Abstract
Background
Anthropometric factors are reported to be risk factors for atrial fibrillation (AF), but it is unclear whether weight change in mid‐life is associated with AF. We aimed to study the possible associations of weight, height, and weight change with the risk of incident AF in men and women.
Methods
Our study cohort included 108 417 persons (51% women) who participated in a population‐based health examination in northern Sweden at 30, 40, 50, or 60 years of age. The health examination included weight and height measurement and collection of data regarding cardiovascular risk factors. Within this cohort, 40 275 participants underwent two health examinations with a 10‐year interval. We identified cases with a first‐ever diagnosis of AF through the Swedish National Patient Registry.
Results
During a total follow‐up of 1 469 820 person‐years, 5154 participants developed incident AF. The mean age at inclusion was 46.3 years, and mean age at AF diagnosis was 66.6 years. After adjustment for potential confounders, height, weight, body mass index (BMI), and body surface area (BSA) were positively associated with risk of incident AF in both men and women. Among participants who underwent two health examinations 10 years apart, 1142 persons developed AF. The mean weight change from baseline was a gain of 4.8%. Weight gain or weight loss was not significantly associated with risk of incident AF.
Conclusions
Height, weight, BMI, and BSA showed positive associations with risk of incident AF in both men and women. Midlife weight change was not significantly associated with AF risk.
Keywords: atrial fibrillation, body height, body weight, weight gain, weight loss
In a prospecitve population‐based cohort study height, weight, BMI, and BSA showed positive associations with risk of incident AF in both men and women. Midlife weight change was not significantly associated with AF risk.

1. INTRODUCTION
Atrial fibrillation (AF) is the most common clinically significant arrhythmia, with a 3% prevalence and an incidence of 4 cases per 1000 person‐years. 1 , 2 Weight, height, body mass index (BMI), and body surface area (BSA) are reportedly associated with left atrial size and thus with an increased risk of AF. 3 , 4 , 5 One study has suggested a causal association between obesity and AF risk. 6 In contrast, other evidence indicates that the association between anthropometric measures and AF risk is predominantly driven by lean body mass. 4 Men and women differ with regards to height, weight, body composition, and the relative importance of various cardiovascular disease risk factors; therefore, it is important to study AF risk factors separately in men and women. 7 Investigating men and women from the same source population facilitates comparison.
There are some studies of the association between weight change and the risk of incident AF. Observational studies have demonstrated that weight loss is associated with increased 8 or decreased AF risk, 3 or not associated with AF risk at all. 9 , 10 , 11 . An American study showed an association between weight change of >5% of the body weight and increased risk of AF 8 and an Australian study found an association between both increase and decrease of BMI and increased risk of AF in women, but not in men. 9 In studies where only men were included, weight increase was associated with an increased risk of AF 12 , 13 or with an increased risk of AF in men with a low physical fitness but not in a men with a high physical fitness. 14 In a study that included women only, there was an association between BMI gain and increased risk of AF. 10 In overweight or obese individuals with type 2 diabetes, a lifestyle intervention that induced modest weight loss did not reduce the risk of incident AF. 15 On the other hand, an Israeli observational study demonstrated that weight loss was associated with decreased AF risk, regardless of baseline weight. 3 Similarly, in overweight and obese individuals with symptomatic AF, weight loss has been associated with a dose‐dependent long‐term freedom from AF. 16
Weight change between early adulthood and midlife and during midlife may have different effects on AF risk. Some studies report that weight gain between early adult life and midlife is associated with an increased risk of AF. 12 , 13 , 14 An American study found that weight gain during midlife was associated with an increased AF risk in men, but not in women. 8 If weight loss during midlife can reduce the risk of incident AF, this could be an important target for primary prevention efforts.
The aims of the study were to investigate the associations between weight and height and the risk of incident AF. We also aimed to investigate the association between mid‐life weight change and the risk of incident AF in men and women.
2. METHODS
2.1. Setting and case definition
This cohort study was conducted in Västerbotten County in northern Sweden (population ~260 000). The study participants participated in a health examination, the Västerbotten Intervention Programme (VIP), between 1 January 1988 and 5 September 2014 (Figure S1). Inhabitants were invited to participate in the VIP at 30, 40, 50, and 60 years of age. The programme included measurements of body height, weight, blood pressure, fasting glucose, oral glucose tolerance test, and serum lipids. Participants also completed a questionnaire about, for example, education level, smoking habits, alcohol use, history of diabetes and myocardial infarction, medication use, and leisure time physical activity. Our present study included only participants with available data on both weight and height. Participants were excluded if they had a diagnosis of AF in the Swedish National Patient Registry registered prior to the health examination. In the cohort, incident AF was defined as a first primary or secondary diagnosis of atrial fibrillation or atrial flutter (ICD‐10 code I48, ICD‐9 code 427D, or ICD‐8 code 427.92) in the Swedish National Patient Registry (including both inpatients and outpatients) registered between the health examination date and 5 September 2014. 1 The study complied with the Declaration of Helsinki and was approved by the Regional Ethics Review board, Umeå, Sweden (approval number 2015‐50‐31M). All participants provided written informed consent. Due to restrictions in the ethics approval, source data cannot be made publicly available.
2.1.1. Cohort used in analyses of the association between weight, height, BMI, BSA, and risk of AF
Each VIP participant were followed until diagnosis of AF, death, migration from the study area, or the end of the study on 5 September 2014. Data from each participant's first health examination were used.
2.1.2. Cohort used in the analysis of the association between weight change and risk of AF
Participants who attended two VIP health examination visits 10 years apart (Figure S1) were included in this cohort. Participants were followed from their second health examination until diagnosis of AF, death, migration from the study area, or end of the study on 5 September 2014.
2.2. Measurements and definitions
Height and weight were measured while participants wore light clothing without shoes. BMI was calculated as the weight (kg) divided by the height squared (m2). BSA was calculated according to the duBois formula (0.007184 × weight (kg)0.425 × height (cm)0.725). 17
Hypertension was defined as systolic blood pressure ≥140 mm Hg, diastolic blood pressure ≥90 mm Hg, or self‐reported use of antihypertensive medications during the two weeks before the health survey. Blood pressure was measured after at least five minutes of rest. For cholesterol measurements, serum samples were obtained after at least four hours of fasting. Diabetes was defined as self‐reported diabetes, treatment with anti‐diabetic medication, fasting plasma glucose ≥7.0 mmol/L, or a 2‐hour oral glucose tolerance plasma glucose level of ≥12.2 mmol/L in capillary samples. Data regarding smoking were collected by questionnaire. Participants were categorized as never smokers or ever smokers (including daily smokers, occasional smokers, and previous smokers). Questionnaire data on alcohol use were collected using the CAGE questions (Cut down, Annoyed, Guilty, Eye opener). Problem drinking was defined as two or more affirmative answers to the CAGE questions. Data on education level was collected by questionnaire. Participants were categorized as having more than secondary school education or having a maximum of secondary school education. Data on physical activity were collected by questionnaire. Physical activity was categorized as regular in participants who stated that they exercised at least once a week, and as not regular in all other participants. Data on myocardial infarction were collected by questionnaire.
For analysis of weight change, participants were categorized in predefined groups according to weight change as a percent of baseline weight: weight decrease of >7%, weight decrease of >3% to 7%, stable weight (weight decrease or increase of ≤3%), weight increase of >3% to 7%, and weight increase of >7%.
Among the total cohort, we were missing data on cholesterol for 876 participants (0.8%), hypertension for 769 participants (0.7%), history of myocardial infarction for 5809 participants (5.4%), diabetes for 432 participants (0.4%), problem drinking for 5559 participants (5.1%), tobacco use for 2165 participants (2.0%), physical activity for 6670 participants (6.2%), and education level for 1782 participants (1.6%).
2.3. Statistical methods
Means with standard deviations (SD), numbers, proportions, and medians with interquartile ranges (IQR) were calculated for baseline characteristics. It was predefined that all analyses were to be stratified by sex. Height, weight, and BSA were analyzed both as continuous variables per SD, and as categorical variables using sex‐specific quartiles. BMI was analyzed both as a continuous variable per SD, and as a categorical variable in World Health Organization categories. Weight change was analyzed both as a continuous variable, and as a categorical variable. We performed multiplicative interaction analyses of the two‐way interaction between sex and each of the anthropometric factors (weight, height, BMI, and BSA) relative to risk of AF. Categorical variables were analyzed with Chi‐square tests and continuous variables with independent samples t tests.
Cox proportional hazards regression analysis was performed to calculate the hazard ratio (HR) and 95% confidence interval (CI) for the association between the determinants and the AF risk. We used two prespecified models: the first was adjusted for age, and the second included adjustments for cardiovascular risk factors (age, cholesterol as a binary variable, using the 5.5 mmol/L as cut‐off, education level, smoking habits, history of myocardial infarction, hypertension, diabetes, alcohol use, and physical activity). Analyses of the associations of height and weight with AF were adjusted for weight and height, respectively.
Cox proportional hazards regression was used to calculate the HR and 95% CI for the association between weight change (as the percent of baseline weight) and the risk of incident AF following the second health examination. The model was adjusted for variables measured at the second examination (model 2). The association between weight change and risk of AF was also analyzed with stratification for sex and body height. Additionally, we performed a sensitivity analysis using a statistical model that included weight change as a quadratic term along with weight change as a linear term, which enabled detection of a possibly U‐shaped association. We also analyzed the association between height, weight, BMI, and BSA measured at baseline and the risk of AF in the weight change cohort.
Categorical variables were classified as present, absent, or missing. We performed a sensitivity analysis of individuals with complete data. We performed another sensitivity analysis where missing values were imputed using multiple imputation by chained equations with ten imputed datasets. This multivariable model was adjusted for age, systolic blood pressure, and cholesterol as continuous variables, antihypertensive treatment, alcohol use, history of myocardial infarction, diabetes, smoking, physical activity, and for education level as categorical variables. Associations between weight or weight change and risk of AF were also adjusted for height, and the association between height and risk of AF was also adjusted for weight.
Baseline characteristics measured at the first health examination for participants in the weight change cohort were compared with those of participants who did not attend a second health examination. Weight change in the weight change cohort was compared with that of individuals excluded from the weight change cohort due to acquisition of AF between the two health examinations. Two‐sided P values of < .05 were considered significant. A power calculation showed that with a sample size of 108 000 participants, a cumulative incidence of AF of 4.8% during the study period, a power of 80% and a significance level of 0.05 (two‐sided), we could detect a HR of 1.14 for increased risk of AF in persons with the highest quartile of height, weight, BMI, or BSA. With a sample size of 40 000 participants that had participated in two health examinations, a cumulative incidence of AF of 2.6% during the study period, a power of 80% and a significance level of 0.05 (two‐sided), we could detect a HR of 1.32 for increased risk of AF in persons with the highest quartile of weight change.
Statistical analyses were performed using IBM SPSS Statistics for Macintosh, Version 25.0 (IBM Corp.).
3. RESULTS
The study cohort included 108 417 persons (51% women), with a total follow‐up of 1 469 820 person‐years. Table 1 shows participant characteristics at the first health examination. The mean participant age at inclusion was 46.3 years (SD 9.3). Incident AF was diagnosed in 1954 women and 3200 men. The mean age at AF diagnosis was 68.6 years (SD 8.7) among women and 65.4 years (SD 9.1) among men. The median follow‐up time was 15.6 (IQR 7.5‐19.9) in women and 14.3 (IQR 6.8‐19.5) years in men. Thus the incidence of AF per 1000 person‐years of follow‐up was 2.5 (95% CI, 2.4‐2.7) among women, and 4.6 (95% CI, 4.4‐4.7) among men. Among both women and men, over one‐fourth of the participants were hypertensive, and half of the participants were ever smokers. Mean height, weight, BMI, and BSA significantly differed between men and women (Table 1) (P < .001 for all comparisons).
TABLE 1.
Baseline characteristics of the study cohort (n = 108 417)
| Men (n = 53 376) | Women (n = 55 041) | |
|---|---|---|
| Age, y | 46.4 (9.3) | 46.3 (9.2) |
| Height, cm | 178.4 (6.7) | 164.8 (6.1) |
| Weight, kg | 83.8 (13.3) | 69.0 (13.0) |
| BMI, kg/m2 | 26.3 (3.8) | 25.4 (4.6) |
| BMI <18.5 | 176 (0.3) | 686 (1.2) |
| BMI 18.5‐24.9 | 20 918 (39.2) | 29 572 (53.7) |
| BMI 25.0‐29.9 | 24 813 (46.5) | 16 799 (30.5) |
| BMI 30.0‐34.9 | 6111 (11.4) | 5668 (10.3) |
| BMI ≥35.0 | 1358 (2.5) | 2316 (4.2) |
| BSA, m2 | 2.0 (0.2) | 1.8 (0.2) |
| Cholesterol, mmol/L | 5.6 (1.2) | 5.5 (1.2) |
| Hypertension | ||
| No hypertension | 36 230 (68.3) | 40 549 (74.2) |
| Hypertension | 16 800 (31.7) | 14 069 (25.8) |
| History of MI | ||
| No history of MI | 49 657 (98.4) | 51 907 (99.6) |
| History of MI | 828 (1.6) | 216 (0.4) |
| Diabetes | ||
| No diabetes | 50 525 (95.0) | 52 899 (96.5) |
| Diabetes | 2632 (5.0) | 1929 (3.5) |
| Problem drinking a | ||
| No problem drinking | 45 405 (89.5) | 50 316 (96.6) |
| Problem drinking | 5355 (10.5) | 1782 (3.4) |
| Tobacco use | ||
| Never smoker | 25 378 (48.6) | 26 899 (49.8) |
| Ever smoker | 26 809 (51.4) | 27 166 (50.2) |
| Physical activity | ||
| Non‐regular | 33 066 (66.0) | 32 931 (63.7) |
| Regular | 17 019 (34.0) | 18 731 (36.3) |
| Education level | ||
| ≤secondary school | 40 172 (76.4) | 36 764 (68.0) |
| >secondary school | 12 387 (23.6) | 17 312 (32.0) |
Values are shown as mean (standard deviation) or number (%).
Abbreviations: AF, atrial fibrillation; BMI, body mass index; BSA, body surface area; MI, myocardial infarction.
Problem drinking defined as 2 or more points on the CAGE scale (Cut down, Annoyed, Guilty, Eye opener).
Univariable and multivariable Cox regression analyses revealed that height, weight, BMI, and BSA at the first health examination were significantly associated with increased AF risk in both men and women (Table 2). Similar results were found in the sensitivity analysis of individuals with a complete dataset, and in the model where missing values were handled using multiple imputation (data not shown). The associations of height, weight, BMI, and BSA with the risk of AF did not significantly differ between men and women. The interaction of sex with weight, height, BMI, and BSA was non‐significant.
TABLE 2.
Association of risk of incident atrial fibrillation with anthropometric factors, per standard deviation (SD) and quartiles (Q), among women (n = 55 041) and men (n = 53 376)
| Women | Men | |||||
|---|---|---|---|---|---|---|
| Model 1 a | Model 2 b | Model 1 a | Model 2 b | |||
| Height, per SD | 1.27 (1.21‐1.33) | 1.19 (1.13‐1.26) | 1.29 (1.24‐1.33) | 1.16 (1.11‐1.21) | ||
| Height, cm | Q1 (<161) | 1 (ref.) | 1 (ref.) | Q1 (<174) | 1 (ref.) | 1 (ref.) |
| Q2 (161‐164) | 1.23 (1.09‐1.39) | 1.15 (1.02‐1.30) | Q2 (174‐177) | 1.22 (1.10‐1.35) | 1.13 (1.01‐1.25) | |
| Q3 (165‐168) | 1.37 (1.20‐1.55) | 1.23 (1.08‐1.40) | Q3 (178‐182) | 1.36 (1.24‐1.50) | 1.16 (1.05‐1.28) | |
| Q4 (≥169) | 1.76 (1.54‐2.00) | 1.50 (1.31‐1.72) | Q4 (≥183) | 1.91 (1.73‐2.12) | 1.47 (1.31‐1.64) | |
| P for trend | <.001 | <.001 | P for trend | <.001 | <.001 | |
| Weight, per SD | 1.37 (1.32‐1.43) | 1.26 (1.20‐1.32) | 1.38 (1.33‐1.42) | 1.27 (1.22‐1.32) | ||
| Weight, kg | Q1 (<60) | 1 (ref.) | 1 (ref.) | Q1 (<75) | 1 (ref.) | 1 (ref.) |
| Q2 (60‐66) | 1.23 (1.06‐1.43) | 1.13 (0.97‐1.31) | Q2 (75‐81) | 1.30 (1.16‐1.44) | 1.18 (1.06‐1.32) | |
| Q3 (67‐74) | 1.40 (1.21‐1.63) | 1.20 (1.03‐1.40) | Q3 (82‐90) | 1.45 (1.31‐1.61) | 1.24 (1.11‐1.38) | |
| Q4 (≥75) | 2.17 (1.89‐2.49) | 1.67 (1.44‐1.93) | Q4 (≥91) | 2.33 (2.11‐2.58) | 1.79 (1.60‐2.01) | |
| P for trend | <.001 | <.001 | P for trend | <.001 | <.001 | |
| Body mass index, per SD | 1.28 (1.23‐1.33) | 1.22 (1.17‐1.28) | 1.27 (1.23‐1.31) | 1.23 (1.19‐1.28) | ||
| Body mass index, kg/m2 | <18.5 | 0.63 (0.33‐1.22) | 0.64 (0.33‐1.24) | <18.5 | 0.46 (0.15‐1.43) | 0.45 (0.14‐1.40) |
| 18.5‐24.9 | 1 (ref.) | 1 (ref.) | 18.5‐24.9 | 1 (ref.) | 1 (ref.) | |
| 25.0‐29.9 | 1.18 (1.06‐1.31) | 1.14 (1.02‐1.26) | 25.0‐29.9 | 1.25 (1.15‐1.35) | 1.21 (1.11‐1.31) | |
| 30.0‐34.9 | 1.58 (1.39‐1.81) | 1.43 (1.25‐1.65) | 30.0‐34.9 | 1.95 (1.75‐2.18) | 1.80 (1.61‐2.02) | |
| ≥35.0 | 2.56 (2.15‐3.05) | 2.19 (1.83‐2.62) | ≥35.0 | 2.69 (2.20‐3.28) | 2.37 (1.93‐2.90) | |
| P for trend | <.001 | <.001 | P for trend | <.001 | <.001 | |
| Body surface area, per SD | 1.42 (1.36‐1.49) | 1.38 (1.32‐1.44) | 1.43 (1.38‐1.48) | 1.39 (1.35‐1.45) | ||
| Body surface area, m2 | Q1 (<1.65) | 1 (ref.) | 1 (ref.) | Q1 (<1.91) | 1 (ref.) | 1 (ref.) |
| Q2 (1.65‐1.74) | 1.37 (1.19‐1.58) | 1.36 (1.18‐1.56) | Q2 (1.91‐2.01) | 1.37 (1.24‐1.52) | 1.35 (1.22‐1.50) | |
| Q3 (1.74‐1.84) | 1.57 (1.37‐1.80) | 1.53 (1.33‐1.75) | Q3 (2.01‐2.12) | 1.68 (1.52‐1.86) | 1.63 (1.47‐1.81) | |
| Q4 (≥1.84) | 2.40 (2.10‐2.73) | 2.23 (1.95‐2.55) | Q4 (≥2.12) | 2.42 (2.19‐2.68) | 2.28 (2.06‐2.53) | |
| P for trend | <.001 | <.001 | P for trend | <.001 | <.001 | |
Associations presented as hazard ratio and 95% confidence interval.
Model 1, Adjusted for age.
Model 2, Adjusted for age, s–cholesterol, education level, smoking, history of myocardial infarction, hypertension, diabetes, alcohol use, and physical activity. Analyses of height are adjusted for weight, and analyses of weight are adjusted for height.
3.1. Weight change cohort
Within our cohort, 21 243 women and 19 032 men underwent two health examinations, ten years apart (Figure S1). The median follow‐up time was 8.9 (IQR 5.8‐11.6) in women and 8.7 (IQR 5.5‐11.5) years in men. After the second health examination, incident AF occurred in 405 women and 737 men. Among these participants, the mean age at AF diagnosis was 63.8 years (SD 5.9) in women and 62.6 years (SD 6.4) in men. The median weight change as the percent of baseline weight was a weight gain of 4.6% (IQR 0‐10.2) in women, and 4.0% (IQR 0‐8.4) in men. Participants in the weight change cohort were overall similar to participants who did not attend a second health examination, but small but significant differences were found, for example, the participants in the weight change cohort weighed 1‐2 kg more.
Within the weight change cohort, weight loss or weight gain during the 10‐year interval between examinations was not significantly associated with the risk of incident AF in men or women (Table 3). Similar results were found in the sensitivity analysis of individuals with a complete dataset, and in the model where missing values were handled using multiple imputation (data not shown). There was no significant interaction between weight change and sex relative to the risk of AF (P = .62). Figure 1 shows the relationship of AF risk with categories of weight change and body height in sex‐specific tertiles. In a sensitivity analysis, the results were not affected by the addition of weight change as a quadratic term to any of the models. Mean weight change significantly differed between the weight change cohort and the individuals excluded from the weight change cohort due to being diagnosed with incident AF between health examinations (n = 355) (4.8% [SD 8.8] compared to 3.7% [SD 8.1], P .01). Analysis of the association between height, weight, BMI, and BSA measured at baseline and the risk of AF in the weight change cohort yielded similar results as the analysis on the cohort of 108 417 participants presented in Table 2.
TABLE 3.
Association between relative weight change and incident atrial fibrillation, stratified by sex
| Women | Men | |||||
|---|---|---|---|---|---|---|
| n (%) | Model 1 a | Model 2 b | n (%) | Model 1 a | Model 2 b | |
| Weight change, per % | 21 243 | 0.997 (0.99‐1.01) | 0.995 (0.98‐1.01) | 19 032 | 1.00 (0.99‐1.01) | 1.00 (0.99‐1.01) |
| Weight decrease >7% | 1540 (7.2) | 1.11 (0.74‐1.66) | 1.06 (0.71‐1.60) | 818 (4.3) | 0.98 (0.67‐1.44) | 0.88 (0.60‐1.30) |
|
Weight decrease >3% to 7%, |
1871 (8.8) | 0.99 (0.67‐1.45) | 1.01 (0.68‐1.48) | 1498 (7.9) | 1.05 (0.80‐1.37) | 1.02 (0.78‐1.33) |
| Stable weight c | 5344 (25.2) | 1 (ref.) | 1 (ref.) | 6015 (31.6) | 1 (ref.) | 1 (ref.) |
|
Weight increase >3% to 7% |
4430 (20.9) | 1.03 (0.78‐1.36) | 1.02 (0.77‐1.35) | 4650 (24.4) | 0.95 (0.78‐1.15) | 0.92 (0.76‐1.12) |
| Weight increase >7% | 8058 (37.9) | 0.92 (0.72‐1.19) | 0.88 (0.68‐1.13) | 6051 (31.8) | 0.97 (0.81‐1.17) | 0.92 (0.76‐1.10) |
Associations presented as hazard ratio with 95% confidence interval.
Model 1, Adjusted for age and height, obtained at the second health examination.
Model 2, Adjusted for variables: age, height, s–cholesterol, education level, smoking, history of myocardial infarction, hypertension, diabetes, alcohol use, and physical activity, obtained at the second health examination.
Stable weight: weight decrease or weight increase of ≤3% of baseline weight.
FIGURE 1.
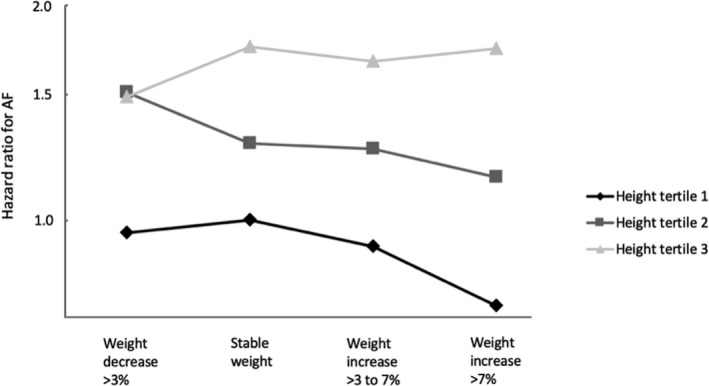
Illustration of the risk of atrial fibrillation in categories of weight change, stratified by sex‐specific tertiles of height. Analysis adjusted for age, sex, s–cholesterol, education level, smoking, history of myocardial infarction, hypertension, diabetes, alcohol use, and physical activity, obtained at the second health examination. Participants in the lowest height tertile with stable weight were used as a reference group. Associations shown as hazard ratios. Associations between weight change and risk of AF are shown as black diamonds for participants in the lowest height tertile, as dark gray squares for participants in the middle height tertile, and as light gray triangles for participants in the highest height tertile
4. DISCUSSION
The present results showed that height, weight, BMI, and BSA were positively associated with risk of incident AF in both men and women. The risk of incident AF was not significantly associated with weight change during the past 10 years among middle‐aged men or women.
Our study cohort comprised a large number of middle‐aged men and women who participated in a population‐based health examination program, with a total follow‐up of 1.5 million person‐years. This large study population enabled analysis of the risk of incident AF associated with anthropometric factors in comparable groups of men and women. The associations between AF and all four anthropometric variables were of similar strength in men and women. Additionally, we found no interaction between sex and the anthropometric factors relative to AF risk.
The presently observed association between height and risk of AF was in agreement with previously reported results. 3 , 5 Associations of BMI and BSA with AF have also been found in other studies. 5 , 7 , 12 Increased atrial size is considered a possible mediator of the association between all studied anthropometric factors and the risk of AF. 5 , 12 , 18 , 19 However, the association between height and AF can only partly be explained by echocardiographic measurements, such as atrial size. 3 Another mechanism that may explain this association include common genetic factors that are associated with both growth pathways and with AF. 20 , 21 Additionally, obesity is reportedly associated with diastolic dysfunction, 22 pericardial fat deposition, 23 and epicardial adiposie tissue 24 which all in turn are associated with increased AF risk. Obesity is also associated with several other established risk factors for AF such as heart failure, coronary heart disease, obstructive sleep apnea, and hypertension. 25
4.1. Weight change cohort
We found that weight gain during the past 10 years was not significantly associated with risk of incident AF among either men or women. In contrast, the population‐based ARIC study found that weight gain was associated with an increased risk of AF among men, but not women. 8 The mean weight change in that study was 4%, similar to the mean weight increase of 4.8% (SD 8.8) in our present investigation. The Malmö Preventive project included only middle‐aged men, and found that weight gain was associated with AF risk. 11 A meta‐analysis showed an association between a weight gain of 5% and increased risk of AF. 26 Differences in studied populations and age of onset of AF might explain the differences between the results of our study compared to the previous studies.
The American Women's Health Study—a randomized study comparing aspirin and vitamin E for primary prevention of cardiovascular disease and cancer—showed that developing obesity in midlife was associated with risk of AF. 14 The study population was restricted to female health professionals, and BMI was calculated using self‐reported weight and height. Furthermore, medical records and ECGs were reviewed only for women with self‐reported AF. These differences between the Women's Health Study and our present study make it difficult to compare the results. Two previous studies reported that weight gain between age 20‐25 and midlife was associated with increased AF risk in men. 10 , 12 It is possible that this observed positive association between weight gain and AF risk was driven by weight gain early in adult life, as opposed to weight gain during midlife, which was investigated in our study.
Our finding that weight loss was not associated with AF risk is in line with the results of several prior studies. 9 , 12 , 13 , 14 , 15 , 26 An intensive lifestyle intervention study that included individuals with type 2 diabetes reported no association between weight loss and risk of AF. 15 However, other studies have found that weight loss was associated with both increased and decreased risk of AF. 3 , 8
One strength of our study was the population‐based study design. A previous analysis of VIP data reported minimal differences in education level and age between participants and non‐participants. 27 The participation rate was slightly lower among persons with low income and persons who are single, compared to persons with a medium‐to‐high income and persons who live with a partner. Height, weight, and weight change measurements, and the collection of data on all other covariates, were performed at health examinations prior to the AF event, eliminating recall bias. Due to the large number of participants and long follow‐up time, we were able to perform sex‐stratified analyses with sufficient statistical power. This is highly important since the association between anthropometric factors and AF reportedly varies between women and men. 7 Another strength of our study is that we do not have any loss to follow‐up due to the personal identification numbers given to every Swedish citizen. A diagnosis of AF in the Swedish National Patient Registry has a high accuracy, with a positive predictive value of 96.5%. 1
Our study also had several limitations. Our use of a questionnaire to collect data regarding factors such as smoking, problem drinking, and leisure time physical activity may have resulted in misclassification of individuals who accidentally or intentionally answered the questionnaire incorrectly. Additionally, our observational study design entails an inherent risk of residual confounding. Another limitation is our use of only two weight measurements to evaluate the association between weight change and incident AF, which may affect our possibility to identify a significant association. It would have been valuable to have echocardiographic measurements in addition to the anthropometric measurements. Notably, we lacked data for some known AF risk factors, such as the presence of structural heart diseases and chronic obstructive pulmonary disease. Finally, the absolute majority of the study participants were of European ancestry and younger than 75 years at the end of follow‐up; therefore, our study results may not be generalizable to other ethnic or age groups. The mean age at AF diagnosis in Sweden is 73.5 years. 2 Thus, our study participants are, on average, younger when receiving their AF diagnosis.
5. CONCLUSIONS
In conclusion, our present results show that higher weight, height, BMI, and BSA were associated with increased risk of AF. In contrast, weight change during midlife was not significantly associated with AF risk in our cohort in which participants were younger than 75 years at the end of follow‐up. The risk estimates were similar for men and women.
CONFLICT OF INTEREST
Authors declare no conflict of interest for this article.
Supporting information
Fig S1
ACKNOWLEDGEMENTS
This work was supported by Västerbotten County Council; the Joint Committee of Northern Sweden Health Care Region; and the Foundation for Medical Research in Skellefteå.
Johansson C, Lind MM, Eriksson M, Johansson L. Weight, height, weight change, and risk of incident atrial fibrillation in middle-aged men and women. J Arrhythmia. 2020;36:974–981. 10.1002/joa3.12409
REFERENCES
- 1. Norberg J, Backstrom S, Jansson JH, Johansson L. Estimating the prevalence of atrial fibrillation in a general population using validated electronic health data. Clin Epidemiol. 2013;9(5):475–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Johansson C, Dahlqvist E, Andersson J, Jansson JH, Johansson L. Incidence, type of atrial fibrillation and risk factors for stroke: a population‐based cohort study. Clin Epidemiol. 2017;24(9):53–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Berkovitch A, Kivity S, Klempfner R, Segev S, Milwidsky A, Erez A, et al. Body mass index and the risk of new‐onset atrial fibrillation in middle‐aged adults. Am Heart J. 2016;17(173):41–8. [DOI] [PubMed] [Google Scholar]
- 4. Fenger‐Gron M, Overvad K, Tjonneland A, Frost L. Lean body mass is the predominant anthropometric risk factor for atrial fibrillation. J Am Coll Cardiol. 2017;69(20):2488–97. [DOI] [PubMed] [Google Scholar]
- 5. Trevisan C, Maggi S, Curreri C, Nante G, Noale M, De Rui M, et al. Anthropometric parameters and the incidence of atrial fibrillation in older people: the PRO.V.A study. Clin Cardiol. 2017;40(7):461–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chatterjee NA, Giulianini F, Geelhoed B, Lunetta KL, Misialek JR, Niemeijer MN, et al. Genetic obesity and the risk of atrial fibrillation: causal estimates from Mendelian randomization. Circulation. 2017;135:741–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Magnussen C, Niiranen TJ, Ojeda FM, Gianfagna F, Blankenberg S, Njølstad I, et al. Sex differences and similarities in atrial fibrillation epidemiology, risk factors, and mortality in community cohorts: results from the BiomarCaRE Consortium (Biomarker for Cardiovascular Risk Assessment in Europe). Circulation. 2017;136:1588–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Huxley RR, Misialek JR, Agarwal SK, Loehr LR, Soliman EZ, Chen LY, et al. Physical activity, obesity, weight change, and risk of atrial fibrillation: the Atherosclerosis Risk in Communities study. Circ Arrhythm Electrophysiol. 2014;7(4):620–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Diouf I, Magliano DJ, Carrington MJ, Stewart S, Shaw JE. Prevalence, incidence, risk factors and treatment of atrial fibrillation in Australia: the Australian Diabetes, Obesity and Lifestyle (AusDiab) longitudinal, population cohort study. Int J Cardiol. 2016;15(205):127–32. [DOI] [PubMed] [Google Scholar]
- 10. Tedrow UB, Conen D, Ridker PM, Cook NR, Koplan BA, Manson JE, et al. The long‐ and short‐term impact of elevated body mass index on the risk of new atrial fibrillation the WHS (Women's Health Study). J Am Coll Cardiol. 2010;55(21):2319–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Grundvold I, Bodegard J, Nilsson PM, Svennblad B, Johansson G, Östgren C, et al. Body weight and risk of atrial fibrillation in 7,169 patients with newly diagnosed type 2 diabetes; an observational study. Cardiovasc Diabetol. 2015;15(14):5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Rosengren A, Hauptman PJ, Lappas G, Olsson L, Wilhelmsen L, Swedberg K. Big men and atrial fibrillation: effects of body size and weight gain on risk of atrial fibrillation in men. Eur Heart J. 2009;30(9):1113–20. [DOI] [PubMed] [Google Scholar]
- 13. Johnson LS, Juhlin T, Engstrom G, Nilsson PM. Risk factor changes and incident atrial fibrillation among middle‐aged men in the Malmo Preventive Project cohort. Eur Heart J Cardiovasc Pharmacother. 2016;2(2):81–7. [DOI] [PubMed] [Google Scholar]
- 14. Grundvold I, Skretteberg PT, Liestol K, Gjesdal K, Erikssen G, Kjeldsen SE, et al. Importance of physical fitness on predictive effect of body mass index and weight gain on incident atrial fibrillation in healthy middle‐age men. Am J Cardiol. 2012;110(3):425–32. [DOI] [PubMed] [Google Scholar]
- 15. Alonso A, Bahnson JL, Gaussoin SA, Bertoni AG, Johnson KC, Lewis CE, et al. Effect of an intensive lifestyle intervention on atrial fibrillation risk in individuals with type 2 diabetes: the Look AHEAD randomized trial. Am Heart J. 2015;170(4):770–7.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Pathak RK, Middeldorp ME, Meredith M, Mehta AB, Mahajan R, Wong CX, et al. Long‐term effect of goal‐directed weight management in an atrial fibrillation cohort: a long‐term follow‐up study (LEGACY). J Am Coll Cardiol. 2015;65(20):2159–69. [DOI] [PubMed] [Google Scholar]
- 17. Du Bois D, Du Bois EF. A formula to estimate the approximate surface area if height and weight be known. Arch Intern Med. 1916;17(6_2):863–71. [PubMed] [Google Scholar]
- 18. Psaty BM, Manolio TA, Kuller LH, Kronmal RA, Cushman M, Fried LP, et al. Incidence of and risk factors for atrial fibrillation in older adults. Circulation. 1997;96(7):2455–61. [DOI] [PubMed] [Google Scholar]
- 19. Zong P, Zhang L, Shaban NM, Pena J, Jiang L, Taub CC. Left heart chamber quantification in obese patients: how does larger body size affect echocardiographic measurements? J Am Soc Echocardiogr. 2014;27(12):1267–74. [DOI] [PubMed] [Google Scholar]
- 20. Gudbjartsson DF, Arnar DO, Helgadottir A, Gretarsdottir S, Holm H, Sigurdsson A, et al. Variants conferring risk of atrial fibrillation on chromosome 4q25. Nature. 2007;448(7151):353–357. [DOI] [PubMed] [Google Scholar]
- 21. Benjamin EJ, Rice KM, Arking DE, Pfeufer A, van Noord C, Smith AV, et al. Variants in ZDHX3 are associated with atrial fibrillation in individuals of European ancestry. Nat Genet. 2009;41(8):879–881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Rosenberg MA, Manning WJ. Diastolic dysfunction and risk of atrial fibrillation: a mechanistic appraisal. Circulation. 2012;126(19):2353–62. [DOI] [PubMed] [Google Scholar]
- 23. Thanassoulis G, Massaro JM, O'Donnell CJ, Hoffmann U, Levy D, Ellinor PT, et al. Pericardial fat is associated with prevalent atrial fibrillation: The Framingham Heart Study. Circ Arrhthm Electrophysiol. 2010;3(4):345–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Goudis CA, Korantzopoulos P, Ntalas IV, Kallergis EM, Ketikoglou DG. Obesity and atrial fibrillation: a comprehensive review of the pathophysiological mechanisms and links. J Cardiol. 2015;66(5):361–369. [DOI] [PubMed] [Google Scholar]
- 25. Mangani JW, Hylek EM, Apovian CM. Obesity begets atrial fibrillation: a contemporary summary. Circulation. 2013;128(4):401–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Jones NR, Taylor KS, Taylor CJ, Aveyard P. Weight change and the risk of incident atrial fibrillation: a systematic review and meta‐anaylsis. Heart. 2019;105(23):1799–1805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Norberg M, Blomstedt Y, Lonnberg G, Nyström L, Stenlund H, Wall S, et al. Community participation and sustainability–evidence over 25 years in the Vasterbotten Intervention Programme. Glob Health Action. 2012;17(5):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Fig S1


