Abstract
Cholestasis is a clinical condition resulting from the imapairment of bile flow. This condition could be caused by defects of the hepatocytes, which are responsible for the complex process of bile formation and secretion, and/or caused by defects in the secretory machinery of cholangiocytes. Several mutations and pathways that lead to cholestasis have been described. Progressive familial intrahepatic cholestasis (PFIC) is a group of rare diseases caused by autosomal recessive mutations in the genes that encode proteins expressed mainly in the apical membrane of the hepatocytes. PFIC 1, also known as Byler’s disease, is caused by mutations of the ATP8B1 gene, which encodes the familial intrahepatic cholestasis 1 protein. PFIC 2 is characterized by the downregulation or absence of functional bile salt export pump (BSEP) expression via variations in the ABCB11 gene. Mutations of the ABCB4 gene result in lower expression of the multidrug resistance class 3 glycoprotein, leading to the third type of PFIC. Newer variations of this disease have been described. Loss of function of the tight junction protein 2 protein results in PFIC 4, while mutations of the NR1H4 gene, which encodes farnesoid X receptor, an important transcription factor for bile formation, cause PFIC 5. A recently described type of PFIC is associated with a mutation in the MYO5B gene, important for the trafficking of BSEP and hepatocyte membrane polarization. In this review, we provide a brief overview of the molecular mechanisms and clinical features associated with each type of PFIC based on peer reviewed journals published between 1993 and 2020.
Keywords: Progressive familial intrahepatic cholestasis, ATP8B1/familial intrahepatic cholestasis 1, ABCB11/bile salt export pump, ABCB4/multidrug resistance class 3, Intrahepatic cholestasis, Bile
Core Tip: Progressive familial intrahepatic cholestasis (PFIC) is an expanding group of genetically-based diseases caused by various autosomal recessive mutations which result in the inability to appropriately form and excrete bile from hepatocytes. There are three classic types of PFIC. Type 1 is caused by mutations of the ATP8B1 gene, encoding familial intrahepatic cholestasis 1. PFIC 2 is the result of mutations in the ABCB11 gene, reducing the apical membrane expression of bile salt export pump. PFIC 3 is caused by variations in the gene that encodes the multidrug resistance class 3 glycoprotein. With advances in DNA sequencing, new forms of this disease have been documented. Alterations in the NR1H4, tight junction protein 2, and MYO5B genes have been linked with new phenotypes of PFIC. Here, we provide a molecular and clinical overview of PFIC.
INTRODUCTION
Among the liver functions, bile production is one of the most important. Hepatocytes and cholangiocytes work together in order to produce a mixture of organic and inorganic compounds that aid in the digestion process and excrete metabolites[1]. The formation of the final product, bile, is divided into two main processes: The formation and conjugation of the bile acids by the hepatocytes and the alkalization and dilution by the cholangiocytes. The bile can be stored in the gallbladder and secreted into the duodenum after a meal by a cholecystokinin stimulus. The detergent properties of bile give it the ability to break lipids into small particles, helping digestion[1].
The impairment of bile flow is responsible for generating a clinical condition called cholestasis[2]. Alterations in the bile formation process, at the hepatocyte and/or cholangiocyte level, lead to some complications, potentially resulting in liver tumors or necessitating liver transplantation (LT)[2]. Several point mutations and pathways that contribute to this clinical condition have been elucidated. In hepatocytes, mutations and polymorphisms of the genes responsible for bile acid transporters, as well as farnesoid X receptor (FXR), an important transcription factor for the bile formation, were described[3,4]. In cholangiocytes, defects of the cystic fibrosis transmembrane conductance regulator (CFTR) and alterations in calcium (Ca2+) signaling contribute to cholestasis establishment and development[5,6].
Nowadays, several cholestasis-related diseases have been described. At the biliary level, there are two such diseases: Primary sclerosing cholangitis and primary biliary cirrhosis. These diseases both result in nonfunctional cholangiocytes, leading to viscous bile[7-9]. Problems in the hepatocyte machinery are also related to cholestatic diseases. Through an unclear genetic, hormonal, and environmental mechanism, some women in late term pregnancy develop intrahepatic cholestasis due to the downregulation of some bile transporters of the hepatocyte membrane[10]. Some drugs, such as antimicrobials or acetaminophen, may also induce the impairment of bile flow through hepatocellular damage, resulting in cholestasis[11,12].
Some autosomal recessive mutations of the bile acid membrane transporters that affect hepatocytes result in a group of diseases known as progressive familial intrahepatic cholestasis (PFIC). Historically, three types of PFIC were described: Type 1 due to mutations of the ATP8B1 gene, type 2 resulting from mutations of the ABCB11 gene, and type 3 caused by variations in the ABCB4 gene. Recently, new types of this disease have been reported. Here, we will discuss the recent molecular and clinical findings of not only PFIC 1, PFIC 2 and PFIC 3, but also of the new types that have recently been reported. This group of hereditary diseases causes severe cholestasis in infants and patients regularly have to undergo LT (Table 1).
Table 1.
Overview of progressive familial intrahepatic cholestasis subtypes
|
|
PFIC 1
|
PFIC 2
|
PFIC 3
|
PFIC 4
|
PFIC 5
|
PFICAssociated with MYO5B defects
|
| Gene | ATP8B1 | ABCB11 | ABCB4 | TJP2 | NR1H4 | MYO5B |
| Molecular findings | Impaired hepatocyte bile salt secretion; canalicular cholestasis; giant cell transformation; portal and lobular fibrosis; lack of ductular proliferation | Increased intracellular bile salt concentrations; canalicular cholestasis; lobular and portal fibrosis; hepatocellular necrosis | Absence of PC from the biliary canaliculi; increased free bile salts in canaliculi; cholangiocyte damage; increased cholesterol crystallization nonspecific portal inflammation; portal fibrosis; giant cell hepatitis | Abnormal CLDN1 localization; normal CLDN2 localization; compromised tight junctions; bile salt leakage into the paracellular space; hepatocyte and cholangiocyte damage; giant cell transformation | Undetectable expression of BSEP in the bile canaliculi; intralobular cholestasis with ductular reaction; hepatocellular ballooning; giant cell transformation | Possibly mislocalized BSEP and MDR3 upon staining; portal and lobular fibrosis; giant cell transformation. Intestinal MVID findings (mislocalized apical brush border proteins, villus atrophy, presence of microvillus inclusion bodies) |
| Clinical findings | Low GGT cholestasis; jaundice caused by hyperbilirubinemia; pruritus; hepatosplenomegaly; diarrhea; stunted growth; exocrine pancreatic insufficiency; progressive sensorineural hearing loss; fat-soluble vitamin deficiencies; can lead to cirrhosis and end stage liver disease | Low GGT cholestasis; elevated transaminases; elevated serum bilirubin; elevated AFP; jaundice; pruritus; scleral icterus; hepatomegaly; chronic skin picking; reduced growth | Elevated GGT cholestasis; pruritus; hepatosplenomegaly; variceal bleeding; portal hypertension; jaundice; acholic stools; stunted growth; reduced bone density; learning disabilities; elevated transaminases; elevated bilirubin; elevated AP | Low GGT cholestasis; neurological and respiratory symptoms | Neonatal onset of normal GGT associated cholestasis; elevated serum bilirubin; elevated serum AFP; vitamin K independent coagulopathy; fibrosis progressing into micronodular cirrhosis | Normal GGT cholestasis; jaundice; pruritus; mildly elevated ALT and AST; elevated serum BS; hepatomegaly |
| Clinical outcomes | Medical management is the first line of defense, but if it is insufficient, surgical is next. Bilary diversion has been effective in around 80% of patients. The last resort is LT in patients who develop cirrhosis and liver failure. Extrahepatic symptoms can persist (or worsen) after LT | There is a 15% chance of cirrhosis developing into HCC or cholangiocarcinoma. Genetic defect(s) can aid in determination of success of biliary diversion. LT in PFIC 2 patients might lead to development of BSEP specific allo-reactive antibodies (in approximately 8% of PFIC 2 patients who undergo LT). In these cases, a second LT may be needed if BSEP deficiency develops in the new liver | HCC and cholangiocarcinoma have been associated with PFIC 3. The severity varies, with those retaining MDR3 expression responding better to medical treatment. Biliary diversion procedures aren’t as effective due to severity upon presentation, but LT is curative | There have been some reports of HCC occurrence in PFIC 4 patients, though not much is known about the mechanism. LT has been successful, with no reported recurrence of the PFIC 4 phenotype after LT | PFIC 5 is very rare (only 8 cases reported in the literature so far). LT has been used, but steatosis in the transplanted liver has been reported in some cases | MVID has been associated with MYO5B defects. Lifelong TPN is required but is associated with increased risk of sepsis and small bowel transplant. Combined bowel-liver transplants may reduce the risk of post transplant onset of cholestasis |
| Treatments | Medical: Vitamin supplementation; UDCA; Rifampin; Cholestyramine; CFTR folding correctors | Medical: 4PBA. Surgical: Biliary diversion procedures; LT (might need increased immunosuppression) | Medical: UDCA; Rifampin. Surgical: Biliary diversion; LT | Surgical: LT | Medical: UDCA; Rifampin; ASBT inhibitors; OCA. Surgical: LT | Medical: UDCA; rifampin; Cholestyramine. Surgical: PEBD; isolated or Combined liver-bowel transplant |
PC: Phosphatidylcholine; CLDN1: Claudin-1; CLDN2: Claudin-2; BSEP: Bile salt export pump; MDR3: Multidrug resistance class 3 glycoprotein; MVID: Microvillus inclusion disease; GGT: Gamma-glutamyl transferase; AFP: Alpha-fetoprotein; AP: Alkaline phosphatase; ALT: Alanine transaminase; AST: Aspartate transaminase; BS: Bile salt; HCC: Hepatocellular carcinoma; MYO5B: Myosin VB; TPN: Total parenteral nutrition; UDCA: Ursodeoxycholic acid; CFTR: Cystic fibrosis transmembrane conductance regulator; 4PBA: 4-phenylbutyrate; ASBT: Apical sodium-dependent bile acid transporter; OCA: Obeticholic acid; PEBD: Partial external biliary diversion; PIBD: Partial internal biliary diversion; IE: Ileal exclusion; LT: Liver transplant; PFIC: Progressive Familial Intrahepatic Cholestasis; TJP2: Tight junction protein-2.
BILE FORMATION
Prior to describing the molecular and clinical features of PFIC, it is important to comprehend the bile formation process in order to understand how complex it is and how alterations could lead not only to hepatic disease, but also to dermatologic and neurologic symptoms.
The main primary bile acids produced by the human liver are cholic acid (CA) and chenodeoxycholic acid (CDCA), generated by cholesterol metabolism[13]. In the hepatocytes, these compounds are conjugated with taurine and glycine in order to form bile salts, increasing their solubility. After the bile is secreted into the duodenum, the bile salts are reabsorbed by the brush border membrane of the terminal ileum. These reabsorbed bile salts return to the liver through portal blood circulation. A small amount of bile salts are lost in the feces but are replaced by de novo synthesis in the liver. Daily, the human liver produces around 200 to 600 mg of bile acids, replenishing the bile acid pool in the enterohepatic circulation[13].
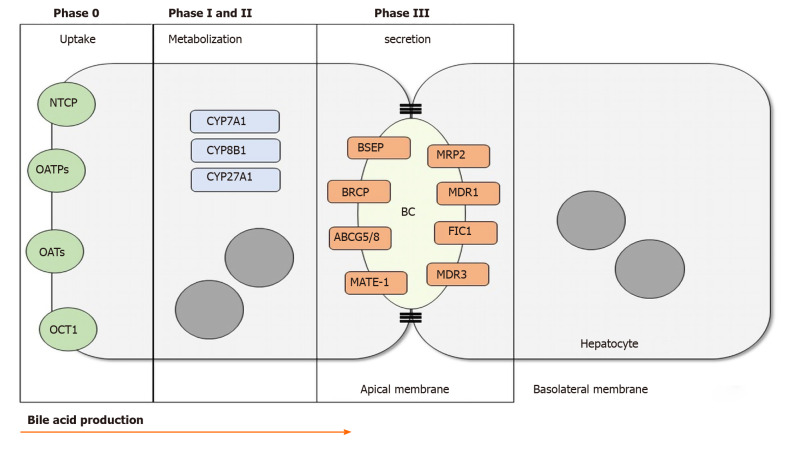
The hepatocellular part of bile formation can be divided into four stages. Stage 0 is characterized by the uptake of the compounds that will be processed to form the bile. This occurs at the basolateral hepatocyte membrane. Stage I is characterized by the activity of cytochrome P450 which turns the lipid compounds into soluble substrates for the next stages. Stage II is marked by the reactions that conjugate the bile acids. Finally, stage III is when the bile salts are secreted into the biliary canaliculus, the virtual space between two hepatocytes[1,13]. There are several different proteins and enzymes related to each stage, as shown in Figure 1.
Figure 1.
Bile acid production. In phase 0, bile acids and other organic products are transported through the basolateral membrane of the hepatocyte. Phase I and II take place in the cytoplasm through activation and metabolization of differents CYPs. In phase III the bile acids are secreted into the biliary canalicus by the transmebrane transporters. NTCP: Na+-taurocholate cotransporting polypeptide; OATPs: Organic anion transporting polypeptides; OATs: Organic anion transporter; OCT1: Organic cation transporter 1; CYP7A1: Cytochrome P450 Family 7 Subfamily A Member 1; CYP8B1: Cytochrome P450 Family 8 Subfamily b Member 1; CYP27A1: Cytochrome P450 Family 27 Subfamily A Member 1; BSEP: Bile salt export pump; BRCP: Breast cancer resistance protein; ABCG5/8: ATP-binding cassette sub-family G member 5/8; MATE-1: Multidrug and toxin extrusion 1; MRP2: Multidrug resistance-associated protein 2; MDR1: Multidrug resistance class 1 glycoprotein; FIC1: Familial intrahepatic cholestasis 1; MDR3: Multidrug resistance class 3 glycoprotein; BC: Biliary canaliculus.
The two main pathways to produce bile acids involve 17 different enzymes located in the cytoplasm, endoplasmic reticulum, mitochondria, and peroxisomes[13]. In the classic pathway, also known as the neutral pathway, cholesterol is first metabolized by CYP7A1, generating CDCA. In the presence of CYP8B1, CDCA is converted into CA. The alternative pathway, known as the acid pathway, begins with cholesterol side chain metabolism by CYP27A1, which is expressed in the mitochondrial membrane[14]. The alternative pathway is important for bile acid production in neonates as well as in diseased livers.
After bile acid synthesis and conjugation with the amino acids taurine and glycine, the bile salts are secreted into the canalicular region, a major step for bile flow. Most of the transporters belong to the ATP-binding cassette transporters (ABC transporters) superfamily[1]. As shown in Figure 1, there are several proteins involved in these steps and each one is responsible for transporting one compound in order to form the final product, the bile. Bile salt export pump (BSEP), multidrug resistance class 1 (MDR1), MDR3, MRP2, Breast Cancer Resistance Protein, sterolins 1 and 2, familial intrahepatic cholestasis 1 (FIC1) and others are examples of these transporters.
After the bile salts are secreted into the canalicular space, the cholangiocytes are responsible for alkalinizing and diluting the bile by secreting bicarbonate (HCO3-) and water[1,15,16]. This process is initiated by the stimulation of secretin, a hormone produced in the S cells of the duodenum in response to the change in pH and presence of peptides after a meal. When this hormone binds to its receptor at the cholangiocyte basolateral membrane, it leads to a cytosolic increase in cAMP which activates CFTR, thereby releasing chloride (Cl-) into the bile duct lumen. This Cl- is exchanged with HCO3-, mediated by the chloride-bicarbonate exchanger which is expressed on the apical membrane of cholangiocytes[15]. The release of HCO3- is enhanced by local increases in Ca2+ signaling that activate the M3 muscarinic receptor. When acetylcholine binds to the M3 muscarinic receptor, InsP3, a second intracellular messenger, is produced. This InsP3 mediates the release of a Ca2+ signal via the type 3 inositol 1,4,5-trisphosphate receptor. The Ca2+ release near the apical membrane activates the Ca2+-dependent chloride channel, which releases more Cl- into the luminal space to be exchanged by the chloride-bicarbonate exchanger[6]. The activation of purinergic receptors also increases bicarbonate secretion, resulting in a pH increase. The presence of bicarbonate in the luminal space creates an osmotic gradient that is important for the flux of water from the cholangiocyte cytosol to the extracellular space. This transport is mediated by small integral membrane proteins known as aquaporins. The water makes the bile less viscous and leads the bile to the gallbladder, where it will be stored until a cholecystokinin stimulus occurs after a meal[1,15,16].
Mutations in genes that encode proteins expressed in the hepatocyte canalicular membrane and involved in Phase III of bile formation are responsible for the development of a cholestatic disease known as PFIC. Now, we will provide a molecular and clinical overview of the three classic types of PFIC and discuss the newer types of this disease.
PFIC 1
PFIC type 1, also known as Byler’s disease, is caused by homozygous or compound heterozygous mutations of the ATP8B1 gene on chromosome 18 (18q21), which encodes the FIC1 protein[17]. FIC1 is part of the type 4 subfamily of P-type adenosine triphosphatases, which are involved in translocating phospholipids in membranes[18]. This protein, present at the apical membrane of hepatocytes, is thought to function as an aminophospholipid translocase, carrying phospholipids, specifically phosphatidylserine (PS) and phosphatidylethanolamine (PE), from the outside, ectoplasmic leaflet of the canalicular membrane, to the inside, cytoplasmic leaflet in hepatocytes[17-19]. When functioning normally, FIC1 protects the hepatocyte from high bile salt concentrations[17]. By maintaining the asymmetry of the plasma membrane, FIC1 guards the canalicular membrane from the detergent nature of hydrophobic bile salts present in the canalicular lumen[17,18].
FIC1 was found to be successful at flopping phospholipids only when it was coexpressed with CDC50 proteins, specifically CDC50A[18]. This coexpression allows the FIC1-CDC50A heterodimer to leave the endoplasmic reticulum and localize to the apical membrane[18]. Mutations in CDC50A may also lead to the PFIC 1 phenotype if FIC1 fails to localize as a result[18]. In CDC50A-depleted intestinal cells, not only the apical membrane expression of FIC1 is reduced, but also the expression of the Solute Carrier Family 10 Member 2 (SLC10A2) also known as apical sodium-dependent bile acid transporter (ASBT)[20]. This transporter is responsible for the reuptake of bile acids in the intestines, so mutations in either the ATP8B1 gene or the CDC50A gene will impair localization of ASBT, reducing bile uptake[20]. This suggests that the FIC1-CDC50A heterodimer is involved in the localization of SLC10A2 to the apical membrane, resulting in bile salt malabsorption, which may be the cause of the diarrhea that PFIC 1 patients experience[20].
There is also evidence that FIC1 is involved in the organization of the apical membrane of polarized cells[21]. In ATP8B1-deficient intestinal Caco-2 cells, the apical actin cytoskeleton was found to be disorganized, there was a loss of microvilli, and the translation of apical membrane protein mRNA was disrupted[21]. The reduced microvilli in the ATP8B1-deficient intestinal cells mirrors the loss of stereocilia on the apical membrane of inner hair cells in mice with an ATP8B1 mutation, which is linked to hearing loss, an event that is also observed in humans[21,22]. These examples support the case for the function of FIC1 in forming or stabilizing microvillar structures throughout the body[21].
In the disease phenotype, the lipid flipping process does not occur, interfering with the secretion of bile salt from the hepatocytes[23]. Lower expression of FIC1 downregulates FXR expression, encoded by the NR1H4 gene[17]. The function of FXR is two-fold. In hepatocytes, elevated bile salts concentrations activate FXR, which induces the expression of the BSEP[17,19]. BSEP allows for the pumping of bile salts out of the hepatocyte into the canalicular lumen[17,19]. Additionally, when expressed in the intestines, FXR represses the expression of ASBT. By doing so, bile salt reabsorption is reduced in the intestines. The bile salt transport defect is secondary in the case of the ATP8B1 mutation. The accumulation of bile salts inside the hepatocytes, coupled with the absence of the protective lipid flipping by FIC1 has a cytotoxic effect, harming the cells and leading to the cholestatic phenotype[17,19].
With the lack of lipid flipping, PS is freely present on the outer leaflet of the canalicular membrane[23]. This gives hydrophobic bile salts that exist in the canalicular space the opportunity to extract the PS from the membrane[23]. In normal hepatocytes, FIC1’s lipid flipping activity is necessary in maintaining the asymmetric and liquid ordered state of the outer leaflet of the canalicular membrane[23]. In diseased hepatocytes with an ATP8B1 mutation, the liquid ordered state becomes disordered because FIC1 is not able to flip the excess aminophospholipids from the outer to the inner leaflet[23]. This makes the membrane more susceptible to phospholipid, cholesterol, and ectoenzyme extraction[23]. Reduced cholesterol presence in the outer leaflet is vital in the functioning of certain integral membrane proteins such as BSEP (implicated in PFIC 2) and MRP1[23].
Each specific mutation comes with its own presentation of cholestatic disease. The G308V mutation was first identified in the Amish population and in homozygotes with this mutation, the FIC1 protein is undetectable in the liver[24]. The D554N mutation was found in Greenland familial cholestasis and also resulted in reduced expression of FIC1[24]. The G1040R mutation, first found in two Saudi families, did not result in a significantly decreased FIC1 expression when compared to the wild-type[24]. As expected, the interaction between FIC1 and CDC50A was undetectable in both the G308V and D554N mutations[24]. It was, however, still present in the G1040R mutation, although at a much lower level than the wild-type[24]. This is likely the reason why these three FIC1 mutant proteins are not localized to the canalicular membrane in WIF-B9 cell models[24].
Clinically, PFIC 1 presents with low gamma-glutamyl transferase (GGT) cholestasis[19]. Recurrent episodes of jaundice caused by hyperbilirubinemia, along with uncontrollable itching, or pruritus also occur[19]. Transaminases, as well as serum bile acids are elevated[19]. In infants with this disease, jaundice with pruritus, as well as hepatosplenomegaly develop early in life[19]. In severe cases, a progressive cholestasis presents along with portal hypertension[19]. Histologically, canalicular cholestasis, giant cell transformation, portal and lobular fibrosis, as well as lack of ductular proliferation are most common[17,19]. Because FIC1 is also expressed in cholangiocytes, enterocytes, and in the pancreas, there are extrahepatic manifestations of PFIC 1, setting it apart from the other types of intrahepatic cholestasis[17,19]. Some of these symptoms include diarrhea, stunted growth, exocrine pancreatic insufficiency, and progressive sensorineural hearing loss[17,19]. Nutritional deficiencies are also common, especially in fat-soluble vitamins A, D, E, and K[19].
Children with PFIC 1 often require vitamin supplementation, as well as other dietary aids in order to manage malnutrition[19]. Ursodeoxycholic acid (UDCA) is the first-line of medication used to treat pruritus[19]. It is a hydrophilic bile acid that is hypothesized to induce the expression of BSEP and MDR3, improving cholestatic symptoms[19]. Rifampin and cholestyramine have also been used in order to reduce pruritus, but have not been as effective as UDCA[19]. In addition, CFTR folding correctors may be a promising future therapy to enhance the trafficking of FIC1 in the hepatocytes[19].
In cases where medical management is inadequate, surgery is the next option. The purpose of these surgeries is to bypass the enterohepatic circulation, thereby lowering the amount of bile salts that are reabsorbed by the digestive system. Partial external biliary diversion (PEBD), partial internal biliary diversion (PIBD), and ileal exclusion (IE) have led to improvement in some cases[19]. PEBD has been shown to improve liver function, reduce serum bile acid levels, and slow progression of liver fibrosis in 80% of patients with either PFIC 1 or PFIC 2[25]. In this procedure, an external stomal conduit is created to divert bile flow outside of the body[19]. PIBD diverts bile flow from the gall bladder to the colon in order to bypass the terminal ileum, where bile salt reabsorption occurs[19]. This procedure has been shown to improve pruritus, as well as decrease bilirubin and plasma bile acid levels[19]. IE bypasses the distal 15% of the ileum to prevent bile acid reabsorption[19]. It provides relief from pruritus and may be used as a bridge to transplant[26]. Ileal adaptation may occur, reducing the effectiveness of this surgery[25].
When no other treatment is successful, liver transplant is the final option. Lobular fibrosis may develop into cirrhosis and end stage liver disease[19]. There is no known association between FIC1 deficiency and tumorigenesis[19]. At this point, LT is indicated, which will improve cholestatic symptoms[19]. However, since FIC1 is expressed outside of the liver, extrahepatic symptoms may persist, or worsen[19]. Refractory diarrhea, coupled with the development of fatty liver changes in the allograft and fibrosis, may lead to re-transplant[27]. Ileal diversion during transplant may be used to potentially prevent these developments[19].
PFIC 2
Homozygous or compound heterozygous mutations in the ABCB11 gene (chromosome 2q31), which encodes the BSEP, results in PFIC 2[17]. BSEP is an adenosine triphosphate-binding cassette transporter involved in actively transporting bile salts out of hepatocytes into biliary canaliculi[17]. It is crucial in maintaining enterohepatic circulation of bile salts[17]. Mutations in BSEP interrupt the process of pumping bile out of hepatocytes, leading to increased intracellular bile salt concentrations which consequently damage the hepatocytes[17].
FXR, activated in a heterodimer with retinoid X receptor α (RXRα), has been linked to upregulating enzymes and bile salt transporters when bound to bile salts in the cell[28]. BSEP is one of these bile salt transporters that FXR activates[28]. CDCA and the FXR/RXRα heterodimer collectively work together to transactivate the ABCB11 promoter[28]. The level of activation of the ABCB11 gene is dependent on the concentration of CDCA[28]. When more bile acids are present in the hepatocyte, more BSEP is made to maintain the equilibrium.
There are over 200 mutations that have been linked with causing PFIC 2[19]. Some of these mutations cause splicing defects while most cause protein processing defects[29]. The majority of missense mutations resulted in a reduction of BSEP levels in vitro[29]. Some common mutations — G238V, D482G, G982R, R1153C, R1286Q, and ΔGly — cause the mutant protein to be retained in the endoplasmic reticulum[30]. Proteosomes are the main cellular component that breaks down these mutant proteins, but the D482G mutant protein can also be broken down by lysosomes[30]. The degradation occurs through Endoplasmic Reticulum Associated Degradation, part of the quality control system of the endoplasmic reticulum[30]. This process involves ubiquitination, retro-translocation out of the endoplasmic reticulum, and degradation by the proteasome[30].
PFIC 2 presents with low GGT cholestasis as well as highly elevated transaminases, serum bilirubin, and alpha-fetoprotein (AFP) levels[17,19]. Jaundice, pruritus, scleral icterus, hepatomegaly, chronic skin picking, and reduced growth are also common[17,19]. Liver histology shows canalicular cholestasis, lobular and portal fibrosis, and hepatocellular necrosis[17]. Unlike PFIC 1, there is up to a 15% chance of cirrhosis developing into either hepatocellular carcinoma (HCC) or cholangiocarcinoma in patients with PFIC 2[19].
Treatment of PFIC 2 is similar to PFIC 1. A promising therapy includes treatment with cell surface BSEP-enhancer molecules such as 4-phenylbutyrate (4PBA)[19]. 4PBA has been shown to improve liver function and pruritus in patients with certain mutations of BSEP[19]. Bile salt excretion was improved, although not completely[31]. 4PBA helps increase cell surface expression of the mutant BSEPs, likely altering a post-transcriptional mechanism[32]. This therapy works best when the mutation results in normal transport ability, but impaired membrane trafficking[32,33]. Biliary diversion procedures have a better response in cases where there is still some BSEP function than in cases where function is nonexistent[19].
LT is the ultimate therapeutic option, but there is an additional risk in patients with PFIC 2. In severe cases, patients might develop allo-reactive antibodies specific to BSEP in the allograft, causing BSEP deficiency in the transplanted liver[19]. This happens in up to 8% of PFIC 2 patients who undergo LT[27]. Increased immuno-suppression may prevent these antibodies from developing, but if the disease recurs, plasmapheresis to remove anti-BSEP antibodies, B-cell depleting antibody therapy, or allogeneic hematopoietic stem cell transplant may help[19,27]. If none of these therapies succeed, a second LT might be required[27].
There have been many scientific advances offering hope for potential treatments for BSEP deficiency. One group led by Jillian Ellis developed a zebrafish model of BSEP deficiency[34]. Treating these zebrafish with rapamycin partially restored bile excretion as well as lengthened their lifespans[34]. The rapamycin likely localizes other transporters, such as MDR1, to the canaliculus in order to compensate for the lack of BSEP[34]. Rapamycin is an autophagy inducer, so the increased trafficking of MDR1 in rapamycin treated zebrafish might be the result of increased autophagy[34]. Another group led by Imagawa et al[33] generated a BSEP-deficient cellular model using patient cells[33]. They generated induced pluripotent stem cells (iPSCs) from peripheral blood mononuclear cells[33]. Then, they were able to differentiate these iPSCs into hepatocyte-like cells that mimicked the BSEP deficiency of the patient from whom they were derived[33]. These cells had abnormal BSEP expression, as well as reduced biliary excretion[33]. When the cells were treated with 4PBA, there was an increase in membrane expression of BSEP[33]. In vitro models such as this one provide a practical way to test the effect of many different therapeutic agents on cells derived from individual patients themselves. Advances in stem cell research might make it possible to determine what drugs a patient will respond best to, based on their specific mutation, without having to test them all on the patient.
PFIC 3
Mutations in the ABCB4 gene, located on chromosome 7q21, which encodes the MDR3 glycoprotein, cause not only PFIC 3, but also other cholestasis related diseases such as gallbladder disease 1/low phospholipid-associated cholelithiasis (GBD1/LPAC), parenteral nutrition-associated liver disease, transient neonatal cholestasis and intrahepatic cholestasis of pregnancy[17]. This phosphatidylcholine (PC) flippase is localized only to the canalicular membrane of hepatocytes and consists of two cytoplasmic nucleotide binding domains and two transmembrane domains, each with six transmembrane segments. This structure facilitates the transport of PC out of the hepatocyte and into the biliary canaliculi. This process is supported by the over-expression of MDR3 in polarized cells, increasing the rate of transport of fluorescently labeled PC, but not other phospholipids. MDR2, the mouse homolog of human MDR3, has been used in knockout (KO) mice in order to model MDR3 deficiency[35]. The absence of the PC flippase leads to hepatocyte necrosis, dilated biliary canaliculus, periportal inflammation and bile duct proliferation[36]. These MDR2 KO animals develop HCC earlier, within 4-6 mo, due to an accumulation of carcinogenic factors[37-39]. The expression of human MDR3 in the MDR2 KO animals restores the function and histological features of the hepatocytes[38,40].
Several variations in the ABCB4 gene have been described. Delaunay and colleagues used site-directed mutagenesis in a cell line model to show that the mutations I541F, L556R, and Q855L result in protein retention in the endoplasmic reticulum[41]. Another four mutations have no effect on MDR3 function: T424A and N510S had a normal pattern of canalicular expression with lower protein stability, while T175A and R652G had no detectable defect. Degiorgio et al[42] analyzed ABCB4 gene mutations in 68 PFIC 3 patients. The authors found 29 mutations in the coding region, including 23 missense, four nonsense, and two short insertion mutations in the gene. 10 of these mutations were present in the transmembrane domains (TM), specifically in TM7, involved in PC translocation[42]. Delaunay et al[41] devised a classification system for the various forms of these mutations[41]. Class I includes mutations that result in defective synthesis, class II covers variations that prevent protein maturation, class III mutants result in mature but defective proteins, class IV variations are unstable, and class V mutants have unknown pathogenicity[41]. These classifications are useful in determining potential therapies for patients, depending on their genotype.
When MDR3 does not function properly, PC is absent from the biliary canaliculi. The free hydrophobic bile acids in this space will damage cholangiocytes, leading to cholestasis[19]. In addition, cholesterol is more likely to crystallize into stones, damaging liver structures by obstructing small bile ducts[25]. PFIC 3 often presents later on in life than both PFIC 1 and PFIC 2, as late as adulthood in some cases[19]. In early onset, pruritus, hepatosplenomegaly, variceal bleeding caused by portal hypertension, alcoholic stools, stunted growth, jaundice, reduced bone density, and learning disabilities have all been associated with MDR3 deficiency[17,19]. Adult onset can present anywhere from cholelithiasis to benign recurrent intrahepatic cholestasis (BRIC)[19]. Unlike PFIC 1 and 2, PFIC 3 presents with elevated GGT[19]. Transaminases, bilirubin, and alkaline phosphatase are elevated as well[17,19]. Although serum bile salt and cholesterol can be in the normal range, biliary phospholipids will be reduced[17]. Histological findings include nonspecific portal inflammation, portal fibrosis, cholestasis with bile duct proliferation, and slight giant cell hepatitis[17,19]. MDR3 expression upon staining will depend on the type of protein defect, with functional protein defects staining normally and truncated protein defects not staining[17,19]. Additionally, hepatocellular carcinoma and cholangiocarcinoma have been associated with PFIC 3[19].
UDCA treatment has been effective in 70% of cases where the patient retains MDR3 expression and residual PC secretion[17]. Rifampin could also improve pruritus[19]. Chemical chaperones could improve MDR3 trafficking, depending on the specific mutation[43]. Biliary diversion procedures are not as successful with MDR3 deficiency because of the severity upon presentation[19]. LT is currently the only known curative treatment[19]. Gene therapy via adeno-associated virus has shown promise in restoring phospholipid excretion and preventing liver injury in murine models of PFIC 3[44]. This may be an alternative therapy for MRD3 deficiency in the near future.
NEWER TYPES OF PFIC
PFIC 4
PFIC 4 is caused by a loss of function mutation in the tight junction protein 2 (TJP2), also called zona occludens 2, present on chromosome 9q21[17]. In the liver, this protein is involved in forming tight junctions by interacting with transmembrane tight junction proteins and the actin cytoskeleton[45]. Tight junctions are essential in the liver because they help prevent the leakage of biliary components into the liver parenchyma[46]. Claudin-1 (CLDN1) and claudin-2 (CLDN2) are two integral tight junction proteins that are expressed in the liver, aiding in the formation of tight junctions[45]. Normally, these two proteins are localized to the canalicular membrane, but in TJP2 mutation, CLDN1 fails to localize, especially in the parenchyma of the hepatic lobule[45,47]. These compromised tight junctions then allow cytotoxic bile salts to leak into the paracellular space, causing damage to the surrounding hepatocytes and cholangiocytes[19,45].
This disease presents with severe cholestasis and low GGT levels[17]. Patients lack mutations in ATP8B1 and ABCB11 genes, excluding the diagnoses of PFIC 1 and PFIC 2. Extrahepatic symptoms have been reported in some patients, mainly in the neurological and respiratory systems[45]. This is likely related to the expression of TJP2 in every epithelial cell in the body[45]. The reason the liver is consistently affected by TJP2 deficiency is because of the particularly damaging nature of the bile acids present in the canaliculi[47]. Pathological findings include intracellular cholestasis, giant cell transformation, lack of TJP2 with staining, abnormal CLDN1 localization, and normal CLDN2 localization[19,47]. The lack of TJP2 could be tied to the nonsense mediated mRNA decay (NMD) process[45]. This process degrades mutant transcripts, such as TJP2 in the case of a mutation[45]. Even though the protein could have residual function, it is still degraded[45]. TJP2 was found to have low mRNA expression in PFIC 4 patients[45]. Western blotting revealed no TJP2 expression, indicating a possible link between the mutation in TJP2 and NMD[45].
There have been several reports of HCC occurrence in patients suffering with TJP2 deficiency[48-50]. The exact mechanism of tumorigenesis has not yet been elucidated. In terms of treatment, there have been no reported cases of recurrence after LT[17].
PFIC 5
Loss of function mutations in the NR1H4 gene (chromosome 12q23), encoding FXR, result in PFIC 5[4,17]. FXR is a nuclear receptor activated by bile acids and directly involved in the expression of both BSEP and MDR3, proteins affected in PFIC 2 and PFIC 3 respectively[4]. This protein is activated by elevated bile acids levels in the ileum, inducing expression of fibroblast growth factor 19 (FGF19)[51]. In the liver, FGF19 binds to the fibroblast growth factor receptor 4/β-Klotho complex, which in turn represses cytochrome P450 7A1 (CYP7A1)[51]. Repression of this enzyme reduces de novo bile acid synthesis[51].
Features of this disease include neonatal onset of normal GGT associated cholestasis, elevated serum bilirubin, elevated serum AFP levels, undetectable expression of BSEP in the bile canaliculi, and vitamin K independent coagulopathy[4]. This vitamin K independent coagulopathy is unique to PFIC 5 and has been shown to be a direct result of FXR mutation[4]. Three fibrinogen genes, as well as some coagulation factors have been linked with FXR-dependent induction, which does not occur in PFIC 5 patients[4]. Histological findings are intralobular cholestasis with ductular reaction, hepatocellular ballooning, fibrosis progressing into micronodular cirrhosis, and giant cell transformation[19]. NR1H4/FXR associated PFIC is very rare as only eight cases have been reported in the literature[4,52,53]. LT has been used, but steatosis in the transplanted liver has been reported in some of these cases[4,53]. UDCA and rifampicin may be used in order to improve pruritus, but ASBT inhibitors, as well as FXR agonists such as obeticholic acid, both show promise as future medical therapies[51,54].
PFIC associated with MYO5B defects
The MYO5B gene (chromosome 18q21.1) encodes an actin-associated molecular motor known as MYO5B[17]. The interaction between MYO5B and RAS-related GTP-binding protein 11A (RAB11A) is essential for the polarization of epithelial cells, as well as localizing BSEP to the canalicular membrane[55]. Diminished activity of the MYO5B/RAB11A recycling endosome pathway is related to disrupting the localization of BSEP[56]. Mutations of this gene are associated with microvillus inclusion disease (MVID) which affects the enterocytes and leads to diarrhea, as well as malabsorption[57]. Mislocalized apical brush border proteins, villus atrophy, and the presence of microvillus inclusion bodies are all associated with MVID[57-60]. Total parenteral nutrition (TPN) is required throughout life, but it has been associated with a high risk of sepsis and small bowel transplant[61].
MVID has been associated with cholestatic liver disease, which possibly occurs as a result of TPN[57]. In fact, MYO5B gene mutations may account for 20% of the idiopathic low-GGT associated cholestasis in pediatric patients[60]. This cholestasis presents with low to normal GGT levels, jaundice, pruritus, mildly elevated Alanine transaminase and Aspartate transaminase, elevated serum BS levels, hepatomegaly, portal and lobular fibrosis, and giant cell transformation[17,19,60]. Staining of BSEP and MDR3 may show that they are present, but not localized to the canalicular membrane, although this may not occur in every patient[19,60].
There is evidence for different phenotypes resulting from the type of MYO5B gene mutation[55,60]. Mutations with limited effects on MYO5B functionality are linked with canalicular transport defects in hepatocytes but are not known to have as significant of an effect on enterocytes[60]. In more damaging mutations, the hepatocyte defects may be greater, but there will also be enterocyte disruption[60]. Enterocytes will not uptake bile acids as efficiently, reducing the amount of bile product taken back to the liver via enterohepatic circulation, potentially alleviating or protecting against cholestatic symptoms[60]. There seems to be an inverse relationship between cholestasis and intestinal function. UDCA, rifampin, and cholestyramine have been used to treat pruritus in these patients[19]. If medical treatment is not sufficient, PEBD may be successful[19]. When symptoms necessitate transplant, combined bowel-liver transplant should be considered in order to avoid post-transplant onset of cholestasis[60].
HOW TO STUDY PFIC?
After the identification of mutations in patients, several models have been created to better understand not only the pathophysiology of PFIC, but also to investigate drugs and treatments. The first model is the use of a cell line with mutations in PFIC related genes to study the mechanisms of these variants on both the molecular and clinical level. van der Velden et al[62], using different cell lines, studied some FIC1 mutations[62]. Delaunay et al[41], studied MDR3 gene mutations using HepG2 as a model. A second option to study PFIC are knockout mice[63]. Some studies have reported the use of ABCB11-/-mice as a model for PFIC 2, however the clinical findings are less severe than those of humans[63]. MDR2 KO mice are used to model complications resulting from MDR3 deficiency in humans and are also used to study treatments for all MDR3-related diseases[35,40].
In 2012, when Shinya Yamanaka and John B. Gurdon were awarded The Nobel Prize in Physiology or Medicine for their discovery in pluripotency, a new method to study PFIC became available. The possibility of transforming any somatic cell into an iPSCs was revolutionary. Okita and colleagues used plasmids to express c-Myc, SRY-box transcription factor 2 (Sox2), Kruppel-like factor 4, and octamer-binding transcription factor 4 (Oct4) in reprogrammed mature cells[64]. This last set of genes is known as Yamanaka’s Factors. However, there are many different protocols to generate iPSCs. Si-Tayeb et al[65] used the expression of Oct4, Sox2, Nanog, and Lin-28 homolog A in a lentivirus delivery system to generate iPS cells[65]. In this way, Imagawa et al[33] generated a BSEP-deficient cellular model using patient cells and tested the effects of 4PBA treatment on protein expression and localization[33]. With this system of reprogramming cells, and access to patient cells with different mutations, a new model became available to better understand the various types of PFIC. The possibility of differentiating these cells into liver cells, such as hepatocytes and cholangiocytes, gives researchers the opportunity to test potential treatments in vitro. This can be extremely useful since very few patients have these rare diseases and are available to enroll in clinical trials. Large scale drug trials may not be feasible.
CONCLUSION
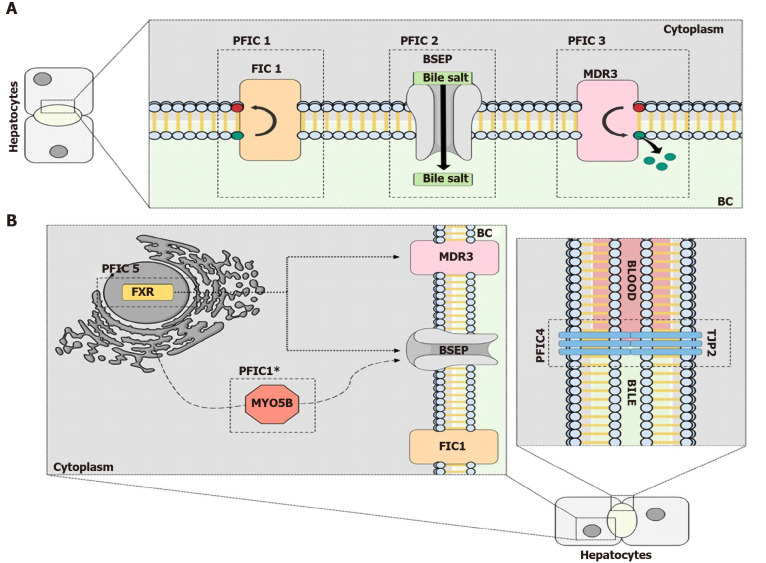
PFIC is a rare disease that affects the bile secretion process of the hepatocytes due to alterations in the proteins involved, as shown in Figure 2. Better understanding of the molecular alterations in this context is necessary to elucidate new targets and pathways that could be the focus of new treatments in the future. With advances in inducing pluripotent cells, a new model that is more precise and closer to humans has become available. Also, the progress in gene editing will help in the development of new models and potentially be used as a treatment method in the future.
Figure 2.
The molecular mechanisms behind progressive familial intrahepatic cholestasis. A: The classic types of Progressive Familial Intrahepatic Cholestasis (PFIC). PFIC 1 is related to mutations in the genes that encode the flippase familial intrahepatic cholestasis 1, which flips phospholipids in the plasma membrane. Mutations in the bile salt export pump (BSEP) protein, a bile salt transporter, results in PFIC 2. The third type of PFIC is caused by mutations in the gene that encodes the Multidrug resistance class 3 glycoprotein (MDR3) protein, another lipid flippase; B: The newer types of PFIC. Mutations in the tight junction protein-2 protein, which prevents the mixing of blood and bile acids, are responsible for PFIC 4. PFIC 5 is a result of mutations in the farnesoid X receptor protein, a transcription factor important for BSEP and MDR3 ecpression. Mutations in Myosin VB result in a PFIC phenotype because the trafficking of the BSEP protein from the endoplasmic reticulum to the plasma membrane is disrupted. PFIC: Progressive Familial Intrahepatic Cholestasis; FIC1: Familial intrahepatic cholestasis 1; BSEP: Bile salt export pump; MDR3: Multidrug resistance class 3 glycoprotein; TJP2: Tight junction protein-2; FXR: Farnesoid X receptor.
Footnotes
Conflict-of-interest statement: A.S.-G., is co-founder and have a financial interest in Von Baer Wolff, Inc. a company focused on biofabrication of autologous human hepatocytes from stem cells technology and programming liver failure and their interests are managed by the Conflict of Interest Office at the University of Pittsburgh in accordance with their policies
Manuscript source: Invited manuscript
Peer-review started: September 11, 2020
First decision: October 27, 2020
Article in press: November 29, 2020
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Cao ZF, Zhang XQ S-Editor: Fan JR L-Editor: A P-Editor: Ma YJ
Contributor Information
Sriram Amirneni, Department of Pathology, University of Pittsburgh, Pittsburgh, PA 15213, United States.
Nils Haep, Department of Pathology, University of Pittsburgh, Pittsburgh, PA 15213, United States.
Mohammad A Gad, Department of Pathology, University of Pittsburgh, Pittsburgh, PA 15213, United States.
Alejandro Soto-Gutierrez, Department of Pathology, University of Pittsburgh, Pittsburgh, PA 15213, United States; Pittsburgh Liver Research Center, University of Pittsburgh, Pittsburgh, PA 15213, United States.
James E Squires, Pittsburgh Liver Research Center, University of Pittsburgh, Pittsburgh, PA 15213, United States; Division of Gastroenterology, Hepatology, and Nutrition, UPMC Children's Hospital of Pittsburgh, Pittsburgh, PA 15224, United States.
Rodrigo M Florentino, Department of Pathology, University of Pittsburgh, Pittsburgh, PA 15213, United States. rodrigomachado@pitt.edu.
References
- 1.Boyer JL. Bile formation and secretion. Compr Physiol. 2013;3:1035–1078. doi: 10.1002/cphy.c120027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hirschfield GM, Heathcote EJ, Gershwin ME. Pathogenesis of cholestatic liver disease and therapeutic approaches. Gastroenterology. 2010;139:1481–1496. doi: 10.1053/j.gastro.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 3.Alvarez L, Jara P, Sánchez-Sabaté E, Hierro L, Larrauri J, Díaz MC, Camarena C, De la Vega A, Frauca E, López-Collazo E, Lapunzina P. Reduced hepatic expression of farnesoid X receptor in hereditary cholestasis associated to mutation in ATP8B1. Hum Mol Genet. 2004;13:2451–2460. doi: 10.1093/hmg/ddh261. [DOI] [PubMed] [Google Scholar]
- 4.Gomez-Ospina N, Potter CJ, Xiao R, Manickam K, Kim MS, Kim KH, Shneider BL, Picarsic JL, Jacobson TA, Zhang J, He W, Liu P, Knisely AS, Finegold MJ, Muzny DM, Boerwinkle E, Lupski JR, Plon SE, Gibbs RA, Eng CM, Yang Y, Washington GC, Porteus MH, Berquist WE, Kambham N, Singh RJ, Xia F, Enns GM, Moore DD. Mutations in the nuclear bile acid receptor FXR cause progressive familial intrahepatic cholestasis. Nat Commun. 2016;7:10713. doi: 10.1038/ncomms10713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Marson FAL, Bertuzzo CS, Ribeiro JD. Classification of CFTR mutation classes. Lancet Respir Med. 2016;4:e37–e38. doi: 10.1016/S2213-2600(16)30188-6. [DOI] [PubMed] [Google Scholar]
- 6.Trampert DC, Nathanson MH. Regulation of bile secretion by calcium signaling in health and disease. Biochim Biophys Acta Mol Cell Res. 2018;1865:1761–1770. doi: 10.1016/j.bbamcr.2018.05.010. [DOI] [PubMed] [Google Scholar]
- 7.Dyson JK, Hirschfield GM, Adams DH, Beuers U, Mann DA, Lindor KD, Jones DE. Novel therapeutic targets in primary biliary cirrhosis. Nat Rev Gastroenterol Hepatol. 2015;12:147–158. doi: 10.1038/nrgastro.2015.12. [DOI] [PubMed] [Google Scholar]
- 8.Arsenijevic A, Stojanovic B, Milovanovic J, Arsenijevic D, Arsenijevic N, Milovanovic M. Galectin-3 in Inflammasome Activation and Primary Biliary Cholangitis Development. Int J Mol Sci. 2020;21:5097. doi: 10.3390/ijms21145097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jiang X, Karlsen TH. Genetics of primary sclerosing cholangitis and pathophysiological implications. Nat Rev Gastroenterol Hepatol. 2017;14:279–295. doi: 10.1038/nrgastro.2016.154. [DOI] [PubMed] [Google Scholar]
- 10.Palmer KR, Xiaohua L, Mol BW. Management of intrahepatic cholestasis in pregnancy. Lancet. 2019;393:853–854. doi: 10.1016/S0140-6736(18)32323-7. [DOI] [PubMed] [Google Scholar]
- 11.Sundaram V, Björnsson ES. Drug-induced cholestasis. Hepatol Commun. 2017;1:726–735. doi: 10.1002/hep4.1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hoofnagle JH, Björnsson ES. Drug-Induced Liver Injury - Types and Phenotypes. N Engl J Med. 2019;381:264–273. doi: 10.1056/NEJMra1816149. [DOI] [PubMed] [Google Scholar]
- 13.Chiang JY. Bile acid metabolism and signaling. Compr Physiol. 2013;3:1191–1212. doi: 10.1002/cphy.c120023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chiang JY. Bile acids: regulation of synthesis. J Lipid Res. 2009;50:1955–1966. doi: 10.1194/jlr.R900010-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Banales JM, Huebert RC, Karlsen T, Strazzabosco M, LaRusso NF, Gores GJ. Cholangiocyte pathobiology. Nat Rev Gastroenterol Hepatol. 2019;16:269–281. doi: 10.1038/s41575-019-0125-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tabibian JH, Masyuk AI, Masyuk TV, O'Hara SP, LaRusso NF. Physiology of cholangiocytes. Compr Physiol. 2013;3:541–565. doi: 10.1002/cphy.c120019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vitale G, Gitto S, Vukotic R, Raimondi F, Andreone P. Familial intrahepatic cholestasis: New and wide perspectives. Dig Liver Dis. 2019;51:922–933. doi: 10.1016/j.dld.2019.04.013. [DOI] [PubMed] [Google Scholar]
- 18.Paulusma CC, Folmer DE, Ho-Mok KS, de Waart DR, Hilarius PM, Verhoeven AJ, Oude Elferink RP. ATP8B1 requires an accessory protein for endoplasmic reticulum exit and plasma membrane lipid flippase activity. Hepatology. 2008;47:268–278. doi: 10.1002/hep.21950. [DOI] [PubMed] [Google Scholar]
- 19.Henkel SA, Squires JH, Ayers M, Ganoza A, Mckiernan P, Squires JE. Expanding etiology of progressive familial intrahepatic cholestasis. World J Hepatol. 2019;11:450–463. doi: 10.4254/wjh.v11.i5.450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.van der Mark VA, de Waart DR, Ho-Mok KS, Tabbers MM, Voogt HW, Oude Elferink RP, Knisely AS, Paulusma CC. The lipid flippase heterodimer ATP8B1-CDC50A is essential for surface expression of the apical sodium-dependent bile acid transporter (SLC10A2/ASBT) in intestinal Caco-2 cells. Biochim Biophys Acta. 2014;1842:2378–2386. doi: 10.1016/j.bbadis.2014.09.003. [DOI] [PubMed] [Google Scholar]
- 21.Verhulst PM, van der Velden LM, Oorschot V, van Faassen EE, Klumperman J, Houwen RH, Pomorski TG, Holthuis JC, Klomp LW. A flippase-independent function of ATP8B1, the protein affected in familial intrahepatic cholestasis type 1, is required for apical protein expression and microvillus formation in polarized epithelial cells. Hepatology. 2010;51:2049–2060. doi: 10.1002/hep.23586. [DOI] [PubMed] [Google Scholar]
- 22.Stapelbroek JM, Peters TA, van Beurden DH, Curfs JH, Joosten A, Beynon AJ, van Leeuwen BM, van der Velden LM, Bull L, Oude Elferink RP, van Zanten BA, Klomp LW, Houwen RH. ATP8B1 is essential for maintaining normal hearing. Proc Natl Acad Sci USA. 2009;106:9709–9714. doi: 10.1073/pnas.0807919106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Paulusma CC, Groen A, Kunne C, Ho-Mok KS, Spijkerboer AL, Rudi de Waart D, Hoek FJ, Vreeling H, Hoeben KA, van Marle J, Pawlikowska L, Bull LN, Hofmann AF, Knisely AS, Oude Elferink RP. Atp8b1 deficiency in mice reduces resistance of the canalicular membrane to hydrophobic bile salts and impairs bile salt transport. Hepatology. 2006;44:195–204. doi: 10.1002/hep.21212. [DOI] [PubMed] [Google Scholar]
- 24.Folmer DE, van der Mark VA, Ho-Mok KS, Oude Elferink RP, Paulusma CC. Differential effects of progressive familial intrahepatic cholestasis type 1 and benign recurrent intrahepatic cholestasis type 1 mutations on canalicular localization of ATP8B1. Hepatology. 2009;50:1597–1605. doi: 10.1002/hep.23158. [DOI] [PubMed] [Google Scholar]
- 25.Srivastava A. Progressive familial intrahepatic cholestasis. J Clin Exp Hepatol. 2014;4:25–36. doi: 10.1016/j.jceh.2013.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Van Vaisberg V, Tannuri ACA, Lima FR, Tannuri U. Ileal exclusion for pruritus treatment in children with progressive familial intrahepatic cholestasis and other cholestatic diseases. J Pediatr Surg. 2020;55:1385–1391. doi: 10.1016/j.jpedsurg.2019.09.018. [DOI] [PubMed] [Google Scholar]
- 27.van der Woerd WL, van Mil SW, Stapelbroek JM, Klomp LW, van de Graaf SF, Houwen RH. Familial cholestasis: progressive familial intrahepatic cholestasis, benign recurrent intrahepatic cholestasis and intrahepatic cholestasis of pregnancy. Best Pract Res Clin Gastroenterol. 2010;24:541–553. doi: 10.1016/j.bpg.2010.07.010. [DOI] [PubMed] [Google Scholar]
- 28.Plass JR, Mol O, Heegsma J, Geuken M, Faber KN, Jansen PL, Müller M. Farnesoid X receptor and bile salts are involved in transcriptional regulation of the gene encoding the human bile salt export pump. Hepatology. 2002;35:589–596. doi: 10.1053/jhep.2002.31724. [DOI] [PubMed] [Google Scholar]
- 29.Byrne JA, Strautnieks SS, Ihrke G, Pagani F, Knisely AS, Linton KJ, Mieli-Vergani G, Thompson RJ. Missense mutations and single nucleotide polymorphisms in ABCB11 impair bile salt export pump processing and function or disrupt pre-messenger RNA splicing. Hepatology. 2009;49:553–567. doi: 10.1002/hep.22683. [DOI] [PubMed] [Google Scholar]
- 30.Wang L, Dong H, Soroka CJ, Wei N, Boyer JL, Hochstrasser M. Degradation of the bile salt export pump at endoplasmic reticulum in progressive familial intrahepatic cholestasis type II. Hepatology. 2008;48:1558–1569. doi: 10.1002/hep.22499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Naoi S, Hayashi H, Inoue T, Tanikawa K, Igarashi K, Nagasaka H, Kage M, Takikawa H, Sugiyama Y, Inui A, Nagai T, Kusuhara H. Improved liver function and relieved pruritus after 4-phenylbutyrate therapy in a patient with progressive familial intrahepatic cholestasis type 2. J Pediatr 2014; 164: 1219-1227. :e3. doi: 10.1016/j.jpeds.2013.12.032. [DOI] [PubMed] [Google Scholar]
- 32.Hayashi H, Sugiyama Y. 4-phenylbutyrate enhances the cell surface expression and the transport capacity of wild-type and mutated bile salt export pumps. Hepatology. 2007;45:1506–1516. doi: 10.1002/hep.21630. [DOI] [PubMed] [Google Scholar]
- 33.Imagawa K, Takayama K, Isoyama S, Tanikawa K, Shinkai M, Harada K, Tachibana M, Sakurai F, Noguchi E, Hirata K, Kage M, Kawabata K, Sumazaki R, Mizuguchi H. Generation of a bile salt export pump deficiency model using patient-specific induced pluripotent stem cell-derived hepatocyte-like cells. Sci Rep. 2017;7:41806. doi: 10.1038/srep41806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ellis JL, Bove KE, Schuetz EG, Leino D, Valencia CA, Schuetz JD, Miethke A, Yin C. Zebrafish abcb11b mutant reveals strategies to restore bile excretion impaired by bile salt export pump deficiency. Hepatology. 2018;67:1531–1545. doi: 10.1002/hep.29632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Smit JJ, Schinkel AH, Oude Elferink RP, Groen AK, Wagenaar E, van Deemter L, Mol CA, Ottenhoff R, van der Lugt NM, van Roon MA. Homozygous disruption of the murine mdr2 P-glycoprotein gene leads to a complete absence of phospholipid from bile and to liver disease. Cell. 1993;75:451–462. doi: 10.1016/0092-8674(93)90380-9. [DOI] [PubMed] [Google Scholar]
- 36.Popov Y, Patsenker E, Fickert P, Trauner M, Schuppan D. Mdr2 (Abcb4)-/- mice spontaneously develop severe biliary fibrosis via massive dysregulation of pro- and antifibrogenic genes. J Hepatol. 2005;43:1045–1054. doi: 10.1016/j.jhep.2005.06.025. [DOI] [PubMed] [Google Scholar]
- 37.Jacquemin E. Role of multidrug resistance 3 deficiency in pediatric and adult liver disease: one gene for three diseases. Semin Liver Dis. 2001;21:551–562. doi: 10.1055/s-2001-19033. [DOI] [PubMed] [Google Scholar]
- 38.Oude Elferink RP, Paulusma CC. Function and pathophysiological importance of ABCB4 (MDR3 P-glycoprotein) Pflugers Arch. 2007;453:601–610. doi: 10.1007/s00424-006-0062-9. [DOI] [PubMed] [Google Scholar]
- 39.Trauner M, Fickert P, Wagner M. MDR3 (ABCB4) defects: a paradigm for the genetics of adult cholestatic syndromes. Semin Liver Dis. 2007;27:77–98. doi: 10.1055/s-2006-960172. [DOI] [PubMed] [Google Scholar]
- 40.Smith AJ, de Vree JM, Ottenhoff R, Oude Elferink RP, Schinkel AH, Borst P. Hepatocyte-specific expression of the human MDR3 P-glycoprotein gene restores the biliary phosphatidylcholine excretion absent in Mdr2 (-/-) mice. Hepatology. 1998;28:530–536. doi: 10.1002/hep.510280234. [DOI] [PubMed] [Google Scholar]
- 41.Delaunay JL, Durand-Schneider AM, Dossier C, Falguières T, Gautherot J, Davit-Spraul A, Aït-Slimane T, Housset C, Jacquemin E, Maurice M. A functional classification of ABCB4 variations causing progressive familial intrahepatic cholestasis type 3. Hepatology. 2016;63:1620–1631. doi: 10.1002/hep.28300. [DOI] [PubMed] [Google Scholar]
- 42.Degiorgio D, Colombo C, Seia M, Porcaro L, Costantino L, Zazzeron L, Bordo D, Coviello DA. Molecular characterization and structural implications of 25 new ABCB4 mutations in progressive familial intrahepatic cholestasis type 3 (PFIC3) Eur J Hum Genet. 2007;15:1230–1238. doi: 10.1038/sj.ejhg.5201908. [DOI] [PubMed] [Google Scholar]
- 43.Gordo-Gilart R, Andueza S, Hierro L, Jara P, Alvarez L. Functional Rescue of Trafficking-Impaired ABCB4 Mutants by Chemical Chaperones. PLoS One. 2016;11:e0150098. doi: 10.1371/journal.pone.0150098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Aronson SJ, Bakker RS, Shi X, Duijst S, Ten Bloemendaal L, de Waart DR, Verheij J, Ronzitti G, Oude Elferink RP, Beuers U, Paulusma CC, Bosma PJ. Liver-directed gene therapy results in long-term correction of progressive familial intrahepatic cholestasis type 3 in mice. J Hepatol. 2019;71:153–162. doi: 10.1016/j.jhep.2019.03.021. [DOI] [PubMed] [Google Scholar]
- 45.Sambrotta M, Thompson RJ. Mutations in TJP2, encoding zona occludens 2, and liver disease. Tissue Barriers. 2015;3:e1026537. doi: 10.1080/21688370.2015.1026537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.González-Mariscal L, Gallego-Gutiérrez H, González-González L, Hernández-Guzmán C. ZO-2 Is a Master Regulator of Gene Expression, Cell Proliferation, Cytoarchitecture, and Cell Size. Int J Mol Sci. 2019;20:4128. doi: 10.3390/ijms20174128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sambrotta M, Strautnieks S, Papouli E, Rushton P, Clark BE, Parry DA, Logan CV, Newbury LJ, Kamath BM, Ling S, Grammatikopoulos T, Wagner BE, Magee JC, Sokol RJ, Mieli-Vergani G University of Washington Center for Mendelian Genomics; Smith JD; Johnson CA; McClean P; Simpson MA; Knisely AS; Bull LN; Thompson RJ. Mutations in TJP2 cause progressive cholestatic liver disease. Nat Genet. 2014;46:326–328. doi: 10.1038/ng.2918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vij M, Shanmugam NP, Reddy MS, Sankaranarayanan S, Rela M. Paediatric hepatocellular carcinoma in tight junction protein 2 (TJP2) deficiency. Virchows Arch. 2017;471:679–683. doi: 10.1007/s00428-017-2204-1. [DOI] [PubMed] [Google Scholar]
- 49.Wei CS, Becher N, Friis JB, Ott P, Vogel I, Grønbæk H. New tight junction protein 2 variant causing progressive familial intrahepatic cholestasis type 4 in adults: A case report. World J Gastroenterol. 2020;26:550–561. doi: 10.3748/wjg.v26.i5.550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhou S, Hertel PM, Finegold MJ, Wang L, Kerkar N, Wang J, Wong LJ, Plon SE, Sambrotta M, Foskett P, Niu Z, Thompson RJ, Knisely AS. Hepatocellular carcinoma associated with tight-junction protein 2 deficiency. Hepatology. 2015;62:1914–1916. doi: 10.1002/hep.27872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cariello M, Piccinin E, Garcia-Irigoyen O, Sabbà C, Moschetta A. Nuclear receptor FXR, bile acids and liver damage: Introducing the progressive familial intrahepatic cholestasis with FXR mutations. Biochim Biophys Acta Mol Basis Dis. 2018;1864:1308–1318. doi: 10.1016/j.bbadis.2017.09.019. [DOI] [PubMed] [Google Scholar]
- 52.Chen HL, Li HY, Wu JF, Wu SH, Chen HL, Yang YH, Hsu YH, Liou BY, Chang MH, Ni YH. Panel-Based Next-Generation Sequencing for the Diagnosis of Cholestatic Genetic Liver Diseases: Clinical Utility and Challenges. J Pediatr 2019; 205: 153-159. :e6. doi: 10.1016/j.jpeds.2018.09.028. [DOI] [PubMed] [Google Scholar]
- 53.Himes RW, Mojarrad M, Eslahi A, Finegold MJ, Maroofian R, Moore DD. NR1H4-related Progressive Familial Intrahepatic Cholestasis 5: Further Evidence for Rapidly Progressive Liver Failure. J Pediatr Gastroenterol Nutr. 2020;70:e111–e113. doi: 10.1097/MPG.0000000000002670. [DOI] [PubMed] [Google Scholar]
- 54.Carino A, Biagioli M, Marchianò S, Fiorucci C, Bordoni M, Roselli R, Di Giorgio C, Baldoni M, Ricci P, Monti MC, Morretta E, Zampella A, Distrutti E, Fiorucci S. Opposite effects of the FXR agonist obeticholic acid on Mafg and Nrf2 mediate the development of acute liver injury in rodent models of cholestasis. Biochim Biophys Acta Mol Cell Biol Lipids. 2020;1865:158733. doi: 10.1016/j.bbalip.2020.158733. [DOI] [PubMed] [Google Scholar]
- 55.Qiu YL, Gong JY, Feng JY, Wang RX, Han J, Liu T, Lu Y, Li LT, Zhang MH, Sheps JA, Wang NL, Yan YY, Li JQ, Chen L, Borchers CH, Sipos B, Knisely AS, Ling V, Xing QH, Wang JS. Defects in myosin VB are associated with a spectrum of previously undiagnosed low γ-glutamyltransferase cholestasis. Hepatology. 2017;65:1655–1669. doi: 10.1002/hep.29020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Girard M, Lacaille F, Verkarre V, Mategot R, Feldmann G, Grodet A, Sauvat F, Irtan S, Davit-Spraul A, Jacquemin E, Ruemmele F, Rainteau D, Goulet O, Colomb V, Chardot C, Henrion-Caude A, Debray D. MYO5B and bile salt export pump contribute to cholestatic liver disorder in microvillous inclusion disease. Hepatology. 2014;60:301–310. doi: 10.1002/hep.26974. [DOI] [PubMed] [Google Scholar]
- 57.Overeem AW, Li Q, Qiu YL, Cartón-García F, Leng C, Klappe K, Dronkers J, Hsiao NH, Wang JS, Arango D, van Ijzendoorn SCD. A Molecular Mechanism Underlying Genotype-Specific Intrahepatic Cholestasis Resulting From MYO5B Mutations. Hepatology. 2020;72:213–229. doi: 10.1002/hep.31002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gonzales E, Taylor SA, Davit-Spraul A, Thébaut A, Thomassin N, Guettier C, Whitington PF, Jacquemin E. MYO5B mutations cause cholestasis with normal serum gamma-glutamyl transferase activity in children without microvillous inclusion disease. Hepatology. 2017;65:164–173. doi: 10.1002/hep.28779. [DOI] [PubMed] [Google Scholar]
- 59.Jarasvaraparn C, He M, Granadillo JL, Kulkarni S, Stoll J, Liss K. MYO5B Pathogenic Variants Found to Cause Intestinal Symptoms Without Microvillus Inclusion Disease In A Child Who Previously Underwent Liver Transplantation For PFIC-like Cholestasis. J Pediatr Gastroenterol Nutr. :2020. doi: 10.1097/MPG.0000000000002792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.van IJzendoorn SCD, Li Q, Qiu YL, Wang JS, Overeem AW. Unequal effects of MYO5B mutations in liver and intestine determine the clinical presentation of low-GGT cholestasis. Hepatology. :2020. doi: 10.1002/hep.31430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Engevik AC, Kaji I, Engevik MA, Meyer AR, Weis VG, Goldstein A, Hess MW, Müller T, Koepsell H, Dudeja PK, Tyska M, Huber LA, Shub MD, Ameen N, Goldenring JR. Loss of MYO5B Leads to Reductions in Na+ Absorption With Maintenance of CFTR-Dependent Cl- Secretion in Enterocytes. Gastroenterology 2018; 155: 1883-1897. :e10. doi: 10.1053/j.gastro.2018.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.van der Velden LM, Stapelbroek JM, Krieger E, van den Berghe PV, Berger R, Verhulst PM, Holthuis JC, Houwen RH, Klomp LW, van de Graaf SF. Folding defects in P-type ATP 8B1 associated with hereditary cholestasis are ameliorated by 4-phenylbutyrate. Hepatology. 2010;51:286–296. doi: 10.1002/hep.23268. [DOI] [PubMed] [Google Scholar]
- 63.Mariotti V, Strazzabosco M, Fabris L, Calvisi DF. Animal models of biliary injury and altered bile acid metabolism. Biochim Biophys Acta Mol Basis Dis. 2018;1864:1254–1261. doi: 10.1016/j.bbadis.2017.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Okita K, Matsumura Y, Sato Y, Okada A, Morizane A, Okamoto S, Hong H, Nakagawa M, Tanabe K, Tezuka K, Shibata T, Kunisada T, Takahashi M, Takahashi J, Saji H, Yamanaka S. A more efficient method to generate integration-free human iPS cells. Nat Methods. 2011;8:409–412. doi: 10.1038/nmeth.1591. [DOI] [PubMed] [Google Scholar]
- 65.Si-Tayeb K, Noto FK, Nagaoka M, Li J, Battle MA, Duris C, North PE, Dalton S, Duncan SA. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology. 2010;51:297–305. doi: 10.1002/hep.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]