Abstract
Background
Inflammation is involved in cerebral ischemia/reperfusion (I/R)-induced neurological damage. Saikosaponin A (SSa), extracted from Radix bupleuri, has been reported to exert anti-inflammatory effects. This article aimed to investigate whether SSa could ameliorate neuroinflammation mediated by ischemic stroke and the underlying mechanism.
Methods
Rat middle cerebral artery occlusion (MCAO) model was employed in this study, and the cognitive and motor functions were detected by behavioral tests. Inflammatory cytokines in the serum were detected by ELISA kits. The expression levels of Toll-like receptor 4 (TLR4), nuclear factor-kappa B (NF-κB), and high-mobility group box 1 (HMGB1) in the brain tissues were assayed with Western blot.
Results
Our results showed that SSa pretreatment could significantly reduce brain damage, improve neurological function recovery, and decrease the water content of brain tissues when compared with the model group. SSa pretreatment significantly reduced the serum HMGB1 level and downregulated the contents of inflammatory cytokines including tumor necrosis factor-α, interleukin-1 beta, and interleukin-6. Furthermore, SSa pretreatment could attenuate the decreased TLR4 and nucleus NF-κB in the brain of MCAO rats. The protein level of HMGB1 in the nucleus was significantly upregulated in the SSa pretreatment group.
Conclusion
Our results suggested that the pretreatment with SSa provided significant protection against cerebral I/R injury in rats via its anti-inflammation property by inhibiting the nucleus HMGB1 release.
Keywords: ischemia, neuroprotection, stroke, toll-like receptors, HMGB1
1. Introduction
Cerebral ischemia/reperfusion (I/R), or cerebral ischemic injury, is a common refractory disease with potentially serious and long-term disability. It is one of the main causes of death all over the world [1]. In cerebral I/R, the blood supply to the brain is initially limited and subsequently recovered with blood flow oxygenation [2,3]. It results in brain tissue death, such as cerebral infarction, caused by insufficient blood and oxygen supply due to cerebral vascular occlusion. Cerebrovascular dysfunction induced by cerebral I/R is considered to be a very important cause of nervous system damage in ischemic stroke. Finding effective methods to prevent cerebral I/R injury is of great significance in the ischemic stroke treatment.
It is well known that the pathophysiology of ischemic stroke includes not only inflammatory components but also thrombotic components. Due to the impeded blood supply, pathophysiological events, such as oxidative stress, abnormal energy metabolism, abnormal ion balance, glutamate excitotoxicity, inflammation, and apoptosis, occur in the disease [4]. Leukocyte recruitment is reported to occur in the global and focal cerebral I/R models. Cerebral I/R triggers the inflammatory cascade and subsequently causes cytotoxic neuronal cell death, which is also called secondary brain injury [5,6]. The inflammatory response associated with brain damage is primarily dependent on Toll-like receptor 2 (TLR2) and Toll-like receptor 4 (TLR4) [7]. The endogenous TLR ligand, high-mobility group box 1 (HMGB1), is involved in the activation of inflammatory cytokine expression in infiltrating macrophages [8].
HMGB1 release triggers inflammatory processes to exacerbate neuronal damage in the post-ischemic brain during acute ischemic brain injury induced by N-methyl-d-aspartate [9,10]. Consistently, infarct formation is inhibited, and the blood–brain barrier (BBB) is protected by the administration of the HMGB1 MAb in the postischemic brain [11]. In addition, HMGB1 knockdown mediated by siRNA, as well as HMGB1 inhibition mediated by HMGB1 A-box, significantly reduces cerebral infarction volume in the rat middle cerebral artery occlusion (MCAO) model [12,13]. Mechanically, the release of HMGB1 leads to the activation of nuclear factor-kappa B (NF-κB), which usually induces increased pro-inflammatory cytokines, adhesion molecules, and chemokines and exacerbates cellular oxidative stress [14,15]. It is reasonable to reduce HMGB1 to inhibit NF-κB activation. Therefore, to reduce the extracellular HMGB1 level and subsequently inhibit reactive oxygen species, inflammatory necrotic cells and inflammatory cytokine activity remain important effective targets for brain I/R injury treatment.
Saikosaponin A (SSa), derived from Radix bupleuri, is widely used in Chinese traditional medicine and has anti-inflammatory effects. SSa suppresses inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), and proinflammatory cytokines by inhibiting NF-κB activation in carrageenan-induced paw edema, showing effective anti-inflammatory activity [16]. Furthermore, SSa has also been demonstrated with anti-inflammatory and antifibrotic actions on CCl4-induced liver damage [17]. Saikosaponin-d (SSd) alleviates CCl4-induced acute hepatocellular injury, possibly by inhibiting oxidative stress and NLRP3 inflammasome activation in the HL-7702 cell line [18]. Another study showed that SSd attenuated lipopolysaccharide-induced depressive-like behaviors via inhibiting microglia activation and neuroinflammation [19]. However, the neuroprotective effect of SSa in MCAO rats has not been studied yet. As far as we know, our research is the first to investigate the effects of SSa on cerebral I/R injury. Therefore, the aim of this article was to explore whether SSa could ameliorate ischemic stroke-induced neuroinflammation and brain damage, and whether the underlying mechanism was related to the HMGB-1/TLR4/NF-κB signaling pathway.
2. Materials and methods
2.1. Animal model and drug administration
Male Sprague-Dawley rats (200 ± 20 g), obtained from Shanghai Laboratory Animal Center (Shanghai, China), were used to build the MCAO model to imitate ischemic re-infusion condition. SSa was purchased from Xi’an Imaherb Biotech Co., Ltd (Xi’an, China), with 99% purity and batch no. 83-67-0. The SSa was dissolved in 0.9% physiological saline as a stock solution, stored at −20°C, and diluted with medium with a specific dosage for each group. The animals were allowed to get access to food freely. The experimental procedures are illustrated in Figure 1. The animals were divided into five groups including three doses of SSa groups, animals in sham and MCAO (model) groups with no SSa treatment. Each group has 14 animals. Different doses of SSa were added to the chow diet 1 week before MACO, which were 0.001% (S1 group), 0.01% (S10 group), and 0.1% (S100 group) weight/food weight ratio, respectively. Through the midabdominal line incision, the operation exposed the right common carotid artery, external carotid artery, and internal carotid artery (ICA). A 0.26 mm nylon filament coated with silicon resin was introduced into the right ICA and advanced about 18–20 mm to the starting point of the middle cerebral artery to occlude. MCAO was considered successful after observing the left hemiparesis. After 1 h of MCAO, reperfusion of blood for 24 h was established by carefully withdrawing the nylon filament. The sham rats received similar surgical procedures only without nylon filament on the ICA to block the blood flow. None of the rats in the Sham group died (0 of 14 rats). Mortality was 28.6% (4 of 14 rats) in the S0 and S1 group, 14.3% (2 of 14 rats) in the S10 group, and 21.4% (3 of 14 rats) in the S100 group. Thus, the number of animals used in the study was as follows: 7 rats for the Sham subgroup, 5 rats for S0 and S1 subgroup each, 6 rats for the S10 subgroup, and 5–6 rats for the S100 subgroup. The dead rats were excluded from further analysis.
Figure 1.
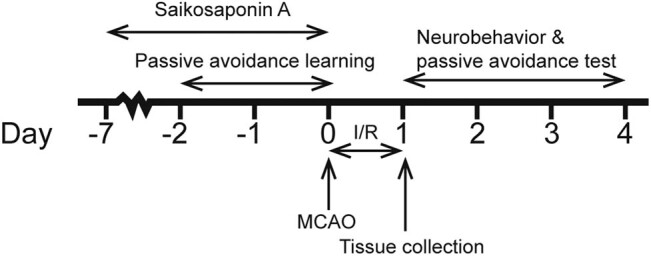
Experiment procedures.
Ethical approval: All experimental procedures were carried out in accordance with the People’s Republic of China Laboratory Animal Use and Care Law, and all experiments were approved by the Animal Care and Use Committee in the Second Hospital of Hebei Medical University.
2.2. Evaluation of motor function
Motor function was evaluated according to the Longa method by an examiner who was blinded to the experimental groups after the rats recovered from unconsciousness. The evaluation was scored as follows: 0 point, no symptoms of neurological deficits; 1 point, the contralateral forepaws could not be fully extended; 2 points, decreased resistance to lateral push; 3 points, unidirectional circling while walking; 4 points, could not walk by themselves; and 5 points, paralyzed. Higher scores indicated more severe neurological dysfunction.
2.3. Cognitive function examination
Rats show preference of darkness. The device was designed to have two rooms, one bright and the other dark. There was one door adjacent to the two rooms and a live metal mesh at the bottom of the darkroom. An animal would be subjected to a 0.5-mA electric shock when it entered the darkroom. Two days before the MCAO, the rats were placed in the bright room and turned to the darkroom. The time spent by the rat to enter the darkroom was measured and considered as an incubation period. The time spent in the dark chamber was recorded to assess learning and memory skills. If the rat did not enter the darkroom within 2 min, the learning exercise was completed. Longer step incubation periods or shorter darkroom times represented better cognitive function. The examination deadline was 5 min. The passive avoidance test was carried out blindly 4 days after MACO.
2.4. Measuring the cerebral infarct and edema
Tissues were collected 24 h after MCAO completed. After deep anesthesia with 10% chlorine hydrate, the rat brains were taken out and placed at −20°C for 15 min. After cutting into 2 mm thick slices on the ice platform, the brain slices were stained with 2% 2,3,5-triphenyltetrazolium chloride (TTC) at 37°C for 30 min and then fixed with 4% formaldehyde. The white area of cerebral infarction and the red non-ischemic area were measured using Image-Pro Plus using the formula: infarct volume% = ([reverse hemisphere volume − side hemisphere noninfarct area]/reverse hemisphere volume) × 100%. The brain was quantified with an electronic scale as wet weight and then dried to a constant weight in a desiccating oven at 105°C as dry weight. Total brain water content was calculated using the formula: ([wet weight − dry weight]/wet weight) × 100%.
2.5. Lactate dehydrogenase (LDH) activity detection
The ischemic brain tissues from each group were taken from the same area from bregma +0.7 to −4.3 mm after 24 h of MCAO. Detection of LDH activity was performed in the brain tissues using a commercial kit according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, China).
2.6. Measurement of inflammatory cytokines in serum
Commercial ELISA kits were used to detect serum inflammatory cytokine levels including interleukin-1 beta (IL-1β), interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), and HMGB1 following the standard manual (Nanjing Jiancheng Bioengineering Institute, China). After 24 h of MACO, 1 ml of blood samples obtained from the rat hearts was centrifuged at the speed of 2,000 × g for 10 min at 4°C, and then, the supernatants were used to detect inflammatory cytokines.
2.7. Western blot for TLR4, NF-κB, and HMGB1 expression
After behavioral testing, ischemic cortex tissue samples were collected for Western blot analysis. The total protein was lysed, and 20 μg protein samples were separated on 10% SDS polyacrylamide gels and then transferred to PVDF membranes. The membranes were blocked for 1 h in 5% skimmed dry milk buffer at room temperature and incubated overnight with the primary antibody (TLR4, NF-κB, and HMGB1; 1:1,000; Milliporesigma, USA). After being washed three times with TBST, the membranes were incubated for 2 h at room temperature with the secondary antibody conjugated with the spicy root peroxidase. The protein expression levels were detected by the electrochemical luminescence detection system and analyzed by the Image-J software.
2.8. Statistical analysis
Data were presented as mean ± SD. Multiple group comparisons were performed by one- or two-way analysis of variance (ANOVA), followed by Turkey’s test. P <0.05 was considered statistically significant.
3. Results
3.1. Effects of SSa on motor function of rats after I/R treatment
The Longa score was used to assess motor function in rats (Figure 2). Rats in the sham group showed good baseline motor function. After MCAO, I/R group rats had lower neurobehavioral scores at different stages than the sham group. After SSa treatment, motor dysfunction was rescued, and neurobehavioral scores were higher than the I/R group, especially in the S10 and S100 groups.
Figure 2.
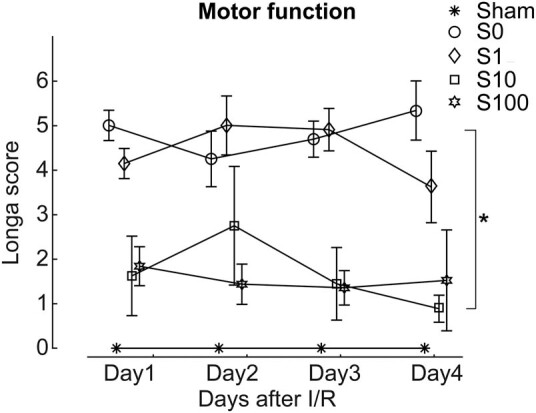
Saikosaponin A protected motor function induced by the I/R treatment. Animals delivered with 0.01% and 0.1% (group s10 and s100) showed better motor function when compared with the model group. However, 0.001% delivery of Saikosaponin A did not show protection effect. *p < 0.05, S10 compared with model; S100 compared with model.
3.2. Effects of SSa on the cognitive function of rats after I/R treatment
The passive avoidance test is an important method to evaluate cognitive function in rats, which avoids the bias caused by motor defects. After MCAO, I/R-treated rats showed a lower lag period and a higher darkroom time when compared with the sham group rats. S10 and S100 pretreatment increased delay time (Figure 3a) and reduced darkroom time (Figure 3b) when compared with the sham group rats, with statistically significant difference between each group.
Figure 3.
Saikosaponin A further ameliorated the learning defects induced by the I/R treatment. (a) The animals learned to stay in a lighted room to avoid electric shock in the darkroom. For 4 consecutive days, the model and S1 groups did not show an advance of learning progress. As comparison, the S10 and S100 groups have significant learning effects when compared with the model group. (b) Animals with learning ability spend less time in the darkroom. For 4 consecutive days, the model and S1 groups stayed longer than the Sham group. In contrast, the S10 and S100 groups have significant learning effects when compared with the model group. * p < 0.05, ** p < 0.01, comparing S10 and S100 in groups A and B with the control group.
3.3. SSa reduces brain ischemia and reperfusion damage in I/R rats
The TTC method was used to assess ischemia levels to demonstrate the effect of SSa on ischemia injury. After MACO, the areas of cerebral infarction in rats treated with S10 and S100 were significantly reduced when compared with the model group rats (Figure 4a). After ischemia and reperfusion, cerebral tissue edema increased the brain water content (Figure 4b). When treated with different doses of SSa, the brain water content decreased significantly when compared with that of the sham group rats.
Figure 4.
Saikosaponin A delivery reduced neurons damage induced by the I/R treatment. (a) Percentage of infarct volume after I/R treatment. Neuronal damage was significantly reduced in the S10 and S100 groups when compared with the model group. * p < 0.05, compared S10 and S100 with the model group. (b) The amount of water in brain tissue was significantly reduced in the S10 and S100 groups when compared with the model group. *p < 0.05, compared S10 and S100 with the model group.
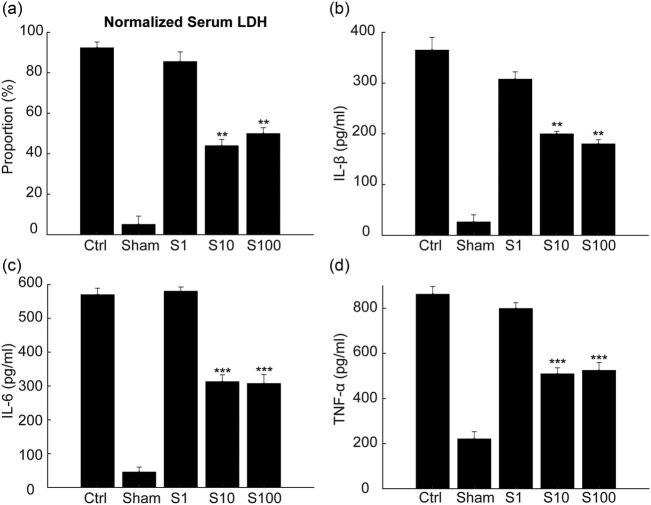
3.4. SSa reduces the increased LDH activities and inflammatory cytokine levels in the I/R rat serum
When an inflammatory response occurs in the tissue, the levels of LDH and inflammatory cytokines in the serum increase accordingly. Rats treated with I/R lacked blood oxygen supply, causing the inflammatory response that further leads to apoptosis and necrosis. Therefore, we tested the levels of LDH and inflammatory cytokines in the serum to verify the protective effect of SSa. The results showed that SSa reduced IL-1β, IL-6, TNF-α, and LDH levels in the serum of MACO rats, indicating that SSa inhibited the inflammatory response in the I/R rats (Figure 5a–d).
Figure 5.
Tissue damage and inflammatory reaction were decreased by administration of Saikosaponin A. (a) 24 h after MCAO, serum LDH levels were significantly decreased in S10 and S100 groups when compared with the model. ** p < 0.01, compared S10 and S100 with the model group. (b–d) ELISA results showed that Saikosaponin A treatment attenuated the increase in serum cytokines. IL-1β (b), IL-6 (c), and TNF-α (d) were all decreased in S10 and S100 groups when compared with the model group. ** p < 0.01, ***p < 0.0001, when compared S10 and S100 with the model group.
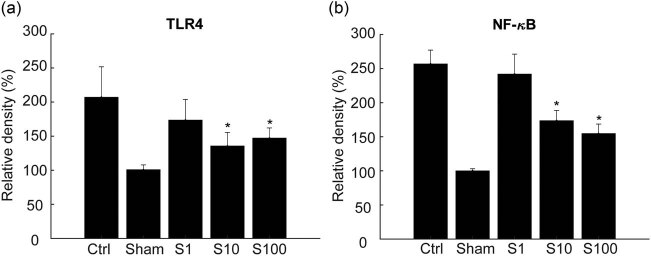
3.5. SSa downregulates the protein expression levels of TLR4 and NF-κB in I/R rats’ brain
The expression levels of TLR4 and NF-κB in ischemic brain tissue increased at 24 h after cerebral ischemia, but SSa significantly downregulated the expression of TLR4 and NF-κB (Figures 6a and b) as indicated by the Western blot analysis. Furthermore, it was found that the SSa treatment (0.01%) significantly decreased the expression of TLR4 and nucleus NF-κB 4 days after stroke (supplementary Figure S1a and b).
Figure 6.
TLR4 and NF-κB expression were reduced by Saikosaponin A treatment. Group S10 and S100 treatment significantly decreased the expression of TLR4 (a) and nucleus NF-κB (b) when compared with the model group. * p < 0.05, compared S10 and S100 with the model group.
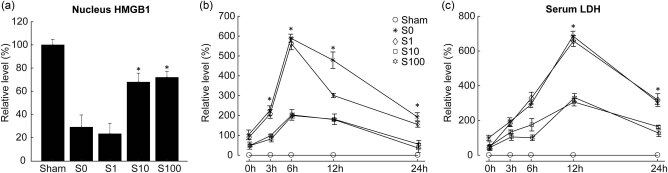
3.6. SSa inhibits nuclear HMGB1 release
HMGB1 released from the nucleus leads to the activation of nuclear NF-κB, resulting in upregulation of proinflammatory cytokines and exacerbation of cellular oxidative stress. Our results indicated that SSa inhibited the release of HMGB1 from the nucleus of the MACO rat brain by Western blot (Figure 7a). In addition, SSa pretreatment reduced HMGB1 release into the serum during reperfusion (Figure 7Bb) and also reduced LDH release (Figure 7c), indicating attenuated nerve damage caused by brain I/R. All these results indicated that SSa reduced brain I/R-induced neuronal damage, likely by inhibiting HMGB1 release.
Figure 7.
Saikosaponin A inhibited release of nucleus HMGB1, thus prevented the inflammatory effect. (a) Rats were sacrificed after MACO to detect the HMGB1 level. S10 and S100 groups restore the amount of HMGB1 as the Sham group, indicating that Saikosaponin A inhibited the release of HMGB1 from nucleus. (b and c) The serum levels of HMGB1 and LDH were detected. S10 and S100 groups reduced inflammation when compared with the model group, indicating that Saikosaponin A suppressed inflammation.
4. Discussion
Ischemic stroke is one of the most common neurological diseases leading to high mortality and disability and has not yet been effectively treated [1]. Therefore, it is necessary to explore novel treatment methods. Timely reperfusion can definitely prevent severe neurological deficits and death, but it may also exacerbate neuronal death and neurological deficits. A variety of pathophysiological mechanisms are related to these adverse reactions, such as intracellular Ca2+ elevation, energy metabolic failure, mitochondrial damage, and inflammatory response [4,20]. A growing body of evidence supports the view that inflammation is detrimental in I/R-induced damage. However, the exact mechanism is still unclear, with respect to how these inflammatory cytokines are regulated by physiological and pathological stimulating regulators.
HMGB-1, released from necrosis and dying cells in the ischemic brain, can trigger TLR4 activation [8]. Activated TLR4 undergoes a series of signal transductions, stimulating NF-κB nuclear translocation and then causing the release of inflammatory factors and I/R injury [21,22]. Therefore, therapeutic strategy targeting at HMGB-1/TLR4/NF-κB signaling may be a promising method to limit neuroinflammatory processes and alleviate stroke damage.
Acute neuroprotection after ischemia is very important, and the neuroprotective strategy aims to reduce ischemia-induced damage. Traditional Chinese Medicine has attracted much attention for its rich clinical experience in the cerebral I/R treatment [23,24]. The main bioactive compound isolated from the dried root of R. bupleuri is Saikosaponin, which has been shown to have many beneficial properties such as anti-inflammatory, antibacterial, antiviral, and immunomodulatory activities. SSa showed potent anti-inflammatory activity by inhibiting NF-κB activation, iNOS, COX-2, and proinflammatory cytokines [16]. Another study demonstrated that SSa treatment attenuated the destruction of the BBB and improved functional recovery after traumatic brain injury. The therapeutic effects were associated with SSa-induced inflammatory inhibition, likely through suppressing the MAPK signaling pathway [25]. Based on the above literature, whether SSa can alleviate cerebral I/R injury attracted our interest, and this study may provide a potentially novel agent for treating I/R injury. Our study used the rat MACO model to explore the role of SSa in inhibiting neuroinflammation and improving brain damage after ischemic stroke, as well as its potential pharmacological mechanisms.
Brain I/R injury can lead to cerebral edema and neurological deficits, accompanied by elevated serum LDH [26]. Our results showed that SSa attenuated the impaired motor and cognitive functions in the MACO rat (Figures 2 and 3), significantly reduced the infarct area (Figure 4a), and decreased the brain water content (Figure 4b). All these results indicated that SSa had significant neuroprotective effects on cerebral I/R damage.
Inflammation caused by tissue damage is a complex process of pathology and physiology involving inflammatory factors secreted by monocytes, macrophages, and microglia [27]. Inflammation has been verified to be involved in the pathogenesis of brain I/R damage. We further studied the effect of SSa on the inflammatory responses induced by I/R injury. The results showed that the SSa pretreatment inhibited the increased serum levels of IL-1β (Figure 5b), IL-6 (Figure 5c), and TNF-α (Figure 5d) as expected.
When cells are injured or near death, their membranes are damaged, and HMGB1 is released from these impaired cells. HMGB1 activates inflammatory response, leading to NF-κB transcription [28,29]. Western blot results showed that nuclear HMGB1 levels were reduced in the model group at 24 h after MCAO, and SSa reversed this response, indicating that SSa inhibited the release of HMGB1 from the nucleus. At the same time, SSa pretreatment decreased the serum HMGB1 level at 3, 6, 12, and 24 h after MACO (Figure 7b), which was also accompanied by delayed inhibition on LDH release by SSa, indicating the degree of neuroinjury (Figure 7c). Simultaneously, SSa pretreatment reduced serum HMGB1 levels at 3, 6, 12, and 24 h after MACO (Figure 7b), accompanied by a delay in SSa inhibition on LDH release as well, indicating the severity of nerve cell damage. All of these data confirmed our hypothesis that SSa might suppress inflammation by inhibiting HMGB1 release. Consistent with these results, brain I/R in the model group also increased the levels of TLR4 and nuclear NF-κB (p-P65), which were significantly extenuated by the SSa pretreatment, suggesting that the inhibition of NF-κB activation may be related to the function of SSa.
In the current study, we demonstrated that SSa exerted protective effect on cerebral I/R injury in a rat MCAO model. SSa pretreatment could significantly reduce brain damage, improve neurological function recovery, and decrease the water content in brain tissue. The SSa pretreatment significantly reduced the serum HMGB1 level and downregulated the contents of inflammatory cytokines. Furthermore, the SSa pretreatment could attenuate nucleus NF-κB increase, indicating that HMGB1 release was inhibited, and anti-inflammatory response might be involved in the neuroprotective effect of SSa on brain I/R injury.
In summary, this study has demonstrated the protective effects of SSa on cerebral I/R injury using a rat MACO model, and the effects of SSa are at least partially associated with its inhibition of HMGB1 release, attenuation of NF-κB activation, and suppression of subsequent activation of inflammatory responses.
Abbreviations
- BBB
blood–brain barrier
- COX-2
cyclooxygenase-2
- HMGB1
high-mobility group box 1
- I/R
ischemia/reperfusion
- ICA
internal carotid artery
- IL-6
interleukin-6
- IL-β
interleukin-1 beta
- iNOS
inducible nitric oxide synthase
- LDH
lactate dehydrogenase
- MCAO
middle cerebral artery occlusion
- NF-κB
nuclear factor-kappa B
- SSa
Saikosaponin A
- TLR2
Toll-like receptor 2
- TLR4
Toll-like receptor 4
- TNF-α
tumor necrosis factor-α
- TTC
2,3,5-triphenyltetrazolium chloride.
Footnotes
Conflict of interest: The authors state no conflict of interest.
References
- [1].Johnston SC, Mendis S, Mathers CD. Global variation in stroke burden and mortality: estimates from monitoring, surveillance, and modelling. Lancet Neurol. 2009;8:345–54. 10.1016/S1474-4422(09)70023-7. [DOI] [PubMed]; Johnston SC, Mendis S, Mathers CD. Global variation in stroke burden and mortality: estimates from monitoring, surveillance, and modelling. Lancet Neurol. 2009;8:345–54. doi: 10.1016/S1474-4422(09)70023-7. [DOI] [PubMed] [Google Scholar]
- [2].Margaill I, Plotkine M, Lerouet D. Antioxidant strategies in the treatment of stroke. Free Radic Biol Med. 2005;39:429–43. 10.1016/j.freeradbiomed.2005.05.003. [DOI] [PubMed]; Margaill I, Plotkine M, Lerouet D. Antioxidant strategies in the treatment of stroke. Free Radic Biol Med. 2005;39:429–43. doi: 10.1016/j.freeradbiomed.2005.05.003. [DOI] [PubMed] [Google Scholar]
- [3].Liesz A, Sun L, Zhou W, Schwarting S, Mracsko E, Zorn M, et al. FTY720 reduces post-ischemic brain lymphocyte influx but does not improve outcome in permanent murine cerebral ischemia. PLoS One. 2011;6:e21312. 10.1371/journal.pone.0021312. [DOI] [PMC free article] [PubMed]; Liesz A, Sun L, Zhou W, Schwarting S, Mracsko E, Zorn M. et al. FTY720 reduces post-ischemic brain lymphocyte influx but does not improve outcome in permanent murine cerebral ischemia. PLoS One. 2011;6:e21312. doi: 10.1371/journal.pone.0021312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Gong G, Bai S, Wu W, Hu L, Liu Y, Niu J, et al. Lrg participates in lipopolysaccharide preconditioning-induced brain ischemia injury via TLR4 signaling pathway. J Mol Neurosci. 2014;54:20–6. 10.1007/s12031-014-0240-8. [DOI] [PubMed]; Gong G, Bai S, Wu W, Hu L, Liu Y, Niu J. et al. Lrg participates in lipopolysaccharide preconditioning-induced brain ischemia injury via TLR4 signaling pathway. J Mol Neurosci. 2014;54:20–6. doi: 10.1007/s12031-014-0240-8. [DOI] [PubMed] [Google Scholar]
- [5].Li W, Suwanwela NC, Patumraj S. Curcumin by down-regulating NF-kB and elevating Nrf2, reduces brain edema and neurological dysfunction after cerebral I/R. Microvasc Res. 2016;106:117–27. 10.1016/j.mvr.2015.12.008. [DOI] [PubMed]; Li W, Suwanwela NC, Patumraj S. Curcumin by down-regulating NF-kB and elevating Nrf2, reduces brain edema and neurological dysfunction after cerebral I/R. Microvasc Res. 2016;106:117–27. doi: 10.1016/j.mvr.2015.12.008. [DOI] [PubMed] [Google Scholar]
- [6].Stoll G, Kleinschnitz C, Nieswandt B. Combating innate inflammation: a new paradigm for acute treatment of stroke? Ann N Y Acad Sci. 2010;1207:149–54. 10.1111/j.1749-6632.2010.05730.x. [DOI] [PubMed]; Stoll G, Kleinschnitz C, Nieswandt B. Combating innate inflammation: a new paradigm for acute treatment of stroke? Ann N Y Acad Sci. 2010;1207:149–54. doi: 10.1111/j.1749-6632.2010.05730.x. [DOI] [PubMed] [Google Scholar]
- [7].Fang H, Wang PF, Zhou Y, Wang YC, Yang QW. Toll-like receptor 4 signaling in intracerebral hemorrhage-induced inflammation and injury. J Neuroinflamm. 2013;10:27. 10.1186/1742-2094-10-27. [DOI] [PMC free article] [PubMed]; Fang H, Wang PF, Zhou Y, Wang YC, Yang QW. Toll-like receptor 4 signaling in intracerebral hemorrhage-induced inflammation and injury. J Neuroinflamm. 2013;10:27. doi: 10.1186/1742-2094-10-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Yu M, Wang H, Ding A, Golenbock DT, Latz E, Czura CJ, et al. HMGB1 signals through toll-like receptor (TLR) 4 and TLR2. Shock. 2006;26:174–9. 10.1097/01.shk.0000225404.51320.82. [DOI] [PubMed]; Yu M, Wang H, Ding A, Golenbock DT, Latz E, Czura CJ. et al. HMGB1 signals through toll-like receptor (TLR) 4 and TLR2. Shock. 2006;26:174–9. doi: 10.1097/01.shk.0000225404.51320.82. [DOI] [PubMed] [Google Scholar]
- [9].Scaffidi P, Misteli T, Bianchi ME. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature. 2002;418:191–5. 10.1038/nature00858. [DOI] [PubMed]; Scaffidi P, Misteli T, Bianchi ME. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature. 2002;418:191–5. doi: 10.1038/nature00858. [DOI] [PubMed] [Google Scholar]
- [10].Wang H, Yang H, Czura CJ, Sama AE, Tracey KJ. HMGB1 as a late mediator of lethal systemic inflammation. Am J Respir Crit Care Med. 2001;164:1768–73. 10.1164/ajrccm.164.10.2106117. [DOI] [PubMed]; Wang H, Yang H, Czura CJ, Sama AE, Tracey KJ. HMGB1 as a late mediator of lethal systemic inflammation. Am J Respir Crit Care Med. 2001;164:1768–73. doi: 10.1164/ajrccm.164.10.2106117. [DOI] [PubMed] [Google Scholar]
- [11].Luan ZG, Zhang H, Yang PT, Ma XC, Zhang C, Guo RX. HMGB1 activates nuclear factor-kappaB signaling by RAGE and increases the production of TNF-alpha in human umbilical vein endothelial cells. Immunobiology. 2010;215:956–62. 10.1016/j.imbio.2009.11.001. [DOI] [PubMed]; Luan ZG, Zhang H, Yang PT, Ma XC, Zhang C, Guo RX. HMGB1 activates nuclear factor-kappaB signaling by RAGE and increases the production of TNF-alpha in human umbilical vein endothelial cells. Immunobiology. 2010;215:956–62. doi: 10.1016/j.imbio.2009.11.001. [DOI] [PubMed] [Google Scholar]
- [12].Jin YC, Kim SW, Cheng F, Shin JH, Park JK, Lee S, et al. The effect of biodegradable gelatin microspheres on the neuroprotective effects of high mobility group box 1 A box in the postischemic brain. Biomaterials. 2011;32:899–908. 10.1016/j.biomaterials.2010.09.054. [DOI] [PubMed]; Jin YC, Kim SW, Cheng F, Shin JH, Park JK, Lee S. et al. The effect of biodegradable gelatin microspheres on the neuroprotective effects of high mobility group box 1 A box in the postischemic brain. Biomaterials. 2011;32:899–908. doi: 10.1016/j.biomaterials.2010.09.054. [DOI] [PubMed] [Google Scholar]
- [13].Kim ID, Shin JH, Kim SW, Choi S, Ahn J, Han PL, et al. Intranasal delivery of HMGB1 siRNA confers target gene knockdown and robust neuroprotection in the postischemic brain. Mol Ther. 2012;20:829–39. 10.1038/mt.2011.291. [DOI] [PMC free article] [PubMed]; Kim ID, Shin JH, Kim SW, Choi S, Ahn J, Han PL. et al. Intranasal delivery of HMGB1 siRNA confers target gene knockdown and robust neuroprotection in the postischemic brain. Mol Ther. 2012;20:829–39. doi: 10.1038/mt.2011.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Liu K, Mori S, Takahashi HK, Tomono Y, Wake H, Kanke T, et al. Anti-high mobility group box 1 monoclonal antibody ameliorates brain infarction induced by transient ischemia in rats. FASEB J. 2007;21:3904–16. 10.1096/fj.07-8770com. [DOI] [PubMed]; Liu K, Mori S, Takahashi HK, Tomono Y, Wake H, Kanke T. et al. Anti-high mobility group box 1 monoclonal antibody ameliorates brain infarction induced by transient ischemia in rats. FASEB J. 2007;21:3904–16. doi: 10.1096/fj.07-8770com. [DOI] [PubMed] [Google Scholar]
- [15].Zhang J, Takahashi HK, Liu K, Wake H, Liu R, Maruo T, et al. Anti-high mobility group box-1 monoclonal antibody protects the blood-brain barrier from ischemia-induced disruption in rats. Stroke. 2011;42:1420–8. 10.1161/STROKEAHA.110.598334. [DOI] [PubMed]; Zhang J, Takahashi HK, Liu K, Wake H, Liu R, Maruo T. et al. Anti-high mobility group box-1 monoclonal antibody protects the blood-brain barrier from ischemia-induced disruption in rats. Stroke. 2011;42:1420–8. doi: 10.1161/STROKEAHA.110.598334. [DOI] [PubMed] [Google Scholar]
- [16].Lu CN, Yuan ZG, Zhang XL, Yan R, Zhao YQ, Liao M, et al. Saikosaponin a and its epimer saikosaponin d exhibit anti-inflammatory activity by suppressing activation of NF-kappaB signaling pathway. Int Immunopharmacol. 2012;14:121–6. 10.1016/j.intimp.2012.06.010. [DOI] [PubMed]; Lu CN, Yuan ZG, Zhang XL, Yan R, Zhao YQ, Liao M. et al. Saikosaponin a and its epimer saikosaponin d exhibit anti-inflammatory activity by suppressing activation of NF-kappaB signaling pathway. Int Immunopharmacol. 2012;14:121–6. doi: 10.1016/j.intimp.2012.06.010. [DOI] [PubMed] [Google Scholar]
- [17].Wu SJ, Tam KW, Tsai YH, Chang CC, Chao JC. Curcumin and saikosaponin a inhibit chemical-induced liver inflammation and fibrosis in rats. Am J Chin Med. 2010;38:99–111. 10.1142/S0192415X10007695. [DOI] [PubMed]; Wu SJ, Tam KW, Tsai YH, Chang CC, Chao JC. Curcumin and saikosaponin a inhibit chemical-induced liver inflammation and fibrosis in rats. Am J Chin Med. 2010;38:99–111. doi: 10.1142/S0192415X10007695. [DOI] [PubMed] [Google Scholar]
- [18].Lin L, Que R, Shen Y, Chen Y, Yan N, Li Y. Saikosaponind alleviates carbontetrachloride induced acute hepatocellular injury by inhibiting oxidative stress and NLRP3 inflammasome activation in the HL7702 cell line. Mol Med Rep. 2018;17:7939–46. 10.3892/mmr.2018.8849. [DOI] [PubMed]; Lin L, Que R, Shen Y, Chen Y, Yan N, Li Y. Saikosaponind alleviates carbontetrachloride induced acute hepatocellular injury by inhibiting oxidative stress and NLRP3 inflammasome activation in the HL7702 cell line. Mol Med Rep. 2018;17:7939–46. doi: 10.3892/mmr.2018.8849. [DOI] [PubMed] [Google Scholar]
- [19].Su J, Pan YW, Wang SQ, Li XZ, Huang F, Ma SP. Saikosaponin-d attenuated lipopolysaccharide-induced depressive-like behaviors via inhibiting microglia activation and neuroinflammation. Int Immunopharmacol. 2020;80:106181. 10.1016/j.intimp.2019.106181. [DOI] [PubMed]; Su J, Pan YW, Wang SQ, Li XZ, Huang F, Ma SP. Saikosaponin-d attenuated lipopolysaccharide-induced depressive-like behaviors via inhibiting microglia activation and neuroinflammation. Int Immunopharmacol. 2020;80:106181. doi: 10.1016/j.intimp.2019.106181. [DOI] [PubMed] [Google Scholar]
- [20].Sahota P, Savitz SI. Investigational therapies for ischemic stroke: neuroprotection and neurorecovery. Neurotherapeutics. 2011;8:434–51. 10.1007/s13311-011-0040-6. [DOI] [PMC free article] [PubMed]; Sahota P, Savitz SI. Investigational therapies for ischemic stroke: neuroprotection and neurorecovery. Neurotherapeutics. 2011;8:434–51. doi: 10.1007/s13311-011-0040-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Eltzschig HK, Eckle T. Ischemia and reperfusion – from mechanism to translation. Nat Med. 2011;17:1391–401. 10.1038/nm.2507. [DOI] [PMC free article] [PubMed]; Eltzschig HK, Eckle T. Ischemia and reperfusion – from mechanism to translation. Nat Med. 2011;17:1391–401. doi: 10.1038/nm.2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Wang X, Wang C, Wang J, Zhao S, Zhang K, Wang J, et al. Pseudoginsenoside-F11 (PF11) exerts anti-neuroinflammatory effects on LPS-activated microglial cells by inhibiting TLR4-mediated TAK1/IKK/NF-kappaB, MAPKs and Akt signaling pathways. Neuropharmacology. 2014;79:642–56. 10.1016/j.neuropharm.2014.01.022. [DOI] [PubMed]; Wang X, Wang C, Wang J, Zhao S, Zhang K, Wang J. et al. Pseudoginsenoside-F11 (PF11) exerts anti-neuroinflammatory effects on LPS-activated microglial cells by inhibiting TLR4-mediated TAK1/IKK/NF-kappaB, MAPKs and Akt signaling pathways. Neuropharmacology. 2014;79:642–56. doi: 10.1016/j.neuropharm.2014.01.022. [DOI] [PubMed] [Google Scholar]
- [23].Luo Y, Yang YP, Liu J, Li WH, Yang J, Sui X, et al. Neuroprotective effects of madecassoside against focal cerebral ischemia reperfusion injury in rats. Brain Res. 2014;1565:37–47. 10.1016/j.brainres.2014.04.008. [DOI] [PubMed]; Luo Y, Yang YP, Liu J, Li WH, Yang J, Sui X. et al. Neuroprotective effects of madecassoside against focal cerebral ischemia reperfusion injury in rats. Brain Res. 2014;1565:37–47. doi: 10.1016/j.brainres.2014.04.008. [DOI] [PubMed] [Google Scholar]
- [24].Gu JH, Ge JB, Li M, Wu F, Zhang W, Qin ZH. Inhibition of NF-kappaB activation is associated with anti-inflammatory and anti-apoptotic effects of Ginkgolide B in a mouse model of cerebral ischemia/reperfusion injury. Eur J Pharm Sci. 2012;47:652–60. 10.1016/j.ejps.2012.07.016. [DOI] [PubMed]; Gu JH, Ge JB, Li M, Wu F, Zhang W, Qin ZH. Inhibition of NF-kappaB activation is associated with anti-inflammatory and anti-apoptotic effects of Ginkgolide B in a mouse model of cerebral ischemia/reperfusion injury. Eur J Pharm Sci. 2012;47:652–60. doi: 10.1016/j.ejps.2012.07.016. [DOI] [PubMed] [Google Scholar]
- [25].Mao X, Miao G, Tao X, Hao S, Zhang H, Li H, et al. Saikosaponin a protects TBI rats after controlled cortical impact and the underlying mechanism. Am J Transl Res. 2016;8:133–41. [PMC free article] [PubMed]; Mao X, Miao G, Tao X, Hao S, Zhang H, Li H. et al. Saikosaponin a protects TBI rats after controlled cortical impact and the underlying mechanism. Am J Transl Res. 2016;8:133–41. [PMC free article] [PubMed] [Google Scholar]
- [26].Chen Y, Wu X, Yu S, Lin X, Wu J, Li L, et al. Neuroprotection of tanshinone IIA against cerebral ischemia/reperfusion injury through inhibition of macrophage migration inhibitory factor in rats. PLoS One. 2012;7:e40165. 10.1371/journal.pone.0040165. [DOI] [PMC free article] [PubMed]; Chen Y, Wu X, Yu S, Lin X, Wu J, Li L. et al. Neuroprotection of tanshinone IIA against cerebral ischemia/reperfusion injury through inhibition of macrophage migration inhibitory factor in rats. PLoS One. 2012;7:e40165. doi: 10.1371/journal.pone.0040165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2018;9:7204–18. 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed]; Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J. et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2018;9:7204–18. doi: 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Bianchi ME, Manfredi AA. High-mobility group box 1 (HMGB1) protein at the crossroads between innate and adaptive immunity. Immunol Rev. 2007;220:35–46. 10.1111/j.1600-065X.2007.00574.x. [DOI] [PubMed]; Bianchi ME, Manfredi AA. High-mobility group box 1 (HMGB1) protein at the crossroads between innate and adaptive immunity. Immunol Rev. 2007;220:35–46. doi: 10.1111/j.1600-065X.2007.00574.x. [DOI] [PubMed] [Google Scholar]
- [29].Kim SW, Lim CM, Kim JB, Shin JH, Lee S, Lee M, et al. Extracellular HMGB1 released by NMDA treatment confers neuronal apoptosis via RAGE-p38 MAPK/ERK signaling pathway. Neurotox Res. 2011;20:159–69. 10.1007/s12640-010-9231-x. [DOI] [PubMed]; Kim SW, Lim CM, Kim JB, Shin JH, Lee S, Lee M. et al. Extracellular HMGB1 released by NMDA treatment confers neuronal apoptosis via RAGE-p38 MAPK/ERK signaling pathway. Neurotox Res. 2011;20:159–69. doi: 10.1007/s12640-010-9231-x. [DOI] [PubMed] [Google Scholar]