Abstract
A morbidly obese middle aged woman in her 40's presented to another hospital with methicillin resistant staphylococcus aureus pneumonia and subsequently developed severe acute respiratory distress syndrome. Her oxygenation demonstrated no improvement with low tidal volume ventilation, paralysis, or prostagladin therapy. She was unable to be manually proned secondary to her habitus. She was subsequently transferred to our facility, where she was initiated on VV-ECMO. Maximal flow through the ECMO circuit was inadequate for oxygenation given significant systemic shunt through her native lungs. In order to optimize lung protective ventilation and treat ARDS, we used an automated kinetic system (Rotoprone Therapy System) to prone the patient. To our knowledge, this is the first description in the literature of using an automated proning system with an ECMO circuit in place. This report describes the technique we used to safely perform axial rotations for two days with fewer providers required than manual proning.
Keywords: Acute respiratory distress syndrome, Prone positioning, Extracorporeal membrane oxygenation
1. Introduction
Rescue measures for severe acute respiratory distress syndrome (ARDS) include both prone positioning (PP) [1] and veno-venous extracorporeal membrane oxygenation (ECMO) [2]. PP and ECMO improve oxygenation and also are thought to decrease the inflammation from ARDS by reducing barotrauma and volu-trauma in the lung [[3], [4], [5]]. There is increasing literature reporting the safety and benefit of combining both PP and ECMO, including the ability to provide lung protective ventilation, quicker ventilator weaning, and expediting decannulation [[6], [7], [8]]. While there is a growing body of literature supporting the use of manual proning, to date no one has reported the use of an automated proning system with a patient on ECMO. The use of an automated proning system can be advantageous in hospitals with low nursing ratios, obese patients, and is often standard in ICUs in the United States [9]. We describe the use of a Rotaprone bed in a patient with ECMO cannulation and where cannulas should be passed to safely rotate patients while minimizing torque on the cannulas.
2. Case report
A 48 year old female with history of hypertension, diabetes mellitus type II, chronic obstructive pulmonary disease, obstructive sleep apnea, and severe obesity (BMI: 51, weight 140kg) presented to another hospital with generalized weakness for four days. She also endorsed worsening shortness of breath from her baseline. She denied fevers, chills, cough, chest pain, and any focal weakness or numbness. Social history was significant for cigarette use of 1 pack per day. Imaging on both chest X-ray and CT Chest were consistent with multi-focal ground glass opacities and consolidations without pleural effusions concerning for pneumonia. Upon admission to the general medicine service, patient was found to be increasingly hypotensive with MAP's below 65 and worsening mental status. She was subsequently intubated for worsening hypoxic respiratory failure. Sputum cultures after intubation grew methicillin resistant staphylococcus aureus. Her course was complicated by severe ARDS refractory to ventilator optimization, paralysis, and epoprostenol. Proning was not attempted given obesity. After one week, patient was transferred to our institution for ECMO evaluation given persistent hypoxia.
Upon arrival, patient's arterial blood gas demonstrated a pH of 7.34, PaCO2 of 59 mmHg, and PaO2 of 64 mmHg while on pressure control ventilation with an inspiratory pressure of 22 cm H20 over PEEP of 20 cm H20 and a FiO2 of 100%. Tidal volumes were approximately 400mL. Patient was cannulated for veno-venous ECMO with 25-Fr right femoral inflow cannula and a 21-Fr right internal jugular outflow cannula. Despite ECMO flow rates to 6L/min, patient required significant ventilator support-pressure control with inspiratory pressure of 10 cm H20, PEEP of 12 cm H20, FiO2 80%, tidal volumes of 300mL, and SaO 2 of 80–92%. Her peripheral arterial blood gas on these ventilator settings and ECMO settings of flow 6L/min, O2 concentration of 100%, sweep of 6L/min was pH 7.21, PaCO2 50 mmHg, PaO2 76 mmHg. The ECMO oxygenator was thought to be functioning well given post-oxygenator blood gas analysis demonstrated a PaO2 of 397 mmHg. Cannula position was adequate on CT imaging with superior ECMO cannula located in the superior vena cava – right atrial junction and the femoral ECMO cannula located in the inferior vena cava. Pre-oxygenator PaO2 was 42 mmHg without significant changes over her hospital course. Given this, recirculation was not felt to be significantly contributing, and additional cannulas were not placed. On day 5, given inability to optimize lung-protective ventilation, a CT scan was performed which demonstrated persistent bilateral dense lower lung consolidations. Echocardiogram demonstrated an ejection fraction of 55% with right ventricular dilation; otherwise the study with within normal limits. Her inability to oxygenate on ECMO was thought to be secondary to profound lung shunting as well as her obesity and high metabolic rate. Given the patient's size, manual proning seemed to be more dangerous and the decision was made to use an automated kinetic system (Rotoprone Therapy System).
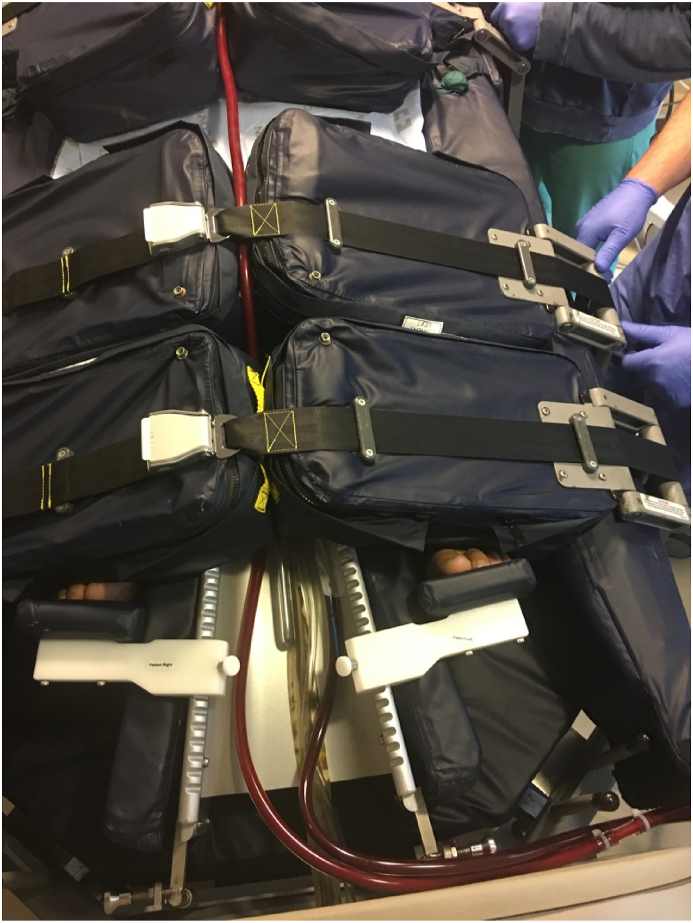
Four feet of extension tubing was added to the ECMO circuit in order to avoid potential tension during axial rotation. Extension tubing was also added to any intravenous lines. Prior to moving to the Rotoprone bed, the ETT holder was removed and the ETT was secured with adhesive tape. Once the patient was transferred to the proning bed, both ECMO cannulas were directed caudally between the U-slot of the bed (Fig. 1).
Fig. 1.
Positioning of ECMO circuits prior to proning. ECMO circuit is toward the patient's left in this picture.
Multiple foam dressings were applied over pressure points as well as any contact points between the ECMO circuit and patient (Fig. 2) in order to avoid skin breakdown.
Fig. 2.
Placement of hydrocolloid foam dressings over the patient's anterior. Particular attention was made to place hydrocolloid dressings over the patient's forehead, cheeks, breats, and contact points between the ECMO circuit and skin.
Accessory padding included with the proning system was used as instructed to further prevent pressure injury. A total of four staff were involved in the proning procedure: one perfusionist monitoring the cannulas, one nurse ensuring stability of intravenous lines, one nurse initiating axial rotation, and one physician monitoring the endotracheal tube and ventilator. The patient was paralyzed with boluses of rocuronium during turning events. During both supine and proned periods, the patient was deeply sedated with propofol and fentanyl in order to avoid agitation and potential derecruitment. While secured in the bed with the back of the bed still on, patients lung compliance worsened and required increased driving pressure. However, this recovered when the back of the bed was removed after the proning maneuver (Fig. 3).
Fig. 3.
Patient proned while in the automated system while attached to the ECMO circuit.
Upon proning the patient, there was significant improvement in oxygenation with improvement in P/F ratio to 250. Patient was proned for a total of 12 hours per day and underwent a total of four axial rotations. After day 8, patient was kept supine for the rest of her ICU stay, with persistent improvements in oxygenation. She did not experience any complications related to mechanical proning. ECMO cannulas were removed on day 13 and she was discharged to rehabilitation on day 41. Patient completed acute rehab and was discharged to home without need for home oxygen or mobile assist devices.
3. Discussion
To our knowledge, this is the first published case of using an automated system to provide PP to a patient while on an ECMO circuit. The choice to employ either ECMO or PP in refractory ARDS has been well described, however there is increasing attention paid to when a single mechanism is not adequate to allow for protective ventilation. ECMO only serves to capture a portion of blood through the circuit. Therefore the ratio of circuit to systemic blood flow (Qe/Qs) operates as a shunt. The shunt fraction is higher in patients with high cardiac output, obesity, or with profound shunt within native lungs. Our patient suffered from obesity, high cardiac output, and dense consolidation causing significant shunt physiology. In these scenarios, it may be necessary to increase ventilator settings to better oxygenate blood shunted through the lungs.
Manual proning was not felt to be an option due to the patient's size and automated proning using the Rotaprone bed was employed. Automated proning requires fewer clinicians to perform, particularly in obese patients, and therefore may decrease strain on hospital staff. Only four providers were need to maintain safety during axial rotation.
An important consideration when performing PP is skin care. Previous studies have noted increased rates of ulcer formation in patients undergoing PP [10]. Skin care may be more difficult in patients requiring an automated proning system given the device may obscure visualization of pressure points. In our case, our ICU team utilized foam dressings, which were added to any potential points of contact with the patient's skin. Skin checks were also performed every shift while supine. There were no adverse effects observed.
When to employ prone positioning while a patient undergoes ECMO is still undergoing study. We believe that if lung protective ventilation cannot be maintained on ECMO, prone positioning should be considered [6]. There is some evidence to suggest that the use of prone ventilation is anti-inflammatory, and it is unclear if that is only related to tidal volume or if the equalization of chest wall compliance is also important [1]. While more study will be needed to determine timing of prone positioning, we conclude that the use of an automated proning system is safe when employing both ECMO and proning.
Author contributions
AL was involved in writing the original draft as well as review and editing of final manuscript. DS, BT, RG were involved in writing-review & editing. SB was involved in writing - review & editing as well as supervision.
Declaration of competing interest
The authors have no conflicts of interest, financial or otherwise, to report regarding the content of this manuscript and no involvement with organization(s) with financial interest in the subject matter.
References
- 1.Abroug F., Ouanes-Besbes L., Elatrous S., Brochard L. The effect of prone positioning in acute respiratory distress syndrome or acute lung injury: a meta-analysis. Areas of uncertainty and recommendations for research. Intensive Care Med. 2008;34(6):1002–1011. doi: 10.1007/s00134-008-1062-3. [DOI] [PubMed] [Google Scholar]
- 2.Brodie D., Bacchetta M. Extracorporeal membrane oxygenation for ARDS in adults. N. Engl. J. Med. 2011;365(20):1905–1914. doi: 10.1056/NEJMct1103720. [DOI] [PubMed] [Google Scholar]
- 3.Thompson B.T., Chambers R.C., Liu K.D. Acute respiratory distress syndrome. N. Engl. J. Med. 2017;377(6):562–572. doi: 10.1056/NEJMra1608077. [DOI] [PubMed] [Google Scholar]
- 4.Galiatsou E., Kostanti E., Svarna E., Kitsakos A., Koulouras V., Efremidis S.C. Prone position augments recruitment and prevents alveolar overinflation in acute lung injury. Am. J. Respir. Crit. Care Med. 2006;174(2):187–197. doi: 10.1164/rccm.200506-899OC. [DOI] [PubMed] [Google Scholar]
- 5.Schmidt M., Zogheib E., Roze H., Repesse X., Lebreton G., Luyt C.E. The PRESERVE mortality risk score and analysis of long-term outcomes after extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. Intensive Care Med. 2013;39(10):1704–1713. doi: 10.1007/s00134-013-3037-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kimmoun A., Roche S., Bridey C., Vanhuyse F., Fay R., Girerd N. Prolonged prone positioning under VV-ECMO is safe and improves oxygenation and respiratory compliance. Ann. Intensive Care. 2015;5(1):35. doi: 10.1186/s13613-015-0078-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Voelker M.T., Jahn N., Bercker S., Becker-Rux D., Koppen S., Kaisers U.X. [Prone positioning of patients during venovenous extracorporeal membrane oxygenation is safe and feasible] Anaesthesist. 2016;65(4):250–257. doi: 10.1007/s00101-015-0131-6. [DOI] [PubMed] [Google Scholar]
- 8.Culbreth R.E., Goodfellow L.T. Complications of prone positioning during extracorporeal membrane oxygenation for respiratory failure: a systematic review. Respir. Care. 2016;61(2):249–254. doi: 10.4187/respcare.03882. [DOI] [PubMed] [Google Scholar]
- 9.Baacke M.G., Neubert T., Spies M., Gotzen L., Stiletto R.J. Rotoprone(®): a new and promising way to prone positioning. Crit. Care. 2002;6(Suppl 1) P19-P. [Google Scholar]
- 10.Girard R., Baboi L., Ayzac L., Richard J.C., Guerin C. Proseva trial g. The impact of patient positioning on pressure ulcers in patients with severe ARDS: results from a multicentre randomised controlled trial on prone positioning. Intensive Care Med. 2014;40(3):397–403. doi: 10.1007/s00134-013-3188-1. [DOI] [PubMed] [Google Scholar]