Abstract
Background
Influenza virus (IV)-related pathophysiology suggests that the prognosis of acute respiratory distress syndrome (ARDS) due to IV could be different from the prognosis of ARDS due to other causes. However, the impact of IV infection alone on the prognosis of ARDS patients compared to that of patients with other causes of ARDS has been poorly assessed.
Methods
We compared the 28-day survival from the diagnosis of ARDS with an arterial oxygen tension/inspiratory oxygen fraction ratio ≤150 mmHg between patients with and without IV infection alone. Data were collected prospectively and analysed retrospectively. We first performed survival analysis on the whole population; second, patients with IV infection alone were compared with matched pairs using propensity score matching.
Results
The cohort admitted from October 2009 to March 2020 consisted of 572 patients, including 73 patients (13%) with IV alone. On the first 3 days of mechanical ventilation, nonpulmonary Sequential Organ Failure Assessment scores were significantly lower in patients with IV infection than in the other patients. After the adjusted analysis, IV infection alone remained independently associated with lower mortality at day 28 (hazard ratio 0.51, 95% CI 0.26–0.99, p=0.047). Mortality at day 28 was significantly lower in patients with IV infection alone than in other patients when propensity score matching was used (20% versus 38%, p=0.02).
Conclusions
Our results suggest that patients with ARDS following IV infection alone have a significantly better prognosis at day 28 and less severe nonpulmonary organ dysfunction than do those with ARDS from causes other than IV infection alone.
Short abstract
Influenza virus infection alone is associated with a better short-term prognosis than are other causes of ARDS https://bit.ly/31W2Mh2
Introduction
Acute respiratory distress syndrome (ARDS) is characterised by acute inflammatory lung injury associated with increased pulmonary vascular permeability, leading to the acute onset of bilateral alveolar infiltrates and hypoxaemia [1]. ARDS is a heterogeneous syndrome with subphenotypes [2]. Despite lung-protective ventilation, specific therapies based on experimental studies have been unsuccessful to improve the outcome of ARDS, which continues to confer high mortality with estimates ranging from 26% to 58% [3–5]. Along these lines, although diffuse alveolar damage (DAD) has been considered as the usual histopathological hallmark of ARDS, more recent reports have revealed that the presence of DAD decreases significantly with the implementation of lung-protective ventilation, and several studies suggest that lung injury within the first week of ARDS is highly dependent on the ARDS aetiology, which influences the prognosis of patients [1, 6]. For instance, autopsy of some patients with COVID-19-associated ARDS pneumonia found interstitial inflammatory infiltrates dominated by lymphocytes along with lung mechanical characteristics that led several authors to classify COVID-19-associated ARDS as “nontypical” [7]. Influenza virus (IV) infection is a major and recurrent cause of ARDS that has been the focus of attention since the 2009 H1N1 pandemic of IV A (H1N1pdm2009) [8–10]. Particularities in histological findings and in cytokine production in the lungs were described with IV-associated ARDS, suggesting that the mechanisms involved in lung injury could be specific; therefore, the prognosis of ARDS due to IV could be different from the prognosis of ARDS due to other causes [11–14]. Among important trials assessing treatments dedicated to ARDS, some were conducted before the 2009 H1N1 pandemic, and there is no systematic research of IV during epidemic periods of influenza [15–18]; some after 2009 and the exact proportion of patients with influenza was not always provided [19–21], or only patients with influenza were included [9, 22, 23]. Thus, the impact of IV infection alone on the prognosis of ARDS patients compared to that of patients with other causes of ARDS remains unclear.
We wanted to focus on IV infection as cause of ARDS with the aim of evaluating the impact of influenza alone on the prognosis of ARDS patients. For that purpose, we assessed the short-term survival of patients admitted to our intensive care unit (ICU) over a 10-year period with ARDS due to IV infection alone or with ARDS due to other causes.
Materials and methods
Patients and setting
This study was a retrospective study performed on data collected prospectively in a mixed 21-bed ICU university. The database regarding ARDS patients admitted to our ICU was initiated in 2005 [15]. The study was approved by the hospital's ethics committee (number 16–117). We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) recommendations for cohort studies (supplemental data). We included all patients aged older than 18 years who were admitted between October 1, 2009, and March 1, 2020, for ARDS (according to the American-European Consensus Conference criteria) with an arterial oxygen tension (PaO2)/inspiratory oxygen fraction (FIO2) ratio ≤150 mmHg [15, 19, 24] after at least 12 h of lung-protective mechanical ventilation (MV) with an FIO2≥50% and a positive end-expiratory pressure (PEEP) level ≥5 cmH2O [25]. Of note, the study period was stopped before admission to the ICU of the first patients with SARS-CoV-2 infection in our ICU. Patients who received noninvasive ventilation only were excluded from the study. Since the 2009 H1N1 pandemic, systematic detection of IV in times of epidemics using real-time PCR with reverse transcription (RT–PCR) from respiratory specimens collected at the time of admission to our ICU is routine practice in patients admitted with respiratory failure and/or fever. Up to January 2016, IV was detected using Argen (bioMérieux, Marcilly, France) according to the manufacturer's recommendations. Then, the Seegene Alplex Respiratory panel (Eurobio, Les Ulis, France) became the routine method and was used in accordance with the manufacturer's protocol. All patients with influenza received a double dose of oseltamivir treatment on the first day in the ICU for a maximum duration of 10 days. All patients received selected digestive decontamination when mechanically ventilated [26].
Ventilatory settings
All patients were ventilated as follows: in assist-control mode, the initial tidal volume (VT) was set at 6 mL·kg−1 of predicted body weight, the PEEP level was selected from the PEEP- FIO2 table proposed by the ARDS Network, and the end-inspiratory plateau pressure was measured to be kept <30 cmH2O until the PaO2/FIO2 ratio was >150 mmHg with a level of PEEP ≤10 cmH2O and FIO2≤60%.
Prospective data collection
ARDS diagnosis and severity
When IV alone was isolated in respiratory samples obtained at ICU admission, ARDS was classified as IV ARDS alone. Aetiological causes of non-IV ARDS alone were listed as IV associated with a co-pathogen, non-IV pneumonia, nonpulmonary sepsis, aspiration and miscellaneous [1, 19–21]. Trauma patients were admitted to another ICU in the hospital (surgical ICU). Based on the Berlin criteria [27], patients were retrospectively categorised according to whether they had severe ARDS (PaO2/FIO2≤100 mmHg) or moderate ARDS (PaO2/FIO2>100 mmHg and ≤150 mmHg) on the first day of ARDS diagnosis. Consequently, patients were prospectively selected based on the 150 mmHg PaO2/FIO2 threshold used in 2005 and were classified retrospectively for severity based on the Berlin criteria.
Baseline characteristics of patients and causes of death
In addition to the diagnosis of IV infection, the following variables recorded upon ICU admission and during the ICU stay were included as control variables because they are potentially associated with ARDS and influenza prognosis [1, 4, 11, 28, 29]. Data collected for all patients were as follows: age, sex, the Simplified Acute Physiology Score (SAPS) II [30] (calculated within 24 h after admission) and the daily Sequential Organ Failure Assessment (SOFA) score [31] (calculated on the first 3 days following ARDS diagnosis). Comorbidities included in the analysis were liver cirrhosis, obesity, diabetes mellitus, aplasia and/or recent chemotherapy for a solid tumour or haematological disease, and previous coronary artery and/or valvular disease with treatment. Obesity was defined by a body mass index ≥30 kg·m−2. The following causes of death were distinguished: primary infection-related organ failure, refractory hypoxaemia, mesenteric ischaemia, central nervous system disorder, end-of-life decision and others [32].
Organ supports used in the ICU
Organ supports assessed for prognostic analysis were prone positioning, renal replacement therapy (RRT), vasopressors (dobutamine, epinephrine and norepinephrine at any dose), and extracorporeal membrane oxygenation (ECMO).
Respiratory parameters
The respiratory parameters recorded and included in the analysis were the lowest values of the PaO2/FIO2 ratio, the highest values of expiratory VT and PEEP applied, the ventilator-measured end-inspiratory plateau pressure and the driving pressure (calculated as the ventilator-measured plateau pressure minus the applied PEEP).
End-points
The primary end-point was to compare the 28-day survival from the diagnosis of ARDS between patients with and without IV infection alone. Secondary end-points included a comparison of pulmonary and nonpulmonary organ dysfunction scores between IV-alone and non-IV-alone ARDS patients on the first 3 days of MV from the diagnosis of ARDS.
Statistical analysis
Data are expressed as percentages for categorical variables and as medians and interquartile range ((IQR) 25–75%) for continuous variables. The Chi-squared test was used to compare categorical variables and the Mann–Whitney U-test or Kruskal–Wallis test was used to compare continuous variables. Survival curves were constructed until day 28 from the diagnosis of ARDS by using the Kaplan–Meier method and compared by the log-rank test. We first used a Cox proportional hazard model to determine whether infection with IV alone was independently associated with prognosis at day 28 in an unadjusted and adjusted analysis. For adjustments, variables were removed in a backward stepwise selection process based on a significance level with a p-value of 0.10. Because patients were admitted over a 10-year period, during which the prognosis of ARDS may have changed, the year of admission was entered in the model as a continuous covariate. Furthermore, because of the collinearity between the SAPS II and SOFA scores, only the SAPS II was considered in the adjusted analysis. Then, we performed a propensity score (PS) matching (1/1 ratio) analysis in order to mitigate confounding bias. We used the E-value methodology to assess the robustness of the 28-day results to unmeasured confounding [33, 34]. The following variables were used in the calculation of the PS: age, year of admission to the ICU, comorbidities (i.e. diabetes mellitus, liver cirrhosis, valvular and/or coronary disease with treatment, COPD, obesity, aplasia and/or recent chemotherapy for a solid tumour or haematological disease), ARDS severity according to the Berlin criteria, and organ supports received in the ICU (i.e. prone positioning, ECMO, vasopressors and RRT). One patient with ARDS due to IV alone was matched with a patient with ARDS due to another cause with the closest absolute PS, and the maximum distance allowed between two matched patients was set at 0.2 (i.e. calliper restriction). Tests were two-sided, and we considered p<0.05 as significant. Statistical analyses were performed using Statview 5.0 (SAS Institute Inc., Cary, NC, USA) and the Statistical Package for Social Sciences, version 20 (SPSS, IBM, Chicago, IL, USA). The PS analysis was performed using R 3.3.3 (R Foundation for Statistical Computing, Vienna, Austria) with the MatchIt package.
Results
Patients
During the study period, 11 778 patients were admitted to our ICU, 6960 patients received MV and 572 ARDS patients met the inclusion criteria. IV was isolated in 103 ARDS patients (18%). Among patients with IV infection, H1N1 A virus was involved in 48 patients (46%), H3N2 A virus in 45 patients (44%) and B virus in 10 patients (10%). At admission to the ICU, IV alone was isolated in 73 patients (13%) and was associated with another pathogen in 30 patients (5%) including Streptococcus pneumoniae (n=11), other group A Streptococcus (n=1), Staphylococcus aureus (n=9), Gram-negative bacilli (n=4) and Aspergillus fumigatus (n=5). Aetiological causes in noninfluenza ARDS patients were as follows: noninfluenza pneumonia (n=233) (40%), aspiration (n=89) (15%), nonpulmonary sepsis (n=81) (14%) and miscellaneous (n=67) (13%). The results for the comparisons of patient characteristics of those with IV alone and those with IV with other pathogens are shown in table 1. Although there was no difference for coexisting conditions, patients admitted to the ICU with IV alone differed significantly from the other patients in terms of severity at the time of admission to the ICU and the need for organ supports during the ICU stay (i.e. prone positioning and RRT) and for treatments (i.e. glucocorticoids and vasopressors).
TABLE 1.
Baseline characteristics, interventions and outcomes of patients with acute respiratory distress syndrome (ARDS)
| Whole population (n=572) | Influenza virus alone | p-value | ||
| Yes (n=73) | No (n=499) | |||
| Baseline characteristics | ||||
| SAPS II score points | 52 (38–67) | 46 (35–61) | 57 (40–69) | 0.003 |
| SOFA score points | 10 (8–13) | 9 (7–11) | 10 (8–14) | <0.0001 |
| Age years | 58 (47–68) | 59 (49–66) | 58 (47–69) | 0.51 |
| Male sex | 364 (63) | 48 (66) | 316 (63) | 0.66 |
| Time of presentation to hospital to the ICU days | 2 (0–4) | 2 (0–4) | 2 (0–5) | 0.74 |
| Coexisting condition | ||||
| Diabetes mellitus | 66 (11) | 8 (11) | 58 (12) | 0.86 |
| Liver cirrhosis | 63 (11) | 9 (12) | 54 (11) | 0.70 |
| Valvular and/or coronary disease with treatment | 94 (17) | 10 (14) | 84 (17) | 0.49 |
| Aplasia and/or recent chemotherapy for solid tumour or haematological disease | 99 (17) | 11 (15) | 87 (17) | 0.62 |
| COPD | 126 (22) | 18 (25) | 108 (22) | 0.56 |
| Obesity | 147 (26) | 19 (26) | 128 (26) | 0.94 |
| Severe ARDS# | 349 (61) | 49 (67) | 300 (60) | 0.25 |
| Interventions | ||||
| Prone positioning | 261 (44) | 41 (56) | 219 (44) | 0.049 |
| Neuromuscular blockers | 568 (99) | 73 (100) | 495 (99) | 0.98 |
| Inhaled nitric oxide | 116 (20) | 20 (27) | 96 (19) | 0.10 |
| Extracorporeal membrane oxygenation | 58 (10) | 11 (15) | 47 (9) | 0.13 |
| Glucocorticoids | 336 (59) | 29 (40) | 307 (62) | 0.004 |
| Vasopressors | 512 (89) | 58 (79) | 452 (91) | 0.003 |
| Renal replacement therapy | 217 (38) | 18 (25) | 199 (39) | 0.01 |
| Outcomes | ||||
| Ventilator associated pneumonia | 66 (11) | 9 (12) | 57 (11) | 0.82 |
| Mortality in the ICU | 230 (40) | 21 (29) | 209 (43) | 0.03 |
| Ventilator-free days at day 28 | 2 (0–16) | 5 (0–15) | 0 (0–17) | 0.14 |
| Mortality at day 28 | 197 (34) | 15 (20) | 182 (36) | 0.007 |
Data are presented as median (interquartile range) or n (%), unless otherwise stated. SAPS: Simplified Acute Physiology Score; SOFA: Sequential Organ Failure Assessment; ICU: intensive care unit. #: arterial oxygen tension/inspiratory oxygen fraction ratio ≤100 mmHg.
Respiratory characteristics
During the first 3 days following the diagnosis of ARDS, the PaO2/FIO2 ratios and arterial carbon dioxide tension values did not differ significantly between the two groups of patients. The applied PEEP levels were significantly higher in patients with IV infection alone, while the driving pressures did not differ significantly between the two groups of patients (supplemental table 1a).
Organ dysfunction
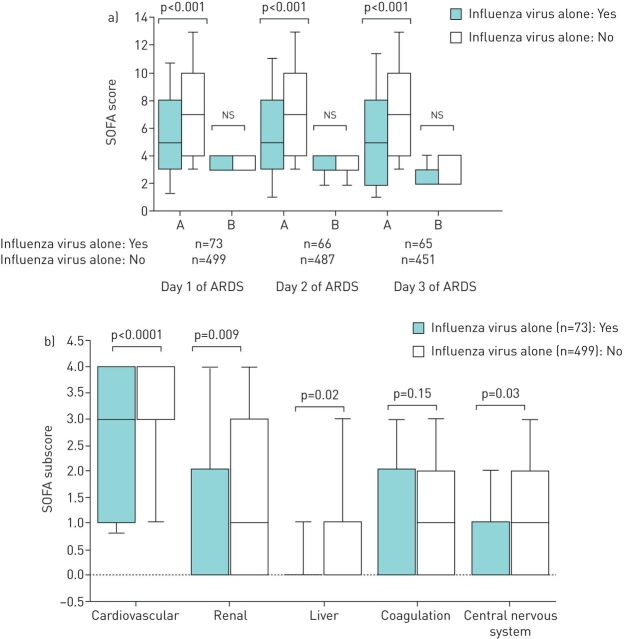
During the first 3 days of MV following the diagnosis of ARDS, the nonpulmonary SOFA scores were significantly lower in patients with ARDS due to IV alone than in other ARDS patients, whereas the SOFA scores for pulmonary dysfunction did not differ significantly (figure 1a). The cardiovascular, liver, renal, and neurological SOFA subscores on the first day of MV were significantly lower in patients with IV infection alone than in other patients (figure 1b).
FIGURE 1.
a) Sequential Organ Failure Assessment (SOFA) scores distinguished between pulmonary and nonpulmonary organ dysfunction on the first 3 days of mechanical ventilation, and b) SOFA subscores on the first day of mechanical ventilation were compared between patients with and without influenza alone. ARDS: acute respiratory distress syndrome; A: SOFA score for extrapulmonary organ dysfunction; B: SOFA score for pulmonary organ dysfunction.
Prognostic analysis and propensity score matching
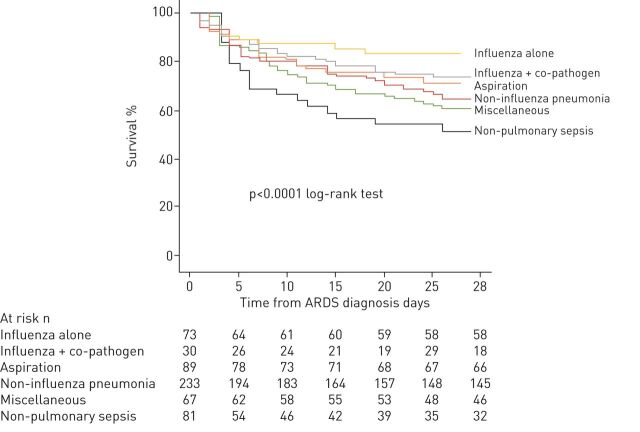
The overall mortality rate at day 28 for the study population was 34% (table 1). Mortality in the ICU and at day 28 and was significantly lower in patients with ARDS due to IV infection alone than in other patients (20% versus 36% and 29% versus 47% respectively, p=0.02 after the two comparisons). Kaplan–Meier survival curves showed that survival differed significantly according to the aetiology of ARDS, and patients with ARDS due to IV alone had highest survival rate (figure 2, p<0.0001 as determined by the log-rank test). The results of the nonadjusted analysis for mortality at day 28 performed on the whole population are listed in table 2.
FIGURE 2.
Cumulative 28-day mortality from admission to the intensive care unit in the whole population. ARDS: acute respiratory distress syndrome.
TABLE 2.
Unadjusted hazard ratios (HRs) for 28-day mortality from the day of acute respiratory distress syndrome (ARDS) diagnosis
| Variables | Unadjusted HR (95% CI) | p-value |
| Influenza virus alone | 0.52 (0.31–0.89) | 0.02 |
| Influenza virus and co-pathogen | 0.76 (0.83–1.56) | 0.46 |
| Non-influenza virus pneumonia | 1.10 (0.83–1.46) | 0.49 |
| Aspiration | 0.64 (0.41–0.99) | 0.048 |
| Nonpulmonary sepsis | 2.40 (1.73–3.32) | <0.001 |
| Miscellaneous | 0.86 (0.55–1.34) | 0.51 |
| Age, per 1-year increment | 1.023 (1.013–1.033) | <0.0001 |
| Male sex | 1.11 (0.83–1.60) | 0.24 |
| Time of presentation to hospital to the ICU, per 1-day increment | 1.24 (0.64–1.86) | 0.59 |
| Prognostic scores | ||
| SAPS II at admission, per 1-point increment | 1.031 (1.024–1.038) | <0.0001 |
| SOFA score on day 1 of ARDS, per 1-point increment | 1.213 (1.171–1.256) | <0.0001 |
| Coexisting condition | ||
| Diabetes mellitus | 1.10 (0.72–1.68) | 0.67 |
| Valvular and/or coronary disease with treatment | 1.20 (0.84–1.72) | 0.31 |
| Aplasia and/or recent chemotherapy for solid tumour or haematological disease | 1.45 (1.06–2.01) | 0.02 |
| Cirrhosis | 2.05 (1.42–2.95) | <0.0001 |
| COPD | 0.76 (0.53–1.07) | 0.14 |
| Obesity | 1.05 (0.77–1.44) | 0.75 |
| MV | ||
| PaO2/FIO2 ratio#, per 1-mmHg increment | 0.990 (0.986–0.995) | <0.0001 |
| Driving pressure#, per 1-point increment | 1.062 (1.035–1.089) | <0.0001 |
| Organ support and treatments | ||
| Treatment with vasopressors | 2.88 (1.47–3.62) | 0.02 |
| Treatment with glucocorticoids | 1.58 (1.14–2.06) | 0.005 |
| Renal replacement therapy | 2.39 (1.84–3.18) | <0.001 |
| Prone positioning | 0.86 (0.64–1.13) | 0.27 |
| Extracorporeal membrane oxygenation | 0.78 (0.45–1.29) | 0.34 |
ICU: intensive care unit; SAPS: Simplified Acute Physiology Score; SOFA: Sequential Organ Failure Assessment; MV: mechanical ventilation; PaO2: arterial oxygen tension; FIO2: inspiratory oxygen fraction. #: worst data recorded between 12 and 24 h of MV from the diagnosis of ARDS, after optimisation of MV.
After adjustments, IV infection alone remained independently associated with a better prognosis at day 28 (hazard ratio (HR) 0.51, 95% CI 0.26–0.99, p=0.047). Extensive results are shown in supplemental table 2a. Results were similar for 90-day mortality from the day of ARDS diagnosis (see supplemental table 2b). The E-value for the HR was 2.56 for the upper limit, and the confidence interval was 1.09. The proportions for causes of death did not differ between IV alone and non-IV alone ARDS (p=0.58) and were distributed as follows: primary infection-related multiple-organ failure (53% versus 50%), refractory hypoxaemia (7% versus 16%), mesenteric ischaemia (13% versus 5%), central nervous system disorder (7% versus 8%), end-of-life decision (0% versus 6%) and others (20% versus 14%).
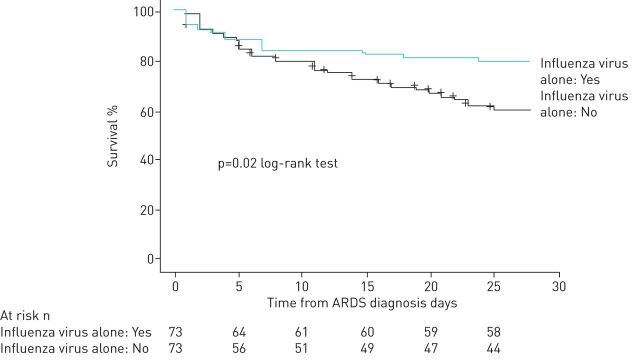
The clinical characteristics of the 73 patients with ARDS due to a cause other than IV alone matched with patients with ARDS due to IV alone are shown in table 3. Patients with ARDS due to IV alone had a significantly lower 28-day mortality (figure 3, p=0.02 as determined by the log-rank test).
TABLE 3.
Baseline characteristics, interventions and outcomes of patients with acute respiratory distress syndrome (ARDS) after propensity score matching
| Influenza virus alone | p-value | ||
| Yes (n=73) | No (n=73) | ||
| Cause of ARDS | |||
| Influenza virus and co-pathogen | 8 (11) | ||
| Aspiration | 14 (19) | ||
| Non-influenza pulmonary infection | 36 (49) | ||
| Nonpulmonary sepsis | 11 (15) | ||
| Miscellaneous | 4 (6) | ||
| Baseline characteristics | |||
| SAPS II score points | 47 (35–61) | 45 (35–59) | 0.87 |
| SOFA score points | 9 (7–11) | 10 (7–12) | 0.18 |
| Age years | 59 (48–65) | 57 (48–68) | 0.84 |
| Male sex | 49 (67) | 42 (58) | 0.23 |
| Coexisting condition | |||
| Diabetes mellitus | 8 (11) | 9 (12) | 0.79 |
| Liver cirrhosis | 9 (12) | 12 (16) | 0.64 |
| Valvular and/or coronary disease with treatment | 10 (14) | 11 (15) | 0.81 |
| Aplasia and/or recent chemotherapy for solid tumour or haematological disease | 11 (15) | 11 (15) | 0.99 |
| COPD | 18 (25) | 12 (16) | 0.69 |
| Obesity | 19 (26) | 24 (33) | 0.36 |
| Severe ARDS# | 50 (68) | 46 (63) | 0.49 |
| Interventions | |||
| Prone positioning | 41 (56) | 39 (53) | 0.99 |
| Neuromuscular blockers | 73 (100) | 73 (100) | >0.99 |
| Inhaled nitric oxide | 20 (27) | 17 (23) | 0.58 |
| Extracorporeal membrane oxygenation | 11 (15) | 13 (18) | 0.65 |
| Glucocorticoids | 30 (41) | 28 (38) | 0.73 |
| Vasopressors | 59 (81) | 65 (90) | 0.16 |
| Renal replacement therapy | 19 (26) | 18 (25) | 0.85 |
| Outcomes | |||
| Ventilator associated pneumonia | 9 (12) | 10 (14) | 0.80 |
| Mortality in the ICU | 21 (29) | 32 (44) | 0.06 |
| Ventilator-free days at day 28 | 6 (0–15) | 0 (0–14) | 0.13 |
| Mortality at day 28 | 15 (20) | 28 (38) | 0.02 |
Data are presented as n (%) or median (interquartile range), unless otherwise stated. SAPS: Simplified Acute Physiology Score; SOFA: Sequential Organ Failure Assessment; ICU: intensive care unit. #: arterial oxygen tension/inspiratory oxygen fraction ratio ≤100 mmHg.
FIGURE 3.
Cumulative 28-day mortality from admission to the intensive care unit in the matched population. ARDS: acute respiratory distress syndrome.
Of note, mortality rates at day 28 did not differ significantly between patients with H1N1pdm2009 virus infection and those with H3N2 virus infection (15% versus 33%, p=0.06).
Discussion
We found in our study that in ARDS patients with a PaO2/FIO2 ratio ≤150 mmHg, a single IV infection remained independently associated with 28-day survival when compared with other causes of ARDS. Importantly, mortality at day 28 remained significantly lower when patients with IV infection alone were matched for both demographic and severity baseline characteristics with patients with non-IV infection alone. The lower mortality observed in patients with IV infection alone did not appear to be related to better lung function, as estimated by blood gas values and data recorded from ventilators, but rather to fewer nonpulmonary organ failures.
Several studies have found that the aetiology of ARDS influences patient outcomes, since the causative agent induces specific lung injury that could be responsible for DAD, which is associated with the outcome of ARDS [1, 35]. Furthermore, comorbid conditions leading to ARDS, such as risk factors for aspiration pneumonia, impact patient outcomes heavily [1, 5, 6, 25, 28]. Causative agents trigger specific injuries to the lung through different mechanisms. For instance, direct caustic actions of a low pH on the airway epithelium during aspiration are followed by an acute neutrophilic inflammatory response that leads to the loss of pulmonary microvascular integrity and extravasation of fluid and protein into the airways and alveoli, which is different from lung injury in IV infection, which is characterised by the replication of IV in the respiratory epithelium followed by the loss of alveolar structure and lung inflammation. Pathological findings in IV-induced lung injury are different from the typical DAD found in ARDS patients and are associated with patient outcome; however, these injuries have been found in IV patients after a long duration of MV [11, 13, 14]. Although no clear data are available, the better outcome in IV-associated ARDS could be supported by its pathophysiological characteristics; it is known in severe respiratory viral infections that T-cells crucially contribute to virus clearance from infected lungs, to the resolution of lung inflammation and thus to a favourable outcome [36, 37]. An immune defect or a superinfection in IV-induced pneumonia is responsible for a longer duration of MV, which could induce lung injury and be responsible for a worse outcome. Steinberg et al. [38] found that alveolar macrophages were increased in ARDS survivors than in nonsurvivors and reached the conclusions that sustained alveolar inflammation was associated with high mortality.
Although lung inflammation following IV infection can spread systematically and lead to multiorgan failure [11, 13, 14], our results for nonpulmonary SOFA subscores show that nonpulmonary organ dysfunction was more pronounced in non-IV alone ARDS patients than in patients with ARDS due to IV alone. It is generally admitted that sepsis-related ARDS is associated with higher mortality than nonsepsis-related ARDS [1, 28]. We found differences in the outcomes not only between patients with extrapulmonary and pulmonary sepsis-related ARDS but also according to the pathogens involved.
Despite the clear specificity highlighted during the IV pandemic in 2009, the proportion of patients with IV infection alone is almost never specified in therapeutic trials conducted before 2009, such as trials assessing neuromuscular blockage [15], fluid management [18] or a protective ventilation strategy [39], or in trials conducted after 2009, such as those assessing prone positioning [20], conducting a large epidemiological survey on general practice [4] or assessing treatment with ECMO [20]. The recent pandemic of COVID-19 has highlighted the specificity of a causative agent responsible for ARDS, since despite ARDS criteria at admission, patients with SARS-CoV2-induced ARDS did not present strict ARDS parameters under MV [40]. We do believe that our results highlight the importance of classification in ARDS patients to achieve specific therapies, including ventilation protocols tailored according to patient subphenotype [2].
The main strength of the study is the large number of ARDS patients who all received lung-protective ventilation during the first days of MV since the diagnosis of ARDS. Our study has several limitations. The study was conducted at a single site; thus, the results may not be applicable to other hospitals. Because of the observational nature of the study and even though we compared matched patients using a PS, we cannot exclude uncontrolled confounders. Nevertheless, the result for the E-value suggests that an unmeasured or unknown confounder would have a substantially greater effect on 28-day mortality with a relative risk exceeding 2.56. In the present study, the unadjusted HRs for the 28-day mortality of cirrhosis and treatment with vasopressors were 2.05 and 2.88, respectively. Finally, we did not perform specific immune signature or biomarker analyses that may support our hypothesis, and we acknowledge that such studies have to be performed before initiation of a therapeutic clinical trial based on specific ARDS causes.
Conclusion
IV was involved in 1 of 10 patients with moderate to severe ARDS admitted to our ICU since the 2009 H1N1 pandemic. We found that IV infection alone was associated with a better short-term survival than other aetiological causes of ARDS encountered in nontrauma patients. Extrapulmonary failure appeared less severe in patients with IV infection alone than in the other patients, explaining in part the better short-term prognosis in these patients.
Supplementary material
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material 00587-2020.SUPPLEMENT (177.4KB, pdf)
Footnotes
Author contributions: All authors contributed to the study conception and design as well as to the acquisition, analysis or interpretation of data. C. Pronier and M. Grégoire performed the laboratory analyses. A. Gacouin and A. Maamar conducted the statistical analysis. M. Lesouhaitier, F. Reizine, A. Gacouin and J.M. Tadié drafted the manuscript, and all authors critically revised the manuscript and approved the final version.
This article has supplementary material available from openres.ersjournals.com
Conflict of interest: A. Gacouin has nothing to disclose.
Conflict of interest: M. Lesouhaitier has nothing to disclose.
Conflict of interest: F. Reizine has nothing to disclose.
Conflict of interest: C. Pronier has nothing to disclose.
Conflict of interest: M. Grégoire has nothing to disclose.
Conflict of interest: B. Painvin has nothing to disclose.
Conflict of interest: A. Maamar has nothing to disclose.
Conflict of interest: V. Thibault has nothing to disclose.
Conflict of interest: Y. Le Tulzo has nothing to disclose.
Conflict of interest: J.M. Tadié has nothing to disclose.
References
- 1.Thompson BT, Chambers RC, Liu KD. Acute respiratory distress syndrome. N Engl J Med 2017; 377: 562–572. doi: 10.1056/NEJMra1608077 [DOI] [PubMed] [Google Scholar]
- 2.Wilson JG, Calfee CS. ARDS subphenotypes: understanding a heterogeneous syndrome. Crit Care Lond Engl 2020; 24: 102. doi: 10.1186/s13054-020-2778-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pham T, Rubenfeld GD. Fifty years of research in ARDS. The epidemiology of acute respiratory distress syndrome. A 50th birthday review. Am J Respir Crit Care Med 2017; 195: 860–870. doi: 10.1164/rccm.201609-1773CP [DOI] [PubMed] [Google Scholar]
- 4.Bellani G, Laffey JG, Pham T, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 2016; 315: 788–800. doi: 10.1001/jama.2016.0291 [DOI] [PubMed] [Google Scholar]
- 5.Phua J, Badia JR, Adhikari NKJ, et al. Has mortality from acute respiratory distress syndrome decreased over time?: a systematic review. Am J Respir Crit Care Med 2009; 179: 220–227. doi: 10.1164/rccm.200805-722OC [DOI] [PubMed] [Google Scholar]
- 6.Pierrakos C, Vincent J-L. The changing pattern of acute respiratory distress syndrome over time: a comparison of two periods. Eur Respir J 2012; 40: 589–595. doi: 10.1183/09031936.00130511 [DOI] [PubMed] [Google Scholar]
- 7.Carsana L, Sonzogni A, Nasr A, et al. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: a two-centre descriptive study. Lancet Infect Dis 2020; 20: 1135–1140. doi: 10.1016/S1473-3099(20)30434-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kumar A, Zarychanski R, Pinto R, et al. Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA 2009; 302: 1872–1879. doi: 10.1001/jama.2009.1496 [DOI] [PubMed] [Google Scholar]
- 9.Noah MA, Peek GJ, Finney SJ, et al. Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 influenza A(H1N1). JAMA 2011; 306: 1659–1668. doi: 10.1001/jama.2011.1471 [DOI] [PubMed] [Google Scholar]
- 10.Australia and New Zealand Extracorporeal Membrane Oxygenation (ANZ ECMO) Influenza Investigators, Davies A, Jones D, et al. Extracorporeal membrane oxygenation for 2009 influenza A(H1N1) acute respiratory distress syndrome. JAMA 2009; 302: 1888–1895. doi: 10.1001/jama.2009.1535 [DOI] [PubMed] [Google Scholar]
- 11.Short KR, Kroeze EJBV, Fouchier RAM, et al. Pathogenesis of influenza-induced acute respiratory distress syndrome. Lancet Infect Dis 2014; 14: 57–69. doi: 10.1016/S1473-3099(13)70286-X [DOI] [PubMed] [Google Scholar]
- 12.Mauad T, Hajjar LA, Callegari GD, et al. Lung pathology in fatal novel human influenza A (H1N1) infection. Am J Respir Crit Care Med 2010; 181: 72–79. doi: 10.1164/rccm.200909-1420OC [DOI] [PubMed] [Google Scholar]
- 13.Herold S, Becker C, Ridge KM, et al. Influenza virus-induced lung injury: pathogenesis and implications for treatment. Eur Respir J 2015; 45: 1463–1478. doi: 10.1183/09031936.00186214 [DOI] [PubMed] [Google Scholar]
- 14.Kalil AC, Thomas PG. Influenza virus-related critical illness: pathophysiology and epidemiology. Crit Care Lond Engl 2019; 23: 258. doi: 10.1186/s13054-019-2539-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Papazian L, Forel JM, Gacouin A, et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med 2010; 363: 1107–1116. doi: 10.1056/NEJMoa1005372 [DOI] [PubMed] [Google Scholar]
- 16.Ferguson ND, Cook DJ, Guyatt GH, et al. High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med 2013; 368: 795–805. doi: 10.1056/NEJMoa1215554 [DOI] [PubMed] [Google Scholar]
- 17.Steinberg KP, Hudson LD, Goodman RB, et al. Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome. N Engl J Med 2006; 354: 1671–1684. doi: 10.1056/NEJMoa051693 [DOI] [PubMed] [Google Scholar]
- 18.National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network, Wiedemann HP, Wheeler AP, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006; 354: 2564–2575. doi: 10.1056/NEJMoa062200 [DOI] [PubMed] [Google Scholar]
- 19.Guerin C, Reignier J, Richard JC, et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 2013; 368: 2159–2168. doi: 10.1056/NEJMoa1214103 [DOI] [PubMed] [Google Scholar]
- 20.Combes A, Hajage D, Capellier G, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med 2018; 378: 1965–1975. doi: 10.1056/NEJMoa1800385 [DOI] [PubMed] [Google Scholar]
- 21.National Heart, Lung, and Blood Institute PETAL Clinical Trials Network, Moss M, Huang DT, et al. Early neuromuscular blockade in the acute respiratory distress syndrome. N Engl J Med 2019; 380: 1997–2008. doi: 10.1056/NEJMoa1901686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pham T, Combes A, Rozé H, et al. Extracorporeal membrane oxygenation for pandemic influenza A(H1N1)-induced acute respiratory distress syndrome: a cohort study and propensity-matched analysis. Am J Respir Crit Care Med 2013; 187: 276–285. doi: 10.1164/rccm.201205-0815OC [DOI] [PubMed] [Google Scholar]
- 23.Brun-Buisson C, Richard J-CM, Mercat A, et al. Early corticosteroids in severe influenza A/H1N1 pneumonia and acute respiratory distress syndrome. Am J Respir Crit Care Med 2011; 183: 1200–1206. doi: 10.1164/rccm.201101-0135OC [DOI] [PubMed] [Google Scholar]
- 24.Maiolo G, Collino F, Vasques F, et al. Reclassifying acute respiratory distress syndrome. Am J Respir Crit Care Med 2018; 197: 1586–1595. doi: 10.1164/rccm.201709-1804OC [DOI] [PubMed] [Google Scholar]
- 25.Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149: 818–824. doi: 10.1164/ajrccm.149.3.7509706 [DOI] [PubMed] [Google Scholar]
- 26.Camus C, Bellissant E, Sebille V, et al. Prevention of acquired infections in intubated patients with the combination of two decontamination regimens. Crit Care Med 2005; 33: 307–314. doi: 10.1097/01.CCM.0000152224.01949.01 [DOI] [PubMed] [Google Scholar]
- 27.Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA 2012; 307: 2526–2533. [DOI] [PubMed] [Google Scholar]
- 28.Ferguson ND, Fan E, Camporota L, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 2012; 38: 1573–1582. doi: 10.1007/s00134-012-2682-1 [DOI] [PubMed] [Google Scholar]
- 29.Paules C, Subbarao K. Influenza. Lancet Lond Engl 2017; 390: 697–708. doi: 10.1016/S0140-6736(17)30129-0 [DOI] [PubMed] [Google Scholar]
- 30.Le Gall J-R, Lemeshow S, Saulnier F. A new simplified acute physiology score (SAPS II) based on a European/North American multicenter study. JAMA 1993; 270: 2957–2963. doi: 10.1001/jama.1993.03510240069035 [DOI] [PubMed] [Google Scholar]
- 31.Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996; 22: 707–710. doi: 10.1007/BF01709751 [DOI] [PubMed] [Google Scholar]
- 32.Daviaud F, Grimaldi D, Dechartres A, et al. Timing and causes of death in septic shock. Ann Intensive Care 2015; 5: 16. doi: 10.1186/s13613-015-0058-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Haneuse S, VanderWeele TJ, Arterburn D. Using the E-value to assess the potential effect of unmeasured confounding in observational studies. JAMA 2019; 321: 602–603. doi: 10.1001/jama.2018.21554 [DOI] [PubMed] [Google Scholar]
- 34.VanderWeele TJ, Ding P. Sensitivity analysis in observational research: introducing the E-value. Ann Intern Med 2017; 167: 268–274. doi: 10.7326/M16-2607 [DOI] [PubMed] [Google Scholar]
- 35.Thille AW, Esteban A, Fernandez-Segoviano P, et al. Comparison of the Berlin definition for acute respiratory distress syndrome with autopsy. Am J Respir Crit Care Med 2013; 187: 761–767. doi: 10.1164/rccm.201211-1981OC [DOI] [PubMed] [Google Scholar]
- 36.Tay MZ, Poh CM, Renia L, et al. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol 2020; 20: 363–374. doi: 10.1038/s41577-020-0311-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhao J, Zhao J, Legge K, et al. Age-related increases in PGD2 expression impair respiratory DC migration, resulting in diminished T cell responses upon respiratory virus infection in mice. J Clin Invest 2011; 121: 4921–4930. doi: 10.1172/JCI59777 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Steinberg KP, Milberg JA, Martin TR, et al. Evolution of bronchoalveolar cell populations in the adult respiratory distress syndrome. Am J Respir Crit Care Med 1994; 150: 113–122. doi: 10.1164/ajrccm.150.1.8025736 [DOI] [PubMed] [Google Scholar]
- 39.Amato MB, Barbas CS, Medeiros DM, et al. Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med 1998; 338: 347–354. doi: 10.1056/NEJM199802053380602 [DOI] [PubMed] [Google Scholar]
- 40.Gattinoni L, Coppola S, Cressoni M, et al. COVID-19 does not lead to a “typical” acute respiratory distress syndrome. Am J Respir Crit Care Med 2020; 201: 1299–1300. doi: 10.1164/rccm.202003-0817LE [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material 00587-2020.SUPPLEMENT (177.4KB, pdf)