Abstract
Background: The coronavirus disease 2019 (COVID-19) pandemic has deeply modified the complex logistical process underlying allogeneic hematopoietic stem cell transplant practices. Aim: In light of these changes, the authors compared data relative to allogeneic transplants carried out from 2018 at their center before (n = 167) and during the pandemic (n = 45). Methods: The authors examined patient characteristics, donor and graft types, cell doses and main transplant outcomes. Moreover, the authors evaluated the rise of costs attributable to additional COVID-19-related procedures as well as the risk of adverse events these procedures conveyed to grafts or recipients. Results: Overall, the number of transplants did not decrease during the pandemic, whereas patients at high relapse risk were prioritized. Transplants were mainly from matched unrelated donors, with a significant decrease in haploidentical related donors. Moreover, the use of bone marrow as a graft for haploidentical transplant was almost abandoned. Cryopreservation was introduced for all related and unrelated apheresis products, with a median storage time of 20 days. Notably, transplant outcomes (engraftment, acute graft-versus-host disease and non-relapse mortality) with cryopreserved products were comparable to those with fresh products. Conclusions: Considering that the emergency situation may persist for months, cryopreserving allogeneic grafts can offer a lifesaving opportunity for patients whose allogeneic transplant cannot be postponed until after the end of the COVID-19 pandemic.
Key Words: allogeneic hematopoietic stem cell transplantation, costs, COVID-19 pandemic, cryopreservation
Introduction
The emergence of the coronavirus disease 2019 (COVID-19) pandemic has triggered an exceptional worldwide health care crisis. Italy was among the first nations in Europe where COVID-19 broke out. On January 27, 2020, the National Transplant Center in Italy issued the first warning about severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection and hematopoietic stem cell (HSC) donation [1]. On February 24, 2020, the requirement for SARS-CoV-2 donor screening was introduced, first in COVID-19 high-risk districts and a few days later in the entire national area [2]. Since March 13, 2020, Italian transplant centers have been strongly recommended by the competent authority to start the conditioning regimen in recipients only after HSC products from either related or unrelated donors have been delivered and cryopreserved [3]. The same recommendations were issued by the European Society for Blood and Marrow Transplantation [4].
Altogether, the COVID-19 pandemic has deeply modified the complex allograft logistical process at multiple stages. On the one hand, it is conceivable that travel restrictions within and across countries, as well as the disease itself or the quarantine following SARS-CoV-2 exposure, could make it more difficult to access HSC donors, either related or unrelated. On the other hand, the graft collection that before the pandemic was strictly organized depending on the conditioning regimen of the recipient has been managed with a more flexible approach thanks to cryopreservation [5]. Because of concerns regarding the negative impact of freezing and thawing on the viability of HSCs, until now the authors have infused allogeneic HSC products soon after collection, whereas only a minimal proportion of grafts have been cryopreserved.
Because of all these considerations, the authors revised data to reflect the effect of the ongoing COVID-19 pandemic on their allogeneic transplant practices. To this end, the authors compared the number and types of allograft cryopreservation procedures carried out in the pre-pandemic and pandemic periods. The authors examined cell doses and main transplant outcomes in patients receiving cryopreserved grafts. Moreover, the authors evaluated the rise of costs attributable to additional COVID-19-related procedures as well as the risk of adverse events these procedures conveyed to grafts or recipients.
Methods
Study population
All allogeneic HSC transplantations performed at the hematology transplant unit of Fondazione Policlinico A. Gemelli IRCCS of Rome (Italy) between January 1, 2018, and August 31, 2020, were retrospectively investigated. Transplants of cord blood units were not included in the study. The authors included in the COVID-19 period all transplants carried out after the first Italian transplant center warning (January 27, 2020) [1]. The ethics committee of Fondazione Policlinico A. Gemelli IRCCS approved the study (protocol 0030921/20).
Donor, recipient and graft variables
Donor variables included demographics, type of donation (related and unrelated), country of origin (if unrelated donors) and ABO match. Patient variables included demographics, diagnosis, status of disease at transplant (complete remission or not), disease risk index [6], hematopoietic cell transplantation comorbidity index (HCT-CI) [7], incidence of acute graft-versus-host disease (aGVHD) [8] and death. Graft variables were source (bone marrow [BM] or peripheral blood stem cells [PBSCs]), total nucleated cell (TNC) content, CD34+ cell content, CD3+ cell content and CD34+ cell viability assessed either fresh or fresh and post-freezing in cryopreserved products. Cell contents were expressed as cell dose (i.e., number of cells per kg of recipient's body weight) and were obtained as previously reported [9].
Study endpoints and definitions
The authors compared characteristics of donors (demographics and country), recipients (demographics and disease-related data) and grafts (PBSCs or BM) in the different transplant periods (COVID-19 or pre-COVID-19). In transplants performed with fresh or cryopreserved grafts, the authors analyzed the following endpoints: TNC and CD34+ cell doses, cumulative incidence of neutrophil engraftment (i.e., day of achievement of an absolute neutrophil count ≥0.5 × 109/L), cumulative incidence of platelet engraftment (i.e., day of achievement of a platelet count ≥20 × 109/L unsupported by transfusion), reticulocyte recovery (i.e., reticulocyte count >2.7%), cumulative incidence of aGVHD (grades II–IV) and non-relapse mortality (NRM) (i.e., death without prior relapse). Adverse events/reactions were defined according to Directive 2004/23/EC [10]. The cost evaluation was conducted considering direct and indirect costs for all activities in the cryopreservation process and calculating the mean values of procedures carried out along with COVID-19-related measures.
Statistical analysis
Continuous variables were expressed as median with relative interquartile range (IQR) and categorical variables as n (%). Univariate analysis for continuous and categorical variables was performed using the Mann–Whitney U test and Fisher exact test as appropriate. Cumulative incidence of neutrophil and platelet engraftment, reticulocyte recovery and aGVHD was calculated with death in the absence of investigated events as competing risk. NRM was calculated with relapse as a competing risk event. A comparison between curves was performed according to the Grey method and expressed as hazard ratio with 95% confidence interval (CI). P< 0.05 was considered statistically significant. Analyses were performed using IBM SPSS Statistics 25.0 and NCSS 10 v 10.0.19.
Results
A total of 212 consecutive transplants from adult donors were included in the study. Although there was no difference regarding age, sex or diagnosis, patients transplanted during COVID-19 showed higher disease risk index and lower HCT-CI scores (Table 1 ). SARS-CoV-2 screening by polymerase chain reaction on nasopharyngeal swabs (with or without serology) was performed in all donors from March 2020 onward. Thirty-five (19 unrelated and 16 related) donors were screened. No positive donors were found.
Table 1.
Characteristics of patients.
| Pre-COVID n = 167 | COVID n = 45 | P value | |
|---|---|---|---|
| Age, years | 56.4 (45.1–62.4) | 49.4 (38.0–60.7) | 0.1437 |
| Male sex | 103 (61.7) | 22 (48.9) | 0.1283 |
| Diagnosis | |||
| Acute myeloid leukemia | 68 (40.7) | 23 (51.1) | |
| Chronic myeloproliferative neoplasms | 35 (21.0) | 7 (15.6) | |
| Acute lymphoblastic leukemia | 21 (12.6) | 8 (17.8) | 0.6176 |
| Myelodysplastic syndrome | 18 (10.8) | 3 (6.7) | |
| Hodgkin/non-Hodgkin lymphoma | 18 (10.8) | 3 (6.7) | |
| Severe aplastic anemia | 3 (1.8) | 1 (2.2) | |
| Multiple myeloma/plasma cell leukemia | 4 (2.4) | 0 (0.0) | |
| High/very high DRI score | 46 (27.5) | 21 (46.7) | 0.0187 |
| Complete remission at transplant | 78 (46.7) | 28 (62.2) | 0.0923 |
| HCT-CI score >2 | 105 (62.9) | 18 (40.0) | 0.0067 |
| Haploidentical transplant | 66 (39.5) | 7 (15.5) | 0.0025 |
| Conditioning regimen | |||
| Myeloablative | 61 (36.5) | 10 (22.2) | 0.0775 |
| Non-myeloablative | 106 (63.5) | 35 (77.8) | |
| Major/bidirectional ABO mismatch | 47 (28.1) | 11 (24.4) | 0.7084 |
Data are expressed as n (%) or median (IQR).
DRI, disease risk index. HCT-CI, hematopoietic cell transplantation-comorbidity index.
Transplant distribution before and during the COVID-19 period
Graft and donor characteristics of transplants performed before and during the COVID-19 pandemic are shown in Table 2 . Allogeneic HSC transplant activities at the authors’ center were not slowed by the pandemic, varying from a mean of 6.8 to 5.6 transplants per month (P = 0.7334). The authors observed a relative increase in transplants from unrelated donors (from 34.7% to 55.6%, P = 0.0155). Compared with the previous period, during the pandemic, the proportion of donations from European countries (96.0% versus 70.7%, P = 0.0103) increased. Overall, fewer transplants from haploidentical familial donors were performed (from 39.5% to 15.5%, P = 0.0025). This resulted from the drastic decrease in BM harvesting procedures usually performed at the authors’ center in the pre-COVID-19 era (from 41.3% to 11.1% of all grafts, P< 0.0001). In addition, haploidentical transplants before the pandemic were frequently carried out in patients with chronic myeloproliferative neoplasms, lymphoma or multiple myeloma. By contrast, during the pandemic, transplants in patients with chronic disease were postponed whenever possible (Table 1). As a result, six out of seven patients (85.7%) receiving a haploidentical transplant during the pandemic were affected by acute myeloid leukemia. In addition, PBSC instead of BM grafts were harvested from haploidentical donors to secure transplant in more urgent cases of patients with high-risk disease. Thirty-two out of 212 grafts underwent cryopreservation and storage before transplant, with a median storage duration of 20 days (IQR, 12–26); all of these were PBSC grafts, and 31 had been collected during the pandemic (Table 2).
Table 2.
Characteristics of donors and grafts.
| Pre-COVID (n = 167) | Missing data | COVID n = 45 | Missing data | P value | |
|---|---|---|---|---|---|
| Donors | |||||
| Age, years | 34.5 (26.2–45.1) | 6 (3.6) | 37 (30.3–47.3) | 2 (4.4) | 0.3185 |
| Male | 113 (68.9) | 3 (1.8) | 33 (72.7) | – | 0.2404 |
| Body weight (kg) | 75 (64-85) | 5 (2.9) | 76 (70.2-88.0) | 1 (2.2) | 0.2182 |
| Related | 109 (65.3) | – | 20 (44.4) | – | 0.0155 |
| Unrelated | 58 (34.7) | 25 (55.6) | |||
| Unrelated donors | – | – | 0.0258 | ||
| Italy Other | 10 (15.3) | 7 (22.6) | |||
| Europe | 38 (58.5) | 23 (74.2) | |||
| Non-Europe | 17 (26.2) | 1 (3.2) | |||
| Grafts | |||||
| PBSC | 98 (58.7) | – | 40 (88.9) | – | < 0.0001 |
| BM | 69 (41.3) | 5 (11.1) | |||
| Fresh | 166 (99.0) | – | 14 (31.1) | – | < 0.0001 |
| Cryopreserveda | 1 (1.0) | 31 (68.9) | |||
Data are expressed as n (%) or median (IQR).
DRI, disease risk index.
No BM grafts were cryopreserved.
Effect of cryopreservation on transplant outcomes
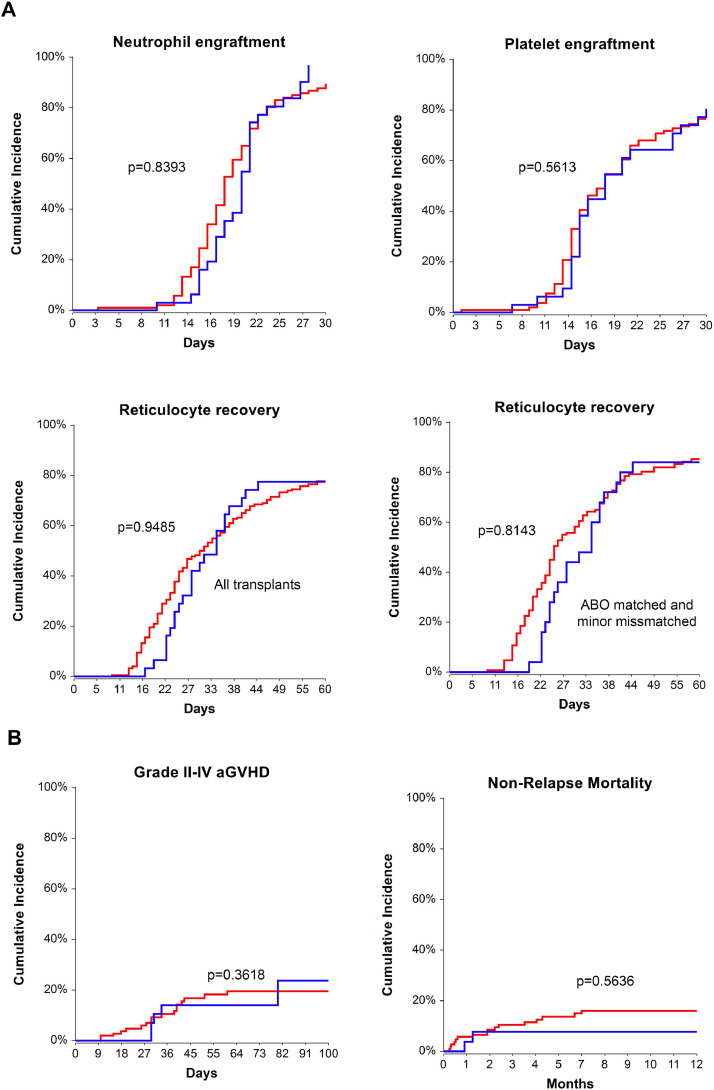
The authors included in the analysis the 31 PBSC grafts cryopreserved from March 2020 onward and one related PBSC product cryopreserved in 2019 to bring forward upon donor request. Table 3 shows patient and graft characteristics of allogeneic transplants performed with fresh or cryopreserved products. Donor- and recipient-related variables were similar in fresh and cryopreserved products. Although patients transplanted with cryopreserved products seemed to be given slightly lower TNC doses than those infused with fresh products, they received similar CD34+ cell doses. Cryopreservation resulted in a slight loss of viable CD34+ cells, decreasing from 7.1 × 106 cells/kg (IQR, 5.4–8.8) to 6.2 × 106 cells/kg (IQR, 4.9–8.2) (P< 0.0001), as evaluated based on CD34+ cell viability at thawing. Despite this, fresh and cryopreserved grafts contained similar CD34+ cell doses. The estimated cumulative incidence of engraftment at day +30 in patients in the fresh and cryopreserved groups was 89.6% (95% CI, 84.0–95.6) and 96.7% (95% CI, 90.7–100) for neutrophils (P= 0.8393) and 76.4% (95% CI, 68.7–84.9) and 80.4% (95% CI, 67.6–95.7) for platelets (P= 0.5613), respectively. Similarly, there was no difference regarding reticulocyte recovery (P= 0.6101) (Figure 1 A). Seventeen patients in the fresh group and five patients in the cryopreserved group developed grade II–IV aGVHD. The day +100 cumulative incidence of grade II–IV aGVHD in the fresh and cryopreserved groups was 19.5% (95% CI, 12.6–30.1) and 23.8% (95% CI, 9.7–58.5) (P= 0.3618), respectively (Figure 1B). Finally, the 1-year NRM was 16.1% (95% CI, 10.2–25.3) in patients receiving fresh grafts and 7.7% (95% CI, 2.0–29.8) in those receiving cryopreserved grafts (P= 0.5636) (Figure 1B).
Table 3.
Donor, recipient and graft characteristics of transplants performed with fresh or cryopreserved PBSC products.
| Fresh | Cryopreserved | P value | |
|---|---|---|---|
| Patients | 106 | 32 | |
| Age, years | 54.6 (43.5–60.6) | 51.5 (41.8–59.0) | 0.4492 |
| Male sex | 65 (61.3) | 16 (50.0) | 0.3071 |
| Diagnosis | |||
| Acute myeloid leukemia | 46 (43.4) | 15 (46.9) | 0.5076 |
| Chronic myeloproliferative neoplasms | 20 (18.9) | 6 (18.7) | |
| Acute lymphoblastic leukemia | 12 (11.3) | 7 (21.9) | |
| Myelodysplastic syndromes | 13 (12.3) | 2 (6.2) | |
| Hodgkin/non-Hodgkin lymphoma | 13 (12.3) | 2 (6.2) | |
| Multiple myeloma/plasma cell leukemia | 2 (1.9) | 0 (0.0) | |
| Myeloablative conditioning regimen | 39 (36.8) | 9 (28.1) | 0.4049 |
| High/very high DRI score | 22 (20.7) | 14 (66.7) | 0.0022 |
| Complete remission | 58 (54.7) | 18 (56.2) | 0.0923 |
| HCT-CI score >2 | 54 (50.9) | 7 (33.3) | 0.1585 |
| Donor age, years | 34.5 (24.9–45.7)* | 39.2 (30.2–50.0)** | 0.0960 |
| Female donor | 29 (27.9) *** | 11 (34.4) | 0.5101 |
| Haploidentical related donor | 0 | 3 (9.4) | 0.0115 |
| Major/bidirectional ABO mismatch | 31 (29.2) | 7 (21.9) | 0.7084 |
| TNC, 108 cells/kg | 8.7 (6.7–10.5) | 7.6 (6.3–8.9) | 0.0264 |
| CD34+ cells, 106 cells/kga | 7.1 (5.3–9.2) | 7.1 (5.4–8.8) | 0.9594 |
| CD3+ cells, 106 cells/kg | 207.0 (151.7–269.0) | 193.7 (144.0–276.4) | 0.8996 |
| Post-thaw CD34+ cells, 106 cells/kgb | – | 6.2 (4.9–8.2) | 0.1859 |
Data are expressed as n (%) or median (IQR).
Missing data in five donors (4.7%).
Missing data in one donor (3.0%).
Missing data in two donors (1.8 %).
At collection.
Post-thaw CD34+ cell doses are compared with CD34+ cell doses infused with fresh products.
DRI, disease risk index. HCT-CI, hematopoietic cell transplantation-comorbidity index.
Figure 1.
Transplant outcomes in patients receiving fresh (red lines) or cryopreserved (blue lines) products. (A) Cumulative incidence of neutrophil, platelet and reticulocyte engraftment. Reticulocyte engraftment is separately reported in total population and ABO matched/minor mismatched patients. (B) Cumulative incidence of grade II–IV acute GVHD and NRM.
Adverse events
During the pandemic, the authors recorded microbial culture positivity for a skin contaminant in one cryopreserved PBSC product. Among all products collected before the pandemic, only BM harvests (three grafts that were transfused fresh) had harbored similar microbial contamination. In addition, during the pandemic, the authors documented one adverse event due to a malfunction in controlled rate cooling during the freezing of one unrelated PBSC unit. This was the result of the failure of the thermocouple probe to correctly detect the cooling of the sample bag and caused an inability to retrieve the record of the temperature curve, whereas chamber cooling went on according to the defined program temperature curve. Therefore, the failure of the probe did not affect product viability. No adverse reactions related to the infusion of cryopreserved grafts were recorded.
Additional costs
Cryopreservation as a result of COVID-19-related measures resulted in additional costs due to the procedure itself, microbial cultures and viability tests performed on cryopreserved units before release. Table 4 shows direct costs the authors calculated based on mean technician hours worked and consumable and disposable requirements for one cryopreservation. Overall, every cryopreservation resulted in an additional cost of €382.62, corresponding to an overall expense of €12,244.
Table 4.
Costs for cryopreservation.
| n | Unitary cost | Cost per procedure | |
|---|---|---|---|
| Personnel, hours worked | 5 | 26 | 130 |
| Consumables and reagents | |||
| Transfer bag | 2 | 1.64 | 3.28 |
| Cryobag | 2.5 | 26.00 | 65.00 |
| DMSO, 10 mL | 2.5 | 4.00 | 10.00 |
| 5% albumin (100 mL) | 1.3 | 26.11 | 33.94 |
| Cryovial | 2 | 0.30 | 0.60 |
| Syringe, 60 mL | 4 | 0.25 | 1.00 |
| Syringe, 5 mL | 1 | 0.06 | 0.06 |
| Spike | 1 | 0.99 | 0.99 |
| Liquid nitrogen (L) | 1 | 8.00 | 8.00 |
| Testing at release | |||
| Microbial culture | 1 | 14.70 | 14.70 |
| Viability test | 1 | 51.28 | 51.28 |
| Total | 318.85 | ||
| Indirect costs (20%) | 63.77 | ||
| Total cost per product | 382.62 |
Costs are expressed in euros. For consumables and reagents, the mean number was calculated based on the amount utilized to cryopreserve 32 overall PBSC products.
DMSO, dimethyl sulfoxide.
Discussion
The COVID-19 pandemic has caused the transplantation community to face exceptional challenges to avoid postponing this life-saving procedure while at the same time ensuring the absolute safety of donors and recipients. In this study, the authors reviewed data relative to allogeneic HSC transplantations performed at their center in the pandemic period to understand how ourtheir daily practices have changed to fulfill the requirements presented by the COVID-19 pandemic.
Despite the increased complexity of the whole process of donor procurement, workout and graft harvesting, the sprouting of the pandemic has not been associated with a decline in allogeneic transplants at the authors’ institution. The authors’ data emphasize that during the pandemic patients at higher relapse risk have been given priority. Likewise, increased COVID-19 mortality in people with multiple comorbidities [11] has led to the prioritization of patients with lower HCT-CI scores. A similar approach has been reported by the Seattle transplant group [12]. The authors developed an algorithm guiding the temporary deferral of transplants deemed non-urgent based on the risk of the underlying malignancy. Indeed, patients have been prioritized according to the risks of recurrence or progression of the malignancy and burden of comorbidity [12].
Overall, the pandemic has significantly influenced the criteria adopted to select donors and has modified the types of stem cell sources. Paralleling the lockdown measures progressively introduced in Italy from mid-February 2020, transplants from related donors have decreased in favor of those from unrelated donors. Multiple reasons can account for this finding. Before the pandemic, the authors used to perform transplants from haploidentical donors using BM as a stem cell source because of the concern regarding the higher incidence of acute and chronic GVHD reported in PBSC haploidentical transplants [13], [14], [15]. Nevertheless, at the onset of the pandemic, the authors’ hospital in Rome was designated a dedicated COVID-19 center, leading to the rearrangement of many departments and the establishment of COVID-19-dedicated pathways, including surgery rooms. Therefore, BM harvesting has been made problematic by increased difficulties in logistics surrounding donor hospital admission, surgery room availability and management of COVID-19-related infectious risk. Moreover, fewer patients with chronic diseases, who were frequent candidates for haploidentical transplant before the pandemic, have received transplantation during the COVID-19 period. Consequently, priority has been given to HLA-matched related and unrelated donors, whereas for patients with no registry donor, haploidentical transplants have been performed using PBSCs as a graft. Indeed, registry donors have represented for the authors’ patients a highly reliable resource, even more so in the pandemic period. Border closures and difficulties related to flight cancellations have prompted the tendency to select donors from European registries, allowing for the organizing of last-minute product transport on wheels.
The most remarkable change in the authors’ practices as a consequence of the COVID-19 pandemic has been to extend cryopreservation to allogeneic PBSC grafts. Until the pandemic, the authors cryopreserved only autologous products unless it was impossible to postpone donation for exceptional reasons related to donors or recipients. Cryopreservation was first introduced to prevent possible delays in unrelated product delivery, whereas it was rapidly adopted for related grafts. In fact, it has allowed more comfortable management of recipient hospital admissions as well as post-donation donor follow-up.
One of the main concerns related to cryopreservation is represented by the loss of viable cells due to unit processing or storage [16,17]. This risk may be exacerbated if products are cryopreserved at cell concentrations greater than 2 × 108cells/mL or if they contain a large amount of granulocytes [18], [19], [20]. In this study, the authors confirmed that a portion of viable CD34+ cells are lost with cryopreservation. Nevertheless, it should be emphasized that the authors’ data were achieved using a single satellite vial, and the results were not confirmed using attached segments. In addition, the authors estimated CD34+ cell viability but not CD34+ cell potency since we did not carry out a colony-forming unit assay on the samples. Despite these limitations, the loss was negligible and did not significantly affect either the CD34+ cell dose or the hematopoietic recovery. Thus, the authors’ data confirm previous observations of allogeneic transplants performed with cryopreserved products, suggesting that this is a safe and feasible procedure [21], [22], [23], [24], [25], [26]. In fact, previous studies comparing transplant outcomes in patients receiving fresh or cryopreserved grafts globally back cryopreservation in the allogeneic scenario [21], [22], [23], [24], [25], [26]. Nonetheless, this might not be true in every clinical setting. In particular, Eapen et al. [27] have recently reported an increased incidence of graft failure and 1-year mortality in severe aplastic anemia patients receiving cryopreserved grafts. Given the short follow-up of patients transplanted with cryopreserved grafts at the authors’ center, they could explore only short-term transplant outcomes. Overall, the authors’ data do not show a difference regarding the incidence of engraftment, aGVHD and NRM in comparison with patients receiving fresh grafts, whereas longer follow-up is necessary to assess whether cryopreserved and fresh transplants elicit superimposable results with regard to chronic GVHD and disease-free and overall survival.
From an ethics perspective, the most important concern raised regarding cryopreservation is that, in exceptional circumstances related to the recipient (i.e., sudden worsening or death), the harvested product might not be transplanted [16,24]. In the present study, the authors found that all cryopreserved products were transfused, and the median storage duration was 20 days. This finding is in agreement with the high relapse risk of the authors’ patients and shows that, at least in our their experience, graft collection is strictly organized to perform the transplant, whereas cryopreservation allows for flexible management of the recipient's hospital admission.
The policy of regularly cryopreserving allogeneic grafts undoubtedly results in additional expenses for the hospital. Considering the disease-related group reimbursement operated by the Italian National Health System for public hospitals, these costs can be considered marginal. Nonetheless, cryopreservation also implies additional risks related to unit processing, such as the risk of bag damage, possible microbial contamination and infusion reactions from dimethyl sulfoxide [16,24]. The authors did not observe adverse reactions among patients receiving cryopreserved products, but we experienced failure of the controlled rate cooling freezer during the processing of one unrelated PBSC unit. Fortunately, in the authors’ case, the failure did not affect product quality. Nevertheless, this episode suggests that both donors and recipients should be made aware of these cryopreservation drawbacks, possibly requiring arrangement of further donations. The authors also recorded microbial contamination of one PBSC product, likely occurring during processing. Notably, before the pandemic, the authors had to cope with similar problems only with BM grafts, whose contamination likely occurred during the harvesting procedure.
Conclusions
The authors’ experience with regard to allogeneic transplants with cryopreserved products suggests that this is a feasible and safe option, allowing compliance with all precautions imposed by the pandemic [24]. After the transient pandemic control during the summer of 2020, several donor registries returned to normal procedures, and freezing products was no longer recommended considering several reports of cryopreserved products never being infused [17]. Presently, however, donors in many countries face a high risk of SARS-CoV-2 infection between workup and collection, and pre-planned cryopreservation is recommended [28]. Considering that this emergency situation may persist for months, cryopreserving allogeneic grafts might offer a lifesaving opportunity for patients whose allogeneic transplant cannot be postponed until after the end of the COVID-19 pandemic.
Declaration of Competing Interest
The authors have no commercial, proprietary or financial interest in the products or companies described in this article.
Acknowledgments
Funding
No funding was received.
Author Contributions
Conception and design of the study: CGV, PC, MB and LT. Acquisition of data: CGV, PC, MB, LT, NO, SG and EM. Analysis and interpretation of data: CGV, PC, MB, LT, NO, SG and EM. Drafting or revising the manuscript: CGV, PC, MB, LT, AB and SS. All authors have approved the final article.
Acknowledgments
The authors are in debt to Claudia Frau for her valuable work in managing relations with patients, donor registry and familial donors.
References
- 1.http://www.trapianti.salute.gov.it/imgs/C_17_cntAvvisi_227_0_file.pdf (last accessed on November 16, 2020).
- 2.http://www.trapianti.salute.gov.it/imgs/C_17_cntAvvisi_225_0_file.pdf (last accessed on November 16, 2020).
- 3.http://www.trapianti.salute.gov.it/imgs/C_17_cntAvvisi_236_0_file.pdf (last accessed on November 16, 2020).
- 4.Ljungman P, Mikulska M, de la Camara R, Basak GW, Chabannon C, Corbacioglu S. Correction: The challenge of COVID-19 and hematopoietic cell transplantation: EBMT recommendations for management of hematopoietic cell transplant recipients, their donors, and patients undergoing CAR T-cell therapy. Bone Marrow Transplant. 2020;8:1. doi: 10.1038/s41409-020-0919-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Algwaiz G, Aljurf M, Koh M, Horowitz MM, Ljungman P, Weisdorf D. Real-World Issues and Potential Solutions in Hematopoietic Cell Transplantation during the COVID-19 Pandemic: Perspectives from the Worldwide Network for Blood and Marrow Transplantation and Center for International Blood and Marrow Transplant Research Health Services and International Studies Committee. Biol Blood Marrow Transplant. 2020;26(12):2181–2189. doi: 10.1016/j.bbmt.2020.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123(23):3664–3671. doi: 10.1182/blood-2014-01-552984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–2919. doi: 10.1182/blood-2005-05-2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant. 1995 Jun;15(6):825–828. [PubMed] [Google Scholar]
- 9.Teofili L, Chiusolo P, Valentini CG, Metafuni E, Bellesi S, Orlando N. Bone marrow haploidentical transplant with post-transplantation cyclophosphamide: does graft cell content have an impact on main clinical outcomes? Cytotherapy. 2020;22(3):158–165. doi: 10.1016/j.jcyt.2020.01.007. [DOI] [PubMed] [Google Scholar]
- 10.https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32004L0023 (last accessed on November 16, 2020).
- 11.Emami A, Javanmardi F, Pirbonyeh N, Akbari A. Prevalence of Underlying Diseases in Hospitalized Patients with COVID-19: a Systematic Review and Meta-Analysis. Arch Acad Emerg Med. 2020;8(1):e35. [PMC free article] [PubMed] [Google Scholar]
- 12.Ueda Oshima M, Sandmaier BM, Petersdorf E, Flowers ME, Hill GR, Lee SJ. Blood and marrow transplantation during the emerging COVID-19 pandemic: the Seattle approach. Bone Marrow Transplant. 2020;26:1–9. doi: 10.1038/s41409-020-01068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bashey A, Zhang MJ, McCurdy SR, St Martin A, Argall T, Anasetti C. Mobilized Peripheral Blood Stem Cells Versus Unstimulated Bone Marrow As a Graft Source forT-Cell-Replete Haploidentical Donor Transplantation Using Post-Transplant Cyclophosphamide. J Clin Oncol. 2017;10(35(26)):3002–3009. doi: 10.1200/JCO.2017.72.8428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ruggeri A, Labopin M, Bacigalupo A, Gülbas Z, Koc Y, Blaise D. Bone marrow versus mobilized peripheral blood stem cells in haploidentical transplants using posttransplantation cyclophosphamide. Cancer. 2018;124(7):1428–1437. doi: 10.1002/cncr.31228. [DOI] [PubMed] [Google Scholar]
- 15.Mussetti A, De Philippis C, Carniti C, Bastos-Oreiro M, Gayoso J, Cieri N. CD3+ graft cell count influence on chronic GVHD in haploidentical allogeneic transplantation using post-transplant cyclophosphamide. Bone Marrow Transplant. 2018;53(12):1522–1531. doi: 10.1038/s41409-018-0183-8. [DOI] [PubMed] [Google Scholar]
- 16.Dholaria B, Malki MMA, Artz A, Savani BN. Securing the graft during pandemic: are we ready for cryopreservation for all? Biol Blood Marrow Transplant. 2020;26(7):e145–e146. doi: 10.1016/j.bbmt.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.https://wmda.info/wp-content/uploads/2020/07/20200611-SEAR-Rapid-Alert.pdf (last accessed on November 14, 2020).
- 18.Félix OMWO, Tunes G, Ginani VC, Simões PC, Barros DP, Delbuono E. The influence of cell concentration at cryopreservation on neutrophil engraftment after autologous peripheral blood stem cell transplantation. Hematol Transfus Cell Ther. 2018;40(3):233–239. doi: 10.1016/j.htct.2018.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Castelhano MV, Reis-Alves SC, Vigorito AC, Rocha FF, Pereira-Cunha FG, De Souza CA. Quantifying loss of CD34+ cells collected by apheresis after processing for freezing and post-thaw. Transfus Apher Sci. 2013;48(2):241–246. doi: 10.1016/j.transci.2013.01.017. [DOI] [PubMed] [Google Scholar]
- 20.Valentini CG, Bianchi M, Orlando N, Autore F, Iachininoto MG, Piccirillo N. Graft Composition and Post-Thawing Cell Viability Influence the Hematopoietic Recovery in Autologous Hematopoietic Stem Cell Transplantation. J Stem Cell Res Ther. 2017;7:379. [Google Scholar]
- 21.Frey NV, Lazarus HM, Goldstein SC. Has allogeneic stem cell cryopreservation been given the 'cold shoulder'? An analysis of the pros and cons of using frozen versus fresh stem cell products in allogeneic stem cell transplantation. Bone Marrow Transplant. 2006;38(6):399–405. doi: 10.1038/sj.bmt.1705462. [DOI] [PubMed] [Google Scholar]
- 22.Kim DH, Jamal N, Saragosa R, Loach D, Wright J, Gupta V. Similar outcomes of cryopreserved allogeneic peripheral stem cell transplants (PBSCT) compared to fresh allografts. Biol Blood Marrow Transplant. 2007;13(10):1233–1243. doi: 10.1016/j.bbmt.2007.07.003. [DOI] [PubMed] [Google Scholar]
- 23.Hamadani M, Zhang MJ, Tang XY, Fei M, Brunstein C, Chhabra S. Graft Cryopreservation Does Not Impact Overall Survival after Allogeneic Hematopoietic Cell Transplantation Using Post-Transplantation Cyclophosphamide for Graft-versus-Host Disease Prophylaxis. Biol Blood Marrow Transplant. 2020;26(7):1312–1317. doi: 10.1016/j.bbmt.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schmidt AH, Buk D, Platz A, van den Brink MRM, Cryopreservation for All Is No Option in Unrelated Stem Cell Transplantation. Comment on Dholaria B Securing the Graft During Pandemic: Are We Ready for Cryopreservation for All? Biol Blood Marrow Transplant. 2020;26:e145–e146. doi: 10.1016/j.bbmt.2020.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Purtill D, Antonenas V, Chiappini P, Tong D, O'Flaherty E, Bajel A. Variable CD34+ recovery of cryopreserved allogeneic HPC products: transplant implications during the COVID-19 pandemic. Blood Adv. 2020;4(17):4147–4150. doi: 10.1182/bloodadvances.2020002431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Medd P, Nagra S, Hollyman D, Craddock C, Malladi R. Cryopreservation of allogeneic PBSC from related and unrelated donors is associated with delayed platelet engraftment but has no impact on survival. Bone Marrow Transplant. 2013;48(2):243–248. doi: 10.1038/bmt.2012.118. [DOI] [PubMed] [Google Scholar]
- 27.Eapen M, Zhang MJ, Tang XY, Lee SJ, Fei MW, Wang HL. Hematopoietic Cell Transplantation with Cryopreserved Grafts for Severe Aplastic Anemia. Biol Blood Marrow Transplant. 2020;26(7):e161–e166. doi: 10.1016/j.bbmt.2020.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.https://www.ebmt.org/sites/default/files/2020-11/EBMT%20COVID-19%20guidelines%20v11.3.pdf (last accessed on November 14, 2020).