Abstract
Background: bladder cancer is one of the most common urinary tract malignancies. Establishment of robust predictors of disease progression and outcome is important for personalizing treatment of non-muscular invasive bladder carcinoma (NMIBC). In this study we evaluated association of PD-L1 expression with other prognostic biomarkers, such as expression of miRNA-145 and miRNA-200a, FGFR3 gene expression, and mutation status in tissue specimens of the luminal subtype of newly diagnosed high and low grade NMIBC. Methods: twenty patients with primary luminal NMIBC were enrolled in the study. Tumor grade and risk level were determined in accordance with European Organization for Research and Treatment of Cancer (EORTC) guidelines and World Health Organization (WHO) classification. Neoplasm molecular subtype and PD-L1 expression level were assessed by immunohistochemistry. We used real-time PCR to evaluate the expression of microRNAs and FGFR3. We detected FGFR3 hotspot mutations in codons 248 and 249 by Sanger sequencing. Results: high grade primary luminal NMIBC showed comparatively higher expression of PD-L1 and microRNA-145 than a low grade tumor, whereas the latter had a higher FGFR3 expression and hotspot mutation rate. The tumor grade (HR = 571.72 [11.03–2.96] p = 0.002), PD-L1 expression (HR = 2.33 [0.92–1.92] p = 0.012), and FGFR3 expression (HR = 0.08 [0.17–0.42] p = 0.003) were associated with relapse-free survival. Conclusions: tumor grade in association with PD-L1 and FGFR3 expression can be considered as a complex predictor for primary luminal NMIBC progression.
Keywords: non-muscular invasive bladder carcinoma, fibroblast growth factor receptor 3, mutation, prognosis, cancer progression, PD-L1, microRNA, expression, tumor relapse
1. Introduction
Bladder cancer remains to be one of the most common human genitourinary tumors worldwide with high cost per patient and high mortality rate [1,2]. Primary morphological features along with involvement of the muscular layer altogether determine two essential divergent pathways of bladder carcinoma development as non-muscular invasive bladder cancer (NMIBC) and muscular invasive bladder carcinoma (MIBC). The first one at the time of diagnosis was proven to be less aggressive than the latter [3,4], but treatment of both requires stringent interventional and surveillance protocols [5]. At the same time, biological and genetic heterogeneity of NMIBC serves as a basis for the disease molecular classification. According to the tumor molecular profiling made by Dadhania and colleagues, CR5/6 and GATA3 expression levels are currently used to designate NMIBC into basal, luminal, and double-negative subtypes [6].
Identification of the pivotal role of immune checkpoint molecules has drastically changed diagnostic, prognostic, and treatment approaches to many malignancies including bladder cancer [7,8,9]. Besides, different laboratories showed that expression levels of the programmed death (PD-1) receptor and its ligand 1 (PD-L1) may broadly vary depending on NMIBC grade and molecular subtype [10]. Moreover, check-point inhibitors’ clinical success closely relates to the tumor immune microenvironment [11], while differences in relapse-free survival (RFS) of patients with luminal, basal, and double-negative NMIBC of high- and low-malignant grade are strictly associated with previously utilized treatment [12].
MicroRNAs are small non-coding molecules involved in different regulatory functions and intracellular signaling [13,14]. Numerous studies have demonstrated prognostic role of microRNAs for NMIBC outcomes not only in tumor tissue-associated form, but also as stable cell-free molecules circulating in different biological fluids [15,16,17]. Yun et al. previously demonstrated an association between NMIBC diagnosis and progression and concentrations of microRNA-145 and microRNA-200a molecules [17]. These and other microRNAs influence tumor progression via the regulation of gene activities, including expression of fibroblast growth factor receptor 3 gene (FGFR3) [18,19]. FGFR3 expression level and mutation status were shown to be determinants of bladder carcinoma progression [20].
In our previous study, we found that PD-L1 expression status was associated with RFS for luminal NMIBCs in the group without previous frontline intervention, and with RFS in the group of patients with luminal relapsed bladder cancer who previously used Bacillus Calmette-Guerin (BCG) [12]. We investigated here the prognostic roles of microRNA-200a, microRNA-145, FGFR3 expression, and mutation status in association with PD-L1 expression in patients with primary luminal NMIBC.
2. Results
2.1. PD-L1 Expression in Primary Luminal NMIBC
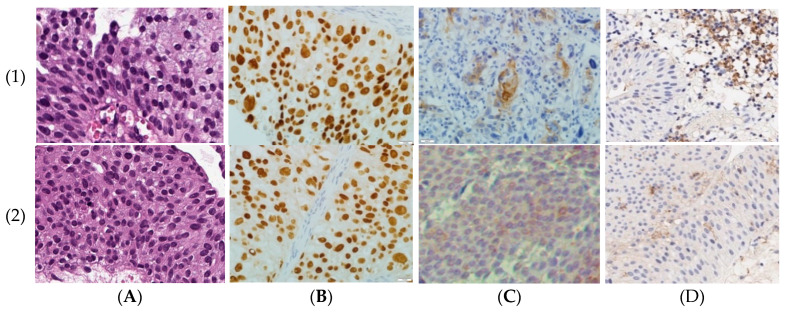
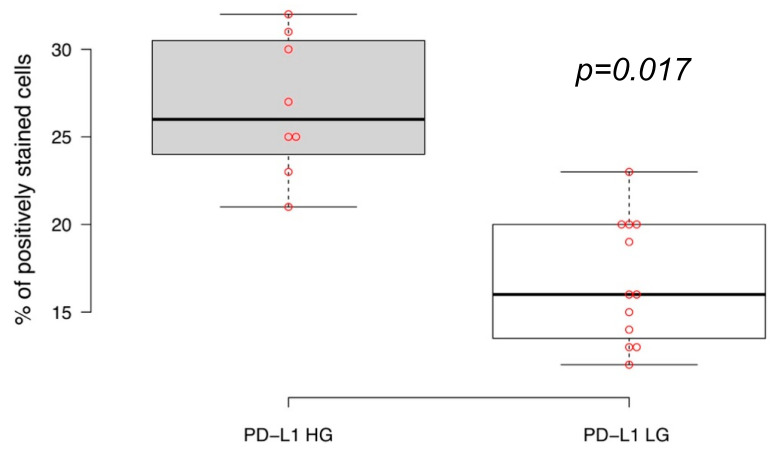
We immunohistochemically assessed PD-L1 expression status of the luminal subtype of non-muscular invasive urothelial carcinomas with different tumor grades (Figure 1 and Figure 2). The proportion of cancer cells positively stained for the PD-L1 membrane expression was statistically higher in GATA3-positive high grade primary NMIBCs (n = 8) than in low grade bladder carcinomas (n = 12), with average values of 26.8% vs. 16.3%, respectively (p = 0.017).
Figure 1.
Morphological characteristic of tumors: (1) high grade primary luminal non-muscular invasive bladder cancer (NMIBC); (2) low grade primary luminal NMIBC. (A) Hematoxylin and eosin staining, ×500. (B) Anti-GATA3 IHC-staining, ×500. (C) Anti-CR5/6 IHC-staining, ×500. (D) Anti-PD-L1 IHC-staining, ×500.
Figure 2.
High-grade (HG) and low grade (LG) non-muscular invasive bladder cancers of luminal molecular subtype express PD-L1 differently. Boxplots show medians of % of tumor cellular membranes positively stained by anti-PD-L1 antibody (Ventana PD-L1 Assay (SP263)); significance of expression differences were estimated by independent t test.
2.2. FGFR3 Expression and Hotspot Mutations in Primary Luminal NMIBC
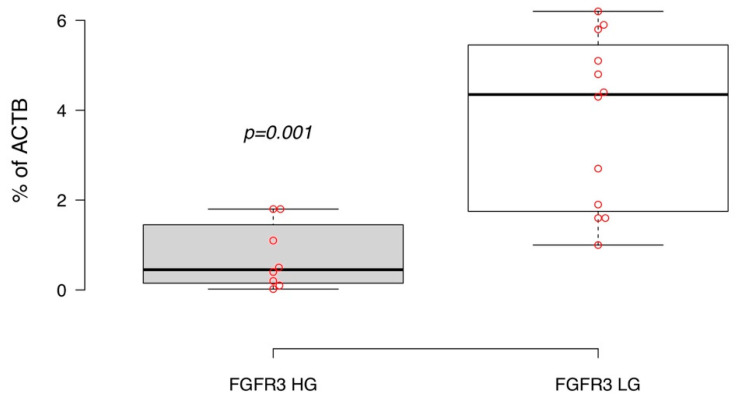
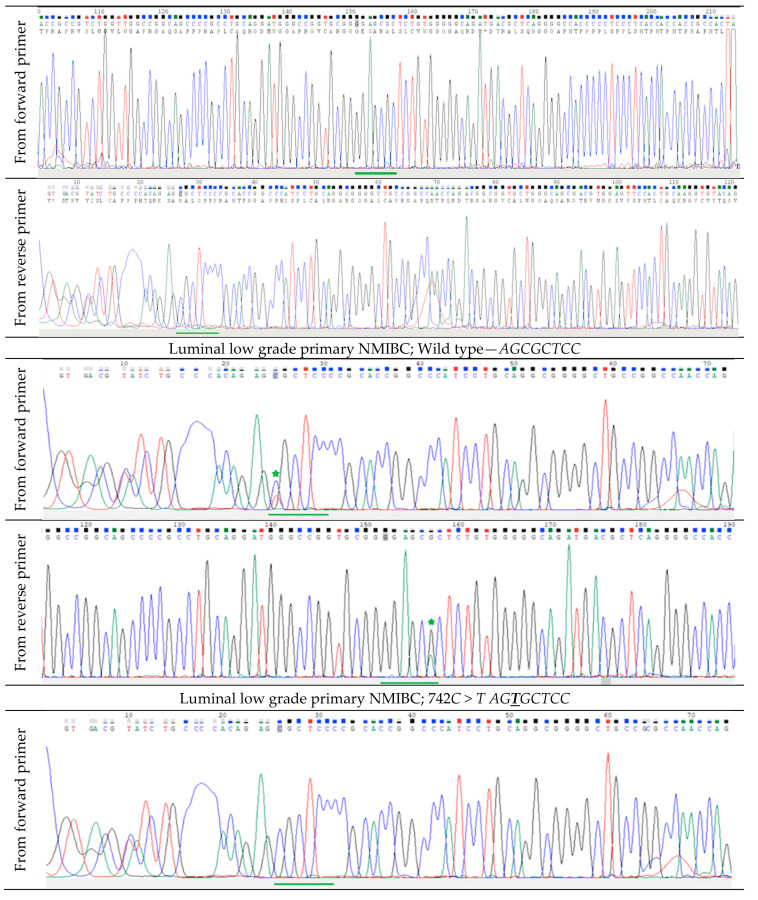
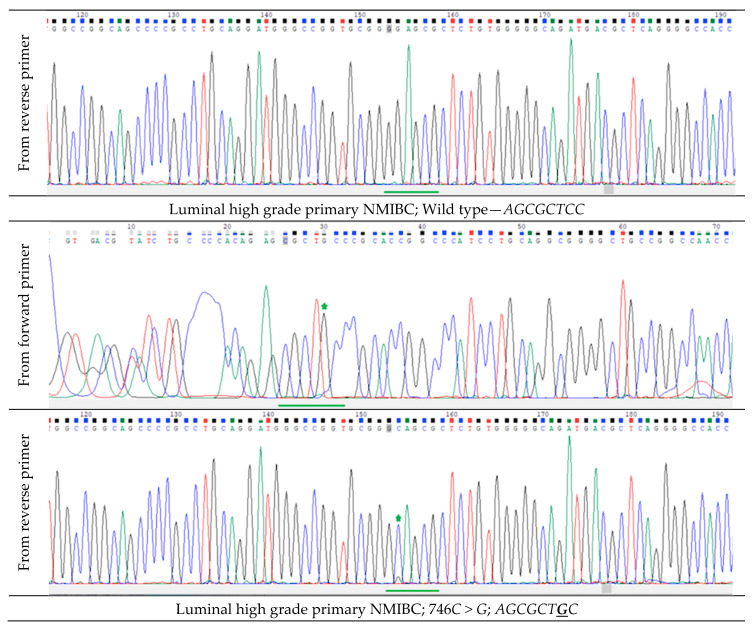
We then assessed the FGFR3 gene transcript expression as the percentage of the ACTB housekeeping gene mRNA concentration. In our experiments, high grade luminal NMIBC was associated with relatively lower cellular expression of FGFR3 compared to low grade non-invasive urothelial cancer (Figure 3, p = 0.001). Detecting FGFR3 hotspot mutations by Sanger sequencing for both grades of primary luminal non-invasive bladder cancer also demonstrated the prevalence of the gene mutation rate in codons 248 and 249 in low grade tumor cells (66.6% of samples had FGFR3 mutations), whereas this proportion was only 25% in high grade luminal NMIBCs (Figure A1). Interestingly, FGFR3 mutation status was not correlated with this gene expression level.
Figure 3.
FGFR3 expression level in primary diagnosed high (HG) and low grade (LG) luminal NMIBC. Boxplots display medians of % of beta-actin gene (ACTB) expression; significance of expression differences were estimated by independent t test.
2.3. MicroRNA-145 and MicroRNA-200a Expression in Primary Luminal NMIBC
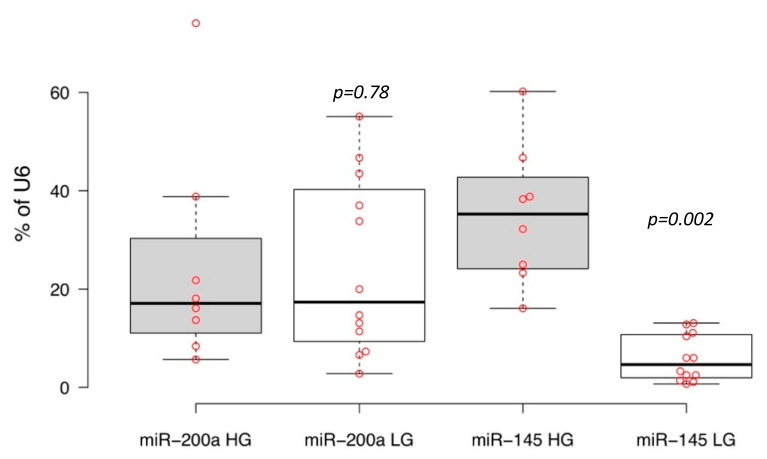
We then assessed expressions of microRNAs-200a and -145 in the tumor samples under investigation by quantitative real-time reverse transcription polymerase chain reaction (PCR). The principle of the microRNA detection method used is described by Chen and co-authors (2005) [21]. This method is based on the individual reverse transcription of mature microRNA using a unique long stem-loop primer. The resulting product was detected by quantitative PCR with a specific fluorescent probe. MicroRNA levels were measured as the percentage of housekeeping U6 small nucleolar RNA expression. We found no statistically significant differences (p = 0.78) between more and less aggressive primary luminal subtypes of bladder cancer for cellular microRNA-200a expression levels; in contrast, the high grade luminal NMIBC tumor tissues had a ~5.9 times higher expression of microRNA-145 than low grade primary tumors (p = 0.002) (Figure 4).
Figure 4.
Expression of microRNA-200a (miR-200a) and microRNA-145 (miR-145) in primary high (HG) and low grade (LG) non-muscular invasive urothelial carcinoma of luminal molecular subtype. Boxplots display medians of percentage of U6 snRNA expression; significance of expression differences were estimated by independent t test.
2.4. PD-L1, FGFR3 and MicroRNAs Expression, and Relapse-Free Survival in Luminal NMIBC
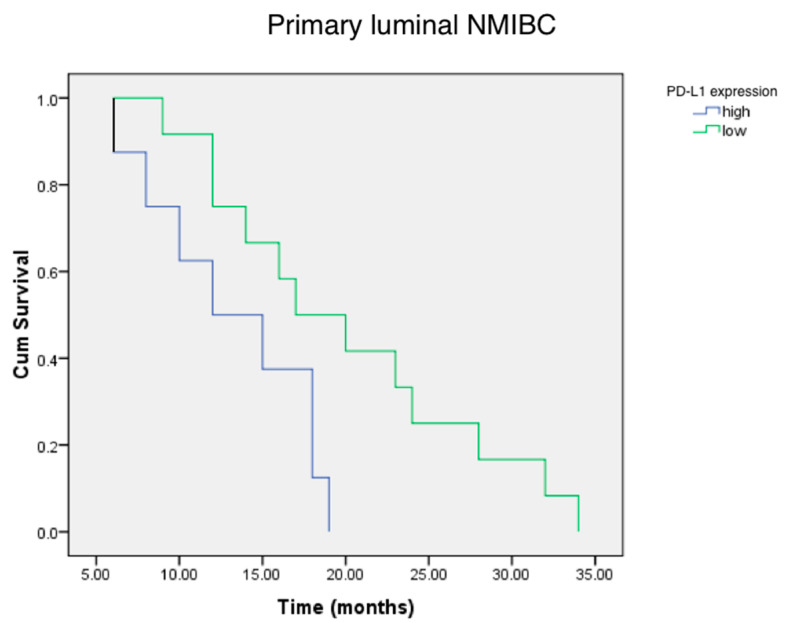
Using the Kaplan–Meier actuarial analysis, we demonstrated that the time to first tumor relapse was lower for the patients with a high PD-L1 status of luminal primary cancer in comparison with less aggressive low PD-L1 expressing luminal NMIBC (Figure 5).
Figure 5.
Cumulative survival (Cum Survival) of patients with high and low PD-L1 expressing the status of primary luminal NMIBC. According to long rank test, there is significant evidence of a difference in relapse times for low and high PD-L1+ status groups (p < 0.05).
Using univariable analysis, we then found that the tumor grade (HR = 571.72 [11.03–2.96] p = 0.002), PD-L1 expression (HR = 2.33 [0.92–1.92] p = 0.012), and FGFR3 expression (HR = 0.08 [0.17–0.42] p = 0.003) were significantly associated with relapse-free survival (Table 1). At the same time, there were no significant link between RFS and miR-145, mir-200a, and FGFR3 gene mutation rate.
Table 1.
Univariable Cox regression models for prediction of recurrence in primary luminal NMIBC.
| Variables | HR | 95% CI | p-Value |
|---|---|---|---|
| Tumor grade | 571.72 | 11.03–2.96 | 0.002 |
| PD-L1 expression | 2.33 | 0.92–1.92 | 0.012 |
| miR-200a expression | 0.98 | 0.95–1.01 | 0.40 |
| miR-145 expression | 0.99 | 0.93–1.06 | 0.96 |
| FGFR3 expression | 0.08 | 0.17–0.42 | 0.003 |
| FGFR3 gene mutations | 1.46 | 0.35–6.00 | 0.593 |
3. Discussion
Temporary cancer molecular biology delivers a broad range of novel molecules and markers that frequently play pivotal roles at some definite stages of tumor development and progression. From a clinician’s point of view, though, it is of great importance to base effective and safe treatment strategies on strong, proven, and, simultaneously and desirably, a limited panel of predictors, which reflects involvement of key pathways in the malignant process. Such internal ambiguity between galloping molecular science and quite conservative clinical medicine requires nontrivial approaches to the issue of the prediction of cancer progression. There is a wide panel of markers with different predictive values for the prognosis of bladder cancer behavior [22,23,24]. During recent decades, we observed how high expert boards tried to combine different clinical hallmarks of the disease and implement molecular novels for guidelines, consensuses, and classifications. As a result, sophisticated actual molecular and clinical classification of non-muscular-invasive bladder cancer illuminated an urgent need for stringent and reliable prognosticators for at least the main molecular subtypes of the tumor, luminal, basal, and double-negative NMIBC.
Based on the recent studies, we narrowed our current research focus on three molecular pathways—immune checkpoint axis PD1/PD-L1, fibroblast growth factor receptor 3 expression status, and tumor-dependent miRs. The prognostic role of PD-L1 for bladder cancer progression and the treatment success of immune checkpoint inhibitors have been rigorously evaluated in many studies [7,8,9,10,11,12]. In our previous study, we demonstrated that PD-L1 expression level correlated with RFS in relapsed aggressive non-muscular invasive bladder carcinoma of the luminal molecular subtype (GATA3-expressing NMIBC) [12,25]. Kang et al. showed that the FGFR3 expression level and its gene mutation status associated it with survival in primary pT1 bladder cancer [19]. At the same time, a research team led by Neuzillet demonstrated a link between FGFR3 mutations, but not expression, and better survival in NMIBC [20]. Meaning of cell-free urine miR-145 and -200a level as marks of emerging bladder cancer was highlighted by Yun [16], whereas many tumor-associated miRs also had predictive value. Having considered the aforementioned, we wondered how relapse-free survival of patients with luminal primary NMIBC might depend on PD-L1 expression, FGFR3 expression, FGFR3 gene hotspot mutation status, and tumor-associated miRs-145 and -200a assessed in complex.
We found out that the more aggressive primary luminal tumor also expressed PD-L1 higher than the less aggressive form. Obtained data corresponded with our previous results [12,25]. Contrast data were received during assessment of FGFR3 expression. High grade primary luminal bladder cancer was associated with low FGFR3 expression, and vice versa. It should be underlined that we found no correlation between the FGFR3 gene mutation rate and the receptor expression, despite the mutations intergroup significant differences between high and less aggressive primary tumors. The fact was disputable with previous observations [20]. At the same time, prior results reflected all of the NMIBC population without taking into consideration its molecular heterogeneity. Probably, an increase in the number of observations would allow to get more definite results. We found out that tumor-related miR-200a expression did not vary in high and low grade primary luminal bladder cancer; hence, the molecule had no further prognostic value. In contrast, we detected reciprocal PD-L1-like tumor-related miR-145 expression in study groups. Therefore, not only cell-free but the intracellular level of miR-145 might be considered as a potential predictive biomolecule. We also found out the inverse relationship between miR-145 expression and FGFR3 regulation in study subgroups. To date, there are no clear proofs of miR-145 to target FGFR3 expression [26], but the revealed correlation needs further investigation. It might be of great importance taking into consideration Zhu and co-authors recent findings of a positive feedback loop for promoting PD-L1 expression in human bladder cancer cells via the ATG7/autophagy/FOXO3A/miR-145 axis [27]. Beside intergroup differences, it was of great importance to evaluate the markers prognostic value for patients’ survival. It turned out that low FGFR3 expression in combination with high PD-L1+ status predicted bad survival of patients with primary luminal non-invasive bladder cancer. Obtained data may be used for developing promising prognosticators in oncology. Further observations should be done to clear the predictive value of well-known and novel molecules in basal and double-negative NMIBC with and without p53 mutations, which will be a focus of our subsequent research.
4. Materials and Methods
4.1. Ethic Procedures
We designed the study in accordance with all requirements to protection of patients’ rights and confidentiality. Study Protocol was reviewed and approved by Ethic Committee of Sechenov University at the Committee Board meeting on 16 September 2019 (Approval No. 9; Rev. No 16/09-1-2019) and Scientific Board of Bio-Ethic Commission of National Research Medical Center of Radiology (Approval No. 12, 2 October 2019; Rev. No. 02/10-2019-E).
4.2. Study Population and Follow-Up Protocol
Twenty patients with confirmed luminal molecular subtype type of non-muscular invasive bladder carcinoma who underwent bladder intervention (endoscopic biopsy or transurethral resection) at Oncology Clinic of Sechenov University or Urology Clinical Hospital of National Research Medical Center of Radiology between January 2014 and May 2015 were enrolled in the study. Informed voluntarily consent was given by all the study participants that met the eligibility criteria as follows: (1) age of 18 years or more at the time of the diagnosis; (2) luminal molecular subtype of primary diagnosed non-muscular invasive urothelial carcinoma; (3) available both no longer than 3 year-frozen neoplasm specimens for real-time PCR and formalin-fixed tumor tissue samples for IHC testing. Patients with primary invasive bladder carcinoma, basal, and double-negative NMIBCs were excluded from the study. Before intervention and tissue samples collection, urine of each patient was examined for possible infection. All patients with urinary infection were excluded from the study.
Table A1 summarizes essential participants’ gender data and also contains clinical and morphological features of diagnosed tumors. Eleven males with average age 56.72 ± 3.1 and nine females whose age averaged at 57.55 ± 2.6 formed the study population. For each case, histologic diagnosis was determined along with assessment of individual risk for the tumor recurrence and progression in accordance with European Organization for Research and Treatment of Cancer (EORTC) risk tables [28] and World Health Organization (WHO) classification [29]. Bladder tumors histologically presented by 18 urothelial papillary carcinomas and 2 micropapillary cancers were designated into low grade (12) and high-grade (8) malignancies.
Intervention implemented and surveillance programs were designed for patients in dependence on the lesion stage, grade, and risk. Participants with low grade NMIBC were treated by intravesical Mitomycin 40 mg weekly for six weeks with following cystoscopy and urine cytology at three months after intervention and subsequent observation nine months later if negative. In this subgroup of patients, diagnostic cystoscopy was carried out once a year in years 4–5 of surveillance. Intervention in high grade NMIBC included immunotherapy of Bacillus Calmette-Guerin (BCG) intravesically, with 100 mg per instillation in the same regimen as mentioned above for Mitomycin. Invasive bladder observations in this subgroup were carried quarterly for years 1 and 2 after primary diagnosis, and thereafter twice a year up to five years if throughout the period relapses did not occur. Relapse-free survival (RFS) was defined as the time from first diagnosis of urothelial carcinoma as the primary tumor date to the date of the first documented tumor relapse, or death due to any cause, whichever occurs first.
4.3. Tissue Sample Preparation and Processing
All extracted tumor specimens no longer than 5–7 min after intervention were divided into three equal parts. The first one underwent histologic tissue processing resulting in formalin-fixed and paraffin-embedded blocks for further histologic examination. The other parts were immediately frozen at −86 °C. Tissue freezing conditions met basic requirements for biological samples preserving, which would ensure equal diagnostic quality of all samples [30]. One of two frozen samples was designated to miRNAs, detecting FGFR3 mutations and gene expression determination. The other frozen fragment of tumor tissue sample underwent immunohistochemical testing for GATA3, KRT5/6, and PD-L1 expression. The molecular panel was chosen for the purpose of multifactorial analysis in accordance with the high prognostic value of the biomarkers as survival predictors.
4.4. DNA and RNA Isolation
Having been thawed, fragments of freshly frozen tumor tissues were homogenized in 600 µL of RLT Plus buffer solution (Qiagen, Hilden, Germany) with 1% beta-mercaptoethanol with the TissueLyser LT homogenizer (Qiagen, Hilden, Germany) using Lysing Matrix A tubes (MP Biomedicals, Irvine, CA, USA). Nucleic acids were isolated using the AllPrep DNA/RNA/miRNA Universal Kit (Qiagen, Hilden, Germany) according to the manufacturer’s recommendations. DNA and RNA concentrations were measured with a Qubit 4 fluorimeter (“Thermo Fisher Scientific”, Waltham, MA, USA) using, respectively, the Qubit dsDNA HS Assay and Qubit RNA BR Assay kits (“Thermo Fisher Scientific”, Waltham, MA, USA).
4.5. Reverse Transcription
For every sample, 100 ng of total RNA was mixed with 20 pm of a reverse transcription primer oligonucleotide (random decamers were used for FGFR3 and ACTB expression, and specific oligonucleotide primers were used for microRNAs, listed below) in 9 µL and incubated at 70 °C for 2 min, and then they were chilled on ice. Reverse transcription was performed at 42 °C for 30 min using the MMLV RT kit (Evrogen, Moscow, Russia) according to the manufacturer’s recommendations. Reverse transcription was then stopped by inactivating reverse transcriptase by incubating the reaction mixture at 70 °C for 10 min. Obtained cDNA was 10-fold diluted and used for RNA expression analyses using real-time PCR experiments.
4.6. Real-Time Polymerase Chain Reaction (PCR)
Real-time PCR experiments were performed using a DTPrime amplifier (DNA technology, Russia). For FGFR3 and ACTB expression, we used qPCRmix-HS SYBR (Evrogen, Moscow, Russia) according to the manufacturer’s recommendations. A measure of 1.5 µL of cDNA solution and 4 pm of each PCR primer were amplified in 25 µL using the following protocol: (1) DNA denaturation at 95 °C for 2 min; (2) 45 cycles of the following: DNA denaturation at 95 °C for 10 s, primer annealing at 67 °C for 3 s, and elongation at 72 °C for 18 s; (3) melting curve analysis.
The following oligonucleotide primers were used for FGFR3: FGFR3RT-F, 5′-CCCAAATGGGAGCTGTCTCG-3′; FGFR3RT-R, 5′-CATCTCAGACACCAGGTCCG-3; for ACTB: b-act-for, 5′-GAGCGGGAAATCGTGCGTGACATT-3′; b-act-rev; 5′-GATGGAGTTGAAGGTAGTTTCGTG-3′.
For microRNAs and for RNU6-1, we used qPCRmix-HS (Evrogen, Moscow, Russia) according to the manufacturer’s recommendations. A measure of 1.5 µL of cDNA solution, 4 pm of each PCR primer, and 3 pm of dual fluorescently labelled detection probe were amplified in 25 µL using the following protocol: (1) DNA denaturation at 94 °C for 2 min; (2) 50 cycles of the following: DNA denaturation at 94 °C for 10 s, and primer annealing and elongation at 53 °C for 20 s; (3) storage at 4 °C.
The following oligonucleotide primers were used: for microRNA-200a: reverse transcription primer 200a-RT, 5′-GTCGTGTCTGAGGCTCACTGAGACCTATTCGCACCTCGACACGACACATCGTT-3′; forward primer 200a-Fw, 5′-CCAGCTAACACTGTCTGGT-3′; reverse primer UR-3, 5′-CTGAGGCTCACTGAGACCT-3′; for microRNA-145: reverse transcription primer 145-RT, 5′-GTCGTGTCTGAGGCTCACTGAGACCTATTCGCACCTGACACGACAGGGATTC-3′; forward primer 145-FW, 5′-CCACAGTCCAGTTTTCCCAG-3′; reverse primer UR-3 (see above); for U6 snRNA: reverse transcription primer U6-RT, 5′-GTCGTGTCTGAGGCTGACTGAGACCTATTCGCACCTGACACGACGGCCATGC-3′; forward primer U6-Fw, 5′-GGCCGCATACAGAGAAGATTA-3′; reverse primer U6-Rv, 5′-CTGAGGCTGACTGAGACCT-3′.
The following fluorescently labelled probes were used for transcript detection: for microRNA-200a: 200a-Pb, 5′-(R6G)-ATTCGCACC(T-BHQ1)CGACACGACACATCGTT-p-3′; for microRNA-145: Pb-145, 5′-(R6G)-ATTCGCACC(T-BHQ1)GACACGACAGGGATTC-p-3′; for U6 snRNA: U6-Pb, 5′-(R6G)-ATTCGCACC(T-BHQ1)GACACGACGGCCATGC-p-3′.
4.7. Detection of FGFR3 Gene Hotspot Mutations
We detected mutations in codons 248 and 249. For this, we PCR-amplified the 7th exon of FGFR3 using 100 ng of genomic DNA, qPCRmix-HS (Evrogen, Moscow, Russia), and 10 pm of the specific primer oligonucleotides (FGFR3-7F, 5′-AGTGGCGGTGGTGGTGAGGGAG-3′; FGFR3-7R, 5′-ACCTTGAGCACGGTAACGTAGGGTGT-3′) by the following protocol: (1) DNA denaturation at 95 °C for 3 min; (2) 32 cycles of the following: DNA denaturation at 95 °C for 20 s, primer annealing at 70 °C for 20 s, and elongation at 72 °C for 20 s; (3) storage at 4 °C. PCR products were purified by agarose electrophoresis and the column purification kit Cleanup Standard (Evrogen, Moscow, Russia). Purified PCR products were Sanger-sequenced at Evrogen (Moscow, Russia) using (separately) both FGFR3-7F and FGFR3-7R primers. Sequencing results were analyzed using Chromas 2.6.6 software (“Technelysium”, Australia). We used non-modified oligonucleotide primers purchased at Evrogen (Moscow, Russia) and fluorescently labeled oligonucleotide probes purchased at DNA-Synthesis (Russia).
4.8. Immunohistochemistry (IHC)
We used an immunohistochemical method for two main purposes. First of all, we determined GATA3 and KRT5/6 expression in tumor slices for triage and the identification of a luminal subtype of primary NMIBC, while the valuation of PD-L1 expression level was the other goal of IHC testing (Figure 1). For this, 4-µm-thick sections of tumor tissue were used. Tissue slices were fixed in 10% formalin, and then were dehydrated and embedded in paraffin using an automated regimen of histological processing on the Spin vacuum tissue processor STP250-V (“Histo-Line Laboratories Srl”, Milan, Italy).
To analyze cell expression of the transcription factor encoded by GATA3, we used a monoclonal antibody against human GATA3 (HG3-31 clone, dilution, 1:100; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). The expression level of cytokeratin 5 and 6 (CR5/6) was assessed in tumor sections stained by a monoclonal antibody against human CR5/6 (D5/16B4 clone, 1:50 dilution, Dako, Denmark). Luminal molecular subtype of urothelial carcinoma was determined in accordance with Wang et al. [31] and Lerner et al. [32] as a >80% cut-off for GATA3 positive nuclear staining along with low or undetectable CR 5/6 cellular cytoplasm staining.
We used the Ventana PD-L1 Assay (SP263) and the OptiView DAB IHC Detection Kit (Cat. No. 760-700/06396500001) with signal amplification (Ventana Medical Systems, Inc., Tucson, AZ, USA) to evaluate the PD-L1 expression level in urothelial carcinoma sections. Negative control was stained by primary Ventana Rabbit Monoclonal Negative Control antibody (Cat. No. 790-4795/06683380001). In each reaction cycle, tonsils tissue specimens served as a positive control. To set up a reaction with the SP263 assay, after the dewaxing and unmasking antigens, rabbit monoclonal antibody PD-L1 (SP263) in working dilution was applied to prepared tumor sections. We used the Ventana BenchMark ULTRA ICH stainer (Ventana Medical Systems Inc., Tucson, AZ, USA) in accordance with the manufacturer’s guidelines for the reaction automated processing [33]. We scored the percentage of PD-L1-reacted cells in five randomly PC-selected high-power fields under light microscope with 500-folds magnification. High PD-L1 status was classified if ≥25% of tumor cells (TC) and/or ≥25% of immune cells (if they represented more than 1% of total cell population) displayed membrane PD-L1 positivity. Cellular membrane staining positivity scored between 0 and 25% for aforementioned cell populations was classified as Low PD-L1+ status. Absence of membrane positive staining appropriated a negative PD-L1 status.
4.9. Statistical Analysis
Data were processed and handled using the SPSS statistical software program, version 22.0 (SPSS, Inc., Chicago, IL, USA). Independent t test and analysis of variance (ANOVA) were used to compare the expression of PD-L1, miR-200a, miR-145, and FGFR3 between groups. The Mann–Whitney U test was used to compare the FGFR3 gene mutations between groups. The Kaplan–Meier method was used to estimate the time to first relapse and relapse-free survival, and differences were assessed using log-rank statistics. The prognostic values of the tumor grade, PD-L1, miR-200a, miR-145, and FGFR3 expressions were analyzed by univariable Cox proportional hazard regression models.
5. Conclusions
Tumor grade in association with PD-L1 and FGFR3 expression can be considered a complex predictor for primary luminal NMIBC progression.
Acknowledgments
We are extremely grateful to all colleagues, hospital officers, laboratory staff for useful advices, assistance and cooperation. We thank our patients for sincere desire to make medicine better.
Abbreviations
| BCG | Bacillus Calmette-Guerin |
| EORTC | European Organization for Research and Treatment of Cancer |
| FGFR3 | Fibroblast growth factor receptor 3 |
| GATA3 | Transcription factor encoded by GATA3 gene |
| IHC | Immunohistochemistry |
| CR5/6 | Cytokeratin 5 and 6 |
| MIBC | Muscular-invasive bladder cancer |
| miR | microRNA |
| NMIBC | Non-muscular invasive bladder cancer |
| PCR | Polymerase chain reaction |
| PD1 | Programmed death receptor 1 |
| PD-L1 | Programmed death receptor ligand 1 |
| RFS | Relapse-free survival |
| TUR | Transurethral resection |
Appendix A
Table A1.
Tumor-related characteristics of the study participants.
| Patient | Tumor Grade and Stage | Gender | Age, Years | Tumor Histology |
|---|---|---|---|---|
| 1 | T1, High grade | Male | 54 | UPC |
| 2 | T1, Low grade | Male | 57 | UPC |
| 3 | T1, High grade | Female | 61 | MC |
| 4 | T1, Low grade | Male | 60 | UPC |
| 5 | T1, Low grade | Female | 51 | UPC |
| 6 | T1, Low grade | Female | 64 | UPC |
| 7 | T1, High grade | Female | 67 | UPC |
| 8 | T1, High grade | Male | 59 | UPC |
| 9 | T1, Low grade | Female | 53 | UPC |
| 10 | T1, High grade | Male | 49 | MP |
| 11 | T1, Low grade | Male | 58 | UPC |
| 12 | T1, Low grade | Female | 50 | UPC |
| 13 | T1, Low grade | Male | 55 | UPC |
| 14 | T1, Low grade | Male | 48 | UPC |
| 15 | T1, Low grade | Male | 63 | UPC |
| 16 | T1, High grade | Female | 70 | UPC |
| 17 | T1, High grade | Male | 69 | UPC |
| 18 | T1, Low grade | Female | 47 | UPC |
| 19 | T1, Low grade | Female | 55 | UPC |
| 20 | T1, High grade | Male | 52 | UPC |
| Total | Low grade (12), High grade (8) | Male (11), Female (9) | Average (Mean ± SD) 57.1 ± 2.8 | UPC (18) MC (2) |
UPC—urothelial papillary carcinoma; MC—micropapillary carcinoma.
Appendix B
Figure A1.
FGFR3 gene hotspot mutations in codons 248 and 249: wild type allele is represented by AGCGCTCC nucleotide sequence; mutant genotypes had substitutions C -> T in position 3 or C -> G in position 7.
Author Contributions
Conceptualization, D.B., D.R., A.B., P.B. and E.B.; methodology, T.D., V.G., S.S., E.S. (Elena Samyshina), A.B., O.T., P.Z. and E.S. (Evgenia Shich); software, H.B., A.K. and O.D.; validation, T.D., D.P., O.T., V.G., A.D. and D.R.; formal analysis, H.B., D.M., O.D., O.T. and E.S. (Elena Samyshina); investigation, D.B., D.M., A.B., D.P., N.P., M.S., A.D., A.K., P.Z. and D.E.; resources, E.S. (Elena Samyshina), T.D., V.G. and S.S.; data curation, E.B., E.S. (Evgenia Shich), N.P., P.B., V.G. and D.E.; writing—D.R., A.K., O.D., N.P., O.T., T.D., H.B., M.S., A.D., P.Z. and E.B.; writing—review and editing, D.B., A.B., P.B. and D.E.; visualization, T.D., V.G., M.S. and D.M.; supervision, E.B.; project administration, D.B. and E.S. (Evgenia Shich); funding acquisition, E.B., A.B. and D.E. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the Russian academic excellence project “5–100” for Sechenov First Moscow State Medical University.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Murphy C.R., Karnes R.J. Bladder Cancer in Males: A Comprehensive Review of Urothelial Carcinoma of the Bladder. J. Men’s Health. 2014;11:18–27. doi: 10.1089/jomh.2014.3503. [DOI] [Google Scholar]
- 2.Botteman M.F., Pashos C.L., Redaelli A., Laskin B.L., Hauser R.A. The health economics of bladder cancer: A comprehensive review of published literature. PharmacoEconomics. 2003;21:1315–1330. doi: 10.1007/bf03262330. [DOI] [PubMed] [Google Scholar]
- 3.Kamat A.M., Hahn N.M., Efstathiou A.J., Lerner S.P., Malmström P.-U., Choi W., Guo C.C., Lotan Y., Kassouf W. Bladder cancer. Lancet. 2016;388:2796–2810. doi: 10.1016/s0140-6736(16)30512-8. [DOI] [PubMed] [Google Scholar]
- 4.Hurst C.D., Knowles M.A. Bladder cancer: Multi-omic profiling refines the molecular view. Nat. Rev. Clin. Oncol. 2017;15:203–204. doi: 10.1038/nrclinonc.2017.195. [DOI] [PubMed] [Google Scholar]
- 5.Eifler J.B., Scarpato K.R., Clark P.E. Management of noninvasive bladder cancers. Curr. Opin. Oncol. 2015;27:185–190. doi: 10.1097/cco.0000000000000173. [DOI] [PubMed] [Google Scholar]
- 6.Dadhania V., Zhang M., Zhang L., Bondaruk J., Majewski T., Siefker-Radtke A., Guo C.C., Dinney C., Cogdell D.E., Zhang S., et al. Meta-analysis of the luminal and basal subtypes of bladder cancer and the identification of signature immunohistochemical markers for clinical use. Oncotarget. 2016;12:105–117. doi: 10.1016/j.ebiom.2016.08.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Blank C., Mackensen A. Contribution of the PD-L1/PD-1 pathway to T-cell exhaustion: An update on implications for chronic infections and tumor evasion. Cancer Immunol. Immunother. 2007;56:739–745. doi: 10.1007/s00262-006-0272-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Massard C., Gordon M.S., Sharma S., Rafii S., Wainberg Z.A., Luke J., Curiel T.J., Colon-Otero G., Hamid O., Sanborn R.E., et al. Safety and Efficacy of Durvalumab (MEDI4736), an Anti–Programmed Cell Death Ligand-1 Immune Checkpoint Inhibitor, in Patients with Advanced Urothelial Bladder Cancer. J. Clin. Oncol. 2016;34:3119–3125. doi: 10.1200/jco.2016.67.9761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Patel S.P., Kurzrock R. PD-L1 Expression as a Predictive Biomarker in Cancer Immunotherapy. Mol. Cancer Ther. 2015;14:847–856. doi: 10.1158/1535-7163.mct-14-0983. [DOI] [PubMed] [Google Scholar]
- 10.Kawahara T., Ishiguro Y., Ohtake S., Kato I., Ito Y., Ito H., Makiyama K., Kondo K., Miyoshi Y., Yumura Y., et al. PD-1 and PD-L1 are more highly expressed in high-grade bladder cancer than in low-grade cases: PD-L1 might function as a mediator of stage progression in bladder cancer. BMC Urol. 2018;18:1–6. doi: 10.1186/s12894-018-0414-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Descamps-Dudez O., Louvet E., Bernard M., Auguste A., Gouy S., Scoazec J.Y., Genestie C., Adam J. Virchows Archiv. Volume 469. Springer; New York, NY, USA: 2016. Heterogeneity in PD-L1 expression and CD8+ infiltrates in low grade versus high grade serous ovarian carcinomas; p. S104. [Google Scholar]
- 12.Blinova E., Enikeev D., Roshchin D., Samyshina E., Deryabina O., Swidsinski A., Blinov D., Kogan E.A., Dudina M., Barakat H., et al. Relapse-Free Survival and PD-L1 Expression in First High- and Low-Grade Relapsed Luminal, Basal and Double-Negative P53-Mutant Non-Muscular Invasive Bladder Cancer Depending on Previous Chemo- and Immunotherapy. Cancers. 2020;12:1316. doi: 10.3390/cancers12051316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Huang T., Li B.-Q., Cai Y.-D. The Integrative Network of Gene Expression, MicroRNA, Methylation and Copy Number Variation in Colon and Rectal Cancer. Curr. Bioinform. 2016;11:59–65. doi: 10.2174/1574893611666151119215823. [DOI] [Google Scholar]
- 14.Liu G., Chen Z., Danilova I.G., Bolkov M.A., Tuzankina I.A., Liu G. Identification of miR-200c and miR141-Mediated lncRNA-mRNA Crosstalks in Muscle-Invasive Bladder Cancer Subtypes. Front. Genet. 2018;9:422. doi: 10.3389/fgene.2018.00422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fang Z., Dai W., Wang X., Chen W., Shen C., Ye G., Li L. Circulating miR-205: A promising biomarker for the detection and prognosis evaluation of bladder cancer. Tumor Biol. 2015;37:8075–8082. doi: 10.1007/s13277-015-4698-y. [DOI] [PubMed] [Google Scholar]
- 16.Yun S.J., Jeong P., Kim W.T., Kim T.H., Lee Y.-S., Song P.H., Choi Y.H., Kim I.Y., Moon S.-K., Kim W.-J. Cell-free microRNAs in urine as diagnostic and prognostic biomarkers of bladder cancer. Int. J. Oncol. 2012;41:1871–1878. doi: 10.3892/ijo.2012.1622. [DOI] [PubMed] [Google Scholar]
- 17.Borkowska E.M., Konecki T., Pietrusiński M., Borowiec M., Jabłonowski Z. MicroRNAs Which Can Prognosticate Aggressiveness of Bladder Cancer. Cancers. 2019;11:1551. doi: 10.3390/cancers11101551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee J.-Y., Ryu D.-S., Kim W.-J., Kim S.-J. Aberrantly expressed microRNAs in the context of bladder tumorigenesis. Investig. Clin. Urol. 2016;57:S52–S59. doi: 10.4111/icu.2016.57.s1.s52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kang H.W., Kim Y.-H., Jeong P., Park C., Kim W.T., Ryu D.H., Cha E.-J., Ha Y.-S., Kim T.-H., Kwon T.G., et al. Expression levels of FGFR3 as a prognostic marker for the progression of primary pT1 bladder cancer and its association with mutation status. Oncol. Lett. 2017;14:3817–3824. doi: 10.3892/ol.2017.6621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Neuzillet Y., Van Rhijn B.W.G., Prigoda N.L., Bapat B., Liu L., Boström P.J., Fleshner N.E., Gallie B., Zlotta A.R., Jewett M.A.S., et al. FGFR3 mutations, but not FGFR3 expression and FGFR3 copy-number variations, are associated with favourable non-muscle invasive bladder cancer. Virchows Archiv. 2014;465:207–213. doi: 10.1007/s00428-014-1596-4. [DOI] [PubMed] [Google Scholar]
- 21.Chen C., Ridzon D.A., Broomer A.J., Zhou Z., Lee D.H., Nguyen J.T., Barbisin M., Xu N.L., Mahuvakar V.R., Andersen M.R., et al. Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res. 2005;33:e179. doi: 10.1093/nar/gni178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Soria F., Krabbe L.-M., Todenhöfer T., Dobruch J., Mitra A.P., Inman B.A., Gust K.M., Lotan Y., Shariat S.F. Molecular markers in bladder cancer. World J. Urol. 2019;37:31–40. doi: 10.1007/s00345-018-2503-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mbeutcha A., Lucca I., Mathieu R., Lotan Y., Shariat S.F. Current Status of Urinary Biomarkers for Detection and Surveillance of Bladder Cancer. Urol. Clin. N. Am. 2016;43:47–62. doi: 10.1016/j.ucl.2015.08.005. [DOI] [PubMed] [Google Scholar]
- 24.Tabayoyong W., Kamat A.M. Current Use and Promise of Urinary Markers for Urothelial Cancer. Curr. Urol. Rep. 2018;19:96. doi: 10.1007/s11934-018-0857-1. [DOI] [PubMed] [Google Scholar]
- 25.Blinova E., Roshchin D., Kogan E.A., Samyshina E., Demura T., Deryabina O., Suslova I., Blinov D., Zhdanov P., Osmanov U., et al. Patient-Derived Non-Muscular Invasive Bladder Cancer Xenografts of Main Molecular Subtypes of the Tumor for Anti-Pd-l1 Treatment Assessment. Cells. 2019;8:526. doi: 10.3390/cells8060526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pashaei E., Guzel E., Ozgurses M.E., Demirel G., Aydin N., Ozen M. A Meta-Analysis: Identification of Common Mir-145 Target Genes that have Similar Behavior in Different GEO Datasets. PLoS ONE. 2016;11:e0161491. doi: 10.1371/journal.pone.0161491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhu J., Li Y., Luo Y., Xu J., Liufu H., Tian Z., Huang C., Li J., Huang C. A Feedback Loop Formed by ATG7/Autophagy, FOXO3a/miR-145 and PD-L1 Regulates Stem-Like Properties and Invasion in Human Bladder Cancer. Cancers. 2019;11:349. doi: 10.3390/cancers11030349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sylvester R.J., Van Der Meijden A.P.M., Oosterlinck W., Witjes J.A., Bouffioux C., Denis L., Newling D.W.W., Kurth K. Predicting Recurrence and Progression in Individual Patients with Stage Ta T1 Bladder Cancer Using EORTC Risk Tables: A Combined Analysis of 2596 Patients from Seven EORTC Trials. Eur. Urol. 2006;49:466–477. doi: 10.1016/j.eururo.2005.12.031. [DOI] [PubMed] [Google Scholar]
- 29.Soukup V., Čapoun O., Cohen D., Hernández V., Babjuk M., Burger M., Compérat E., Gontero P., Lam T., MacLennan S., et al. Prognostic Performance and Reproducibility of the 1973 and 2004/2016 World Health Organization Grading Classification Systems in Non–muscle-invasive Bladder Cancer: A European Association of Urology Non-muscle Invasive Bladder Cancer Guidelines Panel Systematic Review. Eur. Urol. 2017;72:801–813. doi: 10.1016/j.eururo.2017.04.015. [DOI] [PubMed] [Google Scholar]
- 30.Baust J.M., Snyder K.K., Van Buskirk R. Integrating Molecular Control to Improve Cryopreservation Outcome. Biopreserv. Biobank. 2017;15:134–141. doi: 10.1089/bio.2016.0119. [DOI] [PubMed] [Google Scholar]
- 31.Wang C.C., Tsai Y.C., Jeng Y.M. Biological significance of GATA3, cytokeratin 20, cytokeratin 5/6 and p53 expression in muscle-invasive bladder cancer. PLoS ONE. 2019;14:e0221785. doi: 10.1371/journal.pone.0221785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lerner S.P., McConkey D.J., Hoadley K.A., Chan K.S., Kim W.Y., Radvanyi F., Höglund M., Real F.X. Bladder Cancer Molecular Taxonomy: Summary from a Consensus Meeting. Bladder Cancer. 2016;2:37–47. doi: 10.3233/blc-150037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ventana PD-L1 (SP263) Assay Staining in Urothelial Carcinoma. Interpretation Guide. [(accessed on 26 December 2019)]; Available online: https://www.accessdata.fda.gov/cdrh_docs/pdf16/p160046c.pdf.