Abstract
We illustrate a case of post-traumatic recurrent transient prosopagnosia in a paediatric patient with a right posterior inferior temporal gyrus haemorrhage seen on imaging and interictal electroencephalogram abnormalities in the right posterior quadrant. Face recognition area mapping with magnetoencephalography (MEG) and functional MRI (fMRI) was performed to clarify the relationship between the lesion and his prosopagnosia, which showed activation of the right fusiform gyrus that colocalised with the lesion. Lesions adjacent to the right fusiform gyrus can result in seizures presenting as transient prosopagnosia. MEG and fMRI can help to attribute this unique semiology to the lesion.
Keywords: epilepsy and seizures, neuroimaging, neurosurgery, paediatric surgery
Background
Acquired prosopagnosia, the inability to recognise familiar faces in the absence of visual or memory impairments, is due to lesions in bilateral or right-sided fusiform gyrus1–3 and is most often caused by ischaemic lesions but may also be secondary to encephalitis, haemorrhages and tumours.4 There are few reports of seizure-induced prosopagnosia,5 6 and to our knowledge, none in children. Here, we describe a case of prosopagnosia seizure semiology in a child with a right posterior inferior temporal gyrus (ITG) haemorrhage. We report results of face recognition area mapping using magnetoencephalography (MEG) and functional MRI (fMRI), which were performed as part of his presurgical investigation to explore the relationship between the lesion and the unique semiology.
Case presentation
A previously healthy, right-handed, 10-year-old boy presented to the emergency department (ED) because he could not recognise the faces of family members. This was the third such episode over the last 10 days. The first occurred after he had slipped on the ice at school and hit his head on the ground. At that time, he had an immediate headache, but no loss of consciousness, vomiting or convulsions. After getting up, he could not recognise his friends’ faces for about 30 min but had no visual problem per se, and no language impairments. A few days later he had a similar event, but it was shorter and less severe. Between events he would return completely back to normal. The third event occurred 10 days after the first. The father was called to the school because the teacher had noticed the patient was ‘confused’ and unable to recognise his classmates. The father and brother went to the school, but on their arrival, the patient was unable to recognise them. The father remarked that the patient was acting strange, seemed expressionless and that his speech was slow, but not slurred. This last episode, which lasted ~60 min, prompted the family to take him to the ED. Neurological examination was completely normal and ophthalmological evaluation found normal visual acuity and normal visual fields on Goldmann perimetry testing.
Investigations
Electroencephalogram (EEG) 2 weeks after his presentation showed occasional to frequent medium-voltage to high-voltage spike–wave complexes over the right occipital region. A repeat EEG 3 months later showed rare to occasional interictal epileptiform discharges (IEDs) in the right posterior quadrant, which prompted his neurologist to order an MRI to look for a focal lesion. The MRI, done 3 months after presentation, revealed a focal signal abnormality in the cortex and subcortical white matter of the right posterior ITG, which was consistent with a remote haemorrhage (figure 1A, B). The most likely diagnosis to explain the haemorrhage included a traumatic contusion or a very small occult cavernous malformation (CM). A follow-up MRI with susceptibility-weighted imaging confirmed the area of remote bleed and hemosiderin deposition, but because there was no gliosis or volume loss, a tiny CM became the preferred diagnosis and he was referred to neurosurgery for possible surgical intervention and presurgical evaluation. In particular, we wanted to verify using functional neuroimaging (MEG and fMRI) that his area of prior haemorrhage could give rise to his prosopagnosia semiology (see below).
Figure 1.
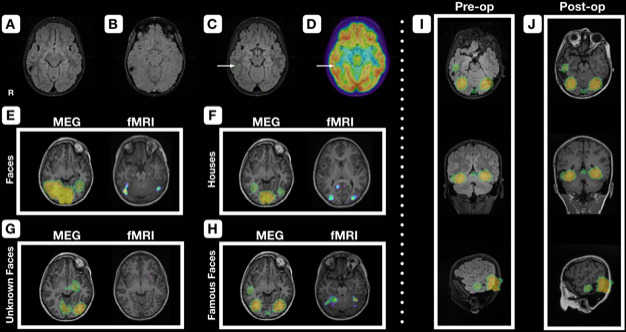
Preoperative MRI and positron emission tomography (PET), face recognition area mapping with magnetoencephalography (MEG) and functional MRI (fMRI), and preoperative and postoperative MRI with MEG-localised face recognition area overlay. A right posterior-inferior temporal focal lesion involving the cortex and subcortical white matter of the inferior temporal gyrus is seen on T2 fluid-attenuated inversion recovery (FLAIR) (A) and susceptibility-weighted imaging (SWI) (B) obtained 3 months after initial presentation. A susceptibility blooming artefact can be seen on SWI, in keeping with a remote bleed and hemosiderin deposition. Another axial cut of the T2 FLAIR (C) more inferior to the previous images shows the location of a small area of T2 FLAIR hyperintensity (arrow) co-localised with fluorodeoxyglucose PET hypometabolism (arrow) (D) at the crown of sulcus anterior-inferior to the hemosiderin staining. For face recognition area mapping, MEG results were displayed as maximal intensity projections while fMRI results were displayed on the patient’s T1 (E–H). Bilateral fusiform activation was seen in both MEG and fMRI in the faces condition (E), with greater activation on the right versus the left. For the houses condition, fusiform activation was greatly decreased with MEG and absent with fMRI (F), while activation was greatest in the primary and secondary visual cortices in MEG and fMRI, respectively. There was left fusiform activation with MEG in the unknown faces condition, but none for fMRI (G). Both modalities showed bilateral fusiform activation for the famous faces conditions with an overall higher amplitude event-related field compared with the unknown faces condition on MEG (H). MEG additionally showed activation of the right fusiform gyrus more anteriorly. The MEG activation map from (H) was overlaid onto the preoperative T2 FLAIR and postoperative T1 MRIs (I, J), demonstrating the adjacency of the MEG-localised face recognition area with the lesion and resection cavity.
Positron emission tomography (PET) co-registration with 3D fluid-attenuated inversion recovery (FLAIR) MRI revealed a small area of combined FLAIR hyperintensity and PET hypometabolism (figure 1C, D), which was anterior-inferior to the hemosiderin staining; this was included in his surgical plan.
Face recognition area mapping
To map the face recognition area with MEG and fMRI, we designed two tasks where the patient had to press a button when he saw (1) any upside-down face or house in a series of pictures of houses and faces, and (2) any familiar face in a series of famous and non-famous faces. The same tasks were repeated in MEG and fMRI, and activation maps were produced for each. Left-sided language lateralisation for presurgical planning was confirmed using fMRI as well (results not displayed). Detailed methods are reported in the Supplemental Methods.
Results
Houses versus faces
Bilateral fusiform activation for the M170 event-related field (ERF) was seen with MEG in the faces condition, with the right side showing greater activation than the left (figure 1E). Similarly, fMRI showed significant activation bilaterally, with greater activation in the right fusiform versus the left. Expectedly, fusiform activation was greatly decreased in the houses condition with MEG, and absent with fMRI; with the houses condition, activation in MEG and fMRI were greatest in the primary and secondary visual cortices, respectively (figure 1F).
Unknown versus famous faces
Activation was maximal in the left fusiform for the unknown faces condition in MEG, while no significant activation was seen with fMRI (figure 1G). Bilateral fusiform activation was seen in the famous faces condition in both MEG and fMRI (figure 1H). Notably, there was an overall higher amplitude ERF seen with MEG in the famous faces condition compared with the unknown faces condition and uniquely in the right fusiform gyrus adjacent to the lesion more anteriorly.
Treatment
The patient was not started on antiepileptic medications on presentation because he had no further events. Eleven months after his initial presentation he underwent a tailored resection of the lesion including the MRI and PET abnormalities. We had made the decision to operate for the following reasons. Based on neuroradiological interpretation by an experienced paediatric neuroradiologist (CSM), the lesion was thought to be a CM. Due to several studies in children that have suggested that CM outcomes, for both re-haemorrhage and seizures, are better after early surgery as compared with medical management or watchful waiting, surgery was offered.7–11 Finally, the parents were very concerned and anxious about possible re-haemorrhage and recurrent seizures and were very motivated to pursue the surgical option.
Outcome and follow-up
The patient had no postoperative neurological or visual field deficits, no trouble recognising faces and no further seizures at last follow-up (10 months postop). Postoperative MRI showed complete resection of the lesion. Pathology showed no CM or abnormal vessels but evidence of perivascular hemosiderin consistent with a traumatic bleed. Importantly, in retrospect, we would not have offered surgery if we had known with certainty that this was simply post-traumatic hemosiderin, and not a CM.
Discussion
Here we describe a patient who likely had focal seizures presenting as prosopagnosia, secondary to a traumatic haemorrhagic contusion adjacent to his right-sided posterior fusiform gyrus. We did not attempt to record seizures because they had abated, at least temporarily, by the time he was referred to neurosurgery. Therefore, we cannot say with 100% certainty that these were seizures (another possibility is transient vasospasm). However, these were indeed recurrent transient events which occurred in the context of (1) EEG abnormalities in the right posterior quadrant, (2) concordant MRI signal changes in the right posterior ITG, including the subcortical white matter and (3) PET hypometabolism encroaching towards the fusiform gyrus. Importantly, face recognition tasks performed during MEG and fMRI showed concordant activation within the fusiform gyrus directly adjacent to the area of haemorrhage and surgical resection (figure 1I, J). Interestingly, the lesion was in continuity with the MEG-localised face recognition area. Together these findings highly suggest that the bleed and/or hemosiderin were inducing seizures, either starting within or spreading to the fusiform gyrus, which manifested as a prosopagnosia semiology.
Few cases in the literature have described seizure-induced prosopagnosia. One case reported a left occipital arachnoid cyst associated with prosopagnosia seizures that disappeared with surgical resection.5 Another case of permanent prosopagnosia was reported following non-convulsive status epilepticus due to a left fusiform venous angioma.6
While the majority of patients with prosopagnosia present with bilateral fusiform lesions,1 a number of cases have described this phenomenon following unilateral, right-sided lesions.3 This appears to be the case in our patient who had a small haemorrhage in the right posterior ITG and right posterior quadrant IEDs, but no contralateral abnormalities on either MRI or EEG. In one report, electrical stimulation of the right inferior occipital gyrus was able to induce transient prosopagnosia,12 which suggests that activity even adjacent to the fusiform gyrus can induce prosopagnosia seizures.
We undertook functional neuroimaging with MEG and fMRI (1) to ensure that the location of his bleed in relation to his face recognition area could explain his prosopagnosia semiology, and (2) to be confident that surgical resection of the lesion would not produce deficits in language or face recognition. Of note, the same stimuli and tasks were used in both imaging modalities. Speech was lateralised to the left hemisphere. Overall, on the main tasks of interest, the results of the two modalities were largely concordant, with the exception of a few regions that were seen on MEG but not fMRI. Perception of faces versus houses localised to bilateral fusiform gyrus with a predominance of the right versus left, which is consistent with previous imaging studies.13 14 Interestingly, in the unknown versus famous faces task, left fusiform activation was seen for unknown faces while bilateral activation was seen for famous aces. This is consistent with prior studies suggesting that the left fusiform is involved in low-level processing of how face-like a picture is, while the right fusiform makes more categorical decisions of whether or not a stimulus is a face.15 Our results suggest that beyond this function, perhaps the right fusiform is also involved in higher-order functions in recognising whether a face is familiar/famous or not.
Overall, our case report adds to the sparse body of literature on prosopagnosia secondary to seizures. Functional neuroimaging not only was helpful to confirm that the proximity of our patient’s bleed to the face recognition area could explain his semiology but also adds evidence to the hypothesis that the right-sided fusiform gyrus is involved in differentiation between famous and non-famous faces.
Learning points.
Seizures may be considered in the differential diagnosis of prosopagnosia.
Lesions adjacent to the right fusiform gyrus can result in transient prosopagnosia secondary to seizures.
Functional neuroimaging techniques such as functional MRI and magnetoencephalography can be useful to localise the face recognition area and help find an aetiology for a patient’s prosopagnosia.
Our results suggest a role of the right fusiform in the differentiation between famous and non-famous faces.
Acknowledgments
Special thanks to Nassima Addour for her clerical assistance in formatting and submitting the paper, and for obtaining informed consent from the patient and parents to present this case report.
Footnotes
Contributors: JL and RWRD conceptualised the study, collected/interpreted the combined data and contributed to significantly drafting the manuscript and creating the figures. JM and SB designed, performed and analysed the magnetoencephalography (MEG) facial recognition task. ESM analysed the EEG that was performed concomitantly with the MEG to look for interictal epileptiform discharges. CSM performed and analysed the high-resolution MRI to look for a cavernous malformation versus traumatic contusion. SA performed exhaustive examination of the pathological specimens to rule out a cavernous malformation. JKC and DK designed, performed and analysed the functional magnetic resonance imaging facial recognition task. All authors contributed significantly to data acquisition/analysis and drafted/revised and approved of the final version of the manuscript.
Funding: This study was funded by Foundation of the Department of Neurosurgery, McGill University, grant number: (Charitable Registration No: 73954 0920 RR 0001).
Competing interests: None declared.
Patient consent for publication: Parental/guardian consent obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Haxby JV, Hoffman EA, Gobbini MI. The distributed human neural system for face perception. Trends Cogn Sci 2000;4:223–33. 10.1016/S1364-6613(00)01482-0 [DOI] [PubMed] [Google Scholar]
- 2.Damasio AR, Damasio H, Van Hoesen GW. Prosopagnosia: anatomic basis and behavioral mechanisms. Neurology 1982;32:331–441. 10.1212/WNL.32.4.331 [DOI] [PubMed] [Google Scholar]
- 3.Landis T, Cummings JL, Christen L, et al. Are unilateral right posterior cerebral lesions sufficient to cause prosopagnosia? clinical and radiological findings in six additional patients. Cortex 1986;22:243–52. 10.1016/S0010-9452(86)80048-X [DOI] [PubMed] [Google Scholar]
- 4.Barton JJ. Disorders of face perception and recognition. Neurol Clin 2003;21:521–48. 10.1016/S0733-8619(02)00106-8 [DOI] [PubMed] [Google Scholar]
- 5.Agnetti V, Carreras M, Pinna L, et al. Ictal prosopagnosia and epileptogenic damage of the dominant hemisphere. A case history. Cortex 1978;14:50–7. 10.1016/S0010-9452(78)80007-0 [DOI] [PubMed] [Google Scholar]
- 6.Wright H, Wardlaw J, Young AW, et al. Prosopagnosia following nonconvulsive status epilepticus associated with a left fusiform gyrus malformation. Epilepsy Behav 2006;9:197–203. 10.1016/j.yebeh.2006.04.010 [DOI] [PubMed] [Google Scholar]
- 7.Dammann P, Wrede K, Jabbarli R, et al. Outcome after conservative management or surgical treatment for new-onset epilepsy in cerebral cavernous malformation. J Neurosurg 2017;126:1303–11. 10.3171/2016.4.JNS1661 [DOI] [PubMed] [Google Scholar]
- 8.Englot DJ, Han SJ, Lawton MT, et al. Predictors of seizure freedom in the surgical treatment of supratentorial cavernous malformations. J Neurosurg 2011;115:1169–74. 10.3171/2011.7.JNS11536 [DOI] [PubMed] [Google Scholar]
- 9.Gross BA, Du R, Orbach DB, et al. The natural history of cerebral cavernous malformations in children. J Neurosurg Pediatr 2016;17:123–8. 10.3171/2015.2.PEDS14541 [DOI] [PubMed] [Google Scholar]
- 10.Kwon C-S, Sheth SA, Walcott BP, et al. Long-Term seizure outcomes following resection of supratentorial cavernous malformations. Clin Neurol Neurosurg 2013;115:2377–81. 10.1016/j.clineuro.2013.08.024 [DOI] [PubMed] [Google Scholar]
- 11.von der Brelie C, Kuczaty S, von Lehe M. Surgical management and long-term outcome of pediatric patients with different subtypes of epilepsy associated with cerebral cavernous malformations. J Neurosurg Pediatr 2014;13:699–705. 10.3171/2014.2.PEDS13361 [DOI] [PubMed] [Google Scholar]
- 12.Jonas J, Descoins M, Koessler L, et al. Focal electrical intracerebral stimulation of a face-sensitive area causes transient prosopagnosia. Neuroscience 2012;222:281–8. 10.1016/j.neuroscience.2012.07.021 [DOI] [PubMed] [Google Scholar]
- 13.Kanwisher N, McDermott J, Chun MM. The fusiform face area: a module in human extrastriate cortex specialized for face perception. J Neurosci 1997;17:4302–11. 10.1523/JNEUROSCI.17-11-04302.1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sergent J, Ohta S, MacDonald B. Functional neuroanatomy of face and object processing. A positron emission tomography study. Brain 1992;115:15–36. 10.1093/brain/115.1.15 [DOI] [PubMed] [Google Scholar]
- 15.Meng M, Cherian T, Singal G, et al. Lateralization of face processing in the human brain. Proc Biol Sci 2012;279:2052–61. 10.1098/rspb.2011.1784 [DOI] [PMC free article] [PubMed] [Google Scholar]


