Abstract
The coronavirus disease 2019 (COVID-19) pandemic is associated with significant morbidity and mortality throughout the world, predominantly due to lung and cardiovascular injury. The virus responsible for COVID-19 — severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) — gains entry into host cells via angiotensin-converting enzyme 2 (ACE2). ACE2 is a primary enzyme within the key counter-regulatory pathway of the renin-angiotensin system (RAS), which acts to oppose the actions of angiotensin (Ang) II by generating Ang-(1–7) to reduce inflammation and fibrosis and mitigate end-organ damage. As COVID-19 spans multiple organ systems linked to the cardiovascular system, it is imperative to understand clearly how SARS-CoV-2 may affect the multifaceted RAS. In addition, recognition of the role of ACE2 and the RAS in COVID-19 has renewed interest in its role in the pathophysiology of cardiovascular disease in general. We provide researchers with a framework of best practices in basic and clinical research to interrogate the RAS using appropriate methodology, especially those who are relatively new to the field. This is crucial, as there are many limitations inherent in investigating the RAS in experimental models and in humans. We discuss sound methodological approaches to quantifying enzyme content and activity (ACE, ACE2), peptides [Ang II, Ang-(1–7)], and receptors (types 1 and 2 Ang II receptors, Mas receptor). Our goal is to ensure appropriate research methodology for investigations of the RAS in patients with SARS-CoV-2 and COVID-19 in order to ensure optimal rigor and reproducibility and appropriate interpretation of results from these investigations.
Keywords: Hypertension, Cardiovascular, Kidney, ACE2, Ang II, Ang-(1–7), Angiotensin
Introduction
The novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is currently causing the coronavirus disease 2019 (COVID-19) pandemic. Initially described in Wuhan, China in December of 2019, COVID-19 has severely affected human health, the global economy, and day-to-day living worldwide. The primary host protein to which SARS-CoV-2 binds to infect human host cells is angiotensin-converting enzyme 2 (ACE2).1 ACE2 is a homolog of angiotensin-converting enzyme (ACE), a key component of the renin-angiotensin system (RAS), and functions to counterbalance the increased blood pressure, pro-inflammatory, and pro-fibrotic effects of the ACE pathway by converting angiotensin (Ang) II to Ang-(1–7) (Figure 1).2–4 Ang II mediates the above effects via the type 1 Ang II receptor (AT1R), while Ang-(1–7) exerts vasodilatory, anti-inflammatory, and anti-fibrotic effects via its Mas receptor.5–8 Not only is ACE2 a cellular entry point for the virus, but it is hypothesized that SARS-CoV-2 may reduce membrane-bound ACE2, based on prior investigations of SARS-CoV.9 In most patients, the initial infection is limited to the oropharynx or nasopharynx, but in some patients, SARS-CoV-2 can additionally infect pulmonary epithelial cells leading to lung injury.10 In a subset of patients, viral infection and replication has been postulated, but not yet demonstrated, to occur within ACE2-expressing vascular endothelial, gastrointestinal (GI) epithelial, and kidney proximal tubular cells, as well as cardiac myocytes, which may contribute to endothelial dysfunction and vascular disease, as well as contribute to clinical manifestations such as diarrhea, acute kidney injury (AKI), and myocarditis (Figure 2). What makes SARS-CoV-2 infections unique (compared to other common respiratory pathogens like influenza and parainfluenza viruses) is the use of ACE2, a specific protein, rather than protein-linked sialic acid, as its receptor. Furthermore, the RAS may be indirectly involved in the pathophysiology of these other respiratory infections, but in SARS-CoV-2 infection, the RAS may play an important and direct role in development and progression of COVID-19.
Figure 1. Balance of angiotensin (Ang) II and Ang-(1–7) in the renin-angiotensin system (RAS).
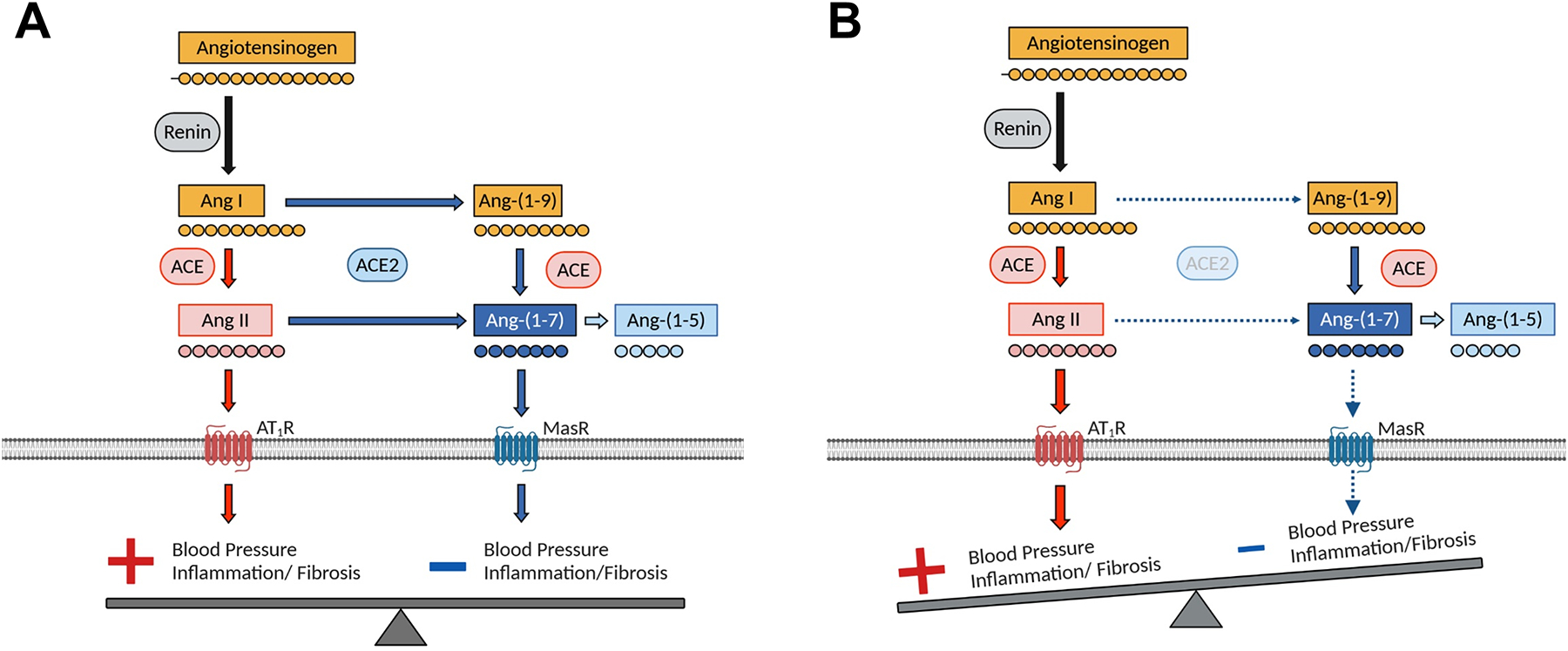
A, In health, the RAS is in balance: blood pressure is normal, and no disease develops. B, With perturbations from various disease states (eg, hypertension or SARS-CoV-2 infection), there is a shift towards the ACE/Ang II pathway and away from the ACE2/Ang-(1–7) pathway. ACE: angiotensin-converting enzyme, ACE2: angiotensin-converting enzyme 2, AT1R: type 1 Ang II receptor. Figure adapted from South et al.2
Figure 2. Spectrum of possible cardiovascular manifestations of COVID-19.
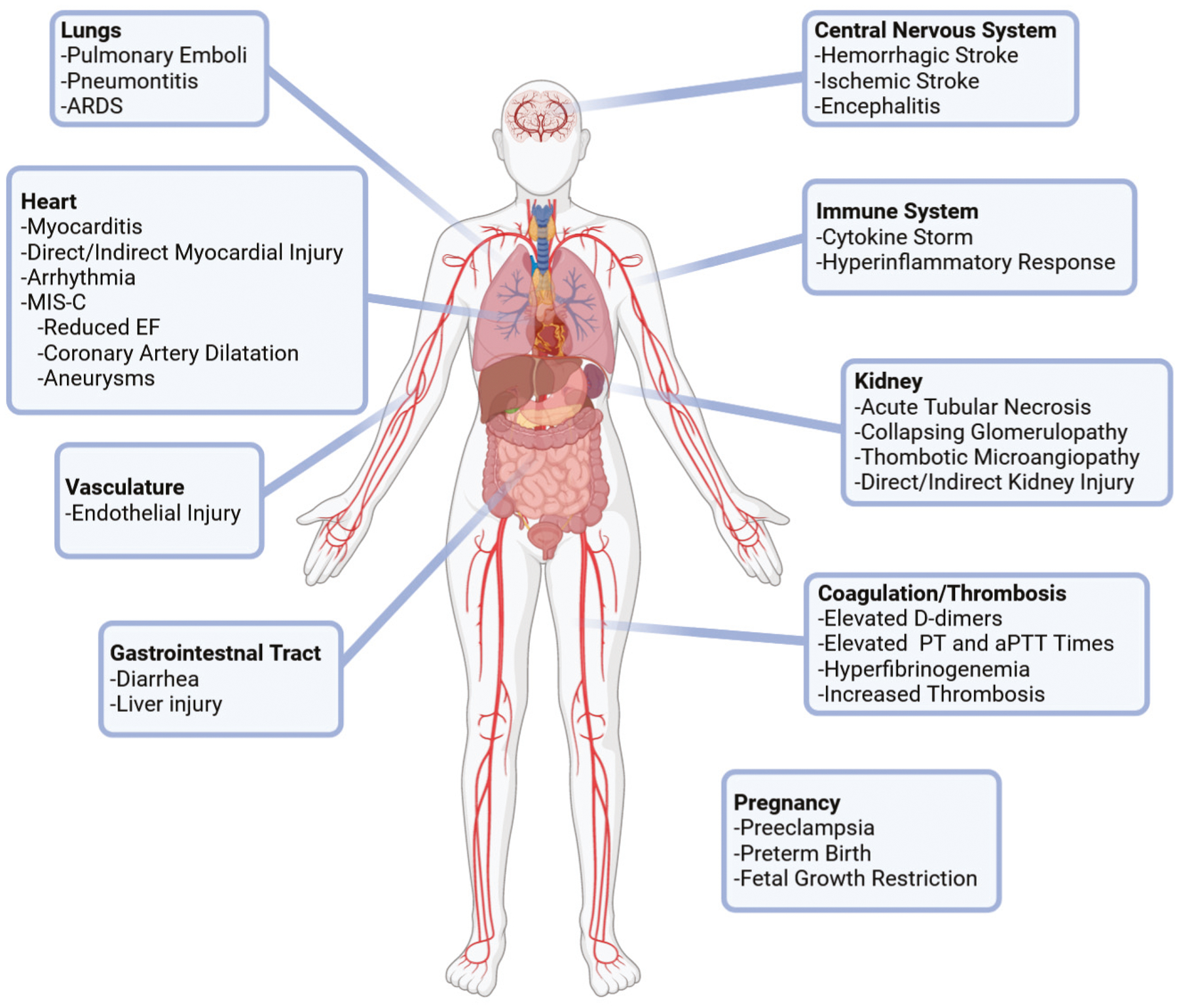
MIS-C: multisystem inflammatory syndrome in children, EF: ejection fraction, PT: prothrombin time, aPTT: activated partial thromboplastin time, D-dimers: dimers of fragment D.
Given concerns that chronic use of ACE inhibitors or Ang II receptor blockers (ARBs) may upregulate ACE2 expression,11, 12 and thereby theoretically increase the risk of SARS-CoV-2 infection, clinical decisions informed by decades of ACE inhibitor/ARB randomized clinical trials (RCTs) in patients with hypertension, kidney disease, and heart disease suddenly were called into question regarding the continuation or initiation of RAS blockade during the COVID-19 pandemic.13, 14 Initially, expert opinions and case series, and later, observational studies, recommended to continue the use of ARBs and ACE inhibitors unless compelling indications to discontinue existed. These statements were made with incomplete insights in an attempt to address this dearth of critical information.15, 16 While this issue has not been resolved, prospective studies and randomized clinical trials and ongoing. Recently, Wysocki et al demonstrated that mice treated with the ACE inhibitor, captopril, or the ARB, telmisartan resulted in no alteration of lung ACE2 expression.17
Given the theoretical importance of this topic, we need to ensure that researchers who may be unfamiliar with the RAS have a guide to avoid the common missteps that occur in interrogating this elaborate system. The RAS is an intricate, multi-enzymatic cascade of peptides that results in multiple G-protein-coupled receptor (GPCR) signaling events, leading to physiological responses. Moreover, the RAS is ubiquitously expressed throughout the body. While research in the RAS has been conducted over the last half a century in cardiovascular and kidney disease, relatively little is known about how this system is perturbed in coronavirus infection. We highlight the myriad of possible ways in which the RAS could play a role in COVID-19 pathophysiology.
In this review, we discuss current knowledge and theory regarding the role of the RAS and ACE2 in SARS-CoV-2 infection and COVID-19; present up-to-date understanding of COVID-19 pathophysiology and related multisystem involvement; and provide multidisciplinary best research practices for future research. Strong clinical evidence coupled with novel basic science evidence is required to confirm how SARS-CoV-2 interacts with and modifies the RAS and how this interaction may influence COVID-19 pathogenesis. Furthermore, standardized and validated techniques for evaluation of RAS components in both clinical and basic science studies are paramount to producing research that is rigorous and reproducible.
Virology of SARS-CoV-2
SARS-CoV-2 is a zoonotic coronavirus that is believed to have begun circulating in humans in November 2019.18 Coronaviruses are enveloped RNA viruses that infect mammals, including humans, and birds, and cause acute and persistent infections.19 A halo of spike (S) proteins gives the viral particles the appearance of a corona, Latin for crown. The coronaviruses are non-segmented positive-strand RNA viruses whose replication is characterized by the transcription of multiple nested subgenomic RNAs, hence their classification in the Nidovirales (nidus is Latin for nest).20
Figure 3 illustrates the replicative cycle of coronaviruses.21 SARS-CoV-2 binds to ACE2 to initiate infection.22 A series of events follows, culminating in the delivery of the encapsidated genome, the nucleocapsid, into the cytoplasm. The genome is unusually long for a RNA virus, about 30,000 nucleotides. The replicase gene codes for a long polyprotein that is cleaved into 16 proteins that include prime targets for antiviral drugs.23
Figure 3. The SARS-CoV-2 replicative cycle.
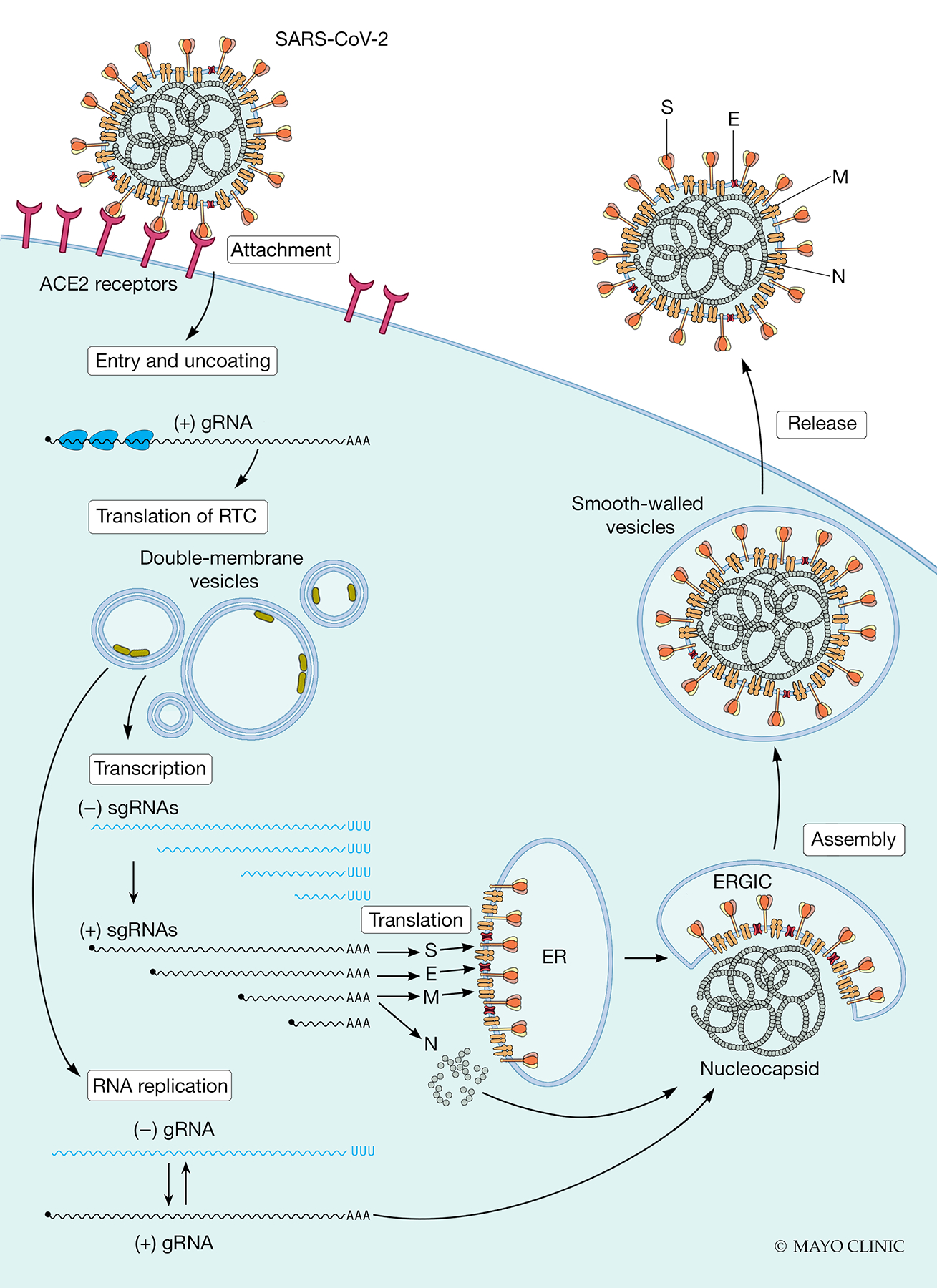
(from left to right). Cell entry of SARS-CoV-2 occurs through recognition of its ACE2 receptor, and after cleavage/activation of the spike (S) protein by cellular proteases, including TMPRSS2. After fusion of the viral envelope with the cellular membrane, the viral positive strand genomic RNA, (+) gRNA, is uncoated and translated into a polyprotein, which is cleaved by viral proteases. The processed non-structural proteins assemble to form the replicase-transcriptase complex (RTC, green blobs). Proteins in this complex elicit the remodeling of membranes to form organelles (double-membrane vesicles) that are dedicated to viral RNA synthesis. The proteins of the RTC then execute viral RNA replication. This process includes production of multiple negative stand (−) subgenomic RNAs (sgRNAs), from which the positive strand (+) sgRNAs are produced. These are also messenger RNAs for the four structural protein S, envelope (E), membrane (M), and nucleocapsid (N). The N protein encapsidates the (+) gRNA to form the nucleocapsid. The S, E, and M proteins, which are co-translationally inserted in the membrane of the endoplasmic reticulum (ER), assemble with the nucleocapsid in the ER-Golgi intermediate compartment (ERGIC). After budding into the ERGIC, viral particles are transported to the plasma membrane in smooth-walled vesicles and released by exocytosis. Figure adapted from Howley and Knipe.238
The other viral components are the membrane (M) protein, which connects the nucleocapsid with the envelope, and the envelope (E) protein, which secures localization of the M protein at the endoplasmic reticulum Golgi intermediate compartment (ERGIC). Following assembly and budding in the ERGIC, exocytosis releases progeny virions.
For SARS-CoV-2, a fraction of the S protein travels to the cell’s plasma membrane, where it can mediate fusion between the infected cell and adjacent uninfected cells that express ACE2. The resultant formation of large, multinucleated syncytiae enables the rapid spread of infection by a route that does not rely on an extracellular phase of completely assembled virions.24
The interaction between the S protein and its receptor is the principal determinant of coronavirus host species range and tissue tropism.1, 25 However, initiation of infection critically depends also on the activation of the S protein by cellular proteases. This process requires at least one protease, located either at the cell surface like transmembrane serine protease 2 (TMPRSS2), or in the endosome, like cathepsins. Furin may also activate the SARS-CoV-2 S protein, which may account for enhanced virulence.26
Clinical Impact of COVID-19 on Systems Relevant to Cardiovascular Medicine
Clinical Impact of SARS-CoV-2 on the Lungs
SARS-CoV-2-induced COVID-19 is largely a lower respiratory tract disease; symptoms and signs include dry cough, pneumonia, dyspnea, hypoxia, and ultimately acute respiratory distress syndrome (ARDS).27 Lung computed tomography has demonstrated pleural effusions, ground-glass opacities (filling of the alveolar spaces with macrophages, fibrin, and proliferating pneumocytes), and progressively diffuse pulmonary hemorrhage.28 Pulmonary vessel hypertrophy, diffuse alveolar damage, and formation of hyaline membranes mainly composed of fibrin have been reported,29, 30 as have pulmonary emboli.31 Moreover, viral load correlates with the ARDS index and the partial pressure of arterial oxygen to percentage of inspired oxygen ratio, and inversely with the lung injury Murray score.28
The pulmonary RAS may mediate COVID-19-related lung injury, as the alveolar epithelium and capillary endothelium express ACE2.1, 32 ACE and ACE2 activity from bronchoalveolar lavage fluid correlate with inflammatory markers in patients with ARDS.33 SARS-CoV-2 infection of epithelial cells and subsequent viral proliferation likely exacerbates alveolar wall destruction and diffuse alveolar damage. SARS-CoV-2 may also target alveolar stem cells.34 Such loss could compromise regenerative processes in the lungs and has stimulated discussion of applying assorted stem cell therapies as therapeutic strategies in COVID-19. Interestingly, viral-induced ACE2 suppression is thought to propagate this acute lung injury by leading to increased lung Ang II content, based on SARS experimental models.9 Mast cell recruitment and degranulation releases chymase, which activates matrix metalloproteinases and further increases Ang II.35–37 Ang II induces alveolar cell apoptosis and lung inflammation and fibrosis, while the ACE2/Ang-(1–7) pathway, and possibly the type 2 Ang II receptor (AT2R), inhibits this process.38–42 Experimental models have shown that acute and chronic lung injury reduce membrane-bound ACE2 and initiate de novo expression of the ACE/Ang II pathway to further drive lung injury.43–45 Thus, direct SARS-CoV-2-induced ACE2/Ang-(1–7) pathway suppression and subsequent ACE/Ang II pathway upregulation may drive COVID-19-related acute lung injury and fibrosis, though this must be confirmed in COVID-19 experimental models and clinical studies.
Clinical Impact of COVID-19 and the Immune System in Cardiovascular Disease
SARS-CoV-2 is thought to magnify interactions between inflammatory responses and cardiovascular disease. Recent data suggests that inflammatory changes within the myocardium are independent of the severity of initial disease presentation and independent of time since disease onset.46 Patients with COVID-19 who manifest the systemic inflammatory response syndrome (SIRS) have increased risk of cardiac injury.47 However, even less ill, non-hospitalized patients who have tested positive for COVID-19 may demonstrate signs of reduced left ventricular ejection fraction (LVEF), native T1 and T2 by cardiac MRI (i.e., signs of myocardial inflammation) and elevated serum troponin.46 Conversely, patients with cardiovascular disease who develop COVID-19 have increased mortality.48 Inflammation associated with COVID-19 appears to contribute to myocarditis and arrhythmias, as well as vascular thrombosis.49, 50 Given the important role of the immune system in cardiovascular disease, understanding how the host immune response to SARS-CoV-2 may enhance cardiovascular risk is paramount.51
COVID-19 has profound effects on both the innate and adaptive immune systems that coalesce to provoke a systemic cytokine release and a resultant cytokine storm in select patients with severe disease. Within the innate immune system, monocyte and macrophage hyperactivation may contribute to increased circulating levels of pro-inflammatory cytokines, including tumor necrosis factor (TNF)-α, interleukin (IL)-6, IL-1, and interferon (IFN)-γ, that are seen in patients with COVID-19 and ARDS, particularly among those admitted to the Intensive Care Unit.52, 53 ACE2 is expressed on macrophages in patients with hypertension,54 and ACE2 overexpression suppresses Ang II-mediated macrophage generation of pro-inflammatory cytokines.55 While it has been shown that SARS-CoV infection reduces membrane-bound ACE2,9 whether macrophages or monocytes contribute to the increased cytokine levels and cardiac risk in COVID-19-associated ARDS is unclear. For example, reductions in ACE2 levels in adipose tissue can secondarily provoke inflammation and cardiac dysfunction.56 Among patients who are critically ill with COVID-19, circulating numbers of neutrophils, the other major innate immune system constituent, are increased compared to those with non-critical illness.57 Autopsy reports suggest that COVID-19-induced formation of neutrophil extracellular traps, web-like structures of DNA and proteins secreted by neutrophils to capture pathogens, may contribute to cytokine storm, vascular thrombosis, and ARDS.58
Within the adaptive immune system, circulating numbers of T cells are similarly reduced in patients with more severe COVID-19.57, 59 As demonstrated in case reports and small retrospective cohort studies, the remaining T cells show more markers of activation,60 higher percentages of pro-inflammatory Th17 cells61 and lower proportions of immunosuppressive T regulatory cells.62 The reduction in circulating T cell numbers could be due to induction of apoptosis signaling pathways in leukocytes.63 Thus, the innate and adaptive immune responses in patients with overlapping COVID-19 and cardiovascular disease are complex and may differentially influence therapeutic considerations in these patients.
Clinical Impact of SARS-CoV-2 on Coagulation
COVID-19 has been associated with a coagulopathy that appears unlikely to be a direct viral effect but rather a consequence of the subsequent inflammatory response.64 Numerous cases of limb ischemia, pulmonary emboli, and strokes in otherwise young, healthy individuals have been reported.65, 66 High rates of venous thrombosis67 arterial thrombosis,66 and microvascular thrombosis68 have been reported in patients with COVID-19. Notably, as is increasingly recognized, venous thromboembolic disease in COVID-19 often occurs despite appropriate prophylactic measures. The endothelium, which can express ACE2,32, 69 is thought to be dysregulated due to inflammatory cytokines shifting to a pro-adhesive and pro-thrombotic state. Microthrombi have also been reported in patients with COVID-19,70 and levels of dimers of fragment D (D-dimers) are markedly increased, with longer prothrombin times and activated partial thromboplastin times in patients with worse outcomes.71, 72 Additional clinical signs include a pro-coagulant profile, including increased clot strength, and hyperfibrinogenemia.73
Previous work has shown that ACE inhibitors enhance release of tissue plasminogen activator and reduce the production of its inhibitor, plasminogen activator 1 (PAI-1).74 A small study of patients with moderate-to-severe COVID-19, in fact, showed improvement in oxygen saturation and reduced alveolar effusion after treatment with plasminogen.30 Moreover, ACE2 is, in general, an antithrombotic enzyme,75 such that loss of ACE2 via internalization with the virus or enhanced shedding76 could exacerbate the coagulopathy. Loss of ACE2 promotes higher levels of Ang II, and Ang II potently upregulates PAI-1 expression, among other pro-coagulant actions. Thus, the RAS may play a role in the development of COVID-19-associated coagulopathy via inflammation-induced endothelial injury.
Clinical Impact of SARS-CoV-2 on the Vasculature
COVID-19 has been associated with endothelial dysfunction, which was originally described in terms of altered vascular responses to acetylcholine,77 but is now characterized by a localized inflammatory response with increased oxidative stress, infiltration of inflammatory cells, and a pro-thrombotic state.78, 79 A recent report studying post-mortem samples obtained from multiple organs of patients with COVID-19 demonstrated inflammatory cell infiltration of the vessel wall, apoptosis, and endothelial cell death.80 Components of the RAS, including ACE2, are expressed in the endothelium and play major role in regulating vascular tone and function.81, 82 It i theoretically is possible that SARS-CoV-2 could infect endothelial cells, though definitive evidence has not been demonstrated. Thus, direct viral downregulation of endothelial ACE2 or secondarily increased Ang II with decreased Ang-(1–7) could further promote a pro-inflammatory response,83 directly via the AT1R, and indirectly through phosphorylation of disintegrin and metalloproteinase domain-containing protein 17 (ADAM17), causing subsequent ACE2 cleavage.84 Taken together, various components of the RAS could mediate multiple aspects of vascular dysfunction that occur in COVID-19, supporting the concept that the RAS provides viable therapeutic targets for further exploration.
Clinical Impact of SARS-CoV-2 on Blood Pressure
A direct effect of SARS-CoV-2 on blood pressure is incompletely described. While hypotension associated with multi-organ dysfunction can occur in severely ill patients with COVID-19,85 it appears to be uncommon at presentation.71 Initial reports suggest that blood pressure at presentation might be higher in patients who go on to develop more severe COVID-19, independent of the presence of baseline hypertension.52, 86 However, this question has not been investigated directly; indeed, most studies have not reported blood pressure.47, 87 Multiple factors that influence blood pressure in acute illness must be accounted for, including fever, inflammatory response, fluid status, and kidney function.
The ACE/Ang II pathway is upregulated in acute illness to maintain adequate organ perfusion. However, if this occurs without an adequate compensatory increase in the ACE2/Ang-(1–7) pathway, deleterious hypertension can occur.88, 89 Indeed, patients with hypertension, heart disease, diabetes mellitus, and chronic kidney disease often demonstrate RAS dysregulation.88, 90 In COVID-19, it is thought that SARS-CoV-2—ACE2 binding and subsequent internalization and downregulation may induce pulmonary RAS dysregulation. Loss of pulmonary endothelial ACE2 could lead to increased circulating Ang II [and decreased Ang-(1–7)] and subsequent unopposed increased vasoconstriction, kidney sodium and water reabsorption (possibly mediated in part by increased aldosterone production and release), and ultimately increased blood pressure.88 Concurrent AKI, and possibly early endothelial dysfunction, could magnify this effect through ACE/Ang II upregulation and other mechanisms. Initial reports demonstrating hypokalemia, possibly due to increased Ang II and aldosterone, support this physiologic theory.86, 91 However, further investigation into these mechanisms in animal and clinical studies is needed, such as serial measurements of RAS components and blood pressure throughout the disease course, including secondary pathways such as chymase-mediated Ang II generation.35, 90
Clinical Impact of SARS-CoV-2 on the Heart
Approximately 20–30% of hospitalized patients with COVID-19 develop acute cardiac injury that appears to be unrelated to viral load and could be related to either direct myocardial injury or secondary to a hyperinflammatory response to the virus in the days to week after acute infection (Figure 3).92 The etiology of COVID-19-related cardiac involvement remains to be determined. COVID-19-related dysfunction manifests as myocardial injury, with elevated serum troponin levels, elevated B-type natriuretic peptide, signs of cardiac ischemia, and decreased left ventricular systolic ejection fraction. The hyperinflammatory response (e.g., stage 3) in adults presents as a myocarditis-like condition.
In its earliest stages, the hyperinflammatory response may be mediated by interferon, and later by activated macrophages and subsequent release of inflammatory cytokines, including IL-1, IL-6, and TNF-α.93 Purported therapies under investigation for the management of cardiac COVID-19 sequelae have included intravenous immunoglobulin, steroids, the anti-IL-6 antibody, tocilizumab,17 anti-IL-1 anakinra, and the antiviral-drug, remdesivir.94, 95 A minority of children appears to be at risk for severe cardiac injury that may be associated with SARS-CoV-2. According to the Centers for Disease Control and Prevention criteria, multisystem inflammatory syndrome in children (MIS-C) is characterized by fever, laboratory evidence of inflammation (≥1 of the following: elevated neutrophil count, C-reactive protein, erythrocyte sedimentation rate, procalcitonin, fibrinogen, D-dimer, ferritin, lactic acid dehydrogenase, or IL-6, reduced lymphocyte count, and hypoalbuminemia), and evidence of multi-organ dysfunction requiring hospitalization (e.g. cardiac, respiratory, hematologic, GI, kidney, dermatologic, or neurologic), as well as evidence of current/recent SARS-CoV-2 infection or recent exposure to a suspected or confirmed SARS-CoV-2 case.94, 96–98 Specifically, MIS-C is associated with depressed left ventricular ejection fraction and coronary artery dilatation, ectasia, or aneurysms.94, 99
Transcriptomic analysis of the heart in patients with hypertrophic obstructive cardiomyopathy revealed that ACE2 was the most robustly expressed transcript, a finding corroborated by quantitative real-time polymerase chain reaction and western blot analysis.100 As with many cardiac diseases, it is generally accepted that ACE2 upregulation is a cytoprotective response that mitigates cardiac compromise. This has several important implications and may explain in part why cardiac disease is a risk factor for COVID-19, as well as why SARS-CoV-2-induced ACE2 suppression could be uniquely damaging to the heart, as this would vitiate this important cytoprotective mechanism. However, it remains unclear if this observation is pertinent to SARS-CoV-2.
Clinical Impact of SARS-CoV-2 on the Central Nervous System
Premorbid cerebrovascular disease is associated with an approximately 2.5-times higher risk of severe COVID-19.101 Conversely, having critical illness due to COVID-19 appears to increase one’s risk of thrombotic events.102 Of 221 patients with COVID-19 from Wuhan, China, acute stroke occurred in 5.9% of patients (ischemic stroke in 11, hemorrhagic stroke and sinus venous thrombosis in one each), with the majority having severe COVID-19.102 However, emergency department stroke code activations dramatically decreased at the onset of the outbreak, raising the possibility that social distancing may limit the spread of non-COVID-19 infections as precipitants of acute stroke pathogenesis, or because patients are delaying care due to fear of exposure to COVID-19 in the hospital.103–105
Numerous mechanisms may contribute to this higher prevalence of cerebrovascular disease during COVID-19.106 Besides a predilection for thrombosis,107 cytokine storm syndrome and hypoxia due to ARDS likely increase the risk of ischemic and hemorrhagic stroke.106, 108 As noted above, the virus may infect endothelial cells directly,80 alter vascular tone of the autonomic nervous system by binding to ACE2,109 and increase blood pressure.106, 109 SARS-CoV-2—ACE2 binding on the capillary endothelial lining may weaken the blood-brain barrier as a means to promote microvascular thrombo-inflammation, as well as allow viral entry into the central nervous system, contributing not only to stroke but also encephalitis.106, 108 SARS-CoV-2 could directly infect the central nervous system, as many cells in the brain express ACE2, though this has yet to be adequately demonstrated.91
Clinical Impact of SARS-CoV-2 on the Kidney
The kidney consequences of COVID-19 are emerging, and AKI is now reported frequently in hospitalized patients.110 In a retrospective analysis of 5449 patients admitted to the hospital in a large New York health care system, 37% had AKI. Of patients admitted to the Intensive Care Unit who required mechanical ventilation, 97% required kidney replacement therapy.111 However, initial reports describing AKI lacked sufficient information to gain much pathophysiologic insight.112 Two reports that included kidney histology from autopsy specimens have provided evidence for severe acute tubular necrosis.113, 114 The mechanisms are unclear and may predominantly involve tubulointerstitial injury from various causes.110 SARS-CoV-2 may have the potential to infect kidney parenchymal cells, as suggested by the presence of virus-like particles in podocytes and tubular cells114 and the presence of viral RNA, but this has not been definitively demonstrated or confirmed.115 The pathophysiology is likely complex, involving viral-mediated injury, cytokine storm, complement activation, hypercoagulation, and microangiopathy, as well as other common contributors to AKI such as hypotension and nephrotoxic medication exposure.112
The ACE2/Ang-(1–7) pathway is highly expressed in the kidneys and co-localizes with ACE/Ang II.116 ACE2 is predominantly expressed on the apical brush border of proximal tubular cells.117 Lower ACE2 expression has been reported in the glomerulus (namely podocytes), distal tubule, interlobular arteriole endothelium, and vascular smooth muscle.117–119 In chronic kidney disease and experimental AKI, ACE expression/activity and Ang II concentration are higher relative to lower ACE2 expression/activity and Ang-(1–7) concentration.120–126 Thus, the RAS could contribute to COVID-19-associated AKI in several ways.112 ACE/Ang II upregulation at the expense of ACE2/Ang-(1–7) downregulation could enhance AKI, independent of direct viral kidney infection, which would propagate inflammation and fibrosis. While SARS-CoV-2 could infect a variety of cell lineages in the kidney that express ACE2, this would be dependent upon viremia and would require priming by TMPRSS2. TMPRSS2, however, is highly expressed in the distal rather than proximal tubule.112, 127 More information clearly is needed to understand AKI pathophysiology in patients with COVID-19.
Clinical Impact of SARS-CoV-2 on the Gastrointestinal Tract
The GI tract is a common target for infection by a variety of coronaviruses, especially in pigs, and the small bowel epithelium abundantly expresses ACE2.128, 129 Anorexia, nausea, vomiting, diarrhea, and abdominal pain are common symptoms, occurring in 30–60% of patients infected with SARS-CoV-2.130, 131 Liver blood tests are often elevated, especially in patients with severe COVID-19, which may indicate SARS-CoV-2-related liver injury, though this has not been proven and is likely multifactorial in etiology.132, 133 Pancreatic blood tests also may be mildly elevated. However, the relationship of abnormal liver and pancreatic tests to the actual infection is unclear.
The gut is the largest immune organ with a tremendous capacity for an inflammatory response that can have a systemic impact. Breakdown of the gut barrier is a common feature of multi-organ failure, accompanying conditions such as ARDS and septic shock, and any infection that involves the gut could amplify inflammation elsewhere. Rapid changes in the microbiome have been described in animal models of infection with delta coronaviruses and recently SARS-CoV-2.128, 134 This dysbiosis and the early disruption of the intestinal barrier together with a potent gut immune response to infection could contribute to systemic cytokine release.135 The apparent intracellular staining of viral nucleocapsid protein in GI epithelial cells and recovery of coronavirus RNA from stool suggests the possibility of a fecal-oral transfer, though this has not been confirmed.136 Similarly, we need to understand if viral ACE2 targeting alters RAS activity in the GI tract, and if that contributes to the GI inflammatory response.137 A focus on mitigating or preventing the impact of the virus on the gut early in the disease may mitigate later systemic consequences of the infection.
Clinical Impact of SARS-CoV-2 on Pregnancy
There is no definitive evidence, to date, that pregnancy increases the risk of more severe COVID-19 or that vertical transmission exists.138–140 However, the RAS could potentially mediate SARS-CoV-2-associated pregnancy and fetal complications. RAS upregulation is crucial to normal pregnancy physiology to maintain maternal hemodynamics and placental blood flow. Both the ACE/Ang II and ACE2/Ang-(1–7) pathways are expressed in the uterus and throughout the placenta.141, 142
Blood pressure tends to be lower early on in the first trimester, in part due to a blunted response to Ang II, increased Ang-(1–7) concentration, and increased production of vasodilatory prostaglandins, but usually increases as pregnancy progresses.143–145 Correspondingly, syncytiotrophoblasts highly express ACE2 in early gestation, but this declines as pregnancy progresses.146
In pathologic states such as preeclampsia, there is a shift towards increased ACE/Ang II pathway expression and decreased ACE2/Ang-(1–7) pathway expression in the placenta and maternal circulation.147 Clinical studies have shown decreased maternal plasma Ang-(1–7) concentration and Ang II hyper-responsiveness in women with preeclampsia.145, 148 Thus, SARS-CoV-2 binding to and the consequent downregulation of uterine/placental ACE2, with increased Ang II relative to decreased Ang-(1–7), could contribute to pregnancy-specific pathology, including preeclampsia, fetal growth restriction, and fetal programming of blood pressure and cardio-metabolic disease.89 Indeed, a recent systematic review of pregnancy outcomes in women with SARS, Middle East respiratory syndrome, or COVID-19 reported a higher pooled proportion of preeclampsia compared to the general population, though the sample size was small (79 total, with 41 women who had COVID-19).149 This preliminary analysis demonstrates the need for further investigation into RAS-mediated mechanisms in the clinical context of preeclampsia and other pregnancy and offspring complications.
Factors Associated with Worse Outcomes in COVID-19: Potential Links to the RAS
Several factors have been associated with worse outcomes in COVID-19. While a majority of these associations are likely attributable to social determinants of health, lack of access to healthcare, racial bias, and the compounding effect of prior health disparities,150 it is possible that biological differences linked to the RAS could play some role. Emerging factors that may be associated with worse COVID-19 outcomes include male sex, racial and/or ethnic minority, obesity, older age, prior history of lung disease, smoking status, and lower socioeconomic status.151–153 The gene for ACE2 is contained on the X chromosome,154 thus differential ACE2 expression may occur with respect to sex. However, data reporting sex differences in ACE2 expression are conflicting. A study performed in rats showed that ACE2 protein expression in the lungs decreases with age, especially in older males compared to older females.155 However, an integrated bioinformatics analysis of single-cell RNA sequencing data in humans indicated that men may have higher ACE2 expression in pulmonary alveolar type II cells compared to women.156 Smoking and chronic obstructive pulmonary disease, which are more common in men, have been associated with higher ACE2 expression, though there are significant methodological limitations to these studies.157–159 Patients with obesity may have higher ACE2 expression in adipose tissue, as obesity is associated with increased ACE/Ang II relative to ACE2/Ang-(1–7) expression.160, 161 However, it is unclear whether differential RAS expression according to these factors has any impact on SARS-CoV-2 infection or COVID-19 severity. It will be important to continue to conduct careful research to evaluate how each of the factors are perturbed in the context of SARS-CoV-2 and whether the RAS is involved.
Best Practices for Basic/Clinical Research of the RAS
Measuring Angiotensin-Converting Enzyme 2 Abundance and Activity
Whereas ACE is a dicarboxypeptidase that cleaves two amino acids from the decapeptide, Ang I, to form the octapeptide, Ang II, ACE2 is a monocarboxypeptidase that cleaves one amino acid, phenylalanine, from Ang II to form the heptapeptide, Ang-(1–7) (Figure 1).3, 162–165 ACE2 also cleaves leucine from Ang I to form Ang-(1–9) (which may have modest biological activity), though with much lower affinity than for Ang II, as well as several other substrates.3, 162–165 ACE2 shares 42% sequence identity with the N- and C-terminal domains of somatic ACE and is the only homologue of ACE that is enzymatically active, but ACE2 is unaffected by all of the ACE inhibitors.3, 166
ACE2 exists in two forms: full length and soluble. Full-length ACE2 has a transmembrane domain, which anchors its extracellular domain to the plasma membrane.167 In polarized epithelia such as in the kidney, lung, and intestine, ACE2 localizes to the apical surface.117, 168 Soluble ACE2, released by sheddases such as ADAM17, lacks the cell membrane-anchoring transmembrane domain and circulates in exceptionally small concentrations in the blood.169 The extracellular domain includes the binding moiety for the S protein of SARS-CoV170, 171 and SARS-CoV-2.1
The relative abundance of full-length ACE2 can be estimated using immunohistochemistry, western blotting, and tissue enzymatic assays (Table 1). Recent studies have provided information on ACE2 localization based on single-cell RNA sequencing analysis.172, 173 However, the levels of messenger RNA do not always correlate with protein levels since ACE2 is frequently regulated post-transcriptionally.172, 173 ACE2 activity can also be measured using high-performance liquid chromatography (HPLC),164, 174 mass spectrometry (MS),175–177 LC-MS/MS165, 178 and an in situ MS-based enzyme activity technique (Table 1).162 These MS and HPLC-based techniques all have in common that they use a natural substrate for ACE2, the peptide Ang II. The products of enzymatic ACE2 cleavage, i.e. Ang-(1–7) and/or the phenylalanine cleaved from Ang II can be quantitatively or semi-quantitatively assessed. A technically less challenging assay, the phenylalanine assay, also uses Ang II as a substrate and measures the formation of phenylalanine to estimate ACE2 activity in biological samples.179–181 This assay is practical but is not specific for ACE2.
Table 1.
Principles and methods used to evaluate ACE2
| Measured parameter | Principle | Methods used | Information |
|---|---|---|---|
| ACE2 activity | Natural ACE2 substrates:
|
|
Quantitative or semiquantitative determination of ACE2 enzyme activity against specific natural substrate |
Artificial ACE2 substrates:
|
Detection of fluorescence formation (fluorescence detecting microplate readers) | Quantitative determination of ACE2 enzyme activity in a high-throughput format | |
| ACE2 protein | Antibody-based assay | Immunohistochemistry Immunofluorescence |
ACE2 protein localization in organs and tissues |
| Immunogold Electron Microscopy | ACE2 protein subcellular distribution | ||
| ELISA | Quantitative determination of ACE2 protein in a high-throughput format | ||
| Western blot | Semi-quantitative determination of ACE2 protein and its immunoreactive fragments |
ACE2 activity assays based on fluorescent substrates are most commonly used. The assays utilize relatively inexpensive intramolecularly quenched fluorescent peptides as the substrate.182–187 They require only regular fluorescence microplate readers and are relatively simple, fast, and convenient to quantitate plasma and tissue ACE2 activity in a high-throughput format. The substrate peptide in these assays usually contains a fluorescent 7-methoxycoumarin group (Mca), which is quenched by energy transfer to a 2,4-dinitrophenyl moiety (Dnp, quencher group). The cleavage of an amide bond between the fluorescent and quencher groups, results in an increase in fluorescence which is used to measure the activity of ACE2 and of other peptidases.188 The two most commonly used peptides for ACE2 activity measurements are Mca-YVADAPK(Dnp)163, 187, 188 and Mca-APK(Dnp).165, 183, 189–192 The Mca-APK(Dnp) substrate is more specific for ACE2 than the Mca-YVADAPK(Dnp) substrate. For evaluation of urine, serum, and kidney ACE2 activity using the intra-molecularly quenched synthetic ACE2-specific substrate, Mca-APK-(Dnp), measurements are performed in black microtiter plates with a 100 μl total volume. MLN-4760 or other inhibitors, such as DX-600, can be used to calculate ACE2-dependent activity. DX-600 is effective in inhibiting human but not rodent ACE2.165, 193 Fluorescence is measured using a microplate fluorescence reader at 320 nm excitation and 420 nm emission wavelength, and ACE2 activity is then calculated from plotting the relative fluorescence units against the linear equations generated for a human or mouse recombinant ACE2 standard.192 It should be emphasized that serum is preferred over plasma for ACE and ACE2 analysis given the potential for assay interference. Many of these substrates are not highly specific for ACE or ACE2, and many of these techniques cannot distinguish between circulating, plasma membrane-bound, and intracellular enzyme activity or content.194
To maintain Ang II at a steady-state level, the formation driven by ACE and the degradation driven by ACE2 should be synchronized under physiological conditions.195 When Ang II formation increases as a result of increased Ang I formation and increased ACE, one would anticipate that ACE2 would eventually increase as well to foster Ang II degradation, otherwise Ang II would increase without a checkpoint. Conversely, if Ang II formation decreases, one would expect a decrease in ACE followed by a decrease in ACE2.195 Overall, the concept of counter-regulation implies that ACE2 changes in the same direction as ACE under physiologic conditions. As such, changes in ACE2 should compensate for changes in ACE in a manner whereby large changes in Ang II are attenuated.195 This, however, has never been fully tested, and other enzymes and substrates are involved, such as aminopeptidase A (Ang II to Ang III [Ang-(2–8)]), neprilysin [Ang I to Ang-(1–7)], and prolyl oligopeptidase [Ang II to Ang-(1–7)]. What is known is that, in pathophysiologic conditions, ACE and ACE2 often change in the opposite direction, similar to Ang II and Ang-(1–7). Discordance in the activity levels of these two enzymes has been well documented in models of diabetic kidney disease.165 The use of the ACE/ACE2 ratio [and Ang II/Ang-(1–7)] is therefore a convenient method to capture relative changes between these two pathways. It is important to emphasize, however, that the ratio may result in misleading conclusions because each enzyme can be regulated independently from the other, so appropriate interpretation is important.
Measurement of RAS Peptides
Dating back to the groundbreaking work of Nussberger and Campbell to develop methods for measuring Ang II,196, 197 appropriate and validated measurements of Ang II and Ang-(1–7) are critical in assessing the RAS, including in interpreting ACE and ACE2 data (Table 2).89, 194, 198 As with ACE and ACE2, Ang II and Ang-(1–7) quantification is difficult and has variable between-laboratory reproducibility. While collection of human samples in hospitalized patients with COVID-19 can be technically challenging, adhering to precise collection, processing, storage, and assay methods by experienced investigators and laboratories whenever possible is necessary to ensure adequate rigor, reproducibility, accuracy, and reliability. Especially relevant to SARS-CoV-2 research, standardized methods and centralized specimen analysis will enable comparisons of RAS measurements across studies and patient populations and enhance the ability to make appropriate inferences.
Table 2.
Comparison of assay limitations to detect Ang peptides
| Limitations | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Low Concen. | Interfering Substances | Ongoing Metabolism | Sequence Homogeneity | Detection Specificity | Posttranslational Modification | Cost and Expertise | ||||
| RIA/ELISA* | X | X | X | X | X | X | ||||
| LC-MS/MS HPLC-MS/MS |
X | X | X | |||||||
Appropriate sample handling and extraction are crucial for all methods of angiotensin peptide detection. Animals should be in a calm environment and medications that acutely alter the RAS be avoided. Blood samples for plasma separation should be collected directly into a peptidase inhibitor cocktail validated for the specific assay being used, while urine typically requires acidification (HCl), peptidase inhibitors, or collection on dry ice. An example of a peptidase inhibitor cocktail tailored to HPLC-RIA includes 1,10-phenanthroline monohydrate, pepstatin, and Na p-hydroxymercuribenzoate.199 Samples should be frozen or extracted immediately. Appropriate sample extraction is required. Particularly for RIA/ELISA, it is critical to perform peptide purification and fractionation (i.e. HPLC-RIA).
Commercially available RIA/ELISA’s utilize antibodies directed against the COOH-terminus and cannot distinguish the NH2-terminus of metabolites.
Limitations to Ang peptide quantification include low concentrations (~fmol/ml), interfering substances, ongoing metabolism (generation and degradation), and sequence homogeneity affecting detection specificity and cross-reactivity (e.g. Ang III with anti-Ang II antibodies).89, 194 Samples should be collected in an EDTA tube with a peptidase inhibitor cocktail validated for the intended assay or possibly ice-cold methanol (Table 2).161, 194, 199 Urine samples should be collected with acidification or inhibitors.161, 194 Samples should be stored at −80°C or immediately extracted. Tissue samples should be rinsed of blood and rapidly frozen in liquid nitrogen or an ethanol/dry ice bath, or immediately extracted.158
RIA and ELISA, if validated with HPLC/MS, can have the requisite sensitivity and specificity, but require optimal conditions and precise methods, including peptide extraction/purification.194,158 The antibodies target each peptide’s unique COOH-terminus, but fail to distinguish the NH2-terminus unless coupled to HPLC. LC-MS/MS and HPLC-MS/MS-based methods are the gold standard but require sufficient expertise and are costly.158 Of note, these assays cannot readily distinguish peptide source (i.e. extracellular vs. intracellular from tissue samples, specific tissue for blood samples). Finally, while normative RAS peptide values do not exist, verified content in various sample sources across species has been established.194
Angiotensin II Type 1 and Type 2 Receptors/Mas Receptor
The main effector peptide of the RAS, Ang II, mediates the classical actions of the RAS via activation of the GPCR AT1R.200 AT1Rs are broadly expressed throughout the body. While humans and primates have a single AT1R, mice and rats have two AT1Rs, termed AT1AR and AT1BR. These are distinct genes encoded on separate chromosomes and have 91% sequence identity.201 The AT1AR has been identified as the closest homologue to the single human AT1R based upon physiologic studies using selective knockout of either the AT1AR or the AT1BR.202, 203 The type 2 Ang II receptor (AT2R) is a GPCR receptor isoform of the RAS, whose activation results in actions opposite of AT1Rs.204 Thus, AT2R activation by Ang II leads to reduced blood pressure, natriuresis, and diminished end organ damage such as fibrosis.204, 205
In the RAS, Ang II generation in humans is largely mediated by ACE and chymase, which are compartmentalized in the blood and tissue.35, 206, 207 New evidence has demonstrated chymase within human cardiomyocytes208 in addition to the AT1R and AT2R on nuclear209–212 and mitochondrial membranes.213, 214 Intracellular Ang II microinjection directly into single kidney proximal tubular cells increases [Ca2+] levels in the presence of extracellular losartan, suggesting activation of intracellular AT1R and AT2R.215
Finally, Ang-(1–7) activation of the Mas receptor initiates a dynamic response that involves an intricate pathway of internalization by either clathrin-coated pits or caveolae through a dynamin-dependent mechanism.216, 217 Endocytic vesicles fuse with early endosomes and stimulate Akt and ERK1/2 phosphorylation, among other signaling events.178 These and similar studies support the function of an intracrine RAS/chymase process that varies from cell to cell and organ to organ, in addition to the known paracrine and autocrine roles of the RAS. This has supplanted the physiologic relevance of previously utilized receptor-binding assays that homogenize tissue and disrupt this important functional compartmentalization, thus leading to significant bias in those studies, which do not account for appropriate methodology. Measurement of ATR is challenging, as commercially available antibodies show a high degree of non-specificity.218 Thus, results from western blot analysis and immunohistochemistry are generally unreliable and proper negative and positive controls should be employed.
Animal Models Relevant to SARS-CoV-2
Gene targeting is a recognized method for establishing the function of a known gene. While more established in mice, genetic manipulation in rats has emerged with new molecular strategies, such as CRISPR/Cas9, but no studies manipulating the ace2 gene in rat models have been published to date. Much has been learned about the components of the RAS by using genetically engineered mice.219 As one example, mice lacking ACE2 were first reported in 2002 and demonstrated a marked cardiac defect.120 Subsequent ACE2 knockout (KO) mouse lines were not able to reproduce this severe contractility defect, but did clearly demonstrate enhanced susceptibility to Ang II-induced hypertension176 and cardiac hypertrophy following transverse aortic constriction.220 These ACE2 KO lines have been further studied in different disease models and to test potential therapeutics, and have been useful in understanding SARS pathology.221 In particular, Kuba et al showed that SARS infection or SARS-CoV S protein dramatically reduced membrane-bound ACE2 in wild-type mice.9 Moreover, while mice treated with SARS-CoV S protein (which binds both human and mouse ACE2) and subsequently subjected to acid instillation in their lungs exhibited worse lung injury and enhanced edema, ACE2 KO mice did not exhibit worsening of lung pathology after S protein exposure.9
More recently, transgenic mice expressing human ACE2 (hACE2) have been generated 222,223 and are available for studies related to viral infection. This is important because the SARS-CoV-2 S protein, much like SARS-CoV, binds human ACE2 with much higher affinity than rodent ACE2.224, 225 These mice express hACE2 in their upper airway, but not alveolar epithelia, as well as in epithelia from liver, kidney, and the GI tract, and in small amounts in the brain.222 When infected with SARS-CoV or SARS-CoV-2, they exhibit macrophage and lymphocyte infiltration in the lungs and pro-inflammatory cytokine and chemokine upregulation in lung and brain, as well as interstitial pneumonia, similar to human disease.222,226 Another novel approach to modeling COVID-19 in mice was recently reported by Hassan et al where wild-type mice were primed for infection by adenoviral delivery of hACE2 to the lungs.227 Lung infection and inflammation are reversed by neutralizing antibodies (a monoclonal chimeric mouse/human antibody constructed with the Fv-human Fc (IgG1) antibody generated after immunization of C57BL/6J mice with SARS-CoV-2 S protein).227 As SARS-CoV-2 entry into cells requires priming by TMPRSS2,1 mice with a deletion of this gene may be of use as well. The range of available models allows investigation of the role of the RAS in COVID-19, and innovation following this pandemic should add to this arsenal.
Best Research Practices for the RAS: Clinical Research
Strong clinical evidence is required to confirm the role of the RAS in COVID-19 and the role of ACE inhibitor and ARB therapy during the pandemic. Key questions include: (1) Does SARS-CoV-2 induce RAS dysregulation; (2) Does RAS dysregulation (e.g. decreased ACE2 and increased Ang II) predict worse outcomes; and (3) Does the RAS mediate the effect of ACE inhibitors/ARBs on outcomes? The answers to these questions may inform risk stratification and treatment strategies. The importance of these questions and need for data in near-real time without the luxury of traditional RCTs has provided a tremendous impetus for collaborations on better-designed observational studies. This section will briefly discuss observational study and RCT methodological considerations.
The explicitly defined study population, exposures, and outcomes of interest inform how one conceptualizes co-morbidities (e.g. hypertension, heart disease) and ACE inhibitor/ARB use as exposures, confounders, effect modifiers, or mediators. Observational studies investigating these topics must adequately address bias to ensure exposure groups are balanced (exchangeability), including confounding (e.g. age), selection, collider, information, and time-dependent biases, including time-varying factors (e.g. kidney function, hypotension).228–231 Methodological considerations include causal diagrams (in particular, directed acyclic graphs), propensity scores, sensitivity analyses, effect modification, and causal mediation.232–237 For example, causal mediation analysis allows us to estimate the direct effect of an exposure (e.g. ACE inhibitor/ARB use) on outcomes and the indirect effect mediated through the RAS (e.g. Ang II, ACE2). Robust observational studies are providing meaningful data sooner and can supplement ongoing RCTs centered on the question of ACE inhibitor/ARB use during COVID-19. Within an RCT, RAS measurements can serve as surrogate outcomes to inform whether the RAS is responsible for the observed treatment effects. Critical appraisal of RCT results is crucial, including determining if the results are generalizable beyond the study population and if major sources of bias have been adequately addressed.
Conclusion
The interaction between the SARS-CoV-2 S protein and ACE2 has generated renewed and great interest in the RAS. Many in the research community are asking questions regarding if and how the RAS modulates SARS-CoV-2 infectivity and COVID-19 morbidity and mortality. We need to ensure that researchers and those who read these studies, especially those who may be unfamiliar with the RAS, have a guide to avoid the common missteps that occur in interrogating this elaborate system.. We highlight the myriad possible ways in which the RAS could play a role in COVID-19 pathophysiology.
The clinical manifestations of COVID-19 include detrimental effects to the heart, central nervous system, lungs, vasculature, kidneys, and GI tract. These system-wide effects are further augmented by derangements of hemolysis, thrombosis, and immune system hyperactivation. Components of the RAS, including ACE2, are expressed throughout each of these organ systems by a variety of cells. Unravelling how the RAS participates in SARS-CoV-2 infectivity and organ damage is a crucial goal. Importantly, as many patients are on inhibitors of the RAS for cardiovascular or kidney disease indications, it is of urgent need to better understand how their risks may be altered.
Fortunately, there are many tools at the disposal of the researcher to examine each of the components of the RAS. However, it is important to note the caveats that exist for each of their uses and to fully understand the strengths and weaknesses of each methodological approach, including enzyme and peptide analysis and animal models and clinical studies. Furthermore, many other components of this system, such as aldosterone, should be measured and considered. Thus, the framework we provide in this review will help guide researchers when developing both clinical and pre-clinical research proposals.
In conclusion, the COVID-19 pandemic has resulted in a dramatic shift in RAS research that traditionally has focused on cardiovascular disease to one that now encompasses another important respiratory viral infection and with further emphasis on the ACE2/Ang-(1–7) pathway. It is important to reflect on lessons learned from the last several decades in research on the RAS and juxtapose this with the pressing need to understand how COVID-19 pathogenesis occurs. The resultant focus on the RAS in SARS-CoV-2 biology will also lead to additional insights, tools, and discoveries into how this important regulatory system potentiates kidney and cardiovascular disease.
Acknowledgments
The authors would like to thank Dr. Vivek Bhalla for organizing this group on behalf of the American Heart Association Council on the Kidney in Cardiovascular Disease. We thank Ritika Revoori and Drs. Bridget Brosnihan, TanYa Gwathmey, and Mark Chappell for help with manuscript preparation, Joanna King for the art of Figure 3, and Dr. Jan Wysocki for providing Table 1. Figures 1 and 2 created with biorender.com.
Sources of Funding
SDC- NIH grants DK118019, HL128355; Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development Grant BX000893; American Heart Association Grant 18TPA34170047.
RC- “COVID-19 Virology” by the Mayo COVID-19 Research Taskforce
ADB- Supported by grants from NIAID (grants AI110173 and AI120698) Amfar (#109593) and Mayo Clinic (HH Shieck Khalifa Bib Zayed Al-Nahyan Named Professorship of Infectious Diseases).
MAP- Grants from Novartis
MAS- Grants from Renal Research Institute
KKG and WRT- NIH grant P01HL095070
AMS- Grants from NIH NHLBI (HL146818, HL148910 and HL148394)
VDG- Grants from NIH: R01-HL136348.
DB- Grants from NIH NIDDK: R01DK104785
Footnotes
Disclosures
DB- Co-inventor of a patent Active Low Molecular Weight Variants of Angiotensin-Converting Enzyme 2, and founder of Angiotensin Therapeutics Inc.
MAP- personal fees for consulting from AstraZeneca, CinCor, Corvidia, DalCor, GlaxoSmithKline, Novartis, Novo Nordisk, Pharmascience, and Sanofi and has equity in DalCor.
ADB is a paid consultant for Abbvie, is a paid member of the DSMB for Corvus Pharmaceuticals, owns equity for scientific advisory work in Zentalis and Nference, and is founder and President of Splissen Therapeutics.
References
- 1.Hoffmann M, Kleine-Weber H, Schroeder S, Kruger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Muller MA, Drosten C, Pohlmann S. Sars-cov-2 cell entry depends on ace2 and tmprss2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280 e278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.South AM, Brady TM, Flynn JT. Ace2 (angiotensin-converting enzyme 2), covid-19, and ace inhibitor and ang ii (angiotensin ii) receptor blocker use during the pandemic: The pediatric perspective. Hypertension. 2020;76:16–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tipnis SR, Hooper NM, Hyde R, Karran E, Christie G, Turner AJ. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J Biol Chem. 2000;275:33238–33243 [DOI] [PubMed] [Google Scholar]
- 4.Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N, Donovan M, Woolf B, Robison K, Jeyaseelan R, Breitbart RE, Acton S. A novel angiotensin-converting enzyme-related carboxypeptidase (ace2) converts angiotensin i to angiotensin 1–9. Circ Res. 2000;87:E1–9 [DOI] [PubMed] [Google Scholar]
- 5.Masi S, Uliana M, Virdis A. Angiotensin ii and vascular damage in hypertension: Role of oxidative stress and sympathetic activation. Vascul Pharmacol. 2019;115:13–17 [DOI] [PubMed] [Google Scholar]
- 6.Sampaio WO, Souza dos Santos RA, Faria-Silva R, da Mata Machado LT, Schiffrin EL, Touyz RM. Angiotensin-(1–7) through receptor mas mediates endothelial nitric oxide synthase activation via akt-dependent pathways. Hypertension. 2007;49:185–192 [DOI] [PubMed] [Google Scholar]
- 7.Santos RA, Simoes e Silva AC, Maric C, Silva DM, Machado RP, de Buhr I, Heringer-Walther S, Pinheiro SV, Lopes MT, Bader M, Mendes EP, Lemos VS, Campagnole-Santos MJ, Schultheiss HP, Speth R, Walther T. Angiotensin-(1–7) is an endogenous ligand for the g protein-coupled receptor mas. Proc Natl Acad Sci U S A. 2003;100:8258–8263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ferrario CM, Chappell MC, Tallant EA, Brosnihan KB, Diz DI. Counterregulatory actions of angiotensin-(1–7). Hypertension. 1997;30:535–541 [DOI] [PubMed] [Google Scholar]
- 9.Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W, Bao L, Zhang B, Liu G, Wang Z, Chappell M, Liu Y, Zheng D, Leibbrandt A, Wada T, Slutsky AS, Liu D, Qin C, Jiang C, Penninger JM. A crucial role of angiotensin converting enzyme 2 (ace2) in sars coronavirus-induced lung injury. Nat Med. 2005;11:875–879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ziegler CGK, Allon SJ, Nyquist SK, Mbano IM, Miao VN, Tzouanas CN, Cao Y, Yousif AS, Bals J, Hauser BM, Feldman J, Muus C, Wadsworth MH 2nd, Kazer SW, Hughes TK, Doran B, Gatter GJ, Vukovic M, Taliaferro F, Mead BE, Guo Z, Wang JP, Gras D, Plaisant M, Ansari M, Angelidis I, Adler H, Sucre JMS, Taylor CJ, Lin B, Waghray A, Mitsialis V, Dwyer DF, Buchheit KM, Boyce JA, Barrett NA, Laidlaw TM, Carroll SL, Colonna L, Tkachev V, Peterson CW, Yu A, Zheng HB, Gideon HP, Winchell CG, Lin PL, Bingle CD, Snapper SB, Kropski JA, Theis FJ, Schiller HB, Zaragosi LE, Barbry P, Leslie A, Kiem HP, Flynn JL, Fortune SM, Berger B, Finberg RW, Kean LS, Garber M, Schmidt AG, Lingwood D, Shalek AK, Ordovas-Montanes J, lung-network@humancellatlas.org HCALBNEa, Network HCALB. Sars-cov-2 receptor ace2 is an interferon-stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell. 2020;181:1016–1035 e1019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ferrario CM, Ahmad S, Groban L. Mechanisms by which angiotensin-receptor blockers increase ace2 levels. Nat Rev Cardiol. 2020;17:378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for covid-19 infection? Lancet Respir Med. 2020;8:e21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.South AM, Tomlinson L, Edmonston D, Hiremath S, Sparks MA. Controversies of renin-angiotensin system inhibition during the covid-19 pandemic. Nat Rev Nephrol. 2020;16:305–307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sparks MA, South A, Welling P, Luther JM, Cohen J, Byrd JB, Burrell LM, Batlle D, Tomlinson L, Bhalla V, Rheault MN, Soler MJ, Swaminathan S, Hiremath S. Sound science before quick judgement regarding ras blockade in covid-19. Clin J Am Soc Nephrol. 2020;15:714–716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Danser AHJ, Epstein M, Batlle D. Renin-angiotensin system blockers and the covid-19 pandemic: At present there is no evidence to abandon renin-angiotensin system blockers. Hypertension. 2020;75:1382–1385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Esler M, Esler D. Can angiotensin receptor-blocking drugs perhaps be harmful in the covid-19 pandemic? J Hypertens. 2020;38:781–782 [DOI] [PubMed] [Google Scholar]
- 17.Wysocki J, Lores E, Ye M, Soler MJ, Batlle D. Kidney and lung ace2 expression after an ace inhibitor or an ang ii receptor blocker: Implications for covid-19. J Am Soc Nephrol. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Andersen KG, Rambaut A, Lipkin WI, Holmes EC, Garry RF. The proximal origin of sars-cov-2. Nat Med. 2020;26:450–452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Coronaviridae Study Group of the International Committee on Taxonomy of V. The species severe acute respiratory syndrome-related coronavirus: Classifying 2019-ncov and naming it sars-cov-2. Nat Microbiol. 2020;5:536–544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim D, Lee JY, Yang JS, Kim JW, Kim VN, Chang H. The architecture of sars-cov-2 transcriptome. Cell. 2020;181:914–921 e910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fehr AR, Perlman S. Coronaviruses: An overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shang J, Ye G, Shi K, Wan Y, Luo C, Aihara H, Geng Q, Auerbach A, Li F. Structural basis of receptor recognition by sars-cov-2. Nature. 2020;581:221–224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Snijder EJ, Limpens R, de Wilde AH, de Jong AWM, Zevenhoven-Dobbe JC, Maier HJ, Faas F, Koster AJ, Barcena M. A unifying structural and functional model of the coronavirus replication organelle: Tracking down rna synthesis. PLoS Biol. 2020;18:e3000715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cifuentes-Munoz N, Dutch RE, Cattaneo R. Direct cell-to-cell transmission of respiratory viruses: The fast lanes. PLoS Pathog. 2018;14:e1007015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Letko M, Marzi A, Munster V. Functional assessment of cell entry and receptor usage for sars-cov-2 and other lineage b betacoronaviruses. Nat Microbiol. 2020;5:562–569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hoffmann M, Kleine-Weber H, Pohlmann S. A multibasic cleavage site in the spike protein of sars-cov-2 is essential for infection of human lung cells. Mol Cell. 2020;78:779–784 e775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in wuhan, china. JAMA. 2020. doi: 10.1001/jama.2020.1585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liu Y, Yang Y, Zhang C, Huang F, Wang F, Yuan J, Wang Z, Li J, Li J, Feng C, Zhang Z, Wang L, Peng L, Chen L, Qin Y, Zhao D, Tan S, Yin L, Xu J, Zhou C, Jiang C, Liu L. Clinical and biochemical indexes from 2019-ncov infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63:364–374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Albarello F, Pianura E, Di Stefano F, Cristofaro M, Petrone A, Marchioni L, Palazzolo C, Schinina V, Nicastri E, Petrosillo N, Campioni P, Eskild P, Zumla A, Ippolito G, Group CIS. 2019-novel coronavirus severe adult respiratory distress syndrome in two cases in italy: An uncommon radiological presentation. Int J Infect Dis. 2020;93:192–197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wu Y, Wang T, Guo C, Zhang D, Ge X, Huang Z, Zhou X, Li Y, Peng Q, Li J. Plasminogen improves lung lesions and hypoxemia in patients with covid-19. QJM. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fabre O, Rebet O, Carjaliu I, Radutoiu M, Gautier L, Hysi I. Severe acute proximal pulmonary embolism and covid-19: A word of caution. Ann Thorac Surg. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ace2 protein, the functional receptor for sars coronavirus. A first step in understanding sars pathogenesis. J Pathol. 2004;203:631–637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schouten LR, van Kaam AH, Kohse F, Veltkamp F, Bos LD, de Beer FM, van Hooijdonk RT, Horn J, Straat M, Witteveen E, Glas GJ, Wieske L, van Vught LA, Wiewel MA, Ingelse SA, Cortjens B, van Woensel JB, Bos AP, Walther T, Schultz MJ, Wosten-van Asperen RM, consortium M. Age-dependent differences in pulmonary host responses in ards: A prospective observational cohort study. Ann Intensive Care. 2019;9:55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yu F, Jia R, Tang Y, Liu J, Wei B. Sars-cov-2 infection and stem cells: Interaction and intervention. Stem Cell Res. 2020;46:101859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dell’Italia LJ, Collawn JF, Ferrario CM. Multifunctional role of chymase in acute and chronic tissue injury and remodeling. Circ Res. 2018;122:319–336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kritas SK, Ronconi G, Caraffa A, Gallenga CE, Ross R, Conti P. Mast cells contribute to coronavirus-induced inflammation: New anti-inflammatory strategy. J Biol Regul Homeost Agents. 2020;34 [DOI] [PubMed] [Google Scholar]
- 37.Trivedi NN, Caughey GH. Mast cell peptidases: Chameleons of innate immunity and host defense. Am J Respir Cell Mol Biol. 2010;42:257–267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shenoy V, Ferreira AJ, Qi Y, Fraga-Silva RA, Diez-Freire C, Dooies A, Jun JY, Sriramula S, Mariappan N, Pourang D, Venugopal CS, Francis J, Reudelhuber T, Santos RA, Patel JM, Raizada MK, Katovich MJ. The angiotensin-converting enzyme 2/angiogenesis-(1–7)/mas axis confers cardiopulmonary protection against lung fibrosis and pulmonary hypertension. Am J Respir Crit Care Med. 2010;182:1065–1072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Uhal BD, Kim JK, Li X, Molina-Molina M. Angiotensin-tgf-beta 1 crosstalk in human idiopathic pulmonary fibrosis: Autocrine mechanisms in myofibroblasts and macrophages. Curr Pharm Des. 2007;13:1247–1256 [DOI] [PubMed] [Google Scholar]
- 40.Uhal BD, Li X, Xue A, Gao X, Abdul-Hafez A. Regulation of alveolar epithelial cell survival by the ace-2/angiotensin 1–7/mas axis. Am J Physiol Lung Cell Mol Physiol. 2011;301:L269–274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang R, Zagariya A, Ibarra-Sunga O, Gidea C, Ang E, Deshmukh S, Chaudhary G, Baraboutis J, Filippatos G, Uhal BD. Angiotensin ii induces apoptosis in human and rat alveolar epithelial cells. Am J Physiol. 1999;276:L885–889 [DOI] [PubMed] [Google Scholar]
- 42.Wagenaar GT, Sengers RM, Laghmani el H, Chen X, Lindeboom MP, Roks AJ, Folkerts G, Walther FJ. Angiotensin ii type 2 receptor ligand pd123319 attenuates hyperoxia-induced lung and heart injury at a low dose in newborn rats. Am J Physiol Lung Cell Mol Physiol. 2014;307:L261–272 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li X, Molina-Molina M, Abdul-Hafez A, Ramirez J, Serrano-Mollar A, Xaubet A, Uhal BD. Extravascular sources of lung angiotensin peptide synthesis in idiopathic pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol. 2006;291:L887–895 [DOI] [PubMed] [Google Scholar]
- 44.Li X, Molina-Molina M, Abdul-Hafez A, Uhal V, Xaubet A, Uhal BD. Angiotensin converting enzyme-2 is protective but downregulated in human and experimental lung fibrosis. Am J Physiol Lung Cell Mol Physiol. 2008;295:L178–185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li X, Zhang H, Soledad-Conrad V, Zhuang J, Uhal BD. Bleomycin-induced apoptosis of alveolar epithelial cells requires angiotensin synthesis de novo. Am J Physiol Lung Cell Mol Physiol. 2003;284:L501–507 [DOI] [PubMed] [Google Scholar]
- 46.Puntmann VO, Carerj ML, Wieters I, Fahim M, Arendt C, Hoffmann J, Shchendrygina A, Escher F, Vasa-Nicotera M, Zeiher AM, Vehreschild M, Nagel E. Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from coronavirus disease 2019 (covid-19). JAMA cardiology. 2020. 10.1001/jamacardio.2020.3557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Shi S, Qin M, Shen B, Cai Y, Liu T, Yang F, Gong W, Liu X, Liang J, Zhao Q, Huang H, Yang B, Huang C. Association of cardiac injury with mortality in hospitalized patients with covid-19 in wuhan, china. JAMA Cardiol. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to covid-19 based on an analysis of data of 150 patients from wuhan, china. Intensive Care Med. 2020;46:846–848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lazzerini PE, Boutjdir M, Capecchi PL. Covid-19, arrhythmic risk and inflammation: Mind the gap! Circulation. 2020 [DOI] [PubMed] [Google Scholar]
- 50.Liu PP, Blet A, Smyth D, Li H. The science underlying covid-19: Implications for the cardiovascular system. Circulation. 2020 [DOI] [PubMed] [Google Scholar]
- 51.Zhu H, Rhee JW, Cheng P, Waliany S, Chang A, Witteles RM, Maecker H, Davis MM, Nguyen PK, Wu SM. Cardiovascular complications in patients with covid-19: Consequences of viral toxicities and host immune response. Curr Cardiol Rep. 2020;22:32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in wuhan, china. Lancet. 2020;395:497–506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kong SL, Chui P, Lim B, Salto-Tellez M. Elucidating the molecular physiopathology of acute respiratory distress syndrome in severe acute respiratory syndrome patients. Virus Res. 2009;145:260–269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Li YQ, Wang SJ, Wang CX, Gao DF, Ding KN, Niu XL. Effect of peroxisome proliferator activated receptor gamma agonist on angiotensin converting enzyme 2 mrna expression in monocyte-derived macrophages of essential hypertensive patients. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2012;34:379–383 [DOI] [PubMed] [Google Scholar]
- 55.Guo YJ, Li WH, Wu R, Xie Q, Cui LQ. Ace2 overexpression inhibits angiotensin ii-induced monocyte chemoattractant protein-1 expression in macrophages. Arch Med Res. 2008;39:149–154 [DOI] [PubMed] [Google Scholar]
- 56.Patel VB, Mori J, McLean BA, Basu R, Das SK, Ramprasath T, Parajuli N, Penninger JM, Grant MB, Lopaschuk GD, Oudit GY. Ace2 deficiency worsens epicardial adipose tissue inflammation and cardiac dysfunction in response to diet-induced obesity. Diabetes. 2016;65:85–95 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zheng Y, Xu H, Yang M, Zeng Y, Chen H, Liu R, Li Q, Zhang N, Wang D. Epidemiological characteristics and clinical features of 32 critical and 67 noncritical cases of covid-19 in chengdu. J Clin Virol. 2020;127:104366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Barnes BJ, Adrover JM, Baxter-Stoltzfus A, Borczuk A, Cools-Lartigue J, Crawford JM, Dassler-Plenker J, Guerci P, Huynh C, Knight JS, Loda M, Looney MR, McAllister F, Rayes R, Renaud S, Rousseau S, Salvatore S, Schwartz RE, Spicer JD, Yost CC, Weber A, Zuo Y, Egeblad M. Targeting potential drivers of covid-19: Neutrophil extracellular traps. J Exp Med. 2020;217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wan S, Xiang Y, Fang W, Zheng Y, Li B, Hu Y, Lang C, Huang D, Sun Q, Xiong Y, Huang X, Lv J, Luo Y, Shen L, Yang H, Huang G, Yang R. Clinical features and treatment of covid-19 patients in northeast chongqing. J Med Virol. 2020;92:797–806 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Thevarajan I, Nguyen THO, Koutsakos M, Druce J, Caly L, van de Sandt CE, Jia X, Nicholson S, Catton M, Cowie B, Tong SYC, Lewin SR, Kedzierska K. Breadth of concomitant immune responses prior to patient recovery: A case report of non-severe covid-19. Nat Med. 2020;26:453–455 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, Liu S, Zhao P, Liu H, Zhu L, Tai Y, Bai C, Gao T, Song J, Xia P, Dong J, Zhao J, Wang FS. Pathological findings of covid-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420–422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, Wang T, Zhang X, Chen H, Yu H, Zhang X, Zhang M, Wu S, Song J, Chen T, Han M, Li S, Luo X, Zhao J, Ning Q. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130:2620–2629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Xiong Y, Liu Y, Cao L, Wang D, Guo M, Jiang A, Guo D, Hu W, Yang J, Tang Z, Wu H, Lin Y, Zhang M, Zhang Q, Shi M, Liu Y, Zhou Y, Lan K, Chen Y. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in covid-19 patients. Emerg Microbes Infect. 2020;9:761–770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Connors JM, Levy JH. Covid-19 and its implications for thrombosis and anticoagulation. Blood. 2020;135:2033–2040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lodigiani C, Iapichino G, Carenzo L, Cecconi M, Ferrazzi P, Sebastian T, Kucher N, Studt JD, Sacco C, Alexia B, Sandri MT, Barco S, Humanitas C-TF. Venous and arterial thromboembolic complications in covid-19 patients admitted to an academic hospital in milan, italy. Thromb Res. 2020;191:9–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Oxley TJ, Mocco J, Majidi S, Kellner CP, Shoirah H, Singh IP, De Leacy RA, Shigematsu T, Ladner TR, Yaeger KA, Skliut M, Weinberger J, Dangayach NS, Bederson JB, Tuhrim S, Fifi JT. Large-vessel stroke as a presenting feature of covid-19 in the young. N Engl J Med. 2020;382:e60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Klok FA, Kruip M, van der Meer NJM, Arbous MS, Gommers D, Kant KM, Kaptein FHJ, van Paassen J, Stals MAM, Huisman MV, Endeman H. Confirmation of the high cumulative incidence of thrombotic complications in critically ill icu patients with covid-19: An updated analysis. Thromb Res. 2020;191:148–150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, Li WW, Li VW, Mentzer SJ, Jonigk D. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in covid-19. N Engl J Med. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chen Y, Guo Y, Pan Y, Zhao ZJ. Structure analysis of the receptor binding of 2019-ncov. Biochem Biophys Res Commun. 2020. 10.1016/j.bbrc.2020.02.071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhang Y, Xiao M, Zhang S, Xia P, Cao W, Jiang W, Chen H, Ding X, Zhao H, Zhang H, Wang C, Zhao J, Sun X, Tian R, Wu W, Wu D, Ma J, Chen Y, Zhang D, Xie J, Yan X, Zhou X, Liu Z, Wang J, Du B, Qin Y, Gao P, Qin X, Xu Y, Zhang W, Li T, Zhang F, Zhao Y, Li Y, Zhang S. Coagulopathy and antiphospholipid antibodies in patients with covid-19. N Engl J Med. 2020;382:e38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with covid-19 in wuhan, china: A retrospective cohort study. Lancet. 2020;395:1054–1062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ranucci M, Ballotta A, Di Dedda U, Bayshnikova E, Dei Poli M, Resta M, Falco M, Albano G, Menicanti L. The procoagulant pattern of patients with covid-19 acute respiratory distress syndrome. J Thromb Haemost. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Roscoe JN, Shapiro VB, Whitaker K, Kim BKE. Classifying changes to preventive interventions: Applying adaptation taxonomies. J Prim Prev. 2019;40:89–109 [DOI] [PubMed] [Google Scholar]
- 75.Fraga-Silva RA, Sorg BS, Wankhede M, Dedeugd C, Jun JY, Baker MB, Li Y, Castellano RK, Katovich MJ, Raizada MK, Ferreira AJ. Ace2 activation promotes antithrombotic activity. Mol Med. 2010;16:210–215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Heurich A, Hofmann-Winkler H, Gierer S, Liepold T, Jahn O, Pohlmann S. Tmprss2 and adam17 cleave ace2 differentially and only proteolysis by tmprss2 augments entry driven by the severe acute respiratory syndrome coronavirus spike protein. J Virol. 2014;88:1293–1307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ludmer PL, Selwyn AP, Shook TL, Wayne RR, Mudge GH, Alexander RW, Ganz P. Paradoxical vasoconstriction induced by acetylcholine in atherosclerotic coronary arteries. N Engl J Med. 1986;315:1046–1051 [DOI] [PubMed] [Google Scholar]
- 78.Godo S, Shimokawa H. Endothelial functions. Arterioscler Thromb Vasc Biol. 2017;37:e108–e114 [DOI] [PubMed] [Google Scholar]
- 79.Yau JW, Teoh H, Verma S. Endothelial cell control of thrombosis. BMC Cardiovasc Disord. 2015;15:130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, Mehra MR, Schuepbach RA, Ruschitzka F, Moch H. Endothelial cell infection and endotheliitis in covid-19. Lancet. 2020;395:1417–1418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lovren F, Pan Y, Quan A, Teoh H, Wang G, Shukla PC, Levitt KS, Oudit GY, Al-Omran M, Stewart DJ, Slutsky AS, Peterson MD, Backx PH, Penninger JM, Verma S. Angiotensin converting enzyme-2 confers endothelial protection and attenuates atherosclerosis. Am J Physiol Heart Circ Physiol. 2008;295:H1377–1384 [DOI] [PubMed] [Google Scholar]
- 82.Sahara M, Ikutomi M, Morita T, Minami Y, Nakajima T, Hirata Y, Nagai R, Sata M. Deletion of angiotensin-converting enzyme 2 promotes the development of atherosclerosis and arterial neointima formation. Cardiovasc Res. 2014;101:236–246 [DOI] [PubMed] [Google Scholar]
- 83.Watanabe T, Barker TA, Berk BC. Angiotensin ii and the endothelium: Diverse signals and effects. Hypertension. 2005;45:163–169 [DOI] [PubMed] [Google Scholar]
- 84.Scott AJ, O’Dea KP, O’Callaghan D, Williams L, Dokpesi JO, Tatton L, Handy JM, Hogg PJ, Takata M. Reactive oxygen species and p38 mitogen-activated protein kinase mediate tumor necrosis factor alpha-converting enzyme (tace/adam-17) activation in primary human monocytes. J Biol Chem. 2011;286:35466–35476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bhatraju PK, Ghassemieh BJ, Nichols M, Kim R, Jerome KR, Nalla AK, Greninger AL, Pipavath S, Wurfel MM, Evans L, Kritek PA, West TE, Luks A, Gerbino A, Dale CR, Goldman JD, O’Mahony S, Mikacenic C. Covid-19 in critically ill patients in the seattle region - case series. N Engl J Med. 2020;382:2012–2022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chen D, Li X, Song Q, Hu C, Su F, Dai J, Ye Y, Huang J, Zhang X. Assessment of hypokalemia and clinical characteristics in patients with coronavirus disease 2019 in wenzhou, china. JAMA Netw Open. 2020;3:e2011122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS, China Medical Treatment Expert Group for C. Clinical characteristics of coronavirus disease 2019 in china. N Engl J Med. 2020;382:1708–1720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.South AM, Diz DI, Chappell MC. Covid-19, ace2, and the cardiovascular consequences. Am J Physiol Heart Circ Physiol. 2020;318:H1084–H1090 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.South AM, Shaltout HA, Washburn LK, Hendricks AS, Diz DI, Chappell MC. Fetal programming and the angiotensin-(1–7) axis: A review of the experimental and clinical data. Clin Sci (Lond). 2019;133:55–74 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Verdecchia P, Cavallini C, Spanevello A, Angeli F. The pivotal link between ace2 deficiency and sars-cov-2 infection. Eur J Intern Med. 2020;76:14–20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lippi G, South AM, Henry BM. Electrolyte imbalances in patients with severe coronavirus disease 2019 (covid-19). Ann Clin Biochem. 2020;57:262–265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Akhmerov A, Marban E. Covid-19 and the heart. Circ Res. 2020;126:1443–1455 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Merad M, Martin JC. Pathological inflammation in patients with covid-19: A key role for monocytes and macrophages. Nat Rev Immunol. 2020;20:355–362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Belhadjer Z, Meot M, Bajolle F, Khraiche D, Legendre A, Abakka S, Auriau J, Grimaud M, Oualha M, Beghetti M, Wacker J, Ovaert C, Hascoet S, Selegny M, Malekzadeh-Milani S, Maltret A, Bosser G, Giroux N, Bonnemains L, Bordet J, Di Filippo S, Mauran P, Falcon-Eicher S, Thambo JB, Lefort B, Moceri P, Houyel L, Renolleau S, Bonnet D. Acute heart failure in multisystem inflammatory syndrome in children (mis-c) in the context of global sars-cov-2 pandemic. Circulation. 2020 [DOI] [PubMed] [Google Scholar]
- 95.Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, Hohmann E, Chu HY, Luetkemeyer A, Kline S, Lopez de Castilla D, Finberg RW, Dierberg K, Tapson V, Hsieh L, Patterson TF, Paredes R, Sweeney DA, Short WR, Touloumi G, Lye DC, Ohmagari N, Oh MD, Ruiz-Palacios GM, Benfield T, Fatkenheuer G, Kortepeter MG, Atmar RL, Creech CB, Lundgren J, Babiker AG, Pett S, Neaton JD, Burgess TH, Bonnett T, Green M, Makowski M, Osinusi A, Nayak S, Lane HC, Members A-SG. Remdesivir for the treatment of covid-19 - preliminary report. N Engl J Med. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Belot A, Antona D, Renolleau S, Javouhey E, Hentgen V, Angoulvant F, Delacourt C, Iriart X, Ovaert C, Bader-Meunier B, Kone-Paut I, Levy-Bruhl D. Sars-cov-2-related paediatric inflammatory multisystem syndrome, an epidemiological study, france, 1 march to 17 may 2020. Euro Surveill. 2020;25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Riphagen S, Gomez X, Gonzalez-Martinez C, Wilkinson N, Theocharis P. Hyperinflammatory shock in children during covid-19 pandemic. Lancet. 2020;395:1607–1608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Feldstein LR, Rose EB, Horwitz SM, Collins JP, Newhams MM, Son MBF, Newburger JW, Kleinman LC, Heidemann SM, Martin AA, Singh AR, Li S, Tarquinio KM, Jaggi P, Oster ME, Zackai SP, Gillen J, Ratner AJ, Walsh RF, Fitzgerald JC, Keenaghan MA, Alharash H, Doymaz S, Clouser KN, Giuliano JS Jr., Gupta A, Parker RM, Maddux AB, Havalad V, Ramsingh S, Bukulmez H, Bradford TT, Smith LS, Tenforde MW, Carroll CL, Riggs BJ, Gertz SJ, Daube A, Lansell A, Coronado Munoz A, Hobbs CV, Marohn KL, Halasa NB, Patel MM, Randolph AG, Overcoming C-I, the CDCC-RT. Multisystem inflammatory syndrome in u.S. Children and adolescents. N Engl J Med. 2020;383:334–346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kaushik S, Aydin SI, Derespina KR, Bansal PB, Kowalsky S, Trachtman R, Gillen JK, Perez MM, Soshnick SH, Conway EE Jr., Bercow A, Seiden HS, Pass RH, Ushay HM, Ofori-Amanfo G, Medar SS. Multisystem inflammatory syndrome in children (mis-c) associated with sars-cov-2 infection: A multi-institutional study from new york city. J Pediatr. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Bos JM, Hebl VB, Oberg AL, Sun Z, Herman DS, Teekakirikul P, Seidman JG, Seidman CE, Dos Remedios CG, Maleszewski JJ, Schaff HV, Dearani JA, Noseworthy PA, Friedman PA, Ommen SR, Brozovich FV, Ackerman MJ. Marked up-regulation of ace2 in hearts of patients with obstructive hypertrophic cardiomyopathy: Implications for sars-cov-2-mediated covid-19. Mayo Clin Proc. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Aggarwal G, Lippi G, Michael Henry B. Cerebrovascular disease is associated with an increased disease severity in patients with coronavirus disease 2019 (covid-19): A pooled analysis of published literature. Int J Stroke. 2020;15:385–389 [DOI] [PubMed] [Google Scholar]
- 102.Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, Chang J, Hong C, Zhou Y, Wang D, Miao X, Li Y, Hu B. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in wuhan, china. JAMA Neurol. 2020. doi: 10.1001/jamaneurol.2020.1127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Baracchini C, Pieroni A, Viaro F, Cianci V, Cattelan AM, Tiberio I, Munari M, Causin F. Acute stroke management pathway during coronavirus-19 pandemic. Neurol Sci. 2020;41:1003–1005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Morelli N, Rota E, Terracciano C, Immovilli P, Spallazzi M, Colombi D, Zaino D, Michieletti E, Guidetti D. The baffling case of ischemic stroke disappearance from the casualty department in the covid-19 era. Eur Neurol. 2020:1–3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Kansagra AP, Goyal MS, Hamilton S, Albers GW. Collateral effect of covid-19 on stroke evaluation in the united states. N Engl J Med. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wu Y, Xu X, Yang L, Liu C, Yang C. Nervous system damage after covid-19 infection: Presence or absence? Brain Behav Immun. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Klok FA, Kruip M, van der Meer NJM, Arbous MS, Gommers D, Kant KM, Kaptein FHJ, van Paassen J, Stals MAM, Huisman MV, Endeman H. Incidence of thrombotic complications in critically ill icu patients with covid-19. Thromb Res. 2020;191:145–147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Baig AM. Neurological manifestations in covid-19 caused by sars-cov-2. CNS Neurosci Ther. 2020;26:499–501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Miller AJ, Arnold AC. The renin-angiotensin system in cardiovascular autonomic control: Recent developments and clinical implications. Clin Auton Res. 2019;29:231–243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Cheng Y, Luo R, Wang K, Zhang M, Wang Z, Dong L, Li J, Yao Y, Ge S, Xu G. Kidney disease is associated with in-hospital death of patients with covid-19. Kidney Int. 2020;97:829–838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Hirsch JS, Ng JH, Ross DW, Sharma P, Shah HH, Barnett RL, Hazzan AD, Fishbane S, Jhaveri KD, Northwell C-RC, Northwell Nephrology C-RC. Acute kidney injury in patients hospitalized with covid-19. Kidney Int. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Batlle D, Soler MJ, Sparks MA, Hiremath S, South AM, Welling PA, Swaminathan S, Covid, Ace2 in Cardiovascular L, Kidney Working G. Acute kidney injury in covid-19: Emerging evidence of a distinct pathophysiology. J Am Soc Nephrol. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Diao B, Wang C, Wang R, Feng Z, Tan Y, Wang H, Wang C, Liu L, Liu Y, Liu Y, Wang G, Yuan Z, Ren L, Wu Y, Chen Y. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (sars-cov-2) infection. MedRxiv. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Su H, Yang M, Wan C, Yi LX, Tang F, Zhu HY, Yi F, Yang HC, Fogo AB, Nie X, Zhang C. Renal histopathological analysis of 26 postmortem findings of patients with covid-19 in china. Kidney Int. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Puelles VG, Lutgehetmann M, Lindenmeyer MT, Sperhake JP, Wong MN, Allweiss L, Chilla S, Heinemann A, Wanner N, Liu S, Braun F, Lu S, Pfefferle S, Schroder AS, Edler C, Gross O, Glatzel M, Wichmann D, Wiech T, Kluge S, Pueschel K, Aepfelbacher M, Huber TB. Multiorgan and renal tropism of sars-cov-2. N Engl J Med. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Ye M, Wysocki J, Naaz P, Salabat MR, LaPointe MS, Batlle D. Increased ace 2 and decreased ace protein in renal tubules from diabetic mice: A renoprotective combination? Hypertension. 2004;43:1120–1125 [DOI] [PubMed] [Google Scholar]
- 117.Ye M, Wysocki J, William J, Soler MJ, Cokic I, Batlle D. Glomerular localization and expression of angiotensin-converting enzyme 2 and angiotensin-converting enzyme: Implications for albuminuria in diabetes. J Am Soc Nephrol. 2006;17:3067–3075 [DOI] [PubMed] [Google Scholar]
- 118.Lely AT, Hamming I, van Goor H, Navis GJ. Renal ace2 expression in human kidney disease. J Pathol. 2004;204:587–593 [DOI] [PubMed] [Google Scholar]
- 119.Soler MJ, Ye M, Wysocki J, William J, Lloveras J, Batlle D. Localization of ace2 in the renal vasculature: Amplification by angiotensin ii type 1 receptor blockade using telmisartan. Am J Physiol Renal Physiol. 2009;296:F398–405 [DOI] [PubMed] [Google Scholar]
- 120.Crackower MA, Sarao R, Oudit GY, Yagil C, Kozieradzki I, Scanga SE, Oliveira-dos-Santos AJ, da Costa J, Zhang L, Pei Y, Scholey J, Ferrario CM, Manoukian AS, Chappell MC, Backx PH, Yagil Y, Penninger JM. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature. 2002;417:822–828 [DOI] [PubMed] [Google Scholar]
- 121.da Silveira KD, Pompermayer Bosco KS, Diniz LR, Carmona AK, Cassali GD, Bruna-Romero O, de Sousa LP, Teixeira MM, Santos RA, Simoes e Silva AC, Ribeiro Vieira MA. Ace2-angiotensin-(1–7)-mas axis in renal ischaemia/reperfusion injury in rats. Clin Sci (Lond). 2010;119:385–394 [DOI] [PubMed] [Google Scholar]
- 122.Mizuiri S, Hemmi H, Arita M, Ohashi Y, Tanaka Y, Miyagi M, Sakai K, Ishikawa Y, Shibuya K, Hase H, Aikawa A. Expression of ace and ace2 in individuals with diabetic kidney disease and healthy controls. Am J Kidney Dis. 2008;51:613–623 [DOI] [PubMed] [Google Scholar]
- 123.Reich HN, Oudit GY, Penninger JM, Scholey JW, Herzenberg AM. Decreased glomerular and tubular expression of ace2 in patients with type 2 diabetes and kidney disease. Kidney Int. 2008;74:1610–1616 [DOI] [PubMed] [Google Scholar]
- 124.Soler MJ, Wysocki J, Batlle D. Ace2 alterations in kidney disease. Nephrol Dial Transplant. 2013;28:2687–2697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wang G, Lai FM, Kwan BC, Lai KB, Chow KM, Li PK, Szeto CC. Expression of ace and ace2 in patients with hypertensive nephrosclerosis. Kidney Blood Press Res. 2011;34:141–149 [DOI] [PubMed] [Google Scholar]
- 126.Yang XH, Wang YH, Wang JJ, Liu YC, Deng W, Qin C, Gao JL, Zhang LY. Role of angiotensin-converting enzyme (ace and ace2) imbalance on tourniquet-induced remote kidney injury in a mouse hindlimb ischemia-reperfusion model. Peptides. 2012;36:60–70 [DOI] [PubMed] [Google Scholar]
- 127.Vaarala MH, Porvari KS, Kellokumpu S, Kyllonen AP, Vihko PT. Expression of transmembrane serine protease tmprss2 in mouse and human tissues. J Pathol. 2001;193:134–140 [DOI] [PubMed] [Google Scholar]
- 128.Li HY, Li BX, Liang QQ, Jin XH, Tang L, Ding QW, Wang ZX, Wei ZY. Porcine deltacoronavirus infection alters bacterial communities in the colon and feces of neonatal piglets. Microbiologyopen. 2020:e1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Camargo SM, Singer D, Makrides V, Huggel K, Pos KM, Wagner CA, Kuba K, Danilczyk U, Skovby F, Kleta R, Penninger JM, Verrey F. Tissue-specific amino acid transporter partners ace2 and collectrin differentially interact with hartnup mutations. Gastroenterology. 2009;136:872–882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Han C, Duan C, Zhang S, Spiegel B, Shi H, Wang W, Zhang L, Lin R, Liu J, Ding Z, Hou X. Digestive symptoms in covid-19 patients with mild disease severity: Clinical presentation, stool viral rna testing, and outcomes. Am J Gastroenterol. 2020;115:916–923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for gastrointestinal infection of sars-cov-2. Gastroenterology. 2020;158:1831–1833 e1833 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sultan S, Lim JK, Altayar O, Davitkov P, Feuerstein JD, Siddique SM, Falck-Ytter Y, El-Serag HB, Aga. Aga institute rapid recommendations for gastrointestinal procedures during the covid-19 pandemic. Gastroenterology. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Xu L, Liu J, Lu M, Yang D, Zheng X. Liver injury during highly pathogenic human coronavirus infections. Liver Int. 2020;40:998–1004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zuo T, Zhang F, Lui GCY, Yeoh YK, Li AYL, Zhan H, Wan Y, Chung A, Cheung CP, Chen N, Lai CKC, Chen Z, Tso EYK, Fung KSC, Chan V, Ling L, Joynt G, Hui DSC, Chan FKL, Chan PKS, Ng SC. Alterations in gut microbiota of patients with covid-19 during time of hospitalization. Gastroenterology. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Miller J, Cantor A, Zachariah P, Ahn D, Martinez M, Margolis K. Gastrointestinal symptoms as a major presentation component of a novel multisystem inflammatory syndrome in children (mis-c) that is related to covid-19: A single center experience of 44 cases. Gastroenterology. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Chen L, Lou J, Bai Y, Wang M. Covid-19 disease with positive fecal and negative pharyngeal and sputum viral tests. Am J Gastroenterol. 2020;115:790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Rubio-Tapia A, Herman ML, Ludvigsson JF, Kelly DG, Mangan TF, Wu TT, Murray JA. Severe spruelike enteropathy associated with olmesartan. Mayo Clin Proc. 2012;87:732–738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Liu D, Li L, Wu X, Zheng D, Wang J, Yang L, Zheng C. Pregnancy and perinatal outcomes of women with coronavirus disease (covid-19) pneumonia: A preliminary analysis. AJR Am J Roentgenol. 2020:1–6 [DOI] [PubMed] [Google Scholar]
- 139.Schwartz DA. An analysis of 38 pregnant women with covid-19, their newborn infants, and maternal-fetal transmission of sars-cov-2: Maternal coronavirus infections and pregnancy outcomes. Arch Pathol Lab Med. 2020 [DOI] [PubMed] [Google Scholar]
- 140.Zaigham M, Andersson O. Maternal and perinatal outcomes with covid-19: A systematic review of 108 pregnancies. Acta Obstet Gynecol Scand. 2020;99:823–829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Anton L, Merrill DC, Neves LA, Diz DI, Corthorn J, Valdes G, Stovall K, Gallagher PE, Moorefield C, Gruver C, Brosnihan KB. The uterine placental bed renin-angiotensin system in normal and preeclamptic pregnancy. Endocrinology. 2009;150:4316–4325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Joyner J, Neves LA, Granger JP, Alexander BT, Merrill DC, Chappell MC, Ferrario CM, Davis WP, Brosnihan KB. Temporal-spatial expression of ang-(1–7) and angiotensin-converting enzyme 2 in the kidney of normal and hypertensive pregnant rats. Am J Physiol Regul Integr Comp Physiol. 2007;293:R169–177 [DOI] [PubMed] [Google Scholar]
- 143.Assali NS, Westersten A. Regional flow-pressure relationship in response to angiotensin in the intact dog and sheep. Circ Res. 1961;9:189–193 [DOI] [PubMed] [Google Scholar]
- 144.Hill CC, Pickinpaugh J. Physiologic changes in pregnancy. Surg Clin North Am. 2008;88:391–401, vii [DOI] [PubMed] [Google Scholar]
- 145.Merrill DC, Karoly M, Chen K, Ferrario CM, Brosnihan KB. Angiotensin-(1–7) in normal and preeclamptic pregnancy. Endocrine. 2002;18:239–245 [DOI] [PubMed] [Google Scholar]
- 146.Pringle KG, Tadros MA, Callister RJ, Lumbers ER. The expression and localization of the human placental prorenin/renin-angiotensin system throughout pregnancy: Roles in trophoblast invasion and angiogenesis? Placenta. 2011;32:956–962 [DOI] [PubMed] [Google Scholar]
- 147.Delforce SJ, Lumbers ER, Ellery SJ, Murthi P, Pringle KG. Dysregulation of the placental renin-angiotensin system in human fetal growth restriction. Reproduction. 2019;158:237–245 [DOI] [PubMed] [Google Scholar]
- 148.Gant NF, Worley RJ, Everett RB, MacDonald PC. Control of vascular responsiveness during human pregnancy. Kidney Int. 1980;18:253–258 [DOI] [PubMed] [Google Scholar]
- 149.Di Mascio D, Khalil A, Saccone G, Rizzo G, Buca D, Liberati M, Vecchiet J, Nappi L, Scambia G, Berghella V, D’Antonio F. Outcome of coronavirus spectrum infections (sars, mers, covid 1 −19) during pregnancy: A systematic review and meta-analysis. Am J Obstet Gynecol MFM. 2020:100107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Novick TK, Rizzolo K, Cervantes L. Covid-19 and kidney disease disparities in the united states. Advances in Chronic Kidney Diseases. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Dietz W, Santos-Burgoa C. Obesity and its implications for covid-19 mortality. Obesity (Silver Spring). 2020;28:1005. [DOI] [PubMed] [Google Scholar]
- 152.Dorn AV, Cooney RE, Sabin ML. Covid-19 exacerbating inequalities in the us. Lancet. 2020;395:1243–1244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Gudbjartsson DF, Helgason A, Jonsson H, Magnusson OT, Melsted P, Norddahl GL, Saemundsdottir J, Sigurdsson A, Sulem P, Agustsdottir AB, Eiriksdottir B, Fridriksdottir R, Gardarsdottir EE, Georgsson G, Gretarsdottir OS, Gudmundsson KR, Gunnarsdottir TR, Gylfason A, Holm H, Jensson BO, Jonasdottir A, Jonsson F, Josefsdottir KS, Kristjansson T, Magnusdottir DN, le Roux L, Sigmundsdottir G, Sveinbjornsson G, Sveinsdottir KE, Sveinsdottir M, Thorarensen EA, Thorbjornsson B, Love A, Masson G, Jonsdottir I, Moller AD, Gudnason T, Kristinsson KG, Thorsteinsdottir U, Stefansson K. Spread of sars-cov-2 in the icelandic population. N Engl J Med. 2020;382:2302–2315 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Liu J, Ji H, Zheng W, Wu X, Zhu JJ, Arnold AP, Sandberg K. Sex differences in renal angiotensin converting enzyme 2 (ace2) activity are 17beta-oestradiol-dependent and sex chromosome-independent. Biol Sex Differ. 2010;1:6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Xie X, Chen J, Wang X, Zhang F, Liu Y. Age- and gender-related difference of ace2 expression in rat lung. Life Sci. 2006;78:2166–2171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Wei X, Xiao Y-T, Wang J, Chen R, Zhang W, Yang Y, Lv D, Qin C, Gu D, Zhang B, Chen W, Hou J, Song N, Zeng G, Ren S. Sex differences in severity and mortality among patients with covid-19: Evidence from pooled literature analysis and insights from integrated bioinformatic analysis. ArXiv. 2020 [Google Scholar]
- 157.Cai G. Bulk and single-cell transcriptomics identify tobacco-use disparity in lung gene expression of ace2, the receptor of 2019-ncov. MedRxiv. 2020 [Google Scholar]
- 158.Chakladar J, Shende N, Li WT, Rajasekaran M, Chang EY, Ongkeko WM. Smoking-mediated upregulation of the androgen pathway leads to increased sars-cov-2 susceptibility. Int J Mol Sci. 2020;21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Leung JM, Yang CX, Tam A, Shaipanich T, Hackett TL, Singhera GK, Dorscheid DR, Sin DD. Ace-2 expression in the small airway epithelia of smokers and copd patients: Implications for covid-19. Eur Respir J. 2020;55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Engin AB, Engin ED, Engin A. Two important controversial risk factors in sars-cov-2 infection: Obesity and smoking. Environ Toxicol Pharmacol. 2020;78:103411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.South AM, Nixon PA, Chappell MC, Diz DI, Russell GB, Jensen ET, Shaltout HA, O’Shea TM, Washburn LK. Association between preterm birth and the renin-angiotensin system in adolescence: Influence of sex and obesity. J Hypertens. 2018;36:2092–2101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Grobe N, Elased KM, Cool DR, Morris M. Mass spectrometry for the molecular imaging of angiotensin metabolism in kidney. Am J Physiol Endocrinol Metab. 2012;302:E1016–1024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Vickers C, Hales P, Kaushik V, Dick L, Gavin J, Tang J, Godbout K, Parsons T, Baronas E, Hsieh F, Acton S, Patane M, Nichols A, Tummino P. Hydrolysis of biological peptides by human angiotensin-converting enzyme-related carboxypeptidase. J Biol Chem. 2002;277:14838–14843 [DOI] [PubMed] [Google Scholar]
- 164.Wysocki J, Ye M, Rodriguez E, Gonzalez-Pacheco FR, Barrios C, Evora K, Schuster M, Loibner H, Brosnihan KB, Ferrario CM, Penninger JM, Batlle D. Targeting the degradation of angiotensin ii with recombinant angiotensin-converting enzyme 2: Prevention of angiotensin ii-dependent hypertension. Hypertension. 2010;55:90–98 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Ye M, Wysocki J, Gonzalez-Pacheco FR, Salem M, Evora K, Garcia-Halpin L, Poglitsch M, Schuster M, Batlle D. Murine recombinant angiotensin-converting enzyme 2: Effect on angiotensin ii-dependent hypertension and distinctive angiotensin-converting enzyme 2 inhibitor characteristics on rodent and human angiotensin-converting enzyme 2. Hypertension. 2012;60:730–740 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Turner AJ, Tipnis SR, Guy JL, Rice G, Hooper NM. Aceh/ace2 is a novel mammalian metallocarboxypeptidase and a homologue of angiotensin-converting enzyme insensitive to ace inhibitors. Can J Physiol Pharmacol. 2002;80:346–353 [DOI] [PubMed] [Google Scholar]
- 167.Wysocki J, Schulze A, Batlle D. Novel variants of angiotensin converting enzyme-2 of shorter molecular size to target the kidney renin angiotensin system. Biomolecules. 2019;9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Ren X, Glende J, Al-Falah M, de Vries V, Schwegmann-Wessels C, Qu X, Tan L, Tschernig T, Deng H, Naim HY, Herrler G. Analysis of ace2 in polarized epithelial cells: Surface expression and function as receptor for severe acute respiratory syndrome-associated coronavirus. J Gen Virol. 2006;87:1691–1695 [DOI] [PubMed] [Google Scholar]
- 169.Wysocki J, Batlle D. Reduced plasma ace2 activity in dialysis patients: Another piece in the conundrum of factors involved in hypertension and cardiovascular morbidity? Nephrol Dial Transplant. 2013;28:2200–2202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang S. The spike protein of sars-cov--a target for vaccine and therapeutic development. Nat Rev Microbiol. 2009;7:226–236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Li W, Moore MJ, Vasilieva N, Sui J, Wong SK, Berne MA, Somasundaran M, Sullivan JL, Luzuriaga K, Greenough TC, Choe H, Farzan M. Angiotensin-converting enzyme 2 is a functional receptor for the sars coronavirus. Nature. 2003;426:450–454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Qi F, Qian S, Zhang S, Zhang Z. Single cell rna sequencing of 13 human tissues identify cell types and receptors of human coronaviruses. Biochem Biophys Res Commun. 2020;526:135–140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Zou X, Chen K, Zou J, Han P, Hao J, Han Z. Single-cell rna-seq data analysis on the receptor ace2 expression reveals the potential risk of different human organs vulnerable to 2019-ncov infection. Front Med. 2020;14:185–192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Ferrario CM, Jessup J, Chappell MC, Averill DB, Brosnihan KB, Tallant EA, Diz DI, Gallagher PE. Effect of angiotensin-converting enzyme inhibition and angiotensin ii receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–2610 [DOI] [PubMed] [Google Scholar]
- 175.Elased KM, Cunha TS, Gurley SB, Coffman TM, Morris M. New mass spectrometric assay for angiotensin-converting enzyme 2 activity. Hypertension. 2006;47:1010–1017 [DOI] [PubMed] [Google Scholar]
- 176.Gurley SB, Allred A, Le TH, Griffiths R, Mao L, Philip N, Haystead TA, Donoghue M, Breitbart RE, Acton SL, Rockman HA, Coffman TM. Altered blood pressure responses and normal cardiac phenotype in ace2-null mice. J Clin Invest. 2006;116:2218–2225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Li N, Zimpelmann J, Cheng K, Wilkins JA, Burns KD. The role of angiotensin converting enzyme 2 in the generation of angiotensin 1–7 by rat proximal tubules. Am J Physiol Renal Physiol. 2005;288:F353–362 [DOI] [PubMed] [Google Scholar]
- 178.Poglitsch M, Domenig O, Schwager C, Stranner S, Peball B, Janzek E, Wagner B, Jungwirth H, Loibner H, Schuster M. Recombinant expression and characterization of human and murine ace2: Species-specific activation of the alternative renin-angiotensin-system. Int J Hypertens. 2012;2012:428950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Keidar S, Strizevsky A, Raz A, Gamliel-Lazarovich A. Ace2 activity is increased in monocyte-derived macrophages from prehypertensive subjects. Nephrol Dial Transplant. 2007;22:597–601 [DOI] [PubMed] [Google Scholar]
- 180.Liu P, Wysocki J, Serfozo P, Ye M, Souma T, Batlle D, Jin J. A fluorometric method of measuring carboxypeptidase activities for angiotensin ii and apelin-13. Sci Rep. 2017;7:45473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Serfozo P, Wysocki J, Gulua G, Schulze A, Ye M, Liu P, Jin J, Bader M, Myöhänen T, García-Horsman JA, Batlle D. Ang ii (angiotensin ii) conversion to angiotensin-(1–7) in the circulation is pop (prolyloligopeptidase)-dependent and ace2 (angiotensin-converting enzyme 2)-independent. Hypertension. 2020;75:173–182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Burrell LM, Risvanis J, Kubota E, Dean RG, MacDonald PS, Lu S, Tikellis C, Grant SL, Lew RA, Smith AI, Cooper ME, Johnston CI. Myocardial infarction increases ace2 expression in rat and humans. Eur Heart J. 2005;26:369–375; discussion 322–364 [DOI] [PubMed] [Google Scholar]
- 183.Donoghue M, Wakimoto H, Maguire CT, Acton S, Hales P, Stagliano N, Fairchild-Huntress V, Xu J, Lorenz JN, Kadambi V, Berul CI, Breitbart RE. Heart block, ventricular tachycardia, and sudden death in ace2 transgenic mice with downregulated connexins. J Mol Cell Cardiol. 2003;35:1043–1053 [DOI] [PubMed] [Google Scholar]
- 184.Douglas GC, O’Bryan MK, Hedger MP, Lee DK, Yarski MA, Smith AI, Lew RA. The novel angiotensin-converting enzyme (ace) homolog, ace2, is selectively expressed by adult leydig cells of the testis. Endocrinology. 2004;145:4703–4711 [DOI] [PubMed] [Google Scholar]
- 185.Ocaranza MP, Godoy I, Jalil JE, Varas M, Collantes P, Pinto M, Roman M, Ramirez C, Copaja M, Diaz-Araya G, Castro P, Lavandero S. Enalapril attenuates downregulation of angiotensin-converting enzyme 2 in the late phase of ventricular dysfunction in myocardial infarcted rat. Hypertension. 2006;48:572–578 [DOI] [PubMed] [Google Scholar]
- 186.Tikellis C, Bialkowski K, Pete J, Sheehy K, Su Q, Johnston C, Cooper ME, Thomas MC. Ace2 deficiency modifies renoprotection afforded by ace inhibition in experimental diabetes. Diabetes. 2008;57:1018–1025 [DOI] [PubMed] [Google Scholar]
- 187.Wysocki J, Ye M, Soler MJ, Gurley SB, Xiao HD, Bernstein KE, Coffman TM, Chen S, Batlle D. Ace and ace2 activity in diabetic mice. Diabetes. 2006;55:2132–2139 [DOI] [PubMed] [Google Scholar]
- 188.Huang L, Sexton DJ, Skogerson K, Devlin M, Smith R, Sanyal I, Parry T, Kent R, Enright J, Wu QL, Conley G, DeOliveira D, Morganelli L, Ducar M, Wescott CR, Ladner RC. Novel peptide inhibitors of angiotensin-converting enzyme 2. J Biol Chem. 2003;278:15532–15540 [DOI] [PubMed] [Google Scholar]
- 189.Wysocki J, Garcia-Halpin L, Ye M, Maier C, Sowers K, Burns KD, Batlle D. Regulation of urinary ace2 in diabetic mice. Am J Physiol Renal Physiol. 2013;305:F600–611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Wysocki J, Goodling A, Burgaya M, Whitlock K, Ruzinski J, Batlle D, Afkarian M. Urine ras components in mice and people with type 1 diabetes and chronic kidney disease. Am J Physiol Renal Physiol. 2017;313:F487–F494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Wysocki J, Ye M, Khattab AM, Fogo A, Martin A, David NV, Kanwar Y, Osborn M, Batlle D. Angiotensin-converting enzyme 2 amplification limited to the circulation does not protect mice from development of diabetic nephropathy. Kidney Int. 2017;91:1336–1346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Xiao F, Burns KD. Measurement of angiotensin converting enzyme 2 activity in biological fluid (ace2). Methods Mol Biol. 2017;1527:101–115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Pedersen KB, Sriramula S, Chhabra KH, Xia H, Lazartigues E. Species-specific inhibitor sensitivity of angiotensin-converting enzyme 2 (ace2) and its implication for ace2 activity assays. Am J Physiol Regul Integr Comp Physiol. 2011;301:R1293–1299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Chappell MC. Biochemical evaluation of the renin-angiotensin system: The good, bad, and absolute? Am J Physiol Heart Circ Physiol. 2016;310:H137–152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Batlle D, Wysocki J, Soler MJ, Ranganath K. Angiotensin-converting enzyme 2: Enhancing the degradation of angiotensin ii as a potential therapy for diabetic nephropathy. Kidney Int. 2012;81:520–528 [DOI] [PubMed] [Google Scholar]
- 196.Campbell DJ, Duncan AM, Kladis A, Harrap SB. Angiotensin peptides in spontaneously hypertensive and normotensive donryu rats. Hypertension. 1995;25:928–934 [DOI] [PubMed] [Google Scholar]
- 197.Nussberger J, Brunner DB, Waeber B, Brunner HR. True versus immunoreactive angiotensin ii in human plasma. Hypertension. 1985;7:I1–7 [DOI] [PubMed] [Google Scholar]
- 198.Nussberger J, Brunner DB, Nyfeler JA, Linder L, Brunner HR. Measurement of immunoreactive angiotensin-(1–7) heptapeptide in human blood. Clin Chem. 2001;47:726–729 [PubMed] [Google Scholar]
- 199.Brosnihan KB, Chappell MC. Measurement of angiotensin peptides: Hplc-ria. Methods Mol Biol. 2017;1527:81–99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Sparks MA, Crowley SD, Gurley SB, Mirotsou M, Coffman TM. Classical renin-angiotensin system in kidney physiology. Compr Physiol. 2014;4:1201–1228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Burson JM, Aguilera G, Gross KW, Sigmund CD. Differential expression of angiotensin receptor 1a and 1b in mouse. Am J Physiol. 1994;267:E260–267 [DOI] [PubMed] [Google Scholar]
- 202.Ito M, Oliverio MI, Mannon PJ, Best CF, Maeda N, Smithies O, Coffman TM. Regulation of blood pressure by the type 1a angiotensin ii receptor gene. Proc Natl Acad Sci U S A. 1995;92:3521–3525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Oliverio MI, Kim HS, Ito M, Le T, Audoly L, Best CF, Hiller S, Kluckman K, Maeda N, Smithies O, Coffman TM. Reduced growth, abnormal kidney structure, and type 2 (at2) angiotensin receptor-mediated blood pressure regulation in mice lacking both at1a and at1b receptors for angiotensin ii. Proc Natl Acad Sci U S A. 1998;95:15496–15501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Ichiki T, Labosky PA, Shiota C, Okuyama S, Imagawa Y, Fogo A, Niimura F, Ichikawa I, Hogan BL, Inagami T. Effects on blood pressure and exploratory behaviour of mice lacking angiotensin ii type-2 receptor. Nature. 1995;377:748–750 [DOI] [PubMed] [Google Scholar]
- 205.Carey RM. At2 receptors: Potential therapeutic targets for hypertension. Am J Hypertens. 2017;30:339–347 [DOI] [PubMed] [Google Scholar]
- 206.Dell’Italia LJ, Meng QC, Balcells E, Wei CC, Palmer R, Hageman GR, Durand J, Hankes GH, Oparil S. Compartmentalization of angiotensin ii generation in the dog heart. Evidence for independent mechanisms in intravascular and interstitial spaces. J Clin Invest. 1997;100:253–258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Wei CC, Tian B, Perry G, Meng QC, Chen YF, Oparil S, Dell’Italia LJ. Differential ang ii generation in plasma and tissue of mice with decreased expression of the ace gene. Am J Physiol Heart Circ Physiol. 2002;282:H2254–2258 [DOI] [PubMed] [Google Scholar]
- 208.Butts B, Ahmed MI, Bajaj NS, Cox Powell P, Pat B, Litovsky S, Gupta H, Lloyd SG, Denney TS, Zhang X, Aban I, Sadayappan S, McNamara JW, Watson MJ, Ferrario CM, Collawn JF, Lewis C, Davies JE, Dell’Italia LJ. Reduced left atrial emptying fraction and chymase activation in pathophysiology of primary mitral regurgitation. JACC Basic Transl Sci. 2020;5:109–122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Eggena P, Zhu JH, Clegg K, Barrett JD. Nuclear angiotensin receptors induce transcription of renin and angiotensinogen mrna. Hypertension. 1993;22:496–501 [DOI] [PubMed] [Google Scholar]
- 210.Gwathmey TM, Shaltout HA, Pendergrass KD, Pirro NT, Figueroa JP, Rose JC, Diz DI, Chappell MC. Nuclear angiotensin ii type 2 (at2) receptors are functionally linked to nitric oxide production. Am J Physiol Renal Physiol. 2009;296:F1484–1493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Re RN, Vizard DL, Brown J, Bryan SE. Angiotensin ii receptors in chromatin fragments generated by micrococcal nuclease. Biochem Biophys Res Commun. 1984;119:220–227 [DOI] [PubMed] [Google Scholar]
- 212.Tang SS, Rogg H, Schumacher R, Dzau VJ. Characterization of nuclear angiotensin-ii-binding sites in rat liver and comparison with plasma membrane receptors. Endocrinology. 1992;131:374–380 [DOI] [PubMed] [Google Scholar]
- 213.Abadir PM, Foster DB, Crow M, Cooke CA, Rucker JJ, Jain A, Smith BJ, Burks TN, Cohn RD, Fedarko NS, Carey RM, O’Rourke B, Walston JD. Identification and characterization of a functional mitochondrial angiotensin system. Proc Natl Acad Sci U S A. 2011;108:14849–14854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Valenzuela R, Costa-Besada MA, Iglesias-Gonzalez J, Perez-Costas E, Villar-Cheda B, Garrido-Gil P, Melendez-Ferro M, Soto-Otero R, Lanciego JL, Henrion D, Franco R, Labandeira-Garcia JL. Mitochondrial angiotensin receptors in dopaminergic neurons. Role in cell protection and aging-related vulnerability to neurodegeneration. Cell Death Dis. 2016;7:e2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Zhuo JL, Li XC, Garvin JL, Navar LG, Carretero OA. Intracellular ang ii induces cytosolic ca2+ mobilization by stimulating intracellular at1 receptors in proximal tubule cells. Am J Physiol Renal Physiol. 2006;290:F1382–1390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Cerniello FM, Carretero OA, Longo Carbajosa NA, Cerrato BD, Santos RA, Grecco HE, Gironacci MM. Mas1 receptor trafficking involves erk1/2 activation through a beta-arrestin2-dependent pathway. Hypertension. 2017;70:982–989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Gironacci MM, Adamo HP, Corradi G, Santos RA, Ortiz P, Carretero OA. Angiotensin (1–7) induces mas receptor internalization. Hypertension. 2011;58:176–181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Herrera M, Sparks MA, Alfonso-Pecchio AR, Harrison-Bernard LM, Coffman TM. Lack of specificity of commercial antibodies leads to misidentification of angiotensin type 1 receptor protein. Hypertension. 2013;61:253–258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Herrera M, Coffman TM. The kidney and hypertension: Novel insights from transgenic models. Curr Opin Nephrol Hypertens. 2012;21:171–178 [DOI] [PubMed] [Google Scholar]
- 220.Yamamoto K, Ohishi M, Katsuya T, Ito N, Ikushima M, Kaibe M, Tatara Y, Shiota A, Sugano S, Takeda S, Rakugi H, Ogihara T. Deletion of angiotensin-converting enzyme 2 accelerates pressure overload-induced cardiac dysfunction by increasing local angiotensin ii. Hypertension. 2006;47:718–726 [DOI] [PubMed] [Google Scholar]
- 221.Imai Y, Kuba K, Rao S, Huan Y, Guo F, Guan B, Yang P, Sarao R, Wada T, Leong-Poi H, Crackower MA, Fukamizu A, Hui CC, Hein L, Uhlig S, Slutsky AS, Jiang C, Penninger JM. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436:112–116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.McCray PB Jr., Pewe L, Wohlford-Lenane C, Hickey M, Manzel L, Shi L, Netland J, Jia HP, Halabi C, Sigmund CD, Meyerholz DK, Kirby P, Look DC, Perlman S. Lethal infection of k18-hace2 mice infected with severe acute respiratory syndrome coronavirus. J Virol. 2007;81:813–821 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Yang XH, Deng W, Tong Z, Liu YX, Zhang LF, Zhu H, Gao H, Huang L, Liu YL, Ma CM, Xu YF, Ding MX, Deng HK, Qin C. Mice transgenic for human angiotensin-converting enzyme 2 provide a model for sars coronavirus infection. Comp Med. 2007;57:450–459 [PubMed] [Google Scholar]
- 224.Wan Y, Shang J, Graham R, Baric RS, Li F. Receptor recognition by the novel coronavirus from wuhan: An analysis based on decade-long structural studies of sars coronavirus. J Virol. 2020;94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Bao L, Deng W, Huang B, Gao H, Liu J, Ren L, Wei Q, Yu P, Xu Y, Qi F, Qu Y, Li F, Lv Q, Wang W, Xue J, Gong S, Liu M, Wang G, Wang S, Song Z, Zhao L, Liu P, Zhao L, Ye F, Wang H, Zhou W, Zhu N, Zhen W, Yu H, Zhang X, Guo L, Chen L, Wang C, Wang Y, Wang X, Xiao Y, Sun Q, Liu H, Zhu F, Ma C, Yan L, Yang M, Han J, Xu W, Tan W, Peng X, Jin Q, Wu G, Qin C. The pathogenicity of sars-cov-2 in hace2 transgenic mice. Nature. 2020 [DOI] [PubMed] [Google Scholar]
- 227.Hassan AO, Case JB, Winkler ES, Thackray LB, Kafai NM, Bailey AL, McCune BT, Fox JM, Chen RE, Alsoussi WB, Turner JS, Schmitz AJ, Lei T, Shrihari S, Keeler SP, Fremont DH, Greco S, McCray PB Jr., Perlman S, Holtzman MJ, Ellebedy AH, Diamond MS. A sars-cov-2 infection model in mice demonstrates protection by neutralizing antibodies. Cell. 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Greenland S, Robins JM. Identifiability, exchangeability, and epidemiological confounding. Int J Epidemiol. 1986;15:413–419 [DOI] [PubMed] [Google Scholar]
- 229.Hernán MA, Hernández-Díaz S, Robins JM. A structural approach to selection bias. Epidemiology. 2004;15:615–625 [DOI] [PubMed] [Google Scholar]
- 230.Shahar E Causal diagrams for encoding and evaluation of information bias. J Eval Clin Pract. 2009;15:436–440 [DOI] [PubMed] [Google Scholar]
- 231.Wolkewitz M, Allignol A, Harbarth S, de Angelis G, Schumacher M, Beyersmann J. Time-dependent study entries and exposures in cohort studies can easily be sources of different and avoidable types of bias. J Clin Epidemiol. 2012;65:1171–1180 [DOI] [PubMed] [Google Scholar]
- 232.Cole SR, Hernan MA. Constructing inverse probability weights for marginal structural models. Am J Epidemiol. 2008;168:656–664 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Knol MJ, VanderWeele TJ. Recommendations for presenting analyses of effect modification and interaction. Int J Epidemiol. 2012;41:514–520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Shrier I, Platt RW. Reducing bias through directed acyclic graphs. BMC Med Res Methodol. 2008;8:70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.VanderWeele TJ. Mediation analysis: A practitioner’s guide. Annu Rev Public Health. 2016;37:17–32 [DOI] [PubMed] [Google Scholar]
- 236.VanderWeele TJ, Ding P. Sensitivity analysis in observational research: Introducing the e-value. Ann Intern Med. 2017;167:268–274 [DOI] [PubMed] [Google Scholar]
- 237.Wu S, Ding Y, Wu F, Hou J, Mao P. Application of propensity-score matching in four leading medical journals. Epidemiology. 2015;26:e19–20 [DOI] [PubMed] [Google Scholar]
- 238.Perlman S, Masters P. Coronaviridae: The viruses and their replication.: Wolters Kluwer; 2020. [Google Scholar]


