INTRODUCTION
A few years ago, an IEEE Spectrum editorial identified 8 “cyborg” technologies paving the way for modern medicine to go bionic.1 These range from well-known implanted electrical devices for stimulating the nervous system to implanted systems to restore senses, deliver drugs, or replace entire organs.
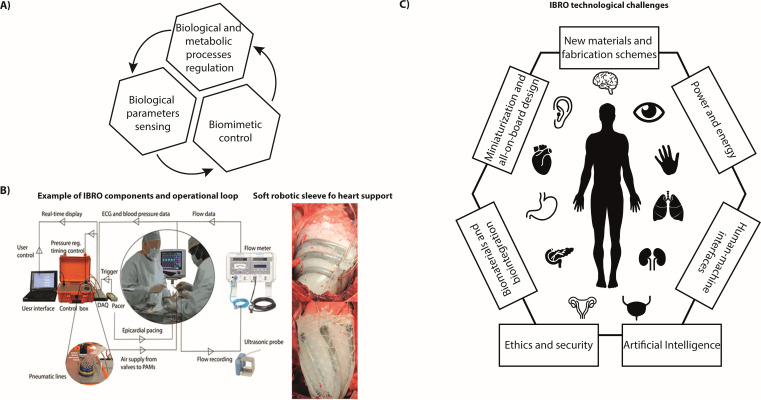
The concept of bionic organs has been linked to science fiction and to human augmentation since a long time. At present, remarkable advancements in powering, sensing and actuation technologies, microfabrication techniques, and communication protocols make credible the translation of this concept from science fiction to clinically viable fully implantable devices. We herein define this new class of machines implantable biorobotic organs (IBROs). By definition, an IBRO should endow awareness, action, and cognition capabilities to favor proper integration with the human body by sensing in-body signals and acting accordingly, thus regulating biological and metabolic processes, in a closed loop fashion as natural organs do [Fig. 1(a)].
FIG. 1.
(a) Schematic block diagram depicting the main building blocks of an IBRO. (b) Example of IBRO for heart support operational workflow and implanted prototype (Reprinted with permission from Roche et al., Sci. Transl. Med. 9(373), eaaf3925 (2017). Copyright 2017 AAAS). (c) Schematic representation of the potential organs to be restored or replaced by IBRO and of the technical and scientific challenges to be faced in the field.
While traditional programmable devices such as pacemakers and neurostimulators do not fall into the IBRO definition due to the inability to modulate their operation according to body needs, some interesting examples of IBRO have been reported in the state-of-the-art. These implanted devices operate well beyond simple electric or mechanical stimulators and are devised to restore lost organ functions such as blood pumping,2 micturition,3 hormone delivery,4 and tissue regeneration.5 A paradigmatic example is represented by a soft robotic sleeve devised to mechanically assist the failing heart. The sleeve presents a bioinspired design and includes multiple individually contracting soft actuators arranged in a layered helical and circumferential fashion, thus mimicking the orientation of mammalian heart muscle fibers. The device is implanted around the heart and actively compresses and twists to act as a cardiac ventricular assist device (VAD). The soft actuators are finely controlled to contract and relax in synchrony with the beating heart by relying on patient performance parameters such as the heart rate, pulmonary artery and ascending aortic pressure and flow rate,2 or native ventricular pressure6 [Fig. 1(b)]. The soft robotic sleeve can be customized to patient-specific needs and may have the potential to act as a bridge to transplantation for patients with heart failure.
Thanks to their ability to replace organ functions and biological/metabolic processes and to be fully implantable, IBRO can act as valid alternatives to transplantation and tissue engineering approaches,7–10 thus overcoming donor shortage issues and pursuing high throughput organ function replacement. Despite being promising, especially in terms of integration with the host, tissue engineering and regenerative medicine are not mature yet to target full organ replacement due to cell viability, diffusion and transport mechanism control, and difficulties in large-scale mechanical or secretory action performances.11 On the other hand, despite being fully artificial, IBRO can rely on consolidated mechatronics and material technologies, potentially enabling us, at the same time, to safely integrate with the host and to replace organ functions without recurring to pharmacological therapy or life-assistive machines.
From an engineering viewpoint, developing this new class of fully implantable robots poses new challenges with respect to both wearable and semi-implantable systems, such as prosthesis or most of the ear implants, and traditional robots.
TECHNICAL CHALLENGES FOR IMPLANTABLE BIOROBOTIC ORGANS
The need for more advanced “robotic” materials, smart powering strategies, friendly and biomimetic control interfaces, biocompatibility, all-on-board integration, miniaturization, safety, and security [Fig. 1(c)] should be mentioned among the first challenges to be faced in the field of IBRO.12
Developing artificial organ counterparts with a robotic approach, thus not using tissue engineering and regenerative medicine technologies, implies selecting appropriate materials and fabrication schemes enabling favorable interaction with the host on the short- and long-term, morphological and biological integration, as well as extended lifetime. Smart materials employment could also enable embedding structural intelligence (i.e., ability to sense and react to certain stimuli and/or to change configuration)13 and therapeutics delivery14 capability while pursuing a compact design. In this regard, the employment of novel fabrication techniques based on soft multimaterial printing15,16 combined with traditional mechatronic fabrication paradigms can be extremely advantageous.
Powering is the paradigmatic bottleneck of all autonomous machines and is particularly crucial when targeting fully implantable devices due to their inaccessibility for battery replacement. While natural tissues put in place metabolic processes to get the energy needed for their operation, IBRO calls for power supply sources that can be electric, chemical, or electromechanical in the case of solutions based on smart materials. Dedicated energy harvesting strategies have been proposed to power medical devices by exploiting in-body natural processes such as heart beating17,18 or the gut environmental conditions.19,20 However, when power densities above few μW/mm2 are needed and energy storage is required, battery integration is the gold standard. In most of the currently available implantable medical devices, lithium-ion batteries are integrated that can last up to ten years before running out. To overcome battery duration issues that nowadays prevent implantable devices from being lifelong, wireless energy transfer based on inductive electromagnetic coupling can represent a possibility to recharge implantable batteries or to provide the required energy, when body energy harvesting is not feasible.21,22 However, problems of distance and possible misalignment between the implant and the external source, together with attenuation issues across tissues, produce significant loss of efficiency that strongly impacts the employment of such powering strategies.
Another critical challenge to be taken into account when designing machines intended for “in-body” operation concerns their interface with the host. Natural organs are governed either by autonomous regulation mechanisms, as in the case of heart beating, metabolic processes, and hormone release, or by voluntary neural commands. Replicating such closed loop control strategies with the host possibly in the loop is a holy grail for IBRO and poses both technological and safety challenges. To make this paradigm true toward autonomous integrated systems, significant advancements in the field of neural interfaces, intentions decoding/feedback encoding algorithms,23 biological triggering sensing, and closed loop regulation protocols24 are needed. Moving to unsupervised autonomous control architectures could rise safety issue due to potentially life-threatening failures (e.g., pump stall in an artificial heart pump and excessive insulin dose delivery in an artificial pancreas) or device hacking.25,26
Additional challenges are imposed by the harsh working environment, namely, the human body. Operational requirements must be combined with a safe interaction with the host, both on the short- and long-terms. Choosing biocompatible materials or coatings, shaping the device to ensure mating with the implant site, and modulating mechanobiological responses by engineering device surfaces27 are crucial requirements to avoid unfavorable host reactions. Beyond pure biocompatibility, selecting the constitutive materials for IBRO should also respond to other needs, such as mechanical resistance (e.g., to impacts), mechanical and chemical stabilities (to corrosion in acidic environments, encrustations,28 and coagulation when in contact with specific biofluids), inertia to electromagnetic interferences, and sealing. Sealing is a particularly crucial issue when complex architectures are devised and where in-out interfaces are needed (e.g., for needle ejection, drug delivery, and wiring). In this sense, material safety and suitability has to be taken into account on both sides: if the human body can be damaged by the artificial guest, the reverse problem is even more complicated. Any leakages from the implant can be fatal for humans, but a non-ideal sealing can compromise the overall functionalities and the electronics, due to liquids or humors penetrating the implanted device, which is not easy to detect and repair.
Despite the large number of challenges to be faced in the design of IBRO, incredible advancements have been witnessed in the past few years, thus making this revolutionary concept closer to real application scenarios. In this direction, examples of implantable soft ventricular assistive devices,6 assisted tissue growth systems,29,30 implantable insulin,31,32 and drug delivery systems33 are particularly interesting and promising, as they smartly faced most of the aforementioned challenges and contributed to increasing the knowledge/awareness of the relevant integration and implantation steps.
CONCLUSIONS AND PERSPECTIVES
In this editorial, we reviewed the challenges characterizing the brand-new field of IBRO. Despite the large amount of effort required to make this kind of technology closer to the clinical practice, biorobotic organs appear as a valid alternative to organ transplantation, tissue engineering, and wearable or semi-implantable medical devices. Compared to the last category, in fact, implantable autonomous machines could replace their natural counterpart by letting the patient forget his/her own pathology and not needing to take care of his own life-supporting device, as a future scenario. This new class of artificial organs will be capable of sensing, controlling, and performing therapeutic or drug delivery actions, all of them with different levels of autonomy. Developing functional artificial organs and translating the existing technology from the bench to the clinical setting require multidisciplinary effort, ranging from materials science to microelectronics and medicine, with an eye to the power consumption which is limiting—at large—many attempts in the field. In order to increase both the level of autonomy and the natural interaction with the host, developing smart solutions to connect such implants to the patient nervous system toward voluntary implant control can represent a fundamental step. Smart recharging, drug refilling strategies, and novel technologies aimed at enhancing system miniaturization, safety, and efficiency go in this direction, thus paving the way for a new generation of fully implantable medical devices for organ function replacement.
References
- 1. Courtland R., “ Microbots on a fantastic voyage,” IEEE Spectrum 52(6), 70–74 (2015). 10.1109/MSPEC.2015.7115573 [DOI] [Google Scholar]
- 2. Roche E. T., Horvath M. A., Wamala I., Alazmani A., Song S.-E., Whyte W., Machaidze Z., Payne C. J., Weaver J. C., Fishbein G., Kuebler J., Vasilyev N. V., Mooney D. J., Pigula F. A., and Walsh C. J., “ Soft robotic sleeve supports heart function,” Sci. Transl. Med. 9(373), eaaf3925 (2017). 10.1126/scitranslmed.aaf3925 [DOI] [PubMed] [Google Scholar]
- 3. Mazzocchi T., Ricotti L., Pinzi N., and Menciassi A., “ Magnetically controlled endourethral artificial urinary sphincter,” Ann. Biomed. Eng. 45(5), 1181–1193 (2017). 10.1007/s10439-016-1784-2 [DOI] [PubMed] [Google Scholar]
- 4. Iacovacci V., Ricotti L., Dario P., and Menciassi A., “ Design and development of a mechatronic system for noninvasive refilling of implantable artificial pancreas,” IEEE/ASME Trans. Mechatronics 20(3), 1160–1169 (2015). 10.1109/TMECH.2014.2327196 [DOI] [Google Scholar]
- 5. Damian D. D., Price K., Arabagi S., Berra I., Machaidze Z., Manjila S., Shimada S., Fabozzo A., Arnal G., Van Story D., Goldsmith J. D., Agoston A. T., Kim C., Jennings R. W., Ngo P. D., Manfredi M., and Dupont P. E., “ In vivo tissue regeneration with robotic implants,” Sci. Rob. 3(14), eaaq0018 (2018). 10.1126/scirobotics.aaq0018 [DOI] [PubMed] [Google Scholar]
- 6. Payne C. J., Wamala I., Abah C., Thalhofer T., Saeed M., Bautista-Salinas D., Horvath M. A., Vasilyev N., Roche E. T., Pigula F. A., and Walsh C. J., “ An implantable extracardiac soft robotic device for the failing heart: mechanical coupling and synchronization,” Soft Rob. 4(3), 241–250 (2017). 10.1089/soro.2016.0076 [DOI] [PubMed] [Google Scholar]
- 7. Hettiaratchi M. H., Schudel A., Rouse T., García A. J., Thomas S. N., Guldberg R. E., and McDevitt T. C., “ A rapid method for determining protein diffusion through hydrogels for regenerative medicine applications,” APL Bioeng. 2, 026110 (2018). 10.1063/1.4999925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kamm R. D., Bashir R., Arora N., Dar R. D., Gillette M. U., Griffith L. G., Kemp M. L., Kinlaw K., Levin M., Martin A. C., McDevitt T. C., Nerem R. M., Powers M. J., Saif T. A., Sharpe J., Takayama S., Takeuchi S., Weiss R., Ye K., Yevick H. G., and Zaman M. H., “ Perspective: The promise of multi-cellular engineered living systems,” APL Bioeng. 2(4), 040901 (2018). 10.1063/1.5038337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Oksdath M., Perrin S. L., Bardy C., Hilder E. F., DeForest C. A., Arrua R. D., and Gomez G. A., “ Review: Synthetic scaffolds to control the biochemical, mechanical, and geometrical environment of stem cell-derived brain organoids,” APL Bioeng. 2, 041501 (2018). 10.1063/1.5045124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Mills R. J. and Hudson J. E., “ Bioengineering adult human heart tissue: How close are we?,” APL Bioeng. 3, 010901 (2019). 10.1063/1.5070106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Iacovacci V., Ricotti L., Menciassi A., and Dario P., “ The bioartificial pancreas (BAP): Biological, chemical and engineering challenges,” Biochem. Pharmacol. 100, 12–27 (2016). 10.1016/j.bcp.2015.08.107 [DOI] [PubMed] [Google Scholar]
- 12. Yang G.-Z., Bellingham J., Dupont P. E., Fischer P., Floridi L., Full R., Jacobstein N., Kumar V., McNutt M., Merrifield R., Nelson B. J., Scassellati B., Taddeo M., Taylor R., Veloso M., Wang Z. L., and Wood R., “ The grand challenges of science robotics,” Sci. Rob. 3(14), eaar7650 (2018). 10.1126/scirobotics.aar7650 [DOI] [PubMed] [Google Scholar]
- 13. McEvoy M. A. and Correll N., “ Materials that couple sensing, actuation, computation, and communication,” Science 347(6228), 1261689 (2015). 10.1126/science.1261689 [DOI] [PubMed] [Google Scholar]
- 14. Ricotti L., Cafarelli A., Iacovacci V., Vannozzi L., and Menciassi A., “ Advanced micro-nano-bio systems for future targeted therapies,” Curr. Nanosci. 11, 144–160 (2015). 10.2174/1573413710666141114221246 [DOI] [Google Scholar]
- 15. Bian L., “ Functional hydrogel bioink, a key challenge of 3D cellular bioprinting,” APL Bioeng. 4(3), 30401 (2020). 10.1063/5.0018548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Skylar-Scott M. A., Mueller J., Visser C. W., and Lewis J. A., “ Voxelated soft matter via multimaterial multinozzle 3D printing,” Nature 575(7782), 330–335 (2019). 10.1038/s41586-019-1736-8 [DOI] [PubMed] [Google Scholar]
- 17. Dagdeviren C., Yang B. D., Su Y., Tran P. L., Joe P., Anderson E., Xia J., Doraiswamy V., Dehdashti B., Feng X., Lu B., Poston R., Khalpey Z., Ghaffari R., Huang Y., Slepian M. J., and Rogers J. A., “ Conformal piezoelectric energy harvesting and storage from motions of the heart, lung, and diaphragm,” Proc. Natl. Acad. Sci. U. S. A. 111(5), 1927–1932 (2014). 10.1073/pnas.1317233111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ouyang H., Liu Z., Li N., Shi B., Zou Y., Xie F., Ma Y., Li Z., Li H., Zheng Q., Qu X., Fan Y., Wang Z. L., Zhang H., and Li Z., “ Symbiotic cardiac pacemaker,” Nat. Commun. 10(1), 1821 (2019). 10.1038/s41467-019-09851-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Nadeau P., El-Damak D., Glettig D., Kong Y. L., Mo S., Cleveland C., Booth L., Roxhed N., Langer R., Chandrakasan A. P., and Traverso G., “ Prolonged energy harvesting for ingestible devices,” Nat. Biomed. Eng. 1(3), 22 (2017). 10.1038/s41551-016-0022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Menciassi A., “ Bioelectronic devices: Gut-powered ingestible biosensors,” Nat. Biomed. Eng. 1(3), 50 (2017). 10.1038/s41551-017-0050 [DOI] [Google Scholar]
- 21. Campi T., Cruciani S., Maradei F., Montalto A., Musumeci F., and Feliziani M., “ Wireless powering of next-generation left ventricular assist devices (LVADs) without percutaneous cable driveline,” IEEE Trans. Microwave Theory Tech. 68(9), 3969–3977 (2020). 10.1109/TMTT.2020.2992462 [DOI] [Google Scholar]
- 22. Tang S. C., Lun T. L. T., Guo Z., Kwok K., and McDannold N. J., “ Intermediate range wireless power transfer with segmented coil transmitters for implantable heart pumps,” IEEE Trans. Power Electron. 32(5), 3844–3857 (2017). 10.1109/TPEL.2016.2584558 [DOI] [Google Scholar]
- 23. Valle G., Mazzoni A., Iberite F., D'Anna E., Strauss I., Granata G., Controzzi M., Clemente F., Rognini G., Cipriani C., Stieglitz T., Petrini F. M., Rossini P. M., and Micera S., “ Biomimetic intraneural sensory feedback enhances sensation naturalness, tactile sensitivity, and manual dexterity in a bidirectional prosthesis,” Neuron 100(1), 37–45 (2018). 10.1016/j.neuron.2018.08.033 [DOI] [PubMed] [Google Scholar]
- 24. Kovatchev B., “ Automated closed-loop control of diabetes: The artificial pancreas,” Bioelectron. Med. 4(1), 14 (2018). 10.1186/s42234-018-0015-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Sackner-Bernstein J., “ Design of hack-resistant diabetes devices and disclosure of their cyber safety,” J. Diabetes Sci. Technol. 11(2), 198–202 (2017). 10.1177/1932296816678264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hempel G., Janosek D. B., and Raziano D. B., “ Hacking humans: A case study and analysis of vulnerabilities in the advancing medical device landscape,” Cyber Secur. 3(4), 351–362 (2020). [Google Scholar]
- 27. Dolan E. B., Varela C. E., Mendez K., Whyte W., Levey R. E., Robinson S. T., Maye E., O'Dwyer J., Beatty R., Rothman A., Fan Y., Hochstein J., Rothenbucher S. E., Wylie R., Starr J. R., Monaghan M., Dockery P., Duffy G. P., and Roche E. T., “ An actuatable soft reservoir modulates host foreign body response,” Sci. Rob. 4(33), eaax7043 (2019). 10.1126/scirobotics.aax7043 [DOI] [PubMed] [Google Scholar]
- 28. Cardona A., Iacovacci V., Mazzocchi T., Menciassi A., and Ricotti L., “ Novel nanostructured coating on PDMS substrates featuring high resistance to urine,” ACS Appl. Bio Mater. 2(1), 255–265 (2019). 10.1021/acsabm.8b00586 [DOI] [PubMed] [Google Scholar]
- 29. Perez-Guagnelli E., Jones J., Tokel A. H., Herzig N., Jones B., Miyashita S., and Damian D. D., “ Characterization, simulation and control of a soft helical pneumatic implantable robot for tissue regeneration,” IEEE Trans. Med. Rob. Bionics 2(1), 94–103 (2020). 10.1109/TMRB.2020.2970308 [DOI] [Google Scholar]
- 30. Latorre M. and Humphrey J. D., “ Critical roles of time-scales in soft tissue growth and remodeling,” APL Bioeng. 2(2), 26108 (2018). 10.1063/1.5017842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Lee S. H., Lee Y. B., Kim B. H., Lee C., Cho Y. M., Kim S.-N., Park C. G., Cho Y.-C., and Choy Y. B., “ Implantable batteryless device for on-demand and pulsatile insulin administration,” Nat. Commun. 8(1), 15032 (2017). 10.1038/ncomms15032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ricotti L., Assaf T., Stefanini C., and Menciassi A., “ System for controlled administration of a substance from a human body-implanted infusion device,” Google patents (16 August 2016).
- 33. Duffy G. P., Robinson S. T., O'Connor R., Wylie R., Mauerhofer C., Bellavia G., Straino S., Cianfarani F., Mendez K., Beatty R., Levey R., O'Sullivan J., McDonough L., Kelly H., Roche E. T., and Dolan E. B., “ Implantable therapeutic reservoir systems for diverse clinical applications in large animal models,” Adv. Healthcare Mater. 9(11), 2000305 (2020). 10.1002/adhm.202000305 [DOI] [PubMed] [Google Scholar]