Abstract
Myelodysplastic syndromes (MDS), a spectrum of heterogeneous hematopoietic stem cell diseases, vary in clinical severity, response to therapy, and propensity toward progression to acute myeloid leukemia. These are acquired clonal disorders resulting from somatic mutations within the hematopoietic stem or progenitor cell population. Understanding the natural history and the risk of developing leukemia and other adverse outcomes is dependent on access to well-annotated biospecimens linked to robust clinical and molecular data. To facilitate the acquisition and distribution of MDS biospecimens to the wider scientific community and support scientific discovery in this disease, the National MDS Natural History study was initiated by the National Heart, Lung, and Blood Institute (NHLBI) and is being conducted in collaboration with community hospitals and academic medical centers supported by the National Cancer Institute (NCI). The study will recruit up to 2000 MDS patients or overlapping myeloproliferative neoplasms (MDS/MPN) and up to 500 cases of idiopathic cytopenia of undetermined significance (ICUS). The National MDS Natural History Study (NCT02775383) will offer the world’s largest disease-focused tissue biobank linked to longitudinal clinical and molecular data in MDS. Here, we report on the study design features and describe the vanguard phase of 200 cases. The study assembles a comprehensive clinical database, quality of life results, laboratory data, histopathology slides and images, genetic information, hematopoietic and germline tissues representing high-quality biospecimens and data from diverse centers across the United States. These resources will be available to the scientific community for investigator-initiated research.
Keywords: Myelodysplastic syndromes, biorepository, natural history, quality of life
Introduction
The myelodysplastic syndromes (MDS) are a heterogeneous collection of clonal, hematopoietic disorders that are manifested clinically by cytopenias, transfusion needs, infection and bleeding events, and variable progression to acute myeloid leukemia (AML). The diagnosis is made primarily by morphological abnormalities within hematopoietic cells in the bone marrow [1,2]. Patients are then classified into one of several subtypes recognized by the World Health Organization (WHO) [3,4] and should undergo assignment of International Prognostic Scoring System (IPSS) risk scores [2,5]. Newer classification systems, such as the revised IPSS [6] (IPSS-R), have been developed that more effectively separate patients into distinct prognostic subgroups compared to the original scoring system [5]. These subgroups are associated with well-defined median lengths of time to AML transformation and overall survival (OS), ranging from a few months to several years [4].
Information about the natural history of MDS derives largely from data collected retrospectively, including the data that led to development of the current prognostic scoring systems. Consequently, complications of MDS and rates of evolution to higher-risk subtypes or to AML are either extrapolated or reported with wide ranges. Epidemiologic risk factors for MDS development or evolution are poorly defined, and assessment of patient-reported outcomes (PROs) are largely limited to companion studies of patients enrolling into clinical trials, and thus are not always reflective of the true spectrum of the MDS experience. Biospecimens are collected at a limited number of specialty centers, and there is a paucity of specimens collected serially, over the course of the disease. MDS progresses through a multi-stage process that can only be understood more completely by conducting a longitudinal analysis of patients over time.
Rigorous biospecimen collection is the foundation for this study and requires a high level of consistency and standardization. Several landmark federal standards have been issued as a framework for establishing effective biorepositories, including The Genomics and Personalized Medicine Act of 2006 [7,8], The President’s Council of Advisors on Science and Technology: Priorities for Personalized Medicine [8,9], and the NCI Best Practices for Biospecimen Resources [9]. All of these accelerate research initiatives for the U.S. population through harmonized biospecimen collection and processing in association with high-quality data capture.
For novel scientific, technological, and clinical practices development in MDS, access to data and biospecimens must occur at the community level, where most patients are being treated. Community- and academic-based recruitment of patients allows for the ascertainment of cases that may have been misdiagnosed, as well as cases with unique pathologies that may actually be precursors to MDS.
Diagnostic difficulties in MDS make uniform tissue acquisition and clinicopathologic interpretation challenging. Recently, the National Comprehensive Cancer Network (NCCN) recommended that the minimal initial evaluation for patients clinically suspected of having MDS include, along with history and physical examination, a complete blood count with leukocyte differential, reticulocyte count, bone marrow aspiration and biopsy for histopathology review as well as genetic testing for detection of recurring MDS-associated mutations or chromosomal abnormalities [10]. However, examination of bone marrow morphology by an experienced hematopathologist represents the diagnostic foundation in this disease [5,11,12].
It is increasingly recognized that PROs are essential to measure and optimize, especially for patients with chronic illness [13]. PROs are reports of health status from the point of view of the patient, without annotation by the clinician, and provide the ability to measure constructs such as physical functioning, mood, symptom burden, and quality of life that cannot be measured through medical tests or procedures [14]. In MDS, quality of life and symptoms such as fatigue are vital outcomes in their own right. Traditional outcomes such as overall survival and PROs also intersect, as has been shown with a recent randomized trial in which PRO monitoring was associated with improved survival [15]. Use of PROs in oncology may exert positive impact on health outcomes through improved physician/patient communication [16]. For MDS, little is known about the natural history of patients’ PROs, and how they may change with different interventions or across the natural evolution of the disease. This information will be critical for improving the quality of healthcare delivery in MDS.
Here, we describe the design and launch of the first publicly funded, prospective, longitudinal U.S. multi-site study designed to enroll MDS patients at the time of diagnosis from both community-based and academic medical centers to obtain high-quality biospecimens linked to epidemiological risk factors, clinical characteristics, and PROs. The primary goal of this study is to facilitate discoveries related to the natural history of MDS, including characteristics associated with misdiagnosis, predictors of progression from pre-malignant conditions to MDS, predictors of progression from MDS to AML, and factors associated with PROs in MDS patients. Criteria for quality control measures and feasibility of the study are based on the vanguard phase that includes the first 200 enrolled participants.
Methods
Study operation
The study is funded by contracts from the NHLBI, with collaboration from the NCI. Study conduct is overseen by a Steering Committee that advises the sponsor on policy issues, identifies obstacles to study conduct, and provides scientific leadership and operational guidance. An Executive Committee oversees day-to-day operations of the study, implementing the protocol as designed by the Steering Committee. Designated subcommittees reporting to the Steering Committee have responsibility for pathology, genetics, and publication guidance. Study operations are coordinated jointly by the Data Coordinating Center (DCC) at the Emmes Corporation in Rockville MD and the Central Laboratory & Biorepository (CL/B) located at the H. Lee Moffitt Cancer Center in Tampa, FL within its affiliate Moffitt Genetics Corporation (M2Gen). The DCC is responsible for the overall coordination and management of the study, operational and analytical methodology, monitoring the clinical sites, and study data management. The CL/B ensures that samples are properly collected, processed, annotated, and stored; and performs central pathology review for the study. Finally, a NHLBI appointed independent Observational Study Monitoring Board acts as an independent advisory group that is responsible for monitoring any safety concerns as well as providing operational recommendations to the sponsor regarding the conduct of the study.
Study design and eligibility
The National MDS Natural History Study enrolls a prospective cohort of patients age 18 years or older with suspected or documented MDS, MDS/MPN, AML with <30% myeloblasts without core binding factor abnormalities, known mutated RUNX1 or acute promyelocytic leukemia, or with idiopathic cytopenias of undetermined significance (ICUS), which includes patients with clonal cytopenias of undetermined significance (CCUS) [17–19]. Eligible patients are either undergoing diagnostic workup for suspicion of MDS, or have been diagnosed with de novo or therapy-related MDS within the past 12-months, have not received hematopoietic growth factors in the past 6-months and have not received any other therapy for MDS. Participants are excluded if they had a solid tumor or hematologic malignancy (except for in situ cancers of the skin [basal or squamous cell], uterine cervix, bladder, breast or prostate) or received radiotherapy or any non-hormonal treatment for cancer within two years of enrollment; have an established hereditary bone marrow failure syndrome, or have aplastic anemia, paroxysmal nocturnal hemoglobinuria, amegakaryocytic thrombocytopenic purpura, or isolated large granular lymphocyte leukemia [20].
Patients are recruited from US based academic and community centers participating in the NCI Community Oncology Research Program (NCORP) [21] and NCI’s National Clinical Trials Network (NCTN) [22]. The study has additional requirements beyond NCI standard regulatory requirements for site and investigator registration as described by the Clinical Trials Support Unit (CTSU) [23]. Participating sites must complete both investigator and study coordinator web-based training. Sites must also complete an inventory describing their process for inventorying specimen kits and handling of biospecimens which requires central laboratory approval. Interactions with the NCI research program are coordinated by Eastern Cooperative Oncology Group – American College of Radiology Imaging Network (ECOG-ACRIN; EA) within the NCTN [24].
Study schedule, eligibility and follow-up
Clinic sites submit unstained diagnostic bone marrow aspirates and biopsies (if performed) and peripheral blood samples to the CL/B, along with the diagnosis determined by the pathologist at the site using the WHO 2016 classification [25]. The study pathologist stains the bone marrow slides and peripheral blood smears at the central lab, conducts a blinded review of the submitted clinical data for diagnostic determination, and a computer algorithm compares the two diagnoses. If the central and local pathologists agree, then study assignment is complete. If there is disagreement due to differences in blast percentage of less than 3%, then the central laboratory pathologist’s diagnosis is used to determine the study assignment. For all other disagreements, a designated pathologist external to the study is consulted. The external pathologist is given access to all the case materials to make an unblinded final determination of the patient’s diagnosis. A Wright-Giemsa stained bone marrow aspirate and a hematoxylin and eosin (H&E) stained core biopsy slide is imaged with an Aperio AT2 digital whole slide scanner (Leica Biosystems Inc., USA). During initial acquisition, images of slide labels are created for each whole slide scan and linked to clinical data at the DCC. Stained slides, unstained slides, and images are accessioned for distribution to approved investigators.
Eligible patients with a diagnosis of MDS, MDS/MPN, ICUS or AML <30% blasts after local and central pathology review, as defined above, are formally assigned to a longitudinal follow-up schedule. Enrolled patients found not to have one of the above study classifications are eligible to be re-screened later for assignment to the longitudinal cohort, but are otherwise not followed. Follow-up of patients assigned to the longitudinal cohort, with peripheral blood samples and clinical status updates, occurs every 6 months or until any of the following: hematopoietic stem cell transplantation, transformation to AML (defined as a blast percentage ≥20% and a ≥ 50% increase from baseline blast percentage [26]), death, or withdrawal of consent (Table 1). Longitudinal follow-up cases also submit specimens whenever bone marrow sampling occurs.
Table 1.
The National MDS Study Sample and Data Submission Schedule.
| Procedure/material | Baseline | Medically indicatedmarrow procedure | Target month | Every 6 months thereafter | |||||
|---|---|---|---|---|---|---|---|---|---|
| 6 | 12 | 18 | 24 | 30 | 36 | ||||
| Medical History and epidemiologic exposures | X | ||||||||
| Tests performed in the prior 6 months: B12 level, serum folate, mean corpuscular volume (MCV), Red cell distribution width (RDW), ferritin, and thyroid-stimulating hormone (TSH), iron studies (iron, Total iron-binding capacity (TIBC), percent saturation). | Xg | ||||||||
| Reticulocyte count | X | ||||||||
| Vulnerable elders survey VES-13 | X | ||||||||
| Medical examinations, events, and proceduresa | X | X | X | X | X | X | X | X | |
| Disease staging (bone marrow aspirate required, and biopsy if performed)b | X | Xb | |||||||
| Peripheral blood sampling (CBC & differential)c | X | Xc | X | X | X | X | X | X | X |
| Germline tissue DNA Collection (12 eyebrow hairs with follicles; 6 hairs from each eyebrow and buccal swab)d | Xd | Xd | Xd | ||||||
| Patient reported outcomes-quality of life | X | Xe | Xe | Xe | Xe | Xe | |||
| Bone marrow aspirate sample collection and shipmentf | X | X | |||||||
| Peripheral blood sample collection and shipmentb | X | Xb,c | X | X | X | X | X | X | X |
| Slides for central pathology review | X | Xb | |||||||
| Redacted pathology and cytogenetic reports | X | Xb | |||||||
No other procedures required by the protocol, but data requested from procedures performed for medical care including peripheral blood counts, therapies and transfusions.
Subsequent disease staging performed as needed per standard of care.
Peripheral blood samples: 2X 10 mL in heparin sodium tubes; 3 mL in red top tube; 8.7 mL in PAXgene DNA extraction tube; 2.5 mL in PAXgene RNA extraction tube. Samples should also be obtained when it is suspected that a patient has evolved to AML (often typified by progressive cytopenias and/or circulating blasts), and whenever a bone marrow aspiration is performed.
Germline DNA will be extracted by the CL/B from eyebrow follicles and buccal swab. The eyebrow follicles and buccal swab are required at baseline, and may be optionally provided at 12 and 24 months. Hair follicles from the hairline may be submitted instead of or as a supplement to the eyebrow hair.
Post baseline, MDS cases and AML cases with <30% blasts without core binding factor or acute promyelocytic leukemia complete 4 questionnaires including QUALMS and FACT-G. Cases entered with ICUS complete only the PROMIS Short Form v1.0 – Fatigue 7a and EQ-5D-5L. After one year, the QOL instruments are completed annually.
Bone marrow: 5–10 mL in a tube with sodium heparin and 3 mL in a purple top tube with EDTA. Subsequent bone marrow samples collected as needed per standard of care.
If the patient is anemic, these tests should be performed in the prior 6 months.
Clinical information and patient reported outcomes
All participants complete a baseline questionnaire including items on demographic information, medical history, detailed tobacco and alcohol use derived in part from the questionnaire used in the Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial, [27] usual occupation and exposure history to environmental chemicals. PROs are measured at baseline, months 6 and 12, and annually thereafter using the following measures: the Quality of Life in Myelodysplasia Scale (QUALMS) [28] (a disease-specific symptom and psychosocial sequalae inventory), the Functional Assessment of Cancer Therapy-General form (FACT-G) [29] (a tool for quality of life assessment in chronic disease originally validated in cancer populations), the Patient Reported Outcomes Measurement Information System (PROMIS) Short Form-Fatigue (a 7 item assessment of a common cytopenia symptom), and the EuroQual EQ-5D-5L (a descriptive questionnaire with a single index value (thermometer) for health status that can be used to develop health utilities for use in health services studies such as decision analyses) [28–31]. ICUS cases do not complete the Qualms and FACT-G post baseline. The Vulnerable Elders Survey VES-13 is included as a baseline frailty assessment [32].
Tissue collection and storage
Biospecimen collection kits are prepared at the CL/B and machine-and human readable bar codes are used for tracking of individual collection tubes and returned to CL/B at indicated schedules (Table 1). Blood, bone marrow, eyebrow hair, and buccal cell smears are collected at the clinical sites and shipped to the CL/B for central processing and storage; in addition, histopathology slides are collected from the sites. Centralized tissue management and tissue processing procedures are performed in association with quality assurance assessments that are approved by the Steering Committee. Standard operating procedures (SOPs) associated with the collection, processing, and storage of samples has been established with quarterly review of a Quality Assurance Committee. A list of the study-associated SOPs is provided in Table S1, and detailed protocols will be available upon request and approval by the NHLBI. The practices provide a culture of Continuous Quality Improvement (CQI) through records management and system-associated documentation, control charts, periodic audits of compliance issues, and root cause analyses of any identified trend [33]. Cell viability and hemolysis based on the Loudon Hospital Specimen Integrity Chart for Hemolysis are used as general indicators of quality [34]. The CL/B collection is maintained at the Moffitt location, with biospecimen inventory management performed using the LabVantage (LabVantage Solutions, Inc, Somerset, NJ) Laboratory Information Management Solution (LIMS) application for electronic tracking, storage, and retrieval. Data elements include subject, diagnosis, tissue type, collection event, collection site, sample preparation, and collection methods. Total cell number per sample, number of aliquots, storage location and viability are recorded for cryopreserved cells. DNA integrity number, 260/280 ratio, total DNA and RNA quantity, concentration, and number of aliquots are tracked through unique Alias ID’s for purified nucleic acids. Ultimately the biospecimen collection will be transferred to the NHLBI Biorepository and made broadly available to the scientific community through the NHLBI’s Biologic Specimen and Data Repository Information Coordinating Center (BioLINCC) program [35].
Sample size
The goals of this study are to facilitate discoveries related to MDS disease natural history, including characteristics associated with misdiagnosis, predictors of progression from pre-malignant conditions to MDS, and from MDS to AML, and factors associated with PROs in MDS patients. A sample size of 2000 MDS cases will provide the ability to discriminate among prognostic subgroups, e.g. related to quality of life, clonal cell sub-populations, or diagnostic subgroups. For example, a 10% improvement in 5-year overall survival among a subgroup of 500 MDS cases would be detectable with greater than 80% power when the survival rate is 50% in the comparison group of 1500 assuming an alpha level of 5% and a uniform accrual rate with no more than 2% losses over 5 years. With the same follow-up assumptions, a 20% improvement would be detectable (power = 88%) from the same baseline level in an analysis of 500 participants with one-quarter of the cases in the subgroup with improvement.
Genetic annotation using a custom gene panel
The National MDS Natural History Study has started to perform molecular analyses on submitted specimens that will be used to annotate biospecimens in the resource. A molecular profiling panel with 96 gene targets identified from the literature as mutated in MDS [36] cases and related proliferative disorders are sequenced in the vanguard cases; these targets are detailed in Table S2. Sequencing was carried out on an Illumina HiSeq4000 sequencer using 150 bp long paired-end reads with a targeted mean read coverage of >600X for each sample.
Data management architecture
The study data management architecture consists of three primary electronic systems: the Oncology Patient Open Enrollment Network (OPEN); Medidata Rave; and GlobalTrace. Clinic site staff use the OPEN system to enroll patients in the study. OPEN initializes each new study participant in the Medidata Rave database, where study data are recorded on electronic case report forms. Clinical data in these systems are collected by ECOG-ACRIN and shared nightly with the Data Coordinating Center at Emmes. The GlobalTrace system is a secure, web-based materials management system designed for specimen tracking in multi-center studies. Clinic sites use GlobalTrace to document and track the shipment of bar-coded specimens to the CL/B. Specimens are managed at the CL/B using the LabVantage LIMS and other systems as described above. The complete database including clinical data and biospecimen inventory resides at the DCC.
Results
Participant baseline characteristics
Feasibility of the study was evaluated during a vanguard stage that piloted the study procedures in the first 200 enrolled participants. All 200 participants were accrued over the course of 12 months starting in June 2016. The rate of participant accrual increased over the initial months and exceeded 20 per month for the last 6 months of the period. Sixty-two sites entered patients with a median of 2 cases per site, while 5 sites entered 10 or more cases during the year (maximum of 14). The majority of sites with case entries were affiliated with the NCORP (43) and they entered 69% of the cases.
The majority of participants entered the study while undergoing evaluation for suspected MDS, with only 10 participants (5%) enrolled within the 6 months following an established, biopsy-supported diagnosis. The 200 vanguard participants included 49 (24.5%) MDS, 10 (5%) MDS/MPN, and 10 (5%) ICUS cases assigned to the longitudinal cohort. AML in 12(6%) and other cancers 23 (13%) were common explanations for assignment to the cross-sectional segment of the study. The other 96 individuals classified in the cross-sectional group had cytopenias without morphologic evidence of MDS and had the following general explanations: autoimmune or inflammatory conditions (12), chronic disease (10), drug-induced (3), iron deficiency (3), aplastic anemia (2), not determined (66).
As detailed in Table 2, the median age of all participants was 72 years (range, 21–90) and 74 years (range, 41–90) for MDS patients. Among all participants, 46% were female, 93% white, 3.5% African-American, 2% Hispanic. The median blood count for white blood cells was 3500 × 109/l, for neutrophils 2240 × 106/l; and for platelets 133 × 109/l. The hemoglobin level was 10.7 g/dl. Red blood cell transfusion rates were 31%, 30%, 20% and 10% in those with MDS, MDS/MPN, ICUS, and other disease classifications, respectively. Therapy-related MDS was observed in 3 of 49 patients (6.8%). MDS cases were classified according to the IPSS-R as follows: Very low (12%), Low (35%) Intermediate (22%), High (12%), Very high (10%), or not determined (8%). Inability to determine the risk in 4 cases was due to a failure of the cytogenetics assay in 1 and cytogenetics compliance problems in 3. All participants have stored images available for future retrieval. An exemplary bone marrow slide image and associated processing steps are shown in Figure 1.
Table 2.
Baseline characteristics.
| MDS | MDS/MPN overlap | ICUS | Cross-sectional | All volunteers | ||
|---|---|---|---|---|---|---|
| Number of patients | 49 | 10 | 10 | 131 | 200 | |
| Sex | ||||||
| Female | 27 (55.1) | 3 (30.0) | 2 (20.0) | 60 (45.8) | 92 (46.0) | |
| Male | 22 (44.9) | 7 (70.0) | 8 (80.0) | 71 (54.2) | 108 (54.0) | |
| Age | ||||||
| Median (Min, Max) | 74 (41,90) | 75 (63,84) | 73 (26,86) | 71 (21,88) | 72 (21,90) | |
| Race | ||||||
| American Indian or Alaska Native | 1 (2.0) | 0 | 0 | 2 (1.5) | 3 (1.5) | |
| Asian | 0 | 0 | 1 (10.0) | 1 (0.8) | 2 (1.0) | |
| Native Hawaiian or Other Pacific Islan | 0 | 0 | 0 | 1 (0.8) | 1 (0.5) | |
| Black or African American | 0 | 1 (10.0) | 0 | 6 (4.6) | 7 (3.5) | |
| White | 48 (98.0) | 9 (90.0) | 8 (80.0) | 121 (92.4) | 186 (93.0) | |
| Unknown | 0 | 0 | 1 (10.0) | 0 | 1 (0.5) | |
| Ethnicity | ||||||
| Hispanic or Latino | 1 (2.0) | 1 (10.0) | 2 (20.0) | 0 | 4 (2.0) | |
| Not Hispanic or Latino | 47 (95.9) | 9 (90.0) | 8 (80.0) | 130 (99.2) | 194 (97.0) | |
| Not Reported | 1 (2.0) | 0 | 0 | 1 (0.8) | 2 (1.0) | |
| Laboratory Results: (median, range) | ||||||
| Neutrophil Count (106/l) | 1648 (300,8900) | 6500 (900,26000) | 1535 (730,3600) | 2510 (150,34000 | 2210 (150,34000 | |
| Hemoglobin (g/dl) | 9.4 (5.9,11.9) | 9.85 (5.5,13.9) | 11.45 (9.3,15) | 11.2 (4.5,15.7) | 10.7 (4.5,15.7) | |
| Platelets (109/l) | 120 (13,635) | 318 (54,846) | 141.5 (48,257) | 133 (17,1591) | 133 (13,1591) | |
| WBC (109/l) | 3.5 (1.6,24.7) | 8.8 (3.7,30.6) | 3.3 (2.2,28.0) | 4.4 (1.1,66.0) | 4.1 (1.1,66.0) | |
| Therapy-related disease | ||||||
| No | 44 (89.8) | 10 (100.0) | 9 (90.0) | 123 (93.9) | 186 (93.0) | |
| Yes | 3 (6.1) | 0 | 0 | 1 (0.8) | 4 (2.0) | |
| Unknown | 2 (4.1) | 0 | 1 (10.0) | 7 (5.3) | 10 (5.0) | |
| Red Blood Cell Transfusion | ||||||
| No | 33 (67.3) | 7 (70.0) | 8 (80.0) | 119 (90.8) | 167 (83.5) | |
| Yes | 16 (32.7) | 3 (30.0) | 2 (20.0) | 12 (9.2) | 33 (16.5) | |
| Alcohol historya | ||||||
| Never | 12 (24.5) | 2 (20.0) | 2 (20.0) | 27 (20.6) | 43 (21.5) | |
| One or fewer drinks per day | 32 (65.3) | 5 (50.0) | 3 (30.0) | 69 (52.7) | 109 (54.5) | |
| More than one drink per day | 5 (10.2) | 3 (30.0) | 5 (50.0) | 32 (24.4) | 45 (22.5) | |
| Unknown | 0 | 0 | 0 | 3 (2.3) | 3 (1.5) | |
| Smoking history | ||||||
| Current smoker | 2 (4.1) | 1 (10.0) | 2 (20.0) | 13 (9.9) | 18 (9.0) | |
| Past smoker | 23 (46.9) | 7 (70.0) | 5 (50.0) | 51 (38.9) | 86 (43.0) | |
| Never smokedb | 24 (49.0) | 2 (20.0) | 3 (30.0) | 66 (50.4) | 95 (47.5) | |
| Unknown | 0 | 0 | 0 | 1 (0.8) | 1 (0.5) | |
| Environmental exposures | ||||||
| No | 25 (51.0) | 5 (50.0) | 4 (40.0) | 75 (57.3) | 109 (54.5) | |
| Yes | 24 (49.0) | 5 (50.0) | 5 (50.0) | 49 (37.4) | 83 (41.5) | |
| Unknown | 0 | 0 | 1 (10.0) | 7 (5.3) | 8 (4.0) | |
| Quality of Life | ||||||
| QUALMS - Median (Min, Max) | 66.3 (39.1,96.6) | 70.0 (37.9,88.6) | 72.0 (46.2,90.9) | 75.0 (39.4,97.0) | 72.3 (37.9,97.0) | |
| Promis Fatigue 7a - Median (Min, Max) | 52.2 (33.4,71.1) | 48.1 (41.9,60.6) | 51.5 (41.9,67.8) | 50.8 (29.4,74.8) | 50.8 (29.4,74.8) | |
| FACT-G - Median (Min, Max) | 83.0 (34.0,103.0 | 78.2 (34.0,94.0) | 83.3 (62.2,105.0 | 83.0 (29.7,108.0) | 83.0 (29.7,108.0) |
The highest amount of alcohol reported at any age for each subject.
Never smoked regularly for 6 months or longer.
Figure 1.
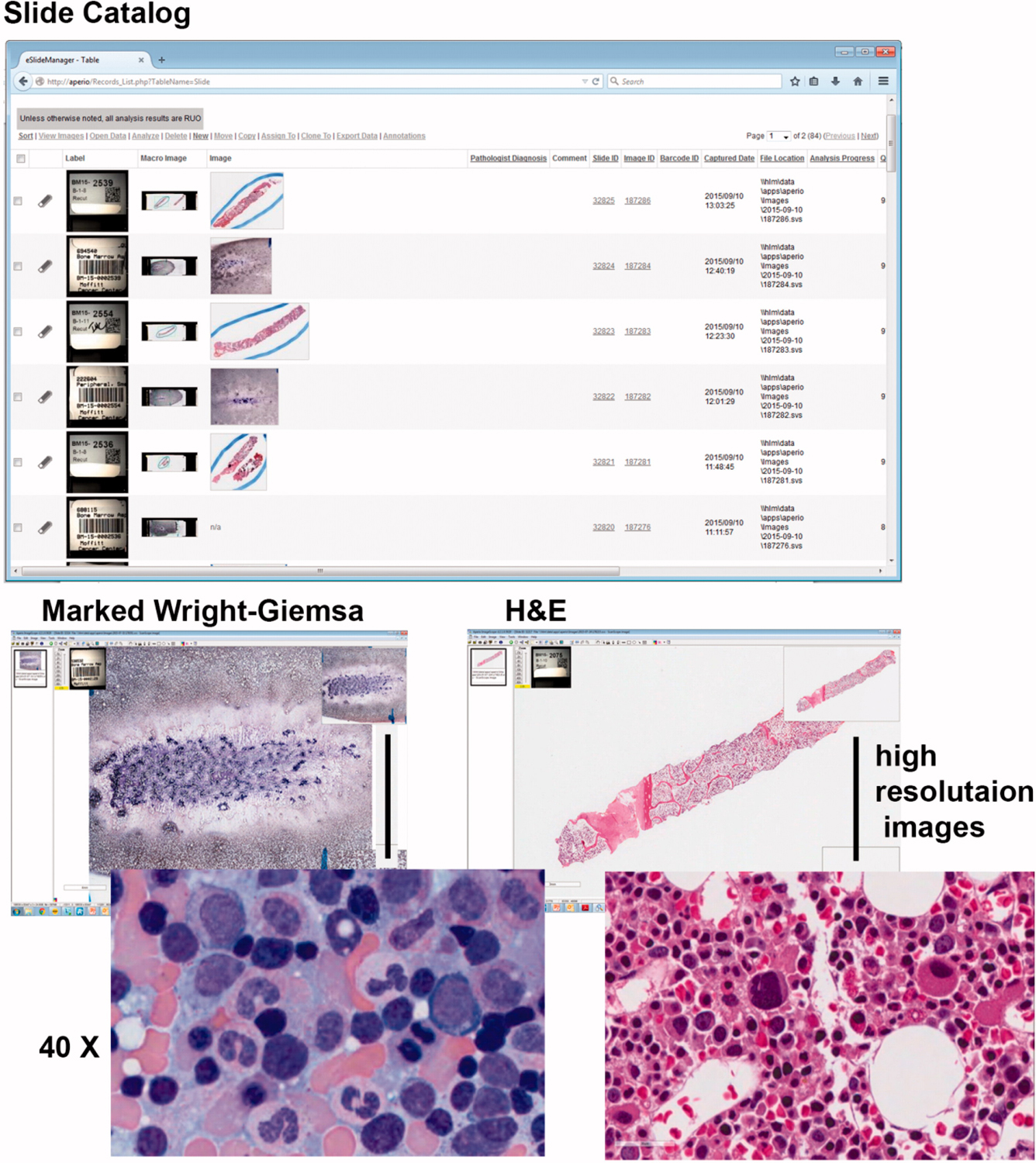
Image acquisition and storage. Pathologist preview slides and mark areas of highest cellularity prior to imaging the Wright-Giemsa slide. The H&E stained bone marrow biopsy is imaged in its entirety. Slides are imaged with an Aperio AT2 digital whole slide scanner (Leica Biosystems Inc., USA) through a 20x/0.75NA objective lens with an optical doubler lens to bring magnification to 40x. Whole slide images are created in SVS file format at a compression ratio of 15–20:1 and stored on secure network drive that is backed up twice daily. Images were accessed through a password-restricted database called Eslide Manager version 12.3.3.5049 (Leica Microsystems Inc, USA).
Over half of participants (52%) reported a history of ever smoking, with 9% current smokers and 43% former smokers. Most patients (77%) drank alcohol at some point in their lives with the following maximum intensity: 54.5% consumed one or fewer drinks per day and 22.5% consumed more than one drink per day at any age. Past environmental exposures occurred in 41.5% of patients, with the most common categories being cutting oils, motor vehicle oils, gasoline, fertilizers, pesticides, and herbicides. Rates of smoking, alcohol use, and environmental exposures were similar for the entire cohort and for the MDS/ICUS population.
Baseline, median PRO scores were 72.7 (range, 37.9–97.0) on the QUALMS, 83.0 (range, 29.7 –108.0) on the FACT-G, and 50.8 (range, 29.4–74.8) on the PROMIS-Fatigue Short Form. While there are no population norms for the QUALMS (it is MDS-specific) the baseline PROMIS-Fatigue Short Form scores are close to the population standard (mean of 50)[37].
Specimen acquisition and processing
Specimen acquisition and processing was successful for 194 participants (97%) during the baseline visit. Marrow aspirate samples were not obtained from 4 participants, and 2 withdrew consent prior to sampling procedures. Complete sample submissions were obtained from 191 cases; partial submissions from 3 (eyebrow hair not submitted, unable to obtain blood sample, and blood sample/missing eyebrow hair, respectively). A total of 14,152 aliquots for 16 specimen types were created and stored. The tissues collected from these participants are described in Table 3. The total number of bone marrow mononuclear cells recovered correlated with the original parent sample volume (r2 = 0.390, p < .0001). Sample viability, especially for cells obtained from the EDTA preservative, went from an average of 82% with 1-day shipping to an average of approximately 50% when shipping duration was four days or greater (Figure S1). Significant levels of hemolysis and clotting were not observed on this study from bone marrow or peripheral blood. The quality and quantity of DNA from four tissues is provided in Table S3.
Table 3.
Specimen resources available.
| Biorepository Collection for The National MDS Natural History Study | |
|---|---|
| Genomic DNA (bone marrow and peripheral blood) | Cell pellets (bone marrow) |
| Germline DNA (epithelial cells and hair follicles) | RNA (peripheral blood) |
| Viably frozen bone marrow cells (red cell depleted, CD34+ and CD34− | Viably frozen peripheral blood mononuclear cells (unseparated and CD3+ lymphocytesa) |
| Plasma (bone marrow and peripheral blood) | Serum |
| Stained hematopathology slides | Annotated, high-quality images |
| Quality measures and genetic annotation according to approved SOPs |
CD3+ T cells isolated on a subset of participants but discontinued due to cell limitations. This population can be isolated from frozen viably stored peripheral blood mononuclear cells upon investigator request and study approval.
Discussion
MDS remains an entity challenging to fully define and describe with respect to clinical, pathobiologic, and genomic evolution. Environmental causes outside of radiation or radiotherapy exposure and previous chemotherapy are not well understood, and the diagnosis itself can be complicated. For example, rates of diagnostic discrepancy between local and central review are still being analyzed and verified. Within the initial 375 patients enrolled in the study, discrepancies may be as high as 40%, with 25% being clinically meaningful [38]. The National MDS Natural History Study is designed to provide resources necessary to address these gaps in knowledge.
The vanguard stage of the National MDS Natural History Study established the feasibility of a large, nationwide, multi-center, prospective cohort study that will create a well-annotated, longitudinal epidemiologic, clinical, PRO data and a biospecimen repository to support future research related to the natural history of MDS. Specifically, the vanguard stage of the study showed the feasibility of recruiting patients with a rare disease through existing, nationwide site networks; that it is possible to identify MDS cases early in the natural history of the disease, when MDS is first suspected; that reconciliation of diagnoses, which are notoriously difficult in MDS, is possible through well-organized, centralized pathological review; that clinic sites have excellent compliance with a complex specimen collection procedure, ultimately facilitating the successful execution of detailed, standardized tissue processing and storage procedures that will facilitate establishment of a high-quality biospecimen inventory; that multiple electronic systems can be successfully integrated to enable enrollment of patients, collection of clinical outcomes, and tracking of biospecimens and all derivatives.
The vanguard study identified an unexpectedly large number of screened individuals without MDS reflecting the diagnostic practices within the population-based study. Based on these findings, the study has been modified to include a subgroup for longitudinal follow-up that fails to meet the current NCCN-based diagnostic guidelines for MDS/ICUS, but may be at risk for disease development. The revised cohort will include those with the presence of dysplasia, select karyotype abnormalities [39] or locally or centrally detected genetic mutations meeting minimally acceptable criteria for allelic variant presence (NRAS, KRAS, CEBPA, RUNX1, GATA2, IDH2, STAG2, ASXL1, SRSF2, TP53, BCOR, PTPN11, FLT3, U2AF1, EZH2, ZRSR2, PHF6, NPM1, WT1, CBL, TET2, IDH1, KIT, DNMT3A, or SF3B1)[40]. It is estimated that this protocol modification will increase the number of followed cases by 50% and permit increased understanding of the importance of genetic variants in these syndromes.
Given the results of the vanguard phase, we expect that the scientific community will be responsive to solicitations for new research proposals when the valuable and growing resource is made widely available. In general, we expect the study to enable investigators to address knowledge gaps in several key areas in MDS, including the improvement of diagnosis and prognosis through addition of genomic information, discovery of temporal relationships between disease evolution and management with quality of life, and risk factors for progression. Future longitudinal follow up will provide prospective information on disease progression and therapeutic outcomes that includes MDS vs. ICUS, lower-risk and higher-risk MDS or MDS to AML progression, biomarkers of response to hypomethylating agents [38], MDS with complex karyotypes, and individual pathologic subtypes of MDS.
Examining participant characteristics at this preliminary vanguard stage has already provided insight into MDS and related conditions in the US population. First, approximately one-quarter of patients with suspected MDS who undergo bone marrow evaluation actually have a myeloid malignancy, ICUS, or another malignancy. Second, the median age of participants diagnosed with MDS is slightly older than for all participants, there is a slight male predominance, and approximately one-third of patients with MDS or MDS/MPN have already received red blood cell transfusions. Third, approximately half of participants have ever smoked cigarettes, approximately one-fifth drink alcohol regularly, and 40% report environmental exposures that may have contributed to MDS development. Moreover, we have begun to amass a unique, longitudinally assembled baseline inventory of PROs specifically for patients with MDS.
Cell viability was impacted by the duration of time from sampling to receipt at the central laboratory. Viability decreased significantly with each additional day of transport. We have previously shown that the normal tissue samples being collected for germline DNA comparisons are also appropriate [41]. A molecular profiling panel of 96 gene targets (Table S2) previously identified as mutated in MDS cases and related proliferative disorders are being sequenced in the study and will be invaluable for annotation of the biospecimens in the resource.
Study entry is conditional on clinical suspicion of MDS. Thus, at baseline, patients have cytopenias and many fit a demographic profile (e.g. advanced age) that could be consistent with this diagnosis. Consequently, this study is not equipped to answer questions about certain pre-clinical MDS conditions, such as clonal hematopoiesis of indeterminate potential. In addition, as the study excludes patients who have received chemotherapy or radiation therapy within the previous two years it will be challenging to address questions concerning fast-onset therapy-related MDS. Still, the National MDS Natural History Study may offer some etiologic insights, e.g. by identifying risk factors for progression from ICUS to MDS; and the cross-sectional cohort may elucidate biological features useful for ruling out MDS among patients who display similar profiles.
In longitudinal studies, informative censoring is a natural consideration and factor of the study design. Patients with established MDS diagnoses will typically be older and have comorbidities. Many investigations that are launched using our study’s resources are likely to have mortality endpoints, and in this case, informative censoring can only occur when vital status is not ascertainable. Fortunately, vital status can often be determined even when patients are otherwise lost to follow-up. Furthermore, we have positioned ourselves advantageously to leverage the existing NCTN and NCORP site networks which have resources and experience to limit losses to follow-up in longitudinal studies. While patient follow-up at the time of AML transformation or hematopoietic cell transplantation is out of this study’s scope, we have coordinated with the Center for International Blood and Marrow Transplant Research to permit linkage of information of individuals who follow this therapeutic route.
One clear advantage of the National MDS Natural History Study is that it should be generalizable to all MDS patients in the US. It is well-known that participants who volunteer for medical research studies are often not accurate representatives of the target populations to which the research results are generalized. We have accounted for this previous limitation by including the NCORP site network, which brings cutting edge cancer research to patients at community medical centers, including minority and underserved communities. Patients from minority groups are under-represented in the vanguard cohort, likely due to excess recruitment from areas of the country where they are under-represented in the population and possibly from racial disparity in care [42]. Future recruitment efforts are focused on areas of the country where minority subjects represent a greater percentage of the potentially eligible population. We also point out that the overall approach of the National MDS Study is oriented toward inclusion of the entire stakeholder community through application of four key design principles, which are replicable for other natural history studies of rare and complex conditions like MDS: (1) Rigorous study design established through a collaborative process that also incorporates consideration for patients’ experience of illness; (2) Establishment of governance structures to oversee implementation of the design across a far-reaching network of clinical sites; (3) Well-coordinated effort between centrally placed laboratory and data processing services; and (4) Independent oversight by a safety monitoring board, which includes expertise in the management of biorepositories.
Finally, although we expect to establish a large bio-specimen archive we acknowledge that the DNA resource created by this proposal would not be of sufficient size for an endeavor like a stand-alone GWAS discovery study. However, our study cohort could contribute as an independent study population for candidate gene validation studies or for validating ‘hits’ from GWAS discovery studies. In addition, the acquisition of serial samples that are linked to uniformly acquired clinical information will provide insight into the contribution of genetic and environmental factors on tumor clonal evolution. Collection of cryopreserved peripheral blood, bone marrow mononuclear cells, and purified CD34+ stem cells will allow for functional testing and RNA expression analysis on specific cell populations.
In summary, the vanguard stage of the National MDS Natural History Study demonstrates the feasibility of a large-scale, prospective cohort study that will establish a longitudinal biospecimen inventory with associated clinical data to provide a resource for the scientific community studying MDS disease natural history. The resource has the potential to address open questions in MDS that require a large sample of patients in the early stage of their disease, which is not otherwise easily obtained in single-center or smaller multi-center initiatives.
Supplementary Material
Acknowledgments
The National MDS Natural History Study has been supported by US Federal Government Contracts HHSN268201400003I and HHSN268201400002I from the National Heart, Lung, and Blood Institute and additional funding by the National Cancer Institute to the participating member clinical centers in the NCORP and NCTN. Many individuals from the biorepository and coordinating centers contributed. In addition, we specifically acknowledge collaborators in study development: Ling Zhang, Eric Padron, Jeff Painter, Steven Kroft, Alexandra Harrington, Worta McCaskill-Stevens, Deborah Pearson, David Cella, Paul Jacobsen, Mary Horowitz, Wael Saber, Jason Thompson, Johannes Goll, Benjamin Ebert, Jason Walker, Robert Fulton, Jaroslaw Maciejewski and Lindsley Coleman. The National MDS Natural History Study thanks the study participants, as well as the investigator teams at the participating clinical sites.
Footnotes
Supplemental data for this article can be accessed here.
This material is published by permission of the Data Coordinating Center for The National MDS Study, operated by The Emmes Company, LLC for the National Heart, Lung and Blood Institute under Contract No. HHSN268201400002I. The US Government retains for itself, and others acting on its behalf, a paid-up, non-exclusive, and irrevocable worldwide license in said article to reproduce, prepare derivative works, distribute copies to the public, and perform publicly and display publicly, by or on behalf of the Government.
Potential conflict of interest
Disclosure forms provided by the authors are available with the full text of this article online at https://doi.org/10.1080/10428194.2019.1616186.
References
- [1].Jacobs RH, Cornbleet MA, Vardiman JW, et al. Prognostic implications of morphology and karyotype in primary myelodysplastic syndromes. Blood. 1986;67:1765–1772. [PubMed] [Google Scholar]
- [2].Kao JM, McMillan A, Greenberg PL. International MDS risk analysis workshop (IMRAW)/IPSS reanalyzed: impact of cytopenias on clinical outcomes in myelodysplastic syndromes. Am J Hematol. 2008;83:765–770. [DOI] [PubMed] [Google Scholar]
- [3].Greenberg P, Anderson J, de Witte T, et al. Problematic WHO reclassification of myelodysplastic syndromes. Members of the International MDS Study Group. J Clin Oncol. 2000;18:3447–3452. [DOI] [PubMed] [Google Scholar]
- [4].Greenberg P, Cox C, LeBeau MM, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood. 1997;89:2079–2088. [PubMed] [Google Scholar]
- [5].Maes B, Meeus P, Michaux L, et al. Application of the International Prognostic Scoring System for myelodysplastic syndromes. Ann Oncol. 1999;10:825–829. [DOI] [PubMed] [Google Scholar]
- [6].Schanz J, Tuchler H, Sole F, et al. New comprehensive cytogenetic scoring system for primary myelodysplastic syndromes (MDS) and oligoblastic acute myeloid leukemia after MDS derived from an international database merge. J Clin Oncol. 2012;30:820–829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Obama B The genomics and Personalized Medicine Act of 2006. Clin Adv Hematol Oncol. 2007;5:39–40. [PubMed] [Google Scholar]
- [8].Overby CL, Tarczy-Hornoch P. Personalized medicine: challenges and opportunities for translational bioinformatics. Per Med. 2013;10:453–462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Vaught J, Rogers J, Myers K, et al. An NCI perspective on creating sustainable biospecimen resources. J Natl Cancer Inst Monogr. 2011;2011:1–7. [DOI] [PubMed] [Google Scholar]
- [10].Greenberg PL, Stone RM, Al-Kali A, et al. Myelodysplastic Syndromes, Version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Net. 2017;15:60–87. [DOI] [PubMed] [Google Scholar]
- [11].Plasilova M, Risitano A, Maciejewski JP. Application of the molecular analysis of the T-cell receptor repertoire in the study of immune-mediated hematologic diseases. Hematology. 2003;8:173–181. [DOI] [PubMed] [Google Scholar]
- [12].Sanz GF, Sanz MA, Greenberg PL. Prognostic factors and scoring systems in myelodysplastic syndromes. Haematologica. 1998;83:358–368. [PubMed] [Google Scholar]
- [13].Rotenstein LS, Huckman RS, Wagle NW. Making patients and doctors happier - the potential of patient-reported outcomes. N Engl J Med. 2017;377:1309–1312. [DOI] [PubMed] [Google Scholar]
- [14].LeBlanc TW, Abernethy AP. Patient-reported outcomes in cancer care - hearing the patient voice at greater volume. Nat Rev Clin Oncol. 2017;14:763–772. [DOI] [PubMed] [Google Scholar]
- [15].Basch EM, Deal AM, Dueck AC, et al. Overall survival results of a randomized trial assessing patient-reported outcomes for symptom monitoring during routine cancer treatment. JAMA. 2017;318:197–198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Yang LY, Manhas DS, Howard AF, et al. Patient-reported outcome use in oncology: a systematic review of the impact on patient-clinician communication. Support Care Cancer. 2018;26:41–60. [DOI] [PubMed] [Google Scholar]
- [17].Kwok B, Hall JM, Witte JS, et al. MDS-associated somatic mutations and clonal hematopoiesis are common in idiopathic cytopenias of undetermined significance. Blood. 2015;126:2355–2361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Schroeder T, Ruf L, Bernhardt A, et al. Distinguishing myelodysplastic syndromes (MDS) from idiopathic cytopenia of undetermined significance (ICUS): HUMARA unravels clonality in a subgroup of patients. Ann Oncol. 2010;21:2267–2271. [DOI] [PubMed] [Google Scholar]
- [19].Steensma DP, Bejar R, Jaiswal S, et al. Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood. 2015;126:9–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Singleton TP, Yin B, Teferra A, et al. Spectrum of clonal large granular lymphocytes (LGLs) of alphabeta T cells: T-cell clones of undetermined significance, T-cell LGL leukemias, and T-cell immunoclones. Am J Clin Pathol. 2015;144:137–144. [DOI] [PubMed] [Google Scholar]
- [21].McCaskill-Stevens W, Lyss AP, Good M, et al. The NCI Community Oncology Research Program: what every clinician needs to know. Am Soc Clin Oncol Edu. 2013. 10.1200/EdBook_AM.2013.33.e84 [DOI] [PubMed]
- [22].Grubbs SS. The new national cancer institute national clinical trials network. Am Soc Clin Oncol Edu. 2012;146–148. [DOI] [PubMed]
- [23].Donovan C CTSU provides tools that link research and practice. ONS News. 2001;16:14–15. [PubMed] [Google Scholar]
- [24].Comis RL, Schnall MD. Opportunities for the ECOG-ACRIN Cancer Research Group within the new National Clinical Trials Network. Semin Oncol. 2015;42:1–3. [DOI] [PubMed] [Google Scholar]
- [25].Bennett JM. Changes in the Updated 2016: WHO classification of the myelodysplastic syndromes and related myeloid neoplasms. Clin Lymphoma Myeloma Leuk. 2016;16:607–609. [DOI] [PubMed] [Google Scholar]
- [26].Sekeres MA, Othus M, List AF, et al. Randomized Phase II study of azacitidine alone or in combination with lenalidomide or with vorinostat in higher-risk myelodysplastic syndromes and chronic myelomonocytic leukemia: North American intergroup study SWOG S1117. J Clin Oncol. 2017;35:2745–2753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Prorok PC, Andriole GL, Bresalier RS, et al. Design of the prostate, lung, colorectal and ovarian (PLCO) cancer screening trial. Control Clin Trials. 2000;21:273S–309S. [DOI] [PubMed] [Google Scholar]
- [28].Abel GA, Efficace F, Buckstein RJ, et al. Prospective international validation of the quality of life in myelodysplasia scale (QUALMS). Haematologica. 2016;101:781–788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Overcash J, Extermann M, Parr J, et al. Validity and reliability of the FACT-G scale for use in the older person with cancer. Am J Clin Oncol. 2001;24:591–596. [DOI] [PubMed] [Google Scholar]
- [30].Abel GA, Buckstein R. Integrating frailty, comorbidity, and quality of life in the management of myelodysplastic syndromes. Am Soc Clin Oncol Edu. 2016;35:e337–344. [DOI] [PubMed] [Google Scholar]
- [31].Abel GA, Klaassen R, Lee SJ, et al. Patient-reported outcomes for the myelodysplastic syndromes: a new MDS-specific measure of quality of life. Blood. 2014;123:451–452. [DOI] [PubMed] [Google Scholar]
- [32].Saliba D, Elliott M, Rubenstein LZ, et al. The Vulnerable Elders Survey: a tool for identifying vulnerable older people in the community. J Am Geriatr Soc. 2001;49:1691–1699. [DOI] [PubMed] [Google Scholar]
- [33].Francois P, Lecoanet A, Caporossi A, et al. Experience feedback committees: a way of implementing a root cause analysis practice in hospital medical departments. PLoS One. 2018;13:e0201067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Dugan L, Leech L, Speroni KG, et al. Factors affecting hemolysis rates in blood samples drawn from newly placed IV sites in the emergency department. J Emerg Nurs. 2005;31:338–345. [DOI] [PubMed] [Google Scholar]
- [35].Ross JS, Ritchie JD, Finn E, et al. Data sharing through an NIH central database repository: a cross-sectional survey of BioLINCC users. BMJ Open. 2016;6:e012769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Makishima H, LaFramboise T, Przychodzen B, et al. Clinical “MUTATOME” of myelodysplastic syndrome; comparison to primary acute myelogenous leukemia. Blood. 2013;122:518. [Google Scholar]
- [37].Brucker PS, Yost K, Cashy J, et al. General population and cancer patient norms for the Functional Assessment of Cancer Therapy-General (FACT-G). Eval Health Prof. 2005;28:192–211. [DOI] [PubMed] [Google Scholar]
- [38].Zhang L, Stablein DM, Epling-Burnette P, et al. Diagnosis of myelodysplastic syndromes and related conditions: rates of discordance between local and central review in the NHLBI MDS Natural History Study. Blood. 2018;132:4370 [Google Scholar]
- [39].Mohamedali AM, Gaken J, Ahmed M, et al. High concordance of genomic and cytogenetic aberrations between peripheral blood and bone marrow in myelodysplastic syndrome (MDS). Leukemia. 2015;29:1928–1938. [DOI] [PubMed] [Google Scholar]
- [40].Papaemmanuil E, Gerstung M, Malcovati L, et al. Clinical and biological implications of driver mutations in myelodysplastic syndromes. Blood. 2013;122:3616–3627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Padron E, Ball MC, Teer JK, et al. Germ line tissues for optimal detection of somatic variants in myelodysplastic syndromes. Blood. 2018;131:2402–2405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Zandberg DP, Hendrick F, Vannorsdall E, et al. Tertiary center referral patterns for patients with myelodysplastic syndrome are indicative of age and race disparities: a single-institution experience. Leuk Lymphoma. 2013;54:304–309. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


