Abstract
Neuronal injuries can lead to various diseases such as neurodegenerative diseases, stroke, trauma, ischemia and, more specifically, glaucoma and optic neuritis. The cellular mechanisms that regulate neuronal death include calcium influx and calcium overload, excitatory amino acid release, oxidative stress, inflammation and microglial activation. Much attention has been paid to the effective prevention and treatment of neuroprotective drugs by natural products. This review summarizes the neuroprotective aspects of natural products, extracted from Panax ginseng, Camellia sinensis, soy and some other plants, and some of their chemical derivatives. Their antioxidative and anti-inflammatory action and their inhibition of apoptosis and microglial activation are assessed. This will provide new directions for the development of novel drugs and strategies to treat neurodegenerative diseases.
Keywords: antioxidant effect; brain injuries, Camellia sinensis, chemical derivatives, curcumin, genistein, natural products, neurodegenerative diseases, neuroprotection, Panax
Introduction
Neuronal damage and death in the central nervous system is a common pathological process in neurological diseases, such as neurodegenerative diseases, stroke, brain injury, glaucoma and optic neuritis (Fei et al., 2015). In the past few decades, the incidence of neurodegenerative diseases, including Parkinson’s disease (PD), Huntington’s disease, amyotrophic lateral sclerosis and Alzheimer’s disease (AD), has increased dramatically worldwide (Wang et al., 2019). These diseases not only seriously threaten the health of older adults, but also greatly increase the social burden. The cellular mechanism that regulates neuronal cell death includes calcium influx and calcium overload, excitatory amino acid release, oxidative stress, inflammation and glial dysfunction (Cheng et al., 2018). However, the underlying mechanism of neuronal death is still not fully understood and there are no effective therapies for these neurological diseases. Therefore, more research and trials of neuroprotective drugs are in progress (Ganguly et al., 2018).
Neuroprotection is not just a treatment to slow disease progression and prevent neuronal death, but also to defer age related neuronal death, a natural phenomenon and extremely complex process (Cai et al., 2017). The neuroprotective effects of natural products and their chemical derivatives have attracted much attention (Araujo-Filho et al., 2016) and are discussed and summarized in this review.
Retrieval Strategy
The PubMed online database was searched for studies from 1942 to 2019, using the following queries in all fields: (Brain injuries (MeSH Terms) OR Neurodegenerative diseases (MeSH Terms)) AND (Natural Products (MeSH Terms)). Non-SCI experiments and reviews were excluded.
We examined the potential usefulness of Panax ginseng, green tea/EGCG/Camellia sinensis, genistein and curcumin in the prevention and treatment of brain injuries or neurodegenerative diseases in human clinical studies in vivo or cells in vitro. We reviewed the molecular mechanisms involved in effective prevention and treatment of neuroprotective drugs from natural products.
Mechanisms of Neuronal Damage and Neuroprotection
The mechanism of neuronal damage and death, from the organ level to the molecular level, has been explored over many years. In 1889, the concept of “brain damage” was first reported; however, the neuronal changes in the brain were not studied. In 1942, Sandler first proposed “neuronal injury” and found that hypoglycemia complicated with poliovirus infection caused neuronal injury. Later on, the study of neuronal damage went down to the molecular level. For example, in 1988, O’Shaughnessy and Lodge found that the activation of excitatory amino acid receptors could lead to neuronal damage by enhanced Ca2+ mobilization and these could be blocked by selective antagonists. In 1995, Ziv et al. also verified the importance of the effects of changes in intracellular calcium concentration on neuronal damage. The theory of the molecular mechanisms of neuronal damage is focused on calcium influx and calcium overload, excitatory amino acid release and free radical formation (Abushouk et al., 2017). Calcium is of particular importance in neuronal damage. Calcium overload can trigger either necrotic or apoptotic cell death of neurons. Normally, nitric oxide plays a physiological role as a neurotransmitter, but in the pathological concentrations it can cause damage to neuronal cells (Gokay et al., 2016). Excitatory amino acids are important neurotransmitters in the central nervous system with their release and uptake controlled under normal circumstances. However, their accumulation can lead to brain damage. Brain tissue is rich in iron and unsaturated fatty acids but lacks the antioxidant systems of catalase, glutathione and vitamin E (Sakr et al., 2015). As a result, neurons are more vulnerable to damage by oxygen free radicals. The expression of reactive oxygen species (ROS, such as peroxides and hydroxyl groups) and reactive nitrogen clusters increase significantly in cerebral ischemia. Extensive production of ROS leads to lipid peroxidation and causes post-ischemic nerve injury (Lei et al., 2015). Glutamate-induced activation of N-methyl-D-aspartic acid receptors can lead to calcium overload, protein kinase activation, nitric oxide synthase and phospholipase, which are associated with the damaged mitochondria and increased free radicals, resulting in neuronal damage (Simoes et al., 2018; Figure 1). Various epidemiological studies have indicated that natural products such as ginsenoside and soy isoflavone glycitein may reduce the risk of neurodegenerative disorders and neuroinflammatory diseases. It has been reported that ginkgo biloba extract 761 and resveratrol can protect neuronal damage by inhibiting the generation or aggregation of β amyloid peptide (Aβ; Yang et al., 2018). Other research into the effects of ginsenosides, including Re, Rb1 and Rg1, showed they have neuroprotective effects and can promote neurite outgrowth in cultured neurons (Li et al., 2017). These and other studies indicate that natural products may contribute towards neuroprotection after neuronal damage.
Figure 1.
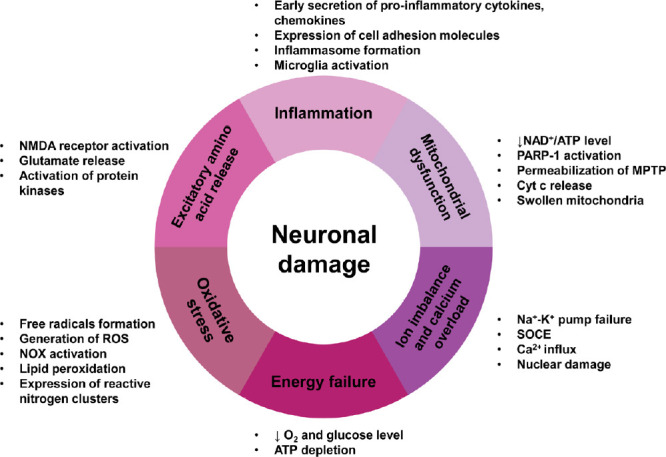
The main molecular mechanisms of neuronal damage.
ATP: Adenosine triphosphate; Cyt c: cytochrome c; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; NAD: nicotinamide adenine dinucleotide free acid; NMDA: N-methyl-D-aspartic acid receptor; NOX: NADPH oxidase; PARP1: poly (ADP-ribose) polymerase-1; ROS: reactive oxygen species.
Neuroprotective Effects of Some Natural Products and Their Chemical Derivatives Ginsenoside from Panax ginseng (WEG)
Ginsenoside from Panax ginseng (WEG)
Ginseng is a perennial plant, belonging to the Panax genus under the family Araliaceae. The herbal medicine of ginseng is extracted from the dried root of the perennial plant.
Among the family of Araliaceae, WEG is a precious herbal medicine used in many countries, especially in Korea, China and Japan, for more than five thousand years. People named the plant WEG because they believed that WEG had properties to resist the adverse influence and restore homeostasis from the damage of multiple physical and chemical factors to the patients. Russian botanist C. A. Meyer named the genus “Panax” which derived from ancient Greek words “pan” and “axos”, meaning “all” and “cure”. Chinese named the plants “ginseng” from ancient literary Chinese “renshen”, which means “human”. Nowadays ginseng has become a popular, worldwide drug with a wide variety of medicinal uses. A recent scientific study demonstrated that ginseng has positive effects on preventing skin aging and increasing vigor (Park et al., 2016b).
A number of compounds have been identified in the root of Ginseng, including ginsenosides, the elements calcium, magnesium and manganese, various vitamins and amino acids (Chen et al., 2019b). However, the two major active ingredients named ginsenosides or ginseng saponins are found exclusively in the Panax species (Chen et al., 2019b). More than 100 different ginsenosides have been named and extracted from whole ginseng of which 40 ginsenosides have been found in WEG (Fernandez-Moriano et al., 2017). All of the identified ginsenosides have a four-ring hydrophobic, steroid-like chemical structure in common. However, depending on the different number of sugar moieties and binding positions, there are two main groups of ginsenosides: 20(S)-protopanaxadiol and 20(S)-protopanaxatriol saponins (Jin et al., 2019). The different numbers of sugar moieties might be responsible for the unique activity of each ginsenoside. The different chemical structures result in each ginsenoside having its own specific tissue-dependent pharmacological effects. Many studies have shown that ginseng has numerous therapeutic effects, including cardiovascular and hepatic benefits (Shi et al., 2019), immunomodulatory effects (Chen et al., 2019a), cancer suppression (Wang et al., 2018), hypoglycemic activity (Jang et al., 2017) and beneficial effects on the central nervous system, such as improved cognitive performance and neurotransmitter modulation (Reay et al., 2019). Many reports have shown that ginsenosides have neuroprotective effects on the central nervous system through their anti-apoptosis, oxidation resistance, and anti-inflammation properties (Zhou et al., 2018). Ginsenosides have been used to treat some neurodegenerative diseases including PD and AD (Razgonova et al., 2019). Several reports demonstrate that ginseng and its active components ginsenosides exhibit neuroprotective effects on dopaminergic neurons both in vitro and in vivo (Cheng et al., 2019; Razgonova et al., 2019). PD, characterized by both the progressive degeneration of the nigrostriatal dopaminergic pathway and the accumulation and aggregation of misfolded α-synuclein, is a progressive neurodegenerative disease affecting 2% of the population aged over 60 years worldwide. Nowadays, most PD treatments provide only symptomatic therapies, and no medicine has been proven to prevent the progressive degeneration. However, ginsenosides Rb1 and Rg1 can attenuate Ca2+ over-influx into mitochondria and increase energy production, leading to partial neuroprotection in vivo. It is reported that in 1-methyl-4-phenylpyridinium (MPP+) treated SH-SY5Y cells, WEG exerted an inhibitory effect on cell death, alleviated the overproduction of ROS, increased Bax/Bcl-2 ratio, downregulated the expression of cytochrome c and prevented the activation of caspase-3 (Bell et al., 2017). These experiments suggested that ginseng has potential therapeutic value in the war against PD.
AD usually affects older adults, destroys memory and thinking skills with gradual neuronal death. Aβ accumulation is widely believed to be the key leading to AD pathogenesis. According to current research, Aβ deposition is caused by the abnormal metabolism of amyloid precursor protein (Wang et al., 2016a). Nowadays therapies in AD include decreasing the generation or aggregation of Aβ, increasing the disaggregation of Aβ in neurons, and using stronger antioxidant and anti-inflammatory drugs. There is still no definitive evidence to document any novel drug effective in preventing the progressive loss of neurons in AD patients. However, studies have suggested that ginsenosides could be protective and trophic against AD.
Korean red ginseng can be used as an adjuvant treatment adding to conventional therapies in AD patients (Heo et al., 2008). The results showed that 15 patients taking 12 weeks of high-dose Korean red ginseng obtained significant improvement in the Alzheimer’s disease Assessment Scale and Clinical Dementia Rating compared with the control group (n = 31) but not using the Korean version of MMSE (Heo et al., 2008). Ginseng may exert its neuroprotective effect by its anti-inflammatory effects. Ginseng extracts can significantly reduce the activation of nuclear factor-κB (NF-κB) and MAP kinase. Lipopolysaccharide (LPS)-induced inducible nitric oxide synthase (iNOS) and LPS-induced activation of STAT signaling pathway can be significantly inhibited by ginsenosides Rh2, Rh3 and compound K (Yu et al., 2017). Therefore, ginseng extracts can be beneficial in treating neuroinflammatory diseases. Another study found that American ginseng selectively inhibits the expression of inflammatory intermediates in the STAT cascades in keratinocytes and the expression of LPS-induced tumor necrosis factor-α and other cytokines in macrophages (Ahn et al., 2016). It has been reported that ginsenoside Rg2 inhibits Na+ influx through the channels and consequently reduce both Ca2+ influx and catecholamine secretion in vivo. Na+ channel blockers have neuroprotective effects on neuronal damage caused by cerebral ischemia, hypoxia, and brain trauma (Leipold, 1995). In conclusion, this natural herb, ginseng, and its components, ginsenosides, provide important natural sources of protection for neuronal damage.
Green tea epigallocatechin-3-gallate
Green tea (Camellia sinensis) is a traditional beverage consumed worldwide, especially in Asian countries. This traditional drink is linked with a low incidence of human cancer, obesity and slowing of the aging process. Thus, the effect of green tea in the prevention and mitigation of neurodegenerative diseases has recently received particular attention (Tyagi et al., 2016). Green tea (Camellia sinensis) is generally composed of four derivatives based on their different structures: epicatechin, epigallocatechin, epicatechin gallate and green tea epigallocatechin-3-gallate (EGCG). However, the most abundant and anti-oxidative component in green tea is (–)-EGCG (Shirani et al., 2016). The potential of EGCG for antioxidant, anti-inflammatory (Ohishi et al., 2016) and fibrogenic instability is responsible for the neuroprotective effects of green tea (Xing et al., 2019). An epidemiological survey has shown that American people drinking two or more cups of tea every day have a low risk of PD (Barranco Quintana et al., 2009). EGCG may prevent ROS by scavenging free radicals, metal chelation and adjusting the oxidation and anti-oxidant enzyme system. EGCG can inhibit Cd2+-induced apoptosis by acting as a ROS scavenger rather than a metal chelating agent (An et al., 2014). EGCG can promote the survival of PC12 cells treated with neurotoxic compounds 6-hydroxydopamine, MPP+, paraquat, levodopa, and hydrogen peroxide (He et al., 2017). It was suggested that green tea polyphenol EGCG not only upregulates the expression of antioxidant enzymes such as superoxide dismutase, catalase and GPX but also effects a variety of apoptotic factors including caspase modulation, Fas ligand (FasL), growth arrest and DNA damage proteins and members of the B cell lymphoma 2 (Bcl-2) family (Wang et al., 2015b). It is believed that these effects lead to EGCG’s antioxidative roles. In addition, EGCG is more effective at scavenging free radicals than other common factors, vitamin C and E (Zahr et al., 2018). Chen et al. (2019a, b) confirmed that orally administered EGCG could modulate other pathways including protein kinase C, extracellular signal-regulated kinase, and Akt/glycogen synthase kinase-3 axis. The neuroprotective effect of EGCG could be exerted through another mechanism that involves scavenging ROS, iron chelation and stabilization of the mitochondrial source of activated caspase. EGCG directly reduced neuronal damage caused by ROS by inhibiting NF-κB (Zhang et al., 2016). Wei et al. (2011) reported that EGCG directly inhibited the expression of nNOS through inhibition of the secretion of nitric oxide and tumor necrosis factor-α by LPS activated microglia, thereby reducing the role of oxidative stress.
The neuroprotective effect of green tea has been confirmed in many clinical trials (Prado Lima et al., 2018; Kim et al., 2019). Green tea, especially EGCG, can be an adjunctive method in the treatment of neurodegenerative diseases and neurological diseases.
Soy isoflavone glycitein
Soybeans and soy products are highly consumed in traditional Asian diets. A previous study demonstrated that the soybeans and soy products can reduce the risk of cancer, heart diseases and diabetes (Namazi et al., 2018). Soy isoflavone glycitein, the most recognized biologically active compound in soybeans and soy products, is considered to have therapeutic potential for different types of neurodegenerative diseases. Soybeans are rich in isoflavones including genistein (4′,5,7-trihydroxyisoflavone), daidzein (4′,7-dihydroxyisoflavone), glycitein (6-methoxydaidzein) and their glycosides. Genistein can defend human cortical neurons against free radical generating toxins such as tertiary-butylhydroperoxide (t-BuOOH) by regulating bcl-2 (Sonee et al., 2004). Glycitein could decrease the expression of β amyloid-induced paralysis in the transgenic Caenorhabditis elegans (Gutierrez-Zepeda et al., 2005). Therefore, soy isoflavones may have therapeutic potential for Aβ-related neurodegenerative disease through combinations of antioxidant activity and inhibition of Aβ deposition. Genistein, which has structural similarities to the sex hormone estrogen, exhibits estrogen agonist and antagonist characteristics and exerts protective effects on hippocampal neurons and dopaminergic neurons in vitro and in vivo (Park et al., 2016a). Genistein can promote the cascade reaction to inhibit production of ROS and expression of BAD mRNA, regulate nuclear factor-erythroid 2 p45-related factor 2/heme oxygenase-1, Nrf 2/HO-1 signal transduction pathway, upregulate the expression of apoptosis suppressing gene and reduce the deposition of Aβ (Bai and Wang, 2019).
All these findings show that soybeans and soy products are potential new therapies for neurodegenerative disorders.
Curcumin
Curcumin, extracted from the root of Zingiberaceae Curcuma longa, is used as a food preservative in ancient Asia (Arablou and Kolahdouz-Mohammadi, 2018). Curcumin, derived mainly from turmeric, has been associated with the beneficial effects of several human ailments for centuries, as recorded in the Ayurveda. Epidemiological studies have shown that the usage of turmeric may be behind the low incidence rate of AD in India compared with that in Caucasians (Tiwari et al., 2016). Turmeric is effective in protecting neurons in the PD mouse. It is believed that turmeric contains a variety of active ingredients, however, the most important component of turmeric is curcumin, which has strong antioxidant properties, radical scavenging and metal chelating activities.
Curcumin can effectively attenuate the production of pro-inflammatory mediators, including cytokines, activator protein 1, NF-κB, iNOS, cyclooxygenase 2 and c-Jun N-terminal kinase (Yang et al., 2018). Curcumin has been shown to directly detoxify reactive nitrogen species such as peroxynitrite in vitro (Liu et al., 2011). Curcumin can protect neuronal mitochondria against peroxynitrite-induced protein nitration. In addition, curcumin suppresses the inflammation in microglia through inhibiting the ganglioside-, LPS-, or interferon-γ-stimulated induction of cyclooxygenase 2 and iNOS, which are key steps in inflammatory processes (Pan et al., 2012). The enhancement of suppressor of cytokine signaling 1 expression and the inhibition of Janus kinase-signal transducer and activator of transcription signaling lead to the anti-inflammatory effect of curcumin on activated microglia (Kim et al., 2003). Furthermore, curcumin can not only reduce oxidative and inflammatory damage, but also decrease the accumulation of Aβ in mutant amyloid precursor protein transgenic plaque-forming mouse model (Du et al., 2019). The molecular structure of curcumin is similar to that of the β-amyloid receptor and studies have shown that low-dose curcumin not only effectively disaggregates Aβ, but also prevents fibril and oligomer formation (Ouberai et al., 2009). This indicates how curcumin may contribute to the prevention or treatment of AD.
It is concluded that curcumin has significant neuroprotective effects, such as anti-inflammatory, antioxidant, and anti-protein-aggregate effects, and has a great potential in the treatment of different neurodegenerative disorders and neuroinflammatory diseases.
Other products and their chemical derivatives
Resveratrol, a natural polyphenol with multiple biological functions (Jeong et al., 2016), is mainly found in grapes, giant knotweed, peanuts and mulberries. Resveratrol can attenuate early neurological dysfunction in transgenic C. elegans (Pandey et al., 2015) and neuropathic pain in mice by activating Sir2 (silent information factor 2) (Shao et al., 2014). In rat models, resveratrol can lessen neuronal damage induced by the dopamine neurotoxin, MPP+ (Chen et al., 2016). In the ischemic model of Mongolian gerbils, the resveratrol delayed neuronal death was accompanied by microglial activation (Bhattacharjee et al., 2016; Liu et al., 2016). Early data have demonstrated that resveratrol potently inhibited the production of NO and iNOS, increased the activity of superoxide dismutase and decreased malondialdehyde concentration (Li et al., 2015b). It showed that resveratrol had neuroprotective effects on LPS-induced and N-methyl-D-aspartic acid-induced neuronal death. This neuroprotective effect may be associated with antioxidant and anti-inflammatory activity. In addition, resveratrol has been shown to activate PI3K/Akt and extracellular signal-regulated kinase 1/2 pathway, resulting in the inactivation of glycogen synthase kinase-3, which may contribute to its neuroprotective effects (Wang et al., 2015a).
Tanshinones are lipid soluble abietane diterpenes extracted from the dried root, Radix Salviae Miltiorrhiza Burge. The main constituents are tanshinone I, tanshinone II A, and tanshinone II B but tanshinone II A is the most abundant component. Studies have shown that tanshinone II A may have neuroprotective effects in ischemic animal models (Lee et al., 2013). Tanshinone II A pretreatment can promote the activation of astrocytes and reduce brain damage in ischemic models. The neuroprotective effects of tanshinone II A may be associated with the increased activity of superoxide dismutase, decreased concentration of malondialdehyde, higher expression of Bcl-2, and lower expression of Bax, nitric oxide and iNOS (Li et al., 2015a). In addition, tanshinone II A can block calcium channels to reduce intracellular Ca2+ accumulation after brain damage (Xu et al., 2016).
Ligustilide is a main active ingredient in Ligusticum and Angelica. Numerous studies have reported that ligustilide can reduce NO production, tumor necrosis factor-α, monocyte chemotactic protein-1, cyclooxygenase-2 and iNOS expression to protect neurons (Su et al., 2011; Ma and Bai, 2012). Moreover, ligustilide can reduce the fatality rate, improve the neurobehavioral defects, attenuate brain edema and alleviate cerebral vasospasm through the blood brain barrier after subarachnoid hemorrhage in mice (Xiao et al., 2015).
Ginkgo biloba extract 761 (EGb761), from the dried leaves of Ginkgo biloba, has been used to treat numerous diseases in traditional Chinese medicine for thousands of years. EGb761 is also used as a natural neuroprotective medication for brain ischemia (Wan et al., 2015). In a neuronal toxicity model in vitro, 12 M bilobalide, a purified terpene lactone component of the EGb761, reduced neuronal death induced by glutamate toxicity, demonstrating that bilobalide may have neuroprotective potential (Chandrasekaran et al., 2003). The retina, as part of the central nervous system, was also protected by EGb761. The vision in normal-tension glaucoma patients was improved after Ginkgo biloba extract treatment compared with the controls. It was suggested that the effect of Ginkgo biloba extract was related to the increased ocular blood flow (Cho et al., 2019).
Scutellarin is a flavonoid compound, extracted from Gerbera jamesonii. Scutellarin may exert its neuroprotective effects by inhibiting Ca2+ overload, resistance to lipid peroxidation, maintenance of the mitochondrial membrane structure and improvement of cerebral energy metabolism after ischemic injury (Tang et al., 2014).
Puerarin, an isoflavone compound, is extracted and isolated from the dry root of Radix Puerariae. In recent years, puerarin has been used to treat glaucoma, diabetic retinopathy and other eye diseases. Some researchers have reported an anti-apoptosis role for puerarin in ischemic models (Wang et al., 2016b). The results showed that pretreatment of puerarin in PC12 cells can reduce MPP+-induced neurotoxicity and apoptosis. The neuroprotective mechanism of puerarin may be linked to inhibition of mitochondrial dysfunction and the activation of caspase-3 (Wang et al., 2016b; Table 1).
Table 1.
Summary of neuroprotective effects of some natural products and their chemical derivatives
| Name | Family | Origin | Main component | Disease | Mechanism (pathway) | Model (disease) |
|---|---|---|---|---|---|---|
| Ginsenoside | Araliaceae | Panax ginseng | Rb1, rg1 | PD | Ca2+, TNF-α, ROS, Bax/Bcl-2, Caspase-3 | In vivo (animal models), in vitro |
| Korean red ginseng | Rg3 | Alzheimer’s disease | Aβ, brain-type Na+ channel | |||
| Ginseng extracts | Rh2, rh3, compound k | Neuroinflammatory diseases | Nf-κb, MAP kinase signaling | |||
| American ginseng | Stat Cascades | |||||
| Epigallocatechin-3-gallate | Green tea | Cancer, obesity and slowing the aging process, PD | GPX, FasL, Bcl-2, PKC, ERK, Akt/GSK-3 axis, ROS, iron chelation, SMAC | In vivo, in vitro (PC12 cells) | ||
| Soy isoflavone glycitein (genistein (4′, 5′7-trihydroxyisoflavone), daidzein (4′, 7-dihydroxyisoflavone), glycitein (6-methoxydaidzein) and their glycosides) | Soybeans and soy products | Neurodegenerative disorders | Bcl-2, cascade, ROS and expression of BAD mRNA, Nrf 2/HO-1 signal transduction pathway | In vivo (animal models), in vitro | ||
| Curcumin | The root of rhizoma curcumae longae | Turmeric | Alzheimer’s disease, PD, | AP-1, NF-κB, iNOS, COX-2, JNK, SOCS-1, JAK-STAT signaling | In vivo (Animal models), in vitro | |
| Resveratrol | Grapes, giant knotweed, peanuts, mulberry and other plants | Sir2, MPP+, NO, iNOS, PI3K/Akt, ERK1/2 pathway | In vivo (animal models), in vitro (PC12 cells) | |||
| Tanshinone | Tanshinone I tanshinone II A, tanshinone II B | Radix salviae miltiorrhiza bunge dried root | Brain ischemia | Bcl-2, Bax, Ca2+ channel | In vivo (animal models) | |
| Ligustilide | Ligusticum and angelica | Brain injury, subarachnoid hemorrhage, brain edema | NO, TNF-α, MCP-1, COX-2, iNOS | In vivo (animal models) | ||
| Ginkgo biloba extract 761 (egb761) | Dried leaves of ginkgo biloba | Bilobalide | Brain ischemia, glaucoma | Glutamate toxicity, ocular blood flow | In vitro (patients) | |
| Scutellarin | Gerbera jamesonii. | Ischemic injury | Ca2+ | In vitro (animal models) | ||
| Puerarin | Radix puerariae dry root | Glaucoma, diabetic retinopathy and other eye diseases | MPP+ | In vivo (animal models), in vitro (PC12 cells) |
Aβ: Amyloid beta-protein; AP-1: activator protein 1; Bax: Bcl-2 associated X protein; COX: cyclooxygenase; ERK: extracellular regulated protein kinases; FasL: Fas ligand; GPX: glutathione peroxidase; GSK: glycogen synthase kinase-3; HO-1: heme oxygenase protein 1; iNOS: inducible nitric oxide synthase; JAK-STAT: Janus kinase-signal transducer and activator of transcription; JNK: c-Jun N-terminal kinase; MCP-1: monocyte chemotactic protein-1; MPP+: 1-methyl-4-phenylpyridinium; NF-κB: nuclear factor kappa-B; NO: nitric oxide; Nrf 2: nuclear respiratory factor-2; PD: Parkinson’s disease; PI3K: phosphatidylinositol-3-kinase; PKC: protein kinase C; Rb1: ginsenoside Rb1; Rg1: ginsenoside Rg1; Rg3: ginsenoside Rg3; Rh2: ginsenoside Rh2; Rh3: ginsenoside Rh3; ROS: reactive oxygen species; Sir2: silent information regulator2; SMAC: second mitochondria-derived activator of caspases; SOCS-1: suppressor of cytokine signaling-1; TNF-α: tumor necrosis factor alpha.
Conclusion
The mortality and morbidity in patients with disease or injury of the central nervous system are still high. Neuronal damage is central to the high figures but its pathophysiology is complex. Although tens of thousands of studies have investigated various neuroprotective mechanisms, including anti-oxidation, inhibition of apoptosis, anti-inflammation and inhibition of microglial activation, the outcomes of neurologically diseased patients or injured people are still poor. As we indicated in our review, evidence has accumulated that natural products and their chemical derivatives can reduce neuronal damage. They and their derivatives provide a new opportunity for the development of novel drugs and strategies for the treatment of these diseases.
Footnotes
Conflicts of interest: The authors have no actual or potential conflicts of interest.
Financial support: This work was supported by the National Natural Science Foundation of China, Nos. 81600738 (to FF), 81771239 (to ZF), 81430043 (to ZF), 81801300 (to NS). The funding sources had no role in paper writing or deciding to submit this paper for publication.
Copyright license agreement: The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Funding: This work was supported by the National Natural Science Foundation of China, Nos. 81600738 (to FF), 81771239 (to ZF), 81430043 (to ZF), 81801300 (to NS).
C-Editor: Zhao M; S-Editors: Wang J, Li CH; L-Editors: Dawes EA, de Souza M, Qiu Y, Song LP; T-Editor: Jia Y
References
- 1.Abushouk AI, Negida A, Ahmed H, Abdel-Daim MM. Neuroprotective mechanisms of plant extracts against MPTP induced neurotoxicity: future applications in Parkinson’s disease. Biomed Pharmacother. 2017;85:635–645. doi: 10.1016/j.biopha.2016.11.074. [DOI] [PubMed] [Google Scholar]
- 2.Ahn S, Siddiqi MH, Aceituno VC, Simu SY, Zhang J, Jimenez Perez ZE, Kim YJ, Yang DC. Ginsenoside Rg5:Rk1 attenuates TNF-alpha/IFN-gamma-induced production of thymus- and activation-regulated chemokine (TARC/CCL17) and LPS-induced NO production via downregulation of NF-kappaB/p38 MAPK/STAT1 signaling in human keratinocytes and macrophages. In Vitro Cell Dev Biol Anim. 2016;52:287–295. doi: 10.1007/s11626-015-9983-y. [DOI] [PubMed] [Google Scholar]
- 3.An Z, Qi Y, Huang D, Gu X, Tian Y, Li P, Li H, Zhang Y. EGCG inhibits Cd(2+)-induced apoptosis through scavenging ROS rather than chelating Cd(2+) in HL-7702 cells. Toxicol Mech Methods. 2014;24:259–267. doi: 10.3109/15376516.2013.879975. [DOI] [PubMed] [Google Scholar]
- 4.Arablou T, Kolahdouz-Mohammadi R. Curcumin and endometriosis: review on potential roles and molecular mechanisms. Biomed Pharmacother. 2018;97:91–97. doi: 10.1016/j.biopha.2017.10.119. [DOI] [PubMed] [Google Scholar]
- 5.Araujo-Filho HG, Quintans-Junior LJ, Barreto AS, Almeida JR, Barreto RS, Quintans JS. Neuroprotective effect of natural products on peripheral nerve degeneration: a systematic review. Neurochem Res. 2016;41:647–658. doi: 10.1007/s11064-015-1771-2. [DOI] [PubMed] [Google Scholar]
- 6.Bai Z, Wang Z. Genistein protects against doxorubicin-induced cardiotoxicity through Nrf-2/HO-1 signaling in mice model. Environ Toxicol. 2019;34:645–651. doi: 10.1002/tox.22730. [DOI] [PubMed] [Google Scholar]
- 7.Barranco Quintana JL, Allam MF, Del Castillo AS, Navajas RF. Parkinson’s disease and tea: a quantitative review. J Am Coll Nutr. 2009;28:1–6. doi: 10.1080/07315724.2009.10719754. [DOI] [PubMed] [Google Scholar]
- 8.Bell M, Sopko NA, Matsui H, Hannan JL, Bivalacqua TJ. RhoA/ROCK activation in major pelvic ganglion mediates caspase-3 dependent nitrergic neuronal apoptosis following cavernous nerve injury. Neural Regen Res. 2017;12:572–573. doi: 10.4103/1673-5374.205091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bhattacharjee S, Zhao Y, Dua P, Rogaev EI, Lukiw WJ. microRNA-34a-mediated down-regulation of the microglial-enriched triggering receptor and phagocytosis-sensor TREM2 in age-related macular degeneration. PLoS One. 2016;11:e0150211. doi: 10.1371/journal.pone.0150211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cai W, Zhang K, Li P, Zhu L, Xu J, Yang B, Hu X, Lu Z, Chen J. Dysfunction of the neurovascular unit in ischemic stroke and neurodegenerative diseases: an aging effect. Ageing Res Revi. 2017;34:77–87. doi: 10.1016/j.arr.2016.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chandrasekaran K, Mehrabian Z, Spinnewyn B, Chinopoulos C, Drieu K, Fiskum G. Neuroprotective effects of bilobalide, a component of Ginkgo biloba extract (EGb 761) in global brain ischemia and in excitotoxicity-induced neuronal death. Pharmacopsychiatry 36 Suppl. 2003;1:S89-94. doi: 10.1055/s-2003-40447. [DOI] [PubMed] [Google Scholar]
- 12.Chen J, Bai Q, Zhao Z, Sui H, Xie X. Resveratrol improves delayed r-tPA treatment outcome by reducing MMPs. Acta Neurol Scand. 2016;134:54–60. doi: 10.1111/ane.12511. [DOI] [PubMed] [Google Scholar]
- 13.Chen LX, Qi YL, Qi Z, Gao K, Gong RZ, Shao ZJ, Liu SX, Li SS, Sun YS. A Comparative study on the effects of different parts of panax ginseng on the immune activity of cyclophosphamide-induced immunosuppressed mice. Molecules. 2019a;24:E1096. doi: 10.3390/molecules24061096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen W, Balan P, Popovich DG. Review of ginseng anti-diabetic studies. Molecules. 2019b;24:E4501. doi: 10.3390/molecules24244501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cheng SY, Wang SC, Lei M, Wang Z, Xiong K. Regulatory role of calpain in neuronal death. Neural Regen Res. 2018;13:556–562. doi: 10.4103/1673-5374.228762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cheng Z, Zhang M, Ling C, Zhu Y, Ren H, Hong C, Qin J, Liu T, Wang J. Neuroprotective effects of ginsenosides against cerebral ischemia. Molecules. 2019;24:E1102. doi: 10.3390/molecules24061102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cho HK, Kim S, Lee EJ, Kee C. Neuroprotective effect of ginkgo biloba extract against hypoxic retinal ganglion cell degeneration in vitro and in vivo. J Med Food. 2019;22:771–778. doi: 10.1089/jmf.2018.4350. [DOI] [PubMed] [Google Scholar]
- 18.Du S, Zhang Y, Yang J, Liu X, Wang Y, Xu B, Jia J. Curcumin alleviates beta amyloid-induced neurotoxicity in HT22 Cells via upregulating SOD2. J Mol Neurosci. 2019;67:540–549. doi: 10.1007/s12031-019-01267-2. [DOI] [PubMed] [Google Scholar]
- 19.Fei F, Li J, Rao W, Liu W, Chen X, Su N, Wang Y, Fei Z. Upregulation of homer1a promoted retinal ganglion cell survival after retinal ischemia and reperfusion via interacting with Erk pathway. Cell Mol Neurobiol. 2015;35:1039–1048. doi: 10.1007/s10571-015-0198-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fernandez-Moriano C, Gonzalez-Burgos E, Iglesias I, Lozano R, Gomez-Serranillos MP. Evaluation of the adaptogenic potential exerted by ginsenosides Rb1 and Rg1 against oxidative stress-mediated neurotoxicity in an in vitro neuronal model. PLoS One. 2017;12:e0182933. doi: 10.1371/journal.pone.0182933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ganguly U, Chakrabarti SS, Kaur U, Mukherjee A, Chakrabarti S. Alpha-synuclein, proteotoxicity and parkinson’s disease: search for neuroprotective therapy. Curr Neuropharmacol. 2018;16:1086–1097. doi: 10.2174/1570159X15666171129100944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gokay NS, Yilmaz I, Komur B, Demiroz AS, Gokce A, Dervisoglu S, Gokay BV. A comparison of the effects of neuronal nitric oxide synthase and inducible nitric oxide synthase inhibition on cartilage damage. BioMed Res Int. 2016;2016:7857345. doi: 10.1155/2016/7857345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gutierrez-Zepeda A, Santell R, Wu Z, Brown M, Wu Y, Khan I, Link CD, Zhao B, Luo Y. Soy isoflavone glycitein protects against beta amyloid-induced toxicity and oxidative stress in transgenic Caenorhabditis elegans. BMC Neurosci. 2005;6:54. doi: 10.1186/1471-2202-6-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.He Y, Tan D, Mi Y, Bai B, Jiang D, Zhou X, Ji S. Effect of epigallocatechin-3-gallate on acrylamide-induced oxidative stress and apoptosis in PC12 cells. Hum Exp Toxicol. 2017;36:1087–1099. doi: 10.1177/0960327116681648. [DOI] [PubMed] [Google Scholar]
- 25.Heo JH, Lee ST, Chu K, Oh MJ, Park HJ, Shim JY, Kim M. An open-label trial of Korean red ginseng as an adjuvant treatment for cognitive impairment in patients with Alzheimer’s disease. Eur J Neurol. 2008;15:865–868. doi: 10.1111/j.1468-1331.2008.02157.x. [DOI] [PubMed] [Google Scholar]
- 26.Jang SH, Park J, Kim SH, Choi KM, Ko ES, Cha JD, Lee YR, Jang H, Jang YS. Red ginseng powder fermented with probiotics exerts antidiabetic effects in the streptozotocin-induced mouse diabetes model. Pharm Biol. 2017;55:317–323. doi: 10.1080/13880209.2016.1237978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jeong SI, Shin JA, Cho S, Kim HW, Lee JY, Kang JL, Park EM. Resveratrol attenuates peripheral and brain inflammation and reduces ischemic brain injury in aged female mice. Neurobiol Aging. 2016;44:74–84. doi: 10.1016/j.neurobiolaging.2016.04.007. [DOI] [PubMed] [Google Scholar]
- 28.Jin S, Jeon JH, Lee S, Kang WY, Seong SJ, Yoon YR, Choi MK, Song IS. Detection of 13 ginsenosides (Rb1, Rb2, Rc, Rd, Re, Rf, Rg1, Rg3, Rh2, F1, compound K, 20(S)-protopanaxadiol, and 20(S)-protopanaxatriol) in human plasma and application of the analytical method to human pharmacokinetic studies following two week-repeated administration of red ginseng extract. Molecules. 2019;24:E2618. doi: 10.3390/molecules24142618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kim HY, Park EJ, Joe EH, Jou I. Curcumin suppresses Janus kinase-STAT inflammatory signaling through activation of Src homology 2 domain-containing tyrosine phosphatase 2 in brain microglia. J Immunol. 2003;171:6072–6079. doi: 10.4049/jimmunol.171.11.6072. [DOI] [PubMed] [Google Scholar]
- 30.Kim J, Funayama S, Izuo N, Shimizu T. Dietary supplementation of a high-temperature-processed green tea extract attenuates cognitive impairment in PS2 and Tg2576 mice. Biosci Biotechnol Biochem. 2019;83:2364–2371. doi: 10.1080/09168451.2019.1659721. [DOI] [PubMed] [Google Scholar]
- 31.Lee JC, Park JH, Park OK, Kim IH, Yan BC, Ahn JH, Kwon SH, Choi JH, Kim JD, Won MH. Neuroprotective effects of tanshinone I from Danshen extract in a mouse model of hypoxia-ischemia. Anat Cell Biol. 2013;46:183–190. doi: 10.5115/acb.2013.46.3.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lei X, Chao H, Zhang Z, Lv J, Li S, Wei H, Xue R, Li F, Li Z. Neuroprotective effects of quercetin in a mouse model of brain ischemic/reperfusion injury via anti-apoptotic mechanisms based on the Akt pathway. Mol Med Rep. 2015;12:3688–3696. doi: 10.3892/mmr.2015.3857. [DOI] [PubMed] [Google Scholar]
- 33.Leipold RJ. Description and simulation of a tubular, plug-flow model to predict the effect of bile sequestrants on human bile salt excretion. J Pharm Sci. 1995;84:670–672. doi: 10.1002/jps.2600840528. [DOI] [PubMed] [Google Scholar]
- 34.Li J, Wen PY, Li WW, Zhou J. Upregulation effects of Tanshinone IIA on the expressions of NeuN, Nissl body, and IkappaB and downregulation effects on the expressions of GFAP and NF-kappaB in the brain tissues of rat models of Alzheimer’s disease. Neuroreport. 2015a;26:758–766. doi: 10.1097/WNR.0000000000000419. [DOI] [PubMed] [Google Scholar]
- 35.Li W, Tan C, Liu Y, Liu X, Wang X, Gui Y, Qin L, Deng F, Yu Z, Hu C, Chen L. Resveratrol ameliorates oxidative stress and inhibits aquaporin 4 expression following rat cerebral ischemia-reperfusion injury. Mol Med Rep. 2015b;12:7756–7762. doi: 10.3892/mmr.2015.4366. [DOI] [PubMed] [Google Scholar]
- 36.Li Y, Guan Y, Wang Y, Yu CL, Zhai FG, Guan LX. Neuroprotective effect of the Ginsenoside Rg1 on cerebral ischemic injury in vivo and in vitro is mediated by PPARgamma-regulated antioxidative and anti-inflammatory pathways. Evid Based Complement Alternat Med. 2017;2017:7842082. doi: 10.1155/2017/7842082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Liu L, Zhang Q, Cai Y, Sun D, He X, Wang L, Yu D, Li X, Xiong X, Xu H, Yang Q, Fan X. Resveratrol counteracts lipopolysaccharide-induced depressive-like behaviors via enhanced hippocampal neurogenesis. Oncotarget. 2016;7:56045–56059. doi: 10.18632/oncotarget.11178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liu W, Fan Z, Han Y, Lu S, Zhang D, Bai X, Xu W, Li J, Wang H. Curcumin attenuates peroxynitrite-induced neurotoxicity in spiral ganglion neurons. Neurotoxicology. 2011;32:150–157. doi: 10.1016/j.neuro.2010.09.003. [DOI] [PubMed] [Google Scholar]
- 39.Ma Z, Bai L. The anti-inflammatory effect of Z-Ligustilide in experimental ovariectomized osteopenic rats. Inflammation. 2012;35:1793–1797. doi: 10.1007/s10753-012-9499-5. [DOI] [PubMed] [Google Scholar]
- 40.Namazi N, Saneei P, Larijani B, Esmaillzadeh A. Soy product consumption and the risk of all-cause, cardiovascular and cancer mortality: a systematic review and meta-analysis of cohort studies. Food Funct. 2018;9:2576–2588. doi: 10.1039/c7fo01622k. [DOI] [PubMed] [Google Scholar]
- 41.Ohishi T, Goto S, Monira P, Isemura M, Nakamura Y. Anti-inflammatory action of green tea. Antiinflamm Antiallergy Agents Med Chem. 2016;15:74–90. doi: 10.2174/1871523015666160915154443. [DOI] [PubMed] [Google Scholar]
- 42.O’Shaughnessy CT, Lodge D. N-methyl-D-aspartate receptor-mediated increase in intracellular calcium is reduced by ketamine and phencyclidine. Eur J Pharmacol. 1988;153:201–209. doi: 10.1016/0014-2999(88)90607-3. [DOI] [PubMed] [Google Scholar]
- 43.Ouberai M, Dumy P, Chierici S, Garcia J. Synthesis and biological evaluation of clicked curcumin and clicked KLVFFA conjugates as inhibitors of beta-amyloid fibril formation. Bioconjug Chem. 2009;20:2123–2132. doi: 10.1021/bc900281b. [DOI] [PubMed] [Google Scholar]
- 44.Pan J, Li H, Ma JF, Tan YY, Xiao Q, Ding JQ, Chen SD. Curcumin inhibition of JNKs prevents dopaminergic neuronal loss in a mouse model of Parkinson’s disease through suppressing mitochondria dysfunction. Transl Neurodegener. 2012;1:16. doi: 10.1186/2047-9158-1-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pandey AK, Bhattacharya P, Shukla SC, Paul S, Patnaik R. Resveratrol inhibits matrix metalloproteinases to attenuate neuronal damage in cerebral ischemia: a molecular docking study exploring possible neuroprotection. Neural Regen Res. 2015;10:568–575. doi: 10.4103/1673-5374.155429. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 46.Park S, Kim DS, Kang S, Moon BR. Fermented soybeans, Chungkookjang, prevent hippocampal cell death and beta-cell apoptosis by decreasing pro-inflammatory cytokines in gerbils with transient artery occlusion. Exp Biol Med. 2016a;241:296–307. doi: 10.1177/1535370215606811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Park SY, Shin YK, Kim HT, Kim YM, Lee DG, Hwang E, Cho BG, Yin CS, Kim KY, Yi TH. A single-center, randomized, double-blind, placebo-controlled study on the efficacy and safety of “enzyme-treated red ginseng powder complex (BG11001)” for antiwrinkle and proelasticity in individuals with healthy skin. J Ginseng Res. 2016b;40:260–268. doi: 10.1016/j.jgr.2015.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Prado Lima MG, Schimidt HL, Garcia A, Dare LR, Carpes FP, Izquierdo I, Mello-Carpes PB. Environmental enrichment and exercise are better than social enrichment to reduce memory deficits in amyloid beta neurotoxicity. Proc Natl Acad Sci U S A. 2018;115:E2403-2409. doi: 10.1073/pnas.1718435115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Razgonova MP, Veselov VV, Zakharenko AM, Golokhvast KS, Nosyrev AE, Cravotto G, Tsatsakis A, Spandidos DA. Panax ginseng components and the pathogenesis of Alzheimer’s disease (Review) Mol Med Rep. 2019;19:2975–2998. doi: 10.3892/mmr.2019.9972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Reay JL, van Schaik P, Wilson CJ. A systematic review of research investigating the physiological and psychological effects of combining Ginkgo biloba and Panax ginseng into a single treatment in humans: Implications for research design and analysis. Brain Behav. 2019;9:e01217. doi: 10.1002/brb3.1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sakr HF, Abbas AM, El Samanoudy AZ. Effect of vitamin E on cerebral cortical oxidative stress and brain-derived neurotrophic factor gene expression induced by hypoxia and exercise in rats. J Physiol Pharmacol. 2015;66:191–202. [PubMed] [Google Scholar]
- 52.Shao H, Xue Q, Zhang F, Luo Y, Zhu H, Zhang X, Zhang H, Ding W, Yu B. Spinal SIRT1 activation attenuates neuropathic pain in mice. PLoS One. 2014;9:e100938. doi: 10.1371/journal.pone.0100938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Shi ZY, Zeng JZ, Wong AST. Chemical structures and pharmacological profiles of Ginseng Saponins. Molecules. 2019;24:E2443. doi: 10.3390/molecules24132443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Shirani A, Okuda DT, Stuve O. Therapeutic advances and future prospects in progressive forms of multiple sclerosis. Neurotherapeutics. 2016;13:58–69. doi: 10.1007/s13311-015-0409-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Simoes AP, Silva CG, Marques JM, Pochmann D, Porciuncula LO, Ferreira S, Oses JP, Beleza RO, Real JI, Kofalvi A, Bahr BA, Lerma J, Cunha RA, Rodrigues RJ. Glutamate-induced and NMDA receptor-mediated neurodegeneration entails P2Y1 receptor activation. Cell Death Dis. 2018;9:297. doi: 10.1038/s41419-018-0351-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sonee M, Sum T, Wang C, Mukherjee SK. The soy isoflavone, genistein, protects human cortical neuronal cells from oxidative stress. Neurotoxicology. 2004;25:885–891. doi: 10.1016/j.neuro.2003.11.001. [DOI] [PubMed] [Google Scholar]
- 57.Su YW, Chiou WF, Chao SH, Lee MH, Chen CC, Tsai YC. Ligustilide prevents LPS-induced iNOS expression in RAW 264.7 macrophages by preventing ROS production and down-regulating the MAPK, NF-kappaB and AP-1 signaling pathways. Int Immunopharmacol. 2011;11:1166–1172. doi: 10.1016/j.intimp.2011.03.014. [DOI] [PubMed] [Google Scholar]
- 58.Tiwari SK, Agarwal S, Tripathi A, Chaturvedi RK. Bisphenol-a mediated inhibition of hippocampal neurogenesis attenuated by curcumin via canonical wnt pathway. Mol Neurobiol. 2016;53:3010–3029. doi: 10.1007/s12035-015-9197-z. [DOI] [PubMed] [Google Scholar]
- 59.Tyagi N, De R, Begun J, Popat A. Cancer therapeutics with epigallocatechin-3-gallate encapsulated in biopolymeric nanoparticles. Int J Pharm. 2016;518:220–227. doi: 10.1016/j.ijpharm.2016.12.030. [DOI] [PubMed] [Google Scholar]
- 60.Wan L, Cheng Y, Luo Z, Guo H, Zhao W, Gu Q, Yang X, Xu J, Bei W, Guo J. Neuroprotection, learning and memory improvement of a standardized extract from Renshen Shouwu against neuronal injury and vascular dementia in rats with brain ischemia. J Ethnopharmacol. 2015;165:118–126. doi: 10.1016/j.jep.2015.02.027. [DOI] [PubMed] [Google Scholar]
- 61.Wang G, Dai F, Yu K, Jia Z, Zhang A, Huang Q, Kang C, Jiang H, Pu P. Resveratrol inhibits glioma cell growth via targeting oncogenic microRNAs and multiple signaling pathways. Int J Oncol. 2015a;46:1739–1747. doi: 10.3892/ijo.2015.2863. [DOI] [PubMed] [Google Scholar]
- 62.Wang J, Xie Y, Feng Y, Zhang L, Huang X, Shen X, Luo X. (-)-Epigallocatechingallate induces apoptosis in B lymphoma cells via caspase-dependent pathway and Bcl-2 family protein modulation. Int J Oncol. 2015b;46:1507–1515. doi: 10.3892/ijo.2015.2869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang KW, Ye XL, Huang T, Yang XF, Zou LY. Optogenetics-induced activation of glutamate receptors improves memory function in mice with Alzheimer’s disease. Neural Regen Res. 2019;14:2147–2155. doi: 10.4103/1673-5374.262593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wang Y, Fu XT, Li DW, Wang K, Wang XZ, Li Y, Sun BL, Yang XY, Zheng ZC, Cho NC. Cyanidin suppresses amyloid beta-induced neurotoxicity by inhibiting reactive oxygen species-mediated DNA damage and apoptosis in PC12 cells. Neural Regen Res. 2016a;11:795–800. doi: 10.4103/1673-5374.182707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wang Z, Qi F, Cui Y, Zhao L, Sun X, Tang W, Cai P. An update on Chinese herbal medicines as adjuvant treatment of anticancer therapeutics. Biosci Trends. 2018;12:220–239. doi: 10.5582/bst.2018.01144. [DOI] [PubMed] [Google Scholar]
- 66.Wang Z, Yu J, Wu J, Qi F, Wang H, Wang Z, Xu Z. Scutellarin protects cardiomyocyte ischemia-reperfusion injury by reducing apoptosis and oxidative stress. Life Sci. 2016b;157:200–207. doi: 10.1016/j.lfs.2016.01.018. [DOI] [PubMed] [Google Scholar]
- 67.Wei IH, Tu HC, Huang CC, Tsai MH, Tseng CY, Shieh JY. (-)-Epigallocatechin gallate attenuates NADPH-d/nNOS expression in motor neurons of rats following peripheral nerve injury. BMC Neurosci. 2011;12:52. doi: 10.1186/1471-2202-12-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Xiao W, Yu A, Liu D, Shen J, Xu Z. Ligustilide treatment promotes functional recovery in a rat model of spinal cord injury via preventing ROS production. Int J Clin Exp Pathol. 2015;8:12005–12013. [PMC free article] [PubMed] [Google Scholar]
- 69.Xing L, Zhang H, Qi R, Tsao R, Mine Y. Recent advances in the understanding of the health benefits and molecular mechanisms associated with green tea polyphenols. J Agric Food Chem. 2019;67:1029–1043. doi: 10.1021/acs.jafc.8b06146. [DOI] [PubMed] [Google Scholar]
- 70.Xu QQ, Xu YJ, Yang C, Tang Y, Li L, Cai HB, Hou BN, Chen HF, Wang Q, Shi XG, Zhang SJ. Sodium tanshinone iia sulfonate attenuates scopolamine-induced cognitive dysfunctions via improving cholinergic system. BioMed Res Int. 2016;2016:9852536. doi: 10.1155/2016/9852536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Yang JY, Zhong X, Kim SJ, Kim DH, Kim HS, Lee JS, Yum HW, Lee J, Na HK, Surh YJ. Comparative effects of curcumin and tetrahydrocurcumin on dextran sulfate sodium-induced colitis and inflammatory signaling in mice. J Cancer Prev. 2018;23:18–22. doi: 10.15430/JCP.2018.23.1.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Yang X, Zheng T, Hong H, Cai N, Zhou X, Sun C, Wu L, Liu S, Zhao Y, Zhu L, Fan M, Zhou X, Jin F. Neuroprotective effects of Ginkgo biloba extract and Ginkgolide B against oxygen-glucose deprivation/reoxygenation and glucose injury in a new in vitro multicellular network model. Front Med. 2018;12:307–318. doi: 10.1007/s11684-017-0547-2. [DOI] [PubMed] [Google Scholar]
- 73.Yu Q, Zeng KW, Ma XL, Jiang Y, Tu PF, Wang XM. Ginsenoside Rk1 suppresses pro-inflammatory responses in lipopolysaccharide-stimulated RAW264.7 cells by inhibiting the Jak2/Stat3 pathway. Chin J Nat Med. 2017;15:751–757. doi: 10.1016/S1875-5364(17)30106-1. [DOI] [PubMed] [Google Scholar]
- 74.Zahr SK, Yang G, Kazan H, Borrett MJ, Yuzwa SA, Voronova A, Kaplan DR, Miller FD. A Translational repression complex in developing mammalian neural stem cells that regulates neuronal specification. Neuron. 2018;97:520–537. doi: 10.1016/j.neuron.2017.12.045. [DOI] [PubMed] [Google Scholar]
- 75.Zhang Y, Xu YY, Sun WJ, Zhang MH, Zheng YF, Shen HM, Yang J, Zhu XQ. FBS or BSA inhibits EGCG induced cell death through covalent binding and the reduction of intracellular ROS production. BioMed Res Int. 2016;2016:5013409. doi: 10.1155/2016/5013409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zhou YD, Hou JG, Liu W, Ren S, Wang YP, Zhang R, Chen C, Wang Z, Li W. 20(R)-ginsenoside Rg3, a rare saponin from red ginseng, ameliorates acetaminophen-induced hepatotoxicity by suppressing PI3K/AKT pathway-mediated inflammation and apoptosis. Int Immunopharmacol. 2018;59:21–30. doi: 10.1016/j.intimp.2018.03.030. [DOI] [PubMed] [Google Scholar]


