Abstract
Nigella sativa seed and its active compounds have been historically recognized as an effective herbal panacea that can establish a balanced inflammatory response by suppressing chronic inflammation and promoting healthy immune response. The essential oil and other preparations of N. sativa seed have substantial therapeutic outcomes against immune disturbance, autophagy dysfunction, oxidative stress, ischemia, inflammation, in several COVID‐19 comorbidities such as diabetes, cardiovascular disorders, Kawasaki‐like diseases, and many bacterial and viral infections. Compelling evidence in the therapeutic efficiency of N. sativa along with the recent computational findings is strongly suggestive of combating emerged COVID‐19 pandemic. Also, being an available candidate in nutraceuticals, N. sativa seed oil could be immensely potential and feasible to prevent and cure COVID‐19. This review was aimed at revisiting the pharmacological benefits of N. sativa seed and its active metabolites that may constitute a potential basis for developing a novel preventive and therapeutic strategy against COVID‐19. Bioactive compounds of N. sativa seed, especially thymiquinone, α‐hederin, and nigellidine, could be alternative and promising herbal drugs to combat COVID‐19. Preclinical and clinical trials are required to delineate detailed mechanism of N. sativa's active components and to investigate their efficacy and potency under specific pathophysiological conditions of COVID‐19.
Keywords: autophagy, COVID‐19, immune response, inflammation, Nigella sativa, oxidative stress, SARS‐CoV‐2 infection
1. INTRODUCTION
The recent outbreak of novel coronavirus is of serious global concern. At the end of December 2019, novel coronavirus (2019‐nCoV) or Severe acute respiratory syndrome coronavirus‐2 (SARS‐CoV‐2) was identified in the Wuhan City of Hubei Province of China (Huang et al., 2020). Due to its highly contagious nature, within a very short time, this virus has spread over 210 countries and territories around the world and the coronavirus disease (COVID‐19) has been stated as a pandemic by WHO on March 11, 2020 (WHO, 2020b). As of August 4, 2020, 18,142,718 people had been infected by this virus and the number of deaths had totaled to 691,013 (WHO, 2020a). The majority of the COVID‐19 patients in China were presented in the hospital with a severe infection of the lower respiratory tract in the form of pneumonia, which is similar to SARS‐CoV and the middle east respiratory syndrome coronavirus (MERS‐CoV) patients (Ralph et al., 2020). It has also been noticed that younger patients who have strong immunity suffer from relatively less illness, while older patients who have several other health issues suffer a high illness. This virus triggers infection in the respiratory tract, nervous system, gastrointestinal tract, kidney, and liver of the patients (Ralph et al., 2020). Several neurologic signs including CAM‐ICU (confusion assessment method for the intensive care unit) positive signs (acute change or a fluctuation in mental status, inattention, disorganized thinking, and altered level of consciousness), agitation, corticospinal tract signs, perfusion abnormalities, cerebral ischemic stroke (Helms et al., 2020), and gastrointestinal disorders (Gu, Han, & Wang, 2020) were reported in severe COVID‐19 condition. In some cases, acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) may be developed which leads to a high mortality rate (X. Yang et al., 2020). Since the disease was identified, the highest efforts have continued to control this deadly outbreak. Despite it is far from the discovery of an effective vaccine against the COVID‐19. Although a significant number of compounds have been proposed and the existing drugs have also been tested for repurposing, no specific therapy has been approved yet (Sohag et al., 2020). In this reality, it is important to consider all kinds of possible options to fight against the COVID‐19.
Since the history of mankind, herbs and their formulations have been used as traditional therapies and modern medicines (Farooq et al., 2016; Hassanien, Assiri, Alzohairy, & Oraby, 2015; Pandiri & Moni, 2018). Being a divine panacea, Nigella sativa L. (Ranunculaceae) has drawn special attention in traditional medicine as well as in modern medicinal research (Ramadan, 2007). Significant levels of natural antioxidant properties and antimicrobial activities were demonstrated in cold‐pressed black cumin (Ramadan, Asker, & Tadros, 2012). The black cumin seed has a well‐defined profile of edible glycolipids (Ramadan & Mörsel, 2003). Besides, physicochemical properties and stability of black cumin seed oil have been investigated (Kiralan, Özkan, Bayrak, & Ramadan, 2014). Among various ingredients of N. sativa seed, volatile oils and alkaloids are mostly associated with biological activities. The significant constituents of its volatile oil are nigellone, thymoquinone, thymohydroquinone, dithymoquinone, thymol, carvacrol, α‐ and β‐pinene, d‐limonene, d‐citronellol, p‐cymene, carvacrol, t‐anethole, 4‐terpineol, and longifolene (Ahmad et al., 2013; Enomoto et al., 2001). The notable alkaloids are isoquinoline (nigellicimine and nigellicimine n‐oxide) and pyrazol (nigellidine and nigellicine) (Ahmad et al., 2013). Thymoquinone constitutes the major bioactive principle with a range of therapeutic benefits (Haseena, Aithal, Das, & Saheb, 2015) including antioxidant (Hosseinzadeh, Taiari, & Nassiri‐Asl, 2012), antiinflammatory (El Gazzar, El Mezayen, Marecki, et al., 2006; El Gazzar, El Mezayen, Nicolls, Marecki, & Dreskin, 2006), anticancer (Gali‐Muhtasib et al., 2008), antibacterial (Halawani, 2009), antifungal activity (Abdel Azeiz, Saad, & Darweesh, 2013), and anticonvulsant activity (Hosseinzadeh & Parvardeh, 2004). Immunomodulatory effects of black cumin seed have also been reported (Haq et al., 1995; Haq, Lobo, Al‐Tufail, Rama, & Al‐Sedairy, 1999). Also, several studies reported the antiviral effect of the black cumin seed (Barakat, El Wakeel, & Hagag, 2013; Onifade, Jewell, & Adedeji, 2013; Salem & Hossain, 2000b; Umar et al., 2016). Moreover, antiischemic effects of black cumin seed, oil, and its active constituents in different organs including brain, kidneys, heart, liver, and intestine have been reported owing to their immunomodulatory and antioxidant properties (Oskouei, Akaberi, & Hosseinzadeh, 2018). Administration of α‐hederin (0.02 mg/kg) in ovalbumin (OVA)‐sensitized rats as asthma model affected the interleukin (IL)‐2 and IL‐17 secretion pathways, altering miRNA‐133a expression (Ebrahimi et al., 2016).
Recently, a molecular docking‐based study identified nigellidine and α‐hederin among the compounds of N. sativa as novel inhibitors of SARS‐CoV‐2 (Salim & Noureddine, 2020). Even, traditional practice that involves taking black cumin seed formula and its steam has been found to relieve the symptoms of the disease. All these evidence strongly suggests the therapeutic potentials of N. sativa seed and its active constituents against COVID‐19. To gain further insight into the therapeutic benefits, we have revisited the pharmacological potentials of N. sativa seed and its bioactive constituents, and present a comprehensive outline on prospects of these natural products for the prevention and cure of COVID‐19. The main goal of our current effort is to explore the possibility of any alternatives to prevent this deadly pandemic.
2. COVID‐19 PATHOPHYSIOLOGY AND POSSIBLE INTERVENTION BY N. SATIVA SEED
Over the past two decades, substantial research progress has been made on the health‐benefiting effects of N. sativa seed and its major active compound, thymoquinone on various physiological systems, including the immune system. However, the precise mechanism of how N. sativa may mediate protective effects against viral infection has not been clearly understood. The following sections summarize the current knowledge on N. sativa in mediating the protection against SARS‐CoV‐2‐associated pathological conditions including immune dysfunction, autophagy dysfunction, oxidative stress, inflammation, and the co‐morbidities such as diabetes, hyperglycemia, cardiovascular disorders, bacterial infection, and viral infection (Table 1).
TABLE 1.
Pharmaceutical effects of N. sativa on various pathophysiological conditions
| Models | Types and doses | Effects of N. sativa or its active components on mechanisms involved | Ref. |
|---|---|---|---|
| Mouse (blood) | Hydroethanolic extract of N. sativa seed (200 mg/kg/day) | Increases concentration of IL‐6, IL‐10 and TNFα and balances Th1/Th2 lymphocytes ratio | Crotzer and Blum (2010), Gholamnezhad, Boskabady, and Hosseini (2014), and Pan et al. (2016) |
| Mouse (blood) | N. sativa oil (100 mg/100 ml/mouse for 7 days) | Increases the serum level of interferon‐gamma, numbers of CD4+ helper T cells and macrophages against murine cytomegalovirus infection | Salem and Hossain (2000b) and Umar et al. (2016) |
| Guinea‐pigs (lung and trachea) | Hydroethanolic extract of N. sativa seed (0.125 mg/ml and 0.25 mg/ml) | Increases anti‐inflammatory activity and decreases release of histamine with improved tracheal responsiveness | Boskabady, Keyhanmanesh, Khamneh, and Ebrahimi (2011), Ikhsan, Hiedayati, Maeyama, and Nurwidya (2018), Keyhanmanesh, Bagban, Nazemieh, Mirzaei Bavil, and Alipour (2013), and Saadat, Mohammadi,Fallahi, Keyhanmanesh, and Aslani (2015) |
| Mouse (lung and blood) | N. sativa oil (29. 5 ml kg−1 day−1 for 17 days) | Reduces the serum levels of IgG1, IgG2a, IL‐2, IL‐12, IL‐10, IFN‐γ, and inflammatory cells in lung tissue of mouse model of allergic asthma | Abbas, Abdel‐Aziz, Zalata, and Abd Al‐Galel Tel (2005) |
| Human (blood) | N. sativa seed powder (3 g/day for 8 weeks) |
Oxidative stress: ‐decreases plasma MDA levels ‐increases activity in erythrocyte GSH‐Px, GST, and SOD |
Mostafa, Moustafa, Mirghani, AlKusayer, and Moustafa (2013) and Sultan et al. (2015) |
| Rat (blood) | Ethanolic extract of N. sativa (different doses for 5‐week period) |
Diabetes and hyperglycemia: ‐decreases plasma glucose, serum MDA, IL‐6, immunoglobulin A, G, and M; ‐increases SOD, GST, and catalase expression ‐develops pancreatic β‐cells degeneration, inflammation, and congestion |
Ikhsan et al. (2018) |
| Rat (blood and urine) |
N. sativa oil (0.6 ml kg−1 day−1) |
Cardiovascular disorders and hypertension: ‐suppresses AA (arachidonic acid) induced platelet aggregation and blood coagulation by increasing discharge of chloride, sodium, potassium and urea followed by diuresis. |
Zaoui et al. (2000) |
| Rat (stomach and colon) |
N. sativa oil (2.5 ml/kg, orally) |
Protects stomach lining against the injurious effects of alcohol and other toxins and stress in colitis and gastritis | Abdel‐Sater (2009), Kanter, Demir, Karakaya, and Ozbek (2005) |
| Rat (heart) | Thymoquinone (10 mg/100 μl/kg, i.p) | Converts LC3I to LC3II in autophagy | |
|
Staphylococcus aureus S. epidermidis |
Thymoquinone (MICs values ranged from 8 to 32 μg/ml) | Promotes bacterial biofilm inhibition | Xiao, Ke, Shi, Zeng, and Cao (2018) |
| Staphylococcus aureus strain | MICs: N. sativa essential oil (0.03125%), thymoquinone (0.0625 mM), carvacrol (0.5 to 1 mM) | Inhibits bacterial biofilm formation | Mouwakeh, Telbisz, Spengler, Mohacsi‐Farkas, and Kisko (2018) |
| Rat | α‐Hederin (0.02 mg/kg) | Decreased IL‐17 mRNA levels and increased miRNA‐133a gene expression | Ebrahimi et al. (2016) |
Abbreviations: GSH‐Px, glutathione peroxidase; GST, glutathione S‐transferase; IgG, immunoglobulin G; IL, interleukin; LC, light chain; MDA, malondialdehyde; MIC, minimum inhibitory concentration; N. sativa, Nigella sativa; SOD, superoxide dismutase; Th, T helper type; TNF‐α, tumor necrosis factor‐α; TQ, thymoquinone.
2.1. Immune response
The innate and adaptive immune systems are immensely essential to defense SARS‐CoV‐2. Recent studies showed that angiotensin‐converting enzyme 2 (ACE‐2) on the surface of host cells (lungs, arteries, heart, kidney, and intestines) is the entry point of some coronaviruses (e.g., HCoV‐NL63, SARS‐CoV, SARS‐CoV‐2) and could act as cell surface receptor for SARS‐CoV‐2 (Xu et al., 2020; Zhou, Yang, et al., 2020; Zhou, Yu, et al., 2020). The invasion of virus stimulates the immune responsive cells (T‐cells, B‐cells, natural killer cells, neutrophils, dendritic cells, and monocytes/macrophages). Macrophages, dendritic cells, and Langerhans cells are those classical antigen‐presenting cells (APCs) for SARS‐CoV‐2 which process the invading virus (Human leukocyte antigen) (Li, Fan, et al., 2020; Li, Geng, Peng, Meng, & Lu, 2020). Many pathogen recognition receptors (e.g., TLR, toll‐like receptor) can recognize the pathogen‐associated molecular patterns (PAMPs) of SARS‐CoV‐2 (Li, Fan, et al., 2020; Li, Geng, et al., 2020). After pathogen recognition, the pattern recognition receptors (PRRs) drive the cytokine secretion from APCs and also help in expressing some effective molecules important for APCs functioning. Once being exposed to SARS‐CoV‐2, the antigen–antibody interactions at appropriate B cell surfaces drive virus‐specific cellular differentiation, maturation, antibody isotype switching, and cell residence in mucosal sites (Hurwitz, 2020). T‐cell receptor (TCR) recognizes the presented antigen and stimulates T‐cells to differentiate into helper T‐cells (CD4+) and cytotoxic T‐cells (CD8+). The helper T‐cells (Th2) secrete cytokines IL‐4 and IL‐5, which stimulate B‐cell differentiation into plasma cells (Moens & Tangye, 2014). IL‐2 is another T‐cell‐produced cytokine that is also found to be critically implicated in ERK1/2‐triggered plasma cell differentiation (Le Gallou et al., 2012). Plasma cells and memory B‐cells may also be produced from direct interaction of SARS‐CoV‐2 antigen with BCR (B‐cell receptor). Then, SARS‐CoV‐2 antigen‐specific antibodies are produced by plasma cells that can kill SARS‐CoV‐2. On the other hand, interferons (IFNs) and inflammatory factors may limit viral spreads (Ben Addi et al., 2008). Dendritic cells could be very efficient for effective activation of B‐ and T‐lymphocytes that provide quick responses through inducing innate and adaptive immunity against SARS‐CoV‐2 (Li, Fan, et al., 2020; Li, Geng, et al., 2020). Cytotoxic T‐cells (CD8+ cells) produce IFNs and granzymes, which may induce natural killer cells to kill SARS‐CoV‐2 (Shahabi et al., 2020) or directly destroy virus‐infected cells (Yi, Lagniton, Ye, Li, & Xu, 2020). Maladaptive inflammatory response was found in some severe COVID‐19 patients due to activation of complement systems where C3 and C5 signaling pathways accumulate various proinflammatory cytokines (such as IL‐1, IL‐6, IL‐8, IL‐21, and TNF‐α) (Risitano et al., 2020).
Previous studies in rats and guinea‐pigs showed that N. sativa seed extracts and its compounds such as thymoquinone, nigellone, and thymohydroquinone possessed significant antihistamine effects (Chakravarty, 1993; Gilani, Aziz, Khurram, Chaudhary, & Iqbal, 2001; Marozzi, Kocialski, & Malone, 1970). El‐Kadi and Kandil reported that N. sativa seeds have immuno‐potentiating properties in human T cells in vitro (El‐Kadi & Kandil, 1987). By inhibiting TANK‐binding kinase 1 (TBK1), thymoquinone downregulates interferon regulatory factor 3 (IRF‐3) activation, which has a critical role in viral and bacterial innate immune responses by regulating the production of Type I interferons (Aziz, Son, & Cho, 2018). T‐lymphocytes were found to be activated by N. sativa seeds to secrete the interleukin, IL‐3 (Haq et al., 1995). A stimulatory effect on macrophages was also observed through a direct effect of N. sativa seeds or via IL‐1 (Haq et al., 1995). Purified proteins from N. sativa seeds are shown to have suppressive and stimulatory effects on cultured lymphocytes to produce cytokines (e.g., IL‐1) (Haq et al., 1999). Besides, the serum level of IL‐10 in rheumatoid arthritis patients increased when N. sativa oil capsule was administered (Hadi, Kheirouri, Alizadeh, Khabbazi, & Hosseini, 2016).
2.2. Autophagy
Autophagy is a natural, regulated, and catabolic mechanism that mediates the degradation of damaged‐cellular components through the actions of the lysosomal system. Autophagy is involved in several physiological processes, namely, cell differentiation and development, starvation and degradation of cellular structures and thus maintaining homeostasis (Klionsky et al., 2016). The alteration of immune system is common in infections with highly pathogenic viruses which facilitates immune dysfunction. Therefore, to fight against viruses like SARS‐CoV infections in the pulmonary system, a powerful immune response is needed (Li, Fan, et al., 2020; Li, Geng, et al., 2020). Being a part of the cell surveillance system, autophagy plays an important role in immune responses (Crotzer & Blum, 2010; Pan et al., 2016).
SARS‐CoV‐2 infection suppresses autophagy (Gassen et al., 2020; Hannan, Rahman, et al., 2020). While autophagy machinery is essential for their replication, some viruses evolved strategies to evade autophagy flux (Gassen et al., 2019). Pharmacological agents that induce autophagy may, therefore, have antiviral effects against SARS‐CoV‐2 (Dong & Levine, 2013). A study indicates that thymoquinone acts as a cardioprotective agent by promoting autophagy (Xiao et al., 2018). The conversion of LC3I to LC3II is an indicator of autophagic activity (Sohn et al., 2017). The study shows that pretreatment of thymoquinone augmented the expression of LC3II while expression of p62 was inhibited (Xiao et al., 2018), indicating the activation of autophagy. In addition, the effects of thymoquinone on autophagy in myocardium were partly eliminated when treated with chloroquine (autophagy inhibitor), supporting the probable contribution of autophagy in thymoquinone‐facilitated cardioprotective properties.
Interestingly, some existing drugs having autophagy modulatory effects were found to be promising against SARS‐CoV‐2 infections (Shojaei, Koleini, et al., 2020; Shojaei, Suresh, Klionsky, Labouta, & Ghavami, 2020). Instead of antagonizing the viral effect, these drugs also suppress autophagy flux in a similar way the virus does, leading to increased accumulation of autophagosomes and subsequent activation of the apoptotic pathway that results in apoptotic death of SARS‐CoV‐2‐infected cells, and thus hinder the virus replication (Shojaei, Suresh, et al., 2020). Based on this autophagy‐dependent mechanism, it can be anticipated that suppressing the autophagy flux can also be a possible therapeutic option to fight against COVID‐19. To the existing knowledge, it is not clear whether N. sativa or thymoquinone has similar autophagy modulatory effect, and therefore, further research is being proposed to underpin this possibility too.
2.3. Inflammation and cytokine storm
Cytokine storm syndrome (CSS) might happen to patients with severe SARS‐CoV‐2 infection requiring intensive care (Mehta et al., 2020). Excess production of immune cells (Channappanavar & Perlman, 2017) and mediators of pro‐inflammatory and inflammatory cytokines cause cytokine storms (Huang et al., 2020; Li, Fan, et al., 2020; Li, Geng, et al., 2020; Mehta et al., 2020). SARS‐CoV‐2‐ and SARS‐CoV‐infected patients showed critical autoimmune inflammation and life‐threatening edema associated with mucins and fibrin (Mehta et al., 2020). In SARS‐CoV‐2 infection, the severity of infection is augmented due to the reduction of the antiviral defenses related to early innate immunity and subsequent elevation of inflammatory cytokines storm (Blanco‐Melo et al., 2020). The disease severity and poor prognosis are found to be involved with excessive production of proinflammatory cytokines, such as IL‐1, IL‐6, IL‐12, IFN‐γ, and TNF‐α in respiratory systems. Among these innate cytokines, an exessive induction of IL‐6 has been diagnosed as a main culprit in patients with COVID‐19 (Costela‐Ruiz, Illescas‐Montes, Puerta‐Puerta, Ruiz, & Melguizo‐Rodriguez, 2020). In the maximum cases of severe COVID‐19 patients, the cytokine storm was associated with elevated levels of erythematosus sedimentation rate (ESR) and CRP, those results with ARDS, respiratory failures, thrombocytopenia, and thrombosis (Zhang et al., 2020).
Moreover, the characteristic inappropriate inflammatory response in SARS‐CoV‐2 infection is highly related to an attenuated function of type I and II IFNs along with an increased expression of IL‐6 (Blanco‐Melo et al., 2020) and C‐X‐C motif chemokine 10 (CXCL10), chemokine ligand 2 (CCL2), CCL3, and CCL4 (Huang et al., 2020). Cytokines, such as IL‐1β, TNFα, and IL‐17 are also responsible for Th17 type responses, causing vascular permeability and leakage developing a sign of severe SARS‐CoV‐2 infection (Wu & Yang, 2020). On the other hand, IL‐37 is known to impede immune activities, inhibit MHC‐II and TNF, IL‐1, IL‐6, IL‐8 modulate inflammation during SARS‐CoV‐2 infection (Conti et al., 2020). NF‐kB pathway positively regulates cytokines and chemokines (Conti et al., 2020). Upon recognition of the PAMPs of SARS‐CoV‐2 by pathogen recognition receptors (e.g., TLR) on the surface of infected‐host cells or immune cells, NF‐kB pathway is activated upregulating the synthesis and secretion of cytokines and chemokines (Conti et al., 2020). To mitigate SARS‐CoV‐2, it is necessary to inhibit Th17 type responses, cytokine secretion, and the master inflammatory regulator, NF‐kB pathway. Several previous reports also corroborated that NF‐kB suppression or regulation is positively related to the enhancement of IFN‐mediated antiviral activity (Mahase, 2020).
Since phytochemicals have potential roles to modulate or suppress NF‐kB activation and thereby inflammation, these could be alternatively used to fight against SARS‐CoV‐2 (Rahman, Biswas, & Kirkham, 2006). Phytochemicals also modulate MAP kinase, phosphatidylinositide kinase, JAK/STAT, TLR, and other pro‐inflammatory signaling molecules (Yahfoufi, Alsadi, Jambi, & Matar, 2018). The volatile oil of N. sativa seed exhibited a remarkable pain‐relieving effect in acetic acid‐induced writhing, formalin, and tail‐flick tests (Zakaria, Jais, & Ishak, 2018). There was a significant increase in antiinflammatory activity and decrease in level of histamine release with improved tracheal responsiveness in animal models experimented with various extracts, oil, and another bioactive compound, α‐hederin of N. sativa (Boskabady et al., 2011; Ikhsan et al., 2018; Keyhanmanesh et al., 2013; Saadat et al., 2015). It has been reported that 5‐lipoxygenase and leukotriene C4 synthase in human blood cells were inhibited by N. sativa oil and thymoquinone (Houghton, Zarka, de las Heras, & Hoult, 1995). Both enzymes can produce inflammatory mediators called leukotrienes and prostaglandins (Houghton et al., 1995; Mansour & Tornhamre, 2004). In addition, N. sativa seed fixed oil decreased the serum IgG1, IgG2a, IL‐2, IL‐12, IL‐10, IFN‐γ, and inflammatory cells in lung tissue of murine model of allergic asthma (Abbas et al., 2005). Considering the antiinflammatory actions of N. sativa seed and its different extracts, these might be potentially used for the prevention as well as cure of SARS‐CoV‐2 viral infection.
2.4. Oxidative stress
Oxidative stress is considered as a biomarker for various disease conditions, such as neurological disorder, cancer, aging, and endocrine illness (Lupoli, Vannocci, Longo, Niccolai, & Pastore, 2018). In addition, many viral infections, such as human immunodeficiency virus (HIV), respiratory syncytial virus (RSV), influenza A, hepatitis viruses, Epstein–Barr virus (EBV), and other viruses induce oxidative stress (Ivanov, Bartosch, & Isaguliants, 2017). Oxidative stress in chronic viral hepatitis triggers liver fibrosis, cirrhosis, cancer, and metabolic dysfunction (Ivanov et al., 2017). In acute respiratory viral infections, oxidative stress increases lung tissue injury and epithelial barrier dysfunction and further it plays an important role in secondary infections (Ivanov et al., 2017). For example, in the presence of inflammatory signals, the influenza viruses damage lungs and airways. The virus activates monocytes and polymorphonuclear leukocytes to produce reactive oxygen species (ROS), a mediator of oxidative stress (Jacoby & Choi, 1994). The produced ROS then contributes to the pathogenesis of influenza virus infection (Peterhans, Grob, Burge, & Zanoni, 1987).
Many viral infections elicit “cytokine storm,” which is responsible for activation of endothelial cells in lung capillary, infiltration of neutrophil, and increased oxidation. Some immune cells notably macrophages and neutrophils produce a lot of ROS (Loffredo et al., 2019; Perrone, Belser, Wadford, Katz, & Tumpey, 2013). Increased oxidative stress is responsible for pulmonary injuries, such as ALI and ARDS (Hecker, 2018; Yan et al., 2019). Among the pandemic viruses, coronaviruses and influenza viruses cause lethal lung injuries and death from ARDS which is a vital reason for fatality (Chen, Liu, et al., 2020; Chen, Zhou, et al., 2020; Fowler Iii et al., 2017; D. Wang et al., 2020). Undoubtedly, elevated oxidative stress is an indication of ARDS that generates cellular injury, organ failure, and death. ARDS may lead to severe hypoxemia which cooperates with frequent uncontrolled inflammation, oxidative injury, and damage to the alveolar–capillary barrier (Meng, Zhao, & Zhang, 2019). According to recent clinical reports, the therapeutic time for COVID‐19‐infected patients is longer than 14 days. In addition, out of 29 patients with COVID‐19 pneumonia, 27 (93%) showed increased CRP, a marker of inflammation and oxidative stress (Chen, Liu, et al., 2020; Chen, Zhou, et al., 2020). An early clinical study in Wuhan reported that 63 out of 73 patients (86.3%) with COVID‐19 had a remarkable CRP level (Chen, Liu, et al., 2020; Chen, Zhou, et al., 2020). The elevated level of CRP was likely due to acute inflammatory response and subsequent cytokine storms during COVID‐19 pathogenesis (Huang et al., 2020).
Natural antioxidants that are derived from plants are valuable to treat various diseases including viral infections. Among different medicinal plants, antioxidant activities of N. sativa has been found much effective in various preclinical models (Ahmad & Beg, 2016; Omidi, Khorram, Mesgari, Asghari‐Jafarabadi, & Tarighat‐Esfanjani, 2017; Ozdemir, Kantekin‐Erdogan, Tat, & Tekin, 2018; Sultan et al., 2015). A study in 30 postmenopausal women revealed that consumption of both Allium sativum and N. sativa seed continuously for 2 months improved antioxidant status (Mostafa et al., 2013). In most of these cases, there was a significant decrease in plasma malondialdehyde (MDA) levels with increased activity in erythrocyte glutathione peroxidase (GSH‐Px), Glutathione‐S‐transferase (GST), and superoxide dismutase (Mostafa et al., 2013; Sultan et al., 2015). In a clinical trial of 50 obese volunteers, N. sativa seed oil in corporation with a low‐calorie diet showed a significant increase in antioxidant capabilities (Namazi, Mahdavi, Alizadeh, & Farajnia, 2015). Correspondingly, one observation related to thymoquinone and N. sativa oil administration demonstrated improvement of cisplatin‐induced abnormality of enzymatic and nonenzymatic antioxidant defense mechanism in rat intestine (Shahid, Farooqui, Khan, & Khan, 2018).
3. SARS‐COV‐2 INFECTION AND COMORBODITIES
3.1. Diabetes and hyperglycemia
Diabetes and unrestricted glycemia are associated with increasing severity and deaths in patients infected by different viruses including 2009 pandemic influenza A (H1N1) (Schoen, Horvat, Guerreiro, de Castro, & de Giassi, 2019), SARS‐CoV (J. K. Yang et al., 2006) and MERS‐CoV (Banik, Alqahtani, Booy, & Rashid, 2016). Hyperglycemia was identified in patients with SARS‐CoV in 2003, where transient impairment of pancreatic islet cell function was noticed (Yang, Lin, Ji, & Guo, 2010). In initial studies in patients with COVID‐19, hyperglycemia was reported in 51% of cases (Chen, Liu, et al., 2020; Chen, Zhou, et al., 2020). However, we should not overlook hyperglycemia, as it can lead to patient immune suppression and any further complications (Butler, Btaiche, & Alaniz, 2005). Patients with diabetes and cardiovascular co‐morbidity have an increased risk of severe COVID‐19 complications, including ARDS, pneumonia, and multi‐organ failure (L.‐P. Guo, Fan, et al., 2020; T. Guo, Liu, et al., 2020; Gupta, Ghosh, Singh, & Misra, 2020).
A mixture of N. sativa and Cinnamomum cassia improves serum glucose concentrations, lipid profile, and renal function both in streptozotocin (STZ)‐induced diabetic rats and other animals (Kaur, Invally, Khan, & Jadhav, 2018). It is also evident that N. sativa decreases fasting plasma glucose, hemoglobin A1c, TBARBs, and increases total antioxidant capacity including SOD and glutathione levels (Kaatabi et al., 2015). For instance, reductions in fasting plasma glucose and glycated hemoglobin (HbA1c) was observed in patients with type‐2 diabetes when N. sativa was administered (Bamosa et al., 2015). Improved insulin levels, lipid profile, increased pancreatic, and hepatic antioxidant enzymes with increased glycogen contents and average pancreatic islet extent can be achieved by black cumin seed oil (Abdelrazek, Kilany, Muhammad, Tag, & Abdelazim, 2018). Modulating oxidative status (El Rabey, Al‐Seeni, & Bakhashwain, 2017), diminishing inflammation (El Rabey et al., 2017), controlling the glucose homeostasis and serum lipid profiles (Daryabeygi‐Khotbehsara, Golzarand, Ghaffari, & Djafarian, 2017), rising in good cholesterol (HDL‐c), with concomitant elimination of bad cholesterols (LDL‐c, TC, and TG) as well as body mass (Daryabeygi‐Khotbehsara et al., 2017; Kaur et al., 2018) are very potential aspects of N. sativa as an antidiabetic agent.
3.2. Cardiovascular disorder and hypertension
A study conducted in Wuhan, which showed a result of an experiment with 150 confirmed patients of COVID‐19, 7% of fatality was attributed in myocarditis with circulatory collapse, while in 33% myocarditis had played an important role in the final adverse outcome (Ruan, Yang, Wang, Jiang, & Song, 2020). Effective lipid‐lowering therapy and cholesterol reduction might significantly suppress the pathology related to coronavirus infection. Statins, particularly, found to prevent SARS‐CoV‐2‐induced acute coronary syndrome through stabilizing atherosclerotic plaques (Madjid, Safavi‐Naeini, Solomon, & Vardeny, 2020). Thus, statins and other lipid‐lowering therapies found to exert immunomodulatory and antiinflammatory properties against SARS‐CoV‐2 infection burdens through efficient blocking of virus attachment and internalization (Jeon & Lee, 2018). N. sativa and its active constituent thymoquinone can operate centrally on cardiovascular actions and can act as antihypertensive agents in anesthetized rats (El Tahir, Ashour, & Al‐Harbi, 1993). Methanol soluble portion of N. sativa oil can show suppressing results in AA (arachidonic acid) induced platelet aggregation and blood coagulation (Enomoto et al., 2001). Besides, dichloromethane extract of N. sativa can partly contribute to antihypertensive activity by increasing discharge of chloride, sodium, potassium, and urea followed by diuresis (Zaoui et al., 2000). Black cumin seed oil resulted in lower levels of harmful LDL‐cholesterol, and higher levels of beneficial HDL cholesterol in animal model indicating a protective role against atherosclerosis (Al‐Naqeep, Al‐Zubairi, Ismail, Amom, & Esa, 2011; Nader, El‐Agamy, & Suddek, 2010). A reduction in serum cholesterol, triglyceride and glucose levels, and leucocyte and platelet counts, and a rise in Hb and PCV levels compared to control were observed after administrating N. sativa orally, which indicates that N. sativa oil has beneficial effects on hyperglycemia, hyperlipidemia, and certain types of anemia (Zaoui et al., 2002).
3.3. Autoimmune and autoinflammatory diseases following COVID‐19
Children were alarmingly affected by COVID‐19, where autoimmune and autoinflammatory diseases lead to overlapping symptoms with pediatric inflammatory multisystemic syndrome (PIMS), which includes Kawasaki‐like diseases (Galeotti & Bayry, 2020; Verdoni et al., 2020), hyperinflammatory shock (Riphagen, Gomez, Gonzalez‐Martinez, Wilkinson, & Theocharis, 2020), and multisystem inflammatory syndrome in children (MIS‐C) (Belhadjer et al., 2020). Due to association of Kawasaki‐like disease symptoms with SARS‐CoV‐2 infection, the complex syndrome is reported as “Kawa‐COVID‐19” or KD‐COVID‐19 (Pouletty et al., 2020; Toubiana et al., 2020; Whittaker et al., 2020) from several studies in France, Italy, United Kingdom, United States of America, and Spain. KD‐COVID‐19 pediatric patients have remarkably increased CRP, IL‐6, IL‐8, and TNF‐α (Waltuch et al., 2020).
N. sativa might have beneficial roles to control the emerging fetal incidence of PIMS or Kawasaki‐like disease associated with COVID‐19 by modulating immune response. N. sativa extracts and/or its bioactive compounds such as thymoquinone, nigellone, and α‐hederin exhibits antihistaminic, antieosinophilic, antileukotrienes, antiimmunoglobulin and reduced‐proinflammatory cytokines (IL‐2, IL‐4, IL‐5, IL‐6, IL‐12, and IL‐13) in in vitro/in vivo models (A. Koshak, Koshak, & Heinrich, 2017). In addition, thymoquinone‐rich extract produced potent favorable immunomodulation in asthma inflammation by suppressing IL‐2, IL‐6, and PGE2 in T‐lymphocytes as well as IL‐6 and PGE2 in monocytes (Koshak, Yousif, Fiebich, Koshak, & Heinrich, 2018). Though the exact mechanism of action in Kawasaki‐like disease is unclear, there has been a clear indication that an increase in circulating inflammatory molecules is seen during this incidence with an event of myocarditis (Belhadjer et al., 2020; Toubiana et al., 2020). Antihypertensive agents as well as antiinflammatory action of N. sativa along with its different extracts have proven beneficial effects for both prevention and cure of this condition (Abbas et al., 2005; Ojha et al., 2015). In addition, cytokine storm might affect patients with Kawasaki‐like disease (Henderson et al., 2020; W. Wang, Gong, Zhu, Fu, & Zhang, 2015) and thymoquinone can prevent this incidence by its antiinflammatory and antioxidative activities (L.‐P. Guo, Fan, et al., 2020; T. Guo, Liu, et al., 2020).
3.4. Bacterial co‐infection in COVID‐19
Some studies have recently shown that many COVID‐19 patients are developing secondary bacterial coinfections such as bacterial pneumonia and sepsis, which are considered a serious threat to severe COVID‐19 patients. A retrospective cohort study published in Lancet included 191 COVID‐19 patients in Wuhan, China where 27 (50%) of the 54 deaths in hospital had secondary bacterial lung infections and the number of bacterial lung‐infected COVID‐19 patients was 28 (Zhou, Yang, et al., 2020; Zhou, Yu, et al., 2020). Staphylococcus aureus is the main pathogen of secondary infections in influenza but Streptococcus pneumoniae and Haemophilus influenzae are also common (Low, 2008). Although, data are not available yet which pathogens are associated with secondary infections in COVID‐19, the most commonly detected co‐pathogens detected in a study of 30 COVID‐19 patients in Qingdao, China were influenza A (60%) and influenza B (53%), followed by Mycoplasma pneumoniae (23%) and Legionella pneumophila (20%) (Xing et al., 2020). Another study found that about 50% of patients died because of viral infection, while the other 50% were caused by secondary infections (Ruan et al., 2020). In the case of H1N1 influenza and pneumonia patients, bacterial coinfection was common which ranges from 12% to 19% (MacIntyre et al., 2018). Additionally, bacterial co‐infection was accompanied by other serious illnesses (MacIntyre et al., 2018). Neutrophil‐lymphocyte ratio (NLR) is a well‐known marker of systemic inflammation and infection, and has been studied as an indicator of bacterial infection comprise of pneumonia (Curbelo et al., 2017; Liu et al., 2016). In another study, severely infected‐COVID‐19 patients were found with elevated NLR (an increased neutrophil count and a decreased lymphocyte count) during the severe phase that implied the potential critical condition and serious disturbance of the internal environment (D. Wang et al., 2020).
A range of antibiotics can be used in secondary bacterial infection. Azithromycin has been found to prevent secondary bacterial complications in post‐Zika and ‐Ebola virus infections (Madrid et al., 2015; Retallack et al., 2016). Moreover, in case of other respiratory viral infections, Azithromycin reveals to reduce the severity of bacterial co‐infections (Bacharier et al., 2015). More than 20 years, bacteria‐induced Whipple's diseases were treated with hydroxychloroquine (Lagier & Raoult, 2018). Some studies provided insightful information on the antiviral effects of chloroquine against SARS coronavirus (Keyaerts, Vijgen, Maes, Neyts, & Van Ranst, 2004; Rolain, Colson, & Raoult, 2007; Savarino, Di Trani, Donatelli, Cauda, & Cassone, 2006). The synergistic effect of hydroxychloroquine and azithromycin works against SARS‐CoV‐2 and prevent bacterial super‐infections. In accordance with the report, COVID‐19 patients treated with the combination of hydroxychloroquine and azithromycin were improved compared with patients treated with hydroxychloroquine only, and control group (Gautret et al., 2020). However, no effective drugs have been offered to treat COVID‐19 patients yet.
A wide range of Gram‐positive and Gram‐negative bacteria was suppressed by thymoquinone, a molecule obtained from N. sativa seeds (Abdallah, 2017). N. sativa showed a significantly higher zone of inhibitions in various bacteria (Hasan, Nawahwi, & Malek, 2013; Maryam, Fatimah, Ebtesam, Abdulrahman, & Ineta, 2016). N. sativa essential oil and its bioactive compounds (thymoquinone, carvacrol, and p‐cymene) were found resistance modifiers against S. aureus and inhibited bacterial biofilm formation (Mouwakeh et al., 2018). A noticeable bactericidal activity of thymoquinone was observed in Gram‐positive cocci associated with minimum inhibitory concentration (MIC) ranging from 8 to 32 μg/ml. In addition, the molecule revealed MIC at 22 μg/ml for S. aureus and 60 μg/ml for S. epidermidis (Chaieb et al., 2011). All of these can become a scientific basis for the investigation of potential uses of N. sativa seed as curative options for COVID‐19 patients coinfected with bacteria.
4. POSSIBLE PROTECTIVE ROLE OF N. SATIVA SEED AGAINST COVID‐19
N. sativa extract contains several important active sites like thymoquinone (27.8%–57.0%), ρ‐simen (7.1%–15.5%), karvakrol (5.8%–11.6%), t‐anetol (0.25%–2.3%), 4‐terpineol (2.0%–6.6%), and longifoline (1.0%–8.0%) reported to exert antiviral, antitumor, and antimicrobial activities (Salem & Hossain, 2000b). A recent report is corroborated with the beneficial effects of N. sativa extracts to suppress SARS‐CoV‐2 virus load through the augmented induction of IL‐8 (Ulasli et al., 2014).
Black seed oil was shown to increase the number and action of CD4+ T cells and augmented levels of IFN‐γ thus overpowering on viral load in mice infected with cytomegalovirus (Forouzanfar, Bazzaz, & Hosseinzadeh, 2014). After administration of N. sativa oil, hepatitis C virus (HCV)‐infected patients improved health conditions such as total protein, red blood cell, and platelet count, decreased fasting blood glucose, and postprandial glucose in both diabetic and non‐diabetic HCV individuals (Barakat et al., 2013). Treatment with N. sativa for half a year was found to recover a 46‐year‐old HIV patient (Onifade et al., 2013). Furthermore, treating with a mixture of N. sativa and honey, a mid‐aged woman with HIV positive got rid of HIV and the number of CD4+ T cells was good enough to declare that she is recovered from HIV infection (Onifade et al., 2013). Altogether, it has been strongly suggested that black cumin seed could be a potential natural product to treat several incurable infectious diseases such as HIV but the efficiency of black cumin is yet to be determined.
In HIV‐infected patients, ROS may activate viral replication while oxidants are known to contribute to the loss of CD4 T cells by apoptosis. But antioxidants can suppress the apoptosis, which indicates a relation between antiviral and antioxidant actions (Peterhans, 1997). In a study with hepatitis C virus (HCV)‐infected individual surprisingly showed a decrease in viral load and an augmented total antioxidant activity, total protein and albumin, improved RBC and platelet counts when supplemented with black seed oil (Barakat et al., 2013). To investigate the antiviral effect of black seed oil, MCMV (murine cytomegalovirus) has been used as a model in a study where it was concluded that N. sativa oil can produce antiviral effect against MCMV infection by increasing level of serum of IFN‐γ, number of CD4+ helper T‐cells, suppressor function, and numbers of macrophages (Salem & Hossain, 2000b). The viral infection is known to be controlled by several components of immune system including natural killer cell (NK cells), and specific cells including CD4 and CD8 T‐cells (Salem & Hossain, 2000a) and it is proved that N. sativa oil can induce antiviral cellular response by rising CD4 cells (Salem & Hossain, 2000b). The antiviral effects of thymoquinone against avian influenza virus (H9N2) and murine cytomegalovirus infection models were confirmed. N. sativa seed fixed oil had stimulatory effects on CD4+ T‐lymphocytes in murine BALB/c cytomegalovirus model (Salem & Hossain, 2000b; Umar et al., 2016).
Coronavirus spike protein can utilize angiotensin converting enzyme 2 (ACE2) like a receptor for entering into cells (Kuhn, Li, Choe, & Farzan, 2004). The α‐hederin gives better energy score when compared to chloroquine, hydroxychloroquine, and favipiravir. Preventive potentials of N. sativa seed oil against COVID is yet to be revealed. The molecular docking system discovered that N. sativa may inhibit COVID‐19 through its main compound nigellidine and α‐hederin. So further insights into in vivo and in vitro experiments with those active compounds are required (Salim & Noureddine, 2020).
5. IMPLICATIONS AND LIMITATIONS
COVID‐19 pandemic poses a serious threat to human life. The host‐directed immunotherapies might be an effective adjunct therapy in severe cases to reduce exacerbated pathogenesis and inflammation‐associated lung damage. There are several immunotherapeutic approaches targeting either inflammatory mediators, passively to reduce SARS‐CoV‐2 invasions, or prevent viral entry (Bonam, Kaveri, Sakuntabhai, Gilardin, & Bayry, 2020). Several trials are still ongoing and yet to define clearly the precise roles of those drugs that block IL‐1, IL‐2, IL‐6, IL‐18, and IFNγ to reduce the COVID‐19 pathogenesis (Alijotas‐Reig et al., 2020; Halyabar et al., 2019). Although scientists across the globe have been paying their utmost efforts to find a possible solution, no suitable therapy against COVID‐19 has yet been available. Along with the current drug development strategy that mainly focused on repurposing conventional therapeutics, the alternative approach, for example, pharmacological intervention by natural products, also demands comprehensive attention. In this perspective, it is important to explore possible alternatives utilizing unconventional resources. In this review, we revisited the pharmacological properties such as antioxidation, antiinflammation, immunomodulation, and autophagy induction of a popular panacea, black cumin seed, and its metabolites that may play a significant role in intervening the pathophysiology of COVID‐19, including immune disturbance, oxidative stress, and autophagy dysfunction (Figure 1). Supplementation with N. sativa oil and administration of its active compounds also returned with many beneficial outcomes in several COVID‐19 comorbidities such as diabetes, cardiovascular disorder, rheumatoid arthritis, and many bacterial and viral diseases (Figure 1). Recently, a clinical trial of 30 participants has been initiated by honey and N. sativa administration against SARS‐CoV‐2 infection (NCT04347382) (Sohaib Ashraf, 2020). This trial may help establish a new therapy for the treatment of this pandemic leading to a decrease in mortality, viral load, and complications rate by SARS‐Cov‐2 viral infection. However, more studies of the precise mechanism of actions of N. sativa with employing appropriate preclinical and clinical models would be immensely essential toward prevention and surveillance of COVID‐19.
FIGURE 1.
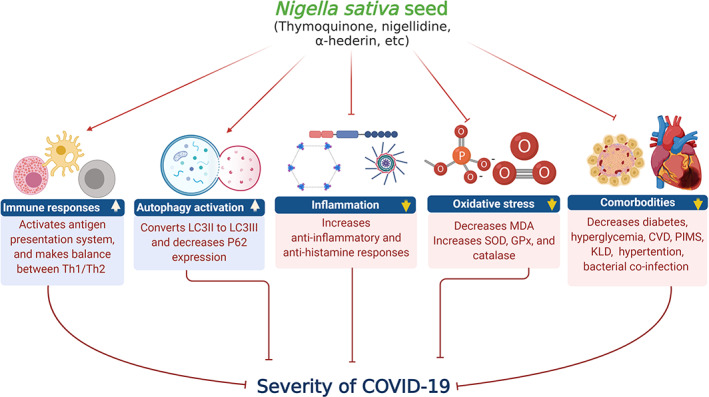
Mechanisms involved in pharmacological effects of N. sativa on COVID‐19. Black cumin seed or its active compounds activate immune cells, initiate antigen presentation system, and make balance between Th1 and Th2 cytokines. The bioactive compounds of black cumin seed increase conversion of LC3II to LC3III, a marker of autophagy activation. Besides, black cumin decreases inflammation, oxidative stress (MDA) and increases anti‐inflammation, ant‐histamine responses, SOD, and GPx. Comorbodities increase the severity of SARS‐CoV‐2 infection. Black cumin may also be beneficial through improving comorbodity situation in SARS‐CoV‐2‐infected patients. CVD, cardiovascular disease; KLD, Kawasaki‐like diseases; PIMS, pediatric inflammatory multisystemic syndrome [Colour figure can be viewed at wileyonlinelibrary.com]
Thymoquinone shows a set of therapeutic benefits with its antioxidant, antiinflammatory, antimicrobial, and anticonvulsant activities. Thymoquinone substantially attenuates pro‐inflammatory response by suppressing TNF‐α, LPS, or other inflammatory stimuli‐induced NF‐κB activation (Note, 2018). Thymoquinone is shown to inhibit the production of pro‐inflammatory cytokines (El Gazzar, 2007; El Gazzar, El Mezayen, Marecki, et al., 2006; El Gazzar, El Mezayen, Nicolls, et al., 2006) and the expression of inflammatory enzymes (such as cyclooxygenase‐2,5‐lipoxygenase and leukotriene C4 synthase, and thus ameliorates airway inflammation (Banerjee et al., 2009; El Mezayen et al., 2006; Mansour & Tornhamre, 2004). These evidences demonstrate the capacity of thymoquinone in attenuating inflammatory response that often follows SARS‐CoV‐2 infection. The antiinflammatory potentials of thymoquinone that we highlighted in this review, however, do not necessarily undermine that of other natural compounds, including curcumin, honokiol, zerumbone, escin, pinitol, and tocotrienol that can also be evaluated for their promising druggability against COVID‐19. Apart from various beneficial effects, thymoquinone also possesses some pharmacological drawbacks that limit its clinical use. Such disadvantages include hydrophobicity (Kazan, Yesil‐Celiktas, & Zhang, 2019), poor bioavailability (Kalam et al., 2017), lipophilicity, and heat and light sensitivity (Goyal et al., 2017). The modern drug delivery system may potentially minimize this limitation. Significant toxicological effects were not observed while administrated with thymoquinone at concentrations of 0.01%, 0.02%, and 0.03% for 90 days (30, 60, or 90 mg/kg/day) but administration of higher concentrations (2 and 3 g/kg/day) resulted in a low order acute oral toxicity of thymoquinone in mice (Badary, Al‐Shabanah, Nagi, Al‐Bekairi, & Elmazar, 1998). Although thymoquinone showed slight toxicity at a higher dose (10 mg/kg), (Alam & Galav, 2013), it is relatively well‐tolerated below this dose. Another component, α‐hederin also has come into view as novel therapeutic potential against viral diseases. Hopefully, inhibition of COVID‐19 by these two compounds (α‐hederin and nigellidine) of black cumin was reported in a molecular docking study, but further studies are warranted to evaluate the efficiency and explore the specific cellular and molecular mechanisms of their antiviral effects alone or in combination with other drugs.
6. CONCLUDING REMARKS
Where the whole world is hoping for an effective drug or vaccine to combat COVID‐19, we have tried to draw our attention to the pharmacological potentials of black cumin seed and its bioactive compounds for further scientific concern to adopt an alternative strategic plan. Black cumin seed and most its bioactive components already have a proven history of boosting up immune systems and can be easily accessible for all classes of people along with self‐awareness and ‐protection (Hannan, Islam, & Uddin, 2020) against COVID‐19. This review can offer some valuable information for the bonafide public domain or organization, where firstly, N. sativa seed and oil can be considered as a first‐aid kit as a preventive measure against COVID‐19; and secondly, the bioactive compounds, thymoquinone, α‐hederin, or nigellidine could be tested preclinically and clinically for drug development, efficacy, and potency under a specific pathophysiological condition in pursuit to control the deadliest pandemic.
CONFLICT OF INTEREST
No conflict of interest from authors regarding the publication of this manuscript.
AUTHOR CONTRIBUTIONS
This work was a collaboration among all the authors. MJU designed outlines and drafted the manuscript. MNI, KSH, PPS, JF, and MJU wrote the initial draft of the manuscript. MJU, MAH, MMR, and DTC reviewed the scientific contents described in the manuscript. All authors read and approved the final submitted version of the manuscript.
ACKNOWLEDGEMENTS
This work is acknowledged to the RP‐Grant 2020 of Ewha Womans University, Republic of Korea. MAH acknowledges postdoctoral support from Korea Research Fellowship (KRF) Program (2018H1D3A1A01074712) through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT, Republic of Korea.
Islam MN, Hossain KS, Sarker PP, et al. Revisiting pharmacological potentials of Nigella sativa seed: A promising option for COVID‐19 prevention and cure. Phytotherapy Research. 2021;35:1329–1344. 10.1002/ptr.6895
Funding information National Research Foundation of Korea, Grant/Award Number: 2018H1D3A1A01074712; RP‐Grant 2020 of Ewha Womans University, Republic of Korea
REFERENCES
- Abbas, A. T. , Abdel‐Aziz, M. M. , Zalata, K. R. , & Abd Al‐Galel Tel, D. (2005). Effect of dexamethasone and Nigella sativa on peripheral blood eosinophil count, IgG1 and IgG2a, cytokine profiles and lung inflammation in murine model of allergic asthma. The Egyptian Journal of Immunology, 12(1), 95–102. [PubMed] [Google Scholar]
- Abdallah, E. M. (2017). Black seed (Nigella sativa) as antimicrobial drug: A mini‐review. Novel Approaches in Drug Designing & Development, 3(2), 1–5. [Google Scholar]
- Abdel‐Sater, K. A. (2009). Gastroprotective effects of Nigella sativa oil on the formation of stress gastritis in hypothyroidal rats. International Journal of Physiology, Pathophysiology and Pharmacology, 1(2), 143–149. [PMC free article] [PubMed] [Google Scholar]
- Abdel Azeiz, A. Z. , Saad, A. H. , & Darweesh, M. F. (2013). Efficacy of thymoquinone against vaginal candidiasis in prednisolone‐induced immunosuppressed mice. Journal of American Science, 9(2013), 55–159. [Google Scholar]
- Abdelrazek, H. M. A. , Kilany, O. E. , Muhammad, M. A. A. , Tag, H. M. , & Abdelazim, A. M. (2018). Black seed thymoquinone improved insulin secretion, hepatic glycogen storage, and oxidative stress in streptozotocin‐induced diabetic male wistar rats. Oxidative Medicine and Cellular Longevity, 2018, 8104110–8104165. 10.1155/2018/8104165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmad, A. , Husain, A. , Mujeeb, M. , Khan, S. A. , Najmi, A. K. , Siddique, N. A. , … Anwar, F. (2013). A review on therapeutic potential of Nigella sativa: A miracle herb. Asian Pacific Journal of Tropical Biomedicine, 3(5), 337–352. 10.1016/S2221-1691(13)60075-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmad, S. , & Beg, Z. H. (2016). Evaluation of therapeutic effect of omega‐6 linoleic acid and thymoquinone enriched extracts from Nigella sativa oil in the mitigation of lipidemic oxidative stress in rats. Nutrition, 32(6), 649–655. 10.1016/j.nut.2015.12.003 [DOI] [PubMed] [Google Scholar]
- Al‐Naqeep, G. , Al‐Zubairi, A. S. , Ismail, M. , Amom, Z. H. , & Esa, N. M. (2011). Antiatherogenic potential of Nigella sativa seeds and oil in diet‐induced hypercholesterolemia in rabbits. Evidence‐based Complementary and Alternative Medicine, 2011, 213628–213628. 10.1093/ecam/neq071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alam, M. , & Galav, V. (2013). Subacute 28 days repeated toxicity assessment of thymoquinone (volatile oil of black seed) in Wistar rats. Indian Journal of Scientific Research, 4(1), 75–79. [Google Scholar]
- Alijotas‐Reig, J. , Esteve‐Valverde, E. , Belizna, C. , Selva‐O, Callaghan, A. , Pardos‐Gea, J. , Quintana, A. , … Miro‐Mur, F. (2020). Immunomodulatory therapy for the management of severe COVID‐19. Beyond the anti‐viral therapy: A comprehensive review. Autoimmunity Reviews, 19(7), 102569. 10.1016/j.autrev.2020.102569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aziz, N. , Son, Y. J. , & Cho, J. Y. (2018). Thymoquinone suppresses IRF‐3‐mediated expression of type I interferons via suppression of TBK1. International Journal of Molecular Sciences, 19(5), 1–13. 10.3390/ijms19051355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bacharier, L. B. , Guilbert, T. W. , Mauger, D. T. , Boehmer, S. , Beigelman, A. , Fitzpatrick, A. M. , … Martinez, F. D. (2015). Early administration of azithromycin and prevention of severe lower respiratory tract illnesses in preschool children with a history of such illnesses: A randomized clinical trial. JAMA, 314(19), 2034–2044. 10.1001/jama.2015.13896 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badary, O. A. , Al‐Shabanah, O. A. , Nagi, M. N. , Al‐Bekairi, A. M. , & Elmazar, M. M. A. (1998). Acute and subchronic toxicity of thymoquinone in mice. Drug Development Research, 44(2–3), 56–61. [DOI] [Google Scholar]
- Bamosa, A. , Kaatabi, H. , Badar, A. , Al‐Khadra, A. , Al Elq, A. , Abou‐Hozaifa, B. , … Al‐Almaie, S. (2015). Nigella sativa: A potential natural protective agent against cardiac dysfunction in patients with type 2 diabetes mellitus. Journal of Family and Community Medicine, 22(2), 88–95. 10.4103/2230-8229.155380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banerjee, S. , Kaseb, A. O. , Wang, Z. , Kong, D. , Mohammad, M. , Padhye, S. , … Mohammad, R. M. (2009). Antitumor activity of gemcitabine and oxaliplatin is augmented by thymoquinone in pancreatic cancer. Cancer Research, 69(13), 5575–5583. 10.1158/0008-5472.CAN-08-4235 [DOI] [PubMed] [Google Scholar]
- Banik, G. R. , Alqahtani, A. S. , Booy, R. , & Rashid, H. (2016). Risk factors for severity and mortality in patients with MERS‐CoV: Analysis of publicly available data from Saudi Arabia. Virologica Sinica, 31(1), 81–84. 10.1007/s12250-015-3679-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barakat, E. M. , El Wakeel, L. M. , & Hagag, R. S. (2013). Effects of Nigella sativa on outcome of hepatitis C in Egypt. World Journal of Gastroenterology, 19(16), 2529–2536. 10.3748/wjg.v19.i16.2529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belhadjer, Z. , Meot, M. , Bajolle, F. , Khraiche, D. , Legendre, A. , Abakka, S. , … Bonnet, D. (2020). Acute heart failure in multisystem inflammatory syndrome in children (MIS‐C) in the context of global SARS‐CoV‐2 pandemic. Circulation, 142, 429–436. 10.1161/CIRCULATIONAHA.120.048360 [DOI] [PubMed] [Google Scholar]
- Ben Addi, A. , Lefort, A. , Hua, X. , Libert, F. , Communi, D. , Ledent, C. , … Robaye, B. (2008). Modulation of murine dendritic cell function by adenine nucleotides and adenosine: Involvement of the A(2B) receptor. European Journal of Immunology, 38(6), 1610–1620. 10.1002/eji.200737781 [DOI] [PubMed] [Google Scholar]
- Blanco‐Melo, D. , Nilsson‐Payant, B. E. , Liu, W. C. , Uhl, S. , Hoagland, D. , Moller, R. , … tenOever, B. R. (2020). Imbalanced host response to SARS‐CoV‐2 drives development of COVID‐19. Cell, 181(5), 1036–1045 e1039. 10.1016/j.cell.2020.04.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonam, S. R. , Kaveri, S. V. , Sakuntabhai, A. , Gilardin, L. , & Bayry, J. (2020). Adjunct immunotherapies for the management of severely ill COVID‐19 patients. Cell Reports Medicine, 1(2), 100016. 10.1016/j.xcrm.2020.100016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boskabady, M. H. , Keyhanmanesh, R. , Khamneh, S. , & Ebrahimi, M. A. (2011). The effect of Nigella sativa extract on tracheal responsiveness and lung inflammation in ovalbumin‐sensitized Guinea pigs. Clinics (São Paulo, Brazil), 66(5), 879–887. 10.1590/s1807-59322011000500027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler, S. O. , Btaiche, I. F. , & Alaniz, C. (2005). Relationship between hyperglycemia and infection in critically ill patients. Pharmacotherapy, 25(7), 963–976. 10.1592/phco.2005.25.7.963 [DOI] [PubMed] [Google Scholar]
- Chaieb, K. , Kouidhi, B. , Jrah, H. , Mahdouani, K. , & Bakhrouf, A. (2011). Antibacterial activity of Thymoquinone, an active principle of Nigella sativa and its potency to prevent bacterial biofilm formation. BMC Complementary and Alternative Medicine, 11, 29. 10.1186/1472-6882-11-29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakravarty, N. (1993). Inhibition of histamine release from mast cells by nigellone. Annals of Allergy, 70(3), 237–242. [PubMed] [Google Scholar]
- Channappanavar, R. , & Perlman, S. (2017). Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Seminars in Immunopathology, 39(5), 529–539. 10.1007/s00281-017-0629-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, L. , Liu, H. G. , Liu, W. , Liu, J. , Liu, K. , Shang, J. , … Wei, S. (2020). Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia. Zhonghua Jie He He Hu Xi Za Zhi, 43(3), 203–208. 10.3760/cma.j.issn.1001-0939.2020.03.013 [DOI] [PubMed] [Google Scholar]
- Chen, N. , Zhou, M. , Dong, X. , Qu, J. , Gong, F. , Han, Y. , … Zhang, L. (2020). Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet, 395(10223), 507–513. 10.1016/S0140-6736(20)30211-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conti, P. , Ronconi, G. , Caraffa, A. , Gallenga, C. E. , Ross, R. , Frydas, I. , & Kritas, S. K. (2020). Induction of pro‐inflammatory cytokines (IL‐1 and IL‐6) and lung inflammation by Coronavirus‐19 (COVI‐19 or SARS‐CoV‐2): Anti‐inflammatory strategies. Journal of Biological Regulators and Homeostatic Agents, 34(2), 327–331. 10.23812/CONTI-E [DOI] [PubMed] [Google Scholar]
- Costela‐Ruiz, V. J. , Illescas‐Montes, R. , Puerta‐Puerta, J. M. , Ruiz, C. , & Melguizo‐Rodriguez, L. (2020). SARS‐CoV‐2 infection: The role of cytokines in COVID‐19 disease. Cytokine & Growth Factor Reviews, 54, 62–75. 10.1016/j.cytogfr.2020.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crotzer, V. L. , & Blum, J. S. (2010). Autophagy and adaptive immunity. Immunology, 131(1), 9–17. 10.1111/j.1365-2567.2010.03321.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curbelo, J. , Luquero Bueno, S. , Galvan‐Roman, J. M. , Ortega‐Gomez, M. , Rajas, O. , Fernandez‐Jimenez, G. , … Aspa, J. (2017). Inflammation biomarkers in blood as mortality predictors in community‐acquired pneumonia admitted patients: Importance of comparison with neutrophil count percentage or neutrophil‐lymphocyte ratio. PLoS One, 12(3), e0173947. 10.1371/journal.pone.0173947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daryabeygi‐Khotbehsara, R. , Golzarand, M. , Ghaffari, M. P. , & Djafarian, K. (2017). Nigella sativa improves glucose homeostasis and serum lipids in type 2 diabetes: A systematic review and meta‐analysis. Complementary Therapies in Medicine, 35, 6–13. 10.1016/j.ctim.2017.08.016 [DOI] [PubMed] [Google Scholar]
- Dong, X. , & Levine, B. (2013). Autophagy and viruses: Adversaries or allies? Journal of Innate Immunity, 5(5), 480–493. 10.1159/000346388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebrahimi, H. , Fallahi, M. , Khamaneh, A. M. , Ebrahimi Saadatlou, M. A. , Saadat, S. , & Keyhanmanesh, R. (2016). Effect of alpha‐Hederin on IL‐2 and IL‐17 mRNA and miRNA‐133a levels in lungs of ovalbumin‐sensitized male rats. Drug Development Research, 77(2), 87–93. 10.1002/ddr.21292 [DOI] [PubMed] [Google Scholar]
- El‐Kadi, A. , & Kandil, O. (1987). The black seed (Nigella sativa) and immunity: Its effect on human T cell subset. Federation Proceedings, 46(4), 1222. [Google Scholar]
- El Gazzar, M. , El Mezayen, R. , Marecki, J. C. , Nicolls, M. R. , Canastar, A. , & Dreskin, S. C. (2006). Anti‐inflammatory effect of thymoquinone in a mouse model of allergic lung inflammation. International Immunopharmacology, 6(7), 1135–1142. 10.1016/j.intimp.2006.02.004 [DOI] [PubMed] [Google Scholar]
- El Gazzar, M. , El Mezayen, R. , Nicolls, M. R. , Marecki, J. C. , & Dreskin, S. C. (2006). Downregulation of leukotriene biosynthesis by thymoquinone attenuates airway inflammation in a mouse model of allergic asthma. Biochimica et Biophysica Acta, 1760(7), 1088–1095. 10.1016/j.bbagen.2006.03.006 [DOI] [PubMed] [Google Scholar]
- El Gazzar, M. A. (2007). Thymoquinone suppressses in vitro production of IL‐5 and IL‐13 by mast cells in response to lipopolysaccharide stimulation. Inflammation Research, 56(8), 345–351. 10.1007/s00011-007-7051-0 [DOI] [PubMed] [Google Scholar]
- El Mezayen, R. , El Gazzar, M. , Nicolls, M. R. , Marecki, J. C. , Dreskin, S. C. , & Nomiyama, H. (2006). Effect of thymoquinone on cyclooxygenase expression and prostaglandin production in a mouse model of allergic airway inflammation. Immunology Letters, 106(1), 72–81. 10.1016/j.imlet.2006.04.012 [DOI] [PubMed] [Google Scholar]
- El Rabey, H. A. , Al‐Seeni, M. N. , & Bakhashwain, A. S. (2017). The antidiabetic activity of Nigella sativa and propolis on streptozotocin‐induced diabetes and diabetic nephropathy in male rats. Evidence‐based Complementary and Alternative Medicine, 2017, 5439614–5439645. 10.1155/2017/5439645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- El Tahir, K. E. , Ashour, M. M. , & al‐Harbi, M. M. (1993). The cardiovascular actions of the volatile oil of the black seed (Nigella sativa) in rats: Elucidation of the mechanism of action. General Pharmacology, 24(5), 1123–1131. 10.1016/0306-3623(93)90359-6 [DOI] [PubMed] [Google Scholar]
- Enomoto, S. , Asano, R. , Iwahori, Y. , Narui, T. , Okada, Y. , Singab, A. N. , & Okuyama, T. (2001). Hematological studies on black cumin oil from the seeds of Nigella sativa L. Biological & Pharmaceutical Bulletin, 24(3), 307–310. 10.1248/bpb.24.307 [DOI] [PubMed] [Google Scholar]
- Farooq, A. , Gulzar, M. , Hussain, M. A. , Gokhan, Z. , Khalid, M. A. , Ashraf, M. , & Gilani, A. (2016). Capparis spinosa L.: A plant with high potential for development of functional foods and nutraceuticals/pharmaceuticals. International Journal of Pharmacology, 12(3), 201–219. [Google Scholar]
- Forouzanfar, F. , Bazzaz, B. S. , & Hosseinzadeh, H. (2014). Black cumin (Nigella sativa) and its constituent (thymoquinone): A review on antimicrobial effects. Iranian Journal of Basic Medical Sciences, 17(12), 929–938. [PMC free article] [PubMed] [Google Scholar]
- Fowler Iii, A. A. , Kim, C. , Lepler, L. , Malhotra, R. , Debesa, O. , Natarajan, R. , … Kasirajan, V. (2017). Intravenous vitamin C as adjunctive therapy for enterovirus/rhinovirus induced acute respiratory distress syndrome. World Journal of Critical Care Medicine, 6(1), 85–90. 10.5492/wjccm.v6.i1.85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galeotti, C. , & Bayry, J. (2020). Autoimmune and inflammatory diseases following COVID‐19. Nature Reviews Rheumatology, 16(8), 413–414. 10.1038/s41584-020-0448-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gali‐Muhtasib, H. , Ocker, M. , Kuester, D. , Krueger, S. , El‐Hajj, Z. , Diestel, A. , … Schneider‐Stock, R. (2008). Thymoquinone reduces mouse colon tumor cell invasion and inhibits tumor growth in murine colon cancer models. Journal of Cellular and Molecular Medicine, 12(1), 330–342. 10.1111/j.1582-4934.2007.00095.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gassen, N. C. , Jan, P. , Thomas, B. , Frederik, D. , Jackson, E. , Katja, W. , … Marcel, A. M. (2020). Analysis of SARS‐CoV‐2‐controlled autophagy reveals spermidine, MK‐2206, and niclosamide as putative antiviral therapeutics. bioRxiv, 04(15), 997254. 10.1101/2020.04.15.997254 [DOI] [Google Scholar]
- Gassen, N. C. , Niemeyer, D. , Muth, D. , Corman, V. M. , Martinelli, S. , Gassen, A. , … Rein, T. (2019). SKP2 attenuates autophagy through Beclin1‐ubiquitination and its inhibition reduces MERS‐coronavirus infection. Nature Communications, 10(1), 5770. 10.1038/s41467-019-13659-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gautret, P. , Lagier, J. C. , Parola, P. , Hoang, V. T. , Meddeb, L. , Mailhe, M. , … Raoult, D. (2020). Hydroxychloroquine and azithromycin as a treatment of COVID‐19: Results of an open‐label non‐randomized clinical trial. International Journal of Antimicrobial Agents, 56, 105949. 10.1016/j.ijantimicag.2020.105949 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Gholamnezhad, Z. , Boskabady, M. H. , & Hosseini, M. (2014). Effect of Nigella sativa on immune response in treadmill exercised rat. BMC Complementary and Alternative Medicine, 14, 437. 10.1186/1472-6882-14-437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilani, A. H. , Aziz, N. , Khurram, I. M. , Chaudhary, K. S. , & Iqbal, A. (2001). Bronchodilator, spasmolytic and calcium antagonist activities of Nigella sativa seeds (Kalonji): A traditional herbal product with multiple medicinal uses. The Journal of the Pakistan Medical Association, 51(3), 115–120. [PubMed] [Google Scholar]
- Goyal, S. N. , Prajapati, C. P. , Gore, P. R. , Patil, C. R. , Mahajan, U. B. , Sharma, C. , … Ojha, S. K. (2017). Therapeutic potential and pharmaceutical development of thymoquinone: A multitargeted molecule of natural origin. Frontiers in Pharmacology, 8, 656. 10.3389/fphar.2017.00656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu, J. , Han, B. , & Wang, J. (2020). COVID‐19: Gastrointestinal manifestations and potential fecal‐oral transmission. Gastroenterology, 158(6), 1518–1519. 10.1053/j.gastro.2020.02.054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo, L.‐P. , Liu, S.‐X. , Yang, Q. , Liu, H.‐Y. , Xu, L.‐L. , Hao, Y.‐H. , & Zhang, X.‐Q. (2020). Effect of thymoquinone on acute kidney injury induced by sepsis in BALB/c mice. BioMed Research International, 2020(1594726), 1–7. 10.1155/2020/1594726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo, T. , Fan, Y. , Chen, M. , Wu, X. , Zhang, L. , He, T. , … Lu, Z. (2020). Effect of thymoquinone on acute kidney injury induced by sepsis in BALB/c mice. JAMA Cardiology, 5, 811–818. 10.1001/jamacardio.2020.1017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta, R. , Ghosh, A. , Singh, A. K. , & Misra, A. (2020). Clinical considerations for patients with diabetes in times of COVID‐19 epidemic. Diabetes and Metabolic Syndrome: Clinical Research and Reviews, 14(3), 211–212. 10.1016/j.dsx.2020.03.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadi, V. , Kheirouri, S. , Alizadeh, M. , Khabbazi, A. , & Hosseini, H. (2016). Effects of Nigella sativa oil extract on inflammatory cytokine response and oxidative stress status in patients with rheumatoid arthritis: A randomized, double‐blind, placebo‐controlled clinical trial. Avicenna Journal of Phytomedicine, 6(1), 34–43. [PMC free article] [PubMed] [Google Scholar]
- Halawani, E. (2009). Antibacterial activity of thymo‐quinone and thymohydroquinone of Nigella sativa L. and their interaction with some antibiotics. Advances in Biology Research, 3(2009), 148–152. [Google Scholar]
- Halyabar, O. , Chang, M. H. , Schoettler, M. L. , Schwartz, M. A. , Baris, E. H. , Benson, L. A. , … Henderson, L. A. (2019). Calm in the midst of cytokine storm: A collaborative approach to the diagnosis and treatment of hemophagocytic lymphohistiocytosis and macrophage activation syndrome. Pediatric Rheumatology Online Journal, 17(1), 7. 10.1186/s12969-019-0309-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hannan, M. A. , Islam, M. N. , & Uddin, M. J. (2020). Self‐confidence as an immune‐modifying psychotherapeutic intervention for COVID‐19 patients and understanding of its connection to CNS‐endocrine‐immune axis. Journal of Advanced Biotechnology and Experimental Therapeutics, 3(4), 14–17. 10.5455/jabet.2020.d151 [DOI] [Google Scholar]
- Hannan, M. A. , Rahman, M. A. , Rahman, M. S. , Sohag, A. A. M. , Dash, R. , Hossain, K. S. , … Uddin, M. J. (2020). Intermittent fasting, a possible priming tool for host defense against SARS‐CoV‐2 infection: Crosstalk among calorie restriction, autophagy and immune response. Immunology Letters, 226, 38–45. 10.1016/j.imlet.2020.07.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haq, A. , Abdullatif, M. , Lobo, P. I. , Khabar, K. S. , Sheth, K. V. , & al‐Sedairy, S. T. (1995). Nigella sativa: Effect on human lymphocytes and polymorphonuclear leukocyte phagocytic activity. Immunopharmacology, 30(2), 147–155. 10.1016/0162-3109(95)00016-m [DOI] [PubMed] [Google Scholar]
- Haq, A. , Lobo, P. I. , Al‐Tufail, M. , Rama, N. R. , & Al‐Sedairy, S. T. (1999). Immunomodulatory effect of Nigella sativa proteins fractionated by ion exchange chromatography. International Journal of Immunopharmacology, 21(4), 283–295. 10.1016/s0192-0561(99)00010-7 [DOI] [PubMed] [Google Scholar]
- Hasan, N. A. , Nawahwi, M. Z. , & Malek, H. A. (2013). Antimicrobial activity of Nigella sativa seed extract. Sains Malaysiana, 42(2), 114–143. [Google Scholar]
- Haseena, S. , Aithal, M. , Das, K. K. , & Saheb, S. H. (2015). Phytochemical analysis of Nigella sativa and its effect on reproductive system. Journal of Pharmaceutical Sciences and Research, 7(8), 514–517. [Google Scholar]
- Hassanien, M. F. , Assiri, A. M. , Alzohairy, A. M. , & Oraby, H. F. (2015). Health‐promoting value and food applications of black cumin essential oil: An overview. Journal of Food Science and Technology, 52(10), 6136–6142. 10.1007/s13197-015-1785-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hecker, L. (2018). Mechanisms and consequences of oxidative stress in lung disease: Therapeutic implications for an aging populace. American Journal of Physiology. Lung Cellular and Molecular Physiology, 314(4), L642–L653. 10.1152/ajplung.00275.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helms, J. , Kremer, S. , Merdji, H. , Clere‐Jehl, R. , Schenck, M. , Kummerlen, C. , … Meziani, F. (2020). Neurologic features in severe SARS‐CoV‐2 infection. The New England Journal of Medicine, 382(23), 2268–2270. 10.1056/NEJMc2008597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson, L. A. , Canna, S. W. , Schulert, G. S. , Volpi, S. , Lee, P. Y. , Kernan, K. F. , … Nigrovic, P. A. (2020). On the alert for cytokine storm: Immunopathology in COVID‐19. Arthritis & Rhematology, 72(7), 1059–1063. 10.1002/art.41285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosseinzadeh, H. , & Parvardeh, S. (2004). Anticonvulsant effects of thymoquinone, the major constituent of Nigella sativa seeds, in mice. Phytomedicine, 11(1), 56–64. 10.1078/0944-7113-00376 [DOI] [PubMed] [Google Scholar]
- Hosseinzadeh, H. , Taiari, S. , & Nassiri‐Asl, M. (2012). Effect of thymoquinone, a constituent of Nigella sativa L., on ischemia‐reperfusion in rat skeletal muscle. Naunyn‐Schmiedeberg's Archives of Pharmacology, 385(5), 503–508. 10.1007/s00210-012-0726-2 [DOI] [PubMed] [Google Scholar]
- Houghton, P. J. , Zarka, R. , de las Heras, B. , & Hoult, J. R. (1995). Fixed oil of Nigella sativa and derived thymoquinone inhibit eicosanoid generation in leukocytes and membrane lipid peroxidation. Planta Medica, 61(1), 33–36. 10.1055/s-2006-957994 [DOI] [PubMed] [Google Scholar]
- Huang, C. , Wang, Y. , Li, X. , Ren, L. , Zhao, J. , Hu, Y. , … Cao, B. (2020). Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet, 395(10223), 497–506. 10.1016/S0140-6736(20)30183-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurwitz, J. L. (2020). B cells, viruses, and the SARS‐CoV‐2/COVID‐19 pandemic of 2020. Viral Immunology, 33(4), 251–252. 10.1089/vim.2020.0055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikhsan, M. , Hiedayati, N. , Maeyama, K. , & Nurwidya, F. (2018). Nigella sativa as an anti‐inflammatory agent in asthma. BMC Research Notes, 11(1), 744. 10.1186/s13104-018-3858-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivanov, A. V. , Bartosch, B. , & Isaguliants, M. G. (2017). Oxidative stress in infection and consequent disease. Oxidative Medicine and Cellular Longevity, 2017, 3496043–3496043. 10.1155/2017/3496043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacoby, D. B. , & Choi, A. M. (1994). Influenza virus induces expression of antioxidant genes in human epithelial cells. Free Radical Biology & Medicine, 16(6), 821–824. 10.1016/0891-5849(94)90198-8 [DOI] [PubMed] [Google Scholar]
- Jeon, J. H. , & Lee, C. (2018). Cholesterol is important for the entry process of porcine deltacoronavirus. Archives of Virology, 163(11), 3119–3124. 10.1007/s00705-018-3967-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaatabi, H. , Bamosa, A. O. , Badar, A. , Al‐Elq, A. , Abou‐Hozaifa, B. , Lebda, F. , … Al‐Almaie, S. (2015). Nigella sativa improves glycemic control and ameliorates oxidative stress in patients with type 2 diabetes mellitus: Placebo controlled participant blinded clinical trial. PLoS One, 10(2), e0113486. 10.1371/journal.pone.0113486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalam, M. A. , Raish, M. , Ahmed, A. , Alkharfy, K. M. , Mohsin, K. , Alshamsan, A. , … Shakeel, F. (2017). Oral bioavailability enhancement and hepatoprotective effects of thymoquinone by self‐nanoemulsifying drug delivery system. Materials Science & Engineering. C, Materials for Biological Applications, 76, 319–329. 10.1016/j.msec.2017.03.088 [DOI] [PubMed] [Google Scholar]
- Kanter, M. , Demir, H. , Karakaya, C. , & Ozbek, H. (2005). Gastroprotective activity of Nigella sativa L oil and its constituent, thymoquinone against acute alcohol‐induced gastric mucosal injury in rats. World Journal of Gastroenterology, 11(42), 6662–6666. 10.3748/wjg.v11.i42.6662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaur, G. , Invally, M. , Khan, M. K. , & Jadhav, P. (2018). A nutraceutical combination of Cinnamomum cassia & Nigella sativa for Type 1 diabetes mellitus. Journal of Ayurveda and Integrative Medicine, 9(1), 27–37. 10.1016/j.jaim.2017.02.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kazan, A. , Yesil‐Celiktas, O. , & Zhang, Y. S. (2019). Fabrication of thymoquinone‐loaded albumin nanoparticles by microfluidic particle synthesis and their effect on planarian regeneration. Macromolecular Bioscience, 19(11), e1900182. 10.1002/mabi.201900182 [DOI] [PubMed] [Google Scholar]
- Keyaerts, E. , Vijgen, L. , Maes, P. , Neyts, J. , & Van Ranst, M. (2004). In vitro inhibition of severe acute respiratory syndrome coronavirus by chloroquine. Biochemical and Biophysical Research Communications, 323(1), 264–268. 10.1016/j.bbrc.2004.08.085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyhanmanesh, R. , Bagban, H. , Nazemieh, H. , Mirzaei Bavil, F. , & Alipour, M. R. (2013). The main relaxant constituents of Nigella sativa methanolic fraction on Guinea pig tracheal chains. Iranian Journal of Allergy, Asthma, and Immunology, 12(2), 136–143. [PubMed] [Google Scholar]
- Kiralan, M. , Özkan, G. , Bayrak, A. , & Ramadan, M. F. (2014). Physicochemical properties and stability of black cumin (Nigella sativa) seed oil as affected by different extraction methods. Industrial Crops and Products, 57, 52–58. [Google Scholar]
- Klionsky, D. J. , Abdelmohsen, K. , Abe, A. , Abedin, M. J. , Abeliovich, H. , Acevedo Arozena, A. , … Zughaier, S. M. (2016). Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy, 12(1), 1–222. 10.1080/15548627.2015.1100356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koshak, A. , Koshak, E. , & Heinrich, M. (2017). Medicinal benefits of Nigella sativa in bronchial asthma: A literature review. Saudi Pharmaceutical Journal, 25(8), 1130–1136. 10.1016/j.jsps.2017.07.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koshak, A. E. , Yousif, N. M. , Fiebich, B. L. , Koshak, E. A. , & Heinrich, M. (2018). Comparative immunomodulatory activity of Nigella sativa L. preparations on proinflammatory mediators: A focus on asthma. Frontiers in Pharmacology, 9, 1075. 10.3389/fphar.2018.01075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhn, J. H. , Li, W. , Choe, H. , & Farzan, M. (2004). Angiotensin‐converting enzyme 2: A functional receptor for SARS coronavirus. Cellular and Molecular Life Sciences, 61(21), 2738–2743. 10.1007/s00018-004-4242-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagier, J. C. , & Raoult, D. (2018). Whipple's disease and Tropheryma whipplei infections: When to suspect them and how to diagnose and treat them. Current Opinion in Infectious Diseases, 31(6), 463–470. 10.1097/QCO.0000000000000489 [DOI] [PubMed] [Google Scholar]
- Le Gallou, S. , Caron, G. , Delaloy, C. , Rossille, D. , Tarte, K. , & Fest, T. (2012). IL‐2 requirement for human plasma cell generation: Coupling differentiation and proliferation by enhancing MAPK‐ERK signaling. Journal of Immunology, 189(1), 161–173. 10.4049/jimmunol.1200301 [DOI] [PubMed] [Google Scholar]
- Li, G. , Fan, Y. , Lai, Y. , Han, T. , Li, Z. , Zhou, P. , … Wu, J. (2020). Coronavirus infections and immune responses. Journal of Medical Virology, 92(4), 424–432. 10.1002/jmv.25685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, X. , Geng, M. , Peng, Y. , Meng, L. , & Lu, S. (2020). Molecular immune pathogenesis and diagnosis of COVID‐19. Journal of Pharmaceutical Analysis, 10(2), 102–108. 10.1016/j.jpha.2020.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, X. , Shen, Y. , Wang, H. , Ge, Q. , Fei, A. , & Pan, S. (2016). Prognostic significance of neutrophil‐to‐lymphocyte ratio in patients with sepsis: A prospective observational study. Mediators of Inflammation, 2016, 8191254–8191258. 10.1155/2016/8191254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loffredo, L. , Martino, F. , Zicari, A. M. , Carnevale, R. , Battaglia, S. , Martino, E. , … Violi, F. (2019). Enhanced NOX‐2 derived oxidative stress in offspring of patients with early myocardial infarction. International Journal of Cardiology, 293, 56–59. 10.1016/j.ijcard.2019.05.014 [DOI] [PubMed] [Google Scholar]
- Low, D. (2008). Reducing antibiotic use in influenza: Challenges and rewards. Clinical Microbiology and Infection, 14(4), 298–306. 10.1111/j.1469-0691.2007.01910.x [DOI] [PubMed] [Google Scholar]
- Lupoli, F. , Vannocci, T. , Longo, G. , Niccolai, N. , & Pastore, A. (2018). The role of oxidative stress in Friedreich's ataxia. FEBS Letters, 592(5), 718–727. 10.1002/1873-3468.12928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacIntyre, C. R. , Chughtai, A. A. , Barnes, M. , Ridda, I. , Seale, H. , Toms, R. , & Heywood, A. (2018). The role of pneumonia and secondary bacterial infection in fatal and serious outcomes of pandemic influenza a(H1N1)pdm09. BMC Infectious Diseases, 18(1), 637. 10.1186/s12879-018-3548-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madjid, M. , Safavi‐Naeini, P. , Solomon, S. D. , & Vardeny, O. (2020). Potential effects of coronaviruses on the cardiovascular system: A review. JAMA Cardiology, 5, 831–840. 10.1001/jamacardio.2020.1286 [DOI] [PubMed] [Google Scholar]
- Madrid, P. B. , Panchal, R. G. , Warren, T. K. , Shurtleff, A. C. , Endsley, A. N. , Green, C. E. , … Tanga, M. J. (2015). Evaluation of ebola virus inhibitors for drug repurposing. ACS Infectious Diseases, 1(7), 317–326. 10.1021/acsinfecdis.5b00030 [DOI] [PubMed] [Google Scholar]
- Mahase, E. (2020). Covid‐19: What treatments are being investigated? BMJ, 368, m1252. 10.1136/bmj.m1252 [DOI] [PubMed] [Google Scholar]
- Mansour, M. , & Tornhamre, S. (2004). Inhibition of 5‐lipoxygenase and leukotriene C4 synthase in human blood cells by thymoquinone. Journal of Enzyme Inhibition and Medicinal Chemistry, 19(5), 431–436. 10.1080/14756360400002072 [DOI] [PubMed] [Google Scholar]
- Marozzi, F. J., Jr. , Kocialski, A. B. , & Malone, M. H. (1970). Studies on the antihistaminic effects of thymoquinone, thymohydroquinone and quercetin. Arzneimittel‐Forschung, 20(10), 1574–1577. [PubMed] [Google Scholar]
- Maryam, A. J. , Fatimah, A. A. , Ebtesam, A. K. , Abdulrahman, A. S. , & Ineta, B. E. (2016). In‐vitro studies on the effect of Nigella sativa Linn., seed oil extract on multidrug resistant Gram positive and Gram negative bacteria. Journal of Medicinal Plants, 4(2), 195–199. [Google Scholar]
- Mehta, P. , McAuley, D. F. , Brown, M. , Sanchez, E. , Tattersall, R. S. , Manson, J. J. , & Hlh Across Speciality Collaboration, UK . (2020). COVID‐19: Consider cytokine storm syndromes and immunosuppression. Lancet, 395(10229), 1033–1034. 10.1016/S0140-6736(20)30628-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng, L. , Zhao, X. , & Zhang, H. (2019). HIPK1 interference attenuates inflammation and oxidative stress of acute lung injury via autophagy. Medical Science Monitor, 25, 827–835. 10.12659/MSM.912507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moens, L. , & Tangye, S. G. (2014). Cytokine‐mediated regulation of plasma cell generation: IL‐21 takes center stage. Frontiers in Immunology, 5, 65. 10.3389/fimmu.2014.00065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mostafa, R. M. , Moustafa, Y. M. , Mirghani, Z. , AlKusayer, G. M. , & Moustafa, K. M. (2013). Antioxidant effect of garlic (Allium sativum) and black seeds (Nigella sativa) in healthy postmenopausal women. SAGE Open Medicine, 1, 2050312113517501. 10.1177/2050312113517501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mouwakeh, A. , Telbisz, A. , Spengler, G. , Mohacsi‐Farkas, C. , & Kisko, G. (2018). Antibacterial and resistance modifying activities of Nigella sativa essential oil and its active compounds against Listeria monocytogenes . In Vivo, 32(4), 737–743. 10.21873/invivo.11302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nader, M. A. , el‐Agamy, D. S. , & Suddek, G. M. (2010). Protective effects of propolis and thymoquinone on development of atherosclerosis in cholesterol‐fed rabbits. Archives of Pharmacal Research, 33(4), 637–643. 10.1007/s12272-010-0420-1 [DOI] [PubMed] [Google Scholar]
- Namazi, N. , Mahdavi, R. , Alizadeh, M. , & Farajnia, S. (2015). Oxidative stress responses to Nigella sativa oil concurrent with a low‐calorie diet in obese women: A randomized, double‐blind controlled clinical trial. Phytotherapy Research, 29(11), 1722–1728. 10.1002/ptr.5417 [DOI] [PubMed] [Google Scholar]
- Note, E. S. (2018). Targeting nuclear factor‐kappab activation pathway by thymoquinone: Role in suppression of antiapoptotic gene products and enhancement of apoptosis. Molecular Cancer Research, 16(9), 1441. 10.1158/1541-7786.MCR-18-0820 [DOI] [PubMed] [Google Scholar]
- Ojha, S. , Azimullah, S. , Mohanraj, R. , Sharma, C. , Yasin, J. , Arya, D. S. , & Adem, A. (2015). Thymoquinone protects against myocardial ischemic injury by mitigating oxidative stress and inflammation. Evidence‐based Complementary and Alternative Medicine, 2015(143629), 1–12. 10.1155/2015/143629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omidi, H. , Khorram, S. , Mesgari, M. , Asghari‐Jafarabadi, M. , & Tarighat‐Esfanjani, A. (2017). Effects of separate and concurrent supplementation of Nano‐sized clinoptilolite and Nigella sativa on oxidative stress, anti‐oxidative parameters and body weight in rats with type 2 diabetes. Biomedicine & Pharmacotherapy, 96, 1335–1340. 10.1016/j.biopha.2017.11.077 [DOI] [PubMed] [Google Scholar]
- Onifade, A. A. , Jewell, A. P. , & Adedeji, W. A. (2013). Nigella sativa concoction induced sustained seroreversion in HIV patient. African Journal of Traditional, Complementary, and Alternative Medicines, 10(5), 332–335. [PMC free article] [PubMed] [Google Scholar]
- Oskouei, Z. , Akaberi, M. , & Hosseinzadeh, H. (2018). A glance at black cumin (Nigella sativa) and its active constituent, thymoquinone, in ischemia: A review. Iranian Journal of Basic Medical Sciences, 21(12), 1200–1209. 10.22038/ijbms.2018.31703.7630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozdemir, N. , Kantekin‐Erdogan, M. N. , Tat, T. , & Tekin, A. (2018). Effect of black cumin oil on the oxidative stability and sensory characteristics of mayonnaise. Journal of Food Science and Technology, 55(4), 1562–1568. 10.1007/s13197-018-3075-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan, H. M. , Chen, L. X. , Xu, Y. H. , Han, W. D. , Lou, F. , Fei, W. Q. , … Sui, X. B. (2016). Autophagy‐associated immune responses and cancer immunotherapy. Oncotarget, 7(16), 21235–21246. 10.18632/oncotarget.6908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pandiri, I. , & Moni, A. (2018). Ocimum herb species: A potential treatment strategy for diabetic kidney disease. Journal of Advanced Biotechnology and Experimental Therapeutics, 1(3), 88–91. [Google Scholar]
- Perrone, L. A. , Belser, J. A. , Wadford, D. A. , Katz, J. M. , & Tumpey, T. M. (2013). Inducible nitric oxide contributes to viral pathogenesis following highly pathogenic influenza virus infection in mice. The Journal of Infectious Diseases, 207(10), 1576–1584. 10.1093/infdis/jit062 [DOI] [PubMed] [Google Scholar]
- Peterhans, E. (1997). Oxidants and antioxidants in viral diseases: Disease mechanisms and metabolic regulation. The Journal of Nutrition, 127(5 Suppl), 962S–965S. 10.1093/jn/127.5.962S [DOI] [PubMed] [Google Scholar]
- Peterhans, E. , Grob, M. , Burge, T. , & Zanoni, R. (1987). Virus‐induced formation of reactive oxygen intermediates in phagocytic cells. Free Radical Research Communications, 3(1–5), 39–46. 10.3109/10715768709069768 [DOI] [PubMed] [Google Scholar]
- Pouletty, M. , Borocco, C. , Ouldali, N. , Caseris, M. , Basmaci, R. , Lachaume, N. , … Melki, I. (2020). Paediatric multisystem inflammatory syndrome temporally associated with SARS‐CoV‐2 mimicking Kawasaki disease (Kawa‐COVID‐19): A multicentre cohort. Annals of the Rheumatic Diseases, 79(8), 999–1006. 10.1136/annrheumdis-2020-217960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman, I. , Biswas, S. K. , & Kirkham, P. A. (2006). Regulation of inflammation and redox signaling by dietary polyphenols. Biochemical Pharmacology, 72(11), 1439–1452. 10.1016/j.bcp.2006.07.004 [DOI] [PubMed] [Google Scholar]
- Ralph, R. , Lew, J. , Zeng, T. , Francis, M. , Xue, B. , Roux, M. , … Kelvin, A. A. (2020). 2019‐nCoV (Wuhan virus), a novel coronavirus: Human‐to‐human transmission, travel‐related cases, and vaccine readiness. Journal of Infection in Developing Countries, 14(1), 3–17. 10.3855/jidc.12425 [DOI] [PubMed] [Google Scholar]
- Ramadan, M. F. (2007). Nutritional value, functional properties and nutraceutical applications of black cumin (Nigella sativa L.): An overview. International Journal of Food Science & Technology, 42(10), 1208–1218. 10.1111/j.1365-2621.2006.01417.x [DOI] [Google Scholar]
- Ramadan, M. F. , Asker, M. M. S. , & Tadros, M. (2012). Antiradical and antimicrobial properties of cold‐pressed black cumin and cumin oils. European Food Research and Technology, 234, 833–844. [Google Scholar]
- Ramadan, M. F. , & Mörsel, J. T. (2003). Analysis of glycolipids from black cumin (Nigella sativa L.), coriander (Coriandrum sativum L.) and Niger (Guizotia abyssinica Cass.) oilseeds. Food Chemistry, 80(2), 197–204. [DOI] [PubMed] [Google Scholar]
- Retallack, H. , Di Lullo, E. , Arias, C. , Knopp, K. A. , Laurie, M. T. , Sandoval‐Espinosa, C. , … DeRisi, J. L. (2016). Zika virus cell tropism in the developing human brain and inhibition by azithromycin. Proceedings of the National Academy of Sciences of the United States of America, 113(50), 14408–14413. 10.1073/pnas.1618029113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riphagen, S. , Gomez, X. , Gonzalez‐Martinez, C. , Wilkinson, N. , & Theocharis, P. (2020). Hyperinflammatory shock in children during COVID‐19 pandemic. Lancet, 395(10237), 1607–1608. 10.1016/S0140-6736(20)31094-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Risitano, A. M. , Mastellos, D. C. , Huber‐Lang, M. , Yancopoulou, D. , Garlanda, C. , Ciceri, F. , & Lambris, J. D. (2020). Complement as a target in COVID‐19? Nature Reviews. Immunology, 20(6), 343–344. 10.1038/s41577-020-0320-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolain, J. M. , Colson, P. , & Raoult, D. (2007). Recycling of chloroquine and its hydroxyl analogue to face bacterial, fungal and viral infections in the 21st century. International Journal of Antimicrobial Agents, 30(4), 297–308. 10.1016/j.ijantimicag.2007.05.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan, Q. , Yang, K. , Wang, W. , Jiang, L. , & Song, J. (2020). Clinical predictors of mortality due to COVID‐19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Medicine, 46, 846–848. 10.1007/s00134-020-05991-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saadat, S. , Mohammadi, M. , Fallahi, M. , Keyhanmanesh, R. , & Aslani, M. R. (2015). The protective effect of alpha‐hederin, the active constituent of Nigella sativa, on tracheal responsiveness and lung inflammation in ovalbumin‐sensitized Guinea pigs. The Journal of Physiological Sciences, 65(3), 285–292. 10.1007/s12576-015-0367-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salem, M. L. , & Hossain, M. S. (2000a). In vivo acute depletion of CD8(+) T cells before murine cytomegalovirus infection upregulated innate antiviral activity of natural killer cells. International Journal of Immunopharmacology, 22(9), 707–718. 10.1016/s0192-0561(00)00033-3 [DOI] [PubMed] [Google Scholar]
- Salem, M. L. , & Hossain, M. S. (2000b). Protective effect of black seed oil from Nigella sativa against murine cytomegalovirus infection. International Journal of Immunopharmacology, 22(9), 729–740. 10.1016/s0192-0561(00)00036-9 [DOI] [PubMed] [Google Scholar]
- Salim, B. , & Noureddine, M. (2020). Identification of compounds from Nigella sativa as new potential inhibitors of 2019 novel coronasvirus (COVID‐19): Molecular docking study. ChemRxiv, 03(31), 1–12. 10.26434/chemrxiv.12055716.v1 [DOI] [Google Scholar]
- Savarino, A. , Di Trani, L. , Donatelli, I. , Cauda, R. , & Cassone, A. (2006). New insights into the antiviral effects of chloroquine. The Lancet Infectious Diseases, 6(2), 67–69. 10.1016/S1473-3099(06)70361-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoen, K. , Horvat, N. , Guerreiro, N. F. C. , de Castro, I. , & de Giassi, K. S. (2019). Spectrum of clinical and radiographic findings in patients with diagnosis of H1N1 and correlation with clinical severity. BMC Infectious Diseases, 19(1), 964. 10.1186/s12879-019-4592-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahabi, F. , Mosaddeghi, P. , Monica, N. , Zahra, D. , Mitra, F. , Mohsen, M. , … Seyed, M. M. (2020). Therapeutic approaches for COVID‐19 based on the dynamics of interferon‐mediated immune responses. Preprint(2020030206), 1–26. 10.20944/preprints202003.0206.v1 [DOI] [Google Scholar]
- Shahid, F. , Farooqui, Z. , Khan, A. A. , & Khan, F. (2018). Oral Nigella sativa oil and thymoquinone administration ameliorates the effect of long‐term cisplatin treatment on the enzymes of carbohydrate metabolism, brush border membrane, and antioxidant defense in rat intestine. Naunyn‐Schmiedeberg's Archives of Pharmacology, 391(2), 145–157. 10.1007/s00210-017-1444-6 [DOI] [PubMed] [Google Scholar]
- Shojaei, S. , Koleini, N. , Samiei, E. , Aghaei, M. , Cole, L. K. , Alizadeh, J. , … Ghavami, S. (2020). Simvastatin increases temozolomide‐induced cell death by targeting the fusion of autophagosomes and lysosomes. The FEBS Journal, 287(5), 1005–1034. 10.1111/febs.15069 [DOI] [PubMed] [Google Scholar]
- Shojaei, S. , Suresh, M. , Klionsky, D. J. , Labouta, H. I. , & Ghavami, S. (2020). Autophagy and SARS‐CoV‐2 infection: A possible smart targeting of the autophagy pathway. Virulence, 11(1), 805–810. 10.1080/21505594.2020.1780088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sohag, A. A. M. , Hannan, M. A. , Rahman, S. , Hossain, M. , Hasan, M. , Khan, M. K. , … Uddin, M. J. (2020). Revisiting potential druggable targets against SARS‐CoV‐2 and repurposing therapeutics under preclinical study and clinical trials: A comprehensive review. Drug Development Research, 1–23. 10.1002/ddr.21709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sohaib Ashraf, S. Z. (2020). Efficacy of Nigella sativa and natural honey against COVID‐19 (WUHAN). ClinicalTrials.gov Identifier: NCT04347382. Retrieved from https://clinicaltrials.gov/ct2/show/NCT04347382
- Sohn, M. , Kim, K. , Uddin, M. J. , Lee, G. , Hwang, I. , Kang, H. , … Ha, H. (2017). Delayed treatment with fenofibrate protects against high‐fat diet‐induced kidney injury in mice: The possible role of AMPK autophagy. American Journal of Physiology. Renal Physiology, 312(2), F323–F334. 10.1152/ajprenal.00596.2015 [DOI] [PubMed] [Google Scholar]
- Sultan, M. T. , Butt, M. S. , Karim, R. , Ahmed, W. , Kaka, U. , Ahmad, S. , … Zia‐Ul‐Haq, M. (2015). Nigella sativa fixed and essential oil modulates glutathione redox enzymes in potassium bromate induced oxidative stress. BMC Complementary and Alternative Medicine, 15, 330. 10.1186/s12906-015-0853-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toubiana, J. , Poirault, C. , Corsia, A. , Bajolle, F. , Fourgeaud, J. , Angoulvant, F. , … Allali, S. (2020). Kawasaki‐like multisystem inflammatory syndrome in children during the covid‐19 pandemic in Paris, France: Prospective observational study. BMJ, 369, m2094. 10.1136/bmj.m2094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulasli, M. , Gurses, S. A. , Bayraktar, R. , Yumrutas, O. , Oztuzcu, S. , Igci, M. , … Arslan, A. (2014). The effects of Nigella sativa (Ns), Anthemis hyalina (Ah) and Citrus sinensis (Cs) extracts on the replication of coronavirus and the expression of TRP genes family. Molecular Biology Reports, 41(3), 1703–1711. 10.1007/s11033-014-3019-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umar, S. , Shah, M. A. , Munir, M. T. , Yaqoob, M. , Fiaz, M. , Anjum, S. , … Umar, W. (2016). Synergistic effects of thymoquinone and curcumin on immune response and anti‐viral activity against avian influenza virus (H9N2) in turkeys. Poultry Science, 95(7), 1513–1520. 10.3382/ps/pew069 [DOI] [PubMed] [Google Scholar]
- Verdoni, L. , Mazza, A. , Gervasoni, A. , Martelli, L. , Ruggeri, M. , Ciuffreda, M. , … Antiga, D. L. (2020). An outbreak of severe Kawasaki‐like disease at the Italian epicentre of the SARS‐CoV‐2 epidemic: An observational cohort study. Lancet, 395(10239), 1771–1778. 10.1016/S0140-6736(20)31103-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waltuch, T. , Gill, P. , Zinns, L. E. , Whitney, R. , Tokarski, J. , Tsung, J. W. , & Sanders, J. E. (2020). Features of COVID‐19 post‐infectious cytokine release syndrome in children presenting to the emergency department. The American Journal of Emergency Medicine, S0735‐6757(20), 30403–30404. 10.1016/j.ajem.2020.05.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, D. , Hu, B. , Hu, C. , Zhu, F. , Liu, X. , Zhang, J. , … Peng, Z. (2020). Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus‐infected pneumonia in Wuhan, China. Journal of the American Medical Association, 323, 1061–1069. 10.1001/jama.2020.1585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, W. , Gong, F. , Zhu, W. , Fu, S. , & Zhang, Q. (2015). Macrophage activation syndrome in Kawasaki disease: More common than we thought? Seminars in Arthritis and Rheumatism, 44(4), 405–410. 10.1016/j.semarthrit.2014.07.007 [DOI] [PubMed] [Google Scholar]
- Whittaker, E. , Bamford, A. , Kenny, J. , Kaforou, M. , Jones, C. E. , Shah, P. , … Consortia, P. (2020). Clinical characteristics of 58 children with a pediatric inflammatory multisystem syndrome temporally associated with SARS‐CoV‐2. Journal of the American Medical Association, 324, 259–269. 10.1001/jama.2020.10369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO . (2020a). Coronavirus disease 2019 (COVID‐19) situation report – 197, August 4, 2020.
- WHO . (2020b). WHO Director‐General's opening remarks at the media briefing on COVID‐19, March 11, 2020
- Wu, D. , & Yang, X. O. (2020). TH17 responses in cytokine storm of COVID‐19: An emerging target of JAK2 inhibitor Fedratinib. Journal of Microbiology, Immunology, and Infection, 53(3), 368–370. 10.1016/j.jmii.2020.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao, J. , Ke, Z. P. , Shi, Y. , Zeng, Q. , & Cao, Z. (2018). The cardioprotective effect of thymoquinone on ischemia‐reperfusion injury in isolated rat heart via regulation of apoptosis and autophagy. Journal of Cellular Biochemistry, 119(9), 7212–7217. 10.1002/jcb.26878 [DOI] [PubMed] [Google Scholar]
- Xing, Q. , Li, G. , Xing, Y. , Chen, T. , Li, W. , Ni, W. , … Pan, S. (2020). Precautions are needed for COVID‐19 patients with coinfection of common respiratory pathogens. medRxiv, 20027698(03), 1–22. 10.1101/February29,2020.20027698 [DOI] [Google Scholar]
- Xu, X. , Chen, P. , Wang, J. , Feng, J. , Zhou, H. , Li, X. , … Hao, P. (2020). Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Science China. Life Sciences, 63(3), 457–460. 10.1007/s11427-020-1637-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yahfoufi, N. , Alsadi, N. , Jambi, M. , & Matar, C. (2018). The immunomodulatory and anti‐inflammatory role of polyphenols. Nutrients, 10(11), 1618. 10.3390/nu10111618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan, X. , Fu, X. , Jia, Y. , Ma, X. , Tao, J. , Yang, T. , … Wei, J. (2019). Nrf2/Keap1/ARE signaling mediated an antioxidative protection of human placental mesenchymal stem cells of fetal origin in alveolar epithelial cells. Oxidative Medicine and Cellular Longevity, 2019, 2654910–2654912. 10.1155/2019/2654910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, J. K. , Feng, Y. , Yuan, M. Y. , Yuan, S. Y. , Fu, H. J. , Wu, B. Y. , … Chan, J. C. (2006). Plasma glucose levels and diabetes are independent predictors for mortality and morbidity in patients with SARS. Diabetic Medicine, 23(6), 623–628. 10.1111/j.1464-5491.2006.01861.x [DOI] [PubMed] [Google Scholar]
- Yang, J. K. , Lin, S. S. , Ji, X. J. , & Guo, L. M. (2010). Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetologica, 47(3), 193–199. 10.1007/s00592-009-0109-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, X. , Yu, Y. , Xu, J. , Shu, H. , Xia, J. , Liu, H. , … Shang, Y. (2020). Clinical course and outcomes of critically ill patients with SARS‐CoV‐2 pneumonia in Wuhan, China: A single‐centered, retrospective, observational study. The Lancet Respiratory Medicine, 8, 475–481. 10.1016/S2213-2600(20)30079-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi, Y. , Lagniton, P. N. P. , Ye, S. , Li, E. , & Xu, R. H. (2020). COVID‐19: What has been learned and to be learned about the novel coronavirus disease. International Journal of Biological Sciences, 16(10), 1753–1766. 10.7150/ijbs.45134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zakaria, A. , Jais, M. R. , & Ishak, R. (2018). Analgesic properties of Nigella sativa and Eucheuma cottonii extracts. Journal of Natural Science, Biology and Medicine, 9(1), 23–26. 10.4103/jnsbm.JNSBM_131_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaoui, A. , Cherrah, Y. , Alaoui, K. , Mahassine, N. , Amarouch, H. , & Hassar, M. (2002). Effects of Nigella sativa fixed oil on blood homeostasis in rat. Journal of Ethnopharmacology, 79(1), 23–26. 10.1016/s0378-8741(01)00342-7 [DOI] [PubMed] [Google Scholar]
- Zaoui, A. , Cherrah, Y. , Lacaille‐Dubois, M. A. , Settaf, A. , Amarouch, H. , & Hassar, M. (2000). Diuretic and hypotensive effects of Nigella sativa in the spontaneously hypertensive rat. Thérapie, 55(3), 379–382. [PubMed] [Google Scholar]
- Zhang, W. , Zhao, Y. , Zhang, F. , Wang, Q. , Li, T. , Liu, Z. , … Zhang, S. (2020). The use of anti‐inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID‐19): The perspectives of clinical immunologists from China. Clinical Immunology, 214, 108393. 10.1016/j.clim.2020.108393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, F. , Yu, T. , Du, R. , Fan, G. , Liu, Y. , Liu, Z. , … Cao, B. (2020). Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: A retrospective cohort study. Lancet, 395(10229), 1054–1062. 10.1016/S0140-6736(20)30566-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, P. , Yang, X. L. , Wang, X. G. , Hu, B. , Zhang, L. , Zhang, W. , … Shi, Z. L. (2020). A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 579(7798), 270–273. 10.1038/s41586-020-2012-7 [DOI] [PMC free article] [PubMed] [Google Scholar]


