Abstract
Relapse was the major cause of treatment failure in patients with acute lymphoblastic leukemia (ALL) after allogeneic hematopoietic stem cell transplantation (allo-HSCT). We aimed to identify the efficacy and safety of preemptive interferon-α (IFN-α) treatment in ALL patients who had minimal residual disease (MRD) after allo-HSCT. Multiparameter flow cytometry and polymerase chain reaction assays were applied for MRD monitoring. Recombinant human IFN-α-2b injections were administered subcutaneously twice weekly in every 4 weeks cycle. Twenty-four (35.3%), 5 (7.4%), 6 (8.8%), and 13 (19.1%) patients achieved MRD negativity at 1, 2, 3, and > 3 months, respectively, after treatment. Seven patients showed grade ≥ 3 toxicities after IFN-α treatment. The 4-year cumulative incidence of total acute graft-versus-host disease (aGVHD), severe aGVHD, total chronic GVHD (cGVHD), and severe cGVHD after treatment was 14.7%, 2.9%, 40.0%, and 7.5%, respectively. The 4-year cumulative incidences of relapse and non-relapse mortality after treatment was 31.9% and 6.0%, respectively. The 4-year probabilities of disease-free survival and overall survival after IFN-α treatment were 62.1% and 71.1%, respectively. Thus, preemptive IFN-α treatment could protect against relapse and improve long-term survival for ALL patients who had MRD after allo-HSCT. The study was registered at https://clinicaltrials.gov as #NCT02185261 (09/07/2014).
Subject terms: Leukaemia, Acute lymphocytic leukaemia
Introduction
Despite considerable advances in the allogeneic hematopoietic stem cell transplantation (allo-HSCT)1, relapse remains the major cause of transplant failure in patients with acute lymphoblastic leukemia (ALL)2. Thus, identifying patients who were at higher risks for relapse after allo-HSCT is of great importance. Minimal residual disease (MRD) helped to identify patients who still harbored higher levels of disease but were below the detection capabilities of morphological analysis. Multiparameter flow cytometry (MFC) identified cells with leukemia-associated immunophenotypes (LAIPs) and polymerase chain reaction (PCR) assays detected leukemia-associated genetic abnormalities, both could be applied for monitoring MRD in leukemia patients. MRD monitoring was proved to predict impending relapse after allo-HSCT by numerous studies3–5.
Impending relapse could be prevented by the early detection of MRD and timely treatments. Thus, preemptive intervention, which could spare patients in remission from further therapies, was reasonable for patients with MRD. Chemotherapy plus donor leukocyte infusion (Chemo-DLI) was the most effective preemptive intervention for MRD6,7, however, it may lead to some severe complications (e.g., graft-versus-host disease [GVHD] and pancytopenia)8. In addition, it was out of choices for some patients because of related donor refusal or unavailability of the second donation from an unrelated donor. Preemptive tyrosine kinase inhibitor (TKIs) treatment was proved to be a useful intervention9,10, but only applied to patients with Philadelphia chromosome (Ph)-positive ALL. Chimeric antigen receptor (CAR) T-cell immunotherapy was another potential preemptive intervention11–13. However, it might also lead to several complications (e.g. life-threatening neurological toxicity and cytokine release syndrome)14,15, and remissions after CAR T-cell treatment was relatively brief because of poor CAR T cell persistence and/or leukemia cell resistance16.
Interferon-α (IFN-α) had shown activity in acute leukemia through immune activation17, which rekindled the interest of using IFN-α as an immunotherapy for patients receiving allo-HSCT18. Our pilot studies showed that IFN-α was a safe agent for allo-HSCT recipients19. We further confirmed that preemptive IFN-α treatment can clear the MRD effectively in patients with acute leukemia and high-risk myelodysplastic syndrome after allo-HSCT4,20–22. IFN-α could also be used as a salvage treatment for patients with unsatisfactory response to preemptive Chemo-DLI23. However, the sample of ALL patients enrolled in these studies was relatively small, and no study had identified the efficacy of preemptive IFN-α treatment in a disease-specific population of patients with ALL after allo-HSCT. In addition, the follow-ups of these patients were short. Thus, the long-term efficacy of preemptive IFN-α treatment remains unknown in ALL patients following allo-HSCT.
Therefore, we aimed to identify the safety and long-term efficacy of preemptive IFN-α treatment in ALL patients following allo-HSCT.
Results
Patient characteristic
The characteristics of the 68 ALL patients receiving preemptive IFN-α treatment are summarized in Table 1. Besides of WT1, 22 patients monitored other molecular markers regularly before and after allo-HSCT (TCR: 7, EVI1: 2, E2A-PBX1: 6, SET-NUP214: 1; SIL-TAL1: 3, MLL: 3), and 11 of them showed WT1 and these molecular makers positive simultaneously before IFN-α treatment (TCR: 5, EVI1: 1, E2A-PBX1: 2, SET-NUP214: 1; SIL-TAL1: 1, MLL: 1). The cycles of IFN-α treatment was 2 (range 0.5–14) cycles, and 3 patients received IFN-α treatment for more than 6 cycles. The reasons for discontinuing IFN-α treatment included MRD turned negative (n = 21), grade ≥ 3 toxicities (infectious: n = 3; hematologic: n = 3; pulmonary: n = 1), GVHD (n = 28), and relapse (n = 12). The duration of follow-up after IFN-α treatment was 953 (range 63–1639) days.
Table 1.
Patient characteristics between IFN-α group in the present study and non-IFN-α groups in the historical cohort.
| Characteristics | IFN-α group (n = 68) | Non-IFN-α group (n = 18) | P value |
|---|---|---|---|
| Sex, male/female, n | 44/24 | 11/7 | 0.778 |
| Median age at allo-HSCT, years (range) | 23 (9–54) | 25 (7–45) | 0.975 |
| Median WBC at diagnosis, × 109/L (range) | 13 (1–647) | 13 (1–256) | 0.766 |
| Median time from diagnosis to allo-HSCT, months (range) | 6 (3–48) | 6 (4–36) | 0.271 |
| First CR induction courses, n (%) | 0.118 | ||
| 1 | 54 (79.4) | 18 (100.0) | |
| > 1 | 14 (20.6) | 0 (0.0) | |
| Pre-HSCT cycles of chemotherapy, courses (range) | 4 (2–23) | 7 (3–17) | < 0.001 |
| Median time from allo-HSCT to MRD positivity, days (range) | 166 (26–735) | 130 (38–586) | 0.811 |
| Time from allo-HSCT to MRD positivity, n (%) | 0.602 | ||
| Early-onset MRD | 22 (32.4) | 7 (38.9) | |
| Late-onset MRD | 46 (67.6) | 11 (61.1) | |
| Median time from allo-HSCT to IFN-α treatment, days (range) | 193 (36–748) | – | |
| Median time from MRD to IFN-α treatment, days (range) | 13 (0–147) | – | |
| Lineage, n (%) | 0.001 | ||
| B | 47 (69.1) | 5 (27.8) | |
| T | 21 (30.9) | 13 (72.2) | |
| Cytogenetics, n (%) | 0.651 | ||
| 11q23 | 4 (5.9) | 2 (11.1) | |
| At least five abnormalities | 7 (10.3) | 1 (5.6) | |
| Low hypodiploidy-near triploidy | 3 (4.4) | 1 (5.6) | |
| High hyperdiploidy | 2 (2.9) | 1 (5.6) | |
| t(1;19) | 1 (1.5) | 0 (0.0) | |
| Other abnormalities | 5 (7.4) | 3 (16.6) | |
| Normal | 46 (67.6) | 10 (55.5) | |
| Disease status at allo-HSCT, n (%) | 0.709 | ||
| CR1 | 59 (86.8) | 15 (83.3) | |
| CR2 | 9 (13.2) | 3 (16.7) | |
| Disease risk index before allo-HSCT, n (%) | 0.709 | ||
| Intermediate risk | 59 (86.8) | 15 (83.3) | |
| High risk | 9 (13.2) | 3 (16.7) | |
| Donor–recipient relationship, n (%) | 0.087 | ||
| Mother–child | 5 (7.4) | 4 (22.2) | |
| Others | 63 (92.6) | 14 (77.8) | |
| Donor-recipient sex matched, n (%) | 0.747 | ||
| Female to male | 13 (19.1) | 4 (22.2) | |
| Others | 55 (80.9) | 14 (77.8) | |
| Donor type | 0.735 | ||
| HLA-identical sibling donor | 12 (17.6) | 4 (22.2) | |
| HLA-haploidentical related donor | 56 (82.4) | 14 (77.8) | |
| Number of HLA-A, -B, -DR mismatches, n (%) | 0.222 | ||
| 0–1 | 15 (22.1) | 7 (38.9) | |
| 2–3 | 53 (77.9) | 11 (61.1) | |
| Graft type, n (%) | – | ||
| Bone marrow and peripheral blood | 68 (100.0) | 18 (100.0) | |
| MRD status after allo-HSCT, n (%) | 0.057 | ||
| PCR positive once | 29 (42.6) | 3 (16.7) | |
| PCR positive twice | 18 (26.5) | 6 (33.3) | |
| MFC positive once | 5 (7.4) | 0 (0.0) | |
| MFC positive twice | 5 (7.4) | 1 (5.6) | |
| PCR positive and MFC positive simultaneously | 11 (16.1) | 8 (44.4) | |
| MRD level, n (%) | 0.778 | ||
| Low level | 24 (35.3) | 7 (38.9) | |
| High level | 44 (64.7) | 11 (61.1) | |
| Discontinuing immunosuppressant before IFN-α treatment, n (%) | 46 (67.6) | – |
allo-HSCT allogeneic hematopoietic stem cell transplantation, HLA human leukocyte antigen, IFN-α interferon-α, MFC multiparameter flow cytometry, MRD minimal residual disease, PCR polymerase chain reaction, WBC white blood cell.
Statistical significance was set at P < 0.05.
GVHD
Ten patients had aGVHD after IFN-α treatment (Table 2). The cumulative incidence of total and severe aGVHD (≥ grade III) at 4 years after treatment was 14.7% (95% CI 6.2–23.2%) and 2.9% (95% CI 0.0–6.9%), respectively.
Table 2.
Characteristics of aGVHD after preemptive IFN-α treatment.
| Characteristics of aGVHD | IFN-α group (n = 68) |
|---|---|
| Time from aGVHD to immunotherapy, days (range) | 12 (1–64) |
| Severity of aGVHD, n (%) | |
| None | 58 (85.3) |
| Grade I | 3 (4.4) |
| Grade II | 5 (7.4) |
| Grade III | 2 (2.9) |
| Site of aGVHD, n (%) | |
| Skin | 9 (13.2) |
| Liver | 0 (0.0) |
| Gut | 4 (5.9) |
| Number of sites, n (%) | |
| 0 | 58 (85.3) |
| 1 | 7 (10.3) |
| 2 | 3 (4.4) |
Data was present as n (%) or median (range).
aGVHD acute graft-versus-host disease, IFN-α interferon-α.
Twenty-seven patients had cGVHD after IFN-α treatment (Table 3). The cumulative incidence of total and severe cGVHD at 4 years after IFN-α treatment was 40.0% (95% CI 28.2–51.8%) and 7.5% (95% CI 1.1–13.9%), respectively.
Table 3.
Characteristics of cGVHD after preemptive IFN-α treatment.
| Characteristics of cGVHD | IFN-α group (n = 68) |
|---|---|
| Time from cGVHD to immunotherapy, days (range) | 43 (1–404) |
| Severity of cGVHD, n (%) | |
| None | 41 (60.3) |
| Mild | 10 (14.7) |
| Moderate | 12 (17.6) |
| Severe | 5 (7.4) |
| Type of cGVHD, n (%) | |
| None | 41 (60.3) |
| Classical cGVHD | 24 (35.3) |
| Overlap syndrome | 3 (4.4) |
| Site of cGVHD, n (%) | |
| Skin | 21 (30.9) |
| Mouth | 8 (11.8) |
| Eye | 4 (5.9) |
| Liver | 7 (10.3) |
| Gut | 5 (7.4) |
| Lung | 5 (7.4) |
| Number of sites, n (%) | |
| 0 | 41 (60.3) |
| 1 | 14 (20.6) |
| 2 | 7 (10.3) |
| ≥ 3 | 6 (8.8) |
Data was present as n (%) or median (range).
cGVHD chronic graft-versus-host disease, IFN-α interferon-α.
MRD evolution and relapse
After preemptive IFN-α treatment, 24 (35.3%), 5 (7.4%), 6 (8.8%), and 13 (19.1%) patients achieved MRD negativity at 1, 2, 3, and > 3 months, respectively, and 6 of them showed relapse. More than 80% of the patients with MRDsin+ achieved MRD negativity after IFN-α treatment, which was higher than that of the patients with MRDco+ (82.4% vs. 58.8%, P = 0.033). Twenty (29.4%) patients did not achieve MRD negativity after IFN-α treatment, and 15 of them showed relapse. The rate of achieving MRD negativity was 65.2% and 81.8% for the patients who discontinued and did not discontinue immunosuppressive agents before IFN-α treatment (P = 0.160). The cumulative incidences of achieving MRD negativity at 3 months, 6 months, 12 months, and 24 months after IFN-α treatment were 48.8% (95% CI 36.7–60.9%), 59.4% (95% CI 47.5–71.3%), 65.7% (95% CI 54.0–77.4%), and 70.7% (95% CI 59.3–82.1%), respectively.
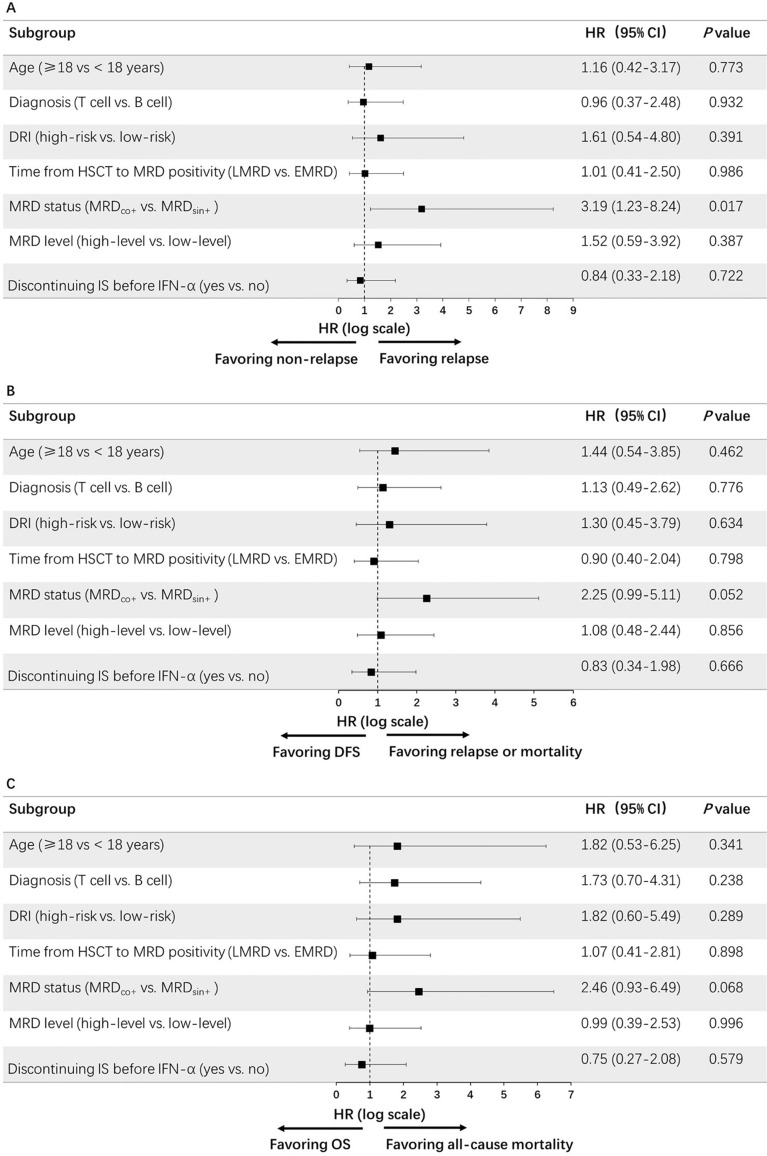
Twenty-one patients experienced relapse after IFN-α treatment. The duration from IFN-α treatment to relapse was 110 (range 14–890) days. The cumulative incidence of relapse (CIR) at 4 years after IFN-α treatment was 31.9% (95% CI 20.5–43.3%). The 4-year CIR after IFN-α treatment was higher in the MRDco+ group compared to that of the MRDsin+ group (47.4% vs. 17.8%, P = 0.011). The MRDsin+ status before IFN-α treatment was the only factor which was associated with a low risk of relapse in univariate analysis (Fig. 1A).
Figure 1.
Univariate analysis for prognostic factors of preemptive IFN-α treatment: (A) relapse; (B) disease-free survival, and (C) overall survival.
NRM
Four patients died of NRM (Supplementary Table 3). The duration from IFN-α treatment to NRM was 112 (range 77–575) days. The cumulative incidence of NRM at 4 years after IFN-α treatment was 6.0% (95% CI 3.1–8.9%).
Survival
The probability of DFS at 4 years after IFN-α treatment was 62.1% (95% CI 50.2–74.0%). The probability of OS at 4 years after IFN-α treatment was 71.1% (95% CI 60.0–82.2%). The MRDsin+ status before IFN-α treatment tended to be associated with a better DFS and OS in univariate analysis (Fig. 1B, C).
Clinical outcomes of MRD-positive patients receiving preemptive Chemo-DLI
We also analyzed the data of patients who received preemptive Chemo-DLI during the same period (Supplementary Table 1 and Supplementary Fig. 1). The median cycle of Chemo-DLI was 1 (range 1–2 cycles), and 3 patients received Chemo-DLI for more than 1 cycle. The characteristics of patients in the Chemo-DLI group were summarized in Supplementary Table 2. The characteristic of aGVHD and cGVHD after Chemo-DLI were showed in Supplementary table 4and Supplementary table 5, respectively. Twelve patients experienced relapse after Chemo-DLI and the median time from Chemo-DLI to relapse was 42 (range 9–1027) days. The 4-year CIR after Chemo-DLI was 60.1% (95% CI 48.1–72.1%). No patients died of NRM after Chemo-DLI. The probabilities of DFS and OS at 4 years after Chemo-DLI were 39.9% (95% CI 16.5–63.3%) and 67.4% (95% CI 44.7–90.1%), respectively.
Patients receiving preemptive IFN-α treatments had better survival than those without preemptive interventions in the historical cohort
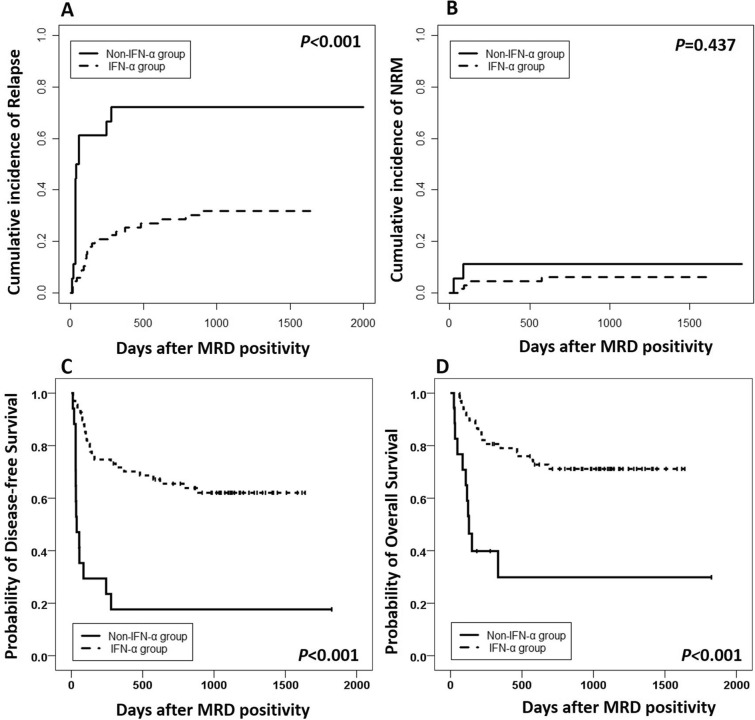
To further confirm the efficacy of preemptive IFN-α treatment, a historical cohort between March 1, 2009 and May 31, 2013 including MRD-positive patients without any interventions was enrolled as controls (n = 18)22. T-ALL was more common in historical cohort and they had more cycles of chemotherapy before transplantation; however, the other patient characteristics were comparable between the present and the historical cohorts (Table 1). The cumulative incidences of relapse and survival were worse in the historical cohort than those receiving preemptive IFN-α treatment in the present study (Fig. 2A, C, D), and the cumulative incidence of NRM rates were comparable between the present and the historical cohorts (Fig. 2B). After adjusted by the MRD status, preemptive IFN-α treatment could also decrease the risk of relapse and improve survival (Supplementary table 6).
Figure 2.
Cumulative incidence of survival after MRD positivity between the preemptive IFN-α treatment group in the present study and those who had MRD but did not receive interventions in the historical cohort: (A) relapse; (B) non-relapse mortality; (C) disease-free survival, and (D) overall survival.
Clinical outcomes of patients receiving prolonged IFN-α treatment
Three patients received IFN-α treatment for more than 6 cycles at the request of themselves, 2 received 7 cycles and 1 received 14 cycles IFN-α treatment. They achieved MRD negative at 84 days, 148 days, and 396 days after IFN-α treatment, respectively. The relapse, NRM, DFS, and OS rates were 0.0% versus 31.9% (P = 0.272), 0.0% versus 6.0% (P = 0.642), 100.0% versus 62.1% (P = 0.230), and 100% versus 71.1% (P = 0.301), respectively, for those with and without prolonged IFN-α treatment.
Discussion
In our study, the cumulative incidence of relapse and NRM at 4 years after preemptive IFN-α treatment were 31.9% and 6.0%, respectively; and the probability of DFS and OS at 4 years after preemptive IFN-α treatment were 62.1% and 71.1%, respectively. Our study is the first to study the efficacy of preemptive IFN-α treatment in a disease-specific population of patients with ALL. These results identify the undefined role of this intervention strategy in ALL patients following allo-HSCT.
The graft-versus-leukemia (GVL) effect had been described in ALL since 1970s24, which was further supported by a large scale study recently25. IFN-α can exert an immunomodulatory effect, promote the GVL effect, and clear MRD after allo-HSCT4,18,21. Moreover, it also showed the growth-inhibitory or cytotoxic effects on human ALL cell in vitro26–28. Based on these results, IFN-α emerges as a useful agent which can clear MRD through different mechanisms. In fact, IFN-α had been used as adjuvant29–31 or maintenance treatments in ALL patients32,33, which was reported to help to achieve CR again in ALL patients who experienced relapse after allo-HSCT34,35. Sumi et al.33 also reported that IFN-α helped to achieve sustained molecular CR in an ALL patient with continuing detection of MRD following allo-HSCT. However, the evidences of IFN-α as a treatment option for ALL was generally derived from single case report or small sample studies, and its clinical utility in ALL has not been consistently established. In the present study, we observed that more than 70% of the patients achieved MRD negativity after preemptive IFN-α treatment. Up to now, this is the largest study confirming that IFN-α can indeed induce clinically relevant anti-leukemic responses in ALL patients.
MFC relying on the identification of cells with LAIPs and is widely believed to be sensitive for relapse prediction in ALL patients3,36–38. Thus, a patient was considered as MRDsin+ status when a single BM sample was tested positive by MFC for LAIPs in our study. In addition, the relapse rate of ALL patients who had one positive LAIPs result was 80.0% after allo-HSCT39.
Approximately 60% of the patients used WT1 as an MRD marker in the present study. WT1 is still an important genetic marker for ALL patients40–43. In addition, Zhao et al.39 reported that the relapse rate of ALL patients who had one positive WT1 result after allo-HSCT was 63.9%, and the sensitivity and specificity of WT1 was 62.2% and 90.6%, respectively, for indicating ALL relapse in allo-HSCT recipients. Thus, it is reasonable to use WT1 as the triggering marker for preemptive IFN-α treatment in ALL patients. However, some authors suggested that the sensitivity and specificity of WT1 monitoring might be relatively low44,45. Thus, MFC was used in the detection of MRD simultaneously, compensating for the relatively low sensitivity of WT1 expression. On the other hand, WT1 was not a specific molecular marker of leukemia. It is inevitable that some patients may receive IFN-α treatment because of high WT1 expressions which were actually not relevant to leukemia (i.e., receiving prophylactic IFN-α treatment), but Klingemann et al.46 demonstrated that even prophylactic IFN-α treatment could also decrease the risk of relapse after allo-HSCT. In addition, only few severe toxicities were observed during IFN-α treatment, which might minimize the impact of the relatively low specificity of WT1 expression.
The 4-year CIR of patients who had MRDsin+ after preemptive IFN-α treatment was only 17.8% in the present study. Thirty-five patients with MRDsin+ were tested repeatedly 2 weeks after obtaining the first positive results. Among the 35 patients who showed MRDsin+ but did not receive IFN-α treatment, although immunosuppressions were tapered in 21 patients, only 1 patient achieved MRD negativity and the other 34 patients were tested positive for 2 consecutive BM samples (i.e., MRDco+). This indicated that MRDsin+ and MRDco+ might be different stages of the ALL progression. Reducing immunosuppression alone could not clear the MRD effectively and the preemptive IFN-α treatment for MRDsin+ patients could help to control the disease more timely. In addition, Zhao et al.39 reported that patients with MRDco+ had higher relapse rate (WT1 + twice: 100%; MFC + twice: 87.5%; MFC + and WT1+: 100%) compared to that of MRDsin+ (WT1 + once: 63.9%; MFC + once: 80.0%), suggesting that MRDco+ may represent a higher risk of relapse compared with MRDsin+. Our results also showed that the clinical outcomes seemed to be better in the MRDsin+ group than the MRDco+ group among patients receiving preemptive IFN-α treatment. Thus, preemptive IFN-α treatment may not completely overcome the poor prognostic significance of MRDco+ status of ALL, and patients with MRDsin+ may benefit more from preemptive IFN-α treatment after allo-HSCT.
We previously reported that preemptive Chemo-DLI could significantly decrease relapse and improve survival of patients with MRD7. In this study, the 4-year CIR, DFS, and OS rate of Chemo-DLI were 60.1%, 39.9%, and 67.4%, respectively. However, approximately 40% of our patients received preemptive IFN-α treatment for MRDsin+ and most of them could clear the MRD. These patients would not be classified as MRDco+ and they did not need to receive Chemo-DLI. Thus, it would be premature to derive conclusions regarding the superiority of IFN-α treatment over Chemo-DLI in patients with MRD.
A limitation to our current study was that it was not a randomized trial and the number of patients in historical control was relatively small. In addition, the ratio of PCR positive and MFC positive simultaneously seemed to be higher in non-IFN-α group although P value was 0.057, which meant that the risk of relapse may not be totally equivalent between IFN group and non-IFN-α group. In future, prospective, randomized trial may further confirm the efficacy of preemptive IFN-α in these patients. Secondly, the sensitivity of PCR for WT1 transcript and MFC for LAIPs was only 10−3–10−4. With a deep detection limit and high specificity, next-generation sequencing for MRD may represent a promising tool for the ALL patients47, and it may further improve the efficacy of preemptive IFN-α treatment. Thirdly, IFN-α can exert anti-leukemia effect through activating NK cells; however, we did not examine the number of NK cells in the present study, and we would identify the association between the number of NK cells and MRD negativity in our future study. Lastly, besides of the CAR-T therapy, several monoclonal antibodies (MoAbs) can also target certain surface antigens on ALL cells resulting in their destruction. However, the efficacy of these MoAbs in allo-HSCT recipients with MRD was unclear. Our future prospective studies can further compare the efficacy among MRD-directed preemptive Chemo-DLI, IFN-α treatment, MoAbs, and CAR-T therapy in ALL patients following allo-HSCT48–50.
In conclusion, preemptive IFN-α treatment could protect against relapse and improved long-term survival of ALL patients who had MRD after allo-HSCT. Because IFN-α may tend to be started in patients with relatively low leukemia burden17, it could not only unlock its therapeutic potential in ALL, but also spare the patients in remission from further therapy. Moreover, IFN-α is a simple treatment with increased accessibility as it could be performed on an outpatient basis. Based on our results, future randomized clinical trials are needed to further compare the efficacy of preemptive IFN-α treatment and cytotherapies in ALL patients who had MRD after allo-HSCT.
Patients and methods
Patients
From June 1, 2014 to December 31, 2017, consecutive Ph-negative ALL patients receiving allo-HSCT at the Peking University Institute of Hematology (PUIH) and showed MRD positivity were enrolled if they met the following criteria: (1) ALL defined as first or second complete remission (CR) without t(9;22) mutations (Supplementary Table 1)51; (2) regained MRD positivity after allo-HSCT. The patients who had active GVHD, active infections, severe myelosuppression, and organ failure were excluded (Supplementary methods)4. Considering the probable synergistic effect between IFN-α treatment and Chemo-DLI, the patients who received both Chemo-DLI and IFN-α treatment were excluded in this study (Supplementary method; Supplementary Fig. 1). The final follow-up visits for endpoint analysis were conducted on December 31, 2019. Thirty-three patients were previously reported in 20174, and all of them were enrolled and followed further in this study. The study was performed in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Peking University People’s Hospital. All patients or the patients’ guardians gave written informed consent before enrollment. The study was registered at https://clinicaltrials.gov as #NCT02185261.
Transplant regimens
The major preconditioning regimen consisted of cytarabine (Ara-C), busulfan, cyclophosphamide, and semustine. Human leukocyte antigen (HLA)-haploidentical related donor (haplo-RD) and HLA-unrelated donor (URD) groups received rabbit anti-thymocyte globulin. All patients received granulocyte colony-stimulating factor (G-CSF)-mobilized, fresh, and unmanipulated bone marrow cells plus peripheral blood stem cells in the present study (Supplementary methods)52–57.
MRD monitoring after allo-HSCT
Routine MRD monitoring was performed 1, 2, 3, 4.5, 6, 9, and 12 months post-transplantation and at 6-month intervals thereafter. MFC for LAIPs and TaqMan-based reverse transcription-real time PCR for the expressions of Wilms’ tumor gene 1 (WT1) were performed in all patients as a routine clinical test on bone marrow (BM) aspirate samples (Supplementary methods)58. When a single BM sample was tested positive by PCR or MFC, we considered this patient to have an MRD-positive status because the use of multiple methods could ensure sensitivity and specificity in the detection of the MRD39.
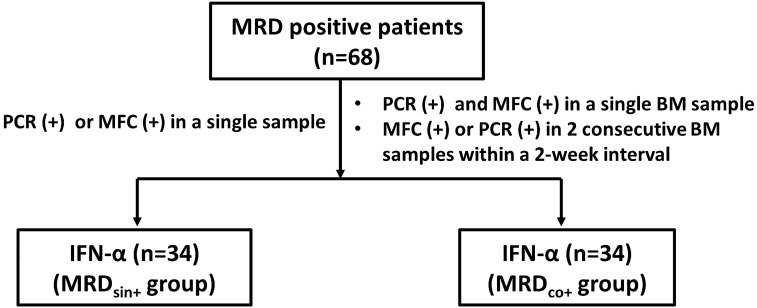
Cases in which a single BM sample was tested positive by PCR or MFC were defined as the MRDsin+ group. Cases in which 2 consecutive BM samples within a 2-week interval were tested positive by PCR or MFC or those in which a single BM sample was tested positive by both PCR and MFC were defined as the MRDco+ group (Supplementary Fig. 1).
Patients in the MRDsin+ group were recommended to receive preemptive IFN-α treatment. For the patients with MRDco+, the efficacy of Chemo-DLI had been confirmed7 but the role of IFN-α treatment was undefined when this study started. Thus, preemptive Chemo-DLI was the first choice for patients with MRDco+, and those who were unable to receive Chemo-DLI (e.g., patient or provider refusal) could receive IFN-α treatment (Fig. 3; Supplementary methods; Supplementary Table 2; Supplementary Fig. 1).
Figure 3.
Diagram of patients enrolled.
Preemptive IFN-α treatment and Chemo-DLI protocol
The detailed protocols for preemptive IFN-α treatment and Chemo-DLI was according to the routine protocols of PUIH which had been described in detailed (Supplementary methods)4,7,21,22. In brief, recombinant human IFN-α-2b injections (Anferon; Tianjin Hualida Biotechnology Co., Ltd., Tianjin, China) were administered subcutaneously for 6 cycles (twice weekly in every 4 weeks cycle), at dosages of 3 million units for patients older than 16 years and at 3 million units per square meter for those younger than 16 years (capped by 3 million units). Prolonged treatment with IFN-α was permitted at the request of patients. MRD status was monitored 1, 2, 3, 4.5, 6, 9, and 12 months after preemptive IFN-α treatment and at 6-month intervals thereafter. The patients who had persistent and increasing levels of MRD or those regained MRD positivity after achieving MRD negativity after IFN-α treatment could receive salvage Chemo-DLI (Supplementary methods)7.
Treatment of GVHD after preemptive immunotherapy
The treatments of acute GVHD (aGVHD) and chronic GVHD (cGVHD) were according to accepted international criteria (Supplementary methods)59–61.
Definitions and assessments
Disease risk index (DRI) before allo-HSCT was described according to the criteria of Armand et al. (i.e., ALL patients in CR1 were categorized into intermediate risk, and ALL patients in CR2 were categorized into high risk group)62. GVHD was diagnosed according to accepted international criteria63,64. Definition of relapse, non-relapse mortality (NRM), early-onset MRD (EMRD), late-onset MRD (LMRD), high-level MRD, and low-level MRD were described in supplementary method4.
Statistical analysis
The primary endpoint was relapse, and secondary endpoints were NRM, disease-free survival (DFS), and overall survival (OS). This study was planned to detect a relapse rate of 55% in patients with MRD receiving preemptive IFN-α treatment, from the reference rate of 75% in patients with PCR or MFC positivity but did not receive interventions in our previous study, controlling for type I and II error rates at 5% and 10%, respectively. Considering an expulsion rate of 15%, a total of 68 patients was planned to be enrolled.
Comparisons of patient characteristics between the groups were performed using the Mann–Whitney U-test for continuous variable and χ2 and Fisher’s exact tests for categorical data. The probability of survival was calculated using the Kaplan–Meier estimator. The incidences of GVHD were calculated using the cumulative incidence function, with death and relapse as competing risks65. Cumulative incidences were estimated for NRM and relapse, to account for competing risks. Relapse was the competing event for NRM and vice versa. Hazard ratios (HRs) for clinical outcomes were estimated from Cox regression analyses. P values were 2-sided. The SPSS 24 (SPSS Inc./IBM, Armonk, NY, USA) and the R software package (version 2.6.1; https://www.r-project.org) were used for data analyses.
Compliance with ethics guidelines
Sining Liu, Xueyi Luo, Xiaohui Zhang, Lanping Xu, Yu Wang, Chenhua Yan, Huan Chen, Yuhong Chen, Wei Han, Fengrong Wang, Jingzhi Wang, Kaiyan Liu, Xiaojun Huang, and Xiaodong Mo declared no potential financial conflict of interest related to this manuscript. Informed consent was obtained from all patients or their guardians. The study was conducted in accordance with the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of Peking University People’s Hospital.
Supplementary information
Acknowledgements
This work was supported by the Capital’s Funds for Health Improvement and Research (Grant Number: 2018-4-4089), CAMS Innovation Fund for Medical Sciences (CIFMS) (Grant Number: 2019-I2M-5-034), Peking University Medicine Fund of Fostering Young Scholars’ Scientific & Technological Innovation (Grant Number: BMU2020PY007), National Key Research and Development Program of China (Grant Number: 2017YFA0104500), the Foundation for Innovative Research Groups of the National Natural Science Foundation of China (Grant Number: 81621001), the Key Program of the National Natural Science Foundation of China (Grant Number: 81530046, 81930004), the Science and Technology Project of Guangdong Province of China (Grant Number: 2016B030230003), Peking University Clinical Scientist Program (Grant Number: BMU2019LCKXJ003), and supported by the Fundamental Research Funds for the Central Universities.
Author contributions
X.M. and X.H. designed the study. S.L., X.L., X.Z., L.X., Y.W., C.Y., H.C., Y.C., W.H., F.W., J.W., K.L. collected the data. S.L., X.L. and X.M. analyzed the data and drafted the manuscript. All authors contributed to the data interpretation, manuscript preparation, and approval of the final version.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-77186-9.
References
- 1.Xu L, et al. The consensus on indications, conditioning regimen, and donor selection of allogeneic hematopoietic cell transplantation for hematological diseases in China-recommendations from the Chinese Society of Hematology. J. Hematol. Oncol. 2018;11:33–33. doi: 10.1186/s13045-018-0564-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yan CH, et al. Causes of mortality after haploidentical hematopoietic stem cell transplantation and the comparison with HLA-identical sibling hematopoietic stem cell transplantation. Bone Marrow Transplant. 2016;51:391–397. doi: 10.1038/bmt.2015.306. [DOI] [PubMed] [Google Scholar]
- 3.Campana D, Pui CH. Minimal residual disease-guided therapy in childhood acute lymphoblastic leukemia. Blood. 2017;129:1913–1918. doi: 10.1182/blood-2016-12-725804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mo XD, et al. IFN-alpha is effective for treatment of minimal residual disease in patients with acute leukemia after allogeneic hematopoietic stem cell transplantation: Results of a registry study. Biol. Blood Marrow Transplant. 2017;23:1303–1310. doi: 10.1016/j.bbmt.2017.04.023. [DOI] [PubMed] [Google Scholar]
- 5.Wang Y, et al. The consensus on the monitoring, treatment, and prevention of leukemia relapse after allogeneic hematopoietic stem cell transplantation in China. Cancer Lett. 2018;438:63–75. doi: 10.1016/j.canlet.2018.08.030. [DOI] [PubMed] [Google Scholar]
- 6.Dominietto A, et al. Donor lymphocyte infusions for the treatment of minimal residual disease in acute leukemia. Blood. 2007;109:5063–5064. doi: 10.1182/blood-2007-02-072470. [DOI] [PubMed] [Google Scholar]
- 7.Yan CH, et al. Risk stratification-directed donor lymphocyte infusion could reduce relapse of standard-risk acute leukemia patients after allogeneic hematopoietic stem cell transplantation. Blood. 2012;119:3256–3262. doi: 10.1182/blood-2011-09-380386. [DOI] [PubMed] [Google Scholar]
- 8.Orti G, et al. Donor lymphocyte infusions in AML and MDS: Enhancing the graft-versus-leukemia effect. Exp. Hematol. 2017;48:1–11. doi: 10.1016/j.exphem.2016.12.004. [DOI] [PubMed] [Google Scholar]
- 9.Chen H, et al. Administration of imatinib after allogeneic hematopoietic stem cell transplantation may improve disease-free survival for patients with Philadelphia chromosome-positive acute lymphobla stic leukemia. J. Hematol. Oncol. 2012;5:29. doi: 10.1186/1756-8722-5-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wassmann B, et al. Early molecular response to posttransplantation imatinib determines outcome in MRD+ Philadelphia-positive acute lymphoblastic leukemia (Ph+ ALL) Blood. 2005;106:458–463. doi: 10.1182/blood-2004-05-1746. [DOI] [PubMed] [Google Scholar]
- 11.Cheng Y, et al. Donor-derived CD19-targeted T cell infusion eliminates B cell acute lymphoblastic leukemia minimal residual disease with no response to donor lymphocytes after allogeneic hematopoietic stem cell transplantation. Engineering. 2019;5:150–155. doi: 10.1016/j.eng.2018.12.006. [DOI] [Google Scholar]
- 12.Ogba N, et al. Chimeric antigen receptor T-cell therapy. J. Natl. Compr. Cancer Netw. 2018;16:1092–1106. doi: 10.6004/jnccn.2018.0073. [DOI] [PubMed] [Google Scholar]
- 13.Park JH, et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N. Engl. J. Med. 2018;378:449–459. doi: 10.1056/NEJMoa1709919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen H, et al. Management of cytokine release syndrome related to CAR-T cell therapy. Front. Med. 2019;13:610–617. doi: 10.1007/s11684-019-0714-8. [DOI] [PubMed] [Google Scholar]
- 15.Maude SL, Teachey DT, Porter DL, Grupp SA. CD19-targeted chimeric antigen receptor T-cell therapy for acute lymphoblastic leukemia. Blood. 2015;125:4017–4023. doi: 10.1182/blood-2014-12-580068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shah NN, Fry TJ. Mechanisms of resistance to CAR T cell therapy. Nat. Rev. Clin. Oncol. 2019;16:372–385. doi: 10.1038/s41571-019-0184-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Anguille S, et al. Interferon-α in acute myeloid leukemia: an old drug revisited. Leukemia. 2011;25:739–748. doi: 10.1038/leu.2010.324. [DOI] [PubMed] [Google Scholar]
- 18.Lin XJ, et al. Effects of preemptive interferon-alpha monotherapy in acute leukemia patients with relapse tendency after allogeneic hematopoietic stem cell transplantation: A case–control study. Ann. Hematol. 2018;97:2195–2204. doi: 10.1007/s00277-018-3429-z. [DOI] [PubMed] [Google Scholar]
- 19.Mo X, et al. Interferon alpha: The salvage therapy for patients with unsatisfactory response to minimal residual disease-directed modified donor lymphocyte infusion. Chin. Med. J. (Engl.) 2014;127:2583–2587. [PubMed] [Google Scholar]
- 20.Mo X, et al. Minimal residual disease-directed immunotherapy for high-risk myelodysplastic syndrome after allogeneic hematopoietic stem cell transplantation. Front Med. 2019;13:354–364. doi: 10.1007/s11684-018-0665-5. [DOI] [PubMed] [Google Scholar]
- 21.Mo X-D, et al. Interferon-α is effective for treatment of minimal residual disease in patients with t(8;21) acute myeloid leukemia after allogeneic hematopoietic stem cell transplantation: results of a prospective registry study. Oncologist. 2018;23:1349–1357. doi: 10.1634/theoncologist.2017-0692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mo X-D, et al. Interferon-α: A potentially effective treatment for minimal residual disease in acute leukemia/myelodysplastic syndrome after allogeneic hematopoietic stem cell transplantation. Biol. Blood Marrow Transplant. 2015;21:1939–1947. doi: 10.1016/j.bbmt.2015.06.014. [DOI] [PubMed] [Google Scholar]
- 23.Mo X, et al. Interferon-alpha salvage treatment is effective for patients with acute leukemia/myelodysplastic syndrome with unsatisfactory response to minimal residual disease-directed donor lymphocyte infusion after allogeneic hematopoietic stem cell transplantation. Front. Med. 2019;13:238–249. doi: 10.1007/s11684-017-0599-3. [DOI] [PubMed] [Google Scholar]
- 24.Weiden PL, et al. Antileukemic effect of graft-versus-host disease in human recipients of allogeneic-marrow grafts. N. Engl. J. Med. 1979;300:1068–1073. doi: 10.1056/NEJM197905103001902. [DOI] [PubMed] [Google Scholar]
- 25.Yeshurun M, et al. The impact of the graft-versus-leukemia effect on survival in acute lymphoblastic leukemia. Blood Adv. 2019;3:670–680. doi: 10.1182/bloodadvances.2018027003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Durrieu L, et al. Human interferon-alpha increases the cytotoxic effect of CD56(+) cord blood-derived cytokine-induced killer cells on human B-acute lymphoblastic leukemia cell lines. Cytotherapy. 2012;14:1245–1257. doi: 10.3109/14653249.2012.714864. [DOI] [PubMed] [Google Scholar]
- 27.Lee WH, Liu FH, Lee YL, Huang HM. Interferon-alpha induces the growth inhibition of human T-cell leukaemia line Jurkat through p38alpha and p38beta. J. Biochem. 2010;147:645–650. doi: 10.1093/jb/mvp213. [DOI] [PubMed] [Google Scholar]
- 28.Manabe A, Yi T, Kumagai M, Campana D. Use of stroma-supported cultures of leukemic cells to assess antileukemic drugs. I. Cytotoxicity of interferon alpha in acute lymphoblastic leukemia. Leukemia. 1993;7:1990–1995. [PubMed] [Google Scholar]
- 29.Visani G, et al. Alpha-interferon improves survival and remission duration in P-190BCR-ABL positive adult acute lymphoblastic leukemia. Leukemia. 2000;14:22–27. doi: 10.1038/sj.leu.2401641. [DOI] [PubMed] [Google Scholar]
- 30.Visani G, Piccaluga P, Malagola M, Isidori A. Efficacy of dasatinib in conjunction with alpha-interferon for the treatment of imatinib-resistant and dasatinib-resistant Ph+ acute lymphoblastic leukemia. Leukemia. 2009;23:1687–1688. doi: 10.1038/leu.2009.96. [DOI] [PubMed] [Google Scholar]
- 31.Wassmann B, et al. Efficacy and safety of imatinib mesylate (Glivec) in combination with interferon-alpha (IFN-alpha) in Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ALL) Leukemia. 2003;17:1919–1924. doi: 10.1038/sj.leu.2403093. [DOI] [PubMed] [Google Scholar]
- 32.Kuang P, et al. Sustaining integrating imatinib and interferon-alpha into maintenance therapy improves survival of patients with Philadelphia positive acute lymphoblastic leukemia ineligible for allogeneic stem cell transplantation. Leuk. Lymphoma. 2016;57:2321–2329. doi: 10.3109/10428194.2016.1144882. [DOI] [PubMed] [Google Scholar]
- 33.Sumi M, et al. Second cord blood transplantation and interferon-alpha maintenance therapy for relapsed Ph(+) acute lymphoblastic leukemia with the T315I mutation. Leuk. Lymphoma. 2017;58:2005–2007. doi: 10.1080/10428194.2016.1266623. [DOI] [PubMed] [Google Scholar]
- 34.Nagafuji K, et al. Interferon-alpha treatment of acute lymphoblastic leukemia relapse after unrelated bone marrow transplantation. Int. J. Hematol. 1998;67:63–68. doi: 10.1016/S0925-5710(97)00081-9. [DOI] [PubMed] [Google Scholar]
- 35.Volodin L, et al. Successful treatment of first and second recurrence of acute lymphoblastic leukemia after related allogeneic bone marrow transplant at unusual sites using single-dose vincristine followed by interferon-alpha2b and granulocyte-macrophage colony-stimulating factor. Leuk. Lymphoma. 2013;54:1107–1109. doi: 10.3109/10428194.2012.734617. [DOI] [PubMed] [Google Scholar]
- 36.Bruggemann M, Kotrova M. Minimal residual disease in adult ALL: Technical aspects and implications for correct clinical interpretation. Blood Adv. 2017;1:2456–2466. doi: 10.1182/bloodadvances.2017009845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Modvig S, et al. Minimal residual disease quantification by flow cytometry provides reliable risk stratification in T-cell acute lymphoblastic leukemia. Leukemia. 2019;33:1324–1336. doi: 10.1038/s41375-018-0307-6. [DOI] [PubMed] [Google Scholar]
- 38.Pemmaraju N, et al. Significance of recurrence of minimal residual disease detected by multi-parameter flow cytometry in patients with acute lymphoblastic leukemia in morphological remission. Am. J. Hematol. 2017;92:279–285. doi: 10.1002/ajh.24629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhao XS, et al. Combined use of WT1 and flow cytometry monitoring can promote sensitivity of predicting relapse after allogeneic HSCT without affecting specificity. Ann. Hematol. 2013;92:1111–1119. doi: 10.1007/s00277-013-1733-1. [DOI] [PubMed] [Google Scholar]
- 40.Chiusa L, et al. Prognostic value of quantitative analysis of WT1 gene transcripts in adult acute lymphoblastic leukemia. Haematologica. 2006;91:270–271. [PubMed] [Google Scholar]
- 41.Heesch S, et al. Prognostic implications of mutations and expression of the Wilms tumor 1 (WT1) gene in adult acute T-lymphoblastic leukemia. Haematologica. 2010;95:942–949. doi: 10.3324/haematol.2009.016386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Qin, Y. Z. et al. The prognostic significance of Wilms' tumor gene 1 (WT1) expression at diagnosis in adults with Ph-negative B cell precursor acute lymphoblastic leukemia. Ann. Hematol. (2019). [DOI] [PubMed]
- 43.Zhang B, et al. The relationship between WT1 expression level and prognosis in patients of acute T lymphoblastic leukemia following allogeneic hematopoietic stem cell transplantation. Zhonghua Xue Ye Xue Za Zhi. 2015;36:642–646. doi: 10.3760/cma.j.issn.0253-2727.2015.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Boublikova L, et al. Wilms' tumor gene 1 (WT1) expression in childhood acute lymphoblastic leukemia: A wide range of WT1 expression levels, its impact on prognosis and minimal residual disease monitoring. Leukemia. 2006;20:254–263. doi: 10.1038/sj.leu.2404047. [DOI] [PubMed] [Google Scholar]
- 45.Schuurhuis GJ, et al. Minimal/measurable residual disease in AML: A consensus document from the European LeukemiaNet MRD Working Party. Blood. 2018;131:1275–1291. doi: 10.1182/blood-2017-09-801498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Klingemann HG, et al. Treatment with recombinant interferon (alpha-2b) early after bone marrow transplantation in patients at high risk for relapse [corrected] Blood. 1991;78:3306–3311. doi: 10.1182/blood.V78.12.3306.3306. [DOI] [PubMed] [Google Scholar]
- 47.Mo X-D, Lv M, Huang X-J. Preventing relapse after haematopoietic stem cell transplantation for acute leukaemia: The role of post-transplantation minimal residual disease (MRD) monitoring and MRD-directed intervention. Br. J. Haematol. 2017;179:184–197. doi: 10.1111/bjh.14778. [DOI] [PubMed] [Google Scholar]
- 48.Barsan V, Ramakrishna S, Davis KL. Immunotherapy for the treatment of acute lymphoblastic leukemia. Curr. Oncol. Rep. 2020;22:11. doi: 10.1007/s11912-020-0875-2. [DOI] [PubMed] [Google Scholar]
- 49.Ali S, et al. Blinatumomab for acute lymphoblastic leukemia: The first bispecific T-cell engager antibody to be approved by the EMA for minimal residual disease. Oncologist. 2020;25:e709–e715. doi: 10.1634/theoncologist.2019-0559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Franquiz MJ, Short NJ. Blinatumomab for the treatment of adult B-cell acute lymphoblastic leukemia: Toward a new era of targeted immunotherapy. Biologics. 2020;14:23–34. doi: 10.2147/BTT.S202746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Giebel S, et al. Hematopoietic stem cell transplantation for adults with Philadelphia chromosome-negative acute lymphoblastic leukemia in first remission: A position statement of the European Working Group for Adult Acute Lymphoblastic Leukemia (EWALL) and the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation (EBMT) Bone Marrow Transplant. 2019;54:798–809. doi: 10.1038/s41409-018-0373-4. [DOI] [PubMed] [Google Scholar]
- 52.Liu Y, et al. Comparison analysis between haplo identical stem cell transplantation and matched sibling donor stem cell transplantation for high-risk acute myeloid leukemia in first complete remission. Sci. China Life Sci. 2019;62:691–697. doi: 10.1007/s11427-018-9361-2. [DOI] [PubMed] [Google Scholar]
- 53.Tang, F. et al. Comparison of the clinical outcomes of hematologic malignancies after myeloablative haploidentical transplantation with G-CSF/ATG and posttransplant cyclophosphamide: Results from the Chinese Bone Marrow Transplantation Registry Group (CBMTRG). Sci China Life Sci. (2019). [DOI] [PubMed]
- 54.Wang Y, et al. Who is the best donor for a related HLA haplotype-mismatched transplant? Blood. 2014;124:843–850. doi: 10.1182/blood-2014-03-563130. [DOI] [PubMed] [Google Scholar]
- 55.Xiao-Jun H, et al. Partially matched related donor transplantation can achieve outcomes comparable with unrelated donor transplantation for patients with hematologic malignancies. Clin. Cancer Res. 2009;15:4777–4783. doi: 10.1158/1078-0432.CCR-09-0691. [DOI] [PubMed] [Google Scholar]
- 56.Wang Y, et al. Haploidentical vs identical-sibling transplant for AML in remission: A multicenter, prospective study. Blood. 2015;125:3956–3962. doi: 10.1182/blood-2015-02-627786. [DOI] [PubMed] [Google Scholar]
- 57.Mo X-D, et al. The role of collateral related donors in haploidentical hematopoietic stem cell transplantation. Sci. Bull. 2018;63:1376–1382. doi: 10.1016/j.scib.2018.08.008. [DOI] [PubMed] [Google Scholar]
- 58.Zhao XS, et al. Wilms' tumor gene 1 expression: An independent acute leukemia prognostic indicator following allogeneic hematopoietic SCT. Bone Marrow Transplant. 2012;47:499–507. doi: 10.1038/bmt.2011.121. [DOI] [PubMed] [Google Scholar]
- 59.Dignan FL, et al. Diagnosis and management of chronic graft-versus-host disease. Br. J. Haematol. 2012;158:46–61. doi: 10.1111/j.1365-2141.2012.09128.x. [DOI] [PubMed] [Google Scholar]
- 60.Dignan FL, et al. Diagnosis and management of acute graft-versus-host disease. Br. J. Haematol. 2012;158:30–45. doi: 10.1111/j.1365-2141.2012.09129.x. [DOI] [PubMed] [Google Scholar]
- 61.Liu, S. N. et al. Prognostic factors and long-term follow-up of basiliximab for steroid-refractory acute GVHD: Updated experienced from a large-scale study. Am. J. Hematol. (2020). [DOI] [PubMed]
- 62.Armand P, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123:3664–3671. doi: 10.1182/blood-2014-01-552984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Filipovich AH, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol. Blood Marrow Transplant. 2005;11:945–956. doi: 10.1016/j.bbmt.2005.09.004. [DOI] [PubMed] [Google Scholar]
- 64.Przepiorka D, et al. Consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15:825–828. [PubMed] [Google Scholar]
- 65.Gooley TA, Leisenring W, Crowley J, Storer BE. Estimation of failure probabilities in the presence of competing risks: New representations of old estimators. Stat. Med. 1999;18:695–706. doi: 10.1002/(SICI)1097-0258(19990330)18:6<695::AID-SIM60>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.