Abstract
Background:
Conceptualizations of delusion formation implicate deficits at feedforward information updating across posterior to prefrontal cortices, resulting in dysfunctional integration of new information about contexts in working memory, and ultimately, failure to update over-familiar prior beliefs. Here, we used functional MRI and machine learning models to address individual variability in feed-forward parietal-prefrontal information updating in schizophrenia patients, and examined relationships with delusional thinking, and polygenic risk for schizophrenia.
Methods:
We studied 66 schizophrenia patients and 143 healthy controls as they performed context updating during working memory (WM). Dynamic causal models of effective connectivity were focused on prefrontal and parietal cortex potentially implicated in delusion processes. We examined polygenic risk for schizophrenia on connectivity in healthy individuals. We then leveraged support vector regression models to define optimal normalized target connectivity tailored for each patient, and tested the extent to which deviation from this target could predict individual variation in delusion severity.
Results:
In schizophrenia patients, updating and manipulating context information was disproportionately less accurate than was WM maintenance, with a task accuracy-by-diagnosis interaction. Also, patients with delusions tended to have relatively reduced parietal-prefrontal feedforward effective connectivity during context updating in WM manipulation. The same connectivity was adversely influenced by polygenic risk for schizophrenia in healthy subjects. Individual patients’ deviation from predicted ‘normal’ feedforward connectivity based on the support vector regression models correlated with delusional severity.
Conclusions:
These computationally-derived observations support a role for feed-forward parietal-prefrontal information processing deficits in delusional psychopathology, and in genetic risk for schizophrenia.
Introduction:
Delusions occur across a range of psychotic disorders, and are characterized by distressing false beliefs that preoccupy, are held with conviction, and disrupt lives. Delusions have complex phenomenology, and possibly just as complex theories of causative and maintenance factors (1–5). Recent conceptualizations of the cognitive architecture of delusions have modeled how aberrant beliefs are over-learnt and become immutable (1, 6–8). It has been argued that the preoccupation and conviction with which beliefs are held may stem from cognitive biases against incorporating into beliefs of evidence otherwise (2, 4). These erroneous beliefs are potentially further reinforced through exaggerated subcortical dopaminergic reinforcement (9, 10). Consistent with this formulation, related hallucinatory phenomena in at-risk mental states were recently observed to result from biasing prior expectations over incoming (contrary) sensory evidence (11). At the level of brain function, these computational biases appear to result from deficits in feedforward information updating, and have also been found in subclinical psychotic experiences (11). More specific to the abstract information processing in delusional beliefs, we previously found that deficits in new information updating in favor of prior beliefs was also associated with reduced feed-forward effective connectivity across posterior to prefrontal cortices (1). These changes were unlikely to be epiphenomena of treatment, because analogous information processing changes also were observed in unaffected siblings of patients with schizophrenia (1).
Our goal, herein, is to extend recent observations linking delusional psychopathology, genetic risk for schizophrenia, and deficits in feedforward parietal-to-prefrontal connectivity during updating of new context information (1). Recent models of delusional and perceptual psychopathology suggest deficits in feedforward cortical integration of new information into belief models, leading to dysfunctional strong priors and consequent belief and perceptual disturbances (1, 12, 13). We hypothesize that these effects should also be reflected in updating and manipulating context information in working memory (WM). Cortical integration of new information into beliefs occur along a hierarchy of overlapping processes, including executive aspects of WM, Bayesian learning and reasoning, and episodic memory (14, 15). We reason that because executive aspects of WM form the base of these hierarchical processes, and involve updating prior information with new content, they should be critically engaged in the updating of new information into belief models putatively dysfunctional in delusions, and should also be associated with the underlying genetic mechanisms of schizophrenia (1, 12, 13). This formulation is supported by the observed relationships between tasks updating new information in contextual WM, and the Bayesian integration of the statistics of this new information in WM, upon which hierarchically more complex belief processing subsequently occurs (1, 14, 15). Recent work has also underscored relationships between deficits in updating or manipulating information in contextual WM, feedforward parietal-prefrontal connectivity, and delusions (1, 13, 16, 17). We therefore predicted that if feedforward parietal-prefrontal connectivity while manipulating prior information held in WM might relate to delusional psychopathology in patients with schizophrenia, these neural functions might also relate to genetic mechanisms of illness, rather than confounds associated with medications or chronic ill health. If so, we would expect that there would be relationships between feedforward parietal-prefrontal connectivity with polygenic risk for disease, particularly in healthy individuals.
We also aim, herein, to examine a substantial issue related to inter-individual variability in cortical function and connectivity, which is particularly pronounced in prefrontal cortical processing we examine here (18–20). As each patient’s optimal neural function for a given cognitive task likely varies, a given patient’s deviation from his or her theoretical optimum should closely relate to cognitive dysfunction and psychopathology, if indeed, the specific neural function is implicated in these disease mechanisms. To examine this, we leverage data in normal control samples who performed analogous context updating tasks. We created support vector regression models of how target feedforward parietal-prefrontal network connectivity during context manipulation in WM may be predicted by connectivity during simple maintenance of information, the latter process found to be less dysfunctional in schizophrenia (21–23). The machine learning models thus derived in healthy subjects were then applied to each patient to determine what his or her target “normal” quantitative feedforward connectivity should be. This novel strategy takes inspiration from related machine learning decoder approaches that utilize surrogate subjects to predict activation patterns for an individual subject (24).
Methods:
Participants:
We examined 143 healthy controls and 66 patients with schizophrenia as they performed an event-related WM task in 3T MRI scanner. Participants were enrolled as part of the Clinical Brain Disorders Branch Sibling Study (25), were between 18 and 45 years of age (Table 1) and right-handed. They were interviewed by a research psychiatrist using the Structured Clinical Interview for DSM-IV, and completed a neurological examination and a battery of neuropsychological tests. Exclusion criteria included an IQ <70, a history of prolonged substance abuse or significant medical problems, and any abnormalities found by EEG or MRI. All participants gave written consent before participation. The study was approved by the NIMH Institutional Review Board. The individuals in this sample were not part of our earlier report (1).
Table 1.
Demographics and behavioral performance for controls and patients with schizophrenia.
| CON | SD | SZ | SD | p | |
|---|---|---|---|---|---|
| (n = 143) | (n = 66) | ||||
| Sex (number of males) | 66 | 44 | 0.01 | ||
| Age (years) | 31.027 | 9.248 | 31.909 | 10.551 | ns |
| Parental Education (years) | 14.23 | 3.42 | 13.82 | 3.67 | ns |
| Education (years) | 16.517 | 2.672 | 14.615 | 2.638 | <0.001 |
| IQ (WAIS) | 111.846 | 8.652 | 96.52 | 10.152 | <0.001 |
| Accuracy | |||||
| Working memory manipulation | 0.933 | 0.081 | 0.776 | 0.155 | <0.001 |
| Working memory maintenance | 0.980 | 0.048 | 0.939 | 0.076 | <0.001 |
| Reaction time (s) | |||||
| Working memory manipulation | 1.813 | 0.280 | 1.955 | 0.373 | 0.003 |
| Working memory maintenance | 1.039 | 0.166 | 1.181 | 0.199 | <0.001 |
CON: Controls; SZ: Patients with schizophrenia; WAIS: Wechsler Adult Intelligence Scale
Cognitive Task Paradigm:
Blood oxygen level-dependent functional MRI data were acquired from subjects after a brief training period (~10 min). The event-related WM paradigm was described in detail in a previous study (26). In each trial, subjects first encoded 2 integer numbers presented over 1s and retained this context information in WM across a jittered interval of 3–5 seconds. In maintenance trials, subjects subsequently responded to cues as to which of the two numbers was “larger” or “smaller” within 2s. In the manipulation trials, subjects had to perform a mental subtraction of 2 or 3 on one of the two context numbers before the “larger” or “smaller” evaluation within 2s. There were 28 trials of WM manipulation and 28 trials of WM maintenance counterbalanced for trial type, and numerical size order, over about 9 minutes.
Imaging Parameters and Analyses
T2*-weighted echo planar imaging (EPI) images with BOLD contrast were obtained with a 3T MRI scanner (GEs, Milwaukee, WI) using a standard GE head coil (64×64×24 matrix with 3.75×3.75×6.0mm spatial resolution, repetition time (TR)=2000 ms, echo time (TE)=28ms, flip angle=90°, field of view (FOV)=24×24cm) while participants performed the cognitive task. The first four scans were discarded to allow for signal saturation.
The functional imaging data were preprocessed and analyzed using the general linear model for event-related designs in statistical parametric mapping (SPM12, Wellcome Trust Centre for Neuroimaging, London, United Kingdom). The functional images were corrected for differences in acquisition time between slices for each whole-brain volume and realigned to correct for head movement. Six movement parameters (translation: x, y, z and rotation: roll, pitch, yaw) were included in the statistical model as covariates of no interest. The functional images were normalized to a standard EPI MNI template and then spatially smoothed using an isotropic Gaussian kernel of 8mm fullwidth half-maximum.
A random-effects, event-related statistical analysis was performed at two levels. In the first level, the onsets and durations of each trial for each task condition were convolved with a canonical hemodynamic response function and modeled using a general linear model on an individual subject basis. The realignment parameters were included as additional regressors of no interest. Data were high-pass filtered at 1/128 Hz. Random-effects analyses at the second (group) level were then conducted based on statistical parameter maps from each individual participant to allow population-level inference. Group level activation maps were thresholded at voxel-level whole brain p<0.05 family-wise error (FWE) correction for multiple comparisons, unless otherwise stated.
Dynamic Causal Modeling
We used Dynamic Causal Modeling, DCM (27) as implemented in SPM12 (DCM10) to examine how parietal and prefrontal brain regions interacted. We focused on the left parietal (–38 –58 38) and prefrontal (–44 30 16) regions implicated previously in context updating dysfunction in delusions (1), and examined the corresponding Human Connectome Project (28) defined parcels in the parietal (area LIPd) and prefrontal (area 46) cortex encompassing these peaks. Time series during WM context updating (manipulation) were extracted from each of the two regions-of-interest (ROI) for each individual, masked by the conjunction of the respective cortical parcel, the group-level activation mask at p<0.05 voxelwise whole brain FWE corrected for multiple comparisons, and an individual subject level activation at p<0.05. Deterministic, bilinear DCM models comprising all 7 possible input and pairing combinations of these two ROIs (29) were then specified (Fig 1B shows this at the DLPFC and parietal cortex, though this also applies to all ROI pairs in the machine learning models below). The strength and direction of regional interactions were then estimated, elucidating how regional neural activity and their interactions were influenced by cognitive inputs during WM context updating, as well as how these neuronal effects were biophysically linked to form blood-oxygen-level-dependent signals (27). Bayesian model averaging (BMA) was used to generate weighted task-related connectivity averages in each direction, for each pair of nodes, based on the posterior model probability (30). Using these BMA results, we then examined task-related modulation of regional connectivity across participants, and relationships with psychopathology and polygenic risk for schizophrenia (31).
Figure 1: Working memory behavioral and feed-forward parietal-prefrontal connectivity.
(A) Relative to controls (N=143), patients with schizophrenia (N=66) were disproportionately less accurate in performing context updating working memory manipulation, than simple maintenance of context information in WM (interaction p<0.001). (B) Patients and controls, combined (or in separate groups, not shown) robustly engaged regions-of-interest (glass-brain) in areas 46 and LIPd (p<0.05 voxelwise whole-brain FWE corrected). These ROIs encompassed regions in the DLPFC (−44 30 16) and parietal cortex (−38 −58 38) where effective connectivity during context updating was previously found to be dysfunctional in schizophrenia patients in relation to delusions (1). Dynamic Causal Models comprising all possible driving inputs (black arrows) and task-related modulation of connectivity (blue arrows) were constructed, estimated and summarized using Bayesian Model Averaging. (C) Feedforward effective connectivity from the parietal to prefrontal cortex during context updating was relatively reduced in schizophrenia patients with delusions (N=35) vs without (N=31, p=0.05). (D) Feedforward effective connectivity from the parietal to prefrontal cortex during context updating was relatively reduced in relation to increased polygenic risk for schizophrenia in healthy controls (N=143, p<0.02).
Polygenic risk for schizophrenia
PRS for schizophrenia were calculated for each control subject as a measure of genomic risk for schizophrenia, based on the most recent GWAS (31). We obtained odds ratios of 102,217 index SNPs from a meta-analysis of PGC2 GWAS using datasets excluding the discovery sample (PGC 2014, non-Lieber sample PGC2 GWAS). These 102K SNPs were LD independent (R2<0.1). We then calculated a weighted sum of risk alleles for schizophrenia, by summing the imputation probability for the reference allele of the index SNP, weighted by the natural log of the odds ratio of association with schizophrenia, at each independent locus across the whole genome (31, 32). In this paper, we used the PRS calculated using the conservative PGC2 GWAS association p-value of 5e-08.
Defining individual patient’s optimal parietal-prefrontal connectivity:
We defined and tested the validity of each individual patient’s putative optimal (normal) parietal-prefrontal connectivity at WM context updating. We first created a linear support vector regression (SVR) model, in healthy controls, of how feedforward parietal-prefrontal network connectivity during context updating may be predicted by connectivities from prefrontal and parietal regions during simple WM maintenance, a process that is less dysfunctional in schizophrenia than context updating (21–23). We included 31 parcels in the frontal and parietal cortex which were robustly engaged during WM maintenance in the patient and control samples (p<0.05 whole-brain voxel-wise FWE corrected in each separate patient or control sample). Time series during WM maintenance were then extracted from the ROI for each individual, masked by the conjunction of the respective cortical parcel, group level activation mask at p<0.05 whole brain voxelwise FWE corrected for multiple comparisons, and the individual subject level activation at p<0.05. Deterministic DCM models comprising all possible pairings across these 31 ROIs (29), giving 465 pairs of regions. From each pair of ROIs, we determined the BMA over all 7 possible models within which a pair of regions could effectively interact (as above and in (29)). This then resulted in a directed matrix of effective connectivity during WM maintenance, predicting in SVR modeling in healthy controls, the target parietal-prefrontal feedforward connectivity during WM context updating. SVR was conducted in Matlab using the implementation “fitrsvm” with a linear kernel, automatic hyperparameter tuning and Sequential Minimal Optimization. We examined the in-sample predictive validity in a leave-one-out correlation analysis of the left-out predicted parietal-prefrontal connectivity versus the actual connectivity. We also examined predictions in more conservative approaches, including 5-fold cross validation in SVR with a held out 20% test-sample, and Elastic Net Regression (33) with alpha 0.5, also with a held out 20% test-sample.
More importantly, we then tested the validity of this modeling approach in the patients. The SVR models thus derived in healthy controls were then applied to each patient to determine what that individual patient’s target “normal” feedforward connectivity should be at context updating during WM manipulation, given the patient’s connectivity pattern during the relatively less dysfunctional WM maintenance. We then examined each patient’s predicted normal connectivity and the deviation from his/her actual connectivity, and the extent to which this deviation correlated with delusional psychopathology, hypothesized previously to be associated with the feedforward connectivity deficits. We then tested the extent to which the connectivity-derived support vector predictions may outperform correlations with delusions derived by simply comparing between patient feedforward connectivity and mean control feedforward connectivity, and also by feedforward connectivity deviations across patients and controls uninformed by machine learning, the latter generated by random pairings between patients and controls, over 10,000 permutations.
Results:
Demographics and Behavior:
Across healthy subjects (N=143) and schizophrenia patients (N=66), age and parental education were similar. Patients had a mean (±SD) duration of illness of 8.20±7.82 years with a mean age of onset 19.5±4.2years, mean total positive and negative symptom score of 54±24 and were on 298±389 mg/day chlorpromazine equivalent dose of antipsychotics. WM manipulation was more difficult (less accurate) than maintenance, and patients generally performed poorer in WM accuracy and reaction time (Table 1, p<0.001). However, WM manipulation was disproportionately more dysfunctional than WM maintenance in patients (WM process-by-diagnosis interaction F(1,217)=63.9, p<0.001, Fig 1A).
Parietal-prefrontal feedforward connectivity
In this report, we focus on the left LIPd and area 46 regions-of-interest (Human Connectome Project parcellation notations (28)), which correspond to regions where we previously found parietal and prefrontal effects associated with delusions and context updating in schizophrenia (1). Peaks in the left LIPd and area 46 ROIs were significantly engaged during context updating in WM manipulation at the group-level in controls and patients (p<0.05 voxel-wise whole-brain FWE corrected for multiple comparisons, in each control and patient group, Supplementary Table S1, Fig 1B). Effective connectivity across these two ROIs during context updating in WM manipulation were engaged at feedforward (parietal to prefrontal) and feedback (prefrontal to parietal) in controls (p<0.001, Supplementary Table S2) and schizophrenia patients (p<0.001, Supplementary Table S4). Connectivity in both directions was relatively reduced in patients between LIPd and area 46 ROIs (p<0.001).
Within patients with schizophrenia (N=66), we examined the hypothesized role of reduced parietal-prefrontal feedforward effective connectivity at left LIPd and area 46 regions during context updating at WM manipulation in delusional psychopathology suggested previously (1). Individuals with delusions on the PANSS (ratings ≥3) tended to have relatively reduced parietal-prefrontal feedforward effective connectivity (t=1.97, p=0.05; Fig 1C). Effects in the opposite feedback direction were not significant.
We then tested whether these feedforward parietal to prefrontal neural functions related to putative genetic mechanisms of illness, rather than to confounds associated with medications or chronic ill health, through effects on polygenic risk for schizophrenia in healthy individuals. Left LIPd to area 46 feedforward effective connectivity, during WM manipulation, was reduced in association with increased polygenic risk for schizophrenia (Fig 1D, r=−0.19, p<0.02). Effects in the opposite feedback direction were not significant.
Defining individual patient’s optimal parietal-prefrontal connectivity:
If indeed dysfunction in parietal-prefrontal feedforward effective connectivity during updating of new information into context may relate to delusions, the degree to which an individual patient’s connectivity may deviate from her “optimum” value should also relate to dysfunction with delusions, particularly if the connectivity optimum reflects a relevant target to normalize psychopathology. To define an individual subject’s parietal-prefrontal connectivity optimum during WM manipulation, we first examined 31 of the most activated parcels in the prefrontal and parietal cortices during WM maintenance in controls (peak activation in each parcel of p<0.05 voxel-wise whole-brain FWE corrected for multiple comparisons, Supplementary Table S3). From these brain parcels, we computed the effective connectivity from each pair of nodes in both directions, giving 930 pair-wise effective connectivity values during WM maintenance. As noted previously, WM maintenance performance accuracy was relatively less dysfunctional in patients (Fig 1A, Cohen’s effect size d=0.67 for maintenance vs d=2.11 for manipulation, relative to controls). However, in healthy controls (N=143), within the WM maintenance prefrontal-parietal connectivity patterns in leave-one-out SVR models, feed-forward parietal-prefrontal connectivity at LIPd to area 46 during WM manipulation was robustly predicted (N=143, r>0.9, p<0.001; Supplementary Fig 1A). More conservative SVR models trained with 80% of the healthy sample (N=116) and tested on a held out 20% sample also gave robust predictions (N=27, r>0.80, p<0.001; Fig 2A). Elastic net regularization and variable selection (alpha 0.5), which effectively tackles model fitting where number of variables exceed that of subjects (33), yielded connectivity predictions in the held-out sample with a reasonably similar effect size as SVR (N=27, r=0.60, p<0.001; Supplementary Fig 1B).
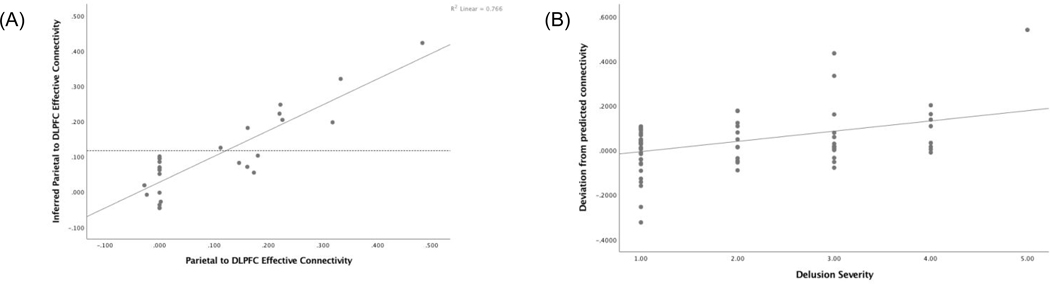
Figure 2: Modeled parietal-prefrontal effective connectivity.
(A) Support vector regression models were created in N=116 and tested in a held out sample of N=27 healthy subjects. Inferred connectivity in the left-out sample corresponded with actual connectivity (r>0.8, p<0.001). (B) In patients with schizophrenia, deviation from predicted ‘normal’ feedforward effective connectivity derived from the independent healthy sample correlated with delusional psychopathology (N=66 patients, r=0.41, p<0.001).
We then reasoned that given WM maintenance connectivity from patients, we might predict their corresponding optimized WM manipulation feedforward connectivity, using the SVR model independently derived from healthy controls. We tested the extent to which this individualized metric may be clinically significant, and found that patients’ deviations from this metric was associated with delusions (r=0.41, p<0.001; Fig 2B). On the other hand, deviation between each patient’s connectivity and mean control connectivity did not correlate with psychopathology (p>0.17). Deviation between each patient’s connectivity and randomly paired controls at parietal-prefrontal connectivity also did not correlate with psychopathology, and yielded an average r=0.01, p=0.52 over 10,000 iterations, with 2/10,000 instances of performance equal or better relative to that of the support vector machine (r>=0.41). We further tested the specificity to delusional psychopathology, and found no significant correlation between each patient’s deviation from modeled ‘optimal’ feedforward connectivity with negative syndrome score (p>0.12), or general symptom score (p>0.8). There was a smaller correlation with positive syndrome score (r=0.25, p=0.03), and within the positive syndrome, correlation with hallucinations (r=0.26, p=0.02), as well as the larger effect with delusions noted above. In a regression of the connectivity deviation against hallucinations and delusions, the hallucinations effect disappeared (p>0.8) while the delusions effect remained (beta=0.41, p=0.01), suggesting a degree of specificity for delusions.
Discussion:
In healthy individuals, we observed that relative deficits in feedforward parietal-prefrontal connectivity during updating of new information into context during WM manipulation is associated with higher polygenic risk for schizophrenia. In schizophrenia patients, we similarly find, in an independent sample, earlier observations that this parietal-prefrontal effective connectivity relates to delusional psychopathology (1, 17), though as expected, given the particular variability at prefrontal parietal function (18–20), these effects are not large (Cohen’s d<0.45). We then examined an estimate of optimal feedforward connectivity during WM manipulation in patients, derived from the connectivity patterns at a less dysfunctional WM maintenance task. Using a machine learning model derived from healthy subjects, larger deviations in feedforward parietal-prefrontal connectivity from predicted optima also related to delusional psychopathology in patients.
These observations are consistent with our previous findings in a non-overlapping sample (1), as well as that of others (12, 17), on the role of feedforward information processing deficits in delusional psychopathology, and genetic risk for schizophrenia. Individualized predictions of feed-forward parietal prefrontal connectivity during integration of new context information in WM may thus serve as a potential biomarker to uncover underlying genetic mechanisms associated with information integration deficits in delusions.
Our results are consistent with a model of hierarchical posterior-cortical-to-PFC networks (14, 15) that integrates feed-forward incorporation of new relevant context information (1, 6, 7, 9, 34). Dysfunction of these circuits relate to the over-estimated likelihoods of pre-existing belief models and their over-reinforcement in delusions, and suggested to be implicated also in the underlying genetic architecture of psychosis (1). Prefrontal dysfunction and aberrant prefrontal-parietal connectivity have also long been implicated in schizophrenia and basic models for psychosis (20, 35–38).
A challenge to defining individual patient targets for potential therapeutics, however, is the substantial inter-individual variability, particularly in prefrontal cortical functional (18, 19) and effective connectivity (39). This would affect how one might define appropriate targets to benchmark new treatment strategies based on brain function. A promising ‘hyperalignment’ decoder approach recently utilized visual inferior temporal voxel patterns from surrogate subjects to predict that for an individual with phobias without exposure to feared stimuli, where the predicted data outperformed that derived at the individual-subject level (24). In terms of feedforward effective connectivity, we utilize an analogous strategy to infer target connectivity associated with context updating based on computationally inferring a patient’s connectivity pattern from models derived from the healthy participants’ data. We infer from these data what the optimized individual patient-specific “normal” feedforward parietal-prefrontal connectivity targets would be. To the extent that patient-specific deviation from optima at this cortical feedforward connectivity may be clinically relevant, we found that these predictions do relate to psychopathology associated with these same connections.
The parietal-prefrontal feedforward connectivity target engaged by context task stimuli similar to that reported here (1, 17), and prefrontal activation (9), have been associated with the cognitive architecture of delusions, as well as psychosis genetic risk (1). However, as it is likely there are other neural connections that influence delusions beyond our ROIs, a larger set of brain regions and larger samples should be explored. This should find some degree of feedback, and circular feedforward and feedback effects, conceptualized in the complex neural dynamics of delusions (40). Our pairwise DCMs across multiple regions (29) in the machine learning models is a pragmatic approach to the computational limitations in estimating large-scale task-modulated effective connectivity. However, this has drawbacks because each pair of brain interactions was considered independently of the others. Recent regression DCM approaches to large networks were focused on linear models that do not as yet include cognitive-task modulatory effects (41) critical for our approach. However, future developments such as task-modulated regression DCMs (41), and augmented modeling of neural interactions (42), could improve the biologic and clinical validity of approaches such as ours. Nevertheless, we posit feedforward parietal-prefrontal connectivity during information updating appears to be at least one target associated with the cognitive and genetic underpinnings of delusional psychopathology in schizophrenia, and perhaps in other psychotic conditions. The extent to which this circuit dysfunction might be causal would be important to determine, potentially with neurofeedback or other therapeutic strategies in functional MRI or EEG. Such new strategies may complement treatment of residual psychopathology, often refractory to pharmacology or cognitive-behavioral therapies.
Supplementary Material
Acknowledgements:
This work was funded by the National Institute of Mental Health Intramural Research Program (DRW, KFB), Lieber Institute for Brain Development, and National Institute of Mental Health grant R01MH101053 (HYT).
Footnotes
Disclosures: The authors report no financial relationships with commercial interests.
References:
- 1.Kaplan CM, et al. (2016) Estimating changing contexts in schizophrenia. Brain 139:2082–2095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Woodward TS, et al. (2014) Symptom Dimensions of the Psychotic Symptom Rating Scales in Psychosis: A Multisite Study. Schizophrenia Bulletin 40(Suppl_4):S265–S274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Corlett PR, Taylor JR, Wang XJ, Fletcher PC, & Krystal JH (2010) Toward a neurobiology of delusions. Prog Neurobiol 92(3):345–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garety PA & Freeman D (1999) Cognitive approaches to delusions: a critical review of theories and evidence. The British journal of clinical psychology / the British Psychological Society 38 ( Pt 2):113–154. [DOI] [PubMed] [Google Scholar]
- 5.Sass L & Byrom G (2015) Phenomenological and neurocognitive perspectives on delusions: A critical overview. World Psychiatry 14(2):164–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Adams RA, Stephan KE, Brown HR, Frith CD, & Friston KJ (2013) The computational anatomy of psychosis. Frontiers in Psychiatry 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fletcher PC & Frith CD (2009) Perceiving is believing: a Bayesian approach to explaining the positive symptoms of schizophrenia. Nat Rev Neurosci 10(1):48–58. [DOI] [PubMed] [Google Scholar]
- 8.Friston K (2012) The history of the future of the Bayesian brain. Neuroimage 62(2):1230–1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Corlett PR, et al. (2007) Disrupted prediction-error signal in psychosis: evidence for an associative account of delusions. Brain 130(9):2387–2400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Menon M, et al. (2011) Exploring the neural correlates of delusions of reference. Biol Psychiatry 70(12):1127–1133. [DOI] [PubMed] [Google Scholar]
- 11.Teufel C, et al. (2015) Shift toward prior knowledge confers a perceptual advantage in early psychosis and psychosis-prone healthy individuals. Proceedings of the National Academy of Sciences 112(43):13401–13406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Powers AR, Mathys C, & Corlett PR (2017) Pavlovian conditioning-induced hallucinations result from overweighting of perceptual priors. Science 357(6351):596–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Baker SC, Konova AB, Daw ND, & Horga G (2019) A distinct inferential mechanism for delusions in schizophrenia. Brain 142(6):1797–1812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Donoso M, Collins AGE, & Koechlin E (2014) Foundations of human reasoning in the prefrontal cortex. Science 344(6191):1481–1486. [DOI] [PubMed] [Google Scholar]
- 15.Brady TF & Alvarez GA (2011) Hierarchical Encoding in Visual Working Memory:Ensemble Statistics Bias Memory for Individual Items. Psychological Science 22(3):384–392. [DOI] [PubMed] [Google Scholar]
- 16.Garety P, et al. (2013) Neuropsychological functioning and jumping to conclusions in delusions. Schizophrenia Research 150(2):570–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fukuda Y, et al. (2019) Reduced parietofrontal effective connectivity during a working-memory task in people with high delusional ideation. Journal of psychiatry & neuroscience : JPN 44(3):195–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dubois J & Adolphs R (2016) Building a Science of Individual Differences from fMRI. Trends in Cognitive Sciences 20(6):425–443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Seitzman BA, et al. (2019) Trait-like variants in human functional brain networks. Proceedings of the National Academy of Sciences 116(45):22851–22861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tan HY, et al. (2006) Dysfunctional prefrontal regional specialization and compensation in schizophrenia. American Journal of Psychiatry 163:1969–1977. [DOI] [PubMed] [Google Scholar]
- 21.Tan HY, Choo WC, Fones CSL, & Chee MWL (2005) fMRI study of maintenance and manipulation processes within working memory in first-episode schizophrenia. American Journal of Psychiatry 162(10):1849–1858. [DOI] [PubMed] [Google Scholar]
- 22.Barch DM, et al. (2001) Selective deficits in prefrontal cortex function in medication-naive patients with schizophrenia. Archives of General Psychiatry 58(3):280–288. [DOI] [PubMed] [Google Scholar]
- 23.Cannon TD, et al. (2005) Dorsolateral Prefrontal Cortex Activity During Maintenance and Manipulation of Information in Working Memory in Patients With Schizophrenia. Arch Gen Psychiatry 62(10):1071–1080. [DOI] [PubMed] [Google Scholar]
- 24.Taschereau-Dumouchel V, et al. (2018) Towards an unconscious neural reinforcement intervention for common fears. Proceedings of the National Academy of Sciences. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Egan MF, et al. (2001) Relative risk for cognitive impairments in siblings of patients with schizophrenia. Biol Psychiatry 50(2):98–107. [DOI] [PubMed] [Google Scholar]
- 26.Tan HY, Callicott JH, & Weinberger DR (2007) Dysfunctional and compensatory prefrontal cortical systems, genes and the pathogenesis of schizophrenia. Cereb Cortex 17 Suppl 1:i171–181. [DOI] [PubMed] [Google Scholar]
- 27.Friston KJ, Harrison L, & Penny W (2003) Dynamic causal modelling. NeuroImage 19(4):1273. [DOI] [PubMed] [Google Scholar]
- 28.Glasser MF, et al. (2016) A multi-modal parcellation of human cerebral cortex. Nature 536:171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nicholson AA, et al. (2017) Dynamic causal modeling in PTSD and its dissociative subtype: Bottom–up versus top–down processing within fear and emotion regulation circuitry. Human Brain Mapping 38(11):5551–5561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Penny WD, Stephan KE, Mechelli A, & Friston KJ (2004) Comparing dynamic causal models. NeuroImage 22(3):1157. [DOI] [PubMed] [Google Scholar]
- 31.Psychiatric-Genetics-Consortium (2014) Biological insights from 108 schizophrenia-associated genetic loci. Nature 511(7510):421–427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Consortium TIS (2009) Common polygenic variation contributes to risk of schizophrenia and bipolar disorder. Nature 460(7256):748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zou H & Hastie T (2005) Regularization and variable selection via the elastic net. Journal of the Royal Statistical Society: Series B (Statistical Methodology) 67(2):301–320. [Google Scholar]
- 34.Friston K, et al. (2016) Active inference and learning. Neuroscience & Biobehavioral Reviews 68:862–879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Callicott JH, et al. (2003) Abnormal fMRI Response of the Dorsolateral Prefrontal Cortex in Cognitively Intact Siblings of Patients With Schizophrenia. American Journal of Psychiatry 160(4):709–719. [DOI] [PubMed] [Google Scholar]
- 36.MacDonald AW III, et al. (2005) Specificity of Prefrontal Dysfunction and Context Processing Deficits to Schizophrenia in Never-Medicated Patients With First-Episode Psychosis. Am J Psychiatry 162(3):475–484. [DOI] [PubMed] [Google Scholar]
- 37.Akil M, et al. (2003) Catechol-O-Methyltransferase Genotype and Dopamine Regulation in the Human Brain. J Neurosci 23(6):2008–2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Seamans JK & Yang CR (2004) The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 74:1–57. [DOI] [PubMed] [Google Scholar]
- 39.Jung K, et al. (2018) Effective connectivity during working memory and resting states: A DCM study. NeuroImage 169:485–495. [DOI] [PubMed] [Google Scholar]
- 40.Jardri R & Denève S (2013) Circular inferences in schizophrenia. Brain 136(11):3227–3241. [DOI] [PubMed] [Google Scholar]
- 41.Frassle S, et al. (2017) Regression DCM for fMRI. Neuroimage 155:406–421. [DOI] [PubMed] [Google Scholar]
- 42.Friston KJ, et al. (2019) Dynamic causal modelling revisited. NeuroImage 199:730–744. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.