Abstract
Cerebral ischemia-reperfusion injury (CIRI), caused by the reperfusion of blocked vessels following ischemic stroke, can lead to secondary brain injury. Throughout CIRI, apoptosis serves an important role. Astragaloside IV is a potential neuroprotectant that alleviates CIRI by inhibiting apoptosis. The calcium-sensing receptor (CaSR) is a G-protein-coupled receptor, the activation of which aggravates ischemia-reperfusion injury. The aim of the present study was to investigate whether the protective effect of Astragaloside IV on CIRI may be associated with the regulation of CaSR. A rat middle cerebral artery occlusion/reperfusion (MCAO/R) model and an oxygen and glucose deprivation/reoxygenation (OGD/R) model of pheochromocytoma (PC12) cells were used to study the neuronal injury induced by CIRI. Neurological function scores (NFS), 2,3,5-triphe-nylterazolium chloride and hematoxylin and eosin staining were used to determine brain damage in rats. Cell viability was measured to evaluate the injury of OGD/R PC12 cells. Western blotting was used to examine the expression of proteins associated with apoptosis and CaSR. The CaSR antagonist NPS-2143 and agonist GdCl3 were used to further confirm the effects of CaSR during the process of apoptosis. The results demonstrated that Astragaloside IV alleviated CIRI by decreasing the NFS of rats, reducing the infarction volume of the brain and promoting the viability of PC12 cells, as well as inhibiting the expression of cleaved caspase-3 and CaSR, which was induced by CIRI. The results of the present study suggested that the activation of CaSR may be involved in CIRI-induced brain damage in rats, and that Astragaloside IV may alleviate CIRI by inhibiting CaSR activation-induced apoptosis.
Keywords: Astragaloside IV, cerebral ischemia-reperfusion injury, calcium-sensing receptor, apoptosis
Introduction
Acute ischemic stroke (AIS) is the most frequent cause of permanent disability in adults worldwide (1). Due to its high incidence, disability and recurrence rate, as well as complications, AIS has become a severe threat to human health and quality of life (2). The most effective treatment for AIS is intravascular recanalization within a certain time window (3-5). Recanalization following AIS is likely to cause cerebral ischemia-reperfusion injury (CIRI), during which excess free radicals attack cells in the tissues that regain blood supply (6,7). CIRI leads to neuronal cell necrosis and apoptosis through a series of pathological processes, such as energy metabolism disorders, excitotoxicity, neuroinflammation, oxidative damage and calcium overload (8). Early treatment with neuroprotective drugs can reduce apoptosis and the release of superoxide free radicals (9).
Apoptosis is one of the most important mechanisms of CIRI. In the core of the ischemic region, the severe death of neurons occurs rapidly, although neurons in the ischemic penumbra can be saved with timely and effective treatment (9,10). Previous studies have confirmed that the number of apoptotic neurons in the ischemic penumbra determines the size of the infarction volume and the severity of the neurological defects (11-13). Neuronal apoptosis is crucial to the treatment and prognosis of CIRI (14,15); the primary task of CIRI prevention and treatment is to inhibit neuronal apoptosis.
Calcium overload is an important link between apoptosis and neuronal necrosis (16,17). A number of studies have demonstrated that extracellular calcium can be absorbed and transported into the mitochondria during ischemia-reperfusion (I/R) injury, leading to increased mitochondrial membrane permeability and promoting oxidative reaction and apoptosis (18-20). Calcium-sensing receptor (CaSR) is a member of the G-protein coupled receptor superfamily and is sensitive to the extracellular application of Ca2+, Gd3+ and other polycationic agonists (21), as well as antagonists such as NPS-2390 and NPS-2143 (22-24). CaSR maintains calcium homeostasis, and its activity serves an important role in increased intracellular calcium levels during I/R injury (17,25,26). Previous studies have reported that the activation of CaSR aggravates myocardial and renal I/R injury and mouse CIRI (19,23,27,28). However, it remains unclear whether increased CaSR expression may induce apoptosis by increasing intracellular calcium overload during CIRI, and whether Astragaloside IV may reduce CaSR expression to alleviate intracellular calcium overload and further inhibit CIRI-induced apoptosis.
Traditional Chinese medicines, such as Buyang Huanwu decoction, have been widely used in the treatment of AIS (29,30). Astragaloside IV (C14H68O14) is one of the main active chemicals and the quality-control marker of the traditional Chinese medicine Astragalus membranaceus, which is the main ingredient of Buyang Huanwu decoction, as recorded in the book 'Correction on the Errors of Medical Works' written by Qingren Wang in the Qing Dynasty (31,32). Numerous studies have demonstrated that Astragaloside IV has anti-inflammatory and antiapoptotic properties, is involved in the regulation of vascular remodeling and energy metabolism, and exerts a neuroprotective effect on CIRI (16,18,27,33-37). Our previous study has demonstrated that Astragaloside IV alleviates CIRI by inhibiting neuronal apoptosis and confirmed that it exerts its role by regulating autophagy (9). In addition, Astragaloside IV has been reported to reduce myocardial injury induced by anoxia/reoxygenation through the inhibition of intracellular calcium overload (27).
Based on the above information, we hypothesized that Astragaloside IV may serve a neuroprotective role by inhibiting apoptosis via the reduction of the expression of CaSR.
Materials and methods
Chemicals and reagents
Dulbecco's modified Eagle's medium (DMEM) and 0.25% Trypsin (without EDTA) were obtained from Thermo Fisher Scientific, Inc. Fetal bovine serum (FBS) was purchased from Zhejiang Tianhang Biotechnology Co., Ltd. Earle's balanced salt solution (1X EBSS without calcium, magnesium or sugar) was obtained from Lea Gene Biotechnology Co., Ltd. Astragaloside IV (purity >98%) was purchased from Shanghai Yuan Ye Bio-Technology Co. Ltd. NPS-2143 was provided by MedChemExpress. GdCl3 (purity >99.99%) was purchased from Shanghai Aladdin Biotechnology Co., Ltd. Cell Counting Kit-8 (CCK-8) kit was obtained from Beijing Zoman Biotechnology Co., Ltd. A calcium assay kit (cat. no. C004-2-1) was obtained from the Nanjing Jiancheng Bioengineering Institute. An Annexin V-FITC apoptosis detection kit was obtained from Vazyme Biotech Co., Ltd. A total protein extraction kit (cat. no. BC3640), BCA protein quantitative kit (cat. no. PC0020) and Antifading Mounting Medium (with DAPI; cat. no. S2110) were purchased from Beijing Solarbio Science & Technology Co., Ltd. Antibodies against B-cell lymphoma 2 (Bcl-2; cat. no. ab201566), cleaved caspase-3 (cat. no. ab49822), CaSR (cat. no. ab19347), β-actin (cat. no. ab8224), GAPDH (cat. no. ab9485), apoptosis-inducing factor (AIF; cat. no. ab2086) and neuronal nuclei (NeuN; cat. no. ab190195) were purchased from Abcam. Antibody against Bcl-2-associated X protein (Bax; cat. no. GB1107) and 2,3,5-triphenyltetrazolium chloride (TTC) dye solution were obtained from Wuhan Servicebio Technology Co., Ltd. Horseradish peroxidase (HRP)-conjugated goat anti-mouse/rabbit immunoglobulin G (IgG) antibodies (cat. nos. bs-0296G-HRP and bs-0295G-HRP) were purchased from Beijing Biosynthesis Biotechnology Co., Ltd. Alexa Fluor 488-conjugated goat anti-mouse (cat. no. GB25303) and Cy3-conjugated donkey anti-rabbit (cat. no. GB21403) IgG antibodies were obtained from Wuhan Servicebio Technology Co., Ltd.
Middle cerebral artery (MCA) occlusion/reperfusion (MACO/R) model
A total of 40 adult Sprague Dawley rats (male; age, 7-8 weeks; weight, 270±20 g) were provided by Beijing Vital River Laboratory Animal Technology Co., Ltd. (permit no. SCXK; 2016-0006). All experimental protocols and animal handling procedures were performed in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals 1996. The experimental protocols were approved by the Committee of Experimental Animals of the Hebei University of Chinese Medicine (Shijiazhuang, China; approval no. DWLL2018032).
The animals were maintained at 23±1°C with 50±10% relative humidity in the Experimental Animal Center for 1 week. The animals were allowed free access to food and water in a 12-h light/dark cycle and randomly divided into four groups: i) Sham; ii) middle cerebral artery occlusion/reperfusion (MCAO/R); iii) MCAO/R + Astragaloside IV (AST-IV); and iv) MCAO/R + CaSR antagonist (NPS-2143). All rats under-went MCAO/R with the exception of the Sham group. Once the MCAO/R rat model was established, the rats were assigned into groups randomly.
The rats were fasted from food for 12 h and water for 4 h and anesthetized by intraperitoneal injection with 2% pentobarbital sodium (40 mg/kg bodyweight) before surgery. The anesthetized rats were placed in a supine position on the operating table and shaved with sterilization around the incision site. Cerebral ischemia/reperfusion was induced by transient occlusion of the left MCA with a nylon monofilament for 2 h and reperfusion for 24 h as previously described (38). Briefly, a midline incision was made in the neck. The left common carotid artery (CCA), left external carotid artery (ECA) and left internal carotid artery (ICA) were isolated and tied at the origin of the ECA and at the distal end of the ECA. The left CCA and ICA were temporarily occluded. Subsequently, a nylon suture was introduced into the ECA and pushed up the ICA until resistance was felt. The filament was inserted 18.5±0.5 mm from the carotid bifurcation, effectively blocking the MCA. After 2 h, the suture was removed, and the ECA was permanently tied. In the Sham group, the arteries were exposed and isolated, but not disturbed. Throughout the experiment, the rats were fixed on a small multi-functional animal laboratory bench that detected the anal temperature of the rats and maintained the temperature at a minimum of 37°C. Based on our previous study, rats in the AST-IV group were intraperitoneally injected with 20 mg/kg Astragaloside IV during reperfusion (9). Rats in the NPS-2143 group were intraperitoneally injected with 10 µmol/kg NPS-2143 during reperfusion (23,24,28), and rats in the Sham group received an equal volume of physiological saline. Room temperature was maintained at 28-32°C throughout the surgery. A total of 36 rats entered further experiments, with 9 rats per group; each group was divided into three sub-groups for further experiments, including TTC, hematoxylin and eosin (H&E) or double immunofluorescence staining and western blotting.
Neurological scoring
Neurological scores were evaluated at 24 h post-reperfusion by a blinded observer using the Zea Longa 5-point scoring system to evaluate the sensorimotor deficits: 0 points, normal performance with no neurological deficits; 1 point, contralateral forepaws could not fully extend; 2 points, circling to the opposite side when walking; 3 points, falling to the opposite side when walking; 4 points, no spontaneous walking and loss of consciousness (39). MCAO/R-treated rats that scored 1-3 points were selected as the experimental subjects. According to the neurological score, three rats were removed: One in the MCAO/R group (dead), one in AST-IV group (scored 4), and one in NPS-2143 group (scored 4). To ensure an equal number of animals in each group, one rat was randomly removed from the Sham group. Finally, 36 rats remained with 9 rats/group. These rats were anesthetized by an intraperitoneal injection of pentobarbital sodium (150 mg/kg bodyweight). After deep anesthesia was confirmed by the lack of response to stimuli such as the toe and tail pinching, the rats were sacrificed by cervical dislocation, and the brain tissues were rapidly removed for experiments.
Measurement of infarct volume
Brains (n=3 per group) were removed at 24 h post-reperfusion and gently frozen at −20°C for 15 min to keep the morphology intact during slicing. Briefly, the brains were sliced into five serial 2-mm coronal sections, incubated in a 2% TTC dye solution for 15 min at 37°C in the dark and transferred to 4% paraformaldehyde in phosphate buffer for fixation at 4°C overnight. The extent of ischemic infarction was traced and the integrated volume was calculated manually by an analyst who was blind to the experimental conditions using Image-Pro Plus 6.0 image analysis software (Media Cybernetics, Inc.). The relative infarction volume was expressed as a percentage of the corrected infarct volume in the whole brain volume as follows: Infarct volume (%)=total infarct volume/total brain volume ×100%.
H&E staining
At 24 h post-reperfusion, the rats (n=3 per group) were sacrificed, and the intact brain tissue was carefully removed and placed in a buffer (cat. no. G1101; Wuhan Servicebio Technology Co., Ltd.) containing 4% formaldehyde. Following fixation, dehydration and clearing, the samples were embedded in paraffin and cut into 5-µm serial coronal sections, mounted on slides and used for H&E staining. The sections were stained with H&E according to the standard protocol (40,41) to observe the morphological changes of injured neurons in the cerebral cortex and the hippocampus following MCAO/R surgery. Images of stained slides were acquired using an Olympus DP72 optical microscope (Olympus Corporation). To quantify the data of brain injury, three randomly selected high-magnification (×400) fields per slide were selected in both the cortex and the hippocampus, and the mean value of each slide was calculated for image analysis.
Double immunofluorescence staining
At 24 h post-reperfusion, the rats (n=3 per group) were sacrificed, and 5-µm serial coronal sections were prepared as aforementioned. The sections were dewaxed with xylene for 15 min, twice, hydrated in a decreasing series of ethanol (100, 100, 85 and 75%, followed by distilled water) at room temperature and treated with an Antigen retrieval solution (Wuhan Servicebio Technology Co., Ltd.) for 10 min at 100°C. Following gentle cooling, the sections were sealed with 3% BSA (cat. no. G5001; Wuhan Servicebio Technology Co., Ltd.) at room temperature. For double immunofluorescence histochemistry, the sections were incubated at 4°C overnight with a mixture of anti-CaSR (1:80) and anti-NeuN (1:50) antibodies. Antibody staining was visualized using Alexa Fluor 488-conjugated goat anti-mouse (1:100) and Cy3-conjugated donkey anti-rabbit secondary antibodies (1:100) for 1 h at room temperature in the dark. Counterstaining of cell nuclei was performed by mounting the sections with Antifading Mounting Medium (with DAPI). Images of the stained slides were captured under a confocal microscope. Three high-magnification (×400) fields were randomly selected for image analysis.
Cell culture
Highly differentiated PC12 pheochromocytoma cells were gifted by Professor Shun-Jiang Xu (The First Hospital of Hebei Medical University, Shijiazhuang, China) (originally purchased from Procell Life Science & Technology Co., Ltd.; cat. no. CL-0481; cell vitality, >95%; cell density, >75%). PC12 cells were cultured in complete culture medium (DMEM supplemented with 10% FBS) at 37°C in a humidified atmosphere containing 5% CO2; the medium was changed every 2 days (42,43). The cultured PC12 cells were used to establish an in vitro model of CIRI.
In vitro model of oxygen-glucose deprivation/reoxygenation (OGD/R)
The OGD/R model has been commonly recognized as the in vitro model simulating CIRI (44-46). First, the complete culture medium was discarded, and the cells were washed twice with 1X PBS. PC12 cells were then added to EBSS to simulate the ischemic state and placed into the tri-gas incubator, which was set to maintain the inside environment at 1% O2, 94% N2 and 5% CO2 at 37°C for 2 h. Subsequently, PC12 cells were washed twice with 1X PBS, and the complete culture medium was added. Finally, PC12 cells were cultured in normal conditions for reoxygenation for 24 h. During the reoxygenation period, 100 µmol/l Astragaloside IV, 25 µmol/l CaSR antagonist NPS-2143 or 300 µmol/l CaSR agonist GdCl3 was added to the PC12 cells.
CCK-8 assay
CCK-8 assay was used to detect the viability of PC12 cells. Cells from one bottle were inoculated into 96-well plates at a density of 3-5×104 cells/well for 24 h and exposed to OGD/R. At 24 h post-reoxygenation, 10 µl/well CCK-8 reagent was added to the cells, followed by 1-h incubation at 37°C. The optical density (OD) was measured using a Varioskan™ LUX multi-function microplate reader (Thermo Fisher Scientific, Inc.) at 450 nm, and cell viability was calculated as follows: Cell viability=(OD value of experimental group-OD value of blank group)/(OD value of control group-OD value of blank group) ×100%. Each group was tested four times in parallel for statistical analysis.
Flow cytometry
PC12 cells were dissociated by 0.25% trypsin (without EDTA) and washed twice with 1X PBS at 4°C. The cells were then centrifuged at 300 × g at 4°C for 5 min and resuspended in 100 µl 1X binding buffer. Subsequently, 5 µl FITC and PI were added to the cells and mixed gently avoiding light for 15 min at room temperature. Finally, 400 µl 1X binding buffer was added to the cells, and the apoptotic rate was measured by an FC 500 MCL flow cytometer (Beckman Coulter, Inc.) and analyzed CXP software (version 2.1; Beckman Coulter, Inc.).
Western blotting
Total protein extraction from the ischemic penumbra area of brain tissue of the CIRI model rats (n=3) or PC12 cells was performed using a total protein extraction kit. The protein concentration was measured using the BCA method. Equal amounts of total protein extracts (30 µg/lane) were separated by 10% SDS-PAGE and transferred to polyvinylidene difluoride membranes by the semi-dry transfer method. The membranes were blocked with 5% nonfat milk in TBS with 0.1% Tween 20 for 1.5 h at room temperature and subsequently incubated overnight at 4°C with anti-CaSR (1:400), anti-cleaved caspase-3 (1:1,000), anti-β-actin (1:1,000), anti-Bcl-2 (1:800) and anti-Bax (1:800) antibodies, followed by incubation with HRP-conjugated goat anti-mouse IgG (1:3,000) or anti-rabbit IgG (1:3,000) antibodies at room temperature for 1 h. The labeled proteins were detected using the enhanced luminol-based ECL reagent kit (Wuhan Servicebio Technology Co., Ltd.) and the FX5 Spectra Imaging System, and the optical density of the bands was measured by ImageJ software (version 1.8.0; National Institute of Health). The in vitro experiment was repeated three times.
Calcium assay
Calcium concentration in the culture medium and PC12 cells was measured using a commercial kit according to the manufacturer's instructions. After 24 h of OGD/R, the cell culture medium of each group was collected and centrifuged at 4,025 × g at 4°C for 5 min, and the supernatant liquid was collected. The working fluid was prepared according to the manufacturer's instructions. The supernatant liquid and all reagents were added to a 96-well plate at the required proportions and mixed. Finally, the OD values of each well were detected at 610 nm using a microplate reader. Each set of experiments was performed in triplicate. The calcium concentration in the culture medium was calculated as follows: Calcium concentration=(OD value of experimental group-OD value of blank group)/(OD value of standard well-OD value of blank group).
To determine the calcium concentration in PC12 cells, cells from each group were collected and lysed with deionized water at a ratio of 1:9 to extract the total cell protein. The BCA method was used to quantify the proteins in each group of cells. The subsequent steps were the same as those used for the determination of the calcium concentration in the cell culture medium. Each set of experiments was performed in triplicate. The calcium concentration in PC12 cells was calculated as follows: Calcium concentration=(OD value of experimental group-OD value of blank group)/(OD value of standard well-OD value of blank group)/protein concentration.
Statistical analysis
Data are presented as the mean ± SD from ≥3 experimental repeats. All data were analyzed by SPSS 21.0 software (IBM Corp.). One-way analysis of variance followed by Bonferroni's post hoc test was used to compare data among the groups. P<0.05 was considered to indicate a statistically significant difference.
Results
In the present study, the neuroprotective effects of Astragaloside IV against CIRI were first observed in vivo and in vitro. Models of CIRI were established by MCAO/R in rats and by OGD/R in PC12 cells. Secondly, a series of experiments were performed to study the protective mechanism of Astragaloside IV.
Astragaloside IV inhibits MCAO/R-induced cerebral damage in rats
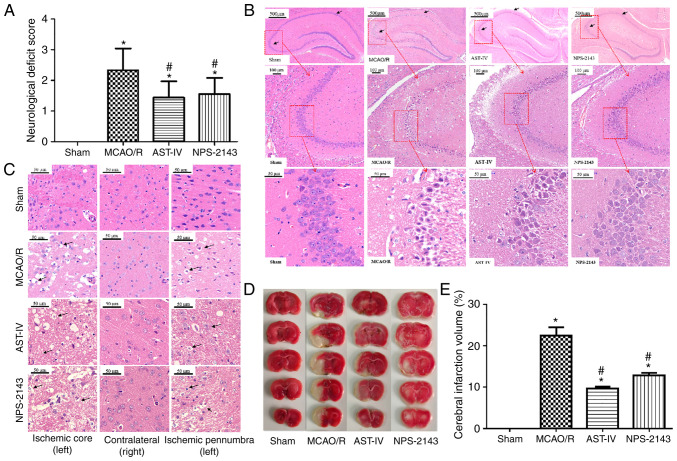
To assess the neuroprotective effects of Astragaloside IV, NFS, TTC and H&E staining were evaluated in a MCAO/R rat model. Rats were administered Astragaloside IV, NPS-2143 or an equal volume of physiological saline for 24 h after reperfusion, and neurological deficits were observed using the Zea Longa Neurological Score, which can effectively evaluate the neurological deficits of MCAO/R rats (39). Rats in the Sham group presented with no neurological deficits; by contrast, a notable increase in neurological deficits was observed in rats of the MCAO/R group (P<0.05 vs. Sham). Treatment with Astragaloside IV and the CaSR inhibitor NPS-2143 significantly decreased the neurological score of MCAO/R rats (P<0.05 vs. MCAO/R; Fig. 1A).
Figure 1.
Neuroprotective effects of Astragaloside IV on a MCAO/R rat model. (A) Neurological function scores of rats at 24 h after post-MCAO/R. n=9. (B) Representative images of H&E staining in the rat hippocampus following CIRI. Magnification, ×50 (top), ×100 (middle) and ×400 (bottom). (C) Representative images of H&E staining in the rat cerebral cortex following CIRI. Magnification, ×400. (D) Representative images of 2,3,5-triphenyltetra-zolium chloride-stained serial coronal brain sections. (E) Infarction volume of rats in different groups at 24 h post-MCAO/R. n=3. *P<0.05 vs. Sham; #P<0.05 vs. MCAO/R. MCAO/R, middle cerebral artery occlusion/reperfusion; CaSR, calcium-sensing receptor; AST-IV, MCAO/R + Astragaloside IV; NPS-2143, MCAO/R + CaSR antagonist; CIRI, cerebral ischemia-reperfusion injury.
H&E staining was used to observe the morphological changes of rat brain tissue and determine the success of model establishment (Fig. 1B and C). Neurons in the hippocampus (Fig. 1B) and the cortex (Fig. 1C) of the rats in the Sham group presented a normal structure, which was similar to that of the nonischemic brain tissue of the MCAO/R rats. The cells exhibited a clear shape, compact structure and clear nucleoli. However, edema and necrosis were observed in the ischemic cortex and hippocampus of the MCAO/R rats: The numbers of neurons decreased, the gaps around neurons increased, and the neurons were swollen or shrunk with disappeared nucleoli and a dissolved nuclear membrane. The pathomorphological changes of the ischemic side brain tissues in the AST-IV and NPS-2143 groups were alleviated compared with those in the MCAO/R group, with fewer apoptotic cells as observed by the cell morphology.
TTC staining revealed no cerebral infarction among rats in the Sham group, whereas the infarction volume in rats of the MCAO/R group was increased (P<0.05 vs. Sham). Astragaloside IV and NPS-2143 significantly reduced the infarction volume of MCAO/R rats (P<0.05 vs. MCAO/R; Fig. 1D and E).
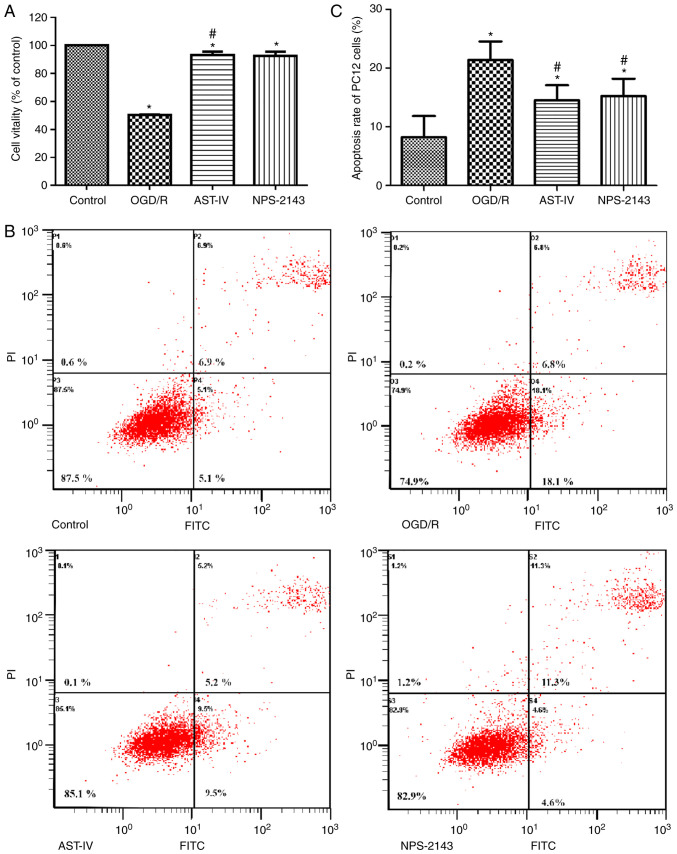
Astragaloside IV increases the viability of PC12 cells and decreases the apoptotic rate following OGD/R
To study the therapeutic potential of Astragaloside IV against OGD/R, the cell viability and apoptotic rates were determined by CCK-8 and flow cytometry assay, respectively. The results demonstrated that the cell viability decreased and the apoptotic rate increased significantly following OGD/R (P<0.05 vs. Control), whereas treatment with Astragaloside IV or the CaSR inhibitor NPS-2143 immediately following reoxygenation improved cell viability and inhibited the apoptotic rate, respectively, compared with those in the OGD/R group (Fig. 2). These results suggested that Astragaloside IV exerted a protective effect on OGD/R PC12 cells, which was similar to that of NPS-2143, suggesting that the protective effect of Astragaloside IV on CIRI maybe associated with CaSR.
Figure 2.
Protective effects of Astragaloside IV on PC12 cells following OGD/R. (A) PC12 cell viability was determined by Cell Counting Kit-8 assay. n=4. (B) Flow cytometry plots of PC12 cells in different groups. (C) Quantitative assessment of the apoptotic rates of PC12 cells. n=3. *P<0.05 vs. Control; #P<0.05 vs. OGD/R. OGD/R, oxygen and glucose deprivation/reoxygenation; AST-IV, OGD/R + Astragaloside IV; NPS-2143, OGD/R + CaSR antagonist.
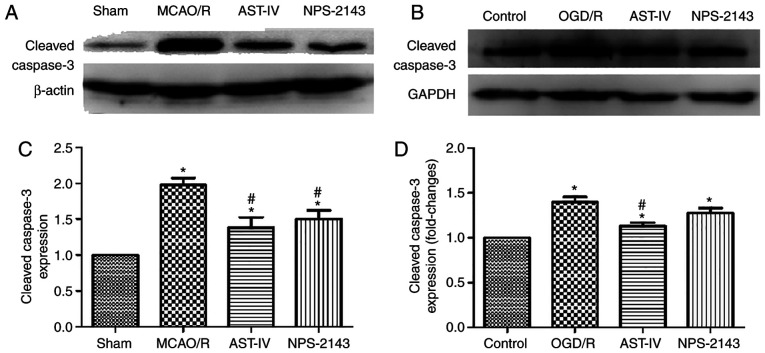
Astragaloside IV reduces the protein expression of cleaved caspase-3
To determine the antiapoptotic effects of Astragaloside IV on OGD/R PC12 cells and MCAO/R rats, the protein expression levels of cleaved caspase-3 were detected as an index of apoptosis by western blotting. The results demonstrated that MCAO/R significantly increased the protein expression levels of cleaved caspase-3 in the rat brain tissue compared with those in the Sham group, and OGD/R had the same effect on PC12 cells (both P<0.05). Compared with that in the OGD/R and MCAO/R groups, treatment with Astragaloside IV or NPS-2143 reduced the levels of cleaved caspase-3 expression (P<0.05). These results suggested that Astragaloside IV may inhibit apoptosis to exert its cerebral anti-ischemic effects. The similar effects of the CaSR inhibitor NPS-2143 to those of Astragaloside IV indicated that the anti-apoptotic effect of Astragaloside IV may be associated with CaSR (Fig. 3).
Figure 3.
Astragaloside IV reduces the protein expression levels of cleaved caspase-3. (A and B) The protein expression levels of cleaved caspase-3 in the rat brain and PC12 cells following different treatments were examined by western blotting. (A) Representative rat brain tissue western blot images from each group. (B) Relative protein expression levels of cleaved caspase-3 in the rat brain. n=3. *P<0.05 vs. Sham; #P<0.05 vs. MCAO/R. (C) Representative western blot images from each group of PC12 cells. (D) Relative expression levels of cleaved caspase-3 in PC12 cells. n=3. *P<0.05 vs. Control; #P<0.05 vs. OGD/R and NPS-2143. MCAO/R, middle cerebral artery occlusion/reperfusion; OGD/R, oxygen and glucose deprivation/reoxygenation; CaSR, calcium-sensing receptor; AST-IV, MCAO/R or OGD/R + Astragaloside IV; NPS-2143, MCAO/R or OGD/R + CaSR antagonist.
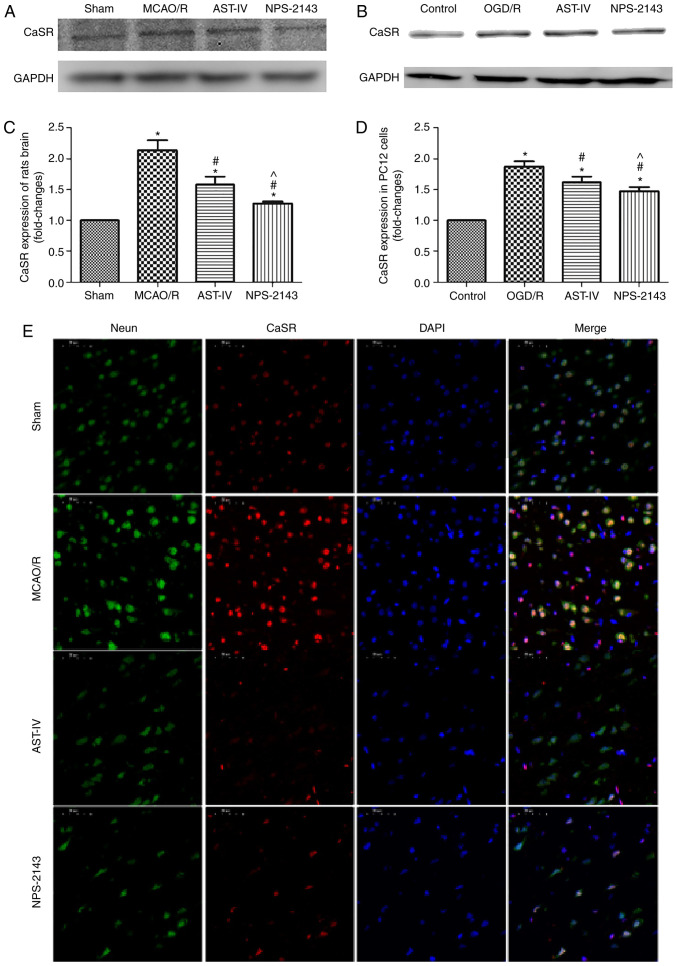
Astragaloside IV decreases the protein expression of CaSR
As aforementioned, Astragaloside IV alleviated CIRI in the rat and PC12 cell models, and its neuroprotective roles were associated with the inhibition of apoptosis and the reduction of the calcium flow into PC12 cells during OGD/R. The possible underlying mechanisms were further investigated. Western blotting results demonstrated that the CaSR protein expression levels in MCAO/R rat brain tissue and OGD/R PC12 cells were increased compared with those in the Sham and Control group, respectively (both P<0.05). Treatment with Astragaloside IV decreased the protein expression levels of CaSR in the MCAO/R brain tissue and OGD/R PC12 cells compared with those in the respective model groups (both P<0.05). In addition, CaSR antagonist NPS-2143 also reduced the expression of CaSR in the brain tissue of MCAO/R rats and in OGD/R PC12 cells (both P<0.05; Fig. 4A-D).
Figure 4.
Astragaloside IV decreased the protein expression of CaSR in vivo and in vitro. The expression of CaSR was detected by western blotting. (A) Representative western blot images from each group in PC12 cells. (B) Relative expression levels of CaSR in PC12 cells. n=3. *P<0.05 vs. control; #P<0.05 vs. OGD/R; ^P<0.05 vs. AST-IV. (C) Representative western blot images from each group of rats. (D) Relative expression levels of CaSR in rats. n=3. *P<0.05 vs. Sham; #P<0.05 vs. MCAO/R. (E) Representative images of double immunofluorescence staining of CaSR and NeuN. Magnification, ×400. CaSR, calcium-sensing receptor; NeuN, neuronal nuclei; MCAO/R, middle cerebral artery occlusion/reperfusion; CaSR, calcium-sensing receptor; OGD/R, oxygen and glucose deprivation/reoxygenation; AST-IV, MCAO/R or OGD/R + Astragaloside IV; NPS-2143, MCAO/R or OGD/R + CaSR antagonist.
Double immunofluorescence staining revealed that the number of neurons in the brain tissue was high, whereas that of CaSR-positive cells was low in the Sham group. The expression of CaSR on neurons in the MCAO/R group appeared to be increased compared with that in the Sham group. Compared with that in the MCAO/R group, the expression of CaSR on neurons in the AST-IV and NPS-2143 groups appeared to be downregulated (Fig. 4E).
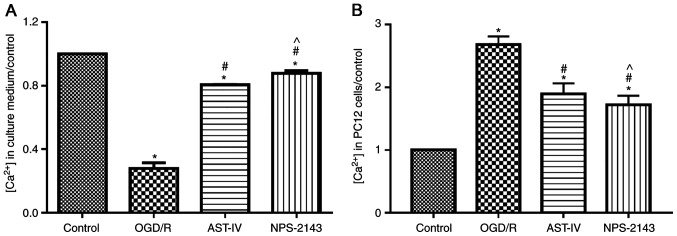
Astragaloside IV inhibits OGD/R-induced calcium flow into PC12 cells
To determine whether Astragaloside IV inhibited calcium overload, the concentration of calcium in the culture medium and PC12 cells was tested using a commercial kit. The results demonstrated that OGD/R promoted the flow of calcium ions into PC12 cells and increased the intracellular calcium concentration compared with that in the control group (P<0.05). By contrast, Astragaloside IV or the CaSR antagonist NPS-2143 reduced the calcium flow into cells and inhibited calcium overload induced by OGD/R (both P<0.05 vs. OGD/R; Fig. 5).
Figure 5.
Astragaloside IV inhibits OGD/R-induced calcium overload in PC12 cells. The calcium concentration in (A) the culture medium and (B) PC12 cells was detected using a commercial kit. n=3, *P<0.05 vs. Control; #P<0.05 vs. OGD/R; ^P<0.05 vs. AST-IV. OGD/R, oxygen and glucose deprivation/reoxygenation; AST-IV, OGD/R + Astragaloside IV; NPS-2143, OGD/R + CaSR antagonist.
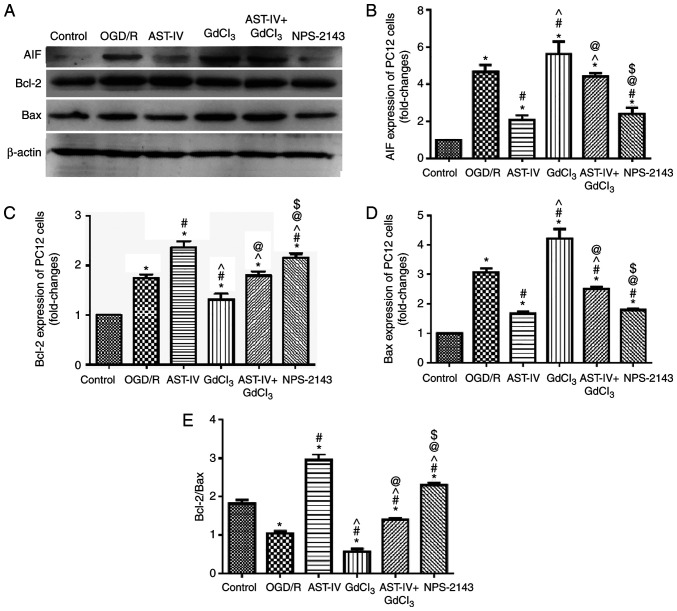
Astragaloside IV decreases the protein expression of AIF and increases the ratio of Bcl-2/Bax during OGD/R
To determine the inhibitory effects of Astragaloside IV on the OGD/R-induced apoptosis of PC12 cells, the expression levels of the proapoptotic proteins AIF and Bax and the antiapoptotic protein Bcl-2 were detected by western blot-ting. OGD/R PC12 cells were treated with Astragaloside IV, the CaSR agonist GdCl3, Astragaloside IV + GdCl3 or the CaSR antagonist NPS-2143. The results demonstrated notable changes in the protein expression levels of AIF, Bax and Bcl-2 among the groups. The expression levels of AIF and Bax increased, whereas those of Bcl-2 decreased significantly in PC12 cells following OGD/R (P<0.05 vs. Control). Treatment with Astragaloside IV decreased the expression levels of AIF and Bax and increased those of Bcl-2 in PC12 cells following OGD/R (P<0.05 vs. OGD/R); the effects of GdCl3 were the opposite to those of Astragaloside IV (P<0.05 vs. OGD/R). These results also revealed that the effects of Astragaloside IV were partly offset by GdCl3 (P<0.05 vs. AST-IV). In addition, the effects of NPS-2143 treatment were similar to those of Astragaloside IV (Fig. 6).
Figure 6.
Astragaloside IV reduces the protein expression levels of AIF and increases the ratio of Bcl-2/Bax in PC12 cells following OGD/R. The expression levels of AIF, Bcl-2 and Bax were detected by western blotting. (A) Representative western blot images in each group. (B-D) Relative expression levels of (B) AIF, (C) Bcl-2 and (D) Bax. (E) Analysis of the Bcl-2/Bax ratio. n=3. *P<0.05 vs. Control; #P<0.05 vs. OGD/R; ^P<0.05 vs. AST-IV; @P<0.05 vs. GdCl3; $P<0.05 vs. AST-IV + GdCl3. Bcl-2, B-cell lymphoma 2; Bcl-2-associated X protein; AIF, apoptosis-inducing factor; OGD/R, oxygen and glucose deprivation/reoxygenation; AST-IV, OGD/R + Astragaloside IV; GdCl3, OGD/R + CaSR agonist; AST-IV + GdCl3, OGD/R + Astragaloside IV + CaSR agonist; NPS-2143, OGD/R + CaSR antagonist.
The changes in the Bcl-2/Bax ratio in the PC12 cells following different treatments were also analyzed; the Bcl-2/Bax ratio was significantly decreased by OGD/R (P<0.05 vs. Control). Both Astragaloside IV and NPS-2143 increased the Bcl-2/Bax ratio in PC12 cells following OGD/R (P<0.05 vs. OGD/R). However, treatment with GdCl3 decreased the Bcl-2/Bax ratio in PC12 cells following OGD/R (P<0.05 vs. OGD/R) and offset the protective effect of Astragaloside IV (P<0.05 vs. GdCl3).
Overall, these results demonstrated that Astragaloside IV and NPS-2143 inhibited the expression levels of pro-apoptotic proteins and increased those of anti-apoptotic proteins, whereas GdCl3 exerted the opposite effects, which may be partly offset by Astragaloside IV. These results suggested that CaSR may be involved in the protective effects of Astragaloside IV over CIRI.
Discussion
Stroke is the most frequent cause of permanent disability in adults worldwide (47,48). Previous studies have demonstrated that brain ischemic injury secondary to arterial occlusion is characterized by neuronal apoptosis (49,50). CIRI triggers complex pathological pathways of the ischemia and reperfusion cascade and ultimately causes irreversible neuronal injury in the ischemic core (51). However, neurons in the penumbra, surrounding the ischemic core, can be salvaged, offering the possibility of rescuing brain tissue following CIRI and reducing post-CIRI disability (15,52). Thus, the use of neuroprotective drugs following reperfusion after ischemic stroke is necessary to reduce apoptosis. The results of the present study demonstrated that Astragaloside IV improved NFS and cerebral infarct volume and significantly reduced the blood-brain barrier permeability in a rat model of MCAO/R through its antiapoptotic effects. The present study also performed further investigation into the neuroprotective mechanism of Astragaloside IV to provide a theoretical basis for its application in patients with stroke.
Apoptosis is one of the main pathological mechanisms of CIRI that begins shortly after cerebral ischemia, is accelerated by reperfusion and partially leads to cerebral infarction (47). The timely and effective inhibition of the apoptosis process can prevent the loss of neuronal cells, reduce the damage to brain tissue caused by ischemia and reperfusion, and reduce the symptoms of neurological defects (5). Our previous study demonstrated that Astragaloside IV attenuated CIRI by promoting the degree of autophagy and downregulating apoptosis in a rat model of MCAO/R and an HT22 cell model of OGD/R (9). The results of the present study revealed that Astragaloside IV relieved brain injury induced by CIRI, reduced apoptosis, inhibited the calcium flow into PC12 cells, decreased CaSR expression and increased the ratio of Bcl-2/Bax compared with those in the corresponding control groups. These results indicated the underlying mechanisms of the neuroprotective effect of Astragaloside IV and demonstrated the pivotal role of CaSR in CIRI.
CaSR is a G protein-coupled receptor that is present throughout the central nervous system (53,54). An increasing number of studies have reported that CaSR is involved in modulating various cellular functions, including cell proliferation, differentiation and apoptosis in the central nervous system by sensing changes in the extracellular calcium concentration (55-58). CaSR can be activated by extracellular calcium, magnesium and CaSR agonists to increase the intracellular calcium concentration (19,57); increased intracellular calcium caused by CaSR contributes to CIRI-induced brain injury. The results of the present study revealed that in PC12 cells subjected to OGD/R, cell viability was decreased and the expression of cleaved caspase-3 was significantly increased compared with those in the control group, accompanied by the upregulation of CaSR, which was reversed by Astragaloside IV and NPS-2143. Similarly, the injury of rat brain tissue induced by MCAO/R was alleviated by treatment with Astragaloside IV or NPS-2143, along with the inhibition of CaSR. These results suggested that Astragaloside IV alleviated CIRI, and that the underlying mechanism may be associated with CaSR.
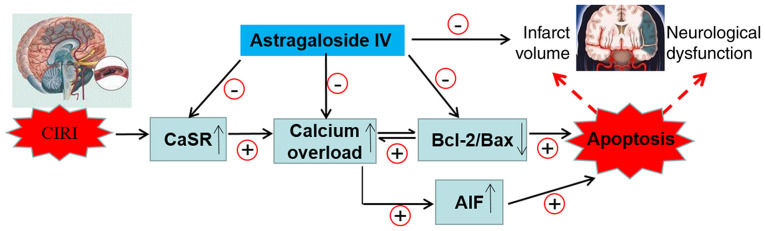
Upregulation of CaSR leads to changes in the expression of AIF, Bcl-2 and Bax that serve a key role in regulating the mitochondrial pathway of apoptosis (59,60). To gain additional insights into the association between apoptosis and the activation of CaSR during CIRI, the intracellular and extracellular concentration of calcium was detected in OGD/R PC12 cells subjected to Astragaloside IV, GdCl3 (a specific CaSR agonist), NPS-2143 (a specific CaSR antagonist) or a combination of Astragaloside IV and GdCl3 treatments, and the protein expression of AIF, Bcl-2 and Bax in vitro was analyzed. The results demonstrated that Astragaloside IV inhibited the calcium flow into the cells, and that the increase in the intracellular calcium concentration in PC12 cells induced the expression of Bcl-2, as well as decreased the protein expression levels of AIF, Bax and CaSR compared with those in the control cells. These effects of Astragaloside IV were similar to those obtained with the CaSR inhibitor NPS-2143 and were partly abolished by the CaSR agonist GdCl3. Therefore, the downregulation of CaSR may contribute to the protective effect of Astragaloside IV on CIRI. In addition, the changes in CaSR expression induced by MCAO/R, the CaSR inhibitor NPS-2143 and the CaSR agonist GdCl3 demonstrated that the decrease in AIF and Bcl-2/Bax may contribute to the mechanism of CaSR in promoting CIRI. These results may help further understand the molecular mechanisms of Astragaloside IV-induced neuroprotection (Fig. 7).
Figure 7.
Neuroprotective mechanisms of Astragaloside IV against CIRI. CIRI, cerebral ischemia-reperfusion injury; CaSR, calcium-sensing receptor; Bcl-2, B-cell lymphoma 2; Bcl-2-associated X protein; AIF, apoptosis-inducing factor.
The results of the present study demonstrated that Astragaloside IV alleviated CIRI in a rat model and decreased the protein expression level of CaSRs. The CaSR inhibitor NPS-2143 exhibited similar effects to those of Astragaloside IV; thus, the effects of the CaSR agonist GdCl3 and Astragaloside IV + GdCl3 on OGD/R-induced injury were further investigated in vitro on PC12 cells. The in vitro experimental results demonstrated that the CaSR agonist GdCl3 aggravated cell injury induced by OGD/R and promoted apoptosis, whereas Astragaloside IV reduced the damage of CaSR agonists. Therefore, Astragaloside IV may inhibit apoptosis by decreasing the protein expression of CaSRs, thus reducing CIRI.
Traditional Chinese medicine glycosides exhibit a wide range of pharmacological activities. Certain glycosides, such as ginsenoside Rg1 and oleuropein, serve an important role in the treatment of central nervous system diseases; however, their blood-brain barrier permeability is low (61,62). By contrast, the small molecular weight of Astragaloside IV (784.97 Da) and the destruction of the blood-brain barrier following CIRI leads to high permeability (63). A previous study on cellular and animal models has demonstrated that Astragaloside IV possesses potent protective effects on the brain (9), which was consistent with the results of the present study. In the present study, the animal sample size was small, as a large number of previous experiments have confirmed the protective effects of Astragaloside IV on CIRI (9); to ensure adherence to animal ethics requirements, a small sample size was used to achieve the experimental purpose. Follow-up experiments with a larger sample size will be performed to further study the neuroprotective mechanism of Astragaloside IV on CIRI, as well as the role of calcium-sensitive receptors in CIRI.
The PC12 cell line used in the present study is a type of highly differentiated PC12-derived neuron-like cell induced by neurotrophic factor (Fig. 8C). Different morphology can be observed in highly differentiated (Fig. 8C), poorly differentiated (Fig. 8B) and undifferentiated (Fig. 8A) PC12 cells. PC12 is a stable cell line that can be directly cultured and passed down, and it has been widely used to study diseases of the nervous system in vitro (43,46,64). Therefore, in the present study, the highly differentiated PC12 cells were used to determine the effects and mechanism of Astragaloside IV on OGD/R-induced apoptosis. Future studies should use primary cultured rat neurons and brain specimens of MCAO/R rats to further study the action of Astragaloside IV on apoptosis in CIRI and explore other potential neuroprotective pathways of Astragaloside IV.
Figure 8.
Morphology of highly differentiated, poorly differentiated and undifferentiated PC12 cells (provided by Procell Life Science & Technology Co., Ltd.).
The results of the present study demonstrated the inhibition of apoptosis may protect neurons by downregulating the expression of CaSR. Astragaloside IV alleviated cerebral injury in MCAO/R rats, improved the viability of PC12 cells following OGD/R, reduced the expression levels of CaSR and inhibited apoptosis compared with those in the corresponding control groups. Astragaloside IV may exert its neuroprotective effect by inhibiting apoptosis through the prevention of the increase of CIRI-induced CaSR expression.
Acknowledgments
The authors would like to thank Professor Shun-Jiang Xu (The First Hospital of Hebei Medical University, Shijiazhuang, China) for providing PC12 cells.
Funding
This study was supported by the Science Foundation of Hebei Province (grant no. H2019423074), Science and Technology Ability Improvement Project of the Hebei University of Chinese Medicine (grant no. KTY2019049), Basic Research Project of Outstanding Young Teachers of the University of Chinese Medicine (grant no. YQ2019007), Hebei Province Graduate Innovative Ability Training Project (grant no. CXZZBS2019156), Research Project of Hebei Administration of Traditional Chinese Medicine (grant no. 2018111) and the Science Foundation for the Higher Education Institutions of Hebei Province (grant no. ZD2018002).
Availability of data and materials
The datasets used and analyzed in the current study are available from the corresponding authors on reasonable request.
Authors' contributions
WJG conceived the study. JLT and YZ performed data analysis. SJD and WJG acquired funding. SJD and YMZ performed the experiments. XHZ was responsible for project administration. YJD established the experimental animal model. WJG supervised the study. XHZ performed the pathomorphology experiments. SJD and YZ wrote the original draft of the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All experiments were approved by the Institutional Animal Care and Use Committee of Hebei University of Chinese Medicine (Shijiazhuang, China; approval no. DWLL2018032).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Hankey GJ. Stroke. Lancet. 2017;389:641–654. doi: 10.1016/S0140-6736(16)30962-X. [DOI] [PubMed] [Google Scholar]
- 2.Writing Group Members. Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, Das SR, de Ferranti S, Després JP, et al. Heart disease and stroke statistics-2016 update: A report from the American heart association. Circulation. 2016;133:e38–e360. doi: 10.1161/CIR.0000000000000350. [DOI] [PubMed] [Google Scholar]
- 3.Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, Brown M, Demaerschalk BM, Hoh B, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: A guideline for healthcare professionals from the American heart association/American stroke association. Stroke. 2019;50:e344–e418. doi: 10.1161/STR.0000000000000211. [DOI] [PubMed] [Google Scholar]
- 4.Qureshi AI, Singh B, Huang W, Du Z, Lobanova I, Liaqat J, Siddiq F. Mechanical thrombectomy in acute ischemic stroke patients performed within and outside clinical trials in the United States. Neurosurgery. 2020;86:E2–E8. doi: 10.1093/neuros/nyz359. [DOI] [PubMed] [Google Scholar]
- 5.Chen YW, Sung SF, Chen CH, Tang SC, Tsai LK, Lin HJ, Huang HY, Po HL, Sun Y, Chen PL, et al. Intravenous thrombolysis administration 3-4.5 h after acute ischemic stroke: A retrospective, multicenter study. Front Neurol. 2019;10:1038. doi: 10.3389/fneur.2019.01038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen CH, Hsieh CL. Effect of acupuncture on oxidative stress iduced by cerebral ischemia-reperfusion injury. Antioxidants (Basel) 2020;9:248. doi: 10.3390/antiox9030248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wu L, Xiong X, Wu X, Ye Y, Jian Z, Zhi Z, Gu L. Targeting oxidative stress and inflammation to prevent ischemia-reperfusion injury. Front Mol Neurosci. 2020;13:28. doi: 10.3389/fnmol.2020.00028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Khandelwal P, Yavagal DR, Sacco RL. Acute ischemic stroke intervention. J Am Coll Cardiol. 2016;67:2631–2644. doi: 10.1016/j.jacc.2016.03.555. [DOI] [PubMed] [Google Scholar]
- 9.Zhang Y, Zhang Y, Jin XF, Zhou XH, Dong XH, Yu WT, Gao WJ. The role of astragaloside IV against cerebral ischemia/reperfusion injury: Suppression of apoptosis via promotion of P62-LC3-autophagy. Molecules. 2019;24:1838. doi: 10.3390/molecules24091838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Liu D, Gu Y, Wang W, Chen W. Astragalin alleviates ischemia/reperfusion-induced brain injury via suppression of endoplasmic reticulum stress. Mol Med Rep. 2020;22:4070–4078. doi: 10.3892/mmr.2020.11448. [DOI] [PubMed] [Google Scholar]
- 11.Liu Y, Yang H, Jia G, Li L, Chen H, Bi J, Wang C. The synergistic neuroprotective effects of combined rosuvastatin and resveratrol pretreatment against cerebral ischemia/reperfusion injury. J Stroke Cerebrovasc Dis. 2018;27:1697–1704. doi: 10.1016/j.jstrokecerebrovasdis.2018.01.033. [DOI] [PubMed] [Google Scholar]
- 12.Gu JH, Ge JB, Li M, Wu F, Zhang W, Qin ZH. Inhibition of NF-κB activation is associated with anti-inflammatory and anti-apoptotic effects of ginkgolide B in a mouse model of cerebral ischemia/reperfusion injury. Eur J Pharm Sci. 2012;47:652–660. doi: 10.1016/j.ejps.2012.07.016. [DOI] [PubMed] [Google Scholar]
- 13.Zhou H, Yang WS, Li Y, Ren T, Peng L, Guo H, Liu JF, Zhou Y, Zhao Y, Yang LC, Jin X. Oleoylethanolamide attenuates apoptosis by inhibiting the TLR4/NF-κB and ERK1/2 signaling pathways in mice with acute ischemic stroke. Naunyn Schmiedebergs Arch Pharmacol. 2017;390:77–84. doi: 10.1007/s00210-016-1309-4. [DOI] [PubMed] [Google Scholar]
- 14.Naito MG, Xu D, Amin P, Lee J, Wang H, Li W, Kelliher M, Pasparakis M, Yuan J. Sequential activation of necroptosis and apoptosis cooperates to mediate vascular and neural pathology in stroke. Proc Natl Acad Sci USA. 2020;117:4959–4970. doi: 10.1073/pnas.1916427117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bai X, Tan TY, Li YX, Li Y, Chen YF, Ma R, Wang SY, Li Q, Liu ZQ. The protective effect of cordyceps sinensis extract on cerebral ischemic injury via modulating the mitochondrial respiratory chain and inhibiting the mitochondrial apoptotic pathway. Biomed Pharmacother. 2020;124:109834. doi: 10.1016/j.biopha.2020.109834. [DOI] [PubMed] [Google Scholar]
- 16.Wang HL, Zhou QH, Xu MB, Zhou XL, Zheng GQ. Astragaloside IV for experimental focal cerebral ischemia: Preclinical evidence and possible mechanisms. Oxid Med Cell Longev. 2017;2017:8424326. doi: 10.1155/2017/8424326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lu FH, Tian Z, Zhang WH, Zhao YJ, Li HL, Ren H, Zheng HS, Liu C, Hu GX, Tian Y, et al. Calcium-sensing receptors regulate cardiomyocyte Ca2+ signaling via the sarcoplasmic reticulum-mitochondrion interface during hypoxia/reoxygenation. J Biomed Sci. 2010;17:50. doi: 10.1186/1423-0127-17-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yin F, Zhou H, Fang Y, Li C, He Y, Yu L, Wan H, Yang J. Astragaloside IV alleviates ischemia reperfusion-induced apoptosis by inhibiting the activation of key factors in death receptor pathway and mitochondrial pathway. J Ethnopharmacol. 2020;248:112319. doi: 10.1016/j.jep.2019.112319. [DOI] [PubMed] [Google Scholar]
- 19.Yin B, Hou XW, Lu ML. Astragaloside IV attenuates myocardial ischemia/reperfusion injury in rats via inhibition of calcium-sensing receptor-mediated apoptotic signaling path-ways. Acta Pharmacol Sin. 2019;40:599–607. doi: 10.1038/s41401-018-0082-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang Y, Qiao L, Xu W, Wang X, Li H, Xu W, Chu K, Lin Y. Paeoniflorin attenuates cerebral ischemia-induced injury by regulating Ca2+/CaMKII/CREB signaling pathway. Molecules. 2017;22:359. doi: 10.3390/molecules22030359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yan L, Zhu T, Sun T, Wang L, Pan S, Tao Z, Yang Z, Cao K. Activation of calcium-sensing receptors is associated with apoptosis in a model of simulated cardiomyocytes ischemia/reperfusion. J Biomed Res. 2010;24:301–307. doi: 10.1016/S1674-8301(10)60042-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kienitz MC, Niemeyer A, Konig GM, Kostenis E, Pott L, Rinne A. Biased signaling of Ca2+-sensing receptors in cardiac myocytes regulates GIRK channel activity. J Mol Cell Cardiol. 2019;130:107–121. doi: 10.1016/j.yjmcc.2019.03.022. [DOI] [PubMed] [Google Scholar]
- 23.Zhen Y, Ding C, Sun J, Wang Y, Li S, Dong L. Activation of the calcium-sensing receptor promotes apoptosis by modulating the JNK/p38 MAPK pathway in focal cerebral ischemia-reperfusion in mice. Am J Transl Res. 2016;8:911–921. [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang ZL, Li ZR, Li JS, Wang SR. Calcium-sensing receptor antagonist NPS-2143 suppresses proliferation and invasion of gastric cancer cells. Cancer Gene Ther. 2019;27:548–557. doi: 10.1038/s41417-019-0128-4. [DOI] [PubMed] [Google Scholar]
- 25.Pearce SH, Thakker RV. The calcium sensing receptor: Insights into extracellular calcium homeostasis in health and disease. J Endocrinol. 1997;154:371–378. doi: 10.1677/joe.0.1540371. [DOI] [PubMed] [Google Scholar]
- 26.Chattopadhyay N, Vassilev PM, Brown EW. Calcium-sensing receptor: Roles in and beyond systemic calcium homeostasis. Biol Chem. 1997;378:759–768. [PubMed] [Google Scholar]
- 27.Meng D, Chen XJ, Bian YY, Li P, Yang D, Zhang JN. Effect of astragalosides on intracellular calcium overload in cultured cardiac myocytes of neonatal rats. Am J Chin Med. 2005;33:11–20. doi: 10.1142/S0192415X05002618. [DOI] [PubMed] [Google Scholar]
- 28.Hu B, Tong F, Xu L, Shen Z, Yan L, Xu G, Shen R. Role of calcium sensing receptor in streptozotocin-induced diabetic rats exposed to renal ischemia reperfusion injury. Kidney Blood Press Res. 2018;43:276–286. doi: 10.1159/000487685. [DOI] [PubMed] [Google Scholar]
- 29.Pan R, Cai J, Zhan L, Guo Y, Huang RY, Li X, Zhou M, Xu D, Zhan J, Chen H. Buyang Huanwu decoction facilitates neuro-rehabilitation through an improvement of synaptic plasticity in cerebral ischemic rats. BMC Complement Altern Med. 2017;17:173. doi: 10.1186/s12906-017-1680-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zheng XW, Shan CS, Xu QQ, Wang Y, Shi YH, Wang Y, Zheng GQ. Buyang Huanwu decoction targets SIRT1/VEGF pathway to promote angiogenesis after cerebral ischemia/reperfusion injury. Front Neurosci. 2018;12:911. doi: 10.3389/fnins.2018.00911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Qi LW, Yu QT, Yi L, Ren MT, Wen XD, Wang YX, Li P. Simultaneous determination of 15 marker constituents in various radix Astragali preparations by solid-phase extraction and high-performance liquid chromatography. J Sep Sci. 2008;31:97–106. doi: 10.1002/jssc.200700286. [DOI] [PubMed] [Google Scholar]
- 32.Sun WX, Zhang ZF, Xie J, He Y, Cheng Y, Ding LS, Luo P, Qing LS. Determination of an astragaloside IV derivative LS-102 in plasma by ultra-performance liquid chromatography-tandem mass spectrometry in dog plasma and its application in a pharmacokinetic study. Phytomedicine. 2019;53:243–251. doi: 10.1016/j.phymed.2018.09.019. [DOI] [PubMed] [Google Scholar]
- 33.Qu YZ, Li M, Zhao YL, Zhao ZW, Wei XY, Liu JP, Gao L, Gao GD. Astragaloside IV attenuates cerebral ischemia-reperfusion-induced increase in permeability of the blood-brain barrier in rats. Eur J Pharmacol. 2009;606:137–141. doi: 10.1016/j.ejphar.2009.01.022. [DOI] [PubMed] [Google Scholar]
- 34.Yang J, Li J, Lu J, Zhang Y, Zhu Z, Wan H. Synergistic protective effect of astragaloside IV-tetramethylpyrazine against cerebral ischemic-reperfusion injury induced by transient focal ischemia. J Ethnopharmacol. 2012;140:64–72. doi: 10.1016/j.jep.2011.12.023. [DOI] [PubMed] [Google Scholar]
- 35.Yin YY, Li WP, Gong HL, Zhu FF, Li WZ, Wu GC. Protective effect of astragaloside on focal cerebral ischemia/reperfusion injury in rats. Am J Chin Med. 2010;38:517–527. doi: 10.1142/S0192415X10008020. [DOI] [PubMed] [Google Scholar]
- 36.Li M, Ma RN, Li LH, Qu YZ, Gao GD. Astragaloside IV reduces cerebral edema post-ischemia/reperfusion correlating the suppression of MMP-9 and AQP4. Eur J Pharmacol. 2013;715:189–195. doi: 10.1016/j.ejphar.2013.05.022. [DOI] [PubMed] [Google Scholar]
- 37.Li M, Qu YZ, Zhao ZW, Wu SX, Liu YY, Wei XY, Gao L, Gao GD. Astragaloside IV protects against focal cerebral ischemia/reperfusion injury correlating to suppression of neutrophils adhesion-related molecules. Neurochem Int. 2012;60:458–465. doi: 10.1016/j.neuint.2012.01.026. [DOI] [PubMed] [Google Scholar]
- 38.Liu P, Tang YY, Yang XS, Dai J, Yang M, Zhang H, Liu Y, Yan H, Song XY. Validation of a preclinical animal model to assess brain recovery after acute stroke. Eur J Pharmacol. 2018;835:75–81. doi: 10.1016/j.ejphar.2018.07.035. [DOI] [PubMed] [Google Scholar]
- 39.Longa EZ, Weinstein PR, Carlson S, Cummins R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke. 1989;20:84–91. doi: 10.1161/01.STR.20.1.84. [DOI] [PubMed] [Google Scholar]
- 40.Zhou P, Lu S, Luo Y, Wang S, Yang K, Zhai Y, Sun G, Sun X. Attenuation of TNF-α-induced inflammatory injury in endothelial cells by ginsenoside Rb1 via inhibiting NF-κB, JNK and p38 signaling pathways. Front Pharmacol. 2017;8:464. doi: 10.3389/fphar.2017.00464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Peng L, Yin J, Wang S, Ge M, Han Z, Wang Y, Zhang M, Xie L, Li Y. TGF-β2/Smad3 signaling pathway activation through enhancing VEGF and CD34 ameliorates cerebral ischemia/reperfusion injury after isoflurane post-conditioning in rats. Neurochem Res. 2019;44:2606–2618. doi: 10.1007/s11064-019-02880-8. [DOI] [PubMed] [Google Scholar]
- 42.Wang Y, Li B, Zhang X. Scutellaria barbata D: Don (SBD) protects oxygen glucose deprivation/reperfusion-induced injuries of PC12 cells by up-regulating Nrf2. Artif Cells Nanomed Biotechnol. 2019;47:1797–1807. doi: 10.1080/21691401.2019.1610413. [DOI] [PubMed] [Google Scholar]
- 43.Zhao DY, Yu DD, Ren L, Bi GR. Ligustilide protects PC12 cells from oxygen-glucose deprivation/reoxygenation-induced apoptosis via the LKB1-AMPK-mTOR signaling pathway. Neural Regen Res. 2020;15:473–481. doi: 10.4103/1673-5374.266059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Meng X, Xie W, Xu Q, Liang T, Xu X, Sun G, Sun X. Neuroprotective effects of radix scrophulariae on cerebral ischemia and reperfusion injury via MAPK pathways. Molecules. 2018;23:2401. doi: 10.3390/molecules23092401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li ZR, Yang L, Zhen J, Zhao Y, Lu ZN. Nobiletin protects PC12 cells from ERS-induced apoptosis in OGD/R injury via activation of the PI3K/AKT pathway. Exp Ther Med. 2018;16:1470–1476. doi: 10.3892/etm.2018.6330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang G, Wang T, Zhang Y, Li F, Yu B, Kou J. Schizandrin protects against OGD/R-induced neuronal injury by suppressing autophagy: Involvement of the AMPK/mTOR pathway. Molecules. 2019;24:3624. doi: 10.3390/molecules24193624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Donnan GA, Fisher M, Macleod M, Davis SM. Stroke. Lancet. 2008;371:1612–1623. doi: 10.1016/S0140-6736(08)60694-7. [DOI] [PubMed] [Google Scholar]
- 48.Georgiopoulos G, Ntaios G, Stamatelopoulos K, Manios E, Korompoki E, Vemmou E, Milionis H, Masi S, Lip GYH, Vemmos K. Comparison of risk scores for the prediction of the overall cardiovascular risk in patients with ischemic stroke: The athens stroke registry. J Stroke Cerebrovasc Dis. 2019;28:104415. doi: 10.1016/j.jstrokecerebrovasdis.2019.104415. [DOI] [PubMed] [Google Scholar]
- 49.Li D, Ai Y. Hydrogen saline suppresses neuronal cell apoptosis and inhibits the p38 mitogenactivated protein kinase-caspase3 signaling pathway following cerebral ischemia-reperfusion injury. Mol Med Rep. 2017;16:5321–5325. doi: 10.3892/mmr.2017.7294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tan XF, Qin T, Li N, Yang YG, Zheng JH, Xie L, Chen MH. High-potassium preconditioning enhances tolerance to focal cerebral ischemia-reperfusion injury through anti-apoptotic effects in male rats. J Neurosci Res. 2019;97:1253–1265. doi: 10.1002/jnr.24483. [DOI] [PubMed] [Google Scholar]
- 51.Dirnagl U, Iadecola C, Moskowitz MA. Pathobiology of ischaemic stroke: An integrated view. Trends Neurosci. 1999;22:391–397. doi: 10.1016/S0166-2236(99)01401-0. [DOI] [PubMed] [Google Scholar]
- 52.Ren Q, Hu Z, Jiang Y, Tan X, Botchway BOA, Amin N, Lin G, Geng Y, Fang M. SIRT1 protects against apoptosis by promoting autophagy in the oxygen glucose deprivation/reperfusion-induced injury. Front Neurol. 2019;10:1289. doi: 10.3389/fneur.2019.01289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rogers KV, Dunn CK, Hebert SC, Brown EM. Localization of calcium receptor mRNA in adult rat central nervous system by in situ hybridization. Brain Res. 1997;744:47–56. doi: 10.1016/S0006-8993(96)01070-0. [DOI] [PubMed] [Google Scholar]
- 54.Ruat M, Molliver ME, Snowman AM, Snyder SH. Calcium sensing receptor: Molecular cloning in rat and localization to nerve terminals. Proc Natl Acad Sci USA. 1995;92:3161–3165. doi: 10.1073/pnas.92.8.3161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bandyopadhyay S, Tfelt-Hansen J, Chattopadhyay N. Diverse roles of extracellular calcium-sensing receptor in the central nervous system. J Neurosci Res. 2010;88:2073–2082. doi: 10.1002/jnr.22391. [DOI] [PubMed] [Google Scholar]
- 56.Ruat M, Traiffort E. Roles of the calcium sensing receptor in the central nervous system. Best Pract Res Clin Endocrinol Metab. 2013;27:429–442. doi: 10.1016/j.beem.2013.03.001. [DOI] [PubMed] [Google Scholar]
- 57.Sun YH, Li YQ, Feng SL, Li BX, Pan ZW, Xu CQ, Li TT, Yang BF. Calcium-sensing receptor activation contributed to apoptosis stimulates TRPC6 channel in rat neonatal ventricular myocytes. Biochem Biophys Res Commun. 2010;394:955–961. doi: 10.1016/j.bbrc.2010.03.096. [DOI] [PubMed] [Google Scholar]
- 58.Tfelt-Hansen J, Hansen JL, Smajilovic S, Terwilliger EF, Haunso S, Sheikh SP. Calcium receptor is functionally expressed in rat neonatal ventricular cardiomyocytes. Am J Physiol Heart Circ Physiol. 2006;290:H1165–H1171. doi: 10.1152/ajpheart.00821.2005. [DOI] [PubMed] [Google Scholar]
- 59.Zhang G, Zhang T, Wu L, Zhou X, Gu J, Li C, Liu W, Long C, Yang X, Shan L, et al. Neuroprotective effect and mechanism of action of tetramethylpyrazine nitrone for ischemic stroke therapy. Neuromolecular Med. 2018;20:97–111. doi: 10.1007/s12017-018-8478-x. [DOI] [PubMed] [Google Scholar]
- 60.Lončarević-Vasiljković N, Milanović D, Pešić V, Tešić V, Brkić M, Lazić D, Avramović V, Kanazir S. Dietary restriction suppresses apoptotic cell death, promotes Bcl-2 and Bcl-xl mRNA expression and increases the Bcl-2/Bax protein ratio in the rat cortex after cortical injury. Neurochem Int. 2016;96:69–76. doi: 10.1016/j.neuint.2016.02.017. [DOI] [PubMed] [Google Scholar]
- 61.Zheng T, Jiang H, Jin R, Zhao Y, Bai Y, Xu H, Chen Y. Ginsenoside Rg1 attenuates protein aggregation and inflammatory response following cerebral ischemia and reperfusion injury. Eur J Pharmacol. 2019;853:65–73. doi: 10.1016/j.ejphar.2019.02.018. [DOI] [PubMed] [Google Scholar]
- 62.Yu H, Liu P, Tang H, Jing J, Lv X, Chen L, Jiang L, Xu J, Li J. Oleuropein, a natural extract from plants, offers neuroprotection in focal cerebral ischemia/reperfusion injury in mice. Eur J Pharmacol. 2016;775:113–119. doi: 10.1016/j.ejphar.2016.02.027. [DOI] [PubMed] [Google Scholar]
- 63.Wang Y, Luo J, Li SY. Nano-curcumin simultaneously protects the blood-brain barrier and reduces M1 microglial activation during cerebral ischemia-reperfusion injury. ACS Appl Mater Interfaces. 2019;11:3763–3770. doi: 10.1021/acsami.8b20594. [DOI] [PubMed] [Google Scholar]
- 64.Hou Y, Wang J, Feng J. The neuroprotective effects of curcumin are associated with the regulation of the reciprocal function between autophagy and HIF-1α in cerebral ischemia-reperfusion injury. Drug Des Devel Ther. 2019;13:1135–1144. doi: 10.2147/DDDT.S194182. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed in the current study are available from the corresponding authors on reasonable request.