Abstract
Paragangliomas are uncommon neuroendocrine neoplasms that occur in characteristic locations. While parasympathetic paragangliomas are mainly located at the head and neck, sympathetic paragangliomas are mostly located below the neck. Among parasympathetic paragangliomas, pheochromocytomas are the most common. Ninety percent of cases of pheochromocytomas arise within the adrenal gland. We report a case of a 63-year-old woman with an extra-adrenal pheochromocytoma of the organ of Zuckerkandl detected by CT and MRI and subsequently confirmed by postoperative histology and immunohistochemistry.
Keywords: Pheochromocytoma, Paraganglioma, Zuckerkandl, MRI, Histopathology, Immunohistochemical
Introduction
Paragangliomas are uncommon neuroendocrine neoplasms that occur in characteristic locations associated with the paraganglionic system [1]. While parasympathetic paragangliomas are mainly located at the head and neck, sympathetic paragangliomas are mostly located below the neck [2]. Among parasympathetic paragangliomas, pheochromocytomas are the most common. Pheochromocytomas and other sympathetic paragangliomas are catecholamine secreting neuroendocrine tumors (NET) [3]. Classic clinical presentations of these tumors include headache, palpitation, and diaphoresis [4]. Approximately 85% of these masses occur within the adrenal glands, whereas 15% are located extra-adrenally along the sympathetic chain [5]. Up to 98% of extra-adrenal diseases is intra-abdominal, most frequently arise from the organ of Zuckerkandl [6]. Since the imaging features, clinical manifestations, histologic appearances of this entity are variable, making diagnosis a challenge. We report a rare case of extra-adrenal pheochromocytoma of the organ of Zuckerkandl, which is confirmed by postoperative histopathology immunohistochemistry.
Case report
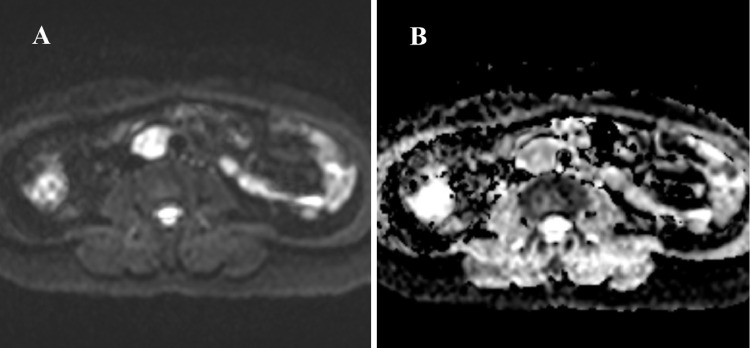
A 63-year-old female patient presented with complaint of epigastric pain for approximately 1 month. In the past 4 years, she was diagnosed with pancreatic carcinoma and had underwent a distal pancreatectomy and splenectomy. The patient also had a history of hypertension and gastroesophageal reflux disease. On physical exam, she appeared in no distress and was mildly hypertensive with otherwise normal vital signs. Abdominal exam revealed no abdominal mass or organomegaly. Laboratory investigations showed normal findings (complete blood count, serum electrolytes, urinalysis, thyroid, liver and renal functions). Imaging was obtained to further evaluate the abdominal pain. Contrast-enhanced abdominal CT scan revealed a 52 × 29 × 27mm well-defined, vividly enhancing soft tissue mass on the right side of aorta, closing to the bifurcation. MRI was then performed, showing a T1W-hypointense, T2W-hyperintense heterogeneous mass within the retroperitoneum, on the right side of aorta. Vivid enhancement after intravenous gadolinium administration was noted (Figs. 1 and 2). The mass showed increased intensity on diffusion-weighted imaging and reduced apparent diffusion coefficient values, which is consistent with restricted diffusion (Fig. 3). The lesion maintained its signal intensity on out-of-phase images (Fig. 4). Also, multiple punctate flow voids were noted on T1- and T2-weighted images. The patient underwent a complete en bloc removal of the mass. Microscopically, hematoxylin––eosin staining of the tumor tissue showed that the mass includes chief cells with basophilic cells and sustentacular cells. Cytoplasmic hyaline globules were also present. Immunohistochemical staining showed the tumor tissue was positive for chromogranin A, synaptophysin, S100, CD34 ,and vimentin. Meanwhile, the tumor tissue was negative for calretinin, HMB45, CK7, CK20, and desmin (Figs. 5–7). Collectively, these results indicated that the diagnosis of the tumor was extra-adrenal pheochromocytoma. Informed consent was given by the patient.
Fig. 6.
Immunohistochemical result show positive for chromogranin A (A), synaptophysin (B), S100 (C), IHC x100.
Fig. 1.
Axial, sagittal and coronal T2-weighted half-Fourier acquisition single-shot turbo spin-echo (HASTE) images demonstrate a hyperintense retroperitoneal mass (arrows) on the right side of aorta, adjacent to the bifurcation (arrowhead).
Fig. 2.
Axial unenhanced T1-weighted fat saturation MR image (B) shows a mass with avid enhancement (arrows) compared to unenhanced image (A).
Fig. 3.
This mass has high-signal intensity on DWI (A) and low ADC values (B), consistent with diffusion restriction.
Fig. 4.
Axial T1-weighted in-phase (A) and out-of-phase (B) MR images show lesion with no signal intensity drop on out-of-phase image.
Fig. 5.
Hematoxylin – eosin staining x100 (A) and x400 (B) of the tumor tissue demonstrates that the mass includes chief cells with basophilic cells and sustentacular cells, creating “Zellballen” pattern.
Fig. 7.
Calretinin (A), HMB45 (B) and desmin (C) negative, IHCx40.
Discussion
Pheochromocytomas (PCCs) and paragangliomas (PGLs) are rare catecholamine-secreting tumors originating from chromaffin cells of the adrenal medulla or extra-adrenal paraganglia [7]. These tumors arise within the adrenal glands in approximately 85% of cases. In the remaining 15% of cases, tumors originate from extra-adrenal paraganglionic chromaffin tissues and are referred to as extra-adrenal PCCs or PGLs [8,9]. Depending on the anatomic site and secretory products, PGLs are categorized into parasympathetic and sympathetic subtypes. According to the World Health Organization Classification of Tumors, updated in 2017, “A pheochromocytoma is an intra-adrenal sympathetic paraganglioma” [10,11]. Approximately 98% of sympathetic PGLs are found below the diaphragm, in association with the celiac, superior mesenteric, inferior mesenteric ganglia, and organ of Zuckerkandl. The organ of Zuckerkandl, which is a group of paraganglia located in the interaortocaval region from the superior mesenteric artery or renal arteries to aortic bifurcation, is the most frequent location [12]. Clinically, diverse presentations of PCC and PGL are related to the hypersecretion of catecholamines, typically producing paroxysmal symptoms, hypertension, and suprarenal or midline abdominal masses. The classic triad of extra-adrenal pheochromocytoma includes paroxysmal attacks of headache, palpitations, and diaphoresis [9,10,13]. Differential diagnosis includes other abdominal masses, especially malignant tumors. Pheochromocytoma has been called the “ten percent” tumors based on the fact that 10% of the cases are bilateral, 10% are extra-adrenal, 10% are clinically silent, and 10% of intra adrenal PCCs are malignant [14].
The imaging findings of PCCs are variable due to the presence of internal necrosis, cystic degeneration, and hemorrhage [15]. Unenhanced CT may show well-defined mass with attenuation ussually higher than 10HU, differentiating it from an adrenal adenoma. Contrast-enhanced CT may demonstrate vivid enhancement with variable washout patterns due to rich capillary network. Although MRI features of PCC are also variable because of the previously mentioned factors, the most common appearance is a mass with “light bulb” hyperintensity on T2W imaging and hypointensity on T1W imaging. Lesion typically show strong enhancement after gadolinium administration [16,17]. However, it is noted that a very high lesion signal intensity on T2W sequences is not pathognomonic for PCCs. Additionally, only approximately 65% of cases are correctively diagnosed as PCCs, whereas 35% are misclassified as malignant lesions or benign adenomas due to the lack of high signal intensity at T2W imaging. Punctate flow-voids representing tumor vessels are referred to as “salt-and-pepper” pattern characteristically seen on T1- and T2-weighted images. The tumors commonly do not contain a large amount of intracellular lipid so they will not demonstrate signal loss on out-of-phase sequences [16], [17], [18].
In histological studies, PCCs and PGLs are made up of chief cells or pheochromocytes and “supporting” cells or sustentacular cells. These cells may present several different patterns, “Zellballen” or cell-nesting and trabecular patterns are seen most [9,13,19]. Immunohistochemically, the chief cells of the tumor are positive for chromogranin A and synaptophysin, which are most common generic neuroendocrine markers, meanwhile, the sustentacular cells express S100 protein [4,20]. The most mentioned pathological differential diagnosis of PGLs presenting in the abdominal cavity are other NET, especially in the setting of concurrent pancreatic carcinoma, such as this case. Both PCCs/PGLs and NET show positive staining for chromogranin A and synaptophysin. However, the absence of positivity for cytokeratins such as CK7, CK20, CAM5.2,…help to distinguish them from NET [4].
The term “malignant pheochromocytoma” and “benign pheochromocytoma” from the 2004 WHO classification of endocrine tumors are no longer used. According to the 2017 WHO classification of endocrine tumors, all PCCs/PGLs could have metastatic potential and several factors may be used to stratify the risk. Currently, no individual scheme stratifying metastatic risk has been officially recommended. However, some immunohistochemical markers, such as Ki-67 proliferative index and SDHB, may be used to identify risk stratification [4,20,21]. A seminal study of PCCs and sympathetic PGLs by Linnoila et al. in 1990 demonstrated 4 adverse factors: extra-adrenal location, confluent necrosis, coarse nodularity, and absence of hyaline globules. The most powerful predictor among these features is extra-adrenal location, which is known to correlate with SDHB [19,20]. In 2005, a scoring system called GAPP was developed by Kimura et al. GAPP scores based on histological, immunohistochemical, and biochemical characteristics to predict the metastatic potential of tumors and the patients’ prognosis with tumors that metastasize [19].
The optimal treatment of pheochromocytoma is surgical resection. Before the operation, antihypertensive medications are often given to lower blood pressure and prevent hypertensive crisis during operation. In patient with malignant disease, palliative chemotherapy, and radiation therapy is indicated [8,22].
Conclusion
PCCs and PGLs are unusual tumors that occur in characteristic anatomic sites and are often detected by imaging examination. The disease presenting with a variability of clinical manifestations and imaging findings makes it challenging to diagnose correctly. Immunohistochemical studies have important roles in both the pathologic diagnosis and prognosis of PCC/PGL.
References
- 1.Asa S.L., Ezzat S., Mete O. The diagnosis and clinical significance of paragangliomas in unusual locations. J Clin Med. 2018;7(9) doi: 10.3390/jcm7090280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lee K.Y., Oh Y.-W., Noh H.J. Extraadrenal paragangliomas of the body: imaging features. Am. J. Roentgenol. 2006;187(2):492–504. doi: 10.2214/AJR.05.0370. [DOI] [PubMed] [Google Scholar]
- 3.Manger W.M., Gifford R.W. Pheochromocytoma. J Clin Hypertens (Greenwich) 2002;4(1):62–72. doi: 10.1111/j.1524-6175.2002.01452.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cheung V.K.Y., Gill A.J., Chou A. Old, new, and emerging immunohistochemical markers in pheochromocytoma and paraganglioma. Endocr Pathol. 2018;29(2):169–175. doi: 10.1007/s12022-018-9534-7. [DOI] [PubMed] [Google Scholar]
- 5.Das C.J., Baruah M.P., Baruah U.M. Radiological imaging in endocrine hypertension. Indian J Endocrinol Metab. 2011;15(Suppl4):S383–S388. doi: 10.4103/2230-8210.86984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Whalen R.K., Althausen A.F., Daniels G.H. Extra-adrenal pheochromocytoma. J. Urol. 1992;147(1):1–10. doi: 10.1016/s0022-5347(17)37119-7. [DOI] [PubMed] [Google Scholar]
- 7.Aygun N., Uludag M. Pheochromocytoma and paraganglioma: from epidemiology to clinical findings. Sisli Etfal Hastan Tip Bul. 2020;54(2):159–168. doi: 10.14744/SEMB.2020.18794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chen H., Sippel R.S., O'Dorisio M.S. The North American Neuroendocrine Tumor Society consensus guideline for the diagnosis and management of neuroendocrine tumors: pheochromocytoma, paraganglioma, and medullary thyroid cancer. Pancreas. 2010;39(6):775–783. doi: 10.1097/MPA.0b013e3181ebb4f0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Whalen R.K., Althausen A.F., Daniels G.H. Extra-adrenal pheochromocytoma. J. Urol. 1992;147(1):1–10. doi: 10.1016/s0022-5347(17)37119-7. [DOI] [PubMed] [Google Scholar]
- 10.Barnes L., Eveson J.W., Reichart P. World Health Organization Classification of Tumours. Pathology and Genetics of Head and Neck Tumours. IARC Press. Lyon; 2005. [Google Scholar]
- 11.Lam A.K. Update on Adrenal Tumours in 2017 World Health Organization (WHO) of Endocrine Tumours. Endocr Pathol. 2017;28(3):213–227. doi: 10.1007/s12022-017-9484-5. [DOI] [PubMed] [Google Scholar]
- 12.Cancer T.I.A., for R. on . World Health Organization; Lyon: 2005. Pathology and Genetics of Head and Neck Tumours. [Google Scholar]
- 13.Thompson L.D.R. Pheochromocytoma: Pathology Case Reviews. 2005;10(5):243–251. [Google Scholar]
- 14.Rha S.E., Byun J.Y., Jung S.E. Neurogenic tumors in the abdomen: tumor types and imaging characteristics. Radiographics. 2003;23(1):29–43. doi: 10.1148/rg.231025050. [DOI] [PubMed] [Google Scholar]
- 15.Katabathina V.S., Rajebi H., Chen M. Genetics and imaging of pheochromocytomas and paragangliomas: current update. Abdom Radiol. 2020;45(4):928–944. doi: 10.1007/s00261-019-02044-w. [DOI] [PubMed] [Google Scholar]
- 16.Blake M.A., Kalra M.K., Maher M.M. Pheochromocytoma: an imaging chameleon. Radiographics. 2004;24(suppl_1):S87–S99. doi: 10.1148/rg.24si045506. [DOI] [PubMed] [Google Scholar]
- 17.Shankar P., Heller M.T. Multi-modality imaging of pheochromocytoma. Radiology Case Reports. 2012;7(4):770. doi: 10.2484/rcr.v7i4.770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Leung K., Stamm M., Raja A. Pheochromocytoma: the range of appearances on ultrasound, CT, MRI, and functional imaging. Am. J. Roentgenol. 2013;200(2):370–378. doi: 10.2214/AJR.12.9126. [DOI] [PubMed] [Google Scholar]
- 19.Tischler A.S. Pheochromocytoma and extra-adrenal paraganglioma: updates. Arch Pathol Lab Med. 2008;132:13. doi: 10.5858/2008-132-1272-PAEPU. [DOI] [PubMed] [Google Scholar]
- 20.Tischler A.S., deKrijger R.R. 15 YEARS OF PARAGANGLIOMA: Pathology of pheochromocytoma and paraganglioma. Endocr. Relat. Cancer. 2015;22(4):T123–T133. doi: 10.1530/ERC-15-0261. [DOI] [PubMed] [Google Scholar]
- 21.Clarke M.R., Weyant R.J., Watson C.G. Prognostic markers in pheochromocytoma. Hum. Pathol. 1998;29(5):522–526. doi: 10.1016/s0046-8177(98)90070-3. [DOI] [PubMed] [Google Scholar]
- 22.Chrisoulidou A., Kaltsas G., Ilias I. The diagnosis and management of malignant phaeochromocytoma and paraganglioma. Endocr Relat Cancer. 2007;14(3):569–585. doi: 10.1677/ERC-07-0074. [DOI] [PubMed] [Google Scholar]