ABSTRACT
Background
The risk of COVID‐19 and related death in people with Parkinson's disease or parkinsonism is uncertain. The aim of the study was to assess the risk of hospitalization for COVID‐19 and death in a cohort of patients with Parkinson's disease or parkinsonism compared with a control population cohort, during the epidemic bout (March–May 2020) in Bologna, northern Italy.
Methods
Participants of the ParkLink study with the clinical diagnosis of Parkinson's disease or parkinsonism and people anonymously matched (ratio 1:10) for sex, age, district, and Charlson Index were included. The hospital admission rate for COVID‐19 (February 26–May 31, 2020) and the death rate for any cause were the outcomes of interest.
Results
The ParkLink cohort included 696 subjects with Parkinson's disease and 184 with parkinsonism, and the control cohort had 8590 subjects. The 3‐month hospitalization rate for COVID‐19 was 0.6% in Parkinson's disease, 3.3% in parkinsonism, and 0.7% in controls. The adjusted hazard ratio (age, sex, district, Charlson Index) was 0.8 (95% CI, 0.3–2.3, P = 0.74) in Parkinson's disease and 3.3 (1.4–7.6, P = 0.006) in parkinsonism compared with controls. Twenty‐nine of the infected subjects died; 30‐day fatality rate was 35.1%, without difference among the 3 groups. Six of 10 Parkinson's disease/parkinsonism patients had the infection during hospitalization or in a nursing home.
Conclusions
Parkinson's disease per se probably is not a risk factor for COVID‐19 hospitalization. Conversely, parkinsonism is an independent risk factor probably because of a more severe health status, entailing higher care dependence and placement in high‐infection‐risk accommodations. © 2020 The Authors. Movement Disorders published by Wiley Periodicals LLC on behalf of International Parkinson and Movement Disorder Society
Keywords: COVID‐19, Parkinson's disease, parkinsonism, cohort studies, frail elderly
After the outbreak start of COVID‐19 in China, in December 2019, 1 the first case in Italy was recorded on February 20, 2020, 2 and soon after the epidemic spread quickly in the northern part of the country. 3 The Emilia‐Romagna region (in northeastern Italy) has been the region in Italy third hardest hit by the COVID‐19 epidemic in number of cases. During the outbreak peak, from March 1 to May 31, 2020, 27,790 cases of infection of SARS‐CoV‐2 were recorded (Italian Ministry of Health). 4 The city of Bologna, the regional capital city, counted 4636 cases in the same period (Local Health Trust data 5 ).
The risk of COVID‐19 infection and related case fatality is higher among vulnerable populations such as elderly affected by chronic conditions. 6 , 7 , 8 Persons with Parkinson's disease (PD) and parkinsonisms (PS) show a paradigmatic pattern of vulnerability, being elderly and often with multiple age‐related comorbidities, including cognitive impairment and higher polypharmacy risk. 9 Whether people with PD or PS have an increased risk of COVID‐19 and an associated higher risk of death is still debatable. 10 , 11 , 12 , 13 , 14 A telephone survey, including an outpatient clinic cohort of people with PD and their family members as controls, reported a similar prevalence of “probable” or “confirmed” COVID‐19. 11 Conversely, another telephone survey suggested a higher prevalence of COVID‐19 compared with that expected for the general population. 15 Trying to overcome the limitations of these studies, we designed a population‐based study investigating the risk for hospital admission of COVID‐19 and the risk of death in people with PD or PS.
ParkLink Bologna is a population‐based record linkage project started in 2016 in the province of the city of Bologna, Italy, including a cohort of people with a diagnosis of PD or PS confirmed by a neurologist according to international criteria. In ParkLink Bologna clinical data of individual patients are linked to administrative databases. 16
The aim of the present study was to assess the risk of hospital admission for COVID‐19 and related death in the ParkLink Bologna cohort of patients with PD or PS compared with a control population cohort during the COVID‐19 epidemic bout in the local health trust of Bologna (March to May 2020).
Methods
The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines 17 were followed.
Study Design
A historical cohort design was applied.
Setting and Study Population
The local health trust (LHT) of Bologna, in northern Italy, had a population of 886,000 (752,104 adults), on December 31, 2019. At the beginning of the outbreak, the area included 9 general hospitals, 3 academic hospitals (1 general, 1 neurologic, 1 orthopedic), 11 private clinics operating within the Italian National Health Service, for a total of about 4400 beds, of which about 120 were intensive care. In response to the COVID‐19 emergency, hospital facilities underwent major reorganization; since mid‐March, 8 COVID‐19 hospitals/wards have been created, with a total of about 400 beds for symptomatic COVID patients and about 150 COVID‐19 intensive care unit beds. 18 Dedicated diagnostic protocols for suspected COVID‐19 patients to be applied at every emergency department access were explicitly implemented by the LHT of Bologna.
This study is based on the ParkLink Bologna project (https://www.ausl.bologna.it/isnb/ricerca/parklink), an ongoing record linkage system for PD and PS approved in 2015 by the local Ethics Committee (see Statements of Ethics section). The project population includes the whole population living in the area covered by the LHT of Bologna during the period of observation, July 1, 2015–June 30, 2030. For the purpose of the present study we included people with PD or PS (ParkLink cohort) and people anonymously matched from the general population (control cohort), alive on December 31, 2019. People included in the ParkLink cohort are both prevalent and incident cases of PD or PS recruited consecutively after signing a form giving consent to link their diagnosis to their stored administrative data for any possible research purpose. Since 2015, all neurologists working in the area of the LHT of Bologna — including 3 hospital outpatient services dedicated to movement disorders and several public and private outpatient services — have been invited to enroll people with PD or PS, during clinical practice. Up to December 2019, 24 neurologists have voluntarily joined the project. The following data have been recorded in an e‐CRF and stored in a secure database linked to the other administrative databases: unique anonymous identification code, date of birth, diagnosis, year of onset, motor symptoms at onset (tremor or bradykinesia), side of onset (unilateral or bilateral), and Hoehn and Yahr score. Diagnosis of PD is defined by the recruiting neurologist according to Gelb criteria. 19 Specified PS, 20 , 21 , 22 , 23 other rare neurological causes affecting the basal ganglia, or unspecified PS are recorded in the system and labeled as parkinsonism tout‐court (PS). People with drug‐induced PS are excluded.
The control cohort consisted of people matched with the ParkLink cohort, with a ratio of 1:10 for age, sex, district of residence, and comorbidity according to the Charlson Index. 24 Subjects who used drugs for PD (levodopa, dopaminergics, monoamine oxidase‐B inhibitors) for at least 180 consecutive days during 2019 were excluded.
Data Sources
Being that the Italian health system is universal, all resident citizens are anonymously recorded in a homogenous way when accessing any public or private health facility and personal data stored by the qualified LHT in secure databases. The ParkLink Bologna project allows, for each individual, the linkage of the clinical diagnosis with administrative databases and the extraction of population‐matched controls. The Unità Operativa Epidemiologia e Statistica of the IRCCS Istituto delle Scienze Neurologiche di Bologna (part of the LHT of Bologna) is in charge of the ParkLink Bologna project and has access to several administrative databases (drug prescription, hospital discharge, copayment exemption, medical home care, mortality). The Charlson Index was based on data from the hospital discharge database, and the index was calculated according to the adapted method for International Classification of Disease‐9‐CM (ICD‐9‐CM). 25 All hospital facilities, both public and private, of the LHT of Bologna provide data on hospital discharge.
Outcomes
Hospital admission rate for COVID‐19 from February 26 to May 31, 2020, was the primary outcome. Diagnosis was obtained from hospital discharge data. The ICD‐9‐CM codes for the diagnosis of COVID‐19 were 079.82 and 480.3, according to the Emilia‐Romagna region regulation (March 10, 2020). Hospital admission rate for any reason was assessed to estimate the general trend of hospitalization in the same time window.
We assessed also the death rate for any cause from inception until June 30, 2020, as reported by the demographic database, and measured as time to event from COVID‐19 hospitalization (index date) to death or the end of follow‐up, whichever came first. When possible, qualitative assessment of hospital charts was carried out for patients with PD/PS and COVID‐19 to clarify in which circumstances COVID‐19 infection occurred.
Statistical Analysis
In the descriptive analysis, the characteristics of the 2 cohorts are presented as mean ± standard deviation (SD) for the continuous variables and with absolute (n) and relative (%) frequency for categorical variables. The chi‐square test and Kruskal‐Wallis test were used to evaluate the univariable association between conditions (PD/PS/controls) and categorical or continuous variables, respectively. To evaluate the outcomes (hospitalization for COVID‐19 and death), 2 analyses were performed. In the first one, the time to entry in the analysis was February 26, 2020, and the time to end point was the date of hospital admission or May 31, 2020. In the second analysis, the time to entry was the date of hospital admission, and the time to end point was the date of death or June 30, 2020. We used univariable and multivariable Cox regression models to estimate the hazard ratio (HR) with 95% confidence interval (95% CI) associating COVID‐19 hospitalization and death in the presence of PD/PS. Results of the univariable analysis are presented as Kaplan–Meier curves. In the multivariable models we included sex, age, Charlson Index, and district of residence as covariates to take into account the impact of the comorbidity. The proportional hazards assumption was tested (P > 0.05) using Schoenfeld residuals. Data linkage and statistical analyses were conducted using Stata SE version 14.2. 26
Results
The ParkLink cohort included 696 subjects with PD (mean age, 75.0 years) and 184 with PS (mean age, 80.5 years), followed up for 2149 and 561 person‐months, respectively. The control cohort included 8590 subjects (mean age, 76.0 years; 26,506 person‐months of follow‐up), after the exclusion of 27 subjects on treatment with drugs for PD. Demographic and clinical characteristic data are summarized by group in Table 1. People with PS were older, with higher Hoehn and Yahr scale score and with more frequent comorbidities than people with PD.
TABLE 1.
Demographic and clinical features of the control cohort and ParkLink cohort (subdivided as subjects with Parkinson's disease and parkinsonism). In bold statistically significant results.
| Demographic | Control cohort | Parkinson's disease | Parkinsonism | P |
|---|---|---|---|---|
| n | 8590 | 696 | 184 | ‐ |
| Age (y), mean (SD, range) | 76.0 (9.1, 42–97) | 75.0 (9.2, 42–95) | 80.5 (7.0, 54–97) | < 0.001 |
| Age distribution, n (%) | < 0.001 | |||
| 40–49 y | 90 (1.1) | 9 (1.3) | 0 (0) | |
| 50–59 y | 427 (5.0) | 42 (6.0) | 1 (0.5) | |
| 60–69 y | 1309 (15.2) | 119 (17.1) | 13 (7.1) | |
| 70–79 y | 3353 (39.0) | 286 (41.1) | 58 (31.5) | |
| 80–89 y | 3145 (36.6) | 227 (32.6) | 98 (53.3) | |
| ≥90 y | 266 (3.1) | 13 (1.9) | 14 (7.6) | |
| Sex, n (%) | 0.911 | |||
| Male | 5000 (58.2) | 409 (58.8) | 105 (57.1) | |
| Female | 3590 (41.8) | 287 (41.2) | 287 (41.2) | |
| District, n (%) | 0.763 | |||
| Bologna | 3676 (42.8) | 296 (42.5) | 81 (44.0) | |
| Reno | 993 (11.6) | 81 (11.6) | 21 (11.4) | |
| Pianura Est | 1657 (19.3) | 129 (18.5) | 41 (22.3) | |
| Pianura Ovest | 1024 (11.9) | 85 (12.2) | 20 (10.9) | |
| Apennino | 619 (7.2) | 57 (8.2) | 6 (3.3) | |
| San Lazzaro | 621 (7.2) | 48 (6.9) | 15 (8.1) | |
| Charlson Index, n (%) | 0.001 | |||
| 0 | 6543 (76.2) | 552 (79.3) | 116 (63.0) | |
| 1 | 897 (10.4) | 62 (8.9) | 32 (17.4) | |
| 2 | 716 (8.3) | 53 (7.6) | 20 (10.9) | |
| ≥3 | 434 (5.1) | 29 (4.2) | 16 (8.7) | |
| Comorbidities, n (%) | ||||
| Myocardial infarction | 154 (1.8) | 13 (1.9) | 3 (1.6) | 0.975 |
| Congestive heart failure | 596 (6.9) | 40 (5.7) | 22 (12.0) | 0.013 |
| Peripheral vascular disease | 69 (0.8) | 6 (0.9) | 1 (0.5) | 0.911 |
| Cerebrovascular disease | 417 (4.8) | 22 (3.2) | 21 (11.4) | < 0.001 |
| Dementia | 569 (6.6) | 35 (5.0) | 24 (13.0) | 0.001 |
| Chronic pulmonary disease | 248 (2.9) | 14 (2.0) | 12 (6.5) | 0.005 |
| Peptic ulcer disease | 46 (0.5) | 3 (0.4) | 2 (1.1) | 0.552 |
| Liver disease | 18 (0.2) | 2 (0.3) | 0 (0.0) | 0.747 |
| Diabetes | 298 (3.5) | 17 (2.4) | 14 (7.6) | 0.003 |
| Renal disease | 205 (2.4) | 16 (2.3) | 5 (2.7) | 0.947 |
| Any malignancy | 441 (5.1) | 40 (5.8) | 5 (2.7) | 0.254 |
| Clinical features at onset, n (%) | ||||
| Unilateral | 568 (81.6) | 68 (37.0) | < 0.0001 | |
| Bilateral | 128 (18.4) | 116 (63.0) | ||
| Tremor (yes) | 449 (72.8) | 74 (52.5) | < 0.0001 | |
| Bradykinesia (yes) | 479 (80.4) | 148 (89.2) | 0.002 | |
| Probability level in PD, n (%) | ||||
| Possible | 231 (33.2) | |||
| Probable | 465 (66.8) | |||
| Parkinsonian etiology in PS, n (%) | ||||
| Vascular parkinsonism | 94 (51.1) | |||
| Progressive supranuclear palsy | 14 (7.6) | |||
| Multiple system atrophy | 9 (4.9) | |||
| Other/undetermined | 67 (36.4) | |||
| Hoehn and Yahr scale at latest observation a | ||||
| Mean (SD) | 2.4 (1.0) | 3.0 (1.1) | < 0.001 | |
| Score 4–5, n (%) | 95 (13.6) | 64 (34.8) | < 0.001 | |
| Therapy, n (%) | ||||
| No therapy | 79 (11.3) | 44 (23.9) | < 0.0001 | |
| Levodopa only | 425 (61.1) | 110 (59.8) | ||
| Dopaminergic only or IMAO B only | 20 (2.9) | 3 (1.6) | ||
| Any combination of drugs | 172 (24.7) | 27 (14.7) |
36% of patients of the ParkLink cohort had the latest recording of the score before 2018.
Risk of Hospital Admission for COVID‐19
The epidemic bout is depicted in Figure 1 as crude number of hospital admissions for COVID‐19 in the whole population of the LHT of Bologna. In our cohorts, the first COVID‐19 patient was admitted on February 27, 2020. Hospital admissions for COVID‐19 were 74: 4 among PD subjects (3 men), 6 among PS subjects (3 men), and 64 in the control cohort (44 men). The 3‐month admission rates were 0.6% in PD (95% CI, 0.2%–1.5%), 3.3% (95% CI, 1.5%–7.1%) in PS, and 0.7% (95% CI, 0.6%–0.9%) in controls. Kaplan–Meier curves showed a progressively higher risk of hospitalization for PS compared with the PD group and the control group during the epidemic bout (Fig. 2). The crude HR compared with the control cohort was 0.8 (95% CI, 0.3–2.1; P = 0.61) for PD and 4.4 (95% CI, 1.9–10.2; P = 0.01) for PS. Univariable analysis also found the following factors associated with hospital admission (Table 2): age, Charlson Index, district of residence, myocardial infarction, congestive heart failure, cerebrovascular disease, dementia, diabetes, renal disease, and any malignancy. After adjustment (Table 2), HR was 0.8 (95% CI, 0.3–2.3; P = 0.74) in PD and 3.3 (95% CI, 1.4–7.6; P = 0.006) in PS. In the same period, the adjusted risk of hospital admission for any reason was slightly increased in PD subjects (HR, 1.51; 95% CI, 1.1–2.1; P = 0.011) but not in PS subjects (HR, 1.2; 95% CI, 0.7–2.2; P = 0.42). Considering only the ParkLink cohort, the univariable analysis with the Hoehn and Yahr scale score as factor found an HR for hospital admission of 1.63 (95% CI, 0.97–2.74; P = 0.064).
FIG. 1.
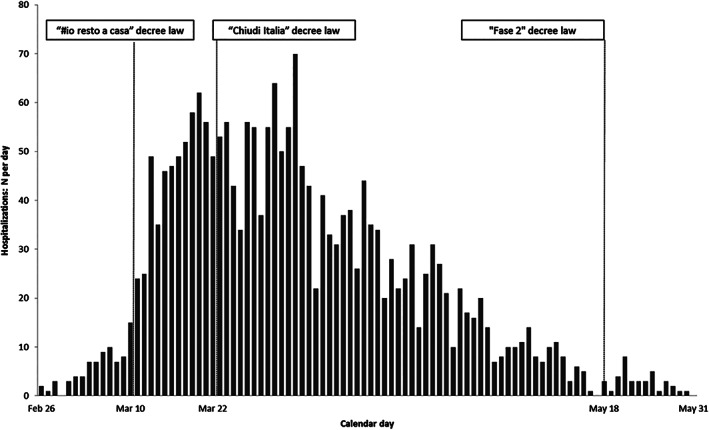
Description of the epidemic bout in the local health trust of Bologna from February 26, 2020, to May 31, 2020: crude number of hospital admissions for COVID‐19 in the whole population. The reported calendar days correspond to the Italian government emergency dispositions: March 10, 2020, “I stay home” decree law (decreto legge “#io resto a casa”); March 22, 2020, “Lock Italy down” decree law (decreto legge “Chiudi Italia”); May 18, 2020, “Phase 2” decree law (decreto legge “Fase 2,” with intraregional mobility permitted for the whole population).
FIG. 2.
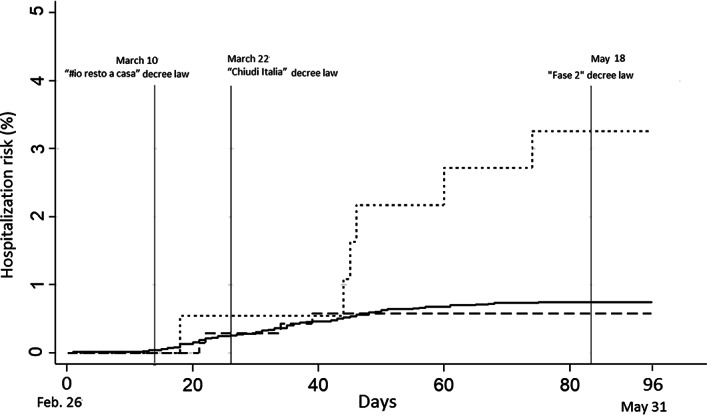
Kaplan‐Meier curves reporting the risk of hospitalization in the group of people with Parkinson's disease (dashed line), people with parkinsonism (dotted line), control cohort (solid line), during the epidemic bout, from February 26, 2020, to May 31, 2020. The reported calendar days correspond to the Italian government emergency dispositions: March 10, 2020, “I stay home” decree law (decreto legge “#io resto a casa”); March 22, 2020, “Lock Italy down” decree law (decreto legge “Chiudi Italia”); May 18, 2020, “Phase 2” decree law (decreto legge “Fase 2”, with intraregional mobility permitted for the whole population).
TABLE 2.
Risk of hospital admission for COVID‐19: univariable and multivariable Cox regression analysis. In bold statistically significant results.
| Number of hospital admission for COVID‐19 | HR (95% CI) | P | Adjusted HR (95% CI) | P | |
|---|---|---|---|---|---|
| Control | 64 | Reference | Reference | ||
| Parkinson's disease | 4 | 0.8 (0.3–2.1) | 0.614 | 0.8 (0.3–2.3) | 0.756 |
| Parkinsonism | 6 | 4.4 (1.9–10.2) | 0.001 | 3.3 (1.4–7.6) | 0.006 |
| Age (mean ± SD, 78.9 ± 8.7 y) | ‐ | 1.04 (1.01–1.07) | 0.008 | 1.02 (1.0–1.05) | 0.172 |
| Sex, n (%) | |||||
| Male | 50 (0.9) | Reference | Reference | ||
| Female | 24 (0.6) | 0.7 (0.4–1.1) | 0.103 | 0.7 (0.4–1.1) | 0.118 |
| District, n (%) | |||||
| Bologna | 38 (0.9) | Reference | Reference | ||
| Reno | 11 (1.0) | 1.1 (0.5–2.1) | 0.841 | 1.0 (0.5–2.0) | 0.981 |
| Pianura Est | 7 (0.4) | 0.4 (0.2–0.9) | 0.029 | 0.4 (0.2–1.0) | 0.041 |
| Pianura Ovest | 9 (0.8) | 0.9 (0.4–1.8) | 0.662 | 0.9 (0.4–1.8) | 0.767 |
| Apennino | 2 (0.3) | 0.3 (0.1–1.3) | 0.108 | 0.3 (0.1–1.3) | 0.122 |
| San Lazzaro | 7 (1.0) | 1.1 (0.5–2.4) | 0.833 | 1.2 (0.5–2.6) | 0.71 |
| Charlson Index, n (%) | |||||
| 0 | 30 (0.4) | Reference | Reference | ||
| 1 | 16 (1.6) | 3.9 (2.1–7.2) | < 0.001 | 3.4 (1.8–6.3) | < 0.001 |
| 2 | 18 (2.3) | 5.5 (3.1–9.9) | < 0.001 | 4.9 (2.7–8.8) | < 0.001 |
| ≥3 | 10 (2.1) | 5.1 (2.5–10.3) | < 0.001 | 4.4 (2.1–9.1) | < 0.001 |
| Comorbidities, n (%) | |||||
| Myocardial infarction | 4 (2.4) | 3.2 (1.2–8.7) | 0.025 | ||
| Congestive heart failure | 14 (2.1) | 3.1 (1.8–5.6) | < 0.001 | ||
| Peripheral vascular disease | 1 (1.3) | 1.7 (0.2–12.2) | 0.6 | ||
| Cerebrovascular disease | 13 (2.8) | 4.2 (2.3–7.7) | < 0.001 | ||
| Dementia | 11 (1.8) | 2.5 (1.3–4.7) | 0.006 | ||
| Chronic pulmonary disease | 4 (1.5) | 1.9 (0.7–5.3) | 0.204 | ||
| Peptic ulcer disease | 0 (0) | na | 1 | ||
| Liver disease | 0 (0) | na | 1 | ||
| Diabetes | 7 (2.1) | 2.9 (1.3–6.4) | 0.007 | ||
| Renal disease | 8 (3.5) | 5.0 (2.4–10.5) | <0.001 | ||
| Any malignancy | 8 (1.7) | 2.2 (1.1–4.7) | 0.03 |
Length of Hospital Stay
The overall mean length of hospital stay was 15.0 ± 11.5 days (range, 1–52 days) in controls, 29.5 ± 29.4 days in PD (range, 11–50 days), 18.7 ± 22.5 days in PS (range, 6–63 days); P = 0.31. Mean length of hospital stay among alive patients was 17.3 ± 12 days (range, 1–52 days) in controls, 35.7 ± 19.9 days in PD (range, 13–50 days), and 26.3 ± 31.8 days in PS (range, 6–63 days); P = 0.28.
Thirty‐Day Fatality Rate After Hospital Admission for COVID‐19
Overall, 29 of the infected subjects died; 30‐day fatality rate was 35.1% (95% CI, 25.5%–47.2%). Twenty‐five controls (39%) died a mean of 16 ± 14.8 days after hospital admission (range, 0–60 days): 1 PD patient (25%) on day 11; and 3 PS patients (50%), 2 on day 6 and 1 on day 21. HR for death compared with controls was 0.59 (95% CI, 0.1–4.4; P = 0.61) in PD and 1.4 (95% CI, 0.43–4.8; P = 0.55) in PS.
Clinical Features of ParkLink Cohort Subjects with COVID‐19
The 4 people with PD and COVID‐19 infection had a mean Hoehn and Yahr scale score of 2.37 ± 0.63 and showed the following comorbidities (Table 3): 2 with congestive heart failure, 1 with cerebrovascular disease, 1 with dementia, and 1 with peripheral vascular disease. One female subject had 3 comorbidities and died after 11 days of hospitalization. The 6 people with PS had a mean Hoehn and Yahr scale score of 3.67 ± 0.82 and had the following diagnoses (and comorbidities; Table 3): 3 vascular parkinsonism (3 cerebrovascular disease, 2 dementia, 1 renal disease), 1 progressive supranuclear palsy (none), 1 multiple system atrophy (cerebrovascular disease, dementia, diabetes), 1 undetermined parkinsonism (perinatal hydrocephalus, dementia). The latter 3 subjects died after hospitalization for 6 days (2 subjects) and 21 days (1 subject). In 6 patients the COVID‐19 infection occurred during hospitalization or as a guest in a nursing home. In the other 2 cases it occurred probably when attending senior citizens/day care centers.
TABLE 3.
Clinical features of ParkLink cohort subjects with COVID‐19
| Patient number | Diagnosis | Sex | Age | Comorbidities | Circumstance of COVID‐19 infection | Outcome (last observation June 30, 2020) |
|---|---|---|---|---|---|---|
| 1 | Parkinson's disease | M | 63 y | — |
Guest in a nursing home. Treated with levodopa‐carbidopa intestinal gel administered by a portable infusion pump through a percutaneous endoscopic gastrostomy with jejunal extension |
Alive |
| 2 | Parkinson's disease | M | 77 y | — | Attending a senior citizens center | Alive |
| 3 | Parkinson's disease | M | 82 y | Congestive heart failure,cerebrovascular disease | During hospitalization for other reason | Alive |
| 4 | Parkinson's disease | F | 84 y | Congestive heart failure, peripheral vascular disease,dementia | Hospitalization the days preceding the last hospital admission for COVID‐19 | Dead after 11 days of hospitalization |
| 5 | Vascular parkinsonism | M | 82 y | Cerebrovascular disease, dementia, renal disease | During hospitalization for other reason | Alive |
| 6 | Vascular parkinsonism | F | 85 y | Cerebrovascular disease |
Guest in a nursing home |
Alive |
| 7 | Vascular parkinsonism | F | 89 y | Cerebrovascular disease, dementia | Unknown | Alive |
| 8 | Progressive supranuclear palsy | M | 80 y | — | Unknown | Dead after 6 days of hospitalization |
| 9 | Multiple system atrophy | F | 77 y | Cerebrovascular disease, dementia, diabetes | Guest in a nursing home | Dead after 6 days of hospitalization |
| 10 | Other (extrapyramidal signs + congenital hydrocephalus) | M | 65 y | Dementia | Attending a senior day care center | Dead after 21 days of hospitalization |
Discussion
During the SARS‐CoV‐2 epidemic bout in Emilia‐Romagna region, Italy, we observed a 3‐month (March–May 2020) hospital admission rate for COVID‐19 of 0.6% in people with PD, not different from a control population. Conversely, people with PS showed a 3‐fold risk (admission rate of 3.3%) after adjustment for comorbidities. Both the ParkLink cohort and the control cohort included mainly elderly people, and we found that COVID‐19 risk is associated with age and number of comorbidities. Finally, the risk of death after hospitalization was high in both cohorts (around 35%). No association with PD or PS was detected, possibly because of the small number of events in these groups.
It has been hypothesized that patients with PD or PS could have a heightened risk of COVID‐19 13 , 27 because of the indirect association with old age, multiple comorbidities, and polypharmacy frequency. 9 Respiratory dysfunction, common among people with PD or PS, may represent a direct risk factor for worse respiratory complications of COVID‐19 infection. 12 , 27 Our data suggest that PD per se is not associated with a higher risk for COVID‐19 hospitalization. A case‐controlled telephonic survey by Fasano et al 11 found that patients with PD (selected from a movement disorder tertiary center in the Lombardy region of Italy) did not have a COVID‐19 risk (7.1%) different from that of family members (7.6%), defined as “probable” or “confirmed”. Conversely, another Italian study reported a higher risk of infection in a movement disorder tertiary center cohort of people with PD, making an indirect comparison of the prevalence found in the cohort with that expected in the general population. Recently, a telephone survey 28 on a population of PD patients in the Piedmont region of Italy reported a prevalence of 0.57% COVID‐19 positivity in a population of PD patients that was indirectly compared with the 0.63% prevalence of the general population. All studies were limited by the method of ascertainment of the infection (self‐reported in most cases), 1 study 11 by the controls (family members) probably sharing the same environmental risk of infection, therefore increasing the risk of recall bias, and 2 studies, 28 by the indirect comparison of the prevalence in the general population without adjusting for other factors.
Our novel observation of a higher risk of COVID‐19 hospitalization for people with PS could be explained by the following hypotheses. First, the category of PS, even if including a heterogeneous spectrum of disorders (some specific such as progressive supranuclear palsy or multiple system atrophy, other less specific “vascular parkinsonism” or “unspecified” PS), can be considered more vulnerable than PD because of a relatively more rapid disease progression, 29 , 30 , 31 , 32 , 33 , 34 reaching milestones such as dysphagia, falls, cognitive impairment, and psychiatric symptoms earlier. Second, people with PS, showing on average higher disability and being cognitively impaired more often than PD patients, have a higher probability of being admitted to nursing homes. 35 Risk of SARS‐CoV‐2 infection in Italy was higher among nursing home guests 36 , 37 ; therefore, people with PS could have had this risk heightened by being part of this population. In our study, indeed at least 6 of 10 patients were infected as guests in the hospital or in the nursing home. Moreover, patients with atypical parkinsonism may more frequently require tracheostomy, percutaneous endoscopic gastrostomy, urinary catheterization, or supplementary nutrition and/or rehydration. These procedures need regular (eg, weekly or monthly) assistance for their revision, checkup and replacement of consumables (eg, cannula, indwelling catheter, drainage bag) from specialized professional nurses and palliative care specialists. Finally, we can hypothesize that during the lockdown period people with PS had a higher risk of hospitalization for any reason; however, we did not find any difference.
Risk factors for severe outcomes in hospitalized COVID‐19 patients are reported to be chronic kidney disease, chronic obstructive pulmonary disease, cerebrovascular disease, and coronary heart disease. 38 However, risk factors for hospitalization for COVID‐19 in the general population are less extensively studied and are reported to be obesity, chronic kidney disease, diabetes, hypertension, and asthma after adjustment for age, sex, and ethnicity. 39 In our 2 cohorts of elderly people (with or without PD/PS), neurologic (cerebrovascular disease, dementia) or nonneurologic comorbidities (myocardial infarction, congestive heart failure, diabetes, renal disease, malignancies) were found to be strongly associated with worse outcomes among hospitalized patients with COVID‐19.
Case fatality after hospital admission for COVID‐19 is extremely variable and affected by many factors, 40 such as case definition, case mix of patients, geographical context, and capacity of the health care system. 41 In Italy, namely, in Lombardy during the epidemic bout, 30‐day fatality rate was 19.7% in patients admitted to a large general hospital (median age, 61 years). 42 A large population‐based study network of hospitalized patients (median age, 69 years) of northern Italy found a 30‐day case fatality rate of 27.6%. 40 In other European countries (Germany, Spain, England), 43 , 44 , 45 the 30‐day case fatality rate ranged between 14% and 27% in patients with varying mean age (63‐73 years). We found an impressive 30‐day fatality rate of 35% that could be explained by the criteria of selection of our cohorts, which included people matched to the target population of PD patients and thus aged people with a relatively higher burden of comorbidities. No difference was found between the ParkLink cohort and the control cohort, possibly because of the insufficient power to detect differences. Two studies 28 , 46 suggested a possibly higher risk of death in PD patients after COVID‐19 infection. However, serious methodological limitations in both studies (eg, PD diagnosis not on a clinical basis, 46 no adjustment for comorbidities, 28 , 46 telephonic ascertainment of COVID‐19 infection 28 ) did not allow drawing conclusions.
Evidence suggests that subjects with chronic and serious neurodegenerative diseases experienced worsening of their clinical status during the COVID‐19 epidemic. A telemedicine assessment survey on carers of 32 patients with frontotemporal dementia during the COVID‐19 emergency in Italy showed worsening in behavior, language, and cognitive function in most cases. 47 Among COVID‐19‐infected patients such impact can be explained as a possible direct cause of the infection itself, but in uninfected patients it may be indirectly because of a generalized restriction of care provision (see Ferini‐Strambi and Salsone 48 ). Indeed, people with PD/PS or with dementia require follow‐up and supportive care provided in specialized health care settings, and in many countries, including Italy, the cancellation of nonurgent outpatient appointments could have caused a drastic reduction of such dedicated care. Population‐based studies on the direct and indirect impact of the COVID‐19 epidemic on neurodegenerative diseases are needed.
The main strength of this study is having the clinical diagnosis of PD and PS performed by a neurologist according to international criteria. In a retrospective study, diagnostic assessment by a qualified neurologist is highly preferable than relying on other methods based on administrative claims data only. Second, it is a population‐based cohort study including patients with PD and PS representative of the entire spectrum of patients (not selected from a movement disorder tertiary centre), compared with a matched general population cohort. Third, the diagnosis of COVID‐19 infection is obtained from hospital discharge–coded administrative data, therefore excluding the risk of recall bias.
Our study has some limitations. The use of ICD codes to label comorbidities could have underestimated the presence of other diseases in patients with a burdensome condition such as PD or PS. In particular, people with PS could have a residual presence of comorbidities not recorded by the SDO system. However, the different risk found for PD and PS suggests that this factor could have biased the results only in a small part. Moreover, the ICD code to diagnose COVID‐19 (079.82, SARS‐associated coronavirus; and 480.3, “Pneumonia due to SARS‐associated coronavirus) has been recommended with little delay compared with the epidemic start in our region and has not been validated for COVID‐19. Nevertheless, coding errors would have the same random probability in the 2 cohorts. In addition, because of the difficulty of the health system in dealing with the epidemic bout, we cannot exclude that some COVID‐19 symptomatic patients were not hospitalized. However, because of the prompt strengthening of the local health facilities dedicated to COVID‐19, we can assume that only mild symptomatic patients have been missed. In any case, there is no reason that the hospitalization risk could be biased in favor/disfavor of one of the groups of patients included. Finally, an updated evaluation of the Hoehn and Yahr scale score was suboptimal (in 36% of the ParkLink cohort patients the last available Hoehn and Yahr evaluation was before 2018); thus, the association of this factor to hospitalization could not be reliably tested.
In conclusion, our study found that PD is not per se a risk factor for COVID‐19 hospitalization, whereas, conversely, being affected by PS is an independent risk factor. A more severe health status of these patients, entailing higher care dependence and placement in accommodations (eg, hospitals, nursing homes) with high infection risk may explain such association. These aspects should be confirmed in larger population‐based cohorts and studied for a longer period.
Statement of Ethics
The Ethics Committee of the Bologna Local Health Trust approved the ParkLink Bologna project on June 25, 2015 (reference code: 15401). All patients included in the ParkLink cohort have given their written informed consent. The persons of the control cohort are anonymous. The approved protocol of the project defines as study population all the inhabitants of the geographical area covered by the LHT of Bologna.
Author Roles
Luca Vignatelli: research project conception, organization and execution; statistical analysis design; writing the first draft of the manuscript.
Corrado Zenesini: research project conception; statistical analysis design and execution; writing the first draft of the manuscript.
Laura Maria Beatrice Belotti: statistical analysis execution; review and critique of the manuscript.
Elisa Baldin: research project conception; statistical analysis design; review and critique of the manuscript.
Giuseppe Bonavina: research project execution; review and critique of the manuscript.
Giovanna Calandra‐Buonaura: research project organization and execution; review and critique of the manuscript.
Pietro Cortelli: research project organization and execution; review and critique of the manuscript.
Carlo Descovich: research project organization; review and critique of the manuscript.
Giovanni Fabbri: research project execution; review and critique of the manuscript.
Giulia Giannini: research project execution; review and critique of the manuscript.
Maria Guarino: research project organization and execution; review and critique of the manuscript.
Roberta Pantieri: research project execution; review and critique of the manuscript.
Giuseppe Samoggia: research project execution; review and critique of the manuscript.
Cesa Scaglione: research project organization; review and critique of the manuscript.
Susanna Trombetti: research project organization; review and critique of the manuscript.
Roberto D'Alessandro: research project conception, organization and execution; review and critique of the manuscript.
Francesco Nonino: research project conception; writing the first draft of the manuscript.
Financial disclosures of all authors (for the preceding 12 months)
None.
Acknowledgments
We thank all study participants, clinicians, patients' associations, and members of the steering committee of ParkLink Bologna Project, who facilitated case recruitment and our research team, and Cecilia Baroncini for the revision of the English language. The ParkLink Bologna group includes Paolo Badiali (Associazione Iniziative Parkinsoniane Imolesi), Francesca Baschieri, Marzio Bellan, Graziano Benfenati, Lidia Bettelli, Marinella Bozzoli (Associazione Amiko Parkinson), Sabina Capellari, Sabina Cevoli, Piero de Carolis, Danilo Di Diodoro, Renata Ferrara, Anna Sandra Gabellini, Pietro Guaraldi, Fabiola Lucchi, Barbara Mostacci, Stefania Alessandra Nassetti, Gaetano Procaccianti, Rita Rinaldi, Giovanni Rizzo, Tommaso Sacquegna, Angelo Spiga (Associazione Parkinsoniani Pianura Est), Luisa Sambati, Elisa Stivanello, Antonella Tempestini, Carmelina Trocino, and Gianni Zecchi (Associazione Iniziativa Parkinsoniani).
Relevant conflicts of interest/financial disclosures: Nothing to report.
Funding agencies: The study was funded by research grants from the IRCCS Istituto delle Scienze Neurologiche di Bologna. We thank the family of Mr Virginio De Marchi for an unrestricted donation in his memory.
References
- 1. Lai CC, Wang CY, Wang YH, Hsueh SC, Ko WC, Hsueh PR. Global epidemiology of coronavirus disease 2019 (COVID‐19): disease incidence, daily cumulative index, mortality, and their association with country healthcare resources and economic status. Int J Antimicrob Agents 2020;55(4):105946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Grasselli G, Pesenti A, Cecconi M. Critical care utilization for the COVID‐19 outbreak in Lombardy, Italy: early experience and forecast during an emergency response. JAMA 2020;323(16):1545–1546. [DOI] [PubMed] [Google Scholar]
- 3. Distante C, Piscitelli P, Miani A. Covid‐19 outbreak progression in Italian regions: approaching the peak by the end of march in northern Italy and first week of April in southern Italy. Int J Environ Res Public Health 2020;17(9):3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ministero_della_Salute . https://github.com/pcm-dpc/COVID-19/blob/master/schede-riepilogative/regioni/dpc-covid19-ita-scheda-regioni-20200531.pdf. Last access October 27, 2020.
- 5. AUSL_di_Bologna . https://public.tableau.com/profile/dsp.ausl.bologna#!/vizhome/ReportCovid-19AziendaUSLBologna31_05_2020mobile/ReportCovid-19AUSLBO31_05. Last access October 27, 2020.
- 6. Onder G, Rezza G, Case‐Fatality Rate BS. Characteristics of patients dying in relation to COVID‐19 in Italy. JAMA 2020;323(18):1775–1776. [DOI] [PubMed] [Google Scholar]
- 7. Palmieri L, Vanacore N, Donfrancesco C, et al. Clinical characteristics of hospitalized individuals dying with COVID‐19 by age group in Italy. J Gerontol A Biol Sci Med Sci 2020;75(9):1796–1800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Clark A, Jit M, Warren‐Gash C, et al. Global, regional, and national estimates of the population at increased risk of severe COVID‐19 due to underlying health conditions in 2020: a modelling study. Lancet Glob Health 2020;8(8):e1003–e1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. McLean G, Hindle JV, Guthrie B, Mercer SW. Co‐morbidity and polypharmacy in Parkinson's disease: insights from a large Scottish primary care database. BMC Neurol 2017;17(1):126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Antonini A, Leta V, Teo J, Chaudhuri KR. Outcome of Parkinson's disease patients affected by COVID‐19. Mov Disord 2020;35(6):905–908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Fasano A, Cereda E, Barichella M, et al. COVID‐19 in Parkinson's disease patients living in Lombardy, Italy. Mov Disord 2020;35(7):1089–1093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Helmich RC, Bloem BR. The impact of the COVID‐19 pandemic on Parkinson's disease: hidden sorrows and emerging opportunities. J Parkinsons Dis 2020;10(2):351–354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Papa SM, Brundin P, Fung VSC, et al. Impact of the COVID‐19 pandemic on Parkinson's disease and movement disorders. Mov Disord Clin Pract 2020;35(5):711–715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Spina Tensini F, Spina Tensini T, Franklin GL, Teive HAG. May Parkinson's disease be a protective factor against CNS involvement by SARS‐CoV2? Parkinsonism Relat Disord 2020;77:20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Del Prete E, Francesconi A, Palermo G, et al. Prevalence and impact Q12 of COVID‐19 in Parkinson's disease: evidence from a multi‐center survey in Tuscany region. J Neurol 2020;1–9. 10.1007/s00415-020-10002-6 [DOI] [PMC free article] [PubMed]
- 16. Zenesini C, Baldin E, Vignatelli L, et al. Use of antidepressants and the risk of Parkinson's disease in the local health Trust of Bologna: a historical cohort study. J Neurol Sci 2019;405:116421. [DOI] [PubMed] [Google Scholar]
- 17. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet 2007;370(9596):1453–1457. [DOI] [PubMed] [Google Scholar]
- 18. Emilia‐Romagna_region . Emergenza Covid‐19. Piano per la gestione dei letti ospedalieri. Rev. 17/03/2020. https://salute.regione.emilia-romagna.it/assistenza-ospedaliera/intro2020. Last access October 27, 2020.
- 19. Gelb DJ, Oliver E, Gilman S. Diagnostic criteria for Parkinson disease. Arch Neurol 1999;56(1):33–39. [DOI] [PubMed] [Google Scholar]
- 20. Gilman S, Wenning GK, Low PA, et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 2008;71(9):670–676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Litvan I, Agid Y, Calne D, et al. Clinical research criteria for the diagnosis of progressive supranuclear palsy (Steele‐Richardson‐Olszewski syndrome): report of the NINDS‐SPSP international workshop. Neurology 1996;47(1):1–9. [DOI] [PubMed] [Google Scholar]
- 22. Armstrong MJ, Litvan I, Lang AE, et al. Criteria for the diagnosis of corticobasal degeneration. Neurology 2013;80(5):496–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Zijlmans JC, Daniel SE, Hughes AJ, Révész T, Lees AJ. Clinicopathological investigation of vascular parkinsonism, including clinical criteria for diagnosis. Mov Disord 2004;19(6):630–640. [DOI] [PubMed] [Google Scholar]
- 24. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987;40(5):373–383. [DOI] [PubMed] [Google Scholar]
- 25. Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD‐9‐CM administrative databases. J Clin Epidemiol 1992;45(6):613–619. [DOI] [PubMed] [Google Scholar]
- 26. StataCorp . Stata Statistical Software. In: 14 R, ed. College Station, TX: StataCorp LP; 2015.
- 27. Bhidayasiri R, Virameteekul S, Kim JM, Pal PK, Chung SJ. COVID‐19: an early review of its global impact and considerations for Parkinson's disease patient care. J Mov Disord 2020;13(2):105–114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Artusi CA, Romagnolo A, Imbalzano G, et al. COVID‐19 in Parkinson's disease: report on prevalence and outcome. Parkinsonism Relat Disord 2020;80:7–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Savica R, Grossardt BR, Bower JH, et al. Survival and causes of death among people with clinically diagnosed synucleinopathies with parkinsonism: a population‐based study. JAMA Neurol 2017;74(7):839–846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Glasmacher SA, Leigh PN, Saha RA. Predictors of survival in progressive supranuclear palsy and multiple system atrophy: a systematic review and meta‐analysis. J Neurol Neurosurg Psychiatry 2017;88(5):402–411. [DOI] [PubMed] [Google Scholar]
- 31. Glass PG, Lees AJ, Bacellar A, Zijlmans J, Katzenschlager R, Silveira‐Moriyama L. The clinical features of pathologically confirmed vascular parkinsonism. J Neurol Neurosurg Psychiatry 2012;83(10):1027–1029. [DOI] [PubMed] [Google Scholar]
- 32. Wiblin L, Lee M, Burn D. Palliative care and its emerging role in multiple system atrophy and progressive supranuclear palsy. Parkinsonism Relat Disord 2017;34:7–14. [DOI] [PubMed] [Google Scholar]
- 33. O'Sullivan SS, Massey LA, Williams DR, et al. Clinical outcomes of progressive supranuclear palsy and multiple system atrophy. Brain 2008;131(Pt 5):1362–1372. [DOI] [PubMed] [Google Scholar]
- 34. Jecmenica‐Lukic M, Petrovic IN, Pekmezovic T, Kostic VS. Clinical outcomes of two main variants of progressive supranuclear palsy and multiple system atrophy: a prospective natural history study. J Neurol 2014;261(8):1575–1583. [DOI] [PubMed] [Google Scholar]
- 35. Hosking A, Hommel A, Lorenzl S, et al; for the Care of Late Stage Parkinsonism (CLaSP) Consortium. Characteristics of patients with late‐stage parkinsonism who are nursing home residents compared with those living at home. J Am Med Dir Assoc 2020;S1525‐8610(20)30536‐3. 10.1016/j.jamda.2020.06.025. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 36. Trabucchi M, De Leo D. Nursing homes or besieged castles: COVID‐19 in northern Italy. Lancet Psychiatry 2020;7(5):387–388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Danis K, Fonteneau L, Georges S, et al. High impact of COVID‐19 in long‐term care facilities, suggestion for monitoring in the EU/EEA, May 2020. Euro Surveill 2020;25(22):2000956. 10.2807/1560-7917.ES.2020.25.22.2000956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Fang X, Li S, Yu H, et al. Epidemiological, comorbidity factors with severity and prognosis of COVID‐19: a systematic review and meta‐analysis. Aging 2020;12(13):12493–12503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ko JY, Danielson ML, Town M, et al. Risk factors for COVID‐19‐associated hospitalization: COVID‐19‐associated hospitalization surveillance network and behavioral risk factor surveillance system. medRxiv 2020. 10.1101/2020.07.27.20161810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Giorgi Rossi P, Ferroni E, Alegiani S, et al. Survival of hospitalized COVID‐19 patients in Northern Italy: a population‐based cohort study by the ITA‐COVID19 Network2020. medRxiv 2020. 10.1101/2020.05.15.20103119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Rajgor DD, Lee MH, Archuleta S, Bagdasarian N, Quek SC. The many estimates of the COVID‐19 case fatality rate. Lancet Infect Dis 2020;20(7):776–777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Giacomelli A, Ridolfo AL, Milazzo L, et al. 30‐day mortality in patients hospitalized with COVID‐19 during the first wave of the Italian epidemic: a prospective cohort study. Pharmacol Res 2020;158:104931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Nachtigall I, Lenga P, Jóźwiak K, et al. Clinical course and factors associated with outcomes among 1904 patients hospitalized with COVID‐19 in Germany: an observational study. Clin Microbiol Infect 2020;S1198‐743X(20)30493‐6. 10.1016/j.cmi.2020.08.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Giesen C, Diez‐Izquierdo L, Saa‐Requejo CM, et al. Epidemiological characteristics of the COVID‐19 outbreak in a secondary hospital in Spain. Am J Infect Control 2020;S0196‐6553(20)30700‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Khalil K, Agbontaen K, McNally D, et al. Clinical characteristics and 28‐day mortality of medical patients admitted with COVID‐19 to a Central London teaching hospital. J Infect 2020;81(3):e85–e89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Zhang Q, Schultz JL, Aldridge GM, Simmering JE, Narayanan NS. Coronavirus disease 2019 Case Fatality and Parkinson's disease. Mov Disord 2020;35(11):1914–1915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Capozzo R, Zoccolella S, Frisullo ME, et al. Telemedicine for delivery of care in frontotemporal lobar degeneration during COVID‐19 pandemic: results from southern Italy. J Alzheimers Dis 2020;76(2):481–489. [DOI] [PubMed] [Google Scholar]
- 48. Ferini‐Strambi L, Salsone M. COVID‐19 and neurological disorders: are neurodegenerative or neuroimmunological diseases more vulnerable? J Neurol 2020;1–11. 10.1007/s00415-020-10070-8 [DOI] [PMC free article] [PubMed] [Google Scholar]


