Abstract
Introduction
Antiplatelet therapy for neonates and infants is often extrapolated from the adult experience, based on limited observation of agonist-induced neonatal platelet hypoactivity and poor understanding of flow shear-mediated platelet activation. Therefore, thrombotic events due to device-associated disturbed flow are inadequately mitigated in critically ill neonates with indwelling umbilical catheters and infants receiving cardiovascular implants.
Methods
Whole blood (WB), platelet-rich plasma (PRP), and gel-filtered platelets (GFP) were prepared from umbilical cord and adult blood, and exposed to biochemical agonists or pathological shear stress of 70 dyne/cm2. We evaluated α-granule release, phosphatidylserine (PS) scrambling, and procoagulant response using P-selectin expression, Annexin V binding, and thrombin generation (PAS), respectively. Activation modulation due to depletion of intracellular and extracellular calcium, requisite second messengers, was also examined.
Results
Similar P-selectin expression was observed for sheared adult and cord platelets, with concordant inhibition due to intracellular and extracellular calcium depletion. Sheared cord platelet Annexin V binding and PAS activity was similar to adult values in GFP, but lower in PRP and WB. Annexin V on sheared cord platelets was calcium-independent, with PAS slightly reduced by intracellular calcium depletion.
Conclusions
Increased PS activity on purified sheared cord platelets suggest that their intrinsic function under pathological flow conditions is suppressed by cell-cell or plasmatic components. Although secretory functions of adult and cord platelets retain comparable calcium-dependence, PS exposure in sheared cord platelets is uniquely calcium-independent and distinct from adults. Identification of calcium-regulated developmental disparities in shear-mediated platelet function may provide novel targets for age-specific antiplatelet therapy.
Keywords: Phosphatidylserine, α-Granule release, Umbilical cord platelets, Thrombosis, Shear stress
Introduction
Thrombosis is one of the primary complications associated with pathological flow conditions prevalent in cardiovascular diseases and their associated therapeutic devices.11,27,61 Platelets, along with von Willebrand factor, play a key role in fluid shear-induced thrombosis,10,31,49,50 which is mitigated by antiplatelet and anticoagulation therapy.14,36 Recent studies have shown that aspirin, a well-established antiplatelet therapy, has low efficacy as primary prophylaxis35 and improved outcomes for secondary prevention of cardiovascular disease.1 However, patient age as a factor is often overlooked in pharmacotherapy, with treatment often extrapolated from regimens developed using platelet research on young- to- middle-aged subjects.29 Thrombosis events in neonates and infants are typically uncommon and are associated with umbilical arterial and venous catheters in newborns admitted to neonatal intensive care units (NICU),71 as well as infants implanted with ventricular assist devices, prosthetic heart valves, and systemic-to-pulmonary shunt implants.38 Antiplatelet dosing in neonatal and pediatric patients is extrapolated from adult populations, with limited and sometimes paradoxical data on their efficacy.66
Neonatal platelets are a unique subpopulation of pediatric platelets that have traditionally been observed to be hypoactive.62 Compared to adult platelets, neonatal platelets have impaired calcium mobilization,18 agonist-induced aggregation,25 and surface receptor expression,25,60 but similar agonist-induced thrombin generation.4,13,39 Under flow conditions, adhesion to von Willebrand factor (vWF) and collagen is similar,3,33 likely due to increased numbers of large multimers and shear-induced activity of neonatal vWF.45 This may explain shorter whole blood (WB) closure times observed in the Platelet Function Analyzer (PFA-100).48 Our recent observations suggest that cord platelets exposed to an array of constant shear stresses generate thrombin at similar rates compared to their adult counterparts, albeit at a reduced maximum thrombin potential.40 This may be compensated by an ADP-mediated positive feedback loop via enhanced P2Y1/P2Y12-mediated dense granule trafficking in neonatal platelets.40 However, the mechanisms underlying the discrepancy in activation between shear-exposed adult and neonatal platelets remain unclear.
We hypothesized that the medium in which platelets are suspended plays a significant role in shear-induced platelet activation, with isolated platelets providing the optimal medium for comparing adult and neonatal platelet activation. Using umbilical cord blood-derived platelets as a proxy for neonatal platelets, we evaluated agonist- and shear-induced platelet activation using flow cytometry markers in WB, platelet-rich plasma (PRP), and isolated platelets to determine the role of their surrounding medium. Platelets were exposed to a constant shear stress of 70 dyne/cm2, which significantly increases thrombin generation40 but is below levels associated with significant platelet lysis.23,31 This pathological shear stress may be observed due to disturbed blood flow resulting from the presence of catheters and blood-recirculating devices, but is uncommon in the healthy vasculature.31 We observed a paradox to the neonatal hypoactivity narrative, specifically the comparatively enhanced membrane phosphatidylserine (PS) activity of sheared isolated cord platelets, which was not present in WB or PRP as previously investigated.40 As PS scrambling requires the continuous presence of cytoplasmic calcium (Ca2+),73 we then tested the hypothesis that intracellular and extracellular Ca2+ depletion inhibits cord platelet activation due to shear stress. An expanded understanding of neonatal platelet function, particularly under realistic flow conditions, may help to improve antiplatelet therapy for neonatal patients facing thrombotic complications and can assist in identifying novel targets for age-specific pharmacotherapy.
Materials and Methods
Reagents
All reagents were from Sigma-Aldrich, unless otherwise stated. Flow cytometry antibodies for P-selectin (CD62P-PE), PS (Annexin V-FITC), and GPIIb (CD41a-APC) staining were obtained from BD Biosciences (Franklin Lakes, NJ).
Blood Collection and Preparation
Umbilical cord blood samples were obtained immediately after clamping from mothers who underwent scheduled term elective repeat Caesarean deliveries and were mixed with 10% citrate phosphate dextrose (CPD). Samples were processed within 30 min of receipt, and care was taken to minimize activation during transport. Whole blood (WB), 30 ml, was collected from healthy adult donors of both genders via antecubital venipuncture and immediately mixed with 10% acid-citrate dextrose (ACD-A). Exclusion criteria included aspirin or ibuprofen use 2 weeks prior to donation. Consent was obtained for both collections from the mother and adult donor, respectively, in accordance with Stony Brook University IRB-approved protocols (CORIHS# 2016-5886-R1 and CORIHS# 2012-4427-FAR).
After the collection of WB samples, three suspensions were prepared: WB, PRP, and gel-filtered platelets (GFP). PRP was prepared by centrifuging cord blood for 4.5 min at 225×g and adult blood for 4.5 min at 650×g, respectively, with these centrifugation parameters providing optimal PRP yield. GFP samples were prepared by filtering PRP through a column of Sepharose 2B beads into HEPES-buffered modified Tyrode’s solution with 1 mM sodium citrate and 0.1% bovine serum albumin (“platelet buffer”), as previously described.55,58 GFP and PRP were diluted to a final platelet count of 20,000/µl in platelet buffer, whereas WB was diluted in platelet buffer in a similar proportion to that of the diluted PRP. This platelet count allows measurement of platelet activation parameters directly due to shear stress exposure without non-linear effects resulting from platelet-platelet crosstalk.55 Flow cytometry and thrombin generation assays were optimized for this diluted platelet concentration. All suspensions were mixed with 3 mM CaCl2 10 min prior to experiments. All experiments were completed within six hours of receiving blood samples.
Intracellular and Extracellular Ca2+ Treatment
Intracellular Ca2+-depleted (–) platelets were prepared by treating GFP with 20 μM 1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid (BAPTA-AM, Sigma-Aldrich, Indianapolis, IN) for 30 min at 37 °C on a low-speed orbital shaker, then filtered through a second column of Sepharose 2B beads to remove solution-phase BAPTA. BAPTA-treated platelets were not recalcified prior to experiments. Extracellular calcium-depleted (–) platelets were prepared by not recalcifying GFP prior to experiments. Both sets of platelets were diluted to a final platelet count of 20,000/µL in platelet buffer. Non-BAPTA-treated, recalcified platelets were used as internal controls.
Biochemical Agonist Treatment
Platelet activation under static conditions was stimulated for 5 min at room temperature with three biochemical agonists: calcium ionophore (A23187), thrombin receptor activator peptide 6 (TRAP-6), and thrombin. A23187, which transports calcium ions across platelet membranes and triggers divalent cation release from intracellular storage sites, was prepared in DMSO and added to samples at a final concentration of 10 μM. TRAP-6, a synthetic platelet PAR1 agonist, was dissolved in 0.9% NaCl and added at a final concentration of 20 μM. Thrombin, a PAR1 and PAR4 agonist, was prepared in tris-buffered saline (TBS) with 50% glycerol and added at a final concentration of 1 U/mL.
Exposure of Platelets to High Shear Conditions
GFP, PRP and WB preparations were exposed to 7 Pa (70 dyne/cm2) for 4 min in a computer-controlled hemodynamic shearing device (HSD).72 The HSD is a cone-plate-Couette viscometer that applies uniform shear stress to blood suspensions in contact with its ultra-high-molecular-weight polyethylene (UHMWPE) surfaces,19,41 which are additionally pre-coated with Sigmacote 10 min prior to experiments. All suspensions, when diluted to a platelet count of 20,000/µL, had an approximate viscosity of 0.001 Pa s (1 cP), and were exposed to a shear rate of 7000 s−1. Diluted WB and platelet buffer viscosities were confirmed to be 1.018 ± 0.030 cP and 0.969 ± 0.020 cP, respectively, as measured with a Canon-Fenske viscometer (n = 2). For flow cytometry measurements, 25 µl samples from sheared GFP, PRP and WB suspensions were collected prior to and at the end of exposure using a LabView-controlled syringe pump (PSD/8, Hamilton, Reno, NV) connected to the HSD via a 28-gauge PTFE tube,59 and placed on ice. For thrombin generation measurement, only GFP samples were collected in a similar manner and processed using the Platelet Activation State (PAS) assay.
Platelet Activation State (PAS) Assay
Thrombin generation for timed sheared GFP samples was measured using a modified prothrombinase-based assay.28 PAS absorbance readings were normalized against quiescent GFP that had been sonicated at 10 W for 10 s with a Branson Sonifier 150 with a microprobe (Branson, MO).58 Normalized PAS values represent the PAS as a fraction of the full activation. The change in platelet activation state, ΔPAS, was calculated over the 4 min duration of shear stress exposure.
Flow Cytometry Measurements
Agonist-treated and sheared WB, PRP, and GFP suspensions were assessed for platelet activation using anti-CD62P (PE) and Annexin V (FITC), expanded from our previous investigations.40 The platelet gate was identified as a distinct cell population within the forward vs. side scatter (FSC vs. SSC) plots that exhibited > 95% positivity for anti-CD41a (APC) measurements using untreated and unsheared control platelets. Cells within this defined gate were further analyzed for CD62P and Annexin V activity, defined as percent positive events in response to agonist and shear treatments. Agonist-treated and sheared samples were diluted 1:20 with platelet buffer and 1:10 with FcR blocker, and stained with 20 μL anti-CD62P and anti-CD41a for 15 min in the dark at 4 °C. Separately, samples were diluted 1:20 with Annexin V buffer (BD Biosciences, Franklin Lakes, NJ) and stained with 5 μL Annexin V for 15 min in the dark at room temperature. Samples were immediately supplemented with platelet buffer or Annexin V buffer to a final volume of 500 µL at 4 °C, scanned using FACS (BD Accuri C6, BD Biosciences, Franklin Lakes, NJ), and analyzed with Kaluza 2.0 (Beckman Coulter, Indianapolis, IN). Flow cytometry measurements were compensated for fluorescence overlap by using dyes in non-adjacent flow channels. Doublet exclusion analysis, which calculates the ratio of FSC-A and FSC-H for all cells within the platelet gate, were performed for all shear experiments. A value of 100% indicates all events are single cells and no aggregate formation is present.
Statistical Analysis
Shapiro-Wilk tests of normality were performed for all data groups prior to statistical analyses to select the appropriate parametric or non-parametric test. The hypotheses that different preparations (WB, PRP, and GFP) and calcium treatments affect flow cytometric parameters were compared by agonist or calcium treatment using Kruskal-Wallis ANOVA with Bonferroni correction. Comparison of flow cytometry events, percentage change in sheared platelet flow cytometry markers, and normalized change in PAS between the age groups were performed with Mann–Whitney U test. For intra-age group comparisons, unsheared samples served as the negative control, while adult platelets served as the control for adult-cord platelet comparisons. For calcium treatments, the recalcified and non-BAPTA treated platelet samples were the controls. For all comparisons, p < 0.05 was used to establish significance. Data are presented as means ± standard error of the means. All statistical tests were performed on IBM SPSS Statistics 26 (Armonk, NY).
Results
Subject Demographics
Two sets of healthy adult blood donors participated in this study, which examined the effect of (1) different platelet preparations on shear and biochemical-agonist induced platelet activation and (2) intracellular and extracellular calcium depletion on platelet activation. The first set included six males and six females with a mean age of 37.17 ± 13.04 years (range 27–65). The second set included three males and three females with a mean age of 30.67 ± 11.52 years (range 22–53). No gender information was collected for the two sets of umbilical cord blood samples, with 13 subjects in the first group and six subjects in the second group.
Platelet Surface Activation in GFP, PRP, and WB
Differences in surface receptor activity to agonist-treated adult and cord platelets have not been directly and systematically compared previously in the context of platelet preparation. Scatter analysis (Table 1) indicates that FSC for cord platelets is 15.98–17.27% lower than for adult platelets (p < 0.01), with SSC 5.53–9.16% lower for cord platelets (p > 0.05). This indicates a smaller size but similar complexity for cord platelets. We observed that cord platelet populations exposed to 70 dyne/cm2 for 4 min (“high shear”) do not show significant changes in size and complexity characteristics, as measured by flow cytometry, independent of their preparation (Figs. 1a–1c). Sheared adult platelets show similar FSC and SSC characteristics as cord platelets, except for PRP, which has a noticeable reduction in events within the platelet gate (Figs. 1d–1f). We monitored two platelet surface activity indicators, P-selectin (CD62P) expression and Annexin V binding to PS, in response to several biochemical agonists, as well as 4 min exposure to high shear, in GFP, PRP, and WB samples. Platelet activity was defined as the percentage of platelets staining positive for the antibody. Comparisons between sheared adult and cord platelet activation markers, as well as sample sizes for these populations, are summarized in Table 2.
Table 1.
Forward (FSC) and side scatter (SSC) characteristics of untreated adult and cord platelets.
| FSC | SSC | |||||
|---|---|---|---|---|---|---|
| Adult | Cord | p-value | Adult | Cord | p-value | |
| GFP | 110,743.93 ± 5491.45 | 87,814.91 ± 2523.02 | 0.005 | 9112.00 ± 425.99 | 8157.52 ± 175.94 | 0.077 |
| PRP | 113,799.17 ± 6104.11 | 91,615.03 ± 2896.32 | 0.007 | 9143.78 ± 453.29 | 8275.83 ± 151.02 | 0.269 |
| WB | 118,904.58 ± 5121.43 | 99,905.93 ± 2853.58 | 0.003 | 9050.01 ± 172.51 | 8549.90 ± 191.18 | 0.103 |
Significant p-values are given in bold
Sample sizes are n = 12 for adult and n=13 for cord samples. Data is represented as means ± s.e.m
Figure 1.
Flow cytometric analysis of sheared cord and adult platelets. Representative scatter plots of cord (a–c) and adult (d–f) platelets in GFP, PRP, and WB exposed to 70 dyne/cm2 for 4 min. Gates represent platelets positively staining for CD41a in the unsheared cord and adult platelet population in each respective preparation. Axes represent forward scatter (FSC-A) and side scatter (SSC-A) data.
Table 2.
Effect of blood preparation on shear-induced platelet activation (70 dyne/cm2).
| Activation Marker | Adult value (%) | Cord value (%) | p-value |
|---|---|---|---|
| ΔCD41a | |||
| GFP | − 3.30 ± 1.17 (11) | − 10.29 ± 3.22 (13) | 0.046 |
| PRP | − 56.70 ± 4.69 (11) | − 3.33 ± 0.69 (11) | 0.000 |
| WB | − 23.83 ± 7.03 (10) | − 10.43 ± 2.05 (11) | 0.409 |
| ΔAnnexin V | |||
| GFP | 15.95 ± 3.58 (11) | 23.75 ± 4.13 (11) | 0.200 |
| PRP | 56.62 ± 9.31 (10) | 12.84 ± 3.27 (10) | 0.004 |
| WB | 24.01 ± 7.35 (9) | 11.35 ± 3.70 (11) | 0.141 |
| ΔCD62P | |||
| GFP | 9.63 ± 2.39 (12) | 7.06 ± 1.31 (11) | 0.355 |
| PRP | 10.03 ± 4.64 (10) | 5.05 ± 1.50 (10) | 0.916 |
| WB | 7.72 ± 3.92 (10) | 6.43 ± 1.68 (11) | 0.499 |
| ΔPAS | |||
| GFP | 7.77 ± 1.31 (7) | 6.38 ± 1.66 (6) | 0.534 |
Significant p-values are given in bold
Parentheses represent sample size of each group. Data is represented as means ± s.e.m
GPIIb Expression in Sheared Platelets is Age-Dependent
GPIIb, an integrin constituent of the GPIIb/IIIa complex, was identified with anti-CD41a, and its expression on non-agonist-treated platelets was used to define the platelet population gate for flow cytometry analyses. CD41a expression was reduced for sheared adult and cord platelets in all three preparations compared to their respective untreated controls (Figs. 2a–2c). CD41a expression on shear-treated adult platelets was 56.7 and 23.8% lower than the untreated control in PRP (p < 0.001) and WB (p < 0.01), respectively (Figs. 2b–2c), while only a 3.3% reduction was observed in GFP (p < 0.05). The largest decline in CD41a for cord platelets, 10.4%, was observed in WB (p < 0.01, Fig. 2c). There were significant age-specific differences between the CD41a decline in GFP and PRP (p < 0.05 and p < 0.001, respectively; Fig. 2d). Doublet exclusion analysis in sheared PRP indicates that 96.12 ± 1.47% of adult and 98.92 ± 0.72% of cord events in the pre-defined platelet gate are single cells, with virtually negligible platelet-platelet aggregation. The sharp decline in CD41a on sheared adult platelets in PRP and WB is potentially due to GPIIb/IIIa shedding or other loss of the CD41a epitope. The lack of CD45 activity (not shown) eliminates the possibility of leukocyte-platelet aggregation.
Figure 2.
Integrin αIIb expression in agonist-treated adult and cord GFP, PRP, and WB. Binding to the α chain of integrin αIIb (CD41a) was used to gate adult (n = 11) and cord (n = 12) platelets in (a) GFP, (b) PRP, and (c) WB after treatment with biochemical agonists and 4 min exposure to 70 dyne/cm2. Percentage reduction in CD41a-positive platelets due to shear exposure is summarized in (d). Percentage of CD41a-positive platelets was compared to the untreated control (#p < 0.05, ##p < 0.01, ###p < 0.001) or related adult group (*p < 0.05, ***p < 0.001).
Biochemical agonist-induced changes in CD41a were observed only after 10 μM ionophore and 1 U/mL thrombin treatment. In GFP, ionophore treatment yielded a small decrease in CD41a activity for both adult and cord platelets compared to untreated samples (p < 0.001), but no difference between the two age groups (Fig. 2a). In PRP, the decrease in CD41a expression was similar for both ionophore-treated adult and cord platelets (p < 0.01 and p < 0.001, respectively; Fig. 2b). In WB, both adult and cord platelets showed significantly reduced CD41a expression in response to 10 μM ionophore and 1 U/mL thrombin (p < 0.001 and p < 0.01, respectively), with no differences between the age groups (Fig. 2c).
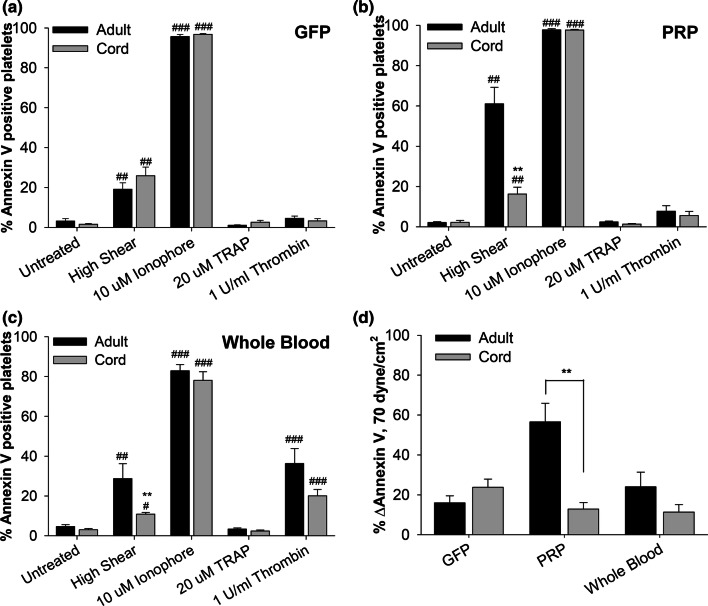
Platelet Preparation Affects Annexin V Binding on Sheared Cord Platelets
PS translocates to the outer platelet membrane upon activation and provides a negatively-charged surface for the formation of the prothrombinase complex, which results in the generation of the potent agonist thrombin. PS scrambling was detected using Annexin V binding. Shear exposure in PRP and WB increased Annexin V binding for both age groups (p < 0.05), but with 3.7-fold and 2.7-fold lower activity, respectively, for cord platelets compared to the adult cohort (p < 0.01, Figs. 3b and 3c). However, GFP exposed to high shear stress significantly increased the proportion of platelets positive for Annexin V (p < 0.01, Fig. 3a), with cord platelets yielding a similar increase in activity as adult platelets (Fig. 3d). This contrasts with the slightly lower normalized thrombin generation rate we observed for cord platelets in response to the shear stress magnitude used in this study.40 The discrepancy in age-specific Annexin V binding for sheared GFP as compared to PRP and WB may be due to modulation by extraplatelet constituents (i.e. leukocytes, red blood cells, plasma proteins, electrolytes), which is not present in the former. This difference prompted the subsequent exploration of the role of intracellular and extracellular Ca2+, which is involved in PS scrambling and α-granule release.
Figure 3.
PS activity on agonist-treated adult and cord GFP, PRP, and WB. Annexin V, a PS activity marker, was measured on adult (n = 9) and cord (n = 10) platelets in (a) GFP, (b) PRP, and (c) WB after treatment with biochemical agonists and 4 min exposure to 70 dyne/cm2. Percentage change in Annexin V-positive platelets due to shear exposure is summarized in (d). Percentage of Annexin V-positive platelets was compared to the untreated control (#p < 0.05, ##p < 0.01, ###p < 0.001) or related adult group (**p < 0.01).
In addition to high shear exposure, biochemical agonist treatment yielded differing responses in Annexin V binding dependent on age group. For all platelet preparations and both age groups, 10 μM ionophore treatment significantly increased the proportion of Annexin V-positive platelets (p < 0.001, Figs. 3a–3c). Treatment of WB with 1 U/mL thrombin significantly increased PS activity for both age cohorts (p < 0.001, Fig. 3c).
Age-Specific P-Selectin Expression is More Sensitive to TRAP Than Shear Stress
P-selectin, expressed on the platelet membrane after release from α-granules, was identified with anti-CD62P. High shear exposure had a minimal effect on CD62P expression in both adult and cord platelets in all preparations (Figs. 4a–4c), with no differences in CD62P increase observed due to age (Fig. 4d). However, platelets for all preparations were more responsive to biochemical agonist stimulation. Treatment with 10 μM ionophore, 20 µM TRAP, and 1 U/mL thrombin yielded significant increases for all preparations and age groups (p < 0.05), with adult CD62P expression generally similar to or slightly higher than that of cord platelets (p > 0.05, Figs. 4a–4c).
Figure 4.
P-selectin expression on agonist-treated adult and cord GFP, PRP, and WB. P-selectin (CD62P), a marker of α-granule release, was measured on adult (n=11) and cord (n = 11) platelets in (a) GFP, (b) PRP, and (c) WB after treatment with biochemical agonists and 4 min exposure to 70 dyne/cm2. Percentage change in CD62P-positive platelets due to shear exposure is summarized in (d). Percentage of CD62P-positive platelets was compared to the untreated control (#p < 0.05, ##p < 0.01, ###p < 0.001) or related adult group (*p < 0.05).
Shear-Mediated PS Scrambling in Cord Platelets is Calcium-Independent
The procoagulant function of platelets is dependent on flipping of negatively-charged PS to the outer membrane leaflet, thereby allowing assembly of coagulation complexes, which results in generation of the potent platelet agonist thrombin.6 This process is not only dependent on intracellular calcium mobilization, but also influx of extracellular calcium.7 Our observation of similar Annexin V expression in shear-activated adult and cord GFP (Fig. 3a), coupled with the paradoxical observation of impaired intracellular calcium mobilization in cord platelets,18 motivated examination of the role of intracellular and extracellular calcium in agonist-treated adult and cord GFP. Prior to agonist treatment, adult and cord GFP were assigned to calcium treatment groups: i) recalcification with 3 mM CaCl2 final (“Control” or “recalcified”); ii) intracellular calcium inhibition with 20 μM BAPTA-AM and no realcification (“–”); and iii) no recalcification (“–”). CD41a, Annexin V, and CD62P expression, and thrombin generation, was measured for all agonist and calcium treatments. Results for the shear treatment and sample sizes for both adult and cord platelet experiments are summarized in Table 3.
Table 3.
Effect of calcium treatment on shear-induced platelet activation (70 dyne/cm2).
| Activation Marker | Adult value (%) | Cord value (%) | p-value |
|---|---|---|---|
| ΔCD41a | |||
| Control | − 0.53 ± 0.16 (6) | − 1.65 ± 0.74 (6) | 0.132 |
| – | − 0.28 ± 0.10 (5) | − 1.10 ± 0.28 (5) | 0.056 |
| – | − 0.22 ± 0.15 (5) | − 1.30 ± 0.44 (6) | 0.017 |
| ΔAnnexin V | |||
| Control | 11.53 ± 4.99 (6) | 6.78 ± ± 1.55 (6) | 1.000 |
| – | 5.54 ± 2.08 (5) | 8.17 ± 1.73 (6) | 0.329 |
| – | 19.88 ± 6.95 (6) | 7.27 ± 1.86 (6) | 0.132 |
| ΔCD62P | |||
| Control | 6.98 ± 1.55 (6) | 3.70 ± 1.10 (6) | 0.180 |
| – | 0.33 ± 0.13 (4) | 0.67 ± 0.43 (6) | 0.762 |
| – | 5.97 ± 2.14 (6) | 2.25 ± 0.39 (6) | 0.132 |
| ΔPAS | |||
| Control | 12.52 ± 5.10 (6) | 14.75 ± 5.85 (6) | 0.589 |
| – | 2.29 ± 0.68 (6) | 7.88 ± 1.01 (6) | 0.002 |
| – | 1.77 ± 0.83 (4) | 2.85 ± 1.14 (5) | 0.556 |
Significant p-values are given in bold
Parentheses represent sample size of each group. Data is represented as means ± s.e.m
CD41a was used to identify platelet populations as described earlier. No differences were observed due to calcium treatment for both age groups and all agonists, except for 10 μM ionophore, which resulted in a 3.5 and 14.5% decrease in CD41a expression compared with untreated adult and cord platelets, respectively (data not shown). However, this decrease was abolished for – and – platelets exposed to ionophore. Changes in CD41a expression for sheared platelets were not significant compared to untreated platelets, but the decrease in CD41a on – cord platelets was significant compared to the adult cohort (p = 0.017, Table 3).
The effect of calcium treatment on Annexin V binding was agonist-dependent (Fig. 5a). Recalcification yielded slight increases in Annexin V binding after shear exposure, with 9.6-fold and 7.4-fold increases in adult and cord platelets, respectively, compared to their untreated controls (p > 0.05). While – slightly reduced Annexin V in sheared adult platelets (p > 0.05), this treatment had no effect on cord platelets (p > 0.05). Significantly higher Annexin V binding was observed for sheared – adult platelets compared to the resting – population (p < 0.05), but not its recalcified sheared control group. However, – treatment had no effect on sheared cord platelets. No differences were observed in Annexin V binding on sheared platelets due to age (Fig. 6b and Table 3). The change in percentage of cells within the platelet gate before and after shear was minimal for all three calcium treatments for both age cohorts of platelets, with a maximum reduction of 5.15 ± 2.59% for untreated sheared cord platelets.
Figure 5.
Role of calcium on surface expression of agonist-treated adult and cord GFP. The role of calcium on agonist-treated adult (n = 6) and cord (n = 6) GFP surface expression was investigated via the BAPTA-induced inhibition of intracellular stores (–) and lack of recalcification (–). Surface expression was measured using (a) Annexin V binding and (b) CD62P expression. Percentage positive events were compared to the non-agonist treated (“untreated”, †p < 0.05, ††p < 0.01, †††p < 0.001) or agonist-treated control (#p < 0.05, ##p < 0.01).
Figure 6.
Role of calcium on surface activity of sheared adult and cord GFP. The effect of BAPTA (–) and lack of recalcification (–) on sheared adult (n = 6) and cord (n = 6) GFP was compared using the change in (a) CD41a, (b) Annexin V, (c) CD62P, and (d) PAS over 4 min exposure to 70 dyne/cm2. These values were compared to the recalcified control (#p < 0.05, ##p < 0.01), or related adult group (*p < 0.05).
As expected, only 10 μM ionophore-exposed adult and cord platelets showed significant increases in Annexin V binding compared to untreated platelets among the biochemical agonists used (p < 0.01). In addition, only – ionophore- and TRAP-treated cord platelets showed a significant decrease in Annexin V binding compared to their recalcified controls (p < 0.05, Fig. 5a).
α-Granule Release in Sheared Adult and Cord Platelets is Intracellular Ca2+-Dependent
The effect of calcium treatment was more prominent on CD62P expression, a hallmark of α-granule release (Fig. 5b and Table 3). For sheared and agonist-treated adult and cord platelets, – treatment significantly reduced CD62P expression (p < 0.05), while – had generally little to no effect (p > 0.05). Both sheared adult and cord platelets yielded a significantly lower change in CD62P expression due to – (p < 0.05), but only sheared cord platelets had reduced CD62P expression due to – (p < 0.01, Fig. 6c). No age-related differences we observed due to Ca2+ treatment or agonist exposure (p > 0.05).
Thrombin Generation Potential on Sheared Cord Platelets is Extracellular Ca2+-Dependent
While flow cytometric markers are excellent at highlighting surface activity on individual platelets in response to agonist and calcium treatment, they do not indicate the thrombin-generating capacity of the platelets. Thrombin generation is also highly dependent on efficient PS scrambling, previously identified with Annexin V binding. We used the difference in normalized PAS (ΔPAS) to quantify the bulk thrombin generation of adult and cord platelets before and after shear exposure (Fig. 6d). Both – and – reduced ΔPAS for sheared adult and cord platelets, but this decrease was only significant for adult platelets (p < 0.05, Fig. 6d). Mean reduction in ΔPAS for sheared BAPTA-treated adult platelets was 81.71% compared to the sheared control, whereas this reduction was 46.58% for cord platelets, with a significant difference between the two age groups (p < 0.05). Reduction in ΔPAS due to – was similar—85.86 and 80.68% for adult and cord platelets, respectively—but the latter was not significant due to the small sample size and variability in the control (p > 0.05). While sheared – adult platelets showed slightly increased Annexin V binding (p > 0.05, Fig. 6b), ΔPAS was significantly reduced (p < 0.05, Fig. 6d), suggesting thrombin generation is heavily dependent on extracellular calcium availability despite the presence of a scrambled PS base for the prothrombinase complex. Sheared – cord platelets also showed a similar dependence of thrombin generation on extracellular calcium despite unchanged PS activity.
Discussion
While neonatal platelets have been shown to be hypoactive in response to a variety of biochemical agonists, their activity after mechanotransduction is poorly understood. The limited experiences with sheared neonatal platelets have primarily focused on collagen-, vWF-, or polystyrene-triggered adhesion and aggregation,3,33,45,57 or collagen-induced closure times,48,53 with increased adhesion and shorter closure times in cord and neonatal blood generally attributed to higher plasma multimeric vWF content. Neonatal and adult platelets have a similar adhesion affinity on both vWF and collagen under fluid shear stress.3 The effect of shear stress alone on neonatal platelets has only recently been studied.40 In the present study, cord platelets exposed to pathological magnitudes of constant flow shear stresses yielded normalized thrombin generation rates, as indicated with PAS, on par with adult platelets. This shear-induced response mirrors prior studies that indicate similar thrombin generating potential between washed adult and cord platelets after stimulation with calcium ionophore A23187 or thrombin.5 However, our normalization treatment, sonication, yields a cord platelet thrombin generation rate 62.5% that of adult platelets,40 which reflects the reduced peak thrombin generation rate46 and concentration5,17,46 found for tissue factor- or thrombin-stimulated platelets in cord plasma. Cord plasma has 44–50% FII, 30–50% FX, and 68–98% FV concentrations compared to adult plasma.42,46 It is important to note here that our thrombin generation assay relies on a modified prothrombinase system where acetylated prothrombin (Ac-FII), FXa, and CaCl2 were added after the shear treatment, and factor V and the anionic phospholipid surface is provided by the platelet.28 These prior observations, coupled with the lower thrombin generation on sonicated cord platelets, suggest either a reduced contribution of platelet factor V to shear-induced thrombin generation or a potential defect in α-granule release.
The α-granule in activated platelets releases P-selectin (CD62P), which is expressed on the outer platelet membrane leaflet. Both peripheral and cord platelets have shown significantly lower P-selectin activity compared to adult platelets after stimulation with the PAR-1 peptide TRAP in PRP9 and WB,60 although basal P-selectin expression was similar.60 These prior studies are confirmed by our observations, where similar reductions in P-selectin expression on cord platelets are observed after treatment with 20 μM TRAP and 1 U/mL thrombin in not only PRP and WB, but also purified platelets (GFP). This indicates an intrinsic cord platelet P-selectin hypoactivity or α-granule impairment. Morphological comparison between neonatal and adult platelets suggests no differences in the number of α-granules, but less granular centralization in TRAP-stimulated neonatal platelets.9 In our sheared GFP, PRP, and WB experiments, we observed similar basal P-selectin expression in cord platelets, but a slightly and non-significantly lower change in P-selectin expression after shear exposure, when compared with adult platelets. The difference in sheared platelet P-selectin expression, although not significant, suggests that a potential granular function impairment is not overcome by shear exposure. This observation is further cemented by a transcriptome analysis of cord platelets indicating that they underexpress genes required for granular fusion to the plasma membrane (STX11, Munc18b)9 and granule exocytosis (RTN1).8 Shear stress and thrombin are physiologically-relevant agonists, while TRAP mimics the PAR-1 antagonism of thrombin. However, using a synthetic agonist, 10 μM ionophore (A23187), yielded similar P-selectin expression on cord and adult platelets in GFP and PRP, whereas adult platelets appear to be slightly more responsive than cord platelets in WB. While prior observations suggest stunted WB neonatal P-selectin expression in response to calcium ionophore stimulation,3 our GFP and PRP observations indicate that a dramatic increase in cytosolic Ca2+ overcomes α-granule release impairment in cord platelets when red blood cells and white blood cells are absent.
Membrane activity, indicated by scrambling to expose anionic PS, is a critical component of thrombin generation. Exposure to constant shear stresses and stress gradients have been shown to promote binding of Annexin V, a marker of PS activity, on adult platelets.22,34,44,51 The distribution of phospholipids, particularly PS, in neonatal platelets has been shown to be similar to that of adults.5 No difference in PS activity was observed between ionophore- and thrombin-stimulated cord and adult platelets in PRP.4,5 However, we observed that shear-treated cord platelets yielded significantly lower Annexin V binding in PRP, and non-significantly lower Annexin V binding in WB. This discrepancy can be attributed to the dependence of PS scrambling on intracellular calcium mobilization.65 Calcium ionophore, which is non-physiologic, allows transport of Ca2+ across the platelet membrane and triggers release of intraplatelet Ca2+ stored in the dense granules.70 Intraplatelet Ca2+ mobilization triggered by collagen and thrombin is impaired in neonatal platelets,18 in part due to underexpression of genes required for calcium transport, metabolism, binding,8 and release.63 Our results suggest that a shear stress magnitude of 70 dyne/cm2 is able to overcome intraplatelet calcium mobilization in isolated platelets, but that yet undetermined extraplatelet constituents in PRP and WB suppress PS scrambling.
The role of intracellular and extracellular Ca2+ on GPIIb expression, α-granule release and PS in agonist-treated isolated platelets was examined through inhibition with BAPTA and lack of recalcification, respectively. Both BAPTA treatment and non-recalcification abolished the significant reduction in GPIIb expression observed on untreated cord platelets exposed to calcium ionophore, suggesting that intracellular calcium mobilization coupled with an abundance of extracellular Ca2+ is sufficient to cause a drop in GPIIb activity. It is unclear if this reduction in activity is indicative of GPIIb/IIIa receptor shedding, conformational change (i.e. inability to bind to the GPIIb antibody), or platelet lysis. Previous analysis of GPIIb/IIIa shedding has only examined the role of pathological shear stresses,12 which we observed occurred only in PRP, suggesting the role of an extraplatelet constituent in such behavior. Lack of intracellular or extracellular Ca2+ does not appear to affect GPIIb activity in isolated cord platelets treated with other biochemical agonists or high shear stress.
While GPIb43 and GPIIb/IIIa20 are traditionally considered the primary platelet mechanoreceptors, our experiments with isolated platelets without the presence of vWF suggest other mechanisms of shear-induced platelet activation.61 Recent investigations have indicated that mechanosensitive cation channel Piezo1, activated by tension in the lipid bilayer in the membrane, contributes to Ca2+ influx under shear stress.24 PS activity in platelet activation is dependent on intracellular Ca2+ mobilization.2 Adult platelet PS activity appears to be sensitive to the availability of intracellular Ca2+ under shear stress, with overcompensation if extracellular Ca2+ is diminished. This requires the involvement of the scramblase TMEM16F, which is dependent on intracellular Ca2+.65 Cord platelet PS activity was largely unaffected by BAPTA treatment, except after ionophore and TRAP treatment, suggesting that membrane scrambling in cord platelets due to calcium flux-induced and PAR-1 activation is sensitive to the availability of intracellular Ca2+. However, we observed that sheared cord platelet PS activity is independent of calcium availability, potentially explaining why thrombin generation is still potent4 despite an intracellular mobilization defect,18 and immature calcium and metabolism pathway (encoded by MCUR1).8 The Ca2+-independent PS activity in sheared cord platelets resembles intrinsic apoptosis-like behavior previously observed for adult platelets above a shear stress of 117 dyne/cm2,34 requiring BAX/BAK regulation, mitochondrial transmembrane potential depolarization, caspase-3 activation, and microparticle formation,54 as well as involvement of the scramblase Xkr8 which may operate independently of Ca2+.64 Furthermore, the slightly reduced and Ca2+-independent PS exposure in sheared cord platelets displays some elements of Scott syndrome, a rare hemorrhagic disorder characterized by impaired Ca2+-dependent phospholipid scrambling, diminished agonist-stimulated PS exposure, and a partial role of TMEM16F in apoptotic PS response.69 Our observation of minimal reduction in cells within the platelet gate after shear exposure suggests that platelets depleted of intracellular and extracellular Ca2+ are mostly intact and their integrity has not been compromised. Therefore, the distinction between activation and apoptosis of cord platelets exposed to pathological shear stress requires further investigation.
P-selectin expression, representing α-granule release, is heavily dependent on intracellular calcium availability in both adult and cord platelets for all biochemical agonists and high shear stress used in this study. A rise in intracellular Ca2+ concentration triggers α-granule secretion,30 aided by the binding of calmodulin.21 Granular contents, including P-selectin, are released into the open canalicular system or membrane after fusion.15 Cord platelets have been observed to have reduced levels of LYN, a protein that mediates release of intracellular Ca2+,63 but larger numbers of α-granules,52 compared to adult platelets. While the non-significantly lower P-selectin expression in agonist-treated cord platelets may be attributed to the previously discussed granular fusion or exocytosis defect,8,9 no age-dependent difference was observed in the role of intracellular Ca2+ on P-selectin expression.
There are a few limitations in our shear study. First, the PAS assay in this study utilizes isolated platelets due to the thrombin-sensitive chromogenic substrate used, as well as fixed adult-derived FII and FXa concentrations. As such, the results may not reflect the thrombin generation which may occur in PRP or WB. Furthermore, cord plasma concentrations of FII and FXa are lower than found in adult plasma, as discussed earlier, which may yield higher than expected thrombin generation. However, this reductionist approach allows us to observe the effects of shear on cord platelets independent of extraplatelet factors (i.e. coagulation factors and inhibitors), and the prothrombinase reaction is observed after the shear treatment. In addition, the use of acetylated prothrombin allows us to bypass the procoagulant effects of thrombin. Second, the PAS assay does not differentiate between procoagualant effects of platelets and their derived microparticles, which have an increased procoagulant activity in neonates.37,56 This distinction requires further study, as our flow cytometric analysis excluded microparticles. In addition, we focused on a single pathologic shear stress, 70 dyne/cm2, which we previously identified from several shear stress conditions as yielding a non-significant difference in normalized PAS between adult and cord platelets.40 We additionally observed that untreated cord platelets are smaller than adult platelets based on FSC characteristics (Table 1), which is different from prior comparisons indicating similar mean platelet volumes.25 However, our analysis considers the percentage, rather than intensities, of gated platelets positive for flow cytometry markers and is not affected by platelet size. Flow cytometric analyses used the CD41a antibody to confirm presence of platelets. Alternatively, anti-CD42b, which binds GPIbα, can also be used for positive platelet identification. While flow scatter analysis of sheared adult and cord platelets suggest no change in size, it is unclear whether shear compromises membrane integrity in a manner similar to pore formation with streptolysin O-permeabilization,16,32 potentially leading to Annexin V binding of phosphatidylserine in the internal platelet membrane leaflet. Recent observations of the lack of caspase-3 activation following decrease of mitochondrial membrane potential after adult platelet exposure to 70 dyne/cm2 suggest this hallmark of apoptosis is unlikely,47 but further experiments with calcein retention26 can confirm if shear induces pore formation in adult and cord platelets. While our focus in this study was on the shear-induced platelet granular secretion and surface expression, plasma-bound agonists and inhibitors, proteins (such as vWF), platelet-RBC interactions, calcium-dependent pathways, and mechanically-sensitive calcium channels may be involved in shear-induced cord platelet activation, and elucidation of their roles will paint a more comprehensive picture of platelet mechanotransduction. Furthermore, a broader understanding of molecular mechanisms of platelet activation, particularly under pathological shear stresses where the ineffectiveness of antiplatelet therapies, particularly aspirin, is documented,67,68 will help identify different pathways of how platelets respond to their mechanical environment.61 Outcomes of these efforts will then be beneficial for developing targets to mitigate thrombotic events in neonates, particularly those in NICUs.
Acknowledgments
The authors gratefully acknowledge Brianne Polehinke for her assistance with the experiments. This work was supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health Grants U01 HL131052 (D. Bluestein) and R01 HL119096 (W.F. Bahou).
Conflict of interest
Jawaad Sheriff, Lisa E. Malone, Cecilia Avila, Amanda Zigomalas, Danny Bluestein, and Wadie F. Bahou declare that they have no conflicts of interest.
Research Involving Human and Animal Participants
All human studies were carried out in accordance with institutional guidelines and approved by the Stony Brook University Institutional Review Board (2012-4427-FAR and 2016-5886-R1). No animal studies were carried out by the authors for this article.
Informed Consent
Informed consent was obtained from all subjects included in the study.
Abbreviations
- BAPTA
1,2-Bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid
- −
Intracellular calcium-depleted
- −
Extracellular calcium-depleted
- CD41a
Integrin αIIb, GPIIb
- CD62P
P-selectin
- GFP
Gel-filtered platelets
- HSD
Hemodynamic shearing device
- NICU
Neonatal intensive care unit
- PAS
Platelet activation state, a measure of normalized thrombin generation
- PRP
Platelet-rich plasma
- PS
Phosphatidylserine
- vWF
von Willebrand Factor
- WB
Whole blood
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Antithrombotic Trialists, C. Baigent C, Blackwell L, Collins R, Emberson J, Godwin J, Peto R, Buring J, Hennekens C, Kearney P, Meade T, Patrono C, Roncaglioni MC, Zanchetti A. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet. 2009;373:1849–1860. doi: 10.1016/S0140-6736(09)60503-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Arachiche A, Kerbiriou-Nabias D, Garcin I, Letellier T, Dachary-Prigent J. Rapid procoagulant phosphatidylserine exposure relies on high cytosolic calcium rather than on mitochondrial depolarization. Arterioscler. Thromb. Vasc. Biol. 2009;29:1883–1889. doi: 10.1161/ATVBAHA.109.190926. [DOI] [PubMed] [Google Scholar]
- 3.Baker-Groberg SM, Lattimore S, Recht M, McCarty OJ, Haley KM. Assessment of neonatal platelet adhesion, activation, and aggregation. J. Thromb. Haemost. 2016;14:815–827. doi: 10.1111/jth.13270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bernhard H, Rosenkranz A, Novak M, Leschnik B, Petritsch M, Rehak T, Kofeler H, Ulrich D, Muntean W. No differences in support of thrombin generation by neonatal or adult platelets. Hamostaseologie. 2009;29(Suppl 1):S94–S97. [PubMed] [Google Scholar]
- 5.Bernhard H, Rosenkranz A, Petritsch M, Kofeler H, Rehak T, Novak M, Muntean W. Phospholipid content, expression and support of thrombin generation of neonatal platelets. Acta Paediatr. 2009;98:251–255. doi: 10.1111/j.1651-2227.2008.01075.x. [DOI] [PubMed] [Google Scholar]
- 6.Bevers EM, Comfurius P, Zwaal RF. Changes in membrane phospholipid distribution during platelet activation. Biochim. Biophys. Acta. 1983;736:57–66. doi: 10.1016/0005-2736(83)90169-4. [DOI] [PubMed] [Google Scholar]
- 7.Bevers EM, Williamson PL. Getting to the outer leaflet: physiology of phosphatidylserine exposure at the plasma membrane. Physiol. Rev. 2016;96:605–645. doi: 10.1152/physrev.00020.2015. [DOI] [PubMed] [Google Scholar]
- 8.Caparros-Perez E, Teruel-Montoya R, Lopez-Andreo MJ, Llanos MC, Rivera J, Palma-Barqueros V, Blanco JE, Vicente V, Martinez C, Ferrer-Marin F. Comprehensive comparison of neonate and adult human platelet transcriptomes. PLoS ONE. 2017;12:e0183042. doi: 10.1371/journal.pone.0183042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Caparros-Perez E, Teruel-Montoya R, Palma-Barquero V, Torregrosa JM, Blanco JE, Delgado JL, Lozano ML, Vicente V, Sola-Visner M, Rivera J, Martinez C, Ferrer-Marin F. Down regulation of the Munc18b-syntaxin-11 complex and beta1-tubulin impairs secretion and spreading in neonatal platelets. Thromb. Haemost. 2017;117:2079–2091. doi: 10.1160/TH17-04-0241. [DOI] [PubMed] [Google Scholar]
- 10.Casa LDC, Gillespie SE, Meeks SL, Ku DN. Relative contributions of von willebrand factor and platelets in high shear thrombosis. J. Hematol. Thromboembolic Dis. 2016;4:1–8. [Google Scholar]
- 11.Casa LDC, Ku DN. Thrombus formation at high shear rates. Annu. Rev. Biomed. Eng. 2017;19:415–433. doi: 10.1146/annurev-bioeng-071516-044539. [DOI] [PubMed] [Google Scholar]
- 12.Chen Z, Mondal NK, Ding J, Koenig SC, Slaughter MS, Griffith BP, Wu ZJ. Activation and shedding of platelet glycoprotein IIb/IIIa under non-physiological shear stress. Mol. Cell. Biochem. 2015;409:93–101. doi: 10.1007/s11010-015-2515-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cvirn G, Gallistl S, Rehak T, Jurgens G, Muntean W. Elevated thrombin-forming capacity of tissue factor-activated cord compared with adult plasma. J. Thromb. Haemost. 2003;1:1785–1790. doi: 10.1046/j.1538-7836.2003.00320.x. [DOI] [PubMed] [Google Scholar]
- 14.Ensor CR, Paciullo CA, Cahoon WD, Jr, Nolan PE., Jr Pharmacotherapy for mechanical circulatory support: a comprehensive review. Ann. Pharmacother. 2011;45:60–77. doi: 10.1345/aph.1P459. [DOI] [PubMed] [Google Scholar]
- 15.Flaumenhaft R. Molecular basis of platelet granule secretion. Arterioscler. Thromb. Vasc. Biol. 2003;23:1152–1160. doi: 10.1161/01.ATV.0000075965.88456.48. [DOI] [PubMed] [Google Scholar]
- 16.Flaumenhaft R, Croce K, Chen E, Furie B, Furie BC. Proteins of the exocytotic core complex mediate platelet alpha-granule secretion: Roles of vesicle-associated membrane protein, SNAP-23, and syntaxin 4. J Biol Chem. 1999;274:2492–2501. doi: 10.1074/jbc.274.4.2492. [DOI] [PubMed] [Google Scholar]
- 17.Fritsch P, Cvirn G, Cimenti C, Baier K, Gallistl S, Koestenberger M, Roschitz B, Leschnik B, Muntean W. Thrombin generation in factor VIII-depleted neonatal plasma: nearly normal because of physiologically low antithrombin and tissue factor pathway inhibitor. J. Thromb. Haemost. 2006;4:1071–1077. doi: 10.1111/j.1538-7836.2006.01947.x. [DOI] [PubMed] [Google Scholar]
- 18.Gelman B, Setty BN, Chen D, Amin-Hanjani S, Stuart MJ. Impaired mobilization of intracellular calcium in neonatal platelets. Pediatr. Res. 1996;39:692–696. doi: 10.1203/00006450-199604000-00022. [DOI] [PubMed] [Google Scholar]
- 19.Girdhar G, Xenos M, Alemu Y, Chiu WC, Lynch BE, Jesty J, Einav S, Slepian MJ, Bluestein D. Device thrombogenicity emulation: a novel method for optimizing mechanical circulatory support device thromboresistance. PLoS ONE. 2012;7:e32463. doi: 10.1371/journal.pone.0032463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Goncalves I, Nesbitt WS, Yuan Y, Jackson SP. Importance of temporal flow gradients and integrin alphaIIbbeta3 mechanotransduction for shear activation of platelets. J. Biol. Chem. 2005;280:15430–15437. doi: 10.1074/jbc.M410235200. [DOI] [PubMed] [Google Scholar]
- 21.Grinstein S, Furuya W. Binding of 125I-calmodulin to platelet alpha-granules. FEBS Lett. 1982;140:49–52. doi: 10.1016/0014-5793(82)80518-8. [DOI] [PubMed] [Google Scholar]
- 22.Haga JH, Slack SM, Jennings LK. Comparison of shear stress-induced platelet microparticle formation and phosphatidylserine expression in presence of alphaIIbbeta3 antagonists. J. Cardiovasc. Pharmacol. 2003;41:363–371. doi: 10.1097/00005344-200303000-00004. [DOI] [PubMed] [Google Scholar]
- 23.Hellums JD. 1993 Whitaker Lecture: biorheology in thrombosis research. Ann. Biomed. Eng. 1994;22:445–455. doi: 10.1007/BF02367081. [DOI] [PubMed] [Google Scholar]
- 24.Ilkan Z, Wright JR, Goodall AH, Gibbins JM, Jones CI, Mahaut-Smith MP. Evidence for shear-mediated Ca(2+) entry through mechanosensitive cation channels in human platelets and a megakaryocytic cell line. J. Biol. Chem. 2017;292:9204–9217. doi: 10.1074/jbc.M116.766196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Israels SJ, Rand ML, Michelson AD. Neonatal platelet function. Semin. Thromb. Hemost. 2003;29:363–372. doi: 10.1055/s-2003-42587. [DOI] [PubMed] [Google Scholar]
- 26.Ivetic N, Arnold DM, Smith JW, Huynh A, Kelton JG, Nazy I. A platelet viability assay (PVA) for the diagnosis of heparin-induced thrombocytopenia. Platelets. 2019;30:1017–1021. doi: 10.1080/09537104.2018.1562169. [DOI] [PubMed] [Google Scholar]
- 27.Jackson SP. Arterial thrombosis—insidious, unpredictable and deadly. Nat. Med. 2011;17:1423–1436. doi: 10.1038/nm.2515. [DOI] [PubMed] [Google Scholar]
- 28.Jesty J, Bluestein D. Acetylated prothrombin as a substrate in the measurement of the procoagulant activity of platelets: elimination of the feedback activation of platelets by thrombin. Anal. Biochem. 1999;272:64–70. doi: 10.1006/abio.1999.4148. [DOI] [PubMed] [Google Scholar]
- 29.Jones CI. Platelet function and ageing. Mamm. Genome. 2016;27:358–366. doi: 10.1007/s00335-016-9629-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Knight DE, Scrutton MC. Direct evidence for a role for Ca2+ in amine storage granule secretion by human platelets. Thromb. Res. 1980;20:437–446. doi: 10.1016/0049-3848(80)90282-0. [DOI] [PubMed] [Google Scholar]
- 31.Kroll MH, Hellums JD, McIntire LV, Schafer AI, Moake JL. Platelets and shear stress. Blood. 1996;88:1525–1541. [PubMed] [Google Scholar]
- 32.Lemons PP, Chen D, Whiteheart SW. Molecular mechanisms of platelet exocytosis: requirements for alpha-granule release. Biochem. Biophys. Res. Commun. 2000;267:875–880. doi: 10.1006/bbrc.1999.2039. [DOI] [PubMed] [Google Scholar]
- 33.Levy-Shraga Y, Maayan-Metzger A, Lubetsky A, Shenkman B, Kuint J, Martinowitz U, Kenet G. Platelet function of newborns as tested by cone and plate(let) analyzer correlates with gestational age. Acta Haematol. 2006;115:152–156. doi: 10.1159/000090928. [DOI] [PubMed] [Google Scholar]
- 34.Leytin V, Allen DJ, Mykhaylov S, Mis L, Lyubimov EV, Garvey B, Freedman J. Pathologic high shear stress induces apoptosis events in human platelets. Biochem. Biophys. Res. Commun. 2004;320:303–310. doi: 10.1016/j.bbrc.2004.05.166. [DOI] [PubMed] [Google Scholar]
- 35.McNeil JJ, Woods RL, Nelson MR, Reid CM, Kirpach B, Wolfe R, Storey E, Shah RC, Lockery JE, Tonkin AM, Newman AB, Williamson JD, Margolis KL, Ernst ME, Abhayaratna WP, Stocks N, Fitzgerald SM, Orchard SG, Trevaks RE, Beilin LJ, Donnan GA, Gibbs P, Johnston CI, Ryan J, Radziszewska B, Grimm R, Murray AM, A.I. Group Effect of aspirin on disability-free survival in the healthy elderly. N Engl J Med. 2018;379:1499–1508. doi: 10.1056/NEJMoa1800722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Metharom P, Berndt MC, Baker RI, Andrews RK. Current state and novel approaches of antiplatelet therapy. Arterioscler. Thromb. Vasc. Biol. 2015;35:1327–1338. doi: 10.1161/ATVBAHA.114.303413. [DOI] [PubMed] [Google Scholar]
- 37.Michelson AD, Rajasekhar D, Bednarek FJ, Barnard MR. Platelet and platelet-derived microparticle surface factor V/Va binding in whole blood: differences between neonates and adults. Thromb. Haemost. 2000;84:689–694. [PubMed] [Google Scholar]
- 38.Monagle P, Chan AKC, Goldenberg NA, Ichord RN, Journeycake JM, Nowak-Gottl U, Vesely SK. Antithrombotic therapy in neonates and children: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141:e737S–e801S. doi: 10.1378/chest.11-2308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Muntean W, Leschnik B, Baier K, Cvirn G, Gallistl S. In vivo thrombin generation in neonates. J. Thromb. Haemost. 2004;2:2071–2072. doi: 10.1111/j.1538-7836.2004.00966.x. [DOI] [PubMed] [Google Scholar]
- 40.Ngo ATP, Sheriff J, Rocheleau AD, Bucher M, Jones KR, Sepp AI, Malone LE, Zigomalas A, Maloyan A, Bahou WF, Bluestein D, McCarty OJT, Haley KM. Assessment of neonatal, cord, and adult platelet granule trafficking and secretion. Platelets. 2020;31:68–78. doi: 10.1080/09537104.2019.1573314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nobili M, Sheriff J, Morbiducci U, Redaelli A, Bluestein D. Platelet activation due to hemodynamic shear stresses: damage accumulation model and comparison to in vitro measurements. ASAIO J. 2008;54:64–72. doi: 10.1097/MAT.0b013e31815d6898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pichler E, Pichler L. The neonatal coagulation system and the vitamin K deficiency bleeding—a mini review. Wien. Med. Wochenschr. 2008;158:385–395. doi: 10.1007/s10354-008-0538-7. [DOI] [PubMed] [Google Scholar]
- 43.Qiu Y, Ciciliano J, Myers DR, Tran R, Lam WA. Platelets and physics: How platelets “feel” and respond to their mechanical microenvironment. Blood Rev. 2015;29:377–386. doi: 10.1016/j.blre.2015.05.002. [DOI] [PubMed] [Google Scholar]
- 44.Receveur, N., D. Nechipurenko, Y. Knapp, A. Yakusheva, E. Maurer, C. V. Denis, F. Lanza, M. Panteleev, C. Gachet, and P. H. Mangin. Shear rate gradients promote a bi-phasic thrombus formation on weak adhesive proteins, such as fibrinogen in a VWF-dependent manner. Haematologica 2019. 10.3324/haematol.2019.235754. [DOI] [PMC free article] [PubMed]
- 45.Rehak T, Cvirn G, Gallistl S, Leschnik B, Kostenberger M, Katzer H, Ribitsch V, Muntean W. Increased shear stress- and ristocetin-induced binding of von Willebrand factor to platelets in cord compared with adult plasma. Thromb. Haemost. 2004;92:682–687. doi: 10.1160/TH04-05-0270. [DOI] [PubMed] [Google Scholar]
- 46.Rice NT, Szlam F, Varner JD, Bernstein PS, Szlam AD, Tanaka KA. Differential contributions of intrinsic and extrinsic pathways to thrombin generation in adult, maternal and cord plasma samples. PLoS ONE. 2016;11:e0154127. doi: 10.1371/journal.pone.0154127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Roka-Moiia Y, Walk R, Palomares DE, Ammann KR, Dimasi A, Italiano JE, Sheriff J, Bluestein D, Slepian MJ. Platelet activation via shear stress exposure induces a differing pattern of biomarkers of activation versus biochemical agonists. Thromb. Haemost. 2020;120:776–792. doi: 10.1055/s-0040-1709524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Roschitz B, Sudi K, Kostenberger M, Muntean W. Shorter PFA-100 closure times in neonates than in adults: role of red cells, white cells, platelets and von Willebrand factor. Acta Paediatr. 2001;90:664–670. [PubMed] [Google Scholar]
- 49.Ruggeri ZM Von. Willebrand factor: looking back and looking forward. Thromb. Haemost. 2007;98:55–62. [PubMed] [Google Scholar]
- 50.Ruggeri ZM, Orje JN, Habermann R, Federici AB, Reininger AJ. Activation-independent platelet adhesion and aggregation under elevated shear stress. Blood. 2006;108:1903–1910. doi: 10.1182/blood-2006-04-011551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sakariassen KS, Holme PA, Orvim U, Barstad RM, Solum NO, Brosstad FR. Shear-induced platelet activation and platelet microparticle formation in native human blood. Thromb. Res. 1998;92:S33–S41. doi: 10.1016/s0049-3848(98)00158-3. [DOI] [PubMed] [Google Scholar]
- 52.Saving KL, Jennings DE, Aldag JC, Caughey RC. Platelet ultrastructure of high-risk premature infants. Thromb. Res. 1994;73:371–384. doi: 10.1016/0049-3848(94)90039-6. [DOI] [PubMed] [Google Scholar]
- 53.Saxonhouse MA, Garner R, Mammel L, Li Q, Muller KE, Greywoode J, Miller C, Sola-Visner M. Closure times measured by the platelet function analyzer PFA-100 are longer in neonatal blood compared to cord blood samples. Neonatology. 2010;97:242–249. doi: 10.1159/000253755. [DOI] [PubMed] [Google Scholar]
- 54.Schoenwaelder SM, Yuan Y, Josefsson EC, White MJ, Yao Y, Mason KD, O’Reilly LA, Henley KJ, Ono A, Hsiao S, Willcox A, Roberts AW, Huang DC, Salem HH, Kile BT, Jackson SP. Two distinct pathways regulate platelet phosphatidylserine exposure and procoagulant function. Blood. 2009;114:663–666. doi: 10.1182/blood-2009-01-200345. [DOI] [PubMed] [Google Scholar]
- 55.Schulz-Heik K, Ramachandran J, Bluestein D, Jesty J. The extent of platelet activation under shear depends on platelet count: differential expression of anionic phospholipid and factor Va. Pathophysiol. Haemost. Thromb. 2005;34:255–262. doi: 10.1159/000093104. [DOI] [PubMed] [Google Scholar]
- 56.Schweintzger S, Schlagenhauf A, Leschnik B, Rinner B, Bernhard H, Novak M, Muntean W. Microparticles in newborn cord blood: slight elevation after normal delivery. Thromb. Res. 2011;128:62–67. doi: 10.1016/j.thromres.2011.01.013. [DOI] [PubMed] [Google Scholar]
- 57.Shenkman B, Linder N, Savion N, Tamarin I, Dardik R, Kennet G, German B, Varon D. Increased neonatal platelet deposition on subendothelium under flow conditions: the role of plasma von Willebrand factor. Pediatr. Res. 1999;45:270–275. doi: 10.1203/00006450-199902000-00019. [DOI] [PubMed] [Google Scholar]
- 58.Sheriff J, Bluestein D, Girdhar G, Jesty J. High-shear stress sensitizes platelets to subsequent low-shear conditions. Ann. Biomed. Eng. 2010;38:1442–1450. doi: 10.1007/s10439-010-9936-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sheriff J, Soares JS, Xenos M, Jesty J, Bluestein D. Evaluation of shear-induced platelet activation models under constant and dynamic shear stress loading conditions relevant to devices. Ann. Biomed. Eng. 2013;41:1279–1296. doi: 10.1007/s10439-013-0758-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sitaru AG, Holzhauer S, Speer CP, Singer D, Obergfell A, Walter U, Grossmann R. Neonatal platelets from cord blood and peripheral blood. Platelets. 2005;16:203–210. doi: 10.1080/09537100400016862. [DOI] [PubMed] [Google Scholar]
- 61.Slepian MJ, Sheriff J, Hutchinson M, Tran P, Bajaj N, Garcia JGN, Scott Saavedra S, Bluestein D. Shear-mediated platelet activation in the free flow: Perspectives on the emerging spectrum of cell mechanobiological mechanisms mediating cardiovascular implant thrombosis. J Biomech. 2017;50:20–25. doi: 10.1016/j.jbiomech.2016.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sola-Visner M. Platelets in the neonatal period: developmental differences in platelet production, function, and hemostasis and the potential impact of therapies. Hematol Am Soc Hematol Educ Program. 2012;506–511:2012. doi: 10.1182/asheducation-2012.1.506. [DOI] [PubMed] [Google Scholar]
- 63.Stokhuijzen E, Koornneef JM, Nota B, van den Eshof BL, van Alphen FPJ, van den Biggelaar M, van der Zwaan C, Kuijk C, Mertens K, Fijnvandraat K, Meijer AB. Differences between platelets derived from neonatal cord blood and adult peripheral blood assessed by mass spectrometry. J. Proteome Res. 2017;16:3567–3575. doi: 10.1021/acs.jproteome.7b00298. [DOI] [PubMed] [Google Scholar]
- 64.Suzuki J, Denning DP, Imanishi E, Horvitz HR, Nagata S. Xk-related protein 8 and CED-8 promote phosphatidylserine exposure in apoptotic cells. Science. 2013;341:403–406. doi: 10.1126/science.1236758. [DOI] [PubMed] [Google Scholar]
- 65.Suzuki J, Umeda M, Sims PJ, Nagata S. Calcium-dependent phospholipid scrambling by TMEM16F. Nature. 2010;468:834–838. doi: 10.1038/nature09583. [DOI] [PubMed] [Google Scholar]
- 66.Tesfamariam B. Distinct characteristics of neonatal platelet reactivity. Pharmacol. Res. 2017;123:1–9. doi: 10.1016/j.phrs.2017.06.003. [DOI] [PubMed] [Google Scholar]
- 67.Valerio L, Sheriff J, Tran PL, Brengle W, Redaelli A, Fiore GB, Pappalardo F, Bluestein D, Slepian MJ. Routine clinical anti-platelet agents have limited efficacy in modulating hypershear-mediated platelet activation associated with mechanical circulatory support. Thromb. Res. 2018;163:162–171. doi: 10.1016/j.thromres.2017.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Valerio L, Tran PL, Sheriff J, Brengle W, Ghosh R, Chiu WC, Redaelli A, Fiore GB, Pappalardo F, Bluestein D, Slepian MJ. Aspirin has limited ability to modulate shear-mediated platelet activation associated with elevated shear stress of ventricular assist devices. Thromb. Res. 2016;140:110–117. doi: 10.1016/j.thromres.2016.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.van Kruchten R, Mattheij NJ, Saunders C, Feijge MA, Swieringa F, Wolfs JL, Collins PW, Heemskerk JW, Bevers EM. Both TMEM16F-dependent and TMEM16F-independent pathways contribute to phosphatidylserine exposure in platelet apoptosis and platelet activation. Blood. 2013;121:1850–1857. doi: 10.1182/blood-2012-09-454314. [DOI] [PubMed] [Google Scholar]
- 70.White JG. Effects of an ionophore, A23187, on the surface morphology of normal erythrocytes. Am. J. Pathol. 1974;77:507–518. [PMC free article] [PubMed] [Google Scholar]
- 71.Will A. Neonatal haemostasis and the management of neonatal thrombosis. Br. J. Haematol. 2015;169:324–332. doi: 10.1111/bjh.13301. [DOI] [PubMed] [Google Scholar]
- 72.Xenos M, Girdhar G, Alemu Y, Jesty J, Slepian M, Einav S, Bluestein D. Device Thrombogenicity Emulator (DTE) - Design optimization methodology for cardiovascular devices: a study in two bileaflet MHV designs. J. Biomech. 2010;43:2400–2409. doi: 10.1016/j.jbiomech.2010.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zwaal RF, Comfurius P, Bevers EM. Surface exposure of phosphatidylserine in pathological cells. Cell. Mol. Life Sci. 2005;62:971–988. doi: 10.1007/s00018-005-4527-3. [DOI] [PMC free article] [PubMed] [Google Scholar]