Abstract
The protein chemerin (tazarotene-induced gene, TIG2; RARRES2) is a relatively new adipokine. Many studies support that circulating chemerin levels associate strongly and positively with body mass index, visceral fat, and blood pressure. Here, we focus on the specific relationship of chemerin and blood pressure with the goal of understanding whether and how chemerin drives (pathological) changes in blood pressure such that it could be interfered with therapeutically. We dissect the biosynthesis of chemerin and how current antihypertensive medications change chemerin metabolism. This is followed with a review of what is known about where chemerin is synthesized in the body and what chemerin and its receptors can do to the physiological function of organs important to blood pressure determination (e.g., brain, heart, kidneys, blood vessels, adrenal, and sympathetic nervous system). We synthesize from the literature our best understanding of the mechanisms by which chemerin modifies blood pressure, with knowledge that plasma/serum levels of chemerin may be limited in their pathological relevance. This review reveals several gaps in our knowledge of chemerin biology that could be filled by the collective work of protein chemists, biologists, pharmacologists, and clinicians.
Keywords: blood pressure, chemerin, hypertension, obesity
CHEMERIN BIOSYNTHESIS (SITES OF AND MECHANISM)
The earliest discoveries around chemerin were made in the immune system, classifying chemerin as a chemokine. Chemerin activates plasmacytoid dendritic cells, natural killer cells, and tissue macrophages.1–7 In 2007, chemerin was identified as an adipokine and the receptor through which it was originally identified to work termed chemerin chemokine-like receptor 1 (CMKLR1) or ChemR23; this receptor is now named Chemerin1.8–13
Context matters relative to the role chemerin plays in biological functions. Prochemerin is made predominantly within the hepatocyte and adipocyte. Figure 1 compares qualitative comparisons of chemerin mRNA levels in some human tissues vs. those in the rat. This figure was drawn to understand whether the rodent (rat) might serve as a model for the human, and to give a quick view of relative organ expression. We do not include tissues involved with reproduction (e.g., ovary, testes, and placenta) or bone in this figure, nor have we included studies done in the mouse. While studies of chemerin mRNA in mouse tissues are more plentiful than those in the rat, we report data in the rat because of its importance as a model in cardiovascular studies. The liver was set to a maximum value with all other values compared with it, given that the absolute magnitude of chemerin mRNA is greatest in the liver of both species. The human levels are reported from the Human Atlas (https://www.proteinatlas.org/ENSG00000106538-RARRES2/tissue). The rat measures, less in number because of the size of the study, were done in the laboratory of one of the authors, Dr Adam Mullick. Where a measure is absent in this specific rat study, the bar is marked with not yet measured or a reference 14,15 is placed for a different study that validates the qualitative expression of mRNA in that specific rat tissue. Secreted prochemerin is cleaved at the N-terminus to form chemerin (ref. 16; Figure 2). This molecule (Chem 20–163 or a 143 amino acid protein) is proteolytically degraded by a host of diverse enzymes that include carboxypeptidases, cathepsins, elastase, Factor XIIIa, Factor XIa, chymase, plasmin, and tryptase, to name a few.7,16–25 Importantly, some of these enzymes are involved in the renin–angiotensin system and are targets of currently used antihypertensive medications. The intersection of therapeutics with chemerin processing and thus ultimate functioning in the body will be later discussed.
Figure 1.
Expression of chemerin mRNA in human and rat organs. For both species, the liver was set to 1 given that the absolute magnitude of chemerin mRNA is greatest in the liver of both species. The human levels are reported from the Human Atlas (https://www.proteinatlas.org/ENSG00000106538-RARRES2/tissue) where mRNA is a conglomerate of that expressed in a Consensus, HPA and GTEx dataset. The rat measures, less in number because of the size of the study, were done in the laboratory of one of the authors, Dr Adam Mullick and were measured relative to RiboGreen expression. Where a measure is absent in the rat study, the bar is marked with not yet measured (NYM) or a reference is placed for a different study that validates the expression of mRNA in that specific tissue.
Figure 2.
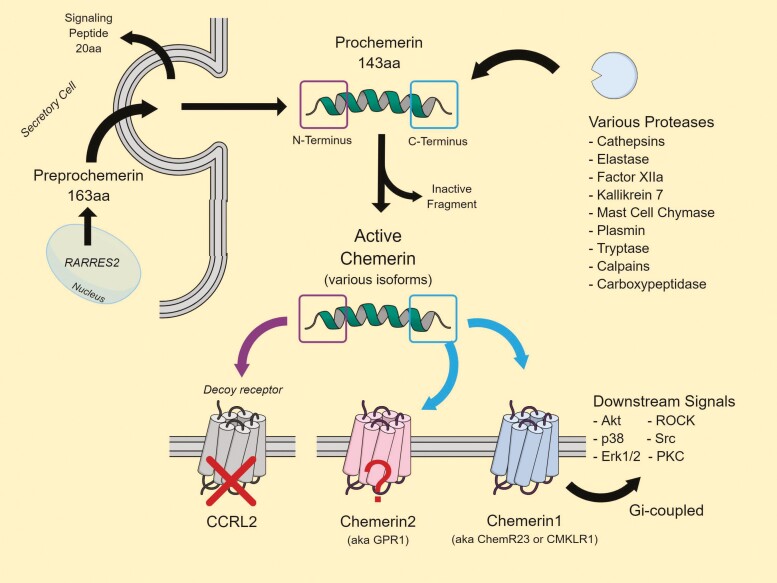
Chemerin secretion, processing and functioning at membrane bound receptors (Chemerin1 and Chemerin2) and chaperone receptor CCRL2. Abbreviations: aa, amino acid; Erk, ERK MAPK; p38, p38 MAPK; RARRES2, retinoic acid receptor responder 2, the chemerin gene.
Chemerin isoforms are named such that number after chemerin refers to the length of the isoform relative to the original 163 amino acid peptide. These include chemerin 125, chemerin 152, chemerin 154, chemerin 155, chemerin 156, chemerin 157, chemerin 158, and chemerin 163 with chemerin 157 producing the most potent responses at Chemerin1. We will term these r/h isoforms to indicate rat/human. All mouse isoforms are 1 amino acid shorter than the rat/human. Chemerin isoforms do not possess identical biological activity. In fact, the majority of circulating chemerin is not bioactive, at least not in the human.26 Chemerin’s overall biological activity at a site is determined by the amount of chemerin 20–163 protein present, and the collective enzymes present that can metabolize chemerin to various isoforms. Biological activity of chemerin isoforms has been compared primarily in cellular chemotaxis and intracellular calcium assays. While mass spectrophotometric measurement of some isoforms has been done,18 determination of a “fingerprint” of chemerin isoforms for a specific tissue has yet to be achieved. It is important to note that most assays which measure chemerin do not discriminate between the above listed chemerin isoforms. The groups of Leung and Morser have contributed mightily to this field, and have made some isoform-specific antibodies and enzyme-linked immunosorbent assays, as well as a study in tissue-specific processing of chemerin (approaching a “fingerprint”) in the obese mouse.18,24,25 Isoforms of chemerin become an issue in determining whether chemerin measures from circulating blood or plasma are a useful biomarker because isoforms can have opposite biological effects, such as anti-inflammatory actions.27,28 Chemerin isoforms have not been stratified to different magnitudes of blood pressure.
The role of chemerin in hypertension is complicated by the possibility that chemerin processing may be directly impacted by antihypertensive drugs. For example, angiotensin-converting enzyme degrades chemerin 20–163 to inactive chemerin 152 due to angiotensin-converting enzyme’s action as a carboxypeptidase.24 Angiotensin-converting enzyme inhibition would thus be predicted to increase the concentration of more active chemerin isoforms. Similarly, chymase (also an angiotensin II producer) metabolizes chemerin to both active and inactive forms.29 The angiotensin-converting enzyme inhibitor fosinopril reduced the elevation of serum and renal chemerin in a streptozocin-induced diabetic nephropathy in the rat.30 Finally, the PPARgamma agonists rosiglitazone, pioglitazone,14 and the angiotensin receptor antagonist irbesartan31 reduced the elevation in chemerin protein and Chemerin1 receptor observed in the kidney of the streptozocin-induced diabetic rat. For these latter studies, it is simply unclear if chemerin levels were reduced because disease was being treated vs. chemerin processing being modified by these drugs.
CHEMERIN RECEPTORS AND TOOLS FOR CHEMERIN BIOLOGY
There are 3 receptors for chemerin—Chemerin1, Chemerin2, and C–C chemokine receptor like 2 (CCRL232,33). All receptors are members of the G-protein-coupled receptor family and are recognized by the International Union of Pharmacology (Figure 2; www.guidetopharmacology.org; target id = 78 for CCRL2; target id = 79 for chemerin receptor1; target id = 82 for chemerin receptor2). We repeat that chemerin receptors have had name changes in the last ten years. Chemerin1 was formerly known as ChemR23 or CMKLR1, and Chemerin2 as G-protein-coupled receptor 1 (GPR1). Chemerin1 and Chemerin2 mediate the direct biological effects of chemerin (Figure 2). CCRL2 is described as a chaperone for chemerin.32 Chemerin isoforms produced endogenously serve as the cognate ligands for individual chemerin receptors (Figure 3). Resolvins also appear to be agonists (substances that stimulate receptor function) at Chemerin1.32 While antibodies are available for chemerin receptors, most of these antibodies have not been validated in experimental samples in which the chemerin receptor protein is knocked out/abolished.
Figure 3.
Experimental tools to investigate the chemerin/chemerin receptor axis. Abbreviations: ELISA, enzyme linked immunosorbent assay; KO, knockout; RT-PCR, reverse transcriptase-polymerase chain reaction. Red capped lines = inhibition. Green arrow = stimulation.
Pharmacological tools to study chemerin receptor function are also available but are not numerous—this is a field in the process of development. The agonist chemerin-9 constitutes residues 149–157 of the full-length peptide, mimics r/h chemerin 157, and retains biological activity at both Chemerin1 and Chemerin 2 receptors34 (guidetopharmacology.org). The Chemerin1 receptor antagonist CCX832 was developed by Chemocentryx and validated within our laboratory32 (Figure 3). These 2 tools—chemerin-9 and CCX832—have been the most commonly used tools in experimental laboratories. Other tools are being developed. For example, Graham et al.35 developed 2-(α-naphthoyl)ethyltrimethylammonium iodide as a Chemerin1 antagonist; this molecule is recognized by IUPHAR as such. Chemerin2 antagonists are not known.
Our team was also involved in the development of a different type of pharmacological tool to reduce chemerin expression vs. mimicking or antagonizing its effects. Dr. Adam Mullick of Ionis Pharmaceuticals created a series of antisense oligonucleotides (ASOs) that facilitate degradation of chemerin pre-mRNA through RNase H1-dependent mechanisms36 (Figure 3). As such, this destroys the ability of the cell to make chemerin protein. Due to hepatic and extrahepatic expression of chemerin, robust inhibition of chemerin at all sites of its expression requires the most potent ASO design. This is achieved by using Generation (Gen) 2.5 ASOs that contain a bicyclic sugar, 2′,4′-methylene bridged nucleic acid with a (S)-constrained ethyl modification (cEt). Importantly, RNase H1-active ASOs are designed as gapmers, as RNA cleavage is only supported at positions containing unmodified sugars. Thus, a typical Gen 2.5 design is a 16mer 3-10-3, with the 3 flanking nucleotides at the 5′ and 3′ ends containing cET modified sugars and a gap of 10 nucleotides containing DNA. As with all ASOs, the nucleotides are connected via a phosphorothioate backbone which is important for nuclease resistance and pharmacokinetic properties necessary for tissue uptake. Lastly, the same Gen 2.5 ASO chemerin with addition of a 5′-Trishexylamino-(THA)-C2GalNAC3 5′endcap (GalNAc) targets the ASO specifically to the liver. This is because of the high affinity of N-acetylgalactosamine (GalNAc) for the asialoglycoprotein receptor, which is highly expressed in hepatocytes.37 These tools allow us to begin to dissect contributions of chemerin to blood pressure regulation and will be discussed within this review.
CHEMERIN’S MECHANISTIC INFLUENCE OVER BLOOD PRESSURE AND HYPERTENSION
As evidenced by the evolution of chemerin’s classification from chemokine to adipokine, chemerin is involved in a wide variety of physiological processes. The cardiovascular system, especially as it relates to hypertension, is one such system in which chemerin has a multiplicity of effects. While there is not yet a clear understanding of how chemerin participates in human hypertension, we can begin to piece this puzzle together by understanding its individual parts. Below, we address each of the systems/organs well-established to play a role in blood pressure (dys)regulation, and how chemerin modifies the function of these important elements of the cardiovascular system (Figure 4). Below, we offer an introduction to chemerin function in cardiovascular organs. Because of reference limitations, we have not been able to exhaustively reference the number of papers for the totality of studies done in each organ. We apologize to our colleagues for this.
Figure 4.
The physiological actions of chemerin in primary organs of the CV system. ? = insufficient basic research.
Our overall opinion at this time is that the mechanistic chemerin research has been deepest in the vascular system; this section of this review is the most thorough in its references and mechanistic discussion. This is in part because this is our expertise. This focus does not mean that vascular work on chemerin is yet sufficient, nor does it mean that no research has been done in other systems. We do not mean to dismiss any organ as being unimportant by citing fewer references. It is a goal of this review to highlight that there is significant important work yet to be done on chemerin in tissues of the cardiovascular system. This is a call to those far more expert than we in these areas to meet the challenge of mechanistic studies in these areas.
The blood vessel
The vasculature, both arteries and veins, contain an endothelium, smooth muscle layer, adventitia, and perivascular adipose tissue (PVAT). The first 3 listed layers are well established. PVAT is gaining ground to be recognized as a vessel tissue layer of its own. In regard to chemerin-stimulated vasoreactivity, the aorta and mesenteric resistance vessels are the most studied at this time.
Chemerin, either as chemerin-9 or full-length recombinant peptide, causes contraction of isolated arteries (rat and human) and potentiates the effect of established vasoconstrictors such as endothelin-1.38–45 Importantly, chemerin-induced contraction is significantly enhanced when the endothelium is removed.38,42 Specific parts of the formally recognized blood vessel—the endothelial cell, the smooth muscle cell, and the sympathetic nerves that innervate these tissues—are next considered.
Endothelium
The presence of a healthy endothelium clearly influences chemerin physiology. In endothelial cells, chemerin increases reactive oxygen species and may lead to decreased nitric oxide production.41,45,46 Nitric oxide synthase inhibitors increased the magnitude of contraction caused by chemerin-9,38 suggesting that chemerin might interact with receptors within the endothelium to reduce contraction or play a role in chemerin disposition. However, the full ability of chemerin-9 to cause contraction was only revealed with the removal of the endothelium, an action that is greater than inhibition of nitric oxide synthase.38 Direct endothelium-dependent contraction and/or relaxation stimulated by chemerin or chemerin-9 has not been reported. If observed, such an action would occur in opposition to the ability of chemerin to increase reactive oxygen species in endothelial cells, typically considered an event that promotes contraction. Understanding how chemerin affects the endothelial cell is important given that endothelial cell dysfunction is a hallmark of cardiovascular diseases, including hypertension.
Smooth muscle
Chemerin1 is present on the smooth muscle cell, with the caveat that antibodies used in the assays that would determine this (immunohistochemistry, Western analyses) have not been validated in a Chemerin1 knockout. As stated above, chemerin-9 and recombinant chemerin cause direct arterial contraction. Chemerin inhibits cyclic adenosine monophosphate production42 and contraction stimulated by chemerin is inhibited by pertussis toxin,40,42 supporting the role of the G protein Gi in chemerin-stimulated signal transduction. Go proteins are also activated by Chemerin1.47 Activation of these pathways supported by these G proteins is consistent with chemerin causing vascular contraction. Multiple groups agree that Chemerin2 (aka GPR1) is not involved in smooth muscle responses to chemerin.40,42 However, chemerin receptor signaling may be more complicated than previously appreciated. Recombinant r/h chemerin 157 and its analog chemerin-9 elicit selective signaling (also known as biased signaling or signaling by biased agonists). This means that different chemerin isoforms elicit different second messenger signaling through the same receptor, making these events appear to be conducted through different receptors. For example, chemerin-9 but not the recombinant, full-length chemerin elevates intracellular calcium in a manner antagonized by CCX 832.40 Finally, chemerin is a mitogen in vascular smooth muscle cells and, when infused in the mouse, elevates blood pressure.48,49 This proliferative effect could influence the long-term remodeling of blood vessels in hypertension.
PVAT
We consider PVAT as an independent layer of the vessel, given that it can be readily dissected from the adventitia. There is increasing support that PVAT plays a role in creating a healthy/unhealthy environment for the vessel. Both chemerin and Chemerin1 receptor can be found in PVAT of experimental rodents and human.38,43 In fat, chemerin plays a beneficial autocrine role in adipogenesis9 but this specific role has not been investigated in PVAT. If there is a PVAT-specific role to chemerin, it makes this layer of the blood vessel more relevant in blood pressure regulation due to the proximity of the site of chemerin synthesis and its receptor expression in vascular smooth muscle cell and nerve.
Microvasculature
Aside from the cell/layer-specific effects of chemerin on the microvasculature, chemerin promotes angiogenesis, which is more elevated in type 2 diabetes mellitus patients with microvasculature complications.50,51 Chemerin’s positive association with angiogenesis is typically viewed through the lens of carcinogenesis, an area with a greater literature.52
The sympathetic nervous system
The cell type in PVAT is predominantly adipocytes, but other cells and tissue types are present. These include sympathetic nerves that may be found in the connective tissue space between the smooth muscle and adipocyte layers.53 Chemerin has at least 2 functions in the vascular sympathetic nerve. First, chemerin made in PVAT promotes electrical-field stimulation (EFS)-induced superior mesenteric arterial contraction (a way to test nerve-mediated contraction) through activation of Chemerin1 receptor.39 EFS-induced contraction was determined to be both nerve- and adrenergic receptor-dependent, suggesting EFS activates sympathetic nerves. Second, exogenous chemerin-9 potentiates EFS-induced arterial contraction. These experimental findings in the rat support that chemerin enhances sympathetic nerve function. This is important in considering the contributions chemerin may make to blood pressure regulation and the multiple levels at which the sympathetic nervous system controls and regulates blood pressure.
The adrenal is part of the sympathetic nervous system and exerts short-term (medulla via epinephrine) and long-term (cortex via mineralocorticoids) control over blood pressure. Chemerin is found in both portions of the adrenal but Chemerin1 is only present in the cortex.54 Chemerin mRNA transcripts are found in the whole adrenal (at rather high levels in human)55 (www.proteinatlas.org) but it is not clear whether 1 or both divisions of the adrenal regulate chemerin-dependent blood pressure responses. The EFS experiments mentioned previously suggest that sympathetic nerve stimulation may lead to the release of chemerin. If chemerin is secreted in the adrenal medulla in response to sympathetic nerve activation, the receptors in the cortex could be activated. While the function of Chemerin1 in the cortex is unknown, the cross-section of chemerin and mineralocorticoids on long-term control over blood pressure warrants further investigation.
It is less clear whether the central nervous system uses chemerin and its receptors to regulate blood pressure, either in the short or long term. Studies in this area investigate the role of chemerin in central control of appetite/feeding behavior.56 Pig, cow, rat, and human brain contain mRNA and protein for chemerin as well as the Chemerin1 and Chemerin2 receptors.57,58 It is not known whether chemerin (including any of its metabolites) made from the periphery can cross the blood brain barrier, but 1 study suggests it might. Chemerin was given intraperitoneally to the rat, and decreased food intake and body weight compared with vehicle control, an event thought of as being centrally dependent.59 This particular area is ripe for mechanistic studies given the important control the central nervous system possesses over blood pressure and feeding.
The heart
Knowledge of chemerin expression, chemerin receptors and chemerin function in the heart is sparse. Chemerin mRNA is expressed in the heart of the rat, in which chemerin is a negative inotrope60 and may induce cellular insulin resistance.61 Chemerin, stimulated by tumor necrosis factor alpha, induced apoptosis in mouse cardiomyocytes.62 Chemerin processing is potentially regulated by peptidase inhibitor 16, a protein upregulated in cardiac disease. Specifically, upregulated peptidase inhibitor 16 inhibited chemerin activation in cardiomyocytes.63 While basic research on the effects of chemerin in the heart are sparse compared with that of the vasculature, epidemiological evidence supporting chemerin’s role in cardiac disease, many times associated with hypertension, has grown. Specifically, there is a positive correlation between chemerin protein serum/plasma concentration and various pathologies associated with the heart. These include atherosclerosis, hypertension, and atrial fibrillation,60 along with the more general designation of cardiovascular disease. Heart failure is a disease in which serum chemerin concentration may be a novel prognostic indicator.64–66
Study of chemerin in cardiac (dys)function is an important new avenue of research. It is unknown whether the association of chemerin levels with cardiac dysfunctions described above are because of direct effects of chemerin on the (human) heart or are the results of the long-term secondary insults of chemerin elsewhere in the body (adipose tissue health, immune cell infiltration, sympathetic nerve control, and elevated blood pressure).
The kidneys
As found in literature for the heart, there are plentiful data from humans suggesting chemerin serum/plasma concentration is positively correlated with renal disease. However, multiple groups support the idea that elevated levels of chemerin in patients with chronic kidney disease are not because of overproduction of chemerin (at least by adipose tissue) but due to poor renal elimination.67–74 Dialysis directly decreased plasma chemerin concentration,67 supporting this idea.
While multiple groups have shown that the Chemerin1 receptor was not expressed in the kidney,9 others support its presence in rat and pig.75 Moreover, chemerin mRNA has been measured in the rat kidney76 and pig kidney.77 In the rat, a renal-specific induction of chemerin expression is observed in a model of hypertensive nephropathy.76 Importantly, in this same model, circulating chemerin levels were not elevated in disease. These cited studies do not, however, describe what the function of chemerin within the kidney might be. This underscores an important point of this review: the local/paracrine actions of chemerin may be the most important to biological function.
The immune system
This subject has been covered by excellent reviews (ref. 78, for example). The inflammatory/anti-inflammatory actions of chemerin remain an important aspect of chemerin’s function, especially given that hypertension has been described as an inflammatory disease. Briefly, active chemerin can serve as a potent chemoattractant for natural killer cells,79 macrophages,1 and dendritic cells.80 Activation of each of these cell types can be linked to an increase in blood pressure.81 Because of chemerin’s chemotactic nature, plasma circulating chemerin is an important component to the immune axis of chemerin. However, there is mounting evidence that plasma chemerin is not directly associated with increases in blood pressure. Finally, forms of chemerin have been described as anti-inflammatory, an area not without controversy.27,28 These findings, coupled with the ability of chemerin to function in potentially biased means at its receptors, have further confused our understanding of chemerin in the inflammatory system.
CHEMERIN MEASURES IN NORMAL AND HUMANS WITH CARDIOVASCULAR DISEASE: WHAT DOES IT MEAN?
Since its discovery, dozens of studies have reported on measurements of circulating (serum or plasma) chemerin, with the idea that such a measure would serve as a potential biomarker for cardiovascular disease. Enzyme-linked immunosorbent assays have been the most used analytic assay. These assays have allowed the field to gain a sense of what is a level of circulating chemerin protein is in humans, both healthy and with disease. Figure 5 reports findings from over 2 dozen human studies, not exhaustive of what is found in the literature.82–105 Each study found circulating chemerin to be in the µg/l range. Children and normal humans were at the low 100 µg/l range. Two points can be made from this figure. First, humans of different ethnicities are represented. Notably, Chinese populations presented with among the lowest level of circulating chemerin protein in what are considered clinically healthy adults (Figure 5). Second, the highest levels of circulating chemerin are associated, generally, with the most severe disease. This is not a perfect association, given that morbidly obese patients had relatively lower levels of circulating chemerin.86
Figure 5.
Compilation of reported (superscripted numbers) chemerin concentration (plasma or serum) in human, from 0 to 400 µg/l. Abbreviations: HTN, hypertensive; Met, metabolic; NT, normotensive; PCOS, polycystic ovarian syndrome; T2DM, type 2 diabetes mellitus.
Importantly, circulating chemerin correlates with the amount of visceral but not subcutaneous fat.106–108 Visceral fat quantity and quality are associated with increased cardiovascular disease in the human,108–111 making observations about chemerin consistent with these general findings. However, circulating chemerin levels have not been used in predictive manner. There are 2 likely reasons for this. First, as relayed above, chemerin exists in multiple isoforms and the biological contributions of each isoform are simply unclear. A significant majority of the enzyme-linked immunosorbent assays used measure a multitude of these isoforms. Second, it is unclear what information circulating chemerin can relay as far as its impact on cardiovascular health because it is, today, impossible to tell which tissues are the greatest contributor to circulating levels or whether high circulating chemerin levels equates to increased chemerin receptor activation.
To this idea, studies in the rat can begin to shed some light. Our laboratory created the global chemerin knockout rat to test the hypothesis that a lack of chemerin would reduce the ability of the knockout to become hypertensive.112 At baseline, blood pressures (mean, systolic, diastolic) were not different between the knockout and wild type of either the male or female rat. With the hypertensive challenge of deoxycorticosterone acetate and salt, the male knockout rats developed a higher blood pressure than the wild type, while the reverse was true in the female. This suggests 2 things. First, the actions of chemerin removal long term (from birth) have consequences that may be different than acute removal (discussed below). Second, there could be sex differences in the role of chemerin in blood pressure regulation. A few groups have demonstrated an upregulation of chemerin and its receptors in rat models of disease. Weng et al.113 used a high fat diet in male Wistar rats and demonstrated that chemerin and Chemerin1 protein expression was higher in the arteries and PVAT of the high fat vs. control group. Similarly, circulating chemerin was elevated in a rat model of preeclampsia.114 In another study, renal chemerin was elevated in a diabetic model,31 and Chemerin1 was upregulated in renal T cells in casein fed progeny of the Dahl S rat.115 This latter study used 2-(α-naphthoyl)ethyltrimethylammonium iodide as a Chemerin1 antagonist. Antagonism of Chemerin1 attenuated the hypertension of the Dahl S rat fed high salt, as well as renal damage.115 These studies are significant in that they support the activation of a chemerin receptor as mediating the disease.
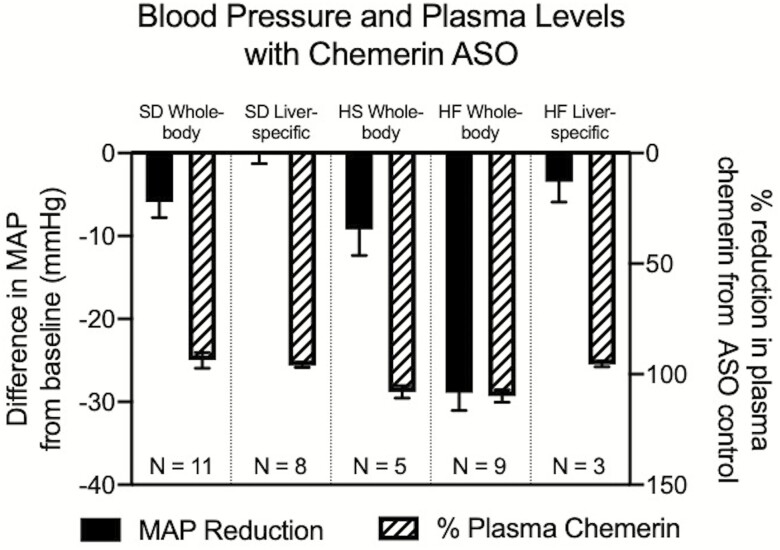
In the rat, as in the human, the liver and adipose tissue are likely the greatest overall contributors to the body load of chemerin (Figure 1). Our group has used the above described ASOs in the rat that cause the degradation of chemerin (pre)mRNA to reduce chemerin protein levels. The ASO against chemerin has been validated. In rats given this ASO, chemerin mRNA expression and chemerin protein in tissues, as well as circulating (plasma) levels of chemerin protein, were abolished compared with chemerin mRNA and protein expression in the tissues and plasma from rats given a control, scrambled ASO. In normal male Sprague-Dawley rats, chemerin ASO treatment reduced mean arterial blood pressure by ~7 mm Hg.116 These data, combined with others discussed below, have been redrawn from 116,117 in Figure 6. Similarly, the GalNAc ASO (liver-specific) against chemerin abolished liver chemerin mRNA, chemerin protein and reduced circulating chemerin levels to near zero, as determined by Western analysis.116 However, this same ASO was unable to reduce blood pressure. This suggests that while hepatic contributions dominate circulating plasma chemerin, liver chemerin is not involved in blood pressure determination.
The chemerin protein important to blood pressure regulation must be from other sources. We argue that adipose tissue is this source. In fact, the chemerin ASO could dramatically reduce the elevated blood pressure of the high fat but not high salt fed Dahl S rat,117 where the Dahl S is a recognized genetic model of hypertension. In the high fat Dahl S rat, the GalNAc ASO did not reduce the blood pressure of the high fat rat but almost abolished circulating chemerin levels (Figure 6).
Figure 6.
Effect of ASOs on mean arterial blood pressure (MAP) and plasma chemerin protein expression in the male Sprague-Dawley (left) and high salt (HS) or high fat (HF) Dahl S male rat. Whole-body ASO knocked down chemerin in all tissues, while the liver-specific ASO knocked down chemerin largely in the liver. Dahl S rats were either on a control, high fat (60% fat), or high salt (4% salt) diet. Data are adapted from refs. 110,111. Bars represent means ± SEM for number of animals indicated by N. Abbreviation: ASO, antisense oligonucleotide.
These findings argue that it is not circulating but rather local chemerin that is most biologically relevant to blood pressure. Figure 7 depicts a working hypothesis of our investigation of fat (primarily adipocytes) as a source of biologically relevant chemerin for blood pressure regulation. PVAT could be particularly important because it contains chemerin that could be secreted to affect both sympathetic nerve and vessel (endothelium, smooth muscle) function. It is also possible that PVAT fat produces particular isoforms of chemerin that are more effective than others in eliciting a biological response. If the human situation is similar, then measures of circulating plasma must be questioned as to their meaningfulness. Admittedly, we know of no way that would allow discrimination between PVAT and other adipose depots as sources of chemerin.
Figure 7.
Working hypothesis of the local actions of chemerin in the vasculature as facilitated by the perivascular adipose tissue (PVAT) as a source of chemerin that could activate chemerin1 receptors on the sympathetic nerve (yellow) or smooth muscle cell to stimulate vascular contraction.
However, these data do not invalidate or diminish the substantial human data correlating blood pressure with circulating levels of chemerin. While local chemerin from the PVAT is likely facilitating the molecular effects leading to the changes in blood pressure, chemerin in the serum is likely secondary to these changes and still a good diagnostic indicator of what is happening on the local level.
WHAT WE NEED TO LEARN ABOUT CHEMERIN AND HYPERTENSION
We share ideas that would move the chemerin field forward, significantly past its strong but largely epidemiological support.
We need more information as to the direct effects of chemerin in the heart, kidney, brain and adrenal. Specifically, we need to learn much more about the basic physiological functions of chemerin, what receptors chemerin utilizes to carry out these functions, and how this system changes in hypertensive disease. Additionally, we need to understand how much of chemerin receptor activation is from systemic (endocrine) vs. local (paracrine) prochemerin processing and metabolite distribution.
We need tools that can selectively activate and inhibit chemerin1, chemerin2, and CCRL2. Possession of receptor subtype tools would help in determining more specific functions of chemerin at the tissue and whole-body level.
We need to understand how important biased actions of chemerin receptors are to biological functions. We introduced the idea that chemerin’s action can be in opposition, an example being that chemerin is described as both inflammatory and inflammatory. Is this because different isoforms stimulate a different collective cadre of receptors (referred above)? Is this because the location of the receptor changes its actions? In this context, understanding whether the cellular machinery available to a chemerin receptor can change the biological outcome is important. Are there cell types, for example, in which chemerin and its isoforms act through receptors in a biased fashion, but others in which biased signaling is not possible?
We should be careful about interpretation of plasma concentration of chemerin in the human. This would require having assays that could discriminate between prochemerin and chemerin metabolites.
We need to determine how adipose tissue specifically contributes to functional chemerin. Adipocyte specific targeting of chemerin is currently a work in progress. There is much interest in being able to reduce adipocyte contributions, in general, but this field has been stymied by technical difficulties.
We need to understand the regulation of prochemerin processing into its myriad of active chemerin metabolites. Which proteases are essential vs. redundant? What is the tissue-dependency?
We need to identify whether there is a genetic association of Loss-of-Function or Gain-of-Function variants in chemerin signaling associated with disease. Work that has found an association of increased plasma chemerin and disease are intriguing but are not nearly as powerful of identifying whether genetic variants in chemerin or the chemerin receptors are associated with improved health or disease.
We need to investigate whether therapeutics that modulate chemerin levels and/or chemerin receptor signaling have utility in treatment of cardiometabolic and/or inflammatory diseases.
FUNDING
American Heart Association Transformative grant 19TPA34850014 (SWW) and National Institutes of Health grant F31 HL 143937 (DJF).
DISCLOSURE
The authors declared no conflict of interest.
REFERENCES
- 1. Wittamer V, Franssen JD, Vulcano M, Mirjolet JF, Le Poul E, Migeotte I, Brézillon S, Tyldesley R, Blanpain C, Detheux M, Mantovani A, Sozzani S, Vassart G, Parmentier M, Communi D. Specific recruitment of antigen-presenting cells by chemerin, a novel processed ligand from human inflammatory fluids. J Exp Med 2003; 198:977–985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Wittamer V, Bondue B, Guillabert A, Vassart G, Parmentier M, Communi D. Neutrophil-mediated maturation of chemerin: a link between innate and adaptive immunity. J Immunol 2005; 175:487–493. [DOI] [PubMed] [Google Scholar]
- 3. Bondue B, Wittamer V, Parmentier M. Chemerin and its receptors in leukocyte trafficking, inflammation and metabolism. Cytokine Growth Factor Rev 2011; 22:331–338 (doi: 10.1016/jcytogfr.2011.11.004). [DOI] [PubMed] [Google Scholar]
- 4. Hart R, Greaves DR. Chemerin contributes to inflammation by promoting macrophage adhesion to VCAM-1 and fibronectin through clustering of VLA-4 and VLA-5. J Immunol 2010; 185:3728–3739. [DOI] [PubMed] [Google Scholar]
- 5. Iannone F, Lapadula G. Chemerin/ChemR23 pathway: a system beyond chemokines. Arthritis Res Ther 2011; 13:104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Schipper HS, Prakken B, Kalkhoven E, Boes M. Adipose tissue-resident immune cells: key players in immunometabolism. Trends Endocrinol Metab 2012; 23:407–415. [DOI] [PubMed] [Google Scholar]
- 7. Zabel BA, Allen SJ, Kulig P, Allen JA, Cichy J, Handel TM, Butcher EC. Chemerin activation by serine proteases of the coagulation, fibrinolytic, and inflammatory cascades. J Biol Chem 2005; 280:34661–34666. [DOI] [PubMed] [Google Scholar]
- 8. Bozaoglu K, Bolton K, McMillan J, Zimmet P, Jowett J, Collier G, Walder K, Segal D. Chemerin is a novel adipokine associated with obesity and metabolic syndrome. Endocrinology 2007; 148:4687–4694. [DOI] [PubMed] [Google Scholar]
- 9. Goralski KB, McCarthy TC, Hanniman EA, Zabel BA, Butcher EC, Parlee SD, Muruganandan S, Sinal CJ. Chemerin, a novel adipokine that regulates adipogenesis and adipocyte metabolism. J Biol Chem 2007; 282:28175–28188. [DOI] [PubMed] [Google Scholar]
- 10. Muruganandam S, Dranse HJ, Rourke JL, McMullen NM, Sinal CJ. Chemerin neutralization blocks hematopoietic stem cell osteoclastogenesis. Stem Cells 2013; 31:2172–2182 (doi: 10.1102/ste.1450). [DOI] [PubMed] [Google Scholar]
- 11. Muruganandan S, Parlee SD, Rourke JL, Ernst MC, Goralski KB, Sinal CJ. Chemerin, a novel peroxisome proliferator-activated receptor gamma (PPARgamma) target gene that promotes mesenchymal stem cell adipogenesis. J Biol Chem 2011; 286:23982–23995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Muruganandan S, Roman AA, Sinal CJ. Role of chemerin/CMKLR1 signaling in adipogenesis and osteoblastogenesis of bone marrow stem cells. J Bone Miner Res 2010; 25:222–234. [DOI] [PubMed] [Google Scholar]
- 13. Roh SG, Song SH, Choi KC, Katoh K, Wittamer V, Parmentier M, Sasaki SI. Chemerin—a new adipokine that modulates adipogenesis via its own receptor. Biochem Biophys Res Commun 2007; 362:1013–1018. [DOI] [PubMed] [Google Scholar]
- 14. Hu W, Yu Q, Zhang J, Liu D. Rosiglitazone ameliorates diabetic nephropathy by reducing the expression of Chemerin and ChemR23 in the kidney of streptozotocin-induced diabetic rats. Inflammation 2012; 35:1287–1293. [DOI] [PubMed] [Google Scholar]
- 15. Helfer G, Ross AW, Thomson LM, Mayer CD, Stoney PN, McCaffery PJ, Morgan PJ. A neuroendocrine role for chemerin in hypothalamic remodelling and photoperiodic control of energy balance. Sci Rep 2016; 6:26830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Mattern A, Zellmann T, Beck-Sickinger AG. Processing, signaling, and physiological function of chemerin. IUBMB Life 2014; 66:19–26. [DOI] [PubMed] [Google Scholar]
- 17. Ge X, Yamaguchi Y, Zhao L, Bury L, Gresele P, Berube C, Leung LL, Morser J. Prochemerin cleavage by factor XIa links coagulation and inflammation. Blood 2018; 131:353–364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Zhao L, Yamaguchi Y, Sharif S, Du XY, Song JJ, Lee DM, Recht LD, Robinson WH, Morser J, Leung LL. Chemerin158K protein is the dominant chemerin isoform in synovial and cerebrospinal fluids but not in plasma. J Biol Chem 2011; 286:39520–39527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Zhao L, Yamaguchi Y, Ge X, Robinson WH, Morser J, Leung LLK. Chemerin 156G, generated by chymase cleavage of prochemerin, is elevated in joint fluids of arthritis patients. Arthritis Res Ther 2018; 20:132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Zhao L, Yamaguchi Y, Shen WJ, Morser J, Leung LLK. Dynamic and tissue-specific proteolytic processing of chemerin in obese mice. PLoS One 2018; 13:e0202780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Du XY, Leung LL. Proteolytic regulatory mechanism of chemerin bioactivity. Acta Biochim Biophys Sin (Shanghai) 2009; 41:973–979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Du XY, Zabel BA, Myles T, Allen SJ, Handel TM, Lee PP, Butcher EC, Leung LL. Regulation of chemerin bioactivity by plasma carboxypeptidase N, carboxypeptidase B (activated thrombin-activable fibrinolysis inhibitor), and platelets. J Biol Chem 2009; 284:751–758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Guillabert A, Wittamer V, Bondue B, Godot V, Imbault V, Parmentier M, Communi D. Role of neutrophil proteinase 3 and mast cell chymase in chemerin proteolytic regulation. J Leukoc Biol 2008; 84:1530–1538. [DOI] [PubMed] [Google Scholar]
- 24. John H, Hierer J, Haas O, Forssmann WG. Quantification of angiotensin-converting-enzyme-mediated degradation of human chemerin 145–154 in plasma by matrix-assisted laser desorption/ionization-time-of-flight mass spectrometry. Anal Biochem 2007; 362:117–125. [DOI] [PubMed] [Google Scholar]
- 25. Parlee SD, McNeil JO, Muruganandan S, Sinal CJ, Goralski KB. Elastase and tryptase govern TNFα-mediated production of active chemerin by adipocytes. PLoS One 2012; 7:e51072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chang SS, Eisenberg D, Zhao L, Adams C, Leib R, Morser J, Leung L. Chemerin activation in human obesity. Obesity (Silver Spring) 2016; 24:1522–1529. [DOI] [PubMed] [Google Scholar]
- 27. Laranjeira S, Regan-Komito D, Iqbal AJ, Greaves DR, Payne SJ, Orlowski P. A model for the optimization of anti-inflammatory treatment with chemerin. Interface Focus 2018; 8:20170007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Pohl R, Rein-Fischboeck L, Meier EM, Eisinger K, Krautbauer S, Buechler C. Resolvin E1 and chemerin C15 peptide do not improve rodent non-alcoholic steatohepatitis. Exp Mol Pathol 2015; 98:295–299. [DOI] [PubMed] [Google Scholar]
- 29. Buechler C, Feder S, Habertl EM, Aslanidis. Chemerin isoforms and activity in obesity. Int J Mol Sci 2019; 20:1128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Huang H, Hu L, Lin J, Zhu X, Cui W, Xu W. Effect of fosinopril on chemerin and VEGF expression in diabetic nephropathy rats. Int J Clin Exp Pathol 2015; 8:11470–11474. [PMC free article] [PubMed] [Google Scholar]
- 31. Yu QX, Zhang H, Xu WH, Hao F, Liu SL, Bai MM, Mu JW, Zhang HJ. Effect of Irbesartan on chemerin in the renal tissues of diabetic rats. Kidney Blood Press Res 2015; 40:467–477. [DOI] [PubMed] [Google Scholar]
- 32. Kennedy AJ, Davenport AP. International union of basic and clinical pharmacology CIII: chemerin receptors CMKLR1 (Chemerin1) and GPR1 (Chemerin2) nomenclature, pharmacology, and function. Pharmacol Rev 2018; 70:174–196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Monnier J, Lewén S, O’Hara E, Huang K, Tu H, Butcher EC, Zabel BA. Expression, regulation, and function of atypical chemerin receptor CCRL2 on endothelial cells. J Immunol 2012; 189:956–967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Wittamer V, Grégoire F, Robberecht P, Vassart G, Communi D, Parmentier M. The C-terminal nonapeptide of mature chemerin activates the chemerin receptor with low nanomolar potency. J Biol Chem 2004; 279:9956–9962. [DOI] [PubMed] [Google Scholar]
- 35. Graham KL, Zhang JV, Lewen S, Burke TM, Dang T, Zoudlova M, Sobel RA, Butcher EG, Zabel BA. A novel CMKLR1 small molecule antagonist suppresses CNS autoimmune inflammatory disease. PLoS One 2014; 9:e112925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Athyros VG, Kakafika AI, Tziomalos K, Karagiannis A, Mikhailidis DP. Antisense technology for the prevention or the treatment of cardiovascular disease: the next blockbuster? Expert Opin Investig Drugs 2008; 17:969–972. [DOI] [PubMed] [Google Scholar]
- 37. Huang Y. Preclinical and clinical advances of GalNAc-decorated nucleic acid therapeutics. Mol Ther Nucleic Acids 2017; 6:116–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Watts SW, Dorrance AM, Penfold ME, Rourke JL, Sinal CJ, Seitz B, Sullivan TJ, Charvat TT, Thompson JM, Burnett R, Fink GD. Chemerin connects fat to arterial contraction. Arterioscler Thromb Vasc Biol 2013; 33:1320–1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Darios ES, Winner BM, Charvat T, Krasinksi A, Punna S, Watts SW. The adipokine chemerin amplifies electrical field-stimulated contraction in the isolated rat superior mesenteric artery. Am J Physiol Heart Circ Physiol 2016; 311:H498–H507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Ferland DJ, Darios ES, Neubig RR, Sjögren B, Truong N, Torres R, Dexheimer TS, Thompson JM, Watts SW. Chemerin-induced arterial contraction is Gi- and calcium-dependent. Vascul Pharmacol 2017; 88:30–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Hanthazi A, Jespers P, vegh G, Degroot G-N, Springael J-Y, Lybaert P, Dewatchter L, McEntee K. Chemerin influences endothelin and serotonin-induced pulmonary artery vasoconstriction in rats. Life Sci 2019; 231:116580 (doi: 10.1016/j.lfs.2019.116580). [DOI] [PubMed] [Google Scholar]
- 42. Kennedy AJ, Yang P, Read C, Kuc RE, Yang L, Taylor EJ, Taylor CW, Maguire JJ, Davenport AP. Chemerin elicits potent constrictor actions via chemokine-like receptor 1 (CMKLR1), not G-protein coupled receptor 1 (GPR1), in human and rat vasculature. J Am Heart Assoc 2016; 5:e004421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kostopoulos CG, Spioglou SG, Varakis JN, Apostolakis E, Papadaki HHC. Chemerin and CMKLR1 expression in human arteries and periadventitial fat: a possible role for local chemerin in atherosclerosis? BMC Cardiovasc Disord 2014; 14:56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Lobato NS, Neves KB, Filgueira FP, Fortes ZB, Carvalho MH, Webb RC, Oliveira AM, Tostes RC. The adipokine chemerin augments vascular reactivity to contractile stimuli via activation of the MEK-ERK1/2 pathway. Life Sci 2012; 91:600–606. [DOI] [PubMed] [Google Scholar]
- 45. Neves KB, Nguyen Dinh Cat A, Lopes RA, Rios FJ, Anagnostopoulou A, Lobato NS, de Oliveira AM, Tostes RC, Montezano AC, Touyz RM. Chemerin regulates crosstalk between adipocytes and vascular cells through Nox. Hypertension 2015; 66:657–666. [DOI] [PubMed] [Google Scholar]
- 46. Neves KB, Lobato NS, Lopes RA, Filgueira FP, Zanotto CZ, Oliveira AM, Tostes RC. Chemerin reduces vascular nitric oxide/cGMP signalling in rat aorta: a link to vascular dysfunction in obesity? Clin Sci (Lond) 2014; 127:111–122. [DOI] [PubMed] [Google Scholar]
- 47. De Henau O, Degroot G-N, Imbault V, Robert V, De Poorter C, Mcheik S, Gales C, Parmentier M, Springael J-Y. Signaling properties of chemerin receptors CMKLR1, GPR1 and CCRL2. PLoS One 2016; 11:e0164179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Kunimoto H, Kazama K, Takai M, Oda M, Okada M, Yamawaki H. Chemerin promotes the proliferation and migration of vascular smooth muscle and increases mouse blood pressure. Am J Physiol Heart Circ Physiol 2015; 309:H1017–H1028. [DOI] [PubMed] [Google Scholar]
- 49. Wen J, Wang J, Guo L, Cai W, Wu Y, Chen W, Tang X. Chemerin stimulates aortic smooth muscle cell proliferation and migration via activation of autophagy in VSMCs of metabolic hypertension rats. Am J Transl Res 2019; 11:1327–1342. [PMC free article] [PubMed] [Google Scholar]
- 50. Gu P, Wang W, Yao Y, Xu Y, Wang L, Zang P, Ma J, Yang C, Liang J, Lu B, Shao J. Increased circulating chemerin in relation to chronic microvascular complications in patients with Type 2 diabetes. Int J Endocrinol 2019; 2019:8693516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Bozaoglu K, Curran JE, Stocker CJ, Zaibi MS, Segal D, Konstantopoulos N, Morrison S, Carless M, Dyer TD, Cole SA, Goring HH, Moses EK, Walder K, Cawthorne MA, Blangero J, Jowett JB. Chemerin, a novel adipokine in the regulation of angiogenesis. J Clin Endocrinol Metab 2010; 95:2476–2485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Wang N, Wang QJ, Feng YY, Shang W, Cai M. Overexpression of chemerin was associated with tumor angiogenesis and poor clinical outcome in squamous cell carcinoma of the oral tongue. Clin Oral Investig 2014; 18:997–1004. [DOI] [PubMed] [Google Scholar]
- 53. Thomas GD. Neural control of the circulation. Adv Physiol Educ 2011; 35:28–32. [DOI] [PubMed] [Google Scholar]
- 54. Moore A, Zabel BA, Burnett R, Watts SW. Chemerin C9 peptide releases catecholamines from rat adrenal medulla. Pharmacologia 2016; 7:290–295. [Google Scholar]
- 55. Uhlen M, Fagerberg L, Hallstrom BM, Lindskog C, Oksvold P, Mardinoglu A, Sivertsson A, Kampf C, Sjostedt E, Asplund A, Olsson I, Edlund K, Lundberg E, Navani S, Szigyarto CA, Odeberg J, Djureinovic D, Takanen JO, Hober S, Alm T, Edqvist PH, Berling H, Tegel H, Mulder J, Rockberg J, Nilsson P, Schwenk JM, Hamsten M, von Feilitzen K, Forsberg M, Persson L, Johansson F, Zwahlen M, von Heijne G, Nielsen J, Ponten F. Tissue-based map of the human proteome. Science 2015; 347:1260419 (doi: 10.1126/science.1260419). [DOI] [PubMed] [Google Scholar]
- 56. Lee TH-Y, Chen KK-Y, Hoo RL-C, Siu PM-F, Yau S-Y. The novel perspectives of adipokines on brain health. Int J Mol Sci 2019; 20:5638 (doi: 10.3390/ijms20225638). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Song SH, Fukui K, Nakajima K, Kozakai T, Sasaki S, Roh SG, Katoh K. Cloning, expression analysis, and regulatory mechanisms of bovine chemerin and chemerin receptor. Domest Anim Endocrinol 2010; 39:97–105. [DOI] [PubMed] [Google Scholar]
- 58. Smolinska N, Kiezun M, Dobrzyn K, Rytelewska E, Kisielewska K, Gudelska M, Zaobidna E, Bogus-Nowakowska K, Wyrebek J, Bors K, Kopij G, Kaminska B, Kaminski T. Expression of chemerin and its receptors in the porcine hypothalamus and plasma chemerin levels during the oestrous cycle and early pregnancy. Int J Mol Sci 2019; 20:3887 (doi: 10.3390/ijms20163887). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Brunetti L, Orlando G, Ferrante C, Recinella L, Leone S, Chiavaroli A, Di Nisio C, Shohreh R, Manippa F, Ricciuti A, Vacca M. Peripheral chemerin administration modulates hypothalamic control of feeding. Peptides 2014; 51:115–121. [DOI] [PubMed] [Google Scholar]
- 60. Kutlay O, Kaygisiz, Kaygisiz B. The effect of chemerin on cardiac parameters and gene expressions in isolated perfused rat heart. Balkan Med J 2019; 36:43–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Zhang R, Liu S, Guo B, Chang L, Li Y. Chemerin induces insulin resistance in rat cardiomyocytes in part through the ERK1/2 signaling pathway. Pharmacology 2014; 94:259–264. [DOI] [PubMed] [Google Scholar]
- 62. Rodríguez-Penas D, Feijóo-Bandín S, García-Rúa V, Mosquera-Leal A, Durán D, Varela A, Portolés M, Roselló-Lletí E, Rivera M, Diéguez C, Gualillo O, González-Juanatey JR, Lago F. The adipokine chemerin induces apoptosis in cardiomyocytes. Cell Physiol Biochem 2015; 37:176–192. [DOI] [PubMed] [Google Scholar]
- 63. Regn M, Laggerbauer B, Jentzsch C, Ramanujam D, Ahles A, Sichler S, Calzada-Wack J, Koenen RR, Braun A, Nieswandt B, Engelhardt S. Peptidase inhibitor 16 is a membrane-tethered regulator of chemerin processing in the myocardium. J Mol Cell Cardiol 2016; 99:57–64. [DOI] [PubMed] [Google Scholar]
- 64. Menzel J, di Giuseppe R, Biemann R, Wittenbecher C, Aleksandrova K, Eichelmann F, Fritsche A, Schulze MB, Boeing H, Isermann B, Weikert C. Association between chemerin, omentin-1 and risk of heart failure in the population-based EPIC-Potsdam study. Sci Rep 2017; 7:14171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Zhou X, Tao Y, Chen Y, Xu W, Qian Z, Lu X. Serum chemerin as a novel prognostic indicator in chronic heart failure. J Am Heart Assoc 2019; 8:e012091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Zhang O, Ji Q, Lin Y, Wang Z, Huang Y, Lu W, Liu X, Zhang J, Liu Y, Zhou YJ. Circulating chemerin levels elevated in dilated cardiomyopathy patients with overt heart failure. Clin Chim Acta 2015; 448:27–32. [DOI] [PubMed] [Google Scholar]
- 67. Blaszak J, Szolkiewicz M, Sucajtys-Szulc E, Konarzewski M, Lizakowski S, Swierczynski J, Rutkowski B. High serum chemerin level in CKD patients is related to kidney function, but not to its adipose tissue overproduction. Ren Fail 2015; 37:1033–1038. [DOI] [PubMed] [Google Scholar]
- 68. Bonomini M, Pandolfi A. Chemerin in renal dysfunction and cardiovascular disease. Vascul Pharmacol 2016; 77:28–34. [DOI] [PubMed] [Google Scholar]
- 69. Dellepiane S, Medica D, Guarena C, Musso T, Quercia AD, Leonardi G, Marengo M, Migliori M, Panichi V, Biancone L, Pizzarelli F, Camussi G, Cantaluppi V. Citrate anion improves chronic dialysis efficacy, reduces systemic inflammation and prevents Chemerin-mediated microvascular injury. Sci Rep 2019; 9:10622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. El-Khashab SO, Gamil M, Ali AY, El-Khashab O, El-Khatib M, Mohamed K, El-Shehaby A, Hamed ST. Chemerin level and the relation to insulin resistance in chronic kidney disease. Saudi J Kidney Dis Transpl 2019; 30:1381–1388. [DOI] [PubMed] [Google Scholar]
- 71. Pfau D, Bachmann A, Lössner U, Kratzsch J, Blüher M, Stumvoll M, Fasshauer M. Serum levels of the adipokine chemerin in relation to renal function. Diabetes Care 2010; 33:171–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Rutkowski P, Sledzinski T, Zielinska H, Lizakowski S, Goyke E, Szrok-Wojtkiewicz S, Swierczynski J, Rutkowski B. Decrease of serum chemerin concentration in patients with end stage renal disease after successful kidney transplantation. Regul Pept 2012; 173:55–59. [DOI] [PubMed] [Google Scholar]
- 73. Salama FE, Anass QA, Abdelrahman AA, Saeed EB. Chemerin: a biomarker for cardiovascular disease in diabetic chronic kidney disease patients. Saudi J Kidney Dis Transpl 2016; 27:977–984. [DOI] [PubMed] [Google Scholar]
- 74. Zylla S, Rettig R, Völzke H, Endlich K, Nauck M, Friedrich N. Serum chemerin levels are inversely associated with renal function in a general population. Clin Endocrinol (Oxf) 2018; 88:146–153. [DOI] [PubMed] [Google Scholar]
- 75. Huang J, Zhang J, Lei T, Chen X, Zhang Y, Zhou L, Yu A, Chen Z, Zhou R, Yang Z. Cloning of porcine chemerin, ChemR23 and GPR1 and their involvement in regulation of lipogenesis. BMB Rep 2010; 43:491–498. [DOI] [PubMed] [Google Scholar]
- 76. Lin S, Teng J, Li J, Sun F, Yuan D, Chang J. Association of chemerin and vascular endothelial growth factor (VEGF) with diabetic nephropathy. Med Sci Monit 2016; 22:3209–3214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Mocker A, Hilgers KF, Cordasic N, Wachtveitl R, Menendez-Castro C, Woelfle J, Hartner A, Fahlbusch FB. Renal chemerin expression is induced in models of hypertensive nephropathy and glomerulonephritis and correlates with markers of inflammation and fibrosis. Int J Mol Sci 2019; 20:6240 (doi: 10.3390/ijms20246240). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Mariani F, Roncucci L. Chemerin/chemR23 axis in inflammation onset and resolution. Inflamm Res 2015; 64:85–95. [DOI] [PubMed] [Google Scholar]
- 79. Della Chiesa M, Sivori S, Castriconi R, Marcenaro E, Moretta A. Pathogen-induced private conversations between natural killer and dendritic cells. Trends Microbiol 2005; 13:128–136. [DOI] [PubMed] [Google Scholar]
- 80. Zabel BA, Silverio AM, Butcher EC. Chemokine-like receptor 1 expression and chemerin-directed chemotaxis distinguish plasmacytoid from myeloid dendritic cells in human blood. J Immunol 2005; 174:244–251. [DOI] [PubMed] [Google Scholar]
- 81. Rodriguez-Iturbe B, Pons H, Johnson RJ. Role of the immune system in hypertension. Physiol Rev 2017; 97:1127–1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Zhang G, Xiao M, Zhang L, Zhao Y, Yang Q. Association of serum chemerin concentrations with the presence of atrial fibrillation. Ann Clin Biochem 2017; 54:342–347. [DOI] [PubMed] [Google Scholar]
- 83. Lu B, Zhao M, Jiang W, Ma J, Yang C, Shao J, Gu P. Independent association of circulating level of chemerin with functional and early morphological vascular changes in newly diagnosed Type 2 diabetic patients. Medicine (Baltimore) 2015; 94:e1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Gu P, Jiang W, Lu B, Shi Z. Chemerin is associated with inflammatory markers and metabolic syndrome phenotypes in hypertension patients. Clin Exp Hypertens 2014; 36:326–332. [DOI] [PubMed] [Google Scholar]
- 85. Yang M, Yang G, Dong J, Liu Y, Zong H, Liu H, Boden G, Li L. Elevated plasma levels of chemerin in newly diagnosed type 2 diabetes mellitus with hypertension. J Investig Med 2010; 58:883–886. [DOI] [PubMed] [Google Scholar]
- 86. Salord N, Gasa M, Mayos M, Fortuna-Gutierrez AM, Montserrat JM, Sánchez-de-la-Torre M, Barceló A, Barbé F, Vilarrasa N, Monasterio C. Impact of OSA on biological markers in morbid obesity and metabolic syndrome. J Clin Sleep Med 2014; 10:263–270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Meric M, Soylu K, Avci B, Yuksel S, Gulel O, Yenercag M, Coksevim M, Uzun A. Evaluation of plasma chemerin levels in patients with non-dipper blood pressure patterns. Med Sci Monit 2014; 20:698–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Wang D, Yuan G-Y, Wang X-Z, Jia J, Di L-L, Yang L, Chen X, Qian F-F, Chen J-J. Plasma chemerin levels in metabolic syndrome. Gen Mol Res 2013; 12:5986–5991. [DOI] [PubMed] [Google Scholar]
- 89. Sledzińska M, Szlagatys-Sidorkiewicz A, Brzezinski M, Kaźmierska K, Sledziński T, Kamińska B. Serum chemerin in children with excess body weight may be associated with ongoing metabolic complications—a pilot study. Adv Med Sci 2017; 62:383–386. [DOI] [PubMed] [Google Scholar]
- 90. Dong B, Ji W, Zhang Y. Elevated serum chemerin levels are associated with the presence of coronary artery disease in patients with metabolic syndrome. Intern Med 2011; 50:1093–1097. [DOI] [PubMed] [Google Scholar]
- 91. Feng X, Li P, Zhou C, Jia X, Kang J. Elevated levels of serum chemerin in patients with obstructive sleep apnea syndrome. Biomarkers 2012; 17:248–253. [DOI] [PubMed] [Google Scholar]
- 92. Yoo HJ, Choi HY, Yang SJ, Kim HY, Seo JA, Kim SG, Kim NH, Choi KM, Choi DS, Baik SH. Circulating chemerin level is independently correlated with arterial stiffness. J Atheroscler Thromb 2012; 19:59–66; discussion 67. [DOI] [PubMed] [Google Scholar]
- 93. Hu W, Feng P. Elevated serum chemerin concentrations are associated with renal dysfunction in type 2 diabetic patients. Diabetes Res Clin Pract 2011; 91:159–163. [DOI] [PubMed] [Google Scholar]
- 94. Coban M, Tasli L, Turgut S, Özkan S, Tunç Ata M, Akın F. Association of adipokines, insulin resistance, hypertension and dyslipidemia in patients with psoriasis vulgaris. Ann Dermatol 2016; 28:74–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Du J, Li R, Xu L, Ma R, Liu J, Cheng J, Zhang Z, Sun H. Increased serum chemerin levels in diabetic retinopathy of Type 2 diabetic patients. Curr Eye Res 2016; 41:114–120. [DOI] [PubMed] [Google Scholar]
- 96. Yin C, Chu H, Li H, Xiao Y. Plasma Sfrp5 and adiponectin levels in relation to blood pressure among obese children. J Hum Hypertens 2017; 31:284–291. [DOI] [PubMed] [Google Scholar]
- 97. Stejskal D, Karpisek M, Hanulova Z, Svestak M. Chemerin is an independent marker of the metabolic syndrome in a Caucasian population—a pilot study. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2008; 152:217–221. [DOI] [PubMed] [Google Scholar]
- 98. Cetin O, Kurdoglu Z, Kurdoglu M, Sahin HG. Chemerin level in pregnancies complicated by preeclampsia and its relation with disease severity and neonatal outcomes. J Obstet Gynaecol 2017; 37:195–199. [DOI] [PubMed] [Google Scholar]
- 99. Duan D-M, Niu J-M, Lei Z, Lin X-H, Chen X. Serum levels of the adipokine chemerin in preeclampsia. J Perinat Med 2012; 40:121–127. [DOI] [PubMed] [Google Scholar]
- 100. Stepan H, Philipp A, Roth I, Kralisch S, Jank A, Schaarschmidt W, Lossner U, Kratzsch J, Bluher M, Stumvol M, Fasshauer. Serum levels of the adipokine chemerin are increased in preeclampsia during the 6 months after pregnancy. Reg Pept 2011; 168:69–72. [DOI] [PubMed] [Google Scholar]
- 101. Wang L, Zhong Y, Ding Y, Shi X, Huang J, Zhu F. Elevated serum chemerin in Chinese women with hyperandrogenic PCOS. Gynecol Endocrinol 2014; 30:746–750. [DOI] [PubMed] [Google Scholar]
- 102. Jialal I, Devaraj S, Kaur H, Adams-Huet B, Bremer AA. Increased chemerin and decreased omentin-1 in both adipose tissue and plasma in nascent metabolic syndrome. J Clin Endocrinol Metab 2013; 98:E514–E517. [DOI] [PubMed] [Google Scholar]
- 103. Habib SS, Eshki A, AlTassan B, Fatani D, Helmi H, AlSaif S. Relationship of serum novel adipokine chemerin levels with body composition, insulin resistance, dyslipidemia and diabesity in Saudi women. Eur Rev Med Pharmacol Sci 2017; 21:1296–1302. [PubMed] [Google Scholar]
- 104. Derosa G, Catena G, Gaudio G, D’Angelo A, Maffioli P. Adipose tissue dysfunction and metabolic disorders: is it possible to predict who will develop type 2 diabetes mellitus? Role of markErs in the progreSsion of dIabeteS in obese paTIeNts (The RESISTIN trial). Cytokine 2020; 127:154947. [DOI] [PubMed] [Google Scholar]
- 105. Pahwa R, Adams-Huet B, Jialal I. The effect of increasing body mass index on cardio-metabolic risk and biomarkers of oxidative stress and inflammation in nascent metabolic syndrome. J Diabetes Complications 2017; 31:810–813. [DOI] [PubMed] [Google Scholar]
- 106. Han J, Kim SH, Suh YJ, Lim HA, Shin H, Cho SG, Kim CW, Lee SY, Lee DH, Hong S, Kim YS, Nam MS. Serum chemerin levels are associated with abdominal visceral fat in Type 2 diabetes. J Korean Med Sci 2016; 31:924–931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Shin HY, Lee DC, Chu SH, Jeon JY, Lee MK, Im JA, Lee JW. Chemerin levels are positively correlated with abdominal visceral fat accumulation. Clin Endocrinol (Oxf) 2012; 77:47–50. [DOI] [PubMed] [Google Scholar]
- 108. Bays H. Central obesity as a clinical marker of adiposopathy; increased visceral adiposity as a surrogate marker for global fat dysfunction. Curr Opin Endocrinol Diabetes Obes 2014; 21:345–351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Booth A, Magnuson A, Foster M. Detrimental and protective fat: body fat distribution and its relation to metabolic disease. Horm Mol Biol Clin Investig 2014; 17:13–27. [DOI] [PubMed] [Google Scholar]
- 110. Emery EM, Schmid TL, Kahn HS, Filozof PP. A review of the association between abdominal fat distribution, health outcome measures, and modifiable risk factors. Am J Health Promot 1993; 7:342–353. [DOI] [PubMed] [Google Scholar]
- 111. Malden D, Lacey B, Emberson J, Karpe F, Allen N, Bennett D, Lewington S. Body fat distribution and systolic blood pressure in 10,000 adults with whole-body imaging: UK Biobank and Oxford BioBank. Obesity (Silver Spring) 2019; 27:1200–1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Watts SW, Darios ES, Mullick AE, Garver H, Saunders TL, Hughes ED, Filipiak WE, Zeidler MG, McMullen N, Sinal CJ, Kumar RK, Ferland DJ, Fink GD. The chemerin knockout rat reveals chemerin dependence in female but not male experimental hypertension. FASEB J 2018; 32:fj201800479 (doi: 10.1096/fj.201800479). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Weng C, Shen Z, Li X, Jiang W, Peng L, Yuan H, Yang K, Wang J. Effects of chemerin/CMKLR1 in obesity-induced hypertension and potential mechanism. Am J Transl Res 2017; 9:3096–3104. [PMC free article] [PubMed] [Google Scholar]
- 114. Nassar SZ, Badae NM. Protective effect of vitamin D supplementation in a rat model of preeclampsia: a possible implication of chemerin. Hypertens Pregnancy 2019; 38:149–156. [DOI] [PubMed] [Google Scholar]
- 115. Abais-Battad JM, Lund H, Fehrenbach DJ, Dasinger JH, Alsheikh AJ, Mattson DL. Parental dietary protein source and the role of CMKLR1 in determining the severity of Dahl salt-sensitive hypertension. Hypertension 2019; 73:440–448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Ferland DJ, Seitz B, Darios ES, Thompson JM, Yeh ST, Mullick AE, Watts SW. Whole-body but not hepatic knockdown of chemerin by antisense oligonucleotide decreases blood pressure in rats. J Pharmacol Exp Ther 2018; 365:212–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Ferland DJ, Flood ED, Garver H, Yeh ST, Riney S, Mullick AE, Fink GD, Watts SW. Different blood pressure responses in hypertensive rats following chemerin mRNA inhibition in dietary high fat compared to dietary high-salt conditions. Physiol Genomics 2019; 51:553–561. [DOI] [PMC free article] [PubMed] [Google Scholar]