Abstract
The outbreak of novel coronavirus disease 2019 (COVID‐19) has become a major pandemic threat worldwide. According to the existing clinical data, this virus not only causes respiratory diseases and affects the lungs but also induces histopathological or functional changes in various organs like the testis and also the male genital tract. The renin‐angiotensin system (RAS), also ACE 2 and TMPRSS2 play an important role in the cellular entry for SARS‐CoV‐2. Because the male genital system presents high ACE 2 expression, the importance of this pathway increases in COVID‐19 cases. As the COVID‐19 pandemic has affected the male genital system in direct or indirect ways and showed a negative impact on male reproduction, this paper focuses on the possible mechanisms underlying the damage caused by COVID‐19 to the testis and also other components of the male genital tract.
Keywords: coronavirus, genital tract, pandemics
Highlights
The male genital system presents high ACE 2 expression therefore, it will be highly important to investigate and clarify the relationship between COVID‐19 and the male genital tract.
1. INTRODUCTION
At the end of December 2019, a novel coronavirus that is significantly contagious than the seasonal flu formally named severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2); also known as coronavirus disease‐19 (COVID‐19) exploded in Wuhan (China) and had spread rapidly worldwide. 1 , 2 The outbreak was declared as a global pandemic in March 2020 by the World Health Organization (WHO). 3
According to the existing clinical data, this virus not only causes respiratory diseases and affects the lungs but also induce histopathological changes in various organs of a nonrespiratory system, such as the kidney, 3 , 4 liver, brain, and heart. 4 SARS‐CoV‐2 was also found in semen analysis of male patients and like many other viruses, such as mumps, hepatitis, herpes simplex, influenza, and human immunodeficiency viruses (HIV), it could infect the testis, the male genital tract and cause damage to male fertility. 4 , 5
There is a very limited number of data about the effects of COVID‐19 on male fertility, so it has become an important topic for researchers. This paper focuses on the possible mechanisms underlying the damage caused by COVID‐19 to the testis and also the male genital tract.
2. CORONAVIRIDAE AND SARS‐COV‐2
Coronaviridae family (Betacoronaviruses) are single‐chain, positive‐sense RNA genome, which is 26–32 kilobases in length with spiny extensions that are seen at the electron microscopic level. Because of these extensions, they received the name “corona,” which means “crown” in Latin. In addition to many subspecies found in animals, there are several subspecies of these viruses found in humans that can be transmitted from person to person and often cause cold, such as HCoV‐229E, HCoV‐OC43, HCoV‐NL63, and HKU1‐CoV. 2 , 6
Coronaviruses have previously caused more serious, highly pathogenic, and lethal diseases, such as middle east respiratory syndrome coronavirus (MERS‐CoV) and severe acute respiratory syndrome coronavirus (SARS‐CoV). 4 , 7
SARS‐CoV‐2 has four main structural proteins: spike surface glycoprotein, small envelope protein, matrix protein, and nucleocapsid protein. This virus binds to host receptors via spike surface glycoprotein from its receptor binding sites. 8 , 9
3. THE RENIN‐ANGIOTENSIN‐ALDOSTERONE SYSTEM (RAAS)
The RAAS is a hormonal cascade system that regulates arterial pressure and fluid balance for homeostatic control in the body. When blood pressure is reduced, this hormonal cascade begins with the biosynthesis of renin from the juxtaglomerular cells in the kidney. Renin stimulates the formation of angiotensin (Ang) I (or Ang 1‐9) from angiotensinogen, released primarily by the liver. 10 , 11 Additionally, angiotensinogen messenger RNA (mRNA) expression has been detected in many other tissues, such as the kidney, brain, heart, adrenal gland, ovary, placenta, and adipose tissue. 12 Angiotensin I is then converted to angiotensin II (or Ang 1‐7) by the angiotensin‐converting enzyme (ACE), found predominantly on the surface of vascular endothelial cells of the lungs. Angiotensin II is the most functional molecule and creates vasoconstriction in blood vessels, which increases blood pressure and also stimulates the release of the hormone aldosterone from the adrenal cortex. Aldosterone increases sodium and water reuptake from kidney tubules into the blood. It increases the amount of fluid in the body and therefore, blood pressure. 10 , 11 Additionally, angiotensin III (or Ang 2‐8) and angiotensin IV (or Ang 3‐8) are known as other products of RAAS. 13 Angiotensin II needs receptors to demonstrate its physiological and pathophysiological actions, so at least four angiotensin II receptor subtypes have been described; AT1R, AT2R, AT3R, and AT4 R locate in different tissues. 11 , 14
ACE 2 has recently been identified enzyme and a novel homolog of ACE, attached to the cell membranes of cells and expressed in 72 different human tissues such as the lungs, gastrointestinal system, heart, kidney, and testis. 15 Unlike ACE, the activity of ACE 2 is not affected by ACE inhibitors 11 and does not catalyze the formation of angiotensin II‐like ACE. 15 ACE 2 catalyzes the generation of vasodilator Ang 1‐7 from vasoconstricting angiotensin II by separating phenylalanine and Ang 1‐9 from angiotensin I, 10 suggesting that ACE 2 may act to provide negative feedback regulation of the RAAS. 11 , 15 Like angiotensin II, also Ang 1‐7 needs a receptor to demonstrate its function and this receptor is termed as MasR. 10 So finally, RAAS regulates blood pressure and renal function by the pressor and depressor arms; the pressor arm is ACE/angiotensin II/AT1R and the depressor arm is ACE 2/Ang 1‐7/MasR. 16 , 17 These two arms show antagonistic biological responses because angiotensin II and Ang 1‐7 differ only in the single amino acid at the C‐terminal biochemically, which gives them the opposite functions 16 (Figure 1).
Figure 1.
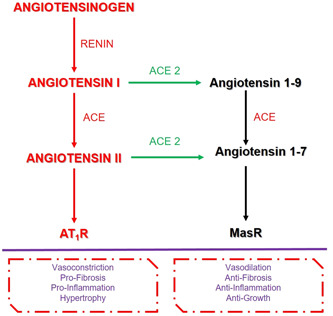
Summary of RAS
Interestingly, ACE 2 has recently been identified as the entry point into cells for some coronaviruses, including SARS and SARS‐CoV‐2. 10 As mentioned above, there are studies that show that this enzyme is detected in different parts of the body, such as enterocytes, renal tubules, gallbladder, cardiomyocytes, male reproductive cells, placental trophoblasts, ductal cells, eye, and vasculature; and hence it might increase the effects of the virus over the entire system. 18
4. THE ACE 2 EXPRESSION IN MALE REPRODUCTIVE SYSTEM
The typical and main components of RAAS have been demonstrated in the male reproductive tract, especially in the testis, epididymis, and also prostate gland. 19 , 20 The RAAS components associated with the regulation of steroidogenesis, testosterone production, spermatogenesis, 21 and sperm contractility in the testis and epididymis. 19
First, among several nonrespiratory organs, ACE 2 is highly expressed in the human testis and also in the male genital tract and seems to play an important role in male reproduction. 21 It was determined that the mRNA expression of ACE 2 was expressed in both germ and somatic cells of the testis. 22 Studies have shown that ACE 2 expression is observed in spermatogonia, Leydig cells, and Sertoli cells, predominantly. 23 , 24 ACE 2 in Leydig cells function in testosterone or steroidogenesis regulation, production, and local vascular regulation to balance the interstitial fluid volume via modulating the conversion of angiotensin II to angiotensin I, also ACE 2 in seminiferous epithelium functions in maintaining healthy spermatogenesis. 24 Additionally, a small percentage of the prostate hillock and club cells express ACE 2. 20 The expression level of ACE 2 was related to age; it has been found that the expression of ACE 2 increases from a young age to the middle age. 22 ACE 2 expression starts to increase around puberty and reaches its maximum expression during reproductive life. 21
Furthermore, the male reproductive tract and also the testis represent both source and target for active angiotensin peptides and their receptors. 19 For instance, Ang 1‐7 and its receptor Mas have been identified in the human testis. 19 , 21 Ang 1‐7 and MasR were detected in the seminiferous tubules (including Sertoli cells), in the interstitial compartment, mainly Leydig cells, and is much more pronounced in Leydig cells. 21
Researchers also investigated the differences in ACE 2/Ang 1‐7/MasR expressions between fertile and infertile men, and accordingly, the expressions were found higher in men with normal spermatogenesis. The testicular samples of infertile men with impaired spermatogenesis expressed MasR and ACE 2 mRNA at lower concentrations. Additionally, neither component of the RAAS was determined in the seminiferous tubules of infertile men with nonobstructive azoospermia. 19 , 21 Studies have indicated that MasR knockout mice showed aberrant testicular steroidogenesis. 21
5. SARS‐COV‐2 RECEPTOR FUNCTION OF ACE 2
It has recently been identified that like SARS and HCoV‐NL63 coronaviruses; also SARS‐CoV‐2 uses the same receptor, ACE 2, for entry into the cells through its surface spike (S) proteins. 7 , 25 , 26 Endocytosis of the virus is achieved through S protein (7). The binding affinity between ACE 2 and SARS‐CoV‐2 is nearly 10 to 20 fold higher than ACE 2 and SARS affinity. 26 According to some studies, the risk of infection with the virus decreased in mice if ACE 2 expression was reduced. 27
After binding of the virus, ACE 2 expression decreases/downregulates on the cell surface. This reduction causes to reduce the conversion of angiotensin II to Ang 1‐7 and also angiotensin I to Ang 1‐9 in the cell, so the amount of angiotensin II increases. This accumulation may lead to toxicity 7 , 26 , 28 and contributed to acute respiratory distress syndrome, inflammation, myocardial injury, neurological frequency, and gastrointestinal reflections 28 (Figure 2).
Figure 2.
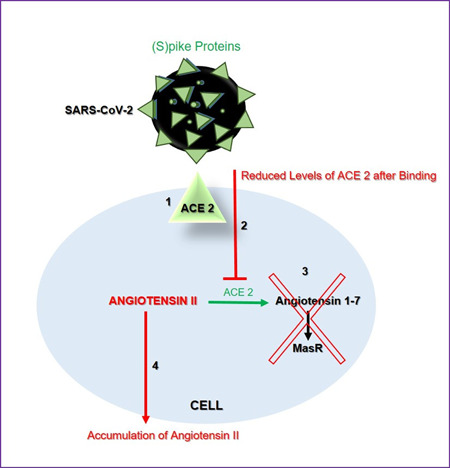
Summary of SARS‐CoV‐2 receptor function of ACE 2. ACE, angiotensin‐converting enzyme; SARS‐CoV‐2, severe acute respiratory syndrome coronavirus 2
SARS‐CoV‐2 causes progressive respiratory failure and alveolar damage in the lungs because of the high expression of ACE 2 in the type II alveolar cells, 26 but as mentioned above, many vital tissues include ACE 2 expressions except the lungs; therefore, these tissues are also in danger of SARS‐CoV‐2 infection. 25
According to data from different countries, the possibility of becoming COVID‐19 increases in young people where the ACE 2 is more expressed, but in contrast, the disease is more severe in the older population. In addition to chronic diseases, the most appropriate mechanism that can explain the severe progression of the disease in the older population may be the increased level of angiotensin II caused by a decreased level of ACE 2. 29 When the systemic effects of increased angiotensin II levels are evaluated, parallel clinical findings are observed with severe COVID‐19 cases, such as increasing pulmonary edema and apoptosis, lung fibroproliferation due to an increase in extracellular matrix production, endothelitis, and thrombus formation. 7
6. SARS‐COV‐2 IN THE MALE REPRODUCTIVE SYSTEM
6.1. Structural and functional changes of testis and epididymis in the case of COVID‐19
According to several studies, it is known that viruses could infect the testis directly. 5 Yang et al. examined the postmortem testis tissues from 12 COVID‐19 patients and searched them by light and electron microscopy. As a result, they observed that swelling, vacuolation, and cytoplasmic dilution in the Sertoli cells additionally, detachment between the basement membrane and seminiferous tubules, loss and cell debris into the lumen of seminiferous tubules. The number of Leydig cells was found significantly lower than the control group. Edema and inflammatory infiltrate composed of T lymphocytes and histiocytes were seen in the interstitial tissue; so taken together, significant seminiferous tubular injury, reduced number of Leydig cells, and lymphocytic inflammation were recognized in the testis of COVID‐19 patients. 3 There have many studies that support these findings and have said that SARS coronaviruses damage multiple organs, including testis and generally cause leukocyte infiltration, impaired spermatogenesis, widespread germ cell destruction with very few or no spermatozoa in the seminiferous tubules, thickened basement membrane, and macrophage (+) stainings in the testis. So, these studies emphasized that, like other SARS viruses, SARS‐CoV‐2 also poses a high risk of injury to the testicular cells and tissue 25 , 30 and damage the blood‐testis barrier. 5
In addition to these structural effects of SARS‐CoV‐2 in the testis, many other studies also provide information that SARS viruses can cause orchitis in humans. 5 , 25 , 30 , 31 Because of the SARS virus and SARS‐CoV‐2 sharing 78% genetic homology and in the same family/genus, it would be correct to predict that SARS‐CoV‐2 will also have such an effect. 25 Also, Xu et al. 32 examined the postmortem testis tissues from six patients who died from SARS‐CoV‐1 infection (at this point, it would be correct to know that SARS‐CoV‐2 is closely related to SARS‐CoV‐1 with 85% identity) and found that all six patients have orchitis. Also, genital complaints, such as scrotal discomfort, were reported in 19% COVID‐19 patients. 33 Similarly, Özveri et al. observed slightly increased vascularity, suggesting spermatic cord inflammation on ultrasound examinations in a COVID‐19 positive patient with a complaint swelling sensation and pain in his testis. No abnormality overlying scrotal skin and sperm count analysis were observed according to their examinations. 30
If we look at the mechanisms of these changes caused by SARS‐CoV‐2 in the testis, as mentioned above, this virus uses ACE 2 for entry into the cells through its surface spike (S) proteins. S proteins have two subunits, S1 and S2, which are responsible for receptor recognition and membrane fusion. Studies have shown that SARS‐CoV‐2 enters into the host cell through the binding of its C‐terminal domain of the S1 subunit to ACE 2. Additionally, some studies have reported that the level of autophagy receptor SQSTM1/p62 in SARS‐CoV‐2 infected cells has increased, suggesting a decrease in autophagy flux. So, SARS‐CoV‐2 itself or via ACE 2 can directly induce or inhibit the autophagy pathway to achieve virus survival. As a result, SARS‐CoV‐2 may cause male reproductive disorders by regulating the level of autophagy in male germ cells. 4 On the contrary, another hypothesis is that testis degeneration in the COVID‐19 cases is attributed to an increase in testicular temperature as an indirect effect of the inflammation. 5
Another molecule effective at entering the cell of the SARS‐CoV‐2 is host proteases like transmembrane serine protease 2 (TMPRSS2), which cleaves the viral S protein to induce a conformational change that allows to a fusion of the virus and host cell membranes. 34 TMPRSS2 is the key molecule for the successful infection process. 35 This protease is more expressed in human tissues than ACE 2; co‐expression of ACE 2 and TMPRSS2 has been shown in the testis, endometrium, and placenta. Researchers investigated the coexpression of these two molecules in the testis and accordingly, they found that ACE 2 is predominantly expressed in myoid cells, spermatogonia, Leydig, and Sertoli cells, while TMPRSS2 is expressed in spermatogonia and (elongated) spermatids of the testicular tissue 34 (Figure 3).
Figure 3.
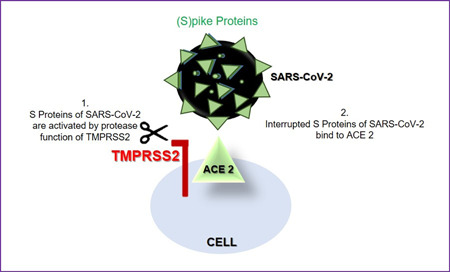
Comechanism of TMPRSS2 and ACE 2 in the fusion process of the testis. ACE, angiotensin‐converting enzyme; SARS‐CoV‐2, severe acute respiratory syndrome coronavirus 2; TMPRSS2, transmembrane serine protease 2
Orchiepididymitis is most frequently caused by viruses and relatively frequent in adolescents; Gagliardi et al. 36 reported the first described case of orchiepididymitis associated with a 14‐year‐old COVID‐19 patient.
6.2. Semen characteristics and prostate connection in the case of COVID‐19
Recent studies have reported that SARS‐CoV‐2 is easily found in human bodily fluids. 35 The presence of a virus in a semen sample is still a topic of discussion and research due to the small number of studies. For example, two different studies have analyzed SARS‐CoV‐2 presence in semen samples and according to these studies, SARS‐CoV‐2 (+) semen samples were found in two patients from 23 cured patients and four patients from 15 patients in the acute phase. Another study reported that SARS‐CoV‐2 was not detected in the semen samples of 34 COVID‐19 patients. 31
It is also known that the prostate gland secretes prostate fluid, one of the main seminal components, and muscles of the gland help in pushing the seminal fluid through the urethra during ejaculation. 31 The critical point is that, as we mentioned above, a small percentage of the prostate hillock and club cells express ACE 2 20 and also TMPRSS2 is highly expressed by the epithelium of the human prostate; 37 so it is more likely to get SARS‐CoV‐2 infection, which may affect its secretions. 31 These mechanisms could explain the SARS‐CoV‐2 (+) semen samples of the studies. 23
If the presence of the virus in semen is definitively proved by studies, assisted reproduction techniques will also be affected. For instance, testing all male patients like HIV or Hepatitis B/C cases, and using appropriate sperm washing techniques, or paying extra attention to sperm freezing for COVID‐19 positive patients. 35
On the contrary, another hypothesis is that poor semen parameters in the COVID‐19 cases are attributed to panic physiology as an indirect effect of the inflammation. 38 We would like to evaluate this connection and also hormonal differences in the case of COVID‐19 under a separate heading.
6.3. Effect of SARS‐CoV‐2 on male fertility via the brain, stress factors—hormonal connection
Like SARS‐CoV‐2, most viruses enter the human body through nasal and oral routes, and viral particles may break the blood‐brain barrier. It has been reported that the brain cells (glial cells and neurons) also express ACE 2 receptors, making them a possible target to induce neuronal death for SARS‐CoV‐2. Importantly, the central nervous system plays a critical role in endocrine control and spermatogenesis. 31 The Hypothalamic‐Pituitary‐Gonadal Axis (HPGa) exerts a vital role in reproduction; in other words, HPGa can inhibit the body's reproductive functions via hormones. 31 , 38
Gonadotropin‐releasing hormone (GnRH) expressing neurons from the hypothalamus secretes GnRH and it activates the release of the follicle‐stimulating hormone (FSH) and luteinizing hormone (LH) from the pituitary gland. A low level of GnRH causes a decrease in FSH and LH, resulting in impaired function of the Sertoli and Leydig cells. 31 Ma et al. 39 showed that COVID‐19 patients had significantly higher serum LH levels but decreased testosterone/LH and FSH levels than healthy men, suggesting potential hypogonadism. Taken together, patients with COVID‐19 have been found to present a reduced testosterone/LH ratio, indicating possible subclinical damage to male gonadal function. 5 Additionally, activation of the HPGa and subsequent alterations in hormone concentrations play a critical role in poor sperm quality. 38
From another point of view, panic physiology in the case of COVID‐19, stress, and negative moods, such as depression and anxiety, are associated with a lower secretion of sex hormone‐binding globulin, higher secretion of cortisol and prolactin, lower sperm count/sperm concentration/semen volume, and higher sperm DNA fragmentation, and also induced sexual dysfunction. 38
Therefore, besides its direct effects on testis, SARS‐CoV‐2 may affect fertility indirectly via the central nervous system. 31
We can summarize the direct and indirect effects of SARS‐CoV‐2 in the male genital tract as listed in the following table:
| Testis | Epididymis | Poor semen quality | Hormones | |
|---|---|---|---|---|
| Direct effects | Structural degenerations orchitis pain autophagy regulation | Orchitis | Virus (+) in semen | Breakdown of HPGa |
| Indirect effects | Increase of testicular temperature | – | Prostate fluid pushing during ejaculation stress hypogonadism breakdown of HPGa | – |
7. CONCLUSION
In conclusion, all preliminary findings mentioned above suggest that the COVID‐19 pandemic affects the male genital system in direct or indirect ways and shows a negative impact on male reproductive health, inducing spermatogenic failure. Additional studies are necessary to answer all the questions and further investigations are warranted, but ACE 2 and TMPRSS2 play an important role in the cellular entry for SARS‐CoV‐2. As the male genital system presents high ACE 2 expression, the importance of this pathway increases in COVID‐19 cases.
CONFLICT OF INTERESTS
The authors declare that there are no conflict of interests.
ACKNOWLEDGMENT
The authors thank all the researchers who contributed to the preparation of this review.
Seymen CM. The other side of COVID‐19 pandemic: Effects on male fertility. J Med Virol. 2021;93:1396–1402. 10.1002/jmv.26667
REFERENCES
- 1. Chen J, Jiang Q, Xia X, et al. Individual variation of the SARS‐CoV‐2 receptor ACE2 gene expression and regulation. Aging Cell. 2020;19(7):e13168. 10.1111/acel.13168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Iliano E, Trama F, Costantini E. Could COVID‐19 have an impact on male fertility? Andrologia. 2020;52(6):e13654. 10.1111/and.13654 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Yang M, Chen S, Huang B, et al. Pathological findings in the testes of COVID‐19 patients: clinical implications. Eur Urol Focus. 2020;6:1124–1129. 10.1016/j.euf.2020.05.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Sun J. The hypothesis that SARS‐CoV‐2 affects male reproductive ability by regulating autophagy. Med Hypotheses. 2020;143:110083. 10.1016/j.mehy.2020.110083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Youssef K, Abdelhak K. Male genital damage in Covid‐19 patients: are available data relevant? Asian J Urol. 2020. 10.1016/j.ajur.2020.06.005. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hasöksüz M, Kiliç S, Saraç F. Coronaviruses and SARS‐CoV‐2. Turk J Med Sci. 2020;50:549–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Eroğlu İ, Uyaroğlu OA, Güven GS. The relation between COVID‐19 and renin angiotensin aldosterone system in the light of current literatüre. Osmangazi J Med. 2020. 10.20515/otd.756606. In press. [DOI] [Google Scholar]
- 8. Wu A, Peng Y, Huang B, et al. Genome composition and divergence of the novel coronavirus (2019‐nCoV) originating in China. Cell Host Microbe. 2020;27(3):325–328. 10.1016/j.chom.2020.02.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ge XY, Li JL, Yang XL, et al. Isolation and characterization of a bat SARS‐like coronavirus that uses the ACE2 receptor. Nature. 2013;503:535–538. 10.1038/nature12711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Steckelings UM, Unger T. The Renin‐Angiotensin‐Aldosterone System. CRC Press; 2014:141–147. [Google Scholar]
- 11. Atlas SA. The renin‐angiotensin‐aldosterone system: patophysiological role and pharmacologic inhibition. J Manag Care Specialty Pharm. 2007;13:9–20. 10.18553/jmcp.2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Morgan L, Broughton PF, Kalsheker N. Angiotensinogen: molecular biology, biochemistry and physiology. Int J Biochem Cell Biol. 1996;28:1211–1222. 10.1016/s1357-2725(96)00086-6 [DOI] [PubMed] [Google Scholar]
- 13. Reudelhuber TL. The renin‐angiotensin system: peptides and enzymes beyond angiotensin II. Curr Opin Nephrol Hypertens. 2005;14:155–159. 10.1097/00041552-200503000-00011 [DOI] [PubMed] [Google Scholar]
- 14. Turgutalp K, Kıykım AA. Local renin‐angiotensin system: physiologic and patophysiologic effects. Mersin Üniversitesi Sağlık Bilimleri Dergisi. 2011;4(1):1–6. [Google Scholar]
- 15. Harmer D, Gilbert M, Borman R, Clarck KL. Quantitive mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Lett. 2002;532:107–110. 10.1016/s0014-5793(02)03640-2 [DOI] [PubMed] [Google Scholar]
- 16. Padda RS, Shi Y, Lo CS, Zhang SL, Chan JSD. Angiotensin‐(1‐7): a novel peptide to treat hypertension and nephropathy in diabetes? J Diabetes Metab. 2015;6(10):1–12. 10.4172/2155-6156.1000615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Silva DG, Braga T, Santos RAS. Angiotensin‐(1‐7): beyond the cardio‐renal actions. Clin Sci. 2013;124:443–456. 10.1042/CS20120461 [DOI] [PubMed] [Google Scholar]
- 18. Hikmet F, Méar L, Edvinsson Å, Micke P, Uhlén M, Lindskog C. The protein expression profile of ACE2 in human tissues. Mol Syst Biol. 2020;16:e9610. 10.15252/msb.20209610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Reis AB, Araújo FC, Pereira VM, Dos Reis AM, Santos RA, Reis FM. Angiotensin (1‐7) and its receptor Mas are expressed in the human testis: Implications for male fertility. J Mol Histol. 2010;41:75–80. 10.1007/s10735-010-9264-8 [DOI] [PubMed] [Google Scholar]
- 20. Song H, Seddighzadeh B, Cooperberg MR, Huang FW. Expression of ACE2, the SARS‐CoV‐2 receptor, and TMPRSS2 in prostate epithelial cells. Eur Urol. 2020;78(2):296–298. 10.1016/j.eururo.2020.04.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Younis JS, Abassi Z, Skorecki K. Is there an impact of the COVID‐19 pandemic on male fertility? The ACE2 connection. Am J Physiol Endocrinol Metab. 2020;318:E878–E880. 10.1152/ajpendo.00183.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Shen Q, Xiao X, Aierken A, et al. The ACE2 expression in Sertoli cells and germ cells may cause male reproductive disorder after SARS‐CoV‐2 infection. J Cell Mol Med. 2020;24:9472–9477. 10.1111/jcmm.15541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Wang Z, Xu X. scRNA‐seq profiling of human testes reveals the presence of the ACE2 receptor, a target for SARS‐CoV‐2 infection in spermatogonia, Leydig and Sertoli cells. Cells. 2020;9:920. 10.3390/cells9040920 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Verma S, Saksena S, Sadri‐Ardekani H. ACE2 receptor expression in testes: implications in coronavirus disease 2019 pathogenesis. Biol Reprod. 2020;2020:1–3. 10.1093/biolre/ioaa080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Chen F, Lou D. Rising concern on damaged testis of COVID‐19 patients. Urology. 2020;142:42. 10.1016/j.urology.2020.04.069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Fu J, Zhou B, Zhang L, et al. Expressions and significances of the angiotensin‑converting enzyme 2 gene, the receptor of SARS‑CoV‑2 for COVID‑19. Mol Biol Rep. 2020;14:1–10. 10.1007/s11033-020-05478-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kuba K, Imai Y, Rao S, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus‐induced lung injury. Nat Med. 2005;11(8):875–879. 10.1038/nm1267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Naik GO. COVID‐19 and the renin‐angiotensin‐aldosterone system. Clin Infect Dis. 2020;81:63–67. 10.1016/j.ando.2020.04.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. AlGhatrif M, Cingolani O, Lakatta EG. The dilemma of coronavirus disease 2019, aging, and cardiovascular disease: insights from cardiovascular aging science. JAMA Cardiol. 2020;5(7):747–748. 10.1001/jamacardio.2020.1329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Özveri H, Eren MT, Kırışoğlu CE, Sarıgüzel N. Atypical presentation of SARS‐CoV‐2 infection in male genitalia. Urol Case Rep. 2020;33:101349. 10.1016/j.eucr.2020.101349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Vishvkarma R, Rajender S. Could SARS‐CoV‐2 affect male fertility? Andrologia. 2020;52(9):e13712. 10.1111/and.13712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Xu J, Qi L, Chi X, et al. Orchitis: a complication of severe acute respiratory syndrome (SARS). Biol Reprod. 2006;74:410e6–416. 10.1016/j.rbmo.2020.04.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Pan F, Xiao X, Guo J, et al. No evidence of SARS‐CoV‐2 in semen of males recovering from COVID‐19. Fertil Steril. 2020;113(6):1135–1139. 10.1016/j.fertnstert.2020.04.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Stanley KE, Thomas E, Leaver M, Wells D. Coronavirus disease‐19 and fertility: viral host entry protein expression in male and female reproductive tissues. Fertil Steril. 2020;114(1):33–43. 10.1016/j.fertnstert.2020.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Carvalho R, Groner MF, Camillo J, Ferreir PRA, Fraietta R. The interference of COVID‐19 in the male reproductive system: important questions and the future of assisted reproduction techniques. Clinics. 2020;75:1–2. 10.6061/clinics/2020/e2183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Gagliardi L, Bertacca C, Centenari C, et al. Orchiepididtmitis in a boy with COVID‐19. Pediatr Infect Dis J. 2020;39(8):e200–e202. 10.1097/INF.0000000000002769 [DOI] [PubMed] [Google Scholar]
- 37. Massarotti C, Garolla A, Maccarini E, et al. SARS‐CoV‐2 in the semen: where does it come from? Andrology. 2020. 10.1111/andr.12839. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Li R, Yin T, Fang F, et al. Potential risks of SARS‐CoV‐2 infection on reproductive health. Reprod Biomed Online. 2020;41(1):89–95. 10.1016/j.rbmo.2020.04.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ma L, Xie W, Li D, Shi L. Effect of SARS‐CoV‐2 infection upon male gonadal function: a single center‐based study. medRxiv. 2020. 10.1101/2020.03.21.20037267. In press. [DOI] [Google Scholar]


