Abstract
Background
There are sparse data on the utilization rate of implantable cardioverter-defibrillator (ICD) and its beneficial effects in Korean patients with heart failure with reduced left ventricular ejection fraction (LVEF).
Methods
Among 5,625 acute heart failure (AHF) patients from 10 tertiary university hospitals across Korea, 485 patients with reassessed LVEF ≤ 35% at least 3 months after the index admission were enrolled in this study. The ICD implantation during the follow-up was evaluated. Mortality was compared between patients with ICDs and age-, sex-, and follow-up duration matched control patients.
Results
Among 485 patients potentially indicated for an ICD for primary prevention, only 56 patients (11.5%) underwent ICD implantation during the follow-up. Patients with ICD showed a significantly lower all-cause mortality compared with their matched control population: adjusted hazard ratio (HR) (95% confidence interval [CI]) = 0.39 (0.16–0.92), P = 0.032. The mortality rate was still lower in the ICD group after excluding patients with cardiac resynchronization therapy (adjusted HR [95% CI] = 0.09 [0.01–0.63], P = 0.015). According to the subgroup analysis for ischemic heart failure, there was a significantly lower all-cause mortality in the ICD group than in the no-ICD group (HR [95% CI] = 0.20 [0.06–0.72], P = 0.013), with a borderline statistical significance (interaction P = 0.069).
Conclusion
Follow-up data of this large, multicenter registry suggests a significant under-utilization of ICD in Korean heart failure patients with reduced LVEF. Survival analysis implies that previously proven survival benefit of ICD in clinical trials could be extrapolated to Korean patients.
Trial Registration
ClinicalTrials.gov Identifier: NCT01389843
Keywords: Implantable Cardioverter-defibrillator, Heart Failure
Graphical Abstract
INTRODUCTION
Sudden deaths are common among patients with heart failure with reduced ejection fraction (HFrEF), and electrical disturbances, such as ventricular tachycardia, bradycardia, and asystole, are considered as major contributors to HFrEF.1 Implantable cardioverter-defibrillator (ICD) is an effective treatment for both bradycardia and potentially lethal ventricular arrhythmias and has proven its beneficial effects on the prevention of sudden cardiac deaths in patients with systolic dysfunction.2,3 In the MADIT-II trial, ICD implantation reduced sudden arrhythmic death in patients with ischemic heart failure.4 Moreover, it has been shown in the SCD-HeFT trial that ICD was better than medical treatment alone with respect to reducing mortality in patients with either ischemic or non-ischemic HFrEF.5 Thus, contemporary guidelines recommend ICD to prevent sudden death in patients with symptomatic HF and left ventricular ejection fraction (LVEF) ≤ 35%, after a sufficient trial of optimal medications, including angiotensin-converting enzyme inhibitor (ACE-I) and beta-blockers.6,7,8,9,10 Although several studies reported that the adherence to these guidelines improved the outcomes in Korean heart failure population,11,12 to the best of our knowledge, there are sparse data on the status of ICD utilization and its outcomes in this population. Therefore, we sought to evaluate the utilization of ICDs and its mortality in Korean systolic heart failure patients using Korean prospective multicentre cohort study registry.
METHODS
Study population and Korean acute heart failure (KorAHF) registry
The KorAHF registry was a prospective, multicentre cohort study that enrolled patients hospitalized for acute heart failure (AHF) from 10 tertiary university hospitals throughout the country from March 2011 to December 2014. Detailed information on the study design and its results have been previously reported [ClinicalTrial.gov NCT01389843].13 Patients with signs or symptoms of heart failure and either lung congestion, objective findings of left ventricular (LV) systolic dysfunction, or structural heart disease were eligible for the study. All patients were scheduled for follow-up at least 3 years after the index hospitalization.
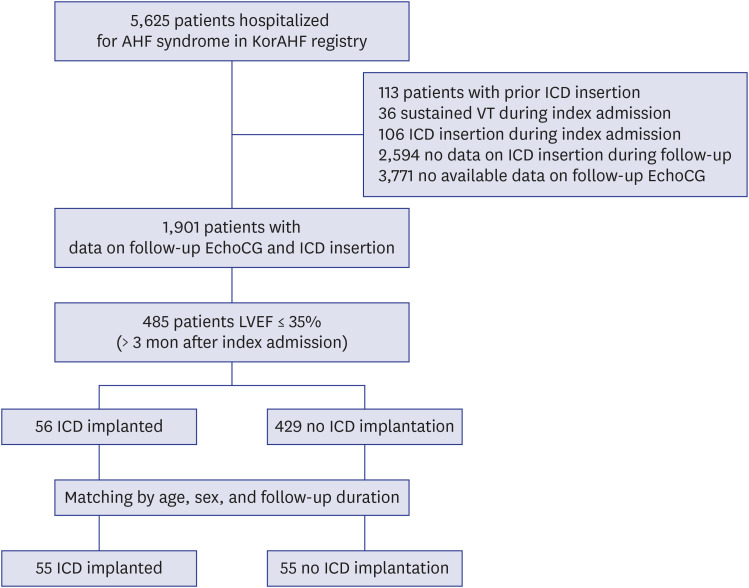
Among the total of 5,625 patients with AHF enrolled in the KorAHF registry, those with the potential for primary prevention ICD implantation were enrolled in this study. Exclusion criteria were as follows: 1) patients who had an ICD implanted before or during the index admission, 2) patients who experienced sustained VT during index admission, 3) patients who lacked follow-up data on ICD implantation and echocardiography after being discharged from index admission. Because the current guidelines recommend optimal medical treatment for at least 3 months prior to ICD implantation,2,3 patients showing reduced LVEF (≤ 35%) for at least 3 months after the index admission were considered eligible for enrolment in this study. A flow chart representing the selection of the study population in Fig. 1.
Fig. 1. Flowchart of the study population.
KorAHF = The Korean Acute Heart Failure registry, ICD = implantable cardioverter-defibrillator, EchoCG = echocardiography, LVEF = left ventricular ejection fraction.
Follow-up and study endpoints
Patients’ follow-up data were collected by the attending physician, with help of a clinical research coordinator, via a web-based system named the Clinical Data Management System (iCReaT) from the Korea National Institute of Health (NIH). Follow-up data were collected up to 60 months. Details for data collection protocol were described in previous studies.13,14,15 The primary endpoint of the KorAHF registry was the all-cause mortality rate. For the purpose of this study, we investigated the rate of ICD implantation among those considered eligible for ICD implantation after the discharge from the index admission. And the post-discharge all-cause mortalities were compared between the patients with (ICD group) and without ICD implantation (no-ICD group).
Analysis in matched population
Additional matched analysis was performed to address the issue of selection bias. First, for each patient from the ICD group, age- (± 2 years tolerance) and sex-matched patients were screened from the no-ICD group. If the follow-up duration of the patient in the no-ICD group was shorter than the time to ICD insertion of the corresponding patient in the ICD group, the patient was excluded. Then individually matched control was randomly selected among these candidates in 1:1 ratio. One patient in the ICD group was excluded from this process due to the lack of matched patient with comparable follow-up duration.
Statistical analysis
Data are presented as the numbers and frequencies for categorical variables and as the means ± standard deviations for continuous variables. To make comparisons among groups, the χ2 test (or Fisher's exact test) was used for categorical variables, and the unpaired Student's t-test (or Mann-Whitney U test) was used for continuous variables. Kaplan–Meier curves were plotted and compared, using the log-rank test for evaluation of post-discharge outcomes. A multivariable Cox proportional-hazards regression models were used to determine the independent effect of ICD insertion time on post-discharge outcomes, respectively. Variables found to be statistically significant (P < 0.1) in the univariate analysis were included in the multivariable model, except for variables with > 10% missing values or variables with a close association with other clinical variables. A two-sided probability value < 0.05 was considered significant. Statistical tests were performed using R programming version 3.6.0 (The R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org).
Ethics statement
The study protocol was approved by the Institutional Review Board (IRB) at each hospital and complied with the Declaration of Helsinki, and the requirement for written informed consent was waived (Seoul National University Bundang Hospital, IRB No. B-1104/125-014).
RESULT
Utilization of ICD in study population
Among the 485 patients whose follow-up echocardiography showed sustained reduced left ventricular systolic function (LVEF ≤ 35%), only 56 patients (11.5%) underwent ICD implantation during the follow-up. The median follow-up 1,046 days (interquartile range [IQR], 618–1,357 days), the median time to ICD implant was 539 days (IQR, 273–832). The median time to follow-up echocardiography from the index admission was 329 days (IQR, 211–413). Cardiac resynchronization therapy defibrillator (CRT-D) was used in 35.7% (20/56) of the ICD group, while CRT pacemaker (CRT-P) was used in only 0.2% (1/429) of the no-ICD group. The clinical characteristics according to the insertion of ICD during follow-up are provided in Table 1. Younger age (ICD vs. no ICD group; 62.9 ± 13.0 vs. 68.7 ± 13.9, P = 0.003), male sex (82.1% vs. 60.1%, P = 0.002), and more depressed LVEF (23.1 ± 7.7% vs. 27.9 ± 9.8%, P < 0.001) at the time of index admission were correlated with future ICD implantation. The prescription rate of beta-blocker or renin-angiotensin system inhibitor (RAS-inhibitor) at the time of discharge tended to be higher in patients who received ICD implantation (ICD vs. no-ICD group; 64.3% vs. 50.1%, P = 0.064 for beta-blocker, 89.3% vs. 76.5%, P = 0.045 for RAS-inhibitor). The etiology of heart failure and proportion of other comorbidities, such as hypertension, diabetes, cerebrovascular disease, and malignancy, were not different between groups.
Table 1. Baseline characteristics and utilization of ICD.
| Characteristics | ICD implant (n = 56) | No ICD (n = 429) | P value | |
|---|---|---|---|---|
| Age, yr | 62.9 ± 13.0 | 68.7 ± 13.9 | 0.003 | |
| Male | 46 (82.1) | 258 (60.1) | 0.002 | |
| BMI | 24.7 ± 4.1 | 23.2 ± 3.6 | 0.003 | |
| Hypertension | 29 (51.8) | 252 (58.7) | 0.397 | |
| Diabetes mellitus | 31 (55.4) | 195 (45.5) | 0.210 | |
| Ischemic heart disease | 20 (35.7) | 176 (41.0) | 0.537 | |
| Valvular heart disease | 3 (5.4) | 46 (10.7) | 0.309 | |
| Cerebrovascular disease | 10 (17.9) | 54 (12.6) | 0.376 | |
| Chronic kidney disease | 6 (10.7) | 82 (19.1) | 0.177 | |
| Malignancy | 3 (5.4) | 36 (8.4) | 0.600 | |
| COPD | 3 (5.4) | 48 (11.2) | 0.269 | |
| De novo heart failure | 27 (48.2) | 165 (38.5) | 0.208 | |
| AF | 9 (16.1) | 104 (24.2) | 0.233 | |
| Previous HF admission | 22 (39.3) | 190 (44.3) | 0.571 | |
| ICU admission | 26 (46.4) | 204 (47.0) | 1.000 | |
| Mechanical ventilation | 6 (10.7) | 43 (10.0) | 1.000 | |
| LVEF, % | 23.1 ± 7.7 | 27.9 ± 9.8 | < 0.001 | |
| LA dimension, mm | 47.9 ± 5.6 | 48.4 ± 9.1 | 0.536 | |
| Hemoglobin, g/dL | 13.6 ± 2.2 | 12.4 ± 2.2 | < 0.001 | |
| Serum creatinine, mg/dL | 1.5 ± 1.2 | 1.6 ± 1.6 | 0.466 | |
| Discharge medication | ||||
| ACEi or ARB, % | 50 (89.3) | 328 (76.5) | 0.045 | |
| Beta-blocker, % | 36 (64.3) | 215 (50.1) | 0.064 | |
| CRT | 20 (35.7) | 1 (0.2) | < 0.001 | |
| Time to f/u echocardiography | 324 ± 120 | 321 ± 136 | 0.878 | |
Data are expressed as mean ± standard deviation or number (%).
ICD = indicates implantable cardioverter-defibrillator, BMI = body mass index, COPD = chronic obstructive pulmonary disease, AF = atrial fibrillation, HF = heart failure, ICU = intensive care unit, LVEF = left ventricular ejection fraction, LA = left atrium, ACEi = angiotensin-converting-enzyme inhibitors, ARB = angiotensin receptor blocker, CRT = cardiac resynchronization therapy.
Predictors for mortality during follow-up
The predictors for all-cause mortality were screened in the study population. These are presented in Table 2. ICD implantation was significantly correlated with favorable mortality outcome in univariate analysis, and also in multivariate analysis (Table 2). The hazard ratio was 0.30 (95% confidence interval [CI], 0.16–0.59; P < 0.001) and 0.43 (95% CI, 0.22–0.82; P = 0.011), before and after the adjustment for other variables, respectively. Older age, female sex, lower body mass index (BMI), hypertension, ischemic heart disease, chronic kidney disease, ADHF, previous history of HF admission, higher baseline LVEF, lower hemoglobin, higher serum creatinine level, no prescribed ACEi or ARB, and beta-blocker were also significantly correlated with the higher all-cause mortality in univariate analysis. In multivariate analysis, age, BMI, chronic kidney disease, previous HF admission, and no prescribed beta-blocker were significantly correlated with mortality.
Table 2. Predictors for all-cause mortality.
| Variables | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | |
| ICD insertion | 0.32 (0.17–0.60) | < 0.001 | 0.43 (0.22–0.82) | 0.011 |
| CRT insertion | 0.57 (0.25–1.29) | 0.177 | ||
| Age | 1.04 (1.03–1.05) | < 0.001 | 1.03 (1.01–1.04) | 0.001 |
| Male | 0.74 (0.56–0.99) | 0.043 | 1.05 (0.76–1.44) | 0.758 |
| BMI | 0.93 (0.90–0.97) | 0.001 | 0.95 (0.91–0.99) | 0.022 |
| Hypertension | 1.63 (1.21–2.19) | 0.001 | 1.23 (0.89–1.70) | 0.201 |
| Diabetes | 1.26 (0.95–1.67) | 0.110 | 1.24 (0.91–1.71) | 0.179 |
| Ischemic heart disease | 1.35 (1.02–1.79) | 0.038 | 0.91 (0.67–1.25) | 0.567 |
| Valvular heart disease | 0.70 (0.41–1.18) | 0.179 | ||
| COPD | 1.51 (0.99–2.30) | 0.056 | 1.13 (0.73–1.75) | 0.577 |
| Cerebrovascular disease | 1.27 (0.86 –1.89) | 0.227 | ||
| Chronic kidney disease | 2.59 (1.90–3.52) | < 0.001 | 2.07 (1.39–3.08) | < 0.001 |
| Malignancy | 1.31 (0.80–2.12) | 0.281 | ||
| ADHF (vs. de novo) | 1.66 (1.22–2.25) | 0.001 | 0.91 (0.57–1.45) | 0.692 |
| Previous HF admission | 1.83 (1.38–2.44) | < 0.001 | 1.76 (1.15–2.68) | 0.009 |
| Atrial fibrillation | 1.04 (0.75–1.46) | 0.797 | ||
| LVEF | 1.02 (1.00–1.03) | 0.023 | 1.00 (0.98–1.02) | 0.987 |
| LA | 1.01 (0.99–1.03) | 0.237 | ||
| Hemoglobin | 0.86 (0.81–0.92) | < 0.001 | 1.02 (0.91–1.06) | 0.632 |
| Creatinine | 1.07 (1.01–1.13) | 0.027 | 0.99 (0.88–1.11) | 0.884 |
| ICU admission | 1.04 (0.79–1.39) | 0.764 | ||
| Mechanical ventilation | 1.29 (0.83–1.99) | 0.255 | ||
| ACEi or ARBa | 0.63 (0.46–0.87) | 0.005 | 0.88 (0.63–1.23) | 0.440 |
| Beta-blockera | 0.50 (0.38–0.67) | < 0.001 | 0.56 (0.41–0.75) | < 0.001 |
HR = hazard ratio, CI = confidence interval, ICD = indicates implantable cardioverter-defibrillator, CRT = cardiac resynchronization therapy, BMI = body mass index, COPD = chronic obstructive pulmonary disease, ADHF = acute decompensated heart failure, HF = heart failure, LVEF = left ventricular ejection fraction, LA = left atrium, ICU = intensive care unit, ACEi = angiotensin-converting-enzyme inhibitors, ARB = angiotensin receptor blocker.
aMedications prescribed at discharge from index admission.
Comparison of mortality in matched population
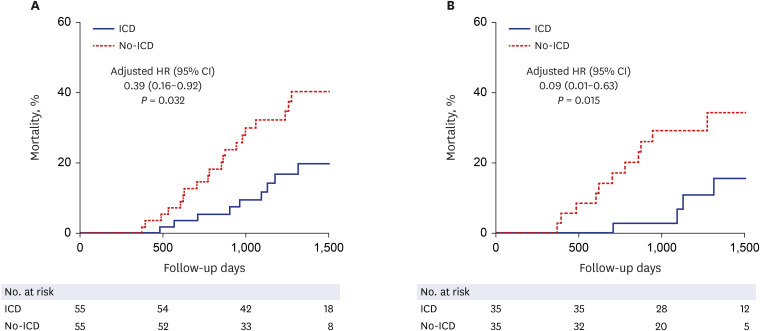
The baseline characteristics for the 1:1 matched population are presented in Table 3. All unmatched variables also became comparable between the ICD and no-ICD groups. Excluding the 20 CRT-D patients and their paired no-ICD group patients, the serum creatinine level was significantly correlated with ICD implantation during the follow-up period. The presence of chronic kidney disease and application of mechanical ventilation during the index admission also tended to be correlated with ICD insertion. After matching, Cox proportional survival analysis revealed that mortality was still significantly lower in the ICD group. HR was 0.45 (95% CI, 0.20–0.98; P = 0.044) and 0.39 (95% CI, 0.16–0.92; P = 0.032) before and after adjustment for other variables, respectively (Table 4). After excluding 20 CRT-D related pairs, the mortality rate was still lower in the ICD group compared with the no-ICD group before (HR [95% CI] = 0.30 [0.10–0.95], P = 0.041) and after adjustments (HR [95% CI] = 0.09 [0.01–0.63], P = 0.015). Kaplan-Meier estimates of survival in the overall and matched study population are shown in Fig. 2.
Table 3. Baseline characteristics in matched population.
| Characteristics | Including CRT-D | Excluding CRT-D | |||||
|---|---|---|---|---|---|---|---|
| ICD implant (n = 55) | No ICD (n = 55) | P value | ICD implant (n = 35) | No ICD (n = 35) | P value | ||
| Age, yr | 63.0 ± 13.1 | 62.9 ± 12.9 | 0.965 | 60.1 ± 13.3 | 60.2 ± 13.1 | 0.957 | |
| Male | 46 (83.6) | 46 (83.6) | 1.000 | 29 (82.9) | 29 (82.9) | 1.000 | |
| BMI | 24.8 ± 4.1 | 24.7 ± 3.9 | 0.896 | 24.6 ± 4.0 | 23.6 ± 2.3 | 0.508 | |
| Hypertension | 28 (50.9) | 31 (56.4) | 0.702 | 17 (48.6) | 19 (54.3) | 0.811 | |
| Diabetes mellitus | 30 (54.5) | 29 (52.7) | 1.000 | 19 (54.3) | 22 (62.9) | 0.627 | |
| Ischemic heart disease | 20 (36.4) | 21 (38.2) | 1.000 | 11 (31.4) | 9 (25.7) | 0.791 | |
| Valvular heart disease | 3 (5.5) | 4 (7.3) | 1.000 | 2 (5.7) | 3 (8.6) | 1.000 | |
| Cerebrovascular disease | 10 (18.2) | 9 (16.4) | 1.000 | 6 (17.1) | 7 (20.0) | 1.000 | |
| Chronic kidney disease | 5 (9.1) | 9 (16.4) | 0.391 | 1 (2.9) | 7 (20.0) | 0.060 | |
| Malignancy | 3 (5.5) | 4 (7.3) | 1.000 | 2 (5.7) | 2 (5.7) | 1.000 | |
| COPD | 3 (5.5) | 8 (14.5) | 0.204 | 1 (2.9) | 3 (8.6) | 0.607 | |
| De novo heart failure | 27 (49.1) | 21 (38.2) | 0.336 | 19 (54.3) | 12 (34.3) | 0.149 | |
| AF | 9 (16.4) | 16 (29.1) | 0.172 | 9 (31.4) | 11 (31.4) | 0.791 | |
| Previous HF admission | 22 (40.0) | 25 (40.0) | 0.700 | 12 (34.3) | 15 (42.9) | 0.623 | |
| ICU admission | 26 (47.3) | 29 (52.7) | 0.703 | 16 (45.7) | 19 (54.3) | 0.633 | |
| Mechanical Ventilation | 6 (10.9) | 4 (7.3) | 0.740 | 5 (14.3) | 0 (0.0) | 0.063 | |
| LVEF (baseline), % | 22.8 ± 7.6 | 25.1 ± 9.3 | 0.181 | 21.8 ± 7.2 | 23.0 ± 9.0 | 0.524 | |
| LA dimension, mm | 47.9 ± 5.7 | 50.1 ± 8.7 | 0.117 | 48.1 ± 5.5 | 50.2 ± 7.9 | 0.199 | |
| Hemoglobin, g/dL | 13.7 ± 2.2 | 13.3 ± 2.6 | 0.415 | 13.9 ± 2.3 | 13.5 ± 2.7 | 0.477 | |
| Serum creatinine, mg/dL | 1.4 ± 1.0 | 1.7 ± 1.2 | 0.193 | 1.2 ± 0.8 | 1.7 ± 1.3 | 0.038 | |
| Discharge medication | |||||||
| ACEi or ARB, % | 50 (90.9) | 46 (83.6) | 0.391 | 34 (97.1) | 29 (82.9) | 0.111 | |
| Beta-blocker, % | 36 (65.5) | 32 (58.2) | 0.556 | 26 (74.3) | 21 (60.0) | 0.309 | |
| CRT | 20 (36.4) | 0 (0) | < 0.001 | NA | NA | NA | |
Data are expressed as mean ± standard deviation or number (%).
CRT = cardiac resynchronization therapy, ICD = indicates implantable cardioverter-defibrillator, BMI = body mass index, COPD = chronic obstructive pulmonary disease, AF = atrial fibrillation, HF = heart failure, ICU = intensive care unit, LVEF = left ventricular ejection fraction, LA = left atrium, ACEi = angiotensin-converting-enzyme inhibitors, ARB = angiotensin receptor blocker.
Table 4. Predictors for all-cause mortality in matched population.
| Variables | Univariate analysis | Multivariate analysis | ||
|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | |
| ICD insertion | 0.45 (0.20–0.98) | 0.044 | 0.39 (0.16–0.92) | 0.032 |
| CRT insertion | 1.15 (0.46–2.84) | 0.769 | ||
| Age | 1.06 (1.02–1.10) | 0.005 | 1.12 (1.04–1.20) | 0.001 |
| Male | 0.36 (0.16–0.83) | 0.016 | 0.54 (0.17–1.70) | 0.290 |
| BMI | 0.91 (0.80–1.04) | 0.149 | 0.95 (0.84–1.07) | 0.381 |
| Hypertension | 1.16 (0.54–2.47) | 0.708 | 2.95 (1.10–7.91) | 0.031 |
| Diabetes | 1.91 (0.86–4.26) | 0.115 | 1.52 (0.58–3.95) | 0.392 |
| Ischemic heart disease | 1.79 (0.84–3.84) | 0.132 | 0.86 (0.63–1.18) | 0.358 |
| Valvular heart disease | NA | NA | ||
| COPD | 3.04 (1.05–8.82) | 0.041 | 2.82 (0.72–11.0) | 0.136 |
| Cerebrovascular disease | 1.92 (0.77–4.77) | 0.160 | ||
| Chronic kidney disease | 3.12 (1.32–7.40) | 0.010 | 1.49 (0.30–7.32) | 0.622 |
| Malignancy | 0.56 (0.08–4.11) | 0.566 | ||
| ADHF (vs. de novo) | 2.32 (0.98–5.49) | 0.056 | 2.21 (0.44–11.2) | 0.338 |
| Previous HF admission | 2.14 (0.99–4.62) | 0.052 | 1.71 (0.42–6.95) | 0.454 |
| Atrial fibrillation | 0.91 (0.37–2.27) | 0.841 | ||
| LVEF (baseline) | 1.00 (0.96–1.04) | 0.914 | 0.97 (0.92–1.02) | 0.224 |
| LA | 1.02 (0.96–1.09) | 0.440 | ||
| Hemoglobin | 0.79 (0.67–0.92) | 0.003 | 1.13 (0.93–1.36) | 0.211 |
| Creatinine | 1.39 (1.07–1.80) | 0.013 | 1.15 (0.69–1.91) | 0.588 |
| ICU admission | 0.78 (0.36–1.67) | 0.523 | ||
| Mechanical ventilation | 1.92 (0.66–5.56) | 0.229 | ||
| ACEi or ARBa | 0.35 (0.14–0.88) | 0.025 | 0.77 (0.23–2.52) | 0.661 |
| Beta-blockera | 0.49 (0.23–1.05) | 0.066 | 0.30 (0.12–0.73) | 0.008 |
HR = hazard ratio, CI = confidence interval, ICD = indicates implantable cardioverter-defibrillator, CRT = cardiac resynchronization therapy, BMI = body mass index, COPD = chronic obstructive pulmonary disease, ADHF = acute decompensated heart failure, HF = heart failure, LVEF = left ventricular ejection fraction, LA = left atrium, ICU = intensive care unit, ACEi = angiotensin-converting-enzyme inhibitors, ARB = angiotensin receptor blocker.
aMedications prescribed at discharge from index admission.
Fig. 2. All-cause mortality according to the ICD implantation during follow-up. (A) In the matched study population, and (B) after excluding patients with CRT-D and their matched controls.
ICD = implantable cardioverter-defibrillator, HR = hazard ratio, CI = confidence interval.
Subgroup analysis
We performed exploratory subgroup analysis in the matched study population. In the ischemic heart failure subgroup, the all-cause mortality rate during the follow-up period was significantly lower in the ICD insertion group than in the ICD non-insertion group (HR [95% CI] = 0.20 [0.06–0.72], P = 0.013), with a borderline interaction P value (interaction P = 0.069). Other than the etiology of HF, the effects of ICD insertion on mortality were not significantly different according to sex, diabetes mellitus, hypertension, chronic kidney disease, atrial fibrillation, or type of HF (Table 5).
Table 5. HRs for mortality with ICD insertion in subgroups of matched population.
| Variables | No. of patients | HR (95% CI) | P value | Interaction P value | |
|---|---|---|---|---|---|
| Etiology of HF | 0.069 | ||||
| Ischemic | 41 | 0.20 (0.06–0.72) | 0.013 | ||
| Non-ischemic | 69 | 0.81 (0.29–2.29) | 0.680 | ||
| Sex | 0.785 | ||||
| Male | 92 | 0.46 (0.19–1.08) | 0.074 | ||
| Female | 18 | 0.34 (0.06–1.74) | 0.195 | ||
| Diabetes mellitus | 0.897 | ||||
| Yes | 56 | 0.40 (0.15–1.04) | 0.059 | ||
| No | 54 | 0.43 (0.11–1.76) | 0.243 | ||
| Hypertension | 0.300 | ||||
| Yes | 58 | 0.67 (0.23–1.93) | 0.455 | ||
| No | 52 | 0.25 (0.08–0.84) | 0.025 | ||
| Chronic kidney disease | 0.209 | ||||
| Yes | 14 | 1.09 (0.24–4.96) | 0.914 | ||
| No | 96 | 0.36 (0.15–0.87) | 0.023 | ||
| Atrial fibrillation | 0.719 | ||||
| Yes | 28 | 0.30 (0.03–2.58) | 0.273 | ||
| No | 85 | 0.44 (0.19–1.01) | 0.052 | ||
| Type of heart failure | 0.271 | ||||
| De novo | 48 | 0.22 (0.04–1.08) | 0.062 | ||
| ADHF | 62 | 0.60 (0.25–1.44) | 0.251 | ||
Hazard ratios for all-cause mortality associated with ICD implantation.
HR = hazard ratio, ICD = indicates implantable cardioverter-defibrillator, CI = confidence interval, HF = heart failure, ADHF = acute decompensated heart failure.
DISCUSSION
In patients with sustained LV dysfunction, ICD implantation is recommended for the improvement of survival. The aims of this study were 1) to speculate the utilization rate of ICDs in Korean heart failure patients and 2) to evaluate the efficacy of ICDs in Korean heart failure patients, using a prospective large multi-center KorAHF registry. Among the 5,625 KorAHF patients, there were 485 identifiable ICD candidates for primary prevention based on their follow-up transthoracic echocardiography. Among them, only 56 (11.5%) patients underwent ICD implantation, implying under-utilization of ICDs in Korean LV dysfunction heart failure patients. In the matched analysis, all-cause mortality was significantly lower in patients with ICD implantation than in patients without ICD implantation. This suggests that there is indeed a beneficial effect of ICD in patients with advanced HF. This is in line with previous randomized trials and their meta-analysis conducted in western countries,16,17,18 which could be extrapolated to Korean patients.
There were several studies reporting the primary prevention ICD implantation rates in patients with advanced heart failure. The TRIUMPH registry data demonstrated that among the patients who survived myocardial infarction (MI) and had LVEF < 40% at discharge, only 35% underwent LVEF reassessment at 6 months and 2.4% received an ICD at 1 year.19 More recently, Pokorney et al.20 reviewed Medicare-insured patients with EF ≤ 35% during index myocardial infarction admission and reported that among those who underwent EF reassessment after discharge from the index admission, only 11% received an ICD within 1 year. In the Swedish Heart Failure Registry, ICD for primary prevention was also underused (ICD implantation rate 9.6%).21 Percent of LVEF improvement was not presented in these two papers. There was a recent report on the increasing annual number of ICD implantation in Korea, but the utilization rates among eligible population was not considered in this paper.22 The current study showed that the ICD implantation rate in Korean heart failure patients eligible for primary prevention ICD was about 11.5% during the overall follow-up. The ICD insertion rate at 1-year follow-up was lower than this, which was 6.6%. The ICD insertion rate in real-world practice might be even lower, considering that the KorAHF only enrolled patients from 10 major tertiary hospitals in Korea. In addition, it should be considered that our study only enrolled patients who were considered eligible for an ICD. Eligibility for an ICD was based on the echocardiographic results, at least 3 months apart from the index admission. Optimal medical treatment was assumed during this period, because the hospitals participating in this KorAHF study were tertiary hospitals with cardiologists specialized in heart failure treatment. In the subgroup of patients who were prescribed both RAS inhibitors and beta-blockers at discharge, ICD insertion rates were 9.1% at 1-year follow-up and 16.7% during overall follow-up, still demonstrating low utilization of ICDs. The low rate of ICD utilization can be attributed to factors from both patients and physicians. In this study, young age, male sex, and higher BMI were correlated with future ICD implantation. Other than underlying diseases, financial status, physicians’ attitude and patients’ preference for ICDs may have also influenced the utilization of ICD, although such data were not available. Several previous studies suggested that there may be a correlation between regional supply of cardiologists or electrophysiologists and ICD usage.23,24,25 However, at least for our study, such correlation was not the cause of low ICD usage, because hospitals participating in KorAHF were all tertiary hospitals with a plethora of qualified cardiologists and electrophysiologists. Referral to an ICD interventionist may be another hindering step. Gupta et al.26 demonstrated that electronic medical record based reminders improved both ICD discussion rate (44% to 67%) and implantation rate (2% to 24%), suggesting that improvement in this referring step can make a real difference in the utilization of ICD.
The beneficial effects of ICDs in patients with advanced systolic heart failure have not sufficiently been addressed in Asian patients, especially in Korean patients.4,5,16,17,18 Although there have been a few studies involving Korean patients with respect to ICD implantation,27,28,29,30 they present the outcomes in a single-arm observational environment, without any comparison to a control population. The present study enrolled patients with long-lasting systolic heart failure with ICD implantation and compared them against their match patients without ICD implantation. To the best of our knowledge, this is the first study demonstrating the benefits of ICDs in Korean patients with systolic heart failure. In the SCD-HeFT study, the benefit of ICD on the prevention of mortality was shown in patients with systolic heart failure, either ischemic or non-ischemic.5 However, a recent DANISH trial raised questions about the usefulness of ICD in non-ischemic cardiomyopathy.31 The subgroup analysis of the present study also demonstrated a trend of differential benefits of ICD according to the etiology of HF. The mortality of the ICD group was significantly lower than that of the control group in the ischemic HF subgroup (HR [95% CI] = 0.20 [0.06–0.72], P = 0.013), while the mortalities were comparable between the groups in the non-ischemic HF subgroup (HR [95% CI] = 0.81 [0.29–2.29], P = 0.680). Interaction P value was borderline (0.069). (Table 5) The benefits of ICD in patients receiving CRT have recently been questioned.32 To address the confounding effect of CRT, additional survival analysis was performed after excluding CRT-D patients and their paired control patients. The mortality was still lower in the ICD group than in the control group (Fig. 2B), suggesting that there is a clear beneficial effect of ICDs in the study population.
This study has several limitations. This is a prospective cohort study and there could be unaddressed biases such as sarcopenia in comparing the outcomes between the ICD and control groups. However, randomized trials there should be ethical issues in conduction randomized trials that evaluate the benefits of ICDs in this population. The matching process was adopted in this study in efforts to address this issue to some degree, though matching variables were limited due to the small number of the eligible patients. At least, there was no significant difference between the matched groups regarding the baseline characteristics including discharge medications. Information on prescription of the new drugs such as angiotensin receptor-neprilysin inhibitor or sodium-glucose cotransporter-2 inhibitor was not collected in the KorAHF registry. Mortality data were collected with assistance from the National Death Records, and survival analysis on specific cause of death, such as sudden cardiac death, was not feasible. After narrowing down the study population, the size of the matched population was rather underpowered to validate the effects of ICD in various subgroups. It was not possible to clarify the reasons for the low ICD utilization in this study.
In conclusion, follow-up data of this large multicenter KorAHF registry suggests a significant under-utilization of ICD in Korean heart failure patients with reduced LVEF. Survival analysis implies that benefits of ICD proven in clinical trials from western countries could be extrapolated to Korean patients.
Footnotes
Funding: This research was funded by grants from the Research of Korea Centers for Disease Control and Prevention (2010-E63003-00, 2011-E63002-00, 2012-E63005-00, 2013-E63003-00, 2013-E63003-01, 2013-E63003-02, and 2016-ER6303-00).
Disclosure: The authors have no potential conflicts of interest to disclosure
- Conceptualization: Choi DJ, Cho Y.
- Data curation: Park JJ, Lee HY, Kim KH, Yoo BS, Kang SM, Back SH, Jeon ES, Kim JJ, Cho MC, Chae SC, Oh BH, Choi DJ.
- Formal analysis: Cho Y, Cho SY.
- Investigation: Park JJ, Lee HY, Oh BH, Choi DJ.
- Methodology: Choi DJ, Cho Y.
- Software: Cho Y, Cho SY.
- Validation: Oh IY, Lee JH, Park JJ, Choi DJ.
- Writing - original draft: Cho Y, Cho SY.
- Writing - review & editing: Oh IY, Lee JH, Park JJ, Lee HY, Kim KH, Yoo BS, Kang SM, Back SH, Jeon ES, Kim JJ, Cho MC, Chae SC, Oh BH, Choi DJ.
References
- 1.Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB, et al. 2017 AHA/ACC/HRS Guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2018;72(14):e91–220. doi: 10.1016/j.jacc.2017.10.054. [DOI] [PubMed] [Google Scholar]
- 2.Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37(27):2129–2200. doi: 10.1093/eurheartj/ehw128. [DOI] [PubMed] [Google Scholar]
- 3.Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Jr, Colvin MM, et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J Am Coll Cardiol. 2017;70(6):776–803. doi: 10.1016/j.jacc.2017.04.025. [DOI] [PubMed] [Google Scholar]
- 4.Moss AJ, Zareba W, Hall WJ, Klein H, Wilber DJ, Cannom DS, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346(12):877–883. doi: 10.1056/NEJMoa013474. [DOI] [PubMed] [Google Scholar]
- 5.Bardy GH, Lee KL, Mark DB, Poole JE, Packer DL, Boineau R, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352(3):225–237. doi: 10.1056/NEJMoa043399. [DOI] [PubMed] [Google Scholar]
- 6.Garg R, Yusuf S Collaborative Group on ACE Inhibitor Trials. Overview of randomized trials of angiotensin-converting enzyme inhibitors on mortality and morbidity in patients with heart failure. JAMA. 1995;273(18):1450–1456. [PubMed] [Google Scholar]
- 7.Hjalmarson A, Goldstein S, Fagerberg B, Wedel H, Waagstein F, Kjekshus J, et al. Effects of controlled-release metoprolol on total mortality, hospitalizations, and well-being in patients with heart failure: the Metoprolol CR/XL Randomized Intervention Trial in congestive heart failure (MERIT-HF) JAMA. 2000;283(10):1295–1302. doi: 10.1001/jama.283.10.1295. [DOI] [PubMed] [Google Scholar]
- 8.Dargie HJ. Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: the CAPRICORN randomised trial. Lancet. 2001;357(9266):1385–1390. doi: 10.1016/s0140-6736(00)04560-8. [DOI] [PubMed] [Google Scholar]
- 9.Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med. 1991;325(5):293–302. doi: 10.1056/NEJM199108013250501. [DOI] [PubMed] [Google Scholar]
- 10.Packer M, Coats AJ, Fowler MB, Katus HA, Krum H, Mohacsi P, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344(22):1651–1658. doi: 10.1056/NEJM200105313442201. [DOI] [PubMed] [Google Scholar]
- 11.Choi KH, Lee GY, Choi JO, Jeon ES, Lee HY, Cho HJ, et al. Effects of angiotensin receptor blocker at discharge in patients with heart failure with reduced ejection fraction: Korean Acute Heart Failure (KorAHF) registry. Int J Cardiol . 2018;257:168–176. doi: 10.1016/j.ijcard.2017.12.002. [DOI] [PubMed] [Google Scholar]
- 12.Youn JC, Kim JJ, Kwon A, Lee HY, Kang SM, Baek SH. Dose response relationship of RAS blocker and beta-blocker in patients with acute heart failure syndrome: data from the Korean Acute Hart Failure (KorAHF) registry. J Card Fail. 2019;25(8):S142. [Google Scholar]
- 13.Lee SE, Cho HJ, Lee HY, Yang HM, Choi JO, Jeon ES, et al. A multicentre cohort study of acute heart failure syndromes in Korea: rationale, design, and interim observations of the Korean Acute Heart Failure (KorAHF) registry. Eur J Heart Fail. 2014;16(6):700–708. doi: 10.1002/ejhf.91. [DOI] [PubMed] [Google Scholar]
- 14.Kang J, Park JJ, Cho YJ, Oh IY, Park HA, Lee SE, et al. Predictors and prognostic value of worsening renal function during admission in HFpEF versus HFrEF: data from the KorAHF (Korean Acute Heart Failure) registry. J Am Heart Assoc. 2018;7(6):e007910. doi: 10.1161/JAHA.117.007910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee SE, Lee HY, Cho HJ, Choe WS, Kim H, Choi JO, et al. Clinical characteristics and outcome of acute heart failure in Korea: results from the Korean Acute Heart Failure registry (KorAHF) Korean Circ J. 2017;47(3):341–353. doi: 10.4070/kcj.2016.0419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hohnloser SH, Kuck KH, Dorian P, Roberts RS, Hampton JR, Hatala R, et al. Prophylactic use of an implantable cardioverter-defibrillator after acute myocardial infarction. N Engl J Med. 2004;351(24):2481–2488. doi: 10.1056/NEJMoa041489. [DOI] [PubMed] [Google Scholar]
- 17.Kadish A, Dyer A, Daubert JP, Quigg R, Estes NA, Anderson KP, et al. Prophylactic defibrillator implantation in patients with nonischemic dilated cardiomyopathy. N Engl J Med. 2004;350(21):2151–2158. doi: 10.1056/NEJMoa033088. [DOI] [PubMed] [Google Scholar]
- 18.Kolodziejczak M, Andreotti F, Kowalewski M, Buffon A, Ciccone MM, Parati G, et al. Implantable cardioverter-defibrillators for primary prevention in patients with ischemic or nonischemic cardiomyopathy: a systematic review and meta-analysis. Ann Intern Med. 2017;167(2):103–111. doi: 10.7326/M17-0120. [DOI] [PubMed] [Google Scholar]
- 19.Miller AL, Gosch K, Daugherty SL, Rathore S, Peterson PN, Peterson ED, et al. Failure to reassess ejection fraction after acute myocardial infarction in potential implantable cardioverter/defibrillator candidates: insights from the Translational Research Investigating Underlying disparities in acute myocardial infarction patients' health status (TRIUMPH) registry. Am Heart J. 2013;166(4):737–743. doi: 10.1016/j.ahj.2013.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pokorney SD, Miller AL, Chen AY, Thomas L, Fonarow GC, de Lemos JA, et al. Reassessment of cardiac function and implantable cardioverter-defibrillator use among medicare patients with low ejection fraction after myocardial infarction. Circulation. 2017;135(1):38–47. doi: 10.1161/CIRCULATIONAHA.116.022359. [DOI] [PubMed] [Google Scholar]
- 21.Schrage B, Uijl A, Benson L, Westermann D, Ståhlberg M, Stolfo D, et al. Association between use of primary-prevention implantable cardioverter-defibrillators and mortality in patients with heart failure: a prospective propensity score-matched analysis from the Swedish heart failure registry. Circulation. 2019;140(19):1530–1539. doi: 10.1161/CIRCULATIONAHA.119.043012. [DOI] [PubMed] [Google Scholar]
- 22.Roh SY, Choi JI, Kim MS, Cho EY, Kim YG, Lee KN, et al. Trends in the use of implantable cardioverter-defibrillators for prevention of sudden cardiac arrest: A South Korean nationwide population-based study. Pacing Clin Electrophysiol. 2019;42(8):1086–1094. doi: 10.1111/pace.13741. [DOI] [PubMed] [Google Scholar]
- 23.Fisher ES, Wennberg DE, Stukel TA, Gottlieb DJ, Lucas FL, Pinder EL. The implications of regional variations in Medicare spending. Part 2: health outcomes and satisfaction with care. Ann Intern Med. 2003;138(4):288–298. doi: 10.7326/0003-4819-138-4-200302180-00007. [DOI] [PubMed] [Google Scholar]
- 24.Fisher ES, Wennberg DE, Stukel TA, Gottlieb DJ, Lucas FL, Pinder EL. The implications of regional variations in Medicare spending. Part 1: the content, quality, and accessibility of care. Ann Intern Med. 2003;138(4):273–287. doi: 10.7326/0003-4819-138-4-200302180-00006. [DOI] [PubMed] [Google Scholar]
- 25.Matlock DD, Peterson PN, Heidenreich PA, Lucas FL, Malenka DJ, Wang Y, et al. Regional variation in the use of implantable cardioverter-defibrillators for primary prevention: results from the national cardiovascular data registry. Circ Cardiovasc Qual Outcomes. 2011;4(1):114–121. doi: 10.1161/CIRCOUTCOMES.110.958264. [DOI] [PubMed] [Google Scholar]
- 26.Gupta A, Gholami P, Turakhia MP, Friday K, Heidenreich PA. Clinical reminders to providers of patients with reduced left ventricular ejection fraction increase defibrillator referral: a randomized trial. Circ Heart Fail. 2014;7(1):140–145. doi: 10.1161/CIRCHEARTFAILURE.113.000753. [DOI] [PubMed] [Google Scholar]
- 27.Kim YH, Kim JS. Clinical characteristics in patients with implantable cardioverter-defibrillator (ICD) Korean Circ J. 2004;34(4):395–404. [Google Scholar]
- 28.Park KH, Lee CH, Jung BC, Cho Y, Bae MH, Kim YN, et al. Effectiveness of implantable cardioverter-defibrillator therapy for heart failure patients according to ischemic or non-ischemic etiology in Korea. Korean Circ J. 2017;47(1):72–81. doi: 10.4070/kcj.2016.0242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Uhm JS, Kim TH, Kim IC, Park YA, Shin DG, Lim YM, et al. Long-term prognosis of patients with an implantable cardioverter-defibrillator in Korea. Yonsei Med J. 2017;58(3):514–520. doi: 10.3349/ymj.2017.58.3.514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yang JH, Byeon K, Yim HR, Park JW, Park SJ, Huh J, et al. Predictors and clinical impact of inappropriate implantable cardioverter-defibrillator shocks in Korean patients. J Korean Med Sci. 2012;27(6):619–624. doi: 10.3346/jkms.2012.27.6.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Køber L, Thune JJ, Nielsen JC, Haarbo J, Videbæk L, Korup E, et al. Defibrillator implantation in patients with nonischemic systolic heart failure. N Engl J Med. 2016;375(13):1221–1230. doi: 10.1056/NEJMoa1608029. [DOI] [PubMed] [Google Scholar]
- 32.Marijon E, Leclercq C, Narayanan K, Boveda S, Klug D, Lacaze-Gadonneix J, et al. Causes-of-death analysis of patients with cardiac resynchronization therapy: an analysis of the CeRtiTuDe cohort study. Eur Heart J. 2015;36(41):2767–2776. doi: 10.1093/eurheartj/ehv455. [DOI] [PMC free article] [PubMed] [Google Scholar]