Abstract
The joint activity of multiple engineered nanoparticles (ENPs) has attracted much attention in recent years. Many previous studies have focused on the combined toxicity of different ENPs with nanostructures of the same dimension. However, the mixture toxicity of multiple ENPs with different dimensions is much less understood. Herein, we investigated the toxicity of the binary mixture of TiO2 nanospherical particles (NPs) and TiO2 nanotubes (NTs) to two freshwater algae with different morphology, namely, Scenedesmus obliquus and Chlorella pyrenoidosa. The physicochemical properties, dispersion stability, and the generation of reactive oxygen species (ROS) were determined in the single and binary systems. Classical approaches to assessing mixture toxicity were applied to evaluate and predict the toxicity of the binary mixtures. The results show that the combined toxicity of TiO2 NPs and NTs to S. obliquus was between the single toxicity of TiO2 NTs and NPs, while the combined toxicity to C. pyrenoidosa was higher than their single toxicity. Moreover, the toxicity of the binary mixtures to C. pyrenoidosa was higher than that to S. obliquus. A toxic unit assessment showed that the effects of TiO2 NPs and NTs were additive to the algae. The combined toxicity to S. obliquus and C. pyrenoidosa can be effectively predicted by the concentration addition model and the independent action model, respectively. The mechanism of the toxicity caused by the binary mixtures of TiO2 NPs and NTs may be associated with the dispersion stability of the nanoparticles in aquatic media and the ROS-induced oxidative stress effects. Our results may offer a new insight into evaluating and predicting the combined toxicological effects of ENPs with different dimensions and of probing the mechanisms involved in their joint toxicity.
Keywords: TiO2 nanoparticles, TiO2 nanotubes, nanotoxicity, freshwater algae, oxidative damage
1. Introduction
In the past decade, research on the health and environmental risk of engineered nanoparticles (ENPs) has increased considerably [1,2,3]. The requests for toxicity data on the effects of ENPs on organisms continue to grow. Most of the toxicity data are derived from a single toxicity [4,5]. In the natural environment, organisms exposed to a mixture of multiple contaminants (n ≥ 2) rather than individual ones is a universal law [6,7]. Many studies have addressed the toxic effects of a mixture of ENPs and other contaminants, e.g., nano-TiO2 and tetracycline [8], nano-TiO2 and hexavalent chromium [9], as well as nano-TiO2 and bisphenol [10]. However, researchers in the field of nanotoxicology have just started to investigate the combined toxic effects of multiple exposures to ENPs.
The existing literature on the combined toxicity of ENPs mainly focus on the modes of joint action (MOJA) [11,12,13,14], which include antagonistic, synergistic, and addition. Moreover, the toxicity effects and MOJA of ENP mixtures are associated with the ENP components and test organisms. Most previous studies have selected ENPs with different components, but their nanostructures have the same dimensions. A few studies have indicated that graphene oxide as a two-dimensional ENP can affect the toxicity of concomitant zinc oxide nanospherical particles (NPs) to aquatic species [15] and human cells [16]. Therefore, toxicological studies on ENPs mixtures are urgently needed to reveal the combined effects induced by ENPs with different dimensional structures.
Nano-TiO2 is one of the most promising ENPs [17], and it is described as the “star” of many ENPs. Compared with TiO2 NPs with zero-dimension, one-dimensional TiO2 nanotubes (NTs) display enhanced excellent properties such as a larger diameter, higher specific surface area, and so on. Thus, the preparation and application of TiO2 NTs have provoked great interest among scientific researchers [18,19]. Freshwater algae, like Scenedesmus obliquus [20,21] and Chlorella pyrenoidosa [22,23] are frequently chosen as a model organism to assess the aquatic toxicity of ENPs, especially as S. obliquus and C. pyrenoidosa have a different morphology, namely, they are flat- and spherical-shaped, respectively. These characteristics mean that these two algae show a distinct sensitivity to ENPs.
In the present study, we investigated the toxicity of TiO2 NPs, TiO2 NTs and the binary mixtures of TiO2 NPs and NTs, to S. obliquus and C. pyrenoidosa. The four main objectives were: (1) to determine the physicochemical properties and to evaluate the stability of TiO2 NPs, TiO2 NTs and TiO2 NPs + NTs in a freshwater model; (2) to investigate the growth inhibition toxicity of TiO2 NPs, TiO2 NTs and the TiO2 NPs + NTs to the algae; (3) to assess the MOJA of TiO2 NPs and NTs and to predict their joint toxicity using the concentration addition (CA) and independent action (IA) models; and (4) to explore the mechanism of cytotoxicity induced by TiO2 NPs, TiO2 NTs, and TiO2 NPs + NTs by determining the generation of reactive oxygen species (ROS).
2. Methods
2.1. Test Material and Test Medium
TiO2 NPs with a diameter of 21 ± 10 nm (powder, P25 grade) and TiO2 NTs with a diameter of 10 nm and a length of 1 μm (wet cake, liquid: water 9.5% wt) were purchased from PlasmaChem GmbH (Berlin, Germany) and Nanjing XFNANO Materials Tech Co. Ltd. (Nanjing, China), respectively. The stock suspensions of the test materials were freshly prepared in ultra-high pure water after 30 min sonication in a water bath sonicator and then stored at 4 °C until use. Algae culture medium was prepared as a test medium at pH 7.8 ± 0.2 according to Organization for Economic Co-operation and Development (OECD) guidelines [24].
2.2. Physicochemical Analysis
The morphology of the single and combined test materials in the test medium was characterized by a transmission electron microscope (TEM, JOEL 2100f, JOEL Ltd., Tokyo, Japan). Zeta potential (ZP) and hydrodynamic diameters (HD) of the particle suspensions at 5 mg/L were analyzed at 0 h and 96 h in the test medium using a ZetaSizer instrument (Nano ZS90, Malvern Instruments Ltd., Worcestershire, UK). The ZP and HD measurements were performed in three independent experiments, and the data presented are the mean of the runs. Based on classical DLVO theory [25], the stability of a particle in the test medium was determined by simulating the total potential energy for interactions between the TiO2 particles.
2.3. Algal Growth Assays
The unicellular freshwater algae S. obliquus and C. pyrenoidosa were obtained from the Chinese Academy of Sciences Institute of Hydrobiology (Wuhan, China). Exponentially growing algae cells (with a final density of 3 × 105 cells/mL for S. obliquus and 4 × 105 cells/mL for C. pyrenoidosa) were added to the control and treated experiments. Internal control experiments were required to eliminate the absorbance effects of the test materials. All flasks containing various treatments were incubated in an artificial growth chamber at a consistent temperature of 24 ± 1 °C for 96 h with a photoperiod of 12-h light (3000–4000 l×) and 12-h dark. The algal cell density was determined using an ultraviolet-visible spectrophotometer (UV1102; Shanghai Tian Mei Scientific Instrument Co., Shanghai, China) after 96 h for S. obliquus and 72 h for C. pyrenoidosa to provide cell numbers and allow the specific growth rate to be calculated. Growth inhibition (%) was calculated by dividing the specific growth rate for a treatment by the mean specific growth rate for the controls. Three replicates were included for each treatment and the data presented are the mean of the runs (n = 3).
2.4. Oxidative Stress Biomarker Assays
2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) purchased from Macklin Biochemical Co., Ltd. (Shanghai, China) was used as a fluorescent probe to measure the intracellular ROS. The 96 h (S. obliquus) and 72 h (C. pyrenoidosa) algal cell suspensions were centrifuged at 15,000 rpm for 10 min at 25 °C (using a D3024 high speed micro-centrifuge (Scilogex, Rocky Hill, CT, USA). After discarding the supernatant, 10 μM DCFH-DA was incubated with algal cells for 30 min under dark conditions at 25 °C. Subsequently, the samples were centrifuged under the same conditions and washed one time with the test medium to remove the loosely bound fluorescent probe. Each test concentration was replicated two times in an independent experiment.
When intracellular ROS are generated, 2′,7′-dichlorofluorescein (DCF) can be converted from DCFH, which is obtained by lipase decomposing DCFH-DA in cells [26]. Thus, the fluorescence intensity (FI) of DCF indicates the extent of intracellular ROS generation. FI was measured using a fluorospectrophotometer (F96PRO, Shanghai Kingdak Scientific Instrument Co., Ltd., Zhejiang, China). The excitation and emission wavelength for the optical measurements were based on previous studies [15,27]. Each sample was measured three times. Data were expressed as a percentage (%) of the fluorescence or the absorbance of the control cells according to the equation:
| %F = 100 − [100(Fc − Ft)/Fc] | (1) |
where %F is the percentage of fluorescence of algal cells; Fc is the mean fluorescence of control cells; and Ft is the mean fluorescence of treated cells.
2.5. Assessment and Prediction for Mixture Toxicity
The logistic model (Equation (2)) was used to fit concentration-response curves (CRCs) for TiO2 NPs, TiO2 NTs, and TiO2 NPs + NTs. The effect concentrations, such as the 10% effect concentration (EC10) and median effect concentration (EC50) of each treatment group were derived from the CRCs. The combined toxicity tests followed the mixture ratios of the effect concentrations of TiO2 NPs and NTs at the same effect level.
| (2) |
where C is the test material’s concentration and θ represents the slope parameter.
The EC50 value was used to calculate a toxic unit (TU), as shown in Equations (3) and (4),
| (3) |
| (4) |
where Ci is the concentration of material i and TUTotal is the sum of the TiO2 NP and NT TUs. Therefore, one TU corresponded to the EC50. The MOJA of TiO2 NPs and NTs were evaluated by plotting the observed TU (TUobs) derived from the toxicity data against the expected TU (TUexp), as described by Kim et al. [28].
The total concentration of a mixture provoking an x% effect (ECxmix) was calculated from the CRCs of the individual components using the CA model, as shown in Equation 5.
| (5) |
where Pi is the fraction of component i in the mixture and ECxi is the concentration of component i that would, when applied singly, provoke the x% effect.
The general equation shown in Equation (6) was used for the IA model,
| (6) |
where E(Cmix) is the effect expected at the total concentration of the mixture (scaled to between 0% and 100%) and E(Ci) is the effect that the ith mixture component would provoke if applied singly at concentration Ci.
2.6. Statistical Analysis
All data are expressed as means ± standard deviation (SD). Statistically significant differences between the test treatments were determined by a t-test at significance levels of p < 0.05, p < 0.01, and p < 0.001.
3. Results and Discussion
3.1. Physicochemical Characterizations
The TEM images are shown in Figure 1. The individual TiO2 NPs and NTs were spherical (Figure 1A) and tubular (Figure 1B) particles, respectively. Additionally, the TEM images show that the TiO2 NPs agglomerated heavily in the test medium, whereas the TiO2 NTs agglomerated slightly. As can be seen from Figure 1C, the morphology of the two TiO2 particles with different shapes in the binary mixture systems was not affected by each other.
Figure 1.
TEM images of the TiO2 NPs (A), TiO2 NTs (B), and TiO2 NPs + NTs (C) in the algae medium.
To characterize the change in the physicochemical properties of TiO2 particles, the ZP and HD values of TiO2 NPs and NTs from single to binary mixtures were measured in the test medium (Table 1). The ZP value of TiO2 NPs at 96 h were significantly increased compared with the ZP value of TiO2 NPs at 0 h (p < 0.01). It was found that there was no obvious change in the ZP value of TiO2 NTs or TiO2 NPs + NTs between 0 h and 96 h (p > 0.05). The measurement of HD showed that the of the TiO2 particles became smaller in size over a 96-h period. Furthermore, the size of the particles in TiO2 NPs + NTs was significantly decreased during 96 h of exposure (p < 0.01). The reason for the reduction in particle size may be due to the sedimentation of large particles.
Table 1.
Zeta potential (ZP) and hydrodynamic diameter (HD) ± standard deviation (n = 3) of the test materials from single to binary mixture systems.
| Test Materials | 0 h | 96 h | ||
|---|---|---|---|---|
| ZP (mV) | HD (nm) | ZP (mV) | HD (nm) | |
| TiO2 NPs | −14.4 ± 0.2 | 969 ± 357 | −13.2 ± 0.3 | 643 ± 84.5 |
| TiO2 NTs | −12.5 ± 1.0 | 748 ± 201 | −13.7 ± 2.3 | 681 ± 94.5 |
| TiO2 NPs + NTs | −12.8 ± 0.5 | 943 ± 99 | −12.9 ± 0.4 | 477 ± 21.1 |
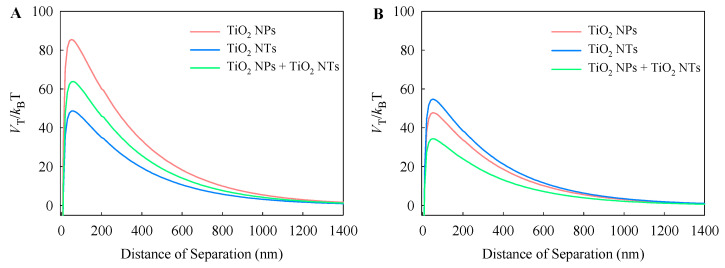
To further evaluate the agglomeration and stability of the TiO2 particles in the test medium, the total potential energy profiles of the TiO2 particles in the single and binary mixture systems at the end of different intervals were calculated using the DLVO theory (Figure 2). At 0 h, the peak values of the total potential energy profiles decreased in the order: TiO2 NPs > TiO2 NPs + NTs > TiO2 NTs (Figure 2A). This means that the TiO2 NPs showed the highest stability in the test medium compared to the other studied systems. However, over 96 h, the stability of TiO2 NPs and TiO2 NPs + NTs obviously decreased, while the stability of TiO2 NTs enhanced slightly (Figure 2B). As mentioned above, the particle size of TiO2 particles in the test medium became smaller with time. Taken together, this suggests that the TiO2 particles were settling due to particle agglomeration.
Figure 2.
Total potential energy (VT) curves for the TiO2 particles in the single and binary systems at 0 h (A) and 96 h (B).
3.2. Toxicity of Single and Mixtures of TiO2 NPs to Algal
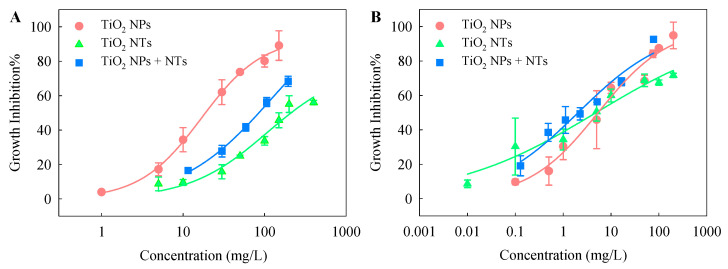
Typical CRCs were observed for the toxic effects of TiO2 NPs, TiO2 NTs, and TiO2 NPs + NTs on the two test species (Figure 3). The effect concentrations determined by the CRCs are listed in Table 2. The CRC analysis indicated a concentration-dependent variation in the individual and combined toxic effects of TiO2 NPs and NTs. For S. obliquus, the CRC for TiO2 NPs was distant from the CRC for TiO2 NTs and started at lower concentrations (Figure 3A). Moreover, the EC10 and EC50 values of TiO2 NPs were lower than those of TiO2 NTs, suggesting that the single toxicity of TiO2 NPs to the algae was higher than TiO2 NTs. Some previous studies have also indicated that the shape of the ENPs is a significant factor in determining the potency and magnitude of the toxicity effect on organisms [29,30,31]. The CRC for TiO2 NPs + NTs was in between that for TiO2 NPs and that for TiO2 NTs. Similarly, the EC50 value derived from the CRC for TiO2 NPs + NTs was between the EC50 value of each components in the binary mixtures. As mentioned above, the stability of TiO2 NPs + NTs in the test medium at the initial time was also between that of TiO2 NPs and that of TiO2 NTs. The findings for the growth inhibition toxicity to S. obliquus combined with the findings for the stability indicate that the higher the initial stability, the stronger the toxicity. Dispersion of ENPs has received special research attention because the environmental behavior and effects of ENPs are greatly dependent on their dispersion status [32]. Previous studies have also suggested that nano-TiO2 aggregates can reduce the light available to the entrapped algal cells and thus inhibits their growth [33,34]. As mentioned above, the TiO2 particles agglomerated under this study. This also means that the agglomeration of particles contributed to the overall growth inhibition toxicity to some degree.
Figure 3.
Concentration-response curves for Scenedesmus obliquus (A) and Chlorella pyrenoidosa (B) exposed to TiO2 NPs, TiO2 NTs, and TiO2 NPs + NTs.
Table 2.
Effect concentrations derived from concentration-response curves of the single nanoparticles and the mixtures a.
| Materials | Scenedesmus obliquus | Chlorella pyrenoidosa | |||
|---|---|---|---|---|---|
| Single Toxicity | EC10 (mg/L) | EC50 (mg/L) | EC10 (mg/L) | EC50 (mg/L) | |
| TiO2 NPs | 2.33 | 19.75 | 0.13 | 5.38 | |
| [1.75–3.44] | [17.02–23.00] | [0.11–0.14] | [3.36–8.23] | ||
| TiO2 NTs | 13.16 | 211.26 | 0.002 | 4.87 | |
| [5.08–25.75] | [152.01–427.73] | [0.002–0.004] | [2.81–9.66] | ||
| Combined Toxicity | EC50 (mg/L) | EC50 (mg/L) | |||
| TiO2
NPs + NTs |
OBS | 85.04 [69.50–102.54] | 2.05 [1.16–3.88] | ||
| IA | 56.30 [56.17–56.42] | 1.08 [0.91–1.34] | |||
| CA | 99.46 [99.09–99.81] | 5.96 [4.97–7.36] | |||
a EC10/EC50 = 10%/mean effect concentration. The 95% two-sided confidence interval is shown in []; OBS = observed combined toxicity; IA = independent action; CA = concentration addition.
For C. pyrenoidosa, the CRC for TiO2 NPs crossed over the CRC for TiO2 NTs (Figure 3B). The EC10 value of TiO2 NPs was higher than that of TiO2 NTs. The EC50 value of TiO2 NPs was slightly higher than that of TiO2 NTs. However, as can be seen from the CRCs, the effect concentrations of TiO2 NPs were lower than those of TiO2 NTs, as the observed effects gradually increased. This means that the differences in the single toxicity of TiO2 NPs and NTs depend on the exposure concentration. Moreover, in the higher exposure concentration range, TiO2 NPs with the higher initial stability showed more toxicity than TiO2 NTs with the lower initial stability. The CRC for TiO2 NPs + NTs intercrossed the other two CRCs and decreased slightly with lower concentrations. The EC50 value derived from the CRC for TiO2 NPs + NTs was lower than that of TiO2 NPs and that of TiO2 NTs. This implies that the joint toxicity of TiO2 NPs and NTs to C. pyrenoidosa was higher than the single toxicity of each component in the binary mixtures.
It was also found that the EC10 and EC50 values of TiO2 NPs, TiO2 NTs, and TiO2 NPs + NTs to C. pyrenoidosa were lower than those to S. obliquus, which implies that the single and binary mixtures exhibited stronger toxicity to C. pyrenoidosa than to S. obliquus. This finding reveals that C. pyrenoidosa is more sensitive to the TiO2 particles than S. obliquus. S. obliquus is usually composed of four cells that are 12–34 μm wide and 10–21 μm long, with a flat shape. C. pyrenoidosa has a spherical shape with a diameter of 3–5 μm. The cells of C. pyrenoidosa are smaller but have a higher specific surface area than those of S. obliquus. This feature allows for a more effective particle uptake by C. pyrenoidosa. Furthermore, our previous study indicated the cell membrane permeability of C. pyrenoidosa was significantly increased after ENP stimulation, compared with the control and S. obliquus [35]. Consequently, the TiO2 particles might interrupt the cell membrane functions of C. pyrenoidosa to a higher degree, and thus trigger more severe growth inhibition toxicity.
3.3. Assessment and Prediction of Joint Toxicity of TiO2 NPs and NTs
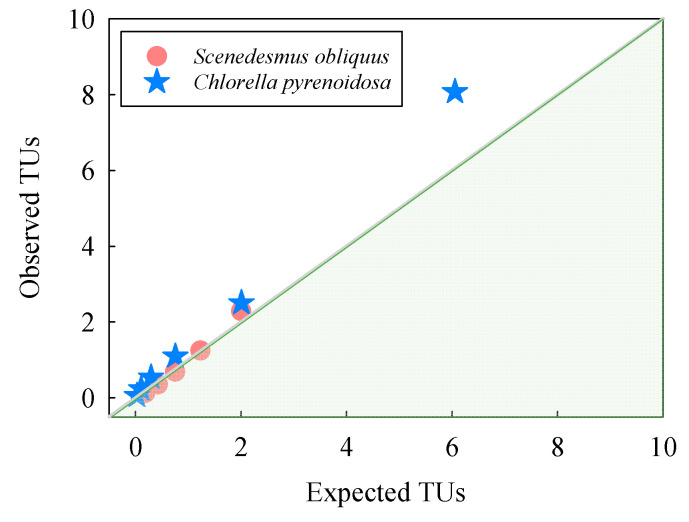
The observed toxicities were converted to TUs and plotted against the expected TUtotal values of the binary mixtures of TiO2 NPs and NTs, as calculated from the sums of the individual TiO2 NPs and NTs. As depicted in Figure 4, the observed TUtotal,obs are almost equal to the expected TUs, indicating that the TiO2 NPs + NTs mixture effects were additive. However, for C. pyrenoidosa, the observed TUtotal,obs of TiO2 NPs + NTs at the highest concentration under this study was obviously higher than the expected TUs, indicating that the joint toxicity was synergistic at higher concentrations of the mixtures.
Figure 4.
Observed toxic units (TUs) as converted from the toxicity data of Scenedesmus obliquus and Chlorella pyrenoidosa following exposure to mixtures of TiO2 NPs and NTs, and subsequently plotted against expected TUs calculated on the basis of the median effect concentrations from the individual constituents present in the mixtures.
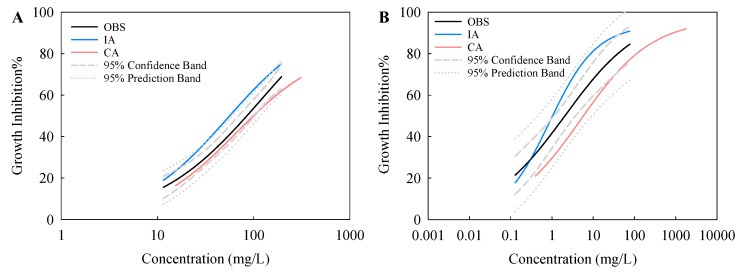
The classical methods, namely, CA and IA, were used to quantitatively assess and predict the combined effects of ENPs [36,37]. The differences between the experimental and predicted joint toxicities to S. obliquus and C. pyrenoidosa are shown in Figure 5 and Table 2. For S. obliquus, the CRC derived from the CA model slightly deviated from the observed CRC, while the CRC derived from the IA model seriously deviated from the observed CRC (Figure 5A). For a direct graphical assessment of the whole concentration-response range, we also depicted the 95% confidence band (CB) and prediction band (PB) of the experimental data points. The CA prediction almost overlapped with the CB of the observed concentration-response data, implying that the CA model showed good predictive quality over the widest range of effects. However, except for the lower effect regions (about <40%), the IA prediction was outside the PB range of the observed concentration-response data. This also means that there was a big difference between the observation and the prediction for the IA model. As can be seen from Table 2, the EC50 value predicted by the CA model (99.46 mg/L) approaches the observed EC50 (85.04 mg/L), and the differences in the EC50 value between the observed and the CA predicted is 17%. However, the differences in the EC50 value between the observed and the CA predicted (56.30 mg/L) is 34%. Generally, the CA model performed better than the IA method although the CA slightly underestimated the observed toxicity of the binary mixtures of TiO2 NPs and TiO2 NTs to S. obliquus. This might be because the predictive power of the CA model was strictly restricted by the concentration addition MOJA of TiO2 NPs and TiO2 NTs to S. obliquus.
Figure 5.
Comparison between regression models of the observed (OBS) combined nanotoxicity and expected toxicity of the mixtures to Scenedesmus obliquus (A) and Chlorella pyrenoidosa (B) according to concentration addition (CA) and independent action (IA). The dashed-line and dotted-line represent the 95% confidence band and 95% prediction band of the experimental data points, respectively.
For C. pyrenoidosa, the CRC derived from the CA and IA models deviated moderately from the observed CRC (Figure 5B). Moreover, the concentration-response data predicted by the CA and IA models were inside the PB range of the observed data. Further, in the higher effect regions (about >65%), the CA prediction was inside the CB range of observed concentration-response data. Except for the range of effects from about 50% to 90%, the IA prediction was inside the CB range of observed concentration-response data. As shown in Table 2, the EC50 value predicted by the CA model was 2.9 times greater than the observed EC50 value. However, the EC50 value predicted by the IA model was 1.9 times lower than the observed EC50 value. Similar to S. obliquus, the CA underestimated the joint toxicity, while the IA model overestimated joint toxicity to C. pyrenoidosa. Generally, the IA model performs better than the CA method. This further implies that the MOJA of TiO2 NPs and TiO2 NTs to C. pyrenoidosa is mainly based on response addition. Taken together, the CA and IA methods provided valid predictions of the toxicity of the mixtures.
3.4. Cellular Oxidative Stress Effects of Single and Mixtures of TiO2 NPs and NTs on Algal Cells
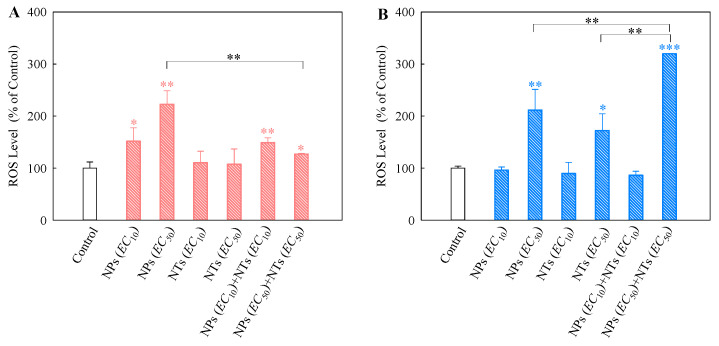
Cellular oxidative stress caused by the elevation of particle-induced ROS is considered the most likely toxic mechanism of nano-TiO2 [38,39,40]. As shown in Figure 6A for S. obliquus, the FI (%) of the TiO2 NPs is significantly higher (p < 0.05) than the control, which indicates a significant increase in ROS. However, there was no significant difference in the ROS level between the TiO2 NTs and control. This implies that TiO2 NPs, but not TiO2 NTs produce ROS in S. obliquus cells. The binary systems of TiO2 NPs and NTs significantly promoted the generation of intracellular ROS. Note that the ROS levels induced by the binary TiO2 NPs + NTs mixtures at the EC50 ratio were significantly lower than the ROS levels induced by single TiO2 NPs at the EC50 value.
Figure 6.
Relative levels of reactive oxygen species (ROS) detected using 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) staining in Scenedesmus obliquus (A) and Chlorella pyrenoidosa (B) exposed to single TiO2 NPs and TiO2 NTs at each EC10 or EC50 value, as well as TiO2 NPs + NTs at the EC10 or EC50 ratios. Statistical significance versus control group: * p < 0.05, ** p < 0.01, and *** p < 0.001.
For C. pyrenoidosa, the TiO2 NPs and NTs at their EC50 value, as well as the TiO2 NPs + NTs at the EC50 ratio significantly increased the ROS levels (Figure 6B). Furthermore, the binary mixtures of TiO2 NPs and NTs induced the generation of intracellular ROS to a higher level than the single TiO2 NPs and NTs, which may intensify the oxidative stress effects on the C. pyrenoidosa cells exposed to the combination of TiO2 NPs + NTs. This also causes more serious apparent toxicity, as we observed in the growth inhibition toxicity testing. In general, TiO2 particle-induced ROS production depended on the particle characteristics, algal cell types, and exposure concentrations. In addition to the mechanisms underlying ROS generation, it also remains unknown as to how the TiO2 NPs interact with the TiO2 NTs and how this interaction regulates the intracellular ROS levels. Nano-TiO2 can cause genotoxicity [41]. Furthermore, the ROS-mediated stress within cells could be the main mechanism for the genotoxicity of nano-TiO2 [42]. Further studies are needed to explore whether the TiO2 NPs and NTs can jointly cause DNA damage due to the production of ROS.
4. Conclusions
To sum up, for the first time we present the toxicity of multiple ENP systems with different dimensions. It was found that the single toxicity varied as a function of the TiO2 dimensions, the test species, and exposure concentrations. The toxicity of the binary mixtures of TiO2 NPs (zero-dimension) and NTs (one-dimension) to two freshwater algae was found to be an additive joint activity according to the TUs. The classical toxicological models (CA and IA) for mixtures predicted the joint toxicities and revealed that the TiO2 NPs and NTs acted as a concentration addition and response addition towards S. obliquus and C. pyrenoidosa, respectively. The mechanisms of TiO2 NPs-NTs joint toxicity were related to the aqueous stability of the TiO2 particles and their ROS-induced oxidative stress effects. Our findings highlight the importance of the dimensions of nanoparticles in assessing the combined risks of multiple ENPs.
Acknowledgments
We thank the editor and the anonymous reviewers for helping to improve the manuscript.
Author Contributions
Z.W. conceived and designed the biological experiments, performed the physicochemical analyses, and co-wrote the paper; S.J. performed the biological experiments. F.Z. designed the experiments, discussed, and co-wrote the paper. D.W. contributed to the general discussion and experimental assistance. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (grant number 31971522) and the Natural Science Foundation of Jiangsu Province (grant number BK20191403).
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Klaper R.D. The known and unknown about the environmental safety of nanomaterials in commerce. Small. 2020;16:e2000690. doi: 10.1002/smll.202000690. [DOI] [PubMed] [Google Scholar]
- 2.Roma J., Matos A.R., Vinagre C., Duarte B. Engineered metal nanoparticles in the marine environment: A review of the effects on marine fauna. Mar. Environ. Res. 2020;161:105110. doi: 10.1016/j.marenvres.2020.105110. [DOI] [PubMed] [Google Scholar]
- 3.Damasco J.A., Ravi S., Perez J.D., Hagaman D.E., Melancon M.P. Understanding nanoparticle toxicity to direct a safe-by-design approach in cancer nanomedicine. Nanomaterials. 2020;10:2186. doi: 10.3390/nano10112186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hou J., Wu Y., Li X., Wei B., Li S., Wang X. Toxic effects of different types of zinc oxide nanoparticles on algae, plants, invertebrates, vertebrates and microorganisms. Chemosphere. 2018;193:852–860. doi: 10.1016/j.chemosphere.2017.11.077. [DOI] [PubMed] [Google Scholar]
- 5.Hua J., Vijver M.G., Richardson M.K., Ahmad F., Peijnenburg W.J. Particle-specific toxic effects of differently shaped zinc oxide nanoparticles to zebrafish embryos (Danio rerio) Environ. Toxicol. Chem. 2014;33:2859–2868. doi: 10.1002/etc.2758. [DOI] [PubMed] [Google Scholar]
- 6.Bhagat J., Nishimura N., Shimada Y. Toxicological interactions of microplastics/nanoplastics and environmental contaminants: Current knowledge and future perspectives. J. Hazard. Mater. 2020;123913 doi: 10.1016/j.jhazmat.2020.123913. [DOI] [PubMed] [Google Scholar]
- 7.Martin O., Scholze M., Ermler S., McPHIE J., Bopp S.K., Kienzler A., Parissis N., Kortenkamp A. Ten years of research on synergisms and antagonisms in chemical mixtures: A systematic review and quantitative reappraisal of mixture studies. Environ. Int. 2020;146:106206. doi: 10.1016/j.envint.2020.106206. [DOI] [PubMed] [Google Scholar]
- 8.Roy B., Suresh P., Chandrasekaran N., Mukherjee A. Antibiotic tetracycline enhanced the toxic potential of photo catalytically active P25 titanium dioxide nanoparticles towards freshwater algae Scenedesmus obliquus. Chemosphere. 2020;128923 doi: 10.1016/j.chemosphere.2020.128923. [DOI] [PubMed] [Google Scholar]
- 9.Thiagarajan V., Seenivasan R., Jenkins D., Chandrasekaran N., Mukherjee A. Combined effects of nano-TiO2 and hexavalent chromium towards marine crustacean Artemia salina. Aquat. Toxicol. 2020;225:105541. doi: 10.1016/j.aquatox.2020.105541. [DOI] [PubMed] [Google Scholar]
- 10.Fu J., Guo Y., Yang L., Han J., Zhou B. Nano-TiO2 enhanced bioaccumulation and developmental neurotoxicity of bisphenol a in zebrafish larvae. Environ. Res. 2020;187:109682. doi: 10.1016/j.envres.2020.109682. [DOI] [PubMed] [Google Scholar]
- 11.Singh D., Kumar A. Assessment of toxic interaction of nano zinc oxide and nano copper oxide on germination of Raphanus sativus seeds. Environ. Monit. Assess. 2019;191:703. doi: 10.1007/s10661-019-7902-5. [DOI] [PubMed] [Google Scholar]
- 12.Singh D., Kumar A. Binary mixture of nanoparticles in sewage sludge: Impact on spinach growth. Chemosphere. 2020;254:126794. doi: 10.1016/j.chemosphere.2020.126794. [DOI] [PubMed] [Google Scholar]
- 13.Yu R., Wu J., Liu M., Zhu G., Chen L., Chang Y., Lu H. Toxicity of binary mixtures of metal oxide nanoparticles to Nitrosomonas europaea. Chemosphere. 2016;153:187–197. doi: 10.1016/j.chemosphere.2016.03.065. [DOI] [PubMed] [Google Scholar]
- 14.Zhang H., Shi J., Su Y., Li W., Wilkinson K.J., Xie B. Acute toxicity evaluation of nanoparticles mixtures using luminescent bacteria. Environ. Monit. Assess. 2020;192:1–8. doi: 10.1007/s10661-020-08444-6. [DOI] [PubMed] [Google Scholar]
- 15.Ye N., Wang Z., Wang S., Peijnenburg W.J. Toxicity of mixtures of zinc oxide and graphene oxide nanoparticles to aquatic organisms of different trophic level: Particles outperform dissolved ions. Nanotoxicology. 2018;12:423–438. doi: 10.1080/17435390.2018.1458342. [DOI] [PubMed] [Google Scholar]
- 16.Wu B., Wu J., Liu S., Shen Z., Chen L., Zhang X.-X., Ren H.-Q. Combined effects of graphene oxide and zinc oxide nanoparticle on human A549 cells: Bioavailability, toxicity and mechanisms. Environ. Sci. Nano. 2019;6:635–645. doi: 10.1039/C8EN00965A. [DOI] [Google Scholar]
- 17.Illarionov G.A., Morozova S.M., Chrishtop V.V., Einarsrud M.-A., Morozov M.I. Memristive TiO2: Synthesis, technologies, and applications. Front. Chem. 2020;8:724. doi: 10.3389/fchem.2020.00724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roy P., Berger S., Schmuki P. TiO2 nanotubes: Synthesis and applications. Angew. Chem. Int. Ed. 2011;50:2904–2939. doi: 10.1002/anie.201001374. [DOI] [PubMed] [Google Scholar]
- 19.Reddy P.V.G., Reddy B.R.P., Reddy M.V.K., Reddy K.R., Shetti N.P., Saleh T.A., Aminabhavi T.M. A review on multicomponent reactions catalysed by zero-dimensional/one-dimensional titanium dioxide (TiO2) nanomaterials: Promising green methodologies in organic chemistry. J. Environ. Manag. 2020;111603 doi: 10.1016/j.jenvman.2020.111603. [DOI] [PubMed] [Google Scholar]
- 20.Chakraborty D., Ethiraj K., Chandrasekaran N., Mukherjee A. Mitigating the toxic effects of CdSe quantum dots towards freshwater alga Scenedesmus obliquus: Role of eco-corona. Environ. Pollut. 2020;116049 doi: 10.1016/j.envpol.2020.116049. [DOI] [PubMed] [Google Scholar]
- 21.Luo Z., Wang Z., Yan Y., Li J., Yan C., Xing B. Titanium dioxide nanoparticles enhance inorganic arsenic bioavailability and methylation in two freshwater algae species. Environ. Pollut. 2018;238:631–637. doi: 10.1016/j.envpol.2018.03.070. [DOI] [PubMed] [Google Scholar]
- 22.Zhao J., Ning F., Cao X., Yao H., Wang Z., Xing B. Photo-transformation of graphene oxide in the presence of co-existing metal ions regulated its toxicity to freshwater algae. Water Res. 2020;176:115735. doi: 10.1016/j.watres.2020.115735. [DOI] [PubMed] [Google Scholar]
- 23.He X., Xie C., Ma Y., Wang L., He X., Shi W., Liu X., Liu Y., Zhang Z. Size-dependent toxicity of ThO2 nanoparticles to green algae Chlorella pyrenoidosa. Aquat. Toxicol. 2019;209:113–120. doi: 10.1016/j.aquatox.2019.02.003. [DOI] [PubMed] [Google Scholar]
- 24.OECD Guidelines for the Testing of Chemicals; Proceedings of the OECD Freshwater Alga and Cyanobacteria, Growth Inhibition Test No. 201; Paris, France. 28 July 2011; [(accessed on 12 December 2020)]. Available online: http://www.oecd.org. [Google Scholar]
- 25.Cosgrove T. Colloid Science Principles, Methods and Applications. Blackwell Publishing Ltd.; Bristol, UK: 2005. [Google Scholar]
- 26.Karlsson H.L., Cronholm P., Gustafsson J., Möller L. Copper oxide nanoparticles are highly toxic: A comparison between metal oxide nanoparticles and carbon nanotubes. Chem. Res. Toxicol. 2008;21:1726–1732. doi: 10.1021/tx800064j. [DOI] [PubMed] [Google Scholar]
- 27.Hu X., Ouyang S., Mu L., An J., Zhou Q. Effects of graphene oxide and oxidized carbon nanotubes on the cellular division, microstructure, uptake, oxidative stress, and metabolic profiles. Environ. Sci. Technol. 2015;49:10825–10833. doi: 10.1021/acs.est.5b02102. [DOI] [PubMed] [Google Scholar]
- 28.Kim K.T., Klaine S.J., Lin S., Ke P.C., Kim S.D. Acute toxicity of a mixture of copper and single-walled carbon nanotubes to Daphnia magna. Environ. Toxicol. Chem. 2010;29:122–126. doi: 10.1002/etc.8. [DOI] [PubMed] [Google Scholar]
- 29.George S., Lin S., Ji Z., Thomas C.R., Li L., Mecklenburg M., Meng H., Wang X., Zhang H., Xia T., et al. Surface defects on plate-shaped silver nanoparticles contribute to its hazard potential in a fish gill cell line and zebrafish embryos. ACS Nano. 2012;6:3745–3759. doi: 10.1021/nn204671v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Demir E. An in vivo study of nanorod, nanosphere, and nanowire forms of titanium dioxide using Drosophila melanogaster: Toxicity, cellular uptake, oxidative stress, and DNA damage. J. Toxicol. Environ. Heal. 2020;83:456–469. doi: 10.1080/15287394.2020.1777236. [DOI] [PubMed] [Google Scholar]
- 31.Demir E. A review on nanotoxicity and nanogenotoxicity of different shapes of nanomaterials. J. Appl. Toxicol. 2020;41:118–147. doi: 10.1002/jat.4061. [DOI] [PubMed] [Google Scholar]
- 32.Zhang D., Qiu J., Shi L., Liu Y., Pan B., Xing B. The mechanisms and environmental implications of engineered nanoparticles dispersion. Sci. Total. Environ. 2020;722:137781. doi: 10.1016/j.scitotenv.2020.137781. [DOI] [PubMed] [Google Scholar]
- 33.Aruoja V., Pokhrel S., Sihtmäe M., Mortimer M., Mädler L., Kahru A. Toxicity of 12 metal based nanoparticles to algae, bacteria and protozoa. Environ. Sci. Nano 2. 2015;2:630–644. doi: 10.1039/C5EN00057B. [DOI] [Google Scholar]
- 34.Aruoja V., Dubourguier H.-C., Kasemets K., Kahru A. Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci. Total Environ. 2009;407:1461–1468. doi: 10.1016/j.scitotenv.2008.10.053. [DOI] [PubMed] [Google Scholar]
- 35.Zhang F., Ye N., Wang S., Meng Y., Fang H., Wang Z., Wang D.G. Dissolved organic matter modulates algal oxidative stress and membrane system responses to binary mixtures of nano-metal-oxides (nCeO2, nMgO and nFe3O4) and sulfadiazine. Nanomaterials. 2019;9:712. doi: 10.3390/nano9050712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu Y., Baas J., Peijnenburg W.J.G.M., Vijver M.G. Evaluating the combined toxicity of Cu and ZnO nanoparticles: Utility of the concept of additivity and a nested experimental design. Environ. Sci. Technol. 2016;50:5328–5337. doi: 10.1021/acs.est.6b00614. [DOI] [PubMed] [Google Scholar]
- 37.Martín-De-Lucía I., Gonçalves S.F., Leganés F., Fernández-Piñas F., Rosal R., Loureiro S. Combined toxicity of graphite-diamond nanoparticles and thiabendazole to Daphnia magna. Sci. Total. Environ. 2019;688:1145–1154. doi: 10.1016/j.scitotenv.2019.06.316. [DOI] [PubMed] [Google Scholar]
- 38.Tsugita M., Morimoto N., Nakayama M. SiO2 and TiO2 nanoparticles synergistically trigger macrophage inflammatory responses. Part. Fibre Toxicol. 2017;14:1–9. doi: 10.1186/s12989-017-0192-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liu Y., Wang S., Wang Z., Ye N., Fang H., Wang D. TiO2, SiO2 and ZrO2 nanoparticles synergistically provoke cellular oxidative damage in freshwater microalgae. Nanomaterials. 2018;8:95. doi: 10.3390/nano8020095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Han B., Pei Z., Shi L., Wang Q., Li C., Zhang B., Su X., Zhang N., Zhou L., Zhao B., et al. TiO2 nanoparticles caused DNA damage in lung and extra-pulmonary organs through ROS-activated FOXO3a signaling pathway after intratracheal administration in rats. Int. J. Nanomed. 2020;15:6279–6294. doi: 10.2147/IJN.S254969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wani M.R., Shadab G. Titanium dioxide nanoparticle genotoxicity: A review of recent in vivo and in vitro studies. Toxicol. Ind. Heal. 2020;36:514–530. doi: 10.1177/0748233720936835. [DOI] [PubMed] [Google Scholar]
- 42.Santonastaso M., Mottola F., Iovine C., Cesaroni F., Colacurci N., Rocco L. In vitro effects of titanium dioxide nanoparticles (TiO2NPs) on cadmium chloride (CdCl2) genotoxicity in human sperm cells. Nanomaterials. 2020;10:1118. doi: 10.3390/nano10061118. [DOI] [PMC free article] [PubMed] [Google Scholar]