Abstract
Coronavirus disease 2019 (COVID-19), caused by the highly contagious novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has resulted in a worldwide pandemic and currently represents a major public health issue. COVID-19 has highlighted the need for clear and accurate guidance on the use of aerosol-generating procedures, such as nebulization, for the treatment of patients with respiratory diseases with or without COVID-19. Despite the lack of evidence, there is heightened concern about the potential risk of transmission of SARS-CoV-2 in the form of aerosolized respiratory droplets during the nebulized treatment of patients with COVID-19. Consequently, the use of metered-dose inhalers (MDIs) has risen considerably as an alternative to nebulized therapy, which has led to inadequate supplies of MDIs in some parts of the United States. In this article, we review and discuss the role of nebulization in patients with SARS-CoV-2 and the treatment of noninfected patients with chronic respiratory diseases. The following two important questions are addressed: (1) should nebulized therapy be used in hospital or home settings by patients infected with SARS-CoV-2; and (2) should nebulized therapy be continued in patients already using it for chronic respiratory disease management in hospital or home settings?
The reviews of this paper are available via the supplemental material section.
Keywords: chronic respiratory disease, COVID-19, handheld inhaler, nebulized therapy, SARS-CoV-2
Introduction
Coronavirus disease 2019 (COVID-19), caused by the highly contagious novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has resulted in a worldwide pandemic and is a significant public health concern.1 The global outbreak of COVID-19 has brought into focus the need for concise and accurate guidance on the use of aerosol-generating procedures, such as nebulization, for the treatment of patients with respiratory diseases with or without COVID-19.
Despite the lack of evidence, there is currently a heightened concern regarding the potential risk of transmission of SARS-CoV-2 in the form of aerosolized respiratory droplets from patients with COVID-19 undergoing nebulized treatment.2,3 The Canadian Paediatric Society and the Global Initiative for Asthma have both advised against the use of nebulized treatment unless absolutely necessary.4,5 The National Institute for Health and Care Excellence (NICE) and Public Health England (PHE) have recommended the continued use of nebulized treatment when applicable.6,7 The Centers of Disease Control and Prevention (CDC) does not advise against the use of nebulized treatment; however, it is categorized as an unknown exposure for healthcare personnel (HCP).8,9 Thus, there is a concern that HCP and healthcare institutions will approach the use of nebulizers in their patients with an overabundance of caution based on the lack of evidence.
As a result of the pandemic, the use of metered-dose inhalers (MDIs) has increased substantially as an alternative to nebulized therapy, resulting in severe shortages of MDIs in some areas of the United States (US).10 Physicians and home-based patients may also misinterpret government recommendations that warn of the possible risk regarding the transmissibility of SARS-CoV-2 by people with suspected COVID-19 who are using nebulized therapy. Therefore, physicians and home-based patients may feel compelled to switch from nebulized therapies to handheld devices. A sudden transition to MDIs may result in unintended consequences for some patients, who may be unable to adequately use their new device or benefit fully from treatment via handheld inhalers. For example, patients may have inadequate hand-breath coordination, poor inspiratory strength, and/or cognitive impairments.11,12 In the home setting, the sudden switch from nebulized treatments to handheld devices could also result in poor medication adherence, resulting in loss of clinical control of disease symptoms, which can lead to severe exacerbations, resulting in emergency room visits or hospitalizations.13–16 Ironically, this substantially increases the risk of exposure to nosocomial SARS-CoV-2.
In this article, we discuss the role of nebulization for patients infected with SARS-CoV-2 and the treatment of noninfected patients with chronic respiratory diseases to address the following two important questions: (1) should nebulized therapy be used in in hospital or home settings by patients infected with SARS-CoV-2; and (2) should nebulized therapy be continued in patients already using it for chronic respiratory disease management in hospital or home settings.
Selection of articles for review
The review topics were divided into specific sections, and articles were selected based on a comprehensive review of the literature. A PubMed search (1 January 2003–7 July 2020) was conducted, including (but not limited to) the following terms/phrases: SARS-CoV-2, COVID-19, SARS, MERS, coronavirus, Severe Acute Respiratory Syndrome, Middle Eastern Respiratory Syndrome, nebulized, nebulization, and handheld inhalers. Additional literature was identified through the review of references found in the primary literature search.
Overview of the transmission routes of respiratory viruses
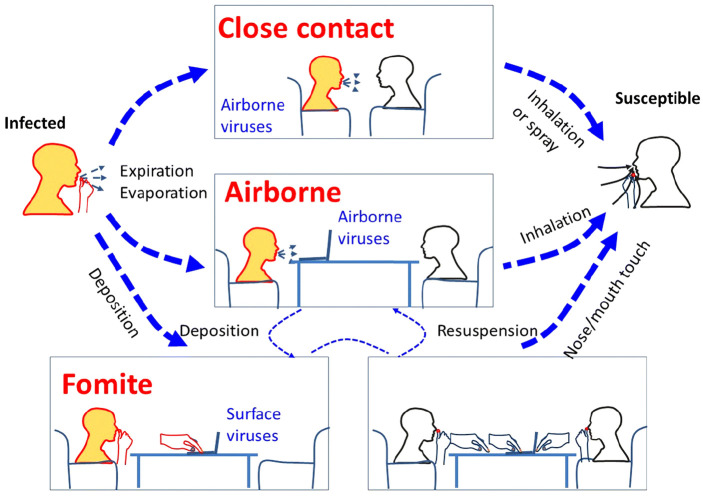
The transmission of viral infections is complex and multifactorial.17 To spread infection, the presence of an inoculum, a sufficient amount of pathogen, and a receptive host is required. In order to be infectious, the pathogen must encounter the correct organ system. The following three routes of transmission spread the majority of respiratory viruses: contact (either direct [skin to skin] or indirect [fomites or surfaces]), droplets, and aerosol transmission (Figure 1).18 Droplets are larger in size than aerosols (generally greater than 5–10 µm in size) and are generated by coughing or sneezing. These generally travel no further than 1 m before settling.17,18 Once they have settled on a surface, the droplets can be a source of contact transmission. Aerosols can be created while breathing and stay suspended in the air longer than droplets, though the life of an aerosol in the air is influenced by many factors, including air exchange rates, temperature gradients, and airflow.17,19,20
Figure 1.
An illustration of various possible transmission routes of respiratory infection between an infected and a susceptible individual. Both close range (i.e., conversational) airborne transmission and longer range (over several meters) transmission routes are illustrated here. The orange head color represents a source and the white head color represents a potential recipient (with the bottom right panel indicating that both heads are potential recipients via self-inoculation from contaminated surface fomite sources). Here “Expiration” also includes normal breathing exhalation, as well as coughing and/or sneezing airflows. Airborne droplets can then settle on surfaces (fomites) from where they can be touched and carried on hands leading to further self-inoculation routes of transmission.
Reproduced from Tellier et al.18 This work is licensed under a Creative Commons Attribution 4.0 International (CC BY 4.0) License. https://creativecommons.org/licenses/by/4.0/.18
Reviews on the transmission of coronaviruses
In specific relation to SARS-CoV-2, the mode of respiratory transmission is not completely understood. Preliminary evidence suggests that, under controlled environmental conditions, SARS-CoV-2 can be viable in aerosols for up to 3 h.21 In addition, another study assessed the aerodynamic nature of SARS-CoV-2 by measuring viral RNA in aerosols in different areas of two hospitals in Wuhan during the COVID-19 outbreak in February and in March 2020.22 The concentration of SARS-CoV-2 RNA detected in aerosols in isolation wards and ventilated patients’ rooms was very low; however, it was higher in the patients’ restroom areas.22 Some medical staff areas had high concentrations of viral RNA initially, but these levels decreased to undetectable levels after the implementation of rigorous sanitization procedures. According to reports from studies in hospital settings in Singapore and Hong Kong, viral RNA was not detected in air samples taken from the rooms of symptomatic patients with COVID-19.23,24 Results from a study in Iran showed that air samples taken 2–5 m from the patients’ beds were negative for SARS-CoV-2 RNA.25 In contrast with the previous studies, a study from the University of Nebraska Medical Center demonstrated SARS-CoV-2 genetic material in approximately 63% of air samples from the rooms of 11 patients infected with COVID-19, with some samples obtained at distances >6 ft from the patient, and in approximately 67% of 12 air samples obtained from the hallways outside patients’ rooms.26 However, it should be noted that this study is a preprint and has not undergone peer review. Whether the levels of virus detected in this study were significant and potentially infectious is also not known.26
The evidence to date suggests that SARS-CoV-2 may be transmitted via droplet and fomite contact. Although airborne transmission is possible, further research is required.
SARS-CoV (SARS) and Middle Eastern Respiratory Syndrome Coronavirus (MERS) are among the most studied respiratory viral infections. The SARS epidemic was linked predominantly to healthcare settings, with ⩾49% of the cases linked to hospitals.27 In addition, super-spreading events, such as those associated with infections in a hotel and an apartment complex in Hong Kong, an airline flight from Hong Kong to Beijing, and an acute care hospital in Toronto, furthered the spread of the SARS outbreak.28–31 A transmission link to HCP was observed when they were in close proximity (<1 m) to the index patient, suggesting direct contact or transmission of the droplet.29,32,33 Samples of air and swabs from frequently touched surfaces in a room occupied by a patient with SARS tested positive by polymerase chain reaction (PCR), but no virus could be cultured from those samples.34
Similar to SARS, MERS outbreaks were predominately linked to healthcare settings, with ⩾31% of the cases linked to hospitals,27,35,36 and healthcare-associated human-to-human transmission was observed.37,38 Super-spreading events have been shown to play a central role in hospital-acquired outbreaks.27,39 MERS-CoV was isolated from environmental surfaces in hospital rooms, suggesting that the virus can be transmitted through contaminated environments and fomites. In addition, potential for the airborne spread of MERS was assessed by an air sample analysis:40 Viral RNA was detected at the inlet of the air ventilation equipment,41 and viruses were extracted from air samples as well as from the surfaces of inaccessible areas (e.g., the ventilator outlet) which had potential for the transmission of aerosols.40
Overall, based on the available experimental and observational data, the evidence supports the potential for the droplet, fomite, and airborne transmission of both SARS and MERS.
Nebulization and viral transmission
Although very limited data exist regarding the risks of transmitting viral infection with nebulized treatment at present, it has been a significant concern since the start of the COVID-19 outbreak. In specific relation to COVID-19, the first case of community-acquired COVID-19 in the US was confirmed in a patient hospitalized in California.9 At the time of patient admission, COVID-19 was not suspected and HCP were not wearing personal protective equipment (PPE). The patient underwent multiple aerosol-generating procedures, including nebulization. In a recent report about that incident, the CDC found that 43 out of 121 HCP that were exposed to this patient with COVID-19 were symptomatic, and subsequently tested for SARS-CoV-2. Of these HCP, three individuals tested positive for the virus. It was reported that all three individuals did not wear PPE when in contact with the patient; therefore, it is difficult to determine, with certainty, whether the HCP who contracted COVID-19 was linked to nebulization. Another study by Tang and colleagues simulated a spontaneously breathing adult patient receiving nebulized therapy with a jet nebulizer and a face mask.42 Air samples obtained from three separate locations indicated 612 viruses per liter near the head, 174 viruses per liter near the abdomen, and 118 viruses per liter near the feet using a live attenuated influenza vaccine as a surrogate virus tracer. The findings of this experimental study showed that the aerosols spread at a decreasing concentration with increasing distance from the patient.42 It is possible that the jet nebulizer with the face mask increased the risk of transmission because viral secretions can enter into the nebulizer’s reservoir.42–45 To reduce viral transmission, jet nebulizers need to be used with the mouthpiece, and HCP should attach filters or one-way valves to the large bore tubing of the nebulizer to prevent viral emissions during aerosol therapy.46 Another option would be to use a mesh nebulizer combined with the mouthpiece and a filter to the other end of the mouthpiece to minimize viral spread to the environment.46
In specific relation to SARS, two cohort studies identified some risks associated with nebulized therapy,47,48 while another reported no such direct association.33 In the two former cohort studies, critical care nurses who assisted with the nebulized therapy of patients with SARS were three times more likely to be infected versus those who did not. However, it should be noted that the use of PPE was not standard practice during the study period. PPE use was highly variable because the nurses were often unaware that the patients were infected with SARS. The latter cohort study by Wong and colleagues reported that medical students conducting bedside clinical trials had an increased risk of SARS, even before nebulized therapy was used.33 A case study found no evidence of SARS-CoV-specific nucleic acid material in air samples taken from the vicinity of a patient with SARS undergoing treatment with a large-volume nebulizer.49
In specific relation to MERS, results from an overcrowded emergency room setting showed increased transmission potential of MERS-CoV from a single patient. MERS-CoV infection was confirmed in 82 individuals (33 patients, 8 HCP, and 41 visitors). This patient did not use nebulized therapy. Results demonstrated that individuals in the same area as the index patient were more likely to get infected.50,51 Furthermore, data from South Korea databases suggested that the MERS outbreak was entirely hospital acquired, and was largely attributable to infection management and policy failures. Nebulization was not listed as a potential source of transmission.50
Overall, the evidence to date has not been definitive in determining whether a direct link exists between nebulization and increased coronavirus transmission. Thus, further research is required to confirm whether this association exists.
Handheld inhalers and viral transmission
At present, there does not appear to be a concern with handheld inhalers and viral transmission. However, abruptly switching patients from nebulized treatment to handheld inhalers may present certain challenges that should be considered. If not accompanied by adequate training for the patient, switching from one device to another can be linked with poor adherence and technique. Suboptimal inhalation delivery could lead to exacerbations and increased healthcare utilization,13–16,52 potentially increasing the risk of nosocomial SARS-CoV-2 exposure. In addition to those with suboptimal peak inspiratory flow, patients who have cognitive, neuromuscular, or ventilatory impairments may not benefit from handheld inhalers.11,12 Furthermore, handheld inhalers often provoke coughing in patients,53,54 thus producing a risk of viral transmission. A study on the use of MDIs with a spacer versus nebulizers for acute airflow obstruction among hospitalized patients in Singapore was analyzed during the SARS outbreak.55 The study demonstrated that the use of a MDI with spacer for acute airflow obstruction was acceptable and preferred by the majority of patients.55 In contrast, nearly two-thirds of nurses thought that older adult patients were more likely to have difficulty using an MDI with a spacer. In addition, more than 80% of nurses stated that nebulized therapy was more effective for treating acute airflow obstruction in the hospital.55
An increase in the use of MDIs has led to shortages in some parts of the US,10 and, therefore, some hospitals and healthcare institutions may consider the need for a common MDI canister protocol during the COVID-19 pandemic. However, deciding whether to implement a common MDI canister protocol requires thoughtful analysis and deliberation. In 2009, the Institute for Safe Medication Practices (ISMP) published an article about the risks and benefits of using a common MDI canister, a patient-specific spacer, and a disinfection procedure between patients to administer doses from the same MDI to multiple patients.56 The following three critical risk factors still exist today: (1) patients undergoing isolation precautions or immunocompromised patients may not be suitable for common canister; (2) methods used to disinfect the mouthpiece (using alcohol wipes) were designed to prevent bacterial contamination, which would not be considered adequate for SARS-CoV-2; and (3) individual noncompliance with the mouthpiece disinfection protocol is of concern.56,57
Currently, there is no evidence to suggest that handheld inhalers will increase viral transmission. However, handheld inhalers as an alternative to nebulizers may have additional challenges in terms of adherence, induction of cough, and fomite transmission.
The right tool for the right patient
In terms of patients with chronic respiratory diseases, the Global Initiative for Chronic Obstructive Lung Disease COVID-19 guidance advises that patients with chronic obstructive pulmonary disease (COPD) maintain regular therapy.58 Similarly, for patients with asthma and/or allergies, the American College of Allergy, Asthma, and Immunology (AAAI) recommends that patients continue their treatment.10 Therefore, patients who are at home receiving nebulized treatment for chronic airway disease and are not experiencing any symptoms of COVID-19 should continue their nebulized treatments. If symptoms develop, patients should self-isolate at home, and, if they are doing that effectively, they can continue their nebulized treatment.
Given the absence of any conclusive data showing that nebulization promotes the transmission of SARS-CoV-2, physicians and healthcare institutions should consider personalizing the prescription of either handheld inhalers or nebulizers for the delivery of bronchodilator treatment for hospitalized patients. For patients who are hospitalized for reasons unrelated to COVID-19, the use of nebulizers should be considered in certain scenarios. Among the most common scenarios are patients with underlying chronic airflow disease hospitalized with an acute exacerbation. These patients are often too tachypneic or have flow limitations to use their handheld inhalers properly. Since nebulizers do not require patient coordination between inhalation and actuation or any special breathing technique, their use should also be considered in patients with cognitive, neuromuscular, or ventilatory impairments and those with suboptimal peak inspiratory flow.11,12 On the other hand, handheld inhalers may be more appropriate for patients who can tolerate MDIs or dry powder inhalers, and for those who do not have any impairments that could affect inhaler technique.
The need for bronchodilators should be carefully evaluated for patients without concomitant COPD or asthma who are hospitalized with COVID-19. A ‘knee-jerk prescription’ of bronchodilators for the relief of dyspnea in these patients may not be indicated.59 There is no description of bronchospasm related to COVID-19, and, therefore, bronchodilators are unlikely to be of benefit if prescribed indiscriminately. If there is evidence of bronchospasm, then a trial of an MDI with a spacer may be appropriate, and, if that is inadequate, then nebulization could be considered, with the HCP using adequate PPE.
Protective measures
Based on the current guidance and limited evidence of increased infection risk with nebulized therapy, the following should be considered (Table 1).10,46,60–64 The use of nebulized treatment at home by a patient with COVID-19 has no known hazards for the patient using the nebulizer; to reduce the risk of affecting other people, patients should follow social distancing guidance and avoid nebulizer use in the presence of others. Furthermore, precautions such as stringent nebulizer hygiene should be implemented, and nebulization should be carried out near open windows or in areas of increased air circulation. Nebulized treatment with long-acting agents, which allows for less frequent dosing, could be considered when applicable to further minimize exposure.
Table 1.
| Home |
|---|
| Avoid nebulization in the presence of other people |
| Stringent nebulizer hygiene |
| Carry out nebulization near open windows/areas of increased air circulation |
| Hospital |
| Adherence to stringent sanitation guidance |
| Use of PPEa in the presence of infected patients |
| Use of negative-pressure rooms |
| Discard/disinfect PPEa after each use |
| Maintenance of least 6 ft from the patient whenever possible |
| Use of filters with nebulizers |
PPE includes N95 respirators/face masks, eye protection, gowns, and gloves.
PPE, personal protective equipment.
From the perspective of public health, hospitals and healthcare facilities should continue to abide by the strict measures that protect HCP from COVID-19, including adherence to stringent sanitization protocols and the use of PPE in the presence of patients with COVID-19.60 In terms of PPE, the CDC currently recommends that HCP wear an N95 respirator or equivalent, eye protection, gown, and gloves when performing an aerosol-generating procedure or providing care in the intensive care unit to patients with known or suspected COVID-19. In the case of a patient undergoing nebulized treatment, the use of negative-pressure rooms, disposing or disinfecting PPE after each use, and maintaining at least 6 ft or greater distance from the patient should be considered.60 In order to minimize viral transmission, it is recommended that nebulizers are used with a mouthpiece and a filter. The COPD Foundation provides additional guidance to use a filter (equivalent to an N95 mask) with nebulizers to filter the patient’s exhaled air, and, therefore, limit the risk of virus transmission.61
Conclusion
In summary, given the absence of any conclusive data that link nebulized treatments to the transmission of SARS-CoV-2, physicians and healthcare institutions should consider “the right tool for the right patient” approach rather than a “one-size-fit- all” approach. Necessary protective measures should also be implemented when applicable.
Supplemental Material
Supplemental material, Author_Response_1 for The use of nebulized pharmacotherapies during the COVID-19 pandemic by Sanjay Sethi, Igor Z. Barjaktarevic and Donald P. Tashkin in Therapeutic Advances in Respiratory Disease
Supplemental material, Reviewer_1_v.1 for The use of nebulized pharmacotherapies during the COVID-19 pandemic by Sanjay Sethi, Igor Z. Barjaktarevic and Donald P. Tashkin in Therapeutic Advances in Respiratory Disease
Supplemental material, Reviewer_2_v.1 for The use of nebulized pharmacotherapies during the COVID-19 pandemic by Sanjay Sethi, Igor Z. Barjaktarevic and Donald P. Tashkin in Therapeutic Advances in Respiratory Disease
Acknowledgments
The authors would like to thank Valerie L. Ravenna, (Theravance Biopharma US, Inc.) for all her efforts in reviewing and compiling the key references for our review paper. The authors acknowledge Gráinne Faherty, for medical writing, and Frederique H. Evans, for editorial assistance (both from Ashfield Healthcare Communications) in the preparation of the document.
Footnotes
Author contribution(s): Sanjay Sethi: Conceptualization; Formal analysis; Writing-review & editing.
Igor Z. Barjaktarevic: Conceptualization; Formal analysis; Writing-review & editing.
Donald P. Tashkin: Conceptualization; Formal analysis; Writing-review & editing.
Conflict of interest statement: SS has consulted for AstraZeneca, Boehringer Ingelheim, Circassia, GlaxoSmithKline, Pulmonx, Nabriva, Novovax, Sunovion, Paratek, Merck and Theravance Biopharma, and has been a speaker for AstraZeneca, Boehringer Ingelheim, Circassia, GlaxoSmithKline, and received research support from Cipla, GlaxoSmithKline and Sanofi.
IZB has consulted for AstraZeneca, Boehringer Ingelheim, CSL Behring, Grifols, Verona Pharma, GE Healthcare, Mylan, Theravance Biopharma, and GlaxoSmithKline and has received research grants from AMGEN, Theravance Biopharma, Mylan, and GE Healthcare.
DPT has served as a consultant and speaker for AstraZeneca, Boehringer Ingelheim, Sunovion, and Mylan, Theravance Biopharma and as a consultant for GlaxoSmithKline.
Funding: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing support was funded by Theravance Biopharma US, Inc. (South San Francisco, CA, USA).
Supplemental material: The reviews of this paper are available via the supplemental material section.
Contributor Information
Sanjay Sethi, University at Buffalo, State University of New York, Clinical and Translational Research Center, 875 Ellicott St., Room 6045A, Buffalo, NY 14215, USA.
Igor Z. Barjaktarevic, David Geffen School of Medicine at UCLA, Los Angeles, CA, USA
Donald P. Tashkin, David Geffen School of Medicine at UCLA, Los Angeles, CA, USA
References
- 1. Lai C, Shih T, Ko W, et al. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents 2020; 55: 105924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Amirav I, Newhouse MT. Transmission of corona virus by nebulizer: a serious, underappreciated risk. CMAJ 2020; 192: E346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Mei-Zahav M, Amirav I. Aerosol treatments for childhood asthma in the era of COVID-19. Pediatr Pulmonol 2020; 55: 1871–1872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Canadian Paediatric Society. Paediatric asthma and COVID-19, https://www.cps.ca/en/documents/position/paediatric-asthma-and-covid-19 (accessed 7 July 2020).
- 5. Global Initiative for Asthma. COVID-19: GINA answers to frequently asked questions on asthma management, https://ginasthma.org/covid-19-gina-answers-to-frequently-asked-questions-on-asthma-management/ (accessed 7 July 2020).
- 6. National Institute for Health and Care Excellence. COVID-19 rapid guideline: community-based care of patients with chronic obstructive pulmonary disease (COPD), https://www.nice.org.uk/guidance/ng168/resources/covid19-rapid-guideline-communitybased-care-of-patients-with-chronic-obstructive-pulmonary-disease-copd-pdf-66141907467973 (accessed 30 April 2020). [PubMed]
- 7. Public Health England. New and emerging respiratory virus threats advisory group (NERVTAG), https://www.gov.uk/government/publications/wuhan-novel-coronavirus-infection-prevention-and-control/covid-19-personal-protective-equipment-ppe (accessed 30 April 2020).
- 8. Centers for Disease Control and Prevention. Healthcare infection prevention and control FAQs for COVID-19, https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-faq.html (accessed 29 April 2020).
- 9. Heinzerling A, Stuckey M, Scheuer T, et al. Transmission of COVID-19 to health care personnel during exposures to a hospitalized patient - Solano County, California, February 2020. MMWR Morb Mortal Wkly Rep 2020; 69: 472–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. American College of Allergy Asthma and Immunology. A message to asthma sufferers about a shortage of albuterol metered dose inhalers, https://acaai.org/news/message-asthma-sufferers-about-shortage-albuterol-metered-dose-inhalers (accessed 23 July 2020).
- 11. Taffet G, Donohue J, Altman P. Considerations for managing chronic obstructive pulmonary disease in the elderly. Clin Interv Aging 2014; 9: 23–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Yawn B, Colice G, Hodder R. Practical aspects of inhaler use in the management of chronic obstructive pulmonary disease in the primary care setting. Int J Chron Obstruct Pulmon Dis 2012; 7: 495–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ari A. Patient education and adherence to aerosol therapy. Respir Care 2015; 60: 941–957. [DOI] [PubMed] [Google Scholar]
- 14. Braido F, Lavorini F, Blasi F, et al. Switching treatments in COPD: implications for costs and treatment adherence. Int J Chron Obstruct Pulmon Dis 2015; 10: 2601–2608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ismaila A, Corriveau D, Vaillancourt J, et al. Impact of adherence to treatment with fluticasone propionate/salmeterol in asthma patients. Curr Med Res Opin 2014; 30: 1417–1425. [DOI] [PubMed] [Google Scholar]
- 16. Vestbo J, Anderson J, Calverley P, et al. Adherence to inhaled therapy, mortality and hospital admission in COPD. Thorax 2009; 64: 939–943. [DOI] [PubMed] [Google Scholar]
- 17. Kutter J, Spronken M, Fraaij P, et al. Transmission routes of respiratory viruses among humans. Curr Opin Virol 2018; 28: 142–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Tellier R, Li Y, Cowling B, et al. Recognition of aerosol transmission of infectious agents: a commentary. BMC Infect Dis 2019; 19: 101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ciuzas D, Prasauskas T, Krugly E, et al. Characterization of indoor aerosol temporal variations for the real-time management of indoor air quality. Atmos Environ 2015; 118: 107–117. [Google Scholar]
- 20. McGrath J, Byrne M, Ashmore M, et al. A simulation study of the changes in PM2.5 concentrations due to interzonal airflow variations caused by internal door opening patterns. Atmos Environ 2014; 87: 183–188. [Google Scholar]
- 21. van Doremalen N, Bushmaker T, Morris D, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med 2020; 382: 1564–1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Liu Y, Ning Z, Chen Y, et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature 2020; 582: 557–560. [DOI] [PubMed] [Google Scholar]
- 23. Cheng V, Wong S-C, Chen J, et al. Escalating infection control response to the rapidly evolving epidemiology of the coronavirus disease 2019 (COVID-19) due to SARS-CoV-2 in Hong Kong. Infect Control Hosp Epidemiol 2020; 41: 493–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Ong S, Tan Y, Chia P, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA 2020; 323: 1610–1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Faridi S, Niazi S, Sadeghi K, et al. A field indoor air measurement of SARS-CoV-2 in the patient rooms of the largest hospital in Iran. Sci Total Environ 2020; 725: 138401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Santarpia J, Rivera D, Herrera V, et al. Transmission potential of SARS-CoV-2 in viral shedding observed at the University of Nebraska Medical Center. medRxiv. Preprint posted online 11 March 2020. DOI: 10.1101/2020.03.23.20039446 [DOI] [Google Scholar]
- 27. Chowell G, Abdirizak F, Lee S, et al. Transmission characteristics of MERS and SARS in the healthcare setting: a comparative study. BMC Med 2015; 13: 210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Braden C. Progress in global surveillance and response capacity 10 years after severe acute respiratory syndrome. Emerg Infect Dis 2013; 19: 864–869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ofner M. Cluster of severe acute respiratory syndrome cases among protected health-care workers – Toronto, Canada, April 2003 (Reprinted from MMWR, vol. 52, pp. 433–436, 2003). JAMA 2003; 289: 2788–2789. [DOI] [PubMed] [Google Scholar]
- 30. Shen Z. Superspreading SARS events, Beijing, 2003. Emerg Infect Dis 2004; 10: 256–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Wong G. MERS, SARS, and Ebola: the role of super-spreaders in infectious disease. Cell Host Microbe 2015; 18: 398–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Varia M. Investigation of a nosocomial outbreak of severe acute respiratory syndrome (SARS) in Toronto, Canada. CMAJ 2003; 169: 285–292. [PMC free article] [PubMed] [Google Scholar]
- 33. Wong T, Lee C, Tam W, et al. Cluster of SARS among medical students exposed to single patient, Hong Kong. Emerg Infect Dis 2004; 10: 269–276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Booth T. Detection of airborne severe acute respiratory syndrome (SARS) coronavirus and environmental contamination in SARS outbreak units. J Infect Dis 2005; 191: 1472–1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Hunter J. Transmission of Middle East Respiratory Syndrome coronavirus infections in healthcare settings, Abu Dhabi. Emerg Infect Dis 2016; 22: 647–656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Oboho I. 2014 MERS-CoV outbreak in Jeddah – a link to health care facilities. N Engl J Med 2015; 372: 846–854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Assiri A. Hospital outbreak of Middle East Respiratory Syndrome coronavirus. N Engl J Med 2013; 369: 407–416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Guery B. Clinical features and viral diagnosis of two cases of infection with Middle East Respiratory Syndrome coronavirus: a report of nosocomial transmission. Lancet 2013; 381: 2265–2272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Oh M. Middle East Respiratory Syndrome coronavirus superspreading event involving 81 persons, Korea. J Korean Med Sci 2015; 30: 1701–1705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Kim S. Extensive viable Middle East Respiratory Syndrome (MERS) coronavirus contamination in air and surrounding environment in MERS isolation wards. Clin Infect Dis 2016; 63: 363–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Bin S. Environmental contamination and viral shedding in MERS patients during MERS-CoV outbreak in South Korea. Clin Infect Dis 2016; 62: 755–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Tang JW, Kalliomaki P, Varila TM, et al. Nebulisers as a potential source of airborne virus. J Infect. Epub ahead of print 15 May 2020. DOI: 10.1016/j.jinf.2020.05.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Rau JL, Ari A, Restrepo RD. Performance comparison of nebulizer designs: constant-output, breath-enhanced, and dosimetric. Respir Care 2004; 49: 174–179. [PubMed] [Google Scholar]
- 44. Ari A. Jet, ultrasonic, and mesh nebulizers: an evaluation of nebulizers for better clinical outcomes. Euras J Pulm 2014; 16: 1–7. [Google Scholar]
- 45. McGrath JA, O’Toole C, Bennett G, et al. Investigation of fugitive aerosols released into the environment during high-flow therapy. Pharmaceutics 2019; 11: 254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Ari A. Practical strategies for a safe and effective delivery of aerosolized medications to patients with COVID-19. Respir Med 2020; 167: 105987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Loeb M, McGeer A, Henry B, et al. SARS among critical care nurses, Toronto. Emerg Infect Dis 2004; 10: 251–255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Raboud J, Shigayeva A, McGeer A, et al. Risk factors for SARS transmission from patients requiring intubation: a multicentre investigation in Toronto, Canada. PLoS One 2010; 5: e10717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Wan G, Tsai Y, Wu Y, et al. A large-volume nebulizer would not be an infectious source for severe acute respiratory syndrome. Infect Control Hosp Epidemiol 2004; 25: 1113–1115. [DOI] [PubMed] [Google Scholar]
- 50. Kim K, Tandi T, Choi J, et al. Middle East Respiratory Syndrome coronavirus (MERS-CoV) outbreak in South Korea, 2015: epidemiology, characteristics and public health implications. J Hosp Infect 2017; 95: 207–213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Nam HS, Park JW, Ki M, et al. High fatality rates and associated factors in two hospital outbreaks of MERS in Daejeon, the Republic of Korea. Int J Infect Dis 2017; 58: 37–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Björnsdóttir U, Gizurarson S, Sabale U. Potential negative consequences of non-consented switch of inhaled medications and devices in asthma patients. Int J Clin Pract 2013; 67: 904–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Arcapta™ Neohaler™ (indacaterol inhalation powder). Prescribing information, https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/022383s000lbl.pdf (accessed 7 July 2020).
- 54. Ventolin HFA (albuterol sulfate) Inhalation aerosol for oral inhalation. Prescribing information, https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/020983s032lbl.pdf (accessed 7 July 2020).
- 55. Khoo S, Tan L, Said N, et al. Metered-dose inhaler with spacer instead of nebulizer during the outbreak of severe acute respiratory syndrome in Singapore. Respir Care 2009; 54: 855–860. [DOI] [PubMed] [Google Scholar]
- 56. ISMP. Shared MDIs: can cross-contamination be avoided? https://www.ismp.org/resources/shared-mdis-can-cross-contamination-be-avoided (accessed 28 April 2020).
- 57. ISMP. Revisiting the need for MDI common canister protocols during the COVID-19 pandemic, https://www.ismp.org/resources/revisiting-need-mdi-common-canister-protocols-during-covid-19-pandemic (accessed 28 April 2020).
- 58. Global Initiative for Chronic Obstuctive Lung Disease. GOLD COVID-19 guidance, https://goldcopd.org/gold-covid-19-guidance/ (accessed 7 July 2020).
- 59. Anesi G. Coronavirus disease 2019 (COVID-19): critical care and airway management issues, https://www.uptodate.com/contents/coronavirus-disease-2019-covid-19-critical-care-and-airway-management-issues (accessed 11 May 2020).
- 60. Centers for Disease Control and Prevention (CDC). Interim infection prevention and control recommendations for patients with suspected or confirmed coronavirus disease 2019 (COVID-19) in healthcare settings, https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html (accessed 30 April 2020).
- 61. COPD Foundation. COVID-19, COPD and you: Important strategies from leading medical experts on managing your health, https://www.copdfoundation.org/Downloads/COVID19_Webinar_Q_&_A_FINAL.pdf (accessed 30 April 2020).
- 62. Wang Q, Yu C. The role of masks and respirator protection against SARS-CoV-2. Infect Control Hosp Epidemiol 2020; 41: 746–747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Yu XY, Sun L, Yao K, et al. Thinking and proposal of new coronavirus aerosol transmission path. Chin Ophthalmol. Epub ahead of print 14 March 2020. DOI: 10.3760/cma.j.cn112142-20200313-00181 [DOI] [Google Scholar]
- 64. Ari A. Use of aerosolised medications at home for COVID-19. Lancet Respir Med. Epub ahead of print 22 June 2020. DOI: 10.1016/S2213-2600(20)30270-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, Author_Response_1 for The use of nebulized pharmacotherapies during the COVID-19 pandemic by Sanjay Sethi, Igor Z. Barjaktarevic and Donald P. Tashkin in Therapeutic Advances in Respiratory Disease
Supplemental material, Reviewer_1_v.1 for The use of nebulized pharmacotherapies during the COVID-19 pandemic by Sanjay Sethi, Igor Z. Barjaktarevic and Donald P. Tashkin in Therapeutic Advances in Respiratory Disease
Supplemental material, Reviewer_2_v.1 for The use of nebulized pharmacotherapies during the COVID-19 pandemic by Sanjay Sethi, Igor Z. Barjaktarevic and Donald P. Tashkin in Therapeutic Advances in Respiratory Disease