Abstract
The emergence of immune checkpoint inhibitors (ICIs) has revolutionized the treatment of recurrent/metastatic (R/M) head and neck squamous cell carcinoma (HNSCC). Biomarkers of the therapeutic efficacy of ICIs have been extensively investigated. In this study, we aimed to analyze whether molecular phenotypes of circulating tumor cells (CTCs) are associated with treatment responses and clinical outcomes in patients with R/M HNSCC treated with nivolumab. Peripheral blood samples were collected before treatment initiation and after four infusions of nivolumab. CTCs isolated by depletion of CD45-positive cells were analyzed to determine the expression of EPCAM, MET, KRT19, and EGFR using real-time quantitative polymerase chain reaction. CTC-positive samples were analyzed to determine the expression of PIK3CA, CCND1, SNAI1, VIM, ZEB2, CD44, NANOG, ALDH1A1, CD47, CD274, and PDCD1LG2. Of 30 patients treated with nivolumab, 28 (93.3%) were positive for CTCs. In 20 CTC-positive patients, molecular alterations in CTCs before and after nivolumab treatment were investigated. Patients with MET-positive CTCs had significantly shorter overall survival than those with MET-negative CTCs (p = 0.027). The expression level of CCND1 in CTCs of disease-controlled patients was significantly higher than that of disease-progressed patients (p = 0.034). In disease-controlled patients, the expression level of CCND1 in CTCs significantly decreased after nivolumab treatment (p = 0.043). The NANOG expression in CTCs was significantly increased in disease-controlled patients after nivolumab treatment (p = 0.036). Our findings suggest that the molecular profiling of CTCs is a promising tool to predict the treatment efficacy of nivolumab.
Subject terms: Cancer immunotherapy, Head and neck cancer, Tumour biomarkers
Introduction
The immune checkpoint inhibitor (ICI), nivolumab, has shown a significant improvement compared with the investigator’s preferred chemotherapy regimen and is currently used as a new therapeutic option in patients with recurrent and metastatic (R/M) head and neck squamous cell carcinoma (HNSCC)1–3. Recently, another ICI, pembrolizumab, has also been reported to have clinical benefit in R/M HNSCC4. Thus, the emergence of ICIs has revolutionized in the treatment of R/M HNSCC; however, the objective response rate is still 20%. Therefore, further improvements and identification of biomarkers for therapeutic efficacy are urgently needed.
To date, a variety of candidate biomarkers for R/M HNSCC and other malignancies have been extensively investigated5–8. Tumor tissue-derived biomarkers such as programmed death-ligand 1 (PD-L1) expression, interferon-γ signature, CD8 T-cell infiltration, and tumor mutational burden are potential sources for the identification of candidate biomarkers. However, the use of tissue specimens in biomarker discovery for ICIs has raised several concerns, such as tissue quality, tissue heterogeneity, and the time of collection. In particular, the tumor microenvironment might fluctuate with disease progression, treatment pressures, and sites of growth.
Recently, accumulating evidence has indicated that liquid biopsy, including circulating tumor cells (CTCs), cell-free DNA, and exosomes, is a reliable method to obtain real-time tumor information9–11. Our previous studies on HNSCC have shown that the presence of CTCs was significantly associated with treatment resistance, locoregional recurrence, and shorter progression-free survival12. Notably, the MET and CD274 expression in CTCs showed a poorer and a better prognosis, respectively. Molecular profiling of CTCs may provide new insights to improve patient selection, maximize efficacy, and predict response as one of the more reliable biomarkers. In the present study, we aimed to analyze whether the molecular characteristics of CTCs can be used as biomarkers to predict treatment efficacy in patients with R/M HNSCC treated with nivolumab; we also aimed to investigate the molecular alterations in CTCs during nivolumab treatment.
Results
Patient characteristics
A total of 30 patients who had histologically confirmed R/M HNSCC were enrolled in this study. Their characteristics are listed in Table 1. The tumor origins included the paranasal sinus (n = 5), oral cavity (n = 3), nasopharynx (n = 2), oropharynx (n = 5), hypopharynx (n = 12), larynx (n = 2), and parotid gland (n = 1). The treatment responses were as follows: complete response (CR) in 0 (0.0%) patients, partial response (PR) in 6 (20.0%), stable disease (SD) in 6 (20.0%), and progressive disease (PD) in 18 (60.0%) patients. The objective response rate and disease control rate were 20.0% and 40.0%, respectively. Eight patients (26.7%) discontinued 4 courses of nivolumab due to rapid disease progression (n = 6) and intolerable immune-related adverse events (n = 2, intestinal lung disease and colitis), respectively. Although we analyzed clinical features between disease-controlled and disease-progressed patients, there was no significant difference in clinical features evaluated.
Table 1.
Patient demographics and clinical characteristics.
| Clinical variable | Disease-controlled patient | Disease-progressed patient | P value |
|---|---|---|---|
| n = 12 | n = 18 | ||
| Age (years): median 67 years old | |||
| < 67 | 6 | 9 | > 0.9999 |
| ≥ 67 | 6 | 9 | |
| Sex | |||
| Male | 12 | 14 | 0.1297 |
| Female | 0 | 4 | |
| Primary site | |||
| Paranasal sinus | 1 | 4 | 0.3884 |
| Oral cavity | 2 | 1 | |
| Nasopharynx | 0 | 2 | |
| Oropharynx | 1 | 4 | |
| Hypopharynx | 7 | 5 | |
| Larynx | 1 | 1 | |
| Parotid gland | 0 | 1 | |
| Local recurrence | |||
| (−) | 7 | 8 | 0.7104 |
| (+) | 5 | 10 | |
| Lymph node metastasis | |||
| (−) | 7 | 8 | 0.7104 |
| (+) | 5 | 10 | |
| Distant metastasis | |||
| (−) | 5 | 8 | > 0.9999 |
| (+) | 7 | 10 | |
| Performance status | |||
| 0 | 10 | 9 | 0.1213 |
| 1 | 2 | 9 | |
| Previous radiotherapy | |||
| (−) | 3 | 3 | 0.6599 |
| (+) | 9 | 15 | |
| PD-L1 expression | |||
| < 1% | 3 | 10 | 0.4197 |
| ≥ 1% | 6 | 8 | |
| Unevaluable | 3 | 0 | |
| Nivolumab treatment | |||
| < 4 times | 3 | 5 | > 0.9999 |
| ≥ 4 times | 9 | 13 | |
Of the 30 patients with R/M HNSCC treated with nivolumab, 28 (93.3%) were positive for CTCs, 20 of whom underwent peripheral blood sample collection at two time points: before treatment initiation and after 4 infusions of nivolumab (Supplementary Fig. 1).
Molecular phenotype of CTCs and their correlation with treatment responses
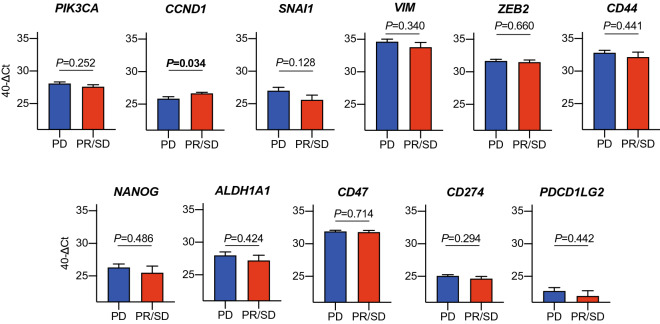
To investigate the relationship between the molecular phenotype of CTCs and treatment responses, the patients with CTCs were divided into two groups according to treatment responses: disease-controlled and disease-progressed patients. First, we analyzed whether the expression of epithelial-related markers in CTCs before treatment initiation is correlated with treatment efficacy and clinical outcome. There was no significant correlation between the expression of epithelial-related genes in CTCs and treatment efficacy (data not shown); however, patients with MET-positive CTCs had a significantly shorter overall survival compared with those with MET-negative CTCs (p = 0.027) (Fig. 1 and Table 2). We then investigated which gene expression in CTCs was associated with treatment responses. Notably, CCND1 expression in CTCs from disease-controlled patients was significantly higher than that in disease-progressed patients (p = 0.034, Fig. 2). Furthermore, we investigated whether the levels of gene expression in the CTCs was associated with the prognosis; however, there was no relationship between gene expression of CTCs and prognosis in patients treated with nivolumab (Supplementary Table 1).
Figure 1.
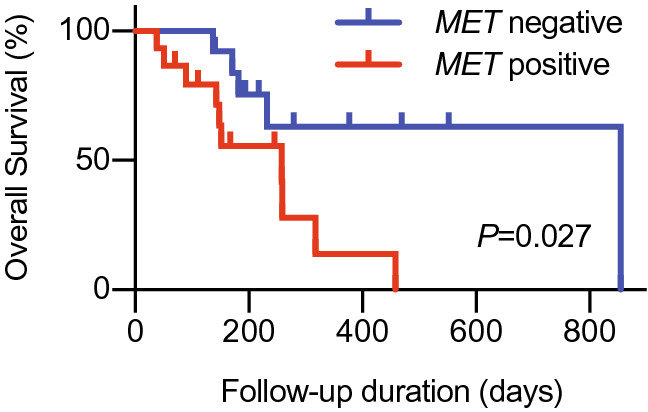
The expression of MET in CTCs and correlation with clinical outcome in R/M HNSCC patients treated with nivolumab. The Kaplan–Meier curve and log-rank test for overall survival were performed to compare the differences between patients with MET-positive CTCs and those with negative CTCs.
Table 2.
Prognostic value of epithelial-related markers in CTCs.
| Gene symbol | P-value | HR | 95% CI | ||
|---|---|---|---|---|---|
| EPCAM | + | 12 | 0.3388 | 1.626 | 0.5335–4.955 |
| − | 16 | ||||
| MET | + | 15 | 0.0265 | 3.045 | 1.082–8.567 |
| − | 13 | ||||
| KRT19 | + | 25 | 0.7516 | 1.265 | 0.3212–4.980 |
| − | 3 | ||||
| EGFR | + | 16 | 0.9933 | 1.004 | 0.3576–2.820 |
| − | 12 | ||||
Figure 2.
Gene expression in CTCs and treatment responses. The patients with CTCs were divided into two groups according to treatment responses: the disease-controlled group and disease-progressed group. Eleven gene expressions in CTCs were compared.
Molecular alterations of CTCs following nivolumab treatment
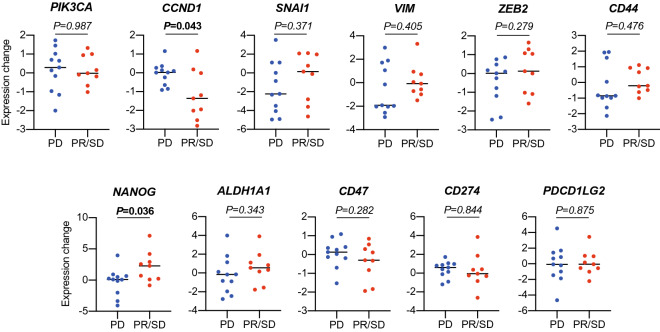
We investigated the change in gene expression in CTCs from individual patients during nivolumab treatment. Although there was no significant change in gene expression including PD-L1 in CTCs in all treated patients by nivolumab treatment (data not shown), the expression level of CCND1 in CTCs in disease-controlled patients, not in disease-progressed patients, was significantly reduced by nivolumab treatment (p = 0.043) (Fig. 3). By contrast, the NANOG expression levels in CTCs in disease-controlled patients were significantly increased by nivolumab treatment (p = 0.036) (Fig. 3).
Figure 3.
Changes in gene expression in CTCs during nivolumab treatment. The patients with CTCs were divided into two groups according to treatment responses: the disease-controlled group and disease-progressed group. The changes in gene expression in CTCs was compared between the two groups.
TCGA analysis
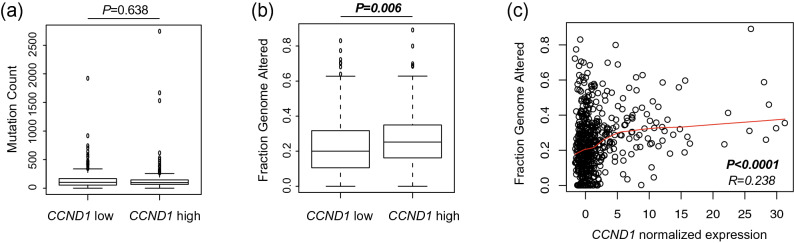
To further elucidate the underlying mechanism of CCND1 that affects the patient’s responses to nivolumab treatment, the relationship between CCND1 expression and genomic alterations in HNSCC was analyzed using the TCGA database. In 515 patients with HNSCC, although no significant difference was observed between CCND1 expression and tumor mutation counts, tumors with high CCND1 expression had significantly higher FGA than those with low CCND1 expression (CCND1 expression and tumor mutation counts; p = 0.638, CCND1 expression and FGA; p = 0.006, Fig. 4a,b). Moreover, there was a weak correlation between the expression of CCND1 and FGA (Fig. 4c, r = 0.238), but not tumor mutation counts (data not shown).
Figure 4.
Genomic alterations in head and neck squamous cell carcinoma. Two genomic alterations, tumor mutation counts (a) and fraction genome altered (b), were compared between tumors with low CCND1 expression and tumors with high CCND1 expression. The cutoff value of high and low CCND1 expression was set as the median. The correlation between the expression of CCND1 and fraction genome altered (c) was evaluated.
Discussion
The primary objective of this study was to explore whether the molecular profiling of CTCs could predict treatment efficacy in patients with R/M HNSCC treated with nivolumab. We succeeded in achieving this objective to some extent, and the following points are of particular importance: (1) patients with MET-positive CTCs have a poorer prognosis compared with those with MET-negative CTCs, (2) CTCs before treatment initiation in the disease-controlled patients showed significantly higher CCND1 expression and significantly reduced by nivolumab treatment compared with those in disease-progressed patients, and (3) the expression of NANOG in CTCs significantly increased in the disease-controlled patients.
The first point probably indicated that MET-positive CTCs are resistant not only to chemotherapy and/or radiotherapy but also to immunotherapy. In our previous study on patients with treatment-naïve HNSCC, patients with MET-positive CTCs had a shorter progression-free survival than those with MET-negative CTCs12. c-MET overexpression and amplification in HNSCC have been shown to drive tumorigenesis, proliferation, invasion, and metastasis, and is commonly associated with resistance to therapy13–16. With regard to the immunological role of c-Met expression in tumor cells, Saigi et al. demonstrated that c-Met activation induces PD-L1 expression via a pathway independent of JAK/STAT activation17. On the contrary, Wang et al. suggested that c-MET expression in tumor cells suppresses the function of NK cells and T cells via the production of indoleamine-2,3-dioxygenase18. In addition, the activation of HGF/Met signaling in HNSCC increases glycolysis, which would result in the suppression of T-cell functions19. Thus, c-Met expression in tumor cells contributes to immune suppression through a variety of mechanisms. Although the clinical application of ICIs is currently being expanded to a variety of malignancies, a combination of c-Met targeting therapies with ICIs should be considered in patients with MET-positive CTCs to improve the therapeutic efficacy of cancer immunotherapy.
The second point suggests that CCND1 expression in tumor cells might be a good target for T cells reactivated by ICIs. Among several gene expressions related to cell growth, epithelial-mesenchymal transition, cancer stemness, and immune regulation, CCND1 expression in CTCs is significantly associated with disease control. CCND1 is known to play a critical role in cell cycle regulation, and high expression of CCND1 is associated with treatment resistance and poor prognosis in HNSCC, as with MET20–22. Therefore, the overexpression of cyclin D1 is associated with genome instability, and consequently results in the increase of somatic mutations and neoantigens. In the TCGA database analysis of HNSCC, no significant difference was observed in the in mutation counts between the low CCDN1 group and high CCDN1 group; however, the FGA was significantly higher in the high CCND1 group than in the low CCND1 group. The FGA is the percentage of genome affected by gains or losses in the copy number, which may increase the neoantigen loads and induce tumor (neoantigen)-specific T cells. Another possibility is that CCND1 protein itself may function as a tumor antigen recognized by T cells. In fact, several epitopes presented by tumor cells and recognized by both CD4 + and CD8 + T cells have been identified23–26. Thus, a preexisting immune response against tumor cells expressing CCND1 may be boosted by nivolumab treatment and thus eliminate such tumor cells. More interestingly, CCND1 expression in CTCs was significantly decreased in patients with disease control after four infusions of nivolumab, suggesting that tumor-specific T cells may lyse tumor cells expressing CCND1.
The last point may indicate that the cancer stemness phenotype represents an immune-resistant phenotype of CTCs. Of patients treated with nivolumab, disease-controlled patients showed significantly increased expression of cancer stemness markers, NANOG. This finding suggests that the induction of an effective antitumor immune response by nivolumab treatment presumably allows the concentration of CTCs to display the cancer stem cell (CSC) phenotype due to lysis of non-CSC tumor cells. Although CSCs are known to be subpopulations that are capable of self-renewal and differentiation27,28, in terms of immunological characterization of CSCs, CSCs can evade immune surveillance by reducing immunogenicity and immune suppressive activity compared with their non-CSC counterparts29–32. Indeed, our previous study revealed that CD44-positive cancer stem-like cells in HNSCC produced various immune suppressive cytokines and enhanced the regulatory T-cell response compared with CD44-negative cells33. Hence, development of new treatment strategies targeting CSC-related molecules is needed to further modulate the antitumor immune responses reactivated by ICIs.
Taken collectively, ICIs can boost the immune responses against tumor cells in certain patients and exert a strong selective pressure on tumor cells in peripheral blood as well as within the tumor microenvironment. Despite the small sample size, molecular alterations in CTCs may in fact be a result of the selective pressure of antitumor immune responses induced by nivolumab. Thus, our findings suggest that the molecular profiling of CTCs may be a promising tool to predict the treatment efficacy of nivolumab and provide new insights into more precise immunotherapies.
Methods
This study was approved by the Gunma University Ethical Review Board for Medical Research Involving Human Subjects (HS2017-152), and all experiments were performed in accordance with the approved guidelines. Written informed consent was obtained from each patient.
Patients and blood collection
Patients who had histologically confirmed R/M HNSCC and treated with nivolumab were eligible for this study. We evaluated several clinical variables, including age, sex, primary site, local recurrence, lymph node metastasis, distant metastasis, performance status, previous radiotherapy, PD-L1 expression, and completion of 4 course of nivolumab. Treatment response was evaluated according to RECIST, version 1.1, during weeks 8–10. If the patients had an unacceptable level of drug-related toxic effects or rapid disease progression before evaluation, the therapeutic effects were evaluated at that point. Blood samples were collected using the K2EDTA Vacutainer (BD Bioscience) before the first nivolumab infusion and if possible, after fourth nivolumab infusion. The samples were analyzed within 4 h of collection.
Detection and gene expression analysis of CTCs
CTC detection and gene expression analysis were performed as described previously34,35. In brief, the peripheral blood mononuclear cells were isolated from blood samples (7.5 mL), and the contaminating erythrocytes were lysed with a red blood cell lysis buffer (Roche). The cell suspension was incubated with a human CD45 depletion cocktail for 15 min and then with magnetic particles for 10 min (EasySep Human CD45 Depletion Kit II, STEMCELL Technologies). Tubes containing cells were placed on a magnet for 10 min twice, and the unbound cells (CTCs) were transferred to new tubes.
Total RNA from the CTCs was extracted using an RNeasy Micro kit (Qiagen) according to the manufacturer’s instructions. cDNA synthesis was performed using the QuantiTect Reverse Transcription kit (Qiagen) with preamplification using the TaqMan PreAmp Master Mix kit (Applied Biosystems). The preamplified products were then analyzed using real-time quantitative polymerase chain reaction (Applied Biosystems) to quantify the 16 target genes. Sixteen primers for the fifteen targets (epithelial cell adhesion molecule [EPCAM], MET, keratin 19 [KRT19], epidermal growth factor receptor [EGFR], phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha [PIK3CA], cyclin D1 [CCND1], snail family transcriptional repressor 1 [SNAI1], vimentin [VIM], zinc finger E-box binding homeobox 2 [ZEB2], CD44, nanog homeobox [NANOG], aldehyde dehydrogenase 1 family member A1 [ALDH1A1], CD47, CD274, and programmed cell death 1 ligand 2 [PDCD1LG2]) and ACTB (β-actin) as control were purchased from Applied Biosystems (TaqMan Gene Expression Assays). All samples were analyzed in triplicates. Detection of at least one of the four epithelial-related genes (EPCAM, MET, KRT19, and EGFR) was defined as CTC positivity. The Ct values of the target genes were normalized to a reference gene (ACTB). The expression levels and changes of the target genes in CTCs were indicated as 40-delta Ct value and − ΔΔCt value, respectively.
The Cancer Genome Atlas (TCGA) analysis
We analyzed the relationship between CCND1 expression and mutation counts and fraction genome altered (FGA) in HNSCC using the TCGA database. The CCND1 expression Z-scores obtained based on the batch-normalized expression values from Illumina HiSeqV2, mutation count, and FGA values were downloaded from cBioPortal (https://www.cbioportal.org/study/clinicalData?id=hnsc_tcga_pan_can_atlas_2018). The cutoff value of high and low CCND1 expression was set as the median.
Statistical analysis
GraphPad Prism version 8.0 for Windows (GraphPad Software, San Diego, CA, USA) was used to perform all statistical analyses. Chi-squared test for independence and Fisher’s exact test were used to examine differences in categorical variables. The Welch’s t-test was used to examine the differences in the means between the two groups. The Kaplan–Meier curves were plotted and compared using the log-rank test. The optimal cut-off values of gene expression in CTCs for overall survival were determined based on receiver operating characteristics curve analysis. In TCGA analysis, the Welch’s t-test was used to determine the significant difference between the high CCND1 group and low CCND1 group. The Pearson correlation coefficient was used to measure the strength of a linear association between two variables, CCND1 expression level and mutation counts or FGA. Two-sided p-values < 0.05 were considered significant.
Supplementary Information
Acknowledgements
This work was supported in part by a Grant-in-Aid for Scientific Research (Grant No. 20K18243 to Hideyuki T., no. 20K09747 to M.S., no. 19K18794 to S.I., no 19K18758 to I.M., and no. 20H03834 to K.C.) from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Abbreviations
- ICI
Immune checkpoint inhibitor
- HNSCC
Head and neck squamous cell carcinoma
- R/M
Recurrent/metastatic
- PD-L1
Programmed death-ligand 1
- CTC
Circulating tumor cell
- TCGA
The Cancer Genome Atlas
- FGA
Fraction genome altered
- CSC
Cancer stem cell
Author contributions
Conception and design: Hideyuki T. and K.C., Acquisition of data: Hiroe T., Y.N., M.U., M.S., S.I., I.M., and T.M., Analysis and interpretation of data: Hiroe T., Hideyuki T., and R. K-I., Writing the manuscript: Hiroe T. and K.C. All authors have read and approved the final manuscript.
Competing interests
The authors declare no competing interest.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-020-78741-0.
References
- 1.Ferris RL, et al. Nivolumab for recurrent squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 2016;375:1856–1867. doi: 10.1056/NEJMoa1602252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cramer JD, Burtness B, Ferris RL. Immunotherapy for head and neck cancer: recent advances and future directions. Oral Oncol. 2019;99:104460. doi: 10.1016/j.oraloncology.2019.104460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kao HF, Lou PJ. Immune checkpoint inhibitors for head and neck squamous cell carcinoma: current landscape and future directions. Head Neck. 2019;41:4–18. doi: 10.1002/hed.25930. [DOI] [PubMed] [Google Scholar]
- 4.Burtness B, et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. Lancet. 2019;394:1915–1928. doi: 10.1016/S0140-6736(19)32591-7. [DOI] [PubMed] [Google Scholar]
- 5.Arora S, et al. Existing and emerging biomarkers for immune checkpoint immunotherapy in solid tumors. Adv. Ther. 2019;36:2638–2678. doi: 10.1007/s12325-019-01051-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Oliva M, et al. Immune biomarkers of response to immune-checkpoint inhibitors in head and neck squamous cell carcinoma. Ann. Oncol. 2019;30:57–67. doi: 10.1093/annonc/mdy507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xiao Q, et al. Genetic and epigenetic biomarkers of immune checkpoint blockade response. J. Clin. Med. 2020;9:286. doi: 10.3390/jcm9010286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gavrielatou N, Doumas S, Economopoulou P, Foukas PG, Psyrri A. Biomarkers for immunotherapy response in head and neck cancer. Cancer Treat. Rev. 2020;84:101977. doi: 10.1016/j.ctrv.2020.101977. [DOI] [PubMed] [Google Scholar]
- 9.Campos CDM, Jackson JM, Witek MA, Soper SA. Molecular profiling of liquid biopsy samples for precision medicine. Cancer J. 2018;24:93–103. doi: 10.1097/PPO.0000000000000311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Soda N, Rehm BHA, Sonar P, Nguyen NT, Shiddiky MJA. Advanced liquid biopsy technologies for circulating biomarker detection. J. Mater. Chem. B. 2019;7:6670–6704. doi: 10.1039/C9TB01490J. [DOI] [PubMed] [Google Scholar]
- 11.Wang J, Chang S, Li G, Sun Y. Application of liquid biopsy in precision medicine: opportunities and challenges. Front. Med. 2017;11:522–527. doi: 10.1007/s11684-017-0526-7. [DOI] [PubMed] [Google Scholar]
- 12.Tada H, Takahashi H, Kuwabara-Yokobori Y, Shino M, Chikamatsu K. Molecular profiling of circulating tumor cells predicts clinical outcome in head and neck squamous cell carcinoma. Oral Oncol. 2020;102:104558. doi: 10.1016/j.oraloncology.2019.104558. [DOI] [PubMed] [Google Scholar]
- 13.Hartmann S, Bhola NE, Grandis JR. HGF/Met signaling in head and neck cancer: impact on the tumor microenvironment. Clin. Cancer Res. 2016;22:4005–4013. doi: 10.1158/1078-0432.CCR-16-0951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lim YC, Kang HJ, Moon JH. C-Met pathway promotes self-renewal and tumorigenecity of head and neck squamous cell carcinoma stem-like cell. Oral Oncol. 2014;50:633–639. doi: 10.1016/j.oraloncology.2014.04.004. [DOI] [PubMed] [Google Scholar]
- 15.Lo Muzio L, et al. Effect of c-Met expression on survival in head and neck squamous cell carcinoma. Tumour Biol. 2006;27:115–121. doi: 10.1159/000092716. [DOI] [PubMed] [Google Scholar]
- 16.Szturz P, et al. Understanding c-MET signalling in squamous cell carcinoma of the head & neck. Crit. Rev. Oncol. Hematol. 2017;111:39–51. doi: 10.1016/j.critrevonc.2017.01.004. [DOI] [PubMed] [Google Scholar]
- 17.Saigi M, et al. MET-oncogenic and JAK2-inactivating alterations are independent factors that affect regulation of PD-L1 expression in lung cancer. Clin. Cancer Res. 2018;24:4579–4587. doi: 10.1158/1078-0432.CCR-18-0267. [DOI] [PubMed] [Google Scholar]
- 18.Wang D, et al. The hepatocyte growth factor antagonist NK4 inhibits indoleamine-2,3-dioxygenase expression via the c-Met-phosphatidylinositol 3-kinase-AKT signaling pathway. Int. J. Oncol. 2016;48:2303–2309. doi: 10.3892/ijo.2016.3486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Boschert V, et al. The influence of met receptor level on HGF-induced glycolytic reprogramming in head and neck squamous cell carcinoma. Int. J. Mol. Sci. 2020;21:471. doi: 10.3390/ijms21020471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ramos-García P, et al. Prognostic and clinicopathological significance of cyclin D1 expression in oral squamous cell carcinoma: a systematic review and meta-analysis. Oral Oncol. 2018;83:96–106. doi: 10.1016/j.oraloncology.2018.06.007. [DOI] [PubMed] [Google Scholar]
- 21.Ramos-García P, et al. Predictive value of CCND1/cyclin D1 alterations in the malignant transformation of potentially malignant head and neck disorders: systematic review and meta-analysis. Head Neck. 2019;41:3395–3407. doi: 10.1002/hed.25834. [DOI] [PubMed] [Google Scholar]
- 22.Feng Z, et al. CCND1 as a predictive biomarker of neoadjuvant chemotherapy in patients with locally advanced head and neck squamous cell carcinoma. PLoS ONE. 2011;6:e26399. doi: 10.1371/journal.pone.0026399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dengjel J, et al. Unexpected abundance of HLA class II presented peptides in primary renal cell carcinomas. Clin. Cancer Res. 2006;12:4163–4170. doi: 10.1158/1078-0432.CCR-05-2470. [DOI] [PubMed] [Google Scholar]
- 24.Dao T, et al. Identification of a human cyclin D1-derived peptide that induces human cytotoxic CD4 T cells. PLoS ONE. 2009;4:e6730. doi: 10.1371/journal.pone.0006730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dengjel J, et al. Identification of a naturally processed cyclin D1 T-helper epitope by a novel combination of HLA class II targeting and differential mass spectrometry. Eur. J. Immunol. 2004;34:3644–3651. doi: 10.1002/eji.200425510. [DOI] [PubMed] [Google Scholar]
- 26.Kondo E, et al. Cyclin D1-specific cytotoxic T lymphocytes are present in the repertoire of cancer patients: implications for cancer immunotherapy. Clin. Cancer Res. 2008;14:6574–6579. doi: 10.1158/1078-0432.CCR-08-0825. [DOI] [PubMed] [Google Scholar]
- 27.Nassar D, Blanpain C. Cancer stem cells: basic concepts and therapeutic implications. Annu. Rev. Pathol. 2016;11:47–76. doi: 10.1146/annurev-pathol-012615-044438. [DOI] [PubMed] [Google Scholar]
- 28.Nguyen LV, Vanner R, Dirks P, Eaves CJ. Cancer stem cells: an evolving concept. Nat. Rev. Cancer. 2012;12:133–143. doi: 10.1038/nrc3184. [DOI] [PubMed] [Google Scholar]
- 29.Clara JA, Monge C, Yang Y, Takebe N. Targeting signalling pathways and the immune microenvironment of cancer stem cells - a clinical update. Nat. Rev. Clin. Oncol. 2020;17:204–232. doi: 10.1038/s41571-019-0293-2. [DOI] [PubMed] [Google Scholar]
- 30.Codony-Servat J, Rosell R. Cancer stem cells and immunoresistance: clinical implications and solutions. Transl. Lung Cancer Res. 2015;4:689–703. doi: 10.3978/j.issn.2218-6751.2015.12.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Qian X, et al. Biology and immunology of cancer stem(-like) cells in head and neck cancer. Crit. Rev. Oncol. Hematol. 2015;95:337–345. doi: 10.1016/j.critrevonc.2015.03.009. [DOI] [PubMed] [Google Scholar]
- 32.Maccalli C, Volontè A, Cimminiello C, Parmiani G. Immunology of cancer stem cells in solid tumours: a review. Eur. J. Cancer. 2014;50:649–655. doi: 10.1016/j.ejca.2013.11.014. [DOI] [PubMed] [Google Scholar]
- 33.Chikamatsu K, Takahashi G, Sakakura K, Ferronse S, Masuyama K. Immunoregulatory properties of CD44+ cancer stem-like cells in squamous cell carcinoma of the head and neck. Head Neck. 2011;33:208–215. doi: 10.1002/hed.21420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chikamatsu K, et al. Expression of immune-regulatory molecules in circulating tumor cells derived from patients with head and neck squamous cell carcinoma. Oral Oncol. 2019;89:34–39. doi: 10.1016/j.oraloncology.2018.12.002. [DOI] [PubMed] [Google Scholar]
- 35.Tada H, Takahashi H, Ida S, Nagata Y, Chikamatsu K. Epithelial- mesenchymal transition status of circulating tumor cells is associated with tumor relapse in head and neck squamous cell carcinoma. Anticancer Res. 2020;40:3559–3564. doi: 10.21873/anticanres.14345. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.