Abstract
When poorly water-soluble drugs are formulated in colloidal lipid emulsions, adequate stability of the emulsion must be ensured. The aim of this work was to investigate different aspects related to drug loading in order to gain a better understanding on how drugs affect the stability of phospholipid-stabilised emulsions. To obtain information on emulsion stability, a rapid and reproduceable shaking test was developed. A passive loading approach was applied for drug loading of the commercially available nanoemulsion Lipofundin® MCT/LCT 10% with seven drugs of different charge and localisation tendency within the emulsion system. Localisation of drug molecules in the droplet interface did not generally lead to destabilisation of the emulsion, whereas the charge of the drug was of decisive importance. Aspects such as the drug concentration, its influence on the pH and the impact of zeta potential changes had an influence on emulsion stability as well. Certain destabilising effects of drugs could be counteracted by modification of the pH. Lipofundin® MCT/LCT 10%, passively loaded with propofol, was compared with two commercially available propofol preparations. No negative effect of the passive loading procedure could be detected.
Keywords: Colloidal fat emulsions, Phospholipids, Emulsion stability, Zeta potential, Drug localisation, Liposomes
Graphical abstract
1. Introduction
Lipid nanoemulsions are used for parenteral nutrition and as delivery system for poorly water-soluble drugs. They are an important formulation option for many new drug candidates among which the fraction of poorly water-soluble substances has increased rapidly in recent years (Leeson, 2016). The application of some lipophilic drugs like propofol, which is classified by the WHO as an essential medicine, can be made possible through this formulation option (Baker, 2005; World Health Organization, 2019).
Since nanoemulsions are thermodynamically unstable systems that are only kinetically stabilised, it is of paramount importance to ensure their stability during the entire shelf life. However, phospholipid-stabilised emulsions can react sensitively to foreign substances such as ions or drugs (Daves and Groves, 1978; Gersonde et al., 2017; Sznitowska et al., 2001; Washington, 1996; Washington and Davis, 1987). Due to their good physiological compatibility, phospholipids are most frequently applied as stabilisers for parenteral emulsions (van Hoogevest and Wendel, 2014). An excess of phospholipids that is used for emulsion production but not required for the stabilisation of the interface of the emulsion droplets remains in the aqueous phase and forms liposomes (Westesen and Wehler, 1992). Since phospholipid-stabilised emulsions are electrostatically stabilised systems, factors that influence the zeta potential of the emulsion droplets are particularly important for emulsion stability. The addition of drugs represents such a factor (Han and Washington, 2005; Sznitowska et al., 2001; Washington, 1996). Thus, the influence of drugs on the stability of phospholipid-stabilised emulsions must be considered particularly carefully in order to assess the possibilities and limitations of their use as carrier systems for active pharmaceutical ingredients.
When lipid emulsions are used for the formulation of drugs, the drug can be localised at different sites in the emulsion system. Drug molecules may be localised in the aqueous phase, within the core or at the surface of the lipid droplets or in further colloidal structures formed by an excess of emulsifier (e.g. micelles or liposomes) (Berton-Carabin et al., 2013; Göke and Bunjes, 2017; Han and Washington, 2005; Kupetz and Bunjes, 2014; Watrobska-Swietlikowska and Sznitowska, 2006). Drug localisation at interfaces may play a particularly important role in colloidal emulsions due to the small droplet sizes and the resulting high interface-to-core ratio. The presence of drug molecules in the droplet interface might influence the function of the emulsifier molecules and thus affect the stability of the emulsion droplets (Watrobska-Swietlikowska and Sznitowska, 2006).
The aim of this study was to elucidate different aspects of drug loading on the physical stability of colloidal lipid emulsions. In order to extend the already existing knowledge, the focus was especially on the influence of drug localisation as well as on potential stabilising effects of the drug molecules. The investigations were based on commercially available, phospholipid-stabilised colloidal emulsions, as these represent a kind of standard for the stability required for the practical use of colloidal lipid emulsions. The use of a commercially available emulsion did, however, lead to the consequence that drug loading could not be performed the classical way (adding the lipophilic drug to the oil phase before emulsification). Thus, a passive loading approach was applied that allows loading of preformed carriers by incubation with the respective drug (Göke and Bunjes, 2018, Göke and Bunjes, 2017; Rosenblatt and Bunjes, 2017). Six drugs with different localisation tendencies within the emulsion system or with different charge were used to investigate the respective influence on emulsion stability. In order to develop a strategy to solve potential stability issues, the quantity of drug loading, the pH of the formulation and the zeta potential of the emulsion droplets were also taken into consideration.
Shaking tests are a well-established tool to perform accelerated studies on the physical stability of emulsions. Freeze-thaw cycles can also be used but are considered less relevant for intravenous emulsions and have been discussed critically with regard to the effect of agents located in the aqueous phase (Han and Washington, 2005). There are also alternative test methods such as, for example, the observation of droplet flocculation after electrolyte addition as indication for instability (Washington et al., 1991) or autoclaving to investigate emulsion stability by heat treatment (Sznitowska et al., 2001). For the investigations in the present study, a shaking test was developed that facilitated a fast stability check on highly stable emulsions. This test enabled to investigate both, effects that decreased or increased emulsion stability, without the need to add further reagents.
2. Material and methods
2.1. Materials
Tetrahydrofuran (HPLC grade), curcumin, cinnarizine and fenofibrate were purchased from Sigma-Aldrich (Steinheim, Germany). Betamethasone valerate was obtained from Caelo (Hilden, Germany). Sodium azide, hydrochloric acid and sodium hydroxide were ordered from Carl Roth (Karlsruhe, Germany), flufenamic acid from TCI (Zwijndrecht, Belgium) and dibucaine from Molekula (München, Germany). Phosphoric acid was from Merck (Darmstadt, Germany). Lipofundin® MCT/LCT 10% and Propofol-Lipuro® 10 mg/mL were from B.Braun (Melsungen, Germany), Disoprivan® 1% was from AstraZeneca (Wedel, Germany).
2.2. Composition of emulsions
Different commercially available emulsions were used in this investigation. In most experimental parts Lipofundin® MCT/LCT 10% (referred to simply as Lipofundin® in the following) was applied. Lipofundin® is a phospholipid-stabilised emulsion composed of 5% soybean oil, 5% medium chain triglycerides, 2.5% glycerol, 0.8% egg lecithin and unspecified quantities of α-tocopherol and sodium oleate (Braun, 2014; Gelbe Liste, 2019).
Furthermore, the propofol-loaded emulsions Propofol-Lipuro® and Disoprivan® with propofol contents of 1% were used. Apart from propofol, Propofol-Lipuro® is composed of 5% soybean oil, 5% medium chain triglycerides and unspecified concentrations of glycerol, egg lecithin and sodium oleate (B. Braun, 2018; Swiss Drug Compendium, 2019). Disoprivan® contains 10% soybean oil, 2.25% glycerol, 0.005% NaEDTA and NaOH for pH adjustment. 1.2% egg lecithin is used as emulsifier (AstraZeneca, 2019; Zeneca Pharmaceuticals, 2019).
2.3. Passive drug loading
Drug loading was carried out according to procedures described previously (Göke and Bunjes, 2017; Rosenblatt and Bunjes, 2017). Lipofundin® was incubated with the bulk material of the respective drugs in closed glass vials under shaking. Depending on the sample volume required for the subsequent test, between 10 mL and 30 mL emulsion were added. In order to ensure good homogenisation during the shaking procedure, the vials were selected in sizes to be filled between 20% and 60%. The drugs fenofibrate (feno), betamethasone valerate (BMV), curcumin (curc), propofol (prop), dibucaine (dibu), cinnarizine (cin) and flufenamic acid (fluf) were used in excess or at defined concentrations, specified in Table 1, Table 2. The structures and physicochemical properties of the drugs are given in Table 3. During the loading period all samples were agitated on a horizontal shaker IKA Vibrax MS3 digital (IKA, Staufen, Germany) with a rotation speed of approximately 300 rpm at 20 °C. The respective incubation times (Table 1, Table 2) depended on the speed of drug loading and the intended final drug concentration. For example, a short loading time of 2 h was chosen for BMV loading because preliminary tests had demonstrated that BMV reached its maximum drug load within approximately 15 min. If necessary, excess of drug was filtered off by 0.45 μm PVDF filters (Carl Roth, Karlsruhe, Germany). Control experiments confirmed for all drugs that there was no considerable loss of drug resulting from adsorption on the filter material.
Table 1.
Conditions of the drug loading procedure applied for the emulsions used in the different shaking series referred to in the table.
| Drug | Concentration initially added | Time of drug loading | Filtration | Final concentration |
|---|---|---|---|---|
| General aspects of the shaking test | ||||
| Dibucaine | 1.5 mg/mL | 72 h | No | 1.5 mg/mL |
| Flufenamic acid | 1.5 mg/mL | 72 h | No | 1.5 mg/mL |
| Influence of drug localisation on emulsion stability | ||||
| Curcumin | 1.5 mg/mL | 40 h | Yes | 0.1 mg/mL |
| Betamethasone valerate | 5 mg/mL | 2 h | Yes | 0.4 mg/mL |
| Fenofibrate | 12 mg/mL | 72 h | Yes | 5.7 mg/mL |
| Propofol | 10 mg/mL | 72 h | No | 10 mg/mL |
| Stability of propofol-containing emulsions | ||||
| Propofol | 10 mg/mL | 72 h | No | 10 mg/mL |
| Influence of dissociable substances on emulsion stability | ||||
| Cinnarizine | 1.5 mg/mL | 72 h | Yes | 0.9 mg/mL |
| Flufenamic acid | 1.5 mg/mL | 72 h | Yes | 1.5 mg/mL |
| Dibucaine | 1.5 mg/mL | 72 h | Yes | 1.5 mg/mL |
| Influence of drug loading quantity on emulsion stability | ||||
| Dibucaine | 1.5 mg/mL | 72 h | No | 1.5 mg/mL |
| Dibucaine | 12 mg/mL | 72 h | Yes | 7.5 mg/mL |
| Flufenamic acid | 1.5 mg/mL | 72 h | No | 1.5 mg/mL |
| Flufenamic acid | 12 mg/mL | 72 h | Yes | 7.1 mg/mL |
| Compensation of drug loading effects through pH adjustment | ||||
| Dibucaine | 12 mg/mL | 72 h | Yes | 7.5 mg/mL |
| Flufenamic acid | 12 mg/mL | 72 h | Yes | 7.1 mg/mL |
Table 2.
Conditions of the drug loading procedure applied for the emulsions used in pH-zeta potential measurements.
| Drug | Concentration initially added | Time of drug loading | Filtration | Final concentration |
|---|---|---|---|---|
| Dibucaine | 12 mg/mL | 72 h | Yes | 7.7 mg/mL |
| Flufenamic acid | 12 mg/mL | 72 h | Yes | 6.9 mg/mL |
| Cinnarizine | 12 mg/mL | 72 h | Yes | 2 mg/mL |
| Fenofibrate | 12 mg/mL | 72 h | Yes | 5.9 mg/mL |
| Propofol | 10 mg/mL | 72 h | No | 10 mg/mL |
| Curcumin | 12 mg/mL | 72 h | Yes | 0.3 mg/mL |
Table 3.
Chemical structure and physicochemical properties of drugs (Scifinder, 2020).
| Drug | Structure | pKa | logP | Melting point [°C] |
|---|---|---|---|---|
| Dibucaine (Dibu) |  |
9.1, 12.9 | 4.8 | 64 |
| Cinnarizine (Cin) | 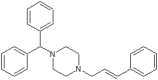 |
7.0 | 5.0 | 120 |
| Flufenamic acid (Fluf) |  |
3.7 | 5.2 | 134 |
| Propofol (Prop) |  |
11.0 | 3.7 | 18 (liquid at room temperature) |
| Curcumin (Curc) |  |
8.1 | 3.1 | 183 |
| Fenofibrate (Feno) |  |
- | 5.8 | 81 |
| Betamethasone valerate (BMV) | 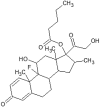 |
12.9 | 4.1 | 183 |
pKa and logP predicted by ACD/Labs Software V11.02 at 25 °C (Scifinder, 2020).
The drug concentrations (mg/mL) reported in this publication refer to the concentration per mL emulsion and not per mL lipid phase.
2.4. Drug quantification
Drug concentrations were determined by UV–Vis spectroscopy in a Specord 40 spectrometer (Analytik Jena, Jena, Germany). The drug-loaded emulsions were dissolved and diluted with tetrahydrofuran/water 9/1 (v/v) to an absorption for which a linear correlation was confirmed by a calibration curve. The measurements were performed at the following wavelengths: 240 nm (BMV), 252 nm (cin), 288 nm (feno), 327 nm (dibu), 347 nm (fluf) and 430 nm (curc). The solvent was measured as reference sample to adjust the baseline. Furthermore, the absorption of the dissolved unloaded emulsion in the same dilution as the sample was measured as blank value and was subtracted from the value of the loaded emulsion before the drug concentration was calculated.
Preliminary tests revealed that BMV precipitates from emulsions or emulsifier solutions within days. This precipitate is probably the less soluble hydrate (Göke and Bunjes, 2017). Because of that, BMV samples were analysed very quickly after saturation was obtained to ensure that no precipitation had yet occurred. After completion of all investigations, polarisation microscopy was applied to control that the BMV loading of the emulsion was stable over the period of investigation and that no drug precipitation had occurred. Polarisation microscopy was performed by a Leica DMLM light microscope with 100× and 200× magnification.
2.5. Shaking test
The shaking stability test was performed in a Retsch MM301 oscillating mill (Retsch, Haan, Germany), equipped with two PTFE beakers, perforated for Eppendorf® tubes. 1.5 mL Eppendorf® tubes were filled with 300 μL of emulsion and shaken with 25 Hz. Each formulation was investigated for different periods of time (at least at 3). The shaking experiment was performed in triplicate for each time period. Furthermore, for each time period, separate samples were prepared (i.e. for one emulsion system investigated after 3 different time periods of shaking, 9 tubes with separate emulsion samples were used). A shaking period was regarded as destabilising when in two out of three samples the emulsion droplets had lost their monomodal size distribution after this shaking time. The mono- or multimodal particle size distribution was evaluated by the results of laser diffraction analysis (cf. Section 2.6).
To adjust the pH of emulsions for stability measurements, 0.1 M NaOH or 1% H3PO4 was used.
2.6. Particle size determination
The particle size distribution was determined by laser diffraction in a HORIBA LA-960 (HORIBA, Oberursel, Germany), applying a manual fraction cell. Samples were diluted in bidistilled water to an appropriate transmission of less than 90%. Each sample was prepared once and measured in 3 runs. Particle size calculations were performed using a refractive index of 1.46 for the dispersed phase (imaginary part: 0.01) and 1.33 for the aqueous phase. The refractive index of 1.46 is the average of the refractive index of soybean oil of 1.475 and medium chain triglycerides of 1.446 (European Pharmacopeia, 2017a, European Pharmacopeia, 2017b). Each sample was prepared once and measured in triplicate. The evaluation is based on the Mie theory and calculated as the volume distribution of the emulsion droplets.
2.7. Zeta potential measurements
Zeta potential measurements were performed by electrophoretic light scattering in a Zetasizer Nano Series ZS (Malvern Panalytical, Kassel, Germany). Samples were diluted 1:400 with a 2.77% glycerol solution. This glycerol concentration is comparable with the composition of the aqueous phase of Lipofundin®. The pH of the glycerol solution was set to different values by 1 M HCl or 1 M NaOH before the dilution, to adjust different pH values in the resulting emulsion. The pH of the resulting dilutions of the emulsions was also determined and these pH values were used for the following analysis. The deviation in zeta potential measurements caused by the changed viscosity of a 2.77% glycerol solution compared to the viscosity of water is less than 10%. Therefore, the viscosity of water at 25 °C (0.8872 mPa*s), pre-set in the measurement instrument, was used as parameter for the calculation. Each sample was diluted once and measured in three runs. In the results section, the mean values and standard deviations of the measurements are displayed.
Due to the problem with drug precipitation, no pH-zeta potential curve was recorded for BMV (cf. Section 2.4).
All drug concentrations given for the emulsions used for the determination of the pH-zeta potential curves represent the concentrations of the emulsions before the dilution with glycerol solution.
2.8. pH measurements
pH measurements were performed directly in the sample using a Mettler Toledo FiveEasy pH meter with an InLab Semi-Micro electrode (Mettler Toledo, Gießen, Germany). Before each series of measurement, the electrode was calibrated with a 2-point calibration at pH 4.01 and 9.21.
3. Results and discussion
3.1. Development and general aspects of the shaking test
In order to investigate the effects of drug loading on the stability of emulsions, a shaking test was developed. The shaking test was subject to various requirements. The process had to allow the emulsions to be examined within a reasonable period of time. Furthermore, the test should allow a reliable identification of stabilising and destabilising effects on the emulsions. For that purpose, parameters had to be found which allow a reproducible comparison of emulsion stability. The newly developed shaking test combines all these aspects. This is exemplified in the following using data of emulsions loaded with two drugs that have different effects on emulsion stability. Three batches were each loaded with 1.5 mg/mL of either dibu (destabilising) or fluf (stabilising). More detailed investigations with dibu and fluf will be described in sections 3.5-3.7.
Without shaking, none of the emulsions showed signs of instability. Upon shaking, instabilities occurred that resulted in an increase of the characteristic particle size values (d90 in particular) as well as a loss in the monomodality of the particle size distributions. For emulsions loaded with the same drug, all three separately loaded batches lost their monomodality in 2 out of 3 replicates at the same time point (Fig. 1A-C and E-G). While the criterion “loss of monomodality” allowed a highly reliable identification of the destabilisation time, the particle size parameters “d90” and “mean” (arithmetic average) in unstable samples were different in each replicate and therefore could not be used for the reproducible identification of instabilities. The reason for this may be that the destabilisation of emulsions does not lead to a predictable, uniform increase in the size of the resulting particles. Thus, the particle size of unstable samples cannot be applied as reproducible parameter. Instead, the shaking period after which the emulsions in 2 out of 3 replicates displayed a multimodal particle size distribution was defined as reproducible instability criterion.
Fig. 1.
Shaking stability data of three emulsions loaded with 1.5 mg/mL dibucaine (A-C) or flufenamic acid (E-G). In stable samples, mean and d90 are very close to each other, thus, they are not always identifiable as separate points. D and H illustrate the conversion of the stability information of A-C and E-G into more compact bar charts. The symbol * indicates samples for which at least 2 out of 3 replicates were no longer monomodal. In the bar charts, stable intervals are shown in green, destabilising intervals in red. The shaking interval during which destabilisation occurred is marked with a colour gradient. Lines represent the investigated shaking times. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
As a consequence, only the time of destabilisation and not the extent was decisive for data evaluation. Thus, the graphs shown in the following will only display the results of the stability evaluation and not the underlying particle sizes. The information about the sample stability after the investigated time periods was pictured as bar charts for better visualisation (see Fig. 1D and especially Fig. 1H for illustration). Only the investigated shaking intervals and the stability results obtained for them are presented. In addition to the last “stable” and the first “unstable” shaking time, the shorter and longer investigated shaking intervals are also of relevance. These confirmed the validity of the destabilisation period as they affirmed that the first observed instability fits into a general trend and did not occur randomly at that sampling time. The investigated shaking times are displayed as lines in the bar charts. Areas shown in green represent time intervals at which the shaking of the emulsion did not yet result in instability. Red intervals represent shaking intervals at which the emulsion exhibited instability. The shaking interval between the last “stable” and the first “unstable” measuring point is marked with a colour gradient. The actual time point of destabilisation is located within this interval. A detailed description of the data evaluation and visualisation can be found in the Supplementary Information.
A destabilising effect of plastic on lipid emulsions is described in the literature (Driscoll et al., 2007). Its use as Eppendorf® tubes in the shaking experiments is, however, not expected to lead to a falsification of the results, since all samples were examined in the same containers. Although these containers might act as an additional stressor in the experiment, the effect would be similar for all samples.
3.2. Characterisation of Lipofundin® as reference sample
The original Lipofundin® emulsion was used as an initial reference sample. Its characterisation revealed a pH of 8.2 and a mean particle size of 155 nm with 100% of the particle size distribution being below 0.45 μm. It can therefore be assumed that the filtration of the loaded samples did not result in a considerable loss of emulsion droplets. Drug loading did not change the size of the emulsion droplets to a relevant extent. The mean particle sizes of unloaded and loaded Lipofundin® emulsions under investigation were 153 ± 3 nm.
The original Lipofundin® emulsion (pH 8.2) was destabilised in a shaking period of between 20 and 30 min (Fig. 2). A higher pH resulted in an increased stability of Lipofundin®. A reduction of the pH to 6.1 led to destabilisaation of the emulsion. A stability lowering effect of pH reduction on phospholipid-stabilised emulsions has already been described (Han et al., 2001; Washington et al., 1990). The dependence of the stability on the pH value correlated very well with the determined pH-zeta potential curve of Lipofundin® (Fig. 3). In agreement with literature data, the zeta potential became more negative with increasing pH while its absolute values decreased significantly with decreasing pH values (Washington, 1996). Disregarding the outlier measured at pH 6, it can be expected that the zeta potential falls below −30 mV near pH 6.5. Based on the DLVO theory, a zeta potential of at least −30 mV is assumed to be important for the long-term stability of charge-stabilised emulsions (Derjaguin, 1940; Riddick, 1968; Verwey, 1947). This explains why a lowering of the pH value to 6.1 led to a drastic destabilisation of the emulsion.
Fig. 2.

Stability of Lipofundin® at different pH values. Stable intervals are shown in green, destabilising intervals in red. The shaking interval during which destabilisation occurred is marked with a colour gradient. Lines represent the investigated shaking times. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
Fig. 3.

pH-zeta potential curves of unloaded Lipofundin® and Lipofundin® loaded with 5.9 mg/mL fenofibrate, 10 mg/mL propofol and 0.3 mg/mL curcumin. The standard deviations at the respective data points are usually not visible due to their small extent.
Deviating from the pH adjustment for the recording of pH-zeta potential curves, phosphoric acid was used instead of HCl to adjust an acidic pH in samples for stability tests. However, the results of the liposome experiments (see Supplementary Information, Supplement 2) indicated that the addition of phosphate, which took place in the form of the phosphate buffer, had no significant effect on the shaking stability of Lipofundin® at the concentration level used here. For this reason, the destabilising effect of a reduced pH in this experiment was attributed to the pH value reduction.
3.3. Influence of drug localisation on emulsion stability
To investigate the influence of drug localisation on the stability of Lipofundin®, four drugs were selected from previous studies, based on their different localisation tendencies. The drugs BMV and curc preferentially localise at the droplet interface, whereas feno and prop are known as matrix-localising (Francke and Bunjes, 2021; Göke and Bunjes, 2017; Kupetz and Bunjes, 2014). The pH values of the drug-loaded emulsions were in a range of pH 8.0 ± 0.1 and thus comparable with pH 8.2 of the unloaded emulsion. All loaded emulsions had a destabilisation interval corresponding to that of the unloaded emulsion (20 to 30 min) (see Supplementary Information, Supplement 3). Thus, an effect on emulsion stability was neither observed for the surface-localising nor for the matrix-localising drugs. It should be noticed that the drug loadings applied were different because BMV and curc have a significantly lower saturation concentration in the emulsion than feno and prop. In order to consider potentially different effects of drug load at saturation and below the saturation concentration, BMV and feno were loaded to the saturation concentration, whereas curc and prop were loaded at a concentration below their maximum concentration. Neither loading with the interface-affine drugs nor with the preferably matrix-localising drugs influenced the stability of the Lipofundin® emulsion in saturation or below.
The loaded concentrations of the interface-localising drugs were significantly lower than those of the matrix-localising drugs, so that no statement can be made as to whether a correspondingly high loading of interface-localising drugs would lead to an effect on the emulsion. However, a drug load comparable to that of feno or prop is not achievable with curc or BMV. A comparison at the same concentration would therefore only be possible at concentrations of 0.3 mg/mL. Since feno and prop had no effect on the stability of Lipofundin® at the significantly higher concentrations of 5.7 mg/mL (feno) and 10 mg/mL (prop) respectively, a deviating effect is not to be expected at lower concentrations.
The drug concentrations under examination were generally quite low. This is due to the low saturation concentration that most poorly water-soluble active ingredients reach in emulsions. However, a considerably higher loading cannot be achieved for these drugs and is therefore of no practical interest.
The finding that these drugs have no effect on emulsion stability is consistent with the pH-zeta potential curves of emulsions loaded with feno, prop and curc (Fig. 3). There was no considerable effect of loading with these drugs on the zeta potential of Lipofundin® over the entire pH range studied. The absence of a shift in the pH-zeta potential curve is surprising for curc, since due to its pKa of 8.1 a partial charge of the substance and a resulting shift of the curve to more negative areas would have been expected, when loaded to an emulsion (Washington, 1996). However, there is a known shift of the pKa of substances at negatively charged phospholipid interfaces above their pKa in aqueous solution. This effect is based on a higher charge density and a reduced dielectric constant in the region of negatively charged interfaces, which leads to an increased occurrence of protons in this region and a resulting shift in the pKa (Cistola et al., 1988; Desai et al., 1994; García-Soto and Fernández, 1983; Langner et al., 1995; Small et al., 1984). Prop should remain uncharged under the conditions applied here. Thus, all drugs examined in this section of the investigation should be present in an uncharged state.
Since BMV does not dissociate in the investigated pH range, no effect on the zeta potential would be expected for this substance, either. The pH-zeta potential curve of curc was recorded at a higher drug concentration (0.3 mg/mL) than used in the stability test (0.1 mg/mL) to magnify potential effects which were, however, not observed. The absence of an effect on emulsion stability despite interface localisation is in good agreement with results of a previous study on the influence of methyl paraben and benzyl alcohol (Han and Washington, 2005). In the shaking test used there, methyl paraben and benzyl alcohol did not have any effect on emulsion stability, in spite of a fraction of 17.1% (benzyl alcohol) and 24% (methyl paraben) of the respective drug content being present in the interface. The concentration of 0.1% methyl paraben used in these tests was even 2.5 times higher than that of BMV applied in the present stability tests.
3.4. Stability of propofol-containing emulsions
In order to investigate whether the procedure of passive drug loading has a distinct effect on the stability of emulsions, the stability of the passively loaded prop emulsion was compared to that of two commercially available emulsions. Propofol-Lipuro® was selected as commercially available product because it is produced by the same company as Lipofundin® and has a comparable composition as far as can be seen from the available information (Braun, 2014, B. Braun, 2018; Gelbe Liste, 2019; Swiss Drug Compendium, 2019). Disoprivan® was selected because results of stability tests on the product marketed as Diprivan® in the USA are available in the literature (Han et al., 2001). Since the lipophilic phase of Disoprivan® is not composed of an oil mixture but of pure soybean oil, it furthermore provided stability information on a differently composed system loaded with prop.
All emulsions, including the passively loaded Lipofundin® contained 10 mg/mL prop (AstraZeneca, 2019; B. Braun, 2018). Before shaking, all investigated emulsions had a monomodal particle size distribution in a size range of 130 to 161 nm. The mean particle size was 130 nm for Disoprivan® and 145 nm for Propofol-Lipuro® whereas that of the passively loaded Lipofundin® was 161 nm and thus equivalent to the 160 nm of the drug-free Lipofundin®. Lipofundin®, Disoprivan® and the passively prop-loaded Lipofundin® exhibited comparable shaking stability (Fig. 4). In contrast, Propofol-Lipuro® had a substantially lower stability.
Fig. 4.

Stability of different propofol-loaded (10 mg/mL) emulsions and drug-free Lipofundin®. Stable intervals are shown in green, destabilising intervals in red. The shaking interval during which destabilisation occurred is marked with a colour gradient. Lines represent the investigated shaking times. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
The passively loaded Lipofundin® thus had a significantly better stability than Propofol-Lipuro®, the commercially available product from the same company. On the one hand, this might be caused by a different composition or concentration of the phospholipids used for the two products, which is not stated in the publicly available information in detail. According to the pH effects described above, the pH may be another factor that may influence the stability of Propofol-Lipuro®. The passively loaded emulsion had a pH of 8.1, whereas Propofol-Lipuro® had a lower pH of 7.5. The remaining shelf-life of the emulsions can be excluded as a cause of differences in stability since both formulations reached their expiration date in the same month and the experiments were carried out approximately 10 months before this date. Variances in free fatty acid content are nevertheless possible and may be another aspect. For example, the two products might have been exposed to different conditions (e.g. heat) which may have led to the formation of free fatty acids. In the present investigations, the lower pH appears to be the most likely explanation for the lower stability of Propofol-Lipuro® compared to the passively loaded Lipofundin® emulsion.
For the higher stability of Disoprivan® as compared to Propofol-Lipuro®, the pH value (pH 7.4) cannot be used as an explanation as it is even below the pH value of all other investigated prop-emulsions. However, the cause for the better stability of Disoprivan® might be the composition or concentration of phospholipids. In order to obtain indications of a potential effect, the influence of additional liposomes on emulsion stability was investigated. The results indicated a decrease in emulsion stability with an increasing concentration of liposomes in the aqueous phase (Supplementary Information, Supplement 2). The increased stability of Disoprivan® compared to a generic product was already demonstrated by Han and Washington (Han et al., 2001). In that study, a relationship between emulsion stability and pH values or the presence of a preservative used in the generic product was taken into consideration. Both factors can be excluded in the experiment carried out here for the comparison of Disoprivan® and Propofol-Lipuro®, since the pH values were ruled out as the cause for the different stability of these two emulsions and Propofol-Lipuro® does not contain any preservatives.
The comparison between the passively loaded Lipofundin® emulsion and Disoprivan® illustrates that drug-loaded emulsions with a different lipid matrix can nevertheless exhibit comparable stability.
3.5. Influence of dissociable substances on emulsion stability
To investigate the influence of the drug charge on emulsion stability, Lipofundin® was loaded with dibu (1.5 mg/mL), cin (0.9 mg/mL) and fluf (1.5 mg/mL). The pH values of the drug-loaded emulsions were pH 9.0 (dibu), pH 8.0 (cin) and pH 6.7 (fluf). Even if the changed pKa at the interface is taken into account, these drugs are present in the charged state in the drug-loaded emulsions (Cistola et al., 1988; Langner et al., 1995; Small et al., 1984). In order to exclude special effects caused by saturation with drug (e.g. drug precipitation due to slight temperature or pH effects), all three drugs were only loaded to an extent that was clearly below their saturation concentration. The positively charged drugs dibu and cin led to a substantial destabilisation of the emulsion in the shaking test. In contrast, the fluf-loaded emulsion demonstrated increased stability in comparison to drug free Lipofundin® (Fig. 5A). The influence of charged substances on emulsion stability is already reported in the literature (Han et al., 2001; Washington, 1996). The addition of dissociable substances results in a shift of the zeta potential (Han and Washington, 2005; Washington, 1996; Washington and Davis, 1987). In contrast to previous studies, where the design of the shaking tests only allowed the investigation of destabilising effects of drugs (Han and Washington, 2005; Sznitowska et al., 2001), the shaking test developed for the study presented here allowed also to identify stabilising effects, as shown for fluf.
Fig. 5.
A) Influence of loading with dissociable drugs on Lipofundin® stability. Stable intervals are shown in green, destabilising intervals in red. The shaking interval during which destabilisation occurred is marked with a colour gradient. Lines represent the investigated shaking times. B) pH-zeta potential curves of Lipofundin®, loaded with 7.7 mg/mL dibucaine, 2 mg/mL cinnarizine and 6.9 mg/mL flufenamic acid. Arrows indicate the pH of the drug-loaded emulsions used in the stability experiments with emulsions containing 1.5 mg/mL dibucaine, 0.9 mg/mL cinnarizine and 1.5 mg/mL flufenamic acid. The standard deviations at the respective data points are usually not visible due to their small extent. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
In agreement with literature data, the zeta potential curve of the dibu- and cin-loaded emulsions shifted into the more positive range, whereas the zeta potential became more negative in the fluf-loaded emulsion (Fig. 5B) (Washington, 1996). However, the effect appeared not to be sufficient to explain the influence of drug loading on stability. If the expected zeta potential for the respective pH value is determined from the pH-zeta potential curves, the zeta potentials of the dibu-, cin- and fluf-loaded emulsions would all be in the range of −40 mV. In contrast to the observations, this zeta potential should lead to a good emulsion stability (Riddick, 1968). Moreover, the drug concentrations in the emulsions used for the shaking test were lower compared to the drug concentrations applied in the determination of pH-zeta potential curves. Therefore, it can be expected that the zeta potentials of the emulsions loaded with less drug were influenced by the drug loading to a lower extent. Thus, these results indicate that the effects on stability were not only attributable to changes in the zeta potential. For this reason, the influence of the extent of drug loading on the stability of the emulsion was also considered.
3.6. Influence of drug loading quantity on emulsion stability
In the literature, a concentration-dependent effect of drug loading on emulsion stability is described (Sznitowska et al., 2001). In order to investigate the influence of the quantity of drug loaded to the emulsion, dibu and fluf were loaded at 1.5 mg/mL as well as at their maximum concentration of 7.5 mg/mL (dibu) or 7.1 mg/mL (fluf), respectively. The dibucaine-loaded emulsions displayed a comparably poor stability at the two different drug concentrations (Fig. 5A). The fluf-loaded emulsion had a much lower stability when loaded to a higher concentration.
As Lipofundin® is an unbuffered system, loading with dissociable drugs had an influence on the pH value. Thus, the influence of the pH value on the stability on Lipofundin® has to be considered. The unloaded emulsion had a pH value of 8.2. Dibu-loading led to a pH of 9 (1.5 mg/mL) or 9.1 (7.5 mg/mL), respectively. According to the pH-zeta potential curve of a dibu-loaded emulsion (Fig. 5B) a less negative zeta potential of about −40 mV has to be expected for both concentrations. The zeta potential might thus be in the limit range of the zeta potential required for the stabilisation of emulsions (Riddick, 1968).
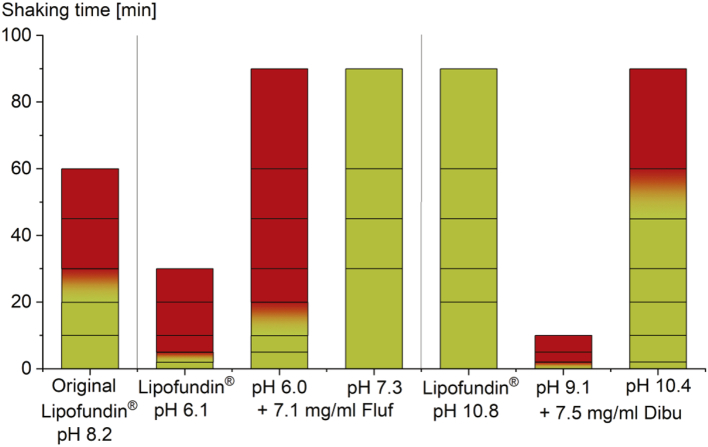
The loading with the lower fluf concentration caused a pH of 6.7, the higher concentration a pH of 6.0. However, the pH-zeta potential curve in Fig. 5B reveals that even at a pH of 6, a zeta potential of approximately −40 mV is still reached due to the significant reduction of the zeta potential by fluf. In order to investigate whether the observed effects persist even after exclusion of influence of the pH, the emulsion loaded with 7.1 mg/mL fluf was compared with the stability of the Lipofundin® emulsion, whose pH value had been adjusted to pH 6 corresponding to the pH of the drug-loaded emulsion (Fig. 6). The result indicates that the presence of fluf leads to a slight stabilisation of the emulsion even at this high concentration. Thus, loading with fluf partly counteracted the destabilisation of the emulsion triggered by the influence of a lower pH value. Therefore, stabilising effects of fluf on the emulsion are also recognisable after the exclusion of pH effects.
Fig. 6.
Influence of pH adjustment on 7.5 mg/mL dibucaine- or 7.1 mg/mL flufenamic acid-loaded Lipofundin®. Comparison of drug-free und drug loaded Lipofundin® at comparable pH. Stable intervals are shown in green, destabilising intervals in red. The shaking interval during which destabilisation occurred is marked with a colour gradient. Lines represent the investigated shaking times. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
The observed effect of emulsion stabilisation by the acidic drug fluf correlates very well with the observation that amino acids, at pH values where they are negatively charged, lead to a stabilisation of phospholipid-stabilised emulsions (Washington et al., 1991). The authors of the respective study assumed an effect caused by Van der Waals forces, since also their investigations did not reveal an exclusive correlation to electrostatic effects. In contrast, depending on the emulsifier used, a destabilising effect was observed for indomethacin (Trotta et al., 2002). However, since neither the zeta potential nor the pH value of the emulsion is stated in the publication, it is not certain whether indomethacin was negatively charged under the conditions studied. Our investigations pointed out that the destabilising effect of drug-induced pH reduction can predominate the stabilising effect of acidic substances. Thus, these effects have to be considered as well.
3.7. Compensation of drug loading effects by pH adjustment
From an application point of view, it is of high interest to determine which interventions can be taken to counteract the destabilising effect caused by basic drugs. Thus, it was investigated if the dependence of the emulsion stability on the pH value can be utilised to compensate negative effects of the drug loading by an increase of the pH.
In this investigation, the pH of the emulsion loaded with 7.5 mg/mL dibu was increased to 10.4. This high pH value was chosen to provoke a substantial effect by the pH-adjustment and is of course outside the physiological limits and not applicable in practice. According to the literature, a pH of 9 should not be exceeded for parenteral administration (Roethlisberger et al., 2017). The dibu-loaded emulsion with the increased pH value showed a distinctly longer destabilisation period compared with the same emulsion at pH 9.1 (Fig. 6). Since dibu has a pKa of 9.1, the drug molecules may already be partially undissociated at a pH value of 10.4, so that fewer charges may be present that can influence the stability. Nevertheless, compared with the unloaded Lipofundin® adjusted to a similar pH, a lower stability of the 7.5 mg/mL dibu-loaded emulsion was observed (Fig. 6). Thus, dibu destabilises the emulsion even at a higher pH value. However, pH adjustment may be a possibility to counteract and thus reduce the destabilising effects. As a prerequisite, the pH value of the drug-loaded and pH-adjusted emulsion would have to remain within the physiological range. Thus, pH adjustment might be a solution in the formulation of other drugs or for lower dibu concentrations. In addition to adjusting the pH value a change to a formulation with a more negatively charged phospholipid type and a resulting increase in the negative zeta potential may be used to compensate the destabilising effects of positively charged drugs (Washington, 1996).
A comparable stabilising effect of pH adjustment is also achievable when the pH of the highly loaded fluf emulsion is increased to 7.3 (Fig. 6). The pH of the fluf-loaded emulsion was below the pH of the original Lipofundin® emulsion, the fluf-loaded emulsion was, however, much more stable. This strengthens the conclusion that fluf increases the stability of the emulsion. Due to its low pKa value of 3.7, the fluf molecules are at least partially charged at both pH 6 and pH 7.3, even if an increase in pKa at interfaces is taken into consideration. However, the proportion of negatively charged fluf molecules is probably higher at pH 7.3, resulting in more additional negative charges on the emulsion droplets. Since the calculated solubility of fluf in water at 25 °C increases significantly from 0.024 g/L in unbuffered water at pH 4.18 to 5.1 g/L at pH 7 (Scifinder, 2020), a large part of the 7.1 mg/mL fluf is probably dissolved in the aqueous phase in the emulsions set to pH 7.3.
4. Conclusion
The developed shaking test enables a quick and reproducible investigation of drug loading-induced effects on emulsion stability. The results show that drug loading can influence the stability of phospholipid-stabilised colloidal lipid emulsions in a positive or a negative way. The investigations on prop-loaded emulsions demonstrate that the method of passive drug loading as such does not generally have negative effects on emulsion stability. On the other hand, the respective investigation reveals how important the exact composition of a formulation is for the stability of the emulsions and how apparently similarly formulated emulsions can behave significantly different. The localisation of drugs in the interface of the emulsion droplets does not seem to generally have a negative effect on the emulsion stability either. Instead, the charge of the drugs seems to be of greater importance for their influence on stability. Depending on the type of charge (positive or negative), the drug may have a stabilising or a destabilising effect on the emulsion. However, these effects seem to be not only due to loading-induced changes of the zeta potential of the emulsion droplets but may also be caused by the presence of the drug molecules as such. A possibility to improve the stability of an instable drug-loaded emulsion may be a balanced combination of drug loading and pH value. This adaption may allow the formulation of drugs in phospholipid-stabilised emulsions despite their destabilising effect. In practice, a pH adjustment can, of course, only be carried out within the limits of physiological compatibility and chemical stability.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgement
This work has been carried out within the framework of the SMART BIOTECS alliance between the Technische Universität Braunschweig and the Leibniz Universität Hannover, an initiative supported by the Ministry of Science and Culture of Lower Saxony, Germany.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ijpx.2020.100060.
Appendix A. Supplementary data
Supplementary material
References
- AstraZeneca . 2019. Fachinformation (Summary of Product Characteristics) Disoprivan®. [Google Scholar]
- Braun B. B. Braun; 2014. Fachinformation (Summary of Product Characteristics) Lipofundin® MCT/LCT 10% [Google Scholar]
- Braun B. B. Braun; 2018. Fachinformation (Summary of Product Characteristics) Propofol-Lipuro® 10 mg/ml. [Google Scholar]
- Baker M.T. Propofol-the challenges of formulation. Anesthesiology. 2005;103:860–876. doi: 10.1097/00000542-200510000-00026. [DOI] [PubMed] [Google Scholar]
- Berton-Carabin C.C., Coupland J.N., Elias R.J. Effect of the lipophilicity of model ingredients on their location and reactivity in emulsions and solid lipid nanoparticles. Colloids Surfaces A Physicochem. Eng. Asp. 2013;431:9–17. doi: 10.1016/j.colsurfa.2013.04.016. [DOI] [Google Scholar]
- Cistola D.P., Hamilton J.A., Jackson D., Small D.M. Ionization and phase behavior of fatty acids in water: Application of the Gibbs phase rule. Biochemistry. 1988;27:1881–1888. doi: 10.1021/bi00406a013. [DOI] [PubMed] [Google Scholar]
- Daves W.H., Groves M.J. The effect of electrolytes on phospholipid-stabilized soyabean oil emulsions. Int. J. Pharm. 1978;1:141–150. doi: 10.1016/0378-5173(78)90016-9. [DOI] [Google Scholar]
- Derjaguin B. On the repulsive forces between charged colloid particles and on the theory of slow coagulation and stability of lyophobe sols. Trans. Faraday Soc. 1940;35:203–215. doi: 10.1039/TF9403500203. [DOI] [Google Scholar]
- Desai S., Hadlock T., Messam C., Chafetz R., Strichartz G. Ionization and adsorption of a series of local anesthetics in detergent micelles: Studies of drug fluorescence. J. Pharmacol. Exp. Ther. 1994;271:220–228. [PubMed] [Google Scholar]
- Driscoll D.F., Silvestri A.P., Bistrian B.R., Mikrut B.A. Stability of total nutrient admixtures with lipid injectable emulsions in glass versus plastic packaging. Am. J. Heal. Pharm. 2007;64:396–403. doi: 10.2146/ajhp060062. [DOI] [PubMed] [Google Scholar]
- European Pharmacopeia . 9th ed. EDQM; Strasbourg, France: 2017. Monography Soybean Oil. [Google Scholar]
- European Pharmacopeia . 9th ed. EDQM; Strasbourg, France: 2017. Monography Medium Chain Triglycerides. [Google Scholar]
- Francke N.M., Bunjes H. 2021. Drug localization and its effect on the physical stability of poloxamer 188-stabilized colloidal lipid emulsions. In preparation. [DOI] [PubMed] [Google Scholar]
- García-Soto J., Fernández M.S. The effect of neutral and charged micelles on the acid-base dissociation of the local anesthetic tetracaine. BBA - Biomembr. 1983;731:275–281. doi: 10.1016/0005-2736(83)90019-6. [DOI] [PubMed] [Google Scholar]
- Gelbe Liste . Medizinische Medien Informations GmbH; 2019. Lipofundin®. https://www.gelbe-liste.de/produkte/Lipofundin-MCT-10-Emulsion-zur-Infusion-250ml Glasflasche_357386#! (accessed 11.21.19) [Google Scholar]
- Gersonde F., Eisend S., Haake N., Kunze T. Physicochemical compatibility and emulsion stability of propofol with commonly used analgesics and sedatives in an intensive care unit. Eur. J. Hosp. Pharm. 2017;24:293–303. doi: 10.1136/ejhpharm-2016-001038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Göke K., Bunjes H. Drug solubility in lipid nanocarriers: Influence of lipid matrix and available interfacial area. Int. J. Pharm. 2017;529:617–628. doi: 10.1016/j.ijpharm.2017.07.025. [DOI] [PubMed] [Google Scholar]
- Göke K., Bunjes H. Parameters influencing the course of passive drug loading into lipid nanoemulsions. Eur. J. Pharm. Biopharm. 2018;126:123–131. doi: 10.1016/j.ejpb.2017.05.009. [DOI] [PubMed] [Google Scholar]
- Han J., Washington C. Partition of antimicrobial additives in an intravenous emulsion and their effect on emulsion physical stability. Int. J. Pharm. 2005;288:263–271. doi: 10.1016/j.ijpharm.2004.10.002. [DOI] [PubMed] [Google Scholar]
- Han J., Davis S.S., Washington C. Physical properties and stability of two emulsion formulations of propofol. Int. J. Pharm. 2001;215:207–220. doi: 10.1016/S0378-5173(00)00692-X. [DOI] [PubMed] [Google Scholar]
- Kupetz E., Bunjes H. Lipid nanoparticles: Drug localization is substance-specific and achievable load depends on the size and physical state of the particles. J. Control. Release. 2014;189:54–64. doi: 10.1016/j.jconrel.2014.06.007. [DOI] [PubMed] [Google Scholar]
- Langner M., Isac T., Hui S.W. Interaction of free fatty acids with phospholipid bilayers. Biochim. Biophys. Acta. 1995;1236:73–80. doi: 10.1016/0005-2736(95)00037-4. [DOI] [PubMed] [Google Scholar]
- Leeson P.D. Molecular inflation, attrition and the rule of five. Adv. Drug Deliv. Rev. 2016;101:22–33. doi: 10.1016/j.addr.2016.01.018. [DOI] [PubMed] [Google Scholar]
- Riddick T.M. ZETA-METER, Inc.; New York: 1968. The Concept; pp. 2–3. (Control of Colloid Stability through Zeta Potential. With a Closing Chapter on its Relationship to Cardiovascular Disease). [Google Scholar]
- Roethlisberger D., Mahler H.C., Altenburger U., Pappenberger A. If euhydric and isotonic do not work, what are acceptable pH and osmolality for parenteral drug dosage forms? J. Pharm. Sci. 2017;106:446–456. doi: 10.1016/j.xphs.2016.09.034. [DOI] [PubMed] [Google Scholar]
- Rosenblatt K.M., Bunjes H. Evaluation of the drug loading capacity of different lipid nanoparticle dispersions by passive drug loading. Eur. J. Pharm. Biopharm. 2017;117:49–59. doi: 10.1016/j.ejpb.2017.03.010. [DOI] [PubMed] [Google Scholar]
- Scifinder Scifinder [WWW Document] 2020. https://scifinder.cas.org/scifinder/view/scifinder/scifinderExplore.jsf URL. accessed 6.15.20.
- Small D.M., Cabral D.J., Cistola D.P., Parks J.S., Hamilton J.A. The ionization behavior of fatty acids and bile acids in micelles and membranes. Hepatology. 1984;4:77S–79S. doi: 10.1002/hep.1840040814. [DOI] [PubMed] [Google Scholar]
- Swiss Drug Compendium Propofol-Lipuro® 10 mg/ml [WWW Document] 2019. https://www.compendium.ch/product/1006291-propofol-lipuro-1-1-g-100ml/mpro URL. accessed 11.21.19.
- Sznitowska M., Janicki S., Dabrowska E., Zurowska-Pryczkowska K. Submicron emulsions as drug carriers: Studies on destabilization potential of various drugs. Eur. J. Pharm. Sci. 2001;12:175–179. doi: 10.1016/S0928-0987(00)00115-9. [DOI] [PubMed] [Google Scholar]
- Trotta M., Pattarino F., Ignoni T. Stability of drug-carrier emulsions containing phosphatidylcholine mixtures. Eur. J. Pharm. Biopharm. 2002;53:203–208. doi: 10.1016/S0939-6411(01)00230-2. [DOI] [PubMed] [Google Scholar]
- van Hoogevest P., Wendel A. The use of natural and synthetic phospholipids as pharmaceutical excipients. Eur. J. Lipid Sci. Technol. 2014;116:1088–1107. doi: 10.1002/ejlt.201400219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verwey E.J.W. Theory of the stability of lyophobic colloids. J. Phys. Colloid Chem. 1947;51:631–636. doi: 10.1021/j150453a001. [DOI] [PubMed] [Google Scholar]
- Washington C. Stability of lipid emulsions for drug delivery. Adv. Drug Deliv. Rev. 1996;20:131–145. doi: 10.1016/0169-409X(95)00116-O. [DOI] [Google Scholar]
- Washington C., Davis S.S. Ageing effects in patenteral fat emulsions: the role of fatty acids. Int. J. Pharm. 1987;39:33–37. doi: 10.1016/0378-5173(87)90195-5. [DOI] [Google Scholar]
- Washington C., Athersuch A., Kynoch D.J. The electrokinetic properties of phospholipid stabilized fat emulsions. IV. The effect of glucose and of pH. Int. J. Pharm. 1990;64:217–222. doi: 10.1016/0378-5173(90)90271-5. [DOI] [Google Scholar]
- Washington C., Connolly M.A., Manning R., Skerratt M.C.L. The electrokinetic properties of phospholipid stabilized fat emulsions. V. The effect of amino acids on emulsion stability. Int. J. Pharm. 1991;77:57–63. doi: 10.1016/0378-5173(91)90301-4. [DOI] [Google Scholar]
- Watrobska-Swietlikowska D., Sznitowska M. Partitioning of parabens between phases of submicron emulsions stabilized with egg lecithin. Int. J. Pharm. 2006;312:174–178. doi: 10.1016/j.ijpharm.2006.01.005. [DOI] [PubMed] [Google Scholar]
- Westesen K., Wehler T. Physicochemical characterization of a model intravenous oil-in-water emulsion. J. Pharm. Sci. 1992;81:777–786. doi: 10.1002/jps.2600810812. [DOI] [PubMed] [Google Scholar]
- World Health Organization . 2019. World Health Organization Model List of Essential Medicines, 21st List [WWW Document]https://apps.who.int/iris/bitstream/handle/10665/325771/WHO-MVP-EMP-IAU-2019.06-eng.pdf?ua=1 URL. (accessed 7.29.20) [Google Scholar]
- Zeneca Pharmaceuticals Professional Information Brochure Diprivan® [WWW Document] 2019. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/019627_S035_DIPRIVAN_PRNTLBL.pdf URL.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material