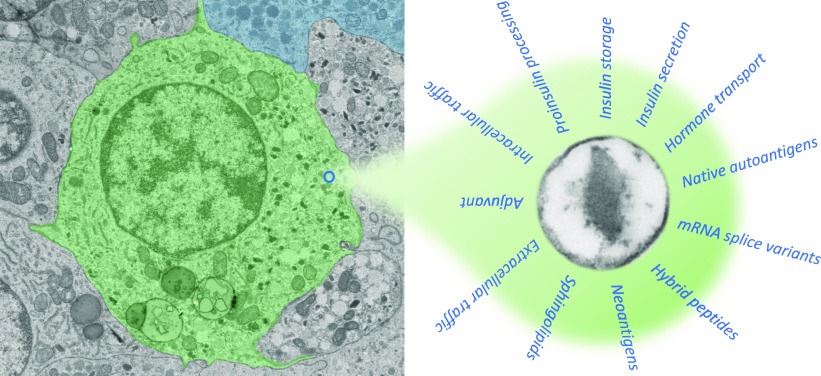
Insulin secretory granules of pancreatic β-cells are complex intracellular organelles comprising many proteins with different catalytic activities and messenger functions (1). These subcellular organelles have a distinctive morphology in electron micrographs as a sphere of around 200–300 nm in diameter comprising a crystalline core of insulin surrounded by a halo of less dense material in a phospholipid bilayer envelope (Fig. 1) (2). The granule is more than just a hormone repository in the cell. It is the site of proteolytic conversion of proinsulin into insulin. This dynamic structure is also involved in intercellular transport and communication through the secretion of other biologically active molecules. Around 10% of the granule population turns over every hour during active secretion. An important contribution to our understanding of the regulation of insulin secretion has come from studies of the granules, and in particular the elucidation of the structure and function of many granule proteins (1).
Figure 1.
Multitasking by insulin granules. Insulin secretory granules (right) are unique features of β-cells (green; left) in pancreatic islets. Light organelles represent immature granules, whereas mature granules are dark and dense spheres containing insulin crystals, which are distinct from somatostatin granules in delta cells (blue). Native proteins as well as posttranscriptionally or posttranslationally modified proteins and sphingolipids have now been identified in these characteristic organelles that act as autoantigenic targets or immune modulators of islet-specific autoreactive CD4 and CD8 T cells. (Electron microscopy graph kindly shared by Dr. Ben Giepmans [2].)
But there is something else about granules. Their exclusivity to β-cells makes them likely to contain immunogenic targets in the selective autoimmune-mediated β-cell destruction causing type 1 diabetes (T1D). Three decades ago, an initial dialogue between β-cell physiologists, biochemists, and immunologists led to the first evidence pointing that there is something about these granules that stimulated islet autoimmunity (3). Until then, immunological studies in T1D were largely biased to insulin and proinsulin, which were the most obvious study subjects at that time. Yet, stronger and more frequent T-cell responses associated with development of T1D have never been reported than when purified insulin secretory granules were used to activate T cells; indeed, autoimmunity to granules has proved to correlate best with development of T1D and recurrence of islet autoimmunity and loss of β-cells following clinical islet transplantation thus far (4,5). Yet, the burden of purifying sufficient numbers of precious insulin secretory granules from β-cells, and the attraction and convenience of using recombinant proteins recognized by islet autoantibodies as T-cell antigens instead, distracted attention from using granules further, preventing us from seeing the forest for the trees. Also, the nature of components of insulin granules stimulating T-cell autoimmunity remained largely unknown.
On the 30th anniversary of the discovery of insulin secretory granules as important source of targets of T1D-associated T-cell autoimmunity, Azoury et al. (6) revisit this characteristic β-cell organelle in a hunt for new islet autoantigens in this issue of Diabetes, discovering several known and new granule antigens recognized by T cells. In addition to enzymes unique to β-cells and involved in the processing proinsulin to insulin, several other granule-associated proteins that are shared with other endocrine tissues proved to be processed and presented by HLA class I and recognized by CD8 T cells of both healthy individuals and those with T1D (6). Recently, several new universes of islet proteome were revealed as targets of islet autoimmunity, including posttranslationally modified islet proteins, hybrid peptides joining two different islet proteins into epitopes, and neoantigens resulting from alternative splicing or ribosomal infidelity creating completely new proteins not normally encoded in the β-cell genome (7–10). Indeed, all types of β-cell proteome (native, modified, and neo) united in insulin secretory granules and proved to act as T-cell targets. The nature of the newly identified CD8 T-cell autoantigens underscores their potential relevance to T1D, given that their distribution is limited to neuroendocrine tissues, warranting future studies to tie this into the immunopathogenesis of T1D. Indeed, similar T cells were found in inflamed islets in NOD mice that could transfer experimental autoimmune diabetes in nondiabetic recipient mice, supporting their diabetogenic potential (6). These findings confirm and extend studies in both humans and mice pointing to granules as source of a wide range of identifiable native and neo islet autoantigens and add CD8 T-cell targets to those recognized by CD4 T cells and autoantibodies (3–5,11).
Insulin granules also contain nonprotein ingredients such as sphingolipids, including sulfatides that contribute to formation and structure of insulin crystals, which possess immunogenic and anti-inflammatory properties. Sulfatides themselves are targets of islet-specific autoantibodies as well as anti-inflammatory natural killer T cells that bridge the innate and adaptive immune system (12). Sulfatides also possess anti-inflammatory properties that can inhibit T-cell autoimmunity and potentially make granules less immunogenic, adding to the magic of insulin secretory granules and their constituents (13). Abnormal islet sphingolipid metabolism in T1D can be therapeutically corrected by the drug fenofibrate and may represent a novel therapeutic opportunity (12).
Insulin secretory granules serve as autoimmune messengers between β-cells and blood and beyond, not just as a source of islet autoantigens, but also in trafficking of β-cell components outside of β-cells (14). Islet resident macrophages sampling large areas of the islet were found to be in close contact with both β-cells and blood vessels and have direct access to the vessel lumen. These macrophages captured intact dense core insulin granules from β-cells and presented insulin peptides to insulin-reactive T cells (14). This process amplified after glucose challenge. By their nature, insulin secretory granules act as so-called hapten-carrier complexes that are efficiently phagocytosed by macrophages, where haptens are small molecules that elicit an immune response only when attached to a large carrier such as a protein or organelle such as granules acting as adjuvant.
The report by Azoury et al. highlights a conundrum that frequencies of circulating CD8 T cells did not differ between health and disease. While this may partly reflect that T cells reactive with islet neoantigens not expressed in thymus escape selection and shaping of the T-cell repertoire that otherwise would lead to central immune tolerance (7), it is important to note that peripheral mechanisms of immune tolerance exist too that can regulate islet autoimmunity and keep islet-specific T cells in check. This is illustrated by studies on functional T-cell autoimmunity to secretory granules that did show strong association with T1D (4,5). T-cell function and activity is subject to inhibition by regulatory T cells in healthy individuals (15). The presence of islet-specific autoreactive T cells in healthy subjects could also explain the development of T1D in cancer patients successfully treated with immunotherapy blocking this immune regulation to unleash immunity against tumor antigens (so-called immune checkpoint inhibitors such as pembrolizumab and nivolumab) (16,17).
The diversity of β-cell targets of the immune system has become daunting. Yet, it offers opportunities to assess whether certain patterns of islet autoimmunity correlate with distinct T1D endotypes, to improve diagnosis and offer precision and personalized medicine in the future (18).
Article Information
Acknowledgments. In loving memory of my mentor Dr. John C. Hutton, pioneer in studies of the insulin secretory granule.
B.O.R. is director of the Wanek Family Project for Type 1 Diabetes.
Footnotes
See accompanying article, p. 2678.
References
- 1.Hutton JC. The insulin secretory granule. Diabetologia 1989;32:271–281 [DOI] [PubMed] [Google Scholar]
- 2.de Boer P, Pirozzi NM, Wolters AHG, et al. Large-scale electron microscopy database for human type 1 diabetes. Nat Commun 2020;11:2475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Roep BO, Arden SD, de Vries RR, Hutton JC. T-cell clones from a type-1 diabetes patient respond to insulin secretory granule proteins. Nature 1990;345:632–634 [DOI] [PubMed] [Google Scholar]
- 4.Roep BO, Kallan AA, Duinkerken G, et al. T-cell reactivity to beta-cell membrane antigens associated with beta-cell destruction in IDDM. Diabetes 1995;44:278–283 [DOI] [PubMed] [Google Scholar]
- 5.Roep BO, Stobbe I, Duinkerken G, et al. Auto- and alloimmune reactivity to human islet allografts transplanted into type 1 diabetic patients. Diabetes 1999;48:484–490 [DOI] [PubMed] [Google Scholar]
- 6.Azoury ME, Tarayrah M, Afonso G, et al. Peptides derived from insulin granule proteins are targeted by CD8+ T cells across MHC class I restrictions in humans and NOD mice. Diabetes 2020;69:2678–2690 [DOI] [PubMed] [Google Scholar]
- 7.de Jong VM, Abreu JR, Verrijn Stuart AA, et al. Alternative splicing and differential expression of the islet autoantigen IGRP between pancreas and thymus contributes to immunogenicity of pancreatic islets but not diabetogenicity in humans. Diabetologia 2013;56:2651–2658 [DOI] [PubMed] [Google Scholar]
- 8.van Lummel M, Duinkerken G, van Veelen PA, et al. Posttranslational modification of HLA-DQ binding islet autoantigens in type 1 diabetes. Diabetes 2014;63:237–247 [DOI] [PubMed] [Google Scholar]
- 9.Delong T, Wiles TA, Baker RL, et al. Pathogenic CD4 T cells in type 1 diabetes recognize epitopes formed by peptide fusion. Science 2016;351:711–714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kracht MJ, van Lummel M, Nikolic T, et al. Autoimmunity against a defective ribosomal insulin gene product in type 1 diabetes. Nat Med 2017;23:501–507 [DOI] [PubMed] [Google Scholar]
- 11.Bergman B, Haskins K. Islet-specific T-cell clones from the NOD mouse respond to beta-granule antigen. Diabetes 1994;43:197–203 [DOI] [PubMed] [Google Scholar]
- 12.Holm LJ, Krogvold L, Hasselby JP, et al. Abnormal islet sphingolipid metabolism in type 1 diabetes. Diabetologia 2018;61:1650–1661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Buschard K, Schloot NC, Kaas A, et al. Inhibition of insulin-specific autoreactive T-cells by sulphatide which is variably expressed in beta cells. Diabetologia 1999;42:1212–1218 [DOI] [PubMed] [Google Scholar]
- 14.Zinselmeyer BH, Vomund AN, Saunders BT, Johnson MW, Carrero JA, Unanue ER. The resident macrophages in murine pancreatic islets are constantly probing their local environment, capturing beta cell granules and blood particles. Diabetologia 2018;61:1374–1383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lindley S, Dayan CM, Bishop A, Roep BO, Peakman M, Tree TI. Defective suppressor function in CD4+CD25+ T-cells from patients with type 1 diabetes. Diabetes 2005;54:92–99 [DOI] [PubMed] [Google Scholar]
- 16.Stamatouli AM, Quandt Z, Perdigoto AL, et al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes 2018;67:1471–1480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Samoa RA, Lee HS, Kil SH, Roep BO. Anti-PD-1 therapy-associated type 1 diabetes in a pediatric patient with relapsed classical Hodgkin lymphoma. Diabetes Care 2020;43:2293–2295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Battaglia M, Ahmed S, Anderson MS, et al. Introducing the endotype concept to address the challenge of disease heterogeneity in type 1 diabetes. Diabetes Care 2020;43:5–12 [DOI] [PMC free article] [PubMed] [Google Scholar]