Abstract
Background
As the causative agent of COVID-19, SARS-CoV-2 is a pathogen of immense importance to global public health. Development of innovative direct-acting antiviral agents is sorely needed to address this virus. Peptide-conjugated morpholino oligomers (PPMO) are antisense compounds composed of a phosphorodiamidate morpholino oligomer covalently conjugated to a cell-penetrating peptide. PPMO require no delivery assistance to enter cells and are able to reduce expression of targeted RNA through sequence-specific steric blocking.
Methods
Five PPMO designed against sequences of genomic RNA in the SARS-CoV-2 5′-untranslated region and a negative control PPMO of random sequence were synthesized. Each PPMO was evaluated for its effect on the viability of uninfected cells and its inhibitory effect on the replication of SARS-CoV-2 in Vero-E6 cell cultures. Cell viability was evaluated with an ATP-based method using a 48 h PPMO treatment time. Viral growth was measured with quantitative RT–PCR and TCID50 infectivity assays from experiments where cells received a 5 h PPMO treatment time.
Results
PPMO designed to base-pair with sequence in the 5′ terminal region or the leader transcription regulatory sequence region of SARS-CoV-2 genomic RNA were highly efficacious, reducing viral titres by up to 4–6 log10 in cell cultures at 48–72 h post-infection, in a non-toxic and dose-responsive manner.
Conclusions
The data indicate that PPMO have the ability to potently and specifically suppress SARS-CoV-2 growth and are promising candidates for further preclinical development.
Introduction
There is a pressing need for the development of additional direct-acting antiviral agents to address infections with SARS-CoV-2, the causative agent of COVID-19. Peptide-conjugated morpholino oligomers (PPMO) are single-stranded nucleic acid analogues capable of entering cells without assistance and interfering with gene expression through steric blockade of targeted RNA. PPMO are composed of a phosphorodiamidate morpholino oligomer (PMO) covalently conjugated to a cell-penetrating peptide (CPP).1–3 PPMO are water soluble, nuclease resistant and non-toxic at effective concentrations across a range of in vitro and in vivo applications.1,4 In several in vivo models of respiratory viruses, including influenza A virus,5,6 respiratory syncytial virus7 and porcine reproductive and respiratory syndrome virus,8 intranasally administered PPMO targeted against virus sequence have reduced viral titre and pathology in lung tissue.
The 5′ untranslated region (5′UTR) of the coronavirus genome contains sequences and structures known to be important in various aspects of the virus life cycle, including translation and RNA synthesis.9 In previous studies, PPMO targeted to various sites in the 5′UTR of mouse hepatitis virus10,11 and SARS-CoV12 were effectively antiviral. In the present study we investigated the ability of five PPMO targeted against various sites in the 5′UTR and polyprotein 1a/b translation start site (AUG) region of SARS-CoV-2 to suppress virus growth in cell cultures. We found that PPMO targeting the 5′ terminal region and the leader transcription regulatory sequence (TRS) region of genomic RNA were potent inhibitors of SARS-CoV-2 replication.
Materials and methods
Biosafety
Work with infectious SARS-CoV-2 was approved by the Institutional Biosafety Committee (IBC) and performed in high biocontainment at Rocky Mountain Laboratories (RML), NIAID, NIH. Sample removal from high biocontainment followed IBC-approved Standard Operating Protocols.
PPMO synthesis
PPMO were synthesized by covalently conjugating the CPP (RXR)4 (where R is arginine and X is 6-aminohexanoic acid) to PMO (Gene Tools LLC, Philomath, OR, USA) at the 3′ end through a non-cleavable linker, by methods described previously.3
Cells and viruses
Vero-E6 cells (ATCC) were maintained in DMEM supplemented with 10% FCS, 1 mM l-glutamine, 50 U/mL penicillin and 50 μg/mL streptomycin (growth medium). All cell culture incubations were carried out at 37°C and 5% CO2. SARS-CoV-2 isolate nCoV-WA1-2020 was kindly provided by the CDC (Atlanta, GA, USA). Preparation and quantification of the virus followed methods described previously.13 Briefly, the original virus stock was propagated once at RML in Vero-E6 cells in DMEM supplemented with 2% FBS containing l-glutamine and antibiotics as above (infection medium). The virus stock used in the experiments was passage 4 and was confirmed by next-generation sequencing to be identical to the initial deposited GenBank sequence MN985325.
Cell viability assay
Cell viability was assessed using CellTiterGlo (Promega). Vero-E6 cells grown to approximately 80% confluence in 96-well plates were incubated in the presence of PPMO in infection medium for 48 h, then assayed according to the manufacturer’s instructions. Statistical analysis was carried out using GraphPad Prizm (San Diego, CA, USA).
Antiviral assays
PPMO were resuspended in sterile PBS. Vero-E6 cells were plated in 48-well plates and were approximately 80% confluent on the day of infection. At 5 h before infection, the growth medium was removed and replaced with infection medium containing PPMO. For viral infections, the PPMO-containing medium was aspirated and the cells rinsed twice with DMEM before adding 100 μL of infection medium containing virus at an moi of 0.01. Following a 1 h infection period, the virus-containing inoculum was aspirated and the cells washed twice with infection medium, after which 300 μL of fresh infection medium without PPMO was added. At 12, 24, 48 and 72 h post-infection, all of the medium in each well was collected for quantitative RT–PCR (qRT–PCR) and TCID50 analysis.
Evaluation of virus quantity by qRT–PCR
Supernatants were harvested as described above and viral RNA purified and quantified by using one-step qRT–PCR following methods described previously.13 Briefly, total RNA was isolated with the RNeasy Mini Kit (Qiagen) and RT–PCR carried out using the One-Step RT–PCR Kit (Qiagen) according to the manufacturer’s protocols. Ct values were calculated using standards produced as described previously.13
TCID50 evaluations
Viral supernatants were serially diluted in DMEM and each dilution sample was titrated in triplicate. Subsequently, 100 μL of each virus dilution was transferred to Vero-E6 cells grown in 96-well plates containing 100 μL of DMEM. Following a 7 day incubation period, wells were scored for cytopathic effect. The TCID50 values were calculated via the Reed–Muench formula.
Results
PPMO design
PPMO design was guided by previous studies in which various nidoviruses were targeted with PPMO.10–12,14,15 In this study, five PPMO were designed to target the 5′UTR and first translation start site region of SARS-CoV-2 positive-sense genomic RNA (Table 1). Two of the PPMO target the 5′ terminal region of the genome. Both 5′END-1 and 5′END-2 were designed with the intention of interfering with pre-initiation of translation of genomic and subgenomic mRNAs.
Table 1.
Names, sequences and target locations of SARS-CoV-2 PPMO
| PPMO name | PPMO sequence (5′-3′) | PPMO target location in the SARS-CoV-2 genomea |
|---|---|---|
| NC | CCTCTTACCTCAGTTACAATTTATA | not applicable (negative control) |
| 5′END-1 | CCTGGGAAGGTATAAACCTTTAAT | 1–24 |
| 5′END-2 | TGTTACCTGGGAAGGTATAAACCTT | 5–29 |
| TRS-1 | TTTTAAAGTTCGTTTAGAGAACAG | 59–82 |
| TRS-2 | AAGTTCGTTTAGAGAACAGATCTAC | 53–77 |
| AUGb | AGGCTCTCCATCTTACCTTTCGGT | 251–275 |
Based on GenBank NC_045512.
Bases targeting the AUG translation start site of SARS-CoV-2 1a/b polyprotein are in bold.
Coronaviruses produce a set of nested mRNAs through the process of discontinuous subgenomic mRNA synthesis. The TRS is a 6–10 nt sequence that is critical in the production of negative-strand mRNA templates during this process.16,17 For SARS-CoV (GenBank NC_004718), the leader-TRS core sequence consists of nt 67–72 of the genomic RNA18 and most if not all viral subgenomic mRNAs possess the same 72 nt 5′ leader sequence. Two PPMO were designed to target the genomic 5′UTR region containing the putative SARS-CoV-2 leader-TRS core sequence (nt 70–75, 5′-ACGAAC-3′) and thereby potentially interfere with body-TRS to leader-TRS base-pairing and/or with translocation of the 48S translation preinitiation complex along the 5′UTR of the genomic and subgenomic mRNAs.
The AUG PPMO was designed to target the translation initiation region for ORF1a/b, which codes for the replicase polyprotein, with the intention to block translation initiation.
A negative control PPMO (NC), consisting of a random-sequence PMO conjugated to the same CPP used in the other PPMO, was included (see Table 1), to control for non-specific effects of the PPMO chemistry. NC was screened using BLAST and contains little significant homology to any primate, rodent or viral sequences.
Evaluation of PPMO cytotoxicity
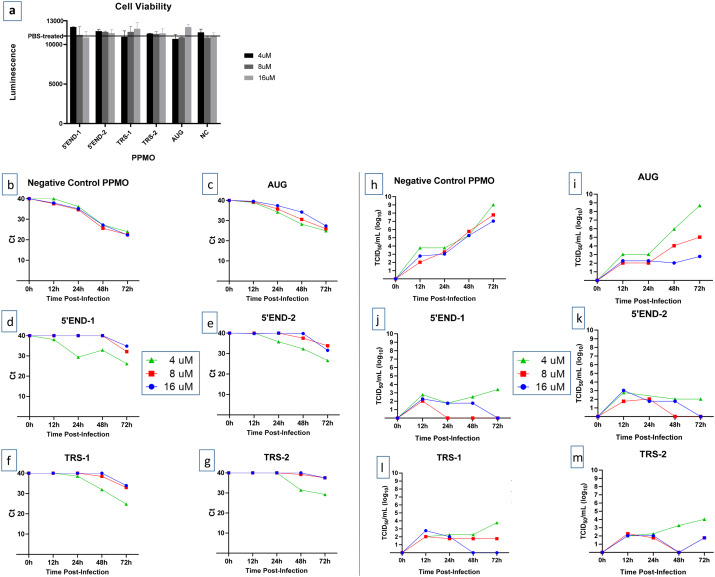
To evaluate the effect of PPMO on cell viability, cells were treated under conditions similar to those used in the antiviral assays described below, but for a longer treatment time and in the absence of virus. Cells were treated in triplicate with increasing doses of PPMO for 48 h then evaluated with a quantitative cell viability assay. At the concentrations used in the antiviral assays described below, none of the PPMO produced more than a 5% cytotoxic effect (Figure 1a).
Figure 1.
Effect of PPMO on cell viability and SARS-CoV-2 growth. (a) Evaluation of the effect of PPMO treatment on cellular ATP level, as an indicator of cell viability, was carried out using uninfected cells incubated for 48 h with increasing concentrations of the indicated PPMO. ATP levels were determined via luminescence readings and are shown compared with PBS-treated cells. For each PPMO concentration, triplicate samples were assayed and the mean±SD is shown. (b–m) Growth curves of SARS-CoV-2. Vero-E6 cells were treated with the indicated concentration of PPMO for 5 h before infection with SARS-CoV-2 (moi of 0.01), then incubated without PPMO after infection. Cell supernatants were collected at 12, 24, 48 and 72 h post-infection and analysed by qRT–PCR (b–g) or TCID50/mL endpoint dilution (h–m) using three technical repeats. Cells treated with PBS had titres similar to those shown for NC PPMO. The limit of virus detection for the TCID50 assay was 101/mL. This experiment was carried out twice, under similar conditions, yielding similar results, and the results from a single experiment are shown. This figure appears in colour in the online version of JAC and in black and white in the print version of JAC.
Evaluation of PPMO antiviral activity
To determine the inhibitory effect of the various PPMO on SARS-CoV-2 replication, Vero-E6 cells were treated with each of the six PPMO described in Table 1 at 4, 8 and 16 μM for 5 h before infection, then incubated without PPMO after infection. Cell supernatants were collected at four timepoints post-infection: 12, 24, 48 and 72 h. Virus growth was evaluated by two methods, qRT–PCR and TCID50 infectivity assay. Using an moi of 0.01, virus growth rose steadily and reached peak growth at 72 h post-infection (Figure 1b and h). Growth of the virus under PBS treatment was highly similar to virus growth under NC PPMO treatment (data not shown). Four of the five PPMO designed to target SARS-CoV-2 RNA were highly effective, suppressing viral titres by 4–6 log10 at the 48 and 72 h timepoints (Figure 1h–m). qRT–PCR analysis showed that in cells treated with 8 or 16 μM of any of the four PPMO targeting the 5′ terminal or TRS regions, virus growth was markedly suppressed at 12–48 h post-infection (Figure 1b–g). The number of cycles required to detect virus from those samples was approximately the same as the number of cycles required to detect the input virus shortly after infection. At 72 h post-infection, the efficacy of the 5′END and TRS PPMO waned, to a minor extent, although still providing considerable suppression of virus growth. Overall, the AUG PPMO was less effective than the other four antiviral PPMO used in this study (Figure 1c and i).
Discussion
In this study, we found that PPMO targeting the 5′ terminal region or leader-TRS region were highly effective at inhibiting the growth of SARS-CoV-2, whereas a PPMO targeting the polyprotein 1a/b AUG translation start site region was less effective. It is unknown if the AUG PPMO was less able to anneal to its target or if duplexing did occur yet was less consequential to virus growth.
To date, little sequence variation in the PPMO target sites in the 5′UTR of SARS-CoV-2 has been identified. Of the whole-genome nucleotide sequences reported in GenBank (at the time of writing), two genotypes contain a single mismatch with the 5′END PPMO and one genome has a single mismatch with the TRS PPMO.19 Previous studies have reported that PPMO having a single base mismatch with their target site retain approximately 90% of their activity compared with those having perfect agreement.15,20
This study demonstrates that PPMO targeted against SARS-CoV-2 can enter cells readily and inhibit viral replication in a sequence-specific, dose-responsive and non-toxic manner. As well, the PPMO exhibited durable potency, considering that only a 5 h PPMO treatment time was used. Our results here, in addition to the in vivo antiviral efficacy demonstrated by PPMO against several respiratory viruses in previous studies,5–8 suggest that further development of the 5′END and TRS PPMO to address SARS-CoV-2 infections is warranted.
Funding
This work was funded by the Intramural Research Program of the National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH).
Transparency declarations
None to declare.
Contributor Information
Kyle Rosenke, Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Hamilton, MT, USA.
Shanna Leventhal, Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Hamilton, MT, USA.
Hong M Moulton, Department of Biomedical Sciences, Carlson College of Veterinary Medicine, Oregon State University, Corvallis, OR, USA.
Susan Hatlevig, Department of Biomedical Sciences, Carlson College of Veterinary Medicine, Oregon State University, Corvallis, OR, USA.
David Hawman, Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Hamilton, MT, USA.
Heinz Feldmann, Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Hamilton, MT, USA.
David A Stein, Department of Biomedical Sciences, Carlson College of Veterinary Medicine, Oregon State University, Corvallis, OR, USA.
References
- 1. Moulton HM, Moulton JD. Morpholinos and their peptide conjugates: therapeutic promise and challenge for Duchenne muscular dystrophy. Biochim Biophys Acta 2010; 1798: 2296–303. [DOI] [PubMed] [Google Scholar]
- 2. Moulton HM, Moulton JD, Antisense morpholino oligomers and their peptide conjugates In: Kurreck J, ed. Therapeutic Oligonucleotides. Royal Society of Chemistry, 2008; 43–79. [Google Scholar]
- 3. Abes S, Moulton HM, Clair P. et al. Vectorization of morpholino oligomers by the (R-Ahx-R)4 peptide allows efficient splicing correction in the absence of endosomolytic agents. J Control Release 2006; 116: 304–13. [DOI] [PubMed] [Google Scholar]
- 4. Stein DA. Inhibition of RNA virus infections with peptide-conjugated morpholino oligomers. Curr Pharm Des 2008; 14: 2619–34. [DOI] [PubMed] [Google Scholar]
- 5. Gabriel G, Nordmann A, Stein DA. et al. Morpholino oligomers targeting the PB1 and NP genes enhance the survival of mice infected with highly pathogenic influenza A H7N7 virus. J Gen Virol 2008; 89: 939–48. [DOI] [PubMed] [Google Scholar]
- 6. Lupfer C, Stein DA, Mourich DV. et al. Inhibition of influenza A H3N8 virus infections in mice by morpholino oligomers. Arch Virol 2008; 153: 929–37. [DOI] [PubMed] [Google Scholar]
- 7. Lai SH, Stein DA, Guerrero-Plata A. et al. Inhibition of respiratory syncytial virus infections with morpholino oligomers in cell cultures and in mice. Mol Ther 2008; 16: 1120–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Opriessnig T, Patel D, Wang R. et al. Inhibition of porcine reproductive and respiratory syndrome virus infection in piglets by a peptide-conjugated morpholino oligomer. Antiviral Res 2011; 91: 36–42. [DOI] [PubMed] [Google Scholar]
- 9. Masters PS, Perlman S, Coronaviridae In: Knipe DM, Howley PM, eds. Fields Virology. Lippincott Williams & Wilkins, 2013; 825–58. [Google Scholar]
- 10. Burrer R, Neuman BW, Ting JP. et al. Antiviral effects of antisense morpholino oligomers in murine coronavirus infection models. J Virol 2007; 81: 5637–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Neuman BW, Stein DA, Kroeker AD. et al. Antisense morpholino-oligomers directed against the 5′ end of the genome inhibit coronavirus proliferation and growth. J Virol 2004; 78: 5891–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Neuman BW, Stein DA, Kroeker AD. et al. Inhibition, escape, and attenuated growth of severe acute respiratory syndrome coronavirus treated with antisense morpholino oligomers. J Virol 2005; 79: 9665–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Munster VJ, Feldmann F, Williamson BN. et al. Respiratory disease in rhesus macaques inoculated with SARS-CoV-2. Nature 2020; 585: 268–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. van den Born E, Stein DA, Iversen PL. et al. Antiviral activity of morpholino oligomers designed to block various aspects of Equine arteritis virus amplification in cell culture. J Gen Virol 2005; 86: 3081–90. [DOI] [PubMed] [Google Scholar]
- 15. Zhang YJ, Stein DA, Fan SM. et al. Suppression of porcine reproductive and respiratory syndrome virus replication by morpholino antisense oligomers. Vet Microbiol 2006; 117: 117–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sawicki SG, Sawicki DL, Siddell SG. A contemporary view of coronavirus transcription. J Virol 2007; 81: 20–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Pasternak AO, Spaan WJ, Snijder EJ. Nidovirus transcription: how to make sense…? J Gen Virol 2006; 87: 1403–21. [DOI] [PubMed] [Google Scholar]
- 18. Yang D, Leibowitz JL. The structure and functions of coronavirus genomic 3′ and 5′ ends. Virus Res 2015; 206: 120–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Khailany RA, Safdar M, Ozaslan M. Genomic characterization of a novel SARS-CoV-2. Gene Rep 2020; 19: 100682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ge Q, Pastey M, Kobasa D. et al. Inhibition of multiple subtypes of influenza A virus in cell cultures with morpholino oligomers. Antimicrob Agents Chemother 2006; 50: 3724–33. [DOI] [PMC free article] [PubMed] [Google Scholar]