Supplemental Digital Content is available in the text.
Keywords: coronavirus disease 2019, hypoxemia, inhaled epoprostenol, prone positioning
Abstract
Objectives:
To evaluate the effects of inhaled epoprostenol and prone positioning, individually and in combination in mechanically ventilated patients with coronavirus disease 2019 and refractory hypoxemia.
Design:
Retrospective study.
Setting:
Academic hospital adult ICUs.
Patients:
Adult patients who received inhaled epoprostenol and prone positioning during invasive ventilation were enrolled. Patients were excluded if inhaled epoprostenol was initiated: 1) at an outside hospital, 2) after prone positioning was terminated, 3) during extracorporeal membrane oxygenation or cardiopulmonary resuscitation, and 4) with Pao2/Fio2 greater than 150 mm Hg.
Interventions:
Inhaled epoprostenol and prone positioning.
Results:
Of the 43 eligible patients, 22 and seven received prone positioning and inhaled epoprostenol alone, respectively, prior to their use in combination, Pao2/Fio2 was not different pre- and post-prone positioning or inhaled epoprostenol individually (89.1 [30.6] vs 97.6 [30.2] mm Hg; p = 0.393) but improved after the combined use of inhaled epoprostenol and prone positioning (84.0 [25.6] vs 124.7 [62.7] mm Hg; p < 0.001). While inhaled epoprostenol and prone positioning were instituted simultaneously in 14 patients, Pao2/Fio2 was significantly improved (78.9 [27.0] vs 150.2 [56.2] mm Hg, p = 0.005) with the combination. Twenty-seven patients (63%) had greater than 20% improvement in oxygenation with the combination of inhaled epoprostenol and prone positioning, and responders had lower mortality than nonresponders (52 vs 81%; p = 0.025).
Conclusions:
In critically ill, mechanically ventilated patients with coronavirus disease 2019 who had refractory hypoxemia, oxygenation improved to a greater extent with combined use of inhaled epoprostenol and prone positioning than with each treatment individually. A higher proportion of responders to combined inhaled epoprostenol and prone positioning survived compared with nonresponders. These findings need to be validated by randomized, prospective clinical trials.
In the U.K. national ICU audit, 72.7% of hospitalized patients with coronavirus disease 2019 (COVID-19) required advanced respiratory support (1). Management of COVID-19 remains largely supportive. Epoprostenol is a naturally occurring prostaglandin that relaxes vascular smooth muscle and also provides anti-inflammatory effects (2, 3). Inhaled epoprostenol (iEPO) has been used off-label for over 2 decades to reduce pulmonary artery pressure and improve oxygenation in critically ill patients (2–5). Hypoxemia in COVID-19 patients is mainly caused by ventilation-perfusion mismatch (6), which might be improved by inhalation of a pulmonary vasodilator. However, due to concerns of virus transmission by aerosol dispersion during nebulization (7), iEPO has been limited to rescue therapy for patients with COVID-19, especially during the early phase of the pandemic. In this report, we describe our preliminary experience with iEPO in mechanically ventilated COVID-19 patients with refractory hypoxemia (defined as Pao2/Fio2 ≤ 150 mm Hg) while on optimal ventilator settings according to the high positive end-expiratory pressure (PEEP) low-Fio2 acute respiratory distress syndrome (ARDS) network protocol (8).
Prior to the COVID-19 pandemic, prone positioning (PP) was shown to improve oxygenation and reduce 28- and 90-day mortality in intubated patients with severe ARDS (9, 10). Thus, PP has been recommended by the Surviving Sepsis Campaign COVID-19 subcommittee (11) and broadly used as an early intervention for intubated COVID-19 patients with refractory hypoxemia (8, 12–17). In this preliminary study, we compared the effects of PP and iEPO both individually and in combination, on oxygenation in COVID-19 patients with refractory hypoxemia.
MATERIALS AND METHODS
After approval by the Institutional Review Board (20050405-IRB01) at the Rush University Medical Center, a retrospective study was implemented to screen consecutive patients with laboratory confirmed COVID-19 admitted to adult ICUs between March 18, 2020, and May 31, 2020. Adult patients who received iEPO and PP during invasive ventilation were enrolled. Patients were excluded if iEPO was initiated at an outside hospital or after PP was terminated, if iEPO was employed during extracorporeal membrane oxygenation (ECMO) or cardiopulmonary resuscitation, or if iEPO was initiated with Pao2/Fio2 greater than 150 mm Hg.
Epoprostenol (Veletri, Actelion Pharmaceuticals US, San Francisco, CA) was prepared in a 50-mL syringe and delivered via continuous nebulization using a vibrating mesh nebulizer (Aeroneb Solo; Aerogen, Galway, Ireland) and a syringe pump, with the nebulizer placed at the inlet of the humidifier (4). iEPO was initiated at 50 ng/kg/min of patient’s predicted body weight, and the dose was maintained for a minimum of 24 hours after initiation. iEPO weaning was determined by the treating physicians and was titrated down by 10 ng/kg/min every 30–60 minutes if the patient’s oxygenation remained stable. PP was initiated if patients’ Pao2/Fio2 ratio could not be maintained greater than or equal to 150 mm Hg with PEEP set greater than or equal to 10 cm H2O and Fio2 greater than or equal to 0.6 (9). The prone position was maintained for at least 16 hours (12).
The primary outcome was to compare oxygenation with PP or iEPO individually with combined use of PP and iEPO. Patient’s position (supine or prone) at which iEPO was initiated was collected, and the number of sessions for PP prior to and after iEPO initiation was also recorded. The ratio of Pao2/Fio2 was compared within 2 hours pre and post either iEPO or PP or in combination. If Pao2 was not available, Spo2 was used as a substitute only in subjects with Spo2 less than or equal to 97%. A greater than 20% improvement of Pao2/Fio2 ratio (or Spo2/Fio2 if Pao2/Fio2 was not available) was considered as a positive response.
Kolmogorov-Smirnov was used to test normality of distribution for considered variables. Continuous variables were expressed as mean (sd) or median (interquartile range [IQR]), depending on the normality of distribution. Paired t test or Wilcoxon sign-rank test was used to compare pre- and post responses with iEPO or PP and changes from supine to iEPO + PP, whereas t test or Mann-Whitney test was used to compare the incremental changes between the responder and nonresponder groups and differences in categorical variables were assessed with the chi-square test. A p value of less than 0.05 was considered statistically significant for all tests. Data analysis was conducted with the SPSS statistical software (SPSS 26.0; SPSS, Chicago, IL).
RESULTS
From March 18, 2020, to May 31, 2020, 336 adult patients with confirmed COVID-19 were admitted to our ICUs. Among the 234 intubated patients, 57 patients received iEPO and PP during invasive ventilation, 14 patients were excluded for the following reasons: 1) iEPO was initiated after PP was terminated (n = 8), 2) iEPO was used during ECMO (n = 2), 3) iEPO was used during cardiopulmonary resuscitation (n = 1), 4) iEPO was initiated at an outside hospital (n = 1), and 5) iEPO was initiated with Pao2/Fio2 greater than 150 mm Hg (n = 2) (Fig. 1). In total, 43 patients (27 male) were eligible, with a mean (sd) age 55.1 (13.8) years and the predominant ethnicities were Hispanic/Latino (54%) and African American (33%). On the day of iEPO initiation, patients’ median (IQR) Sequential Organ Failure Assessment score was 9 (8–11), and the duration from COVID-19 confirmation was 9 days (4–14 d), whereas mechanical ventilation had been used for 68 hours (5.3–203.3 hr). Prior to treatment (namely, in supine position at baseline), Pao2/Fio2 was 86.6 (28.9) mm Hg with PEEP at 16 cm H2O (12–18 cm H2O) with an Fio2 of 1.0 (0.8–1.0), whereas the compliance of respiratory system was 26.5 (7.2) mL/cm H2O.
Figure 1.
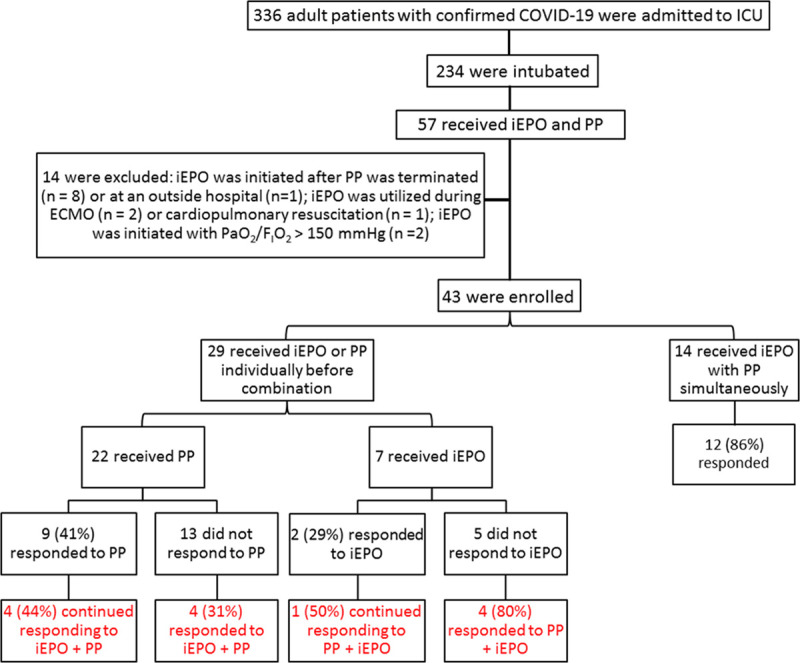
Research flow diagram. COVID-19 = coronavirus disease 2019, ECMO = extracorporeal membrane oxygenation, iEPO = inhaled epoprostenol, PP = prone positioning.
Because of the retrospective nature of this study, it became apparent that patients fell into two categories: the majority (29) received therapy in an additive manner (either PP followed by the addition of iEPO or vice versa), whereas 14 patients started on PP and iEPO at the same time. The response to the approaches of adding one therapy to the other is described first, followed by combined therapy group.
Baseline Versus PP Versus PP + iEPO
Twenty-two patients had a median of 3 (1–5) sessions of PP prior to iEPO initiation. Despite response to PP prior to initiating iEPO in 9 patients (41%), there was no difference in Pao2/Fio2 between pre- versus post-PP (91.7 [31.8] vs 98.0 [31.9] mm Hg; n = 21; p = 0.570) for the entire subset of patients (Fig. 2). In this group, iEPO was initiated 5.4 hours (1.8–17.9 hr) after being placed PP with subsequent significant improvement in Pao2/Fio2 (83.3 [23.5] vs 106.9 [53.4] mm Hg; n = 20; p = 0.034) (Table 1). Among the nine responders to PP, four (44%) showed continued improvements in oxygenation to iEPO with PP. Among the 13 nonresponders to PP, four (31%) responded to iEPO with PP (Fig. 1). Regarding the relationship between the initiation of iEPO to PP, six of the 22 patients used iEPO during their first PP session, four (67%) of whom responded to PP, and three (75%) continued responding to iEPO with PP, whereas of the two patients (50%) who did not respond to initial PP responded to the combination of iEPO with PP.
Figure 2.
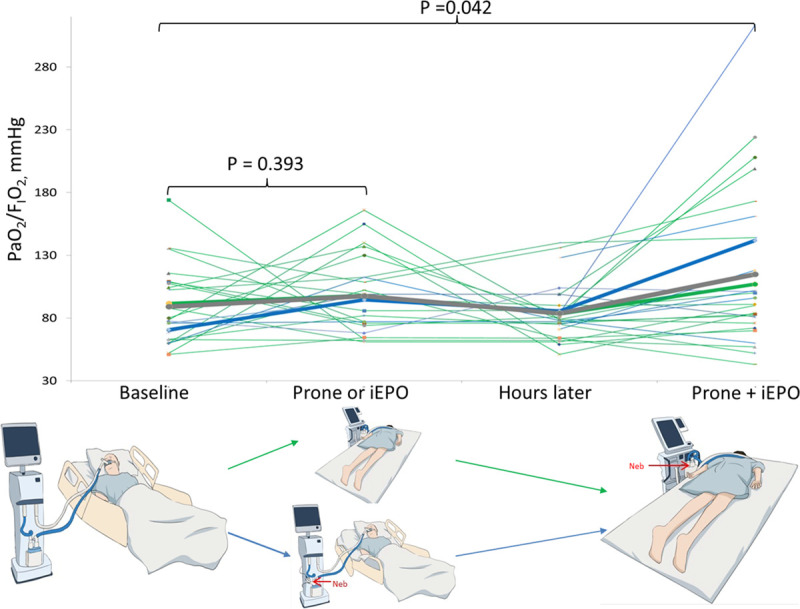
Individual responses to prone positioning (PP) or inhaled epoprostenol (iEPO) and PP + iEPO. Among the 29 patients who received PP (n = 22) or iEPO (n = 7) individually, patients’ Pao2/Fio2 was not significantly different pre- and post-PP (green line) or iEPO (blue line) but improved after the combined use of iEPO and PP (89.1 [30.6] vs 113.3 [66.7] mm Hg; p = 0.042). Green bold line indicates average Pao2/Fio2 for the patients receiving PP, blue bold line indicates average Pao2/Fio2 for the patients receiving iEPO, and gray bold line indicates average Pao2/Fio2 for both group of patients.
TABLE 1.
Pre- and Post-responses to Prone Positioning and Inhaled Epoprostenol for the 22 Intubated Coronavirus Disease 2019 Patients Who Received Inhaled Epoprostenol Under Prone Positioning
| Baseline | PP | p | PP (Few Hours Later) | Inhaled Epoprostenol + PP | p | |
|---|---|---|---|---|---|---|
| Pao2, mm Hga | 76.6 ± 29.0 | 89.0 ± 30.4 | 0.258 | 76.1 ± 18.4 | 96.6 ± 46.4 | 0.041 |
| Pao2/Fio2, mm Hga | 91.7 ± 31.8 | 98.0 ± 31.9 | 0.570 | 83.3 ± 23.5 | 106.9 ± 53.4 | 0.034 |
| Spo2, %b | 93 (91–97) | 92 (90–95) | 0.299 | 92 (86–93) | 94 (88–96) | 0.147 |
| Fio2 | 0.8 (0.7–1.0) | 1.0 (0.9–1.0) | 0.073 | 1.0 (0.8–1.0) | 1.0 (0.8–1.0) | 1.0 |
| Positive end-expiratory pressure, cm H2O | 14 (12–16.5) | 14 (14–18) | 0.548 | 15 (14–18) | 15 (14–16) | 0.107 |
| Static compliance of respiratory system, mL/cm H2Oc | 28 (22–34) | 28 (25–38) | 0.316 | 25 (22–32) | 24 (19–28) | 0.071 |
PP = prone positioning.
aData were available in 21, 21, 20, and 20 patients, respectively.
bData were available in 15 patients.
cData were available in 18, 18, 13, and 13 patients, respectively.
Baseline Versus iEPO Versus iEPO + PP
Seven patients had iEPO initiated in supine position. Patients’ oxygenation (Pao2/Fio2 or Spo2/Fio2) was not significantly different pre- versus post-iEPO (Supplemental Table 1, http://links.lww.com/CCX/A466), but in six patients, Pao2/Fio2 was higher after being placed on PP with iEPO (85.3 [21.1] vs 141.7 [90.0] mm Hg; p = 0.046). Among the seven patients, two responded to iEPO, one of whom continued responding to PP with iEPO, whereas among the remaining five patients who did not respond to iEPO, four responded to the combination of PP with iEPO (Fig. 1).
Baseline Versus iEPO + PP
To summarize, for the aforementioned 29 patients who received PP (n = 22) or iEPO (n = 7) individually prior to the combination, Pao2/Fio2 was not significantly different either pre- versus post-iEPO or PP (89.1 [30.6] vs 97.6 [30.2] mm Hg; n = 24; p = 0.393). In contrast, after combined therapy, their Pao2/Fio2 was higher (89.1 [30.6] vs 113.3 [66.7] mm Hg; p = 0.042) (Fig. 2).
Fourteen patients had iEPO initiated within 51 minutes (22.5–90 min) of PP, so that only the combined effects of therapy could be assessed. With the combined use of iEPO and PP, patients’ Pao2/Fio2 was significantly improved (78.9 [27.0] vs 150.2 [56.2] mm Hg; p = 0.005) (Table 2). Of the 14 patients, 12 (86%) responded to the combined use of iEPO and PP (Fig. 1). Among the 11 patients who had never been proned before, 10 (91%) responded to iEPO with PP.
TABLE 2.
Pre- and Post-responses for the 14 Intubated Coronavirus Disease 2019 Patients Who Received Inhaled Epoprostenol With Prone Positioning Simultaneously
| n | Baseline | Prone Positioning + Inhaled Epoprostenol | p | |
|---|---|---|---|---|
| Pao2, mm Hg | 10 | 71.4 ± 23.3 | 128.2 ± 48.1 | 0.008 |
| Pao2/Fio2, mm Hg | 10 | 78.9 ± 27.0 | 150.2 ± 56.2 | 0.005 |
| Spo2, % | 11 | 90 (81–92) | 93 (90–95) | 0.019 |
| Spo2/Fio2 | 11 | 90 (81–95) | 97 (90–134) | 0.006 |
| Fio2 | 14 | 1.0 (0.88–1.0) | 0.95 (0.78–1.0) | 0.172 |
| Positive end-expiratory pressure, cm H2O | 14 | 18 (13.5–18.5) | 19 (15.5–20) | 0.583 |
| Static compliance of respiratory system, mL/cm H2O | 9 | 23.1 ± 5.9 | 28.6 ± 6.4 | 0.008 |
Taking all patients into account, compared with patients’ Pao2/Fio2 under supine position, the combined use of iEPO and PP significantly improved Pao2/Fio2 (84.0 [25.6] vs 124.7 [62.7] mm Hg; n = 36; p < 0.001) (Fig. 3).
Figure 3.
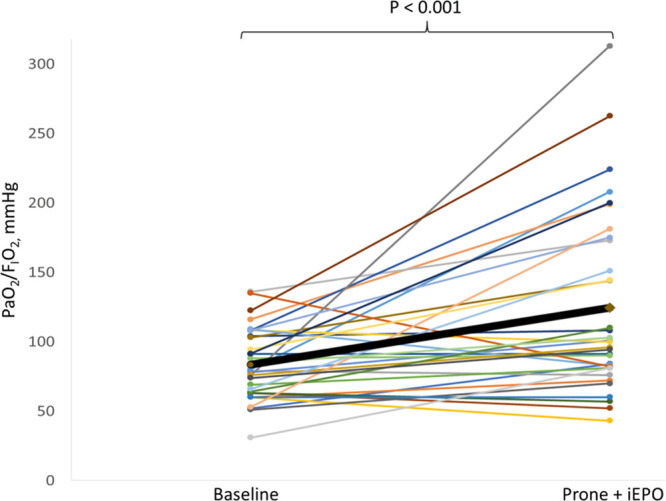
Individual patient’s Pao2/Fio2 at baseline and after the combined use of prone positioning (PP) and inhaled epoprostenol (iEPO). Compared with overall patients’ baseline Pao2/Fio2, the combined use of iEPO and PP significantly improved Pao2/Fio2.
Characteristics of Responders Versus Nonresponders to PP + iEPO
Compared with patients’ oxygenation (Pao2/Fio2 or Spo2/Fio2 if Pao2/Fio2 was not available) in supine position, 27 patients had positive responses (greater than 20% improvement) with the combination of iEPO with PP. Compared with the nonresponders, responders had a trend of fewer PP sessions prior to the combined use of iEPO and PP (1 [0–4] vs 3 [1.3–3.8]; p = 0.054), particularly higher percentage of patients who had never been placed on PP before (44 vs 6%; p = 0.022). Responders had lower mortality than nonresponders (52 vs 81%; p = 0.025) (Table 3).
TABLE 3.
Responders and Nonresponders to Combination of Prone Positioning and Inhaled Epoprostenol
| Responders (n = 27) | Nonresponders (n = 16) | p | |
|---|---|---|---|
| Age, yr | 56.2 ± 13.7 | 53.3 ± 14.3 | 0.831 |
| Gender (male), % | 17 (63) | 10 (63) | 1.0 |
| Ethnicity (Hispanic/Latino), % | 11 (41) | 12 (75) | 0.102 |
| Sequential Organ Failure Assessment score | 10 (8–11) | 9 (8–10.8) | 0.480 |
| Pao2/Fio2 pretreatment, mm Hg | 83.2 ± 26.7 | 92.5 ± 32.5 | 0.515 |
| Positive end-expiratory pressure pretreatment, cm H2O | 16 (12–18) | 15 (14–18) | 0.949 |
| Static compliance of respiratory system pretreatment, mL/cm H2O | 25.4 ± 6.1 | 27.8 ± 8.3 | 0.702 |
| PP sessions prior to iEPO | 1 (0–4) | 3 (1.3–3.8) | 0.054 |
| Had never been placed PP prior to iEPO + PP, % | 12 (44) | 1 (6) | 0.022 |
| Duration between iEPO and PP, hr | 3.5 (1.0–9.0) | 2.3 (1.0–10.1) | 0.890 |
| Duration between coronavirus disease 2019 confirmation to iEPO, d | 9.0 ± 7.5 | 9.7 ± 5.2 | 0.606 |
| Mechanical ventilation hours prior to iEPO | 67 (5–196) | 92 (17–217) | 0.505 |
| 28-d outcome, n (%) | |||
| Mortality | 14 (52) | 13 (81) | 0.025 |
| Extracorporeal membrane oxygenation | 1 (4) | 2 (13) | |
| Discharged from hospital alive | 12 (44) | 1 (6) | |
iEPO = inhaled epoprostenol, PP = prone positioning.
DISCUSSION
In this single-center cohort study, we found that the combined use of iEPO with PP improved oxygenation in intubated COVID-19 patients with refractory hypoxemia, including some patients who did not respond to PP or iEPO individually. To our knowledge, this is the first report on the effects of combining iEPO with PP, which is consistent with other studies on the effects of combining PP with inhaled nitric oxide in patients with ARDS (10, 18).
The mechanism by which oxygenation improves with PP and iEPO might be due to a reduction in ventilation/perfusion mismatch, via the combined benefits of dilating pulmonary vessels in the dorsal areas of the lung with improved ventilation of those areas (10). In ARDS, the iEPO response rate is approximately 60% but the response is lower in patients with sepsis (3) because of endothelial dysfunction and dysregulation of nitric oxide and prostacyclin (19). Similarly, COVID-19 also induces both pulmonary and systemic endothelial dysfunction (6), thereby making it difficult to predict iEPO responsiveness. Our previous cohort study on intubated COVID-19 patients found that over 70% of them responded to the first PP session, whereas the response rates decreased starting from the second PP session (12). In this study, more than 50% of the enrolled patients had previously been placed on PP more than once on maximum ventilator settings (prior to enrollment), which might explain the lower response rate to iEPO and PP.
Compared with the nonresponders in the second and third PP sessions, the responders had lower rates of treatment failure, which was defined as death or escalation to ECMO therapy (12). We also found the responders had lower mortality. The high mortality rate even among the responders in this study might be explained by the higher severity of patients and the use of iEPO as the last resort. Likewise, Garcia et al (16) reported the survival rate of 14.3% in their cohort with veno-venous-ECMO for COVID-19 patients that underwent PP, whereas Shelhamer et al (17) reported mortality of 77.4% in the PP group and 83.9% in the control group that did not undergo PP. The authors explained that the high mortality was contributed by the severe acuity of patients receiving those treatments (PP or ECMO) as rescue therapy. In our study, 14 patients who first received iEPO and PP at the same time had higher response rate (86%) to iEPO with PP, however whether or not earlier use of iEPO and PP in combination could provide additional benefits in treating refractory hypoxemia in mechanically ventilated patients with COVID-19 requires further investigation.
Our study is limited by the lack of a control group and a small sample size. However, our study found improvement in oxygenation when PP was combined with iEPO as rescue therapy in critically ill patients. Our findings suggest that combination has benefit but does not directly address whether patients with an oxygen response to iEPO/iNO might further benefit from combining PP. The combination of PP and iEPO should be considered as supportive therapy for refractory hypoxemia in mechanically ventilated patients in institutions where ECMO is not available or prior to escalation to ECMO therapy. Our findings suggest that nonresponders to the combined use of iEPO and PP should be prioritized as ECMO candidates. Randomized clinical trials are needed to confirm these findings in COVID-19 and non-COVID-19 patients with refractory hypoxemia and to investigate further if it improves survival in such critically ill patients. Seven patients (16%) did not have pre- and posttreatment Pao2/Fio2, so we used Spo2/Fio2 (only Spo2 < 97% were chosen) as an alternative to ensure better correlation with Pao2/Fio2 (20). However, use of Spo2/Fio2 could result in discrepant findings. Thus, Pao2/Fio2 should be preferred as the parameter to assess response to refractory hypoxemia with the combination of iEPO and PP in future studies.
CONCLUSIONS
In mechanically ventilated patients with COVID-19 who had refractory hypoxemia, combined use of iEPO and PP improved oxygenation to a greater extent than with each treatment individually. Responders to combined modalities had lower mortality than nonresponders.
ACKNOWLEDGMENTS
We thank all the respiratory therapists in Respiratory Care Department in the Rush University Medical Center for their dedicated and diligent work during coronavirus disease 2019 pandemic. We also thank Mr. Rongshou Zheng, MPH, for consultancy in data analysis.
Supplementary Material
Footnotes
Dr. Li had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Dr. Li conceived of the idea. Dr. Li and Ms. Augustynovich implemented data collection, Dr. Li performed data analysis and drafted the article, and Dr. Li, Dr. Fink, and Ms. Augustynovich interpreted the data. Drs. Fink and Dhand supervised the process. Mr. Kallet and Dr. Dhand provided critical revision on the article. All authors reviewed and approved the final version.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (http://journals.lww.com/ccxjournal).
This study was funded by Rice Foundation.
Dr. Li discloses research support from Fisher & Paykel Healthcare and Rice foundation, and lecture honorarium from AARC and Fisher & Paykel Healthcare outside the summited work. Dr. Dhand reports remuneration from GSK Pharmaceuticals, Boehringer-Ingelheim, Bayer, Mylan, Teva, and Astra-Zeneca Pharmaceuticals outside the submitted work. Dr. Fink is the Chief Science Officer for Aerogen Pharma, San Mateo, CA. Mr. Kallet has received lecture honoraria from Nihon Kohden in the past 2 years. The remaining authors have disclosed that they do not have any potential conflicts of interest.
This study was approved by the institutional review board in Rush University Medical Center (20050405-IRB01).
REFERENCES
- 1.Intensive Care National Audit and Research Centre: Reports. Available at: https://www.icnarc.org/Our-Audit/Audits/Cmp/Reports. Accessed June 19, 2020
- 2.Elmi-Sarabi M, Deschamps A, Delisle S, et al. : Aerosolized vasodilators for the treatment of pulmonary hypertension in cardiac surgical patients: A systematic review and meta-analysis. Anesth Analg 2017; 125:393–402 [DOI] [PubMed] [Google Scholar]
- 3.Kallet RH, Burns G, Zhuo H, et al. : Severity of hypoxemia and other factors that influence the response to aerosolized prostacyclin in ARDS. Respir Care 2017; 62:1014–1022 [DOI] [PubMed] [Google Scholar]
- 4.Li J, Gurnani PK, Roberts KM, et al. : The clinical impact of flow titration on epoprostenol delivery via high flow nasal cannula for ICU patients with pulmonary hypertension or right ventricular dysfunction: A retrospective cohort comparison study. J Clin Med 2020; 9:464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fuller BM, Mohr NM, Skrupky L, et al. : The use of inhaled prostaglandins in patients with ARDS: A systematic review and meta-analysis. Chest 2015; 147:1510–1522 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Potus F, Mai V, Lebret M, et al. : Novel insights on the pulmonary vascular consequences of COVID-19. Am J Physiol Lung Cell Mol Physiol 2020; 319:L277–L288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dhand R, Li J: Coughs and sneezes: Their role in transmission of respiratory viral infections, including SARS-CoV-2. Am J Respir Crit Care Med 2020; 202:651–659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.The ARDS Clinical Trials Network: Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med 2004; 351:327–336 [DOI] [PubMed] [Google Scholar]
- 9.Guérin C, Reignier J, Richard JC, et al. ; PROSEVA Study Group: Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 2013; 368:2159–2168 [DOI] [PubMed] [Google Scholar]
- 10.Kallet RH: A comprehensive review of prone position in ARDS. Respir Care 2015; 60:1660–1687 [DOI] [PubMed] [Google Scholar]
- 11.Alhazzani W, Møller MH, Arabi YM, et al. : Surviving sepsis campaign: Guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Intensive Care Med 2020; 46:854–887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Weiss T, Cerda F, Scott B, et al. : Prone positioning for patients intubated for severe acute respiratory distress syndrome (ARDS) secondary to COVID-19: A cohort study. Br J Anaesth. 2020 Oct 10. [online ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ziehr DR, Alladina J, Petri CR, et al. : Respiratory pathophysiology of mechanically ventilated patients with COVID-19: A cohort study. Am J Respir Crit Care Med 2020; 201:1560–1564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pan C, Chen L, Lu C, et al. : Lung recruitability in COVID-19-associated acute respiratory distress syndrome: A single-center observational study. Am J Respir Crit Care Med 2020; 201:1294–1297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Carsetti A, Damia Paciarini A, Marini B, et al. : Prolonged prone position ventilation for SARS-CoV-2 patients is feasible and effective. Crit Care 2020; 24:225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Garcia B, Cousin N, Bourel C, et al. ; Lille Intensive Care COVID-19 Group: Prone positioning under VV-ECMO in SARS-CoV-2-induced acute respiratory distress syndrome. Crit Care 2020; 24:428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shelhamer M, Wesson PD, Solari IL, et al. : Prone positioning in moderate to severe acute respiratory distress syndrome due to COVID-19: A cohort study and analysis of physiology. Res Sq. Preprint posted online August 17, 2020. doi:10.21203/rs.3.rs-56281/v1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jolliet P, Bulpa P, Ritz M, et al. : Additive beneficial effects of the prone position, nitric oxide, and almitrine bismesylate on gas exchange and oxygen transport in acute respiratory distress syndrome. Crit Care Med 1997; 25:786–794 [DOI] [PubMed] [Google Scholar]
- 19.Boisrame´-Helms J, Kremer H, Schini-Kerth V, et al. : Endothelial dysfunction in sepsis. Curr Vasc Pharmacol 2013; 11:150–160 [PubMed] [Google Scholar]
- 20.Rice TW, Wheeler AP, Bernard GR, et al. ; National Institutes of Health, National Heart, Lung, and Blood Institute ARDS Network: Comparison of the SpO2/FIO2 ratio and the PaO2/FIO2 ratio in patients with acute lung injury or ARDS. Chest 2007; 132:410–417 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


