Abstract
OBJECTIVE:
This study was designed to show that allergic reactions with gamma cyclodextrin-related caused by sugammadex are not anaphylaxis but an anaphylactoid reaction.
METHODS:
In this retrospective study, 83 morbidly obese patients, who underwent elective laparoscopic sleeve gastrectomy operation, were included. Patient data were obtained from patient files and electronic health records system (SARUS). The patients were divided into two groups as sugammadex (Group S) and neostigmine (Group N). Patient data were recorded, including patient preoperative demographics (age, gender) and preoperative and postoperative (12 hours later) levels of total IgE and ECP (Eosinophil cationic protein) levels.
RESULTS:
There was not a significant change in the total IgE levels in Group S or Group N (p>0.05); however, the levels of ECP significantly decreased in both groups (p<0.001, p=0.01). In the patients in Group S, the preoperative levels of total IgE were significantly positively, and weakly correlated with the preoperative ECP (p=0.311, p=0.045) and postoperative ECP (p=0.310, p=0.046) levels.
CONCLUSION:
Allergic reactions that arise from sugammadex related with gamma-cyclodextrin are non-IgE mediated anaphylactoid reactions. Anaphylactoid reaction was not observed after administration of 2 mg kg-1 sugammadex intravenously in patients with morbid obesity.
Keywords: Sugammadex, immunoglobulin e, cyclodextrins, eosinophil cationic protein, neostigmine, anaphylaxis
Anaphylaxis is an IgE-mediated allergic reaction [1, 2], which may lead to life-threatening consequences if left untreated. Anaphylaxis allergen must be in contact with it beforehand with the patient. The anaphylactoid reaction is a non-IgE mediated reaction and does not require direct exposure to the allergen before. Although sugammadex has been approved by the Food and Drug Administration in the US for its use in adult patients [3], it is not recommended to be used in patients younger than two years old.
Sugammadex is a synthetic γ-dextrin derivative (modified gamma-cyclodextrin), used for reversing neuromuscular blockade by binding to steroidal neuromuscular agents selectively [4–5]. As cyclodextrins are components of several types of flavors, vitamins, colorants, and unsaturated fats [6], they are ingested commonly. Despite the case reports of intraoperative anaphylaxis in patients receiving sugammadex [7], which contains cyclodextrin as a drug carrier molecule, there has been a recent increase in comparative adult studies with neostigmine in the literature, reporting low rates of hypersensitivity and anaphylaxis [8].
Obesity affects several systems in the human body negatively, mostly affecting the distal airways in the respiratory system adversely, leading to emerging symptoms of bronchial asthma [9]. Bronchospasm can be the only symptom of sugammadex-related anaphylaxis [10]. However, the pathogenesis of sugammadex-induced anaphylaxis has not been clarified yet [7–10]. Studies in the literature have demonstrated increases in the levels of total IgE and ECP in hypersensitivity reactions [11].
Total IgE levels [11, 12] and eosinophil counts [11–13] are expected to increase in the blood during bronchial asthma attacks. Serum eosinophil cationic protein (ECP) [11–14] is an inflammatory marker released from eosinophils and is a major finding in attacks of bronchial asthma, in which the levels of ECP may increase [14, 15].
To our knowledge, there are no studies investigating the IgE and ECP levels in the pathogenesis of sugammadex-related hypersensitivity and anaphylaxis in patients with morbid obesity. There is no study in the literature that sugammadex causes an anaphylactic reaction. In this present study, we aimed to examine the effects of sugammadex and neostigmine on the eosinophil cationic protein (ECP) and total IgE levels in bronchial asthma-susceptible morbid obesity patients who underwent sleeve gastrectomy and who had no known history of bronchial asthma or any allergic diseases. This study was designed to show that allergic reactions with gamma cyclodextrin-related sugammadex are not anaphylaxis but an anaphylactoid reaction.
MATERIALS AND METHODS
Study Population
This study was conducted in compliance with the Declarations of Helsinki in the period from the year 2018 to 2019 after obtaining the approval of the Institutional Ethics Committee (Date-Decision no: 04.07.2019–16/14). There was no patient or public participation in this study.
A total of 83 morbidly obese (Body Mass Index (BMI) >40 kg/m2) ASA I -II patients (mean±SD 35.12±12.13 years, 74.7% were females), who underwent elective laparoscopic sleeve gastrectomy operation and who underwent a routine physical examination in the preoperative period to detect chest diseases, were included in this retrospective study. Patients with the morbid obesity with mild obstruction according to spirometry flow-volume curves were included in this study (Forced midexpiratory Flow (FEF25–75 <70%). Patient data were obtained from patient files and electronic patient data system (SARUS).
At the end of the operation, the neuromuscular blockade was reversed by the intravenous administration of 0.05 mg kg-1 neostigmine and 0.02 mg kg-1 atropine to the patients in Group N and 2 mg kg-1 sugammadex to the patients in Group S after the reappearance of the second twitch (T2) on the TOF (Train-of-Four). Patients with a T4/T1 (TOF≥0.9) ratio greater than 90% were included in this study.
The patients were divided into two groups as sugammadex (Group S) and neostigmine (Group N). Patients with available data of preoperative and postoperative levels of total IgE and ECP were included in this study. Patients with a history of allergy (allergic rhinitis, urticaria, and atopic eczema) and renal failure were not included in this study.
Assessments
Data were recorded, including patient demographics (age, gender) and preoperative and postoperative (12 hours later) total IgE (low: 0–100 IU ml-1, high:>100 IU ml-1) and ECP levels (low:0–24 µg L-1, high:>24µg L-1), to make intragroup and intergroup comparisons.
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics for Windows, Version 22.0 (IBM Corp., Armonk, NY). Pearson chi-square and Fisher’s exact tests were performed to analyze the categorical variables. Conformity of the data to normal distribution was assessed using the Shapiro-Wilk test. Student’s t-test and the Mann-Whitney U test was used for comparing the variables that do not follow normal distribution between the groups. Wilcoxon Signed Ranks Test was used for comparing the postoperative and preoperative values of the parameters. Spearman correlation coefficient was used for investigating the correlation between the continuous variables. Data were expressed as n (%), mean±standard deviation (SD) or median (min-max) when appropriate. P-values of <0.05 were considered statistically significant. In the literature, to our knowledge, there is no study evaluating IgE and ECP parameters in sugammadex-induced reactions. Therefore, power analysis could not be calculated.
RESULTS
In this study, 83 patients were included. The mean age was 35.12 years (range, 17 to 61 years). Females accounted for 74.7% of the study population. Sugammadex (Group S) and Neostigmine (Group N) was administered to 50.6% and 49.39% of the patients, respectively. No statistical differences were observed in age, gender, BMI, total IgE and ECP levels, and the levels of Ringer lactate between the groups (p=0.723, p>0.05) (Table 1).
TABLE 1.
Comparison of patients’ demographics and clinical characteristics
| Patient characteristics | Total (n=83) | Group N (n=41) | Group S (n=42) | p |
|---|---|---|---|---|
| Age | 35.12±12.13 | 36.61±13.02 | 33.67±11.17 | 0.272 |
| Gender | ||||
| Male | 21 (25.3) | 9 (22) | 12 (28.6) | |
| Female | 62 (74.7) | 32 (78) | 30 (71.4) | 0.488 |
| BMI | 45.34±5.74 | 44.72±5.42 | 45.94±6.05 | 0.334 |
| Pre-Total IgE | ||||
| 0-100 IU ml-1 | 62 (74.7) | 31 (75.6) | 31 (73.8) | 0.850 |
| >100 IU ml-1 | 21 (25.3) | 10 (24.4) | 11 (26.2) | |
| Pre ECP | ||||
| 0-24 µg L-1 | 55 (66.3) | 28 (68.3) | 27 (64.3) | 0.699 |
| >24 µg L-1 | 28 (33.7) | 13 (31.7) | 15 (35.7) | |
| Ringer lactate | 1200 (1000–1600) | 1200 (1000–1350) | 1200 (1000–1600) | 0.723 |
Data are presented as n (%); mean±SD and median (min-max); SD: standard deviation; Student’s t-test; Mann-Whitney U test; Fisher’s Exact test; Pearson chi-square test. BMI: Body mass index; Pre: Preoperative.
The preoperative and postoperative levels of total IgE and ECP were not statistically different between the groups (p>0.05). There was not a significant change in the total IgE levels in Group S or Group N (p>0.05); however, the levels of ECP significantly decreased in both groups (p<0.001, p=0.01) (Table 2).
TABLE 2.
Comparison of total IgE and ECP values between groups
| Eosinophil cationic protein, Total IgE values | Group N (n=41) | Group S (n=42) | p |
|---|---|---|---|
| Pre-Total IgE (IU ml-1) | 44.8 (17.3–5210) | 34.05 (17.3–3170) | 0.655 |
| Post Total IgE (IU ml-1) | 49.2 (12.5–4990) | 39.95 (17.3–3540) | 0.844 |
| p | 0.327 | 0.694 | |
| Pre ECP (µg L-1) | 15.3 (3.64–71.5) | 16.4 (3–49.1) | 0.642 |
| Post ECP (µg L-1) | 7.81 (1.31–91.3) | 9.88 (1.65–142) | 0.492 |
| p | <0.001 | 0.010 |
Data are presented as median (min-max); Wilcoxon Signed Ranks test; Mann-Whitney U test; Pre: Preoperative; Post: Postoperative.
A comparison of the preoperative and postoperative values of the patients by the group revealed that total IgE and ECP values did not change significantly (p=0.336, p=0.303) (Table 3).
TABLE 3.
Comparison of differences in the study parameters between the groups
| ECP, Total IgE changes | Group N (n=41) | Group S (n=42) | p |
|---|---|---|---|
| Difference in Total IgE levels | 0 (-220–109.4) | 0 (-65–490) | 0.336 |
| Difference in ECP levels | -6.34 (-46.5–71.4) | -4.83 (-37.49–111.3) | 0.303 |
Data are presented as median (min- max); Mann-Whitney U test.
There was a positive and weak correlation of the preoperative total IgE levels with the preoperative ECP (p=0.221, p=0.044) and postoperative ECP (p=0.240, p=0.029) levels statistically significantly in all patients. The postoperative levels of total IgE and ECP showed a positive and weak correlation (p=0.235, p=0.033). In Group N, no statistically significant correlations were found between the preoperative and postoperative total IgE and ECP levels. In Group S, there was a positive, weak, and statistically significant correlation of the preoperative levels of total IgE with the preoperative levels of ECP (p=0.311, p=0.045) and the postoperative ECP levels (p=0.310, p=0.046) (Table 4).
TABLE 4.
Correlation between the preoperative and postoperative total IgE and ECP levels
| ECP, Total IgE changes correlation IgE-ECP Levels | Total | Group N | Group S | |||
|---|---|---|---|---|---|---|
| ρ | p | ρ | p | ρ | p | |
| Pre IgE-Pre ECP | 0.221 | 0.044 | 0.169 | 0.290 | 0.311 | 0.045 |
| Pre IgE-Post ECP | 0.240 | 0.029 | 0.162 | 0.313 | 0.310 | 0.046 |
| Post IgE-Pre ECP | 0.186 | 0.091 | 0.197 | 0.216 | 0.191 | 0.227 |
| Post IgE-Post ECP | 0.235 | 0.033 | 0.214 | 0.178 | 0.254 | 0.105 |
Spearman correlation test; Pre: Preoperative; Post: Postoperative; (ρ): Spearman’s Rank Correlation Coefficient.
DISCUSSION
In this retrospective cohort study on morbidly obese patients, who underwent sleeve gastrectomy, normal levels of total IgE (0–100 IU ml-1) were found in 62 (74.7%) patients and IgE levels of >100 IU ml-1 were found in 21 (25.3%) in the preoperative period. The normal levels of ECP (0–24 µg L-1) were found in 55 (66.3%) patients and ECP levels of >24 µg L-1 were found in 28 (33.7%) patients. Although we did not include allergic and atopic patients, in our study, morbid obesity is a factor increasing the patient susceptibility to develop symptoms of bronchial asthma [12–15]. Distal airway obstruction is quite common in morbidly obese patients. The most sensitive indicator of distal airway obstruction in the early period is FEF25–75 (FEF25–75 <%70) measured by spirometry. Normal adult spirometry and Distal airway obstruction spirometry (Morbidly obese patient FEF25–75 <%70) [respectively (Figs. 1, 2)]. Although elevated levels of serum IgE and ECP can be seen in allergic patients or patients with an atopic constitution [11, 16], no statistically significant increases were observed in the preoperative and postoperative levels of total IgE and ECP in this study. Besides, no bronchospasm or urticaria-like symptoms were observed clinically in none of the study groups. The preoperative levels of total IgE and ECP were statistically significantly correlated with the postoperative ECP levels in all patients. The correlation was positive and weak. Despite the absence of a statistically significant correlation between the preoperative and postoperative total IgE and ECP values in Group N, there was a positive, weak, and a statistically significant correlation between the preoperative total IgE levels and ECP levels in Group S.
FIGURE 1.

Normal adult spirometry.
FIGURE 2.
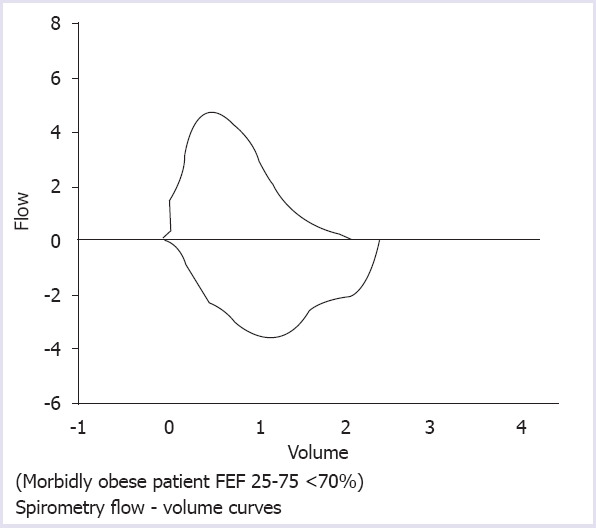
Distal airway obstruction.
Recent studies in the literature have related sugammadex-associated hypersensitivity reactions with γ-cyclodextrin (γCD) [7]. γ-cyclodextrin (γCD) can be ingested with food as it is used as a solvent or stabilizer in several types of food and pharmaceutical products. It is a cyclic oligosaccharide and it is produced after bacterial digestion of starch [17]. γCD has a toxic profile compared to natural αCD and βCD [17]. Ingestion of cyclodextrin containing agents like flavors, vitamins, colorants, and unsaturated fats [6] via food and production of γCD by the intestinal flora starting from childhood can be considered a type of oral immunotherapy [18]. Oral immunotherapy develops natural tolerance [18], and consequently, a normal allergic substance may cause either a minor allergic reaction or none at all. Although γCD is safe to use in foods [19], information about intravenous use is insufficient. Recent comparative studies with sugammadex and neostigmine appear to support our study. It is reported that sugammadex has a favorable tolerability profile in adults, excluding rare cases [8, 20]. In contrast, hydrophobic molecules formed by gamma-cyclodextrin may penetrate body tissues may cause the release of active molecules. Anaphylaxis reactions with sugammadex-rocuronium complex [4, 21, 22] as well as anaphylaxis reactions with rocuronium [23] are available in the literature, which leads to confusion as to which is the real trigger in anaphylaxis. In the literature, the anaphylaxis reaction associated with sugammadex is not IgE/IgG-mediated [24]. Pathophysiology of anaphylaxis and anaphylactoid reactions is presented in Figure 3. There are also studies reporting the use of sugammadex in the treatment of rocuronium-induced anaphylaxis and does not trigger mast cell degranulation but causes a slight increase in the number [25]. McDonnell, Funnell et al. also used sugammadex (respectively 500 mg and 400 mg sugammadex doses) to treat rocuronium-induced anaphylaxis [25, 26]. In the study of Menendez-Ozcoidi et al., erythema, edema, hypotension, tachycardia and desaturation were observed after intravenous administration of 200 mg (3.2 mg/kg) sugammadex in one patient [27]. In a retrospective analysis comparing sugammadex and neostigmine, 2 mg kg-1 and 4 mg kg-1 doses proved to have similar hypersensitivity rates to placebo [8]. In contrast, there are studies showing signs of anaphylaxis with the administration of 4 mg kg-1 and 16 mg kg-1 (recommended dose for an immediate reversal in emergency situations) sugammadex [24]. In our study, with 2 mg kg-1, sugammadex dose did not develop symptoms of anaphylaxis, although the mechanism has not been fully elucidated, we also found that sugammadex was safely administered in obese adult patients. Certain limitations of this study should be noted. First, postoperative tryptase enzyme activity [1] and specific IgE levels, commonly tested to evaluate acute hypersensitivity and anaphylactic reactions, could not be quantified due to the retrospective and single-center design of our study. The positive aspect of our study is that it enrolled morbidly obese patients who were not previously diagnosed with bronchial asthma but had an increased susceptibility to develop its symptoms due to morbid obesity.
FIGURE 3.
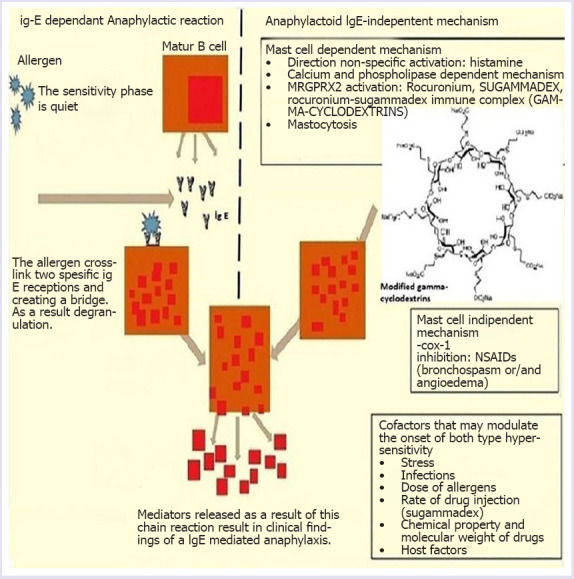
Pathophysiology of anaphylaxis and anaphylactoid reactions.
Conclusion
In conclusion, our retrospective cohort study found out that the preoperative values of total IgE and ECP were positively, weakly, and statistically significantly correlated with the postoperative ECP values in the Group S morbid obesity patients, who underwent sleeve gastrectomy and who were administered sugammadex intravenously to reverse the neuromuscular blockade. No significant elevations in the total IgE and ECP levels were observed after intravenous sugammadex administration. Sugammadex is highly safe in patients, desensitizied naturally to gamma-cyclodextrin. Anaphylactoid reaction was not observed after administration of 2 mg kg-1 sugammadex intravenously in patients with morbid obesity. There is a need for experimental animal studies or prospective larger studies on humans to better evaluate the sugammadex or γ-cyclodextrin-associated may be an anaphylactoid reaction.
Footnotes
Ethics Committee Approval: This study was conducted in accordance with the ethical principles stated in the “Declaration of Helsinki” and permission was obtained from Ethics Committee of Antalya Training and Research Hospital for the use of patient data for publication purposes (Date-Decision no: 04.07. 2019-16/14).
Conflict of Interest: No conflict of interest was declared by the authors.
Financial Disclosure: The authors declared that this study has received no financial support.
Authorship Contributions: Concept – OG; Design – OG; Supervision – OG; Fundings – OG; Materials – MNK; Data collection and/or processing – MNK; Analysis and/or interpretation – OG; Literature review – OG; Writing – OG; Critical review – MNK.
REFERENCES
- 1.O'Donnell R, Hammond J, Soltanifar S. A confirmed case of sugammadex-induced anaphylaxis in a UK hospital. BMJ Case Rep. 2017;2017:bcr2017220197. doi: 10.1136/bcr-2017-220197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sharma S, Tomar S, Dharne M, Ganesan V, Smith A, Yang Y, et al. Deletion of ΔdblGata motif leads to increased predisposition and severity of IgE-mediated food-induced anaphylaxis response. PLoS One. 2019;14:e0219375. doi: 10.1371/journal.pone.0219375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tobias JD. Current evidence for the use of sugammadex in children. Paediatr Anaesth. 2017;27:118–25. doi: 10.1111/pan.13050. [DOI] [PubMed] [Google Scholar]
- 4.Takazawa T, Mitsuhata H, Mertes PM. Sugammadex and rocuronium-induced anaphylaxis. J Anesth. 2016;30:290–7. doi: 10.1007/s00540-015-2105-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Akha AS, Rosa J, 3rd, Jahr JS, Li A, Kiai K. Sugammadex:cyclodextrins development of selective binding agents pharmacology, clinical development and future directions. Anesthesiol Clin. 2010;28:691–708. doi: 10.1016/j.anclin.2010.08.014. [DOI] [PubMed] [Google Scholar]
- 6.Szente L, Szejtli J. Cyclodextrins as food ingredients. Food Sci Technol. 2004;15:137–42. [Google Scholar]
- 7.Hotta E, Tamagawa-Mineoka R, Masuda K, Taura M, Nakagawa Y, Kanehisa F, et al. Anaphylaxis caused by γ-cyclodextrin in sugammadex. Allergol Int. 2016;65:356–8. doi: 10.1016/j.alit.2016.02.013. [DOI] [PubMed] [Google Scholar]
- 8.Min KC, Woo T, Assaid C, McCrea J, Gurner DM, Sisk CM, et al. Incidence of hypersensitivity and anaphylaxis with sugammadex. J Clin Anesth. 2018;47:67–73. doi: 10.1016/j.jclinane.2018.03.018. [DOI] [PubMed] [Google Scholar]
- 9.Mineeva EE, Antonyuk MV, Yurenko AV, Gvozdenko TA. Functional state of the small airways in patients with bronchial asthma associated with obesity. Ter Arkh. 2019;91:60–3. doi: 10.26442/00403660.2019.01.000031. [DOI] [PubMed] [Google Scholar]
- 10.De La Cruz I, Errando C, Calaforra S. Treatment of Anaphylaxis to Rocuronium with Sugammadex:A Case Report with Bronchospasm as the Only Symptom. Turk J Anaesthesiol Reanim. 2019;47:69–72. doi: 10.5152/TJAR.2019.21298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shin YH, Kim HS, Lee EK, Kim YJ, Lee HS, Jang PS, et al. Eosinophil-related markers and total immunoglobulin E as a predictive marker for antibiotic response in chronic rhinosinusitis. Ann Saudi Med. 2015;35:312–7. doi: 10.5144/0256-4947.2015.312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Guo CL, Sun XM, Wang XW, Guo Q. Serum Eosinophil Cationic Protein Is a Useful Marker for Assessing the Efficacy of Inhaled Corticosteroid Therapy in Children with Bronchial Asthma. Tohoku J Exp Med. 2017;242:263–71. doi: 10.1620/tjem.242.263. [DOI] [PubMed] [Google Scholar]
- 13.Vaillant AAJ, Zito PM. Immediate Hypersensitivity Reactions. StatPearls Publishing LLC. 2019 [PubMed] [Google Scholar]
- 14.Kırgezen T, Server EA, Turanoğlu FS, Yiğit Ö, Uzun H, Durmuş S. Salivary Eosinophil Cationic Protein in Allergic Rhinitis. Turk Arch Otorhinolaryngol. 2019;57:91–4. doi: 10.5152/tao.2019.4127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jiang XG, Yang XD, Lv Z, Zhuang PH. Elevated serum levels of TNF-α IL-8, and ECP can be involved in the development and progression of bronchial asthma. J Asthma. 2018;55:111–8. doi: 10.1080/02770903.2017.1318141. [DOI] [PubMed] [Google Scholar]
- 16.Lodin K, Lekander M, Syk J, Alving K, Andreasson A. Associations between self-rated health sickness behaviour and inflammatory markers in primary care patients with allergic asthma:a longitudinal study. NPJ Prim Care Respir Med. 2017;27:67. doi: 10.1038/s41533-017-0068-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Saokham P, Loftsson T. γ-Cyclodextrin. Int J Pharms. 2017;516:278–92. doi: 10.1016/j.ijpharm.2016.10.062. [DOI] [PubMed] [Google Scholar]
- 18.Beyer K, Wahn U. Oral immunotherapy for food allergy in children. Curr Opin Allergy Clin Immunol. 2008;8:553–6. doi: 10.1097/ACI.0b013e32831952c8. [DOI] [PubMed] [Google Scholar]
- 19.Munro IC, Newberne PM, Young VR, Bär A. Safety assessment of gamma-cyclodextrin. Regul Toxicol Pharmacol. 2004;39(Suppl 1):S3–13. doi: 10.1016/j.yrtph.2004.05.008. [DOI] [PubMed] [Google Scholar]
- 20.Honing G, Martini CH, Bom A, van Velzen M, Niesters M, Aarts L, et al. Safety of sugammadex for reversal of neuromuscular block. Expert Opin Drug Saf. 2019;18:883–91. doi: 10.1080/14740338.2019.1649393. [DOI] [PubMed] [Google Scholar]
- 21.Kim GH, Choi WS, Kim JE, Yun MJ, Koo MS, Kwon M, et al. Anaphylactic shock after sugammadex administration induced by formation of a sugammadex-rocuronium complex -a case report. Korean J Anesthesiol. 2019;72:495–9. doi: 10.4097/kja.d.18.00232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Baldo BA. Anaphylaxis caused by sugammadex- rocuronium inclusion complex:What is the basis of the allergenic recognition? J Clin Anesth. 2019;54:48–9. doi: 10.1016/j.jclinane.2018.10.017. [DOI] [PubMed] [Google Scholar]
- 23.Hashimoto M, Sato Boku A, Tachi N, Okumura Y, Kadoi K, Harada J, et al. Two Cases of Rocuronium-Induced Anaphylaxis/Anaphylactic Shock Successfully Treated With Sugammadex. Anesth Prog. 2019;66:151–5. doi: 10.2344/anpr-66-01-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.de Kam PJ, Nolte H, Good S, Yunan M, Williams-Herman DE, Burggraaf J, et al. Sugammadex hypersensitivity and underlying mechanisms:a randomised study of healthy non-anaesthetised volunteers. Br J Anaesth. 2018;121:758–67. doi: 10.1016/j.bja.2018.05.057. [DOI] [PubMed] [Google Scholar]
- 25.McDonnell NJ, Pavy TJ, Green LK, Platt PR. Sugammadex in the management of rocuronium-induced anaphylaxis. Br J Anaesth. 2011;106:199–201. doi: 10.1093/bja/aeq366. [DOI] [PubMed] [Google Scholar]
- 26.Funnell AE, Griffiths J, Hodzovic I. A further case of rocuronium-induced anaphylaxis treated with sugammadex. Br J Anaesth. 2011;107:275–6. doi: 10.1093/bja/aer211. [DOI] [PubMed] [Google Scholar]
- 27.Menéndez-Ozcoidi L, Ortiz-Gómez JR, Olaguibel-Ribero JM, Salvador-Bravo MJ. Allergy to low dose sugammadex. Anaesthesia. 2011;66:217–9. doi: 10.1111/j.1365-2044.2010.06611.x. [DOI] [PubMed] [Google Scholar]


