonist, the addition of the patient’s serum during the acute phase elicited a response significantly blunted compared to that observed in the presence of the healthy control serum (Con.) (P<0.001 vs. Con.). The response in the presence of the patient’s serum obtained at the 6-month follow-up visit (6-m Visit) was more intense than during the acute phase (P<0.001), and similar to that observed in the presence of the healthy control’s serum (P=.n.s.). Data are mean±SEM of three independent experiments. **P<0.01; ***P<0.001; n.s.=not significant. (G) Intracellular Ca+2 mobilization assay with increasing concentrations of pilocarpine. In the presence of the patient’s serum (Acute Phase) the relative fluorescence at increasing concentrations of pilocarpine was significantly reduced compared to the response observed in the presence of the healthy control’s serum (P<0.01 with pilocarpine at 0.1 μM and 1 μM; and P<0.001 with pilocarpine 10 μM), indicating lack of M3 receptor activation. When serum from the patient at 6-month follow-up visit (6-m Visit) was used, the relative fluorescence was significantly increased, and became similar to the values observed in the presence of the healthy control’s serum. Data are mean±SEM of three independent experiments. **P<0.01; ***P<0.001.
Functional studies
The [35S]GTPγS binding assay showed blockade of M3-receptor activation induced by increasing dosages of pilocarpine, a known M3-cholinergic agonist, in the presence of the patient’s serum (acute phase) compared to a healthy control serum (P<0.001). The activation of the M3-receptor was less impaired during the 6-month follow-up, but still significantly inferior to the control serum (P<0.05 vs. control) (Fig.3D, 3E).
The intracellular calcium mobilization assay detected and quantified the variations in intracellular calcium concentration ([Ca]i) during cell activation induced by pilocarpine in the absence or presence of serum from the patient (acute phase and 6-month follow-up) and serum obtained from a healthy control. Increasing concentrations of pilocarpine induced rapid increases in [Ca]i in the absence (EC50=4.4E−8 M; Emax 685±47) and presence of serum from the control (EC50=6.5E−8 M; Emax 677±47). The rise in [Ca]i was significantly attenuated (EC50=3.5E−7 M; Emax 343±25) in the presence of acute phase serum from the patient (P<0.001 vs. control). Cells incubated with serum from the patient at 6-month follow-up had [Ca]i responses similar to the control (EC50=4.5E−8 M; Emax 481±65; P=n.s.)(Fig.3F, 3G).
DISCUSSION
Using ELISA and in vitro functional studies, we identified high levels of antibodies in the serum of a patient with PCD that selectively bind to M3 muscarinic receptors and block their activation, a finding that explains the clinical phenotype. Thus, this is the first report of antibodies blocking M3 acetylcholine receptors conclusively causing a clinical syndrome.
There are 5 subtypes of muscarinic receptors (mAChRs).12–14 M2 and M3 are predominantly found in peripheral organs, whereas M1, M4 and M5 are more abundant in the central nervous system. When activated, M1, M3 and M5 associate with the Gq/11 protein which activates phospholipase-C, and leads to mobilization of intracellular Ca2+ and activation of protein kinase-C.15 Our results indicate that patients with PCD have a selective failure of activation of M3 receptors, with sparing of the other mAChRs.
Blockade of M3 receptors prevents contraction of the smooth muscle of the gastrointestinal tract, the bladder detrusor and the iris sphincter (Fig.1). It also prevents exocrine secretion causing dry eyes, dry mouth, anhidrosis, and impaired pancreatic exocrine function.13, 16 In contrast, heart rate variability during deep breathing was not impaired, nor had the patient resting tachycardia because M2 receptors, which mediate vagal activity at the sinus node, were unaffected. The lack of CNS symptoms, such as amnesia, delirium or stupor, as it occurs when overdosing on non-selective muscarinic receptor blockers that cross the blood-brain barrier,17 is consistent with sparing of M1, M4 and M5 receptors in patients with PCD. Knockout mice lacking M3 muscarinic receptors reproduce the phenotype of PCD with dilated pupils, impaired salivary secretion, impaired intestinal motility and urinary retention.18
Our patient had clinically evident anhidrosis, as suggested by extremely low SSR and ESC values, however the thermoregulatory sweat test (TST, not available at our center), and the quantitative sudomotor axon reflex testing (QSART, refused by the patient and her family), both the gold standard to quantify anhidrosis, could not be performed.
PCD is a rare disorder, with only 11 reported cases so far, although it is conceivable that many cases might be mislabeled as idiopathic anhidrosis, ileus or urinary retention. Antibodies against M3-AChR have been reported in the serum of some patients with Sjögren disease,19 an autoimmune disease affecting lacrimal and salivary glands, but whether they have a causative relationship is unclear. Further studies should determine the binding site of the blocking autoantibodies for the M3 receptor. Comparison of the M1, M2, M3 and M4 receptor structures reveals a high degree of conservation in orthosteric (i.e., primary active) binding sites, with substantial differences in residues contributing to allosteric binding sites, making the latter potential candidates for a selective anti-M3 antibody binding site.20
In conclusion, anti-M3 AChR autoantibodies can cause cholinergic postganglionic failure, and patients with the phenotype should be tested for their presence.
Supplementary Material
Video. Fixed dilated pupils in postganglionic cholinergic dysautonomia. Both pupils are fixed and mydriatic, with no reaction at all to light, consistent with internal ophthalmoplegia.
ACKNOWLEDGEMENTS
We thank Dr. Lourdes Bello-Espinosa (Stony Brook Neurosciences Institutes) and Dr. David S. Goldstein (National Institutes of Health) for their help and advice with this case.
Funding support: NINDS (U54NS065736-01, R01NS026880), NIDA (R37DA008863), Familial Dysautonomia Foundation, and Alfonso Martin Escudero Foundation.
Footnotes
POTENTIAL CONFLICTS OF INTEREST
All authors declare no conflict of interests related to this manuscript.
REFERENCES
- 1.Thomashefsky AJ, Horwitz SJ, Feingold MH. Acute autonomic neuropathy. Neurology. 1972. March;22(3):251–5. [DOI] [PubMed] [Google Scholar]
- 2.Andersen O, Lindberg J, Modigh K, Reske-Nielsen E. Subacute dysautonomia with incomplete recovery. Acta Neurol Scand. 1972;48(4):510–9. [DOI] [PubMed] [Google Scholar]
- 3.Harik SI, Ghandour MH, Farah FS, Afifi AK. Postganglionic cholinergic dysautonomia. Ann Neurol. 1977. April;1(4):393–6. [DOI] [PubMed] [Google Scholar]
- 4.Inamdar S, Easton LB, Lester G. Acquired postganglionic cholinergic dysautonomia: case report and review of the literature. Pediatrics. 1982. December;70(6):976–8. [PubMed] [Google Scholar]
- 5.Vassallo M, Camilleri M, Caron BL, Low PA. Gastrointestinal motor dysfunction in acquired selective cholinergic dysautonomia associated with infectious mononucleosis. Gastroenterology. 1991. January;100(1):252–8. [DOI] [PubMed] [Google Scholar]
- 6.Kaufmann H, Norcliffe-Kaufmann L, Palma JA, et al. Natural history of pure autonomic failure: A United States prospective cohort. Ann Neurol. 2017. February;81(2):287–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gomes I, Bobeck EN, Margolis EB, et al. Identification of GPR83 as the receptor for the neuroendocrine peptide PEN. Sci Signal. 2016. April 26;9(425). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gupta A, Decaillot FM, Gomes I, et al. Conformation state-sensitive antibodies to G-protein-coupled receptors. J Biol Chem. 2007. February 23;282(8):5116–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gomes I, Gupta A, Filipovska J, Szeto HH, Pintar JE, Devi LA. A role for heterodimerization of mu and delta opiate receptors in enhancing morphine analgesia. Proc Natl Acad Sci U S A. 2004. April 6;101(14):5135–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Balaji NS, Crookes PF, Banki F, Hagen JA, Ardill JE, DeMeester TR. A safe and noninvasive test for vagal integrity revisited. Arch Surg. 2002. Aug;137(8):954–8; discussion 8–9. [DOI] [PubMed] [Google Scholar]
- 11.Buchmann SJ, Penzlin AI, Kubasch ML, Illigens BM, Siepmann T. Assessment of sudomotor function. Clin Auton Res. 2019. Feb;29(1):41–53. [DOI] [PubMed] [Google Scholar]
- 12.Caulfield MP, Birdsall NJ. International Union of Pharmacology. XVII. Classification of muscarinic acetylcholine receptors. Pharmacol Rev. 1998. June;50(2):279–90. [PubMed] [Google Scholar]
- 13.Caulfield MP. Muscarinic receptors--characterization, coupling and function. Pharmacol Ther. 1993. June;58(3):319–79. [DOI] [PubMed] [Google Scholar]
- 14.Levey AI, Kitt CA, Simonds WF, Price DL, Brann MR. Identification and localization of muscarinic acetylcholine receptor proteins in brain with subtype-specific antibodies. J Neurosci. 1991. October;11(10):3218–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ishii M, Kurachi Y. Muscarinic acetylcholine receptors. Curr Pharm Des. 2006;12(28):3573–81. [DOI] [PubMed] [Google Scholar]
- 16.Levey AI. Immunological localization of m1-m5 muscarinic acetylcholine receptors in peripheral tissues and brain. Life Sci. 1993;52(5–6):441–8. [DOI] [PubMed] [Google Scholar]
- 17.Richardson JS, Miller PS, Lemay JS, et al. Mental dysfunction and the blockade of muscarinic receptors in the brains of the normal elderly. Prog Neuropsychopharmacol Biol Psychiatry. 1985;9(5–6):651–4. [DOI] [PubMed] [Google Scholar]
- 18.Matsui M, Motomura D, Karasawa H, et al. Multiple functional defects in peripheral autonomic organs in mice lacking muscarinic acetylcholine receptor gene for the M3 subtype. Proc Natl Acad Sci U S A. 2000. August 15;97(17):9579–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nakamura Y, Wakamatsu E, Matsumoto I, et al. High prevalence of autoantibodies to muscarinic-3 acetylcholine receptor in patients with juvenile-onset Sjogren syndrome. Ann Rheum Dis. 2008. January;67(1):136–7. [DOI] [PubMed] [Google Scholar]
- 20.Thal DM, Sun B, Feng D, et al. Crystal structures of the M1 and M4 muscarinic acetylcholine receptors. Nature. 2016. March 17;531(7594):335–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Video. Fixed dilated pupils in postganglionic cholinergic dysautonomia. Both pupils are fixed and mydriatic, with no reaction at all to light, consistent with internal ophthalmoplegia.


