Abstract
Ursolic acid (UA), a natural pentacyclic triterpenoid, possesses widespread biological and pharmacological activities. However, drawbacks such as low bioavailability, poor targeting and rapid metabolism greatly hinder its further clinical application. Recently, with the development of nanotechnology, various UA nanosystems have emerged as promising strategies for effective cancer therapy. This article reviews various types of UA-based nano-delivery systems, primarily with emphasis placed on novel UA-based carrier-free nano-drugs, which are considered to be innovative methods for cancer therapy. Moreover, this review presents carrier-free nano-drugs that co-assembled of UA and photosensitizers that displayed synergistic antitumor performance. Finally, the article also describes the development and challenges of UA nanosystems for future research in this field. Overall, the information presented in this review will provide new insight into the rational utilization of nano-drugs in cancer therapy.
Keywords: Ursolic acid, Nanosytems, Carrier-free, Photosensitizer, Anticancer
Graphical abstract
1. Introduction
Cancer is a devastating disease that exerts influential public health trouble in the world [1,2]. Current treatment options for cancer include surgery, radiotherapy, chemotherapy, hormonal therapy, and immunotherapy or combinational chemotherapy, etc. Among them, surgical resection remains a preferred treatment for patients with solid tumors, such as breast cancer, colon cancer, rectum cancer, lung cancer and so on [3]. However, most surgical approaches exist limitations of low resection rates and high recurrence rates [4]. The majority of patients also received adjuvant chemotherapy after surgical resection to ameliorate the risk of recurrence. Therefore, chemotherapy becomes the first treatment option for metastatic or advanced tumors to improve the survival rate of cancer patients [4]. Current anticancer drugs available in the market are mainly consist of conventional chemotherapeutic drugs, molecular-targeted drugs, immune checkpoint antagonists, and hormonal therapy drugs, such as paclitaxel, sorafenib, tremelimumab, avelumab, dexamethasone and so on [5], [6], [7], [8]. Despite the fact that encouraging progress has been achieved in clinical cancer therapies, current treatments have been identified with some disadvantages (e.g., multidrug resistance (MDR), poor sensitivity and serious side effects) that failed to eradicate tumors or prevent their recurrence [9], [10], [11]. Therefore, it is in urgent need to overcome the shortcomings of current cancer therapies and further explore novel anticancer drugs with superior efficacy and low toxicity for the treatment of neoplastic disease.
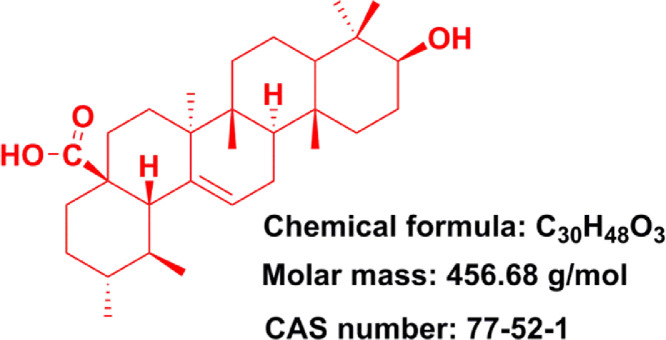
Pentacyclic triterpenoids is an important class of bioactive natural compounds and which has gained much attention because of its superior therapeutic actions and fewer adverse effects in cancer therapy [12], [13], [14]. Ursolic acid (UA) (3b-hydroxy-urs-12-en-28-oic acid, Fig. 1), one of the pentacyclic triterpene carboxylic acids, can be extracted from various plants (e.g., Sanguisorba Officinalis, Rosmarinus Officinalis, Salvia officinalis, etc.) [15], [16], [17]. UA was confirmed to possess multiple pharmacological properties including antidiabetic [18], anti-inflammatory [19], antioxidant [20], antimicrobial [21], anti-hyperlipidaemia, etc [22]. In particular, UA and its derivatives were also found to effectively inhibit a series of cancer cell growth [23], [24], [25], [26], indicating their great potential to be served as promising chemotherapeutic agents for the treatment of human cancers. However, the poor water solubility and permeability of UA greatly hindered its preclinical or clinical applications. Various preclinical trials have revealed that oral administration of UA performed poor intestinal absorption and UA was rapidly eliminated by gut wall/liver metabolism, ultimately resulting in low bioavailability of UA [27,28]. For instance, when administered orally, UA only displayed a half-life of 0.71 ± 0.09 h and Cmax (peak concentration) of 1.10 ± 0.31 µg/ml in rats [27]. When administered intravenously, low molecular weight UA would easily diffuse throughout the body and further resulted in nonspecific distribution [29]. Notably, these disadvantages seriously hindered the clinical application of UA. In recent decades, increasing evidence suggested that constructing nano-drug delivery systems were able to effectively improve bioavailability and enhance the targeting efficiency of agents.
Fig. 1.
Structure of ursolic acid (PubChem CID: 64,945).
The nano-drug delivery systems are widely applied in delivering various agents like drugs, photosensitizers, phytochemicals, biomolecules, and other compounds [30], [31], [32], [33]. Nanoparticles can efficiently permeate into tumor tissues due to the enhanced permeability and retention (EPR) effect of tumor blood vessels. In contrast, normal blood vessels will prevent nanoparticles from permeating [34]. Therefore, nano-drug delivery systems can not only improve the targeted accumulation of therapeutic agents at the tumor sites but also reduce their biodistribution in normal tissues [35]. It has also been reported that MDR and efflux transporters could be overcome through nanoparticles-mediated internalization [36]. Additionally, some investigations suggested that UA-based nano-delivery systems displayed great potential for ameliorating or overcoming the shortcomings of UA [37].
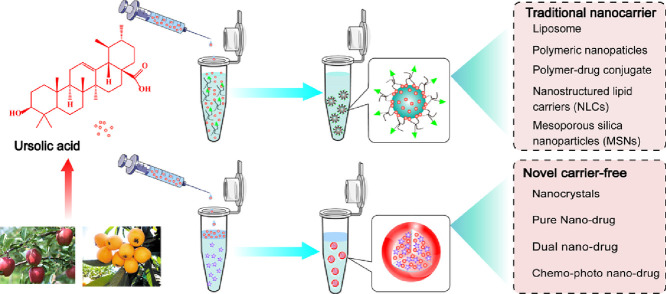
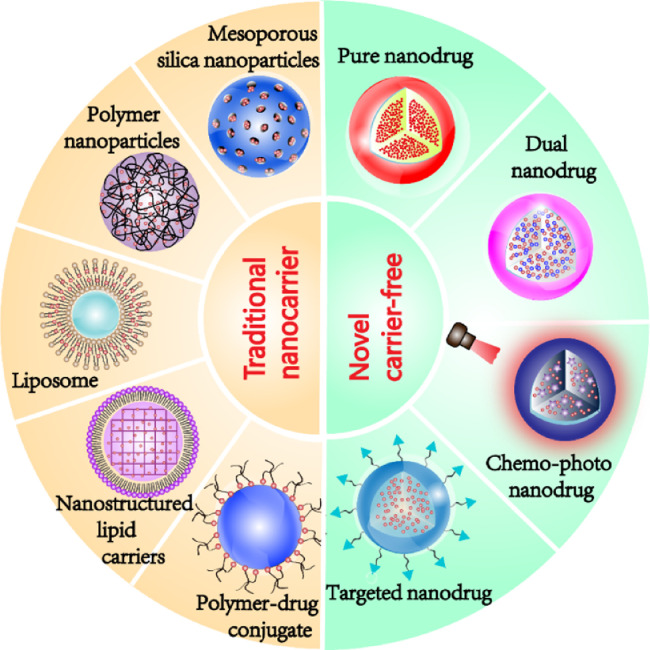
Therefore, this review summarizes the status of preclinical and clinical trials of UA nanosystems in cancer therapy, including traditional nanocarrier-based delivery systems and novel carrier-free delivery systems (Fig. 2). The various UA nanosystems with the properties of improved bioavailability and pharmacokinetic are exhaustively summarized in this review. Besides, this review also discusses the clinical trials of UA liposomes for the treatment of human cancers. The purpose of this review is to offer useful insight into the prospect and challenge of discovering ideal UA nanosystems for clinical cancer therapy.
Fig. 2.
Various types of UA-based nano-delivery systems for cancer therapy.
2. Anti-cancer activities of ursolic acid
Recently, anticancer property of UA has been extensively investigated in area of oncology, especially in breast cancer [38], hepatocellular carcinomas [39], cervical cancer [40], lung cancer [41], melanoma [42], gall bladder carcinomas [43] and prostate cancer [44]. Epidermal growth factor receptor (EGFR), an over-expressed protein in cancer cells, has been identified as a therapeutic target in cancer therapy [45]. Of note, mitogen-activated protein kinases (MAPK) is a downstream effector of EGFR. Studies demonstrated that UA could attenuate the phosphorylation of ERK 1/2, EGFR, p38 MAPK, and c-Jun N-terminal kinase (JNK) so as to induce cancer apoptosis and inhibit cancer cell proliferation and tumor angiogenesis [46]. The phosphoinositide 3-kinase (PI3K) / protein kinase B (AKT), Janus kinase 2 (JAK2)/signal transducer and activator of transcription 3 (STAT3) pathways are three crucial intrinsic pathways (or mitochondrial pathway) involved in apoptosis. Moreover, UA inhibits these cellular signaling pathways by modulating apoptosis-related genes (e.g., Bax, Bad, Bcl-2, Bcl-xL, BID), apoptosis-related protease (e.g., Caspase-3, Caspase-9, Caspase-8, PARP), protein kinases (e.g., JAK2, mTOR, PI3K, AKT), transcription factors STAT3 and nuclear factor (NF)-κB, thus suppressing the development of cancer cell and inducing cancer cell apoptosis [47,48]. Additionally, UA was also found to suppress the expression of pro-inflammatory cytokines, such as tumor necrosis factor (TNF)-α, TNF-γ, interleukin (IL)−2, IL-6, IL-18, which, in turn, influence the inflammatory response [38]. Additionally, some studies revealed that UA significantly down-regulated the expression levels of matrix metalloproteinases (MMP) (e.g., MMP-9, MMP-2) and cell surface adhesive molecules (e.g., ICAM-1, VCAM-1, E-selectin and P-selectin), thereby suppressing tumor invasion and metastasis. The primary mechanisms of action of UA are summarized in Fig. 3. The mechanistic elucidations and signaling pathways will be helpful in selecting proper therapeutic targets and designing ideal UA nanoformulations for cancer therapy.
Fig. 3.
The primary mechanism of anticancer activities of UA.
3. Traditional nanocarrier-based drug delivery systems of ursolic acid
In the past decades, nanotechnology has been extensively applied in pharmaceutical research and a variety of nanocarriers have been developed in order to facilitate drug delivery. The advantages of most nanocarriers are presented below. (i) Improving bioavailability of drugs by enhancing drug solubility, stabilization and loading capacity. (ii) Easily to be modified by surface functionalization due to the large surface-to-volume ratio in nanosized materials. (iii) Controlled or targeted release of the payloads in demand, and (iv) equipped with great biocompatibility and biodegradability. Considerable nanocarriers such as liposomes, chitosan, polymer, dendrimer, mesoporous silica and nanostructured lipid carriers have been developed for optimizing UA-based nanoformulation design (Table 1).
Table 1.
The preparation and characteristics of the traditional carrier of UA nanoparticles.
| Type | Materials or modified | Preparation | Particle size (nm) | zeta potential (mV) | Entrapment efficiency (%) | Drug Loading Capacities (%) | Ref. |
|---|---|---|---|---|---|---|---|
| Liposome | long-circulating and pH-sensitive liposome | The lipid hydration method | 191.1 ± 6.4 | +1.2 ± 1.4 | – | – | [49] |
| PEG-modified | Ethanol injection method | 130–200 | – | 99.56 | – | [50] | |
| Chitosan-modified | An ethanol injection method | 135.4 ± 0.636 | +7.8 | 94.30 | – | [51] | |
| Folate-modified | The thin-film dispersed hydration method | 160.1 ± 12.5 | −21.24 ± 4.2 | 88.9 ± 7.9 | – | [52] | |
| Polymeric NPs | mPEG-PLC | Nano-precipitation method | 144.0 ± 4.0 | −0.99 ± 0.3 | 87 ± 5.3 | 4.75 ± 0.45 | [29] |
| PVP-b-PCL | Nano-precipitation method | 120.0 ± 4.0 | −0.96 ± 0.3 | 82 ± 4.3 | 12 ± 0.45 | [53] | |
| Poly(lactic acid) | Single-emulsion solvent evaporation technique | 246 ± 10 | - 24.6 ± 3.1 | 95.8 ± 2.3 | – | [54] | |
| PLGA | Single-emulsion solvent evaporation technique | 154 ± 4.56 | - 18.4 | 40 ± 3.24 | 4 ± 1.12 | [55] | |
| Polyurethane | interfacial polycondensation technique combined with spontaneous emulsification | 9.77 | – | – | [56] | ||
| Polymer-drug conjugate | Chitosan | Nano-precipitation method | 120 | +41.6 | – | – | [57] |
| Folate-modified chitosan | Nano-precipitation method | 160 | +39.3 | – | – | [58] | |
| Folate-modified PAMAM (G3/G5) | One-pot synthetic approach | 185.6 (G3)276.8 (G5) | +8.1 (G3) +13.4 (G5) | – | – | [59] | |
| PAMAM-G0-LA | self-assembly method | 160.3 ± 10.2 | – | – | – | [60] | |
| 8armPEG and hydrooxycampothecin | self-assembly method | 91 ± 11.45 | 9.50 ± 0.11 | – | 9.22 ± 1.51 | [61] | |
| CMC | self-assembly method | 32.17 ± 2.25 | 6.73 ± 0.51 | – | 32.11 | [62] | |
| Nanostructured lipid carriers | Trierucin/hydrogenated soy phosphatidylcholine/oleic acid | Hot homogenization-ultrasonication method | 171 ± 3 | −20 ± 0.4 | 97.14 ± 1.8 | 4.67 ± 0.09 | [63] |
| phospholipid | solvent emulsification-evaporation and ultrasonic dispersion | 273.8 ± 2.3 | −23.2 ± 1.5 | 86.0 ± 0.4 | – | [64] | |
| solvent emulsification-evaporation and ultrasonic dispersion technology | 207.85 ± 12 | −42 ± 2.12 | – | – | [65] | ||
| Mesoporous silica NPs | MSNs | – | 102.2 ± 6.5 (pH = 10) | −34.6 ± 3.1 | – | – | [66] |
| MSN-CS-FA | – | ∼100 | −25 | 56.70 | 21.8 | [67] | |
| MSN-CS-LA | – | 197 ± 3.5 | +6.3 ± 1.4 | 55.5 ± 1.1 | 20.5 ± 1.1 | [68] |
3.1. Liposome
Liposome is the most well-studied nanocarrier which composed of phospholipids and cholesterol molecules [49,69]. In comparison with free drug, the liposomal drug possesses several attractive features such as controlled release, reduced toxicity, excellent stability, targeted delivery, increased solubility, higher cellular uptake, and multiple routes of administration [70]. De Araújo Lopes et al. developed UA-loaded liposomes which composed of dioleoylphosphatidylethanolamine (DOPE), cholesteryl hemisuccinate (CHEMS), and distearoylphosphatidylethanolamine-polyethylene glycol (DSPE-PEG) 2000. The obtained liposome with an average diameter of 191.1 nm, was benefit for drug accumulation into tumor tissues. Additionally, due to the strong interaction between UA and phospholipids in the liposome bilayer, the characteristics of liposomes remained unchanged for 60 d [49]. In another study performed by Zhao et al., PEG-modified UA liposomes released 53% of the loaded UA in 72 h. The inhibitory cell rates of PEG-UA NPs were found to be 68.27% in vitro, while the inhibition rate of free UA at the same concentration (250 µg/ml) was only 57.03%. Overall, surface-modifications of liposomes with PEG polymer are able to enhance stability of conventional liposomes [50].
Recently, chitosan-modified UA liposomes (CS-UA-L) has been successfully prepared with high tumor targeting, drug controlled release and low side effects [51]. In vitro drug release studies showed that 35.7% of the loaded UA was released within 72 h at pH 7.4, while 100% was released at pH 5.5, indicating a pH-responsive sustained drug release manner. Furthermore, it was also found that the UA accumulation amount in tumor tissues of CS-UA-L-administrated mice was 4.2-fold more than free UA administrated groups [51].
Ligand-targeted UA liposomes have also been designed to enhance therapeutic efficacy but minimize side effects as much as possible [71]. Yang et al. developed UA-loaded FA receptor-targeted liposomes (FTL-UA) with an IC50 against KB cell lines of 22.05 µΜ, whereas the IC50 of FA receptor-blocking group was 65.66 µM. It was shown that FTL-UA effectively targeted KB cells through the FA receptor-mediated pathway [52]. Overall, surface-functionalized liposomes not only facilitate the stability and bioavailability of UA, but also improve its targetability and therapeutic efficacy in vivo.
3.2. Polymeric nanoparticles
A variety of polymeric nanocarriers including chitosan [72], polybutylcyanoacrylate (PBCA), polylactic acid (PLA) and polylactic acid-glycolic acid copolymer (PLGA) [73] have been widely used for the drug delivery. Polymeric nanoparticles show many advantages surpass other nanocarriers such as the simplest preparation method and excellent stability. Zhang et al. [29] proposed that UA-loaded amphiphilic methoxy poly(ethylene glycol)–polycaprolactone (mPEG–PCL) nanoparticles presented a slightly better cell killing activity than free UA (IC50: 46.0 ± 2.8 vs. 92.5 ± 3.1 µΜ). The higher anticancer efficiency of mPEG-PCL nanoparticles were achieved due to the enhanced suppression of COX-2 and activation of Caspase 3. The conjugations of PEG or PEGylated nanoparticles were found to extend blood circulation half-life by reducing the recognition and uptake in the mononuclear phagocyte system (MPS). Subsequently, another UA-loaded amphiphilic molecule poly(N-vinylpyrrolidone)-block-poly(ε-caprolactone) (PVP-b-PCL) nanoparticles were developed and with an IC50 of 32.89 ± 3.23 µM (free UA, IC50 at 59.84 ± 4.12 µM) [53]. Zhang et al. demonstrated that the PVP-b-PCL nanoparticles exhibited longer blood circulation in comparison with mPEG-PCL nanoparticles, which might be ascribed to their evasion of reticuloendothelial system (RES) [53].
In another formulation, UA-loaded PLA nanoparticles have been developed by the emulsification-solvent evaporation technique [54]. 30% of the loaded UA was released from these nanoparticles in 8 h, characterized as a burst release, and 60% was released within 120 h. These PLA nanoparticles showed negligible hemolysis on erythrocytes and lower cytotoxicity against cancer cells than free UA [54]. In another study, a novel UA delivery system using PLGA as nanocarrier (PLGA-UA NPs) was prepared and evaluated with higher cytotoxicity against melanoma cell lines. The nanoparticles showed an initial burst release of 30% loaded UA but followed by a sustained drug release manner for 15 d. Simultaneously, the nanoparticles showed a biphasic clear manner, with 50% of the dosage rapidly cleared in 1 h and the rest was slowly cleared in the blood circulation [55]. Wang et al. reported that gold-UA-loaded PLGA nanoparticles exhibited great cellular uptake and effectively suppressed proliferation, invasion and migration of cervical cancer cells [74]. Oprean et al. developed UA-loaded polyurethane nanoparticles and assessed for their anti-proliferative against breast cancer cells [56]. It was found that these nanoparticles displayed remarkable antiproliferative activities in vitro. The improved drug loading efficiency could be contributed to the great affinity between the hydrophobic UA and polymeric molecules. Accumulating evidence have supported the prolonged blood circulation and enhanced retention concentration of UA at the tumor sites after incorporated into polymeric nanoparticles. In addition, most polymers are biodegradable and biocompatible molecules, indicating their great potential to exhibit minimal toxicity in vivo.
3.3. Polymer-drug conjugates
Growing interests have been focused on polymer-drug conjugates as they present numerous benefits when comparing with individual drug molecules: (i) Improving water solubility of hydrophobic drugs. (ii) Enabling sustained drug release by controlling the breaking of covalent bonds. (iii) Reducing toxicity side effects of drugs with narrow windows for treatment, thereby augmenting their clinical application prospects [75]. Jin et al. reported that the conjugation of UA and chitosan (CH-UA-NPs) could be achieved via covalently linking the carboxyl group of UA to the amino group of chitosan. The CH-UA-NPs significantly alleviated the adverse effects by saving approximately 10-fold of the dosage of free UA [57]. In another study, UA chitosan nanoparticles were further modified by folic acid (FA-UA-CS-NPs) in order to improve its targeted efficiency. Cellular uptake experiments showed that the FA-UA-CS-NPs could selectively target MCF-7 cancer cells through FA receptor-mediated endocytosis [58].
Dendrimers with highly-branched structures and biodegradable properties have been widely used in the preparation of nanoparticles. In a study, UA polymeric conjugations were designed and synthesized by conjugating of polyamidoamine dendrimers (G3/G5) with UA and FA (FA-G3/5-UA) [59]. The results marked that the FA-G3/5-UA significantly enhanced cellular uptake in Hela cells (FA receptor over-expressing cell line) due to the electrostatic absorptive and FA receptor-mediated endocytosis. The IC50 (µM) of FA-G5-UA and free UA was 20.24 and 44.35, respectively. Our group further modified the surface of low-PAMAM (G0/G1) dendrimers with lactobionic acid (LA) in order to develop a novel targeted nanocarrier for UA delivery (UA2-G0-LA NPs) [60]. The in vitro studies showed that the UA2-G0-LA NPs had much higher cytotoxicity against ASGPR-overexpressing cell lines than other groups.
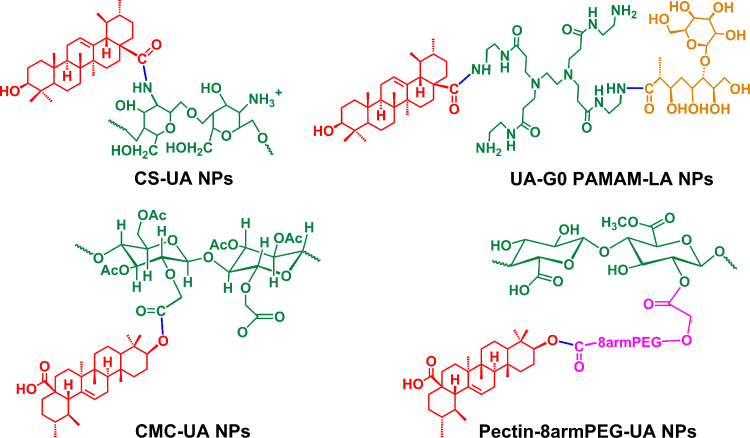
In a recent study, UA was covalently linked to eight-arm-PEG and pectin to form an amphiphilic pro-drug (8armPEG-UA). The 8armPEG-UA and another hydrophobic drug hydroxycamptothecin (HCPT) were self-assembled into nanoparticles (Pec-8PUH NPs) [61]. The hydrophobic drug UA served as the core while the hydrophilic pectin and 8armPEG served as the surface shell, which ensured the enhancement of UA stability and solubility in the neutral aqueous environment [61]. Furthermore, pharmacokinetic experiments revealed that the Pec-8PUH NPs could exhibit prolonged clearance and blood circulation half-life as compared to free drug. Liu et al. reported the nanoparticles (CMC–UA/HCPT NPs) which self-assembled from UA, carboxymethylcellulose (CMC) and HCPT [62]. The blood circulation half-life of CMC-UA/HCPT NPs was 7.3-fold higher than that of free UA [62]. Overall, conjugation of UA with hydrophilic polymers (Summarized in Fig. 4) could improve the solubility of UA and also impart some unique pharmacokinetic properties.
Fig. 4.
Covalent conjugates of UA to polymeric carriers [57, [60], [61], [62].
3.4. Solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs)
Lipid based nanocarriers are able to increase cell membrane permeability and then enhance the cellular uptake of drugs [76]. Based on the diverse internal structures, lipid nanoparticles are typically classified into solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLCs) [77]. The SLNs are only comprised of solid lipid, whereas the NLCs are made up of a mixture of blended solid and liquid lipid [78]. In a study, UA-loaded NLCs which composed of trierucin, hydrogenated soy phosphatidylcholine and oleic acid was prepared by the hot homogenization–ultrasonication method [63]. It was found that the UA NLCs exhibited higher cytotoxicity on K562 leukemic cells and B16 melanoma cells in comparison with free UA [63]. Zhou et al. designed UA loaded phospholipid nanoparticles (UA-PL-NP) by the solvent emulsification–evaporation and ultrasonic dispersion technology and further evaluated their targetability [64]. The AUC0-12 ratio of UA-PL-NP in the liver was 8.6-fold higher than free UA, indicating its great targetability to liver tissues. Then another group optimized the phospholipid nanoparticles by a novel method (Response Surface Methodology) to improve its bioavailability and pharmacokinetic properties. It was shown that the elimination half-life (T1/2el, 8.28 ± 1.98 h) of optimized nanoparticles was higher than free UA (0.69 ± 1.76 h) [65]. To date, UA-loaded solid lipid nanoparticles (UA SLNs) has not been reported in the literature. In summary, UA NLCs not only significantly enhanced bioavailability of UA but also showed superior anti-tumor activities in pre-clinical studies.
3.5. Mesoporous silica nanoparticles
Mesoporous silica nanoparticles (MSNs) is a promising drug delivery system due to its high surface area, narrow pore size distribution, superior chemical property, mechanical stability, and facile surface functionalization [79], [80], [81], [82], [83]. UA-loaded MSNs (UA@MSN-UA) with particle size of 100 nm displayed a higher cytotoxic effect against HepG2 cells than free UA [66]. The nanoparticles released 76% of the loaded UA in 96 h at pH 5.5, whereas 53% was released at pH 7.4, indicating its sustained and pH-responsive release properties. In another study, our group developed active targeting ligand FA and chitosan co-modified UA-loaded MSNs (UA@M-CS-FA) and evaluated the targeted delivery efficiency of UA to FA-receptor positive cancer cells [67]. Mechanistically, UA@M-CS-FA exhibited remarkable inhibition on the migration of cancer cells through regulating P53/MMP-9/PTEN/CD44 proteins. Also, in vivo experiments revealed that UA@M-CS-FA significantly inhibited the tumor proliferation and migration [67]. Similarly, another mesoporous silica nanocarrier (MSN-CS-LA) modified with pH-responsive chitosan and LA was developed for co-delivering UA and sorafenib. This nanoparticle could improve the cellular uptake and internalization of drugs against ASGPR over-expressing cell lines [68]. In conclusion, the development of multifunctional MSNs provides a new way for efficiently delivering UA into tumor tissues.
4. The novel carrier-free nano-drug delivery systems of ursolic acid
The applications of various nanocarriers provide some useful strategies for delivering UA into tumor tissues as well as enhancing its cellular uptake and internalization. Nevertheless, the clinical applications of nanocarriers have been limited due to their potential toxicity, indistinct metabolism and sophisticated preparation process [84,85]. Hence, an increasing attention has been paid to develop novel carrier-free nano-drugs (Table 2) in order to achieve better anti-cancer efficacy as well as reduce the side effects.
Table 2.
The preparation and characteristics of novel carrier-free based nano-drug delivery systems of UA.
| Preparation | Type | Materials or modified | Particle size (nm) | zeta potential (mV) | Entrapment efficiency (%) | Drug Loading Capacities (%) | Ref. |
|---|---|---|---|---|---|---|---|
| Anti-solvent precipitation method | Nanocrystals | UA | 188.0 ± 4.4 | −25.0 ± 5.9 | – | – | [86] |
| UA NPs | UA and TPGS1000 | 127 ± 4.8 | −24.4 | _ | – | [88] | |
| Solvent exchanging method. | UA NPs | UA | ∼158 | −7.71 | 60 | 60 | [87] |
| ICG@UA/PTX NPs | UA, PXT and IGG | 130.8 ± 0.20 | −30.0 ± 0.80 | 91.20 ± 1.2 | – | [89] | |
| UA-LA-ICG NPs | LA and IGG | 116.4 ± 2.4 | −30 ± 1.8 | 90.5 ± 0.7 | 62.9 ± 0.2 | [90] | |
| Apt / UD NPs | DOX and aptamer | ∼108.9 | – | – | – | [91] | |
| Self-assemble method | UA-LMWH NPs | LMWH-UA prodrug | 225.4 ± 4.3 | – | – | – | [92] |
| UA-s-LMWH NPs | pH-activatable sLMWH-UA prodrug | 223.1 ± 0.9 | – | – | – | [93] | |
| UA-Asp-NPs | UA-Asp-prodrug | 115.4 | −20.4 | – | 70.17 | [94] |
4.1. Pure nano-drug
Nanocrystals, one of the broadly used carrier-free delivery systems, was mainly composed of insoluble drugs and certain surfactants [95]. Many clinical trials revealed that nanocrystals could efficiently improve the bioavailability of drugs and ameliorate individual therapeutic differences [96]. In a study by Song et al., the UA nanocrystals were formulated by the anti-solvent precipitation method [86]. It was shown that UA nanocrystals had good aqueous dispensability and could be completely dissolved in 0.5% sodium dodecyl sulfate solution within 120 min. Moreover, the characteristics of nanocrystals remained unchanged for 49 d, which indicated its great stability. Pi et al. evaluated the relative bioavailability of UA nanocrystal and found it was 2.56-fold higher than that of free UA [97]. The nanocrystals may represent a promising delivery system for improving dissolution velocity and anticancer efficiency of UA.
In addition to nanocrystals, there are many different forms of pure nano-drugs. For instance, Wang et al. developed UA nanosuspensions by the anti-solvent precipitation method using a four-stream multi-inlet vortex mixer. In this study, two types of UA nanosuspensions with different particle sizes were evaluated for their anticancer activity [98]. It was shown that the UA nanosuspensions (300 nm) significantly inhibited the growth of the MCF-7 cells, which may be due to the enhanced adhesion of nanoparticles to cell surface. Inspired by the electrostatic and hydrophobic interactions between UA and the nanocarriers, our group has recently developed novel UA nanoparticles by a solvent exchanging method [87]. The UA NPs with particle size of approximately 150 nm was found to effectively permeate into tumor tissues by the EPR effect. The cell viability test showed that the IC50 of UA NPs and free UA were 48.12 and 39.74 µM, respectively [87]. Also, the UA NPs showed less toxicity to normal tissues, indicating its great potential in future clinical applications. In a similar study, in order to improve the bioavailability of UA, a novel UA nanoemulsion which stabilized with TPGS1000 was developed by Qiao et al. [88]. AUC0→12 and Cmax of UA nanoemulsion were found to be approximately 27.5- and 9-fold higher than free UA, respectively. In another study, TPGS1000 was also served as drug stabilizer to prepare UA nanoemulsion by emulsion solvent evaporation method. This nanoemulsion improved the equilibrium solubility of UA in deionized water up to 23.99-fold, and increased the oral bioavailability of UA up to 2.68-fold [99]. Even though the preliminary studies showed the stability of carrier-free nano-drugs in the absence of stabilizers is not as good as that of traditional carrier-based nano-drugs, they still contribute a lot to the development of nano-drugs.
4.2. Carrier-free nano-drug by co-assembly of chemotherapeutic agent and photosensitizer
Phototherapy, which contains photothermal therapy and photodynamic therapy, is characterized by high tumor ablation efficiency. It has been well-reported that co-delivery of chemotherapeutic agents and photosensitizers could enhance the therapeutic effect of cancer. A novel carrier-free nano-drug delivery system (ICG@UA/PTX NPs) for co-delivery of a photosensitizer (Indocyanine green, IGG) and chemotherapy drugs (UA and PTX) was developed by a solvent exchanging method [89]. The carrier-free small molecule nano-drug via self-assembly demonstrated excellent ability of tumor targeting and tumor growth suppressing with no sign of recurrence under NIR laser irradiation [89]. In a recently alternative study, we developed a nano-drug (UA-LA-ICG NPs) by co-assembly of UA, active targeting ligand LA and ICG. The UA-LA-ICG NPs exhibited anti-proliferative activity on asialoglycoprotein receptor (ASGPR)-overexpressing HepG2 cells. It was also shown that UA-LA-ICG NPs + NIR irradiation treatment significantly inhibited the development of tumors in H22 tumor-bearing mice [90].
4.3. Carrier-free nanosize drug by co-assembly of chemotherapeutic agents
Many researchers found that the carrier-free delivery systems based on combination of UA with other chemotherapeutic agents were able to display synergistic anticancer effects. A composite carrier-free system has been developed by the self-assembly of UA, aptamer and doxorubicin (Apt/UD NPs) [91]. In vitro/vivo studies, the Apt/UD NPs showed superior inhibitory and synergistic anticancer effects. Our group developed the prodrug of UA and aspirin (Asp) into a nano-drug via the solvent exchange method [94, 100]. Of note, this nano-drug could release over 80% of UA after 48 h at pH 5.5, while only release of UA about 40% at pH 7.4 [94]. Similarly, the UA-Met-prodrug synthesized by our group exhibited a dose-dependent anti-proliferation effect on various cancer cells and displayed synergistic inhibition of cancer cell metastasis and invasion [101].
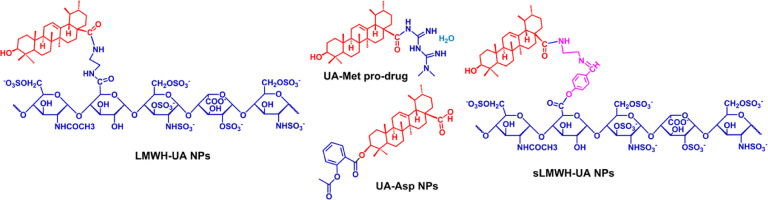
Cheng et al. developed an amphiphilic molecule (LMWH-UA), in order to achieve chemo- and anti-angiogenic combined therapy. The degradation rate of LMWH–UA in plasma and tumor homogenate was 6.64% ± 0.87% and 39.35% ± 5.47%, respectively, which confirmed that the micelles existed excellent stability in plasma and controlled release manner at tumor tissues [92]. Based on the above experiments, Xiong et al. constructed a novel prodrug of UA (sLMWH-UA) by linking with LMWH and UA using the Schiff base (−CH N−) bond. The diameter size of sLMWH-UA was 223.1 ± 0.9 nm, and the critical aggregation concentration was 38.30 µg/ml, indicating its excellent stability in the systemic circulation. The addition of Schiff base bond promoted the micelles to hydrolyze and then release drugs under acidic conditions [93]. Overall, these studies demonstrated that conjugating UA with other chemotherapeutic agents (summarized in Fig. 5) may be a promising strategy for cancer therapy.
Fig. 5.
Structure of conjugates of UA with clinical drugs by the covalent link [92], [93], [94],101].
5. Nanosystem studies with ursolic acid in clinical trials
Clinical trials are essential for validating the efficacy and safety concerns of formulations. Extensive studies have confirmed the significant anticancer effect of UA nanosystems in pre-clinical studies, but the clinical studies of UA nanosystems are comparatively limited. In 2011, the UA liposomes (UA NLs) were firstly evaluated in human pharmacokinetic study by Tianjin Medicinal University Cancer Institute. The clinical data reported that UA NLs achieved a mean Cmax of 3404.6 ± 748.8 ng/ml, and AUC0−∞ value of 9918.4 ± 1215.2 ng·h/ml [102]. Wang et al. evaluated the toxicity and single-dose pharmacokinetics of UA NLs. It was shown that the maximum tolerated dose (MTD) of UA NLs was 98 mg/m2 and the dose-limiting toxicity was hepatotoxicity and diarrhea [103]. In another pharmacokinetic study, Zhu et al. evaluated the single-dose and multiple-dose pharmacokinetic study of UA NLs, in which twenty-four healthy volunteers received a single-dose of UA NLs (37, 74, 98 mg/m2) while eight cancer patients received a multiple-dose of UA NLs (every day 74 mg/m2 of UA NLs for 14 d). The study revealed that the UA NLs exhibited a relatively linear pharmacokinetic behavior at dose levels between 37 and 98 mg/m2 [104]. In a recent clinical study of UA NLs, twenty-one patients with solid tumors were intravenously administered with doses of 56, 74 and 98 mg/m2 UA for 14 d consecutivly over a 21 d treatment cycle. Indeed, 60% of subjects achieved satisfactory therapeutic effect after two treatment cycles [105]. The results of these clinical phases I trials all confirmed that UA NLs could show high tolerance and low toxicity in healthy volunteers. Nevertheless, more extensive studies and investigations should be carried out in patients with different solid tumors.
6. Advantage and challenge of ursolic acid nanosystem
Up to now, UA nanosystems have made significant contribution to cancer therapy. We summarized the in vivo antitumor studies of UA nanoformulations to demonstrate that the applications of nanotechnology are able to improve antitumor efficacy of UA (Table 3). Additionally, we presented the unique advantages and challenges of UA nanosystems for cancer therapy below.
Table 3.
In vivo application of UA nanosystems for cancer treatment.
| Formulation | Animal | Mouse xenograft model | The dose of UA NPs | Route of administration | Observation | Ref. |
|---|---|---|---|---|---|---|
| FA-UA-L | Female Balb/c mouse | KB cells (3.6 × 106 cells/animal), SC | 4.5 mg/kg, once/2 d for 10 d | IV | Tumor-bearing mice treated with FTL-UA had about 55% reduction in tumor volume compared with PBS control group. | [52] |
| CS-UA-L | Female CD-1 mouse | LU14 cells (2 × 106 cells/animal), SC | 80 mg/kg, once a d for 14 d | IV | Free UA showed the modest tumor growth inhibition with an inhibition rate (IR) of 18.25%, and CS-UA-L was up to 61.26%; | [51] |
| UA loaded PVP-b-PCL | Male ICR mice | H22 cells (1 × 107 cells/animal), SC | 50 mg/kg for 10 d | IP | After UA treatment, most tumor lesions were relieved, while that of UA-NPs treated group almost completely disappeared. | [53] |
| gold-UA – PLGA NPs | Athymic nude mice | HeLa (1 × 107 cells/animal), SC | 20 mg/kg for 5 d | IP | Significantly downregulated the tumor volume; No remarkable alteration of the mouse body weight. | [74] |
| CH-UA-NPs | Female Balb/c mice | H22 cells (5 × 106 cells/0.2 ml ascites/mouse), IP | 11 mg/kg once/2 d for 17 d | O | The volume of tumors of nanoparticle-treated group (1.12 ± 0.12 cm3) was dramatically decreased compared with that of the control group (2.36 ± 0.32 cm3, P < 0.01). | [57] |
| FA-CS-UA-NPs | BALB/c nude mice | MCF-5 (2 × 105 cells / animal), SC | 12.5 mg/kg b.w./d for 9 times | IP | The tumor weight in FA-CS-UA-NPs-treated group (2.1 ± 1.02 g) was significantly lower than those of UA and saline control groups (5.26 ± 1.69 g, P < 0.05), and UA-treated group (3.48 ± 0.24 g, P < 0.005). | [58] |
| UA2-G0-LA NPs | Female Balb/c mice | H22 cells (1 × 107cells/animal), SC | 40 mg/kg | IV | The tumor weight and volume of UA-G0-LA NPs group were significantly decreased. | [60] |
| Pec-8PUH NPs | Female Balb/c mice | 4T1 cells (5 × 105 cells/0.2 ml/mouse), SC | 10 mg/kg once/2 d for 8 d | IV | The tumor volumes of Pec-8PUH NPs treated group were extremely smaller than other groups. | [61] |
| CMC–UA/HCPT NPs | Female Balb/c mice | 4T1 cells (5 × 105 cells/0.2 ml/mouse), SC | 10 mg/kg once/2 d for 8 d | IV | Free UA showed the modest tumor growth inhibition with an inhibition rate of 42.9%, and CMC-UA /HCPT NPs was up to 93.5%. | [62] |
| pure UA NPs | Balb/c nude mice | A549 cell (5 × 106 cells/animal), SC | 8 mg/kg once/2 d for 21 d | IV | Efficiently suppressed the A549 tumor growth in vivo without causing any obvious body weight effects in nude mice. | [87] |
| ICG@UA/PT X NPs | KM mice | H22 cells (2 × 106 cells/animal), SC | 2.67 mg/kg of UA, 2.0 mg/kg of ICG once/3 d for 21 d | IV | The tumor volumes of NPs+NIR laser irradiation-treated groups were much smaller than the other group-treated in mice. | [89] |
| LA-ICG-UA NPs | KM mice | H22 cells (1 × 107 cells/animal), SC | 10 mg/kg of UA, 2.5 mg/kg of ICG, once/2 d for 14 d | IV | The combined chemo-photo therapy with UA-LA-ICG NPs + NIR significantly inhibited the tumor growth than chemotherapy or phototherapy alone. | [90] |
| IV: Intravenous; IP: Intraperitoneal; O: Oral; SC: Subcutaneous | ||||||
6.1. Enhancing the bioavailability of UA
Bioavailability is an essential index of pharmacokinetic property that mainly depending on drug solubility, stability, metabolism, and degradation [106]. It has been widely accepted that the poor water solubility greatly limited the bioavailability of chemotherapeutic agents. Using TPGS1000 acted as a drug stabilizer could effectively improve the solubility of drugs and subsequently enhance their oral bioavailability [88]. For nano-delivery system, the rigidity of nanoparticle displays a significant influence on its stability, drug release and accumulation time. Specifically, modifying nanoparticle with water-soluble polyethylene glycol (PEG) can significantly ameliorate its rigidity [45]. In addition, the physicochemical properties of the nanoparticles including morphology, size, surface charge and surface hydrophilicity all have great effects on their circulating half-life and metabolism [107, 108]. A short description of the nano-delivery system of UA that significantly improved its pharmacokinetics and bioavailability is provided in Table 4.
Table 4.
Pharmacokinetics properties and bioavailability of various UA nanosystems.
| UA nanoformulation | Commen | Ref. |
|---|---|---|
| CS-UA-L | The tumor concentration of CS-UA-L was 4.2- and 1.7-fold higher than those of free UA and UA-L groups, respectively. | [51] |
| FTL-UA | A single intravenous dose of 20 mg/kg FTL-UA in mice achieved Cmax of 109.03 ± 8.30 mg/l and T1/2 of drug 7.61 ± 2.41 h, whereas free UA showed low bioavailability (Cmax = 43.82 ± 4.49 mg/l, T1/2 = 0.78 h). AUC value of FTL-UA (218.32 mg/l•h) was 6-fold higher than that of free UA (36.88 mg/l•h). | [52] |
| UA2-G0-LA NPs | AUC0-t of UA2-G0-LA NPs (187.938 ± 25.426 µg/ml•min) was higher than UA and UA2-G0 NPs (98.9 ± 27.929 and 125.001 ± 19.364 µg/ml•min, respectively); Cmax: 1.511 ± 0.336, 2.29 ± 0.394 and 2.896 ± 0.616 µg/ml for UA, UA-G0 and UA2-G0-LA NPs, respectively; Tmax: 8, 18.75 ± 7.5 and 18.75 ± 7.5 min for UA, UA-G0 and UA2-G0-LA NPs, respectively. | [60] |
| CMC-UA/HCPT NPs | CMC-UA NPs and CMC–UA/HCPT NPs could extend the half-life of UA from 1 h to 4.5 h and 7.3 h, respectively. | [62] |
| UA-PL-NP | AUC0-12 ratio of UA-PL-NP 8.6-fold and 1.7-fold higher than free UA in the liver and spleen, respectively. | [64] |
| UA nanosuspensions | AUC0-12 of UA nanosuspensions (35–37 µg/ml•h) was higher than UA (1.4 µg/ml•h) and the Cmax was found to be approximately 9-fold higher than free UA. | [88] |
6.2. Targeted drug delivery
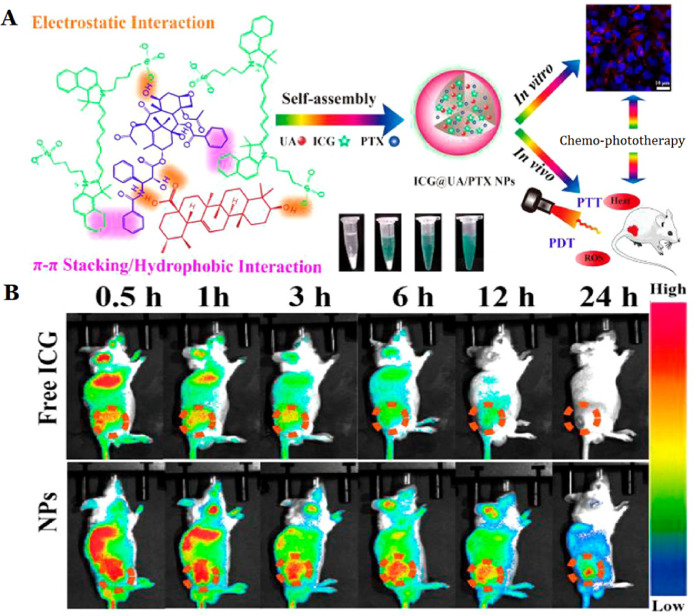
Targeted drug delivery systems are able to selectively deliver drug into the specific sites, thereby enhancing its therapeutic efficacy and reducing toxic effect [109,110]. Targeted drug delivery system can be divided into active and passive targeting systems. The nanoparticles with a size of 10–200 nm are able to effectively permeate into tumor tissues with the help of passive targeting [111]. Our group evaluated the passive tumor-targetability of nanoparticles by NIR fluorescence imaging technology. After 24 h, 30% fluorescence intensity of ICG in nanoparticles at the tumor sites was observed, while there was no fluorescence intensity tumor accumulation of free ICG group (Fig. 6) [89]. However, the size, zeta potential, solubility or dispersion of nanoparticles may also exhibit an impact on its targetability [112]. Additionally, the main factor of passive tumor-targetability of nanoparticles is the heterogeneity of tumor and its stroma, such as a hypoxic gradient [113]. Meanwhile, the increased interstitial fluid pressure is found to hamper the penetration of nanoparticles into neoplastic tissues [114]. Most of the active targeting systems were prepared by surface modification with targeting-ligands (such as protein, sugar, amino acids, enzymes). Modified by multifarious ligands, enhancement of cellular uptake and interaction with tumor surface receptors of nanoparticles have been documented [108]. To obtain effective targeting, the nanoparticles should be stable and also can avoid rapid recognition by RES. Accordingly, it is crucial to optimize the density of targeting-ligands on the surface of nanosystems. In conclusion, the nanosystems with effective passive or active tumor targetability have achieved substantial advantage over the free molecules [115].
Fig. 6.
In vivo targeting efficiency of ICG@UA/PTX NPs compared with that of free ICG. (A) Schematic of ICG-UA/PTX NPs for cancer imaging and chemo-phototherapy. (B) NIR fluorescence images of free ICG and NPs injected into tumor-bearing mice were recorded at 0.5, 1, 3, 6, 12 and 24 h, indicating that the NPs can efficaciously target tumor site. Reproduce with permission [89]. Copyright 2017, American Chemical Society.
6.3. Controlled drug release
Controlled-release drug delivery systems can release drugs with a certain speed at a predetermined time [116,117]. Delivery systems with different drug release manners such as extended-release, sustained-release, delayed-release, and targeted-release, are collectively referred to as controlled-release delivery systems [118]. The plasma drug concentration of nanoparticles will be very low initially and then progressively increased, which led to a broad and flat AUC [119]. By contrast, free drugs will commonly achieve their plasma Cmax during the intravenous infusion period and followed by a rapid drug concentration decrease, and therefore their AUC are likely to be a peak shape with a tail [119]. Accordingly, nanoparticles are of more potential to maintain drug concentration within the therapeutic window than free drugs [120]. To precisely control the release of drug, stimuli-responsive nanomaterials have been developed and utilized. They can release drugs in response to specific triggers, including exogenous stimuli (variations in temperature, magnetic field, ultrasound intensity, light or electric pulses) and endogenous stimuli (variations in enzyme concentration or redox gradients) [121,122]. Nevertheless, these newer delivery systems are still developed at the proof-of-concept level. Taken together, the development of new nanoparticles-based strategy is critical for enhancing drug delivery into tumors.
6.4. Challenge and prospect of UA nanosystem
With significant advances achieved in nanotechnologies, constructing drug-loaded nanosystems is considered to be one of the most promising strategies for cancer therapy. The advantages and disadvantages of different UA nanosystems are sequentially discussed in Table 5. Despite numerous progress has been made in recent years, there are still some challenges in the clinical applications of UA nanosystems. (i) Some nanocarriers of UA possibly aggregate or agglomerate with other biomolecules or biological fluids in vivo [123]. (ii) The stability and storage aspects of UA nanosystems are still needed to be further improved. (iii) Complex preparation methods of UA nanosystems will lead to some problems such as potentially toxic to healthy tissues, indistinct metabolism, as well as biodegradation problems. (iv) Many physiological barriers will certainly limit the deep penetration of UA nanosystems into the tumor tissues and subsequently impact the therapeutic outcome. These challenges must be addressed in order to promote UA nanosystems to become the next-generation nanomedicine.
Table 5.
Summary of the differences, advantages, and disadvantages of different type nanosystems of UA in cancer therapy.
| Different source of nanopariticales | Common features | Unique advantage | Disadvantage | Ref. |
|---|---|---|---|---|
| Liposomes | Improved low solubility, rapid metabolism, poor absorption and limited bioavailability | Biocompatible, biodegradable; Have sustained release and nonimmunogenic properties; Amphipathic. | Short circulation half-life; The leakage of the encapsulated drug. | [49,125] |
| PEG-modified liposome | Improve the blood circulation half-life of liposome and enhance stability. | Reduced interactions with cancer cells. | [50,126] | |
| Polymer nanoparticles | Longer blood circulation half-life; Reliable release profiles | Poor storage properties | [57,127] | |
| Polymer-drug conjugate | Ease of drug administration, improved patient compliance and better long-term prognosis | Unclear release manner. | [93,128] | |
| MSN | Sustained-release property; High drug-loading capacity | An impact on biosafety. | [66,124] | |
| Pure UA nano-drug | High drug-loading capacity; Reduce the potential hazards of the carrier. | Poor stability over traditional carrier nanoparticles. | [87,129] | |
| Dual nano-drugs | Synergistically treat tumor; reduce the doses of main therapeutic drugs; | Poor stability over traditional carrier nanoparticles. | [91,129] | |
| Targeted nanoparticles | Targeting tumor tissue to reduce adverse effects. | Increased immune activation. | [91,126] |
The following several aspects should be considered in promoting UA nanomedicines from preclinical levels to the clinical settings: (i) The implementation of US Food and Drug Administration (FDA)-approved materials as nanocarriers of UA can consequently expedite its clinical transformation. (ii) To address the issues of short blood circulation half-life and nonspecific protein adsorption, the surface of UA nanoparticles can be modified by various materials, such as PEG or polysorbate 80, dysopsonins proteins (e.g., clusterin and albumin), and self-markers (e.g., CD47 peptides) and membrane of erythrocytes, leukocytes, cancer cells or thrombocytes [36,119]. (iii) Additionally, the physicochemical characterization of UA nanoparticles should be carried out under similar clinical conditions. (iv) Moreover, the rational design of nanosystems is important to develop UA nanosystems with high loading capacity, which will further affect its dosage and therapeutic efficacy in clinical treatment [124]. Therefore, the development of nanosystems with active targeting-ligands or stimuli-responsive properties is valuable for improving the targetability and therapeutic efficacy of conventional UA nanosystems. More comprehensive preclinical and clinical trials should be conducted to further validate its therapeutic efficacy. It is also noteworthy that the combination of UA with other therapeutic agents could result in synergistic effects and offer a better therapeutic index. On the other hand, the novel composite nanosystems with two or more anticancer agents will help to reduce adverse effects by reducing the single dosage of chemotherapeutic agent. In addition to these considerations, manufacturing UA nanosystems at an industrial level should consider the cost-benefit and follow GLP and GMP (good laboratory and manufacturing practice).
7. Conclusion
In summary, this review mainly describes the recent advance of UA nanosystems for cancer treatment, with an emphasis on different nanocarrier delivery systems and carrier-free delivery systems. Large amounts of in vitro and in vivo experiments have provided evidence that these UA nanosystems displayed improved efficacy, targetability and reduced systemic toxicity. At present, most of the UA nanosystems have been developed and optimized at the laboratory scale and comprehensive preclinical tests to confirm their potential effect and safety. The phase I clinical trials confirm that UA liposomes are systemically safe, and can effectively improve the bioavailability of UA. Additionally, the potential toxicity of some UA nanosystems in humans remain unclear, hence future studies should focus on complementary toxicity assays of UA nanosystems. It is worth noting that the combination of UA nanosystems with phototherapy or other chemotherapeutic agents resulted in a promising therapeutic effect in cancer therapy. Therefore, a composite system of UA with other anticancer agents can be a promising approach to enhance anticancer efficacy and reduce systemic toxicity.
Conflicts of Interest
The authors declare no conflict of interest.
Acknowledgments
This project was supported by the National Natural Science Foundation of China (81972832, 81472767), Scientific Research Training Program for Undergraduate of Fuzhou University (25055, 25068).
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.ajps.2020.03.001.
Appendix. Supplementary materials
References
- 1.Roy P.S., Saikia B.J. Cancer and cure: a critical analysis. Indian J Cancer. 2016;53(3):441–442. doi: 10.4103/0019-509X.200658. [DOI] [PubMed] [Google Scholar]
- 2.Bray F., Ferlay J., Soerjomataram I., Siegel R.L., Torre L.A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 3.Miller K.D., Siegel R.L., Lin C.C., Mariotto A.B., Kramer J.L., Rowland J.H., et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016;66(4):271–289. doi: 10.3322/caac.21349. [DOI] [PubMed] [Google Scholar]
- 4.Miller K.D., Nogueira L., Mariotto A.B., Rowland J.H., Yabroff K.R., Alfano C.M., et al. Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin. 2019;69(5):363–385. doi: 10.3322/caac.21565. [DOI] [PubMed] [Google Scholar]
- 5.Reyes-Habito C.M., Roh E.K. Cutaneous reactions to chemotherapeutic drugs and targeted therapies for cancer: part I. Conventional chemotherapeutic drugs. J Am Acad Dermatol. 2014;71(2) doi: 10.1016/j.jaad.2014.04.014. 203 e1-12. [DOI] [PubMed] [Google Scholar]
- 6.Lee Y.T., Tan Y.J., Oon C.E. Molecular targeted therapy: treating cancer with specificity. Eur J Pharmacol. 2018;834:188–196. doi: 10.1016/j.ejphar.2018.07.034. [DOI] [PubMed] [Google Scholar]
- 7.Emens L.A. Breast cancer immunotherapy: facts and hopes. Clin Cancer Res. 2018;24(3):511–520. doi: 10.1158/1078-0432.CCR-16-3001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fairchild A., Tirumani S.H., Rosenthal M.H., Howard S.A., Krajewski K.M., Nishino M., et al. Hormonal therapy in oncology: a primer for the radiologist. AJR Am J Roentgenol. 2015;204(6):W620–W630. doi: 10.2214/AJR.14.13604. [DOI] [PubMed] [Google Scholar]
- 9.Li Y.J., Lei Y.H., Yao N., Wang C.R., Hu N., Ye W.C., et al. Autophagy and multidrug resistance in cancer. Chin J Cancer. 2017;36(1):52. doi: 10.1186/s40880-017-0219-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hu L.Y., Mi W.L., Wu G.C., Wang Y.Q., Mao-Ying Q.L. Prevention and treatment for chemotherapy-induced peripheral neuropathy: therapies based on CIPN mechanisms. Curr Neuropharmacol. 2019;17(2):184–196. doi: 10.2174/1570159X15666170915143217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zong S., Wang X., Yang Y., Wu W., Li H., Ma Y., et al. The use of cisplatin-loaded mucoadhesive nanofibers for local chemotherapy of cervical cancers in mice. Eur J Pharm Biopharm. 2015;93:127–135. doi: 10.1016/j.ejpb.2015.03.029. [DOI] [PubMed] [Google Scholar]
- 12.Markov A.V., Zenkova M.A., Logashenko E.B. Modulation of tumour-related signaling pathways by natural pentacyclic triterpenoids and their semisynthetic derivatives. Curr Med Chem. 2017;24(13):1277–1320. doi: 10.2174/0929867324666170112115313. [DOI] [PubMed] [Google Scholar]
- 13.Zhang J., Xu H.Y., Wu Y.J., Zhang X., Zhang L.Q., Li Y.M. Neutrophil elastase inhibitory effects of pentacyclic triterpenoids from eriobotrya japonica (loquat leaves) J Ethnopharmacol. 2019;242:111713. doi: 10.1016/j.jep.2019.01.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Alqahtani A., Hamid K., Kam A., Wong K.H., Abdelhak Z., Razmovski-Naumovski V., et al. The pentacyclic triterpenoids in herbal medicines and their pharmacological activities in diabetes and diabetic complications. Curr Med Chem. 2013;20(7):908–931. [PubMed] [Google Scholar]
- 15.Janicsák G., Veres K., Kakasy A.Z., Máthé I. Study of the oleanolic and ursolic acid contents of some species of the lamiaceae. Biochem System Ecol. 2006;34(5):392–396. [Google Scholar]
- 16.Silva M.G., Vieira I.G., Mendes F.N., Albuquerque I.L., dos Santos R.N., Silva F.O., et al. Variation of ursolic acid content in eight Ocimum species from northeastern Brazil. Molecules. 2008;13(10):2482–2487. doi: 10.3390/molecules13102482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kashyap D., Tuli H.S., Sharma A.K. Ursolic acid (UA): a metabolite with promising therapeutic potential. Life Sci. 2016;146:201–213. doi: 10.1016/j.lfs.2016.01.017. [DOI] [PubMed] [Google Scholar]
- 18.Jang S.M., Yee S.T., Choi J., Choi M.S., Do G.M., Jeon S.M., et al. Ursolic acid enhances the cellular immune system and pancreatic beta-cell function in streptozotocin-induced diabetic mice fed a high-fat diet. Int Immunopharmacol. 2009;9(1):113–119. doi: 10.1016/j.intimp.2008.10.013. [DOI] [PubMed] [Google Scholar]
- 19.Kim M.H., Kim J.N., Han S.N., Kim H.K. Ursolic acid isolated from guava leaves inhibits inflammatory mediators and reactive oxygen species in LPS-stimulated macrophages. Immunopharmacol Immunotoxicol. 2015;37(3):228–235. doi: 10.3109/08923973.2015.1021355. [DOI] [PubMed] [Google Scholar]
- 20.Wojciak-Kosior M., Paduch R., Matysik-Wozniak A., Niedziela P., Donica H. The effect of ursolic and oleanolic acids on human skin fibroblast cells. Folia Histochem Cytobiol. 2011;49(4):664–669. doi: 10.5603/fhc.2011.0050. [DOI] [PubMed] [Google Scholar]
- 21.Broniatowski M., Flasinski M., Hac-Wydro K. Antagonistic effects of alpha-tocopherol and ursolic acid on model bacterial membranes. Biochim Biophys Acta. 2015;1848(10 Pt A):2154–2162. doi: 10.1016/j.bbamem.2015.05.009. [DOI] [PubMed] [Google Scholar]
- 22.Yuliang W., Zejian W., Hanlin S., Ming Y., Kexuan T. The hypolipidemic effect of artesunate and ursolic acid in rats. Pak J Pharm Sci. 2015;28(3):871–874. [PubMed] [Google Scholar]
- 23.Shao J.W., Dai Y.C., Xue J.P., Wang J.C., Lin F.P., Guo Y.H. In vitro and in vivo anticancer activity evaluation of ursolic acid derivatives. Eur J Med Chem. 2011;46(7):2652–2661. doi: 10.1016/j.ejmech.2011.03.050. [DOI] [PubMed] [Google Scholar]
- 24.Yin R., Li T., Tian J.X., Xi P., Liu R.H. Ursolic acid, a potential anticancer compound for breast cancer therapy. Crit Rev Food Sci Nutr. 2018;58(4):568–574. doi: 10.1080/10408398.2016.1203755. [DOI] [PubMed] [Google Scholar]
- 25.Wang J., Jiang Z., Xiang L., Li Y., Ou M., Yang X., et al. Synergism of ursolic acid derivative US597 with 2-deoxy-D-glucose to preferentially induce tumor cell death by dual-targeting of apoptosis and glycolysis. Sci Rep. 2014;4:5006. doi: 10.1038/srep05006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yang X., Li Y., Jiang W., Ou M., Chen Y., Xu Y., et al. Synthesis and biological evaluation of novel ursolic acid derivatives as potential anticancer prodrugs. Chem Biol Drug Des. 2015;86(6):1397–1404. doi: 10.1111/cbdd.12608. [DOI] [PubMed] [Google Scholar]
- 27.Chen Q., Luo S., Zhang Y., Chen Z. Development of a liquid chromatography-mass spectrometry method for the determination of ursolic acid in rat plasma and tissue: application to the pharmacokinetic and tissue distribution study. Anal Bioanal Chem. 2011;399(8):2877–2884. doi: 10.1007/s00216-011-4651-x. [DOI] [PubMed] [Google Scholar]
- 28.Liao Q., Yang W., Jia Y., Chen X., Gao Q., Bi K. LC-MS determination and pharmacokinetic studies of ursolic acid in rat plasma after administration of the traditional chinese medicinal preparation Lu-Ying extract. Yakugaku Zasshi. 2005;125(6):509–515. doi: 10.1248/yakushi.125.509. [DOI] [PubMed] [Google Scholar]
- 29.Zhang H., Li X., Ding J., Xu H., Dai X., Hou Z., et al. Delivery of ursolic acid (UA) in polymeric nanoparticles effectively promotes the apoptosis of gastric cancer cells through enhanced inhibition of cyclooxygenase 2 (COX-2) Int J Pharm. 2013;441(1–2):261–268. doi: 10.1016/j.ijpharm.2012.11.034. [DOI] [PubMed] [Google Scholar]
- 30.Yu M.K., Park J., Jon S. Targeting strategies for multifunctional nanoparticles in cancer imaging and therapy. Theranostics. 2012;2(1):3–44. doi: 10.7150/thno.3463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rajeshkumar S., Bharath L.V. Mechanism of plant-mediated synthesis of silver nanoparticles - A review on biomolecules involved, characterisation and antibacterial activity. Chem Biol Interact. 2017;273:219–227. doi: 10.1016/j.cbi.2017.06.019. [DOI] [PubMed] [Google Scholar]
- 32.Kuai Q., Wang Y., Gao F., Qi Y., Wang R., Wang Y., et al. Peptide self-assembly nanoparticles loaded with panobinostat to activate latent human immunodeficiency virus. J Biomed Nanotechnol. 2019;15(5):979–992. doi: 10.1166/jbn.2019.2764. [DOI] [PubMed] [Google Scholar]
- 33.Jadhav N.R., Nadaf S.J., Lohar D.A., Ghagare P.S., Powar T.A. Phytochemicals formulated as nanoparticles: inventions, recent patents and future prospects. Recent Pat Drug Deliv Formul. 2017;11(3):173–186. doi: 10.2174/1872211311666171120102531. [DOI] [PubMed] [Google Scholar]
- 34.Tiwari S.B., Amiji M.M. A review of nanocarrier-based CNS delivery systems. Curr Drug Deliv. 2006;3(2):219–232. doi: 10.2174/156720106776359230. [DOI] [PubMed] [Google Scholar]
- 35.Chari R.V. Targeted delivery of chemotherapeutics: tumor-activated prodrug therapy. Adv Drug Deliv Rev. 1998;31(1–2):89–104. doi: 10.1016/s0169-409x(97)00095-1. [DOI] [PubMed] [Google Scholar]
- 36.Norouzi M., Amerian M., Amerian M., Atyabi F. Clinical applications of nanomedicine in cancer therapy. Drug Discov Today. 2020;25(1):107–125. doi: 10.1016/j.drudis.2019.09.017. [DOI] [PubMed] [Google Scholar]
- 37.Valdes K., Morales J., Rodriguez L., Gunther G. Potential use of nanocarriers with pentacyclic triterpenes in cancer treatments. Nanomedicine Lond. 2016;11(23):3139–3156. doi: 10.2217/nnm-2016-0251. [DOI] [PubMed] [Google Scholar]
- 38.Luo J., Hu Y.L., Wang H. Ursolic acid inhibits breast cancer growth by inhibiting proliferation, inducing autophagy and apoptosis, and suppressing inflammatory responses via the PI3K/AKT and NF-kappaB signaling pathways in vitro. Exp Ther Med. 2017;14(4):3623–3631. doi: 10.3892/etm.2017.4965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liu L., Zhang J., Li M., Zhang X., Zhang J., Li Z., et al. Inhibition of HepG2 cell proliferation by ursolic acid and polysaccharides via the downregulation of cyclooxygenase-2. Mol Med Rep. 2014;9(6):2505–2511. doi: 10.3892/mmr.2014.2059. [DOI] [PubMed] [Google Scholar]
- 40.Guo J.L., Han T., Bao L., Li X.M., Ma J.Q., Tang L.P. Ursolic acid promotes the apoptosis of cervical cancer cells by regulating endoplasmic reticulum stress. J Obstet Gynaecol Res. 2019;45(4):877–881. doi: 10.1111/jog.13919. [DOI] [PubMed] [Google Scholar]
- 41.Liu K., Guo L., Miao L., Bao W., Yang J., Li X., et al. Ursolic acid inhibits epithelial-mesenchymal transition by suppressing the expression of astrocyte-elevated gene-1 in human nonsmall cell lung cancer A549 cells. Anticancer Drugs. 2013;24(5):494–503. doi: 10.1097/CAD.0b013e328360093b. [DOI] [PubMed] [Google Scholar]
- 42.Harmand P.O., Duval R., Delage C., Simon A. Ursolic acid induces apoptosis through mitochondrial intrinsic pathway and caspase-3 activation in M4Beu melanoma cells. Int J Cancer. 2005;114(1):1–11. doi: 10.1002/ijc.20588. [DOI] [PubMed] [Google Scholar]
- 43.Weng H., Tan Z.J., Hu Y.P., Shu Y.J., Bao R.F., Jiang L., et al. Ursolic acid induces cell cycle arrest and apoptosis of gallbladder carcinoma cells. Cancer Cell Int. 2014;14(1):96. doi: 10.1186/s12935-014-0096-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gai W.T., Yu D.P., Wang X.S., Wang P.T. Anti-cancer effect of ursolic acid activates apoptosis through ROCK/PTEN mediated mitochondrial translocation of cofilin-1 in prostate cancer. Oncol Lett. 2016;12(4):2880–2885. doi: 10.3892/ol.2016.5015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dougherty U., Sehdev A., Cerda S., Mustafi R., Little N., Yuan W., et al. Epidermal growth factor receptor controls flat dysplastic aberrant crypt foci development and colon cancer progression in the rat azoxymethane model. Clin Cancer Res. 2008;14(8):2253–2262. doi: 10.1158/1078-0432.CCR-07-4926. [DOI] [PubMed] [Google Scholar]
- 46.Liu P., Du R., Yu X. Ursolic acid exhibits potent anticancer effects in human metastatic melanoma cancer cells (SK-MEL-24) via apoptosis induction, inhibition of cell migration and invasion, cell cycle arrest, and inhibition of mitogen-activated protein kinase (MAPK)/ERK signaling pathway. Med Sci Monit. 2019;25:1283–1290. doi: 10.12659/MSM.913069. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 47.Meng Y., Lin Z.M., Ge N., Zhang D.L., Huang J., Kong F. Ursolic acid induces apoptosis of prostate cancer cells via the PI3K/Akt/mTOR pathway. Am J Chin Med. 2015;43(7):1471–1486. doi: 10.1142/S0192415X15500834. [DOI] [PubMed] [Google Scholar]
- 48.Kim K., Shin E.A., Jung J.H., Park J.E., Kim D.S., Shim B.S., et al. Ursolic acid induces apoptosis in colorectal cancer cells partially via upregulation of microrna-4500 and inhibition of JAK2/STAT3 phosphorylation. Int J Mol Sci. 2018;20(1):E114. doi: 10.3390/ijms20010114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.de Araujo Lopes S.C, Vinicius Melo Novais M., Salviano Teixeira C., Honorato-Sampaio K., Tadeu Pereira M., Ferreira L.A., et al. Preparation, physicochemical characterization, and cell viability evaluation of long-circulating and pH-sensitive liposomes containing ursolic acid. Biomed Res Int. 2013;2013 doi: 10.1155/2013/467147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhao T., Liu Y., Gao Z., Gao D., Li N., Bian Y., et al. Self-assembly and cytotoxicity study of PEG-modified ursolic acid liposomes. Mater Sci Eng C Mater Biol Appl. 2015;53:196–203. doi: 10.1016/j.msec.2015.04.022. [DOI] [PubMed] [Google Scholar]
- 51.Wang M., Zhao T., Liu Y., Wang Q., Xing S., Li L., et al. Ursolic acid liposomes with chitosan modification: promising antitumor drug delivery and efficacy. Mater Sci Eng C Mater Biol Appl. 2017;71:1231–1240. doi: 10.1016/j.msec.2016.11.014. [DOI] [PubMed] [Google Scholar]
- 52.Yang G., Yang T., Zhang W., Lu M., Ma X., Xiang G. In vitro and in vivo antitumor effects of folate-targeted ursolic acid stealth liposome. J Agric Food Chem. 2014;62(10):2207–2215. doi: 10.1021/jf405675g. [DOI] [PubMed] [Google Scholar]
- 53.Zhang H., Zheng D., Ding J., Xu H., Li X., Sun W. Efficient delivery of ursolic acid by poly(N-vinylpyrrolidone)-block-poly (epsilon-caprolactone) nanoparticles for inhibiting the growth of hepatocellular carcinoma in vitro and in vivo. Int J Nanomed. 2015;10:1909–1920. doi: 10.2147/IJN.S77125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Antonio E., Antunes O.D.R.J., de Araujo I.S., Khalil N.M., Mainardes R.M. Poly(lactic acid) nanoparticles loaded with ursolic acid: characterization and in vitro evaluation of radical scavenging activity and cytotoxicity. Mater Sci Eng C Mater Biol Appl. 2017;71:156–166. doi: 10.1016/j.msec.2016.09.080. [DOI] [PubMed] [Google Scholar]
- 55.Baishya R., Nayak D.K., Kumar D., Sinha S., Gupta A., Ganguly S., et al. Ursolic acid loaded PLGA nanoparticles: in vitro and in vivo evaluation to explore tumor targeting ability on B16F10 melanoma cell lines. Pharm Res. 2016;33(11):2691–2703. doi: 10.1007/s11095-016-1994-1. [DOI] [PubMed] [Google Scholar]
- 56.Oprean C., Zambori C., Borcan F., Soica C., Zupko I., Minorics R., et al. Anti-proliferative and antibacterial in vitro evaluation of the polyurethane nanostructures incorporating pentacyclic triterpenes. Pharm Biol. 2016;54(11):2714–2722. doi: 10.1080/13880209.2016.1180538. [DOI] [PubMed] [Google Scholar]
- 57.Jin H., Pi J., Yang F., Wu C., Cheng X., Bai H., et al. Ursolic acid-loaded chitosan nanoparticles induce potent anti-angiogenesis in tumor. Appl Microbiol Biotechnol. 2016;100(15):6643–6652. doi: 10.1007/s00253-016-7360-8. [DOI] [PubMed] [Google Scholar]
- 58.Jin H., Pi J., Yang F., Jiang J., Wang X., Bai H., et al. Folate-Chitosan nanoparticles loaded with ursolic acid confer anti-breast cancer activities in vitro and in vivo. Sci Rep. 2016;6:30782. doi: 10.1038/srep30782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gao Y., Li Z., Xie X., Wang C., You J., Mo F., et al. Dendrimeric anticancer prodrugs for targeted delivery of ursolic acid to folate receptor-expressing cancer cells: synthesis and biological evaluation. Eur J Pharm Sci. 2015;70:55–63. doi: 10.1016/j.ejps.2015.01.007. [DOI] [PubMed] [Google Scholar]
- 60.Shen Z., Li B., Liu Y., Zheng G., Guo Y., Zhao R., et al. A self-assembly nanodrug delivery system based on amphiphilic low generations of PAMAM dendrimers-ursolic acid conjugate modified by lactobionic acid for HCC targeting therapy. Nanomedicine. 2018;14(2):227–236. doi: 10.1016/j.nano.2017.10.007. [DOI] [PubMed] [Google Scholar]
- 61.Liu Y., Liu K., Li X., Xiao S., Zheng D., Zhu P., et al. A novel self-assembled nanoparticle platform based on pectin-eight-arm polyethylene glycol-drug conjugates for co-delivery of anticancer drugs. Mater Sci Eng C Mater Biol Appl. 2018;86:28–41. doi: 10.1016/j.msec.2017.12.018. [DOI] [PubMed] [Google Scholar]
- 62.Liu YX, Liu KF, Li CX, Wang LY, Liu J, He J, et al. Self-assembled nanoparticles based on a carboxymethylcellulose–ursolic acid conjugate for anticancer combination therapy. RSC Adv. 2017;7(58):36256–36268. [Google Scholar]
- 63.Nahak P., Karmakar G., Chettri P., Roy B., Guha P., Besra S.E., et al. Influence of lipid core material on physicochemical characteristics of an ursolic acid-loaded nanostructured lipid carrier: an attempt to enhance anticancer activity. Langmuir. 2016;32(38):9816–9825. doi: 10.1021/acs.langmuir.6b02402. [DOI] [PubMed] [Google Scholar]
- 64.Zhou X.J., Hu X.M., Yi Y.M., Wan J. Preparation and body distribution of freeze-dried powder of ursolic acid phospholipid nanoparticles. Drug Dev Ind Pharm. 2009;35(3):305–310. doi: 10.1080/03639040802302165. [DOI] [PubMed] [Google Scholar]
- 65.Biswas S., Mukherjee P.K., Harwansh R.K., Bannerjee S., Bhattacharjee P.Enhanced bioavailability and hepatoprotectivity of optimized ursolic acid-phospholipid complex. Drug Dev Ind Pharm2019:1–13. [DOI] [PubMed]
- 66.Li T., Chen X., Liu Y., Fan L., Lin L., Xu Y., et al. pH-Sensitive mesoporous silica nanoparticles anticancer prodrugs for sustained release of ursolic acid and the enhanced anti-cancer efficacy for hepatocellular carcinoma cancer. Eur J Pharm Sci. 2017;96:456–463. doi: 10.1016/j.ejps.2016.10.019. [DOI] [PubMed] [Google Scholar]
- 67.Jiang K., Chi T., Li T., Zheng G., Fan L., Liu Y., et al. A smart pH-responsive nano-carrier as a drug delivery system for the targeted delivery of ursolic acid: suppresses cancer growth and metastasis by modulating P53/MMP-9/PTEN/CD44 mediated multiple signaling pathways. Nanoscale. 2017;9(27):9428–9439. doi: 10.1039/c7nr01677h. [DOI] [PubMed] [Google Scholar]
- 68.Zhao R., Li T., Zheng G., Jiang K., Fan L., Shao J. Simultaneous inhibition of growth and metastasis of hepatocellular carcinoma by co-delivery of ursolic acid and sorafenib using lactobionic acid modified and pH-sensitive chitosan-conjugated mesoporous silica nanocomplex. Biomaterials. 2017;143:1–16. doi: 10.1016/j.biomaterials.2017.07.030. [DOI] [PubMed] [Google Scholar]
- 69.Zhong Y., Wang J., Wang Y., Wu B. Preparation and evaluation of liposome-encapsulated codrug LMX. Int J Pharm. 2012;438(1–2):240–248. doi: 10.1016/j.ijpharm.2012.08.051. [DOI] [PubMed] [Google Scholar]
- 70.Zamboni W.C. Liposomal, nanoparticle, and conjugated formulations of anticancer agents. Clin Cancer Res. 2005;11(23):8230–8234. doi: 10.1158/1078-0432.CCR-05-1895. [DOI] [PubMed] [Google Scholar]
- 71.Sapra P., Tyagi P., Allen T.M. Ligand-targeted liposomes for cancer treatment. Curr Drug Deliv. 2005;2(4):369–381. doi: 10.2174/156720105774370159. [DOI] [PubMed] [Google Scholar]
- 72.Malhotra M., Tomaro-Duchesneau C., Prakash S. Synthesis of TAT peptide-tagged PEGylated chitosan nanoparticles for siRNA delivery targeting neurodegenerative diseases. Biomaterials. 2013;34(4):1270–1280. doi: 10.1016/j.biomaterials.2012.10.013. [DOI] [PubMed] [Google Scholar]
- 73.Bhavna M.d.S., Ali M., Baboota S., Sahni J.K., Bhatnagar A., et al. Preparation, characterization, in vivo biodistribution and pharmacokinetic studies of donepezil-loaded PLGA nanoparticles for brain targeting. Drug Dev Ind Pharm. 2014;40(2):278–287. doi: 10.3109/03639045.2012.758130. [DOI] [PubMed] [Google Scholar]
- 74.Wang S., Meng X., Dong Y. Ursolic acid nanoparticles inhibit cervical cancer growth in vitro and in vivo via apoptosis induction. Int J Oncol. 2017;50(4):1330–1340. doi: 10.3892/ijo.2017.3890. [DOI] [PubMed] [Google Scholar]
- 75.Ekladious I., Colson Y.L., Grinstaff M.W. Polymer-drug conjugate therapeutics: advances, insights and prospects. Nat Rev Drug Discov. 2019;18(4):273–294. doi: 10.1038/s41573-018-0005-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tapeinos C., Battaglini M., Ciofani G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J Control Release. 2017;264:306–332. doi: 10.1016/j.jconrel.2017.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Garces A., Amaral M.H., Sousa Lobo J.M., Silva A.C. Formulations based on solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) for cutaneous use: a review. Eur J Pharm Sci. 2018;112:159–167. doi: 10.1016/j.ejps.2017.11.023. [DOI] [PubMed] [Google Scholar]
- 78.Beloqui A., Solinis M.A., Rodriguez-Gascon A., Almeida A.J., Preat V. Nanostructured lipid carriers: promising drug delivery systems for future clinics. Nanomedicine. 2016;12(1):143–161. doi: 10.1016/j.nano.2015.09.004. [DOI] [PubMed] [Google Scholar]
- 79.Shang F., Sun J., Wu S., Liu H., Guan J., Kan Q. Direct synthesis of acid–base bifunctionalized hexagonal mesoporous silica and its catalytic activity in cascade reactions. J Colloid Interface Sci. 2011;355(1):190–197. doi: 10.1016/j.jcis.2010.10.042. [DOI] [PubMed] [Google Scholar]
- 80.Liu T., Li L., Teng X., Huang X., Liu H., Chen D., et al. Single and repeated dose toxicity of mesoporous hollow silica nanoparticles in intravenously exposed mice. Biomaterials. 2011;32(6):1657–1668. doi: 10.1016/j.biomaterials.2010.10.035. [DOI] [PubMed] [Google Scholar]
- 81.He Q., Gao Y., Zhang L., Zhang Z., Gao F., Ji X., et al. A pH-responsive mesoporous silica nanoparticles-based multi-drug delivery system for overcoming multi-drug resistance. Biomaterials. 2011;32(30):7711–7720. doi: 10.1016/j.biomaterials.2011.06.066. [DOI] [PubMed] [Google Scholar]
- 82.Li L., Tang F., Liu H., Liu T., Hao N., Chen D., et al. In vivo delivery of silica nanorattle encapsulated docetaxel for liver cancer therapy with low toxicity and high efficacy. ACS Nano. 2010;4(11):6874–6882. doi: 10.1021/nn100918a. [DOI] [PubMed] [Google Scholar]
- 83.Lu J., Liong M., Zink J.I., Tamanoi F. Mesoporous silica nanoparticles as a delivery system for hydrophobic anticancer drugs. Small. 2007;3(8):1341–1346. doi: 10.1002/smll.200700005. [DOI] [PubMed] [Google Scholar]
- 84.Liu G., Gao J., Ai H., Chen X. Applications and potential toxicity of magnetic iron oxide nanoparticles. Small. 2013;9(9–10):1533–1545. doi: 10.1002/smll.201201531. [DOI] [PubMed] [Google Scholar]
- 85.Jia L., Zhao Y., Liang X.J. Fast evolving nanotechnology and relevant programs and entities in China. Nano Today. 2011;6(1):6–11. [Google Scholar]
- 86.Song J., Wang Y., Song Y., Chan H., Bi C., Yang X., et al. Development and characterisation of ursolic acid nanocrystals without stabiliser having improved dissolution rate and in vitro anticancer activity. AAPS PharmSciTech. 2014;15(1):11–19. doi: 10.1208/s12249-013-0028-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Fan L., Zhang B., Xu A., Shen Z., Guo Y., Zhao R., et al. Carrier-free, pure nanodrug formed by the self-assembly of an anticancer drug for cancer immune therapy. Mol Pharm. 2018;15(6):2466–2478. doi: 10.1021/acs.molpharmaceut.8b00444. [DOI] [PubMed] [Google Scholar]
- 88.Ge Z.Q., Du X.Y., Huang X.N., Qiao B. Enhanced oral bioavailability of ursolic acid nanoparticles via antisolvent precipitation with TPGS1000 as a stabilizer. J Drug Deliv Sci Technol. 2015;29:210–217. [Google Scholar]
- 89.Guo Y., Jiang K., Shen Z., Zheng G., Fan L., Zhao R., et al. A small molecule nanodrug by self-assembly of dual anticancer drugs and photosensitizer for synergistic near-Infrared cancer theranostics. ACS Appl Mater Interfaces. 2017;9(50):43508–43519. doi: 10.1021/acsami.7b14755. [DOI] [PubMed] [Google Scholar]
- 90.Zhao R., Zheng G., Fan L., Shen Z., Jiang K., Guo Y., et al. Carrier-free nanodrug by co-assembly of chemotherapeutic agent and photosensitizer for cancer imaging and chemo-photo combination therapy. Acta Biomater. 2018;70:197–210. doi: 10.1016/j.actbio.2018.01.028. [DOI] [PubMed] [Google Scholar]
- 91.Jiang K., Han L., Guo Y., Zheng G., Fan L., Shen Z., et al. A carrier-free dual-drug nanodelivery system functionalized with aptamer specific targeting HER2-overexpressing cancer cells. J Mater Chem B. 2017;5(46):9121–9129. doi: 10.1039/c7tb02562a. [DOI] [PubMed] [Google Scholar]
- 92.Cheng W., Dahmani F.Z., Zhang J., Xiong H., Wu Y., Yin L., et al. Anti-angiogenic activity and antitumor efficacy of amphiphilic twin drug from ursolic acid and low molecular weight heparin. Nanotechnology. 2017;28(7) doi: 10.1088/1361-6528/aa53c6. [DOI] [PubMed] [Google Scholar]
- 93.Xiong H., Wu Y., Jiang Z., Zhou J., Yang M., Yao J. pH-activatable polymeric nanodrugs enhanced tumor chemo/antiangiogenic combination therapy through improving targeting drug release. J Colloid Interface Sci. 2019;536:135–148. doi: 10.1016/j.jcis.2018.10.039. [DOI] [PubMed] [Google Scholar]
- 94.Li C., Lin J., Wu P., Zhao R., Zou J., Zhou M., et al. Small molecule nanodrug assembled of dual-anticancer drug conjugate for synergetic cancer metastasis therapy. Bioconjug Chem. 2018;29(10):3495–3502. doi: 10.1021/acs.bioconjchem.8b00657. [DOI] [PubMed] [Google Scholar]
- 95.Müller R.H., Shegokar R., Gohla S., Keck C.M. Intracellular delivery. Springer; 2011. Nanocrystals: production, cellular drug delivery, current and future products; pp. 411–432. [Google Scholar]
- 96.Junghanns J.-U.A., Müller R.H. Nanocrystal technology, drug delivery and clinical applications. Int J Nanomed. 2008;3(3):295. doi: 10.2147/ijn.s595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Pi J., Liu Z., Wang H., Gu X., Wang S., Zhang B., et al. Ursolic acid nanocrystals for dissolution rate and bioavailability enhancement: influence of different particle size. Curr Drug Deliv. 2016;13(8):1358–1366. doi: 10.2174/1567201813666160307142757. [DOI] [PubMed] [Google Scholar]
- 98.Wang Y., Song J., Chow S.F., Chow A.H., Zheng Y. Particle size tailoring of ursolic acid nanosuspensions for improved anticancer activity by controlled antisolvent precipitation. Int J Pharm. 2015;494(1):479–489. doi: 10.1016/j.ijpharm.2015.08.052. [DOI] [PubMed] [Google Scholar]
- 99.Qiu L., Zhao X., Zu Y., Zhang Y., Liu Y., Wu W., et al. Ursolic acid nanoparticles for oral delivery prepared by emulsion solvent evaporation method: characterization, in vitro evaluation of radical scavenging activity and bioavailability. Artif Cells Nanomed Biotechnol. 2019;47(1):610–621. doi: 10.1080/21691401.2019.1573739. [DOI] [PubMed] [Google Scholar]
- 100.Tang Q., Liu Y., Li T., Yang X., Zheng G., Chen H., et al. A novel co-drug of aspirin and ursolic acid interrupts adhesion, invasion and migration of cancer cells to vascular endothelium via regulating EMT and EGFR-mediated signaling pathways: multiple targets for cancer metastasis prevention and treatment. Oncotarget. 2016;7(45):73114–73129. doi: 10.18632/oncotarget.12232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zheng G., Shen Z., Xu A., Jiang K., Wu P., Yang X., et al. Synergistic chemopreventive and therapeutic effects of co-drug UA-Met: implication in tumor metastasis. J Agric Food Chem. 2017;65(50):10973–10983. doi: 10.1021/acs.jafc.7b04378. [DOI] [PubMed] [Google Scholar]
- 102.Xia Y., Wei G., Si D., Liu C. Quantitation of ursolic acid in human plasma by ultra performance liquid chromatography tandem mass spectrometry and its pharmacokinetic study. J Chromatogr B Analyt Technol Biomed Life Sci. 2011;879(2):219–224. doi: 10.1016/j.jchromb.2010.11.037. [DOI] [PubMed] [Google Scholar]
- 103.Wang X.H., Zhou S.Y., Qian Z.Z., Zhang H.L., Qiu L.H., Song Z., et al. Evaluation of toxicity and single-dose pharmacokinetics of intravenous ursolic acid liposomes in healthy adult volunteers and patients with advanced solid tumors. Expert Opin Drug Metab Toxicol. 2013;9(2):117–125. doi: 10.1517/17425255.2013.738667. [DOI] [PubMed] [Google Scholar]
- 104.Zhu Z., Qian Z., Yan Z., Zhao C., Wang H., Ying G. A phase I pharmacokinetic study of ursolic acid nanoliposomes in healthy volunteers and patients with advanced solid tumors. Int J Nanomed. 2013;8:129–136. doi: 10.2147/IJN.S38271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Qian Z., Wang X., Song Z., Zhang H., Zhou S., Zhao J., et al. A phase I trial to evaluate the multiple-dose safety and antitumor activity of ursolic acid liposomes in subjects with advanced solid tumors. Biomed Res Int. 2015;2015 doi: 10.1155/2015/809714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Yallapu M.M., Jaggi M., Chauhan S.C. Curcumin nanomedicine: a road to cancer therapeutics. Curr Pharm Des. 2013;19(11):1994–2010. doi: 10.2174/138161213805289219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kalyane D., Raval N., Maheshwari R., Tambe V., Kalia K., Tekade R.K. Employment of enhanced permeability and retention effect (EPR): nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Mater Sci Eng C Mater Biol Appl. 2019;98:1252–1276. doi: 10.1016/j.msec.2019.01.066. [DOI] [PubMed] [Google Scholar]
- 108.Farokhzad O.C., Langer R. Impact of nanotechnology on drug delivery. ACS Nano. 2009;3(1):16–20. doi: 10.1021/nn900002m. [DOI] [PubMed] [Google Scholar]
- 109.Guan J., Jiang Z., Wang M., Liu Y., Liu J., Yang Y., et al. Short peptide-mediated brain-targeted drug delivery with enhanced immunocompatibility. Mol Pharm. 2019;16(2):907–913. doi: 10.1021/acs.molpharmaceut.8b01216. [DOI] [PubMed] [Google Scholar]
- 110.Srinivasarao M., Galliford C.V., Low P.S. Principles in the design of ligand-targeted cancer therapeutics and imaging agents. Nat Rev Drug Discov. 2015;14(3):203–219. doi: 10.1038/nrd4519. [DOI] [PubMed] [Google Scholar]
- 111.Torchilin V. Tumor delivery of macromolecular drugs based on the EPR effect. Adv Drug Deliv Rev. 2011;63(3):131–135. doi: 10.1016/j.addr.2010.03.011. [DOI] [PubMed] [Google Scholar]
- 112.Yingchoncharoen P., Kalinowski D.S., Richardson D.R. Lipid-based drug delivery systems in cancer therapy: what is available and what is yet to come. Pharmacol Rev. 2016;68(3):701–787. doi: 10.1124/pr.115.012070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Wicki A., Witzigmann D., Balasubramanian V., Huwyler J. Nanomedicine in cancer therapy: challenges, opportunities, and clinical applications. J Control Release. 2015;200:138–157. doi: 10.1016/j.jconrel.2014.12.030. [DOI] [PubMed] [Google Scholar]
- 114.Jain R.K., Stylianopoulos T. Delivering nanomedicine to solid tumors. Nat Rev Clin Oncol. 2010;7(11):653–664. doi: 10.1038/nrclinonc.2010.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Dissanayake S., Denny W.A., Gamage S., Sarojini V. Recent developments in anticancer drug delivery using cell penetrating and tumor targeting peptides. J Control Release. 2017;250:62–76. doi: 10.1016/j.jconrel.2017.02.006. [DOI] [PubMed] [Google Scholar]
- 116.Zhang Y., Wu M., Dai W., Li Y., Wang X., Tan D., et al. Gold nanoclusters for controlled insulin release and glucose regulation in diabetes. Nanoscale. 2019;11(13):6471–6479. doi: 10.1039/c9nr00668k. [DOI] [PubMed] [Google Scholar]
- 117.Yasser M, Teaima M, El-Nabarawi M, El-Monem RA.Cubosomal based oral tablet for controlled drug delivery of telmisartan: formulation, in vitro evaluation and in vivo comparative pharmacokinetic study in rabbits. Drug Dev Ind Pharm2019:1–14. [DOI] [PubMed]
- 118.Bhusal P., Harrison J., Sharma M., Jones D.S., Hill A.G., Svirskis D. Controlled release drug delivery systems to improve post-operative pharmacotherapy. Drug Deliv Transl Res. 2016;6(5):441–451. doi: 10.1007/s13346-016-0305-z. [DOI] [PubMed] [Google Scholar]
- 119.Shi J., Kantoff P.W., Wooster R., Farokhzad O.C. Cancer nanomedicine: progress, challenges and opportunities. Nat Rev Cancer. 2017;17(1):20–37. doi: 10.1038/nrc.2016.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ediriwickrema A., Saltzman W.M. Nanotherapy for cancer: targeting and multifunctionality in the future of cancer therapies. ACS Biomater Sci Eng. 2015;1(2):64–78. doi: 10.1021/ab500084g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mura S., Nicolas J., Couvreur P. Stimuli-responsive nanocarriers for drug delivery. Nat Mater. 2013;12(11):991–1003. doi: 10.1038/nmat3776. [DOI] [PubMed] [Google Scholar]
- 122.Lu Y., Sun W., Gu Z. Stimuli-responsive nanomaterials for therapeutic protein delivery. J Control Release. 2014;194:1–19. doi: 10.1016/j.jconrel.2014.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Bertrand N., Leroux J.C. The journey of a drug-carrier in the body: an anatomo-physiological perspective. J Control Release. 2012;161(2):152–163. doi: 10.1016/j.jconrel.2011.09.098. [DOI] [PubMed] [Google Scholar]
- 124.Shen S., Wu Y., Liu Y., Wu D. High drug-loading nanomedicines: progress, current status, and prospects. Int J Nanomed. 2017;12:4085–4109. doi: 10.2147/IJN.S132780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wong A. Quantitative modeling of the high-throughput production and in vivo kinetics of (drug-encapsulating) liposomes. PLoS ONE. 2010;5(4):e10280. doi: 10.1371/journal.pone.0010280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Wolfram J., Ferrari M. Clinical cancer nanomedicine. Nano Today. 2019;25:85–98. doi: 10.1016/j.nantod.2019.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Sarcan E.T., Silindir-Gunay M., Ozer A.Y. Theranostic polymeric nanoparticles for NIR imaging and photodynamic therapy. Int J Pharm. 2018;551(1–2):329–338. doi: 10.1016/j.ijpharm.2018.09.019. [DOI] [PubMed] [Google Scholar]
- 128.Greco F., Vicent M.J. Combination therapy: opportunities and challenges for polymer-drug conjugates as anticancer nanomedicines. Adv Drug Deliv Rev. 2009;61(13):1203–1213. doi: 10.1016/j.addr.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 129.Yang M.Y., Zhao R.R., Fang Y.F., Jiang J.L., Yuan X.T., Shao J.W. Carrier-free nanodrug: a novel strategy of cancer diagnosis and synergistic therapy. Int J Pharm. 2019;570 doi: 10.1016/j.ijpharm.2019.118663. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.