Abstract
The seasonal influenza vaccine is an important public health tool but is only effective in a subset of individuals. The identification of molecular signatures provides a mechanism to understand the drivers of vaccine-induced immunity. Most previously reported molecular signatures of human influenza vaccination were derived from a single age group or season, ignoring the effects of immunosenescence or vaccine composition. Thus, it remains unclear how immune signatures of vaccine response change with age across multiple seasons. Here we profile the transcriptional landscape of young and older adults over five consecutive vaccination seasons to identify shared signatures of vaccine response as well as marked seasonal differences. Along with substantial variability in vaccine-induced signatures across seasons, we uncovered a common transcriptional signature 28 days post-vaccination in both young and older adults. However, gene expression patterns associated with vaccine-induced antibody responses were distinct in young and older adults; for example, increased expression of Killer Cell Lectin Like Receptor B1 (KLRB1; CD161) 28 days post-vaccination positively and negatively predicted vaccine-induced antibody responses in young and older adults, respectively. These findings contribute new insights for developing more effective influenza vaccines, particularly in older adults.
Keywords: systems vaccinology, seasonal variability, influenza, vaccination, aging, transcriptional profiling
Introduction
Influenza is a major public health burden, particularly in high-risk populations such as older adults. The seasonal inactivated influenza vaccination (IIV) is estimated to be 50–70% effective in randomized controlled trials of young adults (1–5), and efficacy is reduced to under 50% in adults over age 65 (6). Understanding the dynamics of vaccination-induced immune responses, and the factors associated with immunological protection should provide insights important for improving vaccine design.
Systems vaccinology approaches utilizing high-throughput immune profiling techniques have identified signatures of response to influenza vaccination (7–14). These include pre-vaccination transcriptional signatures of apoptosis-related gene modules (9), as well as B cell signaling and inflammatory modules (15). Post-vaccination transcriptional signatures have also been identified, including an early interferon response 1 day post-vaccination and a plasma cell response 3 and 7 days post-vaccination (13). Interferon stimulated genes were upregulated in both monocytes and neutrophils between 15 and 48 hours post-vaccination and correlated with influenza-specific antibody responses (7, 12). In addition, the expression of genes enriched for proliferation and immunoglobulin production 7 days post-vaccination accurately predicted antibody response in an independent cohort (10). Studies of the influence of aging revealed that an early interferon response 1–2 days post-vaccination as well as an oxidative phosphorylation and plasma cell response 7 days post-vaccination were correlated with antibody response in young adults but were diminished or dysregulated in older adults (13, 14).
Notably, previous studies of influenza vaccine response studying the effects of aging used data from a single vaccine season (9) or from two consecutive seasons in which vaccine composition was identical (13, 14); consequently, the generalizability of these signatures is unknown. To date, no comprehensive characterization of vaccine response in both young and older adults has been reported to multiple influenza vaccines which vary in composition. To address this gap, we profiled young and older adults over five consecutive vaccination seasons (2010–11, 2011–12, 2012–13, 2013–14, and 2014–15) hereafter referred to by the first year of each season. We developed a new automated metric to quantify antibody response while accounting for baseline titers and used this novel metric to identify predictive transcriptional signatures of vaccine response using post-vaccination as well as baseline gene expression profiles.
Materials and Methods
Clinical Study Design and Specimen Collection
A total of 317 subjects were recruited at Yale University over the five vaccination seasons between 2010 and 2014 and HAI titers pre- (D0) and post-vaccination (D28) were available from the 294 subjects reported in Table 1. Informed consent was obtained for all subjects under a protocol approved by the Human Subjects Research Protection Program of the Yale School of Medicine. Participants with an acute illness two weeks prior to recruitment were excluded from the study, as were individuals with primary or acquired immune-deficiency, use of immunomodulating medications including steroids or chemotherapy, a history of malignancy other than localized skin or prostate cancer, or a history of cirrhosis or renal failure requiring hemodialysis. Blood samples were collected into Vacutainer sodium heparin tubes and serum tubes (Becton Dickinson) at four different time points, immediately prior to administration of vaccine (D0) and on D2 (2011, 2012, 2013, 2014) or D4 (2010), D7, and D28 post-vaccination.
Table 1.
Vaccine Compositions and Cohorts
| 2010–11 | 2011–12 | 2012–13 | 2013–14 | 2014–15 | |
|---|---|---|---|---|---|
| Vaccine Compositiona | A/California/7/2009 | A/California/7/2009 | A/California/7/2009 | A/California/7/2009 | A/California/7/2009 |
| A/Perth/16/2009 | A/Perth/16/2009 | A/Victoria/361/2011 | A/Texas/50/2012 | A/Texas/50/2012 | |
| B/Brisbane/60/2008 | B/Brisbane/60/2008 | B/Wisconsin/1/2010 | B/Brisbane/60/2008 | B/Brisbane/60/2008 | |
| B/Massachusetts/2/2012 | B/Massachusetts/2/2012 | ||||
| Subjects | 42 | 69 | 92 | 56 | 35 |
| Gender (% Male) | 33 | 42 | 40 | 36 | 51 |
| Age Group (% Older) | 48 | 54 | 49 | 52 | 46 |
| Transcriptomesb | 19 | 39 | 30 | 26 | 20 |
| Young (LR | I | HR)c | 4 | 1 | 6 | 8 | 2 | 6 | 6 | 0 | 9 | 6 | 2 | 5 | 4 | 2 | 5 |
| Older (LR | I | HR)c | 5 | 0 | 3 | 11 | 5 | 7 | 7 | 0 | 8 | 7 | 0 | 6 | 2 | 0 | 7 |
The three vaccine strains in 2009–10 were A/Brisbane/59/2007, A/Brisbane/10/2007, and B/Brisbane/60/2008. A monovalent A/California/7/2009 vaccine was administered to some subjects in March 2010.
Subjects with transcriptional data are a subset of subjects with antibody titers.
Subjects are listed by antibody response category: low responder (LR), indeterminate (I), high responder (HR).
In order to understand the transcriptional program underlying a successful vaccination response, we identified a subset of 134 subjects with extreme (strong or weak) antibody responses to perform transcriptional profiling by microarrays. In the first three seasons, the selection criteria were a four-fold increase to at least 2 strains (strong response) or no four-fold increase to any strain (weak response) as described previously (14). In the fourth and fifth seasons, the adjMFC metric was used in addition to the fold change criteria to account for baseline titers (11). The maxRBA response endpoint was developed after the study completed, however, less than 10% (12/134) of subjects chosen for transcriptional profiling had indeterminate responses by maxRBA (neither high or low responders using a 40% cutoff) (Table 1). These 12 subjects were excluded from the predictive modeling of antibody response.
Peripheral Blood Mononuclear Cell (PBMC) Isolation
Blood samples were collected in sodium heparin tubes (BD Biosciences) from volunteers with prior consent to an IRB approved protocol. About 20 ml of fresh blood were layered on top of 20 ml Histopaque-1077 solution (Sigma-Aldrich). Samples were carefully transferred to a centrifuge (Legend XT, Thermo Fisher Scientific) and centrifuged for 20 minutes at room temperature without brake at 2200 rpm. Buffy coats containing peripheral blood mononuclear cells (PBMC) at the interface were carefully collected to 50 ml RPMI medium with 10% BSA. Samples were centrifuged to pellet cells and the supernatant were discarded. Lymphocyte pellets were re-suspended in 50 ml RPMI containing 10% FBS. A 20-microliter volume of suspended PMBC was mixed with equal volume Trypan Blue solution (Thermo Fisher Scientific) and incubated at room temperature for about 2 minutes. A 20-microliter cell suspension was immediately counted using Haemocytometer under light microscope. Percent viability was determined by the formula (Number of total cells counted – Number of Blue cells counted) x 100. Samples with 95% or more viability were selected for all experiments including RNA extraction.
PBMC RNA Extraction
About 10 million cells (PBMC) in 1 ml RPMI medium from samples with 95% or more viability were taken soon after Histopaque-1077 density gradient separation for RNA extraction. PBMC in RPMI medium were centrifuged for about 10 minutes on a bench top centrifuge at 10,000 rpm. Supernatant was removed carefully from each sample to ensure a clear pellet of PBMC without residual RPMI medium. To each pellet about 700 microliters of QIAzol lysis reagent (Qiagen) was added and mixed by pipetting at least 10 times to ensure proper cell lysis. Lysed cells were immediately frozen at −80° C until further extraction using QIAcube instrument (Qiagen).
QIAcube RNA Extraction Protocol
All RNA samples were extracted using miRNeasy kit (cat. 217004, Qiagen) following the instructions provided using a QIAcube (Qiagen). Briefly, samples lysed in QIAzol reagent were incubated for 5 minutes at room temperature (15° C - 25° C). To each sample about 140 microliters of chloroform was added and shaken vigorously and left at room temperature for about 2–3 minutes. Subsequently, samples were centrifuged at 4° C at 12,000 x g for 15 minutes. Upper aqueous phase containing the RNA species were carefully transferred to a 2 ml collection tube (cat. 990381) Qiagen without touching the interphase and placed in the QIAcube for extraction. For every sample a rotor adapter was prepared with a RNeasy mini spin column at position L1 and a 1.5 ml collection tube at position L3 and placed in the QIAcube rotor. All reagents were prepared by adding proper amount of 100% ethanol (44 ml to buffer RPE and 30 ml to buffer RWT) prior to extraction and placed in respective positions in reagent rack in QIAcube (100% ethanol in position 3, Buffer RWT in position 4, buffer RPE in position 5 and RNase-free water in position 6). RNA extraction was carried out by executing recommended protocol (FIW-50–001-J_FW_MB and PLC program version FIW-50–002-G_PLC_MB) available from the QIAcube web portal. RNA samples with RIN value above 7.0 were used for gene expression analysis.
RNA Isolation and Cell Sorting
Peripheral Blood Mononuclear Cells (PBMCs) isolation and RNA preparation on freshly isolated PBMCs was performed as described (1). For cell sorting, frozen PBMCs were thawed, washed, and stained using antibodies directed against: CD19(HIB19), CD4(SK3), CD20(2H7), CD8(SK1), and CD3(UCHT1) (from BD Biosciences) and using a BD Fortessa instrument. T and B cells were sorted from a subgroup of samples in seasons 2010–2012. We evaluated the purity of 268 / 270 sorts for CD4+ T cells with median purity of 97%, 254 / 254 sorts for CD8+ T cells with median purity of 97%, and 252 / 256 sorts for B cells with median purity of 99%. 90% of the samples had a post-sort purity of at least 90% for all 3 cell types. Very few samples had a purity of less than 80% (0.4%, 1.1%, and 5.6% for CD4 T cells, CD8 T cells, and B cells, respectively).
HAI and VNA Titer Measurements and Response Endpoint Definition
Serum samples were collected pre-vaccination (D0) and 28 days post-vaccination (D28). Hemagglutination inhibition (HAI) assays were performed as previously described (2). Virus neutralization assays (VNA) were performed as described elsewhere (3, 4). Briefly, two-fold dilutions (50 μL) of the RDE-treated sera in sterile Opti-MEM (Invitrogen, Carlsbad, CA) were mixed with 200 PFU of influenza virus (5 μL). The serum-virus samples were then incubated at room temperature for 60 minutes to allow any HA-specific antibodies present in the serum to neutralize the influenza virus. The serum-virus samples (55 μL) were then transferred to MDCK cell cultures cultured in 96-wll flat bottom plates. Following virus absorption for 60 minutes, the serum-virus inocula were removed, and the MDCK cells were cultured for 4 days in Opti-MEM supplemented with 1 μg/ml of tosylsulfonyl phenylalanyl chloromethyl ketone (TPCK)-trypsin (Sigma-Aldrich). Virus production was determined by HA assay. The neutralization titer was defined as the reciprocal of the highest dilution of serum that neutralizes 200 PFU of influenza virus.
To adjust for inverse correlations between HAI titer fold changes and baseline titers, we developed an automated metric: maximum Residual after Baseline Adjustment (maxRBA). First, young and older cohorts were separated, and endpoints were calculated in each season and each age group separately. Any fold changes less than 1 were set to 1 since we did not expect the number of antibodies in the blood to decrease by 2-fold in the 28 days after vaccination and this was likely due to measurement error. Next, the baseline and fold changes were log2 transformed and an exponential curve was fit to the fold change vs. baseline titers for each strain. Then the residuals were calculated and for each subject the maximum residual across all strains was selected as the maxRBA. Finally, “high” and “low” responders were defined as the top and bottom 40th percent of maxRBA, respectively. The maxRBA values presented in Fig. 1 and Supplemental Fig. 3 were calculated by combining young and older adults within each season to allow for comparison across age groups. The code to calculate maxRBA is available in the Calculate_maxRBA() function from the titer R package (https://bitbucket.org/kleinstein/titer).
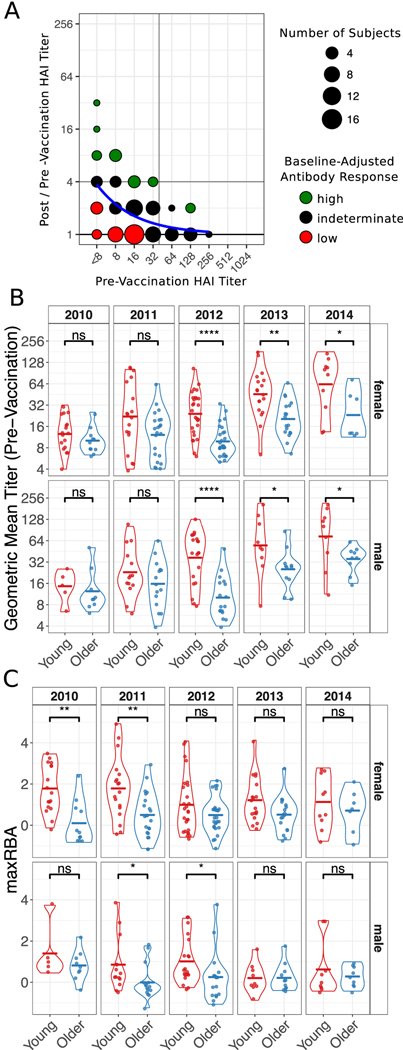
Figure 1. Influenza-Specific Antibody Titers.
(A) An illustration of the maximum Residual after Baseline Adjustment (maxRBA) method for hemagglutination inhibition (HAI) titers. An exponential curve (blue) is fit to the data and the residual is used to stratify subjects into high and low responders. Subjects with largest positive residuals are high responders (green) and subjects with smallest negative residuals are low responders (red). Unlike the illustration, maxRBA is calculated using the maximum residual across all vaccine strains. (B and C) Violin plots of pre-vaccination HAI titers (B) and HAI responses measured by maxRBA (C) are separated by season and gender to compare age groups. Crossbars indicate the mean. Not Significant (ns) p > 0.05, * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001 independent two-sided Wilcoxon rank sum test.
RNA Processing and Microarrays
Each RNA sample was quantified, and integrity assessed by the Agilent 2100 BioAnalyser (Agilent, CA). Samples were processed for cRNA generation using the Illumina TotalPrep cRNA Amplification Kit and subsequently hybridized to the Human HT12-V4.0 BeadChip (Illumina, CA). For gene expression analyses, samples were processed and hybridized to HumanHT-12v4 Expression BeadChip (Illumina San Diego, CA). Arrays were processed at Yale’s Keck Biotechnology Resource Laboratory and raw expression data were output using Illumina GenomeStudio software. Samples from each season were processed in batches and all samples from each subject were run on the same chip to mitigate batch effects. Data from each season are available via ImmPort (https://www.immport.org) under accession numbers SDY63, SDY404, SDY400, SDY520, and SDY640. Microarray data are available through the Gene Expression Omnibus (GEO) Database (https://www.ncbi.nlm.nih.gov/geo/) with accession numbers GSE59635, GSE59654, GSE59743, GSE101709, and GSE101710 for PBMC data and GSE65440, GSE65442, and GSE95584 for B and T cell data. Data from seasons 2010 and 2011 (GSE59635 and GSE59654) were previously published (1) as well as data from D0 and D7 timepoints from the 2012 season (GSE59743) (5). The D0 expression in PBMC (GSE59635, GSE59654, GSE59743, GSE101709, and GSE101710) of a single gene, MINCLE, was also recently published (6). The remainder of the data in this work has not been previously published. The data were quantile normalized and log2 transformed within each season. Multiple probes were collapsed to unique Entrez Gene IDs by selecting the probe with the highest average expression. The Bioconductor package illuminaHumanv4.db version 1.26 was used to map the probes (7).
Gene Module and Differential Expression Analysis
Differentially expressed modules (DEMs) were defined at each post-vaccination time-point using QuSAGE with an FDR < 0.05 (8). Differentially expressed genes (DEGs) were defined at each post-vaccination time-point using two criteria: (1) an absolute fold-change of at least 1.25 relative to the pre-vaccination time point and (2) a significant change in expression by limma (version 3.30.7) after correction for multiple hypothesis testing (FDR < 0.05) (9). Clusters of DEGs were determined by hierarchical clustering using Ward’s minimum variance method with the distance between any 2 genes defined by 1 less the Pearson correlation coefficient. Enrichment of DEG clusters was performed using EnrichR v1.0 with the following databases: GO_Cellular_Component_2018, GO_Biological_Process_2018, GO_Molecular_Function_2018, KEGG_2016, and Reactome_2016 (10). Enrichment of principal components was performed using the geneSetTest function in the limma R package v3.24.15 (9). Control of the false discovery rate was performed according to the procedure in (11) unless otherwise noted.
Meta-Analysis
First, genes were filtered to those that were detected in at least 20% of samples with a p-value below 0.05. For the gene module meta-analysis, QuSAGE activity distributions were taken from the single season analysis and convoluted to create a probability density function for the meta-analysis (12). Modules with an FDR < 0.10 in the meta-analysis were chosen as significant. For the single-gene meta-analysis, a random-effects model was fit for every gene, at each time point, in each age group separately. This model allows for variation in effect sizes across seasons. Effect sizes (mean differences) were calculated for every gene in each season separately and then combined using the rma() function from the metafor R package (13). The restricted maximum-likelihood estimator for the amount of heterogeneity was used (14). Genes with an FDR < 0.05 in the meta-analysis were chosen as significant.
Predicting Antibody Response from Transcriptional Profiles
Because females tended to respond better than males, genes located on the X and Y chromosomes were removed to avoid selection of sex-linked genes that may be confounded with vaccine response. For baseline predictors, the 1,000 genes with the largest coefficient of variation were selected as the initial feature set. For post-vaccination predictors, the log fold-change from D0 was calculated for each gene, and the 1,000 genes with the largest fold-change magnitudes were selected as the initial feature set. The baseline or fold-changes were standardized by subtracting the mean and dividing by the standard deviation. Finally, this preprocessed data from each individual season was combined to form the discovery data. The young adult models were tested on GSE47353 at baseline, day 1, day 7, and day 70 while the older adult models were tested on GSE41080 at baseline and GSE74813 at baseline, day 1, day 7, and day 14 post-vaccination. The Logistic Multiple Network-constrained Regression (LogMiNeR) framework was performed as described (5). Briefly, 5-fold cross validation was used to select the optimal tuning parameters and 50 iterations of cross validation were performed on different splits of the discovery data set. The prior knowledge networks were defined for Reactome (15, 16), Gene Ontology (GO) (17), Blood Transcriptional Modules (BTM) (18) and cell-specific signatures (CELLS) (19) by connecting all pairs of genes within each gene set. The KEGG network incorporated pathway topology and was built using the KEGGgraph R package v1.26.0 (20). ImmuneGlobal (ImmuNet) and ExpOnly_ImmuneGlobal (ImmuNet_Exp) networks were obtained from ImmuNet and edges were restricted to those with confidence of at least 0.1 (21). The STRING network incorporated all experimental evidence from the STRING database v10.0 (22).
Comparison Against Published Signatures
Influenza vaccination signatures were manually curated from several publications (23–28). Differentially expressed genes (DEGs) were defined for each contrast using a significant change in expression by limma (version 3.30.7) (p < 0.001). Differentially expressed modules were defined as described above using QuSAGE but with a p-value cutoff of p < 0.001. Single-gene meta-analysis and gene module meta-analysis were performed as described above for each published signature without filtering lowly expressed genes. HAI-associated comparisons were discretized and tested using maxRBA-defined HR vs LR as described above.
Results
Antibody Titer Dynamics
We evaluated 294 healthy young (21 – 30 years old, n = 147) and older (≥ 65 years old, n = 147) adults over five consecutive influenza vaccination seasons from 2010–2014. All subjects received the standard dose trivalent (2010, 2011, 2012) or quadrivalent (2013, 2014) seasonal inactivated influenza vaccine (IIV). We measured influenza-specific hemagglutination inhibition (HAI) titers pre-vaccination (D0) and 28 days post-vaccination (D28). Over the course of our study, the vaccine composition changed relative to the previous season in three of five seasons (Table 1).
In all seasons, pre-vaccination titers were negatively correlated with the increase in titers post-vaccination (Supplemental Fig. 1). Previous work defined an adjusted maximum fold change (adjMFC) endpoint that removes the nonlinear correlation between fold change and baseline titers (11). However, adjMFC separates subjects into manually defined bins, making it difficult to perform high-throughput analysis. Furthermore, adjMFC does not allow for information sharing between bins as each bin is adjusted independently. To address these limitations, we developed maximum Residual after Baseline Adjustment (maxRBA), which corrects for the dependence on baseline titers for each strain by modeling titer fold changes as an exponential function of pre-vaccination titers and selecting the maximum residual across strains (Fig. 1A). All vaccine strains were approximately equally responsible for the maximum residual in any given season. “High responders” (HR) and “low responders” (LR) were defined as the top and bottom 40th percentiles of the residuals, respectively. maxRBA can be interpreted as the maximum change from expected fold change given the initial titer; it is fully automated, is strain agnostic, and is correlated with plasmablast frequencies seven days post-vaccination (Supplemental Fig. 2A–B). Thus, maxRBA allows a completely automated assessment of the relative strength of each subject’s antibody response independent of pre-existing antibody titers.
Older adults had significantly lower pre-vaccination titers than young adults for three of five seasons (Fig. 1B). The maximum fold change to any vaccine strain showed an increasing trend in young adults compared to older adults (Supplemental Fig. 3C). Because of the inverse relationship between baseline titers and fold change (Supplemental Fig. 1), we adjusted for baseline titers using maxRBA and found that the difference in vaccine response between young and older adults was statistically significant in more seasons (Fig. 1C). Males and females had similar pre-vaccine geometric mean titers (preGMTs) (Supplemental Fig. 3A). However, the antibody response calculated by maxRBA showed a trend toward stronger antibody responses in females compared to males with similar baseline titers in both age groups (Fisher’s combined p = 0.02 (Young), p = 0.12 (Older); Supplemental Fig. 3B). We did not detect any significant difference in baseline titers or titer responses across seasons when stratifying subjects by body mass index, smoking history, aspirin use, or diabetes medication use (p > 0.05 two-sided Wilcoxon rank sum test (discrete) or simple linear regression (continuous)).
We also examined the dynamics of viral titers over the course of the five seasons (Supplemental Fig. 3D). The A/California 7/2009 H1N1 strain was introduced into the seasonal vaccine in 2010 and remained through the 2014 season; however, pre-vaccine titers to this strain were consistently lower in older vs. young adults for 2011–2014. While we did not follow the same subjects across multiple seasons, 50–80% of young and 80–98% of older adults self-reported receiving influenza vaccine in the previous year. Taken together, these results support existing evidence that the capability for antibody persistence is reduced with age (16).
Substantial Seasonal Variability in Vaccine-Induced Signatures
To identify correlates and predictors of vaccine response, we selected a subset of individuals (20 – 40 subjects per season) from young and older adult cohorts who had strong or weak antibody responses according to HAI titers and performed longitudinal transcriptional profiling pre-vaccination (baseline) and 4 (2010 cohort) or 2 days (all other cohorts), 7 days, and 28 days post-vaccination (Table 1; Methods). We first performed differential expression analysis independently in each season without differentiating subjects by antibody response. We compared each post-vaccination time point to baseline and found a vaccine-induced signature that comprised a total of 2,462 significantly differentially expressed genes (DEGs) over all five seasons (FDR < 0.05, Fold Change > 1.25; Supplemental Table 1).
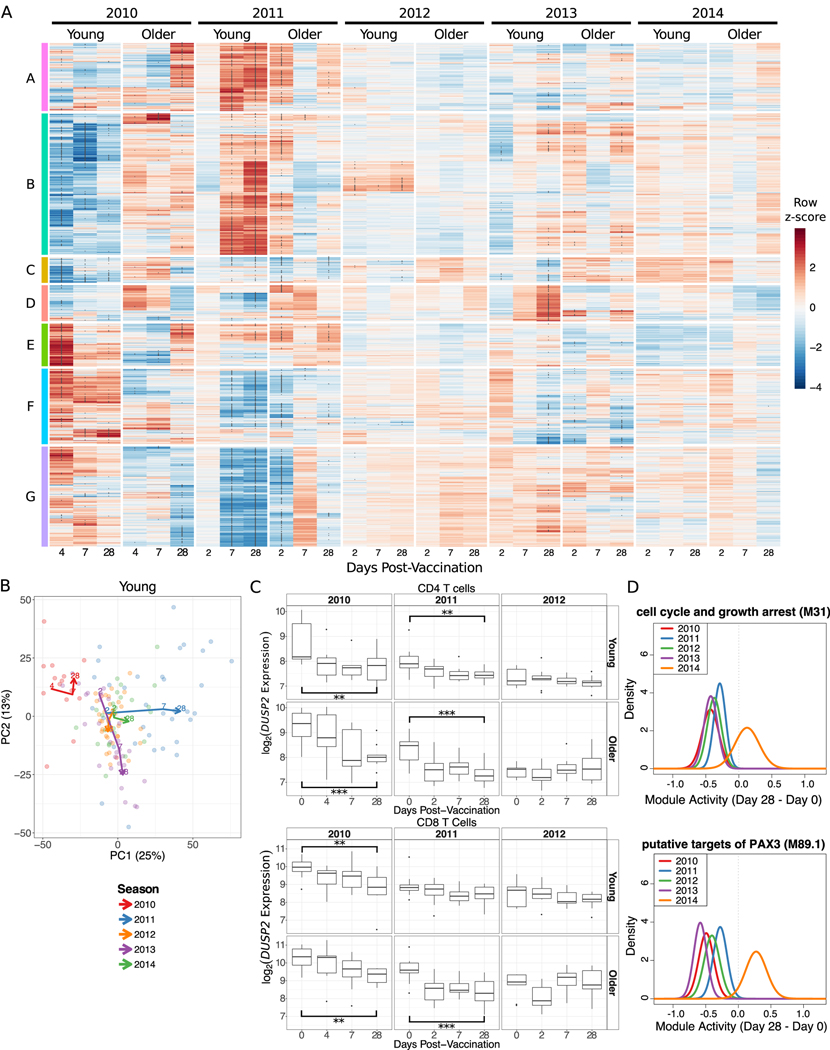
Most of the DEGs were from the first two seasons whereas vaccination in the latter three seasons induced relatively weak changes (Fig. 2A; Supplemental Fig. 4E). In fact, a substantial fraction of DEGs were unique to a single season and not differentially expressed at any time point in another season (Young: 38%, Older: 75%). In young adults, there were 1,330 DEGs shared across two or more seasons while in older adults there were 265 shared DEGs. In both young and older adults, a substantial fraction of these shared genes was differentially expressed 28 days post-vaccination (Supplemental Fig. 4F). To assess whether vaccine-induced changes were consistent between seasons, we divided the 2,462 DEGs into 7 clusters by hierarchical clustering (Fig. 2A; Supplemental Table 2) and tested for their activity in every season using QuSAGE (17) (Supplemental Fig. 5). In young adults, three of the clusters (B, F, G) had significant, but opposite, activity during the 2010 and 2011 seasons, while these clusters were relatively consistent across seasons in older adults. Genes in cluster A were induced strongly in the 2011 season in both age groups and notably enriched for multiple pathways related to mitochondria, including mitochondrial inner membrane, oxidative phosphorylation, respiratory electron transport, citric acid (TCA) cycle and respiratory electron transport, and mitochondrial respiratory chain complex assembly (FDR < 0.05; Supplemental Table 2). These findings reflect our previous identification of a mitochondrial biogenesis signature associated with influenza vaccine antibody response (14). Cluster D was only significantly induced in the 2013 season at 7 and 28 days post-vaccination and was not significantly enriched for any gene sets tested (FDR > 0.05; Supplemental Table 2). The cluster with the most consistent expression pattern across the five seasons was cluster C, which was enriched for pathways related to Toll-like receptor signaling, B and T cell signaling, NF-κB signaling, MAPK signaling, cell senescence or proliferation, and apoptosis (Supplemental Table 2). Interestingly, cluster C contains three genes (DUSP1, DUSP2, CCL3L3) which were significantly downregulated 28 days post-vaccination in four of five seasons. CCL3L3 is a ligand for CCR1, CCR3 and CCR5, known to be chemotactic for monocytes and lymphocytes (18). DUSP1 and DUSP2 are dual specificity phosphatases; DUSP2 dephosphorylates STAT3, leading to inhibition of survival and proliferation signals (19–21), and an age-associated decrease in DUSP1 function contributed to inappropriate IL-10 production in monocytes before and after influenza vaccination (22). To determine whether downregulation of these three genes was a result of changes in cell subset composition or observed in subpopulations of cells, we performed transcriptional profiling on sorted B and T cells in a subset of individuals from three seasons. DUSP1 and DUSP2, but not CCL3L3, were significantly downregulated 28 days post-vaccination over multiple seasons in CD4 and CD8 T cells of young adults (One sided t-test p < 0.01; Fig. 2C, Supplemental Fig. 4C–D). Furthermore, while DUSP2 was only significantly decreased in PBMCs of older individuals in the 2011 season, expression of DUSP2 was significantly decreased 28 days post-vaccination in sorted CD4 and CD8 T cells from older individuals in multiple seasons (Fig. 2C). Thus, the downregulation of DUSP2 28 days post-vaccination is observed in the T cell compartment of both young and older adults.
Figure 2. Substantial Seasonal Variability in Signatures Induced by Influenza Vaccination.
(A) A row-normalized heatmap of the 2,462 significantly differentially expressed genes (DEGs). Clusters A-G were defined by hierarchical clustering. Asterisks within the heatmap indicate genes significantly differentially expressed compared to day 0. (B) The first two principal components from a principal component analysis of all DEGs. Each point is a sample and lines connect the median of the points at each day post-vaccination within each season. (E) DUSP2 expression in sorted CD4 and CD8 T cells. ** p < 0.01, *** p < 0.001 one-sided t-test comparing day 28 and day 0 only. (F) Probability density functions calculated by QuSAGE for two representative gene modules significantly downregulated 28 days post-vaccination in four seasons. M31 contains DUSP1 while M89.1 contains both DUSP1 and DUSP2.
To further assess shared patterns in vaccine-induced changes across five seasons, we performed a principal component analysis (PCA) on gene expression fold changes post-vaccination for all DEGs. The first two components together explained 38% of the variation in young adults’ and 46% of the variation in older adults’ transcriptional changes post-vaccination (Fig. 2B, Supplemental Fig. 4B). Notably, in young adults, the 2011 and 2014 seasons (both with vaccine composition identical to the previous year) had similar trajectories, increasing along the first principal component (PC1) by D28 post-vaccine. Examining the genes contributing to PC1 reveals that four of the top 10 genes (SLMAP, MATR3, MBNL3, RANBP3) increase in expression post-vaccination more in the 2011 and 2014 seasons than in any other season. The shared trajectories along PC1 are not significantly enriched for any blood transcription modules (BTMs) (23), KEGG pathways (24), or cell subset signatures (25) (FDR > 0.05; Supplemental Table 3). The trajectory of the 2010 season was quite distinct from the other seasons in young adults. This season is consistently elevated on PC2, which is significantly enriched for monocytes, TLRs and inflammatory signaling (FDR < 0.05; Supplemental Table 3). The 2012 and 2013 seasons also appear to have similar trajectories, both decreasing in PC2 over time. The vaccines used in these two seasons each introduced multiple new strains while retaining the A/California/7/2009 strain. Five of the top 10 genes (ZNF493, ZNF652, OCIAD1, C21orf58, IL11RA) contributing to PC2 increased in expression 28 days post-vaccination in the 2012 and 2013 seasons while decreasing in expression in the other seasons. This differential expression analysis shows that there are large variations in vaccine-induced transcriptional signatures between seasons which, in young adults, might be explained in part by vaccine composition.
Given the substantial seasonal variation in the number of DEGs, we next performed an analysis of differential expression of gene modules using QuSAGE to quantify the gene module activity of 346 previously defined BTMs (23). There were 262 differentially expressed modules (DEMs) (FDR < 0.05; Supplemental Table 4, Supplemental Fig. 4A). Similar to the gene-level analysis, no significant changes were identified in the 2014 season, but six modules (cell cycle and growth arrest (M31), chemokines and inflammatory molecules in myeloid cells (M86.0), enriched for TF motif TTCNRGNNNNTTC, leukocyte differentiation (M160), putative targets of PAX3 (M89.1), and signaling in T cells (I) (M35.0)) were significantly downregulated in young adults at D28 in four of five seasons (Fig. 2D). These changes were largely driven by decreases in DUSP1/2, EGR1/2, JUN/JUNB, FOS/FOSB, TNF, CD83, and IL1B. Thus, while there was substantial variability in the signatures induced by vaccination across multiple seasons, there is a shared signature consisting of three genes and six modules which was downregulated at D28 in four of five seasons.
Shared Vaccine-Induced Signatures Across Five Seasons
The differential expression approach is limited by fixed fold change and significance cutoffs that may vary between seasons. To increase our power to identify shared signatures across seasons and in older adults, we performed a meta-analysis at the individual gene and gene module level. We identified 338 genes with significantly altered expression post-vaccination (FDR < 0.05; Supplemental Table 5). In young adults, we identified significant genes at D2, D7 and D28 with little overlap among genes on each day. Genes induced on D2 were moderately enriched for innate immune genes from InnateDB (http://innatedb.com/) including MYH9, TYK2, GLRX, and IP6K1 (p = 0.12, hypergeometric test). Some of the genes consistently induced at D7 included IGLL1, CD38, ITM2C, TNFRSF17, MZB1, and TXNDC5. We previously identified TNFRSF17, B cell maturation antigen, as induced seven days following influenza vaccination (26), and it was also identified as a predictive marker gene of antibody response to multiple vaccines including influenza, meningococcal conjugate (MCV4), and yellow fever (YF17D) vaccines (11, 23, 27–29). Consistent with the individual season analysis, the majority of genes identified by the meta-analysis were altered at D28; these D28 DEGs included DUSP1, DUSP2, and CCL3L3, identified in the single-season analysis, and many other downregulated genes including IL1B, CCL3, and JAK1. Thus, there are consistent changes identified across all seasons in young adults at every time point measured.
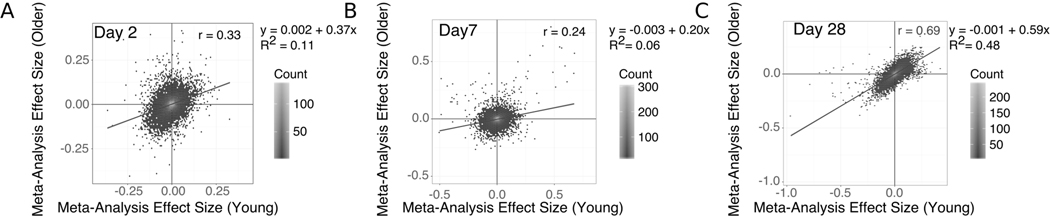
In older adults, we identified 125 genes with significantly altered expression at D28, but no genes with significantly altered expression at D2 or D7 (Supplemental Table 5). The most significantly increased gene at D28 is XRN1, the primary 5’ to 3’ cytoplasmic exonuclease involved in mRNA degradation (30). XRN1 plays a critical role in the control of RNA stability in general, but in addition appears to regulate the response to viral infection at several levels—for example, by targeting viral RNAs for degradation (31), or regulating levels of potential activating ligands such as double-stranded RNA (32). Notably, XRN1 has also been reported to facilitate replication of influenza and other viruses by inhibiting host gene expression (33, 34) - suggesting that dysregulated expression of XRN1 in older adults could influence host response to vaccination. We identified 3 genes shared between both age groups: ARRDC3 and USP30 were downregulated while TNPO1 was upregulated, all at D28. ARRDC3 encodes a member of the arrestin protein family which regulates G protein-mediated signaling and is implicated in regulating metabolism (35). USP30 is a ubiquitin-specific protease that acts as a mitochondrial deubiquitinating enzyme (36). TNPO1 encodes Transportin-1 that serves to import proteins into the nucleus (37). The effect sizes of all genes at D28 were positively correlated between young and older adults with weak positive associations at D2 and D7 (Fig. 3). These results provide additional evidence that transcriptional changes are broadly similar in young and older adults at D28 post-vaccine.
Figure 3. Vaccine-Induced Changes are Correlated Between Young and Older Adults at Day 28.
Scatter plots show the meta-analysis effect sizes of changes post-vaccination for every gene in young vs older adults on days 2 (A), 7 (B) and 28 (C) post-vaccination.
We carried out a gene set level meta-analysis using QuSAGE to combine probability density estimates of gene module activity for each season (38). We identified 186 BTMs significantly altered post-vaccination across five seasons (FDR < 0.05; Supplemental Table 4). The module with the largest increase in activity was plasma cells, immunoglobulins (M156.1) which peaked on D7 with a combined fold change of 1.17 in young adults and 1.08 in older adults at D7. Most BTMs showing significant changes were identified in young adults and, unlike the individual gene level, there was a large overlap between sets at each time point, suggesting the same module changes were sustained over the 28 days following vaccination (Supplemental Fig. 4A). Indeed, a heatmap of module activity shows that in young adults, transcriptional changes continued to intensify at D28 for many modules rather than returning to the baseline state (Supplemental Fig. 6). Older adults showed a qualitatively similar pattern to young adults on D2 and D28, but not D7. The majority (40/59) of the modules significantly altered in older adults on D28 were also significantly altered in young adults at D28 (Supplemental Fig. 4A). The modules downregulated on D28 in both young and older adults were annotated with antigen processing and presentation (M95.0, M95.1, M28, M71, M200, M5.0) and T cell activation (M36, M44, M52). The modules upregulated on D28 included golgi membrane (II) (M237), enriched in DNA interacting proteins (M182), and chaperonin mediated protein folding (I, II) (M204.0, M204.1). Taken together, the high correlation between individual gene changes and overlap of many BTMs suggest a convergence toward a common transcriptional program in young and older adults at D28.
Age-Associated Genes are Induced 7 Days Post-Vaccination
A meta-analysis across all five seasons revealed markedly different baseline transcriptional profiles in young vs. older adults, with 1,072 genes significantly altered (FDR < 0.05, Supplemental Table 6). Of these age-associated genes, 204 genes were also significantly induced by the vaccine in young adults and 125 genes in older adults. We tested whether age-associated genes were enriched for vaccine-induced genes at each time point and found that the overlap was significantly more than expected by chance for the 6 age-associated genes induced on D7 in young adults (p = 0.017, hypergeometric test). Of these 6 overlapping genes, 5 genes (ITM2C, MZB1, IGLL1, TNFRSF17, and TXNDC5) exhibited decreased basal expression in older adults while 1 (SELENOS) exhibited increased basal expression compared to young adults. While these genes were induced in young adults, they were not significantly induced in older adults on D7. Notably, MZB1 and TNFRSF17 are B cell associated genes, suggesting that older adults have decreased B cell activity pre-vaccination and fail to induce the same B cell response as young adults at D7. SELENOS encodes selenoprotein S, which is involved in degrading misfolded endoplasmic reticulum (ER) proteins and influences inflammation via the ER stress response (39, 40). Our results show that age-associated genes are significantly over-represented in the set of genes altered in young adults 7 days post-vaccination.
We next performed a meta-analysis of BTMs between age groups at baseline and identified 120 modules significantly altered with age (FDR < 0.05, Supplemental Table 7). Most of the modules that were decreased with age were associated with adaptive immunity, whereas those that had increased expression with age were mostly innate and inflammatory modules (reflecting age-associated inflammatory dysregulation; Supplemental Fig. 7B). Of these 120 modules, 52 were also significantly altered post-vaccination; however, the overlap at each time point was not significantly more than expected by chance (hypergeometric p > 0.05, Supplemental Fig. 7A). Thus, age-related genes are enriched among the genes induced at D7 in young adults while no gene modules were significantly over-represented.
Post-Vaccination Predictors of Antibody Response
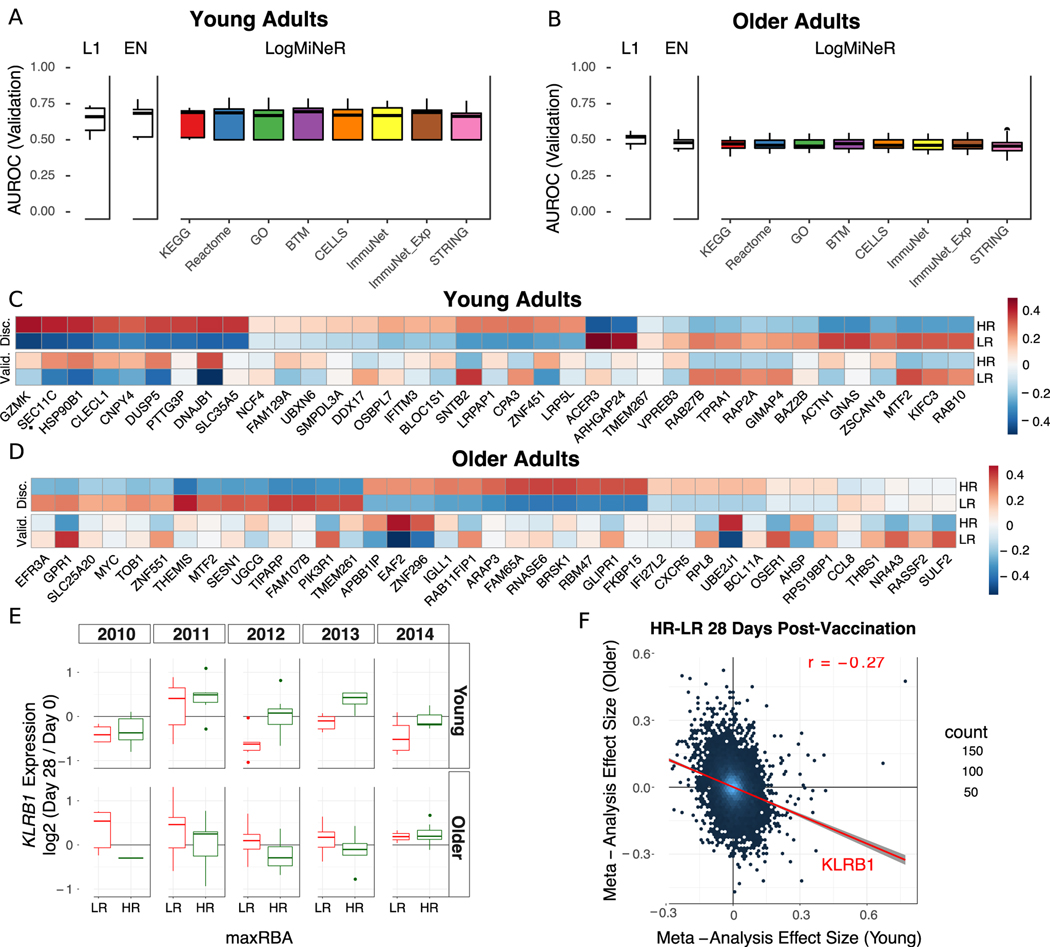
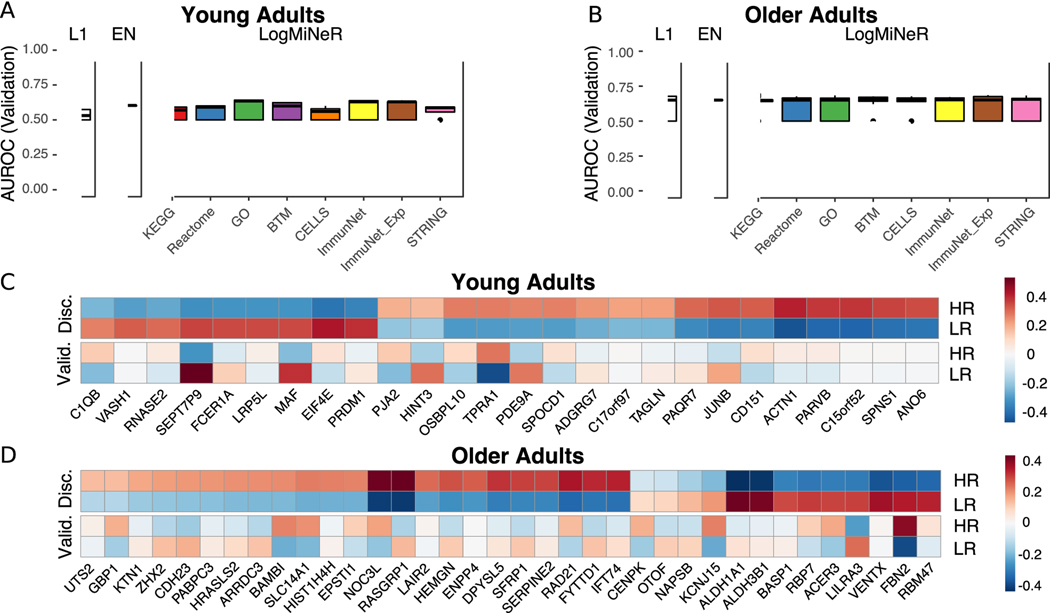
We next asked whether any transcriptional changes post-vaccination could discriminate high antibody responders (HR) from low antibody responders (LR). Regularized logistic regression models with an L1 (Lasso) or L1 and L2 (Elastic Net) penalties were fit to identify genes predictive of antibody response. In addition, to identify biologically interpretable predictors we used the Logistic Multiple Network-constrained Regression (LogMiNeR) framework (26) that facilitates the generation of predictive models with improved biological interpretability over standard methods. We combined the fold changes in gene expression data post-vaccination from five seasons and trained LogMiNeR to predict HR vs. LR in young and older cohorts separately. At each time point, models were trained on all five seasons of data (except for D2, which was not available in the 2010 season; see Methods). Publicly-available data sets from independent groups were used to validate the models. For the models built from expression changes at D2 or D28, no studies at identical time-points were available, so we attempted to validate these models on studies with similar time points (day 1 or 3 in (11) and day 14 in (13)). While we could build predictive models on our data (median AUC ≥ 0.75) they did not validate on other data sets at the (different) time points available (median AUC ≤ 0.55).
For D7 post-vaccine, direct validation data were available in independent datasets. In young adults, D7 models were predictive for HR in the discovery and validation (11) datasets (Fig. 4A). Another MAP kinase phosphatase acting on ERK1/2, DUSP5, was one of 37 genes selected by the Lasso model whose expression was increased in HR (Fig. 4C). DUSP5 is expressed in multiple immune cell types such as B cells (including plasma cells), T cells, dendritic cells, macrophages and eosinophils (41). In murine T cells, DUSP5 appears to promote the development of short-lived effector CD8+ T cells and inhibit memory precursor effector cell generation in an LCMV infection model (42); while optimizing memory precursor cell generation would be the goal of vaccination, the upregulation of DUSP5 in HR could reflect regulation of the balance between short-lived vs. memory precursor effector CD8+ T cells. A sensitivity analysis of the maxRBA cutoff shows that the average expression of predictive genes is consistent across a range of definitions for HR and LR (20th – 40th percentile; Supplemental Fig. 2C–D). Using LogMiNeR, the models were consistently enriched for the B Cell signature as well as the KEGG chemokine signaling pathway (Supplemental Table 8).
Figure 4. Post-Vaccination Transcriptional Predictors of Antibody Response.
(A and B) Boxplots of the area under the receiver operating characteristic curve (AUROC) in the validation data for Lasso (L1), Elastic Net (EN), and Logistic Multiple Network-constrained Regression (LogMiNeR) models built from day 7 transcriptional changes in young (A) and older (B) adults. 50 iterations of cross-validation were performed. x-labels indicate the prior knowledge network for LogMiNeR. (C and D) Heatmaps of Discovery (Disc.) and Validation (Valid.) data showing the z-score of the fold change for individual genes selected by the L1 models in any iteration for young (C) and older (D) adults. (E) Boxplots of KLRB1 expression changes in PBMCs 28 days post-vaccination in low responders (LR) and high responders (HR). (F) A scatter plot of the gene effect sizes comparing HR to LR 28 days post-vaccination in young vs older adults. KLRB1 is indicated as a gene that has a positive effect size in one age group and negative effect size in the other.
In older adults, models predicting antibody responses built from D7 gene expression were highly predictive in the discovery dataset but did not validate on an independent dataset (13) (Fig. 4B, D). Expression of the Solute Carrier Family 25 gene SLC25A20 of mitochondrial transporters contribute to predicting HR vs. LR in older adults. SLC25A20 is the carrier for carnitine and acylcarinitine (43), and so would be expected to be crucial for the transport of fatty acids into mitochondria. The models of response in older adults were significantly enriched for several BTMs of monocyte signatures as well as TLR and Inflammatory Signaling (M16), which positively predicted vaccine response; together with previous studies linking age-associated impairments in TLR function to influenza vaccine antibody response (44, 45), these findings provide additional support for the crucial role of innate immune function in vaccination (Supplemental Table 8).
Notably, none of the models built in young adults at any time point are predictive in older adults (AUC ≤ 0.5). In fact, models built on transcriptional changes at D28 in young adults had a median AUC near 0.8 in young adults, but no more than 0.3 in older adults, suggesting that the same genes predictive of HR in young adults predicted LR in older adults (Supplemental Fig. 8E). The Lasso models making these predictions often chose a single gene, Killer Cell Lectin Like Receptor B1 (KLRB1, also known as CD161), which was driving this inverse pattern (Fig. 4E). KLRB1 is an inhibitory receptor on NK cells (46, 47) and is also a biomarker of Th17 cells (48–50). Notably, changes in KLRB1 expression in sorted CD4 and CD8 T cells at D28 closely mirrored the changes in PBMCs for young, but not older adults (Supplemental Fig. 8A–B). We confirmed this inverse correlation between age groups on a genome-wide scale by performing a meta-analysis comparing HR vs. LR (Supplemental Table 9). We observed a weak negative correlation in effect sizes between young and older adults at D28 (r = −0.27; Fig. 4F). We confirmed this negative correlation in effect sizes between young and older adults using a virus neutralization assay (VNA) in a test sample of blood from seasons 2011 and 2012 (r = −0.32; Supplemental Fig. 8D). Thus, expression changes of many genes at D28 have opposing signs between age groups for the effect size comparing HR vs. LR, and a single gene, KLRB1, predicts response with AUC > 0.7 in opposing directions in young vs. older adults.
Baseline Predictors of Antibody Response
We next sought to identify baseline transcriptional predictors of antibody response. In young adults, LogMiNeR models were predictive above random on discovery and validation (11) (Fig. 5A) datasets. Lasso models included the gene VASH1, known as an angiogenesis inhibitor and mediator of stress resistance in endothelial cells, which was expressed at lower levels in HRs (Fig. 5C); notably, the KEGG gene set leukocyte transendothelial migration was significantly enriched in over 50% of the models when LogMiNeR was used with ImmuNet as prior knowledge (51). Another predictive gene, EIF4E, a translation initiation factor important in type I interferon production, was decreased in HRs. A sensitivity analysis of the maxRBA cutoff shows that the average expression of predictive genes is consistent across a range of definitions for HR and LR (20th – 40th percentile; Supplemental Fig. 2E–F). Finally, the BTMs cell adhesion (M51) and B cell surface signature (S2) were consistently enriched in the models (Supplemental Table 8). In older adults, LogMiNeR models were also predictive on the discovery and one validation dataset (9) (Fig. 5B) but not another (13) (Supplemental Fig. 8C). Two of the individual genes that predict response, ALDH1A1 and ALDH3B1, are aldehyde dehydrogenases which metabolize vitamin A to retinoic acid (Fig. 5D). Recently, aldehyde dehydrogenases were implicated in antiviral innate immunity as mediators of the interferon response through their role in the biogenesis of retinoic acid (52). Multiple monocyte gene sets are enriched in the predictive genes, including the BTM enriched in monocytes (II) (M11.0), which negatively predicts vaccine response (Supplemental Table 8). Thus, these baseline predictive models built from five seasons of transcriptional profiling data provide further evidence for functional distinctions present in subjects prior to vaccination that influence the immunologic response to influenza vaccine in young and older adults.
Figure 5. Baseline Transcriptional Predictors of Antibody Response.
(A and B) Boxplots of the area under the receiver operating characteristic curve (AUROC) in the validation data for Lasso (L1), Elastic Net (EN), and Logistic Multiple Network-constrained Regression (LogMiNeR) models built from baseline (pre-vaccination) transcriptional profiles in young (A) and older (B) adults (9). 50 iterations of cross-validation were performed. x-labels indicate the prior knowledge network for LogMiNeR. (C and D) Heatmaps of Discovery (Disc.) and Validation (Valid.) data showing the z-score of the fold change for individual genes selected by the L1 models in any iteration for young (C) and older (D) adults.
Behavior of Published Signatures Over Five Seasons
To link our findings to previously identified influenza vaccine signatures, we performed a comprehensive assessment of the behavior of 1,603 previously published individual gene and gene module signatures in our data set. We manually curated published signatures from studies that carried out transcriptional profiling on adult cohorts after influenza vaccination (9, 11, 13, 15, 27, 53). We further limited the signatures to shared time points post-vaccination. This set of findings describe 935 response-associated and 653 temporal signatures in B cells and PBMCs as well as 15 age-associated signatures (Supplemental Table 10).
Most of the previously published signatures we validated in our data were single genes induced 7 days post-vaccination in PBMCs or B cells (Supplemental Fig. 9). Of the 135 signatures that showed significant differential expression (p < 0.001), 103 changed in the same direction as the published signature. In PBMCs we validated 26 D7 vaccine-induced genes including four genes independently discovered in our meta-analysis: CD38, ITM2C, TNFRSF17, and SPATS2 (Supplemental Fig. 9B) (11). CD38 is upregulated on the surface of antibody secreting cells, and TNFRSF17, or B cell maturation antigen (BCMA) is a receptor for B cell activating factor (BAFF) expressed on memory B cells and plasma cells (54). Notably, validated vaccine-induced genes in B cells include several associated with mitochondrial function whose expression was upregulated at Day 7, including UQCRQ (ubiquinol cytochrome c reductase, complex III, subunit VII), ME2 (NAD-dependent malic enzyme), TAL (transaldolase 1), and GLDC (glycine decarboxylase) (Supplemental Fig. 9A). We validated several modules significantly associated with antibody response at baseline in young and older adults (Supplemental Fig. 9D) (13). Of these modules, one positively associated with antibody response (enriched in B cells (I) (M47.0)) is enriched in our baseline predictive model of young adults and three negatively associated with antibody response are enriched in our baseline predictive model of older adults (Monocyte surface signature (S4), myeloid cell enriched receptors and transporters (M4.3), enriched in monocytes (II) (M11.0)). Interestingly, these latter three modules are also enriched in predictive models of HR vs LR from D7 fold changes. Finally, there are seven validated single genes whose fold change at D7 is positively associated with antibody response in young adults (Supplemental Fig. 9C) (11). One of these genes, HSP90B1, or gp96 – an ER-based chaperone protein implicated in innate and adaptive immune function – is also selected as a predictive gene of antibody response (55, 56).
Discussion
This study is the first to evaluate the transcriptomic response to influenza vaccination in young and older adults in five consecutive vaccine seasons with three different vaccine compositions. We sought to address whether common signatures of vaccine response or transcriptional predictors of antibody response could be elucidated despite differences in seasonal vaccine composition.
To adjust for the inverse relationship between baseline antibody titers and vaccine-induced antibody production, we developed a novel vaccine response endpoint, maxRBA, to automatically correct for variation in baseline titers; this allowed us to demonstrate an age-associated decrease in antibody response in gender-matched participants. Comparing the transcriptional profiles across five seasons revealed substantial seasonal variability in both the magnitude as well as direction of response. For example, the vaccines administered in the 2010 and 2011 seasons elicited large changes in gene expression, but no statistically significant DEGs were found in the 2014 season despite a comparable sample size. Potentially, the large transcriptional changes observed in 2010 and 2011 could reflect the introduction of the A/California/7/2009 viral pandemic strain to the seasonal vaccine (as well as a change in the H3N2 vaccine strain beginning in 2010—the only year of the five studied when both influenza A strains changed). Notably, a principal component analysis revealed similar vaccine-induced signatures in the 2011 and 2014 seasons and in the 2012 and 2013 seasons. The similarities between the 2011 and 2014 seasons are intriguing because in both seasons the composition of the vaccine was identical to that in the preceding year, perhaps suggesting that these gene signatures reflect a relatively recent recall response. In contrast, the 2012 and 2013 vaccines each contained two strains which had not been present in the previous year’s vaccine. However, we did not observe the same trends in older adults and the design of this study precluded the derivation of strain-specific transcriptional signatures. Nonetheless, our results indicate that changes in vaccine composition, influencing factors such as vaccine strain immunogenicity and the effects of previous vaccination or infection, can alter the transcriptional response to influenza immunization.
Despite substantial inter-season variability, we identified shared vaccine-induced signatures in both young and older adults at D28. We expected D28 expression profiles to be similar to baseline; however, there were numerous transcriptional changes at D28 that were consistent across seasons with different vaccine compositions. Some of the most significant changes identified from single-season differential expression analysis in four out of five seasons were in DUSP1, DUSP2, and CCL3L3; moreover, DUSP2 expression was also decreased in sorted CD4+ and CD8+ T cells from both young and older adults at D28. It is notable that a basal age-related alteration in phosphorylation of DUSP1, a negative regulator of IL-10 production, was associated with increased expression of IL-10 in monocytes from older adults (seen pre- and post-influenza vaccination) (57) and that increased DUSP6 expression was associated with impaired T cell receptor signaling in CD4+ T cells from older adults (58). These results emphasize the importance of modulation of MAP kinase function, such as through phosphatases of the DUSP family, in the regulation of influenza vaccine response. Surprisingly, early response signatures at D2 and D7 post-vaccination were not as consistent across seasons as D28 signatures in a meta-analysis of genes and gene modules. One potential hypothesis that explains this observation is that temporal variations in early responses across seasons were not captured at the time points used, and that responses at D28 are less variable, and thus were captured in every season. It is possible that this common transcriptional program at D28 reflects a convergence towards resolution of the vaccine response in both young and older adults. However, a substantial number of BTMs showed upregulated activity at D28 without evidence of resolution to baseline, particularly in young adults; notably, we previously found evidence of enhanced TNF-alpha and IL-6 production in monocytes 28 days post-influenza immunization (57) that was blunted in monocytes from older adults. Thus, it remains possible that the transcriptional signature we observed also reflects elements of an ongoing immune activated state several weeks after vaccination.
We built predictive models of antibody response from post-vaccination transcriptional responses which were successfully validated in an independent cohort of young adults. Although transcriptional changes were correlated between age groups at D28, models of antibody response built in young adults did not validate in older adults. Strikingly, we identified a genome-wide inverse correlation between the effect size of genes discriminating HR and LR at D28 and confirmed this finding with both HAI and VNA titers. A similar inverse correlation related to age was recently reported using baseline (D0) gene expression signatures (15). We identified a single gene, KLRB1, whose expression alone predicted response in both age groups but in opposite directions. In young adults, changes in KLRB1 expression were also observed in sorted CD4 and CD8 T cells, perhaps reflecting the finding that KLRB1 expression is increased in populations of memory T cells (59). Furthermore, KLRB1hi CD8+ T cells are self-renewing memory cells that are able to reconstitute the memory T cell pool after chemotherapy (60). Thus, KLRB1 induction in young adults may reflect an increase in memory T cell populations. In older adults, these expression patterns were not observed in sorted T cells, implying that KLRB1 expression in another cell type, perhaps NK cells or Th17 cells, was the basis for the predictive performance.
We also built and validated predictive models of antibody response in young and older adults from D0 gene expression data. One of the predictive genes in young adults, VASH1, showed evidence of genetic regulation of gene expression in a previous study of influenza vaccination, suggesting that genotype may have predictive power to explain the antibody response (8). Leukocyte migration and a B cell surface signature were enriched in the predictive models. This is consistent with a recently reported meta-analysis which included baseline transcriptional profiles from the 2010, 2011, and 2012 seasons of the present study and validated a temporally stable B cell receptor signaling gene module that positively predicted response at baseline (15). While the B cell surface signature (S2) module we identified was not the same one identified in the previous study, our findings further support the implication of B cell transcriptional signatures as pre-vaccine biomarkers of antibody response in young adults. In older adults, we incorporated prior knowledge on gene coexpression using LogMiNeR to identify monocyte signatures which were enriched in the predictive models and were negatively associated with antibody response. Our model validated on one older adult cohort (9) but not another (13); this may reflect substantial variability in cohorts of older adults, which would be expected to be more heterogeneous in terms of comorbid medical conditions or medication use compared to young adults. Finally, we linked our findings to previously identified influenza vaccination signatures by performing a comprehensive assessment of 1,603 previously published individual gene and gene module signatures. We present the signatures that validate in any season or a meta-analysis of all seasons of our data to highlight the most consistent set of genes and gene modules associated with vaccination or antibody response in PBMC and B cells.
One limitation of this study is that it was not placebo controlled. Thus, we cannot rule out that some of the changes in gene expression post-vaccination are unrelated to antigen-specific vaccine responses. Participants were specifically recruited from among those presenting to the clinic to receive an influenza vaccination and placebo vaccination was not a practical consideration, especially in older adults who are at higher risk of morbidity and mortality from influenza infection. Nevertheless, the fact that transcriptional changes are occurring post-vaccination means that they have the potential to influence the ultimate quality of the response.
In summary, we recruited nearly 300 young and older adults across five vaccination seasons and, despite substantial seasonal variability in vaccine-induced transcriptional signatures, identified a core transcriptional signature shared between seasons and across age groups 28 days post-vaccination. Our results suggest that vaccine composition, in concert with differences in pre-existing immunity and other individual factors, dramatically influences immune response to inactivated influenza vaccination. A deeper understanding of the cause of the shared transcriptional signatures we consistently identified in both young and older adults 28 days post-vaccination may help to better understand the mechanisms by which the current vaccines function. In addition, we defined a new endpoint (maxRBA) to capture antibody response relative to baseline titer and were able to predict response in young and older adults separately using baseline transcriptional profiles. These signatures, such as the monocyte signature negatively predicting vaccine response in older adults, could be further developed and used to predict the subgroup of individuals who would most likely benefit from vaccination or, on the other hand, the subgroup of predicted non-responders to current vaccines who should be monitored more closely during flu season and enrolled in studies of investigational influenza vaccines. Baseline predictive signatures also open the possibility of modulating the immune system of individuals to mimic the immune state of predicted high responders before vaccination or as part of a combination therapy with vaccination. This work represents a step toward better understanding of how the immune system responds to influenza vaccination in young and older adults and may be beneficial for rationally designing more effective vaccines.
Supplementary Material
Key Points.
Consistent transcriptional signatures identified 28 days post influenza vaccination.
New endpoint (maxRBA) characterizes antibody response relative to baseline.
Genes related to antibody response behave differently in young and older adults.
Acknowledgments
We gratefully acknowledge Dr. Randy Albrecht and Dr. Adolfo Garcia-Sastre at the Icahn School of Medicine at Mount Sinai, who led the Human Immunology Project Consortium (HIPC) core for influenza viral neutralization assays. This work was supported by NIH grant U19 AI089992, K24 AG042489, and by the Claude D. Pepper Older Americans Independence Center at Yale (to H.J.Z. and A.C.S.: P30 AG021342). Computational resources and support were provided by the Yale Center for Research Computing [NIH grants RR19895 and RR029676–01]. H.J.Z. was supported by a GEMSSTAR award from NIA (R03 AG050947). D.G.C. was supported by NIH training grant T32 EB019941. S.A. was supported by the NSF Graduate Research Fellowship Program [grant number DGE-1122492]. Any opinions, findings and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
References
- 1.Ohmit SE, Victor JC, Teich ER, Truscon RK, Rotthoff JR, Newton DW, Campbell S. a, Boulton ML, and Monto AS. 2008. Prevention of symptomatic seasonal influenza in 2005–2006 by inactivated and live attenuated vaccines. J. Infect. Dis. 198: 312–317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Frey S, Vesikari T, Szymczakiewicz-Multanowska A, Lattanzi M, Izu A, Groth N, and Holmes S. 2010. Clinical Efficacy of Cell Culture-Derived and Egg-Derived Inactivated Subunit Influenza Vaccines in Healthy Adults. Clin. Infect. Dis. 51: 997–1004. [DOI] [PubMed] [Google Scholar]
- 3.Jackson L. Gaglani a, M. J., Keyserling HL, Balser J, Bouveret N, Fries L, and Treanor JJ 2010. Safety, efficacy, and immunogenicity of an inactivated influenza vaccine in healthy adults: a randomized, placebo-controlled trial over two influenza seasons. BMC Infect. Dis. 10: 71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Monto AS, Ohmit SE, Petrie JG, Johnson E, Truscon R, Teich E, Rotthoff J, Boulton M, and Victor JC. 2009. Comparative efficacy of inactivated and live attenuated influenza vaccines. N. Engl. J. Med. 361: 1260–1267. [DOI] [PubMed] [Google Scholar]
- 5.Beran J, Wertzova V, Honegr K, Kaliskova E, Havlickova M, Havlik J, Jirincova H, Van Belle P, Jain V, Innis B, and Devaster J-M. 2009. Challenge of conducting a placebo-controlled randomized efficacy study for influenza vaccine in a season with low attack rate and a mismatched vaccine B strain: a concrete example. BMC Infect. Dis. 9: 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goodwin K, Viboud C, and Simonsen L. 2006. Antibody response to influenza vaccination in the elderly: A quantitative review. Vaccine 24: 1159–1169. [DOI] [PubMed] [Google Scholar]
- 7.Bucasas KL, Franco LM, a Shaw C, Bray MS, Wells JM, Niño D, Arden N, Quarles JM, Couch RB, and Belmont JW. 2011. Early patterns of gene expression correlate with the humoral immune response to influenza vaccination in humans. J. Infect. Dis. 203: 921–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Franco LM, Bucasas KL, Wells JM, Niño D, Wang X, Zapata GE, Arden N, Renwick A, Yu P, Quarles JM, Bray MS, Couch RB, Belmont JW, and Shaw C. a. 2013. Integrative genomic analysis of the human immune response to influenza vaccination. Elife 2: e00299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Furman D, Jojic V, Kidd B, Shen-Orr S, Price J, Jarrell J, Tse T, Huang H, Lund P, Maecker HT, Utz PJ, Dekker CL, Koller D, and Davis MM. 2013. Apoptosis and other immune biomarkers predict influenza vaccine responsiveness. Mol. Syst. Biol. 9: 659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tan Y, Tamayo P, Nakaya H, Pulendran B, Mesirov JP, and Haining WN. 2014. Gene signatures related to B-cell proliferation predict influenza vaccine-induced antibody response. Eur. J. Immunol. 44: 285–295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tsang JS, Schwartzberg PL, Kotliarov Y, Biancotto A, Xie Z, Germain RN, Wang E, Olnes MJ, Narayanan M, Golding H, Moir S, Dickler HB, Perl S, and Cheung F. 2014. Global analyses of human immune variation reveal baseline predictors of postvaccination responses. Cell 157: 499–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Obermoser G, Presnell S, Domico K, Xu H, Wang Y, Anguiano E, Thompson-Snipes L, Ranganathan R, Zeitner B, Bjork A, Anderson D, Speake C, Ruchaud E, Skinner J, Alsina L, Sharma M, Dutartre H, Cepika A, Israelsson E, Nguyen P, Nguyen QA, Harrod a. C., Zurawski SM, Pascual V, Ueno H, Nepom GT, Quinn C, Blankenship D, Palucka K, Banchereau J, and Chaussabel D. 2013. Systems scale interactive exploration reveals quantitative and qualitative differences in response to influenza and pneumococcal vaccines. Immunity 38: 831–844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nakaya HI, Hagan T, Duraisingham SS, Lee EK, Kwissa M, Rouphael N, Frasca D, Gersten M, Mehta AK, Gaujoux R, Li GM, Gupta S, Ahmed R, Mulligan MJ, Shen-Orr S, Blomberg BB, Subramaniam S, and Pulendran B. 2015. Systems Analysis of Immunity to Influenza Vaccination across Multiple Years and in Diverse Populations Reveals Shared Molecular Signatures. Immunity 43: 1186–1198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Thakar J, Mohanty S, West AP, Joshi SR, Ueda I, Wilson J, Meng H, Blevins TP, Tsang S, Trentalange M, Siconolfi B, Park K, Gill TM, Belshe RB, Kaech SM, Shadel GS, Kleinstein SH, and Shaw AC. 2015. Aging-dependent alterations in gene expression and a mitochondrial signature of responsiveness to human influenza vaccination. Aging (Albany. NY). 7: 38–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.HIPC-CHI Signatures Project Team, T., and T. HIPC-I Consortium. 2017. Multicohort analysis reveals baseline transcriptional predictors of influenza vaccination responses. Sci. Immunol. 2: eaal4656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Song JY, Cheong HJ, Hwang IS, Choi WS, Jo YM, Park DW, Cho GJ, Hwang TG, and Kim WJ. 2010. Long-term immunogenicity of influenza vaccine among the elderly: Risk factors for poor immune response and persistence. Vaccine 28: 3929–3935. [DOI] [PubMed] [Google Scholar]
- 17.Yaari G, Bolen CR, Thakar J, and Kleinstein SH. 2013. Quantitative set analysis for gene expression: A method to quantify gene set differential expression including gene-gene correlations. Nucleic Acids Res. 41: e170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Proost P, Menten P, Struyf S, Schutyser E, De Meester I, and Van Damme J. 2000. Cleavage by CD26 / dipeptidyl peptidase IV converts the chemokine LD78β into a most efficient monocyte attractant and CCR1 agonist. Blood 96: 1674–1680. [PubMed] [Google Scholar]
- 19.Kwak SP, Hakes DJ, Martell KJ, and Dixon JE. 1994. Isolation and Characterization of a Human Dual Specificity Protein-Tyrosine Phosphatase Gene. J. Biol. Chem. 269: 3596–3604. [PubMed] [Google Scholar]
- 20.Rohan PJ, Davis P, Moskaluk CA, Kearns M, Rohan PJ, Davis P, Moskaluk CA, Kearns M, Krutzsch H, Siebenlist U, and Kelly K. 1993. PAC-1 : A Mitogen-Induced Nuclear Protein Tyrosine Phosphatase. Science (80-. ). 259: 1763–1766. [DOI] [PubMed] [Google Scholar]
- 21.Wei W, Jiao Y, Postlethwaite A, Stuart JM, Wang Y, Sun D, and Gu W. 2013. Dual-specificity phosphatases 2: surprising positive effect at the molecular level and a potential biomarker of diseases. Genes Immun. 14: 1–6. [DOI] [PubMed] [Google Scholar]
- 22.Mohanty S, Joshi SR, Ueda I, Wilson J, Blevins TP, Siconolfi B, Meng H, Devine L, Raddassi K, Tsang S, Belshe RB, Hafler DA, Kaech SM, Kleinstein SH, Trentalange M, Allore HG, and Shaw AC. 2015. Prolonged proinflammatory cytokine production in monocytes modulated by interleukin 10 after influenza vaccination in older adults. J. Infect. Dis. 211: 1174–1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li S, Rouphael N, Duraisingham S, Romero-Steiner S, Presnell S, Davis C, Schmidt DS, Johnson SE, Milton A, Rajam G, Kasturi S, Carlone GM, Quinn C, Chaussabel D, Palucka a K., Mulligan MJ, Ahmed R, Stephens DS, Nakaya HI, and Pulendran B. 2014. Molecular signatures of antibody responses derived from a systems biology study of five human vaccines. Nat. Immunol. 15: 195–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kanehisa M, and Goto S. 2000. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28: 27–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Abbas a R., Baldwin D, Ma Y, Ouyang W, Gurney A, Martin F, Fong S, van Lookeren Campagne M, Godowski P, Williams PM, Chan a C., and Clark HF 2005. Immune response in silico (IRIS): immune-specific genes identified from a compendium of microarray expression data. Genes Immun. 6: 319–331. [DOI] [PubMed] [Google Scholar]
- 26.Avey S, Mohanty S, Wilson J, Zapata H, Joshi SR, Siconolfi B, Tsang S, Shaw AC, and Kleinstein SH. 2017. Multiple network-constrained regressions expand insights into influenza vaccination responses. Bioinformatics 33: i208–i216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nakaya HI, Wrammert J, Lee EK, Racioppi L, Marie-Kunze S, Haining WN, Means AR, Kasturi SP, Khan N, Li G-M, McCausland M, Kanchan V, Kokko KE, Li S, Elbein R, Mehta AK, Aderem A, Subbarao K, Ahmed R, and Pulendran B. 2011. Systems biology of vaccination for seasonal influenza in humans. Nat. Immunol. 12: 786–795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gaucher D, Therrien R, Kettaf N, Angermann BR, Boucher G, Filali-Mouhim A, Moser JM, Mehta RS, Drake DR, Castro E, Akondy R, Rinfret A, Yassine-Diab B, a Said E, Chouikh Y, Cameron MJ, Clum R, Kelvin D, Somogyi R, Greller LD, Balderas RS, Wilkinson P, Pantaleo G, Tartaglia J, Haddad EK, and Sékaly R-P. 2008. Yellow fever vaccine induces integrated multilineage and polyfunctional immune responses. J. Exp. Med. 205: 3119–3131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Querec TD, Akondy RS, Lee EK, Cao W, Nakaya HI, Teuwen D, Pirani A, Gernert K, Deng J, Marzolf B, Kennedy K, Wu H, Bennouna S, Oluoch H, Miller J, Vencio RZ, Mulligan M, Aderem A, Ahmed R, and Pulendran B. 2009. Systems biology approach predicts immunogenicity of the yellow fever vaccine in humans. Nat. Immunol. 10: 116–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mitchell P, and Tollervey D. 2000. mRNA stability in eukaryotes. Curr. Opin. Genet. Dev. 10: 193–198. [DOI] [PubMed] [Google Scholar]
- 31.Molleston JM, and Cherry S. 2017. Attacked from all sides: RNA decay in antiviral defense. Viruses 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu SW, Katsafanas GC, Liu R, Wyatt LS, and Moss B. 2015. Poxvirus decapping enzymes enhance virulence by preventing the accumulation of dsRNA and the induction of innate antiviral responses. Cell Host Microbe 17: 320–331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Khaperskyy DA, Schmaling S, Larkins-Ford J, McCormick C, and Gaglia MM. 2016. Selective Degradation of Host RNA Polymerase II Transcripts by Influenza A Virus PA-X Host Shutoff Protein. PLoS Pathog. 12: 1–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gaglia MM, Covarrubias S, Wong W, and Glaunsinger BA. 2012. A common strategy for host RNA degradation by divergent viruses. J Virol 86: 9527–9530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Patwari P, and Lee RT. 2012. An expanded family of arrestins regulate metabolism. Trends Endocrinol. Metab. 23: 216–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nakamura N, and Hirose S. 2008. Regulation of Mitochondrial Morphology by USP30, a Deubiquitinating Enzyme Present in the Mitochondrial Outer Membrane. Mol. Biol. Cell 19: 1903–1911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Twyffels L, Gueydan C, and Kruys V. 2014. Transportin-1 and Transportin-2: Protein nuclear import and beyond. FEBS Lett. 588: 1857–1868. [DOI] [PubMed] [Google Scholar]
- 38.Meng H, Yaari G, Bolen CR, Avey S, and Kleinstein SH. 2019. Gene set meta-analysis with Quantitative Set Analysis for Gene Expression (QuSAGE). PLOS Comput. Biol. 15: e1006899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Curran JE, Jowett JBM, Elliott KS, Gao Y, Gluschenko K, Wang J, Abel Azim DM, Cai G, Mahaney MC, Comuzzie AG, Dyer TD, Walder KR, Zimmet P, MacCluer JW, Collier GR, Kissebah AH, and Blangero J. 2005. Genetic variation in selenoprotein S influences inflammatory response. Nat. Genet. 37: 1234–1241. [DOI] [PubMed] [Google Scholar]
- 40.Ye Y, Shibata Y, Yun C, Ron D, and Rapoport TA. 2004. A membrane protein complex mediates retro-translocation from the ER lumen into the cytosol. Nature 429: 841–847. [DOI] [PubMed] [Google Scholar]
- 41.Jeffrey KL, Camps M, Rommel C, and Mackay CR. 2007. Targeting dual-specificity phosphatases: manipulating MAP kinase signalling and immune responses. Nat. Rev. Drug Discov. 6: 391–403. [DOI] [PubMed] [Google Scholar]
- 42.Kutty RG, Xin G, Schauder DM, Cossette SM, Bordas M, Cui W, and Ramchandran R. 2016. Dual specificity phosphatase 5 is essential for T cell survival. PLoS One 11: 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Indiveri C, Iacobazzi V, Tonazzi A, Giangregorio N, Infantino V, Convertini P, Console L, and Palmieri F. 2011. The mitochondrial carnitine/acylcarnitine carrier: Function, structure and physiopathology. Mol. Aspects Med. 32: 223–233. [DOI] [PubMed] [Google Scholar]
- 44.van Duin D, Allore HG, Mohanty S, Ginter S, Newman FK, Belshe RB, Medzhitov R, and Shaw AC. 2007. Prevaccine Determination of the Expression of Costimulatory B7 Molecules in Activated Monocytes Predicts Influenza Vaccine Responses in Young and Older Adults. J. Infect. Dis. 195: 1590–1597. [DOI] [PubMed] [Google Scholar]
- 45.Panda A, Qian F, Mohanty S, van Duin D, Newman FK, Zhang L, Chen S, Towle V, Belshe RB, Fikrig E, Allore HG, Montgomery RR, and Shaw AC. 2010. Age-Associated Decrease in TLR Function in Primary Human Dendritic Cells Predicts Influenza Vaccine Response. J. Immunol. 184: 2518–2527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Aldemir H, Prod’homme V, Dumaurier M-J, Retiere C, Poupon G, Cazareth J, Bihl F, and Braud VM. 2005. Cutting Edge: Lectin-Like Transcript 1 Is a Ligand for the CD161 Receptor. J. Immunol. 175: 7791–7795. [DOI] [PubMed] [Google Scholar]
- 47.Rosen DB, Bettadapura J, Alsharifi M, Mathew PA, Warren HS, and Lanier LL. 2005. Cutting Edge: Lectin-Like Transcript-1 Is a Ligand for the Inhibitory Human NKR-P1A Receptor. J. Immunol. 175: 7796–7799. [DOI] [PubMed] [Google Scholar]
- 48.Kleinschek MA, Boniface K, Sadekova S, Grein J, Murphy EE, Turner SP, Raskin L, Desai B, Faubion WA, de Waal Malefyt R, Pierce RH, McClanahan T, and Kastelein RA. 2009. Circulating and gut-resident human Th17 cells express CD161 and promote intestinal inflammation. J. Exp. Med. 206: 525–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Maggi L, Santarlasci V, Capone M, Peired A, Frosali F, Crome SQ, Querci V, Fambrini M, Liotta F, Levings MK, Maggi E, Cosmi L, Romagnani S, and Annunziato F. 2010. CD161 is a marker of all human IL-17-producing T-cell subsets and is induced by RORC. Eur. J. Immunol. 40: 2174–2181. [DOI] [PubMed] [Google Scholar]
- 50.Cosmi L, De Palma R, Santarlasci V, Maggi L, Capone M, Frosali F, Rodolico G, Querci V, Abbate G, Angeli R, Berrino L, Fambrini M, Caproni M, Tonelli F, Lazzeri E, Parronchi P, Liotta F, Maggi E, Romagnani S, and Annunziato F. 2008. Human interleukin 17–producing cells originate from a CD161 + CD4 + T cell precursor. J. Exp. Med. 205: 1903–1916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gorenshteyn D, Zaslavsky E, Fribourg M, Park CY, Wong AK, Tadych A, Hartmann BM, Albrecht RA, García-Sastre A, Kleinstein SH, Troyanskaya OG, and Sealfon SC. 2015. Interactive Big Data Resource to Elucidate Human Immune Pathways and Diseases. Immunity 43: 605–614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cho NE, Bang BR, Gurung P, Li M, Clemens DL, Underhill TM, James LP, Chase JR, and Saito T. 2016. Retinoid regulation of antiviral innate immunity in hepatocytes. Hepatology 63: 1783–1795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Furman D, Chang J, Lartigue L, Bolen CR, Haddad F, Gaudilliere B, Ganio EA, Fragiadakis GK, Spitzer MH, Douchet I, Daburon S, Moreau J-F, Nolan GP, Blanco P, Déchanet-Merville J, Dekker CL, Jojic V, Kuo CJ, Davis MM, and Faustin B. 2017. Expression of specific inflammasome gene modules stratifies older individuals into two extreme clinical and immunological states. Nat. Med. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Darce JR, Arendt BK, Wu X, and Jelinek DF. 2014. Regulated Expression of BAFF-Binding Receptors during Human B Cell Differentiation. J. Immunol. 179: 7276–7286. [DOI] [PubMed] [Google Scholar]
- 55.Pockley AG, and Henderson B. 2018. Extracellular cell stress (Heat shock) proteins—immune responses and disease: An overview. Philos. Trans. R. Soc. B Biol. Sci. 373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Randow F, and Seed B. 2001. Endoplasmic reticulum chaperone gp96 is required for innate immunity but not cell viability. Nat. Cell Biol. 3: 891–896. [DOI] [PubMed] [Google Scholar]
- 57.Mohanty S, Joshi SR, Ueda I, Wilson J, Blevins TP, Siconolfi B, Meng H, Devine L, Raddassi K, Tsang S, Belshe RB, Hafler D. a., Kaech SM, Kleinstein SH, Trentalange M, Allore HG, and Shaw a. C. 2014. Prolonged Proinflammatory Cytokine Production in Monocytes Modulated by Interleukin 10 After Influenza Vaccination in Older Adults. J. Infect. Dis. 211: 1174–1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Li G, Yu M, Lee WW, Tsang M, Krishnan E, Weyand CM, and Goronzy JJ. 2012. Decline in miR-181a expression with age impairs T cell receptor sensitivity by increasing DUSP6 activity. Nat. Med. 18: 1518–1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kirkham CL, and Carlyle JR. 2014. Complexity and Diversity of the NKR-P1:Clr (Klrb1:Clec2) Recognition Systems. Front. Immunol. 5: 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Turtle CJ, Swanson HM, Fujii N, Estey EH, and Riddell SR. 2009. A Distinct Subset of Self-Renewing Human Memory CD8+ T Cells Survives Cytotoxic Chemotherapy. Immunity 31: 834–844. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.