Abstract
Background
Coronavirus disease 2019 (COVID-19) is affecting the whole world and threatening human health. We aim to investigate the immunological characteristics of monocytes in critical patients with COVID-19.
Methods
The number and immune status of monocytes were detected by flow cytometry in 32 COVID-19 patients and 18 healthy individuals.
Results
In critical patients with COVID-19, the absolute number of total monocytes and CD16− monocytes was significantly decreased but CD16+ pro-inflammatory monocytes was increased compared to healthy controls. Antigen presentation potential of monocytes, as measured by HLA-DR expression, was suppressed, while their inflammatory phenotype (CD38 expression) was enhanced. Cytokine levels showed sustained increases in critical patients. And the levels of IL-6 were positively correlated with CD16+ monocytes number. IL-6 and IL-10 levels were negatively correlated with HLA-DR expression of monocytes. During the recovery of COVID-19 patients, the count and immune status of monocyte subsets were restored by degrees. HLA-DR+ monocytes possessed good sensitivity and specificity for predicting the incidence of critical patients with COVID-19.
Conclusions
In critical patients with COVID-19, decline in number and HLA-DR expression of monocytes might lead to decreased antigen presentation potential and thus immunosuppression, while increased CD16+ pro-inflammatory monocytes might mediate hyperinflammation. HLA-DR+ monocytes might be a meaningful assisted indicator to predict the incidence of critical patients with COVID-19.
Keywords: COVID-19, Monocytes, Antigen presentation, Hyperinflammation
1. Introduction
Coronavirus disease 2019 (COVID-19) is highly contagious, which is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The WHO declared its outbreak to be a global public health emergency [1]. Until October 15, 2020, 38,202,956 confirmed cases and 1,087,069 deaths were reported globally [2], far exceeding the total number of SARS cases in 2003. Among these COVID-19 patients, most of them had some common symptoms including fever, cough, myalgia or fatigue at the time of onset. Most patients with COVID-19 were mild and could recover after timely medical intervention. However, about 25% of patients would deteriorate into serious complications, including acute respiratory distress syndrome (ARDS), and even multiple organ failure and death [3], [4]. Like many other infectious diseases, COVID-19 patients can be damaged by the pathogen or excessive immune response incited by the pathogen. However, the exact immune mechanisms causing damage remain unclear. Therefore, exploring the immune mechanism of COVID-19 patients is the focus of current research.
It is increasingly recognized that the innate immune system plays an important role in determining the clinical course of infection [5]. Monocytes are an essential part of the innate immune system. They can directly engulf pathogens or present antigens to T cells to produce cytokines, regulating both humoral and cellular immune responses [6], [7], [8]. Some studies had suggested a significant decrease of monocytes in COVID-19 patients with severe or critical disease [9], [10]. However, the dynamic changes of monocytes in critical patients with COVID-19 is still largely unclear. In this study, we focused on the immunological characteristics of monocytes in critical patients with COVID-19, monitored its dynamic changes in the number and immune status.
2. Materials and methods
2.1. Patients
Thirty-two COVID-19 patients in The First Affiliated Hospital of Wenzhou Medical University were recruited for this study. In the Guideline of the Diagnosis and Treatment of New Coronavirus Pneumonia (trial version 7) issued by the National Health Commission of China [11], the clinical classifications of COVID-19 are as follows: (1) Mild cases: The clinical symptoms were mild, and the imaging examination showed no signs of pneumonia. (2) Moderate cases: Showing fever and respiratory symptoms, accompanied by imaging manifestations of pneumonia. (3) Severe cases: Adult cases meet any of the following criteria: shortness of breath with respiratory rate ≥30 times/min, oxygen saturation ≤93% at rest, partial pressure of arterial oxygen (PaO2)/oxygen concentration (FiO2) ≤ 300 mmHg (1 mmHg = 0.133 kPa). (4) Critical cases: Cases meeting any of the following criteria: respiratory failure requires mechanical ventilation, shock or with other organ failures require ICU care.zai. According to the above classification criteria, all confirmed patients included 13 mild cases (merging the mild and moderate types defined in the guideline, male/female: 6/7; age: 59.69 ± 13.38), 10 severe cases (male/female: 7/3; age: 62.80 ± 13.03) and 9 critical cases (male/female: 6/3; age: 67.78 ± 17.22). There were no statistically significant differences in age and sex between groups. Within three days at admission, blood was collected before treatment for the different groups. And after admission for treatment, blood continued to be collected at intervals for the different groups to observe the trend of the number and function of cells and the therapeutic effect. All patients received standardized treatment in accordance with the above guidelines, including antiviral drugs such as α-interferon (α-IFN), ribavirin, lopinavir/ritonavir, etc., oxygen therapy, fluid management, antibiotics treatment if occasion required, symptomatic relief and supportive treatment. Eighteen healthy individuals with age and gender matching who were not infected with SARS-CoV-2 were used as healthy controls (HCs). The study protocol was approved by the ethics committee of The First Affiliated Hospital of Wenzhou Medical University. Written informed consent was obtained from all subjects.
2.2. Laboratory examination
Specimens from patients included nasopharyngeal swab and blood samples. The presence of SARS-CoV-2 in nasopharyngeal swab was detected by real-time RT-PCR (rRT-PCR) assay. Serum cytokines (IL-2, IL-4, IL-6, IL-10, TNF-α and IFN-γ) were detected with human Th1/2 cytokine kit II and lymphocyte subsets were detected with BD Multitest IMK kit by BD FACSCanto II flow cytometer (BD Biosciences, California, USA). Monocytes subgroups (CD16− and CD16+ monocytes) were detected by HematoFlow with CytoDiff. All tests were performed in accordance with the product manual.
2.3. Fluorescence-activated flow cytometry analysis
To detect the phenotypic and functional characteristics of monocytes, 100 µl of anti-coagulated fresh whole blood of confirmed COVID-19 patients was collected for fluorescence antibody staining. Cells were incubated with fluorescently labeled antibodies of 10 µl/test concentration for 15 min at room temperature. All samples were detected by the BD FACS Canto II flow cytometer and analyzed with Flowjo software (TreeStar, BD, USA). The gate strategy of Monocytes was executed as CD45+ CD36+ CD16+/−. To specifically analyse monocytes and the expression of HLA-DR and CD38 on monocytes, Cells were stained by CytoDiff reagent (CD45-PC7-J33/CD36-FITC-FA6.152/CD16-PC5-3G8/CD2-PE-39C1.5/CD294 (CRTH2)-PE-BM16/CD19-ECD-J3-119) and the following antibodies: allophycocyanin (APC)-conjugated anti-human HLA-DR mAb (L234, BD Biosciences, California, USA), APC-conjugated anti-human CD38 mAb (HB-7, BD Biosciences, California, USA).
2.4. Statistical analysis
GraphPad Prism software (v.7.0a, Inc. San Diego, CA, USA) was used for statistical analysis. Data are expressed as the mean or median for continuous variables with or without normal distribution, respectively. Mann Whitney U test or unpaired test was used to compare different individuals. Multiple comparisons were made among the different groups using the Kruskal-Wallis H nonparametric test followed by Dunn's test as the post-test. Spearman correlation test was used to assess the correlations between variables. The diagnostic value of each indicator was appraised using the receiver operation characteristic (ROC) curves. The 95% confidence interval was utilized to calculate the sensitivity, specificity and consistency, and the cut-off value was selected when the Jordan index was at its maximum. For all the tests, a two-sided P value <0.05 was considered to be significant. The statistical significance is indicated as follows: ****p < 0.0001, ***p < 0.001, **p < 0.01 and *p < 0.05.
3. Results
3.1. Absolute number of total monocytes and subsets in COVID-19 patients
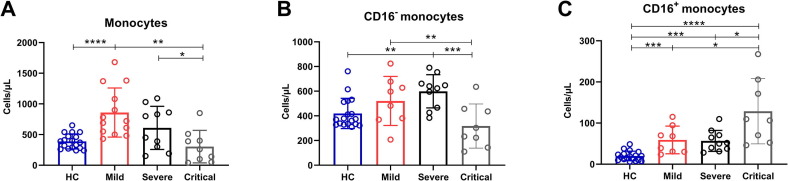
We detected the number of monocytes in COVID-19 patients with different severity and discovered that the significant increase of total monocytes in the Mild group was observed compared to HCs, and the sustained decrease of total monocytes in the Critical group was observed compared to the Mild group and the Severe group (Fig. 1 A). To determine the changes of different monocyte subsets in patients with COVID-19, HematoFlow was performed to determine the absolute number of CD16− and CD16+ monocytes in COVID-19 patients with different severity. We found that CD16− monocytes in the Critical group decreased significantly as compared with the Mild and the Severe group. Furthermore, the absolute number of CD16− monocytes in the Severe group was significantly increased compared to HCs (Fig. 1B). However, compared with the other three groups, CD16+ monocytes in the Critical group continued to increase. And the absolute number of CD16+ monocytes in COVID-19 patients with different severity all significantly increased compared to HCs (Fig. 1C).
Fig. 1.
The absolute number of total monocytes and subsets in COVID-19 patients. (A) The absolute number of circulating total monocytes in HCs and COVID-19 patients with mild, severe and critical illness. (B) The absolute number of CD16− monocytes in HCs and COVID-19 patients with mild, severe and critical illness. (C) The absolute number of CD16+ monocytes in HCs and COVID-19 patients with mild, severe and critical illness. Data are mean ± SEM. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, p < 0.0001.
3.2. Immune status of monocytes in COVID-19 patients
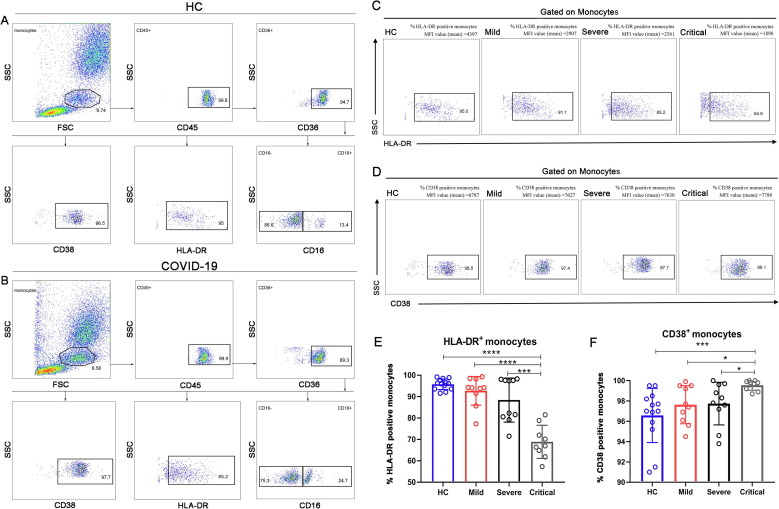
In order to investigate the antigen presentation potential of monocytes, gated as described in Fig. 2 A, B, we firstly detected the expression of HLA-DR on monocytes, and the analysis showed that the expression of HLA-DR in the Critical group was significantly reduced as compared with all other groups (Fig. 2C, E). Similarly, we examined the expression of CD38 on monocytes to understand their activation status, and found that the expression of CD38 in the Critical group relatively increased as compared with all other groups (Fig. 2D, F).
Fig. 2.
Immune status of monocytes in COVID-19 patients. (A, B) A full gating of flow cytometry plots of a HC and a severe COVID-19 patient. (C, D) Flow cytometry plots of HLA-DR/CD38 expression on monocytes in HCs and COVID-19 patients with mild, severe and critical illness are shown. Values in the quadrant represent the percentages of CD38+ monocytes/HLA-DR+ monocytes. MFI, Median Fluorescence Intensity. (E) The expression of HLA-DR on monocytes in HCs and COVID-19 patients with mild, severe and critical illness. (F) The expression of CD38 in HCs and COVID-19 patients with mild, severe and critical illness. The HLA-DR ratio % or CD38 ratio % represent the ratio of HLA-DR or CD38 expression on total monocytes. Calculated at the level of total monocytes. Data are mean ± SEM. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, p < 0.0001.
3.3. Cytokine levels in COVID-19 patients
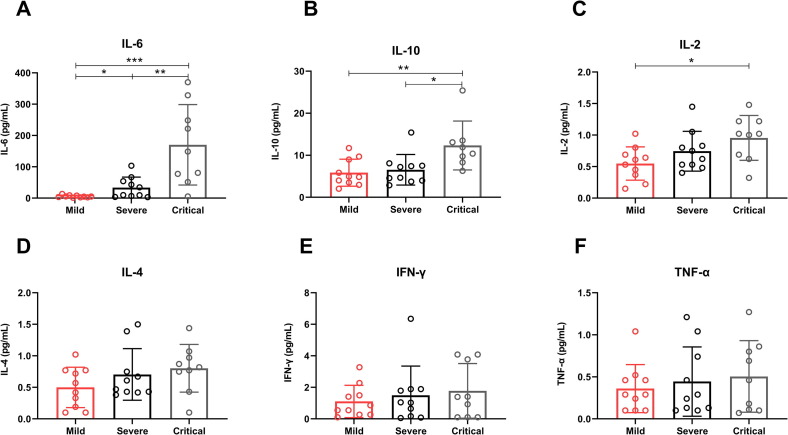
The cytokine levels, including IL-2, IL-4, IL-6, IL-10, IFN-γ and TNF-α were detected in the serum of our patient cohort. The levels of IL-6 and IL-10 showed sustained increases in the Critical group compared with the Mild group and the Severe group, and the IL-6 levels in the Severe group were higher than the Mild group (Fig. 3 A, B). Furthermore, IL-2 levels were also higher in the Critical group than the Mild group (Fig. 3C). While the levels of IL-4, IFN-γ and TNF-α had trends to increase, but no statistical significance as the disease progression of COVID-19 patients (Fig. 3D–F).
Fig. 3.
Cytokine levels in COVID-19 patients. (A) The levels of IL-6 (A), IL-10 (B), IL-2 (C), IL-4 (D), IFN-γ (E) and TNF-α (F) in the serum of COVID-19 patients with mild, severe and critical illness. Data are mean ± SEM.*, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, p < 0.0001.
3.4. The relationship between cytokine levels and monocytes by correlation analysis
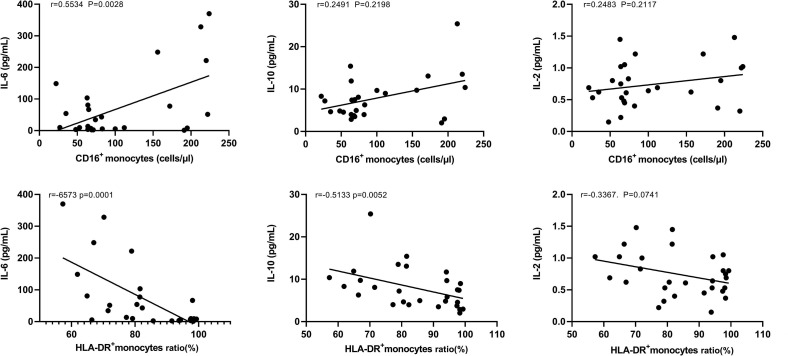
We further analyzed the relationship between the elevated cytokine levels (IL-6, IL-10 and IL-2) and the number of CD16+ monocytes in patients with COVID-19. We found that the levels of IL-6 were positively correlated with the number of CD16+ monocytes (Fig. 4 A). However, IL-10 and IL-2 levels were not related to the number of CD16+ monocytes (Fig. 4B, C). In addition, we found that IL-6 and IL-10 levels were negatively correlated with the expression of HLA-DR on monocytes (Fig. 4D, E). However, IL-2 levels did not correlate with the expression of HLA-DR on monocytes (Fig. 4F). It needed to be emphasized that we removed one outlier sample because we found that the correlations were skewed by the outlier sample, which was from a critical COVID-19 patient.
Fig. 4.
The relationship between cytokine levels and monocytes correlation analysis. (A–C) The relationship between cytokine (IL-6, IL-10 and IL-2) levels and the number of CD16+ monocytes in COVID-19 patients. (D–F) The relationship between cytokine (IL-6, IL-10 and IL-2) levels and the expression of HLA-DR on monocytes in COVID-19 patients.
3.5. Dynamic changes of monocytes in COVID-19 patients during recovery
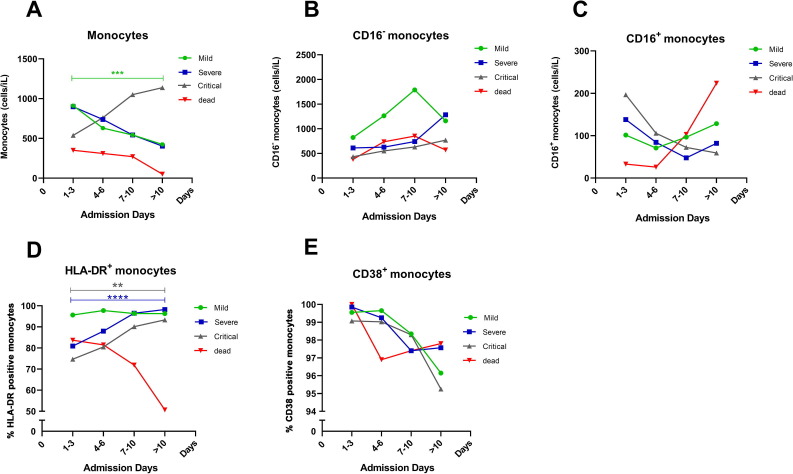
We selected randomly several patients in each group, including 8 mild cases, 4 severe cases, and 4 critical cases to analyzed the dynamic changes of monocytes in COVID-19 patients of different severity during the recovery process. In addition, we also observed the dynamic changes of monocytes in one critical patient before death. We found that with the remission of the disease condition, the absolute number of monocytes in mild and severe patients decreased and monocytes in critical patients increased (Fig. 5 A). We then analyzed the dynamic changes of monocyte subsets in COVID-19 patients. The absolute number of CD16− monocytes increased by degrees and CD16+ monocytes gradually decreased in severe and critical patients as the disease went into remission (Fig. 5B, C). We further analyzed the dynamic changes of the expression of HLA-DR and CD38 on monocytes during recovery in COVID-19 patients, and found that HLA-DR expression on monocytes increased significantly in severe and critical patients (Fig. 5D). However, the expression of CD38 on monocytes decreased in mild, severe and critical patients as the disease went into remission (Fig. 5E). In addition, we found that during the disease progression of one critical patient before death, the absolute number of monocytes and CD16− monocytes were always at a low level, while CD16+ monocytes increased and the expression of HLA-DR on monocytes decreased, but the expression of CD38 on monocytes had no significant changes (Fig. 5A–E).
Fig. 5.
Dynamic changes of monocytes in COVID-19 patients during recovery. (A–C) The absolute number of total monocytes, CD16− monocytes and CD16+ monocytes in COVID-19 patients with mild, severe and critical illness during recovery. (D, E) The expression of HLA-DR and CD38 on monocytes in COVID-19 patients with mild, severe and critical illness as the disease went into remission. (A–E) With one critical patient’s disease progression before death, the dynamic changes of the absolute number of monocytes, CD16− monocytes and CD16+ monocytes and the expression of HLA-DR and CD38 expression on monocytes. Mild patients (green line), severe patients (blue line), critical patients (grey line), one critical patient before death (red line). The X-axis coordinates represented days of admission to hospital of COVID-19 patients for treatment. Except for the red line, every other line represented the average of patient data. The red line represented the dynamic changes of monocytes in one critical patient before death. Selected randomly several patients in each group, including 8 mild cases, 4 severe cases, 4 critical cases and one critical patient before death. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, p < 0.0001. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
3.6. ROC curve analysis of monocytes in COVID-19 patients
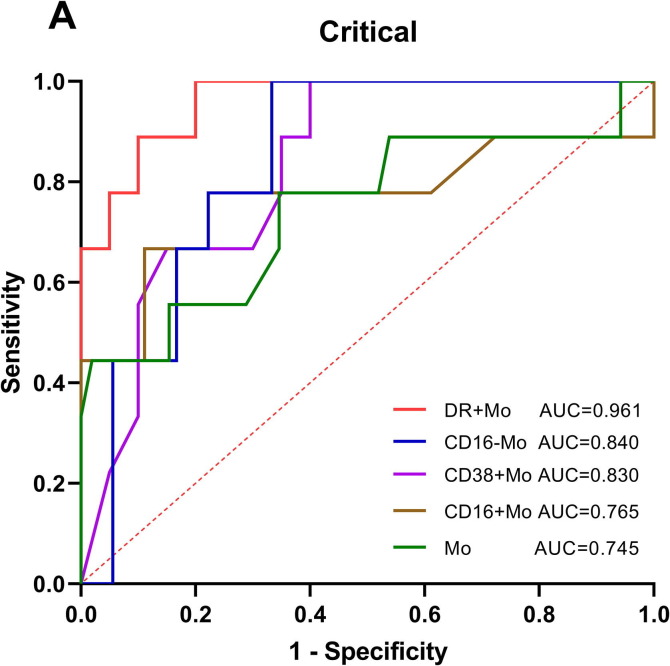
We further used parameters that had different expression in disease severity to predict the critical of COVID-19 patients. Through ROC curve analysis, we found that HLA-DR+ monocytes (AUC = 0.961) showed the best value for predicting the incidence of critical patients with high sensitivity of 100.00% and specificity of 80.00%; followed by CD16− monocytes (0.840), CD38+ monocytes (0.830), CD16+ monocytes (0.765), monocytes (0.745), with sensitivity of 100.00%, 100.00%, 77.78% and 77.78%, and specificity of 66.67%, 60.00%, 77.78% and 65.38%, respectively (Fig. 6 A). The details of the data were listed in Table 1 .
Fig. 6.
ROC curve analysis of monocytes in COVID-19 patients. DR + Mo (AUC = 0.961) showed the best predictive value for predicting the incidence of critical patients with COVID-19. Mo, monocytes; DR + Mo, HLA-DR+ monocytes; CD16-Mo, CD16− monocytes; CD38 + Mo, CD38+ monocytes; CD16 + Mo, CD16+monocytes; These parameters refer to the ability to discriminate critical cases from mild and severe cases; AUC, area under the curve.
Table 1.
ROC curve parameters for predicting the incidence of critical patients.
| Parameters | AUC | P | 95% CI | Cut-off value | Jordan index | Sensitivity | Specificity |
|---|---|---|---|---|---|---|---|
| DR + Mo | 0.961 | <0.0001 | 0.899–1.000 | <81.55 | 0.800 | 100.00% | 80.00% |
| CD16-Mo | 0.840 | 0.0047 | 0.690–0.989 | <6.53 | 0.667 | 100.00% | 66.67% |
| CD38 + Mo | 0.830 | 0.0050 | 0.682–0.979 | >98.50 | 0.600 | 100.00% | 60.00% |
| CD16 + Mo | 0.765 | 0.0270 | 0.534–0.997 | >0.83 | 0.556 | 77.78% | 77.78% |
| Mo | 0.745 | 0.0199 | 0.539–0.950 | <405.00 | 0.432 | 77.78% | 65.38% |
DR + Mo refers to the ability of HLA-DR+ monocytes for predicting the incidence of critical patients; Mo, monocytes; AUC, area under the curve; 95% CI, 95% confidence interval.
4. Discussion
COVID-19 is affecting the whole world, which threatening human health and life. Recent researches on immune cells in COVID-19 were mainly concerned with lymphocytes [12]. However, as an important part of innate immunity, monocytes cannot be ignored in the process of anti-SARS-CoV-2 infection. Currently, it was still largely unclear about the dynamic change of monocytes in critical patients with COVID-19.
In this study, we detected the absolute number of total monocytes in 32 COVID-19 patients with different severity. A significantly increased number of total monocytes in the mild patients and decreased total monocytes in the critical patients were observed in COVID-19 disease compared to HCs. We speculated that monocytes might proliferate to eliminate the viruses in mild patients, while the loss of monocytes in the critical patients suggested that innate immunity might be suppressed to a certain extent when the COVID-19 disease was in critical condition. It was consistent with the study of Benedetta Peruzzi et al., which indicated that not only lymphcytes, but monocytes were also severely affected by SARS-CoV-2 [13].
Monocytes can be divided into two distinct subsets based on the expression of CD16 [14], [15]. The main population of circulating monocytes is CD16− monocytes, whose major function is phagocytosis. CD16+ monocytes are known as pro-inflammatory cells, because they can produce great amounts of cytokines to induce inflammation [7], [16], [17]. Therefore, we further detected CD16− and CD16+ monocytes in COVID-19 patients with different severity. We found that the absolute number of CD16− monocytes in the severe patients was significantly increased compared to HCs. However, the absolute number of CD16− monocytes were significantly decreased in critical patients, indicating that the main phagocytosis function of monocytes in critical patients might be impaired. There was another possibility that the reduction in CD16− monocytes numbers in the critical patients could be due to CD16− monocytes exited from the circulation and transferred to the site of infection. But in our study, we believed that immunosuppression in critical patients led to the decrease in CD16− monocytes numbers. In addition, considering CD16+ monocytes were significantly increased in COVID-19 patients compared to HCs, especially in critical patients. It could be concluded that the decreased monocytes in critical patients might be mainly due to the reduction of CD16− monocytes, and in patients with COVID-19, especially critical patients, CD16+ monocytes might enter into pulmonary circulation, produce large cytokines and cause inflammation of the lungs. CD38 was historically identified as a surface activation marker in T cells and later found to be expressed in additional immune and non-immune cell types, including monocytes. In addition, CD38 can be used as a useful marker to monitor pro-inflammatory monocyte-mediated inflammatory disease status. Stephanie A. Amici et al. believed that anti-CD38 therapy could regulate CD38+ pro-inflammatory monocytes [18]. In our study, we found that the expression of CD38 on circulating monocytes in critical patients with COVID-19 was relatively higher than the other groups, which also suggested that the pro-inflammatory ability of monocytes in critical patients might be enhanced. Some studies found that CD38 played an important role in the infiltration of blood monocytes into tissues [19]. Therefore, we had reasons to speculate that the CD38+ pro-inflammatory monocytes in the peripheral blood of COVID-19 patients might enter into pulmonary circulation to cause inflammation of the lungs. Monocytes cannot only migrate to infected sites and directly engulf pathogens [6], but also present microbial antigens to T cells to initiate and regulate immune responses. The expression of HLA-DR on monocytes plays an essential role in presenting antigens to adaptive immune cells [7], [8]. And the expression of HLA-DR is negatively correlated with the severity of immune dysfunction [20]. The reduced expression of HLA-DR on monocytes has been widely accepted as a reliable indicator of immunosuppression in the ICU patients [21]. To further understand the immune status of monocytes in COVID-19 patients, we detected the antigen presentation potential of monocytes, and found that the expression of HLA-DR on circulating monocytes in critical patients was significantly decreased compared to HCs, mild and severe patients. The results indicated that the antigen presentation potential of monocytes in COVID-19 patients with critical disease might be impaired, and monocytes might not effectively present the antigens to T cells and regulate the immune response against SARS-CoV-2. In recent studies, researchers divided COVID-19 patients into Mild and Severe groups, and also found that COVID-19 patients with severe SARS-CoV-2 symptoms had down-regulated HLA-DR levels of monocytes [22], [23].
We further detected cytokine levels in the serum of COVID-19 patients and found that IL-6, IL-10 and IL-2 levels increased significantly in the critical patients. Through correlation analysis, we further found that IL-6 were positively correlated with the number of CD16+ monocytes. A previous study also found that great amounts of IL-6 could be detected in CD16+ pro-inflammatory monocytes, especially in ICU patients with COVID-19 [9]. These results suggested that in critical patients of COVID-19, increased activated CD16+ pro-inflammatory monocytes might be related to the production of great amounts of cytokines, especially IL-6, which could induce excessive inflammation and lead to immune disorders. On the other hand, it reminded us that IL-6 inhibition might mitigate severe inflammation in COVID-19 patients. A study had confirmed our conjecture that the use of IL-6 blockers can alleviate the inflammatory responses of COVID-19 patients [24]. Besides, we found that the IL-6 and IL-10 levels were negatively correlated with the expression of HLA-DR on monocytes in COVID-19 patients. Ohno Y. et al found that IL-6 could downregulate HLA-DR expression [25]. Therefore, we speculated that excessively secreted IL-6 and IL-10 in critical patients with COVID-19 might inhibit HLA-DR expression on monocytes, which could lead to impaired antigen presentation potential of monocytes. It was consistent with the study of Evangelos J. et al., which found that COVID-19 patient plasma strongly inhibited the expression of HLA-DR on monocytes, and it was partially restored by the IL-6 blocker Tocilizumab [26].
In addition, we analyzed the dynamic changes of monocytes during recovery in COVID-19 patients with different severity. We found that with the remission of the disease condition, the number of total monocytes and CD16− monocytes increased gradually, while CD16+ monocytes decreased in critical patients. Additionally, we also found that during recovery in COVID-19 patients, the expression of HLA-DR on monocytes increased, while CD38 on monocytes exhibited a downward trend. These results might suggest that in COVID-19 patients, the antigen presentation potential of monocytes restoratively enhanced and activation was weakened with the remission of the disease condition. Besides, we found that in the disease progression of one critical patient before death, the number of total monocytes was always at a low level and the expression of HLA-DR on monocytes decreased significantly, while CD16+ monocytes increased significantly, which was the opposite of a well-recovered COVID-19 patient. As the condition of COVID-19 patients deteriorated, the number of monocytes was reduced, their immune function might be suppressed and CD16+ monocytes might play pro-inflammatory function, which might mediate excessive inflammation. Besides, we found that HLA-DR+ monocytes (AUC, 0.961, Se = 100.00%, Sp = 80.00%) were valuable indicators for predicting the incidence of critical patients with COVID-19, which could be much valuable for patient treatment.
In summary, decline in number and HLA-DR expression of monocytes might lead to decreased antigen presentation potential and thus immunosuppression in critical patients with COVID-19 to some extent. However, increased CD16+ pro-inflammatory monocytes might be related to the production of large amounts of cytokines, especially of IL-6, which might mediate hyperinflammation and inhibit antigen presentation potential of monocytes. Besides, HLA-DR+ monocytes might serve as a meaningful assisted indicator to help predict the incidence of critical patients with COVID-19. These findings might provide a new perspective for the diagnosis and treatment of critical patients with COVID-19.
Funding
This work was supported by the Natural Science Foundation of Zhejiang Province of China (grant number LY16H200004).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Zhu N., Zhang D., Wang W. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382(8):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization (WHO), Coronavirus Disease (COVID-19) Outbreak Situation Reports. https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed 15 October 2020).
- 3.Wang D., Hu B., Hu C. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061–1069. doi: 10.1001/jama.2020.1585. (published online ahead of print, 2020 Feb 7) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huang C., Wang Y., Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. (published correction appears in Lancet. 2020 Jan 30) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kaur B.P., Secord E. Innate immunity. Pediatr. Clin. North Am. 2019;66(5):905–911. doi: 10.1016/j.pcl.2019.06.011. [DOI] [PubMed] [Google Scholar]
- 6.Serbina N.V., Jia T., Hohl T.M., Pamer E.G. Monocyte-mediated defense against microbial pathogens. Annu. Rev. Immunol. 2008;26:421–452. doi: 10.1146/annurev.immunol.26.021607.090326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang J., Zhang L., Yu C., Yang X.-F., Wang H. Monocyte and macrophage differentiation: circulation inflammatory monocyte as biomarker for inflammatory diseases. Biomark. Res. 2014;2(1):1. doi: 10.1186/2050-7771-2-1. Published 2014 Jan 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cheadle W.G. The human leukocyte antigens and their relationship to infection. Am. J. Surg. 1993;165(2A Suppl):75S–81S. doi: 10.1016/S0002-9610(05)81210-3. [DOI] [PubMed] [Google Scholar]
- 9.Fu B., Xu X., Wei H. Why tocilizumab could be an effective treatment for severe COVID-19? J. Transl. Med. 2020;18(1):164. doi: 10.1186/s12967-020-02339-3. Published 2020 Apr 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Qin C., Zhou L., Hu Z. Dysregulation of immune response in patients with COVID-19 in Wuhan, China [published online ahead of print, 2020 Mar 12] Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa248. (ciaa248 published online ahead of print, 2020 Mar 12) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.National Health Commission of China, New coronavirus Pneumonia Prevention and Control Program, Available at: http://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989.shtml?spm=C73544894212.P59511941341.0.0, (accessed 5 July 2020).
- 12.Jiang Y., Wei X., Guan J. COVID-19 pneumonia: CD8+ T and NK cells are decreased in number but compensatory increased in cytotoxic potential. Clin. Immunol. 2020;218:108516. doi: 10.1016/j.clim.2020.108516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Peruzzi B., Bencini S., Capone M. Quantitative and qualitative alterations of circulating myeloid cells and plasmacytoid DC in SARS‐CoV‐2 infection. Immunology. 2020 doi: 10.1111/imm.13254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ziegler-Heitbrock H.W., Passlick B., Flieger D. The monoclonal antimonocyte antibody My4 stains B lymphocytes and two distinct monocyte subsets in human peripheral blood. Hybridoma. 1988;7(6):521–527. doi: 10.1089/hyb.1988.7.521. [DOI] [PubMed] [Google Scholar]
- 15.Passlick B., Flieger D., Ziegler-Heitbrock H.W. Identification and characterization of a novel monocyte subpopulation in human peripheral blood. Blood. 1989;74(7):2527–2534. [PubMed] [Google Scholar]
- 16.Wong K.L., Tai J.J., Wong W.C. Gene expression profiling reveals the defining features of the classical, intermediate, and nonclassical human monocyte subsets. Blood. 2011;118(5):e16–e31. doi: 10.1182/blood-2010-12-326355. [DOI] [PubMed] [Google Scholar]
- 17.Ziegler-Heitbrock L. The CD14+ CD16+ blood monocytes: their role in infection and inflammation. J. Leukoc. Biol. 2007;81(3):584–592. doi: 10.1189/jlb.0806510. [DOI] [PubMed] [Google Scholar]
- 18.Amici S.A., Young N.A., Narvaez-Miranda J. CD38 is robustly induced in human macrophages and monocytes in inflammatory conditions. Front. Immunol. 2018;9:1593. doi: 10.3389/fimmu.2018.01593. Published 2018 Jul 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Malavasi F., Deaglio S., Funaro A. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol. Rev. 2008;88(3):841–886. doi: 10.1152/physrev.00035.2007. [DOI] [PubMed] [Google Scholar]
- 20.Zhuang Y., Peng H., Chen Y. Dynamic monitoring of monocyte HLA-DR expression for the diagnosis, prognosis, and prediction of sepsis. Front. Biosci. (Landmark Ed.) 2017;22:1344–1354. doi: 10.2741/4547. Published 2017 Mar 1. [DOI] [PubMed] [Google Scholar]
- 21.Gouel-Chéron A., Allaouchiche B., Guignant C. Early interleukin-6 and slope of monocyte human leukocyte antigen-DR: a powerful association to predict the development of sepsis after major trauma. PLoS One. 2012;7(3) doi: 10.1371/journal.pone.0033095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Silvin A., Chapuis N., Dunsmore G. Elevated calprotectin and abnormal myeloid cell subsets discriminate severe from mild COVID-19. Cell. 2020;182(6):1401–1418.e18. doi: 10.1016/j.cell.2020.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gatti A., Radrizzani D., Viganò P. Decrease of non-classical and intermediate monocyte subsets in severe acute SARS-CoV-2 infection. Cytometry A. 2020;97(9):887–890. doi: 10.1002/cyto.a.24188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xu X., Han M., Li T. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. U.S.A. 2020;117(20):10970–10975. doi: 10.1073/pnas.2005615117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ohno Y., Kitamura H., Takahashi N. IL-6 down-regulates HLA class II expression and IL-12 production of human dendritic cells to impair activation of antigen-specific CD4+ T cells. Cancer Immunol. Immunother. 2016;65(2):193–204. doi: 10.1007/s00262-015-1791-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Giamarellos-Bourboulis E.J., Netea M.G., Rovina N. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe. 2020;27(6):992–1000.e3. doi: 10.1016/j.chom.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]