Abstract
The aim of this study was to investigate the antimalarial activities and toxicity of Pogostemon cablin extracts. In vitro activities against the chloroquine-resistant Plasmodium falciparum K1 strain were assessed by using the Plasmodium lactate dehydrogenase enzyme (pLDH) assay, while in vivo activity against the Plasmodium berghei ANKA strain in mice was investigated using a 4-day suppressive test. The in vitro and in vivo toxicity were determined in Vero cells and mice, respectively. The ethanolic extract possessed antimalarial activity with an IC50 of 24.49 ± 0.01 µg/ml, whereas the aqueous extract showed an IC50 of 549.30 ± 0.07 µg/ml. Cytotoxic analyses of the ethanolic and aqueous extracts revealed a nontoxic effect on Vero cells at a concentration of 80 µg/ml. Based on a preliminary study of in vitro antimalarial activity, the ethanolic extract was chosen as a potential agent for further in vivo antimalarial activity analysis in mice. The ethanolic extract, which showed no toxic effect on mice at a dose of 2000 mg/kg body weight, significantly suppressed parasitemia in mice by 38.41%, 45.12% and 89.00% at doses of 200, 400 and 600 mg/kg body weight, respectively. In conclusion, this study shows that the ethanolic P. cablin extract possesses in vitro and in vivo antimalarial activity without toxic effects.
Keywords: malaria, antimalarial activity, Pogostemon cablin (Blanco) benth, Plasmodium falciparum, Plasmodium berghei, toxicity
Introduction
Malaria is an important parasite-induced tropical disease that threatens many people globally.1,2 The malaria parasites, namely, Plasmodium falciparum, Plasmodium vivax, Plasmodium malariae, Plasmodium ovale and Plasmodium knowlesi, are the 5 species that cause malaria in humans. According to a report published in 2019, approximately 228 million malaria cases occurred worldwide, resulting in 405,000 deaths.3,4 The severe complications associated with malaria are related to mortality incidence and are a significant public health concern.5 At present, treatment of malaria involves mainly chemotherapy. However, emerging resistance to antimalarial drugs of different classes has become an urgent problem.6–10 The treatment failure rates of the first-line drug artemisinin-based combination therapy against P. falciparum were above 10% in the WHO Southeast Asia region and were as high as 93% in Thailand,3 indicating that new therapeutic options are urgently needed. Natural medicinal products serve as potential sources of diverse pharmacological activities based on types of bioactive compounds with differences in chemical structures. Pogostemon cablin (Blanco) Benth., commonly recognized as “Patchouli” or “Phimsen ton” in Thailand, belongs to the Lamiaceae family,11 which is commonly found in South Asian countries.12 This plant is a short-lived perennial herb that grows to 1.2 m tall and is branched with an erect and quadrangular stem.13 It is traditionally used as remedies for colds, fever, abdominal pain, diarrhea, nausea, vomiting, and insect bites and possesses many pharmacological activities, such as antimicrobial, gastroprotective, antioxidant, antiemetic, antithrombotic, blood coagulative and fibrinolytic activities.14–17 However, neither the antimalarial activity nor the toxicity of P. cablin had been studied until this study.
Experimental Section
Plant Material
The dried aerial part of P. cablin was obtained from a traditional Thai drug store in Nakhon Si Thammarat Province, Thailand. The plant was botanically identified by Assoc. Prof. Tanomjit Supavita, an expert botanist, and has a voucher number of SMD143228 at the School of Medicine, Walailak University. The plant material was cleaned to remove dust or dead material and dried at 60°C by using hot air oven. The dried plant material was coarsely chopped using an herb grinder (Jincheng, Model; SF, China) and stored at room temperature until extraction.
Plant Extraction
P. cablin was extracted with ethanol by maceration and with water extraction by the reflux method. Sixty grams of plant powder was soaked in 600 ml of ethanol at room temperature for 72 h, and another 60 grams was extracted with 600 ml of distilled water under reflux for 2 h. These were then filtered and evaporated using a rotary evaporator (Rotavapor®, Buchi, China) at 60°C. The dried crude extracts were collected into screw cap containers and stored in a refrigerator at 4°C.
Phytochemical Analysis
Phytochemical analysis of ethanolic and aqueous extracts of P. cablin was conducted to determine their phytoconstituents, including flavonoids, terpenoids, alkaloids, tannins, anthraquinone, cardiac glycosides, saponins and coumarins. This screening was performed according to methods described by Ngbolua et al and Malar et al.18,19
In vitro Plasmodium Falciparum Cultivation
The culture of P. falciparum was performed using the method demonstrated by Trager and Jensen with some modifications.20 The chloroquine-resistant P. falciparum K1 strain, obtained from Dr. Rapatbhorn Patrapuvich, Department of Drug Research Unit for Malaria, Faculty of Tropical Medicine, Mahidol University, Thailand, was cultivated with noninfected type O-positive red blood cells (2% hematocrit) using RPMI-1640 supplemented with 2 mg/ml sodium bicarbonate, 10 μg/ml hypoxanthine (Sigma-Aldrich, New Delhi, India), 4.8 mg/ml HEPES (Himedia, Mumbai, India), 0.5% AlbuMAX II (Gibco, MA, USA) and 2.5 μg/ml gentamicin (Sigma-Aldrich, New Delhi, India). P. falciparum culture was maintained in 5% CO2 at 37°C. The culture medium was changed daily after assessing parasite density by Giemsa staining.
In vitro Assessment of Antimalarial Activity
The antimalarial activity of ethanolic and aqueous extracts of P. cablin was carried out using an in vitro Plasmodium lactate dehydrogenase (pLDH) assay as described by Makler with some modifications.21 Briefly, the crude extracts (500 mg) were dissolved in 1 ml of dimethyl sulfoxide (DMSO) (Merck, Darmstadt, Germany) and adjusted to obtain a 2-fold serial dilution in the range of 9.77 to 2,500 µg/ml. The parasitized red blood cells (2% hematocrit and 2% parasitemia) were added to a 96-well cell culture plate, and then, the infected red cells were exposed to the ethanolic and aqueous P. cablin extracts (final concentration of each extract: 9.77 µg/ml). Wells containing artesunate (Sigma-Aldrich, New Delhi, India) served as a positive control (final concentration ranging from 0.31 to 10 µg/ml). The plates were incubated in a CO2 incubator at 37°C for 72 h, frozen at −20°C and thawed at 37°C 3 times to achieve complete hemolysis. The lysed cells were transferred to a new 96-well plate containing 100 µl of Malstat reagent and 20 µl of nitroblue tetrazolium/phenazine ethosulfate solution (Calbiochem®, Sigma-Aldrich, New Delhi, India) and kept in the dark for 1 h. Optical density was determined at a 650 nm wavelength using a microplate reader. The percentage of inhibition and the half maximal inhibitory concentration (IC50) were determined from a nonlinear dose dependence curve with concentrations ranging from 9.77 to 2,500 µg/ml.
In vitro Assessment of Cytotoxicity
The 3-(4,5-dimethythiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay was used to assess the toxicity of P. cablin. Vero cells (Elabscience, Wuhan, Hubei, China) were cultured in Dulbecco’s modified Eagle’s medium (CaissonLab, Smithfield, UT) supplemented with 10% fetal bovine serum (CaissonLab, Smithfield, UT). Vero cells (104 cells/ml) were added to a 96-well plate and incubated for 24 h at 37°C and 5% CO2. P. cablin ethanolic and aqueous extracts were added to the plates at 5 concentrations ranging from 5 to 80 µg/ml. The wells containing doxorubicin (Sigma-Aldrich, New Delhi, India) were used as a positive control (final concentration: 0.63 µg/ml). The plates were incubated for 48 h at 37°C and 5% CO2. After the incubation period, the MTT solution was added to the plates, which were then incubated in a CO2 incubator for 2 h. The supernatant was removed, and 100 µl of DMSO was added to each well. Finally, the optical density was determined at a wavelength of 590 nm using a microplate reader. The percentage of survival and 50% cytotoxicity concentration (CC50) were determined by using the dose response curve with concentrations ranging from 0.31 to 10 µg/ml.
Animal Experiments
Healthy ICR mice weighing 20-30 g (body weight) were bought from Nomura Siam International Co., Ltd., Pathumwan, Bangkok, Thailand. The mice were randomly grouped and acclimatized in cages for 1 week prior to the experiment. The experimental animal room temperature was 22°C (± 3°C), and the relative humidity was 50-60%. The lighting environment was set to 12-h light/dark cycles, and the mice had free access to food pellets and clean drinking water provided by the animal care staff.
Four-Day Suppressive Test (Peter’s Test)
The assessment of in vivo antimalarial activity was performed using a 4-day suppressive test to evaluate the schizonticidal activity of the extracts.22 Rodent parasites of Plasmodium berghei strain ANKA were obtained from BEI Resources, NIAID, NIH: P. berghei, strain ANKA, MRA-311, contributed by Thomas F. McCutchan. Five groups of ICR mice (5 mice each) were randomly created, including 3 groups of mice treated with 200, 400 and 600 mg/kg body weight ethanolic P. cablin extract according to previous studies23–25 and positive and negative control groups. Briefly, mice were inoculated intraperitoneally with 200 µl of P. berghei ANKA-infected red cells containing 107 cells/ml on day 0. At 4, 24, 48 and 72 h postinfection, each treated group received 3 doses of the extract via oral gavage. Artesunate at a dose of 6 mg/kg body weight and 7% Tween 80 were administered by oral gavage to the positive and negative control groups, respectively. Each mouse was weighed on the first and last days of the experiment by a sensitive digital balance (Mettler Toledo, Switzerland). At 96 h postinfection, thin blood films were prepared and stained with 10% Giemsa solution and then observed under a light microscope at 100× magnification to determine the percentage of parasitemia. The mean parasitemia was calculated from 10 different fields on each slide. The % parasitemia was calculated as follows:
Finally, the following equation was used to calculate the mean percentage of parasitemia suppression:
where A and B represent the mean percentages of negative parasitemia in the control group and the P. cablin-treated group, respectively.
Acute Toxicity Test
Fifteen ICR mice were randomly divided into 3 groups, including mice treated with 2000 mg/kg P. cablin extract and the negative control and untreated groups. The protocol of acute toxicity followed the 2008 OECD guidelines.26 Before the experiment began, the mice were fasted for 3 h and had free access to only clean drinking water. The ethanolic P. cablin extract was dissolved in 7% Tween 80 to make a stock solution with a concentration of 2000 mg/kg. Mice in the experimental group were treated orally with a single dose of the extract at 2000 mg/kg body weight, whereas mice in the control group were given a 7% Tween 80 solution. Mice were observed during the first 30 min after administration. Physical and behavioral changes, including changes in appetite, sleep, abnormal secretion, diarrhea, rigidity and hair erection, were noted for 14 days after administration. On day 14, 60 mg/kg pentobarbital (Nembutal, Ceva, France) was injected intraperitoneally as an anesthetic. Then, blood was collected using the cardiac puncture technique for biochemical analysis. In addition, kidney and liver tissues were processed and fixed in 10% formalin solution for histopathological examination.
Biochemical Analysis
Heparinized blood was centrifuged at 3,000 ×g for 5 min to obtain plasma samples. The samples were analyzed for biochemical parameters of kidney function (including blood urea nitrogen (BUN) and creatinine (Cr)) and liver function (including alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alkaline phosphatase (ALP)) by standard techniques (kinetic methods for AST, ALT, ALP and BUN and a fixed time method for Cr) using an AU480 chemistry analyzer (Beckman Coulter, USA).
Histopathological Examination
Histopathological examination of formalin-fixed kidney and liver tissues was carried out according to a previous study.27 Briefly, kidney and liver tissues were dehydrated with a gradient series of ethanol solutions and rinsed with xylene 3 times. Then, fixed tissues were placed in a mold that contained a small amount of molten paraffin. In the sectioning process, a 5-µm section was sliced from the paraffin blocks of each tissue sample by a manual rotary microtome (Model RM2235, Leica Biosystems, Germany). Then, the sections were floated in a water bath and then transferred to glass slides. For the staining process, the sections were deparaffinized with xylene 3 times for 10 min each, rehydrated with a descending ethanol series of 100%, 96%, 90%, 80%, and 70% for 10 min each and stained with a hematoxylin and eosin (H&E) solution. Then, the sections were dehydrated by increasing the concentrations of ethanol as follows: 70%, 80%, 90%, 96%, and 100%. The sections were then rinsed with xylene and mounted with glass cover slips. Finally, 2 independent observers who were blinded to the group assignments examined the slides under a light microscope.
Statistical Analysis
The data were analyzed using SPSS for Microsoft Windows version 17.0 (SPSS, IL, USA). Normal distribution was determined by the Koimogorov-Smirnov goodness-of-fit test. The statistical significance of parasitemia suppression and the effect of the extract on kidney and liver functions in the acute toxicity test were tested by 1-way ANOVA followed by post hoc Tukey’s multiple comparison test.
Results
Phytochemical Constituents
The ethanolic P. cablin extract exhibited alkaloids, flavonoids, coumarins and tannins, whereas the aqueous extract exhibited alkaloids, tannins, saponins and coumarins (Table 1).
Table 1.
Phytochemical Constituents of P. cablin in Different Solvent Extracts.
| Phytochemical constituents | Ethanolic extract | Aqueous extract |
|---|---|---|
| Flavonoids | + | − |
| Terpenoids | − | − |
| Alkaloids | + | + |
| Tannins | + | + |
| Anthraquinones | − | − |
| Cardiac glycosides | − | − |
| Saponins | − | + |
| Coumarins | + | + |
+: detected; −: not detected.
In vitro Antimalarial Activity and Cytotoxicity
Quantification of antimalarial activity and cytotoxicity was performed by estimating the concentration of extracts that inhibited 50% of the growth of parasites (IC50) and 50% of the viable cells. The ethanolic and aqueous extracts of P. cablin demonstrated antimalarial activity, with IC50 values of 24.49 ± 0.01 and 549.30 ± 0.07 µg/ml, respectively. Artesunate exhibited antimalarial activity with an IC50 value of 1.28 ± 0.71 µg/ml. The cytotoxicity of the extracts was determined with the highest concentration of 80 µg/ml. According to the classification of cytotoxicity,28 the ethanolic and aqueous extracts of P. cablin exhibited no inhibitory effect at a concentration of 80 µg/ml, indicating no cytotoxic effects on Vero cells.
The 4-day Suppressive Test
The ethanolic P. cablin extract exhibited significant suppressive activity against P. berghei in a dose-dependent manner, with 38.41, 45.12 and 89.00% suppression at doses of 200, 400, and 600 mg/kg, respectively, compared to the activity in the negative control group (p < 0.001) (Table 2). The standard drug artesunate at a 6 mg/kg dose exhibited 96.69% suppression of P. berghei parasitemia compared to the activity in the negative control group, as well as significantly greater suppression than that in the other treated groups (p < 0.001) (Table 2).
Table 2.
Parasitemia Suppressive Activity of the Ethanolic P. cablin Extract Against P. berghei.
|
Group
|
Dose (mg/ml) | % Parasitemia | % Suppression |
|---|---|---|---|
| Negative control | - | 52.39 ± 0.39b, c, d, e | - |
| Artesunate | 6 | 1.73 ± 0.67a, c, d, e | 96.69 ± 0.13a, c, d, e |
| Ethanolic extract | 200 | 32.27 ± 0.00a, b, d, e | 38.41 ± 0.00a, b, d, e |
| 400 | 28.75 ± 0.14a, b, c, e | 45.12 ± 0.27a, b, c, e | |
| 600 | 5.76 ± 0.17a, b, c, d | 89.00 ± 0.32a, b, c, d |
Data are presented as the mean ± SEM (n = 5 per group).
a Compared to negative control, bcompared to artesunate, ccompared to 200 mg/kg extract, dcompared to 400 mg/kg extract, ecompared to 600 mg/kg extract, p < 0.001.
In vivo Acute Toxicity Test
Physical and behavioral changes in mice treated with a dose of 2000 mg/kg extract were observed daily during the 14-day follow-up period. Extract-treated mice showed no signs or symptoms of toxicity, such as hair erection, changed feeding activities, vomiting, diarrhea, abnormal secretion, abnormal sleep and excitement, during the experiment. The mean body weight and percent change in body weight of mice treated with 2000 mg/kg extract and 7% Tween 80 were not significantly different compared to those of the untreated control group (p > 0.05). There was no mortality at all dose levels tested. Based on these results, the lethal dose of the extract was found to be greater than 2000 mg/kg body weight.
Effects of Ethanolic P. cablin Extract on Kidney and Liver Functions
The biochemical parameters of creatinine and ALT in mice treated with 2000 mg/kg extract and 7% Tween 80 did not significantly differ from those in the untreated control group (p < 0.05) (Table 3). The levels of AST in mice treated with 7% Tween 80 and 2000 mg/kg extract were significantly increased compared to those in the untreated mice (p < 0.05). The levels of BUN in mice treated with the 2000 mg/kg extract were significantly less than those in the untreated mice (p < 0.05). The levels of AST and BUN in mice treated with 7% Tween 80 were significantly increased compared with those in untreated mice (p < 0.05) (Table 3).
Table 3.
Effect of Ethanolic P. cablin Extract on Kidney and Liver Functions.
| Parameters | Untreated control | 7% Tween 80 | 2000 mg/kg extract |
|---|---|---|---|
| Liver function test | |||
| AST (U/L) | 127.50 ± 28.99 | 200.50 ± 3.53a | 172.50 ± 46.67a |
| ALT (U/L) | 36.00 ± 14.14 | 36.50 ± 9.19 | 39.5 ± 19.09 |
| ALP (U/L) | 84.50 ± 0.70 | 138.00 ± 31.11a, c | 82.50 ± 31.11b |
| Kidney function test | |||
| BUN (mg/dL) | 23.55 ± 0.21 | 21.35 ± 0.49a, c | 20.00 ± 0.14a, b |
| Creatinine (mg/dL) | 0.55 ± 0.06 | 0.54 ± 0.01 | 0.59 ± 0.02 |
Data are presented as the mean ± SEM (n = 5 per group).
a Compared to untreated control, bcompared to 7% Tween 80, ccompared to 2000 mg/kg extract, p < 0.05.
Histopathological Examination of the Kidney and Liver in the Acute Toxicity Test
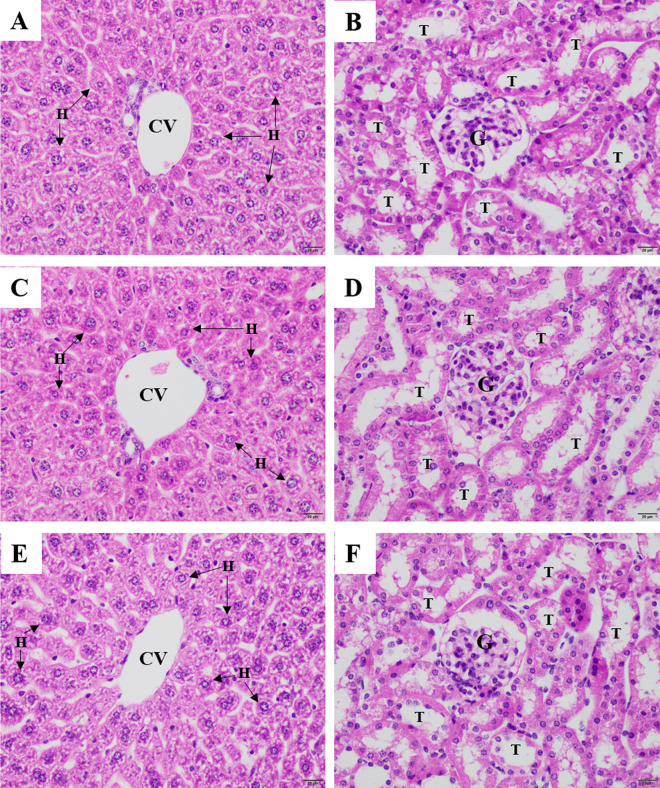
Figure 1 shows the results of the histological examinations of the kidney and liver tissues from mice in the negative control group and from mice treated with 2000 mg/kg body weight ethanolic P. cablin extract. The results demonstrated that mice treated with the 2000 mg/kg body weight extract had normal histological kidney and liver functions compared with those of the negative controls (Figure 1).
Figure 1.
Representative image of H&E staining of the liver and kidneys from the untreated group (A) and (B), the negative control group (C) and (D), and the group treated with 2,000 mg/kg of the ethanolic P. cablin extract (E) and (F). All images were obtained at 400× magnification. Bars = 20 μm. T: tubules, G: glomerulus, CV: central vein, H: hepatocytes.
Discussion
In the extraction processes, the percentage yield was higher for the aqueous extract (13.83%) than for the ethanolic extract (3.45%). The higher yield may be due to the phytoconstituents in the extract being easier to extract in polar solvents such as aqueous solvents.29,30 The in vitro antimalarial activity of ethanolic and aqueous extracts of P. cablin was tested using enzymatic detection of the pLDH enzyme, which is the terminal enzyme in the glycolytic pathway.31 According to a previous study, the in vitro antimalarial activity of extracts was classified as follows: high extract activity at IC50 < 5 µg/ml, promising activity at 5-15 µg/ml, moderate activity at 15-50 µg/ml and inactivity at > 50 µg/ml.32 Our results showed that the ethanolic extract possessed moderate antimalarial activity against the P. falciparum K1 strain with an IC50 value of 24.49 ± 0.01 µg/ml. Phytochemical examination was carried out for the 2 different solvent extracts. The phytochemical constituent analysis of P. cablin revealed alkaloids, tannins and coumarins in both extracts. In contrast, flavonoids were present only in the ethanolic extract, and saponins were present only in the aqueous extract. The absence of terpenoids, anthraquinones and cardiac glycosides was found in both extracts in this study. Our results were similar to those of a previous study that reported chemical constituents from P. cablin.17 Moreover, the ethanolic P. cablin extract has been reported to have 12 secondary metabolites with one important flavonoid compound, tilianin.33 Tilianin is a polyphenol flavonoid compound, and it was previously reported to have pharmacological activities such as antidiabetic, antihyperlipidemic, cardioprotective and anti-inflammatory activities.34–36 Secondary metabolites with pharmacological activity, such as flavonoids and alkaloids, could be accountable for the antimalarial activity.37,38 The potential toxicity of P. cablin extracts was evaluated in Vero cells. According to the cell cytotoxic classification, the CC50 value was used to define the potency of cytotoxicity. A nontoxic effect could be classified as a CC50 value greater than 50 µg/ml.28 Ethanolic and aqueous extracts of P. cablin exhibited a Vero cell survival rate greater than 50% at 80 µg/ml, indicating no cytotoxic effects on Vero cells. This result was consistent with that of a previous study, which reported that the ethanolic P. cablin extract exhibited a noncytotoxic effect, with more than 70% cell viability.39 The selection of the extract with the best antimalarial activity in an animal model that is widely used to predict drug activity was based on the greatest antimalarial activity and the lowest toxicity. According to the results of in vitro studies, the ethanolic P. cablin extract exhibited better antimalarial activity against the P. falciparum K1 strain and had no toxicity toward Vero cells. Therefore, the ethanolic extract was selected for further in vivo antimalarial activity and toxicological assessment in a mouse model. The highest suppression occurred in mice treated with the extract at 600 mg/kg body weight (89.00%), followed by those treated with 400 and 200 mg/kg body weight (45.12 and 38.41%, respectively). These results were consistent with those of a previous study regarding the in vivo antimalarial activity of the Betula alnoides aqueous stem extract, which also exhibited significant suppressive activity against P. berghei in a dose-dependent manner.23 To confirm extract safety, an acute toxicity test was performed by orally administering the extract at a dose of 2000 mg/kg body weight. Manifestations of toxicity or mortality were not observed. Furthermore, biochemical parameters and histopathological changes in the kidney and liver were determined to verify nonvisible toxicity. Biochemical analysis of kidney and liver function is crucial for evaluating the toxicological effects of xenobiotics.40,41 Plasma levels of AST in mice treated with the extract were higher than those in untreated mice, but the levels of ALT and ALP were not different from those in the control mice. Based on these results, ALT is a liver-specific enzyme, rather than AST, and the AST enzyme can be found in many other tissues, such as muscles, and in red blood cells.42 Thus, elevation of AST may occur due to other tissue injuries. This suggestion was confirmed by histopathological changes with normal histological features of the liver. However, the levels of AST and ALP in mice treated with 7% Tween 80 were increased compared with those in the other groups. This evidence might suggest that this vehicle should be used to dissolve crude extracts. For the kidney function test, BUN levels in mice treated with the extract and 7% Tween 80 were statistically less than those of untreated mice; however, this difference may not be clinically significant. In addition, analysis of the histological changes in the kidneys and livers of mice treated with the extract and 7% Tween 80 showed normal features compared to those of control mice. Moreover, the body weights of the treated mice were not different from those of the untreated mice. The observed safety of P. cablin was consistent with a previous study reporting the safety of P. cablin administration with a maximal dose tolerance of 12.5 g/kg.43 Our study confirmed that the ethanolic P. cablin extract exhibited antimalarial activity and no toxic effect on mice at a dose of 2000 mg/kg body weight. Therefore, our study suggests that further work is required to characterize the bioactive molecules in the ethanolic P. cablin extract that are responsible for the activity against malaria parasites.
Conclusions
This is the first report of the in vitro and in vivo antimalarial activities of P. cablin extracts. The ethanolic P. cablin extract possessed antimalarial activity against the malaria parasites P. falciparum K1 and P. berghei ANKA. This study supports the safety of P. cablin as a medicinal plant with no toxicity in vitro and in vivo. Further phytochemical studies on the ethanolic P. cablin extract are recommended to identify the bioactive constituents that can be developed as antimalarial agents.
Acknowledgments
We would like to thank all of the staff at the Animal Experiment Building, Walailak University, for helping us manage the animal experiment. We would also like to thank the staff at the Department of Tropical Pathology, Faculty of Tropical Medicine, Mahidol University, Thailand, for assistance in the tissue processing and histological experiment.
Footnotes
Author Contributions: AP, PC, NH and CP designed the study; AP, PC, NH, SM, KK and CP carried out the experiments; AP drafted the manuscript; PC, NH and CP conducted experiments; AP, SM and KK analyzed the data; PC and CP revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval: Ethical clearance was obtained from the Ethics Committee on Human Rights Related to Research Involving Human Subjects, Walailak University, prior to the recruitment of any participants (Approval number: WUEC-19-122-01). Written informed consent was obtained from all participants before data and sample collection. The animal use protocol was reviewed and approved by the Animal Ethics Committee, Walailak University (Protocol number: WU-AICUC-63001).
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially financially supported by Walailak University. The funder played no role in the study design, data collection, and data analysis and interpretation.
ORCID iD: Chuchard Punsawad, PhD  https://orcid.org/0000-0003-4826-9474
https://orcid.org/0000-0003-4826-9474
References
- 1. Snow RW, Sartorius B, Kyalo D, et al. The prevalence of Plasmodium falciparum in sub-Saharan Africa since 1900. Nature. 2017;550(7677):515–518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. O’Brien SF, Delage G, Seed CR, et al. The epidemiology of imported malaria and transfusion policy in 5 nonendemic countries. Transfus Med Rev. 2015;29(3):162–171. [DOI] [PubMed] [Google Scholar]
- 3. WHO. World Malaria Report. 2019. Geneva, Switzerland. [Google Scholar]
- 4. Cowman AF, Healer J, Marapana D, Marsh K. Malaria: biology and disease. Cell. 2016;167(3):610–624. [DOI] [PubMed] [Google Scholar]
- 5. Luzolo AL, Ngoyi DM. Cerebral malaria. Brain Res Bull. 2019;145:53–58. [DOI] [PubMed] [Google Scholar]
- 6. Hanboonkunupakarn B, White NJ. The threat of antimalarial drug resistance. Trop Dis Travel Med Vaccines. 2016;2(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. World Wide Antimalarial Resistance Network (WWARN) Lumefantrine PK/PD Study Group. Artemether-lumefantrine treatment of uncomplicated Plasmodium falciparum malaria: a systematic review and meta-analysis of day 7 lumefantrine concentrations and therapeutic response using individual patient data. BMC Med. 2015;13:227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. World Wide Antimalarial Resistance Network (WWARN) DP Study Group. The effect of dosing regimens on the antimalarial efficacy of dihydroartemisinin-piperaquine: a pooled analysis of individual patient data. PLoS Med. 2013;10(12):e1001564; discussion e1001564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Adjuik MA, Allan R, Anvikar AR, et al. The effect of dosing strategies on the therapeutic efficacy of artesunate-amodiaquine for uncomplicated malaria: a meta-analysis of individual patient data. BMC Med. 2015;13:66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Nayyar G, Breman J, Newton P, Herrington J. Poor-quality antimalarial drugs in Southeast Asia and sub-Saharan Africa. Lancet Infect Dis. 2012;12:488–496. [DOI] [PubMed] [Google Scholar]
- 11. Hanani E, Hariyanti H, Dayatri DY. Phytochemical identification and antioxidant activity of essential oil of Pogostemon cablin benth. Cultivated in Java Island Indonesia. Int J Pharm. 2019;9(6):e5297. [Google Scholar]
- 12. Ramya HG, Palanimuthu V, Rachna S. An introduction to patchouli (Pogostemon cablin Benth.)—A medicinal and aromatic plant: it’s importance to mankind. Int J Agric Eng. 2013;15(2):243–250. [Google Scholar]
- 13. Swamy M, Sinniah UR. Patchouli (Pogostemon cablin Benth.): Botany, agrotechnology and biotechnological aspects. Industrial Crops and Products. 2016;87:161–176. [Google Scholar]
- 14. Wu H, Li B, Wang X, Jin M, Wang G. Inhibitory effect and possible mechanism of action of patchouli alcohol against influenza A (H2N2) virus. Molecules. 2011;16(8):6489–6501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Abe S, Sato Y, Inoue S, et al. Anti-Candida albicans activity of essential oils including Lemongrass (Cymbopogon citratus) oil and its component, citral. Nihon Ishinkin Gakkai Zasshi. 2003;44(4):285–291. [DOI] [PubMed] [Google Scholar]
- 16. Yang Y, Kinoshita K, Koyama K, et al. Anti-emetic principles of Pogostemon cablin (Blanco) Benth. Phytomedicine. 1999;6(2):89–93. [DOI] [PubMed] [Google Scholar]
- 17. Swamy M, Sinniah U. A comprehensive review on the phytochemical constituents and pharmacological activities of Pogostemon cablin benth.: an aromatic medicinal plant of industrial importance. Molecules. 2015;20:8521–8547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ngbolua K-T-N. Phytochemical screening of some medicinal plants traditionally used by African Women in Kinshasa city (DR Congo) for their intimate hygiene and evaluation of the pH of derived recipes. J Modern Drug Discov Drug Deliv Res. 2014;1:1–7. [Google Scholar]
- 19. Malar G, Chinnachamy C. Phytochemical screening, total flavonoid, total terpenoid and anti-inflammatory activity of aqueous stem extract of Salacia oblonga . J Chem Pharm. 2017;10:550. [Google Scholar]
- 20. Trager W, Jensen JB. Human malaria parasites in continuous culture. 1976. J Parasitol. 2005;91(3):484–486. [DOI] [PubMed] [Google Scholar]
- 21. Makler MT, Hinrichs DJ. Measurement of the lactate dehydrogenase activity of Plasmodium falciparum as an assessment of parasitemia. Am J Trop Med Hyg. 1993;48(2):205–210. [DOI] [PubMed] [Google Scholar]
- 22. Peters W, Portus H, Robinson L. The four-day suppressive in vivo antimalarial test. Ann Trop Med Parasitol. 1995;69:155–171. [Google Scholar]
- 23. Chaniad P, Techarang T, Phuwajaroanpong A, Punsawad C. Antimalarial activity and toxicological assessment of Betula alnoides extract against plasmodium berghei infections in mice. Evid Based Complement Alternat Med. 2019;2019:2324679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Belachew A, Desta A, Tiruneh G. Anti-malarial activity of the root extract of Euphorbia Abyssinia (Euphorbiaceae) against Plasmodium berghei infection in mice. Malar J. 2019;18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Misganaw D, Engidawork E, Nedi T. Evaluation of the anti-malarial activity of crude extract and solvent fractions of the leaves of Olea europaea (Oleaceae) in mice. BMC Complement Altern Med. 2019;19(1):171–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Organisation for Economic Co-operation and Development. Test No. 425: acute oral toxicity: up-and-down procedure. 2008. OECD Publishing, Paris. [Google Scholar]
- 27. Sümer E, Senturk GE, Demirel ÖU, Yesilada E. Comparative biochemical and histopathological evaluations proved that receptacle is the most effective part of Cynara scolymus against liver and kidney damages. J Ethnopharmacol. 2020;249:112458. [DOI] [PubMed] [Google Scholar]
- 28. Berthi W, Gonzalez A, Rios A, Blair S, Cogollo A, Pabon A. Anti-plasmodial effect of plant extracts from Picrolemma huberi and Picramnia latifolia . Malar J. 2018;17(1):151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Razak MFBA, Yong PK, Shah ZM, Abdullah LC, Yee SS, Yaw ITCS. The Effects of varying solvent polarity on extraction yield of Orthosiphon stamineus leaves. J Appl Sci. 2012;12:1207. [Google Scholar]
- 30. Zhang Q-W, Lin L-G, Ye W-C. Techniques for extraction and isolation of natural products: a comprehensive review. Chin Med. 2018;13:20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Makle MT, Hinrichs DJ. Measurement of the lactate dehydrogenase activity of Plasmodium falciparum as an assessment of parasitemia. J Trop Med Hyg. 1993;48(2):205–210. [DOI] [PubMed] [Google Scholar]
- 32. Lusakibanza M, Mesia G, Tona G, et al. In vitro and in vivo antimalarial and cytotoxic activity of five plants used in Congolese traditional medicine. J Ethnopharmacol. 2010;129(3):398–402. [DOI] [PubMed] [Google Scholar]
- 33. Wang D, Yin Z-Q, Zhang Q, Ye W, Zhang X, Zhang J. Nonvolatile chemical constituents from Pogostemon cablin . Zhongguo Zhong Yao Za Zhi. 2010;35(20):2704–2707. [PubMed] [Google Scholar]
- 34. Nam KW, Kim J, Hong JJ, et al. Inhibition of cytokine-induced IkappaB kinase activation as a mechanism contributing to the anti-atherogenic activity of tilianin in hyperlipidemic mice. Atherosclerosis. 2005;180(1):27–35. [DOI] [PubMed] [Google Scholar]
- 35. Yuan Y, Cao W, Hong Y, et al. Tilianin pretreatment prevents myocardial ischemia-reperfusion injury via preservation of mitochondrial function in rat heart. Phytomedicine. 2017;34:106–114. [DOI] [PubMed] [Google Scholar]
- 36. Garcia-Diaz JA, Navarrete-Vazquez G, Garcia-Jimenez S, et al. Antidiabetic, antihyperlipidemic and anti-inflammatory effects of tilianin in streptozotocin-nicotinamide diabetic rats. Biomed Pharmacother. 2016;83:667–675. [DOI] [PubMed] [Google Scholar]
- 37. Amoa Onguéné P, Ntie-Kang F, Lifongo LL, Ndom JC, Sippl W, Mbaze LMA. The potential of anti-malarial compounds derived from African medicinal plants. Part I: a pharmacological evaluation of alkaloids and terpenoids. Malar J. 2013;12(1):449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Rudrapal M, Chetia DD. Plant flavonoids as potential source of future antimalarial leads. Sys Rev Pharm. 2016;8:13–18. [Google Scholar]
- 39. Dechayont B, Ruamdee P, Poonnaimuang S, Mokmued K, Chunthorng-Orn J. Antioxidant and antimicrobial activities of Pogostemon cablin (Blanco) Benth. J Botany. 2017;2017:8310275. [Google Scholar]
- 40. Bariweni M, Yibala O, Ozolua R. Toxicological studies on the aqueous leaf extract of Pavetta crassipes (K. Schum) in rodents. J Pharm Pharmacogn Res. 2018;6:1–16. [Google Scholar]
- 41. Ezeja MI, Anaga AO, Asuzu IU. Acute and sub-chronic toxicity profile of methanol leaf extract of Gouania longipetala in rats. J Ethnopharmacol. 2014;151(3):1155–1164. [DOI] [PubMed] [Google Scholar]
- 42. Zainul N, Zakaria Y, Nik Hassan NF, Muhammad H. Phytochemicals and acute oral toxicity studies of the aqueous extract of Vernonia amygdalina from state of Malaysia. 2016;4. [Google Scholar]
- 43. He J, Peng S, Xie Q, et al. Acute toxicity of patchouli alcohol. Lishizhen Med Mater Med Res. 2012;23(2):274–275. [Google Scholar]