Abstract
In this study, we performed bioinformatics and statistical analyses to investigate the prognostic significance of metabolic genes in clear cell renal cell carcinoma (ccRCC) using the transcriptome data of 539 ccRCC and 72 normal renal tissues from TCGA database. We identified 79 upregulated and 45 downregulated (n=124) metabolic genes in ccRCC tissues. Eleven prognostic metabolic genes (NOS1, ALAD, ALDH3B2, ACADM, ITPKA, IMPDH1, SCD5, FADS2, ACHE, CA4, and HK3) were identified by further analysis. We then constructed an 11-metabolic gene signature-based prognostic risk score model and classified ccRCC patients into high- and low-risk groups. Overall survival (OS) among the high-risk ccRCC patients was significantly shorter than among the low-risk ccRCC patients. Receiver operating characteristic (ROC) curve analysis of the prognostic risk score model showed that the areas under the ROC curve for the 1-, 3-, and 5-year OS were 0.810, 0.738, and 0.771, respectively. Thus, our prognostic model showed favorable predictive power in the TCGA and E-MTAB-1980 ccRCC patient cohorts. We also established a nomogram based on these eleven metabolic genes and validated internally in the TCGA cohort, showing an accurate prediction for prognosis in ccRCC.
Keywords: clear cell renal cell carcinoma, metabolic genes, prognostic model, bioinformatics
INTRODUCTION
Clear cell renal cell carcinoma (ccRCC) is the most common among all renal cell carcinoma (RCC) subtypes, and accounts for 75% of all renal tumors [1]. It represents a hypervascular parenchymal malignancy originating from the proximal tubular cells of the nephron [2]. The prognosis of patients with metastatic ccRCC is poor and the five-year survival rate is less than 10% [3, 4]. In the last two decades, the roles of several genes in the regulation of the growth and progression of ccRCC have been recognized, but, the underlying pathogenetic mechanisms are still not understood fully.
Metabolic reprogramming is a common feature of tumor cells and is associated with tumor growth and progression [5]. The most common metabolic changes observed in several types of cancer cells include upregulation of nucleotide biosynthesis and downregulation of mitochondrial metabolism [6]. Peng et al. investigated the mRNA expression patterns of seven major metabolic processes in the tumor subtypes of 33 different cancers and found that those with increased carbohydrate, nucleotide, and vitamin/co-factor metabolism were consistently associated with worse prognosis [7]. Aberrant regulation of the tricarboxylic acid (TCA) cycle, glutamine metabolism, and lipid metabolism are associated with the growth and progression of RCC [8–11]. Hakimi et al. performed metabolomic profiling of ccRCC tissues and found significant alterations in central carbon metabolism, one-carbon metabolism, and the antioxidant response; metabolic changes in glutathione and cysteine/methionine metabolism pathways were associated with progression and metastasis of ccRCC; a metabolic differentiation group in chromophobe RCC was associated with worse survival outcomes [12]. The investigation related to ccRCC have mainly focused on the metabolic changes involved ccRCC, but, the information regarding the expression patterns of key metabolism-related genes involved in the metabolic reprogramming and the metabolomic characteristics of ccRCC are not clear.
Hence, in the current study, we systematically analyzed the transcriptome data from ccRCC patient tissues to identify the metabolic genes that can accurately predict the prognosis of ccRCC patients.
RESULTS
Identification of differentially expressed metabolic genes in the ccRCC patients
Figure 1 shows the study strategy used to systematically analyze the prognostic prediction values of the metabolic genes in ccRCC. We downloaded the ccRCC transcriptome data for 72 normal renal tissue samples and 539 ccRCC tissue samples from The Cancer Genome Atlas (TCGA) database. Then, we identified differentially expressed metabolic genes in ccRCC using the edgeR package (http://www.bioconductor.org/packages/release/bioc/html/edgeR.html) by screening the 1,466 metabolism-related genes that are listed in the 70 metabolism-related gene sets from the Kyoto Encyclopedia of Genes and Genomes (KEGG) database in the gene set enrichment analysis (GSEA) website (https://www.gsea-msigdb.org/gsea/msigdb/collections.jsp#C2). We used the selection criteria as |log2 fold change (FC)| >1.0 and P < 0.05 and identified 124 differentially expressed metabolic genes, including 79 up-regulated and 45 down-regulated genes (Figure 2).
Figure 1.
The flow chart of the study strategy for identifying metabolic genes with prognostic significance in ccRCC.
Figure 2.
Differentially expressed metabolic genes in ccRCC samples. (A) The heat map shows the expression of 124 differentially expressed metabolic genes in ccRCC and normal renal tissue samples. (B) The volcano plot shows the upregulated or downregulated metabolic genes in the ccRCC samples relative to normal renal tissue samples.
Functional enrichment analysis of the differentially expressed metabolic genes
Next, we performed Gene Ontology (GO) and KEGG functional enrichment analysis of the 124 differentially expressed metabolic genes using the WEB-based Gene Set Analysis Toolkit (WebGestalt) online tool and the results are shown in Table 1. The significantly enriched biological processes were small molecule catabolism, organic acid biosynthesis, organic hydroxy compound metabolism, cellular amino acid metabolism, generation of precursor metabolites and energy, fatty acid derivative metabolism, monosaccharide metabolism, nucleoside phosphate biosynthesis, ribonucleotide metabolism, and fatty acid metabolism. The significantly enriched cellular components were the mitochondrial matrix, ficolin-1-rich granule, and the myelin sheath. The enriched molecular functions were the co-factor binding, oxidoreductase activity acting on paired donors with incorporation or reduction of molecular oxygen, lyase activity, organic acid binding, iron ion binding, monooxygenase activity, transferase activity, transferring glycosyl groups, vitamin binding, oxidoreductase activity acting on the aldehyde or oxo group of donors, and oxidoreductase activity acting on CH-OH group of donors. KEGG analysis showed enrichment in metabolic pathways including those involved in drug metabolism, retinol metabolism, chemical carcinogenesis, purine metabolism, porphyrin and chlorophyll metabolism, steroid hormone biosynthesis, metabolism of xenobiotics by cytochrome P450, and carbon metabolism.
Table 1. KEGG pathway and GO enrichment analysis of differentially expressed metabolic genes.
| GO term | P-value | FDR | |
| Biological processes | small molecule catabolic process | 0 | 0 |
| organic acid biosynthetic process | 6.55E-14 | 2.78E-11 | |
| organic hydroxy compound metabolic process | 3.20E-10 | 9.06E-8 | |
| cellular amino acid metabolic process | 4.93E-10 | 1.05E-7 | |
| generation of precursor metabolites and energy | 6.84E-10 | 1.16E-7 | |
| fatty acid derivative metabolic process | 3.62E-9 | 5.12E-7 | |
| monosaccharide metabolic process | 5.46E-9 | 6.63E-7 | |
| nucleoside phosphate biosynthetic process | 1.17E-8 | 0.000001 | |
| ribonucleotide metabolic process | 1.26E-8 | 0.000001 | |
| fatty acid metabolic process | 2.06E-8 | 0.000002 | |
| Cellular component | mitochondrial matrix | 0.000070 | 0.012090 |
| ficolin-1-rich granule | 0.000265 | 0.022800 | |
| myelin sheath | 0.000774 | 0.044374 | |
| Molecular function | cofactor binding | 6.66E-16 | 1.88E-13 |
| oxidoreductase activity, acting on paired donors, with incorporation or reduction of molecular oxygen | 7.64E-14 | 1.08E-11 | |
| lyase activity | 1.11E-11 | 1.04E-9 | |
| organic acid binding | 9.99E-9 | 6.93E-7 | |
| iron ion binding | 1.23E-8 | 6.93E-7 | |
| monooxygenase activity | 1.75E-8 | 8.24E-7 | |
| transferase activity, transferring glycosyl groups | 4.15E-8 | 0.000002 | |
| vitamin binding | 5.28E-8 | 0.000002 | |
| oxidoreductase activity, acting on the aldehyde or oxo group of donors | 0.000003 | 0.000084 | |
| oxidoreductase activity, acting on CH-OH group of donors | 0.000003 | 0.000095 | |
| KEGG pathway | metabolic pathways | 0 | 0 |
| drug metabolism | 2.43E-12 | 3.71E-10 | |
| retinol metabolism | 3.41E-12 | 3.71E-10 | |
| chemical carcinogenesis | 6.20E-11 | 3.62E-9 | |
| purine metabolism | 6.67E-11 | 3.62E-9 | |
| porphyrin and chlorophyll metabolism | 1.24E-9 | 5.76E-8 | |
| steroid hormone biosynthesis | 3.52E-9 | 1.43E-7 | |
| metabolism of xenobiotics by cytochrome P450 | 3.97E-9 | 1.44E-7 | |
| carbon metabolism | 6.20E-8 | 0.000002 |
Protein-protein interaction (PPI) network and key co-expression modules
We used the STRING database and the cytoscape software to construct a PPI network of the differently expressed metabolic genes in ccRCC tissues. The PPI network consisted of 381 edges and 118 nodes (Figure 3A). We also identified two key co-expression modules using the Molecular Complex Detection (MCODE) plug-in of the Cytoscape software. Module 1 consisted of 36 edges and 9 nodes (Figure 3B), and module 2 consisted of 24 edges and 11 nodes (Figure 3C).
Figure 3.
Protein-protein interaction network and key co-expression modules. (A) The protein-protein interaction (PPI) network shows the interactions between 124 differentially expressed metabolic genes. (B, C) The two key modules consisting of co-expressing differentially expressed metabolic genes, module 1 and module 2 are shown. The red and green circles denote upregulated and downregulated metabolic genes, respectively.
ROC curve analysis of the hub metabolic genes
We identified ten metabolic genes as hub genes using the cytoHubba plug-in of the Cytoscape software and the maximal clique centrality (MCC) algorithm. We then performed the receiver operating characteristic (ROC) curve analysis to evaluate the efficacy of these ten hub genes to discriminate between tumor and normal renal tissue. As shown in Figure 4, the area under the ROC curve (AUC) values showed good diagnostic accuracy, namely, GAPDH (AUC=0.971, P<0.001), POLR3B (AUC=0.943, P<0.001), NME1-NME2 (AUC=0.836, P<0.001), ADCY10 (AUC=0.858, P<0.001), ADCY7 (AUC=0.894, P<0.001), POLR2F (AUC=0.889, P<0.001), NME1 (AUC=0.830, P<0.001), ENTPD2 (AUC=0.781, P<0.001), ADCY8(AUC=0.626, P=0.001), ADCY3 (AUC=0.861, P<0.001).
Figure 4.
ROC curve analysis of hub metabolic genes. The figure shows the ROC curves evaluating the diagnostic accuracy of the 10 hub metabolic genes, namely, (A) GAPDH; (B) POLR3B; (C) NME1-NME2; (D) ADCY10; (E) ADCY7; (F) POLR2F; (G) NME1; (H) ENTPD2; (I) ADCY8; (J) ADCY3 in ccRCC patients.
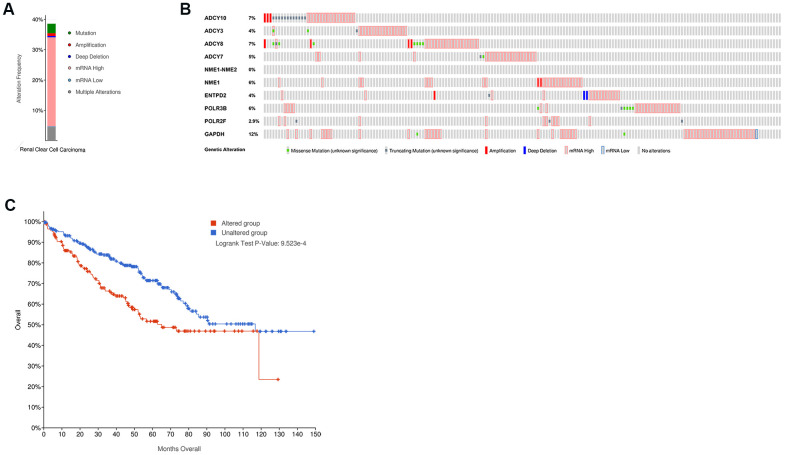
Analysis of mutation frequencies and copy number variations of the hub genes
We used the cBioPortal online tool to analyze the mutation frequencies in the 10 hub genes, namely, GAPDH, POLR3B, NME1-NME2, ADCY10, ADCY7, POLR2F, NME1, ENTPD2, ADCY8, and ADCY3 in ccRCC tissues (TCGA, Firehose Legacy). We observed mutations in these 10 candidate hub genes in 39% (172 of 446) of the ccRCC patients (Figure 5A). The mutation frequency in each of the ten candidate hub genes ranged from 0% to 12% (Figure 5B). Kaplan– Meier survival curve analysis showed that the overall survival (OS) was significantly shorter in the ccRCC patients with mutations in the candidate hub genes compared to the ccRCC patients without mutations in the candidate hub genes (Figure 5C).
Figure 5.
The mutation frequency of the ten hub metabolic genes in the ccRCC patients (TCGA, Firehose Legacy). (A) The overall mutation frequency of the hub metabolic genes in 446 ccRCC patients. (B) The mutation frequency of the individual hub metabolic genes in 446 ccRCC patients. (C) Kaplan–Meier survival curves show the OS of ccRCC patients with mutations in the hub metabolic genes (n=172) compared to those without mutations in the hub metabolic genes (n=274).
Identification of the prognosis-related metabolic genes
We used the caret R package to divide the ccRCC samples (n=539) from TCGA database into the training group (n=381) and the test group (n=158). We first investigated the prognostic significance of the 124 differentially expressed metabolic genes in the training group using univariate Cox regression analysis and identified 46 potential candidate metabolic genes (Supplementary Table 1). Then, we performed the least absolute shrinkage and selection operator (LASSO) regression analysis and identified 12 metabolic genes with potential prognostic value, including NOS1, ALAD, ALDH3B2, ACADM, ITPKA, IMPDH1, SCD5, FADS2, ACHE, CA4, HK3, and VAMP1 (Supplementary Figure 1). Multivariate Cox regression analysis showed that 11 of the 12 genes, namely, NOS1, ALAD, ALDH3B2, ACADM, ITPKA, IMPDH1, SCD5, FADS2, ACHE, CA4, and HK3 independently predicted prognosis of ccRCC patients.
Evaluation of the 11-metabolic gene signature-based prognostic risk score model
We performed stepwise Cox regression analysis using the eleven metabolic genes to construct a prediction model (Table 2). The risk score of each patient was calculated with the following formula: Risk score = (-0.0555 * Exp NOS1) + (0.0180 * Exp ALAD) + (0.1302 * Exp ALDH3B2) + (-0.0948 * Exp ACADM) + (0.0490 * Exp ITPKA) + (0.1788 * Exp IMPDH1) + (-0.1354 * Exp SCD5) + (0.1488 * Exp FADS2) + (0.0594 * Exp ACHE) + (-0.0570 * Exp CA4) + (0.2213 * Exp HK3).
Table 2. Multivariate Cox regression analysis to identify prognosis-related metabolic genes.
| Gene | Coef | Exp(coef) | se(coef) | z | Pr (>|z|) |
| NOS1 | -0.0555 | 0.9460 | 0.0694 | -0.8002 | 0.4236 |
| ALAD | 0.0180 | 1.0181 | 0.2081 | 0.0863 | 0.9312 |
| ALDH3B2 | 0.1302 | 1.1391 | 0.0538 | 2.4218 | 0.0154 |
| ACADM | -0.0948 | 0.9095 | 0.1425 | -0.6653 | 0.5058 |
| ITPKA | 0.0490 | 1.0502 | 0.0583 | 0.8396 | 0.4011 |
| IMPDH1 | 0.1788 | 1.1958 | 0.2212 | 0.8084 | 0.4189 |
| SCD5 | -0.1354 | 0.8734 | 0.0687 | -1.9707 | 0.0488 |
| FADS2 | 0.1488 | 1.1604 | 0.0890 | 1.6715 | 0.0946 |
| ACHE | 0.0594 | 1.0612 | 0.0646 | 0.9189 | 0.3582 |
| CA4 | -0.0570 | 0.9446 | 0.0577 | -0.9884 | 0.3230 |
| HK3 | 0.2213 | 1.2477 | 0.1045 | 2.1166 | 0.0343 |
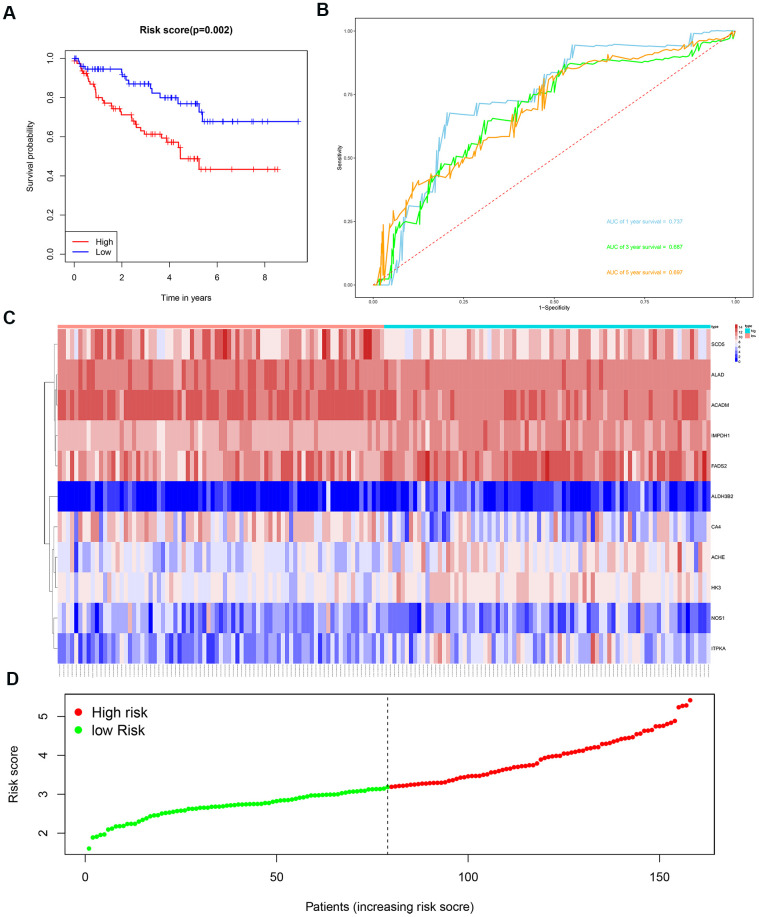
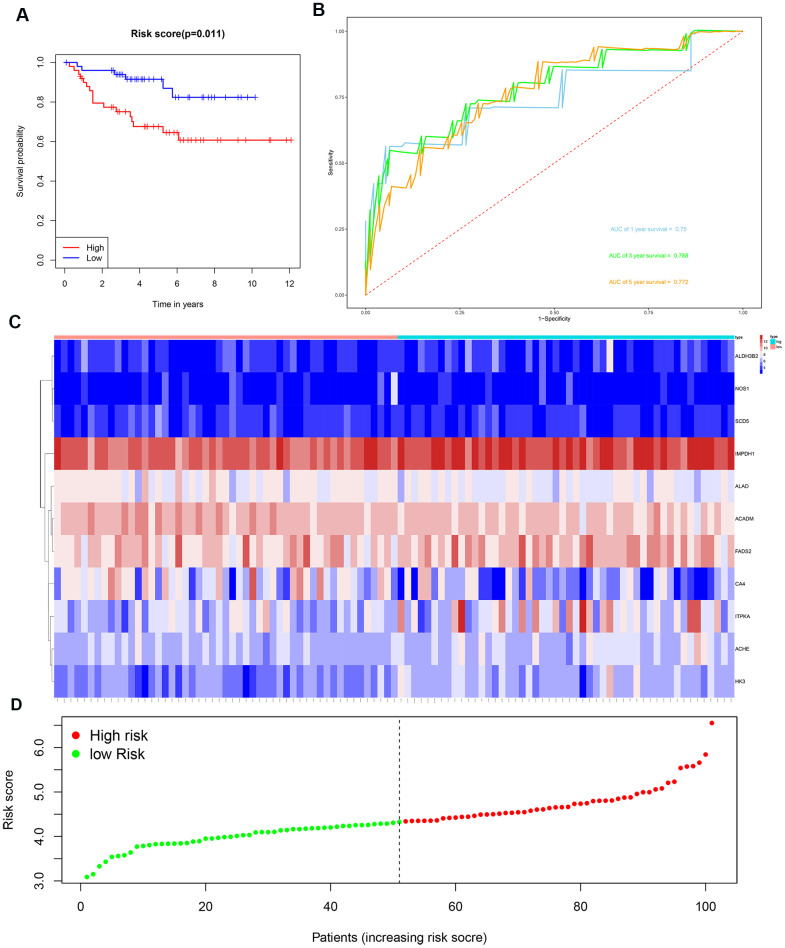
We then divided the 381 patients in the training group into high- and low-risk groups based on the median risk score. Kaplan-Meier survival curve analysis showed that the OS was significantly shorter for the high-risk group ccRCC patients compared to the low-risk group ccRCC patients (Figure 6A). Time-dependent ROC analysis of the 11-metabolic gene signature-based prognostic risk score model showed that AUC values for 1-year, 3-year and 5-year OS were 0.810, 0.738, and 0.771, respectively (Figure 6B). Figures 6C, 6D show the heat map and risk curve analyses of the eleven genes in the high- and low-risk group ccRCC patients, respectively. These results show moderate performance of the 11-metabolic gene signature-based prognostic prediction model. We then calculated the risk scores of the test group patients using the same prognostic risk score formula and assessed the predictive performance of the prognostic risk model. Kaplan-Meier survival curve analysis showed that the OS was significantly shorter for the ccRCC patients in the high-risk group compared to the low-risk group ccRCC patients (Figure 7). We obtained similar results for the E-MTAB-1980 cohort (Figure 8). These results demonstrate stable performance of the 11-metabolic gene signature-based prognostic prediction model.
Figure 6.
Risk score analysis of the 11 metabolic gene signature-based prognostic model in the training group ccRCC patients. (A) Kaplan-Meier survival curve analysis shows the overall survival of high- (n=190) and low-risk (n=191) training group ccRCC patients based on the median risk score calculated using the 11 metabolic genes-based prognostic model. (B) Time dependent ROC curve analysis shows the prognostic performance of the 11-metabolic gene signature-based prognostic model in predicting 1-year, 3-year, and 5-year survival times of the high- and low-risk training group ccRCC patients. (C) Heat map shows the expression of the 11 metabolic genes in high- and low-risk training group ccRCC patients. (D) Risk curve analysis of the 11 metabolic genes in high- and low-risk training group ccRCC patients.
Figure 7.
Risk score analysis of the 11 metabolic gene signature-based prognostic model in the test group ccRCC patients. (A) Kaplan-Meier survival curve analysis shows the overall survival of high- (n=79) and low-risk (n=79) test group ccRCC patients based on the median risk score calculated using the 11 metabolic gene signature-based prognostic model. (B) Time dependent ROC curve analysis shows the prognostic performance of the 11 metabolic gene signature-based prognostic model in predicting 1-year, 3-year, and 5-year survival times of the high- and low-risk test group ccRCC patients. (C) Heat map shows the expression of the 11 metabolic genes in high- and low-risk test group ccRCC patients. (D) Risk curve analysis of the 11 metabolic genes in high- and low-risk test group ccRCC patients.
Figure 8.
Risk score analysis of the 11 metabolic gene signature-related prognostic model in the E-MTAB-1980 cohort. (A) Kaplan-Meier survival curve analysis shows the overall survival of high- (n=50) and low-risk (n=51) ccRCC patients from the E-MTAB-1980 cohort based on the median risk score calculated using the 11 metabolic gene signature-based prognostic model. (B) Time dependent ROC curve analysis shows the prognostic performance of the 11 metabolic gene signature-based prognostic model in predicting 1-year, 3-year, and 5-year survival times of the ccRCC patients from the E-MTAB-1980 cohort. (C) Heat map shows the expression of the 11 metabolic genes in high- and low-risk ccRCC patients from the E-MTAB-1980 cohort. (D) Risk curve analysis of the 11 metabolic genes in high- and low-risk ccRCC patients from the E-MTAB-1980 cohort.
We then constructed a nomogram based on the 11-metabolic gene signature to establish a quantitative method for ccRCC prognosis (Figure 9). We calculated total points for each ccRCC patient by adding up the points for each variable and normalized it to a distribution between 0 and 100. Then, we estimated the one-year, three-year and five-year survival rates for each of the ccRCC patient’s by drawing a line perpendicular to the prognosis axis and the total point’s axis. We then performed Cox regression analysis and observed that age, tumor grade, tumor stage, primary tumor location, lymph node infiltration, distant metastasis and the prognostic risk score were significantly associated with the OS of ccRCC patients (Table 3). Multiple regression analysis showed that age (P=0.003), tumor grade (P=0.016), tumor stage (P<0.001), primary tumor location (P=0.038) and the prognostic risk score (P < 0.001) were independent prognostic factors associated with OS (Table 3).
Figure 9.
The nomogram based on the 11 metabolic genes for predicting the one- year, three-year and five-year OS of ccRCC patients.
Table 3. The prognostic value of different clinical parameters.
| HR | Univariate analysis | P-value | HR | Multivariate analysis | P-value | ||
| 95%CI | 95%CI | ||||||
| Age | 2.03 | 1.40-2.93 | <0.001 | 1.76 | 1.21-2.58 | 0.003 | |
| Gender | 0.89 | 0.61-1.28 | 0.5231 | 0.86 | 0.59-1.26 | 0.429 | |
| Grade | 2.55 | 1.99-3.27 | <0.001 | 1.41 | 1.07-1.88 | 0.016 | |
| Stage | 2.03 | 1.73-2.38 | <0.001 | 2.26 | 1.59-3.21 | <0.001 | |
| T | 2.04 | 1.68-2.48 | <0.001 | 0.68 | 0.47-0.98 | 0.038 | |
| N | 0.83 | 0.69-0.99 | 0.0438 | 0.87 | 0.72-1.05 | 0.149 | |
| M | 1.85 | 1.41-2.43 | <0.001 | 0.74 | 0.44-1.23 | 0.241 | |
| Risk score | 2.72 | 2.16-3.42 | <0.001 | 1.88 | 1.41-2.51 | <0.001 |
HR, hazard ratio; CI, confidence interval.
We used Kaplan–Meier plotter (http://www.proteinatlas.org/) online tool to determine the relationship between these metabolic genes and OS, and survival curve analysis showed that all the eleven metabolic genes were associated with the OS in the ccRCC patients (Figure 10). We used the immunohistochemical-stained results from the Human Protein Atlas database (https://www.proteinatlas.org/) to determine the expression of these 11 metabolic proteins in the ccRCC tissues. The ALDH3B2 and FADS2 protein levels were significantly higher in the ccRCC tissues compared to the normal renal tissue (Figure 11C, 11H). Furthermore, NOS1, ALAD, ACADM, ITPKA, IMPDH1, SCD5, and CA4 levels were significantly reduced in the ccRCC tissues compared to the normal renal tissues (Figure 11A, 11B, 11D–11G, 11I). The expression of HK3 protein was similar in the ccRCC and normal renal tissues (Figure 11J).
Figure 10.
Prognostic value of the prognosis related metabolic genes in ccRCC by Kaplan-Meier plotter. Survival curve analysis of ccRCC patients based on the expression status of (A) NOS1; (B) ALAD; (C) ALDH3B2; (D) ACADM; (E) ITPKA; (F) IMPDH1; (G) SCD5; (H) FADS2; (I) ACHE; (J) CA4; (K) HK3 genes.
Figure 11.

The expression status of the prognosis related metabolic proteins in ccRCC and normal renal tissues in the HPA database. (A) NOS1; (B) ALAD; (C) ALDH3B2; (D) ACADM; (E) ITPKA; (F) IMPDH1; (G) SCD5; (H) FADS2; (I) CA4; (J) HK3.
DISCUSSION
Cancer cells reprogram metabolism to gain survival advantage when competing with normal cells for limited nutrient resources. The ccRCC cells demonstrate the Warburg effect characterized by enhanced glucose uptake and glycolysis in normoxia conditions [13]. Furthermore, oncogenic mutations cause changes in glycolysis, fatty acid biosynthesis, and amino acid metabolism in the ccRCC tissues [14, 15]. However, the genes that drive these metabolic changes in ccRCC tissues are not clear. Moreover, the clinical significance of the metabolic genes in the diagnosis, treatment and prognosis of ccRCC patients is not well characterized.
In the current study, we identify 124 differentially expressed metabolic genes based on transcriptome data analysis of ccRCC and normal kidney tissues from the TCGA database. Functional enrichment analysis shows that these 124 differentially expressed metabolic genes are related to pathways such as small molecule catabolism, amino acid metabolism, fatty acid metabolism, ribonucleotide metabolism, nucleoside phosphate biosynthesis, oxidoreductase activity, mitochondrial matrix, iron ion binding, transferase activity, and glycosyl transfer activity. These pathways are involved in the metabolic reprogramming-related alterations of the biological functions of ccRCC cells. Warburg effect or aerobic glycolysis is the most well characterized metabolic reprogramming process in cancer cells, wherein the cancer cells rely on lactic acid fermentation for their energy needs regardless of oxygenation levels; this metabolic reprogramming helps cancer cells to thrive under nutrient-deprived conditions commonly encountered in the tumor microenvironment and in metastasized tissues [16]. Several studies have shown abnormal expression of glucose transporters, glycolytic enzymes and some key enzymes of the tricarboxylic acid cycle in the ccRCC cells [17–19]. The tryptophan metabolic pathway is also altered in ccRCC cells because it generates metabolites that promote tumor growth and induce immunosuppression, thereby reducing the efficacy of interferon-like immunotherapy [20, 21]. Fatty acid metabolism is also altered in the ccRCC cells [22]. Fatty acid metabolism is a complex process regulated by various enzymes, including acyl-CoA dehydrogenase, hydroxyacyl-CoA dehydrogenase and enoyl-CoA hydratase [23]. Elevated expression of fatty acid synthase is associated with tumor invasiveness and poor OS rates in ccRCC patients [24]. Furthermore, alterations in the tricarboxylic acid (TCA) cycle, electron transport chain (ETC), and glutamine and arginine metabolic pathways contribute to changes in the energy supply and intrinsic antioxidant properties of the ccRCC cells, thereby promoting tumor progression [19, 25]. Therefore, alterations in the expression of metabolic genes modulate several metabolic pathways involved in the synthesis and breakdown of amino acids, fatty acids, and nucleotides, mitochondrial oxidative phosphorylation, and other pathways. Thus, metabolic reprogramming alters the ATP and oxidative stress levels in the tumor cells, and also provides metabolic intermediates that serve to modulate immune cell infiltration in the tumor microenvironment, thereby influencing tumor growth and progression.
In addition, we constructed a PPI network for these differentially expressed genes and screened 10 hub genes including GAPDH, POLR3B, NME1-NME2, ADCY10, ADCY7, POLR2F, NME1, ENTPD2, ADCY8, and ADCY3. Among these genes, ADCY10, ADCY7, ADCY8, and ADCY3 are members of adenylate cyclase family, which catalyzes ATP generation of cAMP [26]. They have different reactions to upstream regulatory pathways and their distribution, and play an important role in tumorigenesis [27]. GAPDH has been found to play an important role in tumor cell survival, tumor angiogenesis, control of tumor cell gene expression, and post-transcriptional regulation of tumor cell mRNA [28]. And we found that ccRCC is associated with cellular amino acid metabolic process, generation of precursor metabolites and energy, organic acid biosynthetic process, nucleoside phosphate biosynthetic process, ribonucleotide metabolic process, and metabolic pathways through the PPI network module analysis.
Moreover, our analysis identified eleven prognosis-related metabolic genes, including NOS1, ALAD, ALDH3B2, ACADM, ITPKA, IMPDH1, SCD5, FADS2, ACHE, CA4, and HK3. NOS1 is a nitric oxide synthase that generates nitric oxide and plays an important role in the development, progression and metastasis of various tumors [29]. NOS1 also generates reactive oxygen species including superoxide ions (O2−) and hydrogen peroxide (H2O2) at low arginine concentrations [30]. The activity of NOS1 is closely related to protein tyrosine nitration [31]. Renaudin et al. demonstrated that NOS1 expression correlated with the pathological grade of renal tumors [32]. ALAD catalyses the second step of heme biosynthesis and is an endogenous inhibitor of 26S proteasome, which is a therapeutic target for several tumors [33–35]. ALAD gene variants are associated with the risk of genitourinary tumors [36]. ALDH3B2 is a member of the aldehyde dehydrogenase family that plays an important role in exogenous drug metabolism [37]. ALDH3B2 polymorphisms are related to the esophageal squamous cell carcinoma in the Chinese population [37]. ALDH3B2 also plays an important role in colorectal carcinogenesis [38, 39]. ACADM is an enzyme that catalyzes the initial step of mitochondrial fatty acid-oxidation pathway [40]. In this study, we demonstrate that ACADM is downregulated in ccRCC tissues. Huang et al. demonstrated that HIF-1 promotes various cancer progression by inhibiting fatty acid oxidation and acyl-CoA dehydrogenases [40]. Niu et al. showed that inhibition of ACDAM activity accelerated breast cancer progression in the mouse model [41]. Amino acid metabolizing enzymes play an important role in the evasion of tumor cells from immune surveillance [42]. IPTKA is a member of the inositol triphosphate kinase family that regulates actin dynamics and Ins (1, 4, 5) P3-mediated calcium signaling [43]. IPTKA is upregulated in several cancers, including those of the breast, lung, colon, liver, prostate and testis [43, 44]. Our study demonstrates that IPTKA mRNA levels are significantly increased in the ccRCC tissues. Guan et al. reported that high expression of IPTKA correlates with higher vascular infiltration and shorter survival time of liver cancer patients [45]. Liu et al. reported that ITPKA gene expression and the IPTKA subtypes correlate with the prognosis of ccRCC patients [46]. IMPDH is a rate-limiting enzyme in the biosynthesis pathway of guanosine triphosphate. It has two subtypes, IMPDH1 and IMPDH2, and is essential for DNA and RNA synthesis and signal transmission in all organisms [47]. In this study, we demonstrate that IMPDH1 mRNA levels are significantly increased in ccRCC tissues. Ruan et al. also reported that high IMPDH1 expression is associated with shorter overall survival and disease-free survival of RCC patients [47]. In a mouse model of non-small cell lung cancer (NSCLC), inhibition of IMPDH1 expression reduced the expression of RNA polymerase-I-dependent pre-ribosomal RNA expression, inhibited the growth of tumor cells, and improved the survival of NSCLC model mice treated with chemotherapy [48]. SCD proteins regulate the biosynthesis of cellular lipid fatty acids. SCD1 and SCD5 are the main subtypes in humans. The monounsaturated fatty acids produced by these enzymes promote the mobility of cell membranes and the growth rate of cancer cells. SCD1 is up-regulated in cancer cells and plays an important role in tumor progression [49]. Our study found that SCD5 mRNA levels were significantly down-regulated in ccRCC tissues. Bellenghi et al. reported that the expression of SCD5 was significantly higher in the primary melanoma cells, but significantly decreased in the metastatic melanoma cells; SCD5 overexpression in the melanoma mouse model significantly reduced the invasiveness of the melanoma cells [49]. Fatty acid desaturase 2 (FADS2) is an enzyme that catalyzes the biosynthesis of highly unsaturated fatty acids (HUFAs) [50]. Our study demonstrates that FADS2 mRNA expression is significantly up-regulated in the ccRCC tissues. Tian et al. showed that FADS2 was overexpressed in the colorectal cancer (CRC) tissues and promoted CRC cell proliferation [50]. The ACHE gene encodes the acetylcholinesterase, an enzyme that hydrolyzes acetylcholine, which is involved in signal transmission [51]. The gene structure and expression of ACHE is altered in various tumors [51, 52]. Motamed-Khorasani et al. reported that high acetylcholinesterase levels correlated with shorter overall survival in ovarian cancer patients [53]. Perry et al. demonstrated that ACHE gene variants were associated with the aggressiveness of human astrocytomas [54]. CA4 is a class of zinc-containing metalloenzymes that catalyze the reversible hydration of carbon dioxide and the dehydration of carbonic acid [55]. The down-regulation of CA4 cognate CAI or CAII is associated with colorectal carcinogenesis [56]. Hexokinase is the first step in catalyzing glycolysis. A recent study found that HK3 plays an important role in acute promyelocytic leukemia [57]. Pudova et al. reported that HK3 overexpression was associated with the epithelial-mesenchymal transition (EMT) of colorectal cancer cells [58]. Our study suggests that these eleven key metabolic genes may participate in the functional regulation of ccRCC by modulating metabolism.
We then used multivariate Cox proportional hazards regression analysis to establish a prognostic risk score model to predict the prognosis of patients with ccRCC. The ROC curve of the risk score model indicates moderate to good performance in predicting one-year OS (AUC= 0.810), three-year OS (AUC=0.738), and five-year OS (AUC=0.771). We then developed a nomogram based on the eleven metabolic genes to help doctors determine the prognosis of the ccRCC patients and decide on the therapeutic strategy. We showed that the prognostic risk score based on these eleven genes was an independent prognostic factor associated with OS. Kaplan-Meier survival curve analysis showed that low expression of NOS1, ALAD, ACADM, SCD5, and CA4 genes and high expression of ALDH3B2, ITPKA, IMPDH1, FADS2, ACHE, and HK3 genes was associated with shorter overall survival of ccRCC patients. These results demonstrate that the prognostic risk signature of these eleven metabolic genes can be used for determining the recurrence risk stratification, treatment outcomes, prognostic prediction. These genes may also serve as potential therapeutic targets.
In the past decade, advances in molecular science and the identification of new molecular biomarkers have shed newer insights into the biology of ccRCC. This has resulted in the development of new targeted therapies and cancer-related biomarkers, including, proliferation markers such as Ki-67, p53 and PTEN, hypoxia-inducible factor pathways, carbonic anhydrase IX, vascular endothelial growth factor (VEGF) and others [59]. Several studies have investigated the relationship between the somatic mutations, variations in gene methylation, differential gene expression, germline variations, and the status of immune biomarkers such as CD8 and PD-L1 with prognosis of ccRCC, and several different prognosis models have been proposed [60–62]. Brannon et al. identified two subtypes of ccRCC based on stratified consensus clustering of gene expression microarray data, and developed a 34-gene classifier for localized ccRCC [63]. Rini et al. analyzed the expression of 732 genes in 942 patients with stage I-III ccRCC, and selected eleven genes of interest including five reference genes to determine a continuous recurrence score [64]. Klatte et al. used tissue microarray technology to determine the correlation between the expressions of Ki-67, p53, VEGFR-1 and VEGF-D with ccRCC patient survival time, and constructed a prognostic model in combination with other clinical factors [65]. Recently, Zhao et al. constructed a prognostic model based on the expression status of three prognostic N6-methyladenosine genes [66]. In this study, we systematically analyzed the expression of metabolic genes that regulate metabolic reprogramming in ccRCC and developed a new prognostic model based on 11 metabolic genes.
This study has some limitations. Firstly, our study is based only on bio-omics data. However, we need to consider that analysis of different patient characteristics on different platforms can lead to patient heterogeneity. Secondly, the prognostic model construction and verification in our study was based on retrospective data analysis. Therefore, our findings need to be verified with a multicenter, large prospective cohort of ccRCC patients. Thirdly, variability in the clinical information from different datasets may reduce the statistical reliability and effectiveness of the Cox regression analysis. Finally, we used bioinformatics techniques to evaluate the diagnostic and prognostic prediction value of several key metabolic genes in ccRCC. However, the specific functions and mechanisms of these key metabolic genes in the growth and progression of ccRCC have not been well characterized, and hence, require further in-depth investigations.
In summary, we systematically studied the biological function and prognostic value of differentially expressed metabolic genes in ccRCC by a series of bioinformatics techniques. We also established a prognostic risk score model based on 11 metabolic genes, which proved to be an independent prognostic factor that can accurately predict the overall survival time of ccRCC patients. Our results will be of great significance in revealing the pathogenesis of ccRCC and developing new therapeutic targets or prognostic molecular markers.
MATERIALS AND METHODS
RNA-seq data analysis of ccRCC patients
We downloaded the transcriptome data consisting of 72 normal renal and 539 ccRCC tissue samples from The Cancer Genome Atlas database (TCGA, https://portal.gdc.cancer.gov/). We also downloaded the clinical data of all the ccRCC patients. We also obtained seventy metabolism-associated gene sets from the GSEA database (https://www.gsea-msigdb.org/gsea/msigdb/collections.jsp#C2). For data analysis, we first pre-processed the raw RNA-seq data with the edgeR package (http://www.bioconductor.org/packages/release/bioc/html/edgeR.html). This included averaging the genes with the same name, removing the genes with average expression of less than 1, and normalizing the expression data with the trimmed mean of M-values (TMM) normalization algorithm. We then identified the differentially expressed metabolic genes using |log2 FC| >1.0 and false discovery rate (FDR) <0.05 as the selection criteria.
Functional enrichment analysis of the differentially expressed metabolic genes
We performed a comprehensive functional and pathway enrichment analysis of the differentially expressed metabolic genes in ccRCC using the WEB-based Gene Set Analysis Toolkit (WebGestalt, http://www.webgestalt.org/). This included identifying the GO terms for the cellular components, biological processes, and molecular functions, as well as the enriched KEGG signaling pathways in which the differentially expressed genes are enriched. GO terms and KEGG pathways with P<0.05 and FDR <0.05 were considered statistically significant.
Construction of protein-protein interaction (PPI) networks and identification of key co-expression modules
The STRING database (https://string-db.org/) was used to identify the protein-protein interactions involving all the differentially expressed metabolic genes. Then, the PPI network was built using the Cytoscape 3.8.0 software (https://cytoscape.org/). The MCODE plug-in was used to select key modules based on the MCODE score and the node number. The cytoHubba plug-in was used to screen the hub genes according to the maximal clique centrality (MCC) algorithm. The GraphPad Prism 5.0 software was used to perform receiver operating characteristic (ROC) curve analyses of the hub genes to assess their ability to differentiate between normal and ccRCC tissues.
Analysis of hub gene mutation frequency and copy number variation
The cBioPortal (https://www.cbioportal.org/) database was used to determine the gene mutations (missense mutations, gene amplifications, and deletions) and copy-number alterations of the hub genes. Then, the correlation between the hub gene mutations and survival time were determined by the survival module in the cBioPortal database.
Identification of prognosis-associated metabolic genes
The caret R package was used to divide the ccRCC patients from TCGA cohort into training and test groups. Univariate Cox regression analysis was then performed for the differentially expressed metabolic genes in the training group using the survival R package. These genes were further screened using the LASSO regression analysis algorithm to identify the potential prognosis-associated metabolic genes, which were then subjected to multivariate Cox regression analysis to identify metabolic genes that can independently predict prognosis of ccRCC patients.
Evaluation of the 11-metabolic gene signature-based prognostic model
The multivariate Cox proportional hazards regression model was established based on the selected metabolic genes to predict the prognosis of patients. The risk score of each patient was calculated according to the following formula:
where, β is the regression coefficient of each gene and Exp is the expression value of each gene. We then divided the training group patients into high- and low-risk groups based on the median risk score. Then, the log-rank test was used to compare the differences in overall survival times between the two groups. Furthermore, we generated ROC curves using the survival ROC package to evaluate the model performance. The rms R package was used to construct a nomogram to predict survival probabilities. Finally, we used the test group and the E-MTAB-1980 dataset (https://www.ebi.ac.uk/arrayexpress/experiments/E-MTAB-1980/) to validate the predictive value of the prognostic model. In addition, we used the Kaplan-Meier plotter (https://kmplot.com/analysis/) online tool to verify the prognostic value of these 11 metabolic genes, and used The Human Protein Atlas (http://www.proteinatlas.org/) online database to detect the protein expression of these 11 metabolic genes.
Supplementary Material
Abbreviations
- ccRCC
Clear cell renal cell carcinoma
- RCC
Renal cell carcinoma
- TCGA
The Cancer Genome Atlas
- GSEA
Gene set enrichment analysis
- KEGG
Kyoto Encyclopedia of Genes and Genomes
- FC
Fold change
- NES
Normalized enrichment score
- PPI
Protein-protein interaction
- MCODE
Molecular Complex Detection
- OS
Overall survival
- LASSO
Least absolute shrinkage and selection operator
- ROC
receiver operating characteristic
- AUC
Area under the receiver operating characteristic curve
- FDR
False discovery rate
Footnotes
AUTHOR CONTRIBUTIONS: Y.W. designed the study and performed the data analysis; X.W., H.F., B.H. and B.L. performed the data analysis; Y.L., Y.R., X.L., Z.L., S.W. and J.L. performed the data analysis and revised the manuscript; T.W. designed the study and revised the manuscript. All authors read and approved the final manuscript.
CONFLICTS OF INTEREST: The authors declare that there are no conflicts of interest.
FUNDING: This study was financially supported by the Medical Youth Top Talent Program of Hubei Provincial.
REFERENCES
- 1.Moch H, Cubilla AL, Humphrey PA, Reuter VE, Ulbright TM. The 2016 WHO classification of tumours of the urinary system and male genital organs-part A: renal, penile, and testicular tumours. Eur Urol. 2016; 70:93–105. 10.1016/j.eururo.2016.02.029 [DOI] [PubMed] [Google Scholar]
- 2.Peng X, Chen Z, Farshidfar F, Xu X, Lorenzi PL, Wang Y, Cheng F, Tan L, Mojumdar K, Du D, Ge Z, Li J, Thomas GV, et al. , and Cancer Genome Atlas Research Network. Complexity of tumor vasculature in clear cell renal cell carcinoma. Cancer. 2009. (Suppl); 115:2282–89. 10.1002/cncr.24238 [DOI] [PubMed] [Google Scholar]
- 3.Choueiri TK, Motzer RJ. Systemic therapy for metastatic renal-cell carcinoma. N Engl J Med. 2017; 376:354–66. 10.1056/NEJMra1601333 [DOI] [PubMed] [Google Scholar]
- 4.Sternberg CN, Davis ID, Mardiak J, Szczylik C, Lee E, Wagstaff J, Barrios CH, Salman P, Gladkov OA, Kavina A, Zarbá JJ, Chen M, McCann L, et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol. 2010; 28:1061–68. 10.1200/JCO.2009.23.9764 [DOI] [PubMed] [Google Scholar]
- 5.Pavlova NN, Thompson CB. The emerging hallmarks of cancer metabolism. Cell Metab. 2016; 23:27–47. 10.1016/j.cmet.2015.12.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gaude E, Frezza C. Tissue-specific and convergent metabolic transformation of cancer correlates with metastatic potential and patient survival. Nat Commun. 2016; 7:13041. 10.1038/ncomms13041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Peng X, Chen Z, Farshidfar F, Xu X, Lorenzi PL, Wang Y, Cheng F, Tan L, Mojumdar K, Du D, Ge Z, Li J, Thomas GV, et al. Cancer Genome Atlas Research Network, Bathe OF, Liang H. Molecular Characterization and Clinical Relevance of Metabolic Expression Subtypes in Human Cancers. Cell Rep. 2018; 23:255–269.e4. 10.1016/j.celrep.2018.03.077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cancer Genome Atlas Research Network. Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature. 2013; 499:43–49. 10.1038/nature12222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Metallo CM, Gameiro PA, Bell EL, Mattaini KR, Yang J, Hiller K, Jewell CM, Johnson ZR, Irvine DJ, Guarente L, Kelleher JK, Vander Heiden MG, Iliopoulos O, Stephanopoulos G. Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia. Nature. 2011; 481:380–84. 10.1038/nature10602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gameiro PA, Yang J, Metelo AM, Pérez-Carro R, Baker R, Wang Z, Arreola A, Rathmell WK, Olumi A, López-Larrubia P, Stephanopoulos G, Iliopoulos O. In vivo HIF-mediated reductive carboxylation is regulated by citrate levels and sensitizes VHL-deficient cells to glutamine deprivation. Cell Metab. 2013; 17:372–85. 10.1016/j.cmet.2013.02.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gameiro PA, Laviolette LA, Kelleher JK, Iliopoulos O, Stephanopoulos G. Cofactor balance by nicotinamide nucleotide transhydrogenase (NNT) coordinates reductive carboxylation and glucose catabolism in the tricarboxylic acid (TCA) cycle. J Biol Chem. 2013; 288:12967–77. 10.1074/jbc.M112.396796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hakimi AA, Reznik E, Lee CH, Creighton CJ, Brannon AR, Luna A, Aksoy BA, Liu EM, Shen R, Lee W, Chen Y, Stirdivant SM, Russo P, et al. An integrated metabolic atlas of clear cell renal cell carcinoma. Cancer Cell. 2016; 29:104–16. 10.1016/j.ccell.2015.12.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ward PS, Thompson CB. Metabolic reprogramming: a cancer hallmark even warburg did not anticipate. Cancer Cell. 2012; 21:297–308. 10.1016/j.ccr.2012.02.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Weiss RH, Lin PY. Kidney cancer: identification of novel targets for therapy. Kidney Int. 2006; 69:224–32. 10.1038/sj.ki.5000065 [DOI] [PubMed] [Google Scholar]
- 15.van der Mijn JC, Panka DJ, Geissler AK, Verheul HM, Mier JW. Novel drugs that target the metabolic reprogramming in renal cell cancer. Cancer Metab. 2016; 4:14. 10.1186/s40170-016-0154-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mullen AR, Wheaton WW, Jin ES, Chen PH, Sullivan LB, Cheng T, Yang Y, Linehan WM, Chandel NS, DeBerardinis RJ. Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature. 2011; 481:385–88. 10.1038/nature10642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ozcan A, Shen SS, Zhai QJ, Truong LD. Expression of GLUT1 in primary renal tumors: morphologic and biologic implications. Am J Clin Pathol. 2007; 128:245–54. 10.1309/HV6NJVRQKK4QHM9F [DOI] [PubMed] [Google Scholar]
- 18.Li B, Qiu B, Lee DS, Walton ZE, Ochocki JD, Mathew LK, Mancuso A, Gade TP, Keith B, Nissim I, Simon MC. Fructose-1,6-bisphosphatase opposes renal carcinoma progression. Nature. 2014; 513:251–55. 10.1038/nature13557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wettersten HI, Hakimi AA, Morin D, Bianchi C, Johnstone ME, Donohoe DR, Trott JF, Aboud OA, Stirdivant S, Neri B, Wolfert R, Stewart B, Perego R, et al. Grade-dependent metabolic reprogramming in kidney cancer revealed by combined proteomics and metabolomics analysis. Cancer Res. 2015; 75:2541–52. 10.1158/0008-5472.CAN-14-1703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee GK, Park HJ, Macleod M, Chandler P, Munn DH, Mellor AL. Tryptophan deprivation sensitizes activated T cells to apoptosis prior to cell division. Immunology. 2002; 107:452–60. 10.1046/j.1365-2567.2002.01526.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Trott JF, Kim J, Abu Aboud O, Wettersten H, Stewart B, Berryhill G, Uzal F, Hovey RC, Chen CH, Anderson K, Graef A, Sarver AL, Modiano JF, Weiss RH. Inhibiting tryptophan metabolism enhances interferon therapy in kidney cancer. Oncotarget. 2016; 7:66540–57. 10.18632/oncotarget.11658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gebhard RL, Clayman RV, Prigge WF, Figenshau R, Staley NA, Reesey C, Bear A. Abnormal cholesterol metabolism in renal clear cell carcinoma. J Lipid Res. 1987; 28:1177–84. [PubMed] [Google Scholar]
- 23.Carracedo A, Cantley LC, Pandolfi PP. Cancer metabolism: fatty acid oxidation in the limelight. Nat Rev Cancer. 2013; 13:227–32. 10.1038/nrc3483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Horiguchi A, Asano T, Asano T, Ito K, Sumitomo M, Hayakawa M. Fatty acid synthase over expression is an indicator of tumor aggressiveness and poor prognosis in renal cell carcinoma. J Urol. 2008; 180:1137–40. 10.1016/j.juro.2008.04.135 [DOI] [PubMed] [Google Scholar]
- 25.Yoon CY, Shim YJ, Kim EH, Lee JH, Won NH, Kim JH, Park IS, Yoon DK, Min BH. Renal cell carcinoma does not express argininosuccinate synthetase and is highly sensitive to arginine deprivation via arginine deiminase. Int J Cancer. 2007; 120:897–905. 10.1002/ijc.22322 [DOI] [PubMed] [Google Scholar]
- 26.Patel TB, Du Z, Pierre S, Cartin L, Scholich K. Molecular biological approaches to unravel adenylyl cyclase signaling and function. Gene. 2001; 269:13–25. 10.1016/s0378-1119(01)00448-6 [DOI] [PubMed] [Google Scholar]
- 27.Chen SL, Hu F, Wang DW, Qin ZY, Liang Y, Dai YJ. Prognosis and regulation of an adenylyl cyclase network in acute myeloid leukemia. Aging (Albany NY). 2020; 12:11864–77. 10.18632/aging.103357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sirover MA. Pleiotropic effects of moonlighting glyceraldehyde-3-phosphate dehydrogenase (GAPDH) in cancer progression, invasiveness, and metastases. Cancer Metastasis Rev. 2018; 37:665–76. 10.1007/s10555-018-9764-7 [DOI] [PubMed] [Google Scholar]
- 29.Fukumura D, Kashiwagi S, Jain RK. The role of nitric oxide in tumour progression. Nat Rev Cancer. 2006; 6:521–34. 10.1038/nrc1910 [DOI] [PubMed] [Google Scholar]
- 30.Xia Y, Dawson VL, Dawson TM, Snyder SH, Zweier JL. Nitric oxide synthase generates superoxide and nitric oxide in arginine-depleted cells leading to peroxynitrite-mediated cellular injury. Proc Natl Acad Sci USA. 1996; 93:6770–74. 10.1073/pnas.93.13.6770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Williams MS, Noguchi S, Henkart PA, Osawa Y. Nitric oxide synthase plays a signaling role in TCR-triggered apoptotic death. J Immunol. 1998; 161:6526–31. [PubMed] [Google Scholar]
- 32.Renaudin K, Denis MG, Karam G, Vallette G, Buzelin F, Laboisse CL, Jarry A. Loss of NOS1 expression in high-grade renal cell carcinoma associated with a shift of NO signalling. Br J Cancer. 2004; 90:2364–69. 10.1038/sj.bjc.6601809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bardag-Gorce F, French SW. Delta-aminolevulinic dehydratase is a proteasome interacting protein. Exp Mol Pathol. 2011; 91:485–89. 10.1016/j.yexmp.2011.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen D, Dou QP. The ubiquitin-proteasome system as a prospective molecular target for cancer treatment and prevention. Curr Protein Pept Sci. 2010; 11:459–70. 10.2174/138920310791824057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Voutsadakis IA, Papandreou CN. The ubiquitin-proteasome system in prostate cancer and its transition to castration resistance. Urol Oncol. 2012; 30:752–61. 10.1016/j.urolonc.2010.03.013 [DOI] [PubMed] [Google Scholar]
- 36.Victery W, Miller CR, Zhu SY, Goyer RA. Effect of different levels and periods of lead exposure on tissue levels and excretion of lead, zinc, and calcium in the rat. Fundam Appl Toxicol. 1987; 8:506–16. 10.1016/0272-0590(87)90136-9 [DOI] [PubMed] [Google Scholar]
- 37.Yin J, Tang W, Long T, Pan H, Liu J, Lv L, Liu C, Shi Y, Zhu J, Sun Y, Shao A, Zhou Q, Ren Z, et al. Association of ALDH3B2 gene polymorphism and risk factors with susceptibility of esophageal squamous cell carcinoma in a Chinese population: a case-control study involving 2,358 subjects. Oncotarget. 2017; 8:110153–65. 10.18632/oncotarget.22656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gao ZG, Yang Y, Han XF, Wang YL, Wang ZJ. ALDH3B2 polymorphism is associated with colorectal cancer susceptibility. J Oncol. 2020; 2020:5179635. 10.1155/2020/5179635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pang B, Xu X, Lu Y, Jin H, Yang R, Jiang C, Shao D, Liu Y, Shi J. Prediction of new targets and mechanisms for quercetin in the treatment of pancreatic cancer, colon cancer, and rectal cancer. Food Funct. 2019; 10:5339–49. 10.1039/c9fo01168d [DOI] [PubMed] [Google Scholar]
- 40.Huang D, Li T, Li X, Zhang L, Sun L, He X, Zhong X, Jia D, Song L, Semenza GL, Gao P, Zhang H. HIF-1-mediated suppression of acyl-CoA dehydrogenases and fatty acid oxidation is critical for cancer progression. Cell Rep. 2014; 8:1930–42. 10.1016/j.celrep.2014.08.028 [DOI] [PubMed] [Google Scholar]
- 41.Niu Z, Shi Q, Zhang W, Shu Y, Yang N, Chen B, Wang Q, Zhao X, Chen J, Cheng N, Feng X, Hua Z, Ji J, Shen P. Caspase-1 cleaves PPARγ for potentiating the pro-tumor action of TAMs. Nat Commun. 2017; 8:766. 10.1038/s41467-017-00523-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Murray PJ. Amino acid auxotrophy as a system of immunological control nodes. Nat Immunol. 2016; 17:132–39. 10.1038/ni.3323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Windhorst S, Kalinina T, Schmid K, Blechner C, Kriebitzsch N, Hinsch R, Chang L, Herich L, Schumacher U, Mayr GW. Functional role of inositol-1,4,5-trisphosphate-3-kinase-a for motility of Malignant transformed cells. Int J Cancer. 2011; 129:1300–09. 10.1002/ijc.25782 [DOI] [PubMed] [Google Scholar]
- 44.Wang YW, Ma X, Zhang YA, Wang MJ, Yatabe Y, Lam S, Girard L, Chen JY, Gazdar AF. ITPKA gene body methylation regulates gene expression and serves as an early diagnostic marker in lung and other cancers. J Thorac Oncol. 2016; 11:1469–81. 10.1016/j.jtho.2016.05.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li J, Zhu YH, Huang P, Zhang B, Sun J, Guan XY. ITPKA expression is a novel prognostic factor in hepatocellular carcinoma. Diagn Pathol. 2015; 10:136. 10.1186/s13000-015-0374-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Liu Q, Zhao S, Su PF, Yu S. Gene and isoform expression signatures associated with tumor stage in kidney renal clear cell carcinoma. BMC Syst Biol. 2013. (Suppl 5); 7:S7. 10.1186/1752-0509-7-S5-S7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ruan H, Song Z, Cao Q, Ni D, Xu T, Wang K, Bao L, Tong J, Xiao H, Xiao W, Cheng G, Xiong Z, Liang H, et al. IMPDH1/YB-1 positive feedback loop assembles cytoophidia and represents a therapeutic target in metastatic tumors. Mol Ther. 2020; 28:1299–313. 10.1016/j.ymthe.2020.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Huang F, Ni M, Chalishazar MD, Huffman KE, Kim J, Cai L, Shi X, Cai F, Zacharias LG, Ireland AS, Li K, Gu W, Kaushik AK, et al. Inosine monophosphate dehydrogenase dependence in a subset of small cell lung cancers. Cell Metab. 2018; 28:369–82.e5. 10.1016/j.cmet.2018.06.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bellenghi M, Puglisi R, Pedini F, De Feo A, Felicetti F, Bottero L, Sangaletti S, Errico MC, Petrini M, Gesumundo C, Denaro M, Felli N, Pasquini L, et al. SCD5-induced oleic acid production reduces melanoma Malignancy by intracellular retention of SPARC and cathepsin B. J Pathol. 2015; 236:315–25. 10.1002/path.4535 [DOI] [PubMed] [Google Scholar]
- 50.Tian J, Lou J, Cai Y, Rao M, Lu Z, Zhu Y, Zou D, Peng X, Wang H, Zhang M, Niu S, Li Y, Zhong R, et al. Risk SNP-mediated enhancer-promoter interaction drives colorectal cancer through both FADS2 and AP002754.2. Cancer Res. 2020; 80:1804–18. 10.1158/0008-5472.CAN-19-2389 [DOI] [PubMed] [Google Scholar]
- 51.Stephenson J, Czepulkowski B, Hirst W, Mufti GJ. Deletion of the acetylcholinesterase locus at 7q22 associated with myelodysplastic syndromes (MDS) and acute myeloid leukaemia (AML). Leuk Res. 1996; 20:235–41. 10.1016/0145-2126(95)00146-8 [DOI] [PubMed] [Google Scholar]
- 52.Soreq H, Lapidot-Lifson Y, Zakut H. A role for cholinesterases in tumorigenesis? Cancer Cells. 1991; 3:511–16. [PubMed] [Google Scholar]
- 53.Motamed-Khorasani A, Jurisica I, Letarte M, Shaw PA, Parkes RK, Zhang X, Evangelou A, Rosen B, Murphy KJ, Brown TJ. Differentially androgen-modulated genes in ovarian epithelial cells from BRCA mutation carriers and control patients predict ovarian cancer survival and disease progression. Oncogene. 2007; 26:198–214. 10.1038/sj.onc.1209773 [DOI] [PubMed] [Google Scholar]
- 54.Perry C, Sklan EH, Birikh K, Shapira M, Trejo L, Eldor A, Soreq H. Complex regulation of acetylcholinesterase gene expression in human brain tumors. Oncogene. 2002; 21:8428–41. 10.1038/sj.onc.1205945 [DOI] [PubMed] [Google Scholar]
- 55.Tashian RE. The carbonic anhydrases: widening perspectives on their evolution, expression and function. Bioessays. 1989; 10:186–92. 10.1002/bies.950100603 [DOI] [PubMed] [Google Scholar]
- 56.Wang N, Chen Y, Han Y, Zhao Y, Liu Y, Guo K, Jiang Y. Proteomic analysis shows down-regulations of cytoplasmic carbonic anhydrases, CAI and CAII, are early events of colorectal carcinogenesis but are not correlated with lymph node metastasis. Tumori. 2012; 98:783–91. 10.1700/1217.13504 [DOI] [PubMed] [Google Scholar]
- 57.Federzoni EA, Valk PJ, Torbett BE, Haferlach T, Löwenberg B, Fey MF, Tschan MP. PU.1 is linking the glycolytic enzyme HK3 in neutrophil differentiation and survival of APL cells. Blood. 2012; 119:4963–70. 10.1182/blood-2011-09-378117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pudova EA, Kudryavtseva AV, Fedorova MS, Zaretsky AR, Shcherbo DS, Lukyanova EN, Popov AY, Sadritdinova AF, Abramov IS, Kharitonov SL, Krasnov GS, Klimina KM, Koroban NV, et al. HK3 overexpression associated with epithelial-mesenchymal transition in colorectal cancer. BMC Genomics. 2018. (Suppl 3); 19:113. 10.1186/s12864-018-4477-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sun M, Shariat SF, Cheng C, Ficarra V, Murai M, Oudard S, Pantuck AJ, Zigeuner R, Karakiewicz PI. Prognostic factors and predictive models in renal cell carcinoma: a contemporary review. Eur Urol. 2011; 60:644–61. 10.1016/j.eururo.2011.06.041 [DOI] [PubMed] [Google Scholar]
- 60.de Martino M, Haitel A, Schatzl G, Klatte T. The protease activated receptor 1 gene variation IVSn -14 A>T is associated with distant metastasis and cancer specific survival in renal cell carcinoma. J Urol. 2013; 190:1392–97. 10.1016/j.juro.2013.03.041 [DOI] [PubMed] [Google Scholar]
- 61.Stewart GD, O’Mahony FC, Laird A, Rashid S, Martin SA, Eory L, Lubbock AL, Nanda J, O’Donnell M, Mackay A, Mullen P, McNeill SA, Riddick AC, et al. Carbonic anhydrase 9 expression increases with vascular endothelial growth factor-targeted therapy and is predictive of outcome in metastatic clear cell renal cancer. Eur Urol. 2014; 66:956–63. 10.1016/j.eururo.2014.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sim SH, Messenger MP, Gregory WM, Wind TC, Vasudev NS, Cartledge J, Thompson D, Selby PJ, Banks RE. Prognostic utility of pre-operative circulating osteopontin, carbonic anhydrase IX and CRP in renal cell carcinoma. Br J Cancer. 2012; 107:1131–37. 10.1038/bjc.2012.360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Brannon AR, Reddy A, Seiler M, Arreola A, Moore DT, Pruthi RS, Wallen EM, Nielsen ME, Liu H, Nathanson KL, Ljungberg B, Zhao H, Brooks JD, et al. Molecular Stratification of Clear Cell Renal Cell Carcinoma by Consensus Clustering Reveals Distinct Subtypes and Survival Patterns. Genes Cancer. 2010; 1:152–163. 10.1177/1947601909359929 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wei X, Choudhury Y, Lim WK, Anema J, Kahnoski RJ, Lane B, Ludlow J, Takahashi M, Kanayama HO, Belldegrun A, Kim HL, Rogers C, Nicol D, et al. Recognizing the continuous nature of expression heterogeneity and clinical outcomes in clear cell renal cell carcinoma. Sci Rep. 2017; 7:7342. 10.1038/s41598-017-07191-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rini B, Goddard A, Knezevic D, Maddala T, Zhou M, Aydin H, Campbell S, Elson P, Koscielny S, Lopatin M, Svedman C, Martini JF, Williams JA, et al. A 16-gene assay to predict recurrence after surgery in localised renal cell carcinoma: development and validation studies. Lancet Oncol. 2015; 16:676–85. 10.1016/S1470-2045(15)70167-1 [DOI] [PubMed] [Google Scholar]
- 66.Klatte T, Seligson DB, LaRochelle J, Shuch B, Said JW, Riggs SB, Zomorodian N, Kabbinavar FF, Pantuck AJ, Belldegrun AS. Molecular signatures of localized clear cell renal cell carcinoma to predict disease-free survival after nephrectomy. Cancer Epidemiol Biomarkers Prev. 2009; 18:894–900. 10.1158/1055-9965.EPI-08-0786 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.