Abstract
Lipoxygenases (LOXs) (EC 1.13.11.12) catalyze the oxygenation of fatty acids and produce oxylipins, including the plant hormone jasmonic acid (JA) and its methyl ester, methyl jasmonate (MeJA). Little information is available about the LOX gene family in aquatic plants. We identified a novel LOX gene family comprising nine LOX genes in the aquatic plant Spirodela polyrhiza (greater duckweed). The reduced anatomy of S. polyrhiza did not lead to a reduction in LOX family genes. The 13-LOX subfamily, with seven genes, predominates, while the 9-LOX subfamily is reduced to two genes, an opposite trend from known LOX families of other plant species. As the 13-LOX subfamily is associated with the synthesis of JA/MeJA, its predominance in the Spirodela genome raises the possibility of a higher requirement for the hormone in the aquatic plant. JA-/MeJA-based feedback regulation during culture aging as well as the induction of LOX gene family members within 6 h of salt exposure are demonstrated.
Keywords: duckweed, lipoxygenases (LOXs), MeJA, phylogenetics, Spirodela polyrhiza, oxylipin, salt
1. Introduction
Duckweeds (Lemnaceae) are a family of aquatic, floating, higher plants with extremely reduced anatomies [1]. Duckweeds are divided into five genera (Spirodela, Lemna, Landoltia, Wolffia, Wolffiella) and 36 species [2]. The plants are small, ranging from ~1.0 cm (Spirodela polyrhiza) down to ~0.5 mm (Wolffia globosa) in length. Duckweeds can flower, but normally propagate vegetatively, with several species doubling their biomass almost daily under optimal conditions in nature or the laboratory [3]. The chromosome numbers reported for various duckweed species range from 2n = 20 to 126, with genome sizes of 150 Mb (S. polyrhiza) to 1881 Mb (Wolffia arrhiza) [4]. Duckweeds have been used as a model system to study photosynthesis dynamics. It was the plant of choice that uncovered the dynamics of the photosystem II D1 protein [5,6,7] and has featured in ecotoxicological and phytoremediation studies [8]. There is also keen biotech interest in duckweeds as an unexploited source of high-quality protein to feed a growing world population [9,10,11,12]. Recent sequencing of full genomes of several duckweed species indicates that duckweeds are moving to the center of interest in genomic studies [13,14,15,16,17]. As more genomes are explored, research is unveiling the mechanistic details adopted for an aquatic lifestyle along with defining challenges faced in an open aquatic environment for maintaining aquatic growth and development. As phytohormones play a role in plant development and stress tolerance, how duckweeds utilize them for growth, stress tolerance, and phytoremediation needs to be explored and unraveled.
The phytohormone jasmonic acid (JA) and its methyl ester (MeJA), along with other compounds collectively called oxylipins, are synthesized via the lipoxygenase (LOX) pathway in plants [18,19,20]. LOXs (linoleate: oxygen oxidoreductase, EC 1.13.11.12) are lipid-oxidizing enzymes widely distributed as a family of non-heme-iron-containing fatty acid dioxygenases in both plants and animals [21,22,23,24]. LOX enzymes catalyze the biosynthesis of JA and MeJA, which provide defense against pathogens, including insects [25]. LOX proteins also positively impact bread making, aroma development, and flavor/color, features that are important to the agri-food industry [26,27]. LOXs are broadly classified into two families, 9-LOX and 13-LOX, based on the preferred addition of molecular oxygen either at carbon atom 9 or at carbon atom 13 of the hydrocarbon backbone [18]. Functional genetic analyses have identified a few phenotypes of different LOX genes. In Arabidopsis, wounding leads to the accumulation of AtLOX2 [28] and AtLOX6 transcripts [29]. Among the tomato LOX genes, LOX1/LOXA plays a role in seed germination [30], LOX2/LOXB in fruit ripening [31,32], LOX3/LOXC in generating volatile C6 and flavor compounds, and LOXD/LOX4 in plant defense [33,34]. Previously, we identified tomato LOX genes that are responsive to MeJA application [35] and to common abiotic stresses [36].
LOX enzymes are encoded in multi-gene families that have been described for soybean [37], Arabidopsis [38], Medicago and grape [39], cucumber [40], rice [41], apple [42], Chinese white pear [43], poplar [44], several legumes [45], pepper [46], cotton [47], tomato [35], tea [48], and radish [49], among others. However, to the best of our knowledge, a LOX gene family has not yet been described in any aquatic plant. We identify and describe the LOX gene family in the resolved genome of greater duckweed (S. polyrhiza). We show that nine LOX genes can be annotated as bona fide LOXs based on accepted LOX classification criteria. Comparative phylogenetics establish the relationship of the nine LOX genes with known LOXs from various species. Remarkably, the Spirodela LOX family is dominated by the 13-LOX, rather than 9-LOX, subfamily. Activation/suppression of specific members of the Spirodela LOX gene family in response to salt stress is presented.
2. Results
2.1. Identification of the LOX Gene Family in the Greater Duckweed, Spirodela polyrhiza
A protein homology search was carried out with available LOX gene family proteins from Arabidopsis, rice, poplar, and tomato [32,38,41]. Sequence similarities led us to the identification of 15 putative LOX genes in the Spirodela genome. However, a comprehensive genome-wide search of the Spirodela genome reduced the number to nine LOX genes containing both PLAT and lipoxygenase domains. Only these were considered as bona fide LOX genes and are summarized in Table 1.
Table 1.
Identification of bona fide lipoxygenase (LOX) genes annotated in the genome of Spirodela polyrhiza.
| Gene Name | Sequence ID | Sequence Coordinates | Genomic (bp *) |
ORF (bp *) |
Protein (aa *) |
Mol. Wt. (kDa *) |
pI | Predicted Subfamily # |
|---|---|---|---|---|---|---|---|---|
| SpLOX1 | Spipo2G0068200 | 5245164:5249140 (−) | 3977 | 2607 | 868 | 97.22 | 5.58 | 9-LOX |
| SpLOX2 | Spipo2G0068500 | 5257861:5263185 (−) | 5325 | 2535 | 844 | 95.40 | 6.12 | 9-LOX |
| SpLOX3 | Spipo4G0070100 | 5947369:5951085 (+) | 3717 | 2421 | 806 | 90.60 | 6.24 | 13-LOX |
| SpLOX4 | Spipo7G0050500 | 4554161:4559214 (+) | 5054 | 2751 | 916 | 102.81 | 7.10 | 13-LOX |
| SpLOX5 | Spipo15G0045200 | 4070955:4073755 (−) | 2801 | 2379 | 792 | 88.63 | 6.04 | 13-LOX |
| SpLOX6 | Spipo28G0005500 | 470166:475175 (−) | 5010 | 2715 | 904 | 102.13 | 6.29 | 13-LOX |
| SpLOX7 | Spipo28G0005600 | 487129:491614 (+) | 4486 | 2715 | 904 | 103.52 | 7.16 | 13-LOX |
| SpLOX8 | Spipo28G0005700 | 497951:504441 (+) | 6491 | 1914 | 637 | 71.66 | 6.99 | 13-LOX |
| SpLOX9 | Spipo0G0030100 | 2519505:2523103 (+) | 3599 | 2763 | 920 | 103.52 | 7.16 | 13-LOX |
* Abbreviations: bp: base pair; aa: amino acids; kDa: kilodaltons; # based on substrate preferences.
The six putative LOX genes that lacked a complete LOX domain and were not considered true LOX genes are listed in Supplementary Table S3. These sequences were also cross verified with the recent genome assembly of S. polyrhiza strain 7498v3 [17] and S. polyrhiza 9509 v3 [50]. A comparative location identifier is given in Supplementary Table S4 for the three genome assemblies used in this study to deduce the genome sequences of the nine Spirodela LOXs. The Spirodela LOX protein lengths varied between 637 and 920 amino acids and the predicted open reading frames ranged from 1914 to 2763 nucleotides. The calculated molecular masses of the nine Spirodela LOXs ranged from 71.66 to 103.52 kDa and varied in pI values from 5.58 to 7.16. SpLOX7 and SpLOX9 have very similar isoelectric points, suggesting them to be isoenzymes. The Spirodela LOX gene family is 1.5-fold larger than that in Arabidopsis.
2.2. Comparative Phylogeny of the S. polyrhiza LOX Gene Family Reveals Dominance of 13-LOX Sub-Family Genes
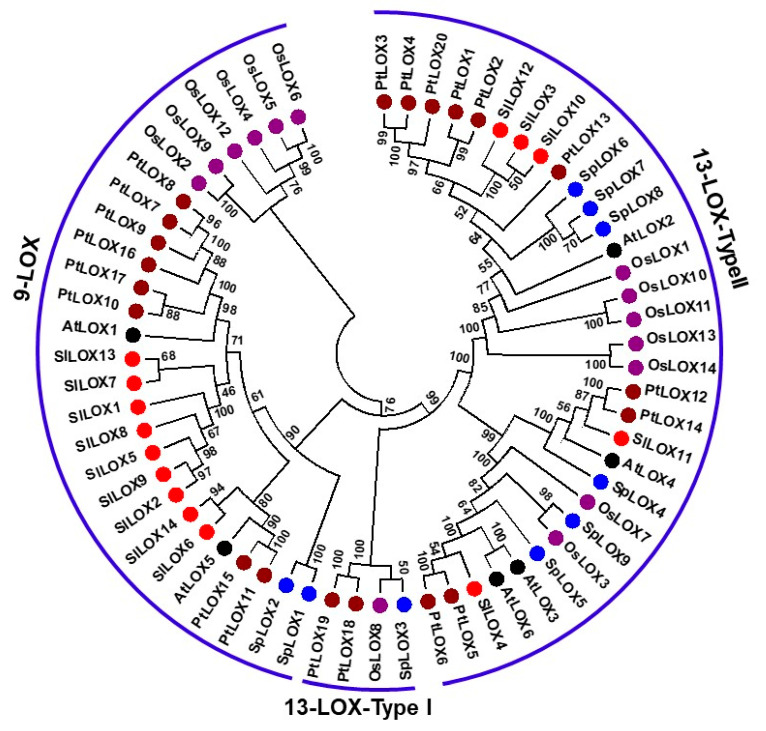
To establish an evolutionary relatedness of the Spirodela LOX gene family to other known LOX gene families, we utilized sixty-three protein sequences belonging to duckweed (9), tomato (14), Arabidopsis (6), rice (14), and poplar (20) LOX genes. Only previously characterized complete LOX gene families were considered for the phylogenetic relationship. A maximum likelihood method based on the JTT matrix-based model divided the nine Spirodela LOX proteins into two distinct subfamilies: 9-LOX and 13-LOX, based on their substrate preferences.
Two Spirodela LOX genes (SpLOX1 and SpLOX2) aligned with the 9-LOX genes previously known from tomato, Arabidopsis, rice, and poplar, while seven Spirodela LOX genes (SpLOX3-9) aligned with previously known 13-LOX genes (Figure 1). The seven Spirodela 13-LOX proteins were sub-divided into two types with respect to their protein structure: type I lack plastid targeting peptides and their sequences are over 75% similar, while type II possess a plastid targeting peptide, but the sequence similarity among them is low. We observed that only poplar PtLOX18 and PtLOX19, rice OsLOX8, and Spirodela SpLOX3 belong to type I (Figure 1).
Figure 1.
Molecular phylogenetic relationships of the Spirodela polyrhiza lipoxygenase (LOX) family with LOX gene families from additional plants. The evolutionary history was inferred using the maximum likelihood method based on the JTT matrix-based model. The bootstrap consensus tree inferred from 1000 replicates is taken to represent the evolutionary history of the taxa analyze. The analysis involved 63 amino acid sequences from five different plants: duckweed (Sp), tomato (Sl), Arabidopsis (At), rice (Os), and poplar (Pt). All positions with less than 95% site coverage were eliminated. Evolutionary analyses were conducted in MEGA7. The bootstrap values of the confidence levels are shown as percentages at branch nodes. The LOX gene family of each species is color coded. LOXs of different species fall into two separate groups: 9-LOX and 13-LOX. The 13-LOX group was further subdivided into type I and type II. Phylogenetic analysis assigned seven LOX proteins from Spirodela to the 13-LOX group and two LOX proteins to the 9-LOX group.
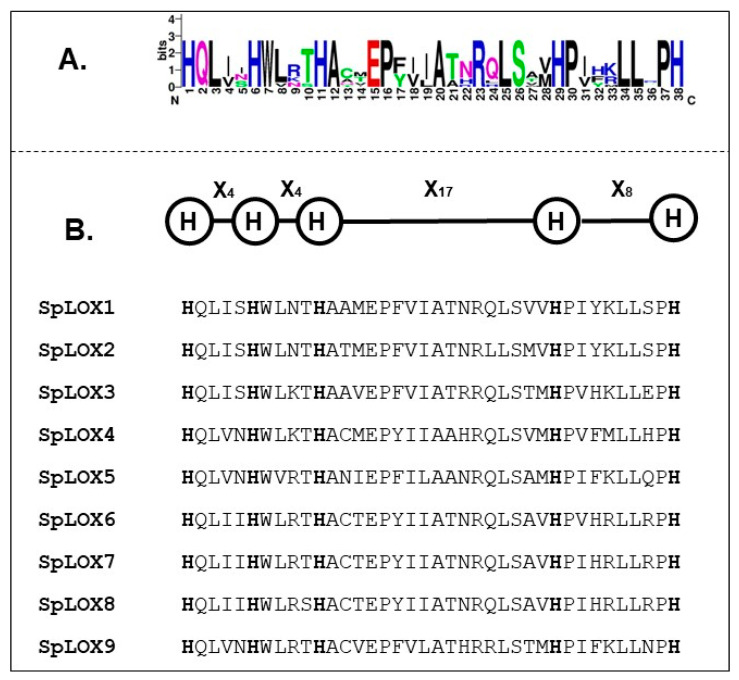
2.3. Analysis of 5-Histidine Conserved Motifs in Spirodela LOX Protein Sequences
All nine Spirodela LOX protein sequences identified here contain the LOX (PF00305), PLAT/LH2 (PF01477), and 5-Histidine signature domains (Table 2). The PLAT (polycystin-1, lipoxygenase, alpha-toxin)/LH2 (lipoxygenase homology) domain is found in a variety of membrane or lipid associated proteins including plant lipoxygenases and forms a beta-sandwich composed of two β-sheets of four β-strands each.
Table 2.
Location of signature domains in Spirodela LOX protein sequences. PLAT, polycystin-1, lipoxygenase, alpha-toxin.
| Gene Name | Sequence ID | PLAT Domain 1, 2 |
LOX Domain |
5-Histidine Domain |
|---|---|---|---|---|
| SpLOX1 | Spipo2G0068200 | 55–162HMMER | 175–846HMMER | 519–556 |
| SpLOX2 | Spipo2G0068500 | 71–165HMMER | 178–822HMMER | 522–559 |
| SpLOX3 | Spipo4G0070100 | 16–131CDART | 135–784HMMER | 479–516 |
| SpLOX4 | Spipo7G0050500 | 115–216HMMER | 229–899HMMER | 567–604 |
| SpLOX5 | Spipo15G0045200 | 28–99HMMER | 112–775HMMER | 447–484 |
| SpLOX6 | Spipo28G0005500 | 123–207HMMER | 220–887HMMER | 557–594 |
| SpLOX7 | Spipo28G0005600 | 121–207HMMER | 222–887HMMER | 557–594 |
| SpLOX8 | Spipo28G0005700 | 23–120HMMER | 129–620HMMER | 466–503 |
| SpLOX9 | Spipo0G0030100 | 138–223 HMMER | 236–903 HMMER | 574–611 |
1 HMMER search tool can be accessed at https://www.ebi.ac.uk/Tools/hmmer/; 2 CDART search tool can be accessed at https://www.ncbi.nlm.nih.gov/Structure/lexington/lexington.cgi.
The lipoxygenases contain six histidine residues, five of which exhibit a conserved pattern in most known lipoxygenase proteins [35]. The positions of the histidine residues in the 38 amino-acid-residue stretch making up the 5-Histidine signature domain are highly conserved in all nine Spirodela LOXs (Figure 2A). This same arrangement, His-(X)4-His-(X)4-His-(X)17-His-(X)8-His, was previously found in other plant LOX protein sequences; for example, in poplar [44], tomato [35], and radish [49] (Figure 2B). The conserved histidine motif has been suggested to play an important role in lipoxygenase enzyme stability and activity [35].
Figure 2.
Identification of conserved histidine (H) residues in the 38 aa signature LOX motif in Spirodela. (A) 38-residue motif among Spirodela LOX sequences. The sequence logo was created with the indigenous nine LOX protein sequences. The average height of each stack indicates the sequence conservation at that position and the height of each residue letter indicates the relative distribution frequency of the corresponding amino acid residue in the 38 amino acid long motif. (B) Sequence alignment of the 38-residue long motif in the Spirodela LOX proteins. The conserved histidine residues are highlighted as bold H.
2.4. Structural Analysis of the Spirodela LOX Genes
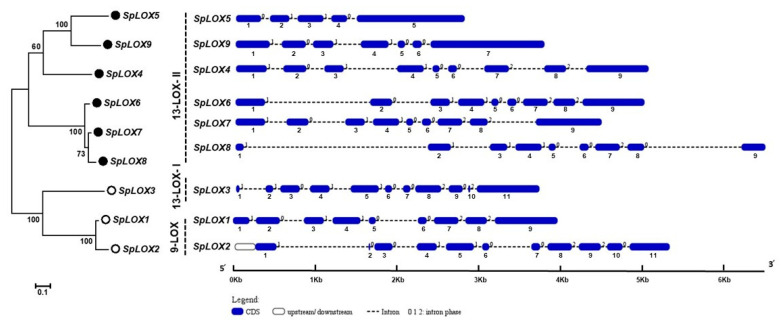
A separate phylogenetic tree was constructed using the nine duckweed LOX coding sequences to compare the exon/intron organization (Figure 3). The intron distribution pattern along with the phase distribution was detected by aligning the Spirodela genomic and coding sequences. Similar exon arrangements have been suggested to indicate a high duplication rates within the LOX family [44]. The Spirodela LOX family has asymmetrical exon positioning and different phases of intron distribution, which suggests that all SpLOX proteins have gone through protein evolution as part of organismal complexity (Figure 3).
Figure 3.
Spirodela LOX family genes’ relatedness and intron-exon arrangements. The evolutionary history was inferred using the maximum likelihood method based on the JTT matrix-based model. The percentage of trees in which the associated taxa clustered together is shown next to the branches. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site. The analysis involved nine amino acid sequences. All positions with less than 95% site coverage were eliminated. Evolutionary analyses were conducted in MEGA7 [51]. The Spirodela LOX subfamilies 9-LOX and 13-LOX are designated, with 13-LOX further separated into type I (13-LOX- I) and type II (13-LOX- II). Schematic genomic organization for each LOX gene was generated using the Gene Structure Display Server (GSDS 2.0; http://gsds.cbi.pku.edu.cn/). Exons (CDS) and introns are represented by blue boxes and black dashed lines, respectively. The sizes of exons and introns are proportional to their sequence lengths.
2.5. Tandem Duplications among Spirodela LOXs
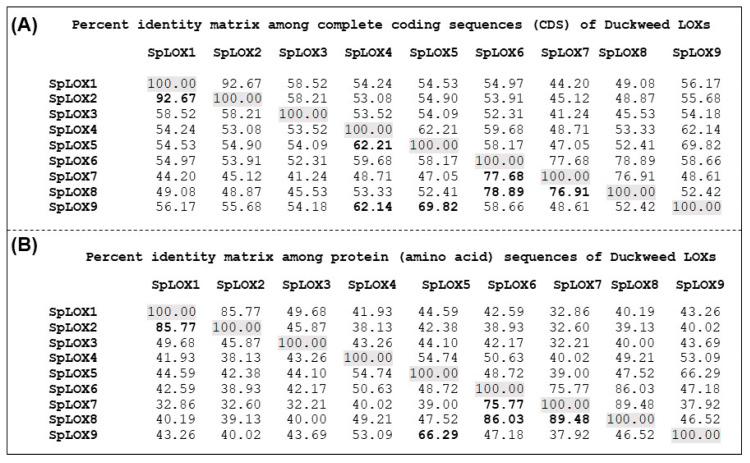
Segmental and tandem duplication events among Spirodela LOXs were assessed based on nucleotide and protein similarities among sequences (Figure 4A,B). Segmental duplication events were not found. However, tandem arrangement of SpLOX1 (Spipo2G0068200) and SpLOX2 (Spipo2G0068500) is indicative of a possible duplication event, with the two sharing 92.67% nucleotide sequence identity and 85.77% amino acid identity. Similarly, the tandem arrangement of SpLOX6 (Spipo28G0005500), SpLOX7 (Spipo28G0005600), and SpLOX8 (Spipo28G0005700) indicates a possible second tandem duplication event. SpLOX6 and SpLOX7 share 77.68% nucleotide sequence identity and 75.77% amino acid identity. The SpLOX6-SpLOX8 pair shares 78.89% nucleotide identity and 86.03% amino acid identity, while the SpLOX7-SpLOX8 pair shares 76.91% nucleotide identity and 89.48% amino acid identity (Figure 4A,B). These results suggest that the duckweed LOX gene family underwent rearrangements and increased in number during evolution, similar to what occurred in the land plant LOX gene families.
Figure 4.
Spirodela LOX sequence identities. (A) Coding DNA sequence and (B) protein sequence identity matrices were generated using EMBOSS stretcher (https://www.ebi.ac.uk/Tools/psa/emboss_stretcher/). The values in bold face are discussed in the text.
2.6. Spirodela LOX Gene Family Transcript Accumulation Changes with Culture Age
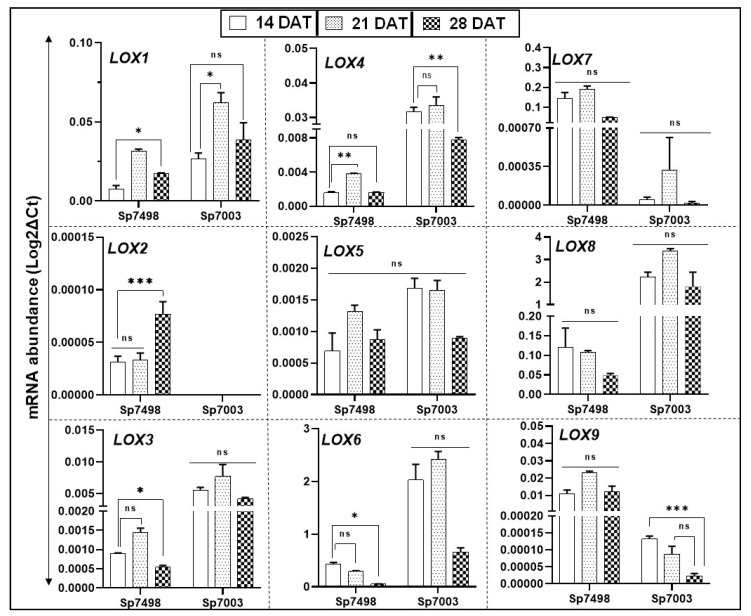
Two clones of S. polyrhiza (Sp7498 and Sp7003) were grown and harvested after 14, 21, and 28 days to assess if LOX gene family transcript abundance remains the same or changes as a function of culture age. Relative gene expression data suggest that LOX gene expression varies to a great extent between clones and to some extent at different plant age stages within a clone (Figure 5).
Figure 5.
qRT-PCR analysis of LOX genes in two clones of Spirodela polyrhiza, Sp7498and Sp7003. Cultures were grown for 28 days in nutrient solution and samples collected at 14, 21, and 28 days of growth. Confluence of plants in the culture flasks was reached after 21 days of growth. mRNA expression profiles of whole plants from the two clones were analyzed. SpACT and Sp18SrRNA housekeeping genes were used to normalize the expression of LOX genes, as described in Materials and Methods. Analysis of variance (ANOVA) with Dunnett’s multiple comparisons test was performed for significant differences in LOX gene expression in the aging plants. Statistical significance between expression data points was assessed against the 14-day expression profiles and categorized as * p < 0.05, ** p < 0.01, and *** p < 0.001 using Graph Pad (version 8.0); ns: not significant.
LOXs 1, 3, and 4 showed strain-independent expression patterns with similar expressions in both strains, while LOXs 5–9 genes showed a strain-dependent expression pattern with a variation in both strains. However, as most of the differences were not statistically significant, they suggest only likely trends (Figure 5). Three patterns were observed for LOX family genes: (i) genes whose expression increases from 14 d to 28 d of growth (LOXs 1 and 2 from Sp7498 and LOX1 from Sp7003); (ii) genes whose expression increases from 14 d to 21 d and then decreases (LOXs 3, 4, 5, and 9 from Sp7498 and LOXs 3, 4, 6, 7, and 8 from Sp7003); and (iii) genes whose expression decreases from 14 d onward to 28 d (LOXs 6, 7, and 8 from Sp7498 and LOXs 5 and 9 from Sp7003). Surprisingly, LOX2 was not expressed in S. polyrhiza 7003 and could not be recorded even at 45 cycles of qRT-PCR.
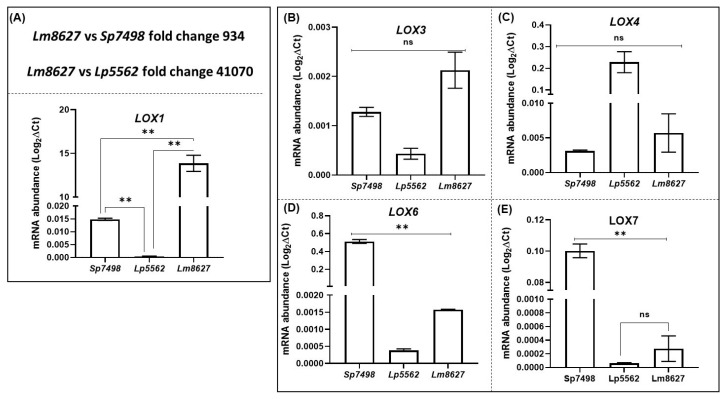
2.7. Expression of Most LOX Genes in Lemna minor and Landoltia punctata is Significantly Lower Compared to Spirodela polyrhiza
LOX gene expression profiles of S. polyrhiza 7498 were then compared with those in L. minor 8627 and L. punctata 5562 at 14 d. Except for LOX1, most of the LOX genes were expressed at low levels in Lemna and Landoltia clones. The expression of LOX1 gene in L. minor was ~4 × 104-fold higher than the LOX1 gene in L. punctata and 9 × 102-fold higher than in S. polyrhiza (Figure 6A). Expression levels of LOX3 and LOX4 genes were not significantly different among these three clones (Figure 6B,C), but expression of LOX6 and LOX7 genes was much lower in both the Lemna and Landoltia clones as compared with that in Spirodela (Figure 6C,D). These results indicate that the expression of LOX genes 2, 5, 8, and 9 is either very low, and thus un-quantifiable, or that they are not expressed in Lemna and Landoltia. Why many LOX genes are not expressed or are expressed at very low levels in Lemna and Landoltia could be a potential scientific question for future studies. On another note, LOX1 expression seems to be Lemna dominated and a future functional study could reveal its putative function.
Figure 6.
Comparative qRT-PCR analysis of LOX genes in Spirodela polyrhiza 7498, Lemna minor 8627, and Landoltia punctata 5562. Plants were taken for expression analysis at different ages of culture. All cultures were grown for 28 days in nutrient solution and samples were collected at 14, 21, and 28 days of growth. The SpACT and Sp18SrRNA housekeeping genes were used to normalize the expression of LOX genes, as described in Materials and Methods. (A) LOX1, (B) LOX3, (C) LOX4, (D) LOX6, and (E) LOX7 expression comparison was made among the three strains. ANOVA with Dunnett’s multiple comparisons test was performed for significant differences in LOX gene expression in the aging tissues among the duckweed species. Statistical significance between aging expression data points was assessed against the 14-day expression profiles and categorized as ** p < 0.01, using Graph Pad (version 8.0); ns: not significant.
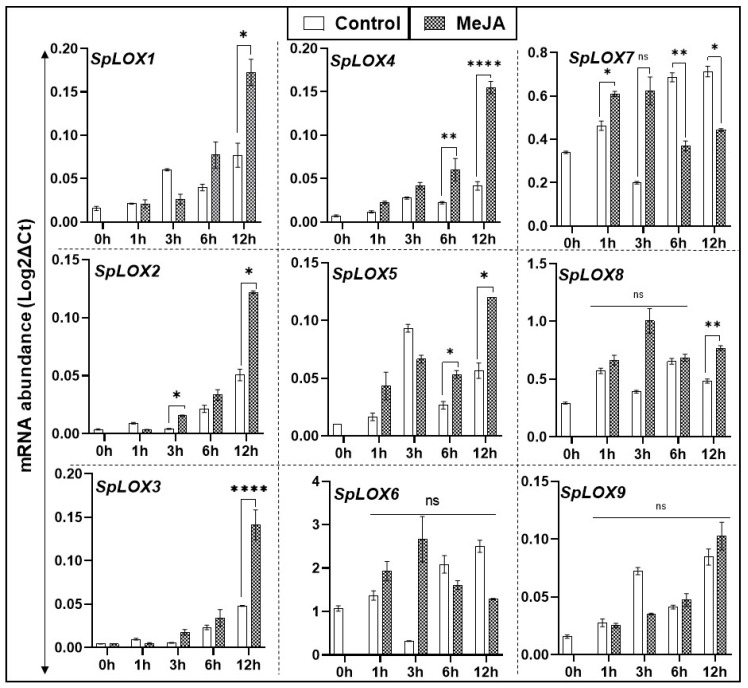
2.8. Methyl Jasmonate Treated Fronds and Transcript Abundance in Spirodela LOX Genes
Exogenous MeJA treatment was found to significantly enhance transcript expression of Spirodela LOXs 2, 4, 5, and 7. MeJA also upregulated LOXs 1, 3, and 8, but only at 12 h after treatment. LOXs 6 and 9 were non-responsive to the treatment (Figure 7). Thus, MeJA dependent and independent members of LOX gene family in Spirodela were identified.
Figure 7.
Methyl jasmonate (MeJA) induced feed-back regulation of Spirodela LOX gene family. MeJA (10 μM) was added to 14-day-old S. polyrhiza 7498 cultures, as previously described [35]. Samples were collected at 0, 1, 3, 6, and 12 h after treatment. Gene expression data were analyzed in treated and untreated fronds by qRT-PCR. Statistical significance between treatment data points was assessed with respect to control for each time point and categorized as * p < 0.05, ** p < 0.01, and **** p < 0.0001 using graph pad (version 8.0); ns: not significant.
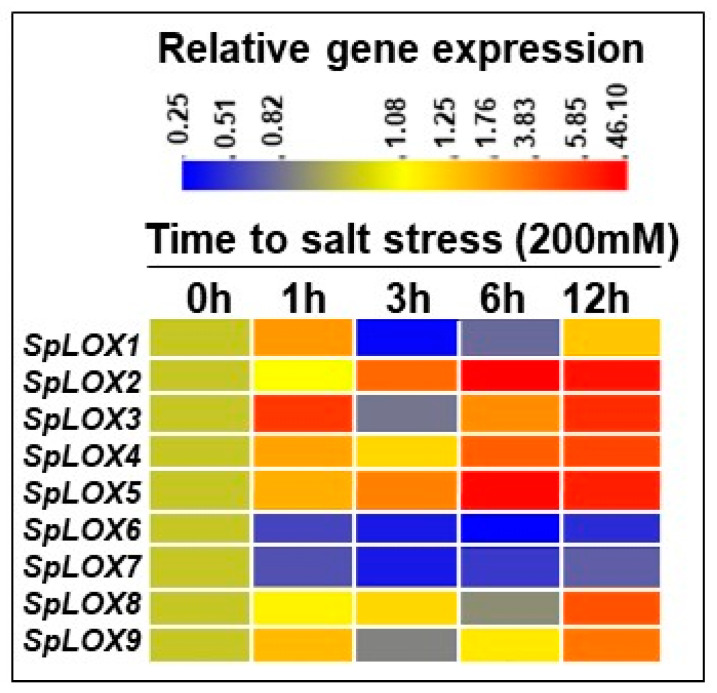
2.9. Salt Stress and Response of Spirodela LOXs
Upregulation of Spirodela LOXs in response to addition of 200 mM NaCl to the growth flasks occurred for LOX 2 (3–12 h); LOXs 3, 4, and 5 (6–12 h); and LOXs 8 and 9 (at 12 h). In comparison, LOXs 6 and 7 were downregulated (Figure 8). LOX 1 was initially upregulated (at 1 h), but remained downregulated thereafter. Thus, salt stress in Spirodela does not seem to favor induction of the 9-LOX pathway over the 13-LOX pathway, or vice versa. To complement these findings, we mined available transcriptome data from a salt experiment study in duckweeds using 100 mM NaCl (Supplementary Figure S1). Expression of only three LOX genes was captured in the transcriptome data. Time-dependent transcriptome analysis showed downregulated expression of LOX genes 1, 5, and 8. This complements the transcriptome data shown for LOX1 and LOX8, and partially for LOX5 (Figure 8).
Figure 8.
Comparative qRT-PCR analysis of LOX genes in Spirodela in response to exogenous salt. Five fronds of S. polyrhiza 7498 were grown for 14 days; then 200 mM NaCl solution was added; and samples were harvested at 0, 1, 3, 6, and 12 h [36]. Gene expression data were analyzed by qRT-PCR. The experiment was repeated three times (n = 3) with each sample size of approximately 100 fronds. Analysis of variance (ANOVA) with Dunnett’s multiple comparisons test was performed for significant differences. Statistical significance between data points was assessed against 0 h versus other time points of expression profiles using graph pad (version 8.0).
3. Discussion
Duckweeds have been used to study photosynthesis dynamics [52], environmental remediation [8], and an biomanufacturing source of protein and amino acids [9,10]. The genomes of several duckweed species were recently resolved [13,14,15,16]. This has generated an interest in understanding and unraveling how close the genomes of these aquatic plants are to those of land plants. For instance, the genome size of Spirodela polyrhiza was found to be similar to that of Arabidopsis thaliana, with the most recent assembly consisting of 18,708 protein-coding genes. Here, we have identified a novel lipoxygenase (LOX) gene family in the duckweed Spirodela polyrhiza 7498 utilizing the two assemblies hosted at Phytozome (http://www.phytozome.net/) [13] and the newest assembly hosted at NCBI [17]. Nine loci were annotated as bona fide LOX genes based on the presence of the critical LOX domain (PLAT/LH2) and the 38-aa long conserved 5-histidine motif [35]. Interestingly, among the nine LOX genes of Spirodela, the 13-LOX subfamily predominated with seven genes, while the 9-LOX subfamily seems to have shrunken to just two genes. The ratio of 13-LOX versus 9-LOX is 3.50 for S. polyrhiza, 2.00 for Arabidopsis, 0.56 for tomato, 1.50 for poplar, and 1.33 for rice (Figure 1).
The 13-LOX subfamily members are involved in the synthesis of the hormone jasmonic acid and its methyl ester MeJA [53]. Thus, the high ratio of 13-LOXs to 9-LOXs in Spirodela, and possibly other duckweeds, may indicate an increased need for 13-LOX genes in regulating the production of JA/MeJA in this aquatic plant. It is important to note that the role of JA in regulating floral induction, evocation, and differentiation in Lemna minor has been suggested previously [54]. These authors showed that the endogenous levels of JA in L. minor were highest during the vegetative stage (day 14 of culture), which dropped by 44% on day 21 of culture at the apical floral induction stage, and was drastically down by 83% on day 28 of culture in the flowering plants (Table 3). This suggests that JA accumulation is negatively correlated with floral induction in Lemna minor and possibly other duckweeds. In this context, as 13-LOX genes are important for the synthesis of JA/MeJA, our findings of a similar trend in expression levels of JA/methyl-JA with ageing of vegetative cultures for six of the seven 13-LOX genes of S. polyrhiza 7498 is interesting and fits well with what was reported [54] for the Lemna flowering stages (Table 3). Very little is known about the function of the LOX family of genes in any aquatic plant, and our studies suggest that the 13-LOX gene family members may play a role in the developmental program of S. polyrhiza.
Table 3.
Summary of jasmonate expression patterns of S. polyrhiza 13-LOX genes during culture aging and decrease in endogenous jasmonate levels in Lemna minor during flower induction. MeJA, methyl jasmonate; JA, jasmonic acid.
| Growth Stages * | Culture Stage *(Days After Inoculation) | Endogenous JA Levels (ng−1 FW) * | 13-LOX Genes with Similar Patterns (Figure 7—This Study) # |
|---|---|---|---|
| Vegetative stage | Day 14 | 389 ± 8 | LOX3, 4, 6, 7, 8, 9 |
| Apical floral induction | Day 21 | 217 ± 13 | |
| Flowering plants | Day 28 | 37.5 ± 1.8 |
* Modified after [54]. # Coordination with quantified expression patterns of JA/MeJA-induced LOX genes in duckweeds (this study).
Another aspect of the other roles JA and MeJA play in plants deals with their defense-related functions. JA/MeJA are known to impact biotic and abiotic responses [51,53]. Here, we also categorized S. polyrhiza LOXs 1, 2, 3, 4, 5, and 8 as upregulated and LOXs 6 and 9 as static genes in response to MeJA treatment. An added novel observation was the finding that the LOX1 gene is highly expressed in L. minor compared with S. polyrhiza or L. punctata. As little or no information is available about molecular mechanisms of JA/MeJA action in duckweeds, future investigations are needed to determine if these aquatic plants follow the land plant-based regulation patterns or utilize as yet unknown processes. In this context, it is important to note that an early study on the germination of photoblastic light-grown as well as dark-grown turions of S. polyrhiza (L.) showed them to be stimulated by JA and MeJA [55]. Moreover, a significant increase in LOX activity in L. minor in response to cadmium-induced toxicity was shown, which was mildly alleviated upon application of salicylic acid [56]. Along with these data, our study paves the way for future research to test whether the role of JA/MeJA is related to defense and/or turion germination in duckweeds. We envision that induction or suppression of lipoxygenase(s) may play a protective role in stress pathways in duckweeds.
Salinity stress has both osmotic and cytotoxic effects on plant growth and development. This is particularly evident in land plants as JAs can alleviate salt stress by increasing the endogenous hormones and the antioxidative system [57,58,59,60]. Downregulation of three LOXs has been shown in a previous transcriptome analysis of duckweed exposed to salt stress [61]. However, our studies demonstrated that salt stress of duckweeds causes a dynamic pattern of the LOX gene family, with induction of several of them within 6 h of salt exposure. These results raise the possibility of a different trend for LOX genes in aquatic plants in response to salt stress as compared with land plants.
4. Materials and Methods
4.1. Plant Material, Growth Conditions, and Salt Stress Treatments
Four duckweed species from three different genera, Spirodela polyrhiza 7003, Spirodela polyrhiza 7498, Lemna minor 8627, and Landoltia punctata 5562, were grown axenically in Hoagland’s E-medium with pH 5.8 (https://www.mobot.org/jwcross/duckweed/media.htm) at room temperature (22 °C ± 2) under white light (50–80 μmol/sec2). Three growth periods (14 ± 1-day, 21 ± 1-day, and 28 ± 1-day) were chosen as described previously [62]. Duckweed clones or strains can be accessed at Rutgers Duckweed Stock Cooperative (RDSC) web page (http://www.ruduckweed.org). All experiments were carried out in triplicate. Samples were taken at indicated time points, washed thrice with distilled water, frozen in liquid nitrogen, and stored at −70 °C until used.
4.2. Sequence Retrieval of the LOX Gene Family from Spirodela polyrhiza
The genome sequence from Spirodela polyrhiza strain 7498v2 hosted at phytozome database (http://www.phytozome.net/) was utilized to retrieve putative duckweed LOX sequences [13]. These sequences were also cross verified with recent genome assembly of Spirodela polyrhiza strain 7498v3 (NCBI accession: GenBank assembly accession: GCA_008360905.1] [17]. A cross verification with another strain assembly was also done with S. polyrhiza 9509 v3 (GenBank assembly accession: GCA_900492545.1) [50]. The multiple bioinformatics approaches were employed to identify and characterize potential LOX gene family members in duckweed, as described earlier [35]. LOX gene family sequences from Arabidopsis (6), rice (12), and poplar (42) were downloaded from the phytozome database (http://www.phytozome.net/) and 14 tomato LOX gene family sequences [35] were downloaded from the Solanaceae [International Tomato Genome Sequencing Consortium (SGN; solgenomics.net) database, version ITAG 2.4]. BLASTp was used to search for similar protein sequences [35]. Putative duckweed LOX protein sequences were retrieved using hidden Markov model (HMM) analysis, with the seed sequence (Pfam # PF00305) (http://pfam.janelia.org) containing a typical LOX domain as query in an HMMER search (https://www.ebi.ac.uk/Tools/hmmer/) [35,63]. Moreover, a key word search of ‘Lipoxygenase” against the S. polyrhiza 7498v2 (Phytozome v13) genome yielded an additional 15 putative annotations. Finally, a cross verification approach of retrieved LOX protein sequences for the presence of signature domains was carried out as described [35,40,45]. Separately, Inter-Pro-Scan (http://www.ebi.ac.uk/Tools/InterProScan/) was used to confirm the presence of the LOX and PLAT/LH2 (polycystin-1, lipoxygenase, α-toxin domain, or the lipoxygenase homology) domains in the retrieved LOX sequences as described [64]. The predicted molecular weight and isoelectric point (PI) for each LOX protein were obtained using tools available at the ExPASy bioinformatics resource portal (https://www.expasy.org).
4.3. Phylogenetic Analysis of the S. polyrhiza LOX Family with Previously Characterized LOX Gene Families
A multiple sequence alignment of the identified tomato LOX protein sequences was performed using the MUSCLE program (http://www.ebi.ac.uk/Tools/msa) [65]. A phylogenetic tree was constructed by the maximum likelihood method with Poisson correction using 1000 boot strap values [66,67]. This analysis involved 54 amino acid sequences from Arabidopsis, tomato, rice, and poplar and nine sequences from S. polyrhiza 7498 (Supplementary Table S1). An evolutionary relatedness tree was generated using the MEGA7 program [68]. The global alignment tool EMBOSS Needle was used for pairwise alignment of S. polyrhiza 7498 LOX proteins to determine sequence identity and similarity.
4.4. Conserved Motifs in LOX Protein Sequences and Subcellular Localizations
The MEME suite [69] was applied to search for conserved motifs in LOX protein sequences. The maximum motif number was set to 20 and motif length to 8–100 aa (http://meme-suite.org/tools/meme/). Searching for the classical 6X histidine signature motif was done manually for each LOX protein and displayed using WebLogo3 (http://weblogo.threeplusone.com/) [35]. The protein sequence identity matrix was generated by EMBOSS stretcher (https://www.ebi.ac.uk/Tools/psa/emboss_stretcher/).
4.5. Gene Structure and Gene Duplication Analyses
Genomic DNA and coding DNA sequences corresponding to each identified tomato LOX gene were retrieved for S. polyrhiza from the phytozome database (http://www.phytozome.net/) and analyzed for intron-exon and intron phase distribution patterns (http://gsds.cbi.pku.edu.cn/). Tandemly duplicated gene pairs were identified as described [70]. Similarity indexes for nucleotides and amino acids were calculated using clustal omega (https://www.ebi.ac.uk/Tools/msa/clustalo/). More than 90% sequence similarity among genes was considered as segmental duplication [71], while tandem duplication events involved five or fewer genes within a 100 kb region.
4.6. Total RNA Extraction, cDNA Preparation, and Quantitative Real Time PCR (qRT-PCR)
Total RNA was extracted from 100 mg of each sample using the plant RNeasy kit according to manufacturer’s instructions (Qiagen, Germantown, MD, USA). RNA samples with an A260/280 ratio of 1.8–2.0 were then electrophoresed on agarose gels to ensure the presence of intact rRNA bands. Methods for cDNA synthesis and qRT-PCR were essentially as described previously [36,72]. An iScript Advanced cDNA synthesis kit and SsoAdvanced universal SYBR green super mix reagents were used for qRT-PCR (Bio-Rad, Hercules, California, USA). The CFX-96 real-time PCR detection system was used for gene expression quantification (Bio-Rad, Hercules, California, USA). Relative gene expression was quantified according to the 2−∆∆CT method [73]. The S. polyrhiza actin (Spipo17G0011400) and 18S rRNA (Spipo23G0000600) genes were used as standard housekeeping genes to normalize the expression of target genes [61]. qRT-PCR data represent the average ± standard deviation from a minimum of three independent biological replicates for each gene. Web-based Primer 3 tool or NCBI Primer-Blast tool was used for primer designing. Each primer sequence was tested with a Blast search in the S. polyrhiza 7498 genome for specific hits. Moreover, each primer pair was tested for its specificity to yield a single amplicon on 1.2% agarose gel. Primers used in this study are listed in Supplementary Table S2.
4.7. Methyl Jasmonate Treatment
Transcript regulation of the S. polyrhiza 7498 LOX gene family in response to MeJA was carried out using a slight modification of an previously published method [35]. MeJA (Millipore, St. Louis, MO, USA, 95%) was diluted 1:10 with 95% ethanol, followed by a further dilution with sterile MilliQ water containing 0.1% Triton X-100, to a final concentration of 10 μM MeJA. Two batches of 14-day grown duckweeds (each batch consisting of 200–300 fronds) were collected from nutrient solution and washed twice with previously autoclaved distilled water. MeJA solution was applied to the plants for 12 h and one batch without MeJA kept as control. Samples were collected in triplicate at time 0, 1, 3, 6, and 12 h post-treatment. A minimum of 10–15 fronds were analyzed for each treatment.
4.8. Salt Treatment
For salt stress treatment, 14-day-old Spirodela polyrhiza 7498 plants were treated with 200 mM salt [36]. A total of 100 fronds were transferred into flasks and then a solution of 200 mM NaCl was added. Control plants were not given the salt treatment. Samples were collected in triplicate at 0, 1, 3, 6, and 12 h post-treatment. A minimum of 10–15 fronds at each time point were sampled and kept frozen in liquid nitrogen and stored at −70 °C until used.
4.9. Data Analysis
The GraphPad (version8.0) suite was used for statistical analysis. ANOVA was performed for significant differences in LOX gene expression. For the MeJA experiment and salt treatment, significant differences were calculated against non-treated control samples at each time point, and categorized at * p < 0.05, ** p < 0.01, and **** p < 0.0001 for each analysis as before [36].
5. Conclusions
Lipoxygenases (LOXs) catalyze synthesis of a group of compounds collectively called oxylipins. Little is known about this family in aquatic plants. In this study, the LOX gene family of the greater duckweed, Spirodela polyrhiza, was identified and characterized by comparative bioinformatics, domain-scan analysis, sequence/phylogenetic analysis, and transcript abundance quantification. Lemna-specific LOX genes were also identified. Jasmonate-mediated changes in expression levels of the Spirodela LOX genes were seen and described. Salt stress identified a different trend of LOX gene expression in the aquatic Spirodela plants as compared with land plants.
Acknowledgments
We thank Shifra Ben-Dor for commenting on the manuscript. We thank Dan Roberts for his support. Mention of trade names or commercial products in this publication is solely for providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Abbreviations
| LOXs | Lipoxygenases |
| PLAT | Polycystin-1, lipoxygenase, alpha-toxin |
| His | Histidine |
| 9-HPOD | 9s-hydroperoxyoctadecadienoic acid |
| 13-HPOD | 13s-hydroperoxyoctadecadienoic acid |
| JA | Jasmonic acid |
| qRT-PCR | Quantitative real-time polymerase chain reaction |
| MeJA | Methyl jasmonate |
Supplementary Materials
The following Supplementary Materials can be found at https://www.mdpi.com/1422-0067/21/24/9527/s1: Supplementary Table S1: Gene sequences from plant species used to deduce evolutionary relatedness with the Spirodela polyrhiza LOX gene family. Supplementary Table S2: List of genes and their primer sequences used for quantitative real-time PCR (qRT-PCR) analysis. Supplementary Table S3: List of genes not considered as true LOXs*. Supplementary Table S4: S. polyrhiza LOXs within three released draft genome sequence. Supplementary Figure S1: Time dependent salt stress transcriptome analysis of LOX gene members in Spirodela polyrhiza.
Author Contributions
Conceived and designed the study: R.K.U.; Computational Bioinformatics: R.K.U.; Performed the experiments: R.K.U.; Analyzed the data: R.K.U.; Funding and reagent availability: A.K.M.; Writing of original draft: R.K.U.; Assisted in analyzing the results and finalized the manuscript: M.E. and A.K.M. All authors approved the final draft. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the in-house ARS Project No. 8042-21000-143-00D to A.K.M.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hillman W.S. The Lemnaceae, or duckweeds. Bot. Rev. 1961;27:221–287. doi: 10.1007/BF02860083. [DOI] [Google Scholar]
- 2.Bog M., Appenroth K.-J., Sree K.S. Duckweed (Lemnaceae): Its Molecular Taxonomy. Front. Sustain. Food Syst. 2019;3:117. doi: 10.3389/fsufs.2019.00117. [DOI] [Google Scholar]
- 3.Landolt E. The Family of Lemnaceae—A Monographic Study (Vol. 1) Volume 71. Geobotanische Institut ETH, Stiftung Rübel; Zurich, Switzerland: 1986. p. 566. [Google Scholar]
- 4.Wang W., Kerstetter R., Michael T. Evolution of genome size in duckweeds (Lemnaceae) J. Bot. 2011;2:1. doi: 10.1155/2011/570319. [DOI] [Google Scholar]
- 5.Mattoo A.K., Pick U., Hoffman-Falk H., Edelman M. The rapidly metabolized 32,000-dalton polypeptide of the chloroplast is the “proteinaceous shield” regulating photosystem II electron transport and mediating diuron herbicide sensitivity. Proc. Natl. Acad. Sci. USA. 1981;78:1572–1576. doi: 10.1073/pnas.78.3.1572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mattoo A.K., Hoffman-Falk H., Marder J.B., Edelman M. Regulation of protein metabolism: Coupling of photosynthetic electron transport to in vivo degradation of the rapidly metabolized 32-kilodalton protein of the chloroplast membranes. Proc. Natl. Acad. Sci. USA. 1984;81:1380–1384. doi: 10.1073/pnas.81.5.1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mattoo A.K., Edelman M. Intramembrane translocation and posttranslational palmitoylation of the chloroplast 32-kDa herbicide-binding protein. Proc. Natl. Acad. Sci. USA. 1987;84:1497–1501. doi: 10.1073/pnas.84.6.1497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ziegler P., Sree K.S., Appenroth K.-J. Duckweeds for water remediation and toxicity testing. Toxicol. Environ. Chem. 2016;98:1127–1154. doi: 10.1080/02772248.2015.1094701. [DOI] [Google Scholar]
- 9.Rusoff L.L., Blakeney E.W., Culley D.D. Duckweeds (Lemnaceae family): A potential source of protein and amino acids. J. Agric. Food Chem. 1980;28:848–850. doi: 10.1021/jf60230a040. [DOI] [PubMed] [Google Scholar]
- 10.Stomp A.-M. Biotechnology Annual Review. Volume 11. Elsevier; Amsterdam, The Netherlands: 2005. The duckweeds: A valuable plant for biomanufacturing; pp. 69–99. [DOI] [PubMed] [Google Scholar]
- 11.Appenroth K.-J., Sree K.S., Fakhoorian T., Lam E. Resurgence of duckweed research and applications: Report from the 3rd International Duckweed Conference. Plant Mol. Biol. 2015;89:647–654. doi: 10.1007/s11103-015-0396-9. [DOI] [PubMed] [Google Scholar]
- 12.Edelman M., Colt M. Nutrient value of leaf vs. seed. Front. Chem. 2016;4:32. doi: 10.3389/fchem.2016.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang W., Haberer G., Gundlach H., Gläßer C., Nussbaumer T., Luo M.C., Lomsadze A., Borodovsky M., Kerstetter R.A., Shanklin J., et al. The Spirodela polyrhiza genome reveals insights into its neotenous reduction fast growth and aquatic lifestyle. Nat. Commun. 2014;5:3311. doi: 10.1038/ncomms4311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Van Hoeck A., Horemans N., Monsieurs P., Cao H.X., Vandenhove H., Blust R. The first draft genome of the aquatic model plant Lemna minor opens the route for future stress physiology research and biotechnological applications. Biotechnol. Biofuels. 2015;8:188. doi: 10.1186/s13068-015-0381-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.An D., Li C., Zhou Y., Wu Y., Wang W. Genomes and Transcriptomes of Duckweeds. Front. Chem. 2018;6 doi: 10.3389/fchem.2018.00230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hoang P.N.T., Michael T.P., Gilbert S., Chu P., Motley S.T., Appenroth K.J., Schubert I., Lam E. Generating a high-confidence reference genome map of the Greater Duckweed by integration of cytogenomic, optical mapping, and Oxford Nanopore technologies. Plant J. 2018;96:670–684. doi: 10.1111/tpj.14049. [DOI] [PubMed] [Google Scholar]
- 17.An D., Zhou Y., Li C., Xiao Q., Wang T., Zhang Y., Wu Y., Li Y., Chao D.-Y., Messing J., et al. Plant evolution and environmental adaptation unveiled by long-read whole-genome sequencing of Spirodela. Proc. Natl. Acad. Sci. USA. 2019;116:18893–18899. doi: 10.1073/pnas.1910401116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wasternack C. Jasmonates: An Update on Biosynthesis, Signal Transduction and Action in Plant Stress Response, Growth and Development. Ann. Bot. 2007;100:681–697. doi: 10.1093/aob/mcm079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wasternack C., Song S. Jasmonates: Biosynthesis, metabolism, and signaling by proteins activating and repressing transcription. J. Exp. Bot. 2017;68:1303–1321. doi: 10.1093/jxb/erw443. [DOI] [PubMed] [Google Scholar]
- 20.Wasternack C., Feussner I. The oxylipin pathways: Biochemistry and function. Annu. Rev. Plant Biol. 2018;69:363–386. doi: 10.1146/annurev-arplant-042817-040440. [DOI] [PubMed] [Google Scholar]
- 21.Andre E., Hou K. The lipoxydases of seeds of Glycine soja (Sieb) and Phaseolus vulgaris (L.) CR Hebd. Seances. Acad. Sci. 1932;195:172–174. [Google Scholar]
- 22.Siedow J.N. Plant lipoxygenase: Structure and function. Annu. Rev. Plant Biol. 1991;42:145–188. doi: 10.1146/annurev.pp.42.060191.001045. [DOI] [Google Scholar]
- 23.Porta H., Rocha-Sosa M. Plant lipoxygenases. Physiological and molecular features. Plant Physiol. 2002;130:15–21. doi: 10.1104/pp.010787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liavonchanka A., Feussner I. Lipoxygenases: Occurrence, functions and catalysis. J. Plant Physiol. 2006;163:348–357. doi: 10.1016/j.jplph.2005.11.006. [DOI] [PubMed] [Google Scholar]
- 25.Rance I., Fournier J., Esquerre-Tugaye M.-T. The incompatible interaction between Phytophthora parasitica var. nicotianae race 0 and tobacco is suppressed in transgenic plants expressing antisense lipoxygenase sequences. Proc. Natl. Acad. Sci. USA. 1998;95:6554–6559. doi: 10.1073/pnas.95.11.6554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Baysal T., Demirdöven A. Lipoxygenase in fruits and vegetables: A review. Enzym. Microb. Technol. 2007;40:491–496. doi: 10.1016/j.enzmictec.2006.11.025. [DOI] [Google Scholar]
- 27.Hayward S., Cilliers T., Swart P. Lipoxygenases: From Isolation to Application. Compr. Rev. Food Sci. Food Saf. 2017;16:199–211. doi: 10.1111/1541-4337.12239. [DOI] [PubMed] [Google Scholar]
- 28.Bell E., Creelman R.A., Mullet J.E. A chloroplast lipoxygenase is required for wound-induced jasmonic acid accumulation in Arabidopsis. Proc. Natl. Acad. Sci. USA. 1995;92:8675–8679. doi: 10.1073/pnas.92.19.8675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chauvin A., Caldelari D., Wolfender J.-L., Farmer E.E. Four 13-lipoxygenases contribute to rapid jasmonate synthesis in wounded Arabidopsis thaliana leaves: A role for lipoxygenase 6 in responses to long-distance wound signals. New Phytol. 2013;197:566–575. doi: 10.1111/nph.12029. [DOI] [PubMed] [Google Scholar]
- 30.Griffiths A., Barry C., Alpuche-Solis A.G., Grierson D. Ethylene and developmental signals regulate expression of lipoxygenase genes during tomato fruit ripening. J. Exp. Bot. 1999;50:793–798. doi: 10.1093/jxb/50.335.793. [DOI] [Google Scholar]
- 31.Kausch K.D., Handa A.K. Molecular Cloning of a Ripening-Specific Lipoxygenase and Its Expression during Wild-Type and Mutant Tomato Fruit Development. Plant Physiol. 1997;113:1041–1050. doi: 10.1104/pp.113.4.1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kausch K.D., Sobolev A.P., Goyal R.K., Fatima T., Laila-Beevi R., Saftner R.A., Handa A.K., Mattoo A.K. Methyl jasmonate deficiency alters cellular metabolome, including the aminome of tomato (Solanum lycopersicum L.) fruit. Amino Acids. 2012;42:843–856. doi: 10.1007/s00726-011-1000-5. [DOI] [PubMed] [Google Scholar]
- 33.Yan L., Zhai Q., Wei J., Li S., Wang B., Huang T., Du M., Sun J., Kang L., Li C.-B., et al. Role of Tomato Lipoxygenase D in Wound-Induced Jasmonate Biosynthesis and Plant Immunity to Insect Herbivores. PLoS Genet. 2013;9:e1003964. doi: 10.1371/journal.pgen.1003964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shen J., Tieman D., Jones J.B., Taylor M.G., Schmelz E., Huffaker A., Bies D., Chen K., Klee H.J. A 13-lipoxygenase, TomloxC, is essential for synthesis of C5 flavour volatiles in tomato. J. Exp. Bot. 2014;65:419–428. doi: 10.1093/jxb/ert382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Upadhyay R.K., Mattoo A.K. Genome-wide identification of tomato (Solanum lycopersicum L.) lipoxygenases coupled with expression profiles during plant development and in response to methyl-jasmonate and wounding. J. Plant Physiol. 2018;231:318–328. doi: 10.1016/j.jplph.2018.10.001. [DOI] [PubMed] [Google Scholar]
- 36.Upadhyay R.K., Handa A.K., Mattoo A.K. Transcript Abundance Patterns of 9- and 13-Lipoxygenase Subfamily Gene Members in Response to Abiotic Stresses (Heat, Cold, Drought or Salt) in Tomato (Solanum lycopersicum L.) Highlights Member-Specific Dynamics Relevant to Each Stress. Genes. 2019;10:683. doi: 10.3390/genes10090683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shin J.H., Van K., Kim D.H., Kim K.D., Jang Y.E., Choi B.-S., Kim M.Y., Lee S.-H. The lipoxygenase gene family: A genomic fossil of shared polyploidy between Glycine max and Medicago truncatula. BMC Plant Biol. 2008;8:133. doi: 10.1186/1471-2229-8-133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bannenberg G., Martínez M., Hamberg M., Castresana C. Diversity of the Enzymatic Activity in the Lipoxygenase Gene Family of Arabidopsis thaliana. Lipids. 2009;44:85. doi: 10.1007/s11745-008-3245-7. [DOI] [PubMed] [Google Scholar]
- 39.Podolyan A., White J., Jordan B., Winefield C. Identification of the lipoxygenase gene family from Vitis vinifera and biochemical characterisation of two 13-lipoxygenases expressed in grape berries of Sauvignon Blanc. Funct. Plant Biol. 2010;37:767–784. doi: 10.1071/FP09271. [DOI] [Google Scholar]
- 40.Liu S.Q., Liu X.H., Jiang L.W. Genome-wide identification, phylogeny and expression analysis of the lipoxygenase gene family in cucumber. Genet. Mol. Res. 2011;10:2613–2636. doi: 10.4238/2011.October.25.9. [DOI] [PubMed] [Google Scholar]
- 41.Umate P. Genome-wide analysis of lipoxygenase gene family in Arabidopsis and rice. Plant Signal. Behav. 2011;6:335–338. doi: 10.4161/psb.6.3.13546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vogt J., Schiller D., Ulrich D., Schwab W., Dunemann F. Identification of lipoxygenase (LOX) genes putatively involved in fruit flavour formation in apple (Malus × domestica) Tree Genet. Genomes. 2013;9:1493–1511. doi: 10.1007/s11295-013-0653-5. [DOI] [Google Scholar]
- 43.Li M., Li L., Dunwell J.M., Qiao X., Liu X., Zhang S. Characterization of the lipoxygenase (LOX) gene family in the Chinese white pear (Pyrus bretschneideri) and comparison with other members of the Rosaceae. BMC Genom. 2014;15:444. doi: 10.1186/1471-2164-15-444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chen Z., Chen X., Yan H., Li W., Li Y., Cai R., Xiang Y. The Lipoxygenase Gene Family in Poplar: Identification, Classification, and Expression in Response to MeJA Treatment. PLoS ONE. 2015;10:e0125526. doi: 10.1371/journal.pone.0125526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Song H., Wang P., Li C., Han S., Lopez-Baltazar J., Zhang X., Wang X. Identification of lipoxygenase (LOX) genes from legumes and their responses in wild type and cultivated peanut upon Aspergillus flavus infection. Sci. Rep. 2016;6:35245. doi: 10.1038/srep35245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sarde S.J., Kumar A., Remme R.N., Dicke M. Genome-wide identification, classification and expression of lipoxygenase gene family in pepper. Plant Mol. Biol. 2018;98:375–387. doi: 10.1007/s11103-018-0785-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Shaban M., Ahmed M.M., Sun H., Ullah A., Zhu L. Genome-wide identification of lipoxygenase gene family in cotton and functional characterization in response to abiotic stresses. BMC Genom. 2018;19:599. doi: 10.1186/s12864-018-4985-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhu J., Wang X., Guo L., Xu Q., Zhao S., Li F., Yan X., Liu S., Wei C. Characterization and Alternative Splicing Profiles of the Lipoxygenase Gene Family in Tea Plant (Camellia sinensis) Plant Cell Physiol. 2018;59:1765–1781. doi: 10.1093/pcp/pcy091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wang J., Hu T., Wang W., Hu H., Wei Q., Wei X., Bao C. Bioinformatics Analysis of the Lipoxygenase Gene Family in Radish (Raphanus sativus) and Functional Characterization in Response to Abiotic and Biotic Stresses. Int. J. Mol. Sci. 2019;20:6095. doi: 10.3390/ijms20236095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Michael T.P., Bryant D., Gutierrez R., Borisjuk N., Chu P., Zhang H., Xia J., Zhou J., Peng H., Baidouri M.E., et al. Comprehensive definition of genome features in Spirodela polyrhiza by high-depth physical mapping and short-read DNA sequencing strategies. Plant J. 2017;89:617–635. doi: 10.1111/tpj.13400. [DOI] [PubMed] [Google Scholar]
- 51.Sharma M., Laxmi A. Jasmonates: Emerging Players in Controlling Temperature Stress Tolerance. Front. Plant Sci. 2016;6 doi: 10.3389/fpls.2015.01129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Edelman M., Mattoo A. The D1 protein: Past and future. In: Demmig-Adams B., Adams W., Mattoo A.K., editors. Photoprotection, Photoinhibition, Gene Regulation and Environment. Springer; Dordecht, The Netherlands: 2006. pp. 23–38. [Google Scholar]
- 53.Wasternack C., Hause B. Jasmonates: Biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann. Bot. 2013;111:1021–1058. doi: 10.1093/aob/mct067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Krajnčič B., Kristl J., Janžekovič I. Possible role of jasmonic acid in the regulation of floral induction, evocation and floral differentiation in Lemna minor L. Plant Physiol. Biochem. 2006;44:752–758. doi: 10.1016/j.plaphy.2006.10.029. [DOI] [PubMed] [Google Scholar]
- 55.Appenroth K.-J., Dathe W., Hertel W., Augsten H. Photophysiology of Turion Germination in Spirodela polyrhiza (L.) SCHLEIDEN. VII. Action of Jasmonic acid. J. Plant Physiol. 1991;138:345–349. doi: 10.1016/S0176-1617(11)80298-1. [DOI] [PubMed] [Google Scholar]
- 56.Lu Q., Zhang T., Zhang W., Su C., Yang Y., Hu D., Xu Q. Alleviation of cadmium toxicity in Lemna minor by exogenous salicylic acid. Ecotoxicol. Environ. Saf. 2018;147:500–508. doi: 10.1016/j.ecoenv.2017.09.015. [DOI] [PubMed] [Google Scholar]
- 57.De Domenico S., Taurino M., Gallo A., Poltronieri P., Pastor V., Flors V., Santino A. Oxylipin dynamics in Medicago truncatula in response to salt and wounding stresses. Physiol. Plant. 2019;165:198–208. doi: 10.1111/ppl.12810. [DOI] [PubMed] [Google Scholar]
- 58.Zhang H., Zhang Q., Zhai H., Li Y., Wang X., Liu Q., He S. Transcript profile analysis reveals important roles of jasmonic acid signalling pathway in the response of sweet potato to salt stress. Sci. Rep. 2017;7:1–12. doi: 10.1038/srep40819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Jiang M., Xu F., Peng M., Huang F., Meng F. Methyl jasmonate regulated diploid and tetraploid black locust (Robinia pseudoacacia L.) tolerance to salt stress. Acta Physiol. Plant. 2016;38:106. doi: 10.1007/s11738-016-2120-z. [DOI] [Google Scholar]
- 60.Faghih S., Ghobadi C., Zarei A. Response of strawberry plant cv. ‘Camarosa’ to salicylic acid and methyl jasmonate application under salt stress condition. J. Plant Growth Regul. 2017;36:651–659. doi: 10.1007/s00344-017-9666-x. [DOI] [Google Scholar]
- 61.Fu L., Ding Z., Sun X., Zhang J. Physiological and Transcriptomic Analysis Reveals Distorted Ion Homeostasis and Responses in the Freshwater Plant Spirodela polyrhiza L. under Salt Stress. Genes. 2019;10:743. doi: 10.3390/genes10100743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Mattoo A.K., Mehta R.A., Baker J.E. Copper-induced ethylene biosynthesis in terrestrial (Nicotiana tabacum) and aquatic (Spirodela oligorrhiza) higher plants. Phytochemistry. 1992;31:405–409. doi: 10.1016/0031-9422(92)90006-C. [DOI] [Google Scholar]
- 63.Finn R.D., Clements J., Eddy S.R. HMMER web server: Interactive sequence similarity searching. Nucleic Acids Res. 2011;39:W29–W37. doi: 10.1093/nar/gkr367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Quevillon E., Silventoinen V., Pillai S., Harte N., Mulder N., Apweiler R., Lopez R. InterProScan: Protein domains identifier. Nucleic Acids Res. 2005;33:W116–W120. doi: 10.1093/nar/gki442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Edgar R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004;32:1792–1797. doi: 10.1093/nar/gkh340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zuckerkandl E., Pauling L. Evolutionary Divergence and Convergence in Proteins. In: Bryson V., Vogel H.J., editors. Evolving Genes and Proteins. Academic Press; Cambridge, MA, USA: 1965. pp. 97–166. [Google Scholar]
- 67.Felsenstein J. Confidence Limits on Phylogenies: An Approach Using the Bootstrap. Evolution. 1985;39:783–791. doi: 10.1111/j.1558-5646.1985.tb00420.x. [DOI] [PubMed] [Google Scholar]
- 68.Kumar S., Stecher G., Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016;33:1870–1874. doi: 10.1093/molbev/msw054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bailey T.L., Boden M., Buske F.A., Frith M., Grant C.E., Clementi L., Ren J., Li W.W., Noble W.S. MEME SUITE: Tools for motif discovery and searching. Nucleic Acids Res. 2009;37:W202–W208. doi: 10.1093/nar/gkp335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yang X., Kalluri U.C., Jawdy S., Gunter L.E., Yin T., Tschaplinski T.J., Weston D.J., Ranjan P., Tuskan G.A. The F-Box Gene Family Is Expanded in Herbaceous Annual Plants Relative to Woody Perennial Plants. Plant Physiol. 2008;148:1189–1200. doi: 10.1104/pp.108.121921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sharp A.J., Locke D.P., McGrath S.D., Cheng Z., Bailey J.A., Vallente R.U., Pertz L.M., Clark R.A., Schwartz S., Segraves R., et al. Segmental Duplications and Copy-Number Variation in the Human Genome. Am. J. Hum. Genet. 2005;77:78–88. doi: 10.1086/431652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Bustin S.A., Benes V., Garson J.A., Hellemans J., Huggett J., Kubista M., Mueller R., Nolan T., Pfaffl M.W., Shipley G.L., et al. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009;55:611–622. doi: 10.1373/clinchem.2008.112797. [DOI] [PubMed] [Google Scholar]
- 73.Livak K.J., Schmittgen T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.