Abstract
Objective:
This study aims to replicate the phenotype of Ltbp1 knockout mice in zebrafish, and to address the function of LTBP1 in craniofacial development.
Methods:
Whole mount in situ hybridization (WISH) of ltbp1 was performed at critical periods of zebrafish craniofacial development to explore the spatial-temporal expression pattern. Furthermore, we generated morpholino based knockdown model of ltbp1 to study the craniofacial phenotype.
Results:
WISH of ltbp1 was mainly detected in the mandibular jaw region, brain trunk, and internal organs such as pancreas and gallbladder. And ltbp1 colocalized with both sox9a and ckma in mandibular region. Morpholino based knockdown of ltbp1 results in severe jaw malformation. Alcian blue staining revealed severe deformity of Meckel's cartilage along with the absence of ceratobranchial. Three-dimension measurements of ltbp1 morphants jaws showed decrease in both mandible length and width and increase in open mouth distance. Expression of cartilage marker sox9a and muscle marker ckma was decreased in ltbp1 morphants.
Conclusions:
Our experiments found that ltbp1 was expressed in zebrafish mandibular jaw cartilages and the surrounding muscles. The ltbp1 knockdown zebrafish exhibited phenotypes consistent with Ltbp1 knockout mice. And loss of ltbp1 function lead to significant mandibular jaw defects and affect both jaw cartilages and surrounding muscles.
Keywords: LTBP1, Craniofacial anomalies, Developmental biology, Zebrafish
Introduction
The latent transforming growth factor-β binding protein-1 (LTBP-1) is an extracellular matrix protein that is structurally similar to the fibrillin family. There are several human and mouse Ltbp1 splice variants including a short (Ltbp-1S) and long (Ltbp-1L) form, which may arise from the two separated promoters, alternative RNA splicing, and differential proteolytic processing.1 LTBP1 is widely expressed among species in variable amounts in different tissues, including heart, lung, liver, placenta, skeletal muscle, and bone.2,3 Current researches suggest that LTBP1 is a multifunctional protein that bind latent transforming growth factor-β (TGF-β) and regulate its function in bone and other connective tissues.4–10 LTBP1 is reported to ease the secretion of latent TGF-β and modulate the activation of latent TGF-β.11–16 It is considered as a stable component of extracellular fibrillar structure that play a critical role in the storage and release of TGF-β as a large latent TGF-β complex.17–19 In addition to regulating the function of TGF-β, LTBP1 function as a structural component of connective tissue microfibrils.20 Studies focusing on its function as matrix proteins find that LTBP1 colocalize with fibrillin 1 in microfibrillar structures not only in the extracellular matrix of primary osteoblasts but also at the surface of newly forming osteoid and bone, suggesting that LTBP1 play an important role in the formation of bone and connective tissue.8
The important function of LTBP1 has been investigated by a series of studies. Targeted deletion of exon 1 or 2 led to Ltbp-1L-null mice dying shortly after birth from defects in heart organogenesis.21 These severe cardiac deformities include persistent truncus arteriosus and interrupted aortic arch, which are associated with imperfect function of cardiac neural crest cells. Similarly, targeted deletion of exon 8 to generate Ltbp1L knockout mice died perinatally, these mice also present heart defects including persistent truncus arteriosus and interrupted aortic arch which are similar to Ltbp1L-null mice.22 These studies indicate the crucial role of LTBP1 in the development of the cardiovascular system. Another Ltbp1 knockout mice is manipulated by targeted deletion of exon 5, which is the first exon shared by the Ltbp1L and Ltbp1S.23 These total knockout mice are viable and fertile, they exhibit craniofacial abnormalities consisting of more compact head structure with shorten maxilla and mandible. Furthermore, the total Ltbp1 knockout mice have shortened long bones. Moreover, LTBP1 knockout mice are less prone to hepatic fibrogenesis.23 These phenomena suggest that loss of LTBP1 function lead to potential changes of the biological activity of TGF-β in fibrogenesis action. Researchers also used the deletion of Ltbp1L exon 5 knockout model to address the function of Ltbp1 in female fertility.24 Since Ltbp1 knockout female mice showed impaired reproduction with subfertility and ovarian cyst formation, the interruption of TGF-β function which would lead to defective follicular wound healing was suggested as the potential cause.
It is known that LTBP1 is critically required for the cardiovascular system organogenesis, bone and connective tissue formation, female reproduction, and craniofacial development.21–24 However, the discrepancy of phenotypes in Ltbp1 gene-manipulated mice is difficult to explain. Moreover, the function of LTBP1 in craniofacial development remains unclear. Since the gene function, fundamental signaling pathways and cellular events in craniofacial morphogenesis have proven to be highly conserved among vertebrates from zebrafish and mouse to man,25,26 we chose zebrafish to observe the phenotype in another model and to address the function of LTBP1 in craniofacial aspect. We performed whole mount in situ hybridization (WISH) of ltbp1 in critical periods of zebrafish craniofacial development to explore the spatial-temporal expression pattern. Furthermore, we generated morpholino based knockdown model of ltbp1 to study the craniofacial phenotype.
This study aims to replicate the phenotype of Ltbp1 knockout mice in zebrafish, and to address the function of LTBP1 in craniofacial development.
Methodology
Zebrafish husbandry
Wild-type zebrafish (Tuebingen) were maintained with standard techniques.27 All experiments were performed in compliance with the Animal Ethics Committee.
Zebrafish embryos were collected from natural mating and kept at 28.5°C. About 300 male/female zebrafish embryos were used for detection of ltbp1 expression pattern at five experimental periods: 1 cell, sphere, shield, 1 day postfertilization (DPF), and 3 DPF stages. About 1800 male/female zebrafish embryos were used for ltbp1 knock-down phenotype study. The animals were divided into three experimental groups: 1. control group (wild-type zebrafish injected with control morpholino); 2. ltbp1 knock-down group (wild-type zebrafish injected with ltbp1 morpholino); 3. Rescue group (wild-type zebrafish co-injected with ltbp1 morpholino and synthesized ltbp1 mRNA). Each group was evaluated at two experimental periods: 3 DPF and 5 DPF stages.
Microinjection of morpholino antisense oligo and mRNA
Morpholino antisense oligos (MO) were designed to block translation and synthesis by Gene Tools, Inc. The ltbp1-MO was targeted to the 5′UTR of the ltbp1 mRNA, and the ltbp1-MO was bound to the 5′UTR (including ATG) of the ltbp1 mRNA. A standard control-MO was used as negative control (Table 1).28 Full-length cDNA was used and amplified to test knockdown specificity of the MO.26,28 Mutations in ltbp1-mRNA were introduced within the first 12 bases of open reading frame (ORF) (ATGCTgGTcTGt) without changing the sequences of amino acid so that mutated ltbp1 mRNA could mimic ltbp1 mRNA and it would not anneal to ltbp1-MO. The MOs and synthesized mRNA were injected into the yolk of embryos at the 1-cell stage. Primers used are described in Table 2. Zebrafish embryos at the 1-cell stage were injected with 1 nL of ltbp1-MO at a concentration ranging from 0.1 to 1 mM and a control-MO was used as a negative control. The final dosages of ltbp1-MO are 0.25 pmol, 0.5 pmol, and 0.75 pmol. Zebrafish embryos at the 1-cell stage were co-injected with ltbp1-mRNA(100 pg/embryo) and ltbp1-MO(0.75 pmol/embryo) for the rescue experiments.
Table 1. Morpholino antisense oligos.
| Morpholino | Sequence |
|---|---|
| ltbp1-MO | 5′-AAACAATGATGTCCCACACGAGCAT-3 |
| control-MO | 5′-CCTCTTACCTCAGTTACAATTTATA- 3 |
Table 2. Primers for the plasmid clone.
| Construct | Primer | Sequence 5′-3′ |
|---|---|---|
| ltbp1-mRNA | ltbp1-mm-ATG-EcoRI | GGAATTCcaccATGCTGGTCTGTGACATCATTGTT |
| ltbp1-R-XhoI | CCGCTCGAGTCACTCTGTGCCCGTACTGGT |
Whole mount in situ hybridization
Probes for ltbp1, sox9a, and ckma were cloned by polymerase chain reaction with primers presented in Table 3, whole mount in situ hybridization (WISH) was performed as described.29 The stained embryos were photographed with a Nikon SMZ1500 stereomicroscope.
Table 3. Primers for the probes.
| probe | Primer | Sequence 5′-3′ |
|---|---|---|
| ltbp1 | PF-ltbp1-11 | ATGCCAGCTCTTTGGAAGTGA |
| PR-ltbp1-743-T7 | taatacgactcactatagggGTTGATATCCACGCAGGCCA | |
| sox9a | PF-sox9a-167 | CTCCTCGACCCCTACCTGAA |
| PR-sox9a-761-T7 | taatacgactcactatagggAGATGTGGGTCTGTTCGCTG | |
| ckma | PF-ckma-404 | GTCACGGTGGATACAAGGCA |
| PR-ckma-1246-T7 | taatacgactcactatagggTCATGCTGTCGATGGACTCG |
Alcian blue staining
Embryos were fixed at 5 DPF, and cartilages were visualized by staining with alcian blue (SigmaAldrich; as described by Kimmel, et al.30 (1998) without using the 1.67% trypsin to digest the tissue.
Statistical analysis
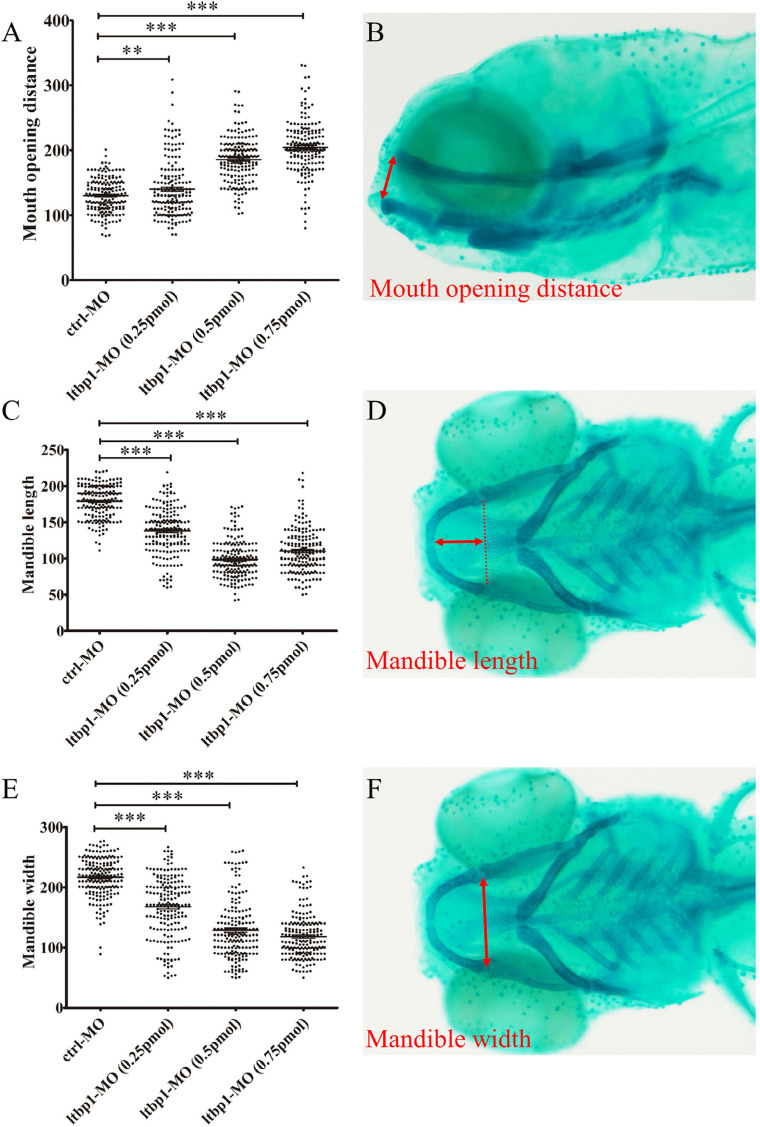
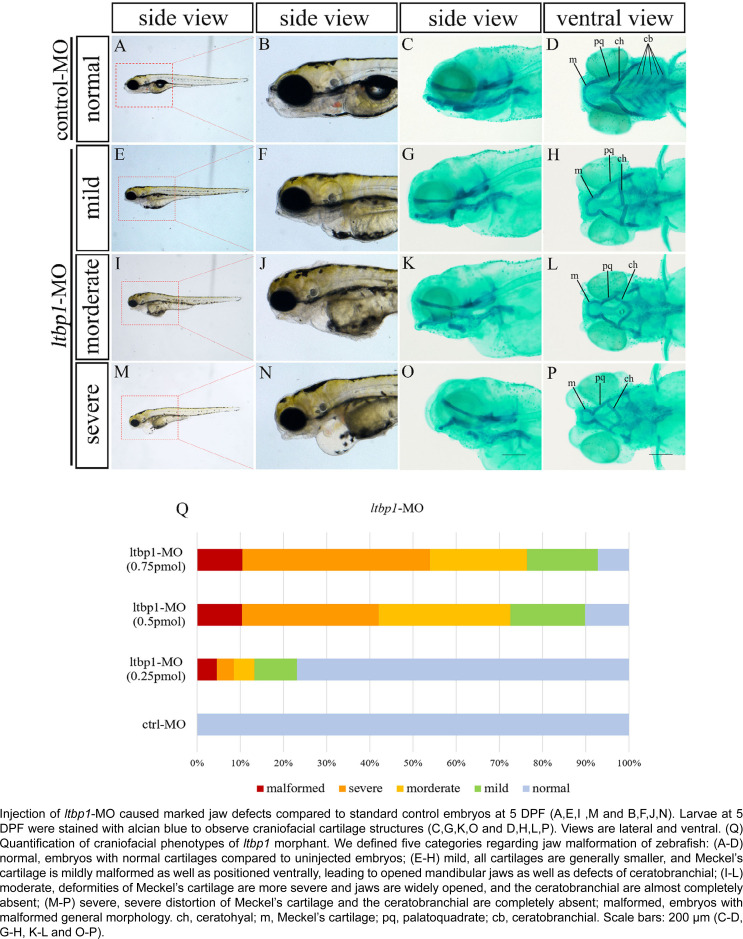
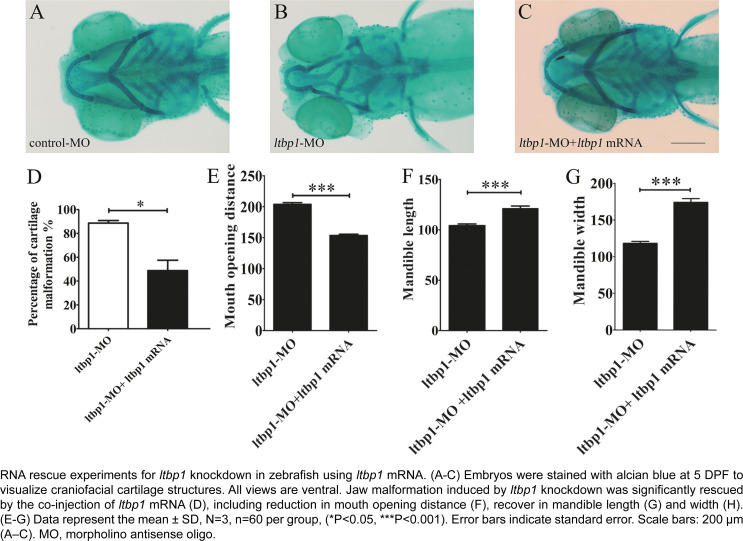
Statistical analysis was performed using GraphPad Prism 5 (GraphPad Software Inc., San Diego, CA, USA). At 5 DPF, embryos were categorized according to zebrafish jaw malformation: normal, embryos with normal cartilages compared to uninjected embryos; mild, all cartilages are generally smaller and Meckel's cartilage is mildly malformed as well as positioned ventrally, leading to mandibular jaws opened along with the defects of ceratobranchial; moderate, deformities of Meckel's cartilage are severe, jaws are widely opened and the ceratobranchial is almost completely absent; severe, increased severity of the Meckel's cartilage distortion and the ceratobranchial is completely absent; malformed, embryos with malformed general morphology. The malformed category was excluded when the percentage of cartilage was estimated. The percentage of cartilage malformation was estimated by considering the total number of mild, moderate, and severe category, and then dividing it by the total number of normal, mild, moderate, and severe category. The mean grey value of WISH staining on the mandibular cartilage (outlined by red dot line) and mandibular surrounding muscle (outlined by red dot line) was measured via ImageJ software. Measurements of lower jaw regions were conducted, including open mouth distance (Figure 3B), mandible length (Figure 3D), and mandible width (Figure 3F), the red double arrow represents the corresponding length. Each specific length was obtained by comparing the length of red double arrow with a scale bar that corresponds with the magnification of the alcian blue images.
Figure 3. Three dimensions (3D) measurements of mandibular jaw malformation.
Data are expressed as mean ± standard deviation (SD). Comparison of different groups was performed by unpaired t-test to estimate data measurements. P-value less than 0.05 (P<0.05) was considered statistically significant.
Results
ltbp1 expresses at mandibular arch skeleton
Since previous studies indicated that LTBP1 is a crucial gene in the early embryo development and it is widely expressed in several systems, we suspected that LTBP1 was expressed in craniofacial tissues. However, the expression pattern of LTBP1 in craniofacial tissues remains unclear. Therefore, we decided to explore the expression pattern of LTBP1 zebrafish ortholog in craniofacial tissues.
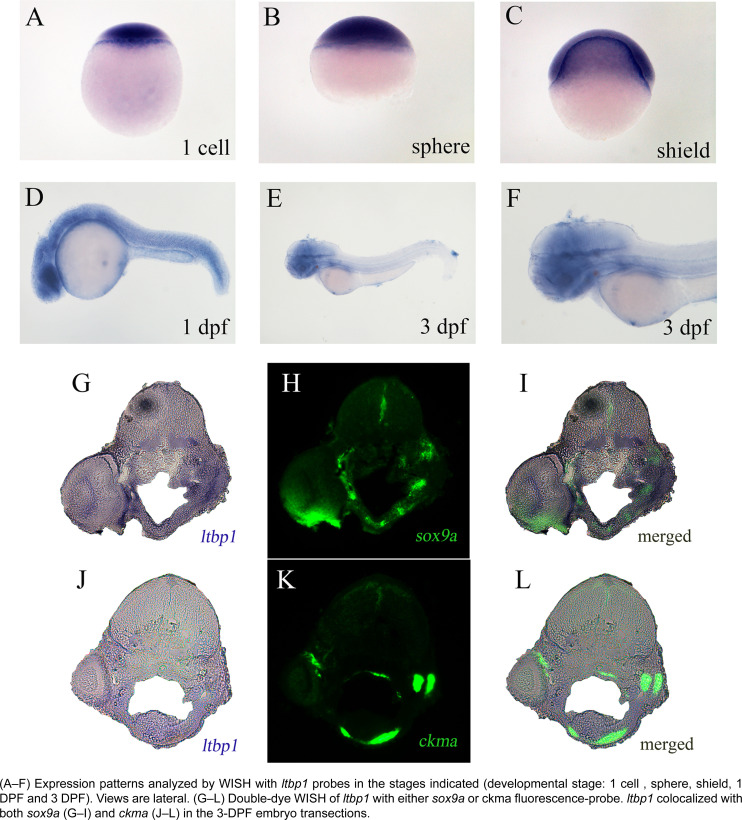
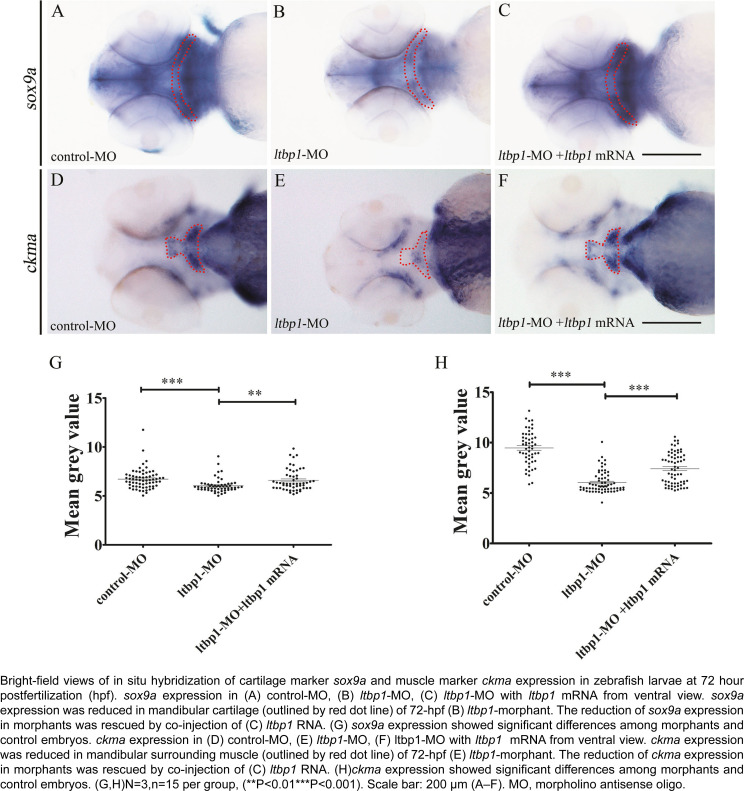
According to WISH of ltbp1 on zebrafish embryos, maternal transcripts were detected during the early developmental stage from 1 cell (Figure 1A) to shield (Figure 1C). At pharyngula period (1 DPF), ltbp1 expressed at pharyngeal arches, brain, the most part of trunk, and the origin of internal organs (Figure 1D). In the developmental stage of protruding mouth (3 DPF), ltbp1 was mainly detected in the mandibular jaw region and brain, but reduced expression also existed in the trunk and internal organs such as pancreas and gallbladder (Figure 1E, F). We performed 2-color WISH of ltbp1 with cartilage marker sox9a and muscle marker ckma, respectively, to further explore the expression pattern of ltbp1 in the mandibular jaw region. sox9a is a marker of cranial neural crest that differs from cartilage, and sox9a participates in both determination of crest-derived chondrogenic lineages and morphogenesis of cartilage (2-3 DPF)31. ckma is a terminal differentiation marker for skeletal muscle,32 and ckma was expressed in muscles of the trunk, pectoral fin, head, and heart.33 According to the observation of the tissue section of zebrafish jaw, ltbp1 colocalized with both sox9a (Figure 1G-N) and ckma (Figure 1J-L). The results indicated that ltbp1 was expressed in craniofacial tissues, and it was mainly concentrated in the zebrafish jaw cartilages and its surrounding muscles.
Figure 1. Expression patterns of ltbp1.
Loss of ltbp1 function Results in Severe Jaw Malformation
Based on the fact that ltbp1 is expressed in the zebrafish mandibular jaw region, we speculated that ltbp1 play a role in the formation of mandibular jaw. The loss of ltbp1 function was mediated by morpholino knockdown to assess the function of ltbp1 during craniofacial development and mandibular jaw formation. We designed translation-blocking morpholino antisense oligonucleotides (MO) for ltbp1 targeting regions in the 5’-UTR of the mRNA. MO with concentration ranging between 0.1 and 1 mM was injected 1 nL into zebrafish embryos in 1-cell stage, and a control-MO was used as a negative control. Embryos injected with control-MO were indistinguishable from uninjected embryos. Embryos injected with ltbp1-MO exhibited several defects, including severe jaw malformation, pericardial edema, generally shortened body length and microphthalmia. At 5 DPF, ltbp1 morphants presented significantly underdeveloped jaw regions and the mandibular jaws were widely opened (Figure 2E-F, I-J and M-N). We performed alcian blue staining to visualize the jaw cartilage skeleton. The staining revealed severe deformity of Meckel's cartilage along with the absence of ceratobranchial (Figure 2C-D, G-H, K-L and O-P). Further measurements of lower jaw regions were conducted, including open mouth distance (Figure 3B), mandible length (Figure 3D) and mandible width (Figure 3F), in order to assess the jaw defects. The measurement of this three dimensions showed that ltbp1 morphants decreased in both mandible length and width and it increased in open mouth distance compared to the control-MO group (Figure 3A,C,E). Interestingly, as ltbp1 MO dosages increase, both penetrance and severity of the jaw defects increases (Figure 2Q).
Figure 2. Depletion of ltbp1 causes severe jaw malformation in zebrafish.
Rescue experiments with full-length ltbp1 mRNA were carried out to confirm the specificity of ltbp1 knockdown phenotype. Co-injection of nonhomologous ltbp1 mRNA with ltbp1 MO significantly rescued the jaw defects (Figure 4A-D). The rescued ltbp1-morphants presented increased jaw length and width, whereas their open mouth distance (Figure 4E-H) decreased, and also decreased the rescue of ceratobranchial (Figure 4C). As most phenotypes were rescued when the mRNA encoding for ltbp1 were co-injected in the ltbp1 MO, the off-targeting effect of ltbp1 MO was ruled out, indicating the specificity of ltbp1 knockdown phenotype.
Figure 4. Specificity of ltbp1 knockdown phenotype.
Loss of ltbp1 function Lead to Abnormal Cartilage and Muscle during Jaw Development
Since ltbp1 is expressed in both jaw cartilages and jaw surrounding muscles, and the loss of ltbp1 function affected jaw development, we examined the two tissue in ltbp1-morphants during jaw formation. Because 3 DPF is the stage of protruding-mouth stage, when the developing cartilages of the pharyngeal skeleton such as Meckel's cartilage, has grown dramatically, we chose 3 DPF as developmental stage to examine the effects of ltbp1 knockdown. We conducted WISH of sox9a and ckma respectively on control-MO embryos, ltbp1-morphants, as well as ltbp1 rescued morphants at 3 DPF. The expression of sox9a was detected in mesenchymal condensation of pharyngeal arches and jaw cartilages in control and rescued morphants, while significantly decreased in ltbp1-morphants (Figure 5A-C and G). Similarly, expression of ckma was shown in mandibular region of craniofacial muscle in control and rescued morphants, but severely reduced in ltbp1-morphants (Figure 5D-F and H). The WISH of sox9a and ckma indicated that ltbp1 function loss affected jaw development in both jaw cartilage and muscle aspects.
Figure 5. Loss of ltbp1 function affects formation of both craniofacial cartilage and muscle.
Discussion
LTBP1 is an important extracellular matrix protein that regulates the bioactivity of TGF-β and also exists as microfibrillar structure of bone and connective tissue. According to recent researches, the expression of ltbp1 is widely distributed in different tissues.2,3 Gene disruption experiments have shown that LTBP1 play roles in several fields, including the development and formation of cardiovascular system, craniofacial system, reproduction system as well as functions in both bone and connective tissue.21–24 In this study, we explored the expression pattern of ltbp1 using zebrafish. The results of ltbp1 WISH showed detectable maternal transcripts from 1 cell to shield stage, which indicated that ltbp1 is necessary during the early embryo development. In the pharyngula period (1 DPF), ltbp1 was extensively expressed in the pharyngeal arches, brain, most part of trunk and the origin of some internal organs. In the developmental stage of protruding mouth (3 DPF), ltbp1 expression was mainly in the mandibular jaw region and brain, while reduced expression was also present in the trunk and internal organs such as pancreas and gallbladder. The ltbp1 WISH results are according with previously reported widespread tissue expression. Since ltbp1 is strongly expressed in the mandibular jaw region at 3 DPF, and the zebrafish jaw region mainly consists of cartilages and muscles. We carried out 2-colour WISH of ltbp1 with cartilage marker sox9a and muscle marker ckma in order to specify the target tissue of ltbp1 expression. The results showed that ltbp1 colocalized with both sox9a and ckma, indicating that ltbp1 concentrates in the zebrafish jaw cartilages and its surrounding muscles.
Considering that the function of a gene is usually associated with its expression pattern, we hypothesize ltbp1 may have its function in the craniofacial development of zebrafish. According to previous studies, different strategies of Ltbp1 knockout mice presented several phenotypes, including heart defects, craniofacial deformities, and impaired reproduction.21–24 However, the discrepancy of the variable phenotypes in Ltbp1 gene-manipulated mice is complicated and ambiguous. To repeat the phenotype in another model, we performed morpholino based knockdown of ltbp1 using zebrafish.
Generally, ltbp1 morphants presented shortened body length, heart edema, and severe jaw malformation. At 5 DPF, ltbp1 morphants manifested significantly underdeveloped jaw regions and the mandibular jaw was widely opened. These phenotypes are consistent with the observation of Ltbp1 knockout mice which were built by different strategies.
In this study, we focused on jaw defects of ltbp1 morphants, since this craniofacial aspect is the most significant observed in this phenotype, remaining unclear. Alcian blue staining of 5 DPF ltbp1 morphants revealed severe deformity of Meckel's cartilage as well as the absence of ceratobranchial. We conducted three dimensional measurements of lower jaw regions to assess the jaw defects, and the ltbp1 morphants decreased both mandible length and width and their open mouth distance increased. Similar phenotype was previously observed in the Ltbp1-null mice reported by Drews, et al.23 (2008), which showed modified facial profile with shortened snouts and shortened maxilla and mandible. We also found that both penetrance and severity of ltbp1-morphants defects of jaw increases as the dosages of ltbp1 MO increases. Meanwhile, most phenotypes were rescued by co-injection of nonhomologous ltbp1 mRNA with ltbp1 MO, therefore the off-targeting effect of ltbp1 MO was ruled out, indicating the specificity of ltbp1 morphant phenotype. Since ltbp1 is expressed in both jaw cartilages and surrounding muscles, we speculated that the jaw deformities could derive from the defects of either or both the two tissues. In order to address possible major aspect, we checked both cartilage and muscle markers in ltbp1-morphants. Both expression of sox9a and ckma was significantly decreased in ltbp1-morphants compared to control-MO and ltbp1 rescued morphants, indicating that knockdown of ltbp1 affected jaw development in both jaw cartilage and muscle aspects.
Considering that some previous studies have found that latent TGF-β is present in the matrix of chondrocytes and that LTBP1 is responsible for storing TGF-β complex in the matrix.6,7 TGF-β1 and their receptors have been found to be related to zebrafish craniofacial bone and cartilage development.34 The cartilage development may be affected by indirect regulating function of LTBP1 and TGF-β1. Besides, LTBP1 may directly affect chondrocytes as an important extracellular matrix protein. Furthermore, LTBP1 may participate in the muscle formation as microfibrillar structures of connective tissue8. Future studies will investigate the biological activity of TGF-β and extracellular fibrillar structure whether they are compromised or not. Although the specific mechanisms require further research, our results have extended the understanding of the important role of LTBP1 especially in craniofacial development, easing future investigations on the mechanisms of LTBP1 gene function.
Conclusion
Our experiments found that ltbp1 is expressed in zebrafish mandibular jaw cartilages and the surrounding muscles in addition to the previously reported tissues. Also, the ltbp1 knockdown zebrafish presented phenotypes consistent with Ltbp1 knockout mice. Lastly, loss of ltbp1 function lead to significant mandibular jaw defects and affect both jaw cartilages and surrounding muscles.
Acknowledgements
We are grateful to Miaomiao Jin, Fang Yuan, and Weilai Huang for helpful discussion and Ruikun Hu, Guanliang Huang for fish maintenance. And we are extremely grateful to Professor Cao Ying for constructive advice for the design of the experiment and the modification of the paper. The authors declare no potential conflicts of interest regarding the authorship and/or publication of this article.
Funding Statement
Funding for this project is provided by the National Natural Science Foundation of China (81371129,81670973,81970921) and Shanghai Sailing Program (20YF1453500).
Footnotes
Funding
Funding for this project is provided by the National Natural Science Foundation of China (81371129,81670973,81970921) and Shanghai Sailing Program (20YF1453500).
Reference
- 1.Koski C, Saharinen J, Keski-Oja J. Independent promoters regulate the expression of two amino terminally distinct forms of latent transforming growth factor-beta binding protein-1 (LTBP-1) in a cell type-specific manner. J Biol Chem. 1999;274(46):32619–32630. doi: 10.1074/jbc.274.46.32619. [DOI] [PubMed] [Google Scholar]; 1- Koski C, Saharinen J, Keski-Oja J. Independent promoters regulate the expression of two amino terminally distinct forms of latent transforming growth factor-beta binding protein-1 (LTBP-1) in a cell type-specific manner. J Biol Chem. 1999;274(46):32619-30. doi:10.1074/jbc.274.46.32619. [DOI] [PubMed]
- 2.Weiskirchen R, Moser M, Günther K, Weiskirchen S, Gressner AM. The murine latent transforming growth factor-beta binding protein (Ltbp-1) is alternatively spliced, and maps to a region syntenic to human chromosome 2p21-22. Gene. 2003;308:43–52. doi: 10.1016/s0378-1119(03)00464-5. [DOI] [PubMed] [Google Scholar]; 2- Weiskirchen R, Moser M, Günther K, Weiskirchen S, Gressner AM. The murine latent transforming growth factor-beta binding protein (Ltbp-1) is alternatively spliced, and maps to a region syntenic to human chromosome 2p21-22. Gene. 2003;308:43-52. doi:10.1016/s0378-1119(03)00464-5 [DOI] [PubMed]
- 3.Faraoni EY, Camilletti MA, Abeledo-Machado A, Laura DR, Ratner F, De Fino F, et al. Sex differences in the development of prolactinoma in mice overexpressing hCGβ: role of TGFβ1. J Endocrinol. 2017;232(3):535–546. doi: 10.1530/JOE-16-0371. [DOI] [PubMed] [Google Scholar]; 3- Faraoni EY, Camilletti MA, Abeledo-Machado A, Laura DR, Ratner F, De Fino F, et al. Sex differences in the development of prolactinoma in mice overexpressing hCGβ: role of TGFβ1. J Endocrinol. 2017;232(3):535-46. doi:10.1530/JOE-16-0371 [DOI] [PubMed]
- 4.Akhurst RJ, Lehnert SA, Faissner A, Duffie E. TGF beta in murine morphogenetic processes: the early embryo and cardiogenesis. Development. 1990;108(4):645–656. doi: 10.1242/dev.108.4.645. [DOI] [PubMed] [Google Scholar]; 4- Akhurst RJ, Lehnert SA, Faissner A, Duffie E. TGF beta in murine morphogenetic processes: the early embryo and cardiogenesis. Development. 1990;108(4):645-56. [DOI] [PubMed]
- 5.Nakajima Y, Miyazono K, Nakamura H. Immunolocalization of latent transforming growth factor-beta binding protein-1 (LTBP1) during mouse development: possible roles in epithelial and mesenchymal cytodifferentiation. Cell Tissue Res. 1999;295(2):257–267. doi: 10.1007/s004410051232. [DOI] [PubMed] [Google Scholar]; 5- Nakajima Y, Miyazono K, Nakamura H. Immunolocalization of latent transforming growth factor-beta binding protein-1 (LTBP1) during mouse development: possible roles in epithelial and mesenchymal cytodifferentiation. Cell Tissue Res. 1999;295(2):257-67. doi:10.1007/s004410051232 [DOI] [PubMed]
- 6.Pedrozo HA, Schwartz Z, Gomez R, Ornoy A, Xin-Sheng W, Dallas SL, et al. Growth plate chondrocytes store latent transforming growth factor (TGF)-beta 1 in their matrix through latent TGF-beta 1 binding protein-1. J Cell Physiol. 1998;177(2):343–354. doi: 10.1002/(SICI)1097-4652(199811)177:2<343::AID-JCP16>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]; 6- Pedrozo HA, Schwartz Z, Gomez R, Ornoy A, Xin-Sheng W, Dallas SL, et al. Growth plate chondrocytes store latent transforming growth factor (TGF)-beta 1 in their matrix through latent TGF-beta 1 binding protein-1. J Cell Physiol. 1998;177(2):343-54. doi:10.1002/(SICI)1097-4652(199811)177:2<343::AID-JCP16>3.0.CO;2-A [DOI] [PubMed]
- 7.Pedrozo HA, Schwartz Z, Mokeyev T, Ornoy A, Xin-Sheng W, Bonewald LF, et al. Vitamin D3 metabolites regulate LTBP1 and latent TGF-beta1 expression and latent TGF-beta1 incorporation in the extracellular matrix of chondrocytes. J Cell Biochem. 1999;72(1):151–165. [PubMed] [Google Scholar]; 7- Pedrozo HA, Schwartz Z, Mokeyev T, Ornoy A, Xin-Sheng W, Bonewald LF, et al. Vitamin D3 metabolites regulate LTBP1 and latent TGF-beta1 expression and latent TGF-beta1 incorporation in the extracellular matrix of chondrocytes. J Cell Biochem. 1999;72(1):151-65. [PubMed]
- 8.Dallas SL, Keene DR, Bruder SP, Saharinen J, Sakai LY, Mundy GR, et al. Role of the latent transforming growth factor beta binding protein 1 in fibrillin-containing microfibrils in bone cells in vitro and in vivo. J Bone Miner Res. 2000;15(1):68–81. doi: 10.1359/jbmr.2000.15.1.68. [DOI] [PubMed] [Google Scholar]; 8- Dallas SL, Keene DR, Bruder SP, Saharinen J, Sakai LY, Mundy GR, et al. Role of the latent transforming growth factor beta binding protein 1 in fibrillin-containing microfibrils in bone cells in vitro and in vivo. J Bone Miner Res. 2000;15(1):68-81. doi:10.1359/jbmr.2000.15.1.68 [DOI] [PubMed]
- 9.Ota T, Fujii M, Sugizaki T, Ishii M, Miyazawa K, Aburatani H, et al. Targets of transcriptional regulation by two distinct type I receptors for transforming growth factor-beta in human umbilical vein endothelial cells. J Cell Physiol. 2002;193(3):299–318. doi: 10.1002/jcp.10170. [DOI] [PubMed] [Google Scholar]; 9- Ota T, Fujii M, Sugizaki T, Ishii M, Miyazawa K, Aburatani H, et al. Targets of transcriptional regulation by two distinct type I receptors for transforming growth factor-beta in human umbilical vein endothelial cells. J Cell Physiol. 2002;193(3):299-318. doi:10.1002/jcp.10170 [DOI] [PubMed]
- 10.Vittal R, Mickler EA, Fisher AJ, Zhang C, Rothhaar K, Gu H, et al. Type V collagen induced tolerance suppresses collagen deposition, TGF-β and associated transcripts in pulmonary fibrosis [published correction appears in PLoS One. 2018;13(12):e0209107] PLoS One. 2013;8(10):e76451. doi: 10.1371/journal.pone.0076451. Published 2013 Oct 21. [DOI] [PMC free article] [PubMed] [Google Scholar]; 10- Vittal R, Mickler EA, Fisher AJ, Zhang C, Rothhaar K, Gu H, et al. Type V collagen induced tolerance suppresses collagen deposition, TGF-β and associated transcripts in pulmonary fibrosis [published correction appears in PLoS One. 2018;13(12):e0209107]. PLoS One. 2013;8(10):e76451. Published 2013 Oct 21. doi:10.1371/journal.pone.0076451 [DOI] [PMC free article] [PubMed]
- 11.Nakajima Y, Miyazono K, Kato M, Takase M, Yamagishi T, Nakamura H. Extracellular fibrillar structure of latent TGF beta binding protein-1: role in TGF beta-dependent endothelial-mesenchymal transformation during endocardial cushion tissue formation in mouse embryonic heart. J Cell Biol. 1997;136(1):193–204. doi: 10.1083/jcb.136.1.193. [DOI] [PMC free article] [PubMed] [Google Scholar]; 11- Nakajima Y, Miyazono K, Kato M, Takase M, Yamagishi T, Nakamura H. Extracellular fibrillar structure of latent TGF beta binding protein-1: role in TGF beta-dependent endothelial-mesenchymal transformation during endocardial cushion tissue formation in mouse embryonic heart. J Cell Biol. 1997;136(1):193-204. doi:10.1083/jcb.136.1.193 [DOI] [PMC free article] [PubMed]
- 12.Nakano M, Arai E, Nakajima Y, Nakamura H, Miyazono K, Hirose T. Immunohistochemical study of chondrolipoma: possible importance of transforming growth factor (TGF)-betas, latent TGF-beta binding protein-1 (LTBP-1), and bone morphogenetic protein (BMP) for chondrogenesis in lipoma. J Dermatol. 2003;30(3):189–195. doi: 10.1111/j.1346-8138.2003.tb00370.x. [DOI] [PubMed] [Google Scholar]; 12- Nakano M, Arai E, Nakajima Y, Nakamura H, Miyazono K, Hirose T. Immunohistochemical study of chondrolipoma: possible importance of transforming growth factor (TGF)-betas, latent TGF-beta binding protein-1 (LTBP-1), and bone morphogenetic protein (BMP) for chondrogenesis in lipoma. J Dermatol. 2003;30(3):189-95. doi:10.1111/j.1346-8138.2003.tb00370.x [DOI] [PubMed]
- 13.Gui Y, Murphy LJ. Interaction of insulin-like growth factor binding protein-3 with latent transforming growth factor-beta binding protein-1. Mol Cell Biochem. 2003;250(1-2):189–195. doi: 10.1023/a:1024990409102. [DOI] [PubMed] [Google Scholar]; 13- Gui Y, Murphy LJ. Interaction of insulin-like growth factor binding protein-3 with latent transforming growth factor-beta binding protein-1. Mol Cell Biochem. 2003;250(1-2):189-95. doi:10.1023/a:1024990409102 [DOI] [PubMed]
- 14.Ge G, Greenspan DS. BMP1 controls TGF beta1 activation via cleavage of latent TGFbeta-binding protein. J Cell Biol. 2006;175(1):111–120. doi: 10.1083/jcb.200606058. [DOI] [PMC free article] [PubMed] [Google Scholar]; 14- Ge G, Greenspan DS. BMP1 controls TGF beta1 activation via cleavage of latent TGFbeta-binding protein. J Cell Biol. 2006;175(1):111-20. doi:10.1083/jcb.200606058 [DOI] [PMC free article] [PubMed]
- 15.Maurya VK, Jha RK, Kumar V, Joshi A, Chadchan S, Mohane JJ, et al. Transforming growth factor-beta 1 (TGF-B1) liberation from its latent complex during embryo implantation and its regulation by estradiol in mouse [published correction appears in Biol Reprod. 2014;91(6):147] Biol Reprod. 2013;89(4):84–84. doi: 10.1095/biolreprod.112.106542. Published 2013 Oct 10. [DOI] [PubMed] [Google Scholar]; 15- Maurya VK, Jha RK, Kumar V, Joshi A, Chadchan S, Mohane JJ, et al. Transforming growth factor-beta 1 (TGF-B1) liberation from its latent complex during embryo implantation and its regulation by estradiol in mouse [published correction appears in Biol Reprod. 2014;91(6):147]. Biol Reprod. 2013;89(4):84. Published 2013 Oct 10. doi:10.1095/biolreprod.112.106542 [DOI] [PubMed]
- 16.Zhao Q, Zheng K, Ma C, Li J, Zhuo L, Huang W, et al. PTPS facilitates compartmentalized LTBP1 S-nitrosylation and promotes tumor growth under hypoxia. Mol Cell. 2020;77(1):95–107.e5. doi: 10.1016/j.molcel.2019.09.018. [DOI] [PubMed] [Google Scholar]; 16- Zhao Q, Zheng K, Ma C, Li J, Zhuo L, Huang W, et al. PTPS facilitates compartmentalized LTBP1 S-nitrosylation and promotes tumor growth under hypoxia. Mol Cell. 2020;77(1):95-107.e5. doi:10.1016/j.molcel.2019.09.018 [DOI] [PubMed]
- 17.Robertson IB, Handford PA, Redfield C. Backbone 1H, 13C and 15N resonance assignment of the C-terminal EGF-cbEGF pair of LTBP1 and flanking residues. Biomol NMR Assign. 2014;8(1):159–163. doi: 10.1007/s12104-013-9474-6. [DOI] [PubMed] [Google Scholar]; 17- Robertson IB, Handford PA, Redfield C. Backbone 1H, 13C and 15N resonance assignment of the C-terminal EGF-cbEGF pair of LTBP1 and flanking residues. Biomol NMR Assign. 2014;8(1):159-63. doi:10.1007/s12104-013-9474-6. [DOI] [PubMed]
- 18.Robertson IB, Handford PA, Redfield C. NMR spectroscopic and bioinformatic analyses of the LTBP1 C-terminus reveal a highly dynamic domain organisation. PLoS One. 2014;9(1):e87125. doi: 10.1371/journal.pone.0087125. [DOI] [PMC free article] [PubMed] [Google Scholar]; 18- Robertson IB, Handford PA, Redfield C. NMR spectroscopic and bioinformatic analyses of the LTBP1 C-terminus reveal a highly dynamic domain organisation. PLoS One. 2014;9(1):e87125. doi:10.1371/journal.pone.0087125 [DOI] [PMC free article] [PubMed]
- 19.Chen H, Cai W, Chu ES, Tang J, Wong CC, Wong SH, et al. Hepatic cyclooxygenase-2 overexpression induced spontaneous hepatocellular carcinoma formation in mice. Oncogene. 2017;36(31):4415–4426. doi: 10.1038/onc.2017.73. [DOI] [PMC free article] [PubMed] [Google Scholar]; 19- Chen H, Cai W, Chu ES, Tang J, Wong CC, Wong SH, et al. Hepatic cyclooxygenase-2 overexpression induced spontaneous hepatocellular carcinoma formation in mice. Oncogene. 2017;36(31):4415-26. doi:10.1038/onc.2017.73 [DOI] [PMC free article] [PubMed]
- 20.Hubmacher D, Wang LW, Mecham RP, Reinhardt DP, Apte SS. Adamtsl2 deletion results in bronchial fibrillin microfibril accumulation and bronchial epithelial dysplasia--a novel mouse model providing insights into geleophysic dysplasia. Dis Model Mech. 2015;8(5):487–499. doi: 10.1242/dmm.017046. [DOI] [PMC free article] [PubMed] [Google Scholar]; 20- Hubmacher D, Wang LW, Mecham RP, Reinhardt DP, Apte SS. Adamtsl2 deletion results in bronchial fibrillin microfibril accumulation and bronchial epithelial dysplasia--a novel mouse model providing insights into geleophysic dysplasia. Dis Model Mech. 2015;8(5):487-99. doi:10.1242/dmm.017046 [DOI] [PMC free article] [PubMed]
- 21.Todorovic V, Frendewey D, Gutstein DE, Chen Y, Freyer L, Finnegan E, et al. Long form of latent TGF-beta binding protein 1 (Ltbp1L) is essential for cardiac outflow tract septation and remodeling. Development. 2007;134(20):3723–3732. doi: 10.1242/dev.008599. [DOI] [PubMed] [Google Scholar]; 21- Todorovic V, Frendewey D, Gutstein DE, Chen Y, Freyer L, Finnegan E, et al. Long form of latent TGF-beta binding protein 1 (Ltbp1L) is essential for cardiac outflow tract septation and remodeling. Development. 2007;134(20):3723-32. doi:10.1242/dev.008599 [DOI] [PubMed]
- 22.Horiguchi M, Todorovic V, Hadjiolova K, Weiskirchen R, Rifkin DB. Abrogation of both short and long forms of latent transforming growth factor-β binding protein-1 causes defective cardiovascular development and is perinatally lethal. Matrix Biol. 2015;43:61–70. doi: 10.1016/j.matbio.2015.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]; 22- Horiguchi M, Todorovic V, Hadjiolova K, Weiskirchen R, Rifkin DB. Abrogation of both short and long forms of latent transforming growth factor-β binding protein-1 causes defective cardiovascular development and is perinatally lethal. Matrix Biol. 2015;43:61-70. doi:10.1016/j.matbio.2015.03.006 [DOI] [PMC free article] [PubMed]
- 23.Drews F, Knöbel S, Moser M, Muhlack KG, Mohren S, Stoll C, et al. Disruption of the latent transforming growth factor-beta binding protein-1 gene causes alteration in facial structure and influences TGF-beta bioavailability. Biochim Biophys Acta. 2008;1783(1):34–48. doi: 10.1016/j.bbamcr.2007.08.004. [DOI] [PubMed] [Google Scholar]; 23- Drews F, Knöbel S, Moser M, Muhlack KG, Mohren S, Stoll C, et al. Disruption of the latent transforming growth factor-beta binding protein-1 gene causes alteration in facial structure and influences TGF-beta bioavailability. Biochim Biophys Acta. 2008;1783(1):34-48. doi:10.1016/j.bbamcr.2007.08.004 [DOI] [PubMed]
- 24.Dietzel E, Weiskirchen S, Floehr J, Horiguchi M, Todorovic V, Rifkin DB, et al. Latent TGF-β binding protein-1 deficiency decreases female fertility. Biochem Biophys Res Commun. 2017;482(4):1387–1392. doi: 10.1016/j.bbrc.2016.12.046. [DOI] [PubMed] [Google Scholar]; 24- Dietzel E, Weiskirchen S, Floehr J, Horiguchi M, Todorovic V, Rifkin DB, et al. Latent TGF-β binding protein-1 deficiency decreases female fertility. Biochem Biophys Res Commun. 2017;482(4):1387-92. doi:10.1016/j.bbrc.2016.12.046 [DOI] [PubMed]
- 25.Mork L, Crump G. Zebrafish craniofacial development: a window into early patterning. Curr Top Dev Biol. 2015;115:235–269. doi: 10.1016/bs.ctdb.2015.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]; 25- Mork L, Crump G. Zebrafish craniofacial development: a window into early patterning. Curr Top Dev Biol. 2015;115:235-269. doi:10.1016/bs.ctdb.2015.07.001 [DOI] [PMC free article] [PubMed]
- 26.Machado RG, Eames BF. Using zebrafish to test the genetic basis of human craniofacial diseases. J Dent Res. 2017;96(11):1192–1199. doi: 10.1177/0022034517722776. [DOI] [PubMed] [Google Scholar]; 26- Machado RG, Eames BF. Using zebrafish to test the genetic basis of human craniofacial diseases. J Dent Res. 2017;96(11):1192-9. doi:10.1177/0022034517722776 [DOI] [PubMed]
- 27.Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF. Stages of embryonic development of the zebrafish. Dev Dyn. 1995;203(3):253–310. doi: 10.1002/aja.1002030302. [DOI] [PubMed] [Google Scholar]; 27- Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF. Stages of embryonic development of the zebrafish. Dev Dyn. 1995;203(3):253-310. doi:10.1002/aja.1002030302 [DOI] [PubMed]
- 28.Stainier DY, Raz E, Lawson ND, Ekker SC, Burdine RD, Eisene JS, et al. Guidelines for morpholino use in zebrafish. PLoS Genet. 2017;13(10):e1007000. doi: 10.1371/journal.pgen.1007000. [DOI] [PMC free article] [PubMed] [Google Scholar]; 28- Stainier DY, Raz E, Lawson ND, Ekker SC, Burdine RD, Eisene JS, et al. Guidelines for morpholino use in zebrafish. PLoS Genet. 2017;13(10):e1007000. doi:10.1371/journal.pgen.1007000 [DOI] [PMC free article] [PubMed]
- 29.Thisse B, Thisse C. In situ hybridization on whole-mount zebrafish embryos and young larvae. Methods Mol Biol. 2014;1211:53–67. doi: 10.1007/978-1-4939-1459-3_5. [DOI] [PubMed] [Google Scholar]; 29- Thisse B, Thisse C. In situ hybridization on whole-mount zebrafish embryos and young larvae. Methods Mol Biol. 2014;1211:53-67. doi:10.1007/978-1-4939-1459-3_5 [DOI] [PubMed]
- 30.Kimmel CB, Miller CT, Kruze G, Ullmann B, BreMiller RA, Larison KD, et al. The shaping of pharyngeal cartilages during early development of the zebrafish. Dev Biol. 1998;203(2):245–263. doi: 10.1006/dbio.1998.9016. [DOI] [PubMed] [Google Scholar]; 30- Kimmel CB, Miller CT, Kruze G, Ullmann B, BreMiller RA, Larison KD, et al. The shaping of pharyngeal cartilages during early development of the zebrafish. Dev Biol. 1998;203(2):245-63. doi:10.1006/dbio.1998.9016 [DOI] [PubMed]
- 31.Yan YL, Willoughby J, Liu D, Crump JG, Wilson C, Miller CT, et al. A pair of sox: distinct and overlapping functions of zebrafish sox9 co-orthologs in craniofacial and pectoral fin development. Development. 2005;132(5):1069–1083. doi: 10.1242/dev.01674. [DOI] [PubMed] [Google Scholar]; 31- Yan YL, Willoughby J, Liu D, Crump JG, Wilson C, Miller CT, et al. A pair of sox: distinct and overlapping functions of zebrafish sox9 co-orthologs in craniofacial and pectoral fin development. Development. 2005;132(5):1069-83. doi:10.1242/dev.01674 [DOI] [PubMed]
- 32.Xu Y, He J, Wang X, Lim TM, Gong Z. Asynchronous activation of 10 muscle-specific protein (MSP) genes during zebrafish somitogenesis. Dev Dyn. 2000;219(2):201–215. doi: 10.1002/1097-0177(2000)9999:9999<::aid-dvdy1043>3.3.co;2-9. [DOI] [PubMed] [Google Scholar]; 32- Xu Y, He J, Wang X, Lim TM, Gong Z. Asynchronous activation of 10 muscle-specific protein (MSP) genes during zebrafish somitogenesis. Dev Dyn. 2000;219(2):201-15. doi:10.1002/1097-0177(2000)9999:9999<::aid-dvdy1043>3.3.co;2-9 [DOI] [PubMed]
- 33.Thisse B, Pflumio S, Fürthauer M, Loppin B, Heyer V, Degrave A, et al. Expression of the zebrafish genome during embryogenesis (NIH R01 RR15402) [Internet] Eugene: ZFIN; 2001. [[cited 2020 Aug 5]]. Available from: https://zfin.org/ZDB-PUB-010810-1. [Google Scholar]; 33- Thisse B, Pflumio S, Fürthauer M, Loppin B, Heyer V, Degrave A, et al. Expression of the zebrafish genome during embryogenesis (NIH R01 RR15402) [Internet]. Eugene: ZFIN; 2001 [cited 2020 Aug 5]. Available from: https://zfin.org/ZDB-PUB-010810-1
- 34.Zhang Y, Ji D, Li L, Yang S, Zhang H, Duan X. ClC-7 Regulates the pattern and early development of craniofacial bone and tooth. Theranostics. 2019;9(5):1387–1400. doi: 10.7150/thno.29761. [DOI] [PMC free article] [PubMed] [Google Scholar]; 34- Zhang Y, Ji D, Li L, Yang S, Zhang H, Duan X. ClC-7 Regulates the pattern and early development of craniofacial bone and tooth. Theranostics. 2019;9(5):1387-400. doi:10.7150/thno.29761 [DOI] [PMC free article] [PubMed]