Objective:
To determine the risk of acquiring perioperative COVID-19 infection in previously COVID-19 negative patients.
Summary of Background Data:
During the initial peak of the COVID-19 pandemic, there was significant concern of hospital acquired COVID-19 infections. Medical centers rapidly implemented systems to minimize perioperative transmission, including routine preoperative testing, patient isolation, and enhanced cleaning.
Methods:
In this retrospective cohort study, medical records of all adult patients who underwent surgery at our quaternary, acute care hospital between March 15 and May 15, 2020 were reviewed. The risk of preoperatively negative patients developing symptomatic COVID-19 within 2–14 days postoperatively was determined. Surgical characteristics, outcomes, and complications were compared between those with and without acquired perioperative COVID-19 infection.
Results:
Among 501 negative patients undergoing index surgeries, 9 (1.8%) developed symptomatic COVID-19 in the postoperative period; all occurred before implementation of routine preoperative testing [9/243, 3.7% vs 0/258, 0%, odds ratio (OR): 0.048, P = 0.036]. No patient who was polymerase-chain-reaction negative on the day of surgery (n = 170) developed postoperative infection. Perioperative infection was associated with preoperative diabetes (OR: 3.70, P = 0.042), cardiovascular disease (OR: 3.69, P = 0.043), angiotensin receptor blocker use (OR: 6.58, P = 0.004), and transplant surgery (OR: 11.00, P = 0.002), and multiple complications, readmission (OR: 5.50, P = 0.029) and death (OR: 12.81, P = 0.001).
Conclusions:
During the initial peak of the COVID-19 pandemic, there was minimal risk of acquiring symptomatic perioperative COVID-19 infection, especially after the implementation of routine preoperative testing. However, perioperative COVID-19 infection was associated with poor postoperative outcome.
Keywords: COVID-19, nosocomial infection, surgical complications
The coronavirus disease 2019 (COVID-19) pandemic is an unprecedented global crisis with significant ramifications for the healthcare system. Ominously, surge modeling forecasted inadequate medical equipment, supplies, and staffing to address the expected needs of seriously ill, ventilator dependent, COVID-19 infected patients. Without drastic reduction in surgical services, life-threatening shortages of personal protective equipment, ventilators, and hospital beds were expected.1–3 Furthermore, early reports suggested that patients with perioperative COVID-19 infection undergoing surgery had significantly worse outcomes.4,5 For the elective surgical patient, there was concern that uninfected patients undergoing surgery might contract the SARS-CoV-2 virus perioperatively. In the face of logistical challenges, resource limitations, and uncertain risk-benefit relationships, governments, hospitals, and surgical specialty societies urged the delay of elective cases.6
As the virus reached the United States, New York City became the epicenter of the COVID-19 pandemic. New York hospitals rapidly implemented systems, based on limited empirical evidence, to enhance safety and minimize surgical risks. At Columbia University, all elective surgeries were canceled. In addition to reducing case volume and performing only urgent and emergent operations, hospitals isolated patients based on COVID-19 status, reorganized dedicated perioperative areas, made the operating room a negative pressure environment, and increased infection prevention and control practices, including COVID-specific operating room and equipment cleaning.1–3,7 As faster and more reliable testing became more widely available, many centers implemented various forms of routine preoperative testing to delay cases in positive patients, when feasible, or isolate them and take special precautions when surgery could not be delayed.8–10 However, the actual risk of patients becoming newly infected in the perioperative period and the efficacy of these measures aimed at reducing transmission remain unknown.
In this study, we investigate the risk of acquiring symptomatic perioperative COVID-19 infection during the initial peak of the pandemic and the impact of routine preoperative COVID-19 testing on this risk. In the cohort of patients with perioperative COVID-19 infection, we assess the association of patient and surgical risk factors in developing the infection, and the impact on postoperative outcomes and adverse events.
METHODS
Setting and Participants
In this retrospective cohort study, we reviewed the medical records of all adult patients who underwent surgery at New York-Presbyterian Hospital—Columbia University Irving Medical Center, a quaternary, acute care hospital in northern Manhattan, between March 15 and May 15, 2020. During this period, surgeries were limited to emergent or urgent cases that were triaged by a multi-disciplinary hospital committee or the operating room medical director (AKL). Three to 5 operating rooms and an adjacent combined preoperative and recovery room were designated for surgical care. Postoperatively, COVID-19 negative patients were segregated from positive patients in the hospital when possible. Suspected or confirmed preoperative COVID-19 patients were isolated. Hospital census data (Fig. 1) of confirmed inpatient COVID-19 hospitalizations showed a rise in cases throughout the study period until a peak around the week of April 13 before gradually declining through May 15, when there were still over 200 inpatient cases. This study was approved by the Columbia University Institutional Review Board (#AAAT0618).
FIGURE 1.
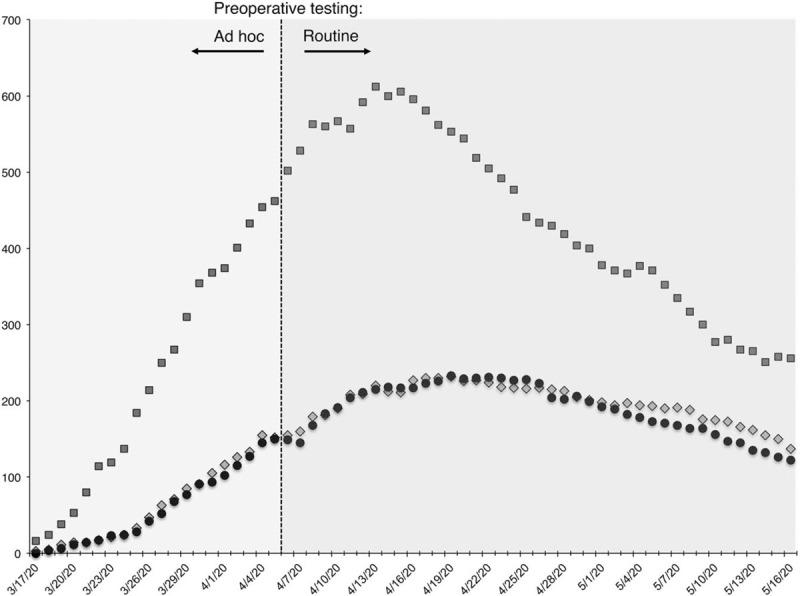
Daily counts of confirmed COVID-19 cases at NYP/CUIMC between March 15 and May 15, 2020. X-axis represents data, Y-axis represents raw count. All cases were confirmed by SARS-CoV-2PCR testing. Squares = Total inpatients; Circles = ICU inpatients; Diamonds = Patients requiring mechanical ventilation. The periods before and after routine preoperative testing took place are indicated. COVID-19 indicates coronavirus disease 2019; CUIMC, Columbia University Irving Medical; ICU, intensive care unit; NYP, New York-Presbyterian.
Preoperative testing for the SARS-COV-2 virus was performed on nasopharyngeal swab specimens with an in-house reverse-transcriptase–polymerase-chain-reaction (PCR) assay. We defined preoperative test as any test within 3 days of the index surgery and excluded patients with earlier test-confirmed COVID-19 infection (Fig. 2). Between March 15th and April 6th, preoperative testing was performed based on clinical indication and availability; after April 6, preoperative testing was mandated for all patients undergoing surgery.
FIGURE 2.
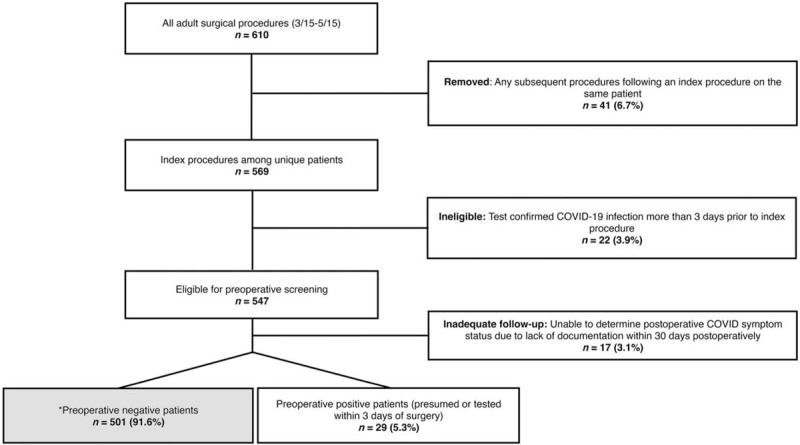
Eligibility for preoperative SARS-CoV-2 testing. ∗ = Preoperative negative patients (n = 501), forming the cohort for determination and analysis of perioperative COVID-19. COVID-19 indicates coronavirus disease 2019.
Preoperatively, patients were presumed positive without a test if they had (i) documentation of 2 or more of 7 core Centers for Disease Control and Prevention defined symptoms11 in the 2 weeks preceding surgery, or (ii) symptom onset and a positive test by postoperative day 1; otherwise they were presumed negative. Postoperatively, patients were determined to have acquired symptomatic COVID-19 if: (i) they had a negative test within 3 days of surgery or were presumed negative preoperatively; and, (ii) they had documentation of 2 or more Centers for Disease Control and Prevention defined symptoms and clinical suspicion for COVID infection occurring in the 2 to 14 days after surgery, with or without a test. Patients without any documented clinical events or encounters after surgery allowing for adequate determination of COVID status based on symptoms were excluded.
Variables and Data Collection
Patient data were manually abstracted from the electronic medical record, including patient characteristics (age, sex, select comorbidities, and medications), surgery characteristics (surgical service, case length, case urgency, anesthesia type), and clinical course (pre- and postoperative length of stay (LOS), intensive care unit admission, adverse events, readmission, and mortality) based on American College of Surgeons National Surgical Quality Improvement Project (NSQIP) definitions12 for a follow-up period of 30 days postoperatively. Pre- and postoperative COVID-19 testing and clinical symptom documentation were also abstracted from the record.
Statistical Methods
Data are presented as mean ± standard deviation or median [interquartile range (IQR)] for continuous variables and frequency (percentage) for categorical variables. For infected patients, the NSQIP surgical risk calculator was used to calculate expected mortality and LOS for comparison to actual outcomes. Univariate analyses were conducted via Firth logistic regression with Bonferroni correction for multiple comparisons to determine patient and surgery characteristics and postoperative outcomes associated with perioperative COVID-19. All analyses were performed in Stata/IC.13
RESULTS
Between March 15 and May 15, there were 610 procedures performed on 569 patients; 22 (3.9%) had confirmed preoperative COVID-19 more than 3 days before their index surgery and were excluded (Fig. 2). Among the remaining 547 patients who were eligible for preoperative PCR test within 3 days of surgery, 29 (5.3%) tested or were suspected positive preoperatively and were thus excluded from further analysis. 17 patients (3.1%) lacked adequate postoperative follow-up for determination of COVID status and were also excluded. Patient and surgical characteristics, and outcomes and adverse events, for the 501 preoperative presumed or test negative patients are presented in Table 1. Common comorbidities included hypertension (52.5%), diabetes mellitus (25.4%), immunocompromised state (20.2%), heart failure, arrhythmia or valvular disease (20.0%), and cardiovascular disease (18.6%). Of the 14 surgical services represented, general surgery accounted for a plurality of cases (31.5%), followed by vascular surgery (10.7%), obstetrics/gynecology (9.4%), orthopedic surgery (8.8%), and neurological surgery (8.8%). Approximately one-quarter of cases (25.7%) were emergent. Median postoperative LOS was 2 days (IQR: 0–5 days); 107 patients (21.4%) required a postoperative intensive care unit stay.
TABLE 1.
Clinical Characteristics, Surgical Details, and Postoperative Course of Patients Without Preoperative COVID-19 (n = 501)
| Sex, n (%) | |
| Male | 229 (45.7) |
| Female | 272 (54.3) |
| Age, mean ± SD | 56.6 ± 16.9 |
| 18–44, n (%) | 129 (25.7) |
| 45–64, n (%) | 192 (38.3) |
| 65–74, n (%) | 102 (20.4) |
| 75 or older, n (%) | 78 (15.6) |
| Body mass index, mean ± SD | 27.0 ± 6.3 |
| <18.5 (underweight), n (%) | 27 (5.4) |
| 30–40 (obese), n (%) | 118 (23.7) |
| 40 or greater (morbid obesity), n (%) | 21 (4.2) |
| Comorbidities, n (%) | |
| Diabetes mellitus | 127 (25.4) |
| Dialysis | 33 (6.6) |
| Hypertension | 263 (52.5) |
| Heart disease (CHF, valvular, arrhythmia) | 100 (20.0) |
| Other cardiovascular disease (CAD, MI, PAD) | 93 (18.6) |
| Lung disease or moderate-to-severe asthma | 70 (14.0) |
| Liver disease | 38 (7.6) |
| Immunocompromised | 101 (20.2) |
| Medications, n (%) | |
| Angiotensin-converting enzyme inhibitors | 53 (10.6) |
| Angiotensin receptor blockers | 58 (11.6) |
| Immunosuppressants | 70 (14.0) |
| Surgical service | |
| Cardiac surgery | 41 (8.2) |
| Gastroenterology | 1 (0.2) |
| General surgery | 158 (31.5) |
| Neurological surgery | 44 (8.8) |
| Obstetrics/gynecology | 47 (9.4) |
| Ophthalmology | 1 (0.2) |
| Oral/maxillofacial surgery | 4 (0.8) |
| Orthopedic surgery | 44 (8.8) |
| Otolaryngology | 19 (3.8) |
| Plastic surgery | 6 (1.2) |
| Thoracic surgery | 41 (8.2) |
| Transplant surgery | 16 (3.2) |
| Urology | 25 (5.0) |
| Vascular surgery | 54 (10.7) |
| Case urgency, n (%) | |
| Emergent | 129 (25.7) |
| Urgent | 372 (74.3) |
| Anesthesia type, n (%) | |
| General | 401 (80.0) |
| Local or regional | 100 (20.0) |
| Case length, min, median (IQR) | 108 (58–194) |
| Preoperative length of stay, d, median (IQR) | 0 (0–1) |
| Postoperative length of stay, d, median (IQR) | 2 (0–5) |
| Postoperative steroids, n (%) | 65 (13.0) |
| Postoperative complications and outcomes, n (%) | |
| Intensive care unit | 107 (21.4) |
| Mechanical ventilation | 42 (8.4) |
| Sepsis | 28 (5.6) |
| Pressors | 58 (11.6) |
| Cardiac complication | 25 (5.0) |
| Pneumonia | 32 (6.4) |
| Surgical site infection | 12 (2.4) |
| Urinary tract infection | 21 (4.2) |
| Venous thromboembolism | 11 (2.2) |
| Renal failure | 33 (6.6) |
| Discharge to rehabilitation or nursing home | 45 (9.3) |
| Readmission | 38 (7.9) |
| Return to operating room | 31 (6.2) |
| Mortality | 14 (2.8) |
CAD indicates coronary artery disease; CHF congestive heart failure; COVID-19, coronavirus disease 2019; IQR, interquartile range; MI, myocardial infarction; PAD, peripheral arterial disease; SD, standard deviation.
Rates of testing and new perioperative infection are presented in Table 2. Among 501 preoperative negative patients, 9 (1.8%) developed symptomatic COVID-19 postoperatively. All suspected cases were ultimately confirmed by PCR. During the period of ad hoc preoperative testing before April 6th, only 24 patients (9.3%) had testing within 3 days of surgery, whereas after routine preoperative testing was implemented, 97.8% were tested. With ad hoc testing, 3.5% had testing on the day of surgery, whereas with routine testing 62.3% were tested on the day of surgery. All new cases occurred in patients who had surgery before routine preoperative testing (9/243, 3.7%) while none occurred after routine testing was implemented [0/258, 0%, odds ratio (OR): 0.048, 95% confidence interval (CI): 0.003–0.825, P = 0.036]. No patient who was PCR negative on the day of surgery (n = 170) acquired perioperative COVID-19. Of note, 6 of the 9 new perioperative cases occurred after surgery performed very early in the study period (between March 15th and 23rd).
TABLE 2.
Perioperative COVID-19 in Eligible Patients Before and After Implementation of Routine Preoperative Screening
| No. (%) | |||
| Ad Hoc Testing | Routine Screening | Total Study Period | |
| (March 15–April 5) | (April 6–May 15) | (March 15–May 15) | |
| (n = 257) | (n = 273) | (n = 530) | |
| Not tested preop / Eligible | 233/257 (90.7) | 6/273 (2.2) | 227/530 (42.8) |
| Postop + / Preop – | 7/221 (3.2) | 0/7 (0.0)∗ | 7/228 (3.1) |
| Tested 1–3 days preop / Eligible | 15/257 (5.8) | 97/273 (35.5) | 112/530 (21.1) |
| Postop + / Preop – | 2/14 (14.3) | 0/89 (0.0) | 2/103 (1.9) |
| Tested same day preop / Eligible | 9/257 (3.5) | 170/273 (62.3) | 179/530 (33.8) |
| Postop + / Preop – | 0/8 (0.0) | 0/162 (0.0) | 0/170 (0.0) |
| Total postop + / Total preop – | 9/243 (3.7)† | 0/258 (0.0)† | 9/501 (1.8) |
Eligibility for screening was defined as absence of positive testing >3 d preoperatively. There were 12 presumed preoperative positive cases, as determined by 2 or more CDC symptoms of COVID-19, all of which occurred before implementation of routine preoperative screening. In practice, all postoperative positive patients were confirmed by SARS-CoV-2 PCR testing.
One patient was tested on the day of surgery but had an invalid specimen; the patient tested negative on repeat postoperative day 1 and was presumed preoperative negative.
OR: 0.048, 95% CI: 0.003–0.825, P = 0.036.
CDC indicates Centers for Disease Control and Prevention; COVID-19, coronavirus disease 2019.
The hospital courses, and clinical and surgical details, of the 9 patients with perioperative COVID-19 are presented in Figure 3 and Table 3. Of note, 5 of 9 patients had symptom onset by postoperative day 2. Six patients had hypertension, 5 diabetes, and 4 cardiovascular disease. Four patients were taking angiotensin-receptor-blockers (ARBs). Postoperatively, the median LOS was 6 days with a range of 0–20 days. Three patients required intensive care, 2 developed pneumonia, 2 required mechanical ventilation, 2 developed sepsis requiring vasopressors, and 2 expired. NSQIP surgical risk predictions12 were available for 8 of the 9 procedures. Median postoperative LOS was 4.5 days (IQR: 0.25–7.5) versus median expected LOS of 2.75 days (IQR: 1.25–3.875). One of these 8 patients (12.5%) died as compared to a cumulative expected mortality risk of 2.8%.
FIGURE 3.
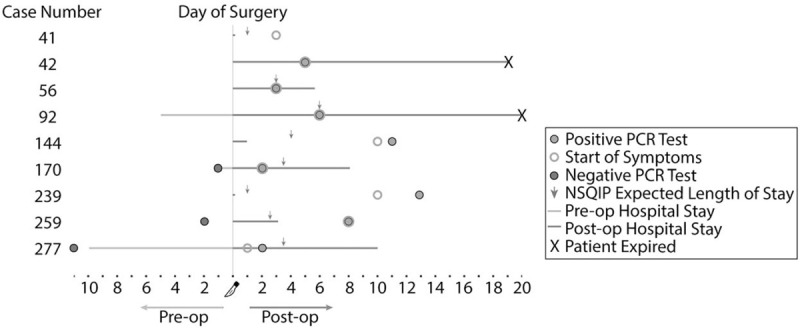
Swimmer plot for patients acquiring symptomatic perioperative COVID-19 infection. Expected length of stay is calculated from the ACS NSQIP Surgical Risk Calculator. ACS NSQIP indicates American College of Surgeons National Surgical Quality Improvement Project; COVID-19, coronavirus disease 2019.
TABLE 3.
Clinical Course and Surgical Characteristics of Patients Acquiring Symptomatic Perioperative COVID-19 Infection
| Case Number | Date of Surgery | Surgery | Urgency | Comorbidities | Chance Serious Complication | Mortality Risk | Clinical Summary |
| 41 | 3/17/20 | Laparoscopic cholecystectomy | Emergent | None | 1.7% | 0.0% | 44F. Uncomplicated procedure for gallstone pancreatitis. Discharged POD0. POD7: outpatient fever and sore throat. No further sequalae. |
| 42 | 3/17/20 | Deceased donor kidney transplant | Urgent | DM, ESRD, HTN, CVD, heart transplant | N/A | N/A | 61M. Uncomplicated procedure. POD5: inpatient respiratory decompensation. Admitted to the ICU POD8 with course complicated by mechanical ventilation, sepsis requiring pressors, renal failure, and UTI. Died POD19. |
| 56 | 3/17/20 | Partial hepatectomy for live donor liver transplant | Urgent | None | 5.8% | 0.0% | 25F. Uncomplicated procedure. POD3: inpatient sore throat and positive test. No further sequalae. |
| 92 | 3/18/20 | Coronary artery bypass graft | Urgent | DM, HTN, CVD | 12.5% | 0.7% | 57M. Triple-vessel CABG complicated by intraoperative MI. Postoperative ICU course complicated by ischemic stroke and COVID pneumonia (POD6), leading to respiratory decompensation and death POD20. |
| 144 | 3/20/20 | Uterine dilation and curettage | Emergent | DM, Obesity, HTN, CVD | 5.7% | 0.2% | 74F. Uncomplicated procedure for bleeding. Discharged POD1. POD10: outpatient shortness of breath, nausea, vomiting, and fatigue. Presented to outside ED POD11 where COVID was confirmed, with no further respiratory sequalae. |
| 170 | 3/23/20 | Transphenoidal excision of pituitary neoplasm | Emergent | Obesity, HTN | 6.3% | 0.1% | 66M. Uncomplicated procedure for pituitary macroadenoma. POD2: febrile, chills, fatigue and positive test. Discharged POD7 with no further sequalae. |
| 239 | 3/31/20 | Chemotherapy port placement | Urgent | DM, HTN, Liver disease | 12.0% | 0.8% | 75M. Uncomplicated ambulatory procedure. POD10: outpatient fatigue. POD13: outside hospital positive test and bacteremia with unclear length of stay, but discharged with repeat negative testing POD29. Improved clinically, and was able to resume chemotherapy treatment. |
| 259 | 4/1/20 | Creation of arteriovenous fistula | Urgent | DM, Obesity, HTN | 10.5% | 1.0% | 49M with new ESRD and volume overload, requiring fistula for urgent hemodialysis. Uncomplicated course. POD8: presented to the ED with fever, cough, and shortness of breath Prolonged hospitalization due to cardiac, renal, and neurologic complications. Discharged POD22. |
| 277 | 4/4/20 | Uterine dilation and evacuation | Emergent | Chronic lung disease, sickle cell disease | 3.4% | 0.0% | 33F, 14-wk gravid, requiring surgical abortion for high-risk pregnancy. Uncomplicated procedure. Overnight POD1 hypotensive and chest X-ray concerning for pneumonia. Confirmed COVID POD2. Discharged POD10 with no further sequalae. |
Chance serious complication and mortality risk are derived from the ACS NSQIP surgical risk calculator.
CVD indicates cardiovascular disease; DM, diabetes mellitus; F, female; HTN, hypertension; ICU, intensive care unit; M, male; POD, postoperative day; UTI, urinary tract infection.
In univariate analysis, several preoperative characteristics were associated with perioperative COVID-19 infection (Table 4) including transplant surgery (OR: 11.00, 95% CI: 2.40–50.48, P = 0.002), use of ARBs (OR: 6.58, 95% CI: 1.84–23.61, P = 0.004), diabetes mellitus (OR: 3.67, 95% CI: 1.05–13.06, P = 0.042), and cardiovascular disease (OR: 3.69, 95% CI: 1.04–13.09, P = 0.043). After correction for multiple comparisons, only transplant surgery remained significant. Perioperative COVID-19 infection was also associated with multiple postoperative outcomes (Table 4) including readmission (OR: 5.50, 95% CI: 1.19–25.46, P = 0.029) and mortality (OR: 12.81, 95% CI: 2.75–59.62, P = 0.001), and numerous adverse events including pneumonia, sepsis, renal failure, cardiac complication, renal failure, and urinary tract infection. After correction for multiple comparisons, only pneumonia and mortality remained significant.
TABLE 4.
Univariate Comparisons of Preoperative Clinical Characteristics and Surgical Details, and Postoperative Course, Complications, and Outcomes, of Patients With Perioperative COVID-19 Infection
| Odds Ratio (95% CI) | P-value | |
| Female sex | 0.68 (0.19–2.41) | 0.554 |
| Age | 0.99 (0.95–1.03) | 0.595 |
| BMI ≥30 | 1.39 (0.37–5.18) | 0.623 |
| Comorbidities | ||
| Diabetes mellitus | 3.70 (1.05–13.06) | 0.042 |
| Dialysis | 2.50 (0.43–14.71) | 0.311 |
| Hypertension | 1.70 (0.46–6.30) | 0.429 |
| Heart disease (CHF, valvular, arrhythmia) | 0.70 (0.12–4.01) | 0.687 |
| Other cardiovascular disease (CAD, MI, PAD) | 3.69 (1.04–13.09) | 0.043 |
| Lung disease or moderate-to-severe asthma | 2.07 (0.48–8.84) | 0.328 |
| Liver disease | 2.14 (0.37–12.55) | 0.398 |
| Immunocompromised | 1.32 (0.31–5.61) | 0.708 |
| Medications | ||
| Angiotensin-converting enzyme inhibitors | 1.48 (0.25–8.60) | 0.662 |
| Angiotensin receptor blockers | 6.58 (1.84–23.61) | 0.004 |
| Immunosuppressants | 1.07 (0.19–6.20) | 0.937 |
| Surgical service | ||
| Cardiac surgery | 1.97 (0.34–11.52) | 0.451 |
| General surgery | 0.65 (0.17–3.04) | 0.651 |
| Neurosurgery | 1.82 (0.31–10.64) | 0.504 |
| Obstetrics/gynecology | 3.28 (0.76–14.17) | 0.112 |
| Oral/maxillofacial surgery | 5.71 (0.29–113.78) | 0.254 |
| Orthopedic surgery | 0.53 (0.03–9.27) | 0.664 |
| Otolaryngology | 1.28 (0.07–22.76) | 0.867 |
| Plastic surgery | 3.94 (0.21–75.06) | 0.362 |
| Thoracic surgery | 0.57 (0.03–10.01) | 0.703 |
| Transplant surgery | 11.00 (2.40–50.48) | 0.002∗ |
| Urology | 0.96 (0.05–17.04) | 0.981 |
| Vascular surgery | 1.45 (0.25–8.42) | 0.679 |
| Emergent case | 2.40 (0.68–8.47) | 0.175 |
| Local or regional anesthesia | 1.34 (0.31–5.68) | 0.696 |
| Case length | 1.00 (1.00–1.01) | 0.814 |
| Preoperative length of stay | 1.02 (0.98–1.05) | 0.315 |
| Postoperative length of stay | 1.04 (0.99–1.09) | 0.170 |
| Postoperative steroids | 2.25 (0.53–9.66) | 0.274 |
| Intensive care unit | 2.00 (0.54–7.47) | 0.302 |
| Mechanical ventilation | 3.72 (0.86–16.15) | 0.070 |
| Sepsis | 5.87 (1.33–25.88) | 0.019 |
| Pressors | 2.58 (0.60–11.07) | 0.203 |
| Cardiac complication | 6.66 (1.50–29.55) | 0.013 |
| Pneumonia | 20.69 (5.62–76.21) | <0.001∗ |
| Surgical site infection | 2.02 (0.11–36.72) | 0.634 |
| Urinary tract infection | 8.09 (1.80–36.32) | 0.006 |
| Venous thromboembolism | 2.20 (0.12–40.18) | 0.594 |
| Renal failure | 4.88 (1.12–21.37) | 0.035 |
| Discharge to rehabilitation or nursing home | 2.24 (0.37–13.56) | 0.381 |
| Readmission | 5.50 (1.19–25.46) | 0.029 |
| Return to operating room | 0.77 (0.04–13.55) | 0.859 |
| Mortality | 12.81 (2.75–59.62) | 0.001∗ |
Bold indicates P < 0.05. Asterisk indicates statistical significance after Bonferroni correction.
BMI indicates body mass index; CAD, coronary artery disease; CHF, congestive heart failure; CI, confidence interval; MI, myocardial infarction; PAD, peripheral arterial disease.
DISCUSSION
The specter of hospital acquired COVID-19 infection is of significant concern to both clinicians and patients. At a Spanish tertiary care center, between February-March 2020, 24.4% of operating room staff tested positive for COVID-19 despite 68% reduction in the number of emergency surgeries.14 Similarly, in Wuhan, China, between January-February 2020, absence of quarantine and personal protection and a “super spreader” was suspected of transmitting COVID-19 to patients and staff in a thoracic surgery department.15 In the community, patients have delayed seeking medical care due to worries regarding nosocomial COVID-19 infection. Studies from Spain and Hong Kong have reported a decline in patients undergoing cardiac diagnostic and therapeutic procedures, and an increase in median time from onset of myocardial infarction symptoms to obtaining medical care, respectively.16,17 This delay or avoidance of necessary health care during the COVID-19 outbreak may be responsible for the observed excess in non-COVID-19 related mortality.
Our study, from a New York City hospital at surge capacity during its initial COVID-19 peak, should serve to allay the fear of nosocomial COVID-19 for surgical patients. We found that only 1.8% of surgical patients, who were preoperatively either asymptomatic and thus presumed COVID-19 negative or tested negative, developed symptomatic COVID-19 infection in the postoperative period. All cases of perioperative infection occurred before the implementation of routine preoperative testing. More than half of these patients developed symptoms within 2 days after surgery, suggesting that their infection may have occurred preoperatively. Consistent with this suggestion, none of the patients who were PCR negative on the day of surgery developed COVID-19 postoperatively. Importantly, the trend in daily confirmed case count in the borough of Manhattan paralleled inpatient COVID-19 census at our hospital, peaking on March 30th, suggesting that the low rate of perioperative infection occurred despite considerable community transmission.18
Risk factors for acquiring COVID-19 with surgery included diabetes, cardiovascular disease, ARB use, and undergoing transplant surgery. The role of ARB use in promoting COVID-19 infection is controversial,19,20 while transplant surgery has been associated with postoperative infection in case reports.21 Diabetes and cardiovascular disease have been more firmly linked to severe presentations in nonsurgical populations.22 Of note, case length was not significantly associated with perioperative infection, suggesting that length of operating room exposure did not increase risk of transmission.
For patients acquiring perioperative COVID-19, postoperative LOS, and mortality rate both exceeded expected values. Surgery with acute COVID-19 infection is known to be associated with greater morbidity and mortality. A recent international multicenter cohort study of surgical patients with perioperative SARS-Co-V-2 infection reported a 30-day mortality rate of 26.4% and 55.5% had pulmonary complications; in general, mortality was higher in older males undergoing emergent major surgeries.23 Similarly, in our series, the 2 deaths occurred in patients with multiple co-morbidities, high preoperative non-COVID-19 expected mortality, and risk factors for severe COVID-19. When deconstructed by individual clinical course, patients who acquired infection divided roughly into 2 groups: rapid decompensation and benign upper respiratory symptoms. This dichotomy of outcomes is consistent with SARS-CoV-2 virus symptom epidemiology, as most patients with symptomatic disease experience benign courses, while a subset of patients experience severe complications.24
A variety of factors likely contributed to the low rate of hospital acquired postoperative COVID-19 seen in our series. These include, but are not limited to, reduction in surgical volume, separation of COVID-19 “+” and “–” patients, use of personal protective equipment for patients and staff, use of viral filters in anesthesia machines, enhanced environmental disinfection, and a “no visitor” policy. Of note, all perioperative infections were restricted to surgeries occurring before the implementation of routine preoperative testing. Thus, routine preoperative testing may have contributed to the low incidence of new postoperative infection. This is plausible as routine preoperative testing identified infected patients for isolation and guided special precautions, which may have further reduced perioperative transmission. Alternatively, preoperative testing may have simply allowed more accurate classification of COVID-19 status. Therefore, it is likely that the observed 1.8% infection rate may ultimately be an overestimate, as early cases before preoperative testing became routine may have gone undetected until the postoperative period.
The relatively low rate of perioperative infection reported herein may serve to increase public confidence in seeking necessary medical care in controlled hospital environments, especially as the rate of community transmission declines over time. Public confidence in the safety of medical centers is instrumental in reducing preventable death and disability from non-COVID-19 medical emergencies during the pandemic. It is also critical in ensuring that preventative screenings and routine care can resume safely in the future, regardless of fluctuations in transmission before widespread vaccine availability. With nearly half of Americans polled recently indicating that someone in their household has delayed seeking care due to the pandemic and that 11% of those experienced a worsening of their condition,25 it is clear that institutions must act quickly to prevent any further harm.
This study is limited by the absence of postoperative SARS-CoV-2 virus testing of all patients and reliance on retrospective chart review to identify symptomatic COVID-19 both before and after surgery for encounters documented within our electronic medical record system; these shortcomings could lead to an underestimate of the true incidence of nosocomial COVID-19 with surgery, though our study likely captured cases rising to the level of clinical significance. Conversely, the possibility of preoperative asymptomatic infection and false negative preoperative testing could artificially elevate our rate of newly acquired postoperative disease, particularly during the period before routine testing. Additionally, due to the small number of new perioperative infection, the study had limited power to identify associated risk factors and outcomes; similarly, multivariable regression was not feasible. The low volume of surgery during this period may limit generalizability as case volume increases. Prospective studies are needed to better determine the true rate of nosocomial COVID-19 and impact of preventative interventions.
In summary, there was minimal risk of acquiring symptomatic perioperative COVID-19 infection during the initial peak of the COVID-19 pandemic. Risk factors for acquiring COVID-19 with surgery included diabetes, cardiovascular disease, ARB use, and undergoing transplant surgery. Perioperative COVID-19 infection was associated with poor postoperative outcome.
Footnotes
Author contributions: BY, AL, and GM conceived of the study. LA, RC, TC, GW, DB, and BY performed data extraction and analysis. CC performed statistical analysis. BY, AL, and GM oversaw reporting of results. All authors discussed findings and contributed to the manuscript.
Mr. Axiotakis and Dr. Youngerman share first authorship for this work.
Drs. Lalwani and McKhann share senior authorship for this work.
The authors report no conflicts of interest.
REFERENCES
- 1.Ross SW, Lauer CW, Miles WS, et al. Maximizing the calm before the storm: tiered surgical response plan for novel coronavirus (COVID-19). J Am Coll Surg 2020; 230:1080–1091.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brindle ME, Gawande A. Managing COVID-19 in surgical systems. Ann Surg 2020; 272:e1–e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Argenziano M, Fischkoff K, Smith CR. Surgery scheduling in a crisis. N Engl J Med 2020; 382:e87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nahshon C, Bitterman A, Haddad R, et al. Hazardous postoperative outcomes of unexpected COVID-19 infected patients: a call for global consideration of sampling all asymptomatic patients before surgical treatment. World J Surg 2020; 44:2477–2481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Knisely A, Zhou ZN, Wu J, et al. Perioperative morbidity and mortality of patients with COVID-19 who undergo urgent and emergent surgical procedures. Ann Surg 2020; In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coccolini F, Perrone G, Chiarugi M, et al. Surgery in COVID-19 patients: operational directives. World J Emerg Surg 2020; 15:25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wong J, Goh QY, Tan Z, et al. Preparing for a COVID-19 pandemic: a review of operating room outbreak response measures in a large tertiary hospital in Singapore. Can J Anaesth 2020; 67:732–745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rosenberg ES, Dufort EM, Blog DS, et al. COVID-19 testing, epidemic features, hospital outcomes, and household prevalence, New York State-March 2020. Clin Infect Dis 2020; ciaa549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Urban MJ, Patel TR, Raad R, et al. Implementation of preoperative screening protocols in otolaryngology during the COVID-19 pandemic. Otolaryngol Neck Surg 2020; 163:265–270. [DOI] [PubMed] [Google Scholar]
- 10.Kamboj M, Pessin M. Comment on Screening of Asymptomatic Surgical Patients for SARS-CoV-2: The Covid19 Subcommittee of the Operating Room Executive Committee. Ann Surg 2020; 272:e243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Symptoms of Coronavirus (COVID-19). [Centers for Disease Control website]. May 20, 2020. Available at: https://www.cdc.gov/coronavirus/2019-ncov/downloads/COVID19-symptoms.pdf. Accessed June 15, 2020. [Google Scholar]
- 12.Bilimoria KY, Liu Y, Paruch JL, et al. Development and evaluation of the universal ACS NSQIP surgical risk calculator: a decision aid and informed consent tool for patients and surgeons. J Am Coll Surg 2013; 217:833–842.e1-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Stata/IC [Statistical Software]. Version 16.1. College Station, Texas: StatCorp, LLC. [Google Scholar]
- 14.Álvarez Gallego M, Gortázar de Las Casas S, Pascual Migueláñez I, et al. SARS-CoV-2 pandemic on the activity and professionals of a General Surgery and Digestive Surgery Service in a tertiary hospital. Cir Esp 2020; 98:320–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li Y-K, Peng S, Li L-Q, et al. Clinical and transmission characteristics of Covid-19 - a retrospective study of 25 cases from a single thoracic surgery department. Curr Med Sci 2020; 40:295–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rodríguez-Leor O, Cid-Álvarez B, Ojeda S, et al. Impacto de la pandemia de COVID-19 sobre la actividad asistencial en cardiología intervencionista en España. REC Interv Cardiol 2020; 2:82–89. [Google Scholar]
- 17.Tam C-CF, Cheung K-S, Lam S, et al. Impact of coronavirus disease 2019 (COVID-19) outbreak on outcome of myocardial infarction in Hong Kong, China. Catheter Cardiovasc Interv 2020; 1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. COVID-19: Data (By Borough). [New York City Department of Health website]. Available at: https://www1.nyc.gov/site/doh/covid/covid-19-data-boroughs.page. Accessed August 15, 2020. [Google Scholar]
- 19.Lubel J, Garg M. Renin-angiotensin-aldosterone system inhibitors in Covid-19. N Engl J Med 2020; 382:e92. [DOI] [PubMed] [Google Scholar]
- 20.Talreja H, Tan J, Dawes M, et al. A consensus statement on the use of angiotensin receptor blockers and angiotensin converting enzyme inhibitors in relation to COVID-19 (corona virus disease 2019). N Z Med J 2020; 133:85–87. [PubMed] [Google Scholar]
- 21.Keller BC, Le A, Sobhanie M, et al. Early COVID-19 infection after lung transplantation. Am J Transplant 2020; 20:2923–2927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cummings MJ, Baldwin MR, Abrams D, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet 2020; 395:1763–1770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.COVIDSurg Collaborative. Mortality and pulmonary complications in patients undergoing surgery with perioperative SARS-CoV-2 infection: an international cohort study. Lancet 2020; 396:27–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020; 323:2052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hamel L, Kearney A, Kirzinger A, et al. KFF Health Tracking Poll - May 2020 [Kaiser Family Foundation Website]. May 27, 2020. Available at: https://www.kff.org/coronavirus-covid-19/report/kff-health-tracking-poll-may-2020/. Accessed June 15, 2020. [Google Scholar]


