Highlights
-
•
Walking pace has been considered a predictor of future adverse health endpoints.
-
•
An inverse association was documented between walking pace and stroke risk.
-
•
With every 1 km/h increment in walking pace, the stroke risk was decreased by 13%.
-
•
Specific cut-off values for walking pace is needed, such as age, gender, etc.
Keywords: Dose response, Meta-analytic review, Stroke incidence, Walking pace
Abstract
Purpose
The extent to which walking pace is associated with a reduced risk for stroke remains unclear. This study examined the association between walking pace and stroke risk based on prospective cohort studies.
Methods
Databases of PubMed, EMBASE, Web of Science, Scopus, and China National Knowledge Internet were searched from the inception dates to January 31, 2019, for prospective cohort studies focusing on walking pace and risk of stroke in adults. Two reviewers independently extracted data and assessed the quality of the studies. The dependent measure was stroke incidence. Using random-effects models, a meta-analysis was performed to estimate the overall relative risks (RR) of stroke incidence and 95% confidence intervals (CIs) for the individuals with the fastest walking paces vs. individuals with the slowest walking paces. A dose-response relationship was also examined.
Results
After screening 1294 titles/abstracts and 14 full-text studies identified in the search, 7 studies (from 8 cohorts) were included in the meta-analysis. The 7 studies included a total of 135,645 participants (95.2% women; mean age 63.6 years) and 2229 stroke events (median follow-up time = 8.0 years). Compared to individuals in the slowest walking-pace category (median = 1.6 km/h), individuals in the fastest walking-pace category (median = 5.6 km/h) had a 44% lower risk of stroke (pooled RR = 0.56, 95%CI: 0.48–0.65). There was also a linear dose-response relationship (RR = 0.87; 95%CI: 0.83–0.91), with the risk of stroke decreased by 13% for every 1 km/h increment in baseline walking pace. We observed similar results across walking-pace assessment, type of stroke ascertainment, stroke subtypes, sex, sample size, and duration of follow-up.
Conclusion
Findings from this meta-analysis indicate that walking pace is inversely associated with the risk of stroke.
Graphical abstract
1. Introduction
Walking pace has been suggested as a predictor of future adverse health endpoints, including, but not limited to, disability,1 cardiovascular disease (CVD),2,3 dementia,4 and all-cause mortality.5,6 Because walking pace can be measured quickly and is sensitive to changes in health condition, this metric has been proposed for inclusion as a functional vital sign for health-risk assessment among individuals, especially older adults.7,8
Stroke is the second leading cause of death worldwide, accounting for 11% of total deaths.9 Also, stroke survivors often experience low quality of life in the long run.10 In addition to the high cost of medical treatment and rehabilitation, stroke may also cause immense physical and psychological pressure for a stroke patient's family. Therefore, identifying the modifiable risk factors and/or early predictors for stroke has great clinical and public health significance. In the past decade, a number of epidemiologic studies have examined the association between walking pace and the risk of stroke, but the findings have been inconsistent and inconclusive. Some prospective cohort studies reported an inverse association between walking pace and the risk of stroke,11, 12, 13, 14 while other studies did not find an association.15, 16, 17 Although a qualitative systematic review suggested that individuals with slower walking paces were more likely to have a stroke,18 no quantitative meta-analysis has yet been conducted.
We aimed to summarize quantitatively the data from the published prospective cohort studies examining the categorical association and dose–response relationship between walking pace and the risk of stroke. Findings may serve as a scientific foundation for the use of walking pace as a diagnosis-assistance tool in the prediction of stroke risk in the general adult population.
2. Methods
2.1. Search strategy
This study followed the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).19 Relevant prospective cohort studies investigating the association between walking pace and the risk of stroke were identified from the inception dates to January 31, 2019. Studies were gathered using the following databases: PubMed, EMBASE, Web of Science, Scopus, and China National Knowledge Internet. The following search terms were employed: “(stroke OR cardiovascular disease OR cerebrovascular disease) AND (cohort OR prospective OR follow-up OR longitudinal) AND (walking pace OR walking speed OR gait) AND (cox OR logistic OR hazard OR risk OR odds ratio)”. Authors also entered these search terms into Google scholar and examined reference lists of potentially relevant studies as additional citations.
2.2. Eligibility criteria
Two authors (MQ and PX) independently conducted the whole-screening process, including screening titles, abstracts, and the full text. Disagreement regarding study inclusion was resolved through group discussion with the involvement of a third author (KH). Studies were reviewed against the following prespecified inclusion criteria: 1) they employed a prospective cohort study design; 2) they reported the relative risk (RR) and corresponding 95% confidence interval (CI) for any stroke subtypes in relation to walking pace (timed walking-pace test or self-report), or these statistics were derivable; and 3) they were published in the English or Chinese language.
2.3. Data extraction
Two authors (MQ and PX) also independently extracted the information from each included study; disagreement was resolved and consensus was reached through group discussion with the involvement of a third author (KH). The following information was extracted: The first author's last name, publication year, country where the study was performed, study name (if available), age at baseline, proportion of males among participants, duration of follow-up, number of participants, number of stroke events, walking-pace categories (e.g., slow, average, or fast), walking-pace assessment (e.g., timed walking-pace test or self-report), type of stroke ascertainment (e.g., medical records or self-report), adjusted RRs and 95%CI for each walking pace category, and variables adjusted in the final model.
2.4. Assessment of study quality
Study quality was assessed based on guidelines developed by the U.S. Preventive Task Force and a modified checklist used in previous studies.4,20 Specifically, study quality was evaluated using the following criteria: 1) allowed adequate adjustment for potential confounders, such as age, sex, smoking status, and blood pressure;21,22 2) reported loss to follow-up rate; 3) contained a clear and proper definition of the exposure and outcome of interest; 4) ascertained outcomes blinded to exposure status; 5) conformed to temporality (i.e., walking pace assessed prior to outcome ascertainment); and 6) structured with at least 5 years of follow-up.23 Each criterion was assigned 1 point, with studies considered to be good quality when the overall score was ≥ 5 points, fair quality when the overall score was between 3 and 4, and poor quality if the overall score was ≤ 2.
2.5. Statistical analysis
The following statistical procedures were employed. First, the association between walking pace and the risk of stroke was analyzed by comparing the fastest to the slowest walking-pace category, and a pooled RR and corresponding 95%CI were calculated by using a random-effects model. The results of included studies that had multiple adjusted confounders were used in this meta-analysis.
Second, a dose–response analysis was performed. This method required the distribution of stroke events, person-years/nonevents, and RRs with corresponding 95%CI for at least 3 categories of walking pace, which was standardized into km/h in the data analyses. We assumed the median pace in each category of walking pace to be the corresponding RR for each included study. Among studies with an open-ended fastest walking-pace category, we assumed the open-ended category to have the same width as the adjacent category (Supplementary Table 1). In studies that reported walking-pace categories as slow, average, or fast,14,16 we identified the pace using the Compendium of Physical Activities,24 which classifies self-reported walking paces of slow, average, or fast as 3.2 km/h, 4.8 km/h, or 5.6 km/h on average on a firm surface, corresponding to 2.8 metabolic equivalents (METs), 3.5 METs or 4.3 METs, respectively. In studies that did not report number of stroke events for each walking-pace category,13,16 we used the total stroke event, number of participants, and RR in each walking-pace category to estimate these data based on the method described by Hamling et al.25 For the studies that did not report the person-years of follow-up for each walking-pace category,12,13,15,16 we estimated them using the product of the average follow-up time and the number of participants in each group.26 Finally, for the single study that reported results by sex,13 the study's sex-specified analyses were considered as 2 independent cohorts.
A dose–response relationship between walking pace and the risk of stroke was estimated using the restricted cubic spline model described by Orsini et al.,27 with 3 knots at 10th, 50th, and 90th percentiles of the exposure distribution. A p value for nonlinearity was calculated by testing the null hypothesis that the coefficient(s) of the nonlinear phases of the spline model was equal to 0. If a linear association was observed, a summary RR was estimated for every 1 km/h increment in walking pace.
Stratified analyses were conducted to assess the effect modification based on a few prespecified factors, including walking-pace assessment (timed walking-pace test vs. self-report), type of stroke ascertainment (medical records vs. self-report), stroke subtypes (ischemic vs. hemorrhagic), sex (male vs. female), sample size (>10,000 participants vs. ≤10,000 participants), and duration of follow-up (>8.6 years vs. ≤8.6 years, mean). Between-study heterogeneity was tested using the Cochran Q test and quantified by I2.28 p < 0.1 was considered significant in the Cochran Q test. Also, values of I2 < 25%, ≥25% to <50%, ≥50% to <75%, and ≥75% were defined as none, low, moderate, or high degrees of heterogeneity, respectively. Publication bias was examined visually by funnel plot and statistically by the Egger test.29 A nonparametric “trim and fill” method was conducted to estimate the overall association of interest if publication bias was observed.30 Two sensitivity analyses were performed to test the robustness of our results: 1) removing 1 primary study from the overall association estimation each time in categorical analysis; and 2) replacing self-report walking pace of slow (3.2 km/h), average (4.8 km/h), or fast (5.6 km/h) in 2 of the included studies14,16 with other multiple options based upon the Compendium of Physical Activities24 in the dose–response analysis.
All analyses were performed using STATA Version 13.0 software (STATA, College Station, TX, USA). A two-sided p < 0.05 was considered to be statistically significant.
3. Results
3.1. Identification and study selection
A total of 1308 studies were retrieved from PubMed, EMBASE, Web of Science, Scopus, and China National Knowledge Internet, and 1301 of these studies were excluded for 1 or more of the following reasons: 1) duplicates (n = 199); 2) not an original study (e.g., review) (n = 95); 3) not a prospective cohort study design (n = 54); 4) not in the English or Chinese language (n = 13); 5) walking pace was not the exposure and/or stroke risk was not the outcome (n = 900); or 6) participants were diagnosed as having cardiovascular diseases at the baseline survey, including myocardial infarction and stroke (n = 40). After exclusion of ineligible studies, a total of 7 studies from 8 cohorts (1 study that analyzed data from men and women separately considered them as 2 cohorts13) remained in the meta-analysis (Fig. 1).
Fig. 1.
Study selection process. CNKI = China National Knowledge Internet.
3.2. Study characteristics
The analysis dataset comprised 135,645 participants (95.2% women; mean age 63.6 years) and 2229 stroke events (Table 1). A total of 6 studies were from the United States,11, 12, 13, 14, 15,17 and 1 was from the United Kingdom.16 All studies were written in English and were graded as “good quality” (Supplementary Table 2). Sample sizes ranged from 148614 to 72,48811 participants (median = 4207), with the follow-up times ranging from 5.2 years12 to 11.9 years15 (median = 8.0 years; mean = 8.6 years). A total of 3 studies enrolled only women,11,12,15 1 enrolled only men,16 and the other studies included both sexes.13,14,17 To confirm the outcome of stroke, 6 studies used medical records,11, 12, 13,15, 16, 17 and 1 study used self-report methods.14 Walking pace was measured by a timed walking-pace test or by self-administered questionnaires. In 2 studies, walking pace was assessed by measuring the time in seconds that it took to walk 4 m17 and 6 m,12 respectively. In the 5 studies using self-administered questionnaires, 3 of them11,13,15 categorized normal walking pace in km/h or miles per hour (mph), and 2 of them14,16 listed walking-pace options as slow, average, or fast. Walking pace in mph was computed as km/h by multiplying mph by 0.62. A total of 6 studies identified at least 3 walking-pace categories (e.g., <3.2 km/h, 3.2–4.7 km/h, and ≥4.8 km/h), and 1 study included 2 walking-pace categories (≤3.6 km/h and >3.6 km/h). All studies were included in the categorical association analysis (fastest vs. slowest category, based on the definition of each primary study) of walking pace and the risk of stroke, and 6 studies with the minimal requirement of at least 3 walking-pace categories were included in the dose–response analysis.
Table 1.
Characteristics results of a meta-analysis of the relationship between walking pace and stroke risk.
| Source | Age at baseline (year)a | Male (%) | Duration of follow-up (year)b | No. of participants | No. of events | Walking-pace categories (km/h)c | Walking-pace assessment | Stroke ascertainment | Adjusted RR (95%CI) | Adjusted variable |
|---|---|---|---|---|---|---|---|---|---|---|
| The Nurses’ Health Study, USA (Hu et al.)11 | 40–65 | 0 | Maximum: 8.0 | 72,488 | Total stroke: 407 Subtypes: Ischemic: 258 Hemorrhagic: 109 Unknown: 40 |
<3.2; 3.2–4.7; ≥4.8 |
Self-report | Medical records | 1.00 (referent); 0.81 (0.63–1.03); 0.49 (0.36–0.68) |
Age, time (4 periods), cigarette smoking, BMI, menopausal status, parental history of myocardial infarction before age 60 years, alcohol consumption, aspirin use, and history of hypertension, diabetes, or hypercholesterolemia |
| The Women's Health Initiative study, USA (McGinn et al.)12 | 65.2 ± 7.2 | 0 | Median: 5.2 | 13,048 | Ischemic stroke: 264 | <3.8; 3.8–4.5; >4.5 |
Timed walking-pace test | Medical records | 1.00 (referent); 0.80 (0.56–1.12); 0.61 (0.43–0.86) |
Age, race/ethnicity, BMI, waist-hip ratio, depression, arthritis, hypertension, smoking, history of CHD, treated diabetes at baseline, hormone, NSAID and aspirin use, grip strength, chair stands, and self-reported general health |
| The Women's Health Study, USA (Sattelmair et al.)15 | 54.6 ± 7.1 | 0 | Mean: 11.9 | 39,315 | Total stroke: 579 Subtypes: Ischemic: 473 Hemorrhagic: 102 Unknown: 4 |
Not walk regularly; <3.2; 3.2–4.7; ≥4.8 |
Self-report | Medical records | 1.00 (referent); 0.82 (0.58–1.17); 0.77 (0.57–1.04); 0.75 (0.52–1.08) |
Age, randomized treatment assignment, smoking, alcohol, saturated fat intake, fruit and vegetable intake, fiber intake, postmenopausal hormone therapy, menopausal status, parental history of myocardial infarction, migraine aura, BMI, history of diabetes, elevated cholesterol, and hypertension |
| The British Regional Heart Study, UK (Jefferis et al.)16 | 68.3 ± 4.8 | 100 | Median: 10.9 | 2995 | Total stroke: 195 | Slow; average; brisk/fast |
Self-report | Medical records | 1.00 (referent); 0.66 (0.43–0.99); 0.64 (0.39–1.07) |
Age, region, alcohol intake, vigorous recreational or sporting activity, smoking history, social class, total cholesterol, HDL-C, log (triglycerides), SBP, antihypertensive medication, BMI, atrial fibrillation, and left ventricular hypertrophy |
| The Framingham Offspring Study, USA (Camargo, et al.)17 | 62.0 ± 9.0 | 46.0 | Median: 8.0 | 2106 | Ischemic stroke: 79 | ≤3.6; >3.6 |
Timed walking-pace test | Medical records | 1.00 (referent); 0.57 (0.29–1.14) |
Age, sex, diabetes mellitus, systolic blood pressure, prevalent cardiovascular disease, atrial fibrillation, smoking, waist-to-hipratio, total cholesterol level, apolipoprotein 4 allele, total plasma homocysteine level, and physical activity |
| The Cardiovascular Health Study, USA (Soares-Miranda et al.)13 | 72.5 ± 5.5 | 39.0 | Maximum: 10.0 | 4207 | Total stroke: 563 Subtypes: Ischemic: 464 Hemorrhagic: 74 Unknown: 25 |
<3.2; 3.2–4.8; >4.8 |
Self-report | Medical records |
Men: 1.00 (referent); 0.75 (0.56–1.02); 0.45 (0.26–0.76) Women: 1.00 (referent); 0.68 (0.54–0.86); 0.48 (0.31–0.75) |
Age, sex, race, education, income, clinical site, smoking, and BMI |
| The Sacramento Aera Latino Study on Aging, USA (Zeki Al Hazzouri et al.)14 | 70.1 ± 6.5 | 42.1 | Mean: 6.0 | 1486 | Total stroke: 142 | Slow; average; fast/very fast |
Self-report | Self-report | 1.00 (referent); 0.69 (0.47–1.02); 0.44 (0.24–0.82) |
Age, sex, education, BMI, IL-6, SBP, elevated depressive symptoms, MMSE, diabetes, atrial fibrillation, CHD, antihypertensive medication, any activities of daily living limitation, and self-rated health |
The mean ± SD or the range of years was reported.
The mean, median, or the maximum follow-up years were reported.
The range or category of walking pace was reported.
Abbreviations: BMI = body mass index; CHD = coronary heart disease; CI = confidence interval; HDL-C = high-density lipoprotein-cholesterol; IL-6 = interleukin-6; km/h = kilometers per hour; MMSE = modified mini-mental state exam score; NSAID = nonsteroidal anti-inflammatory drugs; PA = physical activity; RR = relative risk; SBP = systolic blood pressure.
3.3. The fastest vs. the slowest category of walking pace
The random-effects model indicated an overall inverse association between walking pace and the risk of stroke. Compared to individuals in the slowest walking-pace category (median = 1.6 km/h), individuals in the fastest walking-pace category (median = 5.6 km/h) had a 44% lower risk of stroke (pooled RR = 0.56; 95%CI: 0.48–0.65; n = 7 studies) (Fig. 2); the heterogeneity was not significant among studies (I2 = 0.0; p = 0.62).
Fig. 2.
Meta-analysis for the fastest vs. the slowest walking pace and the risk of stroke. CI = confidence interval; M = men; RR = relative risk; W = women.
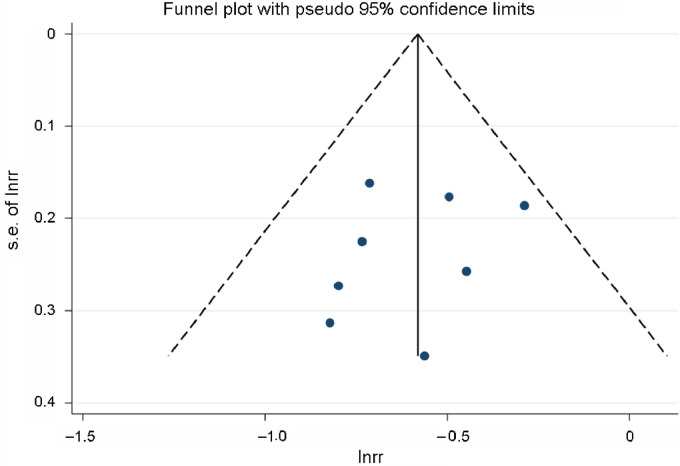
3.4. Publication bias
The funnel plot is shown in Fig. 3. Moreover, the Egger test was used to examine for publication bias. The results indicated that no significant publication bias occurred (p = 0.54).
Fig. 3.
Funnel plot for walking pace and the risk of stroke. rr = relative risk; s.e. = standard error.
3.5. Dose–response relationship analysis
Data from 6 studies including 2150 stroke events were used. A linear dose–response relation was observed between walking pace and the risk of stroke (pnonlinearity = 0.81). The risk of stroke was decreased by 13% (RR = 0.87; 95%CI: 0.83–0.91) for every 1 km/h increment in walking pace (Fig. 4).
Fig. 4.
The linear dose–response relationship between walking pace and the risk of stroke.
3.6. Subgroup and sensitivity analyses
When comparing the fastest to the slowest walking-pace categories (median: 5.6 km/h vs. 1.6 km/h), the observed inverse associations between walking pace and the risk of stroke was not materially modified by walking-pace assessment, type of stroke ascertainment, stroke subtypes, sex, sample size, or duration of follow up (Table 2).
Table 2.
Subgroup analyses according to characteristics of included studies.
| Potential modifiers | No. of studies | RRs (95%CI) | pheterogeneity | I2 (%) |
|---|---|---|---|---|
| Walking pace (high vs. low category) | ||||
| All studies | 7 | 0.56 (0.48–0.65) | 0.62 | 0.0 |
| Walking-pace assessment | ||||
| Timed walking pace | 2 | 0.60 (0.44–0.82) | 0.86 | 0.0 |
| Self-report | 5 | 0.55 (0.46–0.65) | 0.41 | 0.5 |
| Stroke ascertainment | ||||
| Medical records | 6 | 0.57 (0.49–0.67) | 0.58 | 0.0 |
| Self-report | 1 | 0.44 (0.24–0.67) | – | – |
| Stroke subtype | ||||
| Ischemic | 5 | 0.59 (0.45–0.76) | 0.13 | 48.0 |
| Hemorrhagic | 3 | 0.46 (0.29–0.73) | 0.54 | 0.0 |
| Sex | ||||
| Male | 2 | 0.54 (0.38–0.78) | 0.45 | 0.0 |
| Female | 4 | 0.57 (0.47–0.70) | 0.29 | 20.1 |
| Sample size | ||||
| >10,000 | 3 | 0.60 (0.47–0.76) | 0.22 | 33.2 |
| ≤10,000 | 4 | 0.51 (0.40–0.65) | 0.85 | 0.0 |
| Duration of follow-up (year) | ||||
| >8.6 | 3 | 0.59 (0.46–0.75) | 0.31 | 16.0 |
| ≤8.6 | 4 | 0.53 (0.43–0.65) | 0.74 | 0.0 |
Abbreviations: CI = confidence interval; RR = relative risk.
Results from sensitivity analyses indicated that the estimated overall association between walking pace and the risk of stroke was not significantly influenced by excluding any single study (Supplementary Table 3). In addition, the sensitivity analyses of using the multiple assumption of self-report walking pace in the spline analysis did not change the results materially (Supplementary Table 4).
4. Discussion
In this meta-analysis of prospective cohort studies, an inverse association was documented between walking pace and the risk of stroke. Those in the fastest walking-pace category had a 44% lower risk of stroke when compared to those in the slowest walking-pace category. Furthermore, the risk of stroke was decreased by 13% for each 1 km/h increment of walking pace.
As noted in subgroup analyses, associations between walking pace and stroke risk were consistent and had a 40%–56% reduction in risk when analyzed by walking pace, type of stroke, stroke subtypes, study sample sizes, sex distribution, and the duration of follow-up. This suggests that the relationship between walking pace and the risk of stroke is robust, regardless of the method used to assess walking pace, stroke status, and study characteristics.
4.1. Comparison with findings from previous reviews
Findings from this study are comparable to findings in other studies of the association of walking pace with risk of CVD.2,3 These studies have shown a 48% reduction in the risk of CVD when comparing the fastest to the slowest levels of walking pace. Furthermore, findings from the present study are consistent with the findings from a previous qualitative review18 that concluded that older adults with slower walking paces had a higher risks of stroke. However, the conclusions from the qualitative review18 were generated mostly from cross-sectional studies with relatively lower qualities of evidence. Therefore, the present meta-analysis quantitatively combined results from prospective cohort studies and, thus, provides additional information and a relatively higher quality of evidence concerning the relationship between walking pace and the risk of stroke.
4.2. Potential mechanisms
Several explanations might be posited regarding why fast walking pace is associated with lower risk of stroke. First, walking pace is considered one of the most important physical capabilities and an indicator of overall health.31 Apparently healthy individuals who are able to walk at a brisk pace are more likely to participate in daily physical activities than those with lower walking abilities.32,33 Furthermore, brisk walking is an important protective factor for stroke risk, as shown in published studies.34, 35, 36 Second, walking pace is associated with cardiorespiratory fitness. Available evidence indicates that walking pace is highly correlated with Six-Minute Walk Test performance,37 which is considered to be a field measure of cardiorespiratory fitness38 and is strongly and inversely associated with the risk of stroke.39, 40, 41 Third, walking pace is significantly correlated with muscle strength. Muscle loss is strongly associated with higher inflammation levels,42 notably linked to the onset of stroke.43 Finally, walking pace may be indicative of adverse cerebrovascular structure changes. Early changes in cerebral vascular structure, such as white matter hyperintensities, are well-recognized predictors of stroke and dementia as detected by brain magnetic resonance imaging.44 Furthermore, prior research has shown that vascular abnormalities are significantly associated with walking pace.45 Therefore, given the sensitivity of walking pace to changes in brain vascular structure, walking pace may have potential utility as an effective indicator of potential risk of stroke.
4.3. Strengths and limitations
This study has strengths that should be noted. The meta-analysis is based on data from prospective cohort studies, which is regarded as a study design that substantially reduces selection and recall bias. Furthermore, while randomized controlled trials provide the greatest ability to establish causal relationships, it is nearly impossible to assign a walking pace to an individual during a long-term trial. Thus, the pooled results from prospective cohort studies may represent the best empirical data available for understanding the true association between walking pace and the risk of stroke. Additionally, most of the included studies had large sample sizes and long follow-up durations, both of which contributed to more robust data available for the analysis. Our conclusions are strengthened by pooling data for both the categorical association and the dose–response relation analyses of walking pace and stroke risk.
Some limitations need to be acknowledged. First, the walking-pace assessment methods, referent group of walking pace, and stroke ascertainment varied across primary studies. These differences might generate heterogeneity and lower the precision of the overall association estimation. The classification of walking-pace data in 5 of the 7 studies were self-reported, so individual perceptions of walking pace may differ by age, sex, body mass, and physical abilities. Such differences might have increased the variability of the pooled results. Although every attempt was made to address differences in measuring walking pace, misclassification of walking pace cannot be excluded as an alternative explanation of the results. Nevertheless, sensitivity analyses did not indicate any single study or self-report walking-pace assumption that significantly affected the overall association between walking pace and the risk of stroke. Second, we were unable to assess the effects of changes in walking pace on the risk of stroke over the longitudinal study period because the included studies reported walking pace during the baseline survey only. Third, we were unable to consider some subgroup analyses, such as race and geographic locations, in the data analysis because only a limited number of studies contained similar demographic data, and the majority of the studies were from the United States. This limited our ability to detect potential effect modifiers and generalize our findings to a wider population. The relatively small number of included studies also limited the statistical power of this study, especially the ability to examine the potential threshold association between walking pace and the risk of stroke. The current study combined the results from the latest and most comprehensive literature, but additional cumulative evidence is needed to further address the modification of effects on the association between walking pace and stroke risk.
5. Conclusion
Findings from this meta-analysis of prospective cohort studies suggest that a fast walking pace (median = 5.6 km/h) conveys a 44% lower risk of stroke and that for every 1 km/h increment in walking pace, the risk of stroke is decreased by 13%. Because walking is safe, cost-effective, and easy to assess and interpret as a predictive tool, walking pace may be considered an important indicator of stroke occurrence. To increase the generalizability of these findings, well-designed, large-scale prospective cohort studies are needed to determine the age, gender, and population-specified cut-off values for walking pace to improve the effectiveness and efficiency of this early indicator of stroke.
Acknowledgments
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 81703252, to MQ) and Shanghai Key Lab of Human Performance, Shanghai University of Sport (No. 11DZ2261100).
Authors’ contributions
MQ performed the literature search, conceived and designed the study, analyzed the data, and drafted the manuscript; PX performed the literature search, analyzed the data, advised on interpretation of the data, and critically revised the manuscript; RW critically revised the manuscript; KH conceived and designed the study, advised on analysis and interpretation of the data, and critically revised the manuscript; PC advised on analysis and interpretation of the data and critically revised the manuscript. All authors have read and approved the final version of the manuscript, and agree with the order of presentation of the authors
Competing interest
The authors declare that they have no competing interests.
Footnotes
Peer review under responsibility of Shanghai University of Sport.
Supplementary materials associated with this article can be found in the online version at doi:10.1016/j.jshs.2019.09.005.
Contributor Information
Ka He, Email: kk3399@cumc.columbia.edu.
Peijie Chen, Email: chenpeijie@sus.edu.cn.
Appendix. Supplementary materials
References
- 1.Perera S., Patel K.V., Rosano C., Rubin S.M., Satterfield S., Harris T. Gait speed predicts incident disability: a pooled analysis. J Gerontol A Biol Sci Med Sci. 2016;71:63–71. doi: 10.1093/gerona/glv126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hamer M., Chida Y. Walking and primary prevention: a meta-analysis of prospective cohort studies. Br J Sports Med. 2008;42:238–243. doi: 10.1136/bjsm.2007.039974. [DOI] [PubMed] [Google Scholar]
- 3.Veronese N., Stubbs B., Volpato S., Zuliani G., Maggi S., Cesari M. Association between gait speed with mortality, cardiovascular disease and cancer: a systematic review and meta-analysis of prospective cohort studies. J Am Med Dir Assoc. 2018;19 doi: 10.1016/j.jamda.2018.06.007. 981–8.e7. [DOI] [PubMed] [Google Scholar]
- 4.Quan M., Xun P., Chen C., Wen J., Wang Y., Wang R. Walking pace and the risk of cognitive decline and dementia in elderly populations: a meta-analysis of prospective cohort studies. J Gerontol A Biol Sci Med Sci. 2017;72:266–270. doi: 10.1093/gerona/glw121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Liu B., Hu X., Zhang Q., Fan Y., Li J., Zou R. Usual walking speed and all-cause mortality risk in older people: A systematic review and meta-analysis. Gait Posture. 2016;44:172–177. doi: 10.1016/j.gaitpost.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 6.Stamatakis E., Kelly P., Strain T., Murtagh E.M., Ding D., Murphy M.H. Self-rated walking pace and all-cause, cardiovascular disease and cancer mortality: individual participant pooled analysis of 50 225 walkers from 11 population British cohorts. Br J Sports Med. 2018;52:761–768. doi: 10.1136/bjsports-2017-098677. [DOI] [PubMed] [Google Scholar]
- 7.Fritz S., Lusardi M. White paper: “Walking speed: the sixth vital sign.”. J Geriatr Phys Ther. 2009;32:46–49. [PubMed] [Google Scholar]
- 8.Middleton A., Fritz S.L., Lusardi M. Walking speed: the functional vital sign. J Aging Phys Act. 2015;23:314–322. doi: 10.1123/japa.2013-0236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.GBD 2015 Mortality and Causes of Death Collaborators Global, regional, and national life expectancy, all-cause mortality, and cause-specificmortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015. The Lancet. 2016;388:1459–1544. doi: 10.1016/S0140-6736(16)31012-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hankey G.J. Long-term outcome after ischaemic stroke/transient ischaemic attack. Cerebrovasc Dis. 2003;16(Suppl. 1):S14–19. doi: 10.1159/000069936. [DOI] [PubMed] [Google Scholar]
- 11.Hu F.B., Stampfer M.J., Colditz G.A., Ascherio A., Rexrode K.M., Willett W.C. Physical activity and risk of stroke in women. JAMA. 2000;283:2961–2967. doi: 10.1001/jama.283.22.2961. [DOI] [PubMed] [Google Scholar]
- 12.McGinn A.P., Kaplan R.C., Verghese J., Rosenbaum D.M., Psaty B.M., Baird A.E. Walking speed and risk of incident ischemic stroke among postmenopausal women. Stroke. 2008;39:1233–1239. doi: 10.1161/STROKEAHA.107.500850. [DOI] [PubMed] [Google Scholar]
- 13.Soares-Miranda L., Siscovick D.S., Psaty B.M., Longstreth W.T., Jr, Mozaffarian D. Physical activity and risk of coronary heart disease and stroke in older adults: the cardiovascular health study. Circulation. 2016;133:147–155. doi: 10.1161/CIRCULATIONAHA.115.018323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zeki Al Hazzouri A., Mayeda E.R., Elfassy T., Lee A., Odden M.C., Thekkethala D. Perceived walking speed, measured tandem walk, incident stroke, and mortality in older Latino adults: a prospective cohort study. J Gerontol A Biol Sci Med Sci. 2017;72:676–682. doi: 10.1093/gerona/glw169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sattelmair J.R., Kurth T., Buring J.E., Lee I.M. Physical activity and risk of stroke in women. Stroke. 2010;41:1243–1250. doi: 10.1161/STROKEAHA.110.584300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jefferis B.J., Whincup P.H., Papacosta O., Wannamethee S.G. Protective effect of time spent walking on risk of stroke in older men. Stroke. 2014;45:194–199. doi: 10.1161/STROKEAHA.113.002246. [DOI] [PubMed] [Google Scholar]
- 17.Camargo E.C., Weinstein G., Beiser A.S., Tan Z.S., DeCarli C., Kelly-Hayes M. Association of physical function with clinical and subclinical brain disease: the Framingham Offspring Study. J Alzheimers Dis. 2016;53:1597–1608. doi: 10.3233/JAD-160229. [DOI] [PubMed] [Google Scholar]
- 18.Fonseca Alves D.J., Bartholomeu-Neto J., Júnior E.R., Ribeiro Zarricueta B.S., Nóbrega O.T., Córdova C. Walking speed, risk factors, and cardiovascular events in older adults: systematic review. J Strength Cond Res. 2017;31:3235–3244. doi: 10.1519/JSC.0000000000002182. [DOI] [PubMed] [Google Scholar]
- 19.Moher D., Liberati A., Tetzlaff J., Altman D.G., PRISMA Group Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6 doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Harris R.P., Helfand M., Woolf S.H., Lohr K.N., Mulrow C.D., Teutsch S.M. Current methods of the US Preventive Services Task Force: a review of the process. Am J Prev Med. 2001;20(Suppl. 1):S21–35. doi: 10.1016/s0749-3797(01)00261-6. [DOI] [PubMed] [Google Scholar]
- 21.Boysen G., Nyboe J., Appleyard M., Sorensen P.S., Boas J., Somnier F. Stroke incidence and risk factors for stroke in Copenhagen, Denmark. Stroke. 1988;19:1345–1353. doi: 10.1161/01.str.19.11.1345. [DOI] [PubMed] [Google Scholar]
- 22.Wolf P.A., D'Agostino R.B., Belanger A.J., Kannel W.B. Probability of stroke: a risk profile from the Framingham Study. Stroke. 1991;22:312–318. doi: 10.1161/01.str.22.3.312. [DOI] [PubMed] [Google Scholar]
- 23.Li J., Siegrist J. Physical activity and risk of cardiovascular aisease—ameta-analysis of prospective cohort studies. Int J Environ Res Public Health. 2012;9:391–407. doi: 10.3390/ijerph9020391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ainsworth B.E., Haskell W.L., Herrmann S.D., Meckes N., Bassett D.R., Jr, Tudor-Locke C. 2011 Compendium of physical activities: a second update of codes and MET values. Med Sci Sports Exerc. 2011;43:1575–1581. doi: 10.1249/MSS.0b013e31821ece12. [DOI] [PubMed] [Google Scholar]
- 25.Hamling J., Lee P., Weitkunat R., Ambuhl M. Facilitating meta-analyses by deriving relative effect and precision estimates for alternative comparisons from a set of estimates presented by exposure level or disease category. Stat Med. 2008;27:954–970. doi: 10.1002/sim.3013. [DOI] [PubMed] [Google Scholar]
- 26.Moskal A., Norat T., Ferrari P., Riboli E. Alcohol intake and colorectal cancer risk: a dose–response meta-analysis of published cohort studies. Int J Cancer. 2007;120:664–671. doi: 10.1002/ijc.22299. [DOI] [PubMed] [Google Scholar]
- 27.Orsini N., Li R., Wolk A., Khudyakov P., Spiegelman D. Meta-analysis for linear and nonlinear dose–response relations: examples, an evaluation of approximations, and software. Am J Epidemiol. 2012;175:66–73. doi: 10.1093/aje/kwr265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Higgins J.P., Thompson S.G. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–1558. doi: 10.1002/sim.1186. [DOI] [PubMed] [Google Scholar]
- 29.Egger M., Davey Smith G., Schneider M., Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Weinhandl E.D., Duval S. Generalization of trim and fill for application in meta-regression. Res Synth Methods. 2012;3:51–67. doi: 10.1002/jrsm.1042. [DOI] [PubMed] [Google Scholar]
- 31.Cooper R., Kuh D., Hardy R., Mortality Review Group FALCon and HALCyon Study Teams Objectively measured physical capability levels and mortality: systematic review and meta-analysis. BMJ. 2010;341:c4467. doi: 10.1136/bmj.c4467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McHugh J.E., Lawlor B.A. Perceived health status is associated with hours of exercise per week in older adults independent of physical health. J Phys Act Health. 2013;10:1102–1108. doi: 10.1123/jpah.10.8.1102. [DOI] [PubMed] [Google Scholar]
- 33.Granger E., Di Nardo F., Harrison A., Patterson L., Holmes R., Verma A. A systematic review of the relationship of physical activity and health status in adolescents. Eur J Public Health. 2017;27:100–106. doi: 10.1093/eurpub/ckw187. [DOI] [PubMed] [Google Scholar]
- 34.Lee C.D., Folsom A.R., Blair S.N. Physical activity and stroke risk: a meta-analysis. Stroke. 2003;34:2475–2481. doi: 10.1161/01.STR.0000091843.02517.9D. [DOI] [PubMed] [Google Scholar]
- 35.Wendel-Vos G.C., Schuit A.J., Feskens E.J., Boshuizen H.C., Verschuren W.M., Saris W.H. Physical activity and stroke. a meta-analysis of observational data. Int J Epidemiol. 2004;33:787–798. doi: 10.1093/ije/dyh168. [DOI] [PubMed] [Google Scholar]
- 36.Li J., Siegrist J. Physical activity and risk of cardiovascular disease: a meta-analysis of prospective cohort studies. Int J Environ Res Public Health. 2012;9:391–407. doi: 10.3390/ijerph9020391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kamiya K., Hamazaki N., Matsue Y., Mezzani A., Corrà U., Matsuzawa R. Gait speed has comparable prognostic capability to six-minute walk distance in older patients with cardiovascular disease. Eur J Prev Cardiol. 2018;25:212–219. doi: 10.1177/2047487317735715. [DOI] [PubMed] [Google Scholar]
- 38.Solway S., Brooks D., Lacasse Y., Thomas S. A qualitative systematic overview of the measurement properties of functional walk tests used in the cardiorespiratory domain. Chest. 2001;119:256–270. doi: 10.1378/chest.119.1.256. [DOI] [PubMed] [Google Scholar]
- 39.Pandey A., Patel M.R., Willis B., Gao A., Leonard D., Das S.R. Association between midlife cardiorespiratory fitness and risk of stroke: the Cooper Center Longitudinal Study. Stroke. 2016;47:1720–1726. doi: 10.1161/STROKEAHA.115.011532. [DOI] [PubMed] [Google Scholar]
- 40.Kurl S., Laukkanen J.A., Rauramaa R., Lakka T.A., Sivenius J., Salonen J.T. Cardiorespiratory fitness and the risk for stroke in men. Arch Intern Med. 2003;163:1682–1688. doi: 10.1001/archinte.163.14.1682. [DOI] [PubMed] [Google Scholar]
- 41.Hooker S.P., Sui X., Colabianchi N., Vena J., Laditka J., LaMonte M.J. Cardiorespiratory fitness as a predictor of fatal and nonfatal stroke in asymptomatic women and men. Stroke. 2008;39:2950–2957. doi: 10.1161/STROKEAHA.107.495275. [DOI] [PubMed] [Google Scholar]
- 42.Cesari M., Penninx B.W., Pahor M., Lauretani F., Corsi A.M., Rhys Williams G. Inflammatory markers and physical performance in older persons: the InCHIANTI study. J Gerontol A Biol Sci Med Sci. 2004;59:242–248. doi: 10.1093/gerona/59.3.m242. [DOI] [PubMed] [Google Scholar]
- 43.Libby P. Inflammation and cardiovascular disease mechanisms. Am J Clin Nutr. 2006;83:456S–460S. doi: 10.1093/ajcn/83.2.456S. [DOI] [PubMed] [Google Scholar]
- 44.Debette S., Markus H.S. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ. 2010;341:c3666. doi: 10.1136/bmj.c3666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rosano C., Kuller L.H., Chung H., Arnold A.M., Longstreth W.T., Jr, Newman A.B. Subclinical brain magnetic resonance imaging abnormalities predict physical functional decline in high-functioning older adults. J Am Geriatr Soc. 2005;53:649–654. doi: 10.1111/j.1532-5415.2005.53214.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.