Abstract
Targeting the small intestine employing nanotechnology has proved to be a more effective way for site-specific drug delivery. The drug targeting to the small intestine can be achieved via nanoparticles for its optimum bioavailability within the systemic circulation. The small intestine is a remarkable candidate for localized drug delivery. The intestine has its unique properties. It has a less harsh environment than the stomach, provides comparatively more retention time, and possesses a greater surface area than other parts of the gastrointestinal tract. This review focuses on elaborating the intestinal barriers and approaches to overcome these barriers for internalizing nanoparticles and adopting different cellular trafficking pathways. We have discussed various factors that contribute to nanocarriers’ cellular uptake, including their surface chemistry, surface morphology, and functionalization of nanoparticles. Furthermore, the fate of nanoparticles after their uptake at cellular and subcellular levels is also briefly explained. Finally, we have delineated the strategies that are adopted to determine the cytotoxicity of nanoparticles.
KEY WORDS: targeted drug delivery, nanocarriers, small intestine, intestinal barriers, cellular trafficking, cytotoxicity
INTRODUCTION
The oral route is always considered the appropriate and reliable pathway for delivering therapeutic agents due to increased patient compliance, particularly in chronic illness. It ensures convenience, facilitates self-administration, and offers excellent flexibility in dosage regimen as the highly sterile conditions are not required for oral products, their manufacture leading to a decline in production costs (1). Moreover, the oral route seems to have interesting physiological reasons as the surface area (300–400 m) of the gastrointestinal tract (GIT) is extensive for drug absorption through absorptive epithelial cells (2–6).
Despite the numerous advantages of oral delivery, it also possesses some drawbacks such as drug requires crossing manifold compartments of the human body before it reaches the systemic circulation, which is a challenging task (7). Furthermore, after ingestion, the drug faces the harsh acidic pH of the stomach before it arrives at the small intestine via the duodenum, which is said to be the central enzymatic digestion machinery of the human body (8). One of the most promising approaches to overcome the obstacles mentioned earlier of oral drug delivery is nanomedicine use.
Nanomedicines can be defined as either nanoscale (< 100 nm) therapeutic agents or imaging agents that result in systematic enhancement, shielding, specific targeting, controlled release, or reduced drug-induced cytotoxicity (9). The significant clinically used nanomedicines consist of liposomal, polymer-based, protein-based, silica, iron oxide, and gold nanoparticle (NP) (10). The bioavailability of orally delivered drugs is also enhanced by nanocarriers (11,12). Nanoparticles own numerous benefits in oral drug delivery; subsequently they present a large surface area for interactions with the gastrointestinal tract and might be altered in different ways in order to handle the barricades related to oral delivery. The size, shape, and surface chemistry of nanoparticles can significantly impact cellular uptake and treatment (13).
The vital intention of drug delivery based on nanotechnology targets the specific organs, tissues, and cells. Therefore, precise targeting of nanocarriers across the cell membrane is of primary importance (14). Thus, to achieve a more effective drug delivery mode, it needs to thoroughly understand the formulation of an optimized nanomedicine that can target the small intestine. If the intestine is targeted, it will help prevent drug degradation and enhance the small intestine absorption rate that highly improves the impact of orally administered pharmaceuticals. Although by adopting this route, the nanocarriers would more efficiently survive through a healthy environment of GIT but to reach the bloodstream, it has to be overcome intestinal barriers of the small intestine. The internalization of nanoparticles can be achieved by crossing intestinal obstacles such as the mucus layer, the epithelial layer, and tight junctions. The physical and chemical characteristics of nanoparticles also affect their internalization. After their cellular uptake at the cellular and subcellular levels, the fate of these particles is an extensive study (15,16). For obtaining an optimized nanodrug delivery system, cellular-induced cytotoxic response should also be adequately studied (17).
This review article allows understanding the uptake pathways adopted by the nanoparticles for internalization and briefly discusses the barriers and the ways they adapt to restrict the theoretically estimated delivery of nanoparticles and the various approaches adopted by the researchers to overcome these obstacles. The effect of surface chemistry of nanoparticle is quite complicated on their cellular uptake. This discussion would give brief information on the delivery of nanoparticles to the targeted cells and even their interaction with cellular organelles.
INTESTINAL BARRIERS
The most challenging obstacle of the human body for nanoparticles to reach systemic circulation is to overcome the intestinal barriers that include intestinal mucus, epithelial cells, cellular organelles, and narrow junctions between the cells. The villi are considered the main body of the digestive tract’s various structures, enhancing the intestinal absorptive surface area that is about 300–400 m2 (18). The cells, for example, enterocytes, microfold cells, and goblet cells, are closely linked via tight junctions. The portions of the small intestine are categorized as duodenum, jejunum, and ileum. Naturally, it tends the absorption of nutrients.
Furthermore, villi are sheltered with mucus layers having a variable thickness (2). The vital intention of nanotechnology is targeting the specific organs, tissues, and even cells. Therefore, precise targeting nanocarriers’ translocation across the cell membrane is of primary importance (19). The drug has to go through the following barriers
Mucus barrier
Tight junctions
The Mucus Barrier
The mucus layer is considered the primary physical barrier that is usually a hydrogel-type structure which consists of large glycoproteins that belong to the mucin family (20–23). The goblet cells are responsible for mucus secretion, which is also used to shield epithelial cells from the damage caused by bacterial interactions or ingested food (24). In the small intestine, goblet cells’ prominent mucin is MUC2 (25–27). The small intestine consists of a 10- to 200-μm-thick loosely adherent layer from the jejunum to the colon, whereas the average production of mucus is “1” kg per day in a healthy adult (28). The external loosely adhered layer is comparatively less thick (20,29). The mucus layer is formed of parallel and stacked sheets consisting of mucin molecules present between the villi in the small intestine. The mucin two–based sheet has its protein attached to the three neighboring mucin cells building a hexagonal mesh structure (27,30,31). In addition to mucin, many enzymes are also present in the mucus layer that may pose a risk of degradation.
Though, despite being undisputedly extremely essential for the human intestine shielding, it marks the primary barrier to oral nanoparticles (32). Numerous nanomaterials get immobilized by the mucus secretion and cannot reach the epithelial cell layer. Notably, a functionalization with synthetic resin polyethylene glycol (PEG) chains (33,34), the mixture of anionic and cationic charges (35), and therefore the application of self-nanoemulsifying drug delivery systems (SNEDDS) are practical strategies to penetrate the mucosal barrier (36). Anhydrous homogenous liquid mixtures consisting of oil, surfactant, drug, and co-emulsifier having the property of forming oil-in-water nanoemulsion are known to be SNEDDS.
The mucus shows hydrophobic bonding with the nanoparticles and proteins (37). Moreover, the charge-bearing mucin proteins hold the charged particles in the mucus layer due to interaction between the charges (38). The charge density of intestinal mucus mesh can rely upon the intestine’s pH as well as ionic strength of intestinal contents and is also affected by the chyme components within the small intestine. The ionic strength and osmolality changes from hypotonic to isotonic to hypertonic environment among diminutive spaces in the gut subsequently taking nutrients (39).
A greater charge interaction occurs between nanoparticles and mucus in the hypertonic environment and lesser in hypotonic conditions (39). The particle-mucus interactions are partially dependent on feeding state as denser and more impenetrable mucus is related to postprandial levels of calcium, lipids, and bile acids (40). It was observed that dietary and biocompatible polymers could compress the mucosa of the mouse colon, resulting in tightening the mucus structure by up to 80% (41).
However, the outer loosely bounded layer causes the immobilization of nanoparticles, proteins, or pathogens, leading to the digestive tract’s rapid clearance by shedding the mucus layer. However, small solute molecules, such as nutrients, diffuse without hindrance through the mucus layer (39). It was observed the surface of densely charged neutral hydrophilic nanoparticle showed interactions with mucus to a limited extent. In a study, Olmsted et al. perceived densely charge-bearing particles but has neutral, hydrophilic surfaces with mesh size almost about 100 nm (32,39,42). The mucus-penetrating particles (MPPs) were prepared by considering chemical properties of mucus with the capability of quick transferring through the cervical mucus sheet (43,44); for example, either the surface of MPP can be decorated by functionalizing with low molecular weight PEG polymeric chains or particles could be developed with fragile mucosal interactions. The nanoparticles get trapped in the secreted mucus by decreasing the PEG chain density.
Furthermore, the surface coverage has long PEG chains incapable of entering the mucus mesh, almost certainly because of the predicament among the stretched PEG chains and the mucus layer. The mucus-penetrating particles were observed to be bound to the epithelial layer’s surface whereas conventional particles were arrested within the external part of the mucus layer (44). A hydrophilic surface is characteristic of numerous soluble proteins that limited their interaction with mucus. For example, albumin protein has a negative charge; therefore, nanoparticles functionalized with serum albumin show repulsive and weak mucus interactions (45).
Theoretically, the concentrated and controlled delivery can be achieved by the nanoparticle-mucus binding. A feasible technique is used to transfer small and stable molecules in the enterocyte locality. In contrast, practically, the superficially bonded external mucus sheet and peptidases of the small intestine will be a substantial challenge to that strategy. Therefore, MPPs are considered a practical approach, but their worth after oral delivery is still under research (39,46).
Another strategy involves modifying nanoparticles with proteolytic enzymes such as papain, apparently enhancing the mucosal penetration by the breakdown of the mucoglycoprotein substructures (47). The most crucial barrier next to the mucus layer penetration is internalizing the foremost layer of epithelial cells. When the nanoparticles deliver at the apical pole of the epithelial layer, there arise dual opportunities for crossing the basal side. It involves paracellular transport that opens tight junctions, and transcellular delivery occurs among epithelial cells having no cellular uptake. For example, chitosan is considered a favorable polymer for achieving paracellular uptake, which can open tight junctions reversibly (48).
Tight Junction
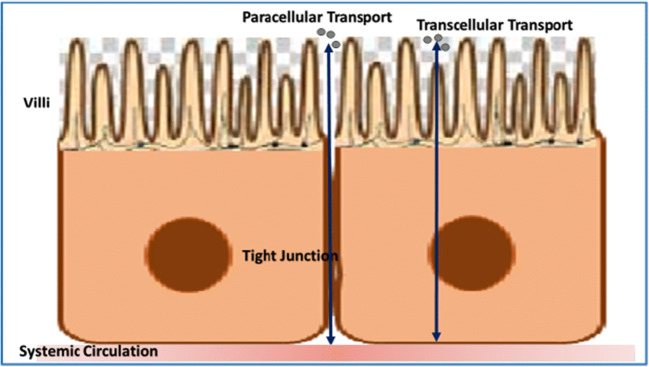
The seal between the two attached epithelial cells that resist paracellular trafficking of small molecules such as water, ions, and solutes is known as a tight junction as shown in Fig. 1 (49,50). They are generally situated in a circle near the apical side of the cell. The separation of the basolateral and apical poles of the epithelial membrane is due to these junctions. Tight junctions are formed by branching strands which further consist of layers of transmembrane proteins having extracellular territories primarily that may be 27-membered claudin family and occludin (51).
Fig. 1.
Tight junction; slit between the two attached epithelial cells that resists paracellular trafficking of small molecules, i.e., water, ions, or solutes
Claudins have two different groups, including the barrier-forming group or the pore-creating group, which control the selective transport of small solute molecules through the epithelium. Moreover, they occur in different sets in variable tissues, and it is indicated that the proteins are liable in various tissues for the paracellular uptake including the small intestine (51). The loops are formed by extracellular parts of these proteins that get attached to conforming loops of neighboring cells, resulting in a seal among cells (52,53). And tricellulin is another type of tight junction protein that occurred between three adjacent cells (49).
Modulation of tight junction permeability has usually adopted by two forms of approaches: a seemingly less controlled tight junction modulation, making leaks that enable the passage of large molecules, and an additional controlled tight junction modulation. To create leaks, absorption enhancers or surface-active agents have been used. For example, fatty acids having medium chain length and their derivatives cause reversible leaks within narrow concentration intervals to occur between tight junctions (54,55). A controlled tight junction modulation appears to be an extra attractive approach than permeation enhancers to increase paracellular amide permeability. Still, the studies showed that it results in more narrow open pathways than absorption enhancers and seems therefore not to be a way to improve nanoparticle delivery.
It was observed by the scientist in animal studies that elevated concentrations of the peptides and also of insulin were required for significant modulation. It remains to be shown if this approach holds promise when scaled up to larger species (56), thus limiting the transport of nanoparticles (57,58).
INTERNALIZATION OF NANOCARRIERS
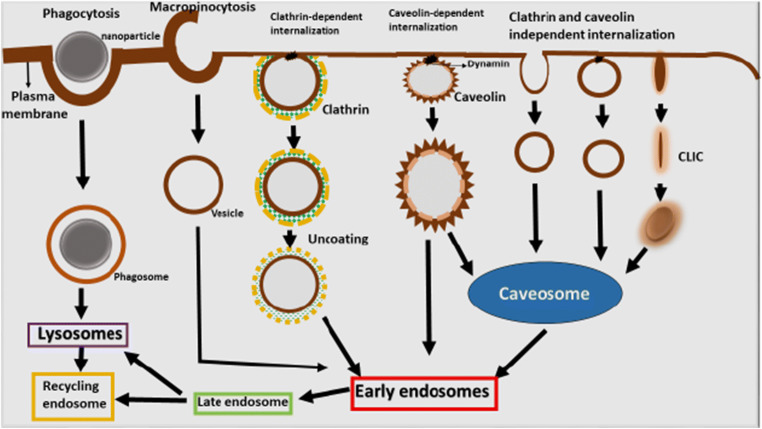
After the nanoparticles overcome the various cellular obstacles, the next challenge is the internalization of nanocarriers in the intestinal cell. Commonly, multiple pathways are adopted by the endothelial and epithelial cells including the uptake of the nanocarriers and interaction with cytosol and other cellular organelles leading to the fate or ejection of nanoparticles from the cell as shown in Fig. 2. Endocytosis is a process of uptake mechanism which is further characterized as pinocytosis and phagocytosis (59). The term endocytosis comprises the development of new vesicles from the cell membrane along with the introduction of proteins, lipids, and extracellular fluids whereas pinocytosis includes the internalization of liquid. At the same time, phagocytosis refers to the incorporation of solid particles (60,61).
Fig. 2.
Diagrammatic illustration of nanocarriers trafficking in the intestinal cell by adopting multiple pathways of endocytosis
The nanoparticles are transferred by intestinal cells through various pathways including:
Clathrin-mediated pathway
Caveolin-mediated pathway
Clathrin- and caveolin-independent pathway
Macropinocytosis (62)
Nature of the adopted endocytosis pathway is estimated by dimensions of the endocytic sac, the properties of the constituents, and the method used for the development of vesicles. The heterogeneity in the endocytic mechanism is responsible for the delivery of nanocarriers to various intracellular localities (63,64).
Clathrin-mediated uptake is occurred by specific receptor-ligand interaction and is also known as non-specific endocytosis. In this pathway, transmembrane receptors produce coated pits and components covered with cytosolic protein retracted to form vesicles that are probably assisted by GTPase dynamic protein. The dynamin protein encloses the neck of lately developed invagination (65). Hydrophobic interaction with the cell membrane is carried out by non-specific endocytosis pathway. The vesicles are 100–120 nm in size. The uncoated vesicles either resulted in the development of early endosomes or reprocessed to the surface of the plasma membrane. Then, these vesicles are converted to developed endosomes and then to lysosomes or multivesicular bodies. The small density lipoprotein is uptaken and transfers by this pathway. Clathrin-dependent endocytosis is also adopted by chitosan and PLGA nanoparticles (66).
Caveola-mediated endocytosis pathway is mainly regulated and comprises complex signaling pathways. In the interior plasma membrane, flask-shaped invaginations are formed known as caveolae which consist of glycophosphingo lipids and high cholesterol level. Caveolin-1 is a dimeric protein that gives shape and structure to the vesicles (67). GTP are dynamin mediated the splitting of caveolae from the membrane. The vesicle size is approximately 50–100 nm. The formation and deposition of caveolae are regulated by cavin-coated proteins besides with caveolins. The caveolae of endothelial cells take part in the trans-endothelial mechanism, which is under investigation for the transfer of nanomaterials to subendothelial tissues. The neutral pH of caveosomes avoids the interaction between caveosomes and the hydrolytic environment. The site-specific delivery of therapeutic material is explored via the uptake of caveosomes by the Golgi apparatus and endoplasmic reticulum (8,68,69).
Clathrin- and caveola-independent endocytosis mechanism is not mediated by receptors or induced stimuli. The internalization and vesicle formation is independent of coat proteins, but actin and its related protein are significant for the development of vesicles. After the delivery of a therapeutic agent to the endosome, they reach the Golgi apparatus or may be recycled by the plasma membrane. In vaccine development, the polyplexes of self-branched and disaccharide relieved nanoparticles composed of chitosan oligomer have been discovered to transport DNA for site-specific delivery and in vivo vascular imaging (8).
The macropinocytosis is autonomous of the action of either receptor or molecular ligand for their activation. The protrusions are formed at the outer part of the plasma membrane under the action of actin. These protrusions show further merging with the cell membrane and result in the synthesis of macropinosomes and help in the delivery of bulks of extracellular fluid. The macropinosomes are > 1 μm in size, typically with irregular shape (70). In comparison, the macropinosomes are seemed to be affected by cytoplasmic pH (71). They either get fused with the compartment of lysosomes or may reutilize their content; therefore, macropinosomes also represent a fate like endosomes. The fragments of an apoptotic cell, bacteria, and viruses are generally ingested by this pathway (72).
Factors Affecting the Internalization of Nanoparticles
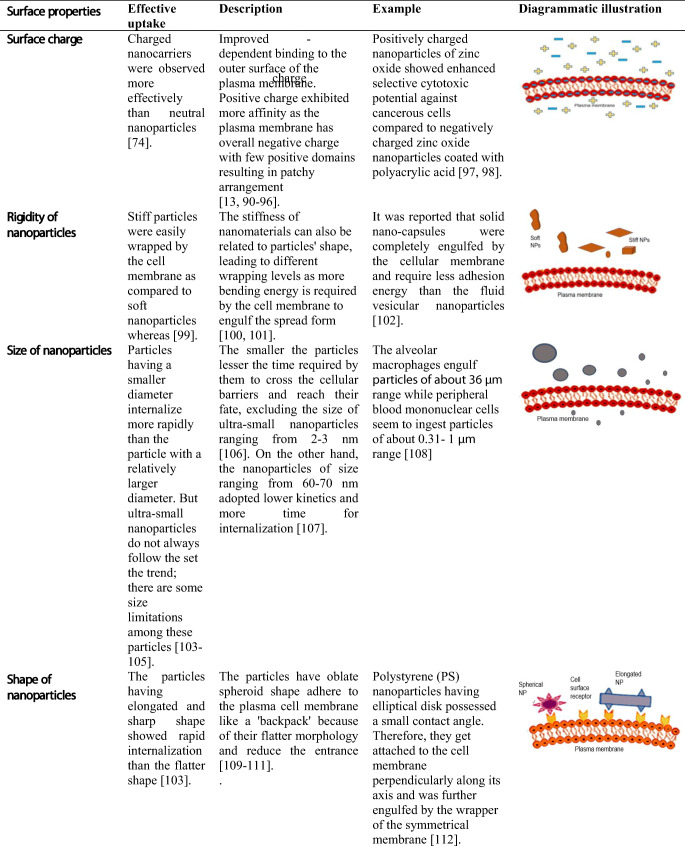
Although nanoparticles are commonly indicated for their enhanced therapeutic and diagnostic effects, they need modification in their properties to achieve better internalization. Moreover, the intestine environment, along with intercellular trafficking and organelle interaction, also affects internalization. The researchers have made several attempts to obtain the best optimal results to target the site of action and minimize the side effects of therapeutic ingredients. These modifications help them in cellular internalization and crossing the barriers and show a high impact on future health care and improved clinical benefits. Their physical and chemical properties most commonly influence the internalization of nanoparticles. These properties include size, surface charge, shape, rigidity, and functionalization of these particles with receptors (73).
The nature of charge present on the nanoparticles has a high impact on the internalization through intestinal epithelial cells. For instance, Mei et al. modified the nanoparticles with a cationic surfactant that create a positive charge, which ensured enhanced retention time at the cellular membrane. The uptake mechanism was boosted, resulting in the high bioavailability of therapeutic agents (74). Similarly, the cancerous cells impart a negative charge due to the overexpression of phospholipids that negatively affect normal cells.
The studies showed that nanoparticles’ stiffness and softness could alter the cellular uptake and their targeting and biodistribution. It was also suggested that rate of cellular uptake is directly proportional to their rigidity since they show latent receptor-mediated diffusion and comparatively more contact surface area with the plasma membrane (75,76). Though nanoparticles’ rigidity can be determined by the relationship among stress and strain given by Young’s modulus, even it is challenging to measure the particles of micro- or nanosize (Yi and Gao 2017).
As far as the size of the nanoparticles is considered, it is a vital feature for trafficking through the cellular compartments. In the endocytosis pathway, the cellular membrane enfolded the particles, so the required considerations for membrane folding the radius of curvature cannot form insignificant, failing to capture the infinitely small particles (77,78). Moreover, the optimum size for nanoparticles necessary for the effective endocytosis pathway is observed by cell nature (79–83).
It is reported that as the nanoparticle comes in contact with the cell membrane, the contact angle showed a significant effect on endocytosis (84,85). Therefore, the different shapes of nanoparticles lead to diverse angles and consequently affect the internalization of nanoparticles (86,87). But, the internalization of spherical nanoparticles is not dependent on their contact angle due to their symmetrical shape. Moreover, engulfment failure occurs when the contact angle and points of attachment to the cell membrane enhance by diminutive axis, i.e., right angle to the membrane (88). Likewise, the polymeric nanoparticles had shown both a high rate of attachment and less internalization rate in comparison to spheroidal and oblate ellipsoidal nanoparticles having a small contact angle having major axis 0.35–2.0 μm and minor axis 0.2–2.0 μm (89).
Some of the important features of the factors are summarized past studies in Table I, showing the researchers’ better options to develop suitable nano formulation.
Table I.
Factors Affecting the Internalization of Nanoparticles
Functionalization to Target Receptors
The advance research is additionally based on enhancing the rate of transcytosis by functionalizing nanoparticles with various proteins. Such as, in Calu-3 airway epithelial cells, a bioconjugation of polymeric NPs with Fc part of immunoglobulin 'G' a constant portion of 'IgG' successfully enhances the rate of transcytosis (113). A study observed 16HBE cells uptake having 60 nm in size and positively charged polysaccharide nanoparticlessignificantly enhance endocytosis after cholesterol depletion. To follow, filipin is supplemented to the medium of cells subjected to nanoparticles, which substantially lower the rate of exocytosis (114).
The transcytosis could be significantly increased in Caco-2 cells by 2-fold via functionalization. The particles were modified with Fc fragments that quickly attached to the neonatal Fc receptor in the epithelium of the intestine. It was observed that Fc-bonded nanoparticles passed over the intestinal barrier in both in vitro experiments by using human epithelium and in vivo in mice (who also express FcRn), resulting in enhancing concentration levels in different organs of the body (115).
The decoration with folic acid to target the folate receptors is considered an attractive opportunity for oral drug delivery. In Caco-2 cells, PLGANPs functionalized with folic acid vastly augmented transcytosis proficiency and are utilized to increase oral bioavailability (116). Subsequently, the cellular uptake of macromolecules is triggered by the interaction of receptors on the cell surface and the ligands (opsonins). Examples of some significant receptors used in phagocytosis include Fc receptor family for IgG (FcγRI, FcγRIIA, and FcγRIIA), the complement receptors (CR1, CR3, and CR4), and α5β1 integrin (117). For example, Fowler and his coworkers revealed that nanoparticles containing insulin targeting the FcRn receptor achieved a prolonged hypoglycemic response in mice when administered orally at a clinical dose. Correspondingly, a bioconjugation with vitamin B12 having a size range of 50-nm or 100-nm nanoparticles not only enhances the trafficking by 3-fold to 7-fold but also results in the adjustment of the pathway and prohibited the nanoparticle transport to lysosomes (118). Roger et al. studied the modification of particles with folic acid to target the folate receptors. It was hypothesized that the trafficking of nanoparticles and their cellular uptake could be additionally enhanced by targeting the folate receptors present on the epithelial cells of the intestine (116).
NANOPARTICLE-INTRACELLULAR ORGANELLE INTERACTION
The intestinal cells consist of a diversity of intracellular organelles and compartments. The nanoparticle interaction with cellular systems and their localization, recognition, and quantification in intestinal cells are of significant prominence to comprehend the impact of physicochemical factors that might stimulate the potential interaction with a definite cell type and their responses (119). Once the nanoparticle enters into the cell, the evaluation of their distribution in different cellular compartments including mitochondria, cytosol, endosomes, lysosomes, or the nucleus gave information about their possible biological effects (120,121), along with indications for the sake of designing of nanocarriers to achieve successful cell targeting and drug delivery. Targeting intracellular organelles or compartments with nanocarriers is widely used for diagnosis and therapy.
It is important to find out whether nanoparticles adhere to the exterior side of the plasma membrane or intracellular, in addition, to extricating the internalized nanoparticles (122–125). There is a definite indication that cellular uptake of nanoparticles is most commonly achieved by the endocytic pathway. Conversely, nanoparticles might be degraded, released, and delivered to new cells that will impact the activity of nanoparticles and cellular responses (126–128). It has been hypothesized that endocytosis of nanoparticle depends not only upon it but also on cell type (129), and for example, intestinal epithelial cells expose altered uptake processes in contrast to phagocytic cells such as macrophages (130).
Moreover, the organelle dysfunctions resulted in different human diseases as their functional activities are closely related to cellular growth, proliferation, differentiation, and cell death. Hence, the nanoparticle design for targeted delivery to the subcellular organelles is of remarkable consideration (131). For the development of optimized nanoparticles that can cross the barriers as well as give significant targeting, peer knowledge about the interface of nanoparticles with the cell and its organelles is required.
Interaction with Mitochondria
The intracellular targets for nanoparticles and therapeutic agents also include mitochondria for the identification, prevention, and treatment of different human diseases, for example, neurodegenerative diseases, cancer, ischemia-reperfusion injury, obesity, and diabetes, as mitochondria are essential for the fabrication of energy. Therefore, it can be utilized for site-specific delivery in tumor or neurodegenerative disease. The idea of the formulation of mitochondriotropic depended on carriers to deliver cargo either to protect the cell or to encourage cell death that has been studied by different researchers (132,133).
Qu and his coworkers combined triphenylphosphonium (PPh3) to the surface of mesoporous silica nanoparticles loaded with doxorubicin (134). It was seen that the combination of delocalized cationic charge at surface ligand and three lipophilic phenyl groups enabled nanoparticle delivery across the mitochondrial membrane. These modified nanoparticles take about 8 h to release from liposomes and interact with mitochondria that led to a decline in levels of ATP in the cell, hence causing mitochondrial dysfunction, which leads to decrease cell feasibility up to 30% when exposed for 24 h in vitro. One of the crucial causes of neurodegenerative diseases, for example, Alzheimer’s disease, is mitochondrial dysfunction. Recently, Kwon et al. fabricated a cerium oxide nanoparticle–based delivery system that can overturn the onset of neuron damage by isolating inert oxygen species generated by improper functioning of mitochondria. It was concluded that cerium oxide nanoparticles which were functionalized with PPh3 and DSPE-PEG2000-methoxy could reuse oxygen atoms and limit further neuronal death in vivo (135). A method was reported by which small spherical gold nanoparticles were formulated to target the mitochondria (136). A recent method was designed for the preparation of polyoxometalate peptide-nanoparticle bioconjugates and the identification of their transport behaviors. The functionalization of the nanoparticle surfaces with peptides enables the significant trafficking of the nanoparticles to the mitochondria (137). Yamada et al. prepared a lipid derivative that is combined with a mitochondrial targeting signal peptide (MTS), which authorizes the targeted uptake of some protein types to mitochondria (138).
Jeena et al. synthesized novel peptide amphiphiles focusing on the mitochondrial targeting that was modified to ensure self-assembly upon gathering in mitochondria (139). The amphiphilic peptides comprising β-sheet develop blocks, conjugated to PPh3 in HeLa cells, observed to primarily accumulate in mitochondria, then resulted in fibrils because of high local concentration. This development of the fibrils disconcerted mitochondrial membranes forming the leaking of mitochondrial matters into the cytoplasm and consequent apoptosis. Forthcoming work utilizing this fabrication would necessitate targeted cell accuracy so as not to stimulate mitochondrial damage in normal healthy cells.
Interaction with Endoplasmic Reticulum
The proper folding and delivery of proteins can also be achieved by the endoplasmic reticulum (137). Furthermore, it is considered a critical site for loading of peptides into MHC primary histocompatibility complex class I molecules and following cytotoxic T cell responses (140). Thus, the endoplasmic reticulum promotes the proper functioning of the cell and its organelles. Cubillos-Ruiz et al. demonstrated exactly how endoplasmic reticulum stress in tumour-mediated dendritic cells endorses cancer growth and diminishes antitumor immunity (141). Estimation of an increment in endoplasmic reticulum stress factor, i.e., XBP1, was related to a shortened dendritic activity which also decreases T cell–dependent immunity. By using nanoparticles PEI (polyethylamine), they were observed to enclosed definite siRNAs.
Moreover, the phagocytic dendritic cells were observed to engulf these nanosized complexes that encouraged approximately 65% gene silencing of XBP1. Silencing of endoplasmic reticulum stress in cancer-related dendritic cells employs intraperitoneal injections of the siRNA-PEI-based nanoparticles. The survival of mice was amplified with aggressive orthotopic ovarian cancers by the use of formulated nanoparticles. This innovative mechanism is responsible for the activation of cell-dependent antitumor immunity by the specific intracellular nanoparticle.
It was revealed by a time-course sampling of gold nanoparticles transported KDEL peptides were immediately confined to the endoplasmic reticulum in the first 5–15 min. In contrast, the majority of peptides were localized to the endoplasmic reticulum within 1 h (142). In this review, it is demonstrated that a delivery system for miR-29b employs PEI-capped gold nanoparticles targeting the endoplasmic reticulum to synergistically stimulate osteoblastic distinction (143).
Interaction with Golgi Apparatus
Golgi apparatus is one of the vital organelles for nanoparticle-based interventions. This cellular organelle is vital for the transport of post-translational amendments of freshly prepared proteins (144). Yu et al. worked on the pathological role of the Golgi apparatus to treat the tumor cells, which are defined as the abnormal growth of cells (145). For this achievement, both COX-2 inhibitor and Brefeldin A were encapsulated into PLGA-PEG nanoparticles. The COX-2 inhibitor such as celecoxib enclosed within the Golgi apparatus, whereas Brefeldin reserved protein delivery from the endoplasmic reticulum to the Golgi apparatus. The nanoparticle loading these two trivial molecules competently impaired the Golgi apparatus in 30 min of time course in vitro in murine metastatic breast tumor cells and displayed increased cytotoxicity. Besides, the combined transfer of these small molecules was detected to reduce the appearance of proteins related to metastasis.
Likewise, the experiments exposed that the Golgi apparatus did not employ the exocytosis in any of the verified cell lines. But, pharmacological inhibitors for microtubule formation and actin polymerization both repressed exocytosis, suggesting a purpose of both for the lysosomal delivery and cell membrane fusion (146). Fluorescent molecules, silica nanoparticles, and carbon quantum dots all were observed to target the Golgi apparatus which were decorated with l-cysteine. l-Cysteine-based chiral carbon quantum dots have the ability to target the Golgi apparatus (147).
Interaction with the Nucleus
By modifying the physicochemical properties of nanoparticles, the cellular organelle targeting can be achieved. For example, a size-dependent association for nanoparticle targeted to the nucleus for therapeutic interventions was observed by many researchers (148–150). The nucleus is a vital organelle to be targeted; its pores possessed a size range of 30 nm. It was observed that nanoparticles of large size labeled with particular nuclear localization order could reach the nucleus (151). Larger nanoparticles with a specific surface can access the nuclear content during mitosis when the nuclear membrane breaks down (152).
It was perceived that gold nanoparticles with a usual diameter of about 4 nm were capable of reaching the nucleus of breast cancer cells, despite the fact the nanoparticles with an average diameter of about 14 nm cannot enter the nuclear envelope and stayed dispersed all over the cell cytoplasm.
The targeting of the cell nucleus by nanoparticles has been described with two different pathways, including active transport of nanoparticles through the complex of nuclear membrane pore, which is assisted by nuclear localization signals (NLS). NLS is an amino acid sequence that “tags” a protein for import into the cell nucleus by nuclear transport. Remarkable examples of these NLS categorizations include small peptides that can be attached to cytoplasmic importins, for instance, importins α and β, that are situated in the perinuclear site (153). After fusing to cytoplasmic importins, nanoparticles with average diameters of 50 nm have been testified to cross the nucleus by adopting active transport by means of the nuclear pore complex (154). A second mechanism of nanoparticle access into the nucleus is achieved by passive diffusion of cytoplasmic nanoparticles via the open channel present in nuclear pore complex. These open channels vary in their diameter in the range of 6–9 nm (148,150,153,155). For adopting passive diffusion, the nanocarriers should be smaller enough to pass the nuclear pore. In recent research, it was reported that functionalized, positively charged, gold nanoparticles with a size range of 14 nm showed higher uptake as compared to particles with a size of 2 nm or 5 nm in diameter (156).
Huo and coworkers determined a size-dependent basis for nuclear uptake of gold nanoparticles in the breast cancer cell and observed that despite better cellular uptake for the larger nanoparticles, the 2-nm nanoparticles delivered DNA payloads to the nucleus 20× times more efficiently than their 14-nm counterparts (157). Nanoparticles with a calculated diameter of 2–6 nm were observed within the nucleus, whereas those larger than 10–16 nm were present outside the nucleus compartment. Based on that observation, the scientists tried to target triplex-forming oligonucleotides to the nucleus with the nanocarriers of 2 nm size which silenced the transcription factor c-myc promoter which is a promising target in anticancer therapy up to 50%. Some studies support the notion of size-dependent particle delivery to the nuclear membrane, as described for gold nanoparticles functionalized with PEG and polyarginine (107).
Tang et al. observed that nuclear delivery of quantum dots can be enhanced by covering them with NLS sequences at 20% density (158). Furthermore, it was confirmed that cellular and nuclear internalization was size-dependent using semiconductor quantum dot nanoparticles.
Interaction with Lysosomes
Lysosomes are considered oval or spherical or may be tubular intracellular vesicles with a pH of 4.5–5 (159). Their size is dependent on the cell type, which may vary between < 1 mm and microns (160). The enzymatic degradation is their primary physiologic function moreover recycling of cellular compounds and other foreign molecules (161). Additionally, they are also involved in repairing of the plasma membrane, signaling, secretions, and processes of energy metabolism (162).
Although they are essential for eukaryotic cells, they are one of the main barriers for transcytosis of nanoparticles. Concerning the mainstream of the nanodelivery system, their ability is abbreviated as EEDD (accessible entrance, difficult discharge). Thus, it was concluded that the endocytosis pathway is comparatively easier than exocytosis. When the nanoparticles are uptaken by the cell, the majority of the cells are subjected to an endolysosomal path that includes the delivery from early to late endosome their fate as maturation or fusion with lysosomes (163,164).
As the nanoparticles reach the acidic compartment of lysosomes, they get exposed to high ionic strength and various potent proteolytic enzymes can also interact with them (165). The nanoparticles get degraded in such a harsh environment which may also alter their colloidal stability. The enzymes also possess the ability to dissolve the outer polymer coating, which was done for improving their resilience. Kreyling et al. decorate radioactively labeled indium to gold nanoparticles and observe in vitro delivery in endosome or lysosomes and how the polymeric coat shell gets separated from the inorganic gold nanoparticle. In vivo study of coated nanoparticles showed they were localized in liver cells. The polymeric coat was subjected to exocytosis, then undergoes renal filtration, resulting in urinary excretion (166).
It was also concluded that fluorescently labeled carboxylated polyvinyl alcohol gold nanoparticles may result in conformational alterations in intracellular lysosomal territories leading to aggregation of nanoparticles moreover appeasing of the fluorescence encodes located on the surface of polymer (167). Ma and colleagues fabricated nanoparticles of gold having various coatings such as coating with human γ-globulin (HGG), human serum albumin (HSA), or human serum fibrinogen (HSF). They examined their cytotoxicity when they get exposed to HeLa cells. It was observed that the rate of degradation of protein corona within the lysosomes is effected by the composition of the corona that also influences the aggregation and the cytotoxicity response of the nanoparticles (168). Besides, the lysosomal embedded metallic nanoparticle dissolution rate is considered they can reduce the concentration of intracellular nanoparticles moreover release of potentially toxic ions (169).
If the nanoparticles achieve the final destination of the eukaryotic cell and they cannot found any other escape, they either would fate by getting degraded by the action of enzymes or may be affected by the nature of the material and accumulated infinitely. However, this is not applicable for internalization through epithelial cells of the small intestine which the first challenge to be crossed by the nanoparticles. For example, in a study, polystyrene nanoparticles with the concentration of about 500 mg/mL were subjected to caco-2 cells and incubated for 24 h, resulting in endolysosomal structures up to 2 mm in diameter as exposed by cLSM and TEM reports. Future work should be performed to reduce nanoparticle accumulation and degradation. It is needed to avoid the endolysosomal pathway altogether or get fused with the plasma membrane (8). Currently, a study on hemagglutinin-2 and/or metformin highlighted the potential endolysosomal escape and the resulting positive increase for exocytosis associated with increased transcytosis efficiency (170).
The co-delivery of molecules to the specific target utilizing an efficient nanoparticle is a new era in nanomedicine technologies. The studies shed light on most recent strategies adopted for intracellular organelle targeting of nanomedicine and highlight efficiency and effectiveness of nanocarrier trafficking to the specific intracellular organelle.
CYTOTOXICITY OF NANOPARTICLES
Nanoparticles have been used in cancer treatment and autoimmune disorders. However, cytotoxic effects of nanoparticles on organs and normal cells are a severely limiting factor that hinders their clinical uses. Moreover, the differences of nanoparticles size, particle shape, and surface area are main components that have a vital concussion on their therapeutic or unwanted actions.
The size of nanoparticles is dependent on the following parameters: surface-to-volume fraction (ratio), sedimentation velocity, attachment efficiency, mass diffusivity, and deposition velocity (171). However, the cytotoxicity of nanoparticles has been affected by changes in NP size (172). Nanoparticle size shows an essential interaction with the biological system (173). Ion release rates are affected by the size of nanoparticles; if the size of nanoparticles becomes smaller, it will lead to more interaction with the surface of the membrane, hence producing higher cytotoxic effects through penetration into the cell (174). Generally, cytotoxicity of nanoparticles that depends upon the size of nanoparticles also be correlated, the ability of nanoparticles to enter the biological system.
Nanoparticle shapes also influence its toxicity. Nanoparticles are available in different shapes (e.g., filament, spherical, non-spherical, plate-shaped, and rod-like) (175). Internalization processes such as endocytosis and phagocytosis depend on the shapes of nanoparticles. Spherical nanoparticles show faster endocytosis than tubular nanoparticles (112). Non-spherical nanoparticles show more toxicity as they are vastly more exposed to blood flow (176).
Various biological aspects such absorption, plasma protein binding, colloidal behavior, and penetration over BBB are also affected by the surface charge presence on the nanoparticles (177). Negatively (−) charged nanoparticles show excellent absorption (through cellular mechanism) than positively (+) charged or neutral nanoparticles. Positively charged nanoparticles cause platelet aggregation and hemolysis, resulting in cytotoxicity through plasma protein (178). The concentration of nanoparticles also effects on cytotoxicity; the toxicity changed with changing concentration (179). For example, the 2 mg/mL silicon concentration on cells had produced cytotoxic action, but in 4 mg/mL, there was no cytotoxic effect observed (180).
Mechanism of Nanoparticle Toxicity
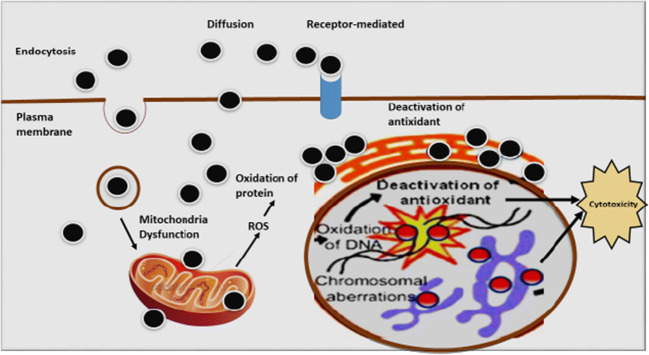
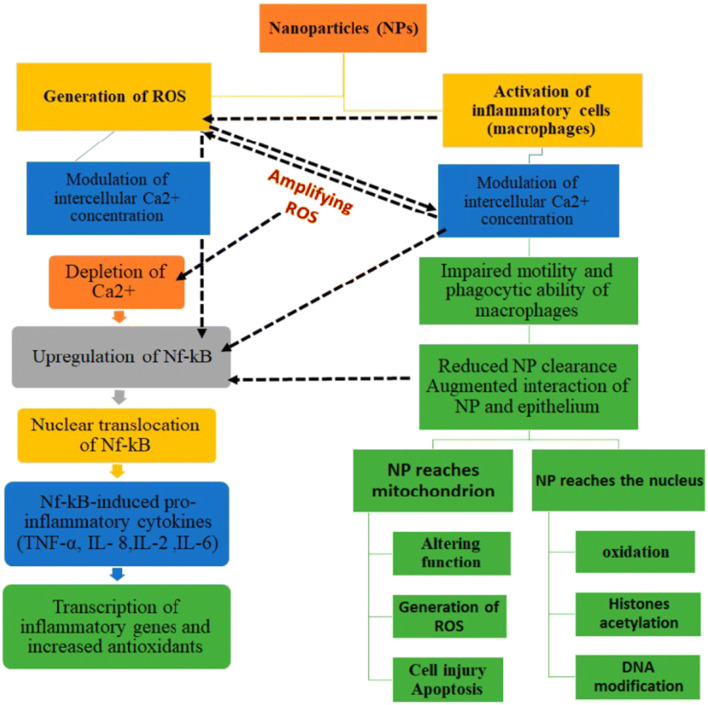
The primary mechanism of nanoparticle toxicity is oxidative stress through the production of reactive O2 species, as illustrated in Fig. 3 (181). The surface properties of the nanoparticles affect many of the biological responses. Growth, adhesion, and different ions are the primary cellular responses. Nanoparticles produce oxidative stress (OS) through physicochemical interaction. So, the nanoparticles have ion, which causes toxicity in cell surface and can be oppressed to treat (remove) cancerous cell (182). The more interaction with the surface of the cell membrane and higher diameter of nanoparticles also increases the toxicity level of nanoparticles. Generally, the cell membrane is made up of the dynamic comprising protein and extracellular polymeric chain (183).
Fig. 3.
Diagrammatic illustration of the mechanism of cytotoxicity caused by nanoparticles
Toxicity is predominantly through an increase of reactive oxygen species (ROS) levels in the cell, as nanoparticles enter the cell through endocytosis. Inflammatory factors such as tumor necrosis factor (TNF-α), II-6, II-8, and II-1 can also be increased by nanoparticles and finally cause mitochondrial damage (184–186). Mitochondria, endoplasmic reticulum, peroxisome, and microsomes are the essential component of intracellular organelles (ROS) in the cell membrane (187,188). Through the mitochondrial electron transport system, an intrinsic source of reactive oxygen species in mitochondria (20). With increased accumulation of calcium (Ca2+) in the cytoplasm, there will be activation of the mitochondrial electron transport chain and ROS generation occurs. Adenosine triphosphate (ATP) water and a small concentration of O2 are produced during the synthesis of mitochondria, resulting in the early stages of ROS production. Superoxide anion, the first reactive oxygen element that is produced by complex I (NADH ubiquinone oxidoreductase) and complex III (co-enzyme Q, bc1 complex, and ubiquinone/cytochrome c reductase) activity in the mitochondrial matrix and intermembrane space, is generated by mitochondria, respectively (189,190). Copper and manganese are those metals that catalyze the conversion of superoxide anions into hydrogen peroxide (191). Alpha ketoglutarate dehydrogenase and monoamine oxidase (MAO) are important sources of reactive oxygen species (ROS) (192,193). Many biological functions have been modulated by nanoparticle-induced ROS. Nanomaterial concentration is correlated with the occurrence of reactive oxygen species (ROS) and oxidative stress to which cell is exposed (173). Oxidative stress causes non-toxicity, as in alteration to cell motility, cytotoxicity, unregulated cell signaling, DNA damage, apoptosis, and cancer proliferation (194,195).
The mechanisms of nanoparticle (NP) toxicity is demonstrated in Fig. 4 and discussed briefly as:
With an increase of reactive O2 species (ROS), nanoparticles may cause oxidation.
Through the perforation process, nanoparticles may damage cell membranes.
Nanoparticles, through interfering cellular ion transport and inhibited cell division, cause damage to the components of the cytoskeleton.
Nanoparticles damage DNA and process of protein synthesis, thus producing mutagenesis.
Cell energy imbalance occurs by mitochondrial damage and by interfering metabolism of mitochondria through nanoparticles.
Nanoparticles also interfere with the formation of lysosomes, triggering the apoptosis, autophagy, and degradation of macromolecules.
Altered transport of substances in and out of cell and structure changes in membranes and proteins is also caused by nanoparticles.
Nanoparticles cause disturbance of the standard mechanism of cell metabolism and organ and tissue metabolism through activation and synthesis of inflammatory mediators.
Fig. 4.
Mechanisms underlying NP cytotoxicity [196]
Nanoparticle Cytotoxicity Assessment Method
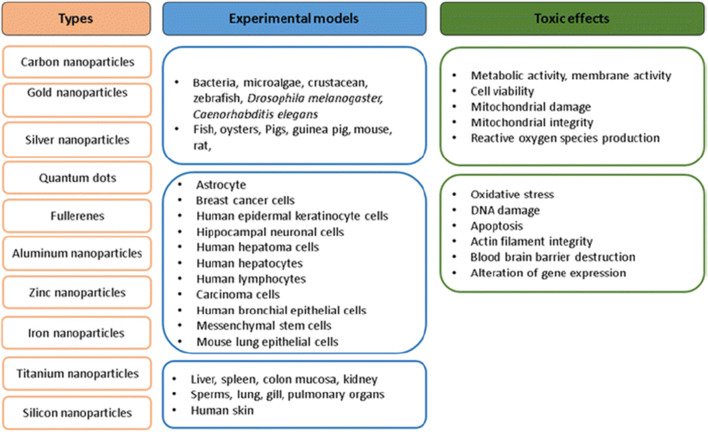
Cytotoxicity of nanoparticles can be assessed through different methods. Nanoparticle types, experimental models, and unwanted effects imposed by nanocarriers are shown in Fig. 5.
Fig. 5.
Nanoparticle types, experimental models used for the studies, and toxic effects of nanoparticles (Vinay Kumar et al., 2017)
In Vitro Assessment Assay
In vitro assessment method is further divided into the following assay: proliferation, apoptosis, necrosis, and OS assays. The proliferation assay measures cellular metabolism through the assessment of metabolically active cells, whereas apoptosis assessment is carried out by different assays including Annexin-V assay, Comet assay, TUNEL assay, and inspection of morphological changes (197,198).
The integrity of the membrane is measured by the necrosis assay. It is also used to determine the viability of the cells. Neutral Red and Trypan Blue are commonly used dyes for membrane integrity measurement (199). The production of reactive ROS and reactive nitrogen species (RNS) lead after exposure of nanoparticles (200). 2′,7′-Dichlorofluorescein diacetate (DCFDA) is a non-fluorescent probe. It is reactive to HO, RO, and ROO in the presence of cellular peroxidases (201). Lipid peroxidation assay is also used for the assessment of oxidative stress (202). In Table II, we elaborate the discussion on the toxicity imposed by the nanoparticles on rat, mouse, pig, guinea pig, human cell lines, and human.
Table II.
Different Assays and Dyes Used for Assessment of Nanoparticle Toxicity
| Assay name | Dye used | Cell lines | advantages | Cytotoxic effects | Reference |
|---|---|---|---|---|---|
| (1) Proliferation assays | (1) 3-(4,5-Dimethyl-thiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) is the most commonly used tetrazolium salt | Rat hippocampal neurons |
(1) Results easily, reproducible (2) Minimum model cell manipulation |
Avoided due to high toxicity | (203–206) |
| (2) Alamar Blue | Rat hepatocyte | ||||
|
(2) Apoptosis assay (a) Annexin assay |
Annexin-v and propidium iodide | Human HepG2 hepatoma cells |
Detection of mutagenicity Used to assess the toxicity of zinc oxide nanoparticles |
(206) |
|
| (b) Comet assay | Dunaliella tertiolecta | Detection of toxicity imposed by silicon dioxide nanoparticles | Selenium nanoparticle toxicity | (209) | |
| (c) TUNEL assay | Goto Kakizaki rats (pancreatic beta cell) | (210) | |||
| (3) Necrotic assay | (a) Neutral red (2-amino-3 methyl-7-dimethyl-aminophenazoniumchloride) | Lysosomes | Measured integrity of the membrane find cell viability | (209) | |
| (b) Trypan Blue | MDCK kidney cells | Maintains membrane stability |
(211) |
||
| (4) Oxidative stress | 2′,7′-Dichlorofluorescein diacetate (DCFDA) | Pc12 cell (rat adrenal medulla) | (202) (214) |
In Vivo Toxicity
Animal models, such as rats and mice, are used for in vivo toxicity. The parameters, such as hematology, absorption, and metabolism, are assessed by the in vivo method. The route of localization of nanoparticles to the tissue or organ is examined through biodistribution studies. Radiolabels are used to detect the nanoparticles in the dead and live animals (215). After exposure, the clearance of NP has been done through examination of biotransformation or elimination of nanoparticles at a different time interval (216). Another method of toxicity assessment of nanoparticles is the examination difference in the chemistry of serum and type of cell after exposure to nanoparticles (217). The cytotoxicity level caused by a nanoparticle is measured through histopathology of the cell (218). Tissues such as lungs, heart, and others that exposed to nanoparticles are used for histopathology examination (219,220).
CONCLUSION
In this review, we presented a detailed overview about internalization of nanoparticles, interaction with intracellular organelles and their cytotoxicity, and also different methods used for the assessment of their toxicity. The internalization of nanoparticles can be achieved by crossing intestinal barriers such as the mucus layer, the epithelial layer, and tight junctions. The physical and chemical properties of nanoparticles also affect their internalization, and the fate of these particles after their cellular uptake at the cellular and subcellular levels is an extensive study. The small size and the relatively large surface area of NPs resulted in increased toxicity when compared to particles in micrometer size. For obtaining an optimized nanodelivery system, their cellular-induced cytotoxic response should also be adequately studied. Nanoparticles have been used in cancer treatment and autoimmune disorders. However, we conclude that the cytotoxicity of nanoparticles has been affected by changes in NP size. Nanoparticle size shows an essential interaction with the biological system. The ion release rate is affected by the size of nanoparticles; if the size of nanoparticles becomes smaller, it will lead to more interaction with the surface of the membrane, hence producing higher cytotoxic effects through penetration into the cell. Generally, cytotoxicity of nanoparticles that depends upon the size of nanoparticles also be correlated, the ability of nanoparticles to enter the biological system.
We also conclude that the shapes of nanoparticles also influence their toxicity. Nanoparticles are available in different shapes (e.g., filament, spherical, non-spherical, plate-shaped, and rod-like). Internalization processes such as endocytosis and phagocytosis depend on the shapes of nanoparticles. Spherical nanoparticles show faster endocytosis than a tubular nanoparticle. In future, the availability of NPs has opened up new technology in the medical field. The distinctive properties of nanomaterials give them many areas of human benefit, including catalysis, medicines, antimicrobials, biosensors, drug delivery, and electronics. Nanoparticles may be attached to the surface of biological membranes by adsorption or electrostatic interactions, and damage the cells by producing reactive oxygen species, leading to protein denaturation, lipid peroxidation, DNA damage, and ultimately cell death.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lipp R. Major advances in oral drug delivery over the past 15 years. Am Pharm Rev. 2013;16:28–35. [Google Scholar]
- 2.Ensign LM, Cone R, Hanes J. Oral drug delivery with polymeric nanoparticles: the gastrointestinal mucus barriers. Adv Drug Deliv Rev. 2012;64(6):557–570. doi: 10.1016/j.addr.2011.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pridgen EM, Alexis F, Farokhzad OC. Polymeric nanoparticle drug delivery technologies for oral delivery applications. Expert Opin Drug Deliv. 2015;12(9):1459–1473. doi: 10.1517/17425247.2015.1018175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hunter AC, et al. Polymeric particulate technologies for oral drug delivery and targeting: a pathophysiological perspective. Maturitas. 2012;73(1):5–18. doi: 10.1016/j.maturitas.2012.05.014. [DOI] [PubMed] [Google Scholar]
- 5.Pawar VK, et al. Targeting of gastrointestinal tract for amended delivery of protein/peptide therapeutics: strategies and industrial perspectives. J Control Release. 2014;196:168–183. doi: 10.1016/j.jconrel.2014.09.031. [DOI] [PubMed] [Google Scholar]
- 6.Malingré MM, Beijnen JH, Schellens JH. Oral delivery of taxanes. Investig New Drugs. 2001;19(2):155–162. doi: 10.1023/A:1010635000879. [DOI] [PubMed] [Google Scholar]
- 7.Mei L, et al. Pharmaceutical nanotechnology for oral delivery of anticancer drugs. Adv Drug Deliv Rev. 2013;65(6):880–890. doi: 10.1016/j.addr.2012.11.005. [DOI] [PubMed] [Google Scholar]
- 8.Reinholz J, Landfester K, Mailänder V. The challenges of oral drug delivery via nanocarriers. Drug Deliv. 2018;25(1):1694–1705. doi: 10.1080/10717544.2018.1501119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bobo D, et al. Nanoparticle-based medicines: a review of FDA-approved materials and clinical trials to date. Pharm Res. 2016;33(10):2373–2387. doi: 10.1007/s11095-016-1958-5. [DOI] [PubMed] [Google Scholar]
- 10.Anselmo AC, Mitragotri S. Nanoparticles in the clinic. Bioeng Translat Med. 2016;1(1):10–29. doi: 10.1002/btm2.10003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Huckaby JT, Lai SK. PEGylation for enhancing nanoparticle diffusion in mucus. Adv Drug Deliv Rev. 2018;124:125–139. doi: 10.1016/j.addr.2017.08.010. [DOI] [PubMed] [Google Scholar]
- 12.Chen M-C, et al. A review of the prospects for polymeric nanoparticle platforms in oral insulin delivery. Biomaterials. 2011;32(36):9826–9838. doi: 10.1016/j.biomaterials.2011.08.087. [DOI] [PubMed] [Google Scholar]
- 13.Banerjee A, et al. Role of nanoparticle size, shape and surface chemistry in oral drug delivery. J Control Release. 2016;238:176–185. doi: 10.1016/j.jconrel.2016.07.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goldberg M, Gomez-Orellana I. Challenges for the oral delivery of macromolecules. Nat Rev Drug Discov. 2003;2(4):289. doi: 10.1038/nrd1067. [DOI] [PubMed] [Google Scholar]
- 15.Kumar S, et al. Shape and size-dependent immune response to antigen-carrying nanoparticles. J Control Release. 2015;220:141–148. doi: 10.1016/j.jconrel.2015.09.069. [DOI] [PubMed] [Google Scholar]
- 16.Champion JA, Katare YK, Mitragotri S. Particle shape: a new design parameter for micro-and nanoscale drug delivery carriers. J Control Release. 2007;121(1-2):3–9. doi: 10.1016/j.jconrel.2007.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gerloff K, et al. Cytotoxicity and oxidative DNA damage by nanoparticles in human intestinal Caco-2 cells. Nanotoxicology. 2009;3(4):355–364. doi: 10.3109/17435390903276933. [DOI] [Google Scholar]
- 18.Schenk M, Mueller C. The mucosal immune system at the gastrointestinal barrier. Best Pract Res Clin Gastroenterol. 2008;22(3):391–409. doi: 10.1016/j.bpg.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 19.Chauhan VP, Jain RK. Strategies for advancing cancer nanomedicine. Nat Mater. 2013;12(11):958. doi: 10.1038/nmat3792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pelaseyed T, et al. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol Rev. 2014;260(1):8–20. doi: 10.1111/imr.12182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Johansson ME, et al. Composition and functional role of the mucus layers in the intestine. Cell Mol Life Sci. 2011;68(22):3635. doi: 10.1007/s00018-011-0822-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Corfield AP. Mucins: a biologically relevant glycan barrier in mucosal protection. Biochimica et Biophysica Acta (BBA)-General Subjects. 2015;1850(1):236–252. doi: 10.1016/j.bbagen.2014.05.003. [DOI] [PubMed] [Google Scholar]
- 23.Jonckheere N, et al. Membrane-bound mucin modular domains: from structure to function. Biochimie. 2013;95(6):1077–1086. doi: 10.1016/j.biochi.2012.11.005. [DOI] [PubMed] [Google Scholar]
- 24.Kim YS, Ho SB. Intestinal goblet cells and mucins in health and disease: recent insights and progress. Curr Gastroenterol Rep. 2010;12(5):319–330. doi: 10.1007/s11894-010-0131-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wiśniewski JR, et al. Extensive quantitative remodeling of the proteome between normal colon tissue and adenocarcinoma. Mol Syst Biol. 2012:8(1). [DOI] [PMC free article] [PubMed]
- 26.Johansson ME, Hansson GC. Mucus and the goblet cell. Dig Dis. 2013;31(3-4):305–309. doi: 10.1159/000354683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ambort D, et al. Calcium and pH-dependent packing and release of the gel-forming MUC2 mucin. Proc Natl Acad Sci. 2012;109(15):5645–5650. doi: 10.1073/pnas.1120269109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lai SK, Wang Y-Y, Hanes J. Mucus-penetrating nanoparticles for drug and gene delivery to mucosal tissues. Adv Drug Deliv Rev. 2009;61(2):158–171. doi: 10.1016/j.addr.2008.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Atuma C, et al. The adherent gastrointestinal mucus gel layer: thickness and physical state in vivo. Am J Physiology-Gastrointest Liver Physiol. 2001;280(5):G922–G929. doi: 10.1152/ajpgi.2001.280.5.G922. [DOI] [PubMed] [Google Scholar]
- 30.Bajka BH, et al. The influence of small intestinal mucus structure on particle transport ex vivo. Colloids Surf B: Biointerfaces. 2015;135:73–80. doi: 10.1016/j.colsurfb.2015.07.038. [DOI] [PubMed] [Google Scholar]
- 31.Hong Z, et al. Atomic force microscopy reveals aggregation of gastric mucin at low pH. Biomacromolecules. 2005;6(6):3458–3466. doi: 10.1021/bm0505843. [DOI] [PubMed] [Google Scholar]
- 32.Olmsted SS, et al. Diffusion of macromolecules and virus-like particles in human cervical mucus. Biophys J. 2001;81(4):1930–1937. doi: 10.1016/S0006-3495(01)75844-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lai SK, et al. Rapid transport of large polymeric nanoparticles in fresh undiluted human mucus. Proc Natl Acad Sci. 2007;104(5):1482–1487. doi: 10.1073/pnas.0608611104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wang JA, Meyer TF, Rudel T. Cytoskeleton and motor proteins are required for the transcytosis of Neisseria gonorrhoeae through polarized epithelial cells. Int J Med Microbiol. 2008;298(3-4):209–221. doi: 10.1016/j.ijmm.2007.05.004. [DOI] [PubMed] [Google Scholar]
- 35.de Sousa IP, et al. Mucus permeating carriers: formulation and characterization of highly densely charged nanoparticles. Eur J Pharm Biopharm. 2015;97:273–279. doi: 10.1016/j.ejpb.2014.12.024. [DOI] [PubMed] [Google Scholar]
- 36.Dünnhaupt S, et al. Nano-carrier systems: strategies to overcome the mucus gel barrier. Eur J Pharm Biopharm. 2015;96:447–453. doi: 10.1016/j.ejpb.2015.01.022. [DOI] [PubMed] [Google Scholar]
- 37.Griffiths PC, et al. Probing the interaction of nanoparticles with mucin for drug delivery applications using dynamic light scattering. Eur J Pharm Biopharm. 2015;97:218–222. doi: 10.1016/j.ejpb.2015.05.004. [DOI] [PubMed] [Google Scholar]
- 38.Lieleg O, Vladescu I, Ribbeck K. Characterization of particle translocation through mucin hydrogels. Biophys J. 2010;98(9):1782–1789. doi: 10.1016/j.bpj.2010.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lundquist P, Artursson P. Oral absorption of peptides and nanoparticles across the human intestine: opportunities, limitations and studies in human tissues. Adv Drug Deliv Rev. 2016;106:256–276. doi: 10.1016/j.addr.2016.07.007. [DOI] [PubMed] [Google Scholar]
- 40.Yildiz HM, et al. Food-associated stimuli enhance barrier properties of gastrointestinal mucus. Biomaterials. 2015;54:1–8. doi: 10.1016/j.biomaterials.2015.02.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Datta SS, Steinberg AP, Ismagilov RF. Polymers in the gut compress the colonic mucus hydrogel. Proc Natl Acad Sci. 2016;113(26):7041–7046. doi: 10.1073/pnas.1602789113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lai SK, et al. Nanoparticles reveal that human cervicovaginal mucus is riddled with pores larger than viruses. Proc Natl Acad Sci. 2010;107(2):598–603. doi: 10.1073/pnas.0911748107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xu Q, et al. Impact of surface polyethylene glycol (PEG) density on biodegradable nanoparticle transport in mucus ex vivo and distribution in vivo. ACS Nano. 2015;9(9):9217–9227. doi: 10.1021/acsnano.5b03876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ensign LM, et al. Mucus-penetrating nanoparticles for vaginal drug delivery protect against herpes simplex virus. Sci Transl Med. 2012;4(138):138ra79–9. [DOI] [PMC free article] [PubMed]
- 45.Swavola JC, Edwards TD, Bevan MA. Direct measurement of macromolecule-coated colloid–mucus interactions. Langmuir. 2015;31(33):9076–9085. doi: 10.1021/acs.langmuir.5b01460. [DOI] [PubMed] [Google Scholar]
- 46.Shan W, et al. Overcoming the diffusion barrier of mucus and absorption barrier of epithelium by self-assembled nanoparticles for oral delivery of insulin. ACS Nano. 2015;9(3):2345–2356. doi: 10.1021/acsnano.5b00028. [DOI] [PubMed] [Google Scholar]
- 47.Müller C, et al. Preparation and characterization of mucus-penetrating papain/poly (acrylic acid) nanoparticles for oral drug delivery applications. J Nanopart Res. 2013;15(1):1353. doi: 10.1007/s11051-012-1353-z. [DOI] [Google Scholar]
- 48.Rosenthal R, et al. The effect of chitosan on transcellular and paracellular mechanisms in the intestinal epithelial barrier. Biomaterials. 2012;33(9):2791–2800. doi: 10.1016/j.biomaterials.2011.12.034. [DOI] [PubMed] [Google Scholar]
- 49.Artursson P, Knight SD. Breaking the intestinal barrier to deliver drugs. Science. 2015;347(6223):716–717. doi: 10.1126/science.aaa6124. [DOI] [PubMed] [Google Scholar]
- 50.Turner JR et al. The role of molecular remodeling in differential regulation of tight junction permeability. in Seminars in cell & developmental biology. 2014. Elsevier. [DOI] [PMC free article] [PubMed]
- 51.Barmeyer, C., J.D. Schulzke, and M. Fromm. Claudin-related intestinal diseases. in Seminars in cell & developmental biology. 2015. Elsevier. [DOI] [PubMed]
- 52.Mrsny RJ, et al. A key claudin extracellular loop domain is critical for epithelial barrier integrity. Am J Pathol. 2008;172(4):905–915. doi: 10.2353/ajpath.2008.070698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Suzuki H, et al. Crystal structure of a claudin provides insight into the architecture of tight junctions. Science. 2014;344(6181):304–307. doi: 10.1126/science.1248571. [DOI] [PubMed] [Google Scholar]
- 54.Aguirre TA, et al. Current status of selected oral peptide technologies in advanced preclinical development and in clinical trials. Adv Drug Deliv Rev. 2016;106:223–241. doi: 10.1016/j.addr.2016.02.004. [DOI] [PubMed] [Google Scholar]
- 55.Maher S, et al. Evaluation of intestinal absorption enhancement and local mucosal toxicity of two promoters. I. Studies in isolated rat and human colonic mucosae. Eur J Pharm Sci. 2009;38(4):291–300. doi: 10.1016/j.ejps.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 56.Taverner A, et al. Enhanced paracellular transport of insulin can be achieved via transient induction of myosin light chain phosphorylation. J Control Release. 2015;210:189–197. doi: 10.1016/j.jconrel.2015.05.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Clayburgh DR, et al. Epithelial myosin light chain kinase–dependent barrier dysfunction mediates T cell activation–induced diarrhea in vivo. J Clin Invest. 2005;115(10):2702–2715. doi: 10.1172/JCI24970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Clayburgh DR, et al. Coordinated epithelial NHE3 inhibition and barrier dysfunction are required for TNF-mediated diarrhea in vivo. J Clin Invest. 2006;116(10):2682–2694. doi: 10.1172/JCI29218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang L, et al. Designing nanoparticles with improved tumor penetration: surface properties from the molecular architecture viewpoint. J Mater Chem B. 2019;7(6):953–964. doi: 10.1039/C8TB03034K. [DOI] [PubMed] [Google Scholar]
- 60.Becker M., An assessment of the effects of shell cross-linked nanoparticle size, core composition, and surface PEGylation on in vivo biodistribution. 2005. [DOI] [PMC free article] [PubMed]
- 61.Liu X, et al. Numerical study of clathrin-mediated endocytosis of nanoparticles by cells under tension. Acta Mech Sinica. 2019;35(3):691–701. doi: 10.1007/s10409-019-00839-0. [DOI] [Google Scholar]
- 62.Mikuličić S, Florin L. The endocytic trafficking pathway of oncogenic papillomaviruses. Papillomavirus Res. 2019;7:135–137. doi: 10.1016/j.pvr.2019.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sahay G, Alakhova DY, Kabanov AV. Endocytosis of nanomedicines. J Control Release. 2010;145(3):182–195. doi: 10.1016/j.jconrel.2010.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Charoo NA, Rahman Z, and Khan MA. Nanoparticles for improvement in oral bioavailability, in Nanoarchitectonics in biomedicine. 2019, Elsevier. p. 371-410.
- 65.Engelberg S, et al. Cancer cell-selective, clathrin-mediated endocytosis of aptamer decorated nanoparticles. Oncotarget. 2018;9(30):20993. doi: 10.18632/oncotarget.24772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Li Z, et al. A review: Using nanoparticles to enhance absorption and bioavailability of phenolic phytochemicals. Food Hydrocoll. 2015;43:153–164. doi: 10.1016/j.foodhyd.2014.05.010. [DOI] [Google Scholar]
- 67.Chettimada S, et al. Caveolae, caveolin-1 and cavin-1: emerging roles in pulmonary hypertension. World J Respirol. 2015;5(2):126. doi: 10.5320/wjr.v5.i2.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Cheng JP, Nichols BJ. Caveolae: one function or many? Trends Cell Biol. 2016;26(3):177–189. doi: 10.1016/j.tcb.2015.10.010. [DOI] [PubMed] [Google Scholar]
- 69.Pelkmans L, et al. Caveolin-stabilized membrane domains as multifunctional transport and sorting devices in endocytic membrane traffic. Cell. 2004;118(6):767–780. doi: 10.1016/j.cell.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 70.Fytianos, K., F. Blank, and L. Müller, Cellular uptake mechanisms and detection of nanoparticle uptake by advanced imaging methods, in Biological responses to nanoscale particles. 2019, Springer. p. 191-211.
- 71.Flatt JW, Butcher SJ. Adenovirus flow in host cell networks. Open Biol. 2019;9(2):190012. doi: 10.1098/rsob.190012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lee, S.-W., B. Alas, and C. Commisso, Detection and quantification of macropinosomes in pancreatic tumors, in Pancreatic cancer. 2019, Springer. p. 171–181. [DOI] [PubMed]
- 73.Zhang S, Gao H, Bao G. Physical principles of nanoparticle cellular endocytosis. ACS Nano. 2015;9(9):8655–8671. doi: 10.1021/acsnano.5b03184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Mei L, Sun H, Song C. Local delivery of modified paclitaxel-loaded poly (ε-caprolactone)/pluronic F68 nanoparticles for long-term inhibition of hyperplasia. J Pharm Sci. 2009;98(6):2040–2050. doi: 10.1002/jps.21581. [DOI] [PubMed] [Google Scholar]
- 75.Anselmo AC, et al. Elasticity of nanoparticles influences their blood circulation, phagocytosis, endocytosis, and targeting. ACS Nano. 2015;9(3):3169–3177. doi: 10.1021/acsnano.5b00147. [DOI] [PubMed] [Google Scholar]
- 76.Guo P, et al. Nanoparticle elasticity directs tumor uptake. Nat Commun. 2018;9(1):130. doi: 10.1038/s41467-017-02588-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Tzlil S, et al. A statistical-thermodynamic model of viral budding. Biophys J. 2004;86(4):2037–2048. doi: 10.1016/S0006-3495(04)74265-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Deserno M, Bickel T. Wrapping of a spherical colloid by a fluid membrane. EPL (Europhysics Letters) 2003;62(5):767. doi: 10.1209/epl/i2003-00438-4. [DOI] [Google Scholar]
- 79.Sperling R, et al. Size determination of (bio) conjugated water-soluble colloidal nanoparticles: a comparison of different techniques. J Phys Chem C. 2007;111(31):11552–11559. doi: 10.1021/jp070999d. [DOI] [Google Scholar]
- 80.Hagendorfer H, et al. Characterization of silver nanoparticle products using asymmetric flow field flow fractionation with a multidetector approach–a comparison to transmission electron microscopy and batch dynamic light scattering. Anal Chem. 2012;84(6):2678–2685. doi: 10.1021/ac202641d. [DOI] [PubMed] [Google Scholar]
- 81.Nazarenus M, et al. In vitro interaction of colloidal nanoparticles with mammalian cells: what have we learned thus far? Beilstein J Nanotechnol. 2014;5(1):1477–1490. doi: 10.3762/bjnano.5.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Maguire CM, et al. Benchmark of nanoparticle tracking analysis on measuring nanoparticle sizing and concentration. J Micro Nano Manuf. 2017;5(4):041002. doi: 10.1115/1.4037124. [DOI] [Google Scholar]
- 83.Hole P, et al. Interlaboratory comparison of size measurements on nanoparticles using nanoparticle tracking analysis (NTA) J Nanopart Res. 2013;15(12):2101. doi: 10.1007/s11051-013-2101-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Albanese A, Tang PS, Chan WC. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012;14:1–16. doi: 10.1146/annurev-bioeng-071811-150124. [DOI] [PubMed] [Google Scholar]
- 85.Sharma G, et al. Polymer particle shape independently influences binding and internalization by macrophages. J Control Release. 2010;147(3):408–412. doi: 10.1016/j.jconrel.2010.07.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kinnear C, et al. Form follows function: nanoparticle shape and its implications for nanomedicine. Chem Rev. 2017;117(17):11476–11521. doi: 10.1021/acs.chemrev.7b00194. [DOI] [PubMed] [Google Scholar]
- 87.Zhao Z, et al. Effect of physicochemical and surface properties on in vivo fate of drug nanocarriers. Adv Drug Deliv Rev. 2019. [DOI] [PubMed]
- 88.Doshi N, Mitragotri S. Macrophages recognize size and shape of their targets. PLoS One. 2010;5(4):e10051. doi: 10.1371/journal.pone.0010051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Toy R, et al. Shaping cancer nanomedicine: the effect of particle shape on the in vivo journey of nanoparticles. Nanomedicine. 2014;9(1):121–134. doi: 10.2217/nnm.13.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Schweiger C, et al. Quantification of the internalization patterns of superparamagnetic iron oxide nanoparticles with opposite charge. J Nanobiotechnol. 2012;10(1):28. doi: 10.1186/1477-3155-10-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Hühn D, et al. Polymer-coated nanoparticles interacting with proteins and cells: focusing on the sign of the net charge. ACS Nano. 2013;7(4):3253–3263. doi: 10.1021/nn3059295. [DOI] [PubMed] [Google Scholar]
- 92.Jiang X, et al. Specific effects of surface amines on polystyrene nanoparticles in their interactions with mesenchymal stem cells. Biomacromolecules. 2010;11(3):748–753. doi: 10.1021/bm901348z. [DOI] [PubMed] [Google Scholar]
- 93.Jiang X, et al. Specific effects of surface carboxyl groups on anionic polystyrene particles in their interactions with mesenchymal stem cells. Nanoscale. 2011;3(5):2028–2035. doi: 10.1039/c0nr00944j. [DOI] [PubMed] [Google Scholar]
- 94.Yang S-H, et al. Role of surface charge in cytotoxicity of charged manganese ferrite nanoparticles towards macrophages. Nanotechnology. 2012;23(50):505702. doi: 10.1088/0957-4484/23/50/505702. [DOI] [PubMed] [Google Scholar]
- 95.Kim ST, et al. The role of surface functionality in determining nanoparticle cytotoxicity. Acc Chem Res. 2013;46(3):681–691. doi: 10.1021/ar3000647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.des Rieux A, et al. An improved in vitro model of human intestinal follicle-associated epithelium to study nanoparticle transport by M cells. Eur J Pharm Sci. 2007;30(5):380–391. doi: 10.1016/j.ejps.2006.12.006. [DOI] [PubMed] [Google Scholar]
- 97.Wingett D, et al. A role of ZnO nanoparticle electrostatic properties in cancer cell cytotoxicity. Nanotechnol Sci Appl. 2016;9:29. doi: 10.2147/NSA.S99747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Louka, P., The role of ZnO nanoparticle electrostatic properties in cancer cell cytotoxicity. 2012. [DOI] [PMC free article] [PubMed]
- 99.Yi X, Gao H. Kinetics of receptor-mediated endocytosis of elastic nanoparticles. Nanoscale. 2017;9(1):454–463. doi: 10.1039/C6NR07179A. [DOI] [PubMed] [Google Scholar]
- 100.Yi X, Shi X, Gao H. Cellular uptake of elastic nanoparticles. Phys Rev Lett. 2011;107(9):098101. doi: 10.1103/PhysRevLett.107.098101. [DOI] [PubMed] [Google Scholar]
- 101.Sun J, et al. Tunable rigidity of (polymeric core)–(lipid shell) nanoparticles for regulated cellular uptake. Adv Mater. 2015;27(8):1402–1407. doi: 10.1002/adma.201404788. [DOI] [PubMed] [Google Scholar]
- 102.Yi X, Gao H. Cell membrane wrapping of a spherical thin elastic shell. Soft Matter. 2015;11(6):1107–1115. doi: 10.1039/C4SM02427C. [DOI] [PubMed] [Google Scholar]
- 103.Chithrani BD, Ghazani AA, Chan WC. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett. 2006;6(4):662–668. doi: 10.1021/nl052396o. [DOI] [PubMed] [Google Scholar]
- 104.Chithrani BD, Chan WC. Elucidating the mechanism of cellular uptake and removal of protein-coated gold nanoparticles of different sizes and shapes. Nano Lett. 2007;7(6):1542–1550. doi: 10.1021/nl070363y. [DOI] [PubMed] [Google Scholar]
- 105.Song Q, et al. Mechanisms of transcellular transport of wheat germ agglutinin-functionalized polymeric nanoparticles in Caco-2 cells. Biomaterials. 2012;33(28):6769–6782. doi: 10.1016/j.biomaterials.2012.05.066. [DOI] [PubMed] [Google Scholar]
- 106.Zhang S, et al. Size-dependent endocytosis of nanoparticles. Adv Mater. 2009;21(4):419–424. doi: 10.1002/adma.200801393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Oh E, et al. Cellular uptake and fate of PEGylated gold nanoparticles is dependent on both cell-penetration peptides and particle size. ACS Nano. 2011;5(8):6434–6448. doi: 10.1021/nn201624c. [DOI] [PubMed] [Google Scholar]
- 108.Hirota K, et al. Optimum conditions for efficient phagocytosis of rifampicin-loaded PLGA microspheres by alveolar macrophages. J Control Release. 2007;119(1):69–76. doi: 10.1016/j.jconrel.2007.01.013. [DOI] [PubMed] [Google Scholar]
- 109.Swiston AJ, et al. Freely suspended cellular “backpacks” lead to cell aggregate self-assembly. Biomacromolecules. 2010;11(7):1826–1832. doi: 10.1021/bm100305h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Stoehr LC, et al. Shape matters: effects of silver nanospheres and wires on human alveolar epithelial cells. Part Fibre Toxicol. 2011;8(1):36. doi: 10.1186/1743-8977-8-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ahmed KBR, et al. Silver nanoparticles: significance of physicochemical properties and assay interference on the interpretation of in vitro cytotoxicity studies. Toxicol in Vitro. 2017;38:179–192. doi: 10.1016/j.tiv.2016.10.012. [DOI] [PubMed] [Google Scholar]
- 112.Champion JA, Mitragotri S. Role of target geometry in phagocytosis. Proc Natl Acad Sci. 2006;103(13):4930–4934. doi: 10.1073/pnas.0600997103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Vllasaliu D, et al. Fc-mediated transport of nanoparticles across airway epithelial cell layers. J Control Release. 2012;158(3):479–486. doi: 10.1016/j.jconrel.2011.12.009. [DOI] [PubMed] [Google Scholar]
- 114.Dombu CY, et al. Characterization of endocytosis and exocytosis of cationic nanoparticles in airway epithelium cells. Nanotechnology. 2010;21(35):355102. doi: 10.1088/0957-4484/21/35/355102. [DOI] [PubMed] [Google Scholar]
- 115.Pridgen EM, et al. Transepithelial transport of Fc-targeted nanoparticles by the neonatal fc receptor for oral delivery. Sci Transl Med. 2013;5(213):213ra167–7. [DOI] [PMC free article] [PubMed]
- 116.Roger E, et al. Folic acid functionalized nanoparticles for enhanced oral drug delivery. Mol Pharm. 2012;9(7):2103–2110. doi: 10.1021/mp2005388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Underhill DM, Goodridge HS. Information processing during phagocytosis. Nat Rev Immunol. 2012;12(7):492. doi: 10.1038/nri3244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Fowler R, et al. Nanoparticle transport in epithelial cells: pathway switching through bioconjugation. Small. 2013;9(19):3282–3294. doi: 10.1002/smll.201202623. [DOI] [PubMed] [Google Scholar]
- 119.Fröhlich E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int J Nanomedicine. 2012;7:5577. doi: 10.2147/IJN.S36111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Muhlfeld C, et al. Interactions of nanoparticles with pulmonary structures and cellular responses. Am J Phys Lung Cell Mol Phys. 2008;294(5):L817–L829. doi: 10.1152/ajplung.00442.2007. [DOI] [PubMed] [Google Scholar]
- 121.Unfried K, et al. Cellular responses to nanoparticles: target structures and mechanisms. Nanotoxicology. 2007;1(1):52–71. doi: 10.1080/00222930701314932. [DOI] [Google Scholar]
- 122.Elsaesser A, et al. Quantification of nanoparticle uptake by cells using microscopical and analytical techniques. Nanomedicine. 2010;5(9):1447–1457. doi: 10.2217/nnm.10.118. [DOI] [PubMed] [Google Scholar]
- 123.Feliu N, et al. Quantitative uptake of colloidal particles by cell cultures. Sci Total Environ. 2016;568:819–828. doi: 10.1016/j.scitotenv.2016.05.213. [DOI] [PubMed] [Google Scholar]
- 124.Gottstein C, et al. Precise quantification of nanoparticle internalization. ACS Nano. 2013;7(6):4933–4945. doi: 10.1021/nn400243d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Vanhecke D, et al. Quantification of nanoparticles at the single-cell level: an overview about state-of-the-art techniques and their limitations. Nanomedicine. 2014;9(12):1885–1900. doi: 10.2217/nnm.14.108. [DOI] [PubMed] [Google Scholar]
- 126.Drasler B, et al. In vitro approaches to assess the hazard of nanomaterials. NanoImpact. 2017;8:99–116. doi: 10.1016/j.impact.2017.08.002. [DOI] [Google Scholar]
- 127.Bourquin J, et al. Biodistribution, clearance, and long-term fate of clinically relevant nanomaterials. Adv Mater. 2018;30(19):1704307. [DOI] [PubMed]
- 128.Oh N, Park J-H. Endocytosis and exocytosis of nanoparticles in mammalian cells. Int J Nanomedicine. 2014;9(Suppl 1):51. doi: 10.2147/IJN.S26592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Mahmoudi M, et al. Cell “vision”: complementary factor of protein corona in nanotoxicology. Nanoscale. 2012;4(17):5461–5468. doi: 10.1039/c2nr31185b. [DOI] [PubMed] [Google Scholar]
- 130.Kuhn DA, et al. Different endocytotic uptake mechanisms for nanoparticles in epithelial cells and macrophages. Beilstein J Nanotechnol. 2014;5(1):1625–1636. doi: 10.3762/bjnano.5.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Ma X, et al. Future of nanotherapeutics: targeting the cellular sub-organelles. Biomaterials. 2016;97:10–21. doi: 10.1016/j.biomaterials.2016.04.026. [DOI] [PubMed] [Google Scholar]
- 132.Lu P, et al. Delivery of drugs and macromolecules to the mitochondria for cancer therapy. J Control Release. 2016;240:38–51. doi: 10.1016/j.jconrel.2015.10.023. [DOI] [PubMed] [Google Scholar]
- 133.Wongrakpanich A, et al. Mitochondria-targeting particles. Nanomedicine. 2014;9(16):2531–2543. doi: 10.2217/nnm.14.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Qu Q, Ma X, Zhao Y. Targeted delivery of doxorubicin to mitochondria using mesoporous silica nanoparticle nanocarriers. Nanoscale. 2015;7(40):16677–16686. doi: 10.1039/C5NR05139H. [DOI] [PubMed] [Google Scholar]
- 135.Kwon HJ, et al. Mitochondria-targeting ceria nanoparticles as antioxidants for Alzheimer’s disease. ACS Nano. 2016;10(2):2860–2870. doi: 10.1021/acsnano.5b08045. [DOI] [PubMed] [Google Scholar]
- 136.Kang JW, et al. High resolution live cell Raman imaging using subcellular organelle-targeting SERS-sensitive gold nanoparticles with highly narrow intra-nanogap. Nano Lett. 2015;15(3):1766–1772. doi: 10.1021/nl504444w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Zhang Z, et al. Mitophagy induced by nanoparticle–peptide conjugates enabling an alternative intracellular trafficking route. Biomaterials. 2015;65:56–65. doi: 10.1016/j.biomaterials.2015.06.029. [DOI] [PubMed] [Google Scholar]
- 138.Yamada Y, Harashima H. Enhancement in selective mitochondrial association by direct modification of a mitochondrial targeting signal peptide on a liposomal based nanocarrier. Mitochondrion. 2013;13(5):526–532. doi: 10.1016/j.mito.2012.09.001. [DOI] [PubMed] [Google Scholar]
- 139.Jeena M, et al. Mitochondria localization induced self-assembly of peptide amphiphiles for cellular dysfunction. Nat Commun. 2017;8(1):26. doi: 10.1038/s41467-017-00047-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Mukai Y, et al. Induction of endoplasmic reticulum–endosome fusion for antigen cross-presentation induced by poly (γ-glutamic acid) nanoparticles. J Immunol. 2011;187(12):6249–6255. doi: 10.4049/jimmunol.1001093. [DOI] [PubMed] [Google Scholar]
- 141.Cubillos-Ruiz JR, et al. ER stress sensor XBP1 controls anti-tumor immunity by disrupting dendritic cell homeostasis. Cell. 2015;161(7):1527–1538. doi: 10.1016/j.cell.2015.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Wang G, et al. KDEL peptide gold nanoconstructs: promising nanoplatforms for drug delivery. Nanomedicine. 2013;9(3):366–374. doi: 10.1016/j.nano.2012.09.002. [DOI] [PubMed] [Google Scholar]
- 143.Pan T, et al. miR-29b-loaded gold nanoparticles targeting to the endoplasmic reticulum for synergistic promotion of osteogenic differentiation. ACS Appl Mater Interfaces. 2016;8(30):19217–19227. doi: 10.1021/acsami.6b02969. [DOI] [PubMed] [Google Scholar]
- 144.Rodrigues J, Banks P, Lydall D. Vps74 connects the Golgi apparatus and telomeres in Saccharomyces cerevisiae. G3: Genes, Genomes, Genetics. 2018;8(5):1807–1816. doi: 10.1534/g3.118.200172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Yu R-Y, et al. Regulating the Golgi apparatus by co-delivery of a COX-2 inhibitor and Brefeldin A for suppression of tumor metastasis. Biomater Sci. 2018;6(8):2144–2155. doi: 10.1039/C8BM00381E. [DOI] [PubMed] [Google Scholar]
- 146.Cordonnier M-N, et al. Actin filaments and myosin I alpha cooperate with microtubules for the movement of lysosomes. Mol Biol Cell. 2001;12(12):4013–4029. doi: 10.1091/mbc.12.12.4013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Li RS, et al. Chiral nanoprobes for targeting and long-term imaging of the Golgi apparatus. Chem Sci. 2017;8(10):6829–6835. doi: 10.1039/C7SC01316G. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Panté N, Kann M. Nuclear pore complex is able to transport macromolecules with diameters of∼ 39 nm. Mol Biol Cell. 2002;13(2):425–434. doi: 10.1091/mbc.01-06-0308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.De la Fuente JM, Berry CC. Tat peptide as an efficient molecule to translocate gold nanoparticles into the cell nucleus. Bioconjug Chem. 2005;16(5):1176–1180. doi: 10.1021/bc050033+. [DOI] [PubMed] [Google Scholar]
- 150.Eibauer M, et al. Structure and gating of the nuclear pore complex. Nat Commun. 2015;6:7532. doi: 10.1038/ncomms8532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Tammam SN, Azzazy HM, Lamprecht A. How successful is nuclear targeting by nanocarriers? J Control Release. 2016;229:140–153. doi: 10.1016/j.jconrel.2016.03.022. [DOI] [PubMed] [Google Scholar]
- 152.Larsen JD, Ross NL, Sullivan MO. Requirements for the nuclear entry of polyplexes and nanoparticles during mitosis. J Gene Med. 2012;14(9-10):580–589. doi: 10.1002/jgm.2669. [DOI] [PubMed] [Google Scholar]
- 153.Yang C, et al. Peptide modified gold nanoparticles for improved cellular uptake, nuclear transport, and intracellular retention. Nanoscale. 2014;6(20):12026–12033. doi: 10.1039/C4NR02535K. [DOI] [PubMed] [Google Scholar]
- 154.Pan L, et al. Nuclear-targeted drug delivery of TAT peptide-conjugated monodisperse mesoporous silica nanoparticles. J Am Chem Soc. 2012;134(13):5722–5725. doi: 10.1021/ja211035w. [DOI] [PubMed] [Google Scholar]
- 155.Maimon T, et al. The human nuclear pore complex as revealed by cryo-electron tomography. Structure. 2012;20(6):998–1006. doi: 10.1016/j.str.2012.03.025. [DOI] [PubMed] [Google Scholar]
- 156.García I, et al. Size-dependent transport and cytotoxicity of mitomycin-gold nanoparticle conjugates in 2D and 3D mammalian cell models. Bioconjug Chem. 2018;30(1):242–252. doi: 10.1021/acs.bioconjchem.8b00898. [DOI] [PubMed] [Google Scholar]
- 157.Huo S, et al. Ultrasmall gold nanoparticles as carriers for nucleus-based gene therapy due to size-dependent nuclear entry. ACS Nano. 2014;8(6):5852–5862. doi: 10.1021/nn5008572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Tang PS, et al. The role of ligand density and size in mediating quantum dot nuclear transport. Small. 2014;10(20):4182–4192. doi: 10.1002/smll.201401056. [DOI] [PubMed] [Google Scholar]
- 159.Ishida Y, et al. A model of lysosomal pH regulation. J Gen Phys. 2013;141(6):705–720. doi: 10.1085/jgp.201210930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Saftig P. Lysosomal membrane proteins, in Lysosomes. 2005, Springer. p. 37-49.
- 161.Saftig P, Klumperman J. Lysosome biogenesis and lysosomal membrane proteins: trafficking meets function. Nat Rev Mol Cell Biol. 2009;10(9):623. doi: 10.1038/nrm2745. [DOI] [PubMed] [Google Scholar]
- 162.Settembre C, et al. Signals from the lysosome: a control centre for cellular clearance and energy metabolism. Nat Rev Mol Cell Biol. 2013;14(5):283. doi: 10.1038/nrm3565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Hofmann D, et al. Mass spectrometry and imaging analysis of nanoparticle-containing vesicles provide a mechanistic insight into cellular trafficking. ACS Nano. 2014;8(10):10077–10088. doi: 10.1021/nn502754c. [DOI] [PubMed] [Google Scholar]
- 164.Hu Y-B, et al. The endosomal-lysosomal system: from acidification and cargo sorting to neurodegeneration. Transl Neurodegen. 2015;4(1):18. doi: 10.1186/s40035-015-0041-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Müller S, Dennemärker J, Reinheckel T. Specific functions of lysosomal proteases in endocytic and autophagic pathways. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics. 2012;1824(1):34–43. doi: 10.1016/j.bbapap.2011.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Kreyling WG, et al. In vivo integrity of polymer-coated gold nanoparticles. Nat Nanotechnol. 2015;10(7):619. doi: 10.1038/nnano.2015.111. [DOI] [PubMed] [Google Scholar]
- 167.Milosevic AM, et al. Assessing the stability of fluorescently encoded nanoparticles in lysosomes by using complementary methods. Angew Chem Int Ed. 2017;56(43):13382–13386. doi: 10.1002/anie.201705422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Ma Z, Bai J, Jiang X. Monitoring of the enzymatic degradation of protein corona and evaluating the accompanying cytotoxicity of nanoparticles. ACS Appl Mater Interfaces. 2015;7(32):17614–17622. doi: 10.1021/acsami.5b05744. [DOI] [PubMed] [Google Scholar]
- 169.Frohlich E. Cellular targets and mechanisms in the cytotoxic action of non-biodegradable engineered nanoparticles. Curr Drug Metab. 2013;14(9):976–988. doi: 10.2174/1389200211314090004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Cui Y, et al. The combination of endolysosomal escape and basolateral stimulation to overcome the difficulties of “easy uptake hard transcytosis” of ligand-modified nanoparticles in oral drug delivery. Nanoscale. 2018;10(3):1494–1507. doi: 10.1039/C7NR06063G. [DOI] [PubMed] [Google Scholar]
- 171.Piao MJ, et al. Silver nanoparticles induce oxidative cell damage in human liver cells through inhibition of reduced glutathione and induction of mitochondria-involved apoptosis. Toxicol Lett. 2011;201(1):92–100. doi: 10.1016/j.toxlet.2010.12.010. [DOI] [PubMed] [Google Scholar]
- 172.Carlson C, et al. Unique cellular interaction of silver nanoparticles: size-dependent generation of reactive oxygen species. J Phys Chem B. 2008;112(43):13608–13619. doi: 10.1021/jp712087m. [DOI] [PubMed] [Google Scholar]
- 173.Nel A, et al. Toxic potential of materials at the nanolevel. Science. 2006;311(5761):622–627. doi: 10.1126/science.1114397. [DOI] [PubMed] [Google Scholar]
- 174.Liu W, et al. Impact of silver nanoparticles on human cells: effect of particle size. Nanotoxicology. 2010;4(3):319–330. doi: 10.3109/17435390.2010.483745. [DOI] [PubMed] [Google Scholar]
- 175.Favi PM, et al. Shape and surface effects on the cytotoxicity of nanoparticles: gold nanospheres versus gold nanostars. J Biomed Mater Res A. 2015;103(11):3449–3462. doi: 10.1002/jbm.a.35491. [DOI] [PubMed] [Google Scholar]
- 176.Li X, et al. Maturation of osteoblast-like SaoS2 induced by carbon nanotubes. Biomed Mater. 2008;4(1):015005. [DOI] [PubMed]
- 177.Georgieva JV, et al. Surface characteristics of nanoparticles determine their intracellular fate in and processing by human blood–brain barrier endothelial cells in vitro. Mol Ther. 2011;19(2):318–325. doi: 10.1038/mt.2010.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Shahbazi M-A, et al. The mechanisms of surface chemistry effects of mesoporous silicon nanoparticles on immunotoxicity and biocompatibility. Biomaterials. 2013;34(31):7776–7789. doi: 10.1016/j.biomaterials.2013.06.052. [DOI] [PubMed] [Google Scholar]
- 179.Hussain S, et al. In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol in Vitro. 2005;19(7):975–983. doi: 10.1016/j.tiv.2005.06.034. [DOI] [PubMed] [Google Scholar]
- 180.Santos HA, et al. In vitro cytotoxicity of porous silicon microparticles: effect of the particle concentration, surface chemistry and size. Acta Biomater. 2010;6(7):2721–2731. doi: 10.1016/j.actbio.2009.12.043. [DOI] [PubMed] [Google Scholar]
- 181.Donaldson K, et al. Combustion-derived nanoparticles: a review of their toxicology following inhalation exposure. Part Fibre Toxicol. 2005;2(1):10. doi: 10.1186/1743-8977-2-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.You Y, et al. High-drug-loading mesoporous silica nanorods with reduced toxicity for precise cancer therapy against nasopharyngeal carcinoma. Adv Funct Mater. 2017;27(42):1703313. doi: 10.1002/adfm.201703313. [DOI] [Google Scholar]
- 183.Coradeghini R, et al. Size-dependent toxicity and cell interaction mechanisms of gold nanoparticles on mouse fibroblasts. Toxicol Lett. 2013;217(3):205–216. doi: 10.1016/j.toxlet.2012.11.022. [DOI] [PubMed] [Google Scholar]
- 184.Asharani P, Hande MP, Valiyaveettil S. Anti-proliferative activity of silver nanoparticles. BMC Cell Biol. 2009;10(1):65. doi: 10.1186/1471-2121-10-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Nair S, et al. Role of size scale of ZnO nanoparticles and microparticles on toxicity toward bacteria and osteoblast cancer cells. J Mater Sci Mater Med. 2009;20(1):235. doi: 10.1007/s10856-008-3548-5. [DOI] [PubMed] [Google Scholar]
- 186.Premanathan M, et al. Selective toxicity of ZnO nanoparticles toward Gram-positive bacteria and cancer cells by apoptosis through lipid peroxidation. Nanomedicine. 2011;7(2):184–192. doi: 10.1016/j.nano.2010.10.001. [DOI] [PubMed] [Google Scholar]
- 187.Trachootham D, Alexandre J, Huang P. Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nat Rev Drug Discov. 2009;8(7):579. doi: 10.1038/nrd2803. [DOI] [PubMed] [Google Scholar]
- 188.Thannickal VJ, Fanburg BL. Reactive oxygen species in cell signaling. Am J Phys Lung Cell Mol Phys. 2000;279(6):L1005–L1028. doi: 10.1152/ajplung.2000.279.6.L1005. [DOI] [PubMed] [Google Scholar]
- 189.Dikalov S. Cross talk between mitochondria and NADPH oxidases. Free Radic Biol Med. 2011;51(7):1289–1301. doi: 10.1016/j.freeradbiomed.2011.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Tahara EB, Navarete FD, Kowaltowski AJ. Tissue-, substrate-, and site-specific characteristics of mitochondrial reactive oxygen species generation. Free Radic Biol Med. 2009;46(9):1283–1297. doi: 10.1016/j.freeradbiomed.2009.02.008. [DOI] [PubMed] [Google Scholar]
- 191.Okado-Matsumoto A, Fridovich I. Subcellular distribution of superoxide dismutases (SOD) in rat liver Cu, Zn-SOD in mitochondria. J Biol Chem. 2001;276(42):38388–38393. doi: 10.1074/jbc.M105395200. [DOI] [PubMed] [Google Scholar]
- 192.Murphy MP. How mitochondria produce reactive oxygen species. Biochem J. 2009;417(1):1–13. doi: 10.1042/BJ20081386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Finkel T. Signal transduction by reactive oxygen species. J Cell Biol. 2011;194(1):7–15. doi: 10.1083/jcb.201102095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Xia T, et al. Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano. 2008;2(10):2121–2134. doi: 10.1021/nn800511k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Zhu X, et al. Biosensing approaches for rapid genotoxicity and cytotoxicity assays upon nanomaterial exposure. Small. 2013;9(9-10):1821–1830. doi: 10.1002/smll.201201593. [DOI] [PubMed] [Google Scholar]
- 196.Marziyeh A, et al. "Health concerns of various nanoparticles: a review of their in vitro and in vivo toxicity." Nanomaterials. 2018;8(9):634. [DOI] [PMC free article] [PubMed]
- 197.Lee KJ, et al. In vivo imaging of transport and biocompatibility of single silver nanoparticles in early development of zebrafish embryos. ACS Nano. 2007;1(2):133–143. doi: 10.1021/nn700048y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Jin Y, et al. Toxicity of luminescent silica nanoparticles to living cells. Chem Res Toxicol. 2007;20(8):1126–1133. doi: 10.1021/tx7001959. [DOI] [PubMed] [Google Scholar]
- 199.Monteiro-Riviere NA, et al. Multi-walled carbon nanotube interactions with human epidermal keratinocytes. Toxicol Lett. 2005;155(3):377–384. doi: 10.1016/j.toxlet.2004.11.004. [DOI] [PubMed] [Google Scholar]
- 200.Strober, W., Trypan blue exclusion test of cell viability. Curr Protoc Immunol, 2015. 111(1): p. A3. B. 1-A3. B. 3. [DOI] [PMC free article] [PubMed]
- 201.Magder S. Reactive oxygen species: toxic molecules or spark of life? Crit Care. 2006;10(1):1–8. doi: 10.1186/cc3992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Wagner AJ, et al. Cellular interaction of different forms of aluminum nanoparticles in rat alveolar macrophages. J Phys Chem B. 2007;111(25):7353–7359. doi: 10.1021/jp068938n. [DOI] [PubMed] [Google Scholar]
- 203.Sayes CM, Reed KL, Warheit DB. Assessing toxicity of fine and nanoparticles: comparing in vitro measurements to in vivo pulmonary toxicity profiles. Toxicol Sci. 2007;97(1):163–180. doi: 10.1093/toxsci/kfm018. [DOI] [PubMed] [Google Scholar]
- 204.Marshall N, Goodwin C, Holt S. A critical assessment of the use of microculture tetrazolium assays to measure cell growth and function. Growth Regul. 1995;5(2):69–84. [PubMed] [Google Scholar]
- 205.Low SP, et al. Evaluation of mammalian cell adhesion on surface-modified porous silicon. Biomaterials. 2006;27(26):4538–4546. doi: 10.1016/j.biomaterials.2006.04.015. [DOI] [PubMed] [Google Scholar]
- 206.Berghe TV, et al. Determination of apoptotic and necrotic cell death in vitro and in vivo. Methods. 2013;61(2):117–129. doi: 10.1016/j.ymeth.2013.02.011. [DOI] [PubMed] [Google Scholar]
- 207.Lu X, et al. In vitro cytotoxicity and induction of apoptosis by silica nanoparticles in human HepG2 hepatoma cells. Int J Nanomedicine. 2011;6:1889. doi: 10.2147/IJN.S24005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Schiavo S, et al. Genotoxic and cytotoxic effects of ZnO nanoparticles for Dunaliella tertiolecta and comparison with SiO2 and TiO2 effects at population growth inhibition levels. Sci Total Environ. 2016;550:619–627. doi: 10.1016/j.scitotenv.2016.01.135. [DOI] [PubMed] [Google Scholar]
- 209.Singh AP, et al. Developments in encapsulation of insulin: is oral delivery now possible? J Pharm Biopharm Res. 2019;1(1):74–93. doi: 10.25082/JPBR.2019.02.005. [DOI] [Google Scholar]
- 210.Mo Y, Lim L-Y. Paclitaxel-loaded PLGA nanoparticles: potentiation of anticancer activity by surface conjugation with wheat germ agglutinin. J Control Release. 2005;108(2-3):244–262. doi: 10.1016/j.jconrel.2005.08.013. [DOI] [PubMed] [Google Scholar]
- 211.Bitensky L. The reversible activation of lysosomes in normal cells and the effects of pathological conditions. in Ciba Foundation Symposium-Anterior Pituitary Secretion (Book I of Colloquia on Endocrinology). 1963. Wiley Online Library.
- 212.Huang M, Khor E, Lim L-Y. Uptake and cytotoxicity of chitosan molecules and nanoparticles: effects of molecular weight and degree of deacetylation. Pharm Res. 2004;21(2):344–353. doi: 10.1023/B:PHAM.0000016249.52831.a5. [DOI] [PubMed] [Google Scholar]
- 213.Kononenko V, et al. Comparative in vitro genotoxicity study of ZnO nanoparticles, ZnO macroparticles and ZnCl2 to MDCK kidney cells: size matters. Toxicol in Vitro. 2017;40:256–263. doi: 10.1016/j.tiv.2017.01.015. [DOI] [PubMed] [Google Scholar]
- 214.Hussain SM, et al. The interaction of manganese nanoparticles with PC-12 cells induces dopamine depletion. Toxicol Sci. 2006;92(2):456–463. doi: 10.1093/toxsci/kfl020. [DOI] [PubMed] [Google Scholar]
- 215.Kim SC, et al. In vivo evaluation of polymeric micellar paclitaxel formulation: toxicity and efficacy. J Control Release. 2001;72(1-3):191–202. doi: 10.1016/S0168-3659(01)00275-9. [DOI] [PubMed] [Google Scholar]
- 216.Li Y-P, et al. PEGylated PLGA nanoparticles as protein carriers: synthesis, preparation and biodistribution in rats. J Control Release. 2001;71(2):203–211. doi: 10.1016/S0168-3659(01)00218-8. [DOI] [PubMed] [Google Scholar]
- 217.Lei R, et al. Integrated metabolomic analysis of the nano-sized copper particle-induced hepatotoxicity and nephrotoxicity in rats: a rapid invivo screening method for nanotoxicity. Toxicol Appl Pharmacol. 2008;232(2):292–301. doi: 10.1016/j.taap.2008.06.026. [DOI] [PubMed] [Google Scholar]
- 218.Baker GL, et al. Inhalation toxicity and lung toxicokinetics of C60 fullerene nanoparticles and microparticles. Toxicol Sci. 2007;101(1):122–131. doi: 10.1093/toxsci/kfm243. [DOI] [PubMed] [Google Scholar]
- 219.Zhu M-T, et al. Comparative study of pulmonary responses to nano-and submicron-sized ferric oxide in rats. Toxicology. 2008;247(2-3):102–111. doi: 10.1016/j.tox.2008.02.011. [DOI] [PubMed] [Google Scholar]
- 220.Kumar V, Sharma N, Maitra S. In vitro and in vivo toxicity assessment of nanoparticles. Int Nano Lett. 2017;7(4):243–256. doi: 10.1007/s40089-017-0221-3. [DOI] [Google Scholar]