Abstract
The rapamycin analogue everolimus (EVR) is a potent inhibitor of the mammalian target of rapamycin (mTOR) and clinically used to prevent allograft rejections as well as tumor growth. The pharmacokinetic and immunosuppressive efficacy of EVR have been extensively reported in patient populations and in vitro studies. However, dose‐dependent ex vivo effects upon acute EVR administration in healthy volunteers are rare. Moreover, immunosuppressive drugs are associated with neuroendocrine changes and psychological disturbances. It is largely unknown so far whether and to what extend EVR affects neuroendocrine functions, mood, and anxiety in healthy individuals. Thus, in the present study, we analyzed the effects of three different clinically applied EVR doses (1.5, 2.25, and 3 mg) orally administered 4 times in a 12‐hour cycle to healthy male volunteers on immunological, neuroendocrine, and psychological parameters. We observed that oral intake of medium (2.25 mg) and high doses (3 mg) of EVR efficiently suppressed T cell proliferation as well as IL‐10 cytokine production in ex vivo mitogen‐stimulated peripheral blood mononuclear cell. Further, acute low (1.5 mg) and medium (2.25 mg) EVR administration increased state anxiety levels accompanied by significantly elevated noradrenaline (NA) concentrations. In contrast, high‐dose EVR significantly reduced plasma and saliva cortisol as well as NA levels and perceived state anxiety. Hence, these data confirm the acute immunosuppressive effects of the mTOR inhibitor EVR and provide evidence for EVR‐induced alterations in neuroendocrine parameters and behavior under physiological conditions in healthy volunteers.
Study Highlights.
WHAT IS THE CURRENT KNOWLEDGE ON THE TOPIC?
☑ Everolimus (EVR) is a potent inhibitor of mTOR and clinically used to prevent organ rejection and to fight tumor growth. EVR is associated with neurobehavioral and psychological changes in patient studies. However, the effects of EVR under normal physiological conditions are unknown.
WHAT QUESTION DID THIS STUDY ADDRESS?
☑ What is the impact of clinically employed doses of EVR on immunological, psychological and neuroendocrine parameters in healthy male individuals?
WHAT DOES THIS STUDY ADD TO OUR KNOWLEDGE?
☑ Our data demonstrate that oral intake of medium and high doses of EVR suppressed T cell proliferation as well as IL‐10 cytokine production in ex vivo mitogen‐stimulated PBMCs. Acute low and medium EVR administration increased state anxiety and noradrenaline concentrations. In contrast, high dose EVR reduced cortisol levels but did not affect state anxiety levels.
HOW MIGHT THIS CHANGE CLINICAL PHARMACOLOGY OR TRANSLATIONAL SCIENCE?
☑ These findings demonstrate a dose dependent impact of EVR on state anxiety and neuroendocrine functions under normal physiological conditons in healthy individuals providing important information for monitoring drug efficacy and unwanted drug side effect for future clinical studies employing immunopharmacotherapy with EVR.
Everolimus (EVR) is a macrolide rapamycin analogue initially developed as an immunosuppressive drug with improved pharmacokinetic properties, including increased solubility, bioavailability, and reduced half‐life in comparison to other rapalogues. 1 , 2 EVR efficiently inhibits the mammalian target of rapamycin (mTOR), a serine/threonine protein kinase and substrate of the PI3K/Akt signaling pathway, crucial for the regulation of cellular homeostasis, including cell growth, proliferation, protein synthesis, cellular stress, and survival. 3 Further, mTOR has been shown to play an essential role in innate and adaptive immune responses either by regulating T cell and B cell proliferation and differentiation as well as inflammatory cytokine and antibody production. 2 , 4 So far, EVR is the only clinically used mTOR inhibitor approved by the US Food and Drug Administration (FDA) for the oral administration and treatment of malignancies, including breast carcinoma, gastrointestinal tract‐derived neuroendocrine tumors, renal cell carcinoma, and to prevent allograft rejection after heart, kidney, and liver transplantations. 1 , 2 , 5
Pharmacokinetic studies with transplant recipients obtaining EVR either with 0.75 mg/dose or 1.5 mg/dose twice a day combined with cyclosporin revealed maximal blood concentration of 11.1 ± 4.6 µg/L and 20.3 ± 8.0 µg/L, respectively, after 1 to 2 hours. 6 , 7 The elimination half‐life amounts for 18–32 hours and steady‐state is achieved between 4 and 7 days. 6 , 7 However, in patient studies, EVR intake is frequently accompanied with other immunosuppressive drugs like cyclosporin affecting the pharmacokinetic profile of EVR. 2 Pharmacokinetics derived from healthy probands receiving a single 2 mg and 4 mg EVR dose revealed maximal EVR blood concentrations of 17.9 ± 5.9 µg/L and 44.2 ± 13.3 µ/L, respectively, reached after 0.5 to 1 hour with an elimination half‐life of 32 hours 8 , 9 and that single doses up to 5 mg are well‐tolerated. 10
Experimental and clinical studies in animal models and patients showed the potent immunosuppressive efficacy of the rapamycin analogue EVR in vitro mediated by blocking lymphocyte proliferation and in vivo by preventing rejections of transplanted organs. 11 , 12 , 13 , 14 Further, impaired renal function exerted by other medication regimens solely using calcineurin inhibitors in patients improved after acute and chronic treatment with EVR. 15 , 16 However, in patient studies, it is difficult to discriminate whether the examined effects solely were induced by EVR because the patients usually obtain a combination therapy consisting of more than one immunosuppressive drug. 11 , 17 , 18 Accumulating clinical evidence document that patients receiving immunosuppressive medication, including calcineurin inhibitors like tacrolimus or cyclosporin A, frequently develop affective and cognitive dysfunctions, such as depression or anxiety, and exhibit neurotoxic side effects, including tremor and peripheral neuropathy potentially manifesting in psychoses and seizures. 19 , 20 , 21 , 22 Although a growing body of studies in experimental animal models and clinical trials documented neurobehavioral effects exerted by mTOR inhibitors, the observed findings are controversial. EVR treatment improves neuropsychological deficits and autism in patients with tuberous sclerosis complex 23 and has beneficial effects on memory and psychiatric symptoms in heart transplant recipients. 19 However mTOR inhibition also has been linked to depressive and anxiety‐like behavior in rodents and the induction of euphoria followed by melancholy, mimicking biopolar disorder in patients with breast cancer. 24 , 25 , 26 , 27 These observations indicate that mTOR inhibition is associated with neurobehavioral and psychiatric symptoms but the impact of comorbidities on the action of EVR is inconclusive since the findings have been acquired from patient studies. Hence, so far, no data exist documenting whether and to what extent clinically used doses of the mTOR inhibitor EVR affect cellular immunosuppressive responses, central functions on behavioral, neuroendocrine and psychological levels, and, thus, may contribute to central side effects, including anxiety in healthy individuals. Against this background, the present study determined systemic blood levels of EVR after an acute, four‐time oral administration of three different, pharmacological relevant doses of the drug along with its effects on T cell specific cytokine production and proliferation as well as behavioral and neuroendocrine parameters in healthy men.
METHODS
Study participants
Healthy male volunteers (n = 22) with a mean age of 28.18 ± 0.69 (age range 22–32 years) and a body mass index of 22.73 ± 0.33 (range 20.99–25.13) were recruited through public advertisement in the surrounding community. All participants had 12 years or more of formal education, which is equivalent to a high school education. All volunteers underwent an extensive physical and psychiatric assessment (self‐reported questionnaires and interviews about their medical history) along with an electrocardiogram, performed and subsequently evaluated by physicians of the Department of Nephrology at the University Hospital Essen. Furthermore, a full blood examination, including assessment of renal, liver, and thyroid parameters, was conducted. Detailed exclusion criteria were described in Supplementary Materials. Participants were completely informed about the study protocol, which was approved by the local ethics committee for human investigations of the University Hospital Essen (17‐7500‐BO) and conducted according to the principles of the Declaration of Helsinki. Additionally, as part of a larger study, it was registered in the German Clinical Trial Register (DRKS00017020). All probands gave written informed consent and received 200 euros for their participation. Due to ethical reasons, all participants of this explorative pilot study had to be completely informed about group allocation. Thus, samples collected from the participants were not randomized and no blinding regarding group assignment was applied. Participants of the three groups (i.e., 1.5/2.25/3.0 mg) did not differ in age, body mass index, or education (Table 1 ).
Table 1.
Sociodemographic, physiological, and psychological characteristics of the three study groups at baseline level
| EVR dose group, mg | ||||
|---|---|---|---|---|
| 1.5 (n = 6) | 2.25 (n = 7) | 3 (n = 6) | P value | |
| Age, years | 27.83 ± 3.66 | 27.86 ± 3.24 | 28.83 ± 3.55 | N.S. |
| BMI, kg/m2 | 22.90 ± 1.63 | 22.62 ± 1.64 | 22.66 ± 1.54 | N.S. |
| School education, > 12 years | 100% | 100% | 100% | N.S. |
| Blood pressure systolic, mmHG | 109.17 ± 10.69 | 112.14 ± 10.35 | 114.17 ± 4.92 | N.S. |
| Diastolic, mmHG | 78.33 ± 12.91 | 80.00 ± 5.77 | 82.50 ± 6.12 | N.S. |
| STAI trait | 34.00 ± 5.29 | 34.57 ± 5.32 | 33.33 ± 5.68 | N.S. |
Data are mean ± SD. Differences between groups were analyzed by Mann–Whitney U test. No significant differences were detected (all P < 0.05).
BMI, body‐mass‐index; EVR, everolimus; N.S., not significant; STAI, State‐Trait Anxiety Inventory.
Study design
Study participants were assigned into three groups according to the administered EVR dose: 1.5 mg (low), 2.25 mg (medium), and 3 mg (high) EVR per intake. Application of the described EVR doses is based on prescribed target trough levels, the findings derived from patient studies and due to ethical reasons. 6 , 7 , 18 We designed and conducted this explorative pilot study against the background of our research activities on behavioral and neuroendocrine effects of immunosuppressive medication. 28 , 29 , 30 , 31 The probands of the different groups received 4 oral EVR administrations in a 12‐hour cycle over 3 consecutive experimental days (Figure 1 ). The study extended over 15 days. Blood was withdrawn from fasted participants before the first EVR administration on day 1 (D1) at 9:00 am for baseline measurements, on day 3 (D3trough) at 6:30 am right before the last administration and at 9:00 am, 2 hours after the last EVR administration (D3peak) as well as 5 and 12 days later on study days 8 (D8) and 15 (D15) to examine residual effects of the drug. EVR blood concentrations have been quantified on D1 for baseline levels, on D3trough to assess EVR trough blood levels, on D3peak for peak blood concentrations, and on D8 to verify that the drug was completely metabolized in the circulation. Additionally, blood collected on D1, D3, D8, and D15 was subjected to further analyses to determine the impact of EVR on cytokine production, immune cell proliferation, and neuroendocrine hormones. Here, D3 stands for 2 hours after the last oral EVR administration when EVR peak levels were detected (D3peak).
Figure 1.
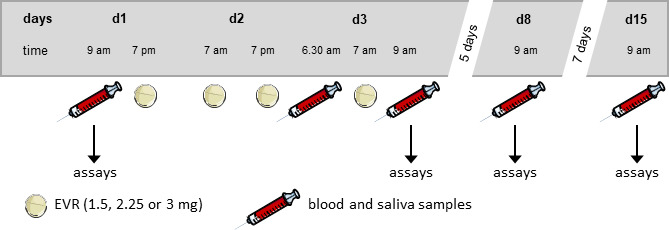
Study design. Three different EVR doses (1.5, 2.25, or 3 mg) were orally administered 4× in a 12‐hour cycle to healthy men. Blood and saliva samples were withdrawn before (day (D)1) and after (D3, D8, and D15) oral administration of EVR and subjected to further analysis. EVR, everolimus.
Drugs
EVR (Certican) tablets were obtained from Novartis Pharmaceuticals (Basel, Switzerland) and orally administered as described in the study design. For in vitro studies, EVR (Cayman Chemicals, Ann Arbor, MI) has been dissolved in dimethylsulfoxide (DMSO; Sigma‐Aldrich, Darmstadt, Germany) to a stock concentration of 10 mM and stored as aliquots at −20°C. EVR stock solution has been further diluted in phosphate‐buffered saline. Cells were treated with a final concentration of 10 nM EVR (DMSO 0.01%) for the indicated time points.
EVR plasma level
EVR plasma concentrations was determined in EDTA anticoagulated whole‐blood using validated liquid chromatography tandem mass spectrometry performed by a laboratory medicine department. The lower limit of quantification for EVR in blood is 1 µg/L.
Isolation of human peripheral blood mononuclear cells
Human peripheral blood mononuclear cells (PBMCs) were isolated from at least six to seven healthy volunteers by Ficoll density‐gradient centrifugation, as previously described, 28 and documented in Supplementary Materials.
Cell culture and treatment
Freshly isolated PBMCs were cultured at defined density ex vivo in growth medium and incubated at 37°C in 5% CO2 for the indicated times. Cultured PBMCs were stimulated with either soluble, monoclonal, murine anti‐human CD3 antibody (50 ng/mL; LEAF Purified anti‐human CD3, Klon HIT3a; BioLegend, San Diego, CA) alone or in combination with murine anti‐human CD28 antibody (50 ng/mL; Ultra LEAF Purified anti‐human CD28, Klon CD28.2; BioLegend). Further, isolated PBMCs from D1 were treated in vitro with 10 nM EVR (DMSO 0.01%; Cayman Chemicals) in presence and absence of anti‐CD3 for the indicated time.
PBMC cytokine secretion
PBMCs (5 × 106 cells/mL) were seeded in 96‐well flat‐bottom tissue culture plates and stimulated with 50 ng/mL of soluble mouse anti‐human CD3 monoclonal antibody for 24 hours (37°C, 5% CO2). Afterward, cells were centrifuged at 450× g for 5 minutes at 4°C and supernatants were collected, transferred to new tubes, and stored in −80°C refrigerator for cytokine analyses. Interleukin (IL)‐2 and IL‐10 levels were assessed in culture supernatants using commercial enzyme‐linked immunosorbent assays (BioLegend), according to the manufacturer’s instructions on a FLUOstar Omega multi‐mode microplate reader (BMG LABTECH GmbH, Ortenberg, Germany). The detection limit is 4 pg/mL and 2 pg/mL, respectively. Intra‐assay and inter‐assay variances were 4.2% and 6.1% for IL‐2 and 4.6% and 6.9% for IL‐10.
EdU proliferation assay of stained PBMCs
Proliferation of CD4+ and CD8+ T cells was determined by flow cytometry using the Click‐iT EdU Pacific Blue Flow Cytometry Assay Kit (Thermo Fisher Scientific, Darmstadt, Germany) according to manufacturer’s instructions and as described in Supplementary Materials .
Endocrine parameters
Plasma noradrenaline (NA) levels were quantified using commercial enzyme‐linked immunosorbent assays (Noradrenaline ELISAFast Track: LDN, Nordhorn, Germany; Cortisol ELISA: IBL International, Hamburg, Germany) with a detection limit of 0.6 ng/mL according to the manufacturer’s protocol. Intra‐assay and inter‐assay variances were 7.4% and 12.5%, respectively. Cortisol was determined in plasma and saliva using commercial ELISA assays (IBL International) according to the manufacturer’s handbook. The detection limit is 0.005 μg/dL and 0.015 µg/dL, respectively. Intra‐assay and inter‐assay variances were 4.8% and 5.9% for cortisol measurements in saliva and 5.6% and 6.9% for cortisol measurements in serum. Storage conditions are described in Supplementary Materials .
Behavioral measures
Sociodemographic data were collected from all participants before the first study day. Trait anxiety was measured before the start of the study with a validated psychological questionnaire (i.e., the trait‐version of the State‐Trait Anxiety Inventory). In the course of the study, heart rate and blood pressure were measured before blood draws (using a blood pressure gauge), and state anxiety was assessed with the state‐version of the State‐Trait Anxiety Inventory. Additionally, at the same time points, the Generic Assessment of Side Effects questionnaire was conducted, assessing psychological and medical indispositions. More details are described in Supplementary Materials .
Statistical analysis
Nonparametric data analyses were performed using PASW statistics (version 22; SPSS, Chicago, IL). Detailed descriptions on statistical analysis are found in Supplementary Materials .
RESULTS
EVR plasma levels during the study days
Circulating EVR levels were obtained in all participants of the low‐dose, medium‐dose, and high‐dose group at baseline (D1), after EVR intake (D3trough as well D3peak levels) and on D8. EVR trough blood levels (P < 0.05) as well as peak levels (P < 0.05) were significantly increased in all three drug concentration groups with significantly more pronounced increases in EVR peak levels in the high‐dose group compared with the low‐dose and medium‐dose groups (P < 0.01 for both groups; Table 2 ). Although EVR trough levels on D3 were significantly increased in the high‐dose group compared with the low‐dose group (P < 0.05), analyses revealed no statistical difference in EVR trough levels between the medium‐dose group compared with low‐dose and high‐dose groups.
Table 2.
Circulating EVR levels during study days
| CEVR, µg/L | EVR dose group, mg | ||
|---|---|---|---|
| 1.5 (n = 6) | 2.25 (n = 7) | 3 (n = 6) | |
| CEVR D1 | < 1 | < 1 | < 1 |
| CEVR D3trough | 2.02 ± 0.72 (35.81%)* | 3.23 ± 1.13 (35.03%)* | 3.75 ± 0.97 (25.73%)* # |
| CEVR D3peak | 5.95 ± 2.04 (34.23%)* | 8.19 ± 3.07 (37.45%)* | 17.67 ± 8.04 (45.52%)* ## §§ |
| CEVR D8 | < 1 | < 1 | < 1 |
Data are mean ± SD (percentage of coefficient of variation). Changes within groups were analyzed by Wilcoxon‐test, differences between groups by Mann–Whitney U test. Significant changes were found within all groups (*P < 0.05 vs. D1), whereas significant differences between groups were detected only between the low‐dose and high‐dose groups for D3trough (# P < 0.05) and D3peak (## P < 0.01) as well as between medium‐dose and high‐dose for D3peak (§§ P < 0.01). D1 = baseline; D3trough = 12 hours after the third dose and right before the fourth dose; D3peak = 2 hours after fourth dose intake; D8 = 5 days after the fourth EVR administration.
CEVR, everolimus concentration; D, days; EVR, everolimus.
Medium‐dose and high‐dose EVR administration suppressed CD4+ and CD8+ T cell proliferation and IL‐10 secretion in healthy men
One of the main objectives of this study was to determine the efficacy of acute orally administration of clinically used EVR doses on inhibition of T cell proliferation and cytokine secretion in healthy men. The fluorescent activated cell sorting analysis revealed a marked significant reduction in CD4+ proliferative T cells on D3 and D8 in the medium‐dose and high‐dose groups (all P < 0.05). However, a significant reduction in CD8+ proliferating T cells on D8 (P < 0.05) but not on D3 was observed in the high‐dose group (Figure 2a ). On D15, proliferation rates of CD4+ and CD8+ T cells in all groups were restored to baseline levels before EVR administration. In contrast, low‐dose administration of EVR (1.5 mg) did not affect CD4+ and CD8+ T cell proliferation during all study days. Differences between groups have only been detected for CD4+ T cells on D8 between the medium‐dose and high‐dose groups (P < 0.05). Similar to the ex vivo conditions but even more pronounced, anti‐proliferative effects were obtained in mitogen‐stimulated drug‐naive PBMCs that were exposed to 10 nM EVR in vitro. Here, EVR significantly suppressed CD4+ and CD8+ T cell proliferation by 90.60% and 79.41% (both P < 0.001) compared with mitogen‐treated cells without EVR (Figure 2b ).
Figure 2.
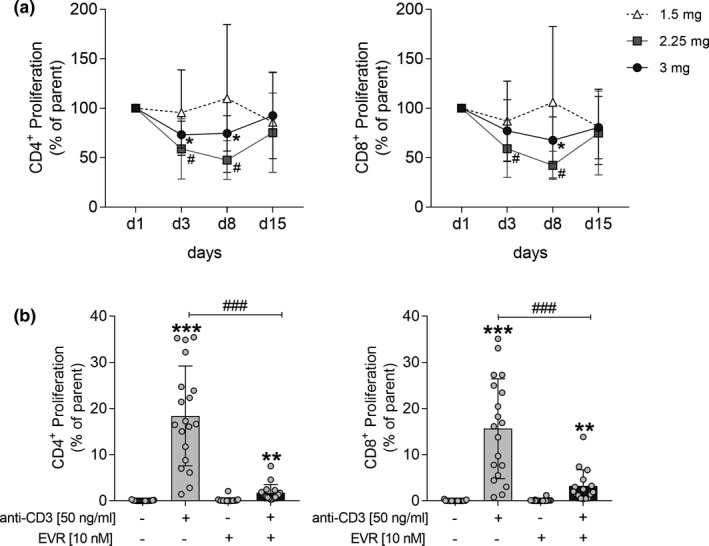
Effect of EVR on T cell proliferation. CD4+ and CD8+ T cell proliferation in peripheral blood mononuclear cells (PBMCs) that were isolated before (day (D)1) and after (D3, D8, and D15) oral administration of EVR in participants of the low‐dose (1.5 mg), medium‐dose (2.25 mg), and high‐dose (3 mg) groups and treated with anti‐CD3 and anti‐CD28 (both 50 ng/mL) for 48 hours (a). CD4+ and CD8 + T cell proliferation in drug‐naive PBMCs isolated on D1 and stimulated with anti‐CD3 (50 ng/mL) in the presence (+) and absence (‐) of EVR (10 nM) for 48 hours (b). Data are represented as mean ± SD. Differences were analyzed by Wilcoxon test. */#/§ P < 0.05 vs. D1 a, **P < 0.01, ***P < 0.001 vs. unstimulated cells, ### P < 0.001 vs. treated cells b. EVR, everolimus.
Further, statistical analysis revealed a significant reduced IL‐10 cytokine secretion from stimulated PBMCs in participants of the low‐dose, medium‐dose, and high‐dose groups on D3 compared with baseline values on D1, respectively (all P < 0.05). Secretion of IL‐10 was already restored to baseline measurements on D8 in PBMC supernatants of the low‐dose and medium‐dose groups, whereas in the high‐dose group it was still significantly reduced on D8 (P < 0.0.5; Figure 3a ). In contrast, T cell enriched PBMCs derived from the high‐dose group released significantly less IL‐2 on D8 in comparison to baseline values on D1 (P < 0.05), whereas low‐doses and medium‐doses did not affect IL‐2 secretion from anti‐CD3 stimulated PBMCs during all study days (Figure 3b ). For IL‐10, no differences between groups were detected, whereas for IL‐2 on D8 a significant difference between the medium‐dose and high‐dose groups was found (P < 0.05). Interestingly, in vitro exposure of the mTOR inhibitor EVR to anti‐CD3 treated but drug‐naive PBMCs significantly decreased IL‐2 as well as IL‐10 production compared with cells stimulated with anti‐CD3 alone (both P < 0.001; Figure 3c,d ).
Figure 3.
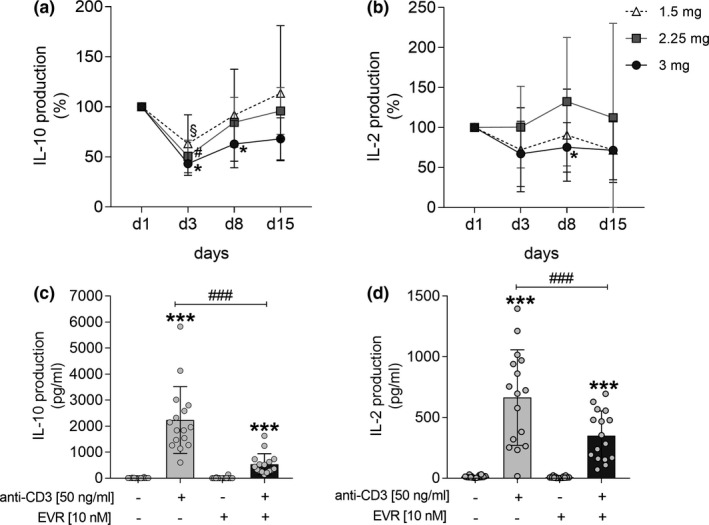
Effect of EVR on cytokine secretion from mitogen‐stimulated PBMCs. IL‐2 and IL‐10 production levels released by PBMCs that were isolated before (day (D)1) and after (D3, D8, and D15) oral administration of EVR in participants of the low‐dose (1.5 mg), medium‐dose (2.25 mg), and high‐dose (3 mg) groups and treated with anti‐CD3 (50 ng/mL) for 24 hours a, b. IL‐2 and IL‐10 cytokine secretion levels from naive PBMCs isolated on D1 and in vitro stimulated with anti‐CD3 (50 ng/mL) in the presence (+) and absence (−) of EVR (10 nM) for 24 hours (c, d). Data are represented as mean ± SD. Differences were analyzed by Wilcoxon test. */# P < 0.05 vs. D1 a, b; **P < 0.01, ***P < 0.001 vs. unstimulated cells, ### P < 0.001 vs. treated cells c, d. EVR, everolimus; PBMC, peripheral blood mononuclear cells.
Acute EVR intake dose‐dependently affected noradrenaline and cortisol levels
The hypothalamic pituitary adrenal (HPA) axis and sympathetic‐adrenomedullary system are stimulated in response to physiological stress resulting in increased glucocorticoid hormones and catecholamines (e.g., cortisol and NA released by the adrenal cortex and glands/medulla). 26 The present findings showed significantly increased systemic NA concentrations 2 hours after last oral administration of the low and medium EVR dose on D3 (both P < 0.05) in comparison to baseline levels on D1 (Figure 4a ). The NA blood levels were still elevated in the medium‐dose group on D8 and significantly reduced in the high‐dose group on D8 and D15 when compared to D1 (all P < 0.05). Furthermore, NA levels of the medium‐dose group were significantly higher than those of low‐dose and high‐dose groups on D8 (P < 0.05). Interestingly, data revealed significantly reduced plasma cortisol levels on D3 and D8 in the high‐dose group (Figure 4b ), which was paralleled by significantly lower cortisol levels analyzed in saliva on D3 (all P < 0.05; Figure 4c ).
Figure 4.
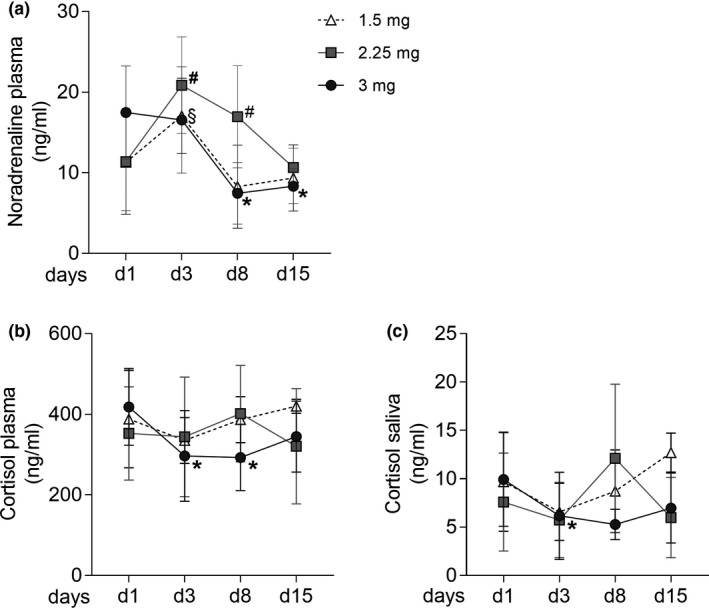
Impact of EVR on systemic levels of neuroendocrine hormones. Noradrenaline plasma level (a) and cortisol concentrations in plasma (b) and saliva (c) before and after oral administration of low‐dose, medium‐dose, and high‐dose EVR in healthy men. Data are expressed as mean ± SD and analyzed by Wilcoxon test. */#/§ P < 0.05 vs. day 1. EVR, everolimus.
Acute EVR administration moderately increased state anxiety levels without affecting subjectively perceived side effects
Subjects of the high‐dose group did not show significant changes in state anxiety levels, whereas participants of the low‐dose and medium‐dose groups showed slightly increased state anxiety levels at D3 (P < 0.05) and in the medium‐dose group also at D15 (P < 0.05). Participants of all three administration groups perceived side effects, which they attributed to the intake of EVR. Nevertheless, the number of participants who did perceive side effects differed between groups. Only one of six in the low‐dose group reported medication attributed side effects, whereas five of seven in the medium‐dose group and three of six in the high‐dose group stated to perceive medication attributed side effects. However, the summed symptom count per measurement time point did not significantly differ between groups. Furthermore, as participants consistently reported only a small number of side effects, none of the groups showed a significant increase of perceived medication‐attributed side effects from baseline (D1) to D3, D8, or D15 (Table 3 ).
Table 3.
State anxiety levels (STAI state) and subjectively perceived side effects (GASE) of the three groups at all time points of measurement
| EVR dose group, mg | |||
|---|---|---|---|
| 1.5 (n = 6) | 2.25 (n = 7) | 3 (n = 6) | |
| STAI state | |||
| D1 | 34.00 ± 5.93 | 32.29 ± 3.35 | 35.00 ± 9.42 |
| D3 | 38.67 ± 7.12* | 36.00 ± 4.00* | 34.67 ± 4.97 |
| D8 | 33.50 ± 5.47 | 33.57 ± 5.38 | 35.40 ± 4.78 |
| D15 | 37.50 ± 9.65 | 35.86 ± 4.88* | 35.00 ± 5.48 |
| GASE | |||
| D1 | 0.00 ± 0.00 | 0.00 ± 0.00 | 0.00 ± 0.00 |
| D3 | 0.17 ± 0.41 | 0.86 ± 0.90 | 0.33 ± 0.52 |
| D8 | 0.83 ± 2.04 | 1.00 ± 1.16 | 0.40 ± 0.55 |
| D15 | 0.67 ± 1.63 | 1.00 ± 1.55 | 1.50 ± 2.51 |
Data are mean ± SD. Changes within groups were analyzed by Wilcoxon‐test, differences between groups by Mann–Whitney U test. Significant changes were found for state anxiety levels within the low‐dose and medium‐dose groups (*P < 0.05 vs. D1), whereas no significant differences between groups were detected. D3 = 2 hours after fourth dose intake.
D, days; EVR, everolimus; GASE, Generic Assessment of Side Effects; STAI, State‐Trait Anxiety Inventory.
DISCUSSION
The mTOR inhibitor EVR is clinically widely used in transplantation medicine to prevent organ rejection and as an anti‐proliferative drug to fight tumor growth. 1 , 2 , 16 However, studies on the pharmacokinetics together with pharmacodynamic data of different doses of EVR in healthy individuals do not exist. Because the knowledge on dose‐dependent effects exerted by EVR under physiological conditions is crucial to better understand the pharmacological action of this drug, we analyzed whether and to what extent three different and clinically relevant doses of EVR affect immunological, psychological, and neuroendocrine parameters in healthy male individuals. We observed that oral intake of medium (2.25 mg) and high doses (3 mg) of EVR efficiently suppressed T cell proliferation as well as IL‐10 cytokine production in ex vivo mitogen‐stimulated PBMCs isolated from healthy volunteers. Although acute low (1.5 mg) and medium (2.25 mg) EVR administration increased state anxiety and systemic NA levels, high (3 mg) dose EVR intake revealed reduced cortisol levels but did not affect perceived state anxiety. Studies in animal models and patients demonstrated the effective immunosuppressive action of EVR mediated by inhibition of lymphocyte proliferation and by preventing graft rejections of solid organ transplantations. 11 , 13 , 14 However, comorbidities and the co‐treatment of EVR with other immunosuppressive drugs, like calcineurin inhibitors and/or steroids, make it hard to discriminate whether the examined effects solely were exerted by the action of EVR. 11 , 17 Therapeutic EVR trough blood levels should be targeted between 3 and 8 ng/mL, which has been shown to associate with reduced biopsy‐proven acute rejection and graft loss. 5 Our data revealed that the mean EVR trough levels of the medium‐dose and high‐dose groups obtained 12 hours after third but right before the last EVR administration were above the therapeutic threshold of 3 ng/mL and, therefore, comparable to therapeutic treatments. 2 , 5 Although EVR has been systemically washed out since study day 8, the observed anti‐proliferative effects even persist 5 days after the last EVR administration in the medium‐dose and high‐dose groups, whereas for IL‐10 suppression this was just the case in the high‐dose group. These findings suggest that higher doses of EVR induce residual immunosuppressive effects even when the drug is completely metabolized. IL‐10 is known as anti‐inflammatory cytokine resulting in the inhibition of pro‐inflammatory factors. Thus, it plays an essential role in the prevention of inflammatory and autoimmune pathologies. 32 However, some studies ascribed IL‐10 also pro‐inflammatory properties that have been associated with increased mitogen‐driven T cell proliferation and the induction of chemotaxis of cytotoxic T cells. 33 , 34 , 35 Our data are consistent with findings of Böhler et al. showing an efficient suppression of T cell derived IL‐10 after EVR treatment in stable renal‐allograft recipients. 11 As shown in previous studies, 12 , 36 we further examined a marked inhibition of proliferative T cells and cytokine production that were directly exposed to EVR in vitro confirming our ex vivo observations. However, in contrast to previously reported findings, 11 the in vitro effects were even more pronounced compared with ex vivo conditions after an acute oral EVR administration in men although EVR concentrations were in a similar range. Thus, although orally administered EVR did not directly affect IL‐2 secretion, in vitro exposure of the drug significantly reduced the production of this pro‐inflammatory cytokine. Thus, we provide further proof for usage of T cell proliferation and IL‐10 production as efficient and valid ex vivo parameters for a potent immunosuppressive action of the drug in future research.
Although impaired mTOR signaling has been implicated in neurocognitive dysfunctions and neurodegenerative processes, 24 , 37 , 38 it remains uncertain whether and to what extent clinically used doses of the mTOR inhibitor EVR affect neurocognitive and psychological parameters in healthy humans. Our data revealed that an acute EVR administration with low and medium doses induced an increase in state anxiety levels, whereas intake of the high doses did not affect state anxiety. These findings indicate that the induced changes in state anxiety apparently depend on the administered EVR dosage. This notion is supported by studies in rats showing that acute one‐time and repeated rapamycin treatment with low doses (1 mg/kg) and high doses (3 mg/kg) resulted in anxiety‐related behavior probably induced by mTOR‐independent mechanisms leading to hyperexcitability of the amygdala. 26 , 27 Moreover, anxiety (e.g., following a drug treatment) has been associated with the perception of drug‐induced side effects. 39 , 40 The fact that participants hardly reported medication‐attributed side effects, although a significant increase of state anxiety was observed in the low‐dose and medium‐dose groups, indicates that acute intake of therapeutic EVR doses indeed does not induce subjectively perceived side effects.
Anxiety, stress, and depression are directly linked to the activation of the sympathetic nervous system reflected by elevated release of NA and specific brain regions that, in turn, stimulates the HPA axis and the release of cortisol. 41 , 42 , 43 Because we observed increased NA levels along with not affected cortisol levels after acute EVR intake in the low‐dose and medium‐dose groups, our data indicate that both EVR doses might lead to an activation of the sympathetic adrenomedullary system probably not accompanied by changes in HPA activity. In contrast, high‐dose EVR treatment reduced plasma and saliva cortisol without directly affecting NA level and state anxiety. Whether the reduction in NA levels on D8 and D15 in the high‐dose group was directly induced by EVR or rather was mediated by secondary factors (e.g., drug withdrawal (rebound effect)) has to be validated in further experiments. Although plasma NA concentration is a useful parameter to assess sympathetic nerve activity, 44 more functional analyses (i.e., pupillometry, heart rate variability, and/or salivary alpha‐amylase activity), 22 , 45 , 46 might provide more details on the effect of the different EVR doses on sympathetic nerve activity in future research. We observed dose‐dependent ex vivo effects of EVR on cytokine production on D3, however, this was not the case for the assessed cortisol and NA levels. These findings indicate specific thresholds for EVR blood concentration that associate with changes in cortisol levels in saliva and plasma as well as NA plasma levels compared with baseline values assessed before drug intake.
Knowledge on the dose‐dependent effects of EVR as well as the cellular immunosuppressive actions will form the basis for future applications of EVR in research protocols, including immune conditioning paradigms. Taste immune conditioning protocols have been demonstrated as effective approaches in animals, healthy humans, and patients aiming to enhance therapeutic potency of the used immunosuppressive compound, such as cyclosporin A, while reducing drug doses along with its associated side effects. 28 , 30 , 31 However, the knowledge whether these conditioning protocols also work with the mTOR inhibitor EVR is highly limited and the subject of future research.
Although outside the scope of the present study, a limitation is that we could not detail the mechanisms via which EVR exerted the examined effects. The rapamycin analogue EVR has been developed as a potent and selective mTOR inhibitor. Thus, mTOR‐specific substrates, like the eIF‐4E‐binding protein (4E‐BP1) and S6 kinase 1 (p70S6K1), are blocked leading to G1 cell cycle arrest and inhibition of cell proliferation. 47 In vitro and ex vivo studies demonstrated that EVR impairs cell proliferation in a dose‐dependent and time‐dependent manner and might reduce cellular adrenocortical cortisol secretion via inhibition of the mTOR signaling pathway. 48 , 49 , 50 These findings suggest that our observed effects potentially were mediated via EVR induced mTOR inhibition rather than by off target or secondary mechanisms. Although the effects presented in our study were examined upon an acute four times administration in healthy individuals, whereas patients usually were treated in the long‐term or even lifelong, our findings might provide important insights in the action of EVR on neuroendocrine and psychological parameters also under pathological conditions.
In conclusion, the present results provide evidence that an acute oral exposure of EVR dose‐dependently mediates a potent inhibition of T cell proliferation, IL‐10 suppression, altered systemic levels of neuroendocrine hormones (i.e., cortisol and NA), and changes in state anxiety in healthy probands. Furthermore, independent of the dosage, EVR seems to induce subjectively perceived side effects only in a minimal extent. Drawbacks of this explorative pilot study are the limited number of study participants and that it was conducted in men only. Taken together, our data not only verify the application of T cell proliferation and IL‐10 secretion as valid parameters for an efficient immunosuppression mediated by the mTOR inhibitor EVR in human studies but rather provide indications for an impact of the drug on behavior.
Author Contributions
T.H., A.L.K., and M.S. wrote the manuscript. M.S., O.W., and T.H. designed the research. T.H., B.W., A.L.K., F.K., and S.H. performed the research. T.H., A.L.K., and M.S. analyzed the data. T.H., B.W., A.L.K., F.K., and S.H. Contributed new reagents/analytical tools.
Conflicts of Interest
O.W. has received research grants for clinical studies, speaker’s fees, honoraria, and travel expenses from Amgen, Astellas, Bristol‐Myers Squibb, Chiesi, Hexal, Janssen‐Cilag, MSD, Novartis, Roche, Pfizer, and Sanofi. All other authors declared no competing interests for this work.
Funding
O.W. is supported by an unrestricted grant of the Rudolf‐Ackermann‐Stiftung (Stiftung für Klinische Infektiologie / Foundatiopn for Clinical Infectiology). This work was supported by grants from the German Research Foundation (FOR 1328; SCHE 341/17‐1; and SCHE 341/17‐2).
Supporting information
Supplementary Material
Acknowledgments
The authors thank Alexandra Kornowski and Dr. Bastian Tebbe for their technical and medical assistance.
References
- 1. Saran, U. , Foti, M. & Dufour, J.‐F. Cellular and molecular effects of the mTOR inhibitor everolimus. Clin. Sci. 129, 895–914 (2015). [DOI] [PubMed] [Google Scholar]
- 2. Sánchez‐Fructuoso, A.I. Everolimus: an update on the mechanism of action, pharmacokinetics and recent clinical trials. Expert Opin. Drug Metab. Toxicol. 4, 807–819 (2008). [DOI] [PubMed] [Google Scholar]
- 3. Schmelzle, T. & Hall, M.N. TOR, a central controller of cell growth. Cell 103, 253–262 (2000). [DOI] [PubMed] [Google Scholar]
- 4. Saemann, M.D. , Haidinger, M. , Hecking, M. , Horl, W.H. & Weichhart, T. The multifunctional role of mTOR in innate immunity: implications for transplant immunity. Am. J. Transplant. 9, 2655–2661 (2009). [DOI] [PubMed] [Google Scholar]
- 5. Salvadori, M. & Bertoni, E. Long‐term outcome of everolimus treatment in transplant patients. Transpl. Res. Risk Manag. 3, 77–90 (2011). [Google Scholar]
- 6. Novartis . Certican tablets: summary of product characteristics. <https://klinischeforschung.novartis.de/dokumente/fachinformationen‐certican‐tabletten/>. Accessed April 15, 2020.
- 7. Shipkova, M. et al. Therapeutic drug monitoring of everolimus: A Consensus report. Ther. Drug Monit. 38, 143–169 (2016). [DOI] [PubMed] [Google Scholar]
- 8. Kovarik, J.M. et al. Effect of food on everolimus absorption: quantification in healthy subjects and a confirmatory screening in patients with renal transplants. Pharmacotherapy 22, 154–159 (2002). [DOI] [PubMed] [Google Scholar]
- 9. Kovarik, J.M. , Hartmann, S. , Figueiredo, J. , Rouilly, M. , Port, A. & Rordorf, C. Effect of rifampin on apparent clearance of everolimus. Ann. Pharmacother. 36, 981–985 (2002). [DOI] [PubMed] [Google Scholar]
- 10. Thudium, K. , Gallo, J. , Bouillaud, E. , Sachs, C. , Eddy, S. & Cheung, W. Bioavailability of everolimus administered as a single 5 mg tablet versus five 1 mg tablets: a randomized, open‐label, two‐way crossover study of healthy volunteers. Clin. Pharmacol. 7, 11–17 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bohler, T. et al. Pharmacodynamic effects of everolimus on anti‐CD3 antibody‐stimulated T‐lymphocyte proliferation and interleukin‐10 synthesis in stable kidney‐transplant patients. Cytokine 42, 306–311 (2008). [DOI] [PubMed] [Google Scholar]
- 12. Levitsky, J. , Miller, J. , Huang, X. , Gallon, L. , Leventhal, J.R. & Mathew, J.M. Immunoregulatory effects of everolimus on in vitro alloimmune responses. PLoS One 11, e0156535 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Schuurman, H.J. et al. SDZ RAD, a new rapamycin derivative: synergism with cyclosporine. Transplantation 64, 32–35 (1997). [DOI] [PubMed] [Google Scholar]
- 14. Schuler, W. et al. SDZ RAD, a new rapamycin derivative: pharmacological properties in vitro and in vivo. Transplantation 64, 36–42 (1997). [DOI] [PubMed] [Google Scholar]
- 15. Salvadori, M. et al. Everolimus with very low‐exposure cyclosporine a in de novo kidney transplantation: a multicenter, randomized, controlled trial. Transplantation 88, 1194–1202 (2009). [DOI] [PubMed] [Google Scholar]
- 16. Pascual, J. et al. Everolimus with reduced calcineurin inhibitor exposure in renal transplantation. J. Am. Soc. Nephrol. 29, 1979–1991 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Dutt, S.B. et al. mTOR inhibition by everolimus does not impair closure of punch biopsy wounds in renal transplant patients. Transplant. Direct 3, e147 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Budde, K. et al. Tolerability and steady‐state pharmacokinetics of everolimus in maintenance renal transplant patients. Nephrol. Dial. Transplant. 19, 2606–2614 (2004). [DOI] [PubMed] [Google Scholar]
- 19. Lang, U.E. , Heger, J. , Willbring, M. , Domula, M. , Matschke, K. & Tugtekin, S.M. Immunosuppression using the mammalian target of rapamycin (mTOR) inhibitor everolimus: pilot study shows significant cognitive and affective improvement. Transpl. Proc. 41, 4285–4288 (2009). [DOI] [PubMed] [Google Scholar]
- 20. Kahan, B. , Flechner, S. , Lorber, M. , Golden, D. , Conley, S. & Van, C.B. Complications of cyclosporine‐prednisone immunosuppression in 402 renal allograft recipients exclusively followed at a single center for from one to five years. Transplantation 43, 197–204 (1987). [DOI] [PubMed] [Google Scholar]
- 21. Bechstein, W.O. Neurotoxicity of calcineurin inhibitors: impact and clinical management. Transpl. Int. 13, 313–326 (2000). [DOI] [PubMed] [Google Scholar]
- 22. Bosch, J.A. , Veerman, E.C. , de Geus, E.J. & Proctor, G.B. Alpha‐amylase as a reliable and convenient measure of sympathetic activity: don't start salivating just yet! Psychoneuroendocrinology 36, 449–453 (2011). [DOI] [PubMed] [Google Scholar]
- 23. Ishii, R. , Wataya‐Kaneda, M. , Canuet, L. , Nonomura, N. , Nakai, Y. & Takeda, M. Everolimus improves behavioral deficits in a patient with autism associated with tuberous sclerosis: a case report. Neuropsychiat. Electrophysiol. 1, 6 (2015). [Google Scholar]
- 24. Russo, E. et al. Everolimus improves memory and learning while worsening depressive‐and anxiety‐like behavior in an animal model of depression. J. Psychiatr. Res. 78, 1–10 (2016). [DOI] [PubMed] [Google Scholar]
- 25. Raymond, E. et al. Safety and pharmacokinetics of escalated doses of weekly intravenous infusion of CCI‐779, a novel mTOR inhibitor, in patients with cancer. J. Clin. Oncol. 22, 2336–2347 (2004). [DOI] [PubMed] [Google Scholar]
- 26. Hadamitzky, M. et al. Acute systemic rapamycin induces neurobehavioral alterations in rats. Behav. Brain Res. 273, 16–22 (2014). [DOI] [PubMed] [Google Scholar]
- 27. Hadamitzky, M. et al. Repeated systemic treatment with rapamycin affects behavior and amygdala protein expression in Rats. Int. J. Neuropsychopharmacol. 21, 592–602 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Albring, A. et al. Preserving learned immunosuppressive placebo response: perspectives for clinical application. Clin. Pharmacol. Ther. 96, 247–255 (2014). [DOI] [PubMed] [Google Scholar]
- 29. Hadamitzky, M. , Luckemann, L. , Pacheco‐Lopez, G. & Schedlowski, M. Pavlovian conditioning of immunological and neuroendocrine functions. Physiolog. Rev. 100, 357–405 (2020). [DOI] [PubMed] [Google Scholar]
- 30. Wirth, T. et al. Repeated recall of learned immunosuppression: evidence from rats and men. Brain Behav. Immun. 25, 1444–1451 (2011). [DOI] [PubMed] [Google Scholar]
- 31. Kirchhof, J. et al. Learned immunosuppressive placebo responses in renal transplant patients. Proc. Natl. Acad. Sci. USA 115, 4223–4227 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Saraiva, M. & O'Garra, A. The regulation of IL‐10 production by immune cells. Nat. Rev. Immunol. 10, 170 (2010). [DOI] [PubMed] [Google Scholar]
- 33. Cohen, S. , Parry, S.L. , Feldmann, M. & Foxwell, B. Autocrine and paracrine regulation of human T cell IL‐10 production. J. Immunol. 158, 5596–5602 (1997). [PubMed] [Google Scholar]
- 34. MacNeil, I.A. , Suda, T. , Moore, K. , Mosmann, T. & Zlotnik, A. IL‐10, a novel growth cofactor for mature and immature T cells. J. Immunol. 145, 4167–4173 (1990). [PubMed] [Google Scholar]
- 35. Jinquan, T. , Larsen, C.G. , Gesser, B. , Matsushima, K. & Thestrup‐Pedersen, K. Human IL‐10 is a chemoattractant for CD8+ T lymphocytes and an inhibitor of IL‐8‐induced CD4+ T lymphocyte migration. J. Immunol. 151, 4545–4551 (1993). [PubMed] [Google Scholar]
- 36. Iwasaki, K. et al. Suppressive effect of everolimus on IL‐2, IL‐10, IL‐21, and IFNgamma levels: implications for the successful minimization of calcineurin inhibitor use in transplantation. Ther. Drug Monit. 41, 371–375 (2019). [DOI] [PubMed] [Google Scholar]
- 37. Wong, M. Mammalian target of rapamycin (mTOR) pathways in neurological diseases. Biomed. J. 36, 40 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cambiaghi, M. et al. Behavioural and EEG effects of chronic rapamycin treatment in a mouse model of tuberous sclerosis complex. Neuropharmacology 67, 1–7 (2013). [DOI] [PubMed] [Google Scholar]
- 39. Benson, S. & Elsenbruch, S. No reason to feel sick? Nocebo responses in the placebo arms of experimental endotoxemia studies. Front. Psychiat. 10, 511 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Barsky, A.J. , Saintfort, R. , Rogers, M.P. & Borus, J.F. Nonspecific medication side effects and the nocebo phenomenon. JAMA 287, 622–627 (2002). [DOI] [PubMed] [Google Scholar]
- 41. Goekoop, J.G. , de Winter, R.F. , Wolterbeek, R. , Van Kempen, G.M. & Wiegant, V.M. Increased plasma norepinephrine concentration in psychotic depression. Therap. Adv. Psychopharmacol. 2, 51–63 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Montoya, A. , Bruins, R. , Katzman, M.A. & Blier, P. The noradrenergic paradox: implications in the management of depression and anxiety. Neuropsychiat. Dis. Treat. 12, 541 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Lenze, E.J. et al. Elevated cortisol in older adults with generalized anxiety disorder is reduced by treatment: a placebo‐controlled evaluation of escitalopram. Am. J. Geriat. Psychiat. 19, 482–490 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Grassi, G. & Esler, M. How to assess sympathetic activity in humans. J. Hypertens. 17, 719–734 (1999). [DOI] [PubMed] [Google Scholar]
- 45. Larsen, R.S. & Waters, J. Neuromodulatory correlates of pupil dilation. Front. Neural. Circuits 12, 21 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Hayano, J. & Yuda, E. Pitfalls of assessment of autonomic function by heart rate variability. J. Physiol. Anthropol. 38, 3 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. O'Reilly, T. & McSheehy, P.M. Biomarker development for the clinical activity of the mTOR inhibitor everolimus (RAD001): processes, limitations, and further proposals. Translat. Oncol. 3, 65 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Gorshtein, A. et al. Mammalian target of rapamycin inhibitors rapamycin and RAD001 (everolimus) induce anti‐proliferative effects in GH‐secreting pituitary tumor cells in vitro. Endoc. Rel. Cancer 16, 1017–1027 (2009). [DOI] [PubMed] [Google Scholar]
- 49. Sukumari‐Ramesh, S. , Singh, N. , Dhandapani, K.M. & Vender, J.R. mTOR inhibition reduces cellular proliferation and sensitizes pituitary adenoma cells to ionizing radiation. Surg Neurol. Int. 2, 22 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. De Martino, M.C. et al. The role of mTOR inhibitors in the inhibition of growth and cortisol secretion in human adrenocortical carcinoma cells. Endocr. Related Cancer 19, 351 (2012). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material


